Translate this page into:
The study of different markers among Chuanxiong Chatiao dosage forms based on bioactive components and antiplatelet aggregation biopotency integrated with chemometrics
⁎Corresponding authors. Y13592925434@126.com (Yi-xin Yao)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University. Production and hosting by Elsevier.
Abstract
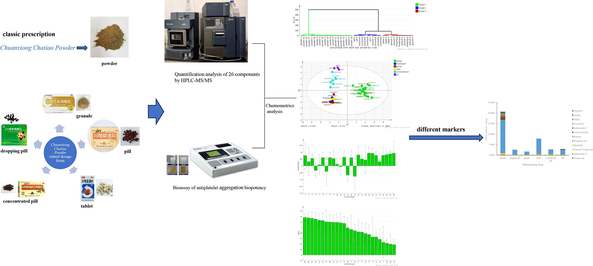
Abstract
High performance liquid chromatography with triple quadrupole mass spectrometry method was developed for the determination of 26 bioactive components of the six Chuanxiong Chatiao dosage forms. Parallel line assay based on quantitative responses (2∙2) method of the biological assay was established to measure the potency of antiplatelet aggregation bioactivity in vitro of the six Chuanxiong Chatiao dosage forms. The different markers were screened by partial least squares based on 26 active components and antiplatelet aggregation among the six Chuanxiong Chatiao dosage forms.
Abstract
Chuanxiong Chatiao powder is a classic prescription in traditional Chinese medicine. A variety of dosage forms have been developed based on powders, such as dropping pills, granules, tablets, pills, and concentrated pills. This classic prescription consists of various active constituents, but due to the selection of a single and unreasonable quality standard indicator, may cause a difference in active constituent and bioactivity among different dosage forms. However, related reports on the comparisons of the different dosage forms are few. Therefore, high-performance liquid chromatography with triple quadrupole mass spectrometry was developed for the determination of 26 bioactive components of the six Chuanxiong Chatiao dosage forms, and a parallel line assay based on a quantitative response method for biological assays was established to measure the antiplatelet aggregation biopotency (AAB) in vitro. The results indicated that the AAB of the powder was greatest, followed by that of the pills, concentrated pills, dropping pills, tablets, granules. The results were analyzed by hierarchical clustering analysis, showing that were some differences among six dosage forms. 12 different markers, including Z- ligustilide, senkyunolide A, butylidenephthalide, asarinin, rosmarinic acid, pulegone, senkyunolide H, notopterol, coniferyl ferulate, hesperidin, vanillic acid, luteolin-7-O-glucoside, were further screened using partial least squares. All of different markers showed a positive association with AAB. There were significant differences in both the sum and composition of different markers among the different dosage forms. This can provide a foundation for the clinical study of the six Chuanxiong Chatiao dosage forms.
Keywords
Chuanxiong Chatiao dosage forms
Bioactive components
Quantification
Antiplatelet aggregation biopotency
Bioassay
Different markers
1 Introduction
Chuanxiong Chatiao powder is a classic prescription in traditional Chinese medicine that is used for treating headaches caused by externally contracted colds that are sometimes accompanied by chills, fever, and a stuffy nose. Chuanxiong Chatiao powder comprises eight traditional herbs: Chuanxiong rhizoma (Chuanxiong, CX), Notopterygii rhizoma et radix (Qianghuo, QH), Angelicae dahuricae radix (Baizhi, BZ), Asari radix et rhizoma (Xixin, XX), Schizonepetae herba (Jinjie, JJ), Saposhnikoviae radix (Fangfeng, FF), Menthae Haplocalycis herba (Bohe, BH), and Glycyrrhizae radix et rhizoma (Gancao, GC) (National Commission of Chinese Pharmacopoeia, 2020a, 2020b). Modern studies have shown that Chuanxiong Chatiao powder has a favorable effect on migraines related to vasodilator headaches, including the improvement of platelet function and blood rheology levels by reducing high shear viscosity, low-cut viscosity, plasma viscosity, and fibrinogen (Wang et al., 2019). Chuanxiong Chatiao powder contains a wide variety of active components (Wang et al., 2020a, 2020b), for example, ferulic acid, senkyunolide I, 5-0-methylvisamidol glycoside, prim-o-glucosylcimifugin, quercetin, chlorogenic acid, caffeic acid, rosmarinic acid, butylidenephthalide, coniferyl ferulate, Z-ligustilide, vanillic acid, quercitrin, and levistilide A can inhibit platelet aggregation (Li et al., 2022; Gao et al., 2016; Fuentes et al., 2014; Zhu et al., 2015; Lu et al., 2015; Zou et al., 1993; Mei et al., 2023; Kong et al., 2020; Kim et al., 1999; Yao, 2017). Additionally, senkyunolide A and senkyunolide H are also closely associated with antiplatelet aggregation (Zhang et al., 2019). Asarinin can inhibit the formation of thrombin (Ku et al., 2013). Imperatorin can promote vasodilatation on arteries precontracted (Zhao et al., 2022). Buddleoside can inhibit the proliferation and migration of vascular smooth muscle cell (Su et al., 2019). Hesperidin can improve vascular morphology and regulate blood pressure (Ohtsuki et al., 2003). Luteolin-7-O-glucoside can protect the vascular endothelium injury (Chen et al., 2022b, 2022a). However, inflammation can be detrimental to cardiovascular diseases as it interacts with the cardiovascular dangerous factors, creating a vicious cycle (Luca et al., 2020). Many components in Chuanxiong Chatiao powder exert significant anti-inflammatory effect, such as hesperidin, buddleoside, Z-ligustilide, senkyunolide A, senkyunolide H, liquiritin, glycyrrhizic acid, luteolin-7-O-glucoside, quercitrin, isoimperatorin, asarinin, imperatorin, pulegone, and notopterol (Choi et al., 2022; Yang et al., 2022; Li et al., 2022; Chen et al., 2021a, 2021b; De Stefano et al., 2021; Sophiya et al., 2022; Zhao et al., 2022; Yang et al., 2019; Liu and Wang, 2022; Azietaku et al., 2017). This indicates that various bioactive ingredients in Chuanxiong Chatiao powder can directly or indirectly act on vasodilator headaches. Furthermore, certain ingredients exhibit antifungal effect that could provide valuabe insights for the development of antifungal drugs. For instance, Z-ligustilide and senkyunolide A in CX show varying inhibitory effects on Botryis cinerea, Fusarium graminearum, Fusarium oxysporum and Colletotrichum gloeosporioides (Chen et al., 2022b, 2022a). Asarinin could influence dihomo-γ-linolenic acid in Mortierella alpina, consequently affecting the content of arachidonic acid in fungi (Shimizu et al., 1992). Caffeic acid display antifungal activity against Candidaalbicans (Sardia et al., 2016). The chlorogenic acid exhibit antifungal activity in vitro by decreasing cell viability, improving potential for mitochondrial depolarization and formation of reactive oxygen species, phosphatidylserine externalization and DNA fragmentation (Rocha da Silva et al., 2022). However, one of the effective components of CX and QH, ferulic acid, which was selected as the quality control indicator in the Chinese Pharmacopoeia 2020 edition is clearly unreasonable (National Commission of Chinese Pharmacopoeia, 2020a, 2020b).
In recent decades, many new dosage forms had been researched using Chuanxiong Chatiao powder, such as dropping pills, granules, tablets, concentrated pills, and pills. Their efficacies were the same as Chuanxiong Chatiao powder in clinical treatment. However, quality control indicators pose issues when one or only a few indicators are used, and applied inconsistently across dosage forms. For example, glycyrrhizic acid was selected as the quality control indicator for dropping pills and concentrated pills, ferulic acid and glycyrrhizic acid for granules, and ferulic acid for tablets and pills, and therefore it is difficult to comprehensively control the quality. (National Commission of Chinese Pharmacopoeia, 2020a, 2020b; State Food and Drug Administration, 2009). As a result of a lack of quality standards, the types and contents of bioactive constituents may differ by different preparation methods, which may eventually lead to differences in clinical efficacy. A study had measured the in vitro dissolution of four analytes of Chuanxiong Chatiao powders and pills, but the number of comparison constituents was too few to be comprehensive (Qu et al., 2022). No other relevant research comparing and analyzing these dosage forms were performed from the perspective of multiple bioactive components.
Based on their clinical effect is closely related to vasodilator headaches, activation of platelet function is one of the important influencing factors, which mainly reflects the aggregation activity of platelets (Wang et al., 2019). Adenosine phosphate (ADP) is a powerful substance causing platelet aggregation (Zhang et al., 2019). Therefore, antiplatelet aggregation bioactivity induced by ADP can be selected to reveal the efficacy of Chuanxiong Chatiao powder and related dosage forms in treating migraines.
