Translate this page into:
Variations in metabolites content and bioactivity to regulate biomarkers of benign prostatic hyperplasia according to the growth stages of Sida rhombifolia
⁎Corresponding authors. jrrho@kunsan.ac.kr (Jung-Rae Rho), jeong.ej@gnu.ac.kr (Eun Ju Jeong)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Abstract
It has been reported that the secondary metabolites produced by plants are influenced by genetic diversity and growth conditions, resulting in significant variations in chemical content even within the same species. In the previous study searching for bioactive materials from plants to improve benign prostatic hyperplasia (BPH), it was found that the methanolic extract of Sida rhombifolia in the family of Malvaceae exhibited the excellent inhibition on the expressions of 5-alpha reductase type 2 (5αR2) and androgen receptor (AR) in human prostate cells. In this study, we aimed to evaluate the change in the contents of major components of S. rhombifolia and the activity of improving BPH according to the growth stages of S. rhombifolia. Plant growth characteristics including plant height, stem diameter, leaf length, leaf width, and number of leaves were examined at intervals of approximately 15 days for 51 days. The contents of 20-Hydroxyecdysone and α-Ecdysone, the main constituents contained in S. rhombifolia on each day after transplantation (DAT) were analyzed using LC-ESI-MS/MS. The inhibitory activities of S. rhombifolia stems or leaves at each DAT on the expressions of AR, 5αR2, proliferating cell nuclear antigen (PCNA) and prostate specific antigen (PSA), were evaluated in human originated prostate cells, RWPE-1 and LNCaP cells activated by Testosterone propionate (TP). Considering the yield of the raw materials, the contents of metabolites, and the bioactivities, it was suggested that the appropriate collection period for S. rhombifolia as a bioactive material to improve BPH might be after 90 DAT.
Keywords
Sida rhombifolia
Benign prostatic hyperplasia
5-Alpha reductase
Secondary metabolites
Growth stages
LC-MS/MS
1 Introduction
Sida rhombifolia L. is a perennial plant belonging to the Malvaceae family, is distributed in tropical and subtropical regions, and is a naturalized plant in Korea (Yang, 2005). S. rhombifolia has been used to treat rheumatism and urinary tract diseases in Ayurveda, a traditional Indian medicine. S. rhombifolia grows up to 50–120 cm tall, and the stem is erect, cylindrical, green or purple, and has many branches. This plant has sparse star-shaped hairs in its juvenile stage, transitioning to an almost glossy upon maturity. As the stem develops, it gradually becomes semi-woody. (Adjanohoun et al., 1996; Cambria et al., 2022). According to the investigation of the constituents of S. rhombifolia, alkaloids, coumarins, flavonoids, steroids, and in particular, ecdysteroids including α-ecdysone (E) and 20-hydroxyecdysone (20HE) have been reported as major components (Prakash et al., 1981; Chaves et al., 2013; Chaves et al., 2017; Jadhav et al., 2007; Goyal et al., 1988; Woldeyes et al., 2012). In the study of the physiological activity of S. rhombifolia, various effects have been reported including vasodilation (Bartsch et al., 2002), hepatoprotection (Dhalwal et al., 2006), arthritis treatment (Gupta et al., 2009), anti-diabetic (Ghosh et al., 2011), and anti-inflammation (Arciniegas et al., 2017).
Primary metabolites perform the functions of photosynthesis and respiration, which are important for the survival of the species, while secondary metabolites are organic molecules that are not involved in the normal growth and development of an organism (Agostini et al., 2012). Secondary metabolites produced by plants are influenced by genetic diversity and environmental factors, resulting in significant differences in chemical content even within the same species (CV Borges et al., 2017). Verma et al (2007) reported that the content of secondary metabolites in Asparagus racemosus, Boerhavia diffusa, and Sida cordifolia varied according to season and plant growth stage which demonstrated changes in the contents of total alkaloids and phenols in the plants.
