Translate this page into:
Teriflunomide mitigates pain-depression dyad in mice: Modulation of PI3K-mediated defensive signaling
⁎Corresponding author at: University Institute of Pharma Sciences, Chandigarh University, Mohali, Punjab 140413, India (L. Singh). lovedeep992s@gmail.com (Lovedeep Singh),
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Abstract
The pain-depression dyad refers to a complex and often bidirectional relationship between physical pain and depression. It suggests that these two experiences can interact and exacerbate each other, leading to a cycle of worsening symptoms. Both pain and depression involve changes in brain chemistry and neural pathways. The phosphoinositide 3-kinase (PI3K) signaling pathway often contributes to mood stabilization and in alleviating pain. Teriflunomide is an active metabolite of leflunomide that positively impacts brain functioning. Thus, the current study was designed to investigate the potential of teriflunomide against reserpine-induced pain-depression dyad and also examined the role of PI3K in the observed effect of teriflunomide. Reserpine (0.5 mg/Kg; i.p) treatment for 3 consecutive days significantly induced the pain-depression dyad along with memory impairment in mice. Administration of reserpine significantly reduced the brain glutathione (GSH) level, uplifting the brain thiobarbituric acid reactive substance (TBARS) and serum tumor necrosis factor-alpha (TNF-α) level. Teriflunomide (10 and 20 mg/Kg) treatment significantly reverted the depressive, pain-inducing and memory-impairing effect of reserpine. Both doses of teriflunomide significantly uplifted the brain GSH level and downregulated the serum TNF-α level. Whereas, only a 20 mg/Kg dose of teriflunomide significantly reduced the brain TBARS level. Furthermore, the PI3K inhibitor (LY294002) treatment significantly alleviated the anti-depressant, anti-nociceptive and memory-improving effect of teriflunomide (20 mg/Kg). LY294002 treatment also reverted the teriflunomide-induced biochemical modulations (20 mg/Kg). LY294002 significantly reduced the brain GSH and uplifted the level of TBARS and serum TNF-α level. Conclusively, the results of the current study delineate that teriflunomide might exert its anti-depressant, anti-nociceptive and memory-improving effect by stimulating the PI3K-mediated protection against oxidative and inflammatory stress.
Keywords
Teriflunomide
Pain-depression-dyad
PI3K
Oxidative stress
TNF-α
Memory
1 Introduction
Pain and depression are two widespread and harmful conditions that exert a substantial socioeconomic burden on society. Clinical observations have long acknowledged the simultaneous presence and interplay between pain and depression (Khan et al., 2020). Pain stands as a pivotal determinant in the development of depression, with their concurrent presence serving to exacerbate the intensity of both conditions (Sheng et al., 2017). Research employing clinical interviews with individuals experiencing chronic pain reveals that the prevalence rate for depression ranges from 30 % to 54 %. (Søndergaard et al., 2017). Individuals experiencing chronic pain have a threefold higher likelihood of meeting the diagnostic criteria for depression compared to those who do not have pain, and the risk of developing depression rises as the number of pain symptoms increases (Taloyan and Löfvander, 2014). Moreover, researchers have also suggested that Individuals who encounter depression during the early stages of their life may undergo cognitive impairment because of prolonged episodes of depression (Sarbu et al., 2024). The co-occurrence of pain and depression can be attributed to shared neurobiological pathways that involve various neurotransmitters, such as serotonin, norepinephrine, Substance-P. (Pinheiro et al., 2015; Rijavec and Novak Grubic, 2012). Moreover, there is supporting evidence indicating that the connection between pain and depression may stem from neuronal tissue damage, which includes the involvement of neuroinflammation and oxidative/nitrosative stress processes (Birmann et al., 2019). It has been well established that a reduced level of brain 5-hydroxytryptamine (5-HT) is a fundamental contributing factor to depression (Moncrieff et al., 2023). Evidence delineates that oxidative stress can result in an increased production of neurotoxic compounds through the oxidation of tryptophan, the precursor of 5-HT. Specific molecules like malondialdehyde, which are associated with strong proinflammatory characteristics, have been linked to depression and pain due to their involvement in pathways such as tryptophan/kynurenine interaction (Bakunina et al., 2015). Furthermore, reports have documented that the activation of the phosphatidylinositol 3-kinase/protein kinase B (PI3K/Akt) cascade mediates the analgesic and anti-depressant effects (Guo et al., 2019; You et al., 2023). Stimulation of the PI3K pathway offers protection to neurons and the brain against ischemic and oxidative stress-induced damage (Uranga et al., 2013).
Pain represents a significant feature that frequently co-occurs in individuals with depressive disorder, commonly denoted as the pain-depression dyad. Those experiencing depressive disorders have been observed to articulate a diminished tolerance to pain (Hedgecock et al., 2019). The reserpine-induced pain-depression dyad is a well-accepted mouse model that exhibits behaviors indicative of both pain and depression (Liu et al., 2014a). It has been documented that reserpine-induced increased pain perception in depression might be attributed to the increased level of Substance P in addition to monoamine depletion (Xu et al., 2013; Liu et al., 2014b). Xu et al found that reserpine treatment exaggerated pain response and uplifted the level of Substance P in comparison to the control group (Xu et al., 2013). Moreover, the depletion of monoamines induced by reserpine is attributed to its blocking action on H+-coupled vesicular monoamine transporters-2 (Singh et al., 2021a). Besides the pain-depression dyad, reserpine also has the potential to induce memory impairment and promote oxidative stress in rodents. This memory impairment effect of reserpine could be due to its monoaminergic depletory action (Tseng et al., 2015). Monoamines play a crucial role in various cognitive functions, including memory (Takano, 2018).