For the purpose of this study, the differences in active composition and bioactivity among the six Chuanxiong Chatiao dosage forms are to be compared. High performance liquid chromatography with triple quadrupole mass spectrometry (HPLC-MS/MS) was used to identify the 26 bioactive components (chlorogenic acid, caffeic acid, vanillic acid, ferulic acid, prim-O-glucosylcimifugin, liquiritin, pulegone, luteolin-7-O-glucoside, senkyunolide I, senkyunolide H, quercitrin, 5-O-methylvisammioside, hesperidin, rosmarinic acid, quercetin, buddleoside, senkyunolide A, glycyrrhizic acid, coniferyl ferulate, Z-ligustilide, butylidenephthalide, imperatorin, asarinin, notopterol, isoimperatorin, and levistilide A) of Chuanxiong Chatiao powder, dropping pills, granules, tablets, concentrated pills, and pills. And a parallel line assay based on a quantitative response (2∙2) method for biological assays was established to measure the antiplatelet aggregation biopotency (AAB) in vitro induced. Meanwhile, the different markers were analyzed among the six dosage forms. This could provide a foundation for the clinical study of the six Chuanxiong Chatiao dosage forms.
2 Materials and methods
2.1 Reagents and chemicals
Standards of vanillic acid, ferulic acid, senkyunolide I, senkyunolide H, coniferyl ferulate, Z-ligustilide, senkyunolide A, butylidenephthalide and levistilide A (Purities ≥ 98 % by HPLC) were obtained from Chengdu Pufei De Biotech Co., Ltd. (Chengdu, China). Chlorogenic acid (96.3 % Pure), caffeic acid (99.7 % Pure), prim-O-glucosylcimifugin (94.6 % Pure), liquiritin (95.0 % Pure), Luteolin-7-O-glucoside (96.6 % Pure), 5-O-Methylvisammioside (97.4 % Pure), quercitrin (93.5 % Pure), hesperidin (95.3 % Pure), quercetin (99.8 % Pure), rosmarinic acid (98.1 % Pure), buddleoside, (98.0 % Pure), pulegone (99.8 % Pure), ammonium glycyrrhizinate (94.4 % Pure), imperatorin (99.0 % Pure), asarinin (100.0 % Pure), notopterol (97.5 % Pure), isoimperatorin (99.2 % Pure) were purchased from national institutes for food and drug control (Beijing, China). Methanol and acetonitrile were HPLC grade, adenosine-5-two sodium phosphate (C10H13N5Na2O10P2, ADP), sodium ferulic acid, and dimethyl sulfoxide (DMSO) were obtained from Sigma-Aldrich Co (St. Louis, MO, USA). Formic acid was analytical grade and purchased from Guangzhou Chemical Reagent Factory (Guangzhou, China). All aqueous solutions were prepared and produced by ultra-Pure water with a Milli-Q system (18.2 MΩ, Millipore, Bedford, MA, USA). Tri Sodium citrate dihydrate C6H5Na3O7·2(H2O) were analytical grade and acquired from Guangzhou Chemical Reagent Factory (Guangzhou, China). Sodium chloride injection (0.9 %, W/V) was obtained from Sichuan Cologne Pharmaceutical Co., Ltd. (Chengdu, China).
2.2 Instrumentation
KQ-500VDE double frequency digital ultrasonic cleaning instrument was obtained from Kunshan Ultrasonic Instrument Co., Ltd. (Kunshan, China). All the analytes were performed using a Waters Acquity HPLC-MS/MS system (Waters Corp., Milford, MA, USA). HPLC system which consisted of an online degasser, an autosampler, and a binary pump solvent management system. Mass spectrometry detection was performed using a Xevo Triple Quadrupole MS (Waters Corp.) equipped with an electrospray ionization source (ESI). The ESI-MS spectra were obtained using multiple reaction monitoring (MRM). Separation was achieved on an Agilent Eclipse Plus Cl8 column (5 μm, 4.6 mm × 250 mm). BSA224S Precision electronic balance was acquired from Beijing Sartorius Scientific Instrument Co., Ltd. (Beijing, China). ZNHW-II Intelligent digital display electric hearting set was acquired from Gongyi Yuhua Instrument Co., Ltd. (Tianjin, China). Frontier™ 5000 Multi Pro multi-function centrifuge was obtained from OHAUS Company (Pine Brook, USA); RE-2000B rotatory evaporator was acquired from Shanghai Yarong Biochemical Instrument Factory (Shanghai, China). SC-2000 Platelets aggregated instrument was obtained from Beijing Succeeder Technology Development Co., Ltd. (Beijing, China).
2.3 Sample collection
CX, QH, BZ, XX, JJ, FF, BH, and GC were collected from the Hehuachi Chinese herbal medicine market of Chengdu, Sichuan province, and the traditional Chinese medicine market of Puning, Guangdong province. There were 20 samples and they were further identified as certified goods by chief pharmacist Liu Maogui, director of the quality management department, Kangmei Pharmaceutical Co., Ltd. CX is a dried rhizome of Ligusticum chuanxiong Hort.; QH is the dried rhizome and root of Notopterygium incisum Ting ex H. T. Chang.; BZ is the dried root of Angelica dahurica (Fisch. ex Hoffm.) Benth. et Hook. f.; XX is the dried rhizome and root of Asarum heterotropoides Fr. Schmidt var. mandshuricum (Maxim.) Kitag.; JJ is the dried aerial part of Schizonepeta tenuifolia Briq.; BH is the dried aerial part of Mentha haplocalyx Briq.; and GC is the dried rhizome and root of Glycyrrhiza uralensis Fisch. Twenty samples of Chuanxiong Chatiao powder were prepared with 120 g of CX, 60 g of QH, 60 g of BZ, 30 g of XX, 120 g of JJ, 45 g of FF, 240 g of BH, and 60 g of GC, which had been pulverized to a fine powder, sifted, and mixed well (National Commission of Chinese Pharmacopoeia, 2020a, 2020b). Chuanxiong Chatiao dropping pills, granules, tablets, concentrated pills, and pills were collected from different factories. All sample information is shown in Table 1.
No.
Sample
Ingredients/Producer
Batch number
1
powder-1
CX-1,QH-1,BZ-1,FF-1,BH-1,JJ-1,XX-1,GC-1
/
2
powder-2
CX-2,QH-2,BZ-2,FF-2,BH-2,JJ-2,XX-2,GC-2
/
3
powder-3
CX-3,QH-3,BZ-3,FF-3,BH-3,JJ-3,XX-3,GC-3
/
4
powder-4
CX-4,QH-4,BZ-4,FF-4,BH-4,JJ-4,XX-4,GC-4
/
5
powder-5
CX-5,QH-5,BZ-5,FF-5,BH-5,JJ-5,XX-5,GC-5
/
6
powder-6
CX-6,QH-6,BZ-6,FF-6,BH-6,JJ-6,XX-6,GC-6
/
7
powder-7
CX-7,QH-7,BZ-7,FF-7,BH-7,JJ-7,XX-7,GC-7
/
8
powder-8
CX-8,QH-8,BZ-8,FF-8,BH-8,JJ-8,XX-8,GC-8
/
9
powder-9
CX-9,QH-9,BZ-9,FF-9,BH-9,JJ-9,XX-9,GC-9
/
10
powder-10
CX-10,QH-10,BZ-10,FF-10,BH-10,JJ-10,XX-10,GC-10
/
11
powder-11
CX-11,QH-11,BZ-11,FF-11,BH-11,JJ-11,XX-11,GC-11
/
12
powder-12
CX-12,QH-12,BZ-12,FF-12,BH-12,JJ-12,XX-12,GC-12
/
13
powder-13
CX-13,QH-13,BZ-13,FF-13,BH-13,JJ-13,XX-13,GC-13
/
14
powder-14
CX-14,QH-14,BZ-14,FF-14,BH-14,JJ-14,XX-14,GC-14
/
15
powder-15
CX-15,QH-15,BZ-15,FF-15,BH-15,JJ-15,XX-15,GC-15
/
16
powder-16
CX-16,QH-16,BZ-16,FF-16,BH-16,JJ-16,XX-16,GC-16
/
17
powder-17
CX-17,QH-17,BZ-17,FF-17,BH-17,JJ-17,XX-17,GC-17
/
18
powder-18
CX-18,QH-18,BZ-18,FF-18,BH-18,JJ-18,XX-18,GC-18
/
19
powder-19
CX-19,QH-19,BZ-19,FF-19,BH-19,JJ-19,XX-19,GC-19
/
20
powder-20
CX-20,QH-20,BZ-20,FF-20,BH-20,JJ-20,XX-20,GC-20
/
21
dropping pill-1
Guizhou Jianxing Pharmaceutical Co., Ltd.
20,171,104
22
dropping pill-2
Guizhou Jianxing Pharmaceutical Co., Ltd.
20,181,104
23
dropping pill-3
Guizhou Jianxing Pharmaceutical Co., Ltd.
20,190,102
24
granule-1
Jilin Zixin Pharmaceutical Industrial Co., Ltd.
171,101
25
granule-2
Jilin Zixin Pharmaceutical Industrial Co., Ltd.
171,103
26
granule-3
Hunan Anbang Pharmaceutical Co., Ltd.
180,602
27
granule-4
Sinopharm Group Dezhong (Foshan) Pharmaceutical Co., Ltd.
18,003
28
granule-5
Guizhou Warmen Pharmaceutical Co., Ltd.
170,801
29
granule-6
Guizhou Warmen Pharmaceutical Co., Ltd.
180,102
30
granule-7
Guizhou Warmen Pharmaceutical Co., Ltd.