Benign prostatic hyperplasia (BPH) is one of the most common urological conditions in older males, occurring when the prostate gland around the urethra grows abnormally, blocking urine output. BPH is known to be mainly caused by changes in the endocrine system, including sex hormones, and aging, although the precise molecular biological mechanisms remain unclear (Pyo et al., 2017). The prevalence of BPH increases after the age of 50 and reaches over 70 % in the 70 s and 90 % in the 90s (Berry et al., 1984). The main clinical symptom observed in BPH patients is Lower Urinary Tract Symptoms (LUTS), which significantly affect the quality of life of patients (Lim, 2017). The elevated levels of dihydrotestosterone (DHT) observed in BPH patients are closely associated with the onset and pathological progression of BPH. The male hormone testosterone (T) is converted into its active form, DHT, by the lipophilic enzyme 5-alpha-reductase (5αR). Two isoenzymes of 5αR exist in the body, 5αR typeⅠ(5αR1) mainly expressed in epithelial cells (skin, liver) and 5αR typeⅡ (5αR2) expressed in stromal cells (mainly prostate) (Carson Ⅲ et al., 2003; Bartsch et al., 2002; Kaefer et al., 1996). It is well known that the overexpression of 5αR2 and the increased conversion to DHT, along with binding to the androgen receptor (AR), cause BPH. Therefore, selective inhibitors for 5αR2, such as finasteride, are considered one of the effective pharmaceuticals for the prevention or treatment of BPH (Wang et al., 2010).
In the search for new substances from natural products to improve BPH, it was found that the extract of S. rhombifolia exhibited excellent regulatory activity on 5αR2 in prostate cells (Jeong et al., 2019; Park, 2023). This study aimed to evaluate the variations in the activity of S. rhombifolia in improving BPH and to analyze the changes in the content of major secondary metabolites at different growth stages. For this, the plant growth characteristics such as plant height, stem diameter, leaf length, leaf width, and number of leaves were examined at intervals of approximately 15 days over a period of 10 weeks. The contents of major constituents of S. rhombifolia, 20HE and E were analyzed using LC-MS/MS analysis, and the inhibitor activities on the expression of AR, 5αR2, proliferating cell nuclear antigen (PCNA), and prostate specific antigen (PSA), biomarkers of BPH, were measured in testosterone propionate (TP) activated RWPE-1 and LNCaP cells.
2 Materials and methods
2.1 Plant material
The seeds of S. rhombifolia were collected from Hallim-eup, Jeju-si, South Korea, in 2019. Plant identification was authenticated by Dr. Soo-Yong Kim in Korea Research Institute of Bioscience and Biotechnology (KRIBB). The cultivation and the investigation of plant growth characteristics were conducted in Naedong Campus of Gyeongsang National University. S. rhombifolia seeds were sown in 72 cell plug trays containing horticultural medium. After growing the seedlings of S. rhombifolia for 51 days, S. rhombifolia was transplanted. Samples were collected at 45, 60, 75, 90, 105, and 120 days after transplantation (DAT). The reason for setting 120 DAT as the final observation point is that, after this point, no leaves were observed due to leaf fall. The flowering was observed around 60 DAT.
2.2 Instrumentation and Reagents
LC-ESI-MS/MS analysis was performed using an Agilent HPLC 1100 series (Santa Clare, CA, USA) and an ABSciex API 3200 mass spectrometer (Applied Biosystems, MDS Sciex, Concord, Canada) equipped with an ESI-Source. Analyst® software (Version 1.6.3) was used to process the data obtained using HPLC and MS/MS. The column used YMC-Triart C18 (50 × 2.1 mm I.D. S-3 ㎛, 12 nm, Japan). The 3-(4,5-Dimethylthiazol-2-yl)-2,5 diphenyltetrazolium bromide (MTT) reagent was purchased from Sigma-Aldrich (St. Louis, MO, USA). Acetonitrile, methanol, and water were HPLC grade and purchased from honeywell burdick & jackson (Muskegon, USA). Formic acid (HPLC grade) was purchased from Fujifilm (Osaka, Japan).
2.3 Investigation of growth characteristics of S. rhombifolia
To investigate the growth characteristics of S. rhombifolia, plant height, stem diameter, leaf length, leaf width, and number of leaves were measured at 5, 30, 45, 52, 60, and 75 DAT. Plant height (cm) was recorded from the ground surface to the tip of the plant. Stem diameter (mm) was measured using the calipers (Digimatic caliper CD-15APX, Mitutoyo, Japan) for stem thickness at 2.5 ∼ 3.0 cm from the ground surface. The length (mm) and width (mm) of the leaf were recorded the longest and widest leaf, respectively. The number of leaves was investigated by the number of leaves per individual.
2.4 Dry yield and extraction of S.rhombifolia
The stems, leaves and flowers of S. rhombifolia collected at 45, 60, 75, 90, 105, 120 DAT were dried at 40 °C for 72 h for estimating dry yield. For the extraction, 500 mg of each dried stems, leaves, and flowers of S. rhombifolia were extracted with 22.5 mL 80 % (v/v) methanol for 4 h using an ultrasonic apparatus. The extract was filtered through a 0.45 ㎛ PVDF filter and the solvent was removed in vacuo.
