Translate this page into:
Synthesize of pluronic-based nanovesicular formulation loaded with Pistacia atlantica extract for improved antimicrobial efficiency
⁎Corresponding author. akbarizade63@gmail.com (Majid Reza Akbarizadeh)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
One of the current concerns to human health is antibiotic resistance, which promotes the use of antibiotics that are more harmful, expensive, and ineffective. In this condition, researchers are turning to innovative options to combat this alarming situation. Combining herbal medicine with nanotechnology has created a new strategy to increase the effectiveness of phytochemical compounds in overcoming antimicrobial resistance. Pistacia atlantica is one of the promising herbs with medicinal benefits, but its poor solubility in biological fluids is challenging. In this regard, we seek to evaluate the antibacterial efficacy of Pistacia atlantica extract-loaded nanovesicle. Cholesterol, Span 40, and Pluronic F127 modified nanoformulation was developed using an environmentally friendly improved heating technique, and it was evaluated for size distribution, zeta potential, morphology, entrapment efficiency (EE%), release behavior, stability, and antimicrobial performance. By using DLS, spherical nanovesicles were identified with a size distribution of 50–150 nm and a zeta potential of −43 mV. The extract's encapsulation efficiency was 72.03%. The developed loaded nanovesicles demonstrated controlled extract release in the tested 96 h and storage stability of at least 12 months at 25 °C. Also, Comparing the two samples, the encapsulated extract had greater antibacterial activity against Candida albicans, Staphylococcus aureus, and Pseudomonas plecoglossicida with MIC of 1320, 570, and 1100 µg/mL, respectively. Besides reducing the misuse of antibiotics by allowing for the controlled release of drugs made from natural sources, we expect the findings described here to help provide alternative plant-based formulations with greater stability and antibacterial activity.
Keywords
Nanovesicle
Pluronic surfactant
Pistacia atlantica extract
Anti-microbial effects
- EE
-
Encapsulation efficiency
- DLS
-
Dynamic light scattering
- MDA-MB-231
-
MDA stands for “M.D. Anderson and MB stands for Metastasis Breast cancer
- PBS
-
Phosphate-buffered saline
- DMSO
-
Dimethyl sulfoxide
- MTT
-
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide
- DMEM
-
Dulbecco's Modified Eagle Medium
- FBS
-
Fetal bovine serum
- TEM
-
Transmission electron microscopy
- kDa
-
Kilodaltons
- We
-
Amount of extract in the vesicular formulation after centrifugation
- Wtotal
-
Amount of extract that we added at the initial of the experiment
- Mt(n)
-
Current cumulative mass of released extract at time t
- Cn
-
Current concentration of extract in the medium
- ƩCm
-
Summed total of the previously measured concentrations
- Vr
-
Volume of the medium
- Vs
-
Volume of the sample removed for analysis
- MIC
-
Minimum Inhibitory Concentration
- CFU
-
Colony forming units
- SPSS
-
Statistical Package for the Social Sciences
- PDI
-
Polydispersity Index
- mV
-
Millivolts
- HLB
-
Hydrophilic-lipophilic balance
- PPO
-
Poly (p-phenylene oxide)
- PEO
-
Polyethylene glycol
- R2
-
Coefficient of determination
- CsA
-
Cyclosporine A
Abbreviations
1 Introduction
Antimicrobial resistance has risen as a result of widespread, improper, irregular, and indiscriminate use of antibiotics drugs (Li et al., 2022), rendering many currently available medicines useless (Dadgostar, 2019). On the other hand, it is urgently necessary to find and isolate novel bioactive chemicals from medicinal plants due to the rising prevalence of drug-resistant infections (Alijani, 2023). Compounds from medicinal plants could provide creative and easy methods to combat pathogenic microbes (Mulat et al., 2019). Pistacia atlantica, which contains many medicinal phenolic components, is one of the beneficial herbal remedies (Fan et al., 2022; Yang et al., 2022). The Pistacia atlantica tree may reach heights of 2 to 7 m. Its fruit is called Chatlangoosh in Turkish, Baneh in Persian, and Hobb al khazra in Arabic. It turns orange in the fall and the tree's crimson flower is used as a dye (Ahmed et al., 2021, Tahmourespour et al., 2022). Pistacia atlantica has many medical and industrial applications (Mahjoub et al., 2018; Ahmed et al., 2021; Benmahieddine et al., 2021). For example, the antimicrobial properties of Pistacia atlantica extract were assessed, and inhibition zones of 25 and 19 mm with a substantial inhibitory impact on S. mitis (ATCC 49456) and S. mutans (ATCC 35668) were observed. Despite the fact that medicinal plants like Pistacia atlantica and their extracts have a variety of purposes, their practical usage has been constrained by issues like solubility and short half-lives (Nikkhah et al., 2020).