181,202
31
tablet-1
Diao Group Tianfu Pharmaceutical Group Co., Ltd.
190,102
32
tablet-2
Diao Group Tianfu Pharmaceutical Group Co., Ltd.
190,103
33
tablet-3
Diao Group Tianfu Pharmaceutical Group Co., Ltd.
180,501
34
concentrated pill-1
Taiji Group Chongqing No.2 Chinese Medicine Factory Co., Ltd.
1,706,007
35
concentrated pill-2
Taiji Group Chongqing No.2 Chinese Medicine Factory Co., Ltd.
1,709,012
36
concentrated pill-3
Taiji Group Chongqing No.2 Chinese Medicine Factory Co., Ltd.
1,802,001
37
concentrated pill-4
Taiji Group Chongqing No.2 Chinese Medicine Factory Co., Ltd.
1,809,007
38
concentrated pill-5
Taiji Group Chongqing No.2 Chinese Medicine Factory Co., Ltd.
1,811,010
39
concentrated pill-6
Lanzhou Taibao Pharmaceutical Co., Ltd.
53,080,503
40
concentrated pill-7
Lanzhou Taibao Pharmaceutical Co., Ltd.
53,080,909
41
pill-1
Lanzhou Foci Pharmaceutical Co., Ltd.
18C5
42
pill-2
Lanzhou Foci Pharmaceutical Co., Ltd.
18F8
43
pill-3
Lanzhou Foci Pharmaceutical Co., Ltd.
18 J16
2.4 Determination of 26 constituents by HPLC-MS/MS
2.4.1 Chromatographic and mass spectrometric conditions
Liquid chromatography analyses were performed using a Waters Acquity HPLC system with an Agilent Eclipse Plus Cl8 column (5 μm, 4.6 mm × 250 mm). The flow rate was set at 1.0 mL.min−1, and the column temperature was kept at 30 ℃. The injection volume was 10 μL. Acetonitrile was selected as mobile phase A, and 0.1 % formic acid –0.5 mmol/L ammonium acetate (V/V) was selected as mobile phase B. The linear gradient elution of A was performed as follows: 0–18 min, 16 % A; 18–30 min, 16 %-25 % A; 30–45 min, 25 % A; 45–55 min, 25 %-42 % A; 55–90 min, 42 % −70 % A.
The ES+ mode conditions of MS parameters were set as follows: desolvation temperature, 500 °C; cone gas flow, 20 L/h; capillary voltage, 2.0 kV; source temperature, 150 °C; and desolvation gas flow, 1000 L/h. The ES- mode conditions were set as follows: desolvation temperature 350 °C; cone gas flow 1 L/h; capillary voltage 2.0 kV; source temperature 72 °C; and desolvation gas flow 650 L/h. The collision energy and cone voltage were set to match the MRM of each component. The summary of MS/MS detection parameters is shown in Table 2.
No.
Component
molecular formula
tR (min)
[M + H]+ (m/z)
[M − H]− (m/z)
[M + NH4]+ (m/z)
MS/MS Fragments Ions
CV (V)
CE (eV)
1
Chlorogenic acid
C16H18O9
6.98
–
353
–
191
31
29
2
Caffeic acid
C9H8O4
7.01
–
179
–
135, 134
32
36
3
Vanillic acid
C8H8O4
10.43
153
–
–
111, 105, 93, 84
31
28
4
Prim-o-glucosylcimifugin
C22H28O11
18.27
469
–
–
307
15
58
5
Ferulic acid
C10H10O4
21.83
195
–
–
117, 89
32
25
6
Liquiritin
C21H22O9
22.18
419
–
–
257
22
35
7
Senkyunolide I
C12H16O4
24.82
225
–
–
189, 119
23
19
8
Senkyunolide H
C12H16O4
25.71
225
–
–
189, 119
23
19
9
Luteolin-7-O-glucoside
C21H20O11
28.88
449
–
–
287
36
52
10
5–0-methylvisamidol glycoside
C22H28O10
30.02
453
–
–
243, 219, 114
36
69
11
Quercitrin
C21H20O11
30.89
–
447
–
300
40
48
12
Hesperidin
C28H34O15
34.32
611
–
–
303, 129
35
31
13
Quercetin
C15H10O7
35.38
303
–
–
153, 69
21
63
14
Rosemarinic acid
C18H16O8
35.43
–
359
–
197, 161
30
37
15
Buddleoside
C28H32O14
48.24
593
–
–
285
33
39
16
Pulegone
C10H18O
54.21
155
–
–
99
37
38
17
Coniferyl ferulate
C20H20O6
69.08
–
355
–
163, 134
22
24
18
Glycyrrhizic acid
C42H62O16
70.09
823
–
–
453
31
40
19
Senkyunolide A
C12H16O2
72.14
193
–
–
91
22
26
20
Z- ligustilide
C12H14O2
75.47
191
–
–
115, 91, 77
21
29
21
Butylidenephthalide
C15H16O3
75.92
245
–
–
131, 103
21
57
22
Imperatorin
C16H14O4
75.96
271
–
–
203, 147, 69
21
39
23
Asarinin
C20H18O6
78.54
–
–
372
173, 135
22
25
24
Notopterol
C21H22O5
78.57
–
353
–
215, 163, 114
28
34
25
Isoimperatorin
C16H14O4
81.49
271
–
–
203, 147, 69
21
40
26
Levistilide A
C24H28O4
81.51
357
–
–
191
21
24
2.4.2 Preparation of sample
Due to the varying dosages of the six Chuanxiong Chatiao dosage forms for each administration, the amount of sample for this study was based on the calculated daily dosage, which was determined by the maximum dosage for each administration and the number of daily doses. Taking a sampling amount of 0.5000 g of Chuanxiong Chatiao Powder, the sampling amounts for the other dosage forms were accordingly calculated as 0.5000 g of pills, 0.2500 g of dropping pills, 0.3333 g of granules, 0.3600 g of tablets, and 0.3750 g of concentrated pills. Samples were weighted accurately, then extracted with 50 mL methanol for 30 min by ultrasound extraction (300 W of efficiency, 45 kHz of frequency), cooled to ordinary temperature and supplemented weightlessness. The extraction solution passed by a filter (0.22 μm mesh size).
2.4.3 Solutions preparation
The 26 reference compounds were respectively prepared by completely dissolving in methanol. All the stock solutions were stored at 4 °C before analysis.
2.5 Biopotency assay in vitro
2.5.1 Test substance solutions
Sodium citrate anticoagulant (3.2 %, w/v), ADP solution (final concentration: 10 μmol/L), a positive drug solution (sodium ferulate, high concentration, 60 mg mL−1; low concentration, 30 mg mL−1), and a sample solution (high concentration, 10 mg mL−1; low concentration, 5 mg mL−1) were used as test substances.
2.5.2 Preparation of the sample solutions for AAB analysis
Approximately 0.5000 g of powder, 0.5000 g of pills, 0.2500 g of dropping pills, 0.3333 g of granules, 0.3600 g of tablets, and 0.3750 g of concentrated pills was respectively weighted accurately, then extracted with 50 mL of methanol for 30 min by ultrasound extraction (300 W, 45 kHz), cooled to room temperature, and the concentration was adjusted. The extraction solution was filtered through filter paper and then concentrated by decompressing. Extracted samples were further dissolved in 1.5 mL of DMSO solution, and then added to physiological saline in 10-mL volumetric flasks. The solution was centrifuged at 8,000 rpm for 15 min, and then the supernatant was collected as a prepared solution.
2.5.3 Test for AAB
Japanese big-eared white rabbits, weighing about 2.5 kg, male, were provided by Chengdu Dashuo Biotechnology Co., Ltd. (Chengdu, China). Blood was collected from the carotid artery of normal rabbits into a centrifuge tube containing 3.2 % trisodium citrate (anticoagulant: blood = 1:9). Blood samples were centrifuged for 10 min twice at 800 rpm at room temperature, and then the platelet-rich plasma (PRP) was collected from the upper layer. The remaining plasma was centrifuged for 10 min at 3500 rpm, and the upper layer of the plasma was designated as the platelet-poor plasma (PPP).
Next, 280 μL of PPP and 10 μL of physiological saline were added into five turbidimetric tubes, and then either 10 μL of physiological saline, 10 μL of a high concentration of the positive drug, 10 μL of a low concentration of the positive drug, 10 μL of a high concentration of the sample solution, or 10 μL of a low concentration of the sample solution was added into the turbidimetric tubes, as corresponding blank solutions.
Additionally, 280 μL of PRP was added into another five turbidimetric tubes, and then either 10 μL of physiological saline, 10 μL of a high concentration of the positive drug, 10 μL of a low concentration of the positive drug, 10 μL of a high concentration of the sample solution, or 10 μL of a low concentration of the sample solution was added into the turbidimetric tubes, as corresponding sample solutions.
The platelet aggregation instrument was incubated to 37 °C and then a blank solution was used to zero the equipment. The platelet maximum aggregation rate was determined by incubating the PRP solution for 60 s, then adding 10 μL of ADP solution. The inhibition rate of platelet aggregation was calculated in the samples or positive drugs using the following formula: platelet inhibition rate = (blank group maximum aggregation rate − sample or positive drug group maximum aggregation rate) / blank group maximum aggregation rate × 100 %. Parallel determination of each sample was performed four times.