2.5 LC-ESI-MS/MS analysis of S. rhombifolia extract
LC-ESI-MS/MS analyses were performed on an Agilent HPLC 1100 series (Santa Clare, CA, USA). A YMC-Triart C18 (50 × 2.1 mm I.D. S-3 ㎛, 12 nm, Japan) was used and maintained at room temperature. The mobile phase consisted of DW (0.1 % formic acid) (A) and acetonitrile (0.1 % formic acid) (B) at a flow rate of 0.3 mL/min, with a gradient elution as follows: 0–2 min, 90 % A; 2–8 min, 90–0 % A; 8–10 min, 0 % A. The injection volume was 1.0 µL. MS/MS analysis was performed using MRM (Multiple Reaction Monitoring) mode after optimization to derive the optimal analysis conditions for 20HE and E. Ionization was used ESI (electrospray ionization) using Z-spray, argon gas was used as collision gas, and nitrogen gas was used as nebulizer gas. Ion source temperature was set at 500℃, nebulizer gas flow at 50 L/min, curtain gas flow at 20 L/min, auxiliary gas flow at 55 L/min, and Ion spray voltage at 4500 kV. ESI was operated in the positive ion mode. Full-scan spectra were acquired over the range of m/z 100–1000. S. rhombifolia extracts were diluted to 25 µg/mL with 50 % acetonitrile solvent and then injected into LC-ESI-MS/MS. 20HE and E were set as major compounds, and the experiment was conducted by diluting 20HE in the range of 10–1000 ng/mL and E in the range of 1–100 ng/mL using 10 % acetonitrile containing 0.1 % formic acid.
2.6 Cell cultures
RWPE-1 (normal human prostatic epithelial cell line) and LNCaP (human prostatic adenocarcinoma cell line) cells were purchased from ATCC (American Type Culture Collection, Manassas, VA, USA). RWPE-1 cells were cultured in keratinocyte serum free medium (K-SFM; Gibco BRL, Grand Island, NY, USA) supplemented with 0.05 mg/mL bovine pituitary extract, 5 ng/mL epidermal growth factor and 1 % (v/v) antibiotics (100 U/mL penicillin and 100 µg/mL streptomycin). LNCaP cells were cultured in RPMI-1640 (GenDEPOT, CM059-050) medium containing 10 % (v/v) Fetal Bovine Serum (FBS), 1 % (v/v) antibiotics (100 U/mL penicillin and 100 µg/mL streptomycin). The cells were incubated at 37℃ and 5 % CO2 saturation.
2.7 Estimation of cell viability
Cell viability was determined using the colorimetric MTT assay. RWPE-1 and LNCaP cells were seeded at densities of 1 × 104 cells/well, respectively, in 96-well plates and incubated for 24 h. Then, the extract of S. rhombifolia (1, 5, 10, 20 µg/mL) was treated and incubated for 24 h. MTT solution (2 mg/mL) in distilled water was added to each well for 3 h at 37℃. The supernatant was aspirated and 100 µL of DMSO was added to solubilize the formazan crystals. After those insoluble crystals were completely dissolved, absorbance at 540 nm was measured using TECAN microplate reader (Infinite M200 Pro, TECAN).
2.8 Western Blotting
RWPE-1 and LNCaP cells were seeded at densities of 6 × 105 cells/well and 4 × 105 cells/well, respectively, in 6-well plates and incubated overnight. The cells were treated with 20 µg/mL of S. rhombifolia extract or finasteride (10 µM), a positive control, for 1 h, followed by treated with 0.5 µM TP. RWPE-1 and LNCaP cells were further incubated for 24 h and 72 h, respectively. The cells were washed with cold phosphate buffered saline (PBS) and cell lysates were extracted with a lysis buffer (M−PER™ Mammalian Protein Extraction Reagent 78501, Thermo Scientific, Waltham, MA, USA) containing protease inhibitor cocktail (Thermo Scientific, Waltham, MA, USA). The protein extracts were centrifuged at 14,000 rpm for 20 min at 4℃. The protein content of cell lysates was quantified using the Bradford assay. Thirty micrograms of harvested protein were separated in 10 % Sodium Dodecyl Sulfate-PolyAcrylamide Gel Electrophoresis (SDS-PAGE) at 100 V and transferred to polyvinylidene fluoride (PVDF) membranes. The membrane was blocked with 5 % skim milk containing 10 mM Tris, 150 mM NaCl, and 0.05 % tween 20 for 1 h at room temperature. Anti-AR (5153 T, Cell Signaling Technology), anti-PCNA (13110S, Cell Signaling Technology), anti-PSA (5365S, Cell Signaling Technology), anti-5αR2 (sc-293232, Santa Cruz Biotechnology), anti-α-tubulin (sc-8035, Santa Cruz Biotechnology) and anti-β-actin (sc8432, Santa Cruz Biotechnology) were used in 1 % skim milk. The membrane was incubated with each primary antibody at 4℃ overnight. After washing three times with Tris Buffered Saline with Tween (TBS-T) buffer, immunoreactive bands were visualized by using immunopure peroxidase-conjugated mouse anti-rabbit IgG or goat anti-mouse IgG (1:10,000 dilution; Santa Cruz Biotechnology). The membrane was incubated with secondary antibodies for 1 h at room temperature. The blots were washed three times with TBS-T buffer. The protein bands were visualized using ECL solution (bio-rad clarity Max western ECL substrate) and calibrated using the Chemidoc Imaging System (Fusion FX5, Vilber Lourmat, France).
