Translate this page into:
The synthesis, characterizations, and lead adsorption studies of chicken eggshell powder and chicken eggshell powder-doped iron (III) oxide-hydroxide
⁎Corresponding author at: Department of Environmental Science, Faculty of Science, Khon Kaen University, Khon Kaen 40002, Thailand; Environmental Applications of Recycled and Natural Materials (EARN) Laboratory, Khon Kaen University, Khon Kaen 40002, Thailand. pornprai@kku.ac.th (Pornsawai Praipipat),
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
The contamination of lead in wastewater causes water quality problems, which is toxic to aquatic organisms and environment, so wastewater treatment is required before discharging to receiving water. Chicken eggshell powder (CP) and chicken eggshell powder-doped iron (III) oxide-hydroxide (CPF) were synthesized, characterized, and investigated lead removal efficiencies by batch experiments, adsorption isotherms, kinetics, and desorption experiments. The specific surface area and pore volume of CPF were higher than CP, whereas the pore diameter size of CPF was smaller than CP. The phase structures of both materials demonstrated semi-crystalline phases with presenting peaks of calcium carbonate. Their surface morphologies were irregular, rough, and uneven surfaces. In both materials, they detected carbon, calcium, oxygen, O—H, N—H, C⚌O, C—O, and C—H. The point of zero charges (pHpzc) of CP and CPF were 4.47 and 4.83. For batch experiments, CPF demonstrated a higher lead removal efficiency than CP because of spending less material dosage and contact time than CP, and both materials had high lead removals at a lead concentration of 50 mg/L by more than 95 %. Thus, the addition of iron (III) oxide-hydroxide helped to increase material efficiency for lead adsorption. CP corresponded to the Langmuir model while CPF corresponded to the Freundlich model. In addition, both materials corresponded to a pseudo-second-order kinetic model relating to a chemisorption process. Moreover, both materials could be reusable for more than 5 cycles for lead adsorption of more than 77 %. Therefore, CPF was a potential material to apply for lead removals in industrial applications.
Keywords
Food waste
Metal oxide
Adsorption
Heavy metal
Wastewater treatment
1 Introduction
Economic, industrial, and technological growth causes an increase in releasing pollutants into the environment resulted to create water pollution. Various types of pollutants such as nutrients, pesticides, dyes, pharmaceutical products, and heavy metals affect the water quality and aquatic environment including human health effects. Especially, heavy metals are the most concerned because of their persistence, bioaccumulation, and toxicity to aquatic organisms and the environment at low concentrations (Ali et al., 2019). Lead is a toxic heavy metal that is normally used in many manufacturing processes of pigment, battery, steel, and plastic industries (Obeng-Gyasi, 2019), so their effluents might have lead contaminated with the above concerns. Moreover, lead toxicity might cause human health effects on misfunctions in nerve, blood, respiration, and reproduction systems (Balali-Mood et al., 2021). Therefore, wastewater treatment is required for removing lead from wastewater to be below 0.02 mg/L following USEPA standards before discharging for a safe environment.
Many methods of chemical precipitation, oxidation–reduction, coagulation-flocculation, ion exchange, and reverse osmosis are generally used for heavy metal eliminations including a lead in wastewater; however, they had disadvantages of incomplete heavy metal removals, expensive operating costs, complicated operations, and creating a big load of toxic sludges (Qasem et al., 2021). While an adsorption method offers an effective method for lead removal with easy operation and suitable operation cost, and this method also is available several choices of adsorbents depending upon the contaminated target metal, budget, and requirement of treated water quality. Several commercial, agricultural, industrial, and waste adsorbents are used for removing heavy metals, dyes, and antibiotics in wastewater such as activated carbon, zeolite, chitosan, rice husk, corn stalk, sawdust, bagasse, bagasse fly ash, coal fly ash, banana peels, lemon peels, pistachio shells, shrimp shells, and eggshells (Alnasrawi et al., 2022; Jangkorn et al., 2022; Kristianto et al., 2019; Mohammed et al., 2020; Mohammed and Kareem, 2021; Praipipat et al., 2022a, 2023a,2023c; Tejada-Tovar et al., 2019; Threepanich and Praipipat, 2022; Praipipat et al., 2023). However, this study will focus on food waste because of its benefits as low-cost adsorbents used for improving water quality along with reducing waste volumes in terms of waste management and disposal. The elimination of heavy metals in wastewater from various food wastes in 2018–2022 is demonstrated in Table 1. Among those adsorbents, chicken eggshells are a good choice because it has good chemical properties of calcium carbonate (CaCO3), carboxyl group (—COOH), and hydroxyl group (—OH) for lead adsorption in wastewater. Even though chicken eggshells have good chemical properties for lead adsorption, they also need to improve in case of high lead concentration for industrial applications.
Materials
Modifications
Heavy ions
qm (mg/g)
references
Avocado seed
–
Pb(II)
18.90
(Boeykens et al., 2019)
Avocado seed
–
Cr(VI)
3.39
(Boeykens et al., 2019)
Avocado seed
Phosphoric acid (H3PO4)
Pb (II)
26.60
(Boeykens et al., 2019)
Avocado seed
Phosphoric acid (H3PO4)
Cr(VI)
5.10
(Boeykens et al., 2019)
Watermelon rind
–
Ni(II)
38.98
(Mathangi and Helen Kalavathy, 2019)
Cassava peel
–
Pb(II)
11.79
(Tejada-Tovar et al., 2019)
Banana peel
–
Pb(II)
18.96
(Tejada-Tovar et al., 2019)
Yam peel
–
Pb(II)
98.36
(Tejada-Tovar et al., 2019)
Pea peel
Biochar
Pb(II)
152.50
(Novoseltseva et al., 2021)
Sweet lemon peel
Biochar
Pb(II)
2840.91
(Kumar, 2020)
Potato peel
Activated carbon
Pb(II)
262
(Kyzas and Mitropoulos, 2018)
Soya bean
Sodium hydroxide (NaOH)
Pb(II)
0.72
(Gaur et al., 2018)
Soya bean
Sodium hydroxide (NaOH)
As(V)
0.08
(Gaur et al., 2018)
Pistachio shell
–
Cd(II)
51.28
(Kayranli, 2022)
Peanut shell
–
Cd(II)
62.11
(Kayranli, 2022)
Almond shell
–
Cd(II)
78.74
(Kayranli, 2022)
Walnut shell
Activated carbon
Cd(II)
14.00
(Mohseni et al., 2022)
Tea waste
–
Pb(II)
1.20
(Çelebi et al., 2020)
Tea waste
–
Cd(II)
2.47
(Çelebi et al., 2020)
Tea waste
–
Ni(II)
1.16
(Çelebi et al., 2020)
Tea waste
–
Zn(II)
1.46
(Çelebi et al., 2020)
Eggshell
–
Ni(II)
13.53
(Kristianto et al., 2019)
Eggshell
Calcined
Ni(II)
769.23
(Kristianto et al., 2019)
Eggshell
Biochar
As(V)
0.89
(Akram et al., 2022)
Eggshell
Potassium permanganate (KMnO4)
Pb(II)
709.13
(Basaleh et al., 2020)
Eggshell
Iron(II) sulfate (FeSO4)
Cd(II)
69.77
(Meng et al., 2022)
Various metal oxides of titanium dioxide (TiO2), aluminum oxide (Al2O3), zinc oxide (ZnO), iron (II or III) oxide (Fe3O4), and iron (III) oxide-hydroxide have been reported by previous studies for improving adsorbent efficiency by increasing surface area and adsorption capacity for heavy metal removals (Aryee et al., 2021; Mahdavi et al., 2015; Ngamsurach et al., 2022a; Praipipat et al., 2023b; Razzaz et al., 2016; Threepanich and Praipipat, 2021). Among those metal oxides, iron (III) oxide-hydroxide is an interesting choice for improving material efficiency with the advantages mentioned above, and it also is low cost, non-toxicity, and chemical stability in a wide pH (Attia et al., 2022; Xu et al., 2015). In previous studies, many studies have used eggshells for removing various heavy metals of cadmium (Cd2+), copper (Cu2+), arsenate (As5+), mercury (Hg2+), nickel (Ni2+), zinc (Zn2+), and lead (Pb2+) with or without material modifications by the calcination process or adding iron (II or III) oxides of magnetite (Fe3O4) and iron (II) sulfate (FeSO4) (Meng et al., 2022; Naghizadeh et al., 2018; Peigneux et al., 2020; Rahmani-Sani et al., 2020). However, no one used iron (III) oxide-hydroxide to modify eggshell adsorbent for eliminating lead (Pb2+), so it is a good idea to investigate the chicken eggshell efficiency by adding iron (III) oxide-hydroxide for offering high adsorbent efficiency to deal high strength of heavy metals in industrial wastewater. Therefore, this study attempts to synthesize chicken eggshell powder materials with or without modification by iron (III) oxide-hydroxide, to compare their lead removal efficiencies through batch experiments, and verify whether adding metal oxide helped to improve material efficiency for lead adsorption.
This study aimed to synthesize chicken eggshell powder (CP) and chicken eggshell powder-doped iron (III) oxide-hydroxide (CPF), to characterize their specific surface area, pore volumes, pore sizes, crystalline structures, surface morphologies, chemical elements, and chemical functional groups by Brunauer-Emmett-Teller (BET), X-ray Diffractometer (XRD), Field Emission Scanning Electron Microscopy and Focus Ion Beam (FESEM-FIB) with Energy Dispersive X-ray Spectrometer (EDX), and Fourier Transform Infrared Spectroscopy (FT-IR), and to examine their lead removal efficiencies by batch experiments with varying doses, contact time, pH, and concentration. Finally, their lead adsorption patterns and mechanisms were studied by linear and nonlinear adsorption isotherms of Langmuir, Freundlich, Temkin, and Dubinin-Radushkevich models and kinetics of pseudo-first-kinetic, pseudo-second-kinetic, elovich, and intraparticle diffusion models. Finally, the material reusability was investigated through desorption experiments.