2.5.4 Calculation of biopotency of AAB in the samples
Because parallel line assays require using a quantitative response (2∙2) method according to the Chinese Pharmacopoeia 2020 edition (National Commission of Chinese Pharmacopoeia, 2020a, 2020b), the ratio was selected as 0.5 between two adjacent doses, and a random design experiment was used to analyze the validity of the test results. The biopotency per milligram (mg) of sodium ferulate was defined as 1 activity unit (U). The estimated biopotency was 1 U/g for the antiplatelet aggregation bioactivity of the sample. The inhibition rates of platelet aggregation of the samples and positive drugs were entered into BS2000 biostatistics software to calculate the AAB of the samples.
2.6 Statistical analysis
The data were analyzed by SNK with one-way ANOVA using SPSS 26.0 (Palo Alto, CA, USA). The differences were considered significant differences for the different subsets when alpha = 0.05, and there were no significant differences in the same subset. Analyses were further conducted using hierarchical clustering analysis (HCA) and partial least squares (PLS) with SIMCA-P 14.0 (Umetrics AB, Umea, Sweden). The parameters of the modeling (R2 and Q2 values) explained the accuracy of the fitting model.
3 Result and discussion
3.1 Optimization of chromatographic and MS condition
Various compositions of the mobile phase, different column temperatures and flow rates were compared and tested for obtaining accurate and sensitive quantitative method by HPLC-MS/MS. As a result, acetonitrile was selected as the mobile phase A, and 0.1 % formic acid–0.5 mmol/L ammonium acetate (V/V) were selected as the mobile phase B, the column temperature was held at 30 °C, the flow rate was determined at 1.0 mL/min. In order to obtain better responses, all analytes were determined with the electrospray ionization (ESI) source by a full-scan mass spectrometry (MS) method, and their ion source parameters such as cone voltage (CV) and collision energy (CE) were optimized under both positive and negative modes. In the meanwhile, the most abundant, specific, and stable fragment ions were obtained by multiple reaction monitoring (MRM) from MS/MS spectrum. The detailed information of retention time (tR), MS information, CV and CE for each analyte was listed in Table 2 and Fig. 1.
MRM chromatogram of the 26 compounds (1. Chlorogenic acid 2. Caffeic acid 3. Vanillic acid 4. Prim-o-glucosylcimifugin 5. Ferulic acid 6. Liquiritin 7. Senkyunolide I 8. Senkyunolide H 9. Luteolin-7-O-glucoside 10. 5-O-methylvisamidol glycoside 11. Quercitrin 12. Hesperidin 13. Quercetin 14. Rosemarinic acid 15. Buddleoside 16. Pulegone 17. Coniferyl ferulate 18. Glycyrrhizic acid 19. Senkyunolide A 20. Z- ligustilide 21. Butylidenephthalide 22. Imperatorin 23. Asarinin 24. Notopterol 25. Isoimperatorin 26. Levistilide A).
3.2 Validation of quantitative analysis in HPLC-MS/MS
Linearity was evaluated by developing calibration curves for the 26 analytes in which mixed standard solutions were diluted and six concentrations of the standard solutions were prepared. Calibration curves for the 26 analytes were formed by plotting the corresponding concentration versus the peak area. The results showed that the R2 value of the calibration curves were above 0.9997 for all components. The limit of detection (LOD) and limit of quantitation (LOQ) for each compound were determined at signal-to-noise (S/N) ratios of 3.
and 10, respectively. The precision of the method was assessed based on the relative standard deviation (RSD) by the determination of intraday and interday variations, using six replicates within a day for the intraday variations, and the experiments were repeated on three continuous days for the interday variations. The results showed that the RSD values of intraday variations of all analytes were within 5 %, and the RSD values of five analytes, namely, pulegone, coniferyl ferulate, senkyunolide A, Z-ligustilide, and levistilide A, were greater than 5.0 %. It was suggested that those analytes should be used immediately after preparation and not be held for very long before analysis. The RSD values of the other analytes were within 5.0 %. The repeatability of the proposed method was assessed based on six replicate injections for the same sample, and the RSD values of all analytes were within 5.0 %. The stability of the samples was evaluated by analyzing six of the same sample solutions at room temperature at different times (0, 3, 6, 9, 12, or 15 h) within a day, and the RSD values were all within 5.0 %. A recovery experiment was conducted with three different concentration levels (low, medium, or high) of the mixed standard solutions added to known concentrations of a sample, and the rate of recovery of the sample and standard was about 1:0.5 (low), 1:1 (medium), and 1:1.5 (high). The entire process was repeated three times at each concentration level. The method recovery was calculated using the formula:
The results showed that the recoveries were within 5.0 %. The detailed results are shown in Table 3.
No.
Linear
R2
Range (mg/ml)
LOD (ng/ml)
LQD (ng/ml)
Precision (RSD, %)
Stability (RSD, %)
Reproducibility (RSD, %)
Recovery
Intra-day
Inter-day
Low
Medium
High
Average /%
RSD/%
Average /%
RSD/%
Average /%
RSD/%
1
y = 6112.7x − 0.115
0.9999
0.17–43.4
1.93
5.98
0.18
0.34
0.46
0.42
101.51
2.59
100.53
1.74
100.21
1.46
2
y = 23725x − 0.0163
1.0000
0.12–15.3
0.67
2.13
0.30
0.35
0.27
0.73
96.81
1.37
95.70
0.92
95.33
0.77
3
y = 25474x − 0.1523
1.0000
0.24–62.4
4.36
13.52
0.53
0.68
0.28
1.05
100.78
3.25
99.54
2.20
99.13
1.84
4
y = 5093.5x − 0.7577
0.9999
0.47–60.2
3.08
9.53
0.40
0.24
0.38
0.28
99.46
0.49
99.40
0.33
99.39
0.27
5
y = 6452.6x − 0.1671
1.0000
0.53–67.8
3.21
9.98
0.35
0.02
0.30
0.56
96.85
1.64
99.30
1.22
100.11
1.09
6
y = 11117x + 0.195
1.0000
0.35–44.6
3.03
9.21
0.17
0.19
0.28
0.48
99.26
1.13
99.66
0.75
99.80
0.63
7
y = 32365x − 0.0421
1.0000
0.18–45.2
4.22
12.97
0.36
0.46
0.28
0.78
101.57
1.39
101.42
0.93
101.37
0.77
8
y = 32319x + 0.8578
0.9999
0.12–14.9
2.10
6.71
0.47
0.40
0.18
1.93
99.43
2.52
98.41
1.70
98.06
1.42
9
y = 6794.3x − 0.0478
0.9998
0.38–48.8
1.22
3.87
0.26
0.42
1.36
0.73
98.68
2.13
97.40
1.44
96.97
1.21
10
y = 5716.5x − 0.3972
1.0000
0.47–60.2
1.77
5.54
0.33
4.93
0.29
1.92
99.42
2.44
98.06
1.65
97.61
1.38
11
y = 6090.2x + 0.6644
0.9999
0.39–50.4
2.41
7.56
0.42
0.28
0.34
0.28
99.23
3.25
99.13
2.17
99.10
1.81
12
y = 10536x − 0.1763
1.0000
0.31–39.6
1.22
3.93
0.30
0.27
0.45
0.80
98.41
3.61
98.97
2.39
99.16
1.99
13
y = 19383x − 0.1701
1.0000
0.13–16.1
1.21
3.95
0.46
0.41
0.33
3.66
99.48
0.65
99.06
0.44
98.92
0.36
14
y = 2569.4x + 0.5664
0.9997
0.80–51.2
5.63
17.78
0.35
0.44
0.48
0.38
101.09
1.60
100.77
1.07
100.66
0.89
15
y = 10176x + 0.4182
0.9999
0.20–25.2
0.93
2.95
0.34
0.74
0.26
1.24
97.58
2.76
99.43
1.80
100.05
1.49
16
y = 5772.7x − 1.5217
1.0000
0.51–64.3
1.13
3.41
0.64
9.67
1.62
0.47
98.47
2.54
95.93
1.74
95.09
1.46
17
y = 5662x − 0.2908
0.9997
0.68–44.8
2.22
6.97
1.18
14.45
2.55
1.47
99.21
2.72
98.57
1.83
98.36
1.53
18
y = 702.52x + 0.1685
0.9997
3.28–210.4
5.32
16.98
0.21
0.60
0.38
0.20
101.20
1.64
100.05
1.10
99.66
0.92
19
y = 4594x − 0.1007
1.0000
0.72–92.6
0.42
1.47
1.54
8.88
1.82
0.94
100.35
2.87
99.22
1.93
98.84
1.62
20
y = 10252x + 2.3748
0.9999
1.49–372.6
1.46
4.83
1.73
12.11
2.09
1.80
100.36
1.36
100.13
0.91
100.05
0.76
21
y = 11223x − 0.2368
0.9999
0.24–30.5
0.77
2.54
0.62
0.41
0.31
1.65
98.79
2.24
98.76
1.50
98.75
1.25
22
y = 10253x − 0.6407
0.9999
0.41–52.4
1.54
4.88
0.20
0.18
0.29
0.47
99.48
3.75
98.05
2.54
97.57
2.12
23
y = 7219x + 0.203
0.9999
0.31–79.8
1.23
3.99
0.62
0.46
0.34
0.91
100.60
2.68
100.76
1.78
100.82
1.48
24
y = 12029x + 0.2116
1.0000
0.13–34.4
2.42
7.63
0.48
0.21
0.29
0.25
99.70
2.88
100.07
1.91
100.19
1.59
25
y = 16067x + 0.1986
1.0000
0.33–42.4
0.92
2.98
0.20
0.38
0.34
0.63
101.51
3.11
98.93
2.13
98.07
1.79
26
y = 16621x + 0.1787
1.0000
0.25–30.6
0.74
2.66
0.63
8.48
1.18
0.61
99.04
0.98
99.78
0.65
100.03
0.54
3.3 Validation of the methodology in the bioassay of AAB
According to the discriminant requirements of the validity test results of the parallel line assay based on a quantitative response (2∙2) method, the validity test of the results of the quantitative determination of the AAB of sample was conducted. The results showed that the regression was extremely significant (P < 0.01), indicating that while the dose of sodium ferulate and the samples increased, the platelet inhibition rate also increased, further revealing that the dose–effect relationship was linear. As far as the parallelism, there was no significant difference (P > 0.05), indicating that the lines formed by the increased platelet inhibition rate in the high-dose and low-dose sample groups were parallel to the lines formed by the increased platelet inhibition rate in the high-dose and low-dose sodium ferulate groups, respectively. Therefore, the assay result was satisfactory. The rate of fiducial limits indicates the precision and stability of the biological assay method. When the rate of fiducial limits is smaller, the precision is better. On the basis of the recommendation of the traditional Chinese medicine biological assay method on the rate of fiducial limits (Xiao et al., 2014), the rate of fiducial limits of AAB in this study was set as not exceeding 30 %.