Research on nutraceuticals may benefit from the creative approach of nanotechnology (Qureshi et al., 2021). The water solubility (Barani et al., 2023), stability (Hamidian et al., 2023), bioavailability (Mostafavi et al., 2022), circulation time (Lu et al., 2022), and target specificity of many drugs and phytochemicals can be enhanced by loading them into biocompatible and biodegradable nanostuctures (Kumar et al., 2018; Kashkooli et al., 2020). For example, Sauraj et al. developed lipophilic 5-fluorouracil prodrug encapsulated xylan-stearic acid conjugates nanoparticles for colon cancer treatment (Kumar et al., 2019). In a related work, the same team also revealed Redox-responsive xylan-SS-curcumin prodrug nanomaterials for dual drug delivery in cancer treatment (Kumar et al., 2020). It is necessary to optimize formulation in order to increase stability (Ibrahim et al., 2021; Wang et al., 2021), bioavailability (Zhou et al., 2021; Xiong et al., 2021), and effectiveness of phytochemicals against microorganisms (Sun et al., 2022; K. Yang et al., 2022; R. Yang et al., 2022). Enhancing the effectiveness of phytochemicals as antimicrobials is now significantly possible because to research developments in nanoformulation (Alijani et al., 2023; Ameen et al., 2021; Deepa et al., 2022) utilizing biocompatible materials (Jeevanandam et al., 2017). It has been shown that antifungal (Sathishkumar et al., 2016; Almansob et al., 2022), anticancer (Darvish et al., 2022; Gangalla et al., 2021; Indhira et al., 2022; Rajadurai et al., 2021), antimicrobial (Ameen et al., 2021; Moghadam et al., 2022; Begum et al., 2022), catalist (Megarajan et al., 2022; Mohammad et al., 2023; Sonbol et al., 2021), antimicrobial efficacy (Rani et al., 2019; Soundararajan et al., 2022) in food manufacturing and clinical application as antibiotics may be improved by encapsulating antimicrobial phytochemicals utilizing micelles, liposomes, dendrimers, polymers, and microemulsions (Kumar et al., 2018; Kashkooli et al., 2020). Vesicular nanosystems in particular show advantageous characteristics, such as biocompatibility, targeted and prolonged delivery capacity (Pan et al., 2023; Zheng et al., 2022), enhanced phytocompound bioavailability and protection, giving them considerable potential for future uses in microbial infection (Prajapati et al., 2021). Because of the above-mentioned favorable characteristics of vesicular nanoplatforms as prospective controlled delivery nano-carriers, researchers are interested in them (Mangalgiri et al., 2021). For example, in lipid formulations, Pistacia atlantica fruit oil was utilized as a carrier. A potential area for treating many illnesses is the creation of lipid nanoparticles using vegetable oils. These therapeutic benefits are the consequence of interactions between active substances and vegetable oil lipid transporters, which have synergistic effects (El Zerey-Belaskri et al., 2022). In another study, it was observed that loading Pistacia atlantica essential oil to solid lipid nanoparticles had an impact on the breast cancer cell line MDA-ability MB-231’s to undergo apoptosis. The results showed that loaded extract may more effectively trigger apoptosis in MDA-MB-231 cells than free extract (Dousti et al., 2022). It should be noted that no similar study was observed for loading the Pistacia atlantica extract into nanovesicles and investigating its antimicrobial properties.
Vesicular nanoplatforms are produced using conventional thin-film methods, which are frequently required for the inclusion of organic solvents during one or more preparation steps. Because of the residual solvents, these techniques put patients at risk for health problems. Evaporation is one of the additional several steps in the time-consuming, expensive process (Yeo et al., 2018). To circumvent these issues caused by thin-film hydration methods, niosome synthesis without the use of organic solvents is required. This calls for a straightforward, cost-effective, and ecologically friendly synthesis process like the Mozafari technique (Mozafari et al., 2008). In the current work, the Mozafari method—an enhanced heating technique was employed to produce vesicles by agitating phospholipids and surfactants in an aqueous media. Then, without using any hazardous chemicals, these components were warmed in the presence of glycerol. Recently, the food-grade antibacterial Nisin was effectively encapsulated and delivered to specific areas using the Mozafari approach (Colas et al., 2007). The aforementioned technique uses a straightforward methodology and a single vessel to produce nanovesicles with improved monodispersity and storage stability at a low cost (Jahanfar et al., 2021).
In this research, for the first time, we aimed to develop a nanovesicular formulation that encapsulate Pistacia atlantica in order to prevent the effects of antibiotic resistance. In addition, the Mozafari technique, a cost-effective method of generating vesicles, replaced the traditional preparation process of nanovesicles. The zeta potential, size, shape, stability, extract entrapment effectiveness, and release behavior of the prepared vesicles were assessed. The antibacterial effects of free extract and encapsulated extract were then evaluated against Candida albicans, Staphylococcus aureus, and Pseudomonas plecoglossicida.
2 Materials and methods
2.1 Materials
Cholesterol, Span 40, Pluronic® F127, phosphate-buffered saline (PBS), glycerol, penicillin, Trypan blue, DMSO, MTT, streptomycin, and chloroform were purchased from Sigma-Aldrich (The Netherlands). The used water was Milli-Q type from Merck Millipore. The fetal bovine serum (FBS), 0.25% trypsin and modified Eagle’s medium (DMEM) were obtained from Gibco (Grand Island, NY, USA). All chemicals were of analytical grade.
2.2 Plant materials
The plant materials (fruits of Pistacia atlantica) that were the focus of this research were gathered in August 2022 from rural areas of Sanandaj Province in Iran's east. To support the reproducibility, voucher specimens for P. atlantica were deposited in University’s public herbarium. The P. atlantica were collected under the license of the collection of plant or seed specimens in the University per applicable institutional, national, and international rules and legislation. It was verified by the university Botanical Survey Prof. Biriji, whose voucher specimen number was 1402-08 deposited at Lorestan University in Iran's Department of Botanical Sciences.
2.3 Extract preparation
P. atlantica fruits (100 g) were air-dried, coarsely ground, and immersed in a solution of methanol: water (60:40) for 120 min. The solution was then percolated for 72 h to ensure a thorough extraction. A semisolid extract was achieved when the extract was agitated, filtered, and evaporated in a rotary evaporator under vacuum (Falahati et al., 2015). The concentrated extract was freeze-dried, in accordance with the British Pharmacopoeia, to produce a dry powdered extract.
2.4 Preparation of extract loaded nanovesicles by improved heating method
The Mozafari method (improved heating method) was used to synthesize extract-loaded nanovesicles in an environmentally friendly manner without the use of solvents or detergents (Jahanfar et al., 2021). Initially, distilled water was added with the Pistacia atlantica extract. The extract solution was then treated with a combination of Span 40, Pluronic F127, cholesterol, and glycerol. The mixture was stirred for 60 min at a speed of 1000 rpm on a hot plate stirrer at temperatures between 40 and 80 °C. The mixture is left at room temperature for one hour, which causes the vesicles to anneal and become more stable. The mixture was kept until use at 4 °C in a dark container with nitrogen. Fig. 1 shows a schematic representation of the Pluronic vesicular nanoplatform loaded with Pistacia atlantica fruits extract and the evolution of its antimicrobial activity.