Till now, the effective management of the pain-depression dyad in a clinical setting remains a complex task (Roughan et al., 2021). Addressing the pain-depression dyad can be challenging. Treating one condition may positively impact the other, but it's often necessary to address both concurrently (Maallo et al., 2021). In the early days, serotonin noradrenaline reuptake inhibitors (duloxetine) and selective serotonin reuptake inhibitors (paroxetine and sertraline) are frequently prescribed as antidepressant medications for managing concurrent chronic pain and depression (IsHak et al., 2018). However, other clinical reports have consistently shown that the pain-relieving effects of antidepressants can be distinguished from their antidepressant properties (Li, 2015). Addressing pain and depression simultaneously in individuals with comorbidities is crucial. By focusing on the shared underlying mechanisms of both pain and depression, we can enhance our ability to effectively manage these concurrent conditions. Teriflunomide (TFN) is an active metabolite of leflunomide, which is primarily utilized to treat rheumatoid arthritis (Lycke et al., 2023) (Fig. 1). Reports have shown that teriflunomide has a positive impact on brain functioning, preserving neuronal activity and protecting mitochondria in brain slices exposed to oxidative stress. Consequently, teriflunomide has been approved by the U.S. Food and Drug Administration (September 12, 2012) as a once-daily pill for managing relapsing forms of multiple sclerosis (Bar-Or et al., 2014). Scientific evidence indicates that depression can arise from the disturbance of homeostatic mechanisms governing synaptic plasticity. This disruption leads to the destabilization and reduction of synaptic connections within the circuits regulating mood and emotion (Duman and Aghajanian, 2012). The alterations in synaptic plasticity elicited by depression are linked to modifications in both the structure and functionality of the hippocampus (Liu et al., 2017). Furthermore, chronic pain has also been reported to induce profound changes in hippocampal plasticity (Grilli, 2017). In the presence of oxidative stress, teriflunomide has been reported to preserve hippocampal neuronal functionality and avert oxidative stress-induced morphological and motility changes in hippocampal mitochondria (Malla et al., 2022). In light of teriflunomide's potential in mitigating oxidative stress-induced alterations in hippocampal neuronal functioning, the present investigation was designed to evaluate its potential against the reserpine-induced pain-depression dyad, with a specific emphasis on oxidative stress. Moreover, the role of PI3K was also studied in the observed effect of teriflunomide.
Chemical structure of leflunomide and teriflunomide.
2 Materials and methods
2.1 Laboratory animals
This study employed Swiss albino mice of both genders, with body weights ranging from 20 to 30 g. They were acclimatized in the central animal house of Chandigarh University. The mice were given a pellet chow diet and allowed to freely access water. The experiments were conducted within the hours of 09:30 to 17:30 in a semi-soundproof laboratory setting. The experimental protocol received proper approval from the Institutional Animal Ethics Committee (Reg. no. CU/2022/IAEC/7/07). The experiments were conducted following the guidelines set forth by the Committee for the Purpose of Control and Supervision of Experiments on Animals, which operates under the Ministry of Environment, Forest and Climate Change, Government of India.
2.2 Drugs and reagents
Reserpine (CAS. No. 50555) was bought from Sisco Research Laboratories Pvt. Ltd, Maharashtra, India. Mouse TNF-α kit (CAS. No. KB2145) was procured from Krishgen Biosystems, Maharashtra, India. PI3K inhibitor, LY294002 was purchased from Sigma-Aldrich, USA. Teriflunomide (CAS. No. 550028) and imipramine (CAS. No. SSH0677) were procured from Tokyo Chemical Industry (TCI) Pvt. Ltd, Hyderabad, Telangana, India. All additional reagents and chemicals meeting the requirement for analytical standards were obtained from authorized suppliers. All reagents employed in the study were prepared freshly prior to usage.
2.3 Experimental design
Reserpine was administered to mice via intraperitoneal injection (i.p) at a dosage of 0.5 mg/Kg for three consecutive days to induce a pain-depression dyad (Singh et al., 2021b) (Fig. 2). The animals were categorized into six groups: Group 1 served as the vehicle control (0.1 % CMC; p.o), Group 2 animals underwent treatment with 0.1 % CMC (p.o) + reserpine (0.5 mg/Kg; i.p). Group 3 received a combination of imipramine (3 mg/Kg i.p) + reserpine. Animals in Group 4 were subjected to teriflunomide treatment (10 mg/Kg; p.o) + reserpine. Group 5 animals were administered teriflunomide (20 mg/Kg; p.o) + reserpine. Group 6 received the PI3K inhibitor (LY294002) at a dose of 25 µg/kg along with teriflunomide (20 mg/Kg; p.o) and reserpine. The 0.1 % CMC, teriflunomide (10 and 20 mg/Kg, p.o) and imipramine (15 mg/Kg; i.p) were given 30 min prior to reserpine treatment for the first three days. Thereafter 0.1 % CMC, teriflunomide and imipramine were given alone for the rest of 2 days in the respective groups. In the mechanistic group, LY294002 was given 30 min before administering teriflunomide (20 mg/Kg, p.o) treatment. The magnitude of depression and pain was assessed on the 4th and 6th days by using various models such as forced swim test (FST), tail suspension test (TST), tail-flick test (TFT), hot water allodynia, and Eddy’s hot plate test. The Eddy’s hot plate test, Hot allodynia, FST and TST were conducted with a 1-hour interval between them. After an additional 2 h, the MWM test was performed. The sequence of tests conducted was: Eddy’s hot plate test > Hot allodynia > FST > TST > MWM. On day 6 after behavioral analysis, the animals were anesthetized using ketamine (50 mg/Kg i.p), and blood samples were taken for the assessment of serum cytokine analysis. The animals were sacrificed and brain tissues were harvested for biochemical analysis (Fig. 2).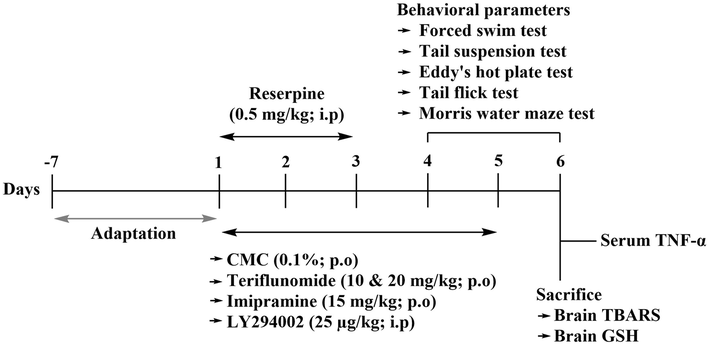
Schematic description of the experimental design.
2.4 Behavioral model
2.4.1 Forced swim test
This experiment was conducted by using the procedure explained by Porsolt (Porsolt et al., 1977). In this examination, the equipment comprises a see-through cylindrical vessel measuring 25 cm in height and 10 cm in diameter. The glass container was filled with water up to such a level that mice did not escape the water container. The temperature of water was consistently held at 25 °C. After the completion of each trial, the water in the container was replaced with fresh water to avoid any bias related to olfaction. Throughout the experiment, each animal was forced to swim for 6 min. During this period the immobility time was recorded (Porsolt et al., 1977).
2.4.2 Tail suspension test
In this test, an animal was suspended at a height of 50 cm from the ground by using adhesive tape. The tape was affixed approximately 1 cm away from the tail's tip. The period of immobility was measured over 4 min out of a total of 6 min. The first 2 min of the test were for the adaptation of mice to the suspension. An animal was considered immobile if it did not show any paw movement (no struggle period) (Liu et al., 2014b).