3.4 Comparison analysis of AAB among six dosage forms
The results of AAB testing were shown in Table 4. In terms of the average value ranking (Fig. 2), powder (6.7760 U g−1) > pills (5.1737 U g−1) > dropping pills (4.7759 U g−1) > concentrated pills (4.5890 U g−1) > tablets (4.2761 U g−1) > granules (3.9586 U g−1). One-way ANOVA was performed, and SNK was selected to compare the differences in the AAB among Chuanxiong Chatiao powder and the five related dosage forms. The results indicated that there were significant differences (P < 0.05) between groups, and there was no significant difference between the dropping pill and concentrated pill. However, significant differences were observed with the other dosage forms. This indicated that the AAB significantly differed between powder and other dosage forms and could be divided into five subgroups, with the AAB of powder the highest, followed by pills, then followed by concentrated pills and dropping pills, then tablets, and finally granules. We concluded that the efficacy of the powder was the strongest, followed by pills, concentrated pills and dropping pills, tablets, granules, indicating that there were some differences among the six dosage forms.
No.
Sample
Biopotency of AAB / U.g−1
Rate of fiducial limits / %
The total concentrations of different markers
1
powder-1
6.8649
7.30
26.0731
2
powder-2
7.1534
8.56
27.0453
3
powder-3
7.1348
7.85
32.5588
4
powder-4
6.752
6.76
14.2627
5
powder-5
6.7594
6.48
19.7953
6
powder-6
6.7319
7.88
24.6263
7
powder-7
6.5102
5.87
18.5370
8
powder-8
6.5343
5.53
18.0996
9
powder-9
6.6473
6.07
16.5261
10
powder-10
6.4635
5.08
14.0298
11
powder-11
6.5507
5.79
18.6742
12
powder-12
6.6108
6.79
20.2527
13
powder-13
6.5827
6.78
16.6562
14
powder-14
6.8085
9.48
26.1420
15
powder-15
6.9001
10.64
24.1747
16
powder-16
7.2347
10.04
20.4620
17
powder-17
6.8649
9.61
12.8324
18
powder-18
6.8881
9.48
16.6425
19
powder-19
6.8935
9.88
24.1147
20
powder-20
6.6334
7.08
21.2151
21
dropping pill-1
4.6040
16.11
2.4811
22
dropping pill-2
4.8594
14.81
3.1021
23
dropping pill-3
4.8642
12.92
2.1622
24
granule-1
4.0375
26.94
1.6225
25
granule-2
3.8962
21.95
1.5620
26
granule-3
3.9389
22.32
2.3310
27
granule-4
3.9973
24.66
1.4198
28
granule-5
3.9959
24.31
1.7416
29
granule-6
4.1096
23.43
2.2126
30
granule-7
3.7346
28.95
1.2240
31
tablet-1
4.2168
18.31
10.1111
32
tablet-2
4.2492
22.71
11.2334
33
tablet-3
4.3623
21.43
2.2516
34
concentrated pill-1
4.7242
18.37
1.5737
35
concentrated pill-2
4.5949
21.84
2.0719
36
concentrated pill-3
4.3929
12.72
3.4857
37
concentrated pill-4
4.6214
12.03
4.8340
38
concentrated pill-5
4.4865
12.93
3.1562
39
concentrated pill-6
4.5616
20.34
1.6490
40
concentrated pill-7
4.7414
19.84
2.1753
41
pill-1
5.1922
11.02
2.7357
42
pill-2
5.1705
11.07
2.6265
43
pill-3
5.1585
10.99
3.3397
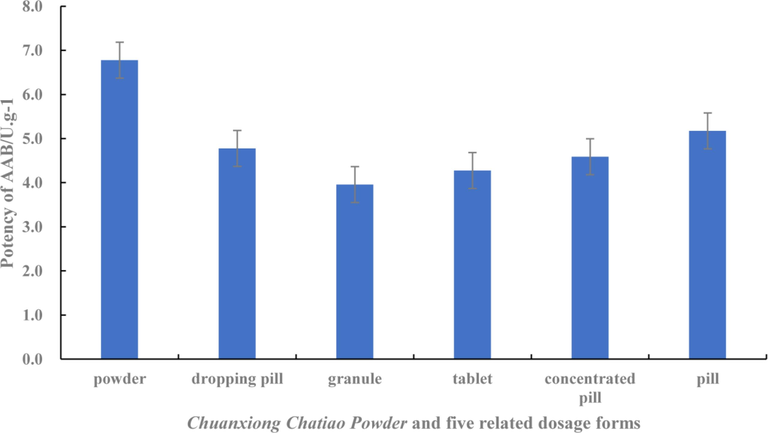
The AAB of Chuanxiong Chatiao Powder and five related dosage forms.
3.5 HCA
To evaluate the difference among Chuanxiong Chatiao powder and five related dosage forms with 26 bioactive ingredients and AAB, HCA was performed. A total of 43 samples were selected as observations while the contents of 26 compounds were selected as independent variables, AAB (Table 4) as dependent variable. In the dendrogram of HCA (Fig. 3), where it can be seen that these samples were sorted into three clusters reflecting the active constituents and AAB differences of three types of samples. Firstly, powder and others were respectively clustered into one category, indicating that the difference was significant between powder (group 1) and others. Secondly, pill and concentrated pill were clustered into one category (group 2), most granules and all tablets were clustered into other category (group 3), indicating that there were significant differences between pill, concentrated pill and granule, tablet, respectively, but there were some similarities between pill and concentrated pill, or granule and tablet. The HCA results demonstrated that the difference is most significant between powder and other dosage forms in the combination of active ingredients and bioactivity, concentrated pills and pills being similar, and tablets and drop pills and granules being similar, there were also significant difference between concentrated pills, pills and tablets, drop pills, granules.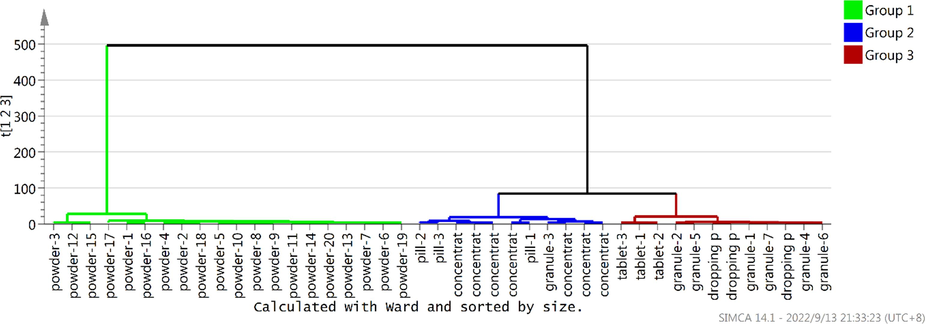
The result of dendrogram by HCA (Group 1, powder-1 ∼powder-20; Group 2, pill-1 ∼ pill-3, concentrated pill-1 ∼ concentrated pill-7, granule-3; Group 3, granule-1, granule-2, granule-4 ∼granule −7, tablet-1∼tablet-3).