Schematic Representation of Pluronic vesicular nanoplatform loaded with Pistacia atlantica fruits extract and evolution of its antimicrobial activity.
3 Characterization of the synthesized niosomes
3.1 Size distribution and morphology
A dynamic light scattering analyzer (Malvern, UK: Zetasizer Nano (ZS), ZEN3600) using a helium laser at a wavelength of 632.8 nm and an angle of 90° was used to examine the size distribution and ζ potential of the Pluronic vesicular nanoplatform loaded with Pistacia atlantica extract. The formulation solution was briefly diluted (1:20) with deionized water to prevent further scattering caused by bigger particles. Three times each of all measurements were made in a chamber that was kept at a constant 25 °C. Transmission electron microscopy (TEM, Zeiss, LEO912-AB, Germany) was used to determine the morphology of formulation by negative staining with a 2% (w/v) uranyl acetate solution.
3.2 Evaluation of the encapsulation efficiency
A UV–vis spectrophotometer (JASCO, V-530, Tokyo, Japan) operating at a wavelength of 410 nm was used to quantify the extract concentration in the vesicular formulation. In a typical procedure, 1 mL of extract-loaded formulation was centrifuged at 20,000 rpm for 60 min. The following formula was used to get the EE% of the loaded formulation: where eW was the amount of extract in the vesicular formulation after centrifugation and Wtotal was the amount of extract that we added at the initial of the experiment.
3.3 In-vitro release
In-vitro release of extract from vesicular formulation was assessed in pH 7.4. Briefly, formulation (0.5 mL) was placed in a dialysis bag (12–14 kDa molecular weight cut-off) in a 100 mL phosphate (pH: 7.4) and. At defined time intervals (0 to 96 h), one mL of the medium was withdrawn and replaced with one ml of fresh buffer. Aliquots of the collected samples were analyzed for their extract content. The cumulative release was calculated using the following equation: where Mt(n) is the current cumulative mass of released extract at time t, n is the number (times) of sampling, Cn is the current concentration of extract in the medium, ƩCm is the summed total of the previously measured concentrations, Vr is the volume of the medium, and Vs corresponds to the volume of the sample removed for analysis. To study their release profiles, various patterns of release kinetics (zero-order, first-order, Higuchi, and Korsmeyer-Peppas model) were used (Verma et al., 2021; Verma et al., 2022; Verma et al., 2022). In the same dialysis bag, the study was also carried out for extract solution with comparable beginning extract concentrations.
3.4 The evaluation of stability
The loaded formulation was synthesized and stored for 12 months at room temperature (25 °C). Physical properties were evaluated at preset intervals (0, 3, 6, 9 and 12 months), including average formulation size (nm) and zeta potential.
3.5 Antibacterial efficacy
Through broth microdilution, the antibacterial activity of extract, empty vesicles, and extract encapsulated in nanovesicles was evaluated against bacterial and fungal strains (Pseudomonas plecoglossicida, Staphylococcus aureus, and Candida albicans). Following that, the minimum inhibitory concentration (MIC) was established. The MIC is the lowest concentration of a substance—typically a drug—that nevertheless allows bacteria to proliferate unnoticeably. After strains were cultivated in Müller-Hinton broth for 24 h, 5 × 105 CFU/mL (5 μl) pathogens, 100 μl of extract, empty vesicle and nanovesicle-encapsulated extract, and 95 μl of pure Hinton broth were added to each well. The 96-well plate was then incubated at 37 °C for 24 h.
3.6 Statistical analysis
Data were coded and processed using the statistical package for the Social Sciences (SPSS) version 25 (IBM Corp., Armonk, NY, USA). ANOVA was performed for normally distributed data in more than two samples. A p value of less than 0.05 was considered statistically significant.
4 Results and discussion
4.1 Physicochemical properties of nanocarrier
The size distribution and zeta values of the extract loaded formulation by DLS at 25 °C are shown in Fig. 2. It was observed that the nanovesicles made using the modified approach ranged in size from 50 to 150 nm. The average size and size distribution of the nanovesicles are significantly influenced by the kind of surfactants and cholesterol type in the vesicular structure. To transport a sufficient number of antibiotics, penetrate the sites of action, and release the entrapped component there, an effective drug carrier must be compact and stable. For instance, Moghaddam et al. recently revealed that the size and PDI of melittin-loaded niosomes may be affected by the surfactant: cholesterol with different molar ratios (Dabbagh Moghaddam et al., 2021).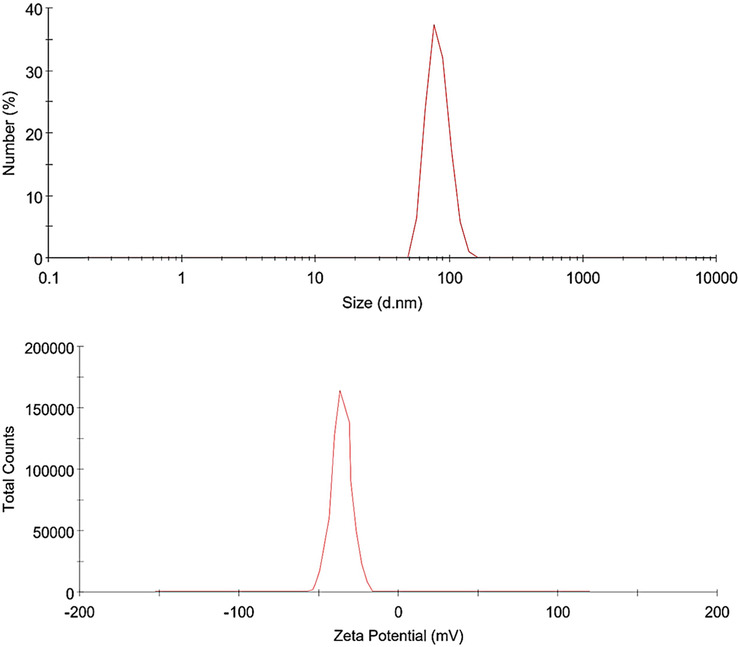
The size distribution and zeta values of extract loaded formulation by DLS at 25 °C.