2.9 Statistical analysis
Growth characteristics investigation, dry yield and quantitative determination statistical analysis were performed with IBS SPSS statistics v26 program (International Business Machines Co, Ltd., USA). The significance level was determined by Duncan’s Multiple Range Test. (p < 0.05). For in vitro experiment was performed with GraphPad Prism 6 program (GraphPad Institute Inc., USA). The significance level was determined by One-Way ANOVA Test (p < 0.05).
3 Results
3.1 Investigation of growth characteristics of S. rhombifolia
To determine the growth characteristics of S. rhombifolia according to the growth stages, plant height, stem diameter, leaf length, leaf width, and number of leaves were investigated. It was found that plant height increased approximately 4.2 times from 95.3 mm to 396.6 mm from 5 DAT to 30 DAT. A significant increase in plant height was observed between 30 DAT and 52 DAT. It was observed that the plant height at 45 DAT increased by approximately 1.7 times compared to that at 30 DAT, which continued to increase up to 1.3 times by 52 DAT (Fig. 1A). Stem diameter was observed to be 1.6 mm at 5 DAT, 4.2 mm at 30 DAT, 7.6 mm at 45 DAT, and 11.2 mm at 52 DAT. The stem diameter at 52 DAT increased about 7 times compared to 5 DAT (Fig. 1B). Leaf length increased from 35.1 mm at 5 DAT to 60.0 mm at 30 DAT, and thereafter exhibited a gradually increasing trend (Fig. 1C). Similar to leaf length, leaf width showed the rapid growth from 5 DAT to 30 DAT. The width of 16.4 mm at 5 DAT increased to 37.7 mm at 30 DAT, indicating an increase of approximately 2.3 times. Furthermore, at 45 DAT, the leaf width increased by approximately 1.1 times compared to that at 30 DAT, followed by a gradual increase thereafter (Fig. 1D). The number of leaves exhibited a gradual increase throughout the growth period, reaching its maximum at 60 DAT, showing an approximately 19.1-fold increase compared to 5 DAT, before gradually decreasing thereafter (Fig. 1E). All parameters except the number of leaves were observed to reach the highest values at 60 DAT and 75 DAT.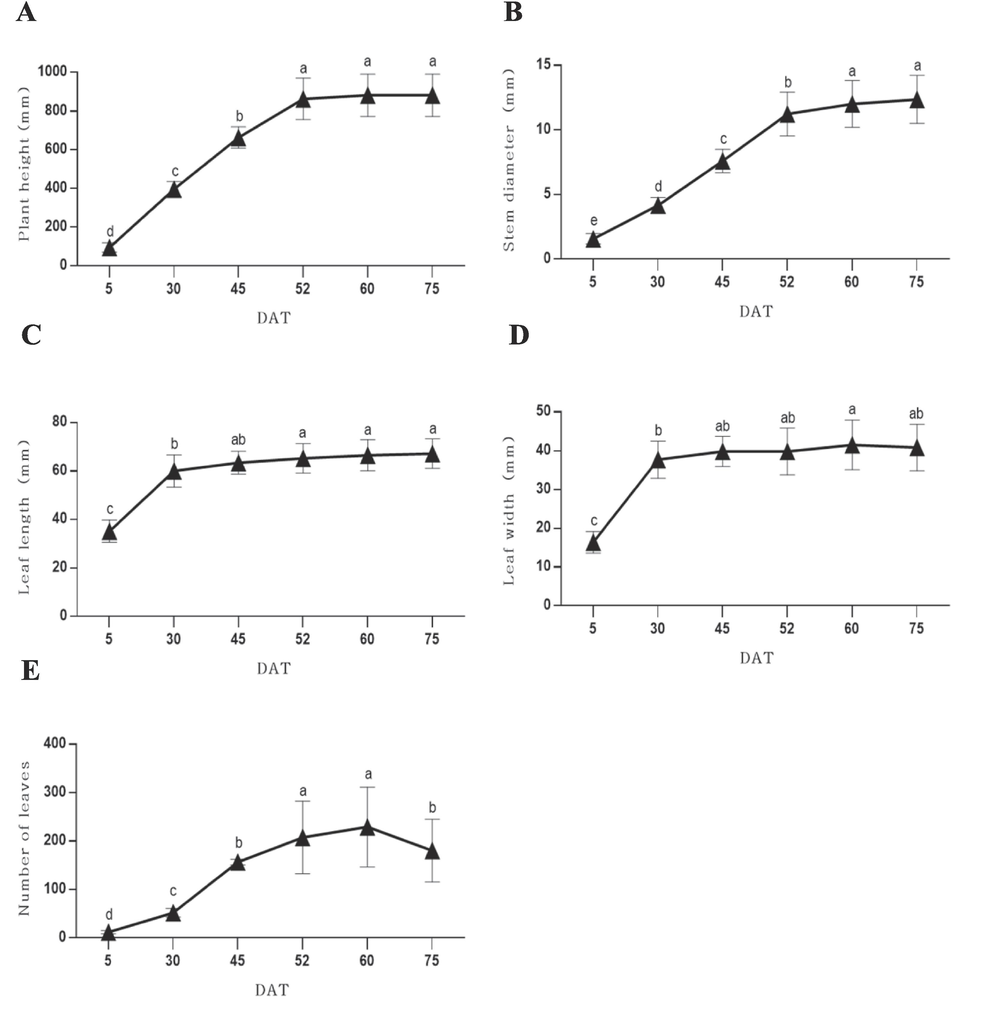
