Translate this page into:
The young fruit of Citrus aurantium L. or Citrus sinensis Osbeck as a natural health food: A deep insight into the scientific evidence of its health benefits
⁎Corresponding authors at: 300 Xueshi Rd., Hanpu Science & Technology Park, Yuelu District, Changsha 410208, China. cshtyh@163.com (Shenghuang Chen), wangwei402@hotmail.com (Wei Wang)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Fruits are consumed as foods or medicines to supply people with nutrition or treat diseases. Zhishi, the dried young fruit of Citrus aurantium L. or Citrus sinensis Osbeck, is one of the most representative health food from the fruit of the Citrus genus. It is widely used in flavorings, canned food, beverages, and medicines because of its outstanding curative effects. The bidirectional regulating effect of Zhishi on the gastrointestinal tract for treating food stagnation or diarrhea has been confirmed. Its active ingredients, including synephrine and N-methyltyramine, have been used clinically as blood pressure boosting and anti-shock drugs. Flavonoids and alkaloids of Zhishi also make it potential weight loss and beauty products due to their definite effectiveness and safety. This paper intends to review the different therapeutic applications of Zhishi and the phytochemicals associated with its medicinal values. Besides, up-to-date information on its botany and analytical methods for the quality control of the medicine is supplied. To conclude, numerous independent research on Zhishi have been conducted in the past decades, but most of them are not deep enough in elucidating its scientific evidence of its health benefits. Further studies may unveil additional pharmacological activities and is beneficial to the mankind.
Keywords
Rutaceae
Fruit
Aurantii Fructus Immaturus
Health food
Citrus aurantium L.
Citrus sinensis Osbeck
1 Introduction
Fruit, one of the most common foods, is loved by people all over the world for its good taste and rich nutrition. Some are also used as medicines because of their therapeutic effect on specific diseases. Herbal material used for both food and Traditional Chinese Medicine (TCM) is also known as dietary herbal medicine or health food (Shan et al., 2015). Dietary herbal medicine can be directly sold and used as food casually but uses of health food in products for healthcare purposes require the approval of the regulatory authority.
With a plantation area of 9 million hectares and a fruit production of 122.3 million tons worldwide in 2009, the Citrus genus became the largest productive fruit crop (Xu et al., 2018). The Citrus genus originates from Southeast Asia, and China has a history of growing them for more than 2,400 years. Among 78 fruit medicines recorded in the Chinese Pharmacopoeia (ChP, 2020 edition) (Chinese Pharmacopoeia Commission, 2020a), 8 originated from the Citrus genus, which makes it the most abundant botanical source for fruit-derived medicines.
Zhishi (ZS), the dried young fruit of C. aurantium and its cultivars (known as Suancheng Zhishi, SCZS) or C. sinensis (known as Tiancheng Zhishi, TCZS) gathered between May to June, is one of the most important Qi-regulating medicine in the clinical practice of TCM system. It is primarily used for the auxiliary treatment of distension, abdominal pain, chest pain, phlegm, severe diarrhea, and organ prolapse induced by Qi stagnation. It possesses broad pharmacological actions such as effects on gastrointestinal and cardiovascular systems. More than 121 compounds, including flavonoids, coumarins, alkaloids, limonoids, and other compounds, along with some minor ingredients in significance such as polysaccharides and organic acids, have been isolated and identified from ZS. Synephrine significantly increases blood pressure without any side effects (Stohs, 2017), which is used as the marker for quality control, according to ChP (Chinese Pharmacopoeia Commission, 2020b).
However, the plant resource of ZS has changed from Poncirus trifoliata (L.) Raf to C. aurantium, and the planting area has also changed from north of the Yangtze River to Jiangxi, Sichuan, and Hunan Province. A variety of adulterations emerged with the historical changes, which led to the impossibility of guaranteeing the safety and effectiveness of ZS in clinic practice (Cai et al, 1999; Li, 2002). Besides, the pharmacological mechanism of Qi-regulating effects of ZS in the TCM theory system remains unclear. To further exploit the therapeutic potentials and supply scientific basis for its development and utilization of ZS, its application in TCM or health food, phytochemistry, quality control, pharmacological activity, toxicology, and clinical use are summarized in this paper.
2 Fruits used in health food or dietary herbal medicine
2.1 Fruits in TCM
Among 590 crude drugs documented in the latest ChP (Chinese Pharmacopoeia Commission, 2020a), 130 were derived from seed or fruit (Fig. 1), accounting for 25 % of the total, only less than root and rhizome, which account for 30 %. They were used for treating a variety of diseases due to their various efficacy in the TCM system. Besides, different parts of the fruit, such as pericarps, clusters, etc., can also be used separately as TCMs (Table 1). For instance, Lajiao (fruit of Capsicum annuum L.) was considered to have therapeutic effects on stomachache, emesis and chilblain (Yan et al., 2018). Goji (fruit of Lycium barbarum L.) had the ability to maintain the function of the eyes and strengthen the activity of the liver, kidneys, and lungs (Cheng et al., 2014). Dazao (fruit of Ziziphus jujuba Mill.) was believed to reinforce the spleen and stomach and was commonly used for the treatment of anorexia, fatigue, and loose stools related to deficiency syndromes of the spleen (Li et al., 2018). *Dietary herbal medicine, #Health food.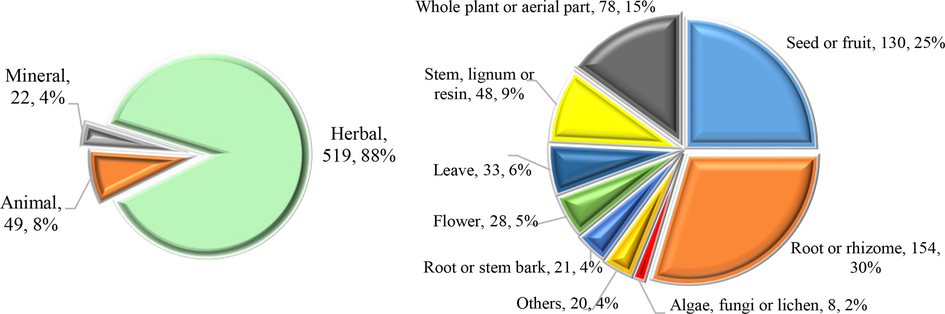
Sources of crude drugs recorded in ChP (2020 edition).
No.
Chinese name
English name
Species
Used parts
Harvesting seasons
Applications
1
Bajiaohuixiang*
Anisi Stellati Fructus
Illicium verum Hook. f.
Mature fruits
Autumn and winter
Vomiting, stomach ache, inflammation, insomnia, and rheumatic pain
2
Dazaojiao
Gleditsiae Sinensis Fructus
Gleditsia sinensis Lam.
Mature fruits
Autumn
Apoplexy, expectorant, and pesticide
3
Dazao*
Jujubae Fructus
Ziziphus jujuba Mill.
Mature fruits
Autumn
Digestive disorders, skin infections, urinary trouble, and cardiovascular diseases
4
Dafupi
Arecae Pericarpium
Areca catechu L.
Pericarps
Winter to the following spring
Parasitic diseases, dyspepsia, diarrhea, edema, and jaundice
5
Shanzhuyu#
Corni Fructus
Cornus officinalis Sieb. et Zucc.
Sarcocarp
Late autumn and early winter
Spontaneous sweating, spermatorrhea, and enuresis
6
Shanzha*
Crataegi Fructus
Crataegus pinnatifida Bge. var. major N. E. Br.
Crataegus pinnatifida Bge.Mature fruits
Autumn
Hypertension and hyperlipidaemia
7
Chuanlianzi
Toosendan Fructus
Melia toosendan Sieb. et Zucc.
Mature fruits
Winter
Irregular menstruation, hyperplasia of mammary glands, and breast-cancer
8
Guangzao
Choerospondiatis Fructus
Choerospondias axillaris (Roxb.) Burtt et Hill
Mature fruits
Autumn
Cardiovascular diseases
9
Nüzhenzi#
Ligustri Lucidi Fructus
Ligustrum lucidum Ait.
Mature fruits
Winter
Osteoporotic bone pain and rheumatic bone
10
Xiaoyelian
Sinopodophylli Fructus
Sinopodophyllum hexandrum (Royle) Ying
Mature fruits
Autumn
Irregular menstruation, hyperplasia of mammary glands, and breast-cancer
11
Xiaohuixiang*
Foeniculi Fructus
Foeniculum vulgare Mill.
Mature fruits
Autumn
Hepatoprotective and remediation of liver toxicity
12
Mugua*
Chaenomelis Fructus
Chaenomeles speciosa (Sweet) Nakai
Nearly mature fruits
Summer and autumn
Weakness of muscles and bones, muscle pain, and arthritis
13
Wuweizi#
Schisandare Chinensis Fructus
Schisandra chinensis (Turcz.) Baill.
Mature fruits
Autumn
Dysphoria and palpitation, insomnia, and dreaminess
14
Niubangzi#
Arctii Fructus
Arctium lappa L.
Mature fruits
Autumn
Throat pain and swelling, and detoxification
15
Maohezi
Terminaliae Belliricae Fructus
Terminalia bellirica (Gaertn.) Roxb.
Mature fruits
Winter
Diabetes, hypertension, and rheumatism
16
Huajuhong
Citri Grandis Exocarpium
Citrus grandis ‘Tomentosa’
Citrus grandis (L.) OsbeckImmature or nearly
mature epicarpsSummer
Cough, copious phlegm, indigestion, hyperglycemia, and hyperlipemia
17
Wumei*
Mume Fructus
Prunus mume (Sieb.) Sieb. et Zucc.
Nearly mature fruits
Summer
Chronic cough and expectoration
18
Huomaren*
Cannabis Fructus
Cannabis sativa L.
Mature fruits
Autumn
Anti-inflammatory and antioxidant
19
Badou
Crotonis Fructus
Croton tiglium L.
Mature fruits
Autumn
Constipation, visceral pain, and intestinal inflammation
20
Shuifeiji
Silybi Fructus
Silybum marianum (L.) Gaertn.
Mature fruits
Autumn
Hepatitis, cirrhosis, toxic liver damage, and jaundice
21
Shuihonghuazi
Polygoni Orientalis Fructus
Polygonum orientate L.
Mature fruits
Autumn
Swelling, indigestion, hypertension, cardiomyopathy, and chronic hepatitis
22
Shiliupi
Granati Pericarpium
Punica granatum L.
Pericarps
Autumn
Diarrhoea, haemostatic and insect repellent
23
Gualou
Trichosanthis Fructus
Trichosanthes kirilowii Maxim.
Trichosanthes rosthornii HarmsMature fruits
Autumn
Cardiovascular disease and cerebral ischaemic diseases
24
Gualoupi
Trichosanthis Pericarpium
Trichosanthes kirilowii Maxim.
Trichosanthes rosthornii HarmsPericarps
Autumn
Qi Stagnation, regulate lipid metabolism, and atherosclerosis
25
Dongguapi
Benincasae Exocarpium
Benincasa hispida (Thunb.) Cogn
Epicarps
Gastrointestinal disease, respiratory disease, and diabetes
26
Dongkuiguo
Malvae Fructus
Malva verticillata L.
Mature fruits
Summer and autumn
Constipation, diuresis, and galactagogue
27
Mudingxiang
Caryophylli Fructus
Eugenia caryophyllata Thunb.
Nearly mature fruits
Invigorate blood circulation, cold pain of heart and abdomen, and galactagogue
28
Difuzi
Kochiae Fructus
Kochia scoparia (L.) Schrad.
Mature fruits
Autumn
Antibacterial, inflammation, gastric ulcer, hypoglycemic, and immunodepression
29
Xiqingguo
Chebulae Fructus Immaturus
Terminalia chebula Retz.
Young fruits
Fevers, diarrhea, asthma, rheumatism, and dysentery
30
Hongdoukou
Galangae Fructus
Alpinia galanga Willd.
Mature fruits
Autumn
Emesis, diarrhea, and indigestion
31
Huajiao*
Zanthoxyli Pericarpium
Zanthoxylum schinifolium Sieb. et Zucc.
Zanthoxylum bungeanum Maxim.
Mature pericarps
Autumn
Gastric diseases, indigestion, diarrhea, and toothache
32
Cang’erzi
Xanthii Fructus
Xanthium sibiricum Patr.
Mature fruits with involucre
Autumn
Allergic rhinitis and other nasal disease
33
Doukou#
Amomi Fructus Rotundus
Amomum kravanh Pierre ex Gagnep.
Amomum compactum Soland ex MatonMature fruits
“Humidness evil” eliminating and qi promoting, stomach cold,vomit, and indigestion
34
Lianqiao
Forsythiae Fructus
Forsythia suspensa (Thunb.) Vahl
Fruits
Autumn
Hepatoprotective, neuroprotective, and cardiovascular protective
35
Wuzhuyu#
Euodiae Fructus
Euodia rutaecarpa (Juss.) Benth.
Euodia rutaecarpa (Juss.) Benth. var. officinalis (Dode) Huang
Euodia rutaecarpa (Juss.) Benth. var. bodinieri (Dode) HuangNearly mature fruits
August to November
Bacterial infection and inflammation
36
Foshou*
Citri Sarcodactylis Fructus
Citrus medica L. var. sarcodactylis Swingle
Fruits
Autumn
Indigestion, hepatic stagnation, “humidness evil” eliminating and copious phlegm
37
Yuganzi*
Phyllanthi Fructus
Phyllanthus emblica L.
Mature fruits
Winter to the following spring
Anti-tumor, anti-inflammatory, anti-bacterial, and anti-viral activities
38
Shaji*
Hippophae Fructus
Hippophae rhamnoides L.
Mature fruits
Autumn and winter
Indigestion, cough, copious phlegm, and blood stasis syndrome
39
Hezi#
Chebulae Fructus
Terminalia chebula Retz.
Terminalia chebula Retz. var. tomentella Kurt.Mature fruits
Autumn and winter
Cancer, diabetic, mutagenic, hepatoprotective, and cardio-protective
40
Buguzhi#
Psoraleae Fructus
Psoralea corylifolia L.
Mature fruits
Autumn
Leucoderma, cardiovascular diseases, nephritis, bone fracture and osteoporosis
41
Chenpi
Citri Reticulatae Pericarpium
Citrus reticulata Blanco
Mature pericarps
Qi stagnation, chest and hypochondriac region pain, and hernia-like pain.
42
Qingpi#
Citri Reticulatae Pericarpium Viride
Citrus reticulata Blanco
Pericarps of young or immature fruit
May to August
Liver Qi stagnation, disperse stagnation
43
Qingguo*
Canarii Fructus
Canarium album Raeusch.
Mature fruits
Autumn
Sore throat, cough and sputum viscosity
44
Luohanguo*
Siraitiae Fructus
Siraitia grosvenorii (Swingle) C. Jeffrey ex A. M. Lu et Z. Y. Zhang
Fruits
Autumn
Immuno-regulation, anti-oxidation, anti-cancer and anti-obesity
45
Shijunzi
Quisqualis Fructus
Quisqualis indica L.
Mature fruits
Autumn
Deworming
46
Jinyingzi#
Rosae Laevigatae Fructus
Rosa laevigata Michx.
Mature fruits
October to November
Chronic diseases
47
Bibo#
Piperis Longi Fructus
Piper longum L.
Nearly mature or mature clusters
Jaundice and allergy
48
Bichengqie
Litseae Fructus
Litsea cubeba (Lour.) Pers.
Mature fruits
Autumn
Abdominal cold pain, cold hernia, and stomach hiccup
49
Caoguo#
Tsaoko Fructus
Amomum tsao-ko Crevost et Lemaire
Mature fruits
Autumn
Stomach disorders and throat infection
50
Chongweizi
Leonuri Fructus
Leonurus japonicus Houtt.
Mature fruits
Autumn
Blood stasis syndrome and edema
51
Hujiao*
Piperis Fructus
Piper nigrum L.
Nearly mature or mature fruits
Last autumn to following spring
Anti-oxidant, anti-depressant, anti-tumor, and anti-inflammatory
52
Nanwuweizi
Schisandrae Sphenantherae Fructus
Schisandra sphenanthera Rehd. et Wils.
Mature fruits
Autumn
Insomnia, lipid peroxidation in liver, and myocardial ischemia
53
Nanheshi
Carotae Fructus
Daucus carota L.
Mature fruits
Autumn
Parasite, hemorrhoid fistula, viral keratitis, and edema
54
Zhiqiao#
Aurantil Fructus
Citrus aurantium L.
Immature fruits
July
Acute lung injury, inflammation, obesity, and gastrointestinal dysfunctions
55
Zhishi#
Aurantil Fructus Immaturus
Citrus aurantium L.
Citrus sinensis OsbeckYoung fruits
May to June
Copious phlegm, cancer, cardiovascular diseases, and gouty arthritis
56
Zhizi*
Gardeniae Fructus
Gardenia jasminoides Ellis
Mature fruits
September to November
Febrile diseases, jaundice, edema, and sprains
57
Gouqizi*
Lycii Fructus
Lycium barbarum L.
Mature fruits
Summer and autumn
Atherosclerosis, cancer, neurodegeneration, and diabetes
58
Sharen*
Amomi Fructus
Amomum villosum Lour.
Amomum villosum Lour. var. xanthioides T. L. Wu et Senjen
Amomum longiligulare T. L. WuMature fruits
Summer and autumn
Digestive diseases, rheumatism, malaria, toothache, and promoting appetite
59
Yadanzi
Bruceae Fructus
Brucea javanica (L.) Merr.
Mature fruits
Autumn
Prostate, lung, and gastrointestinal cancer
60
Xiangyuan*
Citri Fructus
Citrus medica L.
Citrus wilsonii TanakaMature fruits
Autumn
Stomach ache, headache, edema, rheumatism, arthritis, and infectious hepatitis
61
Xiakucao
Prunellae Spica
Prunella vulgaris L.
Clusters
Summer
Liver inflammation, improve eyesight, mammary gland hyperplasia and swelling
62
Yizhi
Alpiniae Oxyphyllae Fructus
Alpinia oxyphylla Miq.
Mature fruits
Summer and autumn
Dementia
63
Yuzhizi
Akebiae Fructus
Abebia quinata (Thunb.) Decne.
Akebia trifoliata (Thunb.) Koidz.
Akebia trifoliata (Thunb.) Koidz. var. australis (Diels) Rehd.Nearly mature fruits
Summer and autumn
Primary dysmenorrhea
64
Sangshen*
Mori Fructus
Morus alba L.
Clusters
April to June
Sore throats, anemia, and hypertension
65
Shechuangzi
Cnidii Fructus
Cnidium monnieri (L.) Cuss.
Mature fruits
Summer and autumn
Atopic dermatitis, asthma, psoriasis, urticaria, ringworm, and osteoporosis
66
Zhuyazao
Gleditsiae Fructus Abnormalis
Gleditsia sinensis Lam.
Infertility fruit
Autumn
Cerebral stroke sequelae
67
Chushizi
Broussonetiae Fructus
Broussonetia papyrifera (L.) Vent.
Mature fruits
Autumn
Alzheimer disease, neurons protect
68
Zisuzi*
Periliae Fructus
Perilla frutescens (L.) Britt.
Mature fruits
Autumn
Cough and asthma with copious phlegm and stiffness in the chest
69
Jili
Tribuli Fructus
Tribulus terrestris L.
Mature fruits
Autumn
Eye trouble, cutaneous pruritus, edema, tracheitis, and blood stasis syndrome
70
Huaijiao
Sophorae Fructus
Sophora japonica L.
Mature fruits
Winter
Heart diseases, neoplasms, inflammation, and hyperlipidemia
71
Lulutong
Liquidambaris Fructus
Liquidambar formosana Hance
Infructescences
Winter
Ovarian cancer, prostatic cancer, and liver cancer
72
Manjingzi
Viticis Fructus
Vitex trifolia L. var. simplicifolia Cham.
Vitex trifolia L.Mature fruits
Autumn
Colds, head-ache, migraine, and eye pain
73
Yingsuqiao
Papaveris Pericarpium
Papaver somniferum L.
Shells
Autumn
Chronic cough, chronic lax, and cramp
74
Lajiao
Capsici Fructus
Capsicum annuum L.
Mature fruits
Summer and autumn
Stomachache, emesis, and chilblain
75
Ruiren
Prinsepiae Nux
Prinsepia uniflora Batal.
Prinsepia uniflora Batal. var. serrata Rehd.
Kernels
Between summer and autumn
Respiratory infections in children
76
Heshi
Carpesii Fructus
Carpesium abrotanoides L.
Mature fruits
Autumn
Anti-diarrheal, anti-inflammatory, abirritatiye, and antibacterial
77
Juhong
Citri Exocarpium Rubrum
Citrus reticulata Blanco
Epicarps
Last autumn and early winter
Respiratory diseases
78
Fupenzi
Rubi Fructus
Rubus chingii Hu
Fruits
Summer
Enuresis, impotence, frequency of micturition, and spermatorrhea
Furthermore, they have therapeutic effects not only when used alone but also used in combination with other drugs. Zhishi Xiebai Guizhi decoction, one of the classical prescriptions, could improve chest, impediment, and heart pains, which are the symptoms of coronary heart disease and myocardial infarction in modern medicine. In this prescription, Trichosanthis Fructus and Allii Macrostemonis Bulbus work as the chief medicine to treat the main cause of the disease. ZS and Magnoliae Officinalis Cortex act as the deputy medicine to enhance the curative effect of chief medicine or treat concomitant symptoms due to the effect of Qi-regulating and phlegm-eliminating. Cinnamomi Ramulus plays an auxiliary effect as an assistant drug (Sang et al., 2021). Zhishi Daozhi pill was used for the treatment of bloating and constipation, in which ZS is used as the deputy medicine for producing a Qi-regulating effect to promote the purgation effect of Rhei Radix et Rhizoma of chief medicine. (Liu and Shen, 2010). Zhishi Xiaopi pill was used primarily for the treatment of dyspepsia to nourish the spleen and stomach with ZS as the chief medicine owing to its Qi-regulating, phlegm-eliminating, and digestion-eliminating effects (Lin et al., 1998).
2.2 Fruits in the diet
According to the National Health Commission of the People’s Republic of China (NHCPRC), 21 fruit-derived medicines in the ChP used for both medicine and food (Chinese Pharmacopoeia Commission, 2020a), including Bajiaohuixiang, Xiaohuixiang, et al., were listed as dietary herbal medicine based on their health benefits, safety evaluation, and especially a time-honored history of applications (Table 1).
However, some fruits in daily life, although they are not included in the list, were still used as dietary herbal medicine or health food in the folk, such as Ningmeng (fruit of Citrus limon (L.) Burm. f.) (Elena et al., 2008), Lizhi (fruit of Litchi chinensis Sonn.) (Zhao et al., 2020), Yingtao (fruit of Cerasus pseudocerasus (Lindl.) G. Don) (Jing et al., 2018) and Putao (fruit of Vitis vinifera L.) (Impei et al., 2015). In addition, Yueju (fruit of Vaccinium vitis-idaea L.) is consumed as food in raw or cooked in lingonberry jam, compote, juice, or syrup (Kowalska, 2021). Huaishi (fruit of Sophora japonica L.) is treated as one of the fat-reducing diets. Suanjiao (fruit of Tamarindus indica L.) is widely used as food flavorings and fruit drinks in Southeast Asia (Dai et al., 2015). These three health products are not recorded in ChP (Chinese Pharmacopoeia Commission, 2020a). In summary, as an essential part of the TCM system, fruit protects people’s health in daily life and plays a crucial role in treating diseases.
3 ZS used in the TCM or health food
3.1 ZS in the diet
The fruit of Citrus genus is one of the top four consumed fruits in the world. The juice content of the fruit of Citrus genus is up to 37.48 %∼52.46 % (Gao et al, 2022). It contains high content of amino acid and vitamin C (Hasan et al., 2022). However, ZS tastes sour and bitter, and as such it is not eaten fresh, so the most of them are processed into concentrated juice (Lv et al., 2015). According to the Food and Agricultural Organization of the United Nations, the fruit of C. aurantium is consumed mainly as fresh or raw materials for juice in the world-leading fruit-producing countries such as Brazil, America, India, Mexico, and Spain (Lv et al., 2015). The fruit of C. sinensis is widely distributed in the Mediterranean basin (such as Southern Italy and Spain) and America, becoming an essential element of the Sicilian kitchen (Barreca et al., 2015).
ZS is commonly used as a health food product in China for the benefits of relaxing the bowels and protecting against gastric mucosa damage. The candied fruit of C. aurantium has become a specialty in China's Hunan Province. According to the data of the State Administration for Market Regulation (National Health Commission of the People’s Republic of China, 2002), there are 48 types of domestic health food containing ZS extract in China and one health product (Baoxianpai JuJu Zhishi pills) imported from Denmark. Daidaihua, flower buds of its varieties Citrus aurantium L. var. amara Engl., were also recorded in the list of dietary herbal medicine, which is frequently used to make tea due to its positive effects on improving appearance and losing weight (Wang et al., 2009; Shen et al., 2019).
3.2 ZS used in TCM
3.2.1 History of ZS used in medicine
ZS has a long history of medicinal use. It was recorded as a medicine in the ShenNongBenCaoJing (the earliest book on TCM in China) in the Han Dynasty for the first time (202 BCE to 220 CE) (Wang, 1985; Li, 2002; Tsai, 2013). It was bitter in taste and cold in nature and was used to treat pruritus, dysentery, and skin disease formed by the combination of pathogenic heat/pathogenic cold and phlegm/blood stasis. Zhiqiao (ZQ), the dried immature fruit of C. aurantium harvested a month later than ZS, was used separately from ZS until the Tang dynasty (618–907 CE) due to their differences in therapeutic effects (Zhu and Li, 2013; Zhao et al., 2020). Officially they were separately recorded in KaiBaoBencao (the first official-revised Materia Medica in the Song dynasty) in 974 CE (Yu et al., 2004; Zhu and Li, 2013; Li et al., 2015). According to BenCaoGangMu (known as the great classic of Oriental Pharmacy), published in 1578 CE of the Ming Dynasty, ZS was bitter, sour, and slightly cold in the medicinal property and had functions of promoting Qi, reliving asthma, removing phlegm, relieving pain, and treating dysentery (Xu et al., 2012). It had been recorded in ChP since 1963 (Chinese Pharmacopoeia Commission, 1963; Tsai, 2013). The history of ZS used in medicine is summarized in Fig. 2.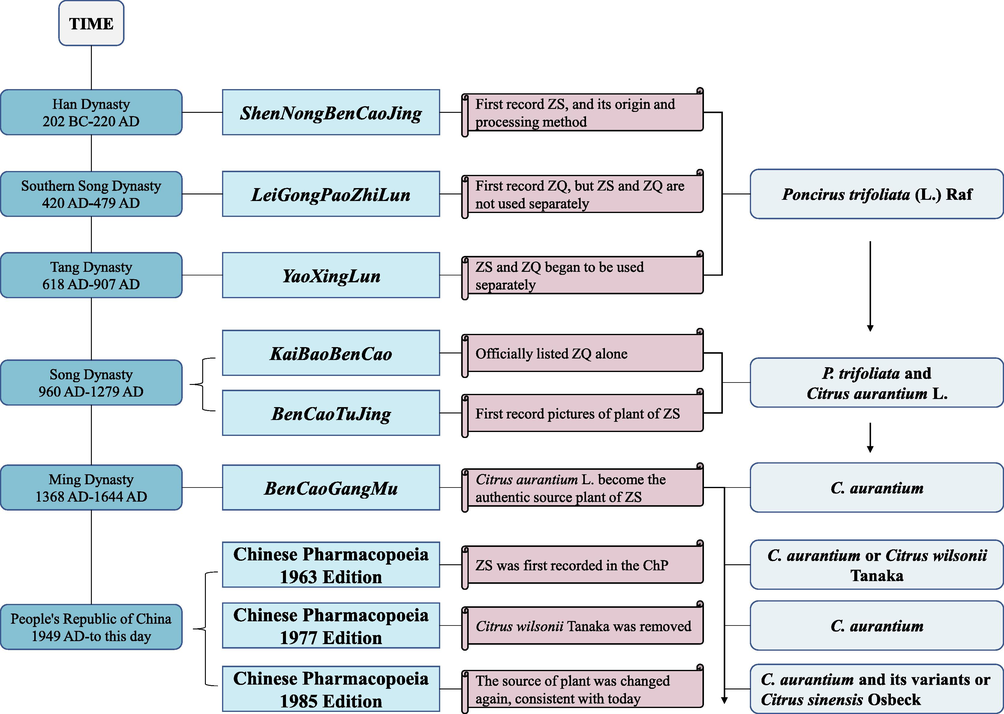
The history of ZS used in medicine and the changes in plant origin of ZS.