2.4.3 Eddy’s hot plate test
In the hot plate experiment, mice were positioned on a consistently heated plate set at 55 °C. Twenty seconds was the cutoff time for this test. Before administering a compound, the mouse's initial latency response (measured in seconds) to the thermal stimulus was checked, i.e., either lifting the hind paw or jumping response with all feet on the plate. The post-treatment latency responses were then assessed on the fourth and sixth days after the administration of the compound to the animals (Linciano et al., 2020).
2.4.4 Tail-flick test
For this test, water was maintained at 55 ± 1 °C and was used for recording the tail-flick response. During the test, each mouse received a terry cloth towel wrap before the tail was gently dipped into the hot water. At least 5 cm of the tail was dipped in the water to record the flick response. The cut-off time for this test was 40 sec. Any pharmacological treatment increasing latency to flick the tail in comparison to the disease control group delineates the analgesic potential of that agent (Latif et al., 2021).
2.4.5 Morris water maze test
The setup comprises a circular drum with a smooth surface, measuring 90 cm in diameter and 50 cm in height, along with a 9 cm-diameter circular platform inside the drum. The platform was positioned within one of the four quadrants of the drum. The circular platform was positioned 1 cm below the water's surface. During the test, the water temperature was maintained within the range of 23 to 25 °C to prevent any temperature-related bias. To conceal the platform a nontoxic white color was mixed with the water to make it translucent. An animal was placed into the drum from the direction opposite to the platform and then allowed to swim within the drum. The time taken to find the platform was recorded as a measure of memory. The animal's visit to the platform was deemed a success if it remained on the platform for a minimum of 15 sec. The maximum time of the test was 5 min. Before each experiment, all the mice underwent rigorous training for a minimum of five days to learn how to find a platform (Morris, 1981).
2.5 Biochemical estimations
2.5.1 Processing of brain samples for oxidative stress analysis
The homogenates of isolated brain samples were prepared in phosphate buffer (pH 7.4). 100 mg of minced brain tissue was homogenized in a 100 mL phosphate buffer (pH of 7.4). After homogenization, the blend underwent centrifugation at a speed of 3000 rpm for 15 min. The obtained supernatant was preserved for determining the levels of thiobarbituric acid reactive species (TBARS) and reduced glutathione (GSH).
2.5.2 Determination of brain TBARS level
In this test, the production of pink color as a result of the reaction of thiobarbituric acid (TBA) with malondialdehyde (MDA) and other aldehydes is indicative of lipid peroxidation. In this procedure, 0.1 mL of brain homogenate supernatant was added to a 2 mL (1:1:1) mixture of thiobarbituric acid- trichloro acetic acid- hydrochloric acid. The test tube containing this mixture was subsequently introduced into the water bath and kept there for 15 min at a temperature of 100 °C. Following the cooling process, the mixture underwent centrifugation for 10 min at a speed of 1000 rotations per minute. The clear supernatant was collected and measured at 535 nm. The findings were quantified as µM/mg of tissue (Niehaus and Samuelsson, 1968).
2.5.3 Determination of brain GSH level
The Beutler et al. approach was used with little modification to estimate the GSH level in the mice brain (Beutler et al., 1963). In this test, the interaction of 5.5′- dithiobis (2-nitrobenzoic acid) (DTNB) with the sulphydryl group of GSH results in the production of the yellow color. In this procedure, 0.5 mL of supernatant, 2 mL of 0.3 M disodium hydrogen phosphate buffer (pH 8.4), and 0.25 mL of freshly prepared DTNB were added. The absorbance was measured at 412 nm for the resultant yellow-colored solution. The findings were reported as µM/mg of the tissue mass.
2.5.4 Estimation of serum TNF-α level
Serum samples underwent the TNF-α estimation using the procedure outlined in the ELISA kits provided by Krishgen Biosystems in Mumbai, India.
2.5.4.1 Standards stock preparation
A 0.5 ug/mL concentration was prepared by adding 1.8 µL of a standard solution of mouse TNF-α in 1998.2 µL of assay diluent (1X). Further to make a 1000 pg/mL middle stock solution, 2 µL of the initial standard (0.5 ug/mL) was diluted with 998 µL of the assay diluent (1X). The different concentrations of the standard (450 pg/mL, 225 pg/mL, 112.5 pg/mL, 56.25 pg/mL, 28.13 pg/mL, and 14.06 pg/mL) were prepared by diluting the middle stock solution.
2.5.4.2 Assay procedure
The plate was initially loaded with 100 µL/well of standards and samples, followed by sealing and incubating at 37˚C for 2 h. The plate was subjected to four wash cycles with wash buffer (1X), and any excess buffer was eliminated by firmly inverting the plate onto absorbent paper and tapping it. Liquid present on the outer bottom surface of the microtiter wells was removed to prevent any potential interference with the subsequent reading process. Additionally, each well received 100 µL of the diluted detection antibody solution, followed by sealing the plate and incubating it at 37˚C for 1 h. The plate underwent four washes using wash buffer at a 1X concentration. To this, 100 µL of a diluted Streptavidin-HRP solution was introduced into each well, followed by sealing the plate and incubating it at 37˚C for 30 min. Subsequently, the plate underwent four wash cycles using 1X wash buffer after the incubation period. Thereafter, 100 µL of the TMB substrate solution was introduced and incubated in the dark at 37 °C for 30 min. Consequently, the wells exhibiting a positive response displayed a blue color. The reaction was halted by introducing 100 µL of stop solution into each well. The blue color of the positive wells transformed into a vibrant yellow color. Finally, the absorbance measurement was conducted at 450 nm, and this was done within 30 min after halting the reaction.
2.6 Statistical analysis
The results were displayed in the form of mean ± standard deviation (SD). Statistical significance was determined using GraphPad Prism 7.00 software, employing two-way analysis of variance (ANOVA) for behavioral data and one-way analysis of variance (ANOVA) for biochemical measurements (GraphPad Software Inc. U.S.A.).