Plant height (A), stem diameter (B), leaf length (C), leaf width (D) and number of leaves (E) according to the growth stages of S. rhombifolia. Growth characteristics were investigated 6 times at 5, 30, 45, 52, 60 and 75 days after transplantation (DAT). Results were expressed as the mean ± S.D. of data. Different letters indicate significant differences at p < 0.001.
3.2 Dry yield of stems and leaves of S. rhombifolia according to growth stages
The stems and leaves of S. rhombifolia collected between 45 DAT and 120 DAT were dried and dry yield was calculated. The dry yield of stems at different growth stages of S. rhombifolia ranged from 26.71 to 69.28 %. The highest dry yield was found at the final growth stage, 120 DAT, indicating 2.6-fold increase compared to yield at 45 DAT (Table 1). In contrast, the dry yield of leaves ranged from 31.90 to 43.61 % with the highest yield recorded at 60 DAT, after which it decreased (Table 2). The flowers of S. rhombifolia were collected at 60 DAT, and the dry yield of flowers was measured as 46.25 %. The dry yield of stems and leaves reached their maximum levels at 75 DAT and 60 DAT, respectively, then slightly decreased thereafter. However, an increasing trend was observed again until the final observation point of growth.
DATa
Fresh weight (g)
Dry weight (g)
Dry yield (%)
45
63.88 ± 12.17cb
17.09 ± 3.40c
26.71 ± 1.05d
60
76.93 ± 27.46bc
26.65 ± 10.41c
34.98 ± 8.85c
75
131.33 ± 48.08a
64.95 ± 17.90a
50.95 ± 5.09b
90
110.67 ± 35.32ab
42.92 ± 13.79b
38.74 ± 0.90c
105
94.00 ± 28.82abc
43.48 ± 13.89b
46.09 ± 1.33b
120
73.33 ± 20.85bc
50.97 ± 14.97ab
69.28 ± 2.01a
Significantc
*
***
***
DATa
Fresh weight (g)
Dry weight (g)
Dry yield (%)
45
24.20 ± 3.87abb
7.72 ± 1.26a
31.90 ± 0.64c
60
20.47 ± 6.58b
8.45 ± 1.92a
43.61 ± 12.89a
75
29.00 ± 11.08ab
9.52 ± 3.68a