3.2.2 Changes in plant origin of ZS
The earliest ZS before the Song Dynasty was the fruit of P. trifoliata, which is known as Lvyi ZS nowadays in Fujian (Cai et al., 1998; Hu et al., 2019; Zhao et al., 2020). BenCaoTuJing (the earliest botanical atlas of medicinal herbs in China) included the first botanical picture of ZS in 1061 CE, which also confirmed that the fruit of P. trifoliata cannot be used as ZS (Xie, 1991; Cai et al., 1998; Xu et al., 2012; Zhao et al., 2020). In the Song Dynasty, the fruit of C. aurantium was gradually used as the authentic ZS (Cai et al., 1998; Xu et al., 2012). At that time, BenCaoGangMu made it clear that the fruit of P. trifoliata was the adulterant of ZS in the market (Xie, 1991; Xu et al., 2012; Hu et al., 2019; Zhao et al., 2020).
ChP recorded ZS for the first time in 1963, and the plant sources were C. aurantium and Citrus wilsonii Tanaka (Chinese Pharmacopoeia Commission, 1963). In the 1977 edition of the ChP, Citrus wilsonii Tanaka was removed (Chinese Pharmacopoeia Commission, 1977). While in the 1985 edition of the ChP, the plant source of ZS was changed to C. aurantium and its varieties, considering that the fruit of some varieties has a long history of being used as ZS (Chinese Pharmacopoeia Commission, 1985). Fruit thinning is usually conducted to ensure the quality of the fruit. For economic purposes, the young fruit of C. sinensis obtained by fruit thinning was gradually used as ZS in the market. It was first recorded as the source of ZS in the 1985 edition of ChP (Chinese Pharmacopoeia Commission, 1985; Xie, 1991; Zhou, 2014) (Fig. 2).
3.2.3 ZS and its common adulterations
C. aurantium and C. sinensis are distributed in the south of the Qinling Mountains. Most are artificially cultivated, while some are semi-wild (Fig. 3) (Wu, 2004). Jiangxi of China is the genuine producing area of SCZS and one of the famous products called “Eyan ZS” (Hu et al., 2019). While TCZS was mainly produced in Sichuan and Guizhou (Fig. 4). There are five cultivars of C. aurantium according to ChP which can be used for ZS (Chinese Pharmacopoeia Commission, 2020b; Xie, 1991) (shown in Table 2). Interestingly, C. sinensis currently cultivated in various countries, are all introduced from Guangdong or Fujian of southern China (Laura et al., 2019).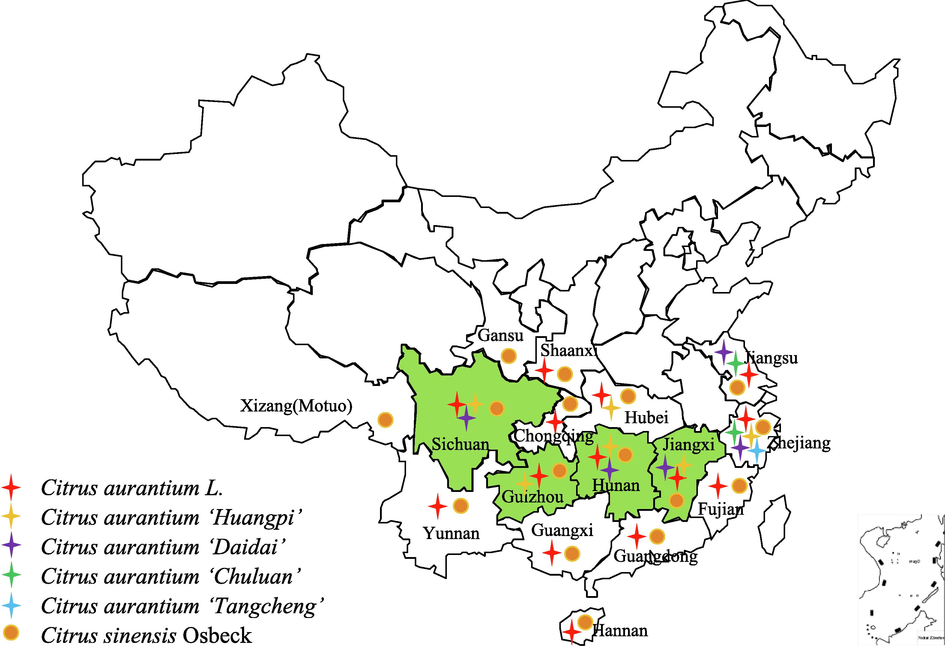
Climatic and ecological adaptability distribution of ZS in China.

(A) Eyan ZS; (B) SCZS; (C) TCZS.
Herbal name
Plant
Diameter (cm)
Ladybug pouch
Number of chambers (pcs)
Mesocarp thickness(cm)
Exocarp
Mesocarp
Transverse section
Carpopodium
Reference
ZS
Citrus aurantium L.
0.5–2.5
Sepia
7–12
0.3–1.2
Dark green or brownish-brown
Yellowish white or yellowish brown
Smooth and slightly bulging
Disk remnants or fruiting pedicel abscission scars
C. aurantium ‘Huangpi’
1.5–2.5
Light brown
8–10
0.5–0.9
Brown or greenish-yellow
Yellowish brown
Slightly curled or flat
Slightly protruding from the peel
(Li et al., 2007)
C. aurantium ‘Daidai’
1.7–2.5
Light brown
9–11
0.5–0.7
Tan or grey-brown
Yellowish brown
Slightly outward
Disc from the peel with slightly radiate ridge
(Liu et al., 2019)
C. aurantium ‘Chuluan’
0.75–1
Pale yellow
10–12
0.6–1.1
Brownish green or greenish black
Off-white
Thick and bulging
Dented into the peel, radial wrinkles all around
(Chen, 1998)
C. aurantium ‘Tangcheng’
*
*
*
*
Surface coarse
Yellowish white
Poor thickness
Protruding from the peel
(Wang et al., 2009)
Citrus sinensis Osbeck
1–2.5
Dark yellow to deep red
8–12
0.2–0.4
Dark brown
Off-white color
Flat
Disc
QP
Citrus reticulata Blanco
0.5–2
Light brown
8–10
0.1–0.2
Grey-green or black-green
Yellowish brown or light yellowish brown
Poor thickness
Disc
(He et al., 2021)
ZQ
Citrus aurantium L.
3–5
Brown or tan
7–12
0.4–1.3
Tan to brown
Yellowish white
Smooth and slightly outward
Dented into the peel
(Gao et al., 2020)
Juyuan
Citrus medica L.
4–10
Yellowish white
10–17
1–3
Yellow or chartreuse
Yellowish white or light yellowish brown
Transverse section with undulating margins
*
(Mondal et al., 2021)
Xiangyuan
Citrus wilsonii Tanaka
1.5–2.5
Brown or light reddish brown
9–11
0.4–0.8
Blackish green or yellowish brown
Light reddish brown
Extremely outward
Disc
(Yan et al., 2021)
Lvyi ZS
Poncirus trifoliata (L.) Raf
0.6–1.6
Yellowish white
5–7
0.3–0.6
Greenish-brown
Off-white color
Flat
Disc with slightly radiate ridge
(Jang et al., 2018)
Honghecheng
Citrus hongheensis Ye et al
3–5.5
Brown
10–12
1–1.3
Yellowish brown
Outer edges green, inner dark brown
Flat or inward
Longitudinal ribs or persistent calyx
(Zhang et al., 2020)
You
Citrus grandis (L.) Osbeck
1.5–3
Light brown
10–18
0.5–1.5
Tan and grey-brown
Light yellowish brown
Inward
Disc
(Anmol et al., 2021)
Xiecheng
Citrus.junos Sieb. ex Tanaka
2.5–4
Pale yellowish white
9–11
0.3–0.5
Turquoise
Pale red
Slightly outward
Coarse with radiate ridge
(Song et al., 2019)
Yichangcheng
Citrus.ichangensis Swingle
2.5–4
Yellowish white
5–7
0.2–0.4
Greyish green or light yellowish brown
Pale yellowish white
Flat
Protruding from the peel
(Ding et al., 2012)
Xiangcheng
Citrus aurantium ‘Xiangcheng’
0.5–2.5
Sepia
9–12
0.6–1.1
Brownish green or greenish black
Yellowish white
Outward and roll
Radiate ridge
(Luo et al., 2008)
Xiucheng
Citrus aurantium’Xiucheng’
0.5–2.5
Brownness
9–12
0.7–1.2
Grey-green or yellowish brown
Yellowish white
Slightly bulging and outward
Not obvious
(Ding and Lu, 1986)
Zhicheng
Poncirus trifoliata × C. aurantium
3.6–6
Milky white or milky yellow
6–8
0.3–0.6
Scarlet
Milky yellowish white
Flat
Slightly dented into the peel with a ring
(Lu, 2015)
The plant source of ZS has undergone a variety of changes in history. In addition, the fruits of many other Citrus plants share similar appearance, chemical constituents, and pharmacology with ZS. These reasons resulted in the phenomenon that the fruits of other Citrus plants were commonly used as adulterants of ZS in the market. Detailed adulterants information is shown in Table 2 and Fig. 5. The adulterants cannot be used as ZS due to the variations of their chemistry and efficacy (Li et al., 2016; Zeng et al., 1997; Zhong et al., 2021; Zhu and Pan, 2005; Li, 2002).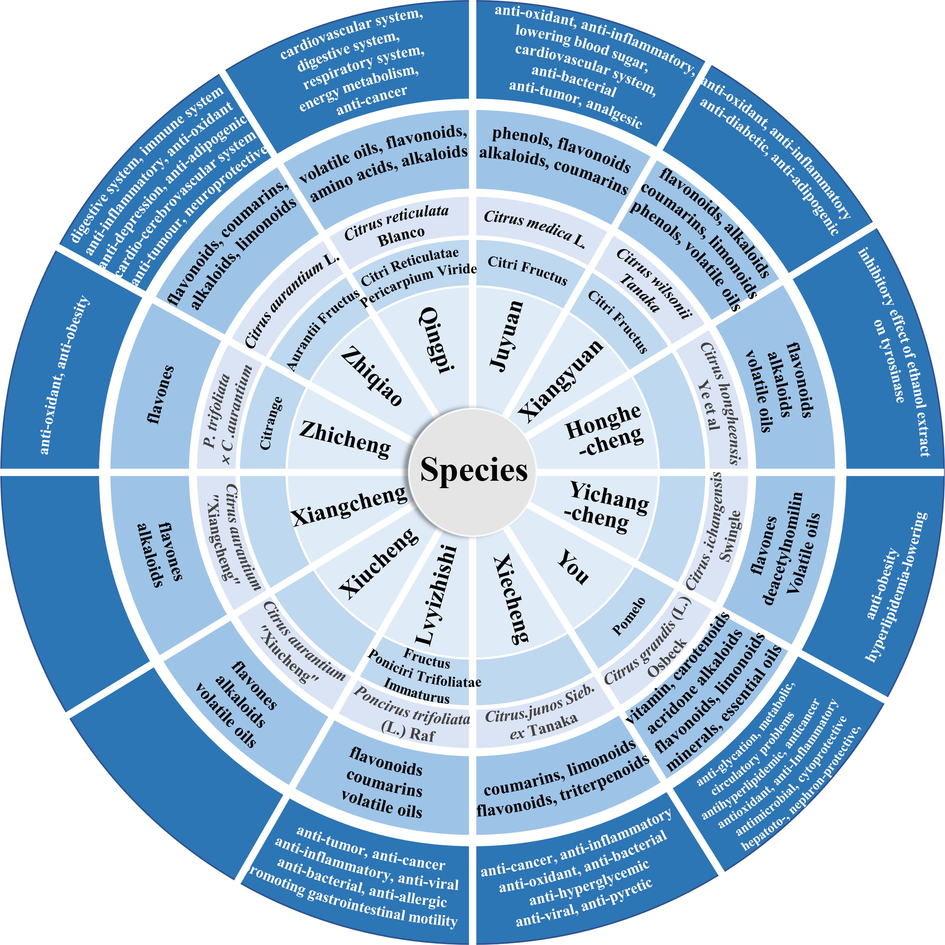
The adulterants of ZS.
3.2.4 Ethnopharmacology
3.2.4.1 Discussion on the medicinal property of ZS
In the TCM theory system, the properties of TCM, also known as the “Yao Xin”, include cold, hot, warm, and cool, which are summarized based on the reactions of medicines acting on the body. ZS was identified as “bitter, pungent, sour, and warm in nature” in both 1995 and 2000 editions of ChP (Chinese Pharmacopoeia Commission, 1995, 2000). The reasons are as follows (Liu and Zhou, 1988, 1993; Guo et al., 2001): (1) All Qi-moving drugs derived from the Citrus genus are warm in nature; (2) ZS is able to stimulate the sympathetic nerve and adrenal system, which belongs to the property of warm medicine; (3) ZS is often used in combination with warm-hot medicines to treat the cold syndrome.
In fact, ZS is actually used to treat the hot syndrome but not the cold syndrome. According to ShenNongBenCaoJing, ZS is bitter in taste and cold in nature. Among the 108 prescriptions containing ZS, 49 were used for the hot syndrome, accounting for about 45.4 % of the total. Forty-six were used for the cold and hot mixed or inconspicuous syndrome, and only 13 were used for the cold syndrome (Wu et al., 2008). Besides, in the 2020 edition of ChP (Chinese Pharmacopoeia Commission, 2020b), ZS is recorded as bitter, pungent, sour, and slightly cold. Therefore, the medicinal properties of ZS are either sightly cold or flat, rather than warm. However, the discussion still needs further experimental verification.
3.2.4.2 Application of ZS
Traditionally, it is believed that ZS used alone can improve stagnation of dyspepsia and gastrointestinal function, reduce chest pain and cure organ prolapse. Using alone or in combination with other herbs, the medicinal effect of ZS is severer than ZQ, which is named as “Po Qi” (Bai et al., 2018). ZS is used for gastrointestinal accumulation, while ZQ is used for the stagnation of Qi in the lungs or stomach.
Among the 100 classical prescriptions published by National Administration of Traditional Chinese Medicine in 2018, five contained ZS (Table 3). According to the database (https://db.yaozh.com/), there are 766 classic prescriptions containing ZS. So far, quality standards of 186 prescriptions containing ZS have been recorded by the NHCPRC. Among them, 40 were recorded in the ChP (Chinese Pharmacopoeia Commission, 2020b). However, only 3 Chinese patent medicines (Zhishi Daozhi pills, Zhishi Xiaopi pills, and Fufang Zhishi pills) are available on the market. Therefore, there is a great potential for the development and use of prescriptions of ZS.
Prescription name
Main herbs
Traditional use
References
Dynasty
Xiaochengqi Decoction
Rhei Radix et Rhizoma, Magnoliae Officinalis Cortex, Aurantii Fructus Immaturus
Reliving chronic constipation and food stagnation
ShangHanLun
Han (B.C.202-A.D.220)
Zhishi Xiebai Guizhi Decoction
Aurantii Fructus Immaturus, Magnoliae Officinalis Cortex, Allii Macrostemonis Bulbus, Cinnamomi Ramulus
Reducing heart and chest pain, expectorant
JinGuiYaoLue
Han (B.C.202-A.D.220)
Houpo Qiwu Decoction
Magnoliae Officinalis Cortex, Glycyrrhizae Radix et Rhizoma, Rhei Radix et Rhizoma, Jujubae Fructus, Aurantii Fructus Immaturus, Cinnamomi Ramulus, Zingiberis Rhizoma Recens
Dissipating cold and painful abdominal Mass
JinGuiYaoLue
Han (B.C.202-A.D.220)
Wendan Decoction
Pinelliae Rhizoma, Bambusae Caulis in Taenias, Aurantii Fructus Immaturus, peel of Citrus reticulata Blanco, Zingiberis Rhizoma Recens, Glycyrrhizae Radix et Rhizoma
Dissipating stagnant qi and eliminating sputum in gallbladder
BeiJiQianJinYaoFang
Tang (A.D.618–907)
Sanhua Decoction
Magnoliae Officinalis Cortex, Rhei Radix et Rhizoma, Aurantii Fructus Immaturus, Notopterygii Rhizoma et Radix
Treating stroke
SuWenBingJiQiYiBaoMing Ji
Jin (CE1115–1234)
3.3 Phytochemistry
A variety of methods have been used for the extraction, separation, and identification of chemical constituents of ZS. Currently, a total of 121 compounds have been isolated and identified from ZS, predominantly flavonoids, coumarins, alkaloids, limonoids, and other phenolic compounds.
3.3.1 Flavonoids
Flavonoids in ZS are mainly divided into four types: flavones, polymethoxy flavonoids, flavonols, and flavanones based on their differences in skeleton and substituents (Fig. 6).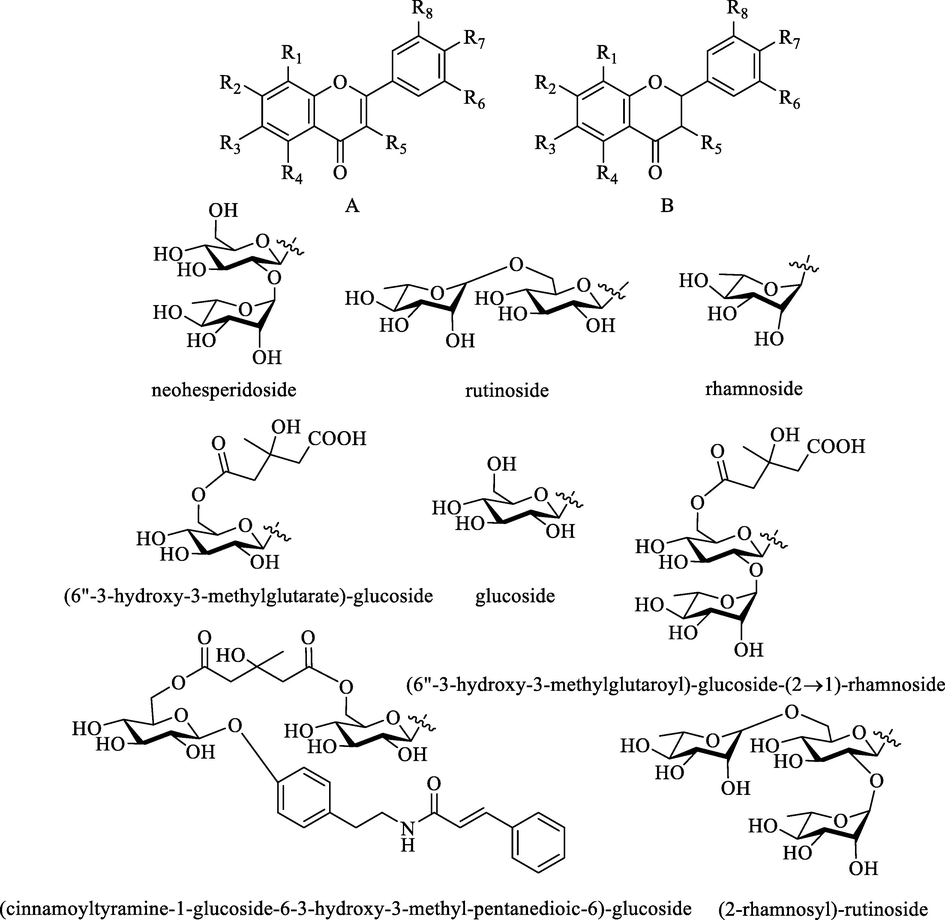
The skeleton of flavonoids and their substituent moieties in ZS.
3.3.1.1 Flavones
Flavones are 2-phenyl chromone derivatives without substituent at the C-3 position. Both C-5 and C-7 positions of its A ring are substituted by hydroxyl groups, while the C-4′ and C-3′ positions of its B ring are often substituted by hydroxyl groups or methoxy groups. Most of the flavones isolated from ZS are glycosides, including O-glycosides (1–7) and C-glycosides (8–15). As shown in Table 4 and Fig. 6, O-glycosides are mostly substituted in the C-7 position, while C-glycosides are mainly substituted in the C-8 position. *Detected by HPLC or LC-MS.
No.
Compound name
Skeleton
R1
R2
R3
R4
R5
R6
R7
R8
Reference
1
Rhoifolin
A
H
O-neohesperidoside
H
OH
H
H
OH
H
(Mencherini et al., 2013)
2
Rhoifolin-4′-glucoside
A
H
O-neohesperidoside
H
OH
H
H
O-glucoside
H
(Mencherini et al., 2013)
3
Luteolin-7-O-neohesperidoside
A
H
O-neohesperidoside
H
OH
H
OH
OH
H
(Mencherini et al., 2013)
4
Neodiosmin
A
H
O-neohesperidoside
H
OH
H
OH
OCH3
H
(Mencherini et al., 2013)
5
Chrysoeriol-7-O-neohesperidoside*
A
H
O-neohesperidoside
H
OH
H
H
OH
OCH3
(Barreca et al., 2015)
6
Diosmin*
A
H
O-rutinoside
H
OH
H
OH
OCH3
H
(Bai et al., 2018)
7
6-C-β-glucosyldiosmin
A
H
OH
O-glucoside
OH
H
OH
OCH3
H
(Zhang et al., 2019)
8
Lucenin-2*
A
glucoside
OH
glucoside
OH
H
H
OH
OH
(Barreca et al., 2015)
9
Apigenin-6,8-di-C-glucoside
A
glucoside
OH
glucoside
OH
H
H
OH
H
(Mencherini et al., 2013; Barreca et al., 2015)
10
Diosmetin-6,8-di-C-glucoside
A
glucoside
OH
glucoside
OH
H
OH
OCH3
H
(Mencherini et al., 2013)
11
Chysoeriol-6,8-di-C-glucoside*
A
glucoside
OH
glucoside
OH
H
H
OH
OCH3
(Barreca et al., 2015; Tong et al., 2018)
12
Scoparin*
A
glucoside
OH
H
OH
H
H
OH
OCH3
(Barreca et al., 2015)
13
3,8-di-C-glucosylapigenin
A
glucoside
OH
H
OH
glucoside
H
OH
H
(Matsubara and Sawabe, 1994)
14
3,8-di-C-glucosyldiosmetin
A
glucoside
OH
H
OH
glucoside
H
OCH3
OH
(Matsubara and Sawabe, 1994)
15
Quercetin-3-hydroxy-3-methylglutaryl-glycoside*
A
H
OH
H
OH
(6″-3-hydroxy-3-methylglutarate)-glucoside
H
OH
OH
(Barreca et al., 2015)
16
Tangeretin
A
OCH3
OCH3
OCH3
OCH3
H
H
OCH3
H
(Han et al., 2010)
17
Nobiletin
A
OCH3
OCH3
OCH3
OCH3
H
H
OCH3
OCH3
(Han et al., 2010)
18
5-demethyl nobiletin
A
OCH3
OCH3
OCH3
OH
H
H
OCH3
OCH3
(Zhang et al., 2015)
19
5-hydroxy-6,7,8,4′-tetramethoxyflavone
A
OCH3
OCH3
OCH3
OH
H
H
OCH3
H
(Jiang et al., 2016)
20
Isosinensetin*
A
OCH3
OCH3
H
OCH3
H
H
OCH3
OCH3
(Bai et al., 2018; Tong et al., 2018)
21
Tetramethyl-O-isoscutellarein*
A
OCH3
OCH3
H
OCH3
H
H
OCH3
H
(Tong et al., 2018)
22
5,7,8,4′-tetramethoxyflavone
A
OCH3
OCH3
H
OCH3
H
H
OCH3
H
(Han et al., 2010)
23
7,8,3′,4′-tetramethoxyflavone*
A
OCH3
OCH3
H
H
H
OCH3
OCH3
H
(Bai et al., 2018)
24
Sinensetin
A
H
OCH3
OCH3
OH
H
H
OCH3
OCH3
(Jiang et al., 2016)
25
5-hydroxy-6,7,3′,4′-tetramethoxyflavone
A
H
OCH3
OCH3
OCH3
H
H
OCH3
OCH3
(Jiang et al., 2016)
26
5,6,7,4′-tetramethoxyflavone*
A
H
OCH3
OCH3
OCH3
H
H
OCH3
H
(Bai et al., 2018)
3.3.1.2 Polymethoxy flavonoids
Polymethoxy flavonoids, unique flavonoids in Citrus plants, refer to a class of natural products containing four or more methoxy groups on the molecular skeleton at the C-5, C-8, C-3′, and C-5′ positions, or more at the C-7 and/or C-4′ positions (16–26). A total of 11 polymethoxy flavonoids (16–26) were isolated from ZS (Table 4).
3.3.1.3 Flavonols
Hydroxyl groups or other oxygen-containing groups are attached to the C-3 position of the skeleton of flavonoids. But flavonols with hydroxyl or methoxy groups in other positions were also isolated from ZS (27–39) (Table 5 and Fig. 6). *Detected by HPLC or LC-MS.
No.
Compound name
Skeleton
R1
R2
R3
R4
R5
R6
R7
R8
Reference
27
Natsudaidai
A
OCH3
OCH3
OCH3
OCH3
OH
H
OCH3
OCH3
(Jiang et al., 2016)
28
3,5,6,7,3′,4′-hexamethoxy flavone*
A
H
OCH3
OCH3
OCH3
OCH3
H
OCH3
OCH3
(Tong et al., 2018)
29
3-methoxynobiletin*
A
OCH3
OCH3
OCH3
OCH3
OCH3
OCH3
OCH3
H
(Bai et al., 2018)
30
Natsudaidain-3-O-glucoside*
A
OCH3
OCH3
OCH3
OCH3
O-glucoside
H
OCH3
OCH3
(Tong et al., 2018)
31
Quercetin-3-O-glycoside*
A
H
OH
H
OH
O-glucoside
H
OH
OH
(Barreca et al., 2015)
32
Rutin*
A
H
OH
H
OH
O-rutinoside
OH
OH
H
(Bai et al., 2018)
33
Nicotiflorin
A
H
OH
H
OH
O-rutinoside
H
OH
H
(Bai et al., 2018)
34
Narcissoside
A
H
OH
H
OH
O-rutinoside
OCH3
OH
H
(Bai et al., 2018)
35
Quercetin-3-O-(2-rhamnosyl)-rutinoside*
A
H
OH
H
OH
O-(2-rhamnosyl)-rutinoside
H
OH
OH
(Barreca et al., 2015)
36
Natsudaidain-3-O-(3-hydroxy-3-methylglutarate)-glucoside*
A
OCH3
OCH3
OCH3
OCH3
O-(6″-3-hydroxy-3-methylglutarate)-glucoside
H
OCH3
OCH3
(Tong et al., 2018)
37
Limocitrin-3-O-(3-hydroxy-3- methylglutarate)-glucoside*
A
OCH3
OH
H
OH
O-(6″-3-hydroxy-3-methylglutarate)-glucoside
H
OH
OCH3
(Tong et al., 2018)
38
5,7,4′-trihydroxy-8,3′-dimethoxyflavone-3-O-6″-(3-hydroxyl-3-methylglutaroyl)-β-d-glucopyranoside
A
OCH3
OH
H
OH
O-(6″-3-hydroxy-3-methylglutaroyl)-glucoside-(2 → 1)-rhamnoside
H
OH
OCH3
(Deng et al., 2020)
39
Citrusauranosides A
A
OCH3
OH
H
OH
O-(cinnamoyltyramine-1-glucoside-6–3-hydroxy-3-methyl-pentanedioic-6)-glucoside
OCH3
OH
H
(Zhang et al., 2019)
3.3.1.4 Flavanones
Flavanones in ZS are present in the forms of glycoside or aglycone. Among the aglycones, naringenin (40) and hesperetin (41) are the most important due to their broad pharmacological activity. Apart from glucosides (42–44), there are other two types of flavanones, including rutinosides (45–51) and neohesperidosides (53–56) (Table 6 and Fig. 6). *Detected by HPLC or LC-MS.