3.6 The analysis of different markers among six dosage forms by PLS
To further compare and differentiate the difference among Chuanxiong Chatiao powder and five related dosage forms base on 26 active compounds and AAB, PLS was performed. A total of 43 samples were selected for analysis, while the contents of 26 compounds were selected as independent variables, AAB as dependent variable. In the PLS, the first three principal components were selected, R2X (cum) was 0.677, R2Y (cum) was 0.958, Q2 (cum) was 0.940, and we generated score scatter plot, permutation, loading scatter plot, coefficient plot and variable importance plot (Fig. 4). In the score scatter plot (Fig. 4A), all samples were mainly divided into four parts, besides it couldn’t be distinguished between granule and tablet, other dosage forms could be better distinguished. It indicated that there was no significant difference between granules and tablets, but the difference were significant among other dosage forms. In the permutation (Fig. 4B), R2 was 0.337, Q2 was −0.205, the values of left are lower than the right, indicating that the model was accurate and predictive. In the coefficient plot (Fig. 4C), the correlations were negative between Prim-O-glucosylcimifugin, Senkyunolide I, Quercitrin, Quercetin, Buddleoside and AAB, the correlation were positive between other ingredients and AAB, respectively. In the variable importance plot (Fig. 4D), the value of VIP in decreasing order was as follows: Z- ligustilide (1.369) > Senkyunolide A (1.357) > Butylidenephthalide (1.338) > Asarinin (1.312) > Rosmarinic acid (1.270) > Pulegone (1.256) > Senkyunolide H (1.210) > Notopterol (1.201) > Coniferyl ferulate (1.191) > Hesperidin (1.190) > Vanillic acid (1.176) > Luteolin-7-O-glucoside (1.154) > Levistilide A (0.991) > Caffeic acid (0.933) > Isoimperatorin (0.889) > Ferulic acid (0.847) > Prim-O-glucosylcimifugin (0.838) > Quercitrin (0.786) > Liquiritin (0.747) > Chlorogenic Acid (0.658) > Quercetin (0.656) > Buddleoside (0.574) > Senkyunolide I (0.462) > 5-O-Methylvisammioside (0.431) > Glycyrrhizic acid (0.398) > Imperatorin (0.376). The results indicated that the correlations were different between 26 bioactive constituents and AAB, further revealed the efficacy of the six Chuanxiong Chatiao dosage forms was the result which combined action of multiple bioactive constituents.Fig. 5..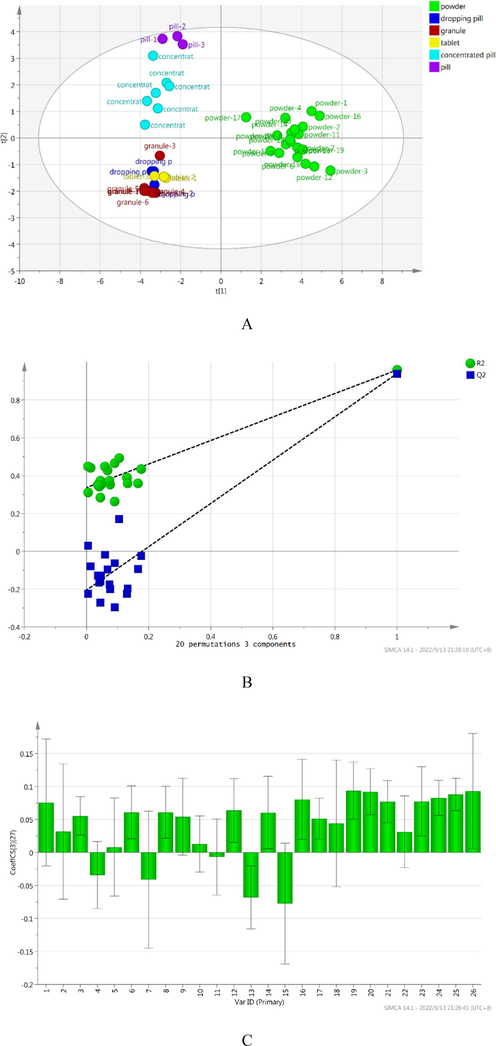
The results of statistical analysis by PLS (A: score scatter plot; B: permutation; C: coefficient plot; D: variable importance plot).
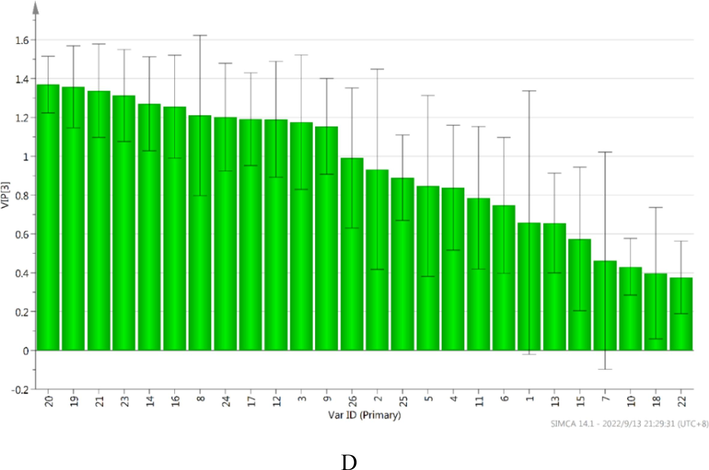
The results of statistical analysis by PLS (A: score scatter plot; B: permutation; C: coefficient plot; D: variable importance plot).
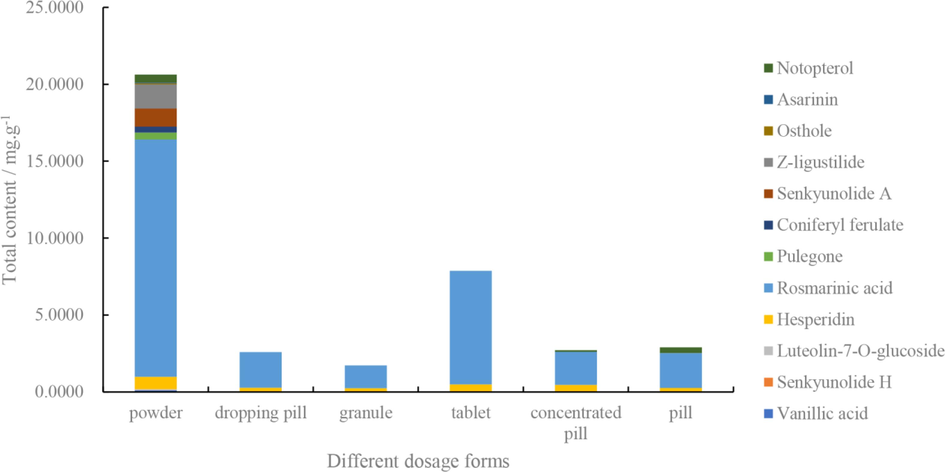
Total concentration of 12 difference markers in different dosage forms.
When the value of variable importance was > 1, Z- ligustilide, senkyunolide A, butylidenephthalide, asarinin, rosmarinic acid, pulegone, senkyunolide H, notopterol, coniferyl ferulate, hesperidin, vanillic acid, luteolin-7-O-glucoside were selected as the different markers. All different markers were positively associated with the AAB.
The results of the concentrations of 12 different markers showed that all constituents were detected in 20 samples of powder. However, the number and concentrations of constituents in the other five dosage forms were lower than in the powder. The common active components among six dosage forms were hesperidin and rosmarinic acid, however, other components were not detected in the tablet, granules and dropping pills. In the pills and concentrated pills, notopterol was detected, while other ingredients were not detected.
The total concentration of the 12 different markers were shown in Table 4. In terms of the average value ranking, powder (20.6360 mg g−1) > tablets (7.8653 mg g−1) > pills (2.9006 mg g−1) > concentrated pills (2.7065 mg g−1) > dropping pills (2.5818 mg g−1) > granules (1.7305 mg g−1). To further compare the differences of the total concentration of the12 different markers among the six Chuanxiong Chatiao dosage forms, SNK with one-way ANOVA was used (Table 5). The results indicated that there were significant differences (P < 0.05) between powders and other dosage forms, and there was no significant difference among the other dosage forms.
different dosage forms
N
Subset for alpha = 0.05
Total concentration
Hesperidin
Rosmarinic acid
Notopterol
1
2
1
2
1
2
1
2
granule
7
1.7305
0.235057
1.495443
0
dropping pill
3
2.5818
0.270733
2.311067
0
concentrated pill
7
2.706543
0.4538
2.154814
0.097957
pill
3
2.900633
0.261567
2.265967
0.3731
tablet
3
7.865367
0.4843
7.3811
0
powder
20
20.636025
0.813025
15.43149
0.53543
Sig.
0.143
1
0.193
1
0.154
1
0.836
0.173
To compare the differences of hesperidin, rosmarinic acid and notopterol among powder, concentrated pills and pills, SNK with one-way ANOVA was used (Table 5). The results indicated that there were significant differences (P < 0.05) in the content of hesperidin and rosmarinic acid between powders and the other two dosage forms, and there was no significant difference between concentrated pills and pills. In the content of notopterol, there were significant differences (P < 0.05) between concentrated pills and the other two dosage forms, and there was no significant difference between pills and powders. Based on the above discussion, it can be concluded that the pharmacodynamic substances vary among the six Chuanxiong Chatiao dosage forms.