32.75 ± 1.26bc
90
32.33 ± 10.31a
10.73 ± 3.46a
33.21 ± 1.05bc
105
21.33 ± 6.15b
8.63 ± 2.73a
40.13 ± 3.27ab
Significantc
NS
NS
**
3.3 LC-ESI-MS/MS analysis of S. rhombifolia
In the previous study reported by Jadhav et al (2007), seven ecdysteroids including 20HE and E were isolated from the aerial part of S. rhombifolia and their structures were determined. Additionally, LC-ESI-MS analysis demonstrated that 20HE and E were the major compounds in S. rhombifolia (Wang et al., 2008). In this study, the change in the contents of 20HE and E in the extract of S. rhombifolia at different growth stages was measured through LC-ESI-MS/MS. Prior to the analysis of extracts using MRM mode to improve detection sensitivity, the detection conditions of each compound, 20HE and E, were optimized and the corresponding values were selected for analysis. The precursor and product ion of 20HE and E for MRM were set m/z 481.5 [M+H]+ and m/z 445.2 [M−2H2O]+, m/z 465.6 [M+H]+ and m/z 429.4 [M−2H2O]+, respectively (Figs. 2 and 3). The parameter values of declustering potential (DP), entrance potential (EP), collision energy (CE), collision cell entrance potential (CEP), and cell exit potential (CXP) optimized for 20HE and E were shown in Table 3. For quantitation of 20HE and E in the extract of S. rhombifolia, calibration curves were obtained with different concentration ranges. 20HE was diluted to 10, 25, 50, 100, 200, 1000 ng/mL, and E was diluted to 1, 5, 25, 50, 100 ng/mL. The linearity was evaluated by the regression coefficient (R2) of the respective calibration curves which showed R2. Calibration curves of 20HE and E showed excellent linearity with regression coefficient (R2) of 0.9999 and 0.9993, respectively (Table 4). DP-declustering potential; EP-entrance potential; CE-collision energy; CEP-cell entrance potential; CXP-cell exit potential.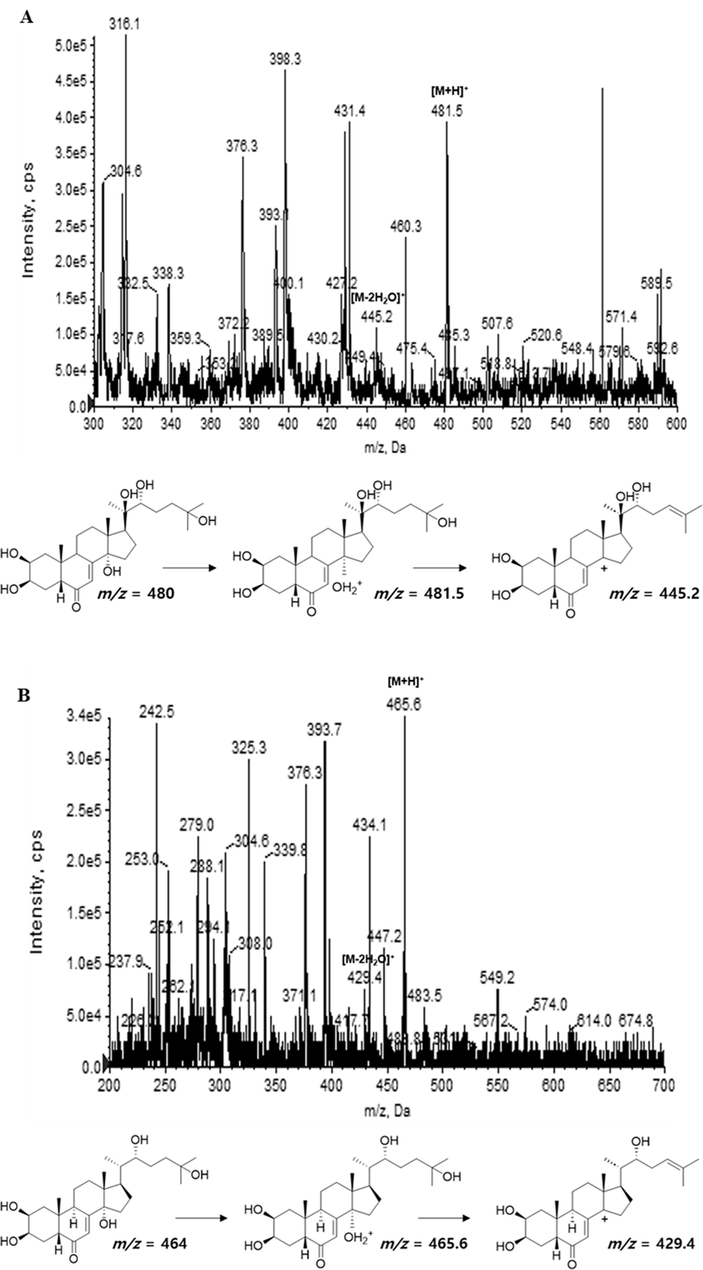
Product ion mass spectra of 20-hydroxyecdysone (A) and α-ecdysone (B) isolated from S. rhombifolia.