No.
Compound name
Skeleton
R1
R2
R3
R4
R5
R6
R7
R8
Reference
40
Naringenin
B
H
OH
H
OH
H
H
OH
H
(Liu et al., 2016)
41
Hesperetin
B
H
OH
H
OH
H
OH
OCH3
H
(Liu et al., 2016)
42
Hesperetin-7-O-β-d-glucoside
B
H
O-glucoside
H
OH
H
H
OCH3
OH
(Zhang et al., 2005; Feng et al., 2012)
43
Isosakuranin
B
H
O-glucoside
H
OH
H
H
OCH3
H
(Feng et al., 2012)
44
Hesperetin-7-O-β-d-glucopyranoside
B
H
O-glucoside
H
OH
H
H
OCH3
OH
(Jiang et al., 2017)
45
Narirutin
B
H
O-rutinoside
H
OH
H
H
OH
H
(Deng et al., 2020)
46
Eriocitrin
B
H
O-rutinoside
H
OH
H
H
OH
OH
(Deng et al., 2020)
47
Didymin*
B
H
O-rutinoside
H
OH
H
H
OCH3
H
(Tong et al., 2018)
48
Isosakuranetin-7-rutinoside*
B
H
O-rutinoside
H
OH
H
H
OCH3
H
(Sommella et al., 2017)
49
Hesperidin
B
H
O-rutinoside
H
OH
H
H
OCH3
OH
(Liu et al., 2016)
50
Methyl hesperidin
B
H
O-rutinoside
H
OH
H
H
OCH3
OCH3
(Yang, 2007)
51
Narirutin-4′-O-glucoside*
B
H
O-rutinoside
H
OH
H
H
O-glucoside
H
(Barreca et al., 2015; Tong et al., 2018)
52
Eriodictin
B
H
O-rhamnoside
H
OH
H
H
OH
OH
(Feng et al., 2012)
53
Naringin
B
H
O-neohesperidoside
H
OH
H
H
OH
H
(Liu et al., 2016)
54
Neoeriocitrin
B
H
O-neohesperidoside
H
OH
H
OH
OH
H
(Mencherini et al., 2013)
55
Poncirin
B
H
O-neohesperidoside
H
OH
H
H
OCH3
H
(Feng et al., 2012)
56
Neohesperidin
B
H
O-neohesperidoside
H
OH
H
OH
OCH3
H
(Liu et al., 2016)
57
Melitidin
B
H
O-(6″-3-hydroxy-3-methylglutaroyl)-glucoside-(2 → 1)-rhamnoside
H
OH
H
H
OH
H
(Mencherini et al., 2013)
58
Brutieridin
B
H
O-(6″-3-Hydroxy-3-methylglutaroyl)-glucoside-(2 → 1)-rhamnoside
H
OH
H
OH
OCH3
H
(Mencherini et al., 2013)
Flavanonols
59
Aromadendrin-7-O-β-d-glucopyranside
B
H
O-glucoside
H
OH
OH
H
OH
H
(Deng et al., 2020)
3.3.2 Coumarins
Coumarins are a class of natural compounds with a benzo-α-pyrone, which are divided into furanocoumarins and pyranocoumarins. They are also another major biologically active compound found in the Citrus genus. They were three types of coumarins in ZS, including typical coumarins (61–69), isocoumarins (70–72) and furanocoumarins (73–77) (Table 7 and Fig. 7). However, the isolation of pyranocoumarins from ZS has not been reported.
Type
No.
Compound name
Skeleton
R1
R2
R3
R4
Reference
Simple coumarins
60
Meranzin hydrate-β-d-glucoside
A
H
H
OCH3
8,3′-β-glucosyloxy-2′-hydroxy-3′-methylbutyl-7-
(Deng et al., 2018)
61
Meranzin hydrate
A
H
H
OCH3
7-methoxy-8-(2′,3′-dihydroxy-isopentyl)-
(Mencherini et al., 2013)
62
Isomeranzin
A
H
H
OCH3
7-methoxy-8-(2-oxo-3-methylbutyl)-
(Zhang et al., 2015)
63
Auraptene
A
H
H
7-[(3,7-dimethyl-2,6-octadienyl)oxy]-(E)-
H
(Satoh et al., 1995)
64
Marmin
A
H
H
7-(6′,7′-dihydroxygeranyloxy)
H
(Satoh et al., 1995)
65
Praealtin D
A
H
H
7-[(2,6,7-trihydroxy-7-methyl-3-methyleneoctyl)oxy]-
H
(Xiong et al., 2016)
66
Umbelliferone
A
H
H
OH
H
(Deng et al., 2018)
67
Scopoletin
A
H
OCH3
OH
H
(Feng et al., 2012)
68
5,7-dihydroxylcoumarin
A
OH
H
OH
H
(Feng et al., 2012)
69
5,7-dihydroxylcoumarin-5-O-β-d-glucopyranoside
A
O-glucoside
H
OH
H
(Zhang et al., 2005)
Isocoumarin
70
5,7-dihydroxychromone
B
H
OH
H
OH
(Zhang et al., 2015)
71
5,7-dihydroxy-chromone-7-neohesperidoside
B
H
O-neohesperidoside
H
OH
(Zhang et al., 2019)
72
5,7-dihydroxy-8-methoxychromone
B
OCH3
OH
H
OH
(Jiang et al., 2016)
Furanocoumarins
73
Xanthotoxol
C
H
OH
(Feng et al., 2012)
74
Bergapten
C
H
OCH3
(Xiong et al., 2016)
75
Citraurancoumarin A
C
OH
6,7-dihydroxy-3,7-dimethyloct-1-en-3-yl-
(Xiong et al., 2016)
76
5[(6′,7′-dihydroxy-3′,7′-dimethyl-2-octenyl) oxy]-psoralen
C
5-[(6′,7′-dihydroxy-3′,7′-dimethyl-2-octenyl)oxy]
H
(Satoh et al., 1995)
77
Citraurancoumarin B
C
7-[(2,6,7-trihydroxy-7-methyl-3-methyleneoctyl)oxy]-
H
(Xiong et al., 2016)
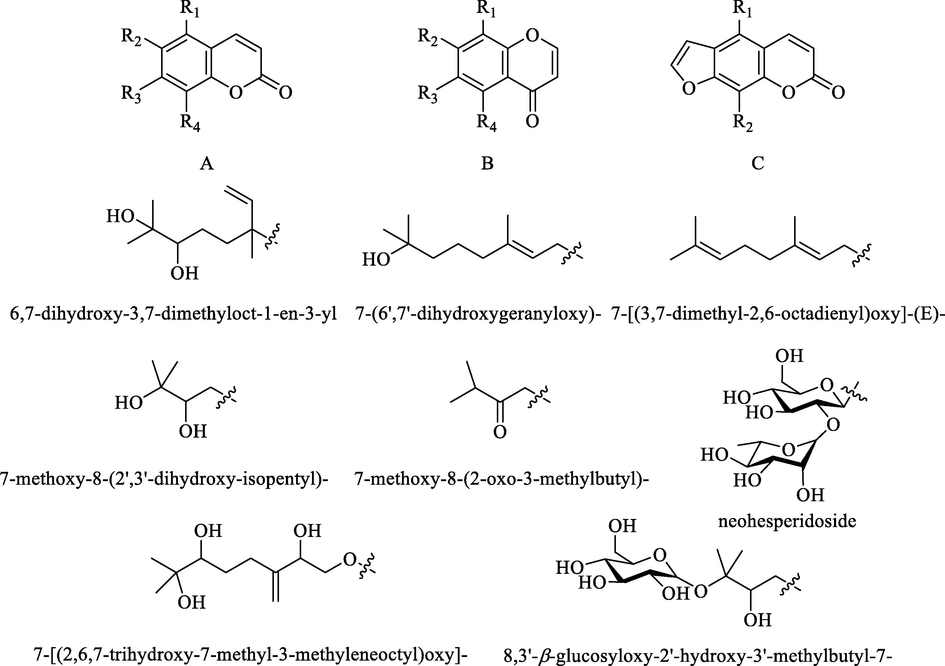
The skeleton of coumarins and their substituent moieties in ZS.
3.3.3 Alkaloids
The alkaloids in ZS possess the effects of blood pressure elevating, contraction of uterine smooth muscle promoting, cardiotonic, and anti-obesity. The intravenous injection of ZS has a significant effect on raising blood pressure. Synephrine (78) and N-methyltyramine (79) were identified from the elevating blood pressure components of ZS (Hu'Nan Medicine Industry Research Institute, 1976). Zhang used silica gel column chromatography, Sephadex LH-20 column chromatography, preparative high-performance liquid chromatography (HPLC), and other methods for the separation and purification of the four alkaloids (81–84) (Zhang et al., 2015). Peng also extracted and separated 4 alkaloids (78–80 and 86) from the ethanol extract of ZS to obtain (Peng et al., 2001). In addition, some other alkaloids (85, 87 and 88) with multiple nitrogen atoms were also isolated (Table 8 and Fig. 8) (Zhang et al., 2005; Deng et al., 2018). *Detected by HPLC or LC-MS.
Type
No.
Compound name
Reference
Alkaloids
78
Synephrine
(Hu'Nan Medicine Industry Research Institute, 1976)
79
N-methyltyramine
80
N-acetyloctopamine
(Peng et al., 2001)
81
N-benzoyl tyramine
(Zhang et al., 2015)
82
N-benzoyl tyramine methyl ether
(Zhang et al., 2015)
83
2-hydroxybenzoic acid N-2-(4-hydroxyphenyl) ethylamide
(Zhang et al., 2015)
84
N-[2-(4-hydroxyphenyl) ethyl]-3-methylbut-2-enamide
(Zhang et al., 2015)
85
Uracil
(Deng et al., 2018)
86
GABA
(Peng et al., 2001)
87
Thymidine
(Deng et al., 2018)
88
Adenosine
(Zhang et al., 2005)
Limonoids
89
Deacetyl nomilin
(Dandekar et al., 2008)
90
Limonin
(Dandekar et al., 2008)
91
Isoobacunoicacid
(Zhang et al., 2019)
92
Nomilin
(Zhao et al., 2017)
93
Obacunone*
(Bai et al., 2018)
94
Isolimonoic acid
(Jayaprakasha et al., 2008)
95
Deacetyl nomilinic acid glucoside
(Dandekar et al., 2008)
96
Ichanexic acid
(Jayaprakasha et al., 2008)
Phenols
97
Phloroglucinol
(Deng et al., 2020)
98
Methyl 3-(2′,4′-dihydroxy phenyl) propanoate
(Deng et al., 2020)
99
Trans-ferulic acid
(Zhang et al., 2019)
100
(3R)-Thunberginol C
(Zhang et al., 2019)
101
Phlorin
(Zhang et al., 2005)
102
6′-O-trans-cinnamoyl-3,5-dihydroxyphenyl-β-d-glucopyranoside
(Deng et al., 2020)
103
Citrusauranosides C
(Zhang et al., 2017)
104
Aurantiside A
(Zhang et al., 2006)
105
1-O-3,5-dihydroxyphenyl-(6-O-4-hydroxybenzoyl)-β-d-glucopyranoside
(Zhang et al., 2017)
106
Citrusauranosides B
(Zhang et al., 2019)
Others
107
Cymol
(Zhang et al., 2019)
108
Benzoic acid
(Xiong et al., 2016)
109
Cinnamic acid
(Zhang et al., 2019)
110
Rimboxo
(Deng et al., 2018)
111
Quinic acid*
(Bai et al., 2018)
112
Citrauranoside A
(Xiong et al., 2016)
113
6′-(β-d-apiosyl)-β-d-glucosyl-columbianetin
(Zhang et al., 2019)
114
Linaloyl glucoside
(Deng et al., 2018)
115
Picraquassioside A
(Zhang et al., 2019)
116
Citrusin C
(Matsubara and Sawabe, 1994)
117
Coniferin
(Sawabe et al., 1986)
118
Syrigin
(Sawabe et al., 1986)
119
Dehydrodiconiferyl alcohol-4-β-d-glucoside
(Matsubara and Sawabe, 1994)
120
Citrusin A
(Matsubara and Sawabe, 1994)
121
Citrusin B
(Matsubara and Sawabe, 1994)
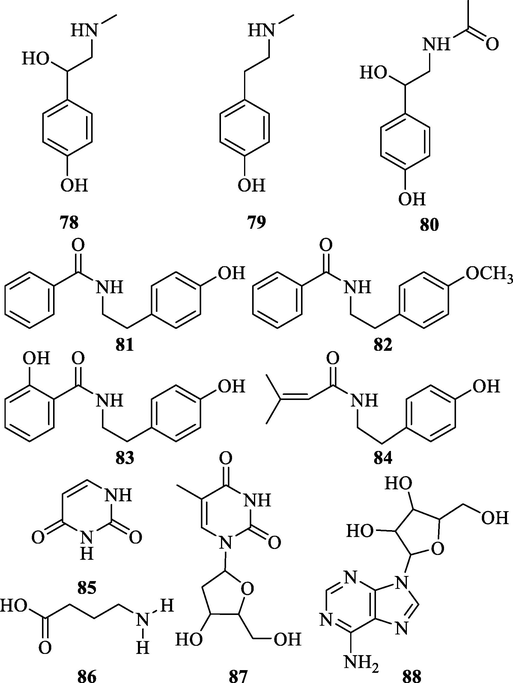
Chemical structures of alkaloids in ZS.
3.3.4 Limonoids
Limonoids are a group of highly oxygenated tetracyclic triterpenoids, which are also present in the form of aglycones and glycosides. Deepak used water-based polystyrene adsorption resin to extract and purify deacetyl nomilin (89), limonin (90), and deacetyl nomilin acid glucoside (95) (Dandekar et al., 2008). In screening constituents of improving gastrointestinal motility from the 70 % ethanol–water extract, isoobacunoicacid (91) was obtained (Zhang et al., 2019). Jayaprakasha isolated isolimonoic acid (94) and ichanexic acid (96) from ZS (Jayaprakasha et al., 2008). In addition, Zhao and Bai used UHPLC-Q-TOF-MS to determine nomilin (92) and obacunone (93) (Zhao et al., 2017; Bai et al., 2018) (Table 8 and Fig. 9).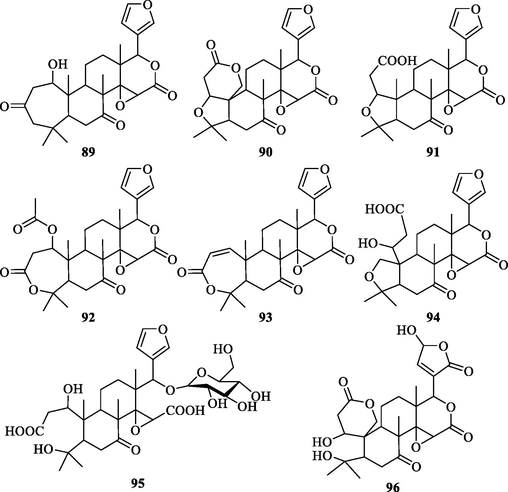
Chemical structures of limonoids in ZS.
3.3.5 Other phenolic compounds
Other phenolic compounds of ZS have also been reported (Table 8 and Fig. 10). Phlorin (1 0 1) was first obtained from ZS in 2005 (Zhang et al., 2005). The new compound (1 0 4) was isolated later in 2006 (Zhang et al., 2006). Two other new compounds (103 and 105) were isolated 11 years later (Zhang et al., 2017). Zhang obtained a new phenolic compound (1 0 6) and four known compounds (99–100 and 103–104) (Zhang et al., 2019). Deng used silica gel column chromatography, HW-40F gel column chromatography, ODS reversed-phase column chromatography, and preparative HPLC to separate phenolic compounds from ZS, leading to the isolation of a new compound (1 0 2) and two firstly reported compounds (97 and 98) in ZS (Deng et al., 2020).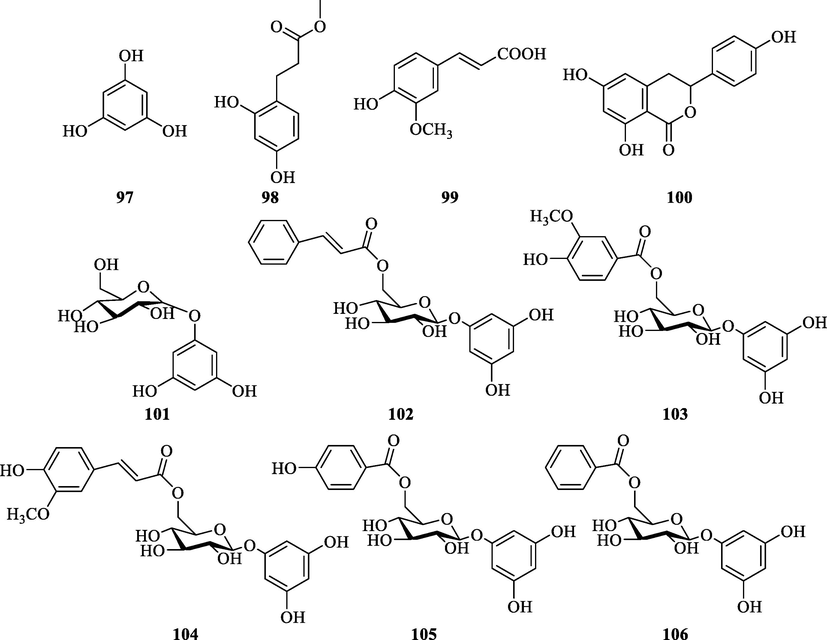
Chemical structures of phenols in ZS.
3.3.6 Other compounds
Phenyl compounds and phenyl glycosides were also present in ZS (Table 8 and Fig. 11). Besides, ZS is rich in the nutrients necessary for human life. For example, 4 cyclic peptides (Matsubara et al., 1991), 5 amino acids (Deng et al., 2018), and 4 polysaccharides were obtained from ZS (Wang et al., 2014).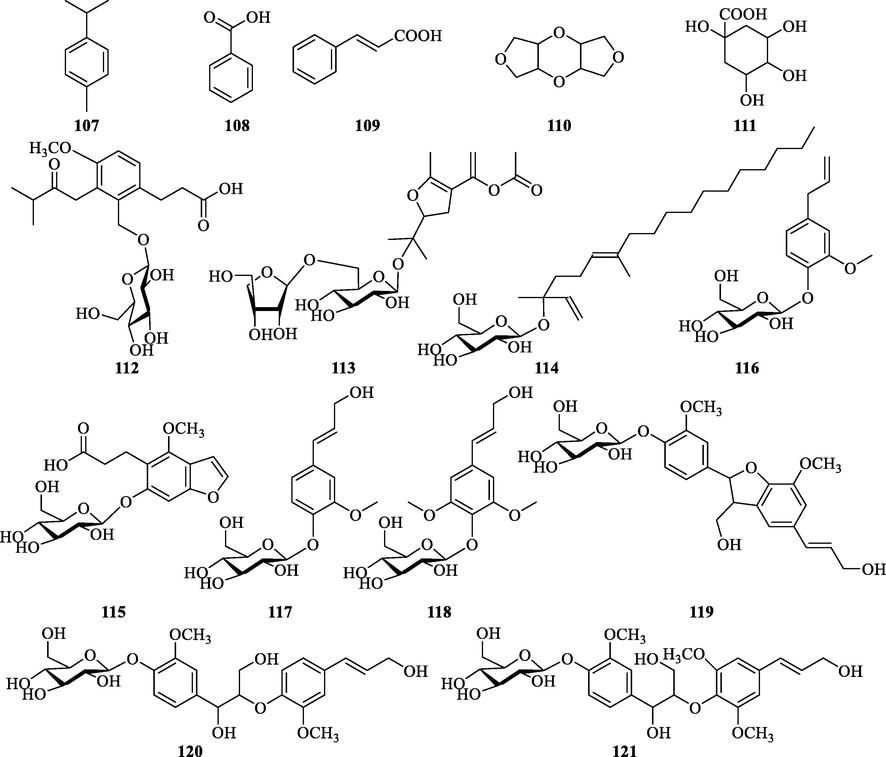
Chemical structures of other compounds isolated from ZS.
3.4 Quality control
Quality control of TCMs has received much attention in recent years. Thin-layer chromatography (TLC) and HPLC are the main techniques used for quality control in pharmacopeias in China and around the world. According to ChP (Chinese Pharmacopoeia Commission, 2020b) and Taiwan Traditional Chinese Medicine Dictionary (the third edition), synephrine (78) was used as an indicator component, whose content in ZS should be no<0.3 %. The fourth issue of Hong Kong Chinese Materia Medica discriminated SCZS from TCZS based on their appearance, micrograph, and HPLC chromatogram. Besides, hesperidin (49) and naringin (53) were used for the quality control of ZS, and the contents of naringin (53) and synephrine (78) should be no<0.66 % and 0.3 %, respectively. In 2021, The Japanese Pharmacopoeia (the 18th edition) prescribed a color reaction method for the identification of ZS. According to Food and Drug Administration (FDA), the content of synephrine (78) in dried fruit of C. aurantium ranged from 0.012 % to 0.25 % (Correll, 2015).
Different countries or regions have various requirements for quality control standards of herbal medicines. Determination of multi-components and application of multi-instruments are the trends of the times for quality control. The literature data for quality control from 1984 to 2022 were summarized. As shown in Fig. 12, synephrine (78), the most representative constituent in ZS, was investigated as a single-marker for quality control of ZS in 22 papers from 1994 to 2022. The results also suggested naringin (53), hesperidin (49), neohesperidin (56), and synephrine (78) were the most investigated compounds in TLC, HPLC, and other technical research.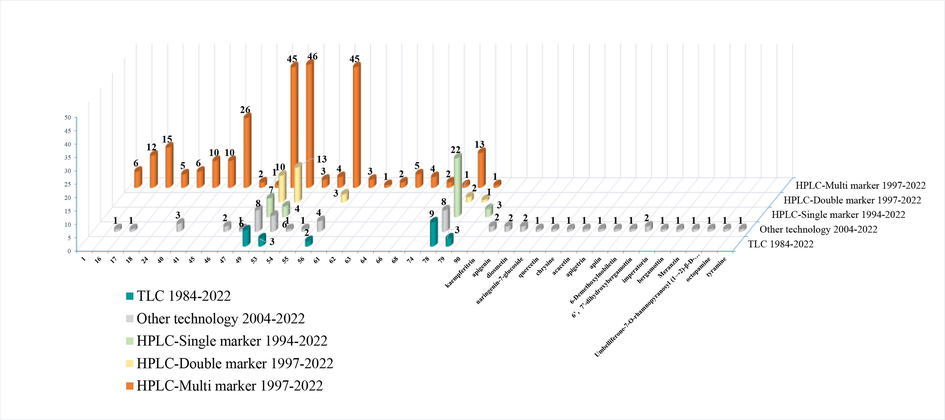
The hotspot compounds in TLC, HPLC, and other technical research for quality control of ZS.
3.4.1 TLC
TLC is a major force in the quality control of TCM, which is more suitable for the detection of the components without UV absorption compared with HPLC (Deng et al., 2019). Yuan compared the TLC methods of ZS, ZQ, Qingpi (QP, the young fruit or peel of Citrus reticulata Blanco), and Chengpi (CP, the mature peel of Citrus reticulata Blanco) in various pharmacopeias and literature and found the method of ZQ in ChP (Chinese Pharmacopoeia Commission, 2020b) is the best one, which allowed discrimination of the four medicines (Yuan et al., 2021). In addition, the contents of total flavonoid (Wang and Luo, 1989) and alkaloid (Wang et al., 1994) were determined by TLC-Scanning. Bagatela established a more robust high-performance TLC (HPTLC) method coupled with desorption electrospray ionization mass spectrometry (DESI-MS) for the analysis of more compounds (Bagatela et al., 2015). Tao described a new approach that applies artificial neural networks (ANN) to raw HPTLC images for the discrimination of ZS, ZQ, QP, and CP (Li et al., 2019).
3.4.2 HPLC/UHPLC
The contents of alkaloids and flavonoids were different among ZS and its adulterants, and no alkaloids was detected in Honghecheng using HPLC method (Chen et al., 2022; Wu et al., 2022; Zeng et al., 1997; Zhu and Pan, 2005). Naringin (53) and neohesperidin (56) were not detected in Zhizhu pills containing TCZS, but they did in those containing SCZS (Song et al., 2016). Shi compared the level of limonin (90) of ZS in different origins, and those from Hunan were the highest in the content (Shi and Liu, 2011). Chuang established a HPLC method coupled with an algorithm to discriminate ZS, ZQ, Gouju (fruit of P. trifoliata), and Xiangyuan (fruit of Citrus wilsonii Tanaka) (Chuang et al., 2007). Furthermore, solid-phase extraction column coated with Strata-X (Zeng et al., 2016) and rapid resolution LC (Wang et al., 2009) were also used for the determination of phenolic compounds and flavonoids. Mid-June proved to be the best harvest time of ZS by UHPLC analysis because the content of total chemical compounds was highest at that time (Deng et al., 2017). Additionally, the chemical differences between peel and pulp of ZS were discovered by UHPLC (Shi et al., 2021).
Fingerprints can provide a comprehensive description and evaluation of the quality of TCMs. The fingerprints of the SCZS and TCZS were established separately, and 17 peaks were identified by comparing retention time and UV spectroscopy with the standard. The mark compounds, naringin (53) and neohesperidin (56), were discovered based on their variable important value, which did not exist in TCZS, and thus can be used for discrimination of these two herbal materials (Zeng et al., 2016). Synephrine (78) was used for fingerprint analysis for the first time in 2011, and this method was then recorded in the Hong Kong Chinese Materia Medica in 2012 (Zhang et al., 2011). However, naringin (53) was detected in TCZS by this method, which was inconsistent with the results of most reported studies (Lu et al., 2022). It may be caused by inaccurate identification of the plant origins of the samples. Gao established a method for simultaneous determination of nine components by using quantitative analysis of multi-components by single-marker (QAMS) (Gao et al., 2020). Qi used chemical pattern recognition technology such as similarity evaluation, cluster analysis (CA), principal component analysis (PCA), and orthogonal partial least square discriminant analysis (OPLS-DA) for the data analysis and quality evaluation of ZS (Qi et al., 2021). Notably, the fingerprint of standard decoction of ZS should be studied separately due to the chemical differences between SCZS and TCZS (Shi et al., 2019).
The content of naringin (53) heightened along with the increase of the diameter of ZS slices (Huang et al., 2008; Lin et al., 2022). With the prolonging of storage time, the content of naringin (53) was increased, while the content of neohesperidin (56) and synephrine (78) decreased (Zhou and Gui, 1997; Xu et al., 2020). In addition, the level of synephrine (78) and flavonoids varied among the different processed products of ZS. For example, the vinegar-fried ZS was the highest in its content, and the alcohol-fried one was the lowest (Cai et al., 2022; Ouyang, 2005). HPLC analyses of the formula granule and decoction of ZS suggested they showed high similarity in chemistry (Zhang et al., 2007). Granules from different manufacturers showed significant variations according to UHPLC analysis results (Chen et al., 2010).