2 Materials and methods
2.1 Raw material
Chicken eggshells were collected from the local restaurants in Khon Kaen province, Thailand.
2.2 Chemicals
All chemicals were analytical grades (AR) without purification before use which were ferric chloride hexahydrate (FeCl3·6H2O) (LOBA, India), sodium hydroxide (NaOH) (RCI Labscan, Thailand), sodium chloride (NaCl) (RCI Labscan, Thailand), hydrochloric acid (HCl) (RCI Labscan, Thailand), lead nitrate (Pb(NO3)2) (QRëC, New Zealand). For a pH adjustment, 1 % NaOH and 1 % HNO3 were used.
2.3 Synthesis of chicken eggshell powder (CP) and chicken eggshell powder-doped iron (III) oxide-hydroxide (CPF)
The synthesis of CP and CPF are demonstrated in Fig. 1a-b which is based on Praipipat, P. et al (Praipipat et al., 2022b), and the details were clearly explained below: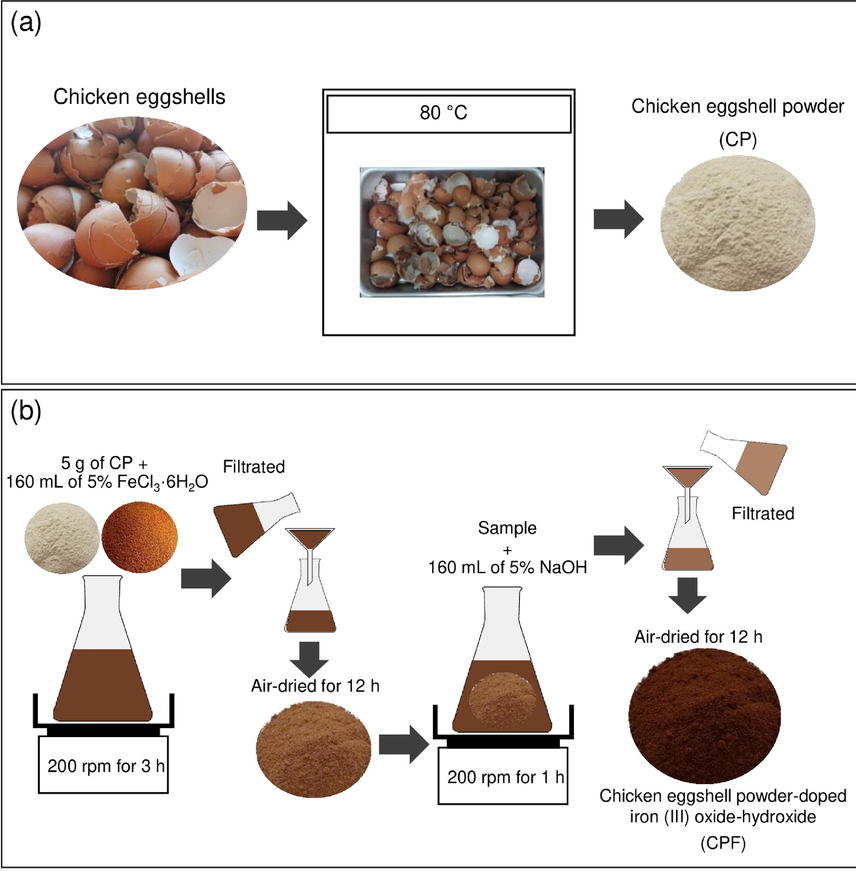
Flow diagram of synthesis methods of (a) CP and (b) CPF.
2.3.1 The synthesis of CP
Chicken eggshells were washed with tap water to eliminate contaminations, and then they were dried overnight in a hot air oven (Binder, FED 53, Germany) at 80 °C. Then, they were ground and sieved in size of 125 µm, and they were kept in a desiccator before use called chicken eggshell powder (CP).
2.3.2 The synthesis of CPF
5 g of CP were added to 500 mL of Erlenmeyer flask containing 160 mL of 5 % FeCl3·6H2O, and they were mixed by an orbital shaker (GFL, 3020, Germany) of 200 rpm for 3 h. Then, they were filtrated and air-dried at room temperature for 12 h. After that, they were added to 500 mL of Erlenmeyer flask containing 160 mL of 5 % NaOH, and they were mixed by an orbital shaker of 200 rpm for 1 h. Then, they were filtered, air-dried at room temperature for 12 h, and kept in a desiccator before use called chicken eggshell powder-doped iron (III) oxide-hydroxide (CPF).
2.4 Characterizations of chicken eggshell powder (CP) and chicken eggshell powder-doped iron (III) oxide-hydroxide (CPF)
Brunauer-Emmett-Teller (BET) (Bel, Bel Sorp mini X, Japan), X-ray Diffractometer (XRD) (PANalytical, EMPYREAN, UK), Field Emission Scanning Electron Microscopy and Focus Ion Beam (FESEM-FIB) with Energy Dispersive X-ray Spectrometer (EDX) (FEI, Helios NanoLab G3 CX, USA, and Fourier Transform Infrared Spectroscopy (FT-IR) (Bruker, TENSOR27, Hong Kong) were used for characterizing the specific surface area, pore size, pore volume, surface morphology, chemical compositions, and chemical functional groups of CP and CPF.
2.5 The point of zero charges of chicken eggshell powder (CP) and chicken eggshell powder-doped iron (III) oxide-hydroxide (CPF)
The study of the point of zero charges of CP and CPF is based on the study of Praipipat, P. et al (Praipipat et al., 2022b). The sample solutions of 0.1 M NaCl were prepared in different pH values from 2 to 12 by using 0.1 M HCl and 0.1 M NaOH. Then, 0.1 g of material was added to 250 mL Erlenmeyer flasks containing 50 mL of each sample solution, and it was shaken by an orbital shaker (GFL, 3020, Germany) at room temperature at 150 rpm for 24 h. After that, the final pH value of the sample solution was measured by a pH meter (Mettler Toledo, SevenGo with InLab 413/IP67, Switzerland) and calculated ΔpH (pHfinal – pHinitial). A point that is the crosses line of ΔpH versus pHinitial equal to zero is the point of zero charge (pHpzc) of material.
2.6 Batch adsorption experiments
The different values of dose (0.1 to 0.6 g), contact time (1 to 6 h), pH (1, 3, 5, 7, 9, 11), and concentration (10 to 70 mg/L) were applied for investigating lead removal efficiencies of chicken eggshell powder (CP) and chicken eggshell powder-doped iron (III) oxide-hydroxide (CPF) through a series of batch adsorption experiments which the control condition was the initial lead concentration of 50 mg/L, a sample volume of 200 mL, pH 5, a shaking speed of 200 rpm, and a temperature of 25 ℃. The optimum value was selected from the lowest value of each affecting factor with the highest lead removal efficiency, and that value was applied to the next affecting factor study. The atomic adsorption spectrophotometer (PerkinElmer, PinAAcle 900F, USA) is used for analyzing lead concentrations, and triplicate experiments were conducted to confirm the results. Lead removal in the percentage (%) is calculated by following Eq. (1)
2.7 Adsorption isotherms
Linear and nonlinear Langmuir, Freundlich, Temkin, and Dubinin-Radushkevich models represented the adsorption process of monolayer or multi-layer or heat or thermodynamic is used for identifying the adsorption pattern of chicken eggshell powder (CP) and chicken eggshell powder-doped iron (III) oxide-hydroxide (CPF), and their adsorption isotherms are calculated by Eqs. (2)–(9) (Dubinin and Radushkevich, 1947; Freundlich, 1906; Langmuir, 1918; Temkin and Pyzhev, 1940):
Langmuir isotherm:
Freundlich isotherm:
Temkin isotherm:
Dubinin-Radushkevich isotherm:
For adsorption isotherm experiments, 0.5 g of CP or 0.3 g of CPF were added to 500 mL Erlenmeyer flasks with variable lead concentrations from 10 to 70 mg/L. The control condition of CP or CPF was a sample volume of 200 mL, a shaking speed of 200 rpm, pH 5, a temperature of 25 °C, and a contact time of 4 h for CP and 2 h for CPF.
2.8 Adsorption kinetics
Linear and nonlinear pseudo-first-order kinetic, pseudo-second-order kinetic, elovich, and intraparticle diffusion models are used for studying the adsorption mechanism of chicken eggshell powder (CP) and chicken eggshell powder-doped iron (III) oxide-hydroxide (CPF), and their adsorption kinetic equations are calculated by Eqs. (10)–(16) (Elovich and Larinov, 1962; Ho and McKay, 1999; Lagergren, 1898; Weber and Morris, 1963):
Pseudo-first-order kinetic model:
Pseudo-second-order kinetic model:
Elovich model:
Intra-particle diffusion model:
For adsorption kinetic experiments, 2.5 g of CP or 1.5 g of CPF were added to 1000 mL of breaker with the lead concentration of 50 mg/L. The control condition of CP and CPF was a sample volume of 1000 mL, a shaking speed of 200 rpm, pH 5, a temperature of 25 °C, and a contact time of 6 h.