3 Results
3.1 Effect on immobility time in the FST
On both day 4 and day 6 of the FST, the administration of reserpine significantly extended the duration of immobility (Day 4: 185.600 ± 10.644 sec and Day 6: 171.400 ± 13.050 sec) when compared to the results observed in the control group (Day 4: 73.296 ± 12.155 sec and Day 6: 76.430 ± 17.430 sec) (Fig. 3A). Standard imipramine administration significantly alleviated the depressive effect of reserpine. A significant reduction in the immobility time was recorded with the imipramine treatment on day 4 and day 6 (Day 4: 95.200 ± 30.392 sec and Day 6: 86.600 ± 22.523 sec). Teriflunomide (10 and 20 mg/Kg) treatment significantly alleviated the depressive effect of reserpine. The observations of day 4 and day 6 showed that teriflunomide (10 mg/Kg) decreased the immobility time (Day 4: 111.400 ± 19.731 sec and Day 6: 98.600 ± 10.738 sec) when compared to the group treated with reserpine alone. Similarly, a 20 mg/kg dose of teriflunomide also reduced the immobility time (Day 4: 100.800 ± 10.060 sec and Day 6: 95.200 ± 4.207 sec) as compared to the reserpine-treated group. In this investigation, PI3K inhibitor, LY2934002 significantly alleviated the anti-depressant effect of teriflunomide (20 mg/Kg). The observations of day 4 and day 6 showed that the LY294002 treatment significantly uplifted the duration of immobility (Day 4: 182.400 ± 24.276 sec and Day 6: 186.400 ± 28.395 sec) as compared to the teriflunomide (20 mg/Kg) treated group (Fig. 3A).![Effect of various pharmacological interventions on immobility time in (A) FST and (B) TST. Two-way analysis of variance (ANOVA) followed by Tukey’s test was used to establish the statistical significance. #: p < 0.05 vs control, *: p < 0.05 vs reserpine and **: p < 0.05 vs TFN (20 mg/Kg). [TFN, teriflunomide].](/content/184/2024/17/4/img/10.1016_j.arabjc.2024.105668-fig3.png)
Effect of various pharmacological interventions on immobility time in (A) FST and (B) TST. Two-way analysis of variance (ANOVA) followed by Tukey’s test was used to establish the statistical significance. #: p < 0.05 vs control, *: p < 0.05 vs reserpine and **: p < 0.05 vs TFN (20 mg/Kg). [TFN, teriflunomide].
3.2 Effect on immobility time in the TST
The reserpine administration resulted in a significant increase in the immobility time on the fourth and sixth day of assessment (Day 4: 240.000 ± 7.906 sec and Day 6: 171.000 ± 22.902 sec) when compared to the control group (Day 4: 93.000 ± 15.604 sec and Day 6: 93.000 ± 15.556 sec) (Fig. 3B). The administration of imipramine at a dosage of 15 mg/Kg significantly reduced the immobility time on the fourth and sixth day (Day 4: 104.400 ± 17.387 sec and Day 6: 97.600 ± 14.450 sec) when compared to the group treated with reserpine. Both doses of teriflunomide (10 and 20 mg/Kg) treatment significantly abolished the depression-inducing effect of reserpine. Teriflunomide (10 mg/kg) significantly reduced the duration of immobility on the 4th and 6th day of observation (Day 4: 134.600 ± 16.227 sec and Day 6: 121.400 ± 9.965 sec) as compared to the group treated with reserpine. Similarly, a 20 mg/kg dose of teriflunomide also reduced the immobility time (Day 4: 124.000 ± 10.724 sec and Day 6: 110.800 ± 8.438 sec) as compared to the reserpine-treated group. Furthermore, LY294002 significantly abolished the anti-depressant effect of teriflunomide (20 mg/Kg). The outcome of this group showed that LY294002 pretreatment significantly increased the immobility time on both days of observation (Day 4: 215.200 ± 14.498 sec and Day 6: 200.800 ± 22.208 sec) when compared to the alone teriflunomide-treated group (Fig. 3B).
3.3 Effect on paw withdrawal latency in the Eddys hot plate test
In this study, the reserpine treatment significantly induced nociception in comparison to the control group (Fig. 4A). The group that received reserpine exhibited a significant reduction in latency time during Eddy's hot plate test (Day 4: 4.680 ± 0.753 sec and Day 6: 4.480 ± 0.756 sec) when compared to the control group (Day 4: 10.002 ± 0.953 sec and Day 6: 9.902 ± 0.770 sec) on both days of assessment. The standard drug imipramine significantly increased the latency time on day 4 and day 6 (Day 4: 8.296 ± 0.541 sec and Day 6: 8.552 ± 0.431 sec) when compared to the reserpine group. At 10 mg/Kg dose, teriflunomide exhibited a significant improvement in latency time on day 4 (5.580 ± 0.760 sec) and day 6 (6.380 ± 0.746 sec) when compared to the group treated with reserpine. Similarly, the higher dose of teriflunomide (20 mg/Kg) also alleviated the nociceptive effect of reserpine. Teriflunomide (20 mg/Kg) significantly increased the latency time on day 4 and day 6 (Day 4: 6.760 ± 0.868 sec and Day 6: 6.880 ± 0.482 sec) when compared to the group treated with reserpine. Pretreatment with PI3K inhibitor, LY294002 significantly abolished the anti-nociceptive effect of teriflunomide. The outcome of this group showed that the LY294002 treatment significantly reduced the latency time (Day 4: 4.060 ± 0.802 sec and Day 6: 3.960 ± 0.915 sec) when compared to the group treated with teriflunomide (20 mg/Kg) (Fig. 4A).![Effect of various pharmacological interventions on (A) paw withdrawal response in Eddy’s hot plate test and (B) tail withdrawal latency in the tail flick test. Two-way analysis of variance (ANOVA) followed by Tukey’s test was used to establish the statistical significance. #: p < 0.05 vs control, *: p < 0.05 vs reserpine and **: p < 0.05 vs TFN (20 mg/Kg). [TFN, teriflunomide].](/content/184/2024/17/4/img/10.1016_j.arabjc.2024.105668-fig4.png)
Effect of various pharmacological interventions on (A) paw withdrawal response in Eddy’s hot plate test and (B) tail withdrawal latency in the tail flick test. Two-way analysis of variance (ANOVA) followed by Tukey’s test was used to establish the statistical significance. #: p < 0.05 vs control, *: p < 0.05 vs reserpine and **: p < 0.05 vs TFN (20 mg/Kg). [TFN, teriflunomide].