3.4.3 Lc-MS/GC–MS
Currently, high-resolution quadrupole time-of-flight tandem mass spectrometry (QTOF-MS/MS) becomes a powerful technology for the identification and determination of herbal materials chemical constituents. In 2020, a total of 295 metabolites were screened using this method, and 89 phytochemicals were identified in the flowers, fruits, roots, leaves, and branches of C. aurantium ‘Daidai’. Among them, sixty-nine were reported for the first time (Yu et al., 2020). Wu developed a simultaneous qualitative and quantitative analysis method for 10 bioactive flavonoids in ZS (Wu et al, 2022). There were 19 different components between raw material and bran-fried product of ZS, which provided a deep insight into the processing mechanism of ZS (Peng et al., 2020). QTOF-MS/MS coupled with genetic algorithm optimized support vector machines (GA-SVM) was successfully employed to discriminate of the fruit of seven Citrus herbs (Duan et al., 2014). Tong developed an online extraction strategy, which eliminated sample pretreatment steps and increased extraction efficiency (Tong et al., 2018). In addition, an UHPLC coupled with linear ion trap-Orbitrap tandem mass spectrometry (UHPLC-LTQ-Orbitrap-MS/MS) method was developed to detect the chemical composition of ZS, and a total of 27 compounds were detected, including 14 flavonoids, 7 coumarins, 5 limonoids, and 1 alkaloid (Yu et al., 2016).
GC–MS was applied in the identification of Citrus genus (Dong et al. 2022). The volatile composition of ZS from China was different from those from Korea (the fruit of P. trifoliate and C. aurantium) according to GC–MS analysis results (Liu et al., 2003). Twenty-nine and thirty-eight constitutes were identified from SCZS and TCZS, respectively. Among them, twenty-four are common in these two herbal materials (Liu et al., 2011). He established HS-GC-IMS-fingerprint for the identification of SCZS and TCZS (He et al., 2022). Sixty-five compounds, including 57 different from SCZS, were identified from Xiangyuan (Yang et al., 2010). Differences in harvesting periods resulted in variations in the levels of volatile components of ZS, and limonene content increased along with fruit growth (Deng et al., 2019). Compared with raw material, bran, honey and bran, or honey and chaff fried ZS caused the generation of 52, 26, and 28 derivatives, respectively (Yu et al., 2015). In addition, Deng established a method for the determination of limonene, gamma terpene, linalool, and 4-terpineol of ZS (Deng et al., 2020).
3.4.4 DNA molecular techniques.
DNA molecular marker and DNA barcode were used for species identification and genetic characteristics research based on PCR amplification technology. The origin and evolution of Citrus genus were studied by analyzing their whole-genome sequences. Both C. aurantium and C. sinensis come from the hybridization of pomelo and mandarin (Wu et al., 2014; Wu et al., 2018). The differences of genetic relationship among C. aurantium, C. sinensis, and other cultivars were revealed by Simple Sequence Repeats (SSR), Inter-SSR (ISSR), Sequence-Related Amplified Polymorphism (SRAP), and Random Amplified Polymorphism DNA (RAPD) technologies (Goh et al., 2022; Liu et al., 2005; Luo et al., 2007; Polat et al., 2012; Sun et al., 2012; Zuo et al., 2005). The trnH-psbA and ITS2 were screened out as promising DNA barcode for the identification of Citrus genus plants (Luo et al., 2010; Mahadani and Ghosh, 2014). In addition, the laser-induced fluorescence spectroscopy technology was also used for the identification of C. sinensis (Kubota et al., 2017; Magalhães et al., 2021).
3.4.5 Other methods
Other technologies, such as flow-injection with ultraviolet spectroscopic (UV) detection and proton nuclear magnetic resonance (Zhang et al., 2016), flow-injection mass spectrometric metabolic fingerprinting (Zhao et al., 2015), paper spray-mass spectrometry (Liu et al., 2017), and electronic tongue technology (Wu et al., 2012), were used to differentiate ZS and its adulteration. Headspace solid-phase microextraction coupled with electronic nose based on mass spectrometry was developed for the discrimination of geographical origins (Italy, South Africa, and Spain) of the fruit of C. sinensis (Centonze et al., 2019). Zhang used the mass constant method to evaluate the quality of ZS (Zhang et al., 2019).
Infrared spectroscopy (IR) could distinguish the different origins of ZS (Song et al., 2009; Huyi et al., 2013), and determine the content of synephrine (Lei et al., 2015). Total alkaloids, volatile oils, and flavonoids were determined by UV (Tang, 2008; Xu et al., 2016). Green chromatography using aqueous solutions of room temperature ionic liquids (Tang et al., 2006) and packed-column supercritical fluid chromatography could determine the content of alkaloids (Lu et al., 2006). Capillary electrophoresis with electrochemical detection and capillary GC were used to determine the active ingredient and organochlorine pesticide residues, respectively (Hui and Wang, 2004; Peng and Ye, 2007). Zhao developed a sensitive and practical indirect competitive enzyme-linked immunosorbent assay based on anti-monoclonal antibodies to determine the content of naringenin (40), the critical biological active compound of ZS (Zhao et al., 2021).
3.5 Biological activities
ZS exhibits anti-depressant and anti-adipogenic activities, which are closely related to the treatment of digestive and cardiovascular systems diseases. Besides, ZS show significant anti-inflammatory and antioxidant activities for being rich in polyphenols.
3.5.1 Effect on the digestive system
3.5.1.1 Gastrointestinal motility regulating
ZS and its ingredients play an important role in exciting gastrointestinal smooth muscle and promoting gastrointestinal movement. Currently, the research on the promotion of gastrointestinal motility of ZS mainly focuses on slow transit constipation (STC), migrating myoelectric complex (MMC), and functional dyspepsia (FD) (Table 9 and Fig. 13). SP: substance P; VIP: vasoactive intestinal peptide; CCK: content of cholecystokinin; SS: somatostatin; A/T: the active phase to the circle; GAS: gastrin; MTL: motilin; Ach: acetylcholine; SCF: stem cell factor; IL: interleukin; c-Kit: gastric sinus tissue; GRP78: glucose regulatory protein; ATF6: activating transcription factor; XIAP: X-linked apoptosis inhibitory protein; PCNA: proliferating cell nuclear antigen.
Bioactivity
Mechanism
Method type
Extracts/compounds
Active concentration/dose
p value
References
STC
Accelerate the frequency of extracorporeal colonic muscle strips
Vitro (Wister rat)
The medicinal serum of ZS
p < 0.05
(Liu et al., 2010)
Decrease the amplitude of colon slow waves and coefficient variation while increase the frequency
Vivo ((SPF) SD rat)
ZS volatile oil
2.7 g/kg for 30 d
p < 0.05 vs model control
(He et al., 2013)
Increase impelling rate of small intestine, shorten the time and add the number of dark stools
Vivo (ICR mice)
ZS decoction
1 g/kg and 2 g/kg
n = 10, p < 0.01 or 0.05 vs model control
(Zhang and He, 2010)
Increase SP and VIP in intestinal tissues
Vivo (Wister rat)
ZS decoction
4 and 8 g/kg/d, i.g for 3 months
n = 10, p < 0.01 or 0.05 vs model control
(Tao, 2011)
Improve the intestinal motility
Vivo (Male SD rat)
ZS decoction
4 g/kg/d, i.g for 2 weeks
n = 14, p < 0.01 or 0.05 vs model control
(Wang et al., 2015)
MMC
Affect time course and peak potentials of phase I–II
Vivo (Dog)
Lemon Terpene
98 %, 0.1 mL/kg, i.g
p < 0.01
(Yang et al., 1995)
Vivo (Female dog)
ZS decoction
100 %, 1 mL/kg, i.g
n = 14, p < 0.01
(Bi et al., 1991)
Short the time duration of phase II and increase slow-wave load peaks, and intensity of peaks in phase III
Vivo (Female sheep)
ZS decoction
25 g, p.o
n = 2, p < 0.01, p < 0.05, respectively
(Kuang, 1997)
Stimulate the electro-reactivity of small intestines by improving CCK and reducing SS in hypothamus
Vivo (SD rat)
ZS decoction
100 %, 8 mL/kg, i.g for 7 d
p < 0.01 vs control
(Song et al., 2006)
Increase the ratio of A/T and SP in the myenteric nerve plexu
Vivo (SD rat)
ZS decoction
1 g/mL, 8 mL/kg, i.g for 5 d
p < 0.05
(Wang et al., 2001)
H1-receptor
Vivo (KM mice)
Carbon-ZS decoction
0.1 g/mL, 0.3 mL, i.g
n = 12, p < 0.01 vs model control
(Liu et al., 2001)
Enhance the electrical activity in the small intestine
Vivo (Dog)
ZS decoction vs atropine
100 %, 1 mL/kg vs 0.5 mg
n = 13, p < 0.01 vs control
(Huang et al., 1996)
Vivo (Dog)
ZS decoction vs propranolol
100 %, 1 mL/kg vs 0.5 mg/mL
(Yang et al., 1996)
Promote intestinal transit
Vivo (SPF mice)
Wall-broken spore power of ZS
1.5 g/kg/d, 2 mL/10 g/each, i.g for 7 d
n = 10, p < 0.05
(Lin et al., 2012)
Promote antral smooth muscle cell contraction and improve the intracellular Ca2+ fluorescence intensity
Vitro ((SPF) SD rat)
The medicinal serum of ZS
10 %, 20 %, 50 %
p < 0.05
(Li et al., 2015)
Promote secretion of gastric acid and gastrointestinal motility and decrease the GAS and MTL
Vivo (Wister rat)
Suspension prepared with water
0.11 g/mL/d, 2 mL/each, i.g for 4 d,
n = 8, p < 0.05
(Tang et al., 2015)
FD
Promote gastric emptying by increasing SP and MTL and reducing VIP
Vivo (SD rat)
Suspension prepared with water
0.11 g/mL, 2 mL/d, i.g for 14 d
n = 40, p < 0.05 vs control
(Zhang and Li, 2018)
Vivo (Wister rat)
ZS decoction
250 g/L, 10 mL/kg, i.g for 4 weeks
n = 10, p < 0.05 vs model control
(Zhu et al., 2005)
Improve the small bowel propulsion and gastric emptying and 49 increase MTL
Vivo (Wister rat)
Compounds 40, 49, 56
100 mg/kg/d, i.g for 2 weeks
p < 0.05 vs model control
(Huang et al., 2012)
Promote gastrointestinal motility of model rats with spleen deficiency by affecting GAS, Ach, MTL, SP and VIP
Vivo (SD rat)
ZS decoction
0.104, 0.208, 0.416 g/mL
n = 10, p < 0.05 or 0.01 vs model control, except 78 had no effect on the GAS
(Hu et al., 2017)
Compounds 53, 56, 78
53: 3.267, 6.535, 13.070 mg/mL
10 mL/kg/d, i.g for 1 week
56: 3.865, 7.730, 15.460
mg/mL
78: 0.252, 0.504, 1.008 mg/mL
Increase SCF and MTL by reducing IL-6 in serum
Vivo (SD rat)
ZS decoction
1.0 g/mL and 2.0 g/mL, 1.5 mL/100 g, 2 times/d, i.g for 4 weeks
n = 8, p < 0.05 vs model control
(Lin et al., 2020)
Promote the proliferation and differentiation of ICCs by starting SCF/c-Kit signaling pathways
Vivo (SD rat)
ZS decoction
0.5, 1.0 and 2.0 g/mL, 1.5 mL/100 g, 2 times/d, i.g for 4 weeks
n = 8, p < 0.05 vs domperidone group
(Deng et al., 2018)
Reduce the ERS injury of ICCs by decreasing expression of GRP78 and ATF6
Vitro ((SPF) SD rat)
The medicinal serum of ZS
20 %
p < 0.05 vs control
(Wang et al., 2018)
Promote ICC proliferation by increasing XIAP and PCNA protein
Vitro ((SPF) SD rat)
The medicinal serum of ZS
10 %
n = 6, p < 0.01 or 0.05
(Teng et al., 2020)
Improve the ramp phase and the tonic phase
Vivo (Male SD rat)
ZS flavonoid
10 and 15 mg/mL, 1 mL/100 g, 1 time/d, i.g for 4 weeks
n = 14, p < 0.05 vs model control
(Wu et al., 2016)
Improve gastric accommodation and decreased mRNA expression of 5-HT and c-fos
Vivo (Male (SPF) SD rat)
ZS flavonoid
100 mg/kg/d, i.g for 2 weeks
n = 8, p < 0.05 vs model control
(Li et al., 2016)
Increase SP and MTL and reduce VIP
Vivo (SD rat)
ZS decoction vs bran-fried ZS with bran decoction
1 g/mL/d, 0.7 mL/100 g, i.g for 6 d
n = 10, p < 0.05
(Lin et al., 2012)
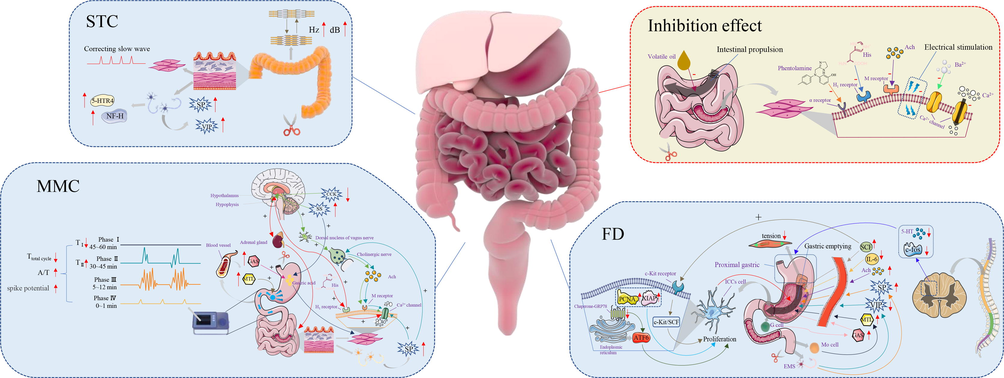
Possible mechanisms of bi-directional regulation effects on the gastrointestinal tract.
STC is a condition of prolonged bowel cycles and difficult defecation caused by dysfunction of the large intestine and abnormal conduction. The medicinal serum (Liu et al., 2010), volatile oil (He et al., 2013), and decoction of ZS (Zhang and He, 2010) improved STC by increasing the content of substance P and vasoactive intestinal peptide (VIP) (Tao, 2011), and promoting the expression of neurofilament-H and 5-HTR4 (Wang et al., 2015).
MMC was divided into four phases, including the stationary, intermittent contractions, strong contractions, and the transition phases. MMC symptoms of dogs and sheep (Kuang, 1997) were improved by affecting the period, frequency, and intensity of phases I–III after treatment with lemon terpene (Yang et al., 1995) and ZS decoction (Bi et al., 1991). ZS stimulated the electro-reactivity of small intestines by improving the content of cholecystokinin and substance P (Wang et al., 2001) and reducing the content of somatostatin (Song et al., 2006). The therapeutic effect of ZS may be related to M and H1 receptors (Liu et al., 2001), as well as intracellular Ca2+ fluorescence intensity (Li et al., 2015), but not to β receptors and H2 receptors (Huang et al., 1996; Yang et al., 1996).
FD is the most common gastrointestinal motility disorder in gastroenterology. The interstitial cells of Cajal (ICC) are pacemakers and regulators of gastrointestinal activity, which participate in the transmission of gastrointestinal neurotransmitters. Compounds 40, 49, 56, and 78 promoted gastric emptying by increasing the content of substance P, motilin, and stem cell factor (SCF) and reducing the content of VIP and interleukin (IL)-6 (Zhu et al., 2005; Huang et al., 2012; Hu et al., 2017; Zhang and Li, 2018; Lin et al., 2020). ZS promoted the proliferation of ICC by starting SCF/c-Kit (gastric sinus tissue) signaling pathways (Deng et al., 2018) and increasing the expression of X-linked apoptosis inhibitory protein and proliferating cell nuclear antigen (PCNA) protein (Teng et al., 2020). Besides, ZS protected against endoplasmic reticulum stress injury of ICCs (Wang et al., 2018). Flavonoid of ZS could improve reduced proximal gastric adaptability (Wu et al., 2016) and visceral hypersensitivity (Li et al., 2016). The absorption and the elimination processes of compounds 40 and 41 increased in model mice compared with normal mice (Xu et al., 2019).
However, ZS has proven to possess bi-directional regulation effects on the gastrointestinal tract. The extracts (Hu et al., 1992; Hu et al., 1994), pure compounds including 16–17, 22, 24, 28, 39–40, and 64 (Takase et al., 1994; He et al., 2018; Zhang et al., 2019), and decoction of ZS (Chen et al., 1981; Cho et al., 1996; Zhang et al., 2006) showed significant inhibitory effects on the contraction of gastrointestinal tract (Yang et al., 1998; Xie et al., 2001). The inhibitory effect of naringenin (40) could be completely antagonized by l-NAME and indomethacin (He et al., 2018), and those of ZS extract could be partly antagonized by phentolamine (Yang et al., 1998; Xie et al., 2001). However, the inhibition of ZS volatile oil could not be antagonized by phentolamine. In addition, it could also inhibit intestinal propulsion in vivo (Hu et al., 1992). Furthermore, the inhibitory effect of ZS was not related to N-receptor, β-receptor, prostaglandin, and NO (Yang et al., 1998; Xie et al., 2001). The smooth muscle relaxes after promoting Ca2+ outflux and inhibiting Ca2+ influx. ZS, similar to verapamil, could inhibit the contraction of smooth muscle by inhibiting Ca2+ influx (Huang et al., 1993; Sheng et al., 1994). Epoxyauraptene, isolated from bran-fried ZS (Lin et al., 2012), could inhibit the contractility of gastrointestinal smooth muscle by inhibiting the function of smooth muscle myosin (Xu et al., 2012). Besides, ZS flavonoid have a therapeutic effect on irritable bowel syndrome (IBS) (Liang et al., 2014) Detailed information and possible mechanisms of bi-directional regulation effects of ZS on the gastrointestinal tract are shown in Table 10 and Fig. 13. His: histamine; AIM: Methanol extracts; AIW: water-fractions; AIC: chloroform-fractions; NO: nitric oxide; MC: mast cells.
Bioactivity
Mechanism
Method type
Extracts/compounds
Active concentration/dose
p value
References
Gastrointestinal motility inhibiting
Relax isolated intestinal smooth muscle
Vitro (Rabbit)
ZS injection
2 × 10-3–4 × 10-3
(Chen et al., 1981)
Decrease the frequency and amplitude of jejunal contraction
Vitro (Wister rat)
Volatile oils
3 μL/mL, 10 μL/mL
(Hu et al., 1992)
Inhibit the contraction of isolated intestinal smooth muscle
Vitro (Wister rat)
ZS flavonoid
20 mg/mL
(Hu et al., 1994)
Counteract the spastic contraction induced by BaCl2, Ach and His in ileum
Vitro (guinea pig)
ZS flavonoid
20 mg/mL
n = 4
Inhibit the isometric contraction of ileum smooth muscle
Vivo (SD rat)
The water extract of ZS
IC50 = 11.8 × 10-2 g/L
(Cho et al., 1996)
Inhibit contraction tension of isolated intestine tissue
Vitro (Male KM mice)
Compounds 16, 17, 22, 24, 28, 39, 64
100 μM
n = 6, p < 0.05 vs control
(Zhang et al., 2019)
Inhibit gastric motor activity
Vivo (Male Wistar rats)
Compound 17
10–50 mg/kg
ED50 = 17.2 mg/kg
n = 4, p < 0.05 vs control
(Takase et al., 1994)
Compound 64
ED50 = 8.0 mg/kg
Exhibit relaxations of contractions in isolated ileum
Vitro (Male guinea pigs)
Compound 17
Ach: IC50 = 3.73 × 10-4 g/mL
transmural electrical stimulation: IC50 = 4.05 × 10-4 g/mL
His: IC50 = 1.48 × 10-4 g/mL
Compound 64
Ach: IC50 = 2.56 × 10-4 g/mL
transmural electrical stimulation: IC50 = 8.12 × 10-4 g/mL
His: IC50 = 3.73 × 10-4 g/mL
Inhibit the active tension on spontaneous contractions of intestine smooth muscle
Vivo (Male SD rat)
Raw medicinal materials
125–500 mg/kg, p.o
n = 8, p < 0.001, p < 0.01 vs control
(He et al., 2018)
Decrease contractile amplitude and frequency of isolated colonic muscle strips and small intestinal longitudinal strips
Vitro (Wister rat)
ZS decoction
10-3 g/mL
n = 12, p < 0.001 vs control
(Xie et al., 2001)
Vitro (Wister rat)
ZS decoction
0.1 g/mL
n = 12, p < 0.001 vs control
(Yang et al., 1998)
Reduce contraction amplitude, frequency and tone in isolated intestinal smooth muscle
Vitro (Rabbit)
ZS decoction
1.2 mg/mL
n = 6, p < 0.01 vs control and model control
(Zhang et al., 2006)
Antagonize the contraction of Ach and His on the smooth muscle of isolated small intestine
Vitro (KM mice)
ZS decoction
Ach group: 0.1 and 0.2 g/mL
Ach: n = 4, p < 0.01
vs control
(Huang et al., 1993)
His group: 0.1 g/mL
His: n = 4, p < 0.05
Shift the dose–response curve of BaCl2 in parallel to the right
BaCl2 group: 0.1 g/mL
BaCl2: n = 5, p < 0.05
Inhibit the isolated intestine contraction induced by Ca2+ after high-K+ depolarization
Vitro (KM mice)
ZS decoction
0.1 g/mL
n = 4
(Sheng et al., 1994)
Relax the smooth muscle, relieve spasm and inhibit peristalsis of intestinal by increasing NO in the serum and decreasing MC of colon and ileum
Vivo (SD rat)
ZS flavonoid
3 g/kg, 1 time/d for 7 d
n = 8, p < 0.05 vs model control
(Liang et al., 2014)
Reduce the resting tension of isolated gastric smooth muscle
Vitro (Male SD rat)
ZS flavonoid
6000–10000 μg/mL
n = 7, p < 0.05 vs control
(Wu et al., 2016)
Direct experimental evidence
Stimulate and inhibit the contractility of isolated jejunal segments in all 6 different low/high contractile states
Vitro (SD rat)
Compound 17
2.5 μM
(Xiong et al., 2014)
Stimulate and inhibit jejunal contractility
Vitro (SD rat)
Compound 17
EC50 = 4.0 μM
IC50 = 30.0 μM
Relax in the fundic circular muscle pre-contracted by U46619
Vitro (Male SD rat)
AIM, AIW and AIC
AIM = 100 μg/mL, AIW = 100 μg/mL, AIC = 10 μg/mL
n = 4–6
(Kim, 2013)
Relax both fundic circular and longitudinal smooth muscle
Compound 41
41 = 10-4 M
Diphasic effects in the longitudinal smooth muscle contracted by U46619
Vitro (Male SD rat)
AIM, AIW
AIM = 100 μg/mL, AIW = 100 μg/mL
n = 4–6, p < 0.05 vs another group
Diastole in isolated pyloric circular smooth muscle strip in different Ca2+ environment
Vitro (Male SD rat)
ZS flavonoid
3000–10000 μg/mL
n = 10, p < 0.01 or 0.05 vs control
(Wu et al., 2016)
Increase the contraction of isolated intestinal strips, decrease after 1–1.5 min
Vitro (Wister rat)
Volatile oil
10 μL/mL
(Hu et al., 1994)
ZS flavonoid
20 mg/mL
The high doge of aroma significantly reduced small intestinal propulsion and increased gastric residual rate, while the high doge of bitter and low doge of sour all promote.
Vivo (KM mice)
Aroma components of ZS
0.06–0.3 g/kg
1 time/d for 3d
n = 10, p < 0.01 or 0.05 vs control
(Chen et al., 2019)
bitter components of ZS
0.82–4.1 g/kg
sour components of ZS
0.25–1.25 g/kg
Aroma on jejunum and ileum, bitter on duodenum, sour on ileum is bi-directionally regulated effect
Vitro (KM mice)
Aroma components of ZS
2.5 × 10-4–4 × 10-3 g/mL
n = 8
bitter components of ZS
4.015 × 10-3–6.425 × 10-2 g/mL
sour components of ZS
3.125 × 10-3–5 × 10-2 g/mL
It is found that promoting and inhibiting effects of ZS on the gastrointestinal tract were mainly investigated in vivo and in vitro, respectively. There was also direct experimental evidence to verify the bi-directional regulation effect of ZS (Table 10) (Hu et al., 1994; Kim, 2013; Xiong et al., 2014; Chen et al., 2019). In addition, when in combination with Baizhu (rhizome of Atractylodes macrocephala Koidz.), ZS showed the inhibiting effect while Baizhu used separately had an enhancing effect on the contractile activity of isolated gastric muscular strips in vitro (Zheng et al., 1998).
3.5.1.2 Anti-gastric ulcer effect
The hot water extract of ZS significantly inhibited gastric ulcers and hemorrhagic lesions (Takase et al., 1994) caused by ethanol and aspirin. Interestedly, marmin (64) and nobiletin (17) showed a stronger anti-gastric ulcer effect than aluminum thioglycolate (Hiroyuki, 1997). Volatile oils of ZS could prevent the formation of pyloric ligation ulcers and reduce the secretion of gastric juice and pepsin activity (Hu et al., 1992). Epoxyauraptene was decreased injury index on ulcerative colitis. Flavonoids of ZS reduced the expression of PCNA and C-erbB-2 protein to protect gastric mucosa (Zhang et al., 2020). ZS had a protective effect on gastric mucosa injury after cerebral infarction (Tang et al., 2014) by regulating the expression of the motilin and VIP (An et al., 2015) (Table 11). APAP: Acetaminophen; PUMA: p53 up-regulated apoptosis regulator.
Bioactivity
Mechanism
Method type
Extracts/compounds
Active concentration/dose
p value
References
Anti-gastric ulcer
Caused by ethanol and aspirin
Vivo
Hot water extract of ZS
250 mg/kg, p.o
ethanol: 69.7–69.9 %
(Hiroyuki, 1997)
aspirin: 37.3–46.8 %
Vivo
Compound 64
25 mg/kg, p.o
67.6 %
Compound 17
70 %
Inhibit ethanol-induced gastric hemorrhagic lesions
Vivo (Male wister rat)
Compound 64
10–50 mg/kg, p.o
ED50 = 17.2 mg/kg
(Takase et al., 1994)
Compound 17
ED50 = 8.0 mg/kg
Prevent the formation of pyloric ligation ulcers, and reduce the secretion of gastric juice and pepsin activity
Vivo (Wister rat)
Volatile oil
20 %, 1.0 mL/100 g, i.p for 48 h
n = 8, p < 0.01 vs control
(Hu et al., 1992)
Protect gastric mucosa by reduced the expression of PCNA and C-erbB-2
Vivo (Male (SPF) SD rat)
ZS flavonoid
150 mg/kg/d, i.g for 14 d
n = 20, p < 0.05 vs model control
(Zhang et al., 2020)
Protect gastric mucosa injury appeared after cerebral infarction by regulated the expression of the MTL and VIP
Vivo (Male wister rat)
Suspension prepared with water
2 mL/d, i.g for 4 d
n = 8, p < 0.01 vs model control or control
(Tang et al., 2014; An et al., 2015)
Decrease injury index on ulcerative colitis.