2.9 Desorption experiments
The desorption experiments are designed to examine the possible material reusability by studying five adsorption–desorption cycles to confirm the abilities of chicken eggshell powder (CP) and chicken eggshell powder-doped iron (III) oxide-hydroxide (CPF) for lead adsorption. After an adsorption process, CP or CPF was added to 500 mL of Erlenmeyer flask containing 200 mL of 0.5 M HNO3 solution, then it was shaken by an incubator shaker (New Brunswick, Innova 42, USA) at 200 rpm for 4 h. After that, it was washed with deionization water and dried at room temperature, and CP or CPF is ready for the next adsorption cycle. The desorption efficiency in percentage is calculated by following Eq (17):
3 Result and discussion
3.1 The physical characteristics of chicken eggshell powder (CP) and chicken eggshell powder-doped iron (III) oxide-hydroxide (CPF)
The physical characteristics of CP and CPF are demonstrated in Fig. 2a-b. CP was a cream color powder matching the color of a chicken eggshell shown in Fig. 2a while CPF was an iron-rust color powder corresponding to a color of iron (III) oxide-hydroxide color shown in Fig. 2b.
The physical characteristics of (a) CP and (b) CPF.
3.2 Characterizations of chicken eggshell powder (CP) and chicken eggshell powder-doped iron (III) oxide-hydroxide (CPF)
3.2.1 BET analysis
The specific surface area, pore volume, and pore diameter size of CP and CPF were determined by the Brunauer-Emmet and Teller technique (BET) with N2 adsorption–desorption isotherm at 77.3 K and degas temperature of 80 °C for 6 h, and the results of Barrett-Joyner-Halenda (BJH) method reported in Table 2.
Material
Specific surface area (m2/g)
Pore volume (cm3/g)
Pore diameter size (nm)
CP
0.692
0.001
3.597
CPF
17.382
0.011
2.617
The specific surface area of CP and CPF were 0.692 and 17.382 m2/g which CPF demonstrated a higher specific surface area than CP. The pore volumes of CP and CPF were 0.001 and 0.011 cm3/g and their pore diameter sizes were 3.597 and 2.617 nm. As a result, CPF had a higher pore volume and a smaller pore diameter size than CP. The specific surface area and pore volume of CPF were higher than CP about 25 times and 11 times of CP which might be from the formation of iron (III) oxide-hydroxide in CPF. Therefore, the addition of iron (III) oxide-hydroxide into chicken eggshell powder increased the surface area and pore volume while the pore diameter size was decreased. Moreover, they were classified to be mesoporous materials since their pore diameter sizes were in a range of 2–50 nm following the classification by the International Union of Pure and Applied Chemistry (IUPAC) (Rouquerol et al., 1994).
3.2.2 XRD analysis
XRD patterns of CP and CPF by XRD analysis were demonstrated in Fig. 3a-b. For CP, it illustrated a semi-crystalline structure by presenting the specific calcium carbonate peaks of 23.16°, 29.53°, 30.65°, 36.08°, 39.51°, 43.26°, 47.63°, 48.61°, 57.43°, 60.70°, 64.68°, and 72.95° matched to JCPDS No. 05–0586 (Praipipat et al., 2022b). For CPF, it demonstrated a semi-crystalline structure by presenting the specific calcium carbonate peaks similarly to CP, and it also detected the specified iron (III) oxide-hydroxide peaks of 33.06°, 41.20°, and 53.68° following JCPDS No. 29–0713 (Huang et al., 2018). As a result, it could be confirmed by the successfully adding of iron (III) oxide-hydroxide to synthesize CPF.
XRD patterns of (a) CP and (b) CPF.
3.2.3 FESEM-FIB and EDX analysis
The surface morphologies of CP and CPF by FESEM-FIB analysis at 2500X magnification with 50 µm illustrated in Fig. 4a-b. Both materials were irregular, rough, and uneven surfaces, so the addition of iron (III) oxide-hydroxide did not affect the surface morphology of CPF.
The surface morphologies and chemical compositions of (a, c) CP and (b, d) CPF.
The chemical compositions of CP and CPF by EDX analysis are demonstrated in Fig. 4c-d. Three main chemical components of carbon (C), oxygen (O), and calcium (Ca) were found in both materials. CP and CPF contained 42.0 and 28.9 wt% O, 35.0 and 27.7 %wt Ca, 22.9 and 18.9 %wt C, 0 and 0.2 %wt Cl, and 0 and 24.3 %wt Fe, respectively. After adding iron (III) oxide-hydroxide, iron (Fe) was detected in CPF which confirmed the successful addition of iron (III) oxide-hydroxide. The addition of iron (III) oxide-hydroxide affected to the decreases of C, O, and Ca by increasing of Fe which might be from the formation of iron (III) oxide-hydroxide in CPF. Moreover, Na also detected in CPF might be from chemicals of ferric chloride hexahydrate (FeCl3·6H2O) used for material synthesis.
3.2.4 FT-IR analysis
The chemical functional groups of CP and CPF were examined by FT-IR analysis, and their FT-IR spectra before and after lead adsorptions are demonstrated in Fig. 5a-b. Five main function groups of O—H, N—H, C⚌O, C—O, and C—H were detected in both materials, whereas Fe–O was only found in CPF which might be from the addition of iron (III) oxide-hydroxide. For O—H, it represented the stretching of the hydroxyl group and acidic hydrogen group, and N—H was the amine and amide presented in the protein fiber of the eggshell membrane (Lulit et al., 2019). C⚌O referred to the stretching of the amide and carbonyl group, and C—O was the stretching of the carbonate group presented calcium oxide (CaO) in eggshells (Awogbemi et al., 2020). In addition, C—H was the bending of calcium carbonate (CaCO3) (Tizo et al., 2018).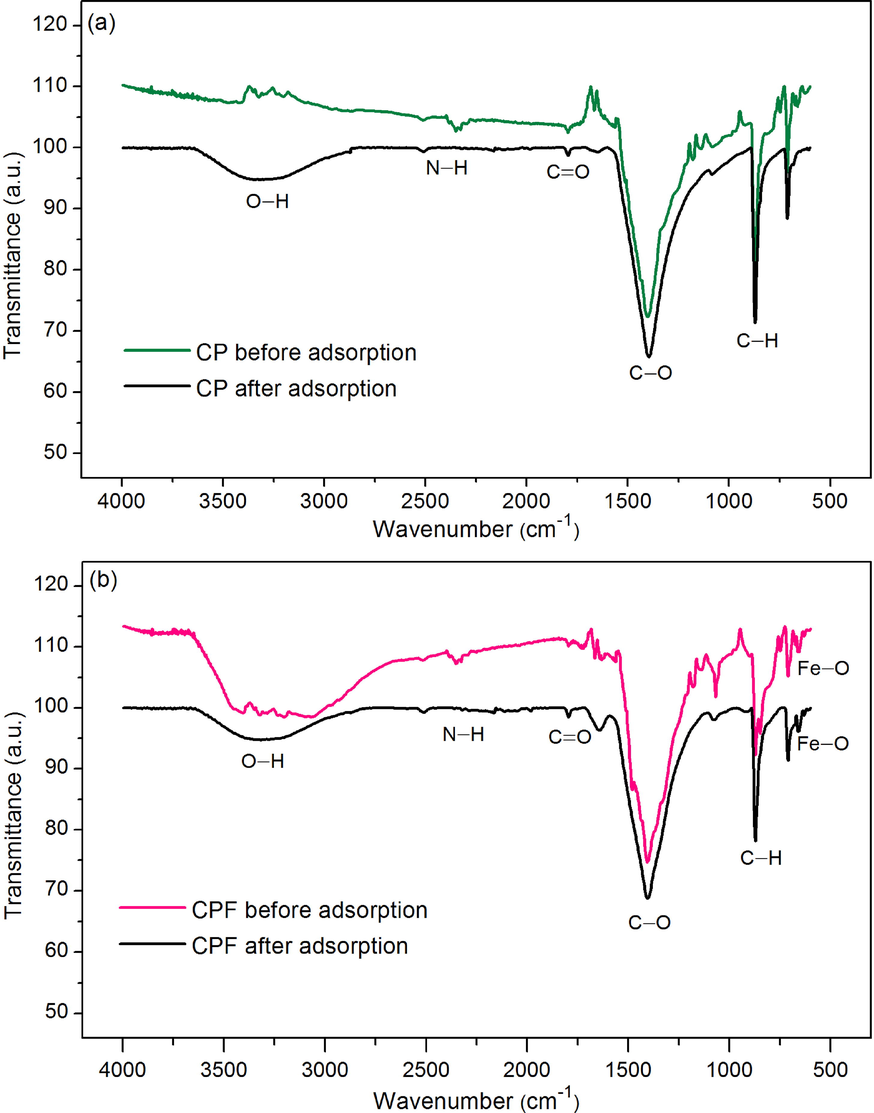
The chemical functional groups before and after lead adsorptions of (a) CP and (b) CPF.
For before lead adsorption, CP detected the stretching of O—H at 3350.05 cm−1, N—H at 2327.00 cm−1, stretching of C⚌O at 1796.26 and 1665.68 cm−1, stretching of C—O at 1402.17 cm−1, and bending of C—H at 872.22 and 711.78 cm−1 shown in Fig. 5a, and CPF detected the stretching of O—H at 3349.52 cm−1, N—H at 2327.32 cm−1, stretching of C⚌O at 1788.75 and 1665.52 cm−1, stretching of C—O at 1407.37 cm−1, bending of C—H at 871.98 and 710.68 cm−1, and Fe–O was 630.28 cm−1 shown in Fig. 5b.