The polydispersity index, or PDI, is a measure of the size distribution quality of nanosystems. The suitability of a nanocarrier formulation for a certain drug delivery pathway is determined by PDI and zeta potential. Controlling and validating these properties is essential for the therapeutic applications of nanocarrier compositions. With a PDI of around 0.18, the extract-loaded formulation is regarded as a monodisperse and stable nanocarrier. In a related investigation, Silva et al. prepared technetium-99 m-labeled niosomes with PDIs ranging from 0.196 to 0.308 showed good homogeneity throughout all the combinations (De Silva et al., 2019).
In the current study, the formulation's zeta potentials were also approximately −43 mV (Fig. 2). An efficient method for identifying nanocarriers is the estimate of the surface charge using the zeta potential approach, which may also be used to understand the behavior and stability of niosomes in biofluids. The zeta potential of colloidal systems is one of the variables used to assess their stability. The system is stabilized against aggregation as the zeta potential rises because the charged particles resist one another. Systems are regarded as stable if their zeta potential is ±30 mV or above (Joseph and Singhvi, 2019).
Fig. 3 presents TEM images of extract-loaded vesicles. successful synthesize of vesicular nanocarrier was verified by the TEM images. The nanovesicles had spherical forms and resembled the typical vesicles micrographs found in other investigations (Guinedi et al., 2005; Aboelwafa et al., 2010). The diameters of the vesicles, which were measured to be between 70 and 110 nm, were in perfect agreement with the DLS measurements of the particle sizes. The bimodal size distribution of the formulation was shown by the particle size distribution histogram. This was further supported by the TEM investigation, which showed multiple vesicles at a 100 nm size. But in contrast to the overall size distribution, it was found that the fraction of bigger vesicles was quite low. Silva et al. reported the similar trend, noting that DLS and TEM sizes for Technetium-99 m-labeled niosomes are 95 and 101 nm, respectively (De Silva et al., 2019).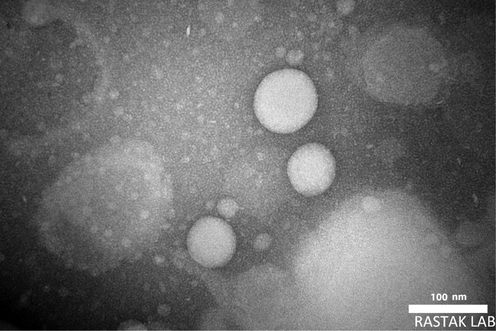
TEM micrograph of extract loaded formulation.
4.2 Encapsulation efficiency of loaded formulation
The EE% of the extract-loaded vesicles was evaluated after the extract that was not entrapped was separated by centrifugation at 15,000 rpm for 30 min at 4 °C. The extract in the loaded formulation had an encapsulation effectiveness (% EE) of 72.03%. Pluronic F127 and Span 40 have molecular weights of 12.600 and 402.6, respectively. According to reports, adding a low molecular weight amphiphile to pluronics has a major impact on whether these polymers associate or dissociate to produce vesicles. Span 40 is a hydrophobic amphiphile with an HLB value of 6.7, and extract has a hydrophobic nature as well. The entrapment location inside the bilayer must thus be shared by the extract and the hydrophobic amphiphile when Pluronic F127, which has the greatest HLB and longest PPO chain length, is mixed with Span 40 (Sezgin-Bayindir et al., 2015). This can explain the low extract loading.
4.3 In-vitro release behavior
Fig. 4 displays the extract release patterns from free extract and extract loaded vesicles in PBS (pH 7.4). The in-vitro drug release profiles showed that extract released from loaded vesicles is noticeably lower than free extract. The findings of earlier research are consistent with the release profile of free extract (Detroja et al., 2011). The amounts released after 96 h are shown in Fig. 4. The release profiles show a common method of drug release, prolonged drug release of extract from nanovesicles (Yang et al., 2008). This activity was attributed to the hydrophilic PEO chains that surround the surface of the vesicles and cause a reduction in surface tension that hinders the release of the extract. At pH 7.4, the extract-loaded formulation released 70.2 ± 6.1% of the extract after 96 h whereas the free extract released completely in 20 h. Extended-release behavior was shown by several research on the in-vitro release behavior of encapsulated formulations. The in-vitro release of free Cyclosporine A and niosomal formulations showed similar findings (Shilakari Asthana et al., 2016; Rasul et al., 2020). It was determined that prepared extract-loaded niosomes are excellent options for boosting extract solubility, which may lead to an improvement in effectiveness and bioavailability.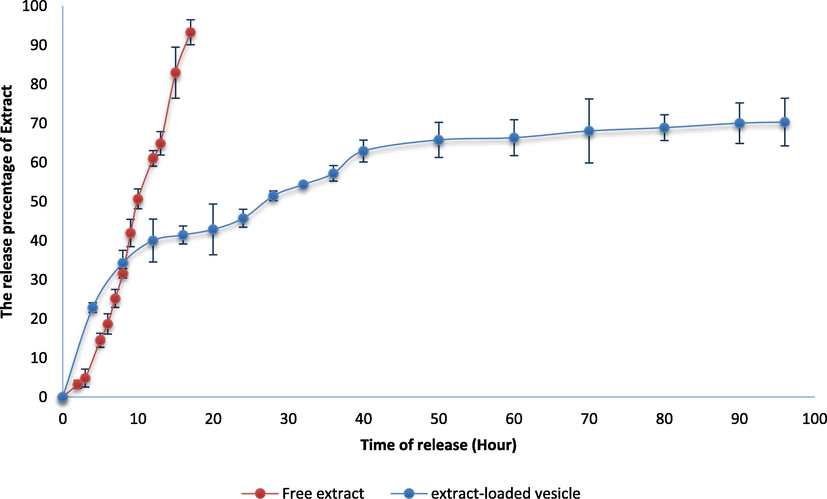
Drug release profiles of extract-loaded formulation and free extract at pH 7.4.