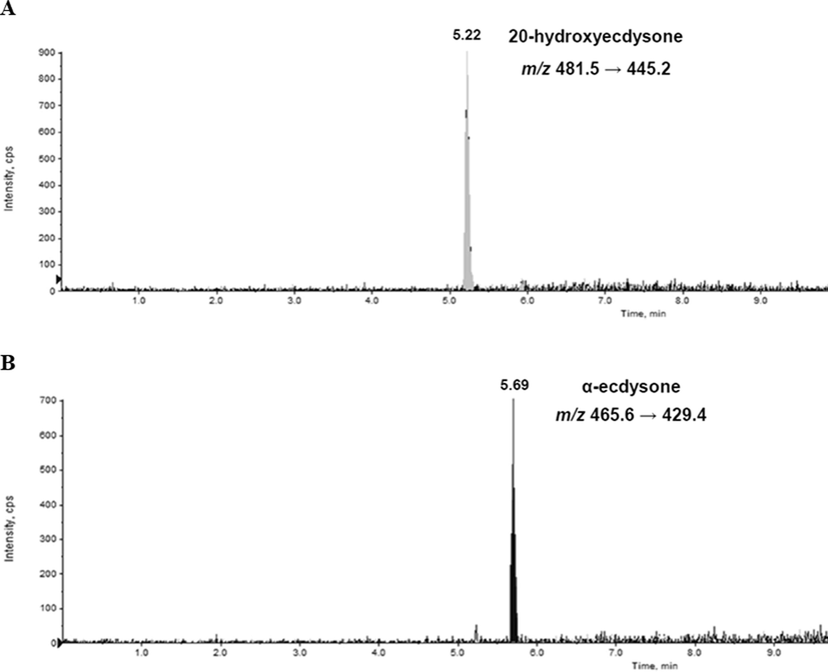
Extracted ion chromatograms of analytes quantified 20-hydroxyecdysone (A) and α-ecdysone (B) isolated from S. rhombifolia in MRM mode.
Compounds
Precursor ion (m/z)
Product ion
(m/z)
Optimized Parameters
DP
EP
CE
CEP
CXP
20-hydroxyecdysone
481.5
445.2
31
6.5
21
18
6
α-ecdysone
465.6
429.4
21
3.5
19
24
6
Compounds
Regression equation
R2
Linearity range (ng/mL)
20-hydroxyecdysone
y = 27.9x – 62.4
0.9999
10–1000
α-ecdysone
y = 14.7x – 22.7
0.9993
1–100
3.4 Quantitative determination of 20HE and E in S. rhombifolia extract
Using the optimized parameters of 20HE and E, the contents of these compounds in the extract of S. rhombifolia at the different growth stages were analyzed. The results of quantitative analysis of 20HE and E in S. rhombifolia extract were summarized in Fig. 3. In stems of S. rhombifolia, the contents of 20HE and E were highest at 60 DAT and decreased thereafter, then tended to increase again at 90 DAT. The contents of 20HE and E in S. rhombifolia stems were found as 6.22 and 1.3 µg/g at 60 DAT, 3.67 and 0.81 µg/g at 90 DAT, 5.09 and 0.79 µg/g at 120 DAT, respectively (Fig. 4A and B). In leaves of S. rhombifolia, similar to that of stems, the contents of 20HE and E were highest at 60 DAT. The contents of 20HE and E in the S. rhombifolia leaves were 3.21 and 0.64 µg/g at 60 DAT, 1.55 and 0.31 µg/g at 75 DAT, 1.75 and 0.23 µg/g at 105 DAT, respectively (Fig. 4C and D). In the case of flowers, the contents of 20HE and E were measured to be 30.73 µg/g and 3.47 µg/g, respectively, based on the average values. It was confirmed that the content of 20HE was much higher in flowers compared to stems (3.67 ∼ 6.22 µg/g) or leaves (1.55 ∼ 3.21 µg/g).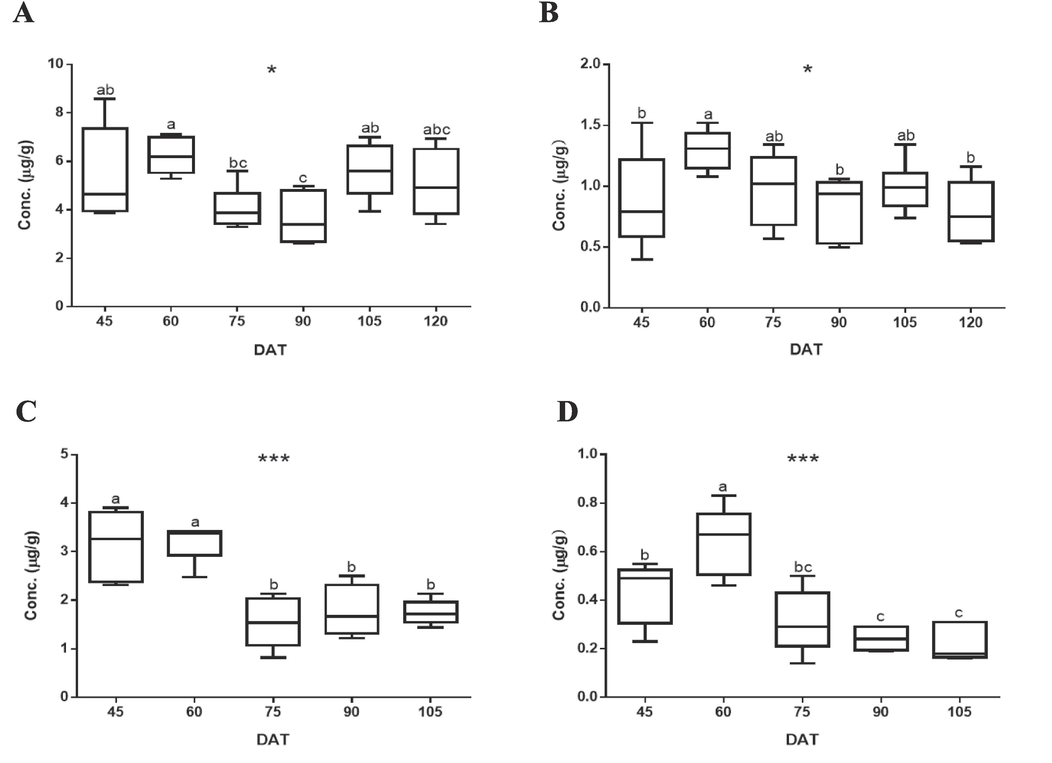
Quantitative determination of (A) 20-hydroxyecdysone and (B) α-ecdysone in the extract of S. rhombifolia stems and (C) 20-hydroxyecdysone and (D) α-ecdysone in the extract of S. rhombifolia leaves by LC-ESI-MS/MS. Results were expressed as the mean ± S.D. of data. DAT: Day After Transplantation. Different letters indicate significant differences at * p < 0.05 and *** p < 0.001.