For after lead adsorption, CP detected the stretching of O—H at 3286.58 cm−1, N—H at 2509.98 cm−1, stretching of C⚌O at 1795.79 cm−1, stretching of C—O at 1396.43 cm−1, and bending of C—H at 871.45 and 711.38 cm−1 shown in Fig. 5a, and CPF detected the stretching of O—H at 3318.96 cm−1, N—H at 2509.43 cm−1, stretching of C⚌O at 1795.64 and 1639.95 cm−1, stretching of C—O at 1406.51 cm−1, bending of C—H at 871.97 and 710.26 cm−1, and Fe–O was 625.49 cm−1 shown in Fig. 5b.
For comparing FT-IR spectra of before and after lead adsorption of CP and CPF, it observed the low frequency of bands of main functional groups after lead adsorption. In addition, the low intensity or shifting band was also observed which might be from adsorbing lead by CP and CPF. Especially, the bands around 3286.58 and 1396.43 cm−1 of CP and 3318.96 and 1406.51 cm−1 of CPF demonstrated the main observations for happening lead adsorption by O—H and C—O groups in CP and CPF by indicating Pb-O stretching for adsorbing lead by CP and CPF.
3.3 The point of zero charges of chicken eggshell powder (CP) and chicken eggshell powder-doped iron (III) oxide-hydroxide (CPF)
The point of zero charge (pHpzc) is generally used for determining which pH value is good for lead adsorption by material. A pH value at the net charge equal to zero of the adsorbent is the pHpzc of the material. The results of pHpzc of CP and CPF are demonstrated in Fig. 6a-b. The pHpzc values of CP and CPF were 4.47 and 4.83, so the addition of iron (III) oxide-hydroxide affected the increase of pHpzc. For lead adsorption, high lead adsorption should occur at the pH solution higher than the pHpzc (pHsolution > pHpzc) of the material because the surface of the material is negatively charged. Therefore, the high lead adsorption by CP and CPF should be found at a pH solution higher than the pH of 4 (pHsolution > 4) similarly reported by other studies (Ngamsurach et al., 2022a; Praipipat et al., 2023a; Threepanich and Praipipat, 2021, 2022).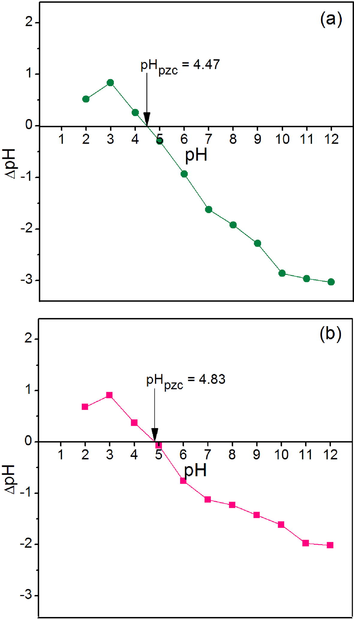
The point of zero charges of (a) CP and (b) CPF.
3.4 Batch adsorption experiments
3.4.1 The effect of dose
The effect of dose was investigated lead removal efficiencies by varying six different dosages from 0.1 to 0.6 g of chicken eggshell powder (CP) and chicken eggshell powder-doped iron (III) oxide-hydroxide (CPF) with the control condition of the lead concentration of 50 mg/L, a sample volume of 200 mL, a contact time of 4 h, pH 5, a temperature of 25 ℃, and a shaking speed of 200 rpm, and the results are demonstrated in Fig. 7a. Lead removal efficiencies of both materials were increased with the increase in dosage which might be from the increase of surface area or active sites of materials for adsorbing lead (Ngamsurach et al., 2022a). Their highest lead removal efficiencies were 95.89 % at 0.5 g for CP and 100 % at 0.3 g for CPF. Therefore, they were optimum doses of CP and CPF that were used for studying the contact time effect.
Batch experiments on the effects of (a) dose, (b) contact time, (c) pH, and (d) concentration of CP and CPF for lead adsorptions.
3.4.2 The effect of contact time
The effect of contact time was investigated lead removal efficiencies by varying six different contact times from 1 to 6 h of CP and CPF with the control condition of the lead concentration of 50 mg/L, a sample volume of 200 mL, pH 5, a temperature of 25 ℃, a shaking speed of 200 rpm, and the optimum dose from 3.4.1, and the results are demonstrated in Fig. 7b. Lead removal efficiencies of both materials were increased with the increase of contact time similar to the dose effect, and the saturated lead adsorption on the material is generally found at the highest lead removal efficiency. Their highest lead removal efficiencies were 94.36 % at 4 h for CP and 100 % at 2 h for CPF. Therefore, they were the optimum contact time of CP and CPF which were used for studying the pH effect.
3.4.3 The effect of pH
The effect of pH was investigated lead removal efficiencies by varying six different pH from 1 to 11 as respective pH conditions of acid, neutral, and alkaline of CP and CPF with the control condition of the lead concentration of 50 mg/L, a sample volume of 200 mL, a temperature of 25 ℃, a shaking speed of 200 rpm, and the optimum dose and contact time from 3.4.1 and 3.4.2, and the results are demonstrated in Fig. 7c. Lead removal efficiencies of both materials were increased with the increase of pH values from 1 to 5, then they were decreased. Their highest lead removal efficiencies were found at pH 5 with lead removal at 95.27 % and 99.23 % for CP and CPF. This result corresponded to the result of pHpzc of CP and CPF and other previous studies that reported the highest lead removal efficiency at pH > 4 (Ngamsurach et al., 2022a; Threepanich and Praipipat, 2022, 2021). Therefore, pH 5 was the optimum pH of CP and CPF which were used for studying the concentration effect.
3.4.4 The effect of concentration
The effect of concentration was investigated lead removal efficiencies by varying seven different concentrations from 10 to 70 mg/L of CP and CPF with the control condition of a sample volume of 200 mL, a temperature of 25 ℃, a shaking speed of 200 rpm, and the optimum dose, contact time, and pH from 3.4.1, 3.4.2, and 3.4.3, and the results are demonstrated in Fig. 7d. Lead removal efficiencies of CP were 100 % from 10 to 30 mg/L after they were decreased to 90.90 % at 70 mg/L whereas lead removal efficiencies of CPF were 100 % from 10 to 40 mg/L after they were decreased to 93.74 % at 70 mg/L. The decreasing of lead removal efficiencies of both materials from the increasing of lead concentrations might be from the active sites of them did not enough to caught up with lead ions similarly reported by other studies (Ngamsurach et al., 2022a; Threepanich and Praipipat, 2022, 2021). For the lead concentration of 50 mg/L, lead removal efficiencies of CP and CPF were 95.35 % and 99.57 %, and CPF demonstrated a higher lead removal efficiency than CP.
In conclusion, 0.5 g, 4 h, pH 5, 50 mg/L and 0.3 g, 2 h, pH 5, 50 mg/L were the optimum conditions in dose, contact time, pH, and concentration of CP and CPF for lead removal efficiencies of 95.35 % and 99.57 %, respectively. Since CPF spent less material dosage and contact time than CP with having a higher lead removal efficiency than CP, so the addition of iron (III) oxide-hydroxide helped to improve material efficiency for lead adsorption similar reported by previous studies (Threepanich and Praipipat, 2021). CPF was recommended to be applied for lead removal in future industrial applications.
3.5 Adsorption isotherms
Linear and nonlinear models of Langmuir, Freundlich, Temkin, and Dubinin-Radushkevich models were used for investigating the adsorption patterns of chicken eggshell powder (CP) and chicken eggshell powder-doped iron (III) oxide-hydroxide (CPF) for lead adsorptions. For linear models, Langmuir, Freundlich, Temkin, and Dubinin-Radushkevich isotherms were plotted by Ce/qe versus Ce, log qe versus log Ce, qe versus ln Ce, and ln qe versus ε2, respectively. For nonlinear models, all isotherms were plotted by Ce versus qe. The plotting graph results are demonstrated in Fig. 8a-f, and the equilibrium isotherm parameters are illustrated in Table 3.
Graphs of (a) linear Langmuir, (b) linear Freundlich, (c) linear Temkin, (d) linear Dubinin- Radushkevich, and (e) nonlinear adsorption isotherms of CP and (f) CPF for lead adsorptions.
Regression methods
Isotherm models
Parameters
CP
CPF
Linear
Langmuir
qm (mg/g)
25.189
42.735
KL (L/mg)
4.315
7.548
R2
0.993
0.991
Freundlich
1/n
0.258
0.172
KF (mg/g)(L/mg)1/n
16.776
31.893
R2
0.942
0.993
Temkin
bT (J/mol)
796.126
749.817
AT (L/g)
349.521
20859.232
R2
0.988
0.904
Dubinin-Radushkevich
qm (mg/g)
20.075
30.631
KDR (mol2/J2)
0.013
0.003
E (kJ/mol)
6.299
12.500
R2
0.941
0.841
Nonlinear
Langmuir
qm (mg/g)
25.493
40.762
KL (L/mg)
4.822
7.212
R2
0.994
0.993
R2adj
0.993
0.991
RMSE
3.185
6.526
Freundlich
1/n
0.215
0.181
KF (mg/g)(L/mg)1/n
16.842
32.109
R2
0.948
0.995
R2adj
0.937
0.994
RMSE
1.937
1.611
Temkin
bT (J/mol)
805.753
751.870
AT (L/g)
346.447
20865.050
R2
0.989
0.907
R2adj
0.987
0.889
RMSE
1.055
6.684
Dubinin-Radushkevich
qm (mg/g)
20.218
31.766
KDR (mol2/J2)
0.015
0.006
E (kJ/mol)
5.695
9.178
R2
0.942
0.844
R2adj
0.931
0.812
RMSE
2.736
7.267
For linear models, the Langmuir maximum adsorption capacities (qm) of CP and CPF were 25.189 and 42.735 mg/g, and Langmuir adsorption constants (KL) of CP and CPF were 4.315 and 7.548 L/mg. For Freundlich isotherm, the 1/n values of CP and CPF were 0.258 and 0.172. Freundlich adsorption constants (KF) of CP and CPF were 16.776 and 31.893 (mg/g)(L/mg)1/n. For Temkin isotherm, bT values of CP and CPF were 796.126 and 749.817 J/mol, and AT values of CP and CPF were 349.521 and 20859.232 L/g. For the Dubinin-Radushkevich model, the maximum adsorption capacities (qm) of CP and CPF were 20.075 and 30.631 mg/g, and the activity coefficient (KDR) values of CP and CPF were 0.013 and 0.003 mol2/J2. The adsorption energy (E) values of CP and CPF were 6.299 and 12.500 kJ/mol. R2 values of CP and CPF on Langmuir and Freundlich models were 0.993, 0.991 and 0.942, 0.993, respectively. In addition, R2 values of CP and CPF on Temkin and Dubinin-Radushkevich models were 0.988, 0.904 and 0.941, 0.841, respectively.