3.4 Effect on tail withdrawal latency in tail flick test
The reserpine-treated group significantly decreased the tail-flick time (Day 4: 3.304 ± 0.330 sec and Day 6: 2.828 ± 0.358 sec) when compared to the control group (Day 4: 9.460 ± 0.737 sec and Day 6: 9.480 ± 0.870 sec) on both days of evaluation (Fig. 4B). On both days of assessment, the standard imipramine treatment significantly increased the tail-flick time (Day 4: 7.988 ± 0.766 sec and Day 6: 8.486 ± 1.528 sec) when compared to the group treated with reserpine. The low dose of teriflunomide (10 mg/Kg) significantly increased the tail-flick time on both days (Day 4: 6.070 ± 0.980 sec and Day 6: 5.200 ± 0.825 sec) when compared to the group treated with reserpine. The higher dose of teriflunomide also significantly increased the tail-flick time on both days of evaluation (Day 4: 5.992 ± 1.255 sec and Day 6: 6.400 ± 0.678 sec). The 20 mg/Kg dose of teriflunomide showed a prominent anti-nociceptive effect. The administration of LY294002, a PI3K inhibitor, significantly reversed the analgesic effect of teriflunomide (20 mg/Kg). The group treated with LY294002 showed a significant reduction in tail-flick time (Day 4: 3.640 ± 0.471 sec and Day 6: 3.822 ± 0.357 sec) when compared to the group treated solely with teriflunomide (20 mg/Kg) (Fig. 4B).
3.5 Effect on latency to find the platform in the MWM test
The reserpine treatment had a significant negative impact on spatial memory. On 4th and 6th day, a significant increase in latency time to locate the platform was observed in the group that received reserpine treatment (Day 4: 115.400 ± 25.696 sec and Day 6: 105.400 ± 16.456 sec)when compared to the control group (Day 4: 20.400 ± 10.714 sec and Day 6: 24.600 ± 8.849 sec) (Fig. 5). At a dose of 15 mg/Kg, imipramine significantly decreased the time to locate the platform (Day 4: 73.400 ± 21.314 sec and Day 6: 52.800 ± 15.786 sec) on both days when compared to the reserpine group. The administration of teriflunomide at a dosage of 10 mg/Kg significantly decreased the time to find the platform on the fourth and sixth day of assessment (Day 4: 84.000 ± 26.077 sec and Day 6: 64.400 ± 9.737 sec) when compared to the group treated with reserpine. However, on the fourth day, the effect of a 10 and 20 mg/kg dose of teriflunomide was also significant from the effect noted in the control group. While the higher dose of teriflunomide (20 mg/Kg) effectively eliminated the detrimental impact of reserpine on spatial memory on day 6. Teriflunomide (20 mg/Kg) significantly decreased the time to locate the platform on both day 4 and day 6 (Day 4: 64.800 ± 12.398 sec and Day 6: 50.200 ± 10.450 sec) when compared to the group treated with reserpine. In this study, the spatial memory-enhancing effect of teriflunomide (20 mg/Kg) was counteracted by a PI3K inhibitor, LY294002. LY294002 significantly increased the time (Day 4: 104.800 ± 14.567 sec and Day 6: 105.000 ± 8.573 sec) to find the platform on day 4 and day 6 of the evaluation (Fig. 5).![Effect of various pharmacological interventions on latency to reach the platform on the MWM test. Two-way analysis of variance (ANOVA) followed by Tukey’s test was used to establish the statistical significance. #: p < 0.05 vs control, *: p < 0.05 vs reserpine and **: p < 0.05 vs TFN (20 mg/Kg). [TFN, teriflunomide].](/content/184/2024/17/4/img/10.1016_j.arabjc.2024.105668-fig5.png)
Effect of various pharmacological interventions on latency to reach the platform on the MWM test. Two-way analysis of variance (ANOVA) followed by Tukey’s test was used to establish the statistical significance. #: p < 0.05 vs control, *: p < 0.05 vs reserpine and **: p < 0.05 vs TFN (20 mg/Kg). [TFN, teriflunomide].
3.6 Effect on the brain GSH level
In the present study, reserpine treatment significantly reduced brain GSH levels (0.506 ± 0.073 µM/mg) when compared to the control group (1.079 ± 0.068 µM/mg) (Fig. 6A). Administration of imipramine significantly reverted the GSH-lowering effect of reserpine. Imipramine significantly upregulated the brain GSH level (0.870 ± 0.199 µM/mg) as compared to the reserpine group. Pretreatment with teriflunomide (10 mg/Kg) significantly reverted the GSH lowering effect of reserpine. Teriflunomide (10 mg/Kg) significantly increased the level of GSH in the brain (0.819 ± 0.071 µM/mg) as compared to the reserpine group. The higher dose of teriflunomide (20 mg/Kg) also reverted the GSH lowering effect of reserpine. However, the effect of the 20 mg/Kg dose of teriflunomide was more prominent (0.966 ± 0.068 µM/mg) as compared to the 10 mg/Kg dose of teriflunomide. The outcome of teriflunomide (20 mg/Kg) + LY294002 showed that LY294002 treatment significantly abolished the GSH upregulating effect of teriflunomide (20 mg/Kg). A significant reduction in the brain GSH level was observed with the LY294002 treatment (0.588 ± 0.134 µM/mg) (Fig. 6A).![Effect of various pharmacological interventions on (A) the brain GSH and (B) TBARS level. One-way analysis of variance (ANOVA) followed by Tukey’s test was used to establish the statistical significance. a = p < 0.05 vs control, b = p < 0.05 vs reserpine, c = p < 0.05 vs imipramine, #: p < 0.05 vs control, *: p < 0.05 vs reserpine and **: p < 0.05 vs TFN (20 mg/Kg). [TFN, teriflunomide].](/content/184/2024/17/4/img/10.1016_j.arabjc.2024.105668-fig6.png)
Effect of various pharmacological interventions on (A) the brain GSH and (B) TBARS level. One-way analysis of variance (ANOVA) followed by Tukey’s test was used to establish the statistical significance. a = p < 0.05 vs control, b = p < 0.05 vs reserpine, c = p < 0.05 vs imipramine, #: p < 0.05 vs control, *: p < 0.05 vs reserpine and **: p < 0.05 vs TFN (20 mg/Kg). [TFN, teriflunomide].
3.7 Effect on the brain TBARS level
In the present study, outcomes showed that reserpine treatment significantly uplifted the level of TBARS in the brain (0.512 ± 0.126 nM/mg) as compared to the control group (0.212 ± 0.052 nM/mg) (Fig. 6B). The standard drug imipramine significantly reduced the level of TBARS (0.285 ± 0.081 nM/mg) when compared to the reserpine-treated group. Whereas, the test drug teriflunomide (10 mg/Kg) had no significant effect on the TBARS level (0.441 ± 0.095 nM/mg) in comparison to the reserpine-treated group. Whereas, a higher dose of teriflunomide (20 mg/Kg) significantly reverted the TBARS uplifting effect of reserpine. A significant reduction in the TBARS level was observed with the 20 mg/Kg dose of teriflunomide (0.238 ± 0.082 nM/mg) as compared to the reserpine group. The outcome of teriflunomide (20 mg/Kg) + LY294002 group showed that LY294002 treatment significantly abolished the TBARS lowering effect of teriflunomide (20 mg/Kg). A significant increase in the brain TBARS level was observed with the LY294002 treatment (0.419 ± 0.084 nM/mg) (Fig. 6B).