Vivo (BALB/C mice)
Compound epoxyauraptene
160 and 320 μmol/L
n = 10, p < 0.05 vs model control
(Lin et al., 2015)
Hepatoprotection
Improve liver cell damage, reduce cell edema, and degeneration of vacuoles
Vivo (KM mice)
70 % ethanol ZS extract
9.59 g/kg/d, i.g for 4 weeks
p < 0.01 or 0.05 vs model control
(Jiao et al., 2007)
Against APAP-induced liver necrosis by inhibited the PUMA and reversing disorder of liver lipid metabolism
Vivo (Male SD rat)
Ethanol ZS extract
6 g/kg/d, i.g for 7 d
n = 10, p < 0.01 or 0.001 vs model control
(Shu et al., 2020)
Protect the restoration of intestinal microbiota composition, reshaped barrier integrity
Vivo (Male C57BL6 mice)
Ethanol extract
8.7 g/kg/d, p.o for 4 weeks
n = 10, p < 0.05 or 0.01 vs model control
(Liu et al., 2020)
Anti-depression
Reduce the number of spontaneous activities
Vivo (Wister mice)
Volatile oil
(10 %, 20 %), 10 mL/kg/d, p.o for 3 d
n = 12, p < 0.05 vs control
(Hu et al., 1994)
Increase the latency period of tonic seizures, increase the sleeping time and the time spent in the open arms of the elevated plus maze
Vivo (Male Swiss mice)
Essential oil
0.5 or 1.0 g/kg, p.o
n = 9, p < 0.05 vs model control
(Carvalho-Freitas and Costa, 2002)
Vivo
Hexanic and dichloromethanic fractions from 70 % hydroethanolic extract
1.0 g/kg, p.o
n = 9, p < 0.05 vs model control
Light-dark box and the marble-burying test mediated by 5-HT1A receptors
Vivo (Male Swiss mice)
Essential oil
0.5 or 1.0 g/kg, p.o 30 min (single dose) or once a day for 15 d (repeated doses)
Light-dark box
Single: n = 8
p < 0.05 vs control.
(Pultrini et al., 2005)
Repeated: n = 7, 5
Marble-burying test
Single: n = 6, 9
p < 0.05 vs control
Repeated: n = 8, 7
Light/dark box procedure
Vivo (Male Swiss mice)
Essential oil
acute (5 mg/kg) or repeated 1 mg/kg/d for 14 d
p < 0.01 or 0.05 vs model control
(Costa et al., 2013)
Restores reward circuitry
Vivo (Male Wister rat)
Compound 61
10 mg/kg/d, i.g for 1 week
n = 6, p < 0.001, 0.01 or 0.05 vs model control
(Liu et al., 2021)
Increase in open box horizontal movement
Vivo ((SPF) SD rat)
ZS flavonoid
3000 mg/kg/d i.g for 1 week
n = 8, p < 0.01 vs control, p < 0.05 vs model control
(Liang et al., 2014)
3.5.1.3 Hepatoprotection
Ethanol extract of ZS could improve liver cell damage and reduce cell edema and cell vacuoles of diabetic rats (Jiao et al., 2007). ZS protected against acetaminophen-induced liver necrosis by inhibiting the p53 up-regulated apoptosis regulator and reversing disorder of liver lipid metabolism (Shu et al., 2020). Ethanol extract of ZS possessed a protective effect on the restoration of intestinal microbiota composition and reshaped barrier integrity of liver-gut axis, which prevented the translocation of microbiota endotoxin product from the intestine to the liver (Liu et al., 2020). Moreover, the ethanol extract of ZS combined with methotrexate which was the disease-modifying agent for the treatment of rheumatoid arthritis significantly ameliorated methotrexate‐induced chronic hepatic injury (He et al., 2018). Si Ni San, a prescription containing ZS, could significantly reduce the hepatocyte damage induced by CCl4 and 2–4-6-trinitrochlorobenzene (Jiang and Xu, 2004) and promote hepatic stem cell differentiation via Wnt/β-catenin signaling pathway (Xu et al., 2022) (Table 11).
3.5.2 Anti-depression activity
The severe depressive disorder has a close and bidirectional association with FD (Koloski et al., 2014). Consequently, ZS has become the research hotspot because it can promote gastrointestinal motility while exerting anti-depressant effects. Volatile oil (Hu et al., 1994) or essential oil from the peel of ZS (Carvalho-Freitas and Costa, 2002) was able to treat depression and could increase the latency period of convulsing mice (Pultrini et al., 2005). However, the essential oil was not active in the forced swim test (Costa et al., 2013). Meranzin hydrate (61) elicited antidepressant effects and restored reward circuitry (Liu et al., 2021). ZS flavonoid increased the rearing score of IBS rats in open box horizontal movement (Liang et al., 2014) (Table 11).
In a clinical trial of cocaine withdrawal, essential oil from the peel of ZS provided an acute anxiolytic effect on the patients exposed to simulated public speaking (n = 17) (Neto et al., 2017). Si Ni San was also used to treat depression in clinical practice (Cong et al., 2021).
3.5.3 Effect on the cardiovascular system
3.5.3.1 Blood pressure elevating
The blood pressure of rats or dogs was significantly increased after intravenous injection of fluid extract (Yan, 1995), alkaloid aqueous solution (Hu et al., 1994), or injection (Chen et al., 1981) of ZS. Synephrine (78) and N-methyltyramine (79) with the blood pressure regulating effect were obtained by bio-guided isolation, and N-methyltyramine (79) significantly increased coronary and renal blood flow and decreased vascular resistance and myocardial oxygen consumption. The combination of compounds 79 and 78 (3:1, v/v) could resist the decrease of blood pressure caused by reserpine. Synephrine (78) affected adrenergic receptors directly, while N-methyltyramine (79) worked mainly through the indirect mechanism of catecholamine release (Guo, 1978). The blood pressure regulating effect of SCZS was severer than TCZS, probably due to the chemical variations (Cui et al., 2010). Furthermore, the raw material of ZS showed a stronger effect on raising blood pressure than its processed products (Yi et al., 2011) (Table 12). ICa-L: l-type calcium; dp/dt: first derivative of the left ventricular pressure; GPIP: common peak isovolumetric pressure; ICAM-1: intercellular adhesion molecule-1; HUVEC: human umbilical vein endothelial cells line; ox-LDL: oxidized low-density lipoprotein; ET-1: endothelin; TG: triglycerides; TCH: total cholesterol; ED: endothelial dysfunction; PLT: platelet; RBC: red blood cell; CIA: collagen‐induced arthritis; Hcy: homocysteine.
Bioactivity
Mechanism
Method type
Extracts/compounds
Active concentration/dose
p value
References
Blood pressure elevating
Elevate blood pressure
Vivo (Rat)
ZS injection
0.5 mL, iv drop
(Chen et al., 1981)
Vivo (Wister rat)
Alkaloid aqueous solution
5 and 10 g/kg
n = 5, p < 0.01
(Hu et al., 1994)
Vivo (Male SD (Ⅱ) rat)
70 % ethanol SCZS extract
2.1 and 4.2 g/kg, 0.84 mL/100 g duodenal administration
n = 10, p < 0.05 and 0.01 vs control
(Cui et al., 2010)
Vivo
70 % ethanol TCZS extract
2.1 and 4.2 g/kg, 0.84 mL/100 g duodenal administration
n = 10, p < 0.05 and 0.01 vs SCZS group
Vivo (Rat)
ZS decoction
2 mg/kg/d, i.g for 5 weeks
n = 10, p < 0.05 vs control
(Yi et al., 2011)
Vivo
Fried-ZS with bran decoction
2 mg/kg/d, i.g for 5 weeks
n = 10, p < 0.05 vs control
Vivo (Dog)
ZS injection
3 mL, iv drop
(Chen et al., 1981)
Vivo (Dog)
ZS fliud extract
100 %, 3 mL, iv drop
n = 6, p < 0.05
(Yan, 1995)
Increase coronary flow
Vivo (Dog)
Compound 79
0.1 mg/kg/min, iv drop
0.2 mg/kg, p < 0.05
(Guo, 1978)
Increase renal blood flow
Vivo (Dog)
Compound 79
0.1 and 0.2 mg/kg/min, iv drop
Cardiotonic effect
Enhance the contractility of the papillary muscles
Vitro (Cat)
ZS injection
12.5, 6.25 mg/mL
high dose: n = 9, p < 0.01
low dose: n = 8, p < 0.05 or 0.01 (78p greater than 0.05)(Jia et al., 1980)
Compound 78
17.5, 8.75 μg/mL
Compound 79
12.5, 6.25 μg/mL
Induce autorhythmic contractions of the papillary muscles
Vitro (Cat)
ZS injection
6.25 mg/mL
n = 17, only 78p < 0.01
Compound 78
8.75 μg/mL
Compound 79
6.25 μg/mL
Increase the contractility and amplitude of the isolated heart
Vitro (Toad)
ZS fliud extract
10 %, 0.05 mL
50 %, 0.1 mL
n = 6
(Yan, 1995)
Increase coronary flow and cardiac contractility in isolated heart
Vitro (Guinea pig)
ZS injection
0.1–0.2 g
(XiangYa School Of Medicine, 1978)
Enhance systolic force, slow heart rate and output per minute in heart and lung preparations
Vitro (Guinea pig)
Perfusion of perfusate
6 mg/mL
Increase the cardiac output, accelerate the blood stream in bulbar conjunctiva
Vivo (Dog)
ZS injection
8 g/kg/h, iv drop
n = 4, p < 0.05 vs control
(Zhao et al., 1989)
Vivo
Compounds 78, 79
4 g/kg/h, iv drop
n = 5, p < 0.01 vs control
Increased the tension of aortic strip
Vitro (Rabbit)
ZS decoction
1 %–200 %
r = 0.82, n = 12, p < 0.001
(Li et al., 2001)
Increase the ICa-L current in ventricular myocytes and promote the opening of calcium channel
Vitro (Guinea pig)
ZS extract
4 × 10-3–1 × 10-1 g/mL
n = 10, p < 0.05 or 0.01 vs control
(Fang et al., 2003)
Vitro (Guinea pig)
Compounds 78, 79
10–100 mmol/L
n = 10, p < 0.05 or 0.01 vs control
(Fang et al., 2004)
Enhance the cardiac function, the peak dp/dt and the percentage in dp/dt /GPIP
Vivo (Dog)
ZS injection
0.5 g/kg, iv drop
236 %, p < 0.01
n = 6, vs control
(Chen et al., 1980)
111 %, p < 0.01
Compound 78
1 mg/kg, iv drop
86 %/68 %, n = 6, p < 0.05 vs control
Compound 79
0.25 mg/kg, iv drop
94 %, p < 0.01
n = 6, vs control
111 %, p < 0.05
Anti-atherosclerotic activity
Inhibit the ICAM-1 expression of HUVEC induced by ox-LDL
Vitro (HUVEC cell)
70 % ethanol extract of ZS
Compound 49
1.0 and 2.0 mg/mL
15.625 μg/mL
n = 4, p < 0.01 or 0.05 vs model control
(Luo et al., 2012)
Compound 56
0.25 mg/mL
Inhibit cyclic strain-induced HUVEC ET-1 secretion
Vitro (HUVEC cell)
Compound 49
10 and 100 mmol/L
n = 6, p < 0.05 vs control
(Chiou et al., 2008)
Reduce TG, TCH and ldl-cholesterol in the hyperlipidemia rats
Vivo (SD rat)
ZS injection
50, 200 mg/kg, ip drop
p < 0.01 or 0.05 vs control
(Ham et al., 2007)
Decrease the blood glucose
Vivo (Male KM mice)
70 % ethanol extract of ZS
5.07 mg/kg/d, i.g for 4 weeks
n = 10, p < 0.05 vs model control
(Jiao et al., 2009)
Reduce ED, and increase serum nitrite and vascular NO bioavailability
Vivo (Male Wistar rat)
Compound 49
100 mg/kg/d, p.o for 2 weeks
n = 10, p < 0.001
(Kumar et al., 2017)
Decrease the TG and TCH in the diabetic mice by abolishing the effect of l-methionine on Ach
Vivo (Male KK-Ay mice and C57BL/6 mice)
Compound 56
50 mg/kg, BW i.g for 6 weeks
n = 10, p < 0.001 or 0.01 vs model control
(Jia et al., 2015)
Antithrombotic activity
Anti-thrombosis
Vitro (Rabbit)
ZS decoction
0.1 g/mL
n = 5
(Ou et al., 1989)
Inhibit PLT and RBC aggregation
Vivo (Male Wister rat)
ZS decoction
1, 2.5 g/kg/d, i.g for 10 d
n = 11, p < 0.001 vs model control
(Ji et al., 2003)
Decrease PLT number in CIA rat
Vivo (Male SD rat)
Ethanol extract
3.6 g/d, i.g for 9 weeks
n = 8, p < 0.01 vs model control
(He et al., 2018)
Reduce serum Hcy and cholesterol in l-methionine-treated rats
Vivo (Male Wistar rat)
Compound 49
100 mg/kg/d, p.o for 2 weeks (22.37 ± 0.30 vs 11.01 ± 1.01 mg/mL) and (182.7 ± 2.15 vs 101.5 ± 2.76 mg/mL), respectively
n = 10, p < 0.001
(Kumar et al., 2017)
3.5.3.2 Cardiotonic effect
ZS extract showed the function of enhancing cardiac contractility and increasing coronary flow in various experimental models, including cat (Jia et al., 1980), toad (Yan, 1995), guinea pig (XiangYa School Of Medicine, 1978), dog (Zhao et al., 1989), and rabbit (Li et al., 2001). Synephrine (78) and N-methyltyramine (79) showed significant cardiotonic effect that could be blocked by phentolamine, verapamil, and incubation with Ca2+-free Krebs solution. It suggested this effect may be related to the activation of adrenergic α receptors, cholinergic M receptors, and verapamil-sensitive Ca2+ channels on the smooth muscle cell membrane (Chen et al., 1980; Li et al., 2001; Fang et al., 2003; Fang et al., 2004). In addition, hesperidin (49) and neohesperidin (56) possessed a cardioprotective effect (Wang et al., 2013) (Table 12).
3.5.3.3 Anti-atherosclerotic activity
ZS extract, as well as hesperidin (49) and neohesperidin (56), had a significant inhibitory effect on intercellular adhesion of human umbilical vein endothelial cells line (HUVEC) (Luo et al., 2012). In addition, hesperidin (49) inhibited the secretion of cyclic strain-induced HUVEC endothelin (ET)-1 secretion (Chiou et al., 2008). ZS injection (Ham et al., 2007) and neohesperidin (56) (Jia et al., 2015) significantly decreased the levels of triglycerides (TG), total cholesterol (TCH), and ldl-cholesterol in diabetic mice or hyperlipidemia rats. The 70 % ethanol extract of ZS was able to decrease blood glucose (Jiao et al., 2009). Hesperidin (49) significantly reduced endothelial dysfunction in rats (Kumar et al., 2017) (Table 12).
3.5.3.4 Antithrombotic activity
ZS decoction and its residue after removing ether-soluble ingredients had a significant inhibitory effect on thrombosis in vitro, indicating that the antithrombotic components of ZS are highly water-soluble substances (Ou et al., 1989). The effects of ZS decoction on inhibitory aggregation of platelet and red blood cell in both healthy and blood stasis rats was prior to those of aspirin and had a dose–effect relationship (Ji et al., 2003). Ethanol extract of ZS significantly decreased the platelet count in collagen‐induced arthritis rats (He et al., 2018). Hesperidin (49) reduced serum homocysteine and serum cholesterol of l-methionine-treated rats (Kumar et al., 2017) (Table 12).
3.5.4 anti-adipogenic activity
Obesity has been considered as major global health challenge in modern society. Synephrine (78), an analog of ephedrine, has been suggested as a safe dietary supplement to reduce obesity without the toxic and side effects of ephedrine analogs (Haaz et al., 2006). As a non-stimulatory thermogenic agent, synephrine tended to bind with β-3 receptor, thus promoted lipolysis, and had no effect on the CNS and cardiovascular system (Ribeiro et al., 2019; Rossato et al., 2011; Stohs et al., 2011a, 2011b; Stohs and Badmaev, 2016; Stohs, 2017). Synephrine (Guo et al., 2019), flavonoid (Kim et al., 2012), and menthol extract of ZS (Raciti et al., 2018) reduced the viability of 3 T3‐L1 cells and inhibited its differentiation, they could also promote lipolysis and reduce lipid accumulation. Synephrine and N-methyltyramine (79) could so stimulate lipolysis in both rats and human adipocytes (Mercader et al., 2011) (Table 13). The association of hypertension with cardiovascular risk in the short- and long-term was unequivocally established (Landsberg et al., 2013). As one of the main components of blood pressure elevating, synephrine and N-methyltyramine were easy to be destroyed by alkaline intestinal fluid after oral administration. Therefore, it elevates blood pressure only by injection (Xu, 2019). C/EBPα: CCAAT/enhancer-binding protein α; PPARγ: peroxisome proliferator-activated receptor γ; PKB/Akt: protein kinase B; GSK3β: glycogen synthase kinase 3β; BAT: brown adipose tissues; iWAT: white adipose tissues; SCD-1: stearoyl-CoA desaturase 1; FAS: fatty acid synthase; ACOX: acylCoA oxidase; LPS: lipopolysaccharide; XOD: Xanthine oxidase; AFIC-CDs: ZS carbonisata-derived carbon dots; MDA: Malondialdehyde; ROS: reactive oxygen species; FRAP: Ferric reducing antioxidant power.
Bioactivity
Mechanism
Method type
Extracts/compounds
Active concentration/dose
p value
References
Anti-adipogenic
Suppress adipogenesis, adipocytes differentiation and lipid accumulation by inhibited expression of C/EBPα and PPARγ, the PKB/Akt pathway and GSK3β activity
Vitro (3 T3-L1 cell)
Compound 78
1, 10 μM for 6 d
P < 0.01 vs control
(Guo et al., 2019)
Vitro (3 T3-L1 cell)
ZS flavonoid
10, 50 μg/ml for 4 or 6 d
p < 0.01 or 0.05 vs control
(Kim et al., 2012)
Reduce 10 % viability of cell
Vitro (3 T3-L1 cell)
Menthol extract of ZS
1000 μg/ml
p < 0.001 vs control
(Raciti et al., 2018)
Stimulate the lipolysis
Vitro (Wistar rat)
Compound 78
0.1–1000 μg/ml, 80 % of the maximal response
p < 0.001 vs model control
(Mercader et al., 2011)
Vitro (Human WAT cells)
Compounds 78
0.1–1000 μg/ml
33 % of the maximal response
p < 0.001 or 0.01 vs control
Compound 79
20 % of the maximal response
Increase the activity of BAT and induce the browning of iWAT by activating AMPK-PGC1α pathway
Vivo (Male C57BL/6J mice)
Polymethoxy flavonoids
0.2 % (w/w) p.o for 12 weeks
n = 10, p < 0.01 vs model control
(Kou et al., 2020)
Inhibit expression of SCD-1 and FAS. ACOX was induced by 56
Vivo (Male KK-Ay mice and C57BL/6 mice)
Compound 56
50 mg/kg, BW for 6 weeks
n = 10, p < 0.05, 0.01, or 0.001 vs control
(Jia et al., 2015)
Anti-inflammatory
Reduce TNF-α, IL-1β and IL-6
Vitro (RAW 264.7 cell)
50 % methanol ZS extract
160 μg/mL
p < 0.001 vs model control
(Zhao et al., 2018)
Compound 40
160 μg/mL
Decrease the serum of IL-1β, IL-6 and TNF-α
Vivo (Male SD rat)
50 % methanol ZS extract
250 mg/kg, i.g
n = 8
Compound 40
100 mg/kg, i.g
Reduce NF-κB, TNF-α, IL-1β and IL-6
Vitro (Male SD rat)
Compound 17
10 and 25 mg/kg
n = 20, p < 0.01 or 0.05 vs control/model control
(Bi et al., 2016)
Inhibit LPS-stimulated NO production by downregulating the protein expressions of TNF-α, NF-κB, COX-2, and iNOS
Vitro (RAW264.7 cell)
Compounds 17, 40, 41
100 μM
n = 6, p < 0.001 vs model control
(He et al., 2018)
Decrease IL-6, IL-1β and TNF-α in serum and colon tissue
Vivo (C57BL/6J mice)
ZS flavonoid
50, 100, 200 mg/kg p.o for 23 d.
n = 10, P < 0.001 vs model control
(Chen et al, 2022)
Enhance IL-10 and reduce TNF-α in mice colon tissue
Vivo (BALB/C mice)
Compound epoxyaurapten
160 and 320 μmol/L, 0.01 mL/g, i.g for 10 d
n = 10, p < 0.05 vs model control
(Lin et al., 2015)
Attenuate mRNA expression of COX-2
Vitro (RAW264.7 cell)
ZS pharmacopuncture
100, 50, 25, 12.5, 6.25 %
(Kim and Park, 2010)
Inhibit both His and dextran-induced edema
Vivo (Male Wistar rat)
70 % EtOH extract of ZS
75–200 mg/kg, i.g for 2 weeks
IC50 = 119.6 mg/kg
n = 6, p < 0.05
(Mencherini et al., 2013)
IC50 = 118.3 mg/kg
Inhibit inflammatory swelling of ankle joints induced by formaldehyde and egg white
Vitro (Rat)
ZS extract
egg white: 15 g/kg
n = 5, p < 0.001 vs control
(Zhang and Wang, 2004)
formaldehyde: 20 g/kg first day, 10 g/kg/d later
n = 6, p < 0.05 vs control
Decrease MPO and NO, and expressions of TNF-α, COX-2, iNOS and NF-κB
Vivo (Male SD rat)
Raw medicinal materials
125–500 mg/kg, p.o
n = 8, p < 0.001 vs model control
(He et al., 2018)
Inhibit the LPS-induced production of NO, TNF-α, IL-6, IL-1β and PGE2 in macrophages and decrease the endotoxin level and TNF-α in RAW 264.7 cells
Vitro (Male Wistar rat)
ZS decoction, transformed ZS
0.5, 1.0 and 2.0 mg/mL
n = 6, p < 0.01 vs model control
(Liu et al., 2017)
Reduce the paw pressure score and volume, ankle diameter by decreased IL-1β and TNF-α
Vivo (RAW 264.7 cell)
AFIC-CDs
2, 4 and 8 mg/kg
n = 8, p < 0.05 or 0.01
(Wang et al., 2019)
Vitro
0.4, 0.8 and 1.6 mg/mL
p < 0.05 or 0.01 vs model control
Reduce XOD activity on serum and liver
Vivo (Male SD rat)
AFIC-CDs
2, 4 and 8 mg/kg
Vitro
2 mg/kg
vs allopurinol
58.83 %
4 mg/kg
64.38 %
8 mg/kg
52.58 %
Decrease the production of iNOS, COX-2, IL-6 and TNF-α induced by LPS by suppressd NF-kB and MAPKs signal pathways
Vitro (L6 skeletal muscle cell)
ZS flavonoid
10, 50, 75 and 100 mg/mL
p < 0.05 vs model control/control
(Kim et al., 2012)
Anti-oxidant
Increase GSH–PX, GSH, and SOD, and reduce MDA
Vitro (Male SD rat)
Compound 17
10 and 25 mg/kg
n = 20, p < 0.01 or 0.05 vs control/model control
(Bi et al., 2016)
Inhibit strain-induced ROS formation
Vitro (HUVEC cell)
Compound 49
10 and 100 μmmol/L
n = 6, p < 0.05 vs control
(Chiou et al., 2008)
Scavenging of ·OH, ·O2-and DPPH·
Vitro
Compound 53
IC50 = 45.94 mg/mL
(Jiao et al., 2008)
SC50 = 10.16 mg/mL
DC50 = 9.62 mg/mL
70 % ethanol ZS extract
IC50 = 2.71 mg/mL
SC50 = 5.02 mg/mL
DC50 = 0.54 mg/mL
Inhibit lipid peroxidation in mouse liver, kidney and heart tissue homogenates
Vitro (Male KM mice)
Compound 53
9.87, 11.04, 10.40 mg/mL
Scavenging of ·OH, ·O2–, DPPH· and H2O2
Vitro
CLB 1–4
6.4 mg/mL > vitamin C (6.4 mg/mL)
(Wang et al., 2014)
Enhance the enzyme activities of SOD, CAT, and GSH-Px in blood, heart, and liver
Vivo (Male C57BL/6N mice)
CALB
100 and 200 mg/kg
Reduce MDA formation in blood, heart, and liver
CALA
100 mg/kg
Scavenging of DPPH·, ABTS+ and ·OH, FRAP
Vitro
Crude juice
DPPH: 14.39 ± 0.19 μM TE
n = 4
(Barreca et al., 2014)
ABTS+:14.30 μM TE (64 %)
·OH: 4.8 μM TE
C- and O-glycosyl flavones
9.50 %
O-glycosyl flavanones
18.20 %
Flavonoid pool
DPPH·: 12.25 ± 0.70 μM TE (85.1 %)
ABTS+: 5.6 μM TE (25 %)
·OH: 32 %
Scavenging ·OH
FA3: contain flavonoids
10–2000 mg/mL
(Liu et al., 2009)
FA4: contain 9.96 % limonin
2000 mg/mL
Scavenging of DPPH· and SOD, against H2O2-induced cell injury and attenuate LPS-induced COX-2 mRNA expression
Vitro (RAW264.7 cell)
ZS pharmacopuncture
6.25 %–50 %
(Kim and Park, 2010)
Ameliorate all biochemical parameters of lung damaged rats caused by chromium
Vivo (Female Wistar rat)
80 % ethanol ZS extract
100 and 300 mg/kg/d, p.o for 10 days
n = 6, p < 0.001 vs model control
(Soudani et al., 2013)
Decrease the content of MDA, increase the content of NO, GSH and the activity of CAT
Vivo (Male KM mice)
70 % ethanol ZS extract
1.69, 3.37, 5.07 mg/kg, 1 time/d, i.g for 4 weeks,
n = 10, p < 0.01 or 0.05 vs model control
(Jiao et al., 2009)
Dietary supplementation of 0.2 % ploymethoxy flavonoids of ZS with high-fat diet (w/w) markedly reduced body weight gain of obese mice and elevated their thermogenesis in the cold tolerance test (4℃) (Kou et al., 2020). Neohesperidin (56) also inhibited lipid accumulation in the liver and decreased the size of epididymal adipocytes of KK-Ay mice (Jia et al., 2015).
3.5.5 anti-inflammatory activity
The constituents (Bi et al., 2016; He et al., 2018), extracts (Zhao et al., 2018), and preparations of ZS (Kim and Park, 2010; Liu et al., 2017; Wang et al., 2019) could reduce the expression of nuclear factor-kappa B (NF-κB), tumor necrosis factor-alpha (TNF-α), IL-1β, IL-6, NO, cyclooxygenase (COX)-2, inducible nitric oxide synthase (iNOS), and myeloperoxidase (MPO) of RAW 264.7 cells through mitogen-activated protein kinases signaling pathway. ZS flavonoids possessed similar effects on lipopolysaccharide-induced L6 skeletal muscle cells (Kim et al., 2012). Epoxyaurapten (Lin et al., 2015) up-regulated IL-10 and down-regulated TNF-α to treat ulcerative colitis. ZS exhibited higher inhibition on xanthine oxidase (Wang et al., 2019), which could induce gouty arthritis by increasing uric acid, than the mature fruit of Citrus medica L. and Citrus medica L. var. sarcodactylis Swingle, QP, and CP). Besides, the ethyl acetate extract showed the strongest inhibition than others (Liu et al., 2016).
ZS extract significantly inhibited inflammatory swelling of joints of rats induced by histamine, dextran (Mencherini et al., 2013), formaldehyde, and egg white (Zhang and Wang, 2004). The carbonized product of ZS significantly reduced the paw pressure score and volume, as well as ankle diameter of gouty arthritis rats (Wang et al., 2019). ZS reduced inflammatory cell infiltration and decreased the level of the inflammatory factor of trinitrobenzene sulfonic acid-induced inflammatory bowel disease rats (He et al., 2018). ZS flavonoid alleviated inflammation by decreased the levels of IL-6, IL-1β and TNF-α in DSS-induced colitis mice (Chen et al., 2022). Combination therapy of ethanol extract of ZS and MTX effectively reduced the inflammatory symptoms (ankle joint, bone destruction) and joint damage by inhibiting the NF‐κB pathway (He et al., 2018) (Table 13).