For nonlinear models, the Langmuir maximum adsorption capacities (qm) of CP and CPF were 25.493 and 40.762 mg/g, and Langmuir adsorption constants (KL) of CP and CPF were 4.822 and 7.212 L/mg. For Freundlich isotherm, the 1/n values of CP and CPF were 0.215 and 0.181. Freundlich adsorption constants (KF) of CP and CPF were 16.842 and 32.109 (mg/g)(L/mg)1/n. For Temkin isotherm, bT values of CP and CPF were 805.753 and 751.870 J/mol, and AT values of CP and CPF were 346.447 and 20865.050 L/g. For the Dubinin-Radushkevich model, the maximum adsorption capacities (qm) of CP and CPF were 20.218 and 31.766 mg/g, and the activity coefficient (KDR) values of CP and CPF were 0.015 and 0.006 mol2/J2. The adsorption energy (E) values of CP and CPF were 5.695 and 9.178 kJ/mol. R2 values of CP and CPF on Langmuir and Freundlich models were 0.994, 0.993 and 0.948, 0.995, respectively. In addition, R2 values of CP and CPF on Temkin and Dubinin-Radushkevich models were 0.989, 0.907 and 0.942, 0.844, respectively. Moreover, R2adj of CP and CPF in nonlinear Langmuir and Freundlich models were 0.993, 0.991 and 0.937, 0.994, respectively. R2adj of CP and CPF in nonlinear Temkin and Dubinin-Radushkevich models were 0.987, 0.889 and 0.931, 0.812, respectively.
For R2 value consideration, since R2 values of CP in both linear and nonlinear Langmuir models were higher than Freundlich, Temkin, and Dubinin-Radushkevich models, its adsorption patterns corresponded to Langmuir isotherm relating to physical adsorption. On the other hand, since R2 values of CPF in both linear and nonlinear Freundlich models were higher than Langmuir, Temkin, and Dubinin-Radushkevich models, its adsorption patterns corresponded to Freundlich isotherm relating to physiochemical adsorption. For the Langmuir model, the parameters of qm and KL were used for explaining the adsorption pattern. Since CPF had higher qm and KL values in both linear and nonlinear models than CP, CPF could remove lead with a faster adsorption rate than CP. For the Freundlich model, KF and 1/n values were used for consideration. Normally, if the 1/n value is in a range of 0–1 that means the adsorbent is favorable adsorption (Eltaweil et al., 2022). Since 1/n values in both linear and nonlinear models of CP and CPF were in a range of 0 > 1/n > 1, both materials were favorable lead adsorption. In addition, the KF values in both linear and nonlinear models of CPF had higher than CP, so CPF could adsorb lead with a faster adsorption rate than CP. For the Temkin model, the AT is generally used for consideration in which a high AT demonstrates stronger energy for equilibrium adsorption of adsorbent. CPF illustrated a higher AT value in both linear and nonlinear models than CP, so CPF had stronger energy for equilibrium lead adsorption than CP. For the Dubinin-Radushkevich model, the parameters of qm and E are normally used for explaining the adsorption pattern. For the qm value, it corresponded to the qm of the Langmuir model that a higher qm value demonstrated a higher adsorption capacity of the adsorbent. For the E value, if the E value is lower than 8 kJ/mol represented the physisorption process, whereas if the E value is in a range of 8–16 kJ/mol represented the chemisorption process. As a result, the adsorption pattern of CP was physisorption with the E value of 5.695 and 6.299 kJ/mol in linear and nonlinear models corresponding to the Langmuir model which was the best fitted model of CP. Oppositely, the adsorption pattern of CPF was chemisorption with the E value of 12.500 and 9.178 kJ/mol in linear and nonlinear models corresponding to the Freundlich model which was the best fitted model of CPF. Moreover, both linear and nonlinear isotherm models were recommended to plot graphs for confirming the results and protecting against data mistranslation (Ngamsurach et al., 2022b; Ngamsurach and Praipipat, 2022a, 2022b, 2021).
Moreover, the comparison of the maximum adsorption capacity (qm) value of eggshell adsorbents with and without modifications for lead adsorption is illustrated in Table 4. For unmodified comparison, CP and CPF had higher qm values than previous studies (Alamillo-López et al., 2020; Hassan et al., 2020; Soares et al., 2016). For modified comparison, both materials had also higher qm values than the eggshell with acidic modification (Maxwell et al., 2020). In addition, their qm values had higher than the eggshell with thermal treatment or calcination process except in the study of Gurav, V. L and Samant, R. A. which CP had a lower qm value than this study. Moreover, CPF had a higher qm value than the eggshells modified with Ag-Fe (Alamillo-López et al., 2020), so the addition of iron (III) oxide-hydroxide into the chicken eggshell of this study demonstrated a high material performance for lead adsorption in aqueous solution.
Materials
qm (mg/g)
References
Eggshell
3.90
(Alamillo-López et al., 2020)
Eggshell
15.91
(Soares et al., 2016)
Eggshell
18.80
(Hassan et al., 2020)
Eggshell modified with HCl
16.95
(Maxwell et al., 2020)
Calcinated hen eggshell with silica gel
0.87
(Bayu et al., 2022)
Calcinated eggshell
29.60
(Gurav and Samant, 2021)
Calcium oxide from hen eggshell
0.88
(Kasirajan et al., 2022)
Eggshell modified with Ag-Fe
27.80
(Alamillo-López et al., 2020)
CP
25.19
This study
CPF
42.74
This study
3.6 Adsorption kinetics
Linear and nonlinear kinetic models of pseudo-first-order kinetic model, pseudo-second-order kinetic model, elovich model, and intraparticle diffusion were used to determine the adsorption mechanism and rate of reaction of lead adsorptions by chicken eggshell powder (CP) and chicken eggshell powder-doped iron (III) oxide-hydroxide (CPF). For linear models, they were plotted by ln (qe-qt) versus time (t), t/qt versus time (t), qt versus ln t, and qt versus time (t0.5) for pseudo-first-order kinetic, pseudo-second-order kinetic, elovich, and intraparticle diffusion models, respectively. For nonlinear models, they were plotted by qt versus time (t). The plotting graph results are illustrated in Fig. 9a-f, and the adsorption kinetic parameters are presented in Table 5.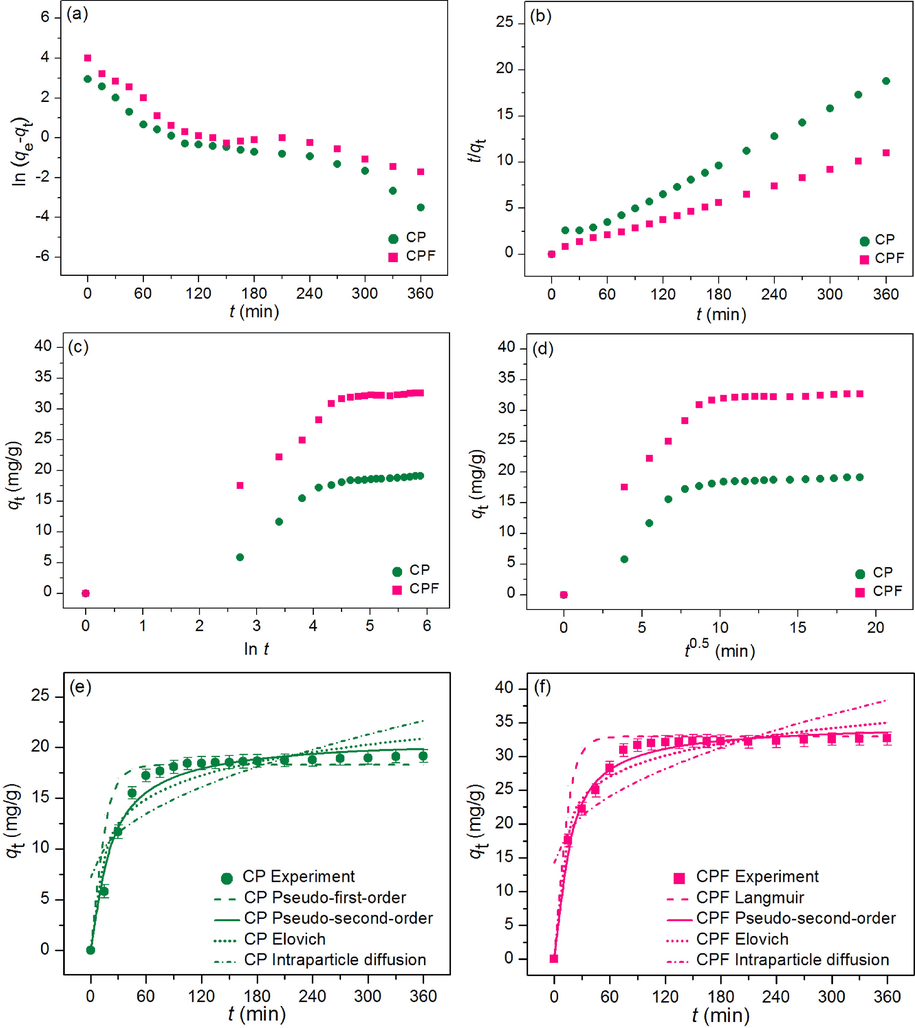
Graphs of (a) linear pseudo-first-order, (b) linear pseudo-second-order, (c) linear elovich model (d) linear intraparticle diffusion, and (e) nonlinear kinetic models of CP and (f) CPF for lead adsorptions.