3.8 Effect on the brain TNF-α level
The reserpine-treated group significantly increased the level of serum TNF-α (458.000 ± 104.370 pg/mL) as compared to the control group (155.667 ± 49.166 pg/mL) (Fig. 7). The standard drug imipramine significantly decreased the level of serum TNF-α (192.667 ± 16.073 pg/mL) in comparison to the reserpine group. Moreover, the outcomes of teriflunomide ( 20 mg/Kg) showed a significant reduction in the serum TNF-α level (267.000 ± 49.122 pg/mL) as compared to the reserpine group. However, no effect was observed on serum TNF-α level with a 10 mg/kg dose of teriflunomide (324.000 ± 50.090 pg/mL) in comparison to the reserpine group. Furthermore, the LY294002 treatment (429.333 ± 92.635 pg/mL) partly reverted the serum TNF-α uplifting effect of teriflunomide (Fig. 7).![Effect of various pharmacological interventions on the serum TNF-α level. One-way analysis of variance (ANOVA) followed by Tukey’s test was used to establish the statistical significance. #: p < 0.05 vs control, *: p < 0.05 vs reserpine and **: p < 0.05 vs TFN (20 mg/Kg). [TFN, teriflunomide].](/content/184/2024/17/4/img/10.1016_j.arabjc.2024.105668-fig7.png)
Effect of various pharmacological interventions on the serum TNF-α level. One-way analysis of variance (ANOVA) followed by Tukey’s test was used to establish the statistical significance. #: p < 0.05 vs control, *: p < 0.05 vs reserpine and **: p < 0.05 vs TFN (20 mg/Kg). [TFN, teriflunomide].
4 Discussion
Depression is a pervasive and enduring medical condition that can impact cognition, emotions, and physical well-being. It manifests through persistent feelings of low mood, diminished energy, sadness, sleep disturbances, and an inability to experience pleasure in life (Cui, 2015). Pain is recognized to have both a sensory dimension (intensity) and an affective dimension (unpleasantness) (Talbot et al., 2019). The coexistence of pain and depression can exert deleterious effects on patient health, as well as impair personal and social functioning (Han and Pae, 2015). Evidence states that Approximately 19 % of adults in Europe and 20.4 % in the United States experience chronic pain, with depression being the most commonly associated comorbidity. The prevalence of depression in European and American societies stands at 7 % and 7.5 %, respectively (Tenti et al., 2022). The use of rodent models has facilitated preclinical research into the mechanisms and manifestations of depression triggered by pain (Singh et al., 2021c). In the present research, reserpine treatment induced the pain-depression-like state in mice. Reserpine significantly increased the immobility time in comparison to the control group of mice in FST and TST. Whereas, decreased the paw and tail withdrawal latency in Eddy’s hot plate and tail flick tests, respectively. A test drug teriflunomide (10 and 20 mg/Kg) significantly reverted the depressive and pain-eliciting effect of reserpine. The results of FST and TST showed a significant decrease in immobility time in comparison to the reserpine group of mice. Whereas, increased the paw and tail withdrawal latency in Eddy’s hot plate and tail flick tests, respectively. Reports delineate that depression can impact cognitive function, including memory. Individuals with depression might experience difficulties with concentration, attention, and memory. This can manifest as forgetfulness, difficulty recalling information, and impaired cognitive abilities. The cognitive symptoms associated with depression are often referred to as depressive cognitive symptoms (Levin et al., 2007). In this study, reserpine treatment uplifted the time to reach the platform in the MWM test. Teriflunomide (10 and 20 mg/Kg) treatment also reverted the memory-impairing effect of reserpine. A significant decrease in time to locate the platform was observed with both doses of teriflunomide. Overall, these results delineate that teriflunomide is an effective analgesic, anti-depressant and memory-consolidating agent at the preclinical step.
LY294002 is a chemical compound that functions as an inhibitor of the PI3K pathway (Huang et al., 2021). The PI3K pathway plays a pivotal role in regulating cell survival, growth and differentiation (Yang et al., 2019). The activation of PI3K is responsible for producing phosphatidylinositol-3,4,5-trisphosphate, which leads to Akt activation. The PI3K/Akt pathway exerts a significant influence on the regulation of multiple signaling pathways, one of which is the glycogen synthase kinase 3 beta (GSK-3β) pathway (Lindahl et al., 2005). GSK3β has been proposed as a potential contributor to the development of major depressive disorder (MDD). GSK3β is involved in the signaling pathways of various neurotransmitters such as serotonin, dopamine, and glutamate, which are implicated in mood regulation (Moreno et al., 2006). PI3K/Akt is known to phosphorylate GSK-3β, resulting in its inactivation (Lindahl et al., 2005). Thus, delineating that PI3K activation negatively regulates the GSK-3β mediated pathophysiological role in depression. Moreover, there have been reports delineating that different pharmacological agents mediate the anti-nociceptive and anti-inflammatory effects via the stimulation of the PI3K/Akt cascade (Zhang et al., 2019; Feng et al., 2020). In the present study, LY294002 treatment significantly alleviated the anti-depressant and anti-nociceptive effect of teriflunomide (20 mg/Kg). Outcomes of LY29002 treatment showed a significant increase in immobility time in FST and TST in comparison to the teriflunomide-treated (20 mg/kg) group. Whereas, decreased the paw and tail withdrawal latency in Eddy’s hot plate and tail flick tests, respectively. Evidence also delineates that activation of the PI3K pathway is associated with the early stages of learning and memory processes. It plays a role in the molecular processes that support the transformation of short-term memories into more enduring long-term memories (Santini et al., 2014). In this investigation, LY294002 treatment significantly alleviated the memory-consolidating effect of teriflunomide (20 mg/Kg). LY294002 treatment significantly increased in time to locate the platform in comparison to the teriflunomide (20 mg/kg) treated group. Thus, these outcomes delineate that teriflunomide might exert its anti-depressant, anti-nociceptive and memory-improving effect by activating the PI3K pathway.