3.5.6 Anti-oxidant activity
Flavonoids (Chiou et al., 2008; Jiao et al., 2008; Barreca et al., 2014), polysaccharides (CLB 1–4) (Wang et al., 2014), and extracts of ZS (Jiao et al., 2008; Kim and Park, 2010) showed scavenging activity on ·OH, ·O2–, DPPH·, and ABTS+ radicals. The scavenging capacity of ·OH, ·O2–, and DPPH· of 70 % ethanol extract of ZS was 2.02–17.81 times higher than naringin (53) (Jiao et al., 2008). The antioxidant effect of ethanol extract and extract after purified by macroporous resin column chromatography had no significant difference, suggesting that polysaccharides and proteins of ZS may highly contribute to the activity (Xie et al., 2009). ZS harvested from June 11 to July 7 possessed the highest antioxidant capacity on H2O2-induced RIN-m5F cells (Tang et al., 2021).
Nobiletin (17) (Bi et al., 2016) and polysaccharides (Wang et al., 2014) exerted antioxidant effect by increasing concentrations of glutathione (GSH)‑PX, GSH, Catalase (CAT) and superoxide dismutase (SOD), and reducing the level of malondialdehyde and NO of isoflurane‑induced cognitive impairment rats and d-galactose-induced aging mice. Pretreatment with 80 % ethanol extract of ZS ameliorated all biochemical parameters of lung-damaged rats caused by chromium (Soudani et al., 2013). The extract of ZS could effectively enhance the kidney antioxidant function of diabetic rats (Jiao et al., 2009) (Table 13).
3.5.7 Anti-tumour activity
ZS had a good inhibitory effect on a variety of cancer cells, including colon cancer (Jayaprakasha et al., 2008), hepatocellular carcinoma (Ma et al., 2013), breast adenocarcinoma (Wang, 2008), gastric cancer (Dong et al., 2011), leukemia (Han et al., 2012), and hepatoblastoma (Lee et al., 2015) by arresting G2/M phase. ZS extract significantly enhanced the cytolytic activity of natural killer cells and the expression of natural killer cell-activating receptors, especially NKp30 and NKp46 (Park et al, 2022). Compounds 17, 18, and 22 play a great role in inhibiting angiogenesis, and the compounds with methoxy in C8 position were better than the others in inhibiting vascular growth (Yang, 2007). ZS flavonoid could inhibit the growth of tumor induced by HepG2 cells (Lee et al., 2015). Nobiletin (17) significantly inhibited the growth of H22 transplantable tumors (Ma et al., 2013) (Table 14). CREB: response element binding protein; BDNF: brain-derived neurotrophic factor; ELT: escape latency time; TSTQ: time spent in the target quadrant.
Bioactivity
Mechanism
Method type
Extracts/compounds
Active concentration/dose
p value
References
Anti-tumour
G2/M cell cycle arrest
Vitro (HT-29 cell)
Compounds 94, 96
1.25, 2.5, 5.0, and 10.0 μM
(Jayaprakasha et al., 2008)
G2 cell cycle arrest, decrease the expression of Bcl-2 and COX-2 and increase Bax and caspase-3
Vitro (SMMC-7721 cell)
Compound 17
IC50 = 26.51 mg/L
(Ma et al., 2013)
Inhibit the growth of H22 transplantable tumor by decreased the expression of COX-2 and Bcl-2/Bax and increased Bax and caspase-3
Vivo (H22 cell)
Compound 17
125, 250, and 500 mg/kg, i.g daily
n = 10, p < 0.01 or 0.05 vs model control
Inhibit angiogenesis
Vivo (Hatching egg)
Compounds 14, 18, 22
n = 15, p < 0.05 vs control
(Yang, 2007)
Vitro (SMMC-7721 cell and BcaP37 cell)
Compounds 16, 17, 22
12.5, 25, 50, 100 μg/mL
(Wang, 2008)
G2/M cell cycle arrest and apoptosis, upregulate p53 and p21 protein
Vitro (AGS cell)
Compound 24
5, 10, 30, 60, 120 μmol/L
p < 0.01 or 0.05 vs control
(Dong et al., 2011)
Inhibit the expression of XIAP and Bcl-xL and the activity of Akt
Vitro (U937 cell)
Crude methanol extracts of the ZS
10–100 μg/ml
p < 0.05 vs control
(Han et al., 2012)
Increase the expressions of caspase 3, P-p38, Bax, and Bak, decrease Bcl-2, Bcl-xL, pAkt, and phosphoinositide-3-kinase/Akt pathway
Vitro (HepG2 cell, Thle2 cell)
ZS flavonoid
25, 50, 75, and 100 μg/ml
IC50 = 75 μg/mL
p < 0.05 vs control
(Lee et al., 2015)
Inhibit tumor induced by HepG2 cell growth in the xenograft model
Vivo (BALB/c mice)
ZS flavonoid
25 and 50 mg/kg
n = 3
Enhances natural killer cytolytic activity
Vivo (C57BL/6 mice)
ZS extract
200 mg/kg, p.o for 10 d
p < 0.05 or p < 0.01
(Park et al., 2022)
Vitro (NK-92, K562 and Y AC-1 cells)
50, 100 µg/mL
p < 0.05 or p < 0.01
Neuroprotective
Via modulation of Akt, Bax, p-CREB and BDNF in aging rats
Vivo (Male SD rat)
Compound 17
10, 25 mg/kg/d, ip drop
n = 20, p < 0.01 or 0.05 vs model control
(Bi et al., 2016)
Prevent rise in ELT and decrease TSTQ
Vivo (Male Wistar rat)
Compound 49
50 and 100 mg/kg, p.o for 2 weeks.
n = 10, p < 0.001
(Kumar et al., 2017)
Decrease the activity of AchE in the cortex and hippocampus and the expression of Bax and caspase–3 protein, increase the expression of Bcl–2 in the hippocampus
Vivo (Male c57Bl/6J mice)
ZS extract
50, 100 mg/kg, p.o for 4 weeks
n = 8, p < 0.05 vs model control
(Lee et al., 2019)
Compound 17
50 mg/kg, p.o for 4 weeks
Increase cerebral blood flow and decrease cerebral vascular resistance
Vivo (Dog)
ZS injection
1 g/kg, iv drop
n = 5, p < 0.01
(XiangYa School Of Medicine, 1978)
The activities of SOD, CAT and GSH-Px were significantly enhanced against 6-OHDA-induced oxidative stress
Vitro (PC12 cell)
ZS flavonoid
3.75, 7.5, 15 μg/mL
p < 0.05
(Liang et al., 2021)
3.5.8 Anti-bacterial activity
Limonin (90), glycosides of its analogues, and ZS flavonoids had sound inhibitory effects on Escherichia coli and Staphylococcus aureus (Liu et al., 2009). ZS flavonoids also could reduce the diversity and abundance of the intestinal microbiota in DSS-induced colitis mice (Chen et al., 2022). Currently, the antibacterial research of ZS mainly focuses on its essential oil. It showed a dose-dependent inhibition on mycelium growth and aflatoxin B1 genesis of Aspergillus flavus at 0.5–2 % (v/v). The effect varied among essential oils from different fruits of the Citrus genus (Restuccia et al., 2019). In addition, it showed inhibition on Fusarium oxysporum, Fusarium solani, Botrytis cinerea, Bipolaris sorokiniana, and Fusarium avenaceum with the inhibitory rate of 38.49 %–68.47 % at 2 μL/mL and 76.13 %–84 % at 4 μL/mL (Metoui et al., 2015).
3.5.9 Neuroprotective activity
ZS flavonoid significantly enhanced SOD, CAT, and GSH-Px activities of 6-hydroxydopamine-treated PC12 cells (Liang et al., 2021). Synephrine (78) was able to bind to the neuromedin U2 receptor of HEK293 cells with high efficacy and potency (Zheng et al., 2014). Synephrine (78) was synthesized in 1927 as a sympathomimetic drug and was included in Nordic Pharmacopoeia and Deutsches Apothekerbuch. ZS extract (Lee et al., 2019), nobiletin (17) (Bi et al., 2016), and hesperidin (49) (Kumar et al., 2017) effectively decreased the escape latency time, path length (length taken to reach the platform), and swim distance. They also increased the time spent in the target quadrant and the number of crossed the platform. Nobiletin (17) might ameliorate cognitive impairment of aging rats via modulation of Akt, B-cell lymphoma 2-associated X protein, p-cAMP response element binding protein, and brain-derived neurotrophic factor (Bi et al., 2016). Furthermore, it could also down-regulate the acetylcholinesterase activity and caspase‑3 protein expression and up-regulate the Bcl‑2 expression of the cortex and hippocampus (Lee et al., 2019). ZS injection could significantly increase cerebral blood flow and decrease cerebrovascular resistance of dogs (XiangYa School Of Medicine, 1978) (Table 14).
3.5.10 Immunomodulatory activity
ZS extract, hesperidin (49), and neohesperidin (56) can significantly increase the content of NO in the supernatant of HUVEC induced by ox-LDL. The extract and hesperidin can also increase NO content in the supernatant of normal HUVEC (Luo et al., 2012). Endothelial NO synthase (eNOS) activity and the phosphorylation of eNOS and Akt of HUVEC were enhanced after being treated with compound 49 (Chiou et al., 2008).
3.5.11 Effect on the reproductive system
ZS extracts, rich in synephrine (78) and N-methyltyramine (79), had the effect of stimulating isolated circular muscles of the vaginal (Tang, 2001) and uterus (n = 7, p < 0.01 or p < 0.05) (Zhang et al., 2007) of rabbit, which resulted in rhythmically compress activities of muscle or increase of the spontaneous muscle contract intension and frequency. This result was consistent with those conducted in vivo (Yan, 1995). The extract showed bidirectional regulation on the isolated mouse uterus. ZS, combined with Kushen (the root of Sophora flavescens Ait.) (2:1, v/v), showed stronger inhibitory activity in the circular muscle of the vaginal (Tang, 2001).
3.5.12 Adrenaline action
Synephrine (78) could promote lipolysis by binding with β-3 receptor (Ribeiro et al., 2019; Rossato et al., 2011; Stohs et al., 2011a, 2011b; Stohs and Badmaev, 2016; Stohs, 2017). A novel cell-based functional assay suggested that the ZS are the possible agonists of β-2 adrenoceptor (Wang et al., 2009). N-methyltyramine (79) could treat gastrointestinal disorders via the regulation of adrenergic receptors, which may be related to the bio-transformation of compound 79 to epinephrine by serial synthase in nerve cells of the small intestine (Ni et al., 2019). ZS injection (1.5 g/kg) and compound 79 (0.1 and 0.2 mg/kg) significantly increased renal blood flow and urine output, and decreased renal vascular resistance of dogs (Guo, 1978).
3.5.13 Others
Hesperetin (41) and naringenin (40) displayed broad-spectrum inhibition against human glucuronosyltransferases (Liu et al., 2016). The methanol extracts of ZS showed obvious inhibitory effects on α-glucosidase and acetylcholinesterase. Flavanone glycosides of ZS were effective on α-glucosidase while polymethoxy flavonoids were effective on acetylcholinesterase (Guo et al., 2021). In addition, ethanol extract of ZS induced the expression of P-glycoprotein and cytochrome P450 3A4 of expression via upregulation of the pregnane X receptor in LS180 cells (Okada et al., 2017).
The volatile oil (Hu et al., 1994) and extract of ZS (10.8 g/kg) (Zhang and Wang, 2004) significantly reduced the incidence of acetic acid-induced torsional reactions in mice (p < 0.05, p < 0.01, respectively). ZS could improve the bioavailability of omeprazole in rats (Yu et al., 2013). Nomilin (89) and limonin (90) showed anti-obesity and anti-hyperglycemic effects via binding to Takeda G protein-coupled receptor 5, a bile acid receptor (Zohra et al., 2020). ZS can significantly increase the growth of Ctenopharyngodon idella (Ding et al., 2005) and Litopenaeus vannamei (Guo et al., 2005). Essential oil of ZS could reduce the survival rate of larvae of Culex pipiens (Michaelakis et al., 2009).
3.6 Toxicity
Safety is the most basic properties of health foods. Toxicity studies (Table 15) indicated ZS is safe for health food purposes. No obvious toxicity of ZS on mice was observed (Lin et al., 2012). Ethanol extract and pure compounds (49 and 58) had no toxicity on HUVEC (Luo et al., 2012), LS180 (Okada et al., 2017), and/or RAW 264.7 cells (Wang et al., 2019). Since 2004, ephedra products have been banned for weight loss by FDA due to their safety problems (Bent et al., 2004). The extracts of the fruit of C. aurantium, which contains synephrine (78) with a similar chemical structure and effect on weight loss, rapidly became its replacement due to its safety (Haaz et al., 2006). Clinical trials indicated that synephrine and C. aurantium extracts did not result in cardiovascular diseases and did not act as stimulants at commonly used doses compared to with more active substances like caffeine (Stohs, 2017). No adverse reactions were observed after sixteen healthy subjects treated with the extracts of C. aurantium for 15 days (Shara, Stohs and Smadi, 2018). The mixture containing synephrine, caffeine and green tea extract did not lead to increased cardiovascular stress (Seifert et al., 2011). Both commercial ZS extract (containing 7.5 % synephrine) and synephrine could reduce the body weight gain of the mice but had no significant effects on the relative weight of their organs and biochemical and hematological parameters (Arbo et al., 2009). Synephrine or combined with caffeine did not produce developmental toxicity in rats (Hansen et al., 2011). No adverse effect was observed in a 90-day subchronic safety/toxicity study (Deshmukh et al., 2017). Synephrine did not exhibit cytotoxicity on the 3 T3-L1 cells even at a high dose (Guo et al., 2019). Synephrine was not cytotoxic, genotoxic, and mutagenic towards human liver cells at specific concentrations (Ribeiro et al., 2019).
Extracts/compounds
Vitro/vivo
Active concentration/dose
Effect
Reference
Wall-broken spore power of ZS
Vivo
(SPF) KM mice
120 g/kg, 40 mL/kg, p.o for 7 d
n = 10, no cytotoxicity
(Lin et al., 2012)
70 % ethanol extract of ZS
Vitro
HUVEC cell
2 mg/mL
n = 4, survival rate greater than 80 %
(Luo et al., 2012)
Compound 49
0.03125 mg/mL
Compound 56
0.25 mg/mL
Ethanol extracts
Vitro
LS180 cell
20 mg/mL
No cytotoxicity
(Okada et al., 2017)
AFIC-CDs
Vitro
RAW 264.7 cell
5000 μg/mL
No cytotoxicity
(Wang et al., 2019)
C. aurantium extract
Vivo
Human
Containing 49 mg compound 78, for 15 d
n = 16, no stimulant (cardiovascular) and adverse effects.
(Shara, Stohs and Smadi, 2018)
Synephrine, caffeine and green tea extract
Vivo
Human
13 mg synephrine + 176 mg caffeine + 55.5 mg green tea extract, for 24 h
n = 23, not increased cardiovascular stress
(Seifert et al., 2011)
ZS extract
Vivo
Male albino CF1 mice
400, 2000 or 4000 mg/kg, p.o for 28 d
n = 9–10, no cytotoxicity
(Arbo et al., 2009)
Compound 78
30 or 300 mg/kg, p.o for 28 d
Compound 78
6 % synephrine extract and caffeineVivo
Sprague-Dawley rats
50, 100 mg/mL
25 mg/ kg 6 % synephrine extract + 25 mg/ kg caffeine
n = 7, not produce developmental toxicity
(Hansen et al., 2011)
ZS extract (contain 7.5 % compound 78)
Vivo
SD rat
1000 mg/kg/d, p.o for 90 d
n = 10, resulted in non-adverse effects
(Deshmukh et al., 2017)
Compound 78
Vitro
3 T3-L1 cell
200 μM for1 or 6 d
No cytotoxicity
(Guo et al., 2019)
Compound 78
Vitro
HepG2 cell
2, 20 and 200 μM
No cytotoxic, genotoxic, and mutagenic
(Ribeiro et al., 2019)
Compound 78
Vivo
CF1 mice
300, 350, 400 mg/kg
Some nonspecific toxic effects
(Schmitt et al., 2012)
ZS flavonoid
Vitro
L6 skeletal muscle cell
150, 200 μg/mL
Affect viability
(Kim et al., 2012)
ZS extract
Vivo (Mice)
i.g for 3 d
n = 10, LD50 = 118.3 g/kg, maximum safe dose = 68.5 g/kg
(Zhang and Wang, 2004)
i.h
LD50 = 58.4 g/kg, the maximum safe dose = 27.5 g/kg
ZS injection
Vivo (Mice)
Tail vein injection
LD50 = 71.8 ± 6.5 g/kg
(XiangYa School Of Medicine, 1978)
i.p
LD50 = 267 ± 37 g/kg
Vivo
Dog
21 g/kg, iv
No cytotoxicity
There were several clinical hazard reports containing extract of the fruit of C. aurantium (Stohs, 2010; Stohs and Ray, 2020). They all emphasized the similarities of the chemical structure and side effects between ephedrine and synephrine. However, ephedrine is a phenylpropanolamine derivative without a substituted hydroxy group, while synephrine is a phenylethanolamine derivatives with a para-substituted hydroxyl group, which led to the difference of their half-life (Stohs et al., 2011b; Stohs, 2017; Stohs et al., 2020a, 2020b). Ephedrine exhibited better effect for binding to α, β-1, and β-2 receptor, while synephrine bound to β-3 receptor (Costa et al., 2022; Stohs et al., 2011b; Stohs and Badmaev, 2016; Stohs et al., 2020). In addition, the para hydroxy group and the lack of the methyl group on the side-chain of synephrine greatly decreased its lipid solubility and the ability of crossing the blood–brain barrier, which resulted in no effect on the CNS and cardiovascular stimulation (Rossato et al., 2011; Stohs et al., 2011a, 2011b; Stohs, 2017).
As a trace amine, synephrine have been detected in human plasma and platelets (Costa et al., 2022; Rossato et al., 2011). On a daily basis, C. aurantium extract (Advantra Z®; synephrine)-containing products and a variety of orange juices were consumed by tens of millions of people worldwide without the report of serious incidents (Stohs et al., 2011a; Stohs, 2017). Under the specified dose, direct experimental evidence and numerous peer reviews have proved the safety of C. aurantium extract and synephrine from human, animal, and cell levels whenever used alone or combined with stimulants such as caffeine (Stohs, 2017) The clinical hazard reports containing C. aurantium extract and synephrine should consider other ingredients, adulteration problems, and the physical condition of patient (Costa et al., 2022; Hansen et al., 2011; Rossato et al., 2011; Stohs, 2017). As a consequence, the hazard of synephrine cannot be inferred through the current scientific assumptions.
However, studies also suggested that ZS could cause damage when used in high doses, which is one of the most important reasons why ZS can be used as a health food but not as dietary herbal medicine. According to ChP, the clinical administrations of ZS for adults are suggested to be 3–10 g daily (Chinese Pharmacopoeia Commission, 2020b). When the dose of synephrine reached to 400 mg/kg, no specific toxic side effects such as erection and salivation was observed in mice (Schmitt et al., 2012). ZS flavonoids could affect viability of L6 skeletal muscle (Kim et al., 2012). According to the NHCPRC, the rat with an oral LD50 value is 5001–15000 mg/kg was listed as grade 2 (practically non-toxic). High LD50 values were also observed when the mice were intragastrically administered and intraperitoneally or intravenously injected (Zhang and Wang, 2004). A high dose of ZS caused high and fast elevation of blood pressure (over 180–200 mmHg), temporary ectopic rhythm, and anuria of anesthetized dogs (XiangYa School Of Medicine, 1978). The oral administration (23.42 g/kg/d) of ZhiZi HouPo Decoction containing ZS caused damage to the liver and kidneys, and that of the SCZS-containing group (n = 5) was more severe to the liver and kidneys of rats than the TCZS-containing group (Zhang and Feng, 2019).
3.7 Clinical research
Zhizhu pills containing SCZS (n = 82) exhibited superior effects on FD patients with spleen-deficiency and Qi-stagnation syndrome than those containing TCZS (n = 78) (Wu et al., 2011). The effective rate on FD patients (n = 60) after 4 weeks of treatment with Weikang pian (flavonoids extracted from ZS) was 94 %–96 %, and no adverse event occurred (Yan et al., 2019). In addition, ZS and its main constituents, including synephrine (78) and N-methyltyramine (79), had a therapeutic effect on septic shock children (Pediatric Septic Shock Research Cooperative Group, 1981; Zhao et al., 1989). Furthermore, ZS extraction could improve the symptoms of atopic dermatitis patients (Kim and Jung, 2014).
ZS capsules could increase the levels of HDL-C and NO and decrease the levels of ET-1, TC, TG, and LDL-C of hyperlipidemia patients (p < 0.01, n = 40). These effects of ZS capsules were significantly better than fluvastatin (p < 0.01) (Wu et al., 2012). ZS increased NO and reduced platelet aggregation, RBC aggregation, CD62p, d-dimer, ET-l, and Ox-LDL of acute coronary syndrome patients (p < 0.05 or 0.01, n = 34) (Ji et al., 2008).
4 Discussion and conclusions
This article presents a systematic review of ZS as an example of the fruit used as both food and medicine. The plant origin of ZS has undergone a variety of changes in history, which caused the phenomenon of the fruit of other Citrus genus plants used as ZS to become quite common. In addition, the plant source of ZS used in the classical prescriptions of different periods needs to be further investigated for their development. It is unable to identify all the adulterants by the current quality control methods. Therefore, systematic research is needed for the authentification of ZS. The fruit of both C. aurantium and C. sinensis can be used as ZS according to ChP, but their difference in the chemical composition may result in variation in pharmacological effect, which should be the focus of later research (Shi et al., 2019; Song et al., 2016; Zeng et al., 2016; Zhang et al., 2011). The harvesting time is a significant factor causing the variations in chemistry and bioactivity, but these variations remain to be further clarified (Deng et al., 2017; Deng et al., 2019; Tang et al., 2021). Thus, it is important to use ZS samples harvested in the right season in clinic practice or academic research.
The discussion on the warm or cold medicinal properties of ZS requires more experimental and clinical evidence. Synephrine, the major compound of ZS, is safe at a specific dose, but long-term safety study should be conducted. Besides, more attention should be paid to other compounds, such as flavonoids, and their bioactivities in digestive and cardiovascular systems. ZS is able to eliminate the food stagnation, constipation, diarrhea, and phlegm caused by the Qi being blocked in the abdominal cavity due to its Qi-regulating effect which is closely related to the bi-directional regulation effects on gastrointestinal tract. For example, ZS stimulate or inhibit gastrointestinal smooth muscle when treating food stagnation or diarrhea, respectively. Whether its phlegm eliminating effect is related to bronchial smooth muscle stimulating activity is not clear. Stimulating smooth muscle is the pharmacological basis for its therapeutic effect on the prolapse of stomach, uterus, and anus.
In summary, as a health product and TCM, ZS possesses extensive health care and therapeutic effects, but more comprehensive studies on its quality control, pharmacology, and safety is necessary to reveal its scientific evidence of health benefits.
Acknowledgements
This work was supported by the National Key Research and Development Program of China (2018YFC1707903), Provincial Natural Science Foundation of Hunan (2022JJ40318), Scientific Research Fund of Hunan University of Chinese Medicine (2021XJJJ006), Pharmaceutical Open Fund of Domestic First-class Disciplines (cultivation) of Hunan Province (2020YX10), Changsha Municipal Science and Technology Bureau (kq1901095), and College Students' innovative projects of the Hunan University of Chinese Medicine.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- The regulatory effect of Citrus Aurantium on the MTL and VIP in the acute stage of cerebral infarction rats. Chi J Chin Med. 2015;30:1461-1463.
- [Google Scholar]
- Phytochemical and therapeutic potential of Citrus grandis (L.) Osbeck: A Review. J Evid Based Integr Med. 2021;26:1-20.
- [Google Scholar]
- Subchronic toxicity of Citrus aurantium L. (Rutaceae) extract and p-synephrine in mice. Regul Toxicol Pharm. 2009;54:114-117.
- [Google Scholar]
- High-performance thin-layer chromatography/desorption electrospray ionization mass spectrometry imaging of the crude extract from the peels of Citrus aurantium L. (Rutaceae) Rapid Commun Mass Sp. 2015;29:1530-1534.
- [Google Scholar]
- Identification and comparison of constituents of Aurantii Fructus and Aurantii Fructus Immaturus by UFLC-DAD-Triple TOF-MS/MS. Molecules. 2018;23:803.
- [Google Scholar]
- First evidence of C- and O-glycosyl flavone in blood orange (Citrus sinensis (L.) Osbeck) juice and their influence on antioxidant properties. Food Chem. 2014;149:244-252.
- [Google Scholar]
- C-and O-glycosyl flavonoids in Sanguinello and Tarocco blood orange (Citrus sinensis (L.) Osbeck) juice: Identification and influence on antioxidant properties and acetylcholinesterase activity. Food Chem. 2015;619:196.
- [Google Scholar]
- Safety and efficacy of Citrus aurantium for weight loss. Am J Cardiol. 2004;94:1359-1361.
- [Google Scholar]
- A myoelectric activity of a small intestine influenced by Zhishi. Chin J Tradit Chin Med Pharm. 1991;6:39-40.
- [Google Scholar]
- Nobiletin ameliorates isoflurane-induced cognitive impairment via antioxidant, anti-inflammatory and anti-apoptotic effects in aging rats. Mol Med Rep. 2016;14:5408-5414.
- [Google Scholar]
- Research on variety verification and resource application of Aurantii Fructus and Aurantii Fructus Immaturus medicinal materials. J Jiangxi Univ Tradit Chin Med. 1998;10:184-186.
- [Google Scholar]
- Studies on Chinese Drugs Zhiqiao and Zhishi Origin Investigation and Merchandise ldentification. Jiangxi College of TCM. 1999;24:259-262.
- [Google Scholar]
- Comparative study between bran stir-fried and sand-scalded Aurantii Fructus Immaturus based on chemometrics. Chin J Mod Appl Pharm. 2022;39:1395-1403.
- [Google Scholar]
- Anxiolytic and sedative effects of extracts and essential oil from Citrus aurantium L. Biol Pharm Bull. 2002;25:1629-1633.
- [Google Scholar]
- Discrimination of geographical origin of oranges (Citrus sinensis L. Osbeck) by mass spectrometry-based electronic nose and characterization of volatile compounds. Food Chem. 2019;277:25-30.
- [Google Scholar]
- Study on the effect of odoriferous components of Citrus aurantium on gastrointestinal motility in mice. Jiangxi J Traditl Chin Med. 2019;50:67-70.
- [Google Scholar]
- Chen, S., Zhou, Q. Y., Chen, L., Liao, X., and Li, R.et al., 2022. The Aurantii Fructus Immaturus flavonoid extract alleviates inflammation and modulate gut microbiota in DSS-induced colitis mice. Frontiers in Nutrition 9.
- Studies of Citrus Aurantium and its hypertensive ingredients on the cardiac functions and hemodynamics in comparison with dopamine and dobutamine. Acta Pharm Sin. 1980;15:71-77.
- [Google Scholar]
- Comparison of the pharmacological effects of Citri Reticulatae Pericarpium Viride, Citri Reticulatae Pericarpium and Aurantii Fructus Immaturus. Jiangsu J Tradit Chin Med 1981:60-61.
- [Google Scholar]
- Content comparison of active flavonoids in Zhishi granules from different manufactories. Jiangxi J Traditl Chin Med. 2010;25:914-916.
- [Google Scholar]
- Development of a HPLC-UV Method for the Separation and Quantification of Hesperidin, Neohesperidin, Neohesperidin Dihydrochalcone and Hesperetin. Nat Prod Res ahead-of-print 2022:1-5.