Regression methods
Kinetic models
Parameters
CP
CPF
Linear
Pseudo-first-order kinetic
qe (mg/g)
7.081
8.344
k1 (min−1)
0.014
0.013
R2
0.893
0.818
Pseudo-second-order kinetic
qe (mg/g)
20.121
33.557
k2 (g/mg·min)
0.003
0.004
R2
0.996
0.999
Elovich
α (mg/g/min)
0.901
0.719
β (g/mg)
0.286
0.179
R2
0.894
0.923
Intraparticle diffusion
ki (mg/g·min0.5)
0.814
1.268
Ci (mg/g)
7.179
14.304
R2
0.651
0.638
Nonlinear
Pseudo-first-order kinetic
qe (mg/g)
7.640
8.503
k1 (min−1)
0.020
0.019
R2
0.892
0.822
R2adj
0.886
0.811
RMSE
2.369
3.769
Pseudo-second-order kinetic
qe (mg/g)
21.023
34.916
k2 (g/mg·min)
0.002
0.002
R2
0.997
0.998
R2adj
0.996
0.997
RMSE
1.064
0.955
Elovich
α (mg/g/min)
0.916
0.722
β (g/mg)
0.291
0.187
R2
0.895
0.928
R2adj
0.889
0.923
RMSE
1.715
3.709
Intraparticle diffusion
ki (mg/g·min0.5)
0.825
1.376
Ci (mg/g)
7.334
14.683
R2
0.656
0.641
R2adj
0.636
0.620
RMSE
3.125
5.003
For linear models, the adsorption capacities (qe) of CP and CPF on a pseudo-first-order kinetic model were 7.081 and 8.344 mg/g, and their reaction rate constants (k1) were 0.014 and 0.013 min−1. For a pseudo-second-order kinetic model, the adsorption capacities (qe) of CP and CPF were 20.121 and 33.557 mg/g, and their reaction of rate constants (k2) were 0.003 and 0.004 g/mg·min. For the elovich model, the initial adsorption rates (α) of CP and CPF were 0.901 and 0.719 mg/g/min, and their extents of surface coverage (β) were 0.286 and 0.179 g/mg. For the intraparticle diffusion model, the reaction of rate constants (ki) of CP and CPF were 0.814 and 1.268 mg/g·min0.5, and their constant Ci values were 7.179 and 14.304 mg/g. R2 values of CP and CPF on pseudo-first-order and pseudo-second-order kinetic models were 0.893, 0.818 and 0.996, 0.999, respectively. In addition, R2 values of CP and CPF on elovich and intraparticle diffusion models were 0.894, 0.923 and 0.651, 0.638, respectively.
For nonlinear models, the adsorption capacities (qe) of CP and CPF on a pseudo-first-order kinetic model were 7.640 and 8.503 mg/g, and their reaction rate constant (k1) were 0.020 and 0.019 min−1. For a pseudo-second-order kinetic model, the adsorption capacities (qe) of CP and CPF were 21.023 and 34.916 mg/g, and their reaction of rate constants (k2) were 0.002 and 0.002 g/mg·min. For the elovich model, the initial adsorption rates (α) of CP and CPF were 0.916 and 0.722 mg/g/min, and their extents of surface coverage (β) were 0.291 and 0.187 g/mg. For the intraparticle diffusion model, the reaction of rate constants (ki) of CP and CPF were 0.825 and 1.376 mg/g·min0.5, and their constant Ci values were 7.334 and 14.683 mg/g. R2 values of CP and CPF on pseudo-first-order and pseudo-second-order kinetic models were 0.892, 0.822 and 0.997, 0.998, respectively. In addition, R2 values of CP and CPF on elovich and intraparticle diffusion models were 0.895, 0.928 and 0.656, 0.641, respectively. Moreover, R2adj values of CP and CPF in nonlinear pseudo-first-order and pseudo-second-order kinetic models were 0.886, 0.811 and 0.996, 0.997, respectively. R2adj values of CP and CPF in nonlinear elovich and intraparticle diffusion models were 0.889, 0.923 and 0.636, 0.620, respectively.
For R2 value consideration, since R2 values of CP and CPF in both linear and nonlinear pseudo-second-order kinetic models were higher than pseudo-first-order kinetic, elovich, and intraparticle diffusion models, so their adsorption rate and mechanism of both materials corresponded to pseudo-second-order kinetic model with relating to a chemisorption process with heterogeneous adsorption. Therefore, the parameters of qe and k2 were used to describe the adsorption rate and mechanism. Since CPF demonstrated higher qe values in both linear and nonlinear models than CP, CPF might adsorb lead higher than CP corresponding to batch experiments. In addition, the k2 value of CPF was also higher values in both linear and nonlinear models than CP, so CPF demonstrated a faster reaction rate for lead adsorption than CP. For a pseudo-first-order kinetic model, the qe and k1 values of CPF in both linear and nonlinear models had also higher than CP similar to a pseudo-second-order kinetic model. For elovich, the parameters of α and β were used for consideration. Normally, if an α value is higher than a β value, the adsorption rate is higher than the desorption rate (Basha et al., 2022). In this study, both α values of CP and CPF had higher than β values in both linear and nonlinear models. For intraparticle diffusion, the Ci value is generally used for determining the type of adsorption mechanism of the adsorbent. If the Ci value is higher than zero (Ci > 0) that means the adsorption kinetic of the adsorbent is multiple steps of the intraparticle diffusion. Oppositely, if the Ci value is equal to zero (Ci = 0) that means the adsorption kinetic of the adsorbent is the sole rate-limiting step of the intraparticle diffusion (Basha et al., 2022). Since Ci values of CP and CPF in both linear and nonlinear models were higher than zero, they had multiple steps of intraparticle diffusion for lead adsorption. Finally, the graph plotting of both linear and nonlinear kinetic models was also recommended for correct data translations (Ngamsurach et al., 2022b; Ngamsurach and Praipipat, 2022a, 2022b, 2021).
3.7 Desorption experiments
The desorption experiments were used for investigating the feasibility for reuse materials because this is a necessary point to estimate the cost and economic feasibility of industrial applications. Lead adsorption tests of chicken eggshell powder (CP) and chicken eggshell powder-doped iron (III) oxide-hydroxide (CPF) for 5 cycles of adsorption–desorption were applied to confirm their abilities, and their results are illustrated in Fig. 10a-b. In Fig. 10a, CP could be reused in 5 cycles with high adsorption and desorption in ranges of 77.60–95.27 % and 70.28–94.92 %, respectively which adsorption and desorption were decreased by approximately 18 % and 25 %, respectively. For CPF, it also confirmed to be reusability in 5 cycles with high adsorption and desorption in ranges of 84.52–99.49 % and 80.07–99.05 %, respectively which adsorption and desorption were decreased by approximately 15 % and 19 %, respectively shown in Fig. 10b. Therefore, both materials are potential materials for lead adsorption with the reusability of more than 5 cycles by more than 77 %, and they can be further applied to industrial applications.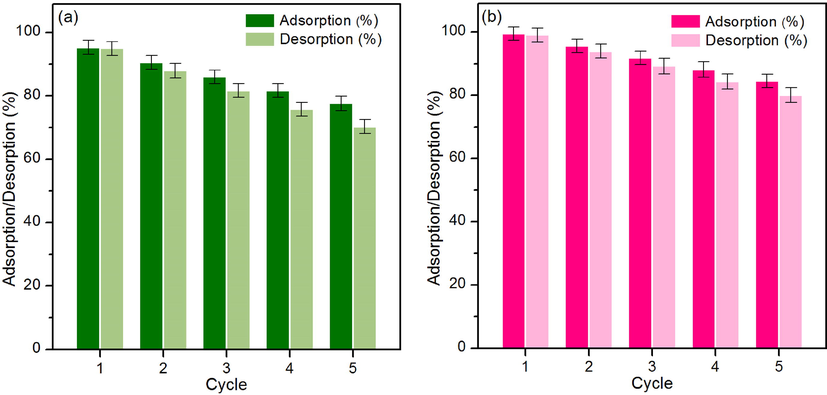
The desorption experiments of (a) CP and (b) CPF.