The results revealed that numerous active components were not detected in the new dosage forms of Chuanxiong Chatiao powder, a situation potentially attributable to a variety of factors, these might include the raw materials used in the production and the production process employed. Regarding the raw materials used in the production, it’s one of crucial factors that the quality of medicinal materials varies. For instance, the content of various active ingredients varied significantly among different production regions in CX, such as Z- ligustilide, senkyunolide A, senkyunolide H, coniferyl ferulate and vanillic acid (Wang et al., 2020a, 2020b; Fang et al., 2015; Yi et al., 2016). A considerable difference was observed in the content of notopterol between different sources of QH (Ma et al., 2020). The content of active components in different parts of BH (stem, leave), differed significantly, including pulegone, rosmarinic acid, hesperidin. (Zhong, 2016; Wu et al., 2016). Furthermore, a significant variation was found in the content of luteolin-7-O-glucoside among different production regions of BH (Zhen et al., 2021). The content of pulegone in JJ was noticeably influenced by various factors, including different parts (spikes, stems, leaves), drying method, and harvest time (Yu, 2013).
In relation to the production process, Chuanxiong Chatiao powder is used as powder in medicine, which maximizes the preservation of all components in the medicinal material. However, most dosage forms are prepared by water extraction. In the process of water extraction, the solubility of liposoluble components is one of most important factors that influence the quality and efficacy of the product, because they have limited solubility in water. Furthermore, certain active components are unstable and susceptible to degradation or transformation during the water extraction process, which can result in a lower content of these active components in the final product. For example, coniferyl ferrate is prone to hydrolysis, resulting in the formation of ferulic acid and coniferol, and further oxidation of coniferol can generate vanillic acid. Z-ligustilide is susceptible to oxidation, causing it to transform into senkyunolide H and senkyunolide I, or polymerize into levistilide A. Senkyunolide A can easily undergo isomerization to generate butylphthalide (Mei et al., 2023; Zhang et al., 2019). Hesperidin is unstable and prone to degradation during high-temperature water extraction (Wang et al., 2014). Rosmarinic acid is susceptible to hydrolysis, resulting in the formation of caffeic acid (Li et al., 2017). The content of pulegone consistently decreases with increasing decoction time (Luo et al., 2016) (Fig. 6). Even though the content of asarinin gradually increases with extended decoction time, it remains significantly lower compared to that in the decoction pieces or medicinal material (Huang et al., 2012).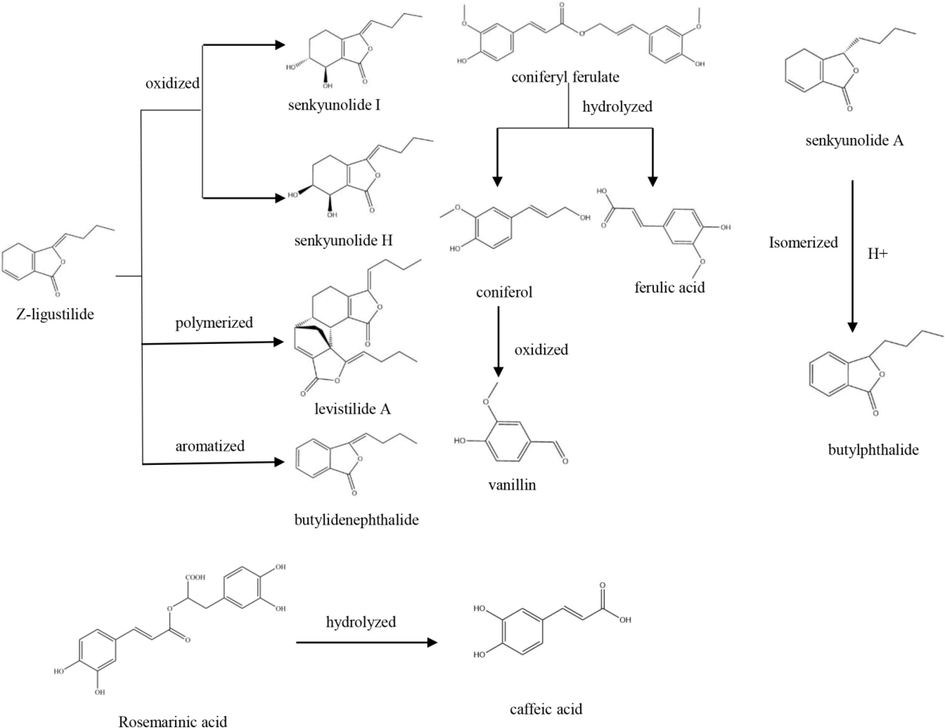
The transformation relation of active components.
Furthermore, some dosage forms employ ethanol for the purpose of extracting liposoluble components, such as concentrated pill, which CX, QH, and FF were extracted using 70 % ethanol. This method aims to extract the active ingredients from the medicinal materials to the greatest extent possible. However, it should be noted that certain liposoluble ingredients, such as Z-ligustilide, senkyunolide A, etc., which are susceptible to degradation after extraction or during product storage (Zhu et al., 2013). Currently, the stability of these extracted active ingredients has not been comprehensively considered and controlled. Meanwhile, in these dosage forms, the initial step typically involves the extraction of volatile oil from BH and JJ, followed by a mixture with other extracts, which is similar to approach used to maintain the stability of active ingredient in the product during storage (Wang et al., 2012; Yu et al., 2013). Additionally, in the drying process of products, such as tablets and granules, the drying temperature has a significant impact on certain active ingredients, particularly the volatile components (Wang et al., 2012). Due to the absence of relevant quality control indicators, the product quality can easily become inconsistent. Pills, being similar to powder, but it might be influenced by the raw materials, storage and drying.
This showed that because of the issues with quality standards, the quality of the six dosage forms was not controlled comprehensively, and led to bioactive components that were different among the six dosage forms, which also caused the AABs to be different. This indicates that it is necessary for developing a comprehensive quality control method based on multiple indicators in the six Chuanxiong Chatiao dosage forms, and that optimizing the process of production would better ensure consistency when used as a clinical medication.
4 Conclusion
The HPLC-MS/MS method was reliable and accurate for the simultaneous determination of 26 bioactive components in the six Chuanxiong Chatiao dosage forms, and the parallel line assay based on a quantitative response (2∙2) method of a biological assay accurately measured AAB in vitro. We screened 12 ingredients as the different markers, including Z- ligustilide, senkyunolide A, butylidenephthalide, asarinin, rosmarinic acid, pulegone, senkyunolide H, notopterol, coniferyl ferulate, hesperidin, vanillic acid, luteolin-7-O-glucoside. All different markers were positively associated with AAB. There are significant differences in the sum and composition of different markers among the six dosage forms. The results of this study provide a foundation for clinical study of the six Chuanxiong Chatiao dosage forms.
Author contributions
Jiajia Xiao designed the study. Jiajia Xiao and Yixin Yao did the experiments. Longbiao Luo and Yuping Weng analyzed the data. Jingqi Wang provided the samples. Guangmou Zhang and Huanzhao Lin provide the standards. Yixin Yao and Lin Yue wrote the manuscript. All authors gave approval to the final edition of research paper.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- A review of the ethnopharmacology, phytochemistry and pharmacology of Notopterygium incisum. J. Ethnopharmacol.. 2017;202:241-255.
- [CrossRef] [Google Scholar]
- Luteoloside protects the vascular endothelium against iron overload injury via the ROS/ADMA/DDAH Ⅱ/eNOS/NO pathway. Chin. J. Nat. Med.. 2022;20(1):22-32.
- [CrossRef] [Google Scholar]
- Network Pharmacology-Based Strategy for Elucidating the Molecular Basis Forthe Pharmacologic Effects of Licorice (Glycyrrhiza spp.) Front. Pharmacol.. 2021;12
- [CrossRef] [Google Scholar]
- Isoimperatorin exerts anti-inflammatory activity by targeting the LPS-TLR4/MD-2-NF-κB pathway. Eur. J. Inflamm.. 2021;19:1-10.
- [CrossRef] [Google Scholar]
- Antifungal effects and active components of Ligusticum chuanxiong. Molecules. 2022;2022(27):4589.
- [CrossRef] [Google Scholar]
- A Comparative Study of Hesperetin, Hesperidin and Hesperidin Glucoside: Antioxidant, Anti-Inflammatory, and Antibacterial Activities In Vitro. Antioxidants. 2022;11:1618.
- [CrossRef] [Google Scholar]
- Anti-Inflammatory and Proliferative Properties of Luteolin-7-O-Glucoside. Int. J. Mol. Sci.. 2021;22:1321.
- [CrossRef] [Google Scholar]
- Fingerprint analysis of processed Rhizoma Chuanxiong by high-performance liquid chromatography coupled with diode array detection. Chin. Med.. 2015;10:2.
- [CrossRef] [Google Scholar]
- Chlorogenic acid inhibits human platelet activation and thrombus formation. PLOS ONE. 2014;9(3):e90699.
- [CrossRef] [Google Scholar]
- Quality assessment of tongtian oral liquid based on full-quantified HPLC fingerprint and bioassay. Chin. J. Exp. Tradit. Med. Form.. 2016;22(14):44-50.