The interplay between pain and depression can arise due to neuronal tissue damage, which is associated with the occurrence of neuroinflammation as well as oxidative and nitrosative stress processes (Birmann et al., 2019). Inflammatory agents in the brain play a crucial role in sustaining the connection between depression and pain, potentially serving as underlying components of this syndrome (Campos et al., 2020). GSH has been reported to play a role in regulating oxidative stress and inflammation. These processes, in turn, may have an indirect impact on pain perception. Individuals with chronic pain conditions might have reduced glutathione levels (Borkum, 2016). Evidence states that GSH mediates protection against oxidative or nitrosative stress, either through its direct interaction with reactive oxygen or nitrogen species or as a necessary cofactor for GSH S-transferases and glutathione peroxidases (Morris et al., 2014). In the present study, reserpine treatment significantly reduced the brain GSH and increased brain TBARS and serum TNF-α levels. Teriflunomide treatment significantly uplifted the brain GSH level (10 and 20 mg/Kg) and reduced the brain TBARS level (20 mg/Kg). Moreover, both doses of teriflunomide also downregulated the serum TNF-α level. Furthermore, the available evidence suggests that the PI3K/Akt pathway plays a pivotal role in removing reactive oxygen species, thus, maintaining the redox balance (Koundouros and Poulogiannis, 2018). The PI3K/Akt pathway has a significant role in controlling the protective response to oxidative stress through the Nrf2-ARE antioxidant pathway (de Sousa et al., 2014). In this study, LY294002 treatment significantly alleviated the GSH-increasing and TBARS-lowering effect of teriflunomide. LY294002 treatment significantly increased and decreased the brain TBARS and GSH levels. Whereas, no significant effect was observed on serum TNF-α levels. Thus, delineating that teriflunomide might alleviate oxidative stress by stimulating the PI3K pathway. Conclusively, the results of the current study delineate that teriflunomide might exert its anti-depressant, anti-nociceptive and memory-improving effect by stimulating the PI3K-mediated protection against oxidative stress. Though this study provides valuable insights into the potential of teriflunomide in alleviating the pain-depression dyad, it does have certain limitations. In this study, only TBARS and GSH estimations were performed to assess the oxidative stress levels in biological systems. Moreover, elucidating the effect of teriflunomide on the brain Nrf-2 and HO-1 could offer valuable insights for potential therapeutic strategies. Additionally, the initial assessment of the compound's impact on TNF-α levels serves as a starting point for prospective studies focused on modulating inflammatory pathways. Even though the broader objectives may not have been entirely achieved, this study has laid the foundation for future research to delve deeper insight into the oxidative and inflammatory stress pathways in the protective effect of teriflunomide against the pain-depression dyad.
Ethics approval
The experimental protocol received proper approval from the Institutional Animal Ethics Committee (IAEC) (Reg. no. CU/2022/IAEC/7/07). The experiments were conducted following the guidelines set by the Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA), under the Ministry of Environment and Forest, Government of India.
Funding
The research leading to these results has received funding from Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia under Researchers Supporting Project (PNURSP2024R342). This study also received funding under the Researchers Supporting Project (RSPD2024R812) from King Saud University, Riyadh, Saudi Arabia.
CRediT authorship contribution statement
Kamal Nabi: Methodology, Validation, Formal analysis, Investigation, Resources, Writing – original draft. Lovedeep Singh: Conceptualization, Methodology, Software, Validation, Formal analysis, Investigation, Resources, Data curation, Writing – original draft, Writing – review & editing, Supervision. Atul Kabra: Resources, Software, Writing – review & editing. Ashwag S. Alanazi: Writing – review & editing, Funding acquisition. Hany W. Darwish: Writing – review & editing, Funding acquisition. Bader Alsuwayt: Writing – review & editing, Funding acquisition.
Acknowledgments
The authors extend their appreciation to Princess Nourah bint Abdulrahman University Researchers Supporting Project number (PNURSP2024R342), Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia for supporting this research. The authors also extend their appreciation to the Researchers Supporting Project number (RSPD2024R812), King Saud University, Riyadh 11451, Saudi Arabia for supporting this research. Additionally, the authors are grateful to the University Institute of Pharma Sciences at Chandigarh University for providing laboratory resources.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Immune mechanisms linked to depression via oxidative stress and neuroprogression. Immunology. 2015;144(3):365-373.
- [Google Scholar]
- Teriflunomide and its mechanism of action in multiple sclerosis. Drugs. 2014;74:659-674.
- [Google Scholar]
- An improved method for the determination of blood glutathione. J. Lab. Clin. Med.. 1963;61:882-888.
- [Google Scholar]
- 3-(4-Chlorophenylselanyl)-1-methyl-1H-indole promotes recovery of neuropathic pain and depressive-like behavior induced by partial constriction of the sciatic nerve in mice. J. Trace Elem. Med Biol.. 2019;54:126-133.
- [Google Scholar]
- Migraine triggers and oxidative stress: a narrative review and synthesis. Headache: The Journal of Head and Face Pain. 2016;56(1):12-35.
- [Google Scholar]
- Neuroinflammation, pain and depression: an overview of the main findings. Front. Psychol.. 2020;11:1825.
- [Google Scholar]
- Oxidative stress in early stage bipolar disorder and the association with response to lithium. J. Psychiatr. Res.. 2014;50:36-41.
- [Google Scholar]
- Synaptic dysfunction in depression: potential therapeutic targets. Science. 2012;338(6103):68-72.
- [Google Scholar]
- Antinociceptive effects of Shenling Baizhu through PI3K-Akt-mTOR signaling pathway in a mouse model of bone metastasis with small-cell lung cancer. Evid. Based Complement. Alternat. Med. 2020
- [Google Scholar]
- Chronic pain and adult hippocampal neurogenesis: translational implications from preclinical studies. J. Pain Res.. 2017;10:2281-2286.
- [Google Scholar]
- Baicalin ameliorates neuroinflammation-induced depressive-like behavior through inhibition of toll-like receptor 4 expression via the PI3K/AKT/FoxO1 pathway. J. Neuroinflammation. 2019;16(1):1-21.
- [Google Scholar]
- Pain and depression: a neurobiological perspective of their relationship. Psychiatry Investig.. 2015;12(1):1-8.
- [Google Scholar]
- Molecular mechanisms and applications of a reserpine-induced rodent model [molecular mechanisms and applications of a reserpine-induced rodent model] SSR Institute of International Journal of Life Sciences. 2019;5(1):2160-2167.
- [Google Scholar]
- LY294002 is a promising inhibitor to overcome sorafenib resistance in FLT3-ITD mutant AML cells by interfering with PI3K/Akt signaling pathway. Front. Oncol.. 2021;11:782065
- [Google Scholar]
- Pain and depression: a systematic review. Harv. Rev. Psychiatry. 2018;26(6):352-363.