- [Google Scholar]
- Chen, P.S. 1998. Investigation, exploitation and protection of Citrus autium var. Documana Bonar Resources. J Zhejiang For Sci & Tech.
- An evidence-based update on the pharmacological activities and possible molecular targets of Lycium barbarum polysaccharides. Drug Des Devel Ther. 2014;17:33-78.
- [Google Scholar]
- Pharmacopoeia of the People’s Republic of China (first ed.). Guangzhou: Chemical Industry Press, Beijing & Guangdong Science and Technology Press; 1995. p. :200.
- Pharmacopoeia of the People’s Republic of China (first ed.). Beijing: Chemical Industry Press; 2000. p. :200.
- Pharmacopoeia of the People’s Republic of China (first ed.). Beijing: China Medical Science Press; 2020.
- Pharmacopoeia of the People’s Republic of China (first ed.). Beijing: China Medical Science Press; 2020. p. :258.
- Chinese Pharmacopoeia Commission. 1963. Pharmacopoeia of the People’s Republic of China, first ed. People’s Medical Publishing House, Beijing, pp. 195.
- Chinese Pharmacopoeia Commission. 1985. Pharmacopoeia of the People’s Republic of China, first ed. People’s Medical Publishing House & Chemical Industry Press, Beijing, pp. 197.
- Chiou, C.S., Lin, J.W., Kao, P.F., Liu, J.C., and Cheng, T.H., et al. 2008. Effects of hesperidin on cyclic strain-induced endothelin-1 release in human umbilical vein endothelial cells. Clin Exp Pharmacol P 35, 938-943.
- Spasmolytic and anti-peptic ulcer activities of crude drugs acting on gastrointestinal tract in rats. Yakhak Hoeji. 1996;40:591-598.
- [Google Scholar]
- Origin identification on the commercial samples of Aurantii Fructus. J Sep Sci. 2007;30:1235-1241.
- [Google Scholar]
- Chinese Pharmacopoeia Commission. 1977. Pharmacopoeia of the People’s Republic of China, first ed. People’s Medical Publishing House, Beijing, pp. 399.
- Antidepressant mechanism of Shugan Lipi recipe in regulating tryptophan metabolism and its common pharmacodynamic substances. Chin J Chin Mater Med. 2021;64:3633-3642.
- [Google Scholar]
- Correll, W. 2015. “Warning letter to TruVision health LLC. FDA Center for Food Safety and Applied Nutrition”.
- Citrus aurantium L. Essential oil exhibits anxiolytic-like activity mediated by 5-HT1A-receptors and reduces cholesterol after repeated oral treatment. Bmc Complem Altern M. 2013;13:42.
- [Google Scholar]
- Natural Sympathomimetic Drugs: From Pharmacology to Toxicology. Biomolecules (Basel, Switzerland). 2022;12:1793.
- [Google Scholar]
- The different effects of 2 species of Fructus Aurantii Immaturus on cardiovascular and respiratory system of rats. Chin J Informa TCM. 2010;17:41-43.
- [Google Scholar]
- Simultaneous extraction of bioactive limonoid aglycones and glucoside from Citrus aurantium L. Using hydrotropy. Z Naturforschung C J Biosci. 2008;63:176-180.
- [Google Scholar]
- UPLC fingerprinting of Citrus aurantium from different origins and harvesting periods. J Chin Med Mater. 2017;40:2051-2054.
- [Google Scholar]
- Study on chemical constituents of ethanol extract from Cirtus aurantium. China Pharm. 2018;29:458-460.
- [Google Scholar]
- Establishment of GC-MS fingerprints of volatile components in Citrus aurantium of different gathering periods. Chin Tradit Pat Med. 2019;41:2160-2163.
- [Google Scholar]
- Determination of four monoterpenoids in Citrus aurantium by GC-MS. Lishizhen Med Mater Med Res. 2020;31:264-266.
- [Google Scholar]
- Discussion about application of thin layer chromatography in current quality standard control. Chin J Exp Tradit Med Form. 2019;25:201-206.
- [Google Scholar]
- Effects of Citrus aurantium L. On interstitial cells of Cajal (ICCs) and SCF/c-Kit signaling pathway in the antrum of rats with functional dyspepsia. Chi J Tradit Chin Med Pharm. 2018;33:2304-2309.
- [Google Scholar]
- Isolation and identification of phenols compounds in Aurantii Fructus Immaturus. China Pharma. 2020;31:1040-1043.
- [Google Scholar]
- Bitter orange (Citrus aurantium L.) extract subchronic 90-day safety study in rats. Toxicol Rep. 2017;4:598-613.
- [Google Scholar]
- Supplementation of houttuynia cordata or ponccirus trifoliate on lymphocyte transformation of grass carp. Fisheries Sci. 2005;24:4-6.
- [Google Scholar]
- Citrus ichangensis peel extract exhibits anti-metabolic disorder effects by the inhibition of PPARγ and LXR signaling in high-fat diet-induced C57BL/6 mouse. Evid-Based Compl Alt. 2012;2012:678592
- [Google Scholar]
- Investigation and quality analysis of Aurantii Fructus Strains in Jiangxi. Chin J Chin Mater Med. 1986;11:267-268.
- [Google Scholar]
- Effects of sinensetin on proliferation and apoptosis of human gastric cancer AGS cells. Chin J Chin Mater Med. 2011;36:790-794.
- [Google Scholar]
- Study on discrimination of decoction pieces from rutaceae by HS-GC-MS. J Chin Med Mater 2022:2920-2925.
- [Google Scholar]
- Characterization and classification of seven Citrus herbs by liquid chromatography-quadrupole time-of-flight mass spectrometry and genetic algorithm optimized support vector machines. J Chromatogr A. 2014;1339:118-127.
- [Google Scholar]
- Genotype and harvest time influence the phytochemical quality of fino lemon juice (Citrus limon (L.) Burm. F.) for industrial use. J Agr Food Chem. 2008;56:1669-1675.
- [Google Scholar]
- Effect of Citrus Aurantium extract on L-type calcium currents in ventricular myocytes of single guinea pigs. J Central South Univ (Med Sci). 2003;28:353-356.
- [Google Scholar]
- Effect of Jiuxinfumai injection on L-type calcium currents in single guinea pig ventricular myocytes. J Central South Univ (Med Sci). 2004;29:521-524.
- [Google Scholar]
- Studies on chemical constituents of Citrus aurantium L. Asia-Pacific Tradit Med. 2012;8:22-24.
- [Google Scholar]
- Application of fingerprint combined with QAMS in quality evaluation of Aurantii Immaturus Fructus. Chin Tradit Herbal Drugs. 2020;51:2548-2556.
- [Google Scholar]
- Aurantii Fructus: A systematic review of ethnopharmacology, phytochemistry and pharmacology. Phychem Rev. 2020;20:909-944.
- [Google Scholar]
- Suitability evaluation of main citrus varieties for juice processing in Hunan province. Food & Ferment Indust. 2022;48:112-121.
- [Google Scholar]
- Distinguishing citrus varieties based on genetic and compositional analyses. PLOS ONE. 2022;17:e267007.
- [Google Scholar]
- Pharmacological study of N-methyltyramine, an effective component of Zhishi injection to raise blood pressure. Chin Tradit Herbal. Drugs. 1978;64
- [Google Scholar]
- P-Synephrine exhibits anti-adipogenic activity by activating the Akt/GSK3β signaling pathway in 3T3-L1 adipocytes. J Food Biochem. 2019;43:e13033.
- [Google Scholar]
- A strategy to discover selective α-glucosidase/acetylcholinesterase inhibitors from five function-similar citrus herbs through LC-Q-TOF-MS, bioassay and virtual screening. J Chromatogr B. 2021;1174:122722
- [Google Scholar]
- Effects of Citrus aurantium on the health of Litopenaeus Vannamei. J Hydroecology. 2005;25:99-101.
- [Google Scholar]
- Discussion on the warm properties of Zhishi. J Pract Tradit Chin Inter Med. 2001;15:28.
- [Google Scholar]
- Citrus aurantium and synephrine alkaloids in the treatment of overweight and obesity: An update. Obes Rev. 2006;7:79-88.
- [Google Scholar]
- Lipid lowing activity of Ponciri Fructus and Aurantii Fructus Immaturus on hyperlipemia rats induced by triton WR-1339. Kor J Herbology. 2007;22:109-116.
- [Google Scholar]
- Citrus aurantium L. Exhibits apoptotic effects on U937 human leukemia cells partly through inhibition of Akt. Int J Oncol. 2012;40:2090-2096.
- [Google Scholar]
- Study on the compounds of polymethoxylated flavonoids in Fructus Aurantii Immaturus. Lishizhen Med Mater Med Res. 2010;21:2469-2470.
- [Google Scholar]
- Developmental toxicity of Citrus aurantium in rats. Birth Defects Res B Dev Reprod Toxicol. 2011;92:216-223.
- [Google Scholar]
- Antimicrobial activity of peels and physicochemical properties of juice prepared from indigenous citrus fruits of Sylhet region. Bangladesh. Heliyon. 2022;8:e9948.
- [Google Scholar]
- He, M.J., Yang, J.X., Wei, Y., and AN, J. 2013. Effects of Fructus Aurantii Immaturus volatile oil on slow transit constipation in rats. Chi J Tradit Chin Med Pharm 28, 1487-1491.
- Simultaneous quantitative analysis of ten bioactive flavonoids in Citri Reticulatae Pericarpium Viride (Qing Pi) by ultrahigh-performance liquid chromatography and high-resolution mass spectrometry combined with chemometric methods. Phytochem Analysis. 2021;32:1152-1161.
- [Google Scholar]
- Citrus aurantium L. And its flavonoids regulate TNBS-induced inflammatory bowel disease through anti-inflammation and suppressing isolated jejunum contraction. Int J Mol Sci. 2018;19:3057.
- [Google Scholar]
- Synergistic enhancement and hepatoprotective effect of combination of total phenolic extracts of Citrus aurantium L. And methotrexate for treatment of rheumatoid arthritis. Phytother Res. 2018;33:1122-1133.
- [Google Scholar]
- Characterization and identification of volatile components in Citrus aurantium L. and Citrus sinensis Osbeck. based on headspace-gas chromatography-ion mobility spectrometry. Journal of Food Safety and Quality. 2022;13:7227-7235.
- [Google Scholar]
- Anti-ulcer effects of Aurantii Fructus Immaturus and Aurantii Fructus and their anti-ulcer active ingredients. Nat Med. 1997;51:190-193.
- [Google Scholar]
- Study on the mechanism of Aurantii Fructus Immaturus and its main active ingredients in promoting gastrointestinal motility of model rats with spleen deficiency. China Pharm. 2017;28:1747-1750.
- [Google Scholar]
- The changes of the origin and authentic production of Aurantii Fructus Immaturus and Aurantii Fructus. J Chin Med Mater. 2019;42:686-689.
- [Google Scholar]
- Effects of volatile oil of Aurantii Fructus and Aurantii Fructus Immaturus on gastrointestinal tract of animals. Jiangxi Med J. 1992;27:158-159.
- [Google Scholar]
- A comparison of pharmacological activities of effective constituents in Zhishi (Citrus aurantium) Chin Tradit Herbal Drugs. 1994;25:419-421.
- [Google Scholar]
- Study on the contents of naringin in Fructus Aurantii Immatures of different specifications. Tradit Chin Drug Res & Clin Pharm. 2008;19:130-131.
- [Google Scholar]
- Influence of Fructus Aurantii Immaturus flavonoids on gastrointestinal motility in rats with functional dyspepsia. Tradit Chin Drug Res & Clin Pharm. 2012;23:612-615.
- [Google Scholar]
- Studies on effect of Citrus Aurantium to the contraction of isolated mouse intestine. J Chin Med Univ. 1993;22:49-52.
- [Google Scholar]
- Effect of atropine on the enhancing action of Fructus Aurantii Immaturus on the myoelectric activity of small intestine in dogs. Chin J Integra Tradit West Med. 1996;16:292-294.
- [Google Scholar]
- Determination of organochlorine pesticide residues in Citrus aurantium and its extracts by capillary gas chromatography. Chin Tradit Herbal Drugs. 2004;35:1368-1370.
- [Google Scholar]
- Hu'Nan Medicine Industry Research Institute, F.R. 1976. A brief report on the chemical research of the elevate blood pressure components of Aurantii Fructus Immaturus. Chin Tradit Herbal Drugs, 6-10.
- Different origin of Citrus aurantium raw herbs and their extracts infrared spectroscopy. Chin J Exp Traditl Med Form. 2013;19:127-131.
- [Google Scholar]
- Metabolic and biological profile of autochthonous Vitis vinifera L. Ecotypes. Food Funct. 2015;6:1526-1538.
- [Google Scholar]
- Phytotherapeutic effects of the fruits of Poncirus trifoliata (L.) Raf. On cancer, inflammation, and digestive dysfunction. Phytother Res. 2018;32:616-624.
- [Google Scholar]
- Novel triterpenoid from Citrus aurantium L. Possesses chemopreventive properties against human colon cancer cells. Bioorgan Med Chem. 2008;16:5939-5951.
- [Google Scholar]
- Effect of immature bitter orange on model rats of blood stasis in prethrombotic state. Shandong J Tradit Chin Med. 2003;22:360-362.
- [Google Scholar]
- Study on the effect of immature bitter orange capsule on the risk factors of pre-thrombotic state in acute coronary syndrome patients. J Tradit Chin Med. 2008;49:1087-1089.
- [Google Scholar]
- Effect of Citrus aurantium and its active ingredients on the contractility and autoregulation of the papillary muscle of the cat. Chin Sci Bull 1980:522-525.
- [Google Scholar]
- Hypoglycemic and hypolipidemic effects of neohesperidin derived from Citrus aurantium L. In diabetic KK-Ay mice. Food Funct. 2015;6:878-886.
- [Google Scholar]
- Flavonoids of Fructus aurantii processed using the Zhangband system. Biomed Res. 2017;28:4386-4389.
- [Google Scholar]
- Determination of a new chromone from Aurantii Fructus Immaturus by DFT/GIAO method. Nat Prod Res. 2016;30:69-73.
- [Google Scholar]
- Effect of Si Ni San and its individual component drugs on experimental liver injury. Chin J Nat Med. 2004;2:45-49.
- [Google Scholar]
- Effects of Citrus aurantium extract on liver antioxidant defense function in experimental diabetic mouse. J Hygiene Res. 2007;36:689-692.
- [Google Scholar]
- Study on the antioxidant effect of Citrus aurantium extract in vitro. J Chin Med Mater. 2008;31:113-116.
- [Google Scholar]
- Effects of Citrus aurantium on kidney antioxidant function and islet in experimental diabetic mice. Chin J Public Health. 2009;25:662-664.
- [Google Scholar]
- Genetic diversity and domestication footprints of Chinese Cherry [Cerasus pseudocerasus (Lindl.) G.Don] as revealed by nuclear microsatellites. Front. Plant Sci. 2018;9:238.
- [Google Scholar]
- Effects of Ponciri Fructus and Aurantii Fructus Immaturus on the gastric fundus motility. J Vet Clin. 2013;30:27-31.
- [Google Scholar]
- Clinical study for the efficacy of external preparation containing Sophorae Radix, Aurantii Immaturus Fructus, Schizonepetae Spica, Forsythia fructus and Portulacea Herba Extract on atopic dermatitis. J Korea Ophthalm & Otolaryng & Dermat. 2014;27:17-36.
- [Google Scholar]
- Antioxidative and anti-inflammatory effects of Aurantii Fructus Immaturus pharmacopuncture. Korea J Acupunt. 2010;27:13-24.
- [Google Scholar]
- Citrus aurantium flavonoids inhibit adipogenesis through the Akt signaling pathway in 3T3-L1 cells. Bmc Complem Altern M. 2012;12:31.
- [Google Scholar]
- Suppressive effect of flavonoids from Korean Citrus aurantium L. On the expression of inflammatory mediators in L6 skeletal muscle cells. Phytother Res. 2012;26:1904-1912.
- [Google Scholar]
- The braine-gut pathway in functional gastrointestinal disorders is bidirectional: A 12-year prospective population-based study. Gut. 2014;61:1284-1290.
- [Google Scholar]
- Citrus aurantium L. Polymethoxyflavones promote thermogenesis of brown and white adipose tissue in high-fat diet induced C57BL/6J mice. J Funct Foods. 2020;67:103860
- [Google Scholar]
- Lingonberry (Vaccinium vitis-idaea L.) fruit as a source of bioactive compounds with health-promoting effects-a review. Int J Mol Sci. 2021;22:5126.
- [Google Scholar]
- Effect of Zhishi on the electrical activity of the small intestine in sheep. Res of TCM. 1997;13:49-50.
- [Google Scholar]
- Laser-induced fluorescence spectroscopy (LIFS) for discrimination of genetically close sweet orange accessions (Citrus sinensis L. Osbeck) Appl Spectrosc. 2017;71:203-214.
- [Google Scholar]
- Hesperidin, a citrus flavonoid, protects against l-methionine-induced hyperhomocysteinemia by abrogation of oxidative stress, endothelial dysfunction and neurotoxicity in wistar rats. Pharm Biol. 2017;55:146-155.
- [Google Scholar]
- Obesity-Related Hypertension: Pathogenesis, Cardiovascular Risk, and Treatment. J Clin Hypertens. 2013;15:14-33.
- [Google Scholar]
- Sanguinello and Tarocco (Citrus sinensis [L.] Osbeck): Bioactive compounds and colour appearance of blood oranges. Food Chem. 2019;270:395-402.
- [Google Scholar]
- Ameliorating effect of Citrus aurantium extracts and nobiletin on β-amyloid (1–42)-induced memory impairment in mice. Mol Med Rep. 2019;20:3448-3455.
- [Google Scholar]
- Flavonoids of Korean Citrus aurantium L. Induce apoptosis via intrinsic pathway in human hepatoblastoma HepG2 Cells. Phytother Res. 2015;29:1940-1949.
- [Google Scholar]
- Determination of Citrus aurantium based on near-infrared spectroscopy. Nat Prod Res Dev. 2015;27:1199-1204.
- [Google Scholar]
- Identification of the authenticity of the traditional Chinese medicine Zhishi. Res Practice Chin Med. 2002;16:36-37.
- [Google Scholar]
- Comparative pharmacokinetics of triterpenic acids in normal and immunosuppressed rats after oral administration of Jujubae Fructus extract by UPLC-MS/MS. J Chromatogr B. 2018;1077–1078:13-21.
- [Google Scholar]
- Actions of immature orange fruit and dried tangerine peel on the tension of isolated aortic muscle strips. Chin Tradit Patent Med. 2001;23:658-660.
- [Google Scholar]
- Effect of drug-containing serum of immature bitter orange on cell contraction, intracellular Ca2+ concentration and calmodulin expression in rat antral smooth muscle cells. World Chin J Digest. 2015;23:1224-1230.
- [Google Scholar]
- Herbal textual research on ZhiShi and ZhiQiao. Wset J Tradit Chin Med. 2015;28:36-38.
- [Google Scholar]
- Application of chemometric algorithms in the high-performance thin-layer chromatography fingerprint of traditional chinese medicines. J Aoac Int. 2019;102:720-725.
- [Google Scholar]
- Effect of extracts from Citrus aurantium on gastrointestinal function of mouse. Food & Machinery 2007:52-54.
- [Google Scholar]
- Comparison of Aurantii Fructus Immaturus and Aurantii Fructus based on multiple chromatographic analysis and chemometrics methods. J Chromatogr A. 2016;1469:96-107.
- [Google Scholar]
- Effect of total flavonoid glycosides from Fructus Aurantii Immaturus on visceral hypersensitivity in rats with functional dyspepsia. J Beijing Univ Tradit Chin Med. 2016;39:1027-1032.
- [Google Scholar]
- Pharmacodynamics study of total flavonoids from Fructus Aurantii Immaturus for treatment of irritable bowel syndrome in rats with liver stasis and spleen deficiency syndrome. Tradit Chin Drug Res & Clin Pharm. 2014;25:576-581.
- [Google Scholar]
- Protective effect of immature bitter orange (Citrus aurantium L.) flavonoids extracts on PC12 cell injury induced by 6-hydroxydopamine. Hunan Agr Sci; 2021. p. :40-44.
- A comparison between Zhishi Xiaopiwan and cisapride in treatment of functional dyspepsia. World J Gastroenterol. 1998;4:544-547.
- [Google Scholar]
- The correlation between diameter and flavonoids contents in the dry fruit of Citrus aurantium. J Chin Med Mater 2022:2912-2919.
- [Google Scholar]
- Effect of the wall-broken spore power of Aurantii Fructus Immaturus on the intestinal transit and acute toxicity test in mice. J Guangdong Pharm Univ. 2012;4:435-437.
- [Google Scholar]
- Effect of Zhishi on IL-6, SCF and MTL in rats with functional dyspepsia. Shanxi J TCM. 2020;36:52-54.
- [Google Scholar]
- Effects of epoxyauraptene on smooth muscle myosin and expression of myosin light chain kinase. Chin Tradit Patent Med. 2012;34:1852-1856.
- [Google Scholar]
- Effect of raw and stir-frying with bran Fructus Aurantii Immaturus on gut hormone of spleen deficiency rats induced by reserpine. Guiding J of TCM and Pha. 2012;18:11-13.
- [Google Scholar]
- Effect of epoxyaurapten from Aurantii Fructus Immaturus on treating ulcerative colitis. Chin J Exp Tradit Med Form. 2015;21:102-105.
- [Google Scholar]
- Metabolic profile and underlying improved bioactivity of Fructus aurantii immaturus by human intestinal bacteria. Food Funct. 2017;8:2193-2201.
- [Google Scholar]
- Rapid analysis of Aurantii Fructus Immaturus (Zhishi) using paper spray ionization mass spectrometry. J Pharmaceut Biomed Anal. 2017;137:204-212.
- [Google Scholar]
- Naturally occurring TPE-CA maintains gut microbiota and bile acids homeostasis via FXR signaling modulation of the liver-gut axis. Front Pharmacol. 2020;11:12.
- [Google Scholar]
- Preliminary study on hydroxyl free radical scavenging activity and antimierobial effect of Fructus Aurantii lmmaturus extraets. Food Sci. 2009;30:123-126.
- [Google Scholar]
- Simultaneous determination of naringin and hesperidin in Zhishi Daozhi Pills by HPLC. China Pharm. 2010;19:43-44.
- [Google Scholar]
- Contrast analysis of chemical constitutes in volatile oil of Citrus aurantium and Citrus sinensis from chongqing. Chin J Exp Tradit Med Form. 2011;17:45-48.
- [Google Scholar]
- Chemical evidence for potent xanthine oxidase inhibitory activity of ethyl acetate extract of Citrus aurantium L. Dried immature fruits. Molecules. 2016;21:302.
- [Google Scholar]
- The regulation of histamine receptor antagonist in the effect of Fructus Aurantii Immaturus on the motor activity of small intestine in mice. J Southeast Univ (Med Sci). 2001;20:144-146.
- [Google Scholar]
- Studies on constituents of Rutaceae plants growing in China comparied with those in Korea. Chin Tradit Herbal Drugs. 2003;34:586-589.
- [Google Scholar]
- Inhibitory effect of hesperetin and naringenin on human UDP-glucuronosyltransferase enzymes: Implications for herb-drug interactions. Biol Pharm Bull. 2016;39:2052-2059.
- [Google Scholar]
- Effects of Zhishi (Fructus Aurantii Immaturus) and its medicinal serum on extracorporeal colonic muscle strips in rats with slow transit constipation. J Beijing Univ Tradit Chin Med. 2010;33:402-405.
- [Google Scholar]
- RAPD analysis of the germplasm resources of Fructus aurantii of Jiangxi. J Jiangxi Uni TCM. 2005;17:55-58.
- [Google Scholar]
- Preliminary discussion on the warm property of Zhishi. Jilin J Chin Med 1988:29-30.
- [Google Scholar]
- Disscussion on the change of nature andtaste of Zhishi and Zhiqiao varieties. Chi J Chin Mater Med. 1993;18:178-179.
- [Google Scholar]
- Meranzin hydrate elicits antidepressant effects and restores reward circuitry. Behav Brain Res. 2021;398:112898
- [Google Scholar]
- Chemical constituents of flavonoids from Citrus aurantium var. Amara. J Chin Med Mater. 2019;42:2565-2569.
- [Google Scholar]
- Identification of the Flavonoid Compounds and evaluation of the anti-oxidant and preventing obesity effects of Citrange (Citrus sinensis x Poncirus trifoliata) Fruits extracts. Southwest University; 2015.
- Determination of the content of synephrine in Fructus Aurantii Immaturus by packed-column supercritical fluid chromatography. Liaoning J Tradit Chin Med. 2006;33:222-223.
- [Google Scholar]
- Study on quality difference of Aurantii Fructus Immaturus from different origins. Chin Trait Herb Drugs. 2022;53:4493-4503.
- [Google Scholar]
- ISSR analysis on the genetic diversity of Citrus aurantium's main current species in the genuine producing region. Acta Agri Uni Jiangxi. 2007;29:124-128.
- [Google Scholar]
- Fingerprint for different cultural species of Aurantii Fructus by HPLC. Chin Tradit Herbal Drugs. 2008;39:430-432.
- [Google Scholar]
- Assessment of candidate plant DNA barcodes using the Rutaceae family. Sci Chin Life Sci. 2010;53:701-708.
- [Google Scholar]
- Effects on icam-1 expression and releasing of huvec induced by ox-ldl of Aurantii Fructus Immaturus extract and its active components. Progress Moder Biomed. 2012;12:6228-6233.
- [Google Scholar]
- Citrus fruits as a treasure trove of active natural metabolites that potentially provide benefits for human health. Chem Cent J. 2015;9:68.
- [Google Scholar]
- Inhibitory effects of nobiletin on hepatocellular carcinoma in vitro and in vivo. Phytother Res. 2013;28:560-567.
- [Google Scholar]
- Discrimination of genetically very close accessions of sweet orange (Citrus sinensis L. Osbeck) by laser-induced breakdown spectroscopy (LIBS) Molecules. 2021;26:3092.
- [Google Scholar]
- Utility of indels for species-level identification of a biologically complex plant group: a study with intergenic spacer in Citrus. MOL BIOL REP. 2014;41:7217-7222.
- [Google Scholar]
- Biologically active compounds in Citrus fruits peels. J Syn Organ Chem Associat. 1994;52:318-327.
- [Google Scholar]
- Structures of new cyclic peptides in young unshiu (Citrus unshiu Marcov.), orange (Citrus sinensis Osbeck.) and amanastu (Cirtus natsudaidai) peelings. Agr Biol Chem. 1991;55:2923-2929.
- [Google Scholar]
- HPLC-PDA-MS and NMR characterization of a hydroalcoholic extract of Citrus aurantium L. Var.amara peel with antiedematogenic activity. J Agr Food Chem. 2013;61:1686-1693.
- [Google Scholar]
- Isopropylnorsynephrine is a stronger lipolytic agent in human adipocytes than synephrine and other amines present in Citrus aurantium. J Physiol Biochem. 2011;67:443-452.
- [Google Scholar]
- Activity antifungal of the essential oils; Aqueous and ethanol extracts from Citrus aurantium L. Nat Prod Res. 2015;29:2238-2241.
- [Google Scholar]
- Citrus essential oils and four enantiomeric pinenes against Culex pipiens (Diptera: Culicidae) Parasitol Res. 2009;105:769-773.
- [Google Scholar]
- Role of Citrus medica L. Fruits extract in combatting the hematological and hepatic toxic effects of carbofuran. Chem Res Toxicol. 2021;34:1890-1902.
- [Google Scholar]
- National Health Commission of the People’s Republic of China, 2002. Circular of the Ministry of Health on Further Regulating the Management of Health Food Raw Materials. http://www.nhc.gov.cn/cms-search/xxgk/getManuscriptXxgk.htm?id=38057 /(accessed 11 March 2003).