The in-vitro release data was used for different kinetic model equations to determine the drug release model from vesicular formulation. The release and regression coefficient constant (R2) were evaluated based on the slope of the applicable plots. The various equation curves of the extract loaded nanovesicle were displayed in Fig. 5. Based on this figure, the Korsmeyer Peppas modeling for the vesicular formulation has a reasonable R2 value. The Korsmeyer-Peppas proposed model in our investigation had release exponent (n) values that were close to those of the aceclofenac niosomal formulation (Diva et al., 2014). On other study, Cyclosporine loaded in the niosome formulation and showed different data compared with our study, so that, the release kinetic behavior demonstrated that all formulations were best represented by a zero-order model and could have a controlled release of drug over time (Diva et al., 2014).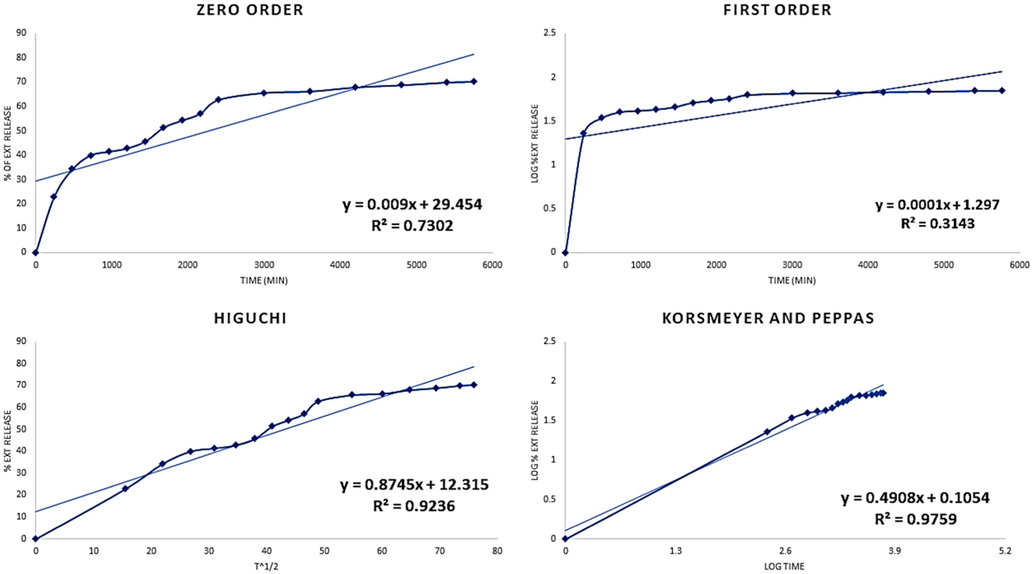
Different release kinetics of extract loaded nanovesicle.
4.4 Stability study
Vesicular systems must maintain their integrity under various circumstances. The stability of the loaded formulation was assessed over a 12-month period at 25 °C using the parameters of particle size and zeta potential (Fig. 6). The size of the extract-loaded vesicle changed little at 25 °C, demonstrating good physical stability. This slight changes might be due to the aggregation or fusing of vesicles, which often happened when molecular mobility increased and smaller vesicles converted into bigger ones (Kopermsub et al., 2011). The presence of cholesterol in vesicles contributes to their high level of stability. Zeta potential values were shown to decrease up to 6 months before increasing once again during the subsequent 6 months. As a result, and in excellent accord with other results, these loaded vesicles may be a stable formulation that they have higher storage period. For instance, the size, zeta, and EE% of a cyclosporine A (CsA) loaded niosome were assessed over the period of 3 months. They demonstrated that after three months of storage, niosomal vesicles are a stable formulation (Rasul et al., 2020).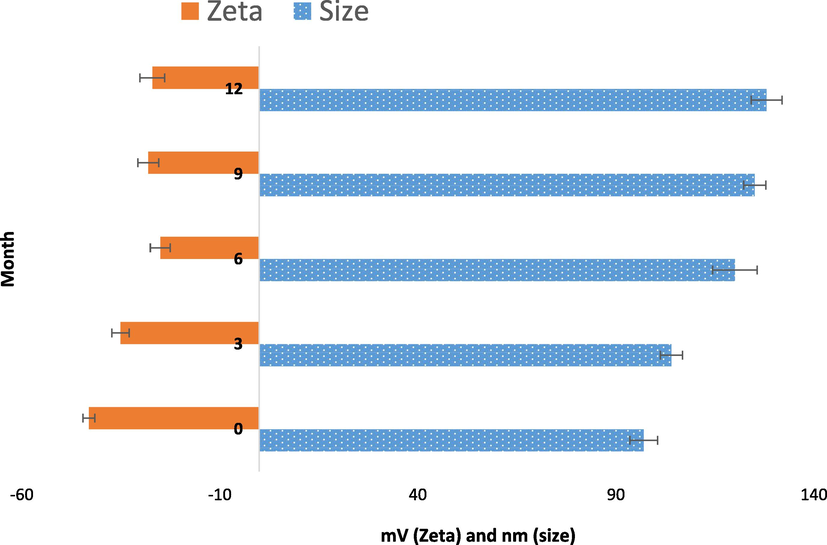
Variations in size and zeta potential of extract-loaded vesicle during storage at a 12-month period in 25 °C.