4 The possible mechanisms of lead adsorption by chicken eggshell powder (CP) and chicken eggshell powder-doped iron (III) oxide-hydroxide (CPF)
The possible mechanisms of lead adsorption on CP and CPF was explained in Fig. 11a-b. The main structure of CP and CPF is composed of calcium carbonate (CaCO3) with a hydroxyl group (–OH) while CPF had also the complex compound of iron (III) oxide-hydroxide CP∙Fe(OH)3 precipitation on its surface from adding iron (III) oxide-hydroxide into CP by sharing electrons with –OH of CP. The possible mechanism of lead adsorptions by CP and CPF might occur from donating a proton (H+) from –OH or CP∙Fe(OH)3 of the main chemical compounds for capturing lead (II) ions (Pb2+) instead of H+ from a process of electrostatic interaction (Wang et al., 2022). Moreover, the ion exchange from donating calcium ions (Ca2+) from CaCO3 for exchanging Ca2+ with Pb2+ on the surface of CP or CPF might possibly occur (Ahmad et al., 2012).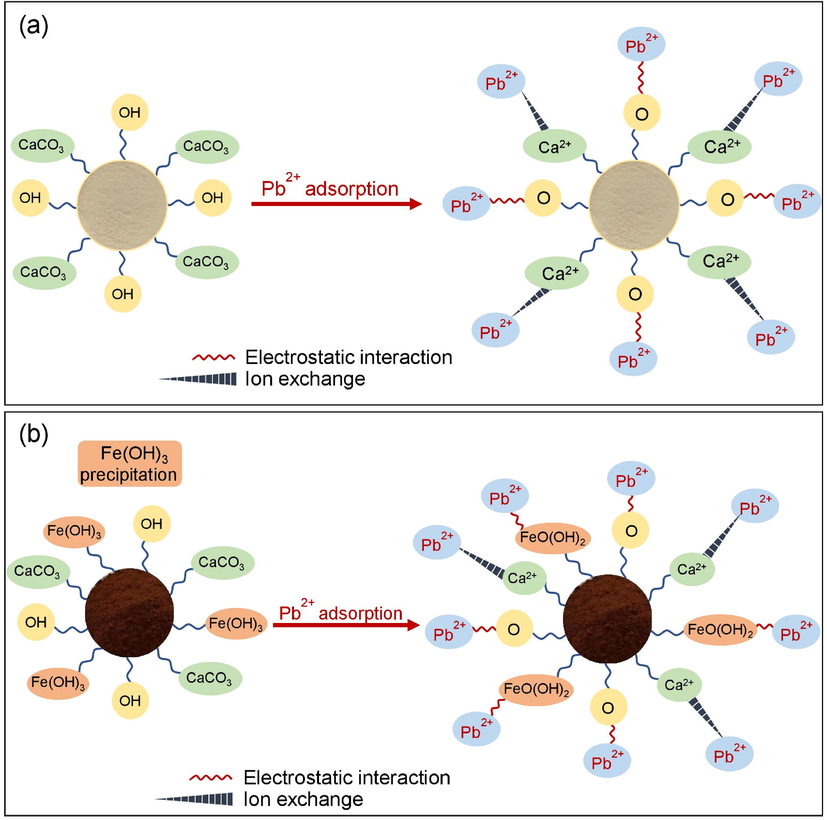
Possible mechanisms of lead adsorption on (a) CP and (b) CPF.
5 Conclusion
Chicken eggshell powder (CP) and chicken eggshell powder-doped iron (III) oxide-hydroxide (CPF) were successfully synthesized. The specific surface area and pore volume of CPF were higher than CP whereas its pore diameter size was smaller than CP. The crystalline structures of both materials illustrated semi-crystalline phases with presenting peaks of calcium carbonate. Their surface morphologies were irregular, rough, and uneven surfaces, and three main chemical elements of carbon (C), calcium (Ca), and oxygen (O) were detected in both materials. Iron (Fe) was only found in CPF which could confirm the addition of iron (III) oxide-hydroxide into CP. Five main chemical functional groups of O—H, N—H, C⚌O, C—O, and C—H were detected in both materials whereas Fe–O was only found in CPF. The point of zero charges (pHpzc) of CP and CPF were 4.47 and 4.83. For batch experiments, the optimum conditions of CP and CPF were 0.5 g, 4 h, pH 5, 50 mg/L and 0.3 g, 2 h, pH 5, 50 mg/L, and their lead removal efficiencies were 95.35 % and 99.57 %. As a result, CPF demonstrated a higher lead removal efficiency than CP because it spent less material dosage and contact time than CP. Thus, adding iron (III) oxide-hydroxide helped to improve material efficiency for lead adsorption. For the adsorption isotherm study, CP corresponded to the Langmuir model related to a physical adsorption process whereas CPF corresponded to the Freundlich model correlated to a physicochemical adsorption process. For the kinetic study, they corresponded to a pseudo-second-order kinetic model related to a chemisorption process with heterogeneous adsorption. Moreover, both CP and CPF could be reusable for more than 5 cycles for lead adsorption of more than 77 %. Therefore, CP and CPF were high-potential materials for lead adsorptions in an aqueous solution, and CPF demonstrated the highest lead removal efficiency. Therefore, CPF was suitable to apply for industrial wastewater treatment applications in the future.
For future works, the competing ions such as sodium (Na+), and magnesium (Mg2+), contaminated in real wastewater should be investigated to confirm the specific lead adsorption by CP or CPF, and the continuous flow study also needs to study for further industrial applications.
Funding
The authors are grateful for the financial support received from Research, Innovation and Academic Services Fund, Faculty of Science, Khon Kaen University.
CRediT authorship contribution statement
Pornsawai Praipipat: Supervision, Conceptualization, Funding acquisition, Investigation, Methodology, Validation, Formal analysis, Visualization, Writing – original draft, Writing – review & editing. Pimploy Ngamsurach: Visualization, Investigation, Writing – original draft. Krissana Pratumkaew: Investigation.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Eggshell and coral wastes as low cost sorbents for the removal of Pb2+, Cd2+ and Cu2+ from aqueous solutions. J. Ind. Eng. Chem.. 2012;18:198-204.
- [CrossRef] [Google Scholar]
- Synthesis and application of egg shell biochar for As(V) removal from aqueous solutions. Catalysts. 2022;12:431.
- [CrossRef] [Google Scholar]
- Efficient removal of heavy metals from aqueous solutions using a bionanocomposite of Eggshell/Ag-Fe. Catalysts. 2020;10:727.
- [CrossRef] [Google Scholar]
- Environmental chemistry and ecotoxicology of hazardous heavy metals: Environmental persistence, toxicity, and bioaccumulation. J. Chem.. 2019;2019:6730305.
- [CrossRef] [Google Scholar]
- Adsorption of methylene blue from aqueous solution using different types of activated carbon. J. Appl. Water Eng. Res. 2022
- [CrossRef] [Google Scholar]
- Fe3O4 and iminodiacetic acid modified peanut husk as a novel adsorbent for the uptake of Cu (II) and Pb (II) in aqueous solution: Characterization, equilibrium and kinetic study. Environ. Pollut.. 2021;268:115729
- [CrossRef] [Google Scholar]
- Iron oxide nanoparticles and their pharmaceutical applications. Appl. Surf. Sci. Adv.. 2022;11:100284
- [CrossRef] [Google Scholar]
- Modification and characterization of chicken eggshell for possible catalytic applications. Heliyon. 2020;6:e05283.
- [Google Scholar]
- Toxic mechanisms of five heavy metals: mercury, lead, chromium, cadmium, and arsenic. Front. Pharmacol.. 2021;12:643972
- [CrossRef] [Google Scholar]
- Metal removal using chemically modified eggshells: Preparation, characterization, and statistical analysis. Desalin. Water Treat.. 2020;173:313-330.
- [CrossRef] [Google Scholar]
- Sulfonated graphene oxide impregnated cellulose acetate floated beads for adsorption of methylene blue dye: optimization using response surface methodology. Sci. Rep.. 2022;12:9339.
- [CrossRef] [Google Scholar]
- Adsorptive capacity of calcinated hen eggshell blended with silica gel for removal of lead II ions from aqueous media: kinetics and equilibrium studies. J. Environ. Public Health. 2022;2022:2882546.
- [CrossRef] [Google Scholar]
- Chromium and Lead adsorption by avocado seed biomass study through the use of Total Reflection X-Ray Fluorescence analysis. Appl. Radiat. Isot.. 2019;153:108809
- [CrossRef] [Google Scholar]
- Adsorption capability of brewed tea waste in waters containing toxic lead(II), cadmium (II), nickel (II), and zinc(II) heavy metal ions. Sci. Rep.. 2020;10:17570.
- [CrossRef] [Google Scholar]
- The equation of the characteristic curve of activated charcoal. Proc. USSR Acad. Sci.. 1947;55:327-329.
- [Google Scholar]
- Theory of adsorption from solutions of non electrolytes on solid (I) equation adsorption from solutions and the analysis of its simplest form,(II) verification of the equation of adsorption isotherm from solutions. Izv. Akad. Nauk. SSSR. Otd. Khim. Nauk. 1962;2:209-216.
- [Google Scholar]
- Graphene oxide incorporated cellulose acetate beads for efficient removal of methylene blue dye; isotherms, kinetic, mechanism and co-existing ions studies. J. Porous Mater. 2022
- [CrossRef] [Google Scholar]
- Adsorptive removal of lead and arsenic from aqueous solution using soya bean as a novel biosorbent: equilibrium isotherm and thermal stability studies. Appl. Water Sci.. 2018;8:98.
- [CrossRef] [Google Scholar]
- Application of waste egg shell for adsorption of Cd(II) and Pb(II) ions to protect environment: equilibrium, kinetic and adsorption studies. Orient. J. Chem.. 2021;37:128-135.