- Anxiolytic effect of Citrus aurantium L. In crack users. Evid-Based Compl Alt. 2017;2017:1-8.
- [Google Scholar]
- Effect of N-methyltyramine on the regulation of adrenergic receptors via enzymatic epinephrine synthesis for the treatment of gastrointestinal disorders. Biomed Pharmacother. 2019;111:1393-1398.
- [Google Scholar]
- Extracts of immature orange (Aurantii fructus immaturus) and citrus unshiu peel (Citri unshiu pericarpium) induce P-glycoprotein and cytochrome P450 3A4 expression via upregulation of pregnane X receptor. Front Pharmacol. 2017;8:84.
- [Google Scholar]
- Antithrombotic effect of 11 kinds of traditional Chinese medicine including Citrus aurantium in vitro. Chin J Integra Tradit West Med. 1989;9:358-359.
- [Google Scholar]
- Study on the contents of synephrine in raw Fructus Aurantii Immaturus and its processed products. China Pharm. 2005;16:395-397.
- [Google Scholar]
- Park, A., Yang, Y., Lee, Y., Jung, H., Kim, T.et al., 2022. Aurantii Fructus Immaturus enhances natural killer cytolytic activity and anticancer efficacy in vitro and in vivo. Frontiers in Medicine 9.
- Pediatric Septic Shock Research Cooperative Group, H.P. 1981. Injectio Fructus Aurantii Immaturus in 100 pediatric septic shock cases. Chin Med J-Peking 94, 523-528.
- Studies on the active constituents of Aurantii Fructus Immaturus. J Nanjing Tcm Univ (Nat. 2001;Sci)17:91-92.
- [Google Scholar]
- Analysis of differential chemical compositions of Aurantii Fructus Immaturus before and after stir-frying with bran and chemical compositions of wheat bran after processing by UPLC-Q-TOF-MS. Chin J Exp Tradit Med Form. 2020;26:144-152.
- [Google Scholar]
- Determination of flavonoids in Fructus Aurantii Immaturus and Fructus Aurantii by capillary electrophoresis with electrochemical detection. J Inst Anal. 2007;26:694-697.
- [Google Scholar]
- Molecular characterization of sour orange (Citrus aurantium) accessions and their relatives using SSR and SRAP markers. Genetics and molecular research. 2012;11:3267-3276.
- [Google Scholar]
- Effects of the essential oil from Citrus aurantium L. In experimental anxiety models in mice. Life Sci. 2005;78:1720-1725.
- [Google Scholar]
- An HPLC fingerprint research on Citrus aurantium based on chemical pattern recognition technology. J Inst Anal. 2021;40:72-78.
- [Google Scholar]
- Citrus aurantium L. Dry extracts promote C/ebpβ expression and improve adipocyte differentiation in 3T3-L1 cells. Plos One. 2018;13:e193704.
- [Google Scholar]
- Efficacy of different citrus essential oils to inhibit the growth and B1 aflatoxin biosynthesis of Aspergillus flavus. Environ Sci Pollut R. 2019;26:31263-31272.
- [Google Scholar]
- Analysis of the cytotoxic, genotoxic, mutagenic, and pro-oxidant effect of synephrine, a component of thermogenic supplements, in human hepatic cells in vitro. Toxicol. 2019;422:25-34.
- [Google Scholar]
- Synephrine: From trace concentrations to massive consumption in weight-loss. Food Chem Toxicol. 2011;49:8-16.
- [Google Scholar]
- Chemical profiling and quality evaluation of Zhishi-Xiebai-Guizhi Decoction by UPLC-Q-TOF-MS and UPLC fingerprint. J Pharmaceut Biomed. 2021;194:113771
- [Google Scholar]
- Studies on the bioactive constituents of Aurantii Fructus Immaturus. Yakugaku Zasshi. 1995;116:244-250.
- [Google Scholar]
- Structure and physiological activity of phenyl propanoid glycosides of Hassaku (Citrus hassaku HORT.) and Orange (Citrus sinensis Osbeck.) peelings. Nippon Nogeikagaku Kaishi. 1986;60:593-599.
- [Google Scholar]
- Toxicological effects of a mixture used in weight loss products: p-synephrine associated with ephedrine, salicin, and caffeine. Int J Toxicol. 2012;31:184-191.
- [Google Scholar]
- Effect of acute administration of an herbal preparation on blood pressure and heart rate in humans. Int J Med Sci. 2011;8:192-197.
- [Google Scholar]
- History and development of“One Root of Medicine and Food”. Chin Bull Life Sci. 2015;27:1061-1069.
- [Google Scholar]
- Safety evaluation of p-synephrine following 15 days of oral administration to healthy subjects: A clinical study. Phytother Res. 2018;32:125-131.
- [Google Scholar]
- Citrus aurantium L. Var. Amara Engl. Inhibited lipid accumulation in 3T3-L1 cells and Caenorhabditis elegans and prevented obesity in high-fat diet-fed mice. Pharmcol Res. 2019;147:104347
- [Google Scholar]
- Influence of Citrus Aurantium on calcium in the contraction of isolated mouse intestine muscle. J Chin Med Univ. 1994;23:12-15.
- [Google Scholar]
- Comparison between peel and pulp of Aurantii Fructus Immaturus by UPLC fingerprint and multicomponent quantitative analysis. Chi J Chin Mater Med. 2021;46:4446-4455.
- [Google Scholar]
- Quality control of standard decoction of Aurantii Fructus Immaturus. Chin J Exp Tradit Med Form. 2019;25:153-159.
- [Google Scholar]
- The determination on limonin in fructus aurantii immaturus and fructus aurantii from different areas. Chin Food Add 2011:126-129.
- [Google Scholar]
- Hepatoprotective effect of Citrus aurantium L. Against APAP-induced Liver Injury by regulating liver lipid metabolism and apoptosis. Int J Biol Sci. 2020;16:752-765.
- [Google Scholar]
- Flavonoid composition of Tarocco (Citrus sinensis L. Osbeck) clone “Lempso” and fast antioxidant activity screening by DPPH-UHPLC-PDA-IT-TOF. Phytochem Anal. 2017;28:521-528.
- [Google Scholar]
- Simultaneous determination of eight components in two kinds of Zhizhu pills using double-wavelength HPLC. Chin J Pharm Anal. 2016;36:1715-1721.
- [Google Scholar]
- Anti-inflammatory activities of isogosferol, a furanocoumarin isolated from Citrus junos seed shells through bioactivity-guided fractionation. Molecules. 2019;24:4088.
- [Google Scholar]
- The species differences of flavonoids in Fructus Aurantii Immaturus and their infrared spectrum relevance. Chin J Hospital Pharm. 2009;29:1547-1549.
- [Google Scholar]
- Mechanism of hypothalamic effect in small intestine electro-activity of rats regulated by Fructus Aurantii Immatures. Chin J Integra Tradit West Med. 2006;26:55-58.
- [Google Scholar]
- Oxidative stress-related lung dysfunction by chromium(VI): Alleviation by Citrus aurantium L. J Physiol Biochem. 2013;69:239-253.
- [Google Scholar]
- Assessment of the adverse event reports associated with Citrus aurantium (bitter orange) from April 2004 to October 2009. J Funct Foods. 2010;2:235-238.
- [Google Scholar]
- Safety, efficacy, and mechanistic studies regarding Citrus aurantium (Bitter Orange) extract and p-synephrine. Phytother Res. 2017;31:1463-1474.
- [Google Scholar]
- A Review of Natural Stimulant and Non-stimulant Thermogenic Agents. Phytother Res. 2016;30:732-740.
- [Google Scholar]
- The safety of Citrus aurantium (bitter orange) and its primary protoalkaloid p-synephrine. Phytother Res. 2011;25:1421-1428.
- [Google Scholar]
- A review of the receptor-binding properties of p-synephrine as related to its pharmacological effects. Oxid Med Cell Longev. 2011;2011:482973
- [Google Scholar]
- Review of Published Bitter Orange Extract and p-Synephrine Adverse Event Clinical Study Case Reports. J Diet Suppl. 2020;17:355-363.
- [Google Scholar]
- p-Synephrine, ephedrine, p-octopamine and m-synephrine: Comparative mechanistic, physiological and pharmacological properties. Phytother Res. 2020;34:1838-1846.
- [Google Scholar]
- A new strategy employed for identification of sweet orange cultivars with RAPD markers. Genet Mol Res. 2012;11:2071-2080.
- [Google Scholar]
- Pharmacological profile of gastric mucosal protection by marmin and nobiletin from a traditional herbal medicine. Aurantii Fructus Immaturus. Jpne J Pharm. 1994;1:66.
- [Google Scholar]
- Study on the effect on rabbit's vaginal smooth muscle contract in vitro with the challenges of Fructus Aurantii immaturus (FAI) or Radix Sophorae flavescentis (RSF) Lishizhen Med Mater Med. 2001;12:973-974.
- [Google Scholar]
- Comparision of the content of total alkaloids and aetherolea in different species of Aurantii Fructus Immaturus. Cuiding J Tradit Chin Med Pharm. 2008;14:112-114.
- [Google Scholar]
- The protective effect of Citrus aurantium on gastric tissue in the acute stage of cerebral infarction. Chin J Inter Med on Cardio-Cerebrovascular Disease. 2014;12:1255-1256.
- [Google Scholar]
- Determination of octopamine, synephrine and tyramine in Citrus herbs by ionic liquid improved 'green' chromatography. J Chromatogr A. 2006;1125:182-188.
- [Google Scholar]
- Regulatory effects of Citrus Aurantium (Zhishi) on gastrin and motilin in acute stage of cerebral infarction rats. Chin J Tradit Med Sci Tech. 2015;22:39-40.
- [Google Scholar]
- The levels of bioactive ingredients in Citrus aurantium L. At different harvest periods and antioxidant effects on H2O2-induced RIN-m5F cells. J Sci Food Agr. 2021;101:1479-1490.
- [Google Scholar]
- Effect of Zhishi on intestinal neurotransmitters in rats with slow-transmission constipation SP, VIP. Informa Tradit Chin Med. 2011;28:14-15.
- [Google Scholar]
- Efficacy of Zhizhuwan decoction and its ingredient-contained serums on proliferation and apoptosis of rat colon interstitial cells of Cajal. Chin J Exp Tradit Med Form. 2020;26:120-126.
- [Google Scholar]
- Online extraction-high performance liquid chromatography–diode array detector-quadrupole time-of-flight tandem mass spectrometry for rapid flavonoid profiling of Fructus aurantii immaturus. J Chromatogr B. 2018;1077–1078:1-6.
- [Google Scholar]
- Identification and quality control of Aurantii Immaturus Fructus and misused related species. Chin Med Univ; 2013.
- Analysis on the source and flow of the use of Aurantii Fructus Immaturus and Aurantii Fructus. Jiangxi J Tradit Chin Med 1985:47-48.
- [Google Scholar]
- Studies on extraction, separation, identification and biological ablity of flavonoids from Fructus aurantii immaturus. Zhejiang Gongshang Univ; 2008. p. :p..
- Study of evaluation on resources and materia madica of Fructus Aurantii in Zhejiang province. J Zhejiang Chine Med Univ. 2009;34:603-604.
- [Google Scholar]
- A system for screening agonists targeting β2-adrenoceptor from Chinese medicinal herbs. J Zhejiang Univ-Sc B. 2009;10:243-250.
- [Google Scholar]
- Effect and mechanism of drug-containing serum of Zhishi (immature bitter orange) on endoplasmic reticulum stress injury in rat gastric interstitial cells of cajal. Shandong Med J. 2018;58:34-37.
- [Google Scholar]
- Mechanism of aqueous fructus aurantii immaturus extracts in neuroplexus of cathartic colons. World J Gastroenterol. 2015;21:9366-9538.
- [Google Scholar]
- Study on quantitative determination of total flavonoid in Qingpi, Zhiqiao and Zhishi by TLC-densitometric method. Chi J Chin Mater Med. 1989;14:38-41.
- [Google Scholar]
- Chemical composition and clinical application of Daidaihua. Acta Chin Med. 2009;32:276-278.
- [Google Scholar]
- Determination of flavonoids in Fructus Aurantii Immaturus by RRLC. Chin J Exp Tradit Med Form. 2009;15:1-3.
- [Google Scholar]
- Structural characterization and antioxidant activities of polysaccharides from Citrus aurantium L. Int J Biol Macromol. 2014;67:112-123.
- [Google Scholar]
- Study on the effect of fructus aurantii immaturus on myoelectric activity of gastrointestinal tract in rats. J Southeast Univ (Med Sci). 2001;20:153-154.
- [Google Scholar]
- Determination of synephrine and N-methyltyramine in Zhishi (Zhiqiao) by thin-layer scanning. Chin J Pharm Anal. 1994;14:48-50.
- [Google Scholar]
- Study on screening cardioprotective components from Zhishi Xiebai Guizhi Tang. Chin J Chin Mater Med. 2013;38:1601-1605.
- [Google Scholar]
- Antihyperuricemic and anti-gouty arthritis activities of Aurantii fructus immaturus carbonisata-derived carbon dots. Nanomedicine (Lond.). 2019;14:2925-2939.
- [Google Scholar]
- Flora reipublicae popularis sinicae. Beijing: Science Press; 2004.
- Application of electronic tongue in the source identification of Aurantii Fructus Immaturus. Chin Pharm J. 2012;47:808-812.
- [Google Scholar]
- The influence of Fructus Aurantii Immaturus on blood lipids and vascular endothelial function in patients with hyperlipidemia. Chin J Integra Med Cardio-Cerebrovascular Disease. 2012;10:283-285.
- [Google Scholar]
- To compare the efficacy of two kinds of Zhizhu pills in the treatment of functional dyspepsia of spleen-deficiency and qi-stagnation syndrome: A randomized group sequential comparative trial. Bmc Gastroenterol. 2011;11:81.
- [Google Scholar]
- Simultaneous qualitative and quantitative analysis of 10 bioactive flavonoids in Aurantii Fructus Immaturus (Zhishi) by ultrahigh-performance liquid chromatography and high-resolution tandem mass spectrometry combined with chemometric methods. Phytochem Anal. 2022;33:710-721.
- [Google Scholar]
- Flavonoids variations in five medicinal materials of genus Citrus. Chin Tradit Patent Med. 2022;44:3225-3228.
- [Google Scholar]
- Textual research on the clinical application of modern Chinese medicine on the cold and warm properties of Fructus Aurantii Immaturus. Lishizhen Med Mater Med Res. 2008;19:3077-3079.
- [Google Scholar]
- Sequencing of diverse mandarin, pummelo and orange genomes reveals complex history of admixture during citrus domestication. Nat Biotechnol. 2014;32:656-662.
- [Google Scholar]
- Effect of Aurantii Fructus Immaturus flavonoid on the contraction of isolated gastric smooth muscle strips in rats. Evid-Based Compl Alt. 2016;2016:5616905.
- [Google Scholar]
- Effects of Aurantii Fructus Immatures flavonoid on the proximal gastric accommodation of functional dyspepsia rats. Chin J Integra Tradit West Med Digest. 2016;24:188-193.
- [Google Scholar]
- XiangYa School Of Medicine, C. 1978. Pharmacological study on the raising blood pressure of Citrus aurantium. Chin Sci Bull, 58-62.
- Research on duration and changes of Zhi Shi and Zhi Qiao as ancient and present drugs. Chin J Integra Med Cardio-Cerebrovascular Disease 1991:19-22.
- [Google Scholar]
- Antioxidation in vitro of flavonoids from Fructus aurantii immatures. Chem Ind Forest Products. 2009;29:33-36.
- [Google Scholar]
- Effect of Fructus Aurantii Immaturus on contractile activity of colonic muscle strips in rats. J Shandong Med Univ. 2001;39:437-438.
- [Google Scholar]
- A new phenolic glycoside and two new monoterpenoid furocoumarins from Aurantii Fructus Immaturus. Nat Prod Res. 2016;30:1571-1576.
- [Google Scholar]
- Characteristics of nobiletin-induced effects on jejunal contractility. Fitoterapia. 2014;94:1-9.
- [Google Scholar]
- Pharmacology of traditional Chinese Medicine (third edition.). Shanghai: Shanghai Scientific & technical Publishers; 2019. p. :174. ISBN 978-7-5478-4239-3
- SiNiSan alleviates liver injury by promoting hepatic stem cell differentiation via Wnt/β-catenin signaling pathway. Phyto med. 2022;99:153969
- [Google Scholar]
- Extraction of the total flavonoids of Fructus Aurantii Immaturus and its content analysis. J Univ South Chin (Sci Tech). 2016;30:93-97.
- [Google Scholar]
- Comparative pharmacokinetics of naringin and neohesperidin after oral administration of flavonoid glycosides from Aurantii Fructus Immaturus in normal and gastrointestinal motility disorders mice. Chin Herbal Med. 2019;11:314-320.
- [Google Scholar]
- Effect of epoxyaurapten on the function of myosin and the contractility of intestinal smooth muscle. Chin J Exp Tradit Med Form. 2012;12:170-174.
- [Google Scholar]
- The structure and function of the global citrus rhizosphere microbiome. Nat Commun. 2018;9:4894.
- [Google Scholar]
- Effects of different storage time on the quality of immature fruit of Citrus Aurantium. Eval anal drug-use hospitals Chin. 2020;20:397-402.
- [Google Scholar]
- Textual research on the materia medica of Chinese medicine Zhishi. J Chin Med Mater. 2012;35:998-1002.
- [Google Scholar]
- Pharmacological study of Aurantii Fructus and Aurantii Fructus Immaturus (third report: Comparison of the effects of Aurantii Fructus tincture and infusion with adrenaline) J Shandong Univ (Nat Sci) 1995:122-128.
- [Google Scholar]
- Research on biomarkers of different growth periods and different drying processes of Citrus wilsonii Tanaka based on plant metabolomics. Front Plant Sci. 2021;12:700367
- [Google Scholar]
- Efficacy of weikang pian in patients with functional dyspepsia: A double-blind, randomized, placebo-controlled clinical trial. Evid-Based Compl Alt. 2019;2019:4827046.
- [Google Scholar]
- Preparative isolation and purification of capsaicin and dihydrocapsaicin from Capsici Fructus using supercritical fluid extraction combined with high-speed countercurrent chromatography. J Sci Food Agr. 2018;98:2498-2506.
- [Google Scholar]
- Study of the polymethoxylated flavonoids in Fructus aurantii immaturus. Jilin Univ; 2007.
- Lemon terpene has the effect of enhancing the electrical activity of the small intestine in dogs. Basic. Clin Med. 1995;82
- [Google Scholar]
- Effect of propranolol on the small intestinal myoelectric activities increased by Fructus Aurantii Immaturns. J Southeast Univ (Med Sci). 1996;15:223-225.
- [Google Scholar]
- Comparison of volatile oils from both Fructus Aurantii and Fructus Aurantii Immaturus of Citrus wilsonii in shaanxi by GC-MS. J Food Sci Biotechn. 2010;29:219-229.
- [Google Scholar]
- Action of Zhishi on the smooth muscle of isolated small intestine in rats. J Northwest Normal Univ (Nat Sci). 1998;34:69-72.
- [Google Scholar]
- Yi, L.B., Lin, G.M., Hou, Y., Zhang, F., and Jia, T.Z. 2011. “Study on the raising blood pressure effect of Citrus aurantium L. And its processed product”. In: 2011 Annual Academic Conference of the Chinese Medicine Association of China (CMA) (ed.).).
- Yu, L.Y., Chen, M.F., Liu, J., Huang, X.Q., and He, W., et al. 2020. Systematic detection and identification of bioactive ingredients from Citrus aurantium L. Var. Amara using HPLC-Q-TOF-MS combined with a screening method. Molecules 25, 357.
- The effect of Citrus aurantium on the pharmacokinetics of omeprazole in rats. Pharm Clin Chin Mater Med. 2013;29:80-83.
- [Google Scholar]
- Textual Research on the Materia Medica of Zhiqiao. Res Practice Chin Med. 2004;18:23-24.
- [Google Scholar]
- Analysis of chemical constituents from Citrus aurantium by UHPLC-LTQ-Orbitrap-MS/MS. Chi J Chin Mater Med. 2016;41:3371-3378.
- [Google Scholar]
- Analysis on volatile oil in different processed products of Aurantii Fructus Immaturus from jiangxi by GC-MS. Chin J Exp Tradit Med Form. 2015;21:12-18.
- [Google Scholar]
- Research on discrimination of Zhishi (Aurantii Fructus Immaturus) from its commonly known adulterants based on thin layer chromatography. J Hunan Univ Chin Med. 2021;41:1534-1539.
- [Google Scholar]
- Determination of synephrine and n-methyltyramine in zhiqiao and zhishi by HPLC. Chi J Chin Mater Med. 1997;22:362-363.
- [Google Scholar]
- Study on HPLC fingerprint and chemical constituent difference of different species of Aurantii Fructus Immaturus. Chi J Chin Mater Med. 2016;41:3272-3278.
- [Google Scholar]
- Preparation and quantification of the total phenolic products in Citrus fruit using solid-phase extraction coupled with high-performance liquid chromatography with diode array and UV detection. J Sep Sci. 2016;39:3806-3817.
- [Google Scholar]
- Zhang, Y., Zhao, S., and Ye, W. 2005. “Studies on Chemical Constituents of Citrus aurantium L. ”. In: The 8th National Symposium on Traditional Chinese Medicine and Natural Medicines and the 5th National Symposium on Medicinal Plants and Phytopharmacy (ed.).).
- The effects of different varieties of Aurantii Fructus Immaturus on the potential toxicity of Zhi-Zi-Hou-Po Decoction based on spectrum-toxicity correlation analysis. Molecules. 2019;24:4254.
- [Google Scholar]
- Effect of Fructus Aurantii Immaturus decoction on defecating function of constipated mice induced by compound diphenoxylate. Inter J Tradit Chin Med. 2010;32:209-210.
- [Google Scholar]
- Bioactive constituents obtained from the fruits of Citrus aurantium. J Nat Med-Tokyo. 2019;73:146-153.
- [Google Scholar]
- The study on Fructus Aurantii Immatures consolidating the mechanism of improving gastric dysmotility in functional dyspepsia rats. Shaanxi J Tradit Chin Med. 2018;39:998-1000.
- [Google Scholar]
- Contrasting analysis on HPLC spectrum of Fructus Aurantii Immaturus formula granule with corresponding cut crude drug and decoction. Chin J Informa TCM. 2007;14:38-39.
- [Google Scholar]
- Characterization of the complete chloroplast genome of Citrus hongheensis, a key protected wild plant in Yunnan province of China. Mitochondrial Dna B. 2020;5:3514-3515.
- [Google Scholar]
- A new phenolic glycoside from fruits of Citrus aurantium. Chin Tradit Herbal Drugs 2006:1295-1297.
- [Google Scholar]
- Application of quality constant method in grade evaluation of Aurantii Fructus Immaturus pieces. Chi J Chin Mater Med. 2019;44:3268-3274.
- [Google Scholar]
- Experimental study on the effect of Zhishi IV in the treatment of sores. Hubei J Tradit Chin Med. 2004;26:52-53.
- [Google Scholar]
- Isolation and identification of chemical constituents from Aurantii Fructus Immaturus. J Shenyang Pharm Univ. 2015;32:22-25.
- [Google Scholar]
- Two new phenolic glycosides isolated from the fruits of Citrus aurantium. Chin J Nat Med. 2017;15:41-44.
- [Google Scholar]
- Antidepressant-like effect of Citrus sinensis (L.) Osbeck essential oil and its main component limonene on mice. J Agr Food Chem. 2019;67:13817-13828.
- [Google Scholar]
- A new coumarin glucoside from the fruits of Citrus aurantium. Chin J Nat Med. 2005;3:141-143.
- [Google Scholar]
- Study on the effect on rabbit isolated vaginal smooth muscle contract in vitro with the challenges of Fructus Aurantii Immaturus. Acta Med Sin. 2007;20:8-9.
- [Google Scholar]
- Protective effect of total flavonoid glycosides of Zhishi on gastric mucosa in rats with bile reflux gastritis. Chin J Clin Pharm. 2020;36:3760-3763.
- [Google Scholar]
- Differentiation of Aurantii fructus immaturus and Fructus poniciri trifoliatae immaturus by flow-injection with ultraviolet spectroscopic detection and proton nuclear magnetic resonance using partial least-squares discriminant analysis. Anal Lett. 2016;49:711-722.
- [Google Scholar]
- Analysis of HPLC fingerprints of Citrus aurantium and Citrus sinensis and the contents of naringin and synephrine. China Pharm. 2011;22:3711-3714.
- [Google Scholar]
- Effect of Zhishi decoction on the activity of rabbit isolated intestinal smooth muscle. Chin J Tradit Med Sci Tech. 2006;13:335-336.
- [Google Scholar]
- Differentiation of Aurantii Fructus Immaturus from Poniciri Trifoliatae Fructus Immaturus using flow-injection mass spectrometric (FIMS) metabolic fingerprinting method combined with chemometrics. J Pharmaceut Biomed. 2015;107:251-257.
- [Google Scholar]
- Anti-shock effects of synthetic effective compositions of Fructus Aurantii Immaturus. Chin Med J (Engl). 1989;102:91-93.
- [Google Scholar]
- Chemotaxonomic classification applied to the identification of two closely-related Citrus TCMs using UPLC-Q-TOF-MS-Based metabolomics. Molecules. 2017;22:1721.
- [Google Scholar]
- Anti-inflammatory effects of Zhishi and Zhiqiao revealed by network pharmacology integrated with molecular mechanism and metabolomics studies. Phytomedicine. 2018;50:61-72.
- [Google Scholar]
- Herbal textual research on Aurantii Fructus Immaturus and Aurantii Fructus in Chinese classical prescriptions. Modern Chin Med. 2020;22:1175-1184.
- [Google Scholar]
- Nutrient components, health benefits, and safety of litchi (Litchi chinensis Sonn.): A review. Compr Rev Food Sci F. 2020;19:2139-2163.
- [Google Scholar]
- A sensitive and practical ELISA for analyzing naringenin in pummelo and herb samples. Food Chem. 2021;362:130223
- [Google Scholar]
- P-synephrine: A novel agonist for neuromedin U2 receptor. Biol Pharm Bull. 2014;37:764-770.
- [Google Scholar]
- Action of traditional Chinese medicines of Zhishi and Baizhu on contractile activity of isolated gastric muscular strips in rats. J Gansu Sci. 1998;10:65-68.
- [Google Scholar]
- Quality evaluation of aurantii fructus based on fingerprint qualitative analysis, multi-component quantitative analysis and chemometrics. Chin J Exp Tradit Med Form. 2021;28:138-145.
- [Google Scholar]
- Discussing the identification method of the authenticity of Chinese medicine Citrus aurantium L. Guide Chin Med. 2014;12:278-279.
- [Google Scholar]
- Studies on the quality of baked Fructus Aurantii Immaturus with bran of different storage periods. Chi J Chin Mater Med. 1997;22:88-89.
- [Google Scholar]
- Differences in Zhishi between ShenNongBenCaoJing and ShangHanLun of Song dynasty. Lishizhen Med Mater Med Res. 2013;24:427-428.
- [Google Scholar]
- Comparative analysis of the contents of naringin, hesperidin and sypephrine in four kinds of Zhishi Chinese herbs. J Fujian Normal Univ (Natl Sci). 2005;21:91-93.
- [Google Scholar]
- Effect of immature bitter orange on gastric emptying in rats with functional dyspepsia. Chin J Clin Pharm. 2005;14:291-294.
- [Google Scholar]
- Relationship between the limonoid content in different parts of the sour orange (Citrus aurantium L.) and the ligand activity of a bile acid receptor, TGR5. Horticulture J. 2020;89:384-393.
- [Google Scholar]
- Gentic relationships among Fructus Aurantii cultivars revealed by ISSR. Chin J of Chin Mater Med. 2005;30:1416-1419.
- [Google Scholar]