4.5 Antimicrobial activity of formulations
The antibacterial effects of synthesized extract loaded vesicles were assessed by measuring the minimum inhibitory concentration (MIC). Pistacia atlantica extract has been shown in prior investigations to have an antibacterial effect. For instance, the lowest inhibitory concentration, minimum bactericidal concentration, and disc diffusion techniques were used to assess the antibacterial activity of the Pistacia atlantica extract against six pathogenic bacteria. The essential oil displayed stronger antibacterial activity against the tested Gram-positive bacteria than the Gram-negative bacteria, according to the results of both antimicrobial techniques. Fig. 7 represents the MIC of free vesicles, extracts, and vesicles/extracts against Candida albicans, Staphylococcus aureus, and Pseudomonas plecoglossicida. There was no evidence of antibacterial action in the free vesicles. The vesicle/extract demonstrated more potent antibacterial action against all three pathogens than free extract did. It's interesting to note that Staphylococcus aureus, with a MIC of 570 g/mL, had the best antibacterial action. The Staphylococcus aureus (a gram-positive bacteria) is responsible for a wide range of clinical illnesses. This bacterium frequently causes infections, both in situations where they are contracted in the community and in hospitals. Many authors mentioned that the uptake of vesicles is higher in gram-positive bacteria. These results suggest that, in comparison to free extract, lower vesicle/extract concentrations are needed to inhibit microbial growth. This may be because nanovesicles have the capacity to protect extract from microbial enzyme effects and facilitate vesicle interaction on the surface of the pathogen membrane. For instance, Mansouri et al found that streptomycin sulfate-loaded niosomes showed more antibacterial activity than free streptomycin sulfate, with MIC values decreasing between 4- and 8-folds (Mansouri et al., 2021). Similar studies by Kashef et al. showed that ciprofloxacin encapsulated niosomes were more efficient against staphylococcus aureus that forms biofilms and is ciprofloxacin resistant (Kashef et al., 2020).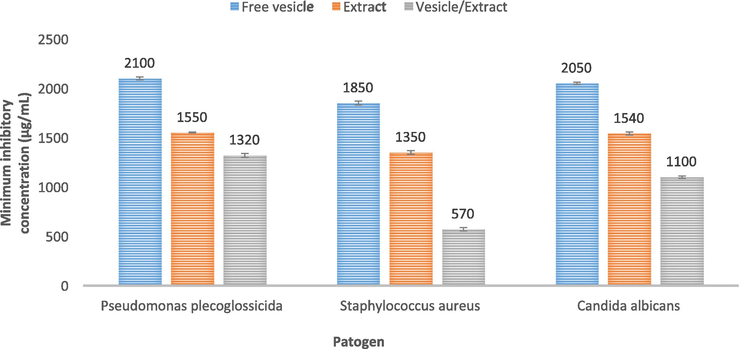
The minimum inhibitory concentration (MIC) of free Vesicle, extract and Vesicle/extract against Pseudomonas plecoglossicida, Staphylococcus aureus, and Candida albicans.
5 Conclusion
Here, an environmentally friendly method for preparing nanovesicles for the delivery of Pistacia atlantica extract was successfully used. In comparison to free extracts, the developed loaded nanovesicles showed improved stability and antibacterial efficacy, enabling high encapsulation efficiency and controlled release. In comparison to the free extract, the loaded vesicles also showed an improved potency against Candida albicans, Staphylococcus aureus, and Pseudomonas plecoglossicida. This work advances the use of drugs obtained from nature in the treatment of infectious disorders brought on by bacteria that are multidrug resistant. To determine the plant extracts' active ingredients and to learn more about the underlying process, more research is required. We see a chance to expand on the information we've obtained here about plant extracts and nanovesicles to additional bioactive substances for better stability and antibacterial effectiveness. The suggested vesicle-loaded extract release is anticipated to aid decrease the misuse of antibiotics by permitting nature-derived materials and controlled drug release, even if the therapeutic efficacy of pathogen elimination in reality has yet to be proved in clinics.
Acknowledgement
Authors would like to acknowledge Zabol University of Medical Sciences for supporting this research.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Comparative study on the effects of some polyoxyethylene alkyl ether and sorbitan fatty acid ester surfactants on the performance of transdermal carvedilol proniosomal gel using experimental design. AAPS PharmSciTech. 2010;11:1591-1602.
- [Google Scholar]
- Four Pistacia atlantica subspecies (atlantica, cabulica, kurdica and mutica): A review of their botany, ethnobotany, phytochemistry and pharmacology. J. Ethnopharmacol.. 2021;265:113329.
- [Google Scholar]
- Biosynthesis of core–shell α-Fe2O3@Au nanotruffles and their biomedical applications. Biomass Conversion and Biorefinery. 2023;13:1-6.
- [Google Scholar]
- A review on metal-based nanoparticles and their toxicity to beneficial soil bacteria and fungi. Ecotoxic Environ Safety. 2021;213:112027.
- [Google Scholar]
- review on metal-based nanoparticles and their toxicity to beneficial soil bacteria and fungi. Ecotox Enviro Safety. 2021;213:112027.
- [Google Scholar]
- Aptamer-conjugated carbon nanotubes or graphene for targeted cancer therapy and diagnosis. Aptamers Engineered Nanocarriers for Cancer Therapy. 2023;2023:277-294.
- [Google Scholar]
- Influence of plant and environment parameters on phytochemical composition and biological properties of Pistacia atlantica Desf. Biochem. Syst. Ecol.. 2021;95:104231.
- [Google Scholar]
- Microscopical investigations of nisin-loaded nanoliposomes prepared by Mozafari method and their bacterial targeting. Micron. 2007;38:841-847.
- [Google Scholar]
- Delivery of melittin-loaded niosomes for breast cancer treatment: an in vitro and in vivo evaluation of anti-cancer effect. Cancer Nanotechnol.. 2021;12:1-35.
- [Google Scholar]
- Antimicrobial resistance: implications and costs. Infect. Drug Resistance.. 2019;12:3903.
- [Google Scholar]
- Biosynthesis of Zn-doped CuFe2O4 nanoparticles and their cytotoxic activity. Scientific Reports. 2022;12:9442.
- [Google Scholar]
- Characterization, optimization, and in vitro evaluation of Technetium-99m-labeled niosomes. Int. J. Nanomed.. 2019;14:1101.
- [Google Scholar]
- Green synthesis of silver nanoparticles from vegetable waste of pea Pisum sativum and bottle gourd Lagenaria siceraria: Characterization and antibacterial properties. Fronti Envir Sci. 2022;10:941554.
- [Google Scholar]
- Enhanced antihypertensive activity of candesartan cilexetil nanosuspension: formulation, characterization and pharmacodynamic study. Sci. Pharm.. 2011;79:635-652.