- [CrossRef] [Google Scholar]
- Content changes of methyleugenol, safrole and asarinin in Asari Radix et Rhizoma related to decocting time. Chin. Tradit. Pat. Med.. 2012;34(10):1971-1974.
- [Google Scholar]
- Metabolism of quercitrin by human intestinal bacteria and its relation to some biological activities. Biol. Pharm. Bull.. 1999;22(7):749-751.
- [CrossRef] [Google Scholar]
- Antiplatelet aggregation effect of vanillic acid in vitro and in vivo. Chin. J. Pharmacov.. 2020;17(6):342-347.
- [CrossRef] [Google Scholar]
- Antithrombotic activities of epi-sesamin in vitro and in vivo. Am. J. Chin. Med.. 2013;41(6):1313-1327.
- [CrossRef] [Google Scholar]
- Study on thermal stability of rosmarinic acid in different solvents and it’s degradation products. Chin. J. Mod. Appl. Pharm.. 2017;34(4):483-487.
- [CrossRef] [Google Scholar]
- Research advances in cardio- cerebrovascular diseases of Ligusticum chuanxiong hort. Front. Pharmacol.. 2022;12
- [CrossRef] [Google Scholar]
- The genus Asarum: A review on phytochemistry, ethnopharmacology, toxicology and pharmacokinetics. J. Ethnopharmacol.. 2022;282:114642
- [CrossRef] [Google Scholar]
- Inhibitory effect of caffeic acid on ADP-induced thrombus formation and platelet activation involves mitogen-activated protein kinases. Sci. Rep.. 2015;5(1):1-13.
- [CrossRef] [Google Scholar]
- Inflamm-ageing: the role of inflammation in age-dependent cardiovascular disease. Eur. Heart J.. 2020;41:2974-2982.
- [CrossRef] [Google Scholar]
- Scientific connotation of decocting method “taking when fragrance volatilized fiercely” for Yinqiao Powder based on dynamic changes of volatile components during decocting process. Chin. Tradit. Herb. Drugs. 2016;47(15):2650-2655.
- [CrossRef] [Google Scholar]
- Rapid discrimination of Notopterygium incisum and Notopterygium franchetii based on characteristic compound profiles detected by UHPLC-QTOF-MS/MS coupled with multivariate analysis. Phytochem. Analysis.. 2020;31(3):355-365.
- [CrossRef] [Google Scholar]
- Exploring the quality markers of the production region of Ligusticum chuanxiong through antiplatelet aggregation bioactivity and multiple active components, and correlation analysis of the quality markers and geographical factors. Ind. Crop. Prod.. 2023;200:116845
- [CrossRef] [Google Scholar]
- Pharmacopoeia, Pharmacopoeia of People’s Republic of China. Beijing: Chemical Industry Press, one part; 2020. p. :541-547.
- Pharmacopoeia, Pharmacopoeia of People’s Republic of China. Beijing: Chemical Industry Press, four part; 2020. p. :206.
- Glucosyl hesperidin improves serum cholesterol composition and inhibits hypertrophy in vasculature. J. Nutr. Sci. Vitaminol.. 2003;49(6):447-450.
- [Google Scholar]
- Exploring the scientific rationality of the phenomenon of “different dosage forms of the same prescription” of Chinese proprietary medicine based on biopharmaceutical properties of powder and pill of Chuanxiong Chatiao prescription. Front. Pharmacol.. 2022;13:893552
- [CrossRef] [Google Scholar]
- Evaluation of the antifungal effect of chlorogenic acid against strains of Candida spp. resistant to fluconazole: apoptosis induction and in silico analysis of the possible mechanisms of action. J. Med. Microbiol.. 2022;71(5):001526
- [CrossRef] [Google Scholar]
- Synthesis, antifungal activity of caffeic acid derivative esters, and their synergism withfluconazole and nystatin against Candida spp. Diagn. Micro. Infec. Dis.. 2016;86:387-391.
- [CrossRef] [Google Scholar]
- Inhibition of Δ5-desaturase in polyunsaturated fatty acid biosynthesis by (−)-asarinin and (−)-epiasarinin. Phytochemistry. 1992;31(3):757-760.
- [CrossRef] [Google Scholar]
- Quercitrin neutralizes sPLA2IIa activity, reduces the inflammatory IL-6 level in PC3 cell lines, and exhibits anti-tumor activity in the EAC-bearing mice model. Front. Pharmacol.. 2022;13
- [CrossRef] [Google Scholar]
- Effects of buddleoside on hypertensive vascular remodeling through Ang II/AT1 signaling pathway in aorta. Chin. Tradit. Herb. Drugs. 2019;50(20):5003-5010.
- [CrossRef] [Google Scholar]
- Advances in studies on varieties, harvest processing and packaging storage of Menthae Haplocalycis Herba. Chin. J. Exp. Tradit. Med. Form.. 2012;18(13):307-309.
- [Google Scholar]
- Studies on degradation kinetics of Hesperidin in aqueous. Lishizhen Med. Mater. Med. Res.. 2014;25(09):2088-2090.
- [CrossRef] [Google Scholar]
- A Chinese prescription Chuanxiong Chatiao San for migraine: a systematic review and meta-analysis of randomized controlled trials. Evid-Based. Compl. Alt.. 2019;2301680
- [CrossRef] [Google Scholar]
- General situation of modern research of Chuanxiong Chatiaosan. Chin. J. Exp. Tradit. Med. Form.. 2020;26(13):228-234.
- [CrossRef] [Google Scholar]
- Simultaneous determination of 20 bioactive components in Chuanxiong Rhizoma from different production origins in Sichuan province by ultra-high-performance liquid chromatography coupled with triple quadrupole mass spectrometry combined with multivariate statistical analysis. Electrophoresis. 2020;41:1606-1616.
- [CrossRef] [Google Scholar]
- Study on the chemical constituents of different parts of Mentha haplocalyx in Jiangxi by GC-MS. China Pharm.. 2016;27(21):2959-2961.
- [CrossRef] [Google Scholar]
- Studies and application of biological evaluation in the quality standardization of Chinese medicines. World. Sci. Technol. / Mod. Tradit. Chin. Med. Mater. Med.. 2014;16(3):514-518.
- [CrossRef] [Google Scholar]
- Correction to: linarin protects against cadmium-induced osteoporosis via reducing oxidative stress and inflammation and altering RANK/RANKL/OPG pathway. Biol. Trace Elem. Res.. 2022;200:3701.
- [CrossRef] [Google Scholar]
- Pulegone inhibits inflammation via suppression of NLRP3 inflammasome and reducing cytokine production in mice. Immunopharm. Immunot.. 2019;41(3):420-427.
- [CrossRef] [Google Scholar]
- Yao, Y., 2017. Quality Assessment of Chuanxiong Rhizome Based on Antiplatelet Aggregation Bioactivity and Medicinal composition. Dissertation. Master. Chengdu: Chengdu University of traditional Chinese medicine. https://kns.cnki.net/KCMS/detail/detail.aspx?dbname=CMFD201802&filename=1018954193.nh.
- The variation in the major constituents of the dried rhizome of Ligusticum chuanxiong (Chuanxiong) after herbal processing. Chin. Med.. 2016;11:26.
- [CrossRef] [Google Scholar]
- Study on harvest time, pretreating, storage and prepared slices classification of Schizonepeta tenuifolia. In: Dissertation Doctor. Nanjing: Nanjing University of Chinese Medicine; 2013.
- [Google Scholar]
- Simultaneous determination of night effective constituents and correlation analysis of multi-constituents and antiplatelet aggregation bioactivity In Vitro in Chuanxiong Rhizoma subjected to different decoction times. J. Anal. Methods. Chem.. 2019;8970624
- [CrossRef] [Google Scholar]
- The Angelica dahurica: a review of traditional uses, phytochemistry and pharmacology. Front. Pharmacol.. 2022;13
- [CrossRef] [Google Scholar]
- Correlation study on non-volatile ingredients of Menthae Haplocalycis Herba formula granules and traditional herbal pieces by UPLC–MS /MS. Chin. J. Chin. Mater. Med.. 2021;46(05):1134-1140.
- [CrossRef] [Google Scholar]
- Zhong, K., 2016. The differential Study on the Chemical Constituents of Mentha haplocalyx between its stems and leaves. Dissertation. Master. Beijing: Beijing University of Chinese Medicine. https://kns.cnki.net/KCMS/detail/detail.aspx?dbname=CMFD201602&filename=1016069990.nh.
- Investigation of Stability of Lactones in Volatile Oil from Chuanxiong Rhizoma. Chin. J. Exp. Tradit. Med. Form.. 2013;19(20):14-17.
- [CrossRef] [Google Scholar]
- Synthesis and anti-platelet aggregation activity of quercetin-3-O-acetate and quercetin-3-O-propionate. Nat. Prod. Res. Dev.. 2015;27:1346-1351.
- [CrossRef] [Google Scholar]
- Antithrombotic and antiplatelet effects of rosmarinic acid, a water-soluble component isolated from Radix Salviae miltiorrhizae (Danshen) Acta. Pharm. Sin.. 1993;28(4):241-245.
- [CrossRef] [Google Scholar]