- [CrossRef] [Google Scholar]
- Bio-sorption for tannery effluent treatment using eggshell wastes; kinetics, isotherm and thermodynamic study. Egypt. J. Pet.. 2020;29:273-278.
- [CrossRef] [Google Scholar]
- Pseudo-second order model for sorption processes. Process Biochem.. 1999;34:451-465.
- [CrossRef] [Google Scholar]
- Biocompatible FeOOH-Carbon quantum dots nanocomposites for gaseous NOx removal under visible light: Improved charge separation and High selectivity. J. Hazard. Mater.. 2018;354:54-62.
- [CrossRef] [Google Scholar]
- Comparative lead adsorptions in synthetic wastewater by synthesized zeolite A of recycled industrial wastes from sugar factory and power plant. Heliyon. 2022;8:e09323.
- [Google Scholar]
- Adsorption of lead (Pb-II) using CaO-NPs synthesized by solgel process from hen eggshell: Response surface methodology for modeling, optimization and kinetic studies. South African J. Chem. Eng.. 2022;40:209-229.
- [CrossRef] [Google Scholar]
- Cadmium removal mechanisms from aqueous solution by using recycled lignocelluloses. Alexandria Eng. J.. 2022;61:443-457.
- [CrossRef] [Google Scholar]
- Adsorption of Ni(II) ion onto calcined eggshells: a study of equilibrium adsorption isotherm. Indones. J. Chem.. 2019;19:143-150.
- [CrossRef] [Google Scholar]
- Experimental and kinetic study of removal of lead (Pb+2)from battery effluent using sweet lemon (Citrus limetta) peel biochar adsorbent. Environ. Dev. Sustain.. 2020;22:4379-4406.
- [Google Scholar]
- Zero-cost agricultural wastes as sources for activated carbons synthesis: lead ions removal from wastewaters. Proc.. 2018;2:652.
- [CrossRef] [Google Scholar]
- About the theory of so-called adsorption of soluble substances. K. Sven. Vetenskapsakademiens Handl.. 1898;24:1-39.
- [Google Scholar]
- The adsorption of gases on plane surfaces of glass, mica and platinum. J. Am. Chem. Soc.. 1918;40:1361-1403.
- [Google Scholar]
- Synthesis of nano-calcium oxide from waste eggshell by sol-gel method. Sustainability. 2019;11:2-10.
- [Google Scholar]
- Heavy metals removal from aqueous solutions by Al2O3 nanoparticles modified with natural and chemical modifiers. Clean Technol. Environ. Policy. 2015;17:85-102.
- [CrossRef] [Google Scholar]
- Study of mathematical models for the removal of Ni2+ from aqueous solutions using Citrullus lanatus rind, an agro-based waste. Water Environ. J.. 2019;33:276-291.
- [CrossRef] [Google Scholar]
- Application of activated eggshell as effective adsorbent in the removal of lead (II) ion from fertilizer plant eEffluent. J. Mater. Sci. Res. Rev.. 2020;5:18-36.
- [Google Scholar]
- Removal of Cd2+ in water by FeSO4 magnetic modified eggshell adsorbent. Desalin. Water Treat.. 2022;247:161-172.
- [CrossRef] [Google Scholar]
- Simultaneous adsorption of tetracycline, amoxicillin, and ciprofloxacin by pistachio shell powder coated with zinc oxide nanoparticles. Arab. J. Chem.. 2020;13:4629-4643.
- [CrossRef] [Google Scholar]
- Enhancement of ciprofloxacin antibiotic removal from aqueous solution using zno nanoparticles coated on pistachio shell. Desalin. Water Treat.. 2021;213:229-239.
- [CrossRef] [Google Scholar]
- The effect of activated carbon produced from remnants of walnut shell and ox tongue leftover on removing cadmium metal from water. Desalin. Water Treat.. 2022;256:176-193.
- [CrossRef] [Google Scholar]
- Fabrication, characterization and theoretical investigation of novel Fe3O4@egg-shell membrane as a green nanosorbent for simultaneous preconcentration of Cu (II) and Tl (I) prior to ETAAS determination. Environ. Nanotechnol. Monit. Manage.. 2018;10:171-178.
- [CrossRef] [Google Scholar]
- Synthesis of powdered and beaded chitosan materials modified with ZnO for removing lead (II) ions. Sci. Rep.. 2022;12:17184.
- [CrossRef] [Google Scholar]
- Modified beaded materials from recycled wastes of bagasse and bagasse fly ash with iron (III) oxide-hydroxide and zinc oxide for the removal of reactive blue 4 dye in aqueous solution. ACS Omega. 2022;7:34839-34857.
- [CrossRef] [Google Scholar]
- Modified alginate beads with ethanol extraction of Cratoxylum formosum and Polygonum odoratum for antibacterial activities. ACS Omega. 2021;6:32215-32230.
- [CrossRef] [Google Scholar]
- Antibacterial activities against Staphylococcus aureus and Escherichia coli of extracted Piper betle leaf materials by disc diffusion assay and batch experiments. RSC Adv.. 2022;12:26435-26454.
- [CrossRef] [Google Scholar]
- Comparative antibacterial activities of Garcinia cowa and Piper sarmentosum extracts against Staphylococcus aureus and Escherichia coli with studying on disc diffusion assay, material characterizations, and batch experiments. Heliyon. 2022;8:e11704.
- [Google Scholar]
- Production of high-performance lead(II) ions adsorbents from pea peels waste as a sustainable resource. Waste Manag. Res.. 2021;39:584-593.
- [CrossRef] [Google Scholar]
- Sources of lead exposure in various countries. Rev. Environ. Health. 2019;34:25-34.
- [CrossRef] [Google Scholar]
- Development and characterization of magnetic eggshell membranes for lead removal from wastewater. Ecotoxicol. Environ. Saf.. 2020;192:110307
- [CrossRef] [Google Scholar]
- Comparative reactive blue 4 dye removal by lemon peel bead doping with iron (III) oxide-hydroxide and zinc oxide. ACS Omega. 2022;7:41744-41758.
- [CrossRef] [Google Scholar]
- Zeolite A powder and beads from sugarcane bagasse fly ash modified with iron(III) oxide-hydroxide for lead adsorption. Sci. Rep.. 2023;13:1873.
- [CrossRef] [Google Scholar]
- Chicken and duck eggshell beads modified with iron (III) oxide-hydroxide and zinc oxide for reactive blue 4 dye removal. Arab. J. Chem.. 2022;15:104291
- [CrossRef] [Google Scholar]
- Powdered and beaded sawdust materials modified iron (III) oxide - hydroxide for adsorption of lead (II) ion and reactive blue 4 dye. Sci. Rep.. 2023;13:531.
- [CrossRef] [Google Scholar]
- Modification of sugarcane bagasse with iron(III) oxide-hydroxide to improve its adsorption property for removing lead(II) ions. Sci. Rep.. 2023;13:1467.
- [CrossRef] [Google Scholar]
- Reactive blue 4 adsorption efficiencies on bagasse and bagasse fly ash beads modified with titanium dioxide (TiO2), magnesium oxide (MgO), and aluminum oxide (Al2O3) Ind. Crop. Prod.. 2023;191:115928
- [CrossRef] [Google Scholar]
- Removal of heavy metal ions from wastewater: a comprehensive and critical review. npj Clean Water. 2021;4
- [CrossRef] [Google Scholar]
- Use of chicken feather and eggshell to synthesize a novel magnetized activated carbon for sorption of heavy metal ions. Bioresour. Technol.. 2020;297:122452
- [CrossRef] [Google Scholar]
- Chitosan nanofibers functionalized by TiO2 nanoparticles for the removal of heavy metal ions. J. Taiwan Inst. Chem. Eng.. 2016;58:333-343.
- [CrossRef] [Google Scholar]
- Recommendations for the characterization of porous solids (Technical report) Pure Appl. Chem.. 1994;66:1739-1758.
- [CrossRef] [Google Scholar]
- Evaluation of eggshell-rich compost as biosorbent for removal of Pb(II) from aqueous solutions. Water. Air. Soil Pollut.. 2016;227:150.
- [CrossRef] [Google Scholar]
- Characterization of residual biomasses and its application for the removal of lead ions from aqueous solution. Appl. Sci.. 2019;9:4486.
- [CrossRef] [Google Scholar]
- Kinetics of ammonia synthesis on promoted iron catalysts. Acta physiochim. URSS. 1940;12:327-356.
- [Google Scholar]
- Powdered and beaded lemon peels-doped iron (III) oxide-hydroxide materials for lead removal applications: Synthesis, characterizations, and lead adsorption studies. J. Environ. Chem. Eng.. 2021;9:106007
- [CrossRef] [Google Scholar]
- Efficacy study of recycling materials by lemon peels as novel lead adsorbents with comparing of material form effects and possibility of continuous flow experiment. Environ. Sci. Pollut. Res.. 2022;29:46077-46090.
- [CrossRef] [Google Scholar]
- Efficiency of calcium carbonate from eggshells as an adsorbent for cadmium removal in aqueous solution. Sustain. Environ. Res.. 2018;28:326-332.
- [CrossRef] [Google Scholar]
- Preparation and application of granular bentonite-eggshell composites for heavy metal removal. J. Porous Mater.. 2022;29:817-826.
- [CrossRef] [Google Scholar]
- Comparing the adsorption behaviors of Cd, Cu and Pb from water onto Fe-Mn binary oxide, MnO2 and FeOOH. Front. Environ. Sci. Eng.. 2015;9:385-393.
- [CrossRef] [Google Scholar]