- [Google Scholar]
- In vitro drug release profile of aceclofenac niosomes formed with different ratios of cholestrol using sorbitan esters. Int. J. Chem. Sc.. 2014;12:237-247.
- [Google Scholar]
- Loading Pistacia atlantica essential oil in solid lipid nanoparticles and its effect on apoptosis of breast cancer cell line MDA-MB-231. Pharm. Dev. Technol.. 2022;27:63-71.
- [Google Scholar]
- From traditional knowledge to modern formulation: potential and prospects of Pistacia atlantica Desf. Essential and fixed oils uses in cosmetics. Cosmetics. 2022;9:109.
- [Google Scholar]
- Evaluation of the antifungal activities of various extracts from Pistacia atlantica Desf. Current Medical Mycol.. 2015;1:25.
- [Google Scholar]
- Inter-component synergetic corrosion inhibition mechanism of Passiflora edulia Sims shell extract for mild steel in pickling solution: Experimental, DFT and reactive dynamics investigations. Sustain Chem Pharm. 2022;29:100821.
- [Google Scholar]
- Optimization and characterization of exopolysaccharide produced by Bacillus aerophilus rk1 and its in vitro antioxidant activities. J King Saud University-Science. 2021;33(5):101470.
- [Google Scholar]
- Preparation and evaluation of reverse-phase evaporation and multilamellar niosomes as ophthalmic carriers of acetazolamide. Int. J. Pharm.. 2005;306:71-82.
- [Google Scholar]
- Evaluation of cytotoxicity, loading, and release activity of paclitaxel loaded-porphyrin based metal-organic framework (PCN-600) Heliyon. 2023;9(1):e12634
- [Google Scholar]
- J Mol Liq. 2021;329:115615.
- [Google Scholar]
- Biomimetic facile synthesis of zinc oxide and copper oxide nanoparticles from Elaeagnus indica for enhanced photocatalytic activity. Environ Res. 2022;212:113323.
- [Google Scholar]
- Entrapment of rosemary extract by liposomes formulated by Mozafari method: physicochemical characterization and optimization. Heliyon.. 2021;7:e08632.
- [Google Scholar]
- Nanoformulation and application of phytochemicals as antimicrobial agents. Antimicrobial nanoarchitectonics. Elsevier; 2017. p. :61-82.
- Multifunctional nanocrystals for cancer therapy: a potential nanocarrier. Nanomater. Drug Delivery Therapy. 2019:91-116.
- [Google Scholar]
- The antimicrobial activity of ciprofloxacin-loaded niosomes against ciprofloxacin-resistant and biofilm-forming staphylococcus aureus. Infect. Drug Resistance.. 2020;13:1619.
- [Google Scholar]
- Controlled anti-cancer drug release through advanced nano-drug delivery systems: Static and dynamic targeting strategies. J. Control. Release. 2020;327:316-349.
- [Google Scholar]
- Potential use of niosomes for encapsulation of nisin and EDTA and their antibacterial activity enhancement. Food Res. Int.. 2011;44:605-612.
- [Google Scholar]
- pH-responsive prodrug nanoparticles based on xylan-curcumin conjugate for the efficient delivery of curcumin in cancer therapy. Carbohydr. Polym.. 2018;188:252-259.
- [Google Scholar]
- Lipophilic 5-fluorouracil prodrug encapsulated xylan-stearic acid conjugates nanoparticles for colon cancer therapy. Int. J. Biol. Macromol.. 2019;128:204-213.
- [Google Scholar]
- Redox responsive xylan-SS-curcumin prodrug nanoparticles for dual drug delivery in cancer therapy. Mater. Sci. Eng. C. 2020;107:110356.
- [Google Scholar]
- Antimicrobial mechanisms of spice essential oils and application in food industry. Food Chem. 2022;382:132312.
- [Google Scholar]
- Met1-specific motifs conserved in OTUB subfamily of green plants enable rice OTUB1 to hydrolyse Met1 ubiquitin chains. Nature Communications. 2022;13(1):4672.
- [Google Scholar]
- Pistacia atlantica Desf. A review of its traditional uses, phytochemicals and pharmacology. J. Med. Life. 2018;11:180.
- [Google Scholar]
- A brief review on niosome drug delivery system. Res. J. Pharmaceut. Dosage Forms Technol.. 2021;13:23-24.
- [Google Scholar]
- Streptomycin sulfate–loaded niosomes enables increased antimicrobial and anti-biofilm activities. Front. Bioeng. Biotechnol.. 2021;9:745099.
- [Google Scholar]
- Synthesis of N-myristoyltaurine stabilized gold and silver nanoparticles: Assessment of their catalytic activity, antimicrobial effectiveness and toxicity in zebrafish. Environ Resea. 2022;212:113159.
- [Google Scholar]
- Nickel oxide nanoparticles synthesis using plant extract and evaluation of their antibacterial effects on Streptococcus mutans. Bioprocess and Biosystems Engineering. 2022;45:1201-1210.
- [Google Scholar]
- Eco-friendly synthesis of carbon nanotubes and their cancer theranostic applications. Mat Advanc. 2022;3(12):4765-4782.
- [Google Scholar]
- Encapsulation of food ingredients using nanoliposome technology. Int. J. Food Prop.. 2008;11:833-844.
- [Google Scholar]
- Medicinal plant compounds for combating the multi-drug resistant pathogenic bacteria: a review. Curr. Pharm. Biotechnol.. 2019;20:183-196.
- [Google Scholar]
- Physical stability of microliposomes in bene (Pistacia atlantica) oil with different formulations. Quality Assurance Saf. Crops Foods.. 2020;12:41-49.
- [Google Scholar]
- Inhibitory effect of cinnamaldehyde on Fusarium solani and its application in postharvest preservation of sweet potato. Food Chem. 2023;408:135213.
- [Google Scholar]
- Antimicrobial application potential of phytoconstituents from turmeric and garlic. In: Bioactive Natural Products for Pharmaceutical Applications. Springer; 2021. p. :409-435.