- [Google Scholar]
- Twin studies of the covariation of pain with depression and anxiety: a systematic review and re-evaluation of critical needs. Neurosci. Biobehav. Rev.. 2020;111:135-148.
- [Google Scholar]
- Phosphoinositide 3-kinase/Akt signaling and redox metabolism in cancer. Front. Oncol.. 2018;8:160.
- [Google Scholar]
- Bergapten attenuates nitroglycerin-induced migraine headaches through inhibition of oxidative stress and inflammatory mediators. ACS Chem. Nerosci.. 2021;12(18):3303-3313.
- [Google Scholar]
- Cognitive deficits in depression and functional specificity of regional brain activity. Cogn. Ther. Res.. 2007;31:211-233.
- [Google Scholar]
- Pain and depression comorbidity: a preclinical perspective. Behav. Brain Res.. 2015;276:92-98.
- [Google Scholar]
- Identification of a potent and selective 5-HT1A receptor agonist with in vitro and in vivo antinociceptive activity. ACS Chem. Nerosci.. 2020;11(24):4111-4127.
- [Google Scholar]
- Prevalence of suicidality during pregnancy and the postpartum. Archives of Women’s Mental Health.. 2005;8:77-87.
- [Google Scholar]
- The role of neural plasticity in depression: from hippocampus to prefrontal cortex. Neural Plast.. 2017;2017:6871089.
- [Google Scholar]
- Attenuation of reserpine-induced pain/depression dyad by gentiopicroside through downregulation of GluN2B receptors in the amygdala of mice. NeuroMol. Med.. 2014;16:350-359.
- [Google Scholar]
- Teriflunomide concentrations in cerebrospinal fluid and plasma in patients with multiple sclerosis: a pharmacokinetic study. CNS Drugs. 2023;37(2):181-188.
- [Google Scholar]
- A lateralized model of the pain-depression dyad. Neurosci. Biobehav. Rev.. 2021;127:876-883.
- [Google Scholar]
- Teriflunomide preserves neuronal activity and protects mitochondria in brain slices exposed to oxidative stress. Int. J. Mol. Sci.. 2022;23(3):1538.
- [Google Scholar]
- The serotonin theory of depression: a systematic umbrella review of the evidence. Mol. Psychiatry. 2023;28(8):3243-3256.
- [Google Scholar]
- Sex differences in depressive response during monoamine depletions in remitted depressive subjects. J. Clin. Psychiatry. 2006;67(10):1618.
- [Google Scholar]
- Spatial localization does not require the presence of local cues. Learn. Motiv.. 1981;12(2):239-260.
- [Google Scholar]
- The glutathione system: a new drug target in neuroimmune disorders. Mol. Neurobiol.. 2014;50:1059-1084.
- [Google Scholar]
- Formation of malonaldehyde from phospholipid arachidonate during microsomal lipid peroxidation. Eur. J. Biochem.. 1968;6(1):126-130.
- [Google Scholar]
- Symptoms of depression and risk of new episodes of low back pain: a systematic review and meta-analysis. Arthritis Care Res.. 2015;67(11):1591-1603.
- [Google Scholar]
- Behavioral despair in mice: a primary screening test for antidepressants. Arch. Int. Pharmacodyn. Ther.. 1977;229(2):327-336.
- [Google Scholar]
- Depression and pain: often together but still a clinical challenge-a review. Psychiatr. Danub.. 2012;24(4):346-352.
- [Google Scholar]
- Comorbid chronic pain and depression: shared risk factors and differential antidepressant effectiveness. Front. Psych.. 2021;12:643609
- [Google Scholar]
- Mechanisms of translation control underlying long-lasting synaptic plasticity and the consolidation of long-term memory. Prog. Mol. Biol. Transl. Sci.. 2014;122:131-167.
- [Google Scholar]
- Early depressive manifestations in patients with dementia caused by Alzheimer's disease. Exp. Ther. Med.. 2024;27(2):1-15.
- [Google Scholar]
- The link between depression and chronic pain: neural mechanisms in the brain. Neural Plast.. 2017;2017:9724371.
- [Google Scholar]
- Skimmetin/osthole mitigates pain-depression dyad via inhibiting inflammatory and oxidative stress-mediated neurotransmitter dysregulation. Metab. Brain Dis.. 2021;36:111-121.
- [Google Scholar]
- Biological evaluation of aegle marmelos fruit extract and isolated aegeline in alleviating pain-depression dyad. in silico analysis of aegeline on MAO-A and iNOS. ACS Omega. 2021;6(3):2034-2044.
- [Google Scholar]
- Prevalence of depression and anxiety in patients with chronic non-malignant pain–a Danish register-linkage cohort study. Eur. Psychiatry. 2017;41(S1):313.
- [Google Scholar]
- Cognitive function and monoamine neurotransmission in schizophrenia: evidence from positron emission tomography studies. Front. Psych.. 2018;9:28.
- [Google Scholar]
- The sensory and affective components of pain: are they differentially modifiable dimensions or inseparable aspects of a unitary experience? A systematic review. British Journal of Anaesthesia. 2019;123:263-272.
- [Google Scholar]
- Depression and gender differences among younger immigrant patients on sick leave due to chronic back pain: a primary care study. Prim. Health Care Res. Dev.. 2014;15(1):5-14.
- [Google Scholar]
- A narrative review of the assessment of depression in chronic pain. Pain Manag. Nurs.. 2022;23(2):158-167.
- [Google Scholar]
- (−) Epigallocatechin-3-gallate prevents the reserpine-induced impairment of short-term social memory in rats. Behav. Pharmacol.. 2015;26(8–9):741-747.
- [Google Scholar]
- Enhanced phosphatidylinositol 3-kinase (PI3K)/Akt signaling has pleiotropic targets in hippocampal neurons exposed to iron-induced oxidative stress. J. Biol. Chem.. 2013;288(27):19773-19784.
- [Google Scholar]
- Ferulic acid increases pain threshold and ameliorates depression-like behaviors in reserpine-treated mice: behavioral and neurobiological analyses. Metab. Brain Dis.. 2013;28:571-583.
- [Google Scholar]
- Targeting PI3K in cancer: mechanisms and advances in clinical trials. Mol. Cancer. 2019;18(1):26.
- [Google Scholar]
- Taselisib moderates neuropathic pain through PI3K/AKT signaling pathway in a rat model of chronic constriction injury. Brain Res. Bull.. 2023;199:110671
- [Google Scholar]
- Antinociceptive and anti-inflammatory effects of cryptotanshinone through PI3K/Akt signaling pathway in a rat model of neuropathic pain. Chem. Biol. Interact.. 2019;305:127-133.
- [Google Scholar]