- [Google Scholar]
- Physical impact of nano-layer on nano-fluid flow due to dispersion of magnetized carbon nano-materials through an absorbent channel with thermal analysis. J Mol Liq. 2021;325:115211.
- [Google Scholar]
- Efficient, highly stable Zn-doped NiO nanocluster electrocatalysts for electrochemical water splitting applications. J Sol-Gel Sci Tech. 2019;89(2):500-510.
- [Google Scholar]
- In vitro characterization and release studies of combined nonionic surfactant-based vesicles for the prolonged delivery of an immunosuppressant model drug. Int. J. Nanomed.. 2020;15:7937.
- [Google Scholar]
- Anti-acne, anti-dandruff and anti-breast cancer efficacy of green synthesised silver nanoparticles using Coriandrum sativum leaf extract. J Photochem Photobio. 2016;163:69-76.
- [Google Scholar]
- Development and characterization of mixed niosomes for oral delivery using candesartan cilexetil as a model poorly water-soluble drug. AAPS PharmSciTech. 2015;16:108-117.
- [Google Scholar]
- In vitro and in vivo evaluation of niosomal formulation for controlled delivery of clarithromycin. Scientifica. 2016:2016.
- [Google Scholar]
- Isolation of exopolysaccharide, galactan from marine Vibrio sp. BPM 19 to template the synthesis of antimicrobial platinum nanocomposite. Process Biochem. 2022;122:267-274.
- [Google Scholar]
- Extraction, Structure and Immunoregulatory Activity of Low Molecular Weight Polysaccharide from Dendrobium officinale. Polymers. 2022;14(14):2899.
- [Google Scholar]
- Anti-adherence and anti-bacterial activities of Pistacia atlantica resin extract against strongly adherent Streptococcus mutans strains. Dental Res. J.. 2022;19
- [Google Scholar]
- Fabrication of GO–MnO2 nanocomposite using hydrothermal process for cationic and anionic dyes adsorption: Kinetics, isotherm, and reusability. J. Environ. Chem. Eng.. 2021;9:106045.
- [Google Scholar]
- Simultaneous capturing of mixed contaminants from wastewater using novel one-pot chitosan functionalized with EDTA and graphene oxide adsorbent. Environ. Pollut.. 2022;304:119130.
- [Google Scholar]
- Multifunctional β-cyclodextrin-EDTA-chitosan polymer adsorbent synthesis for simultaneous removal of heavy metals and organic dyes from wastewater. Environ. Pollut.. 2022;292:118447.
- [Google Scholar]
- Approaches for expedition of discharging of PCM involving nanoparticles and radial fins. J Mol Liq. 2021;329:115052.
- [Google Scholar]
- Dysfunction of FadA-cAMP signalling decreases Aspergillus flavus resistance to antimicrobial natural preservative Perillaldehyde and AFB1 biosynthesis. Environ microbio. 2022;24(3):1590-1607.
- [Google Scholar]
- Membrane-Targeting Neolignan-Antimicrobial Peptide Mimic Conjugates to Combat Methicillin-Resistant Staphylococcus aureus (MRSA) Infections. J Med Chem.. 2022;65(24):16879-16892.
- [Google Scholar]
- Bisphenol A and its analogues in paired urine and house dust from South China and implications for children’s exposure. Chemosphere. 2022;294:133701.
- [Google Scholar]
- Synthesis and application of poly (ethylene glycol)–cholesterol (Chol–PEGm) conjugates in physicochemical characterization of nonionic surfactant vesicles. Colloids Surf. B Biointerfaces. 2008;63:192-199.
- [Google Scholar]
- Brief effect of a small hydrophobic drug (cinnarizine) on the physicochemical characterisation of niosomes produced by thin-film hydration and microfluidic methods. Pharmaceutics.. 2018;10:185.
- [Google Scholar]
- Photoclick Reaction Constructs Glutathione-Responsive Theranostic System for Anti-Tuberculosis. Front Mol Biosci. 2022;9:845179.
- [Google Scholar]
- Zhou, SS., Almarashi, A., Talabany, ZJ.,, Selim, MM., Issakhov, A., Li, YM., Yao, SW., Li, Z., 2021. Augmentation of performance of system with dispersion of nanoparticles inside PCM. J Mol Liq. 2021. 333, 115921.
- Xiong, P., Almarashi, A., Dhahad, A., Alawee, W., Issakhov, A., Chu, Y., 2021. Nanoparticles for phase change process of water utilizing FEM. J Mol Liq. 2021, 334, 116096.
- Begum, S., Shamim, S., F Ameen, F., Z Hussain, Z., SA Bhat, SA., T Qadri, T., Hussain, M., 2022. A Combinatorial Approach towards Antibacterial and Antioxidant Activity Using Tartaric Acid Capped Silver Nanoparticles. Processes. 10(4), 716.
- Almansob, A., AH Bahkali, AH., Ameen, F., 2022. Efficacy of Gold Nanoparticles against Drug-Resistant Nosocomial Fungal Pathogens and Their Extracellular Enzymes: Resistance Profiling towards Established Antifungal Agents. Nanomaterials. 12(5), 814.
- Rajadurai, UM ., Hariharan, A., Durairaj, S., Ameen, F., Dawoud, S Alwakeel, S., 2021. Assessment of behavioral changes and antitumor effects of silver nanoparticles synthesized using diosgenin in mice model. J Drug Delivery Sci Tech. 66, 102766.
- Mohammad, WT., Alijani, H. Faris, P., Salarkia, E., Naderifar, M., 2023. Plant-mediated synthesis of sphalerite (ZnS) quantum dots, Th1-Th2 genes expression and their biomedical applications. South Afr J Botany. 155, 127-139.
- Sonbol, H., F Ameen, F., S AlYahya, S., A Almansob, A., S Alwakeel. S., 2021. Padina boryana mediated green synthesis of crystalline palladium nanoparticles as potential nanodrug against multidrug resistant bacteria and cancer cells. Scientific reports 11 (1), 1-19.