3.5 Activities of stems of S. rhombifolia to improve the expressions of AR, 5αR2, PCNA and PSA in testosterone propionate-activated RWPE-1 and LNCaP cells
The protective activities of stems, leaves, and flowers of S. rhombifolia on the expressions of proteins related to BPH were evaluated in TP-activated RWPE-1 and LNCaP cells. Prior to evaluating the activities, the toxicities of each extract (stems, leaves and flowers of S. rhombifolia) were measured using MTT assay. The treatment of cells with each extract of S. rhombifolia showed no toxicity against RWPE-1 or LNCaP cells at the concentration range from 1 to 20 μg/mL (data not shown). The regulatory effects of the extract of S. rhombifolia on the expressions of AR, 5αR2, PCNA and PSA, biomarkers of BPH, were measured using Western blot assay. Finasteride, a selective 5αR2 inhibitor, was applied as a positive control.
In RWPE-1 cells, when treated with TP, the expression levels of intracellular AR, 5αR2, and PCNA were increased by 26 %, 37 %, and 33 %, respectively, compared to those of non-treated cells (NC) (Fig. 5). The treatment of the cells with the extract of S. rhombifolia stems significantly reduced the expressions of these proteins. The reduction of AR expression was observed from 75 DAT, with a subsequent decrease thereafter. In cells treated with the extract at 120 DAT, the last stage of growth, AR expression decreased to 49 % compared to that of the TP-only treated group. The reduction of 5αR2 expression was observed at all stages of growth, particularly at 120 DAT, where the most remarkable activity on 5αR2 was demonstrated, resulting in a decrease of 41 % compared to the TP-only treated group. The reduction of PCNA expression was found at 45 DAT and from 90 to 120 DAT. Based on the above results, it was found that the period with the highest activity of S. rhombifolia for suppressing three biomarkers associated with BPH, 5αR2, AR, and PCNA, in RWPE-1 cells occurred between 90 and 120 DAT.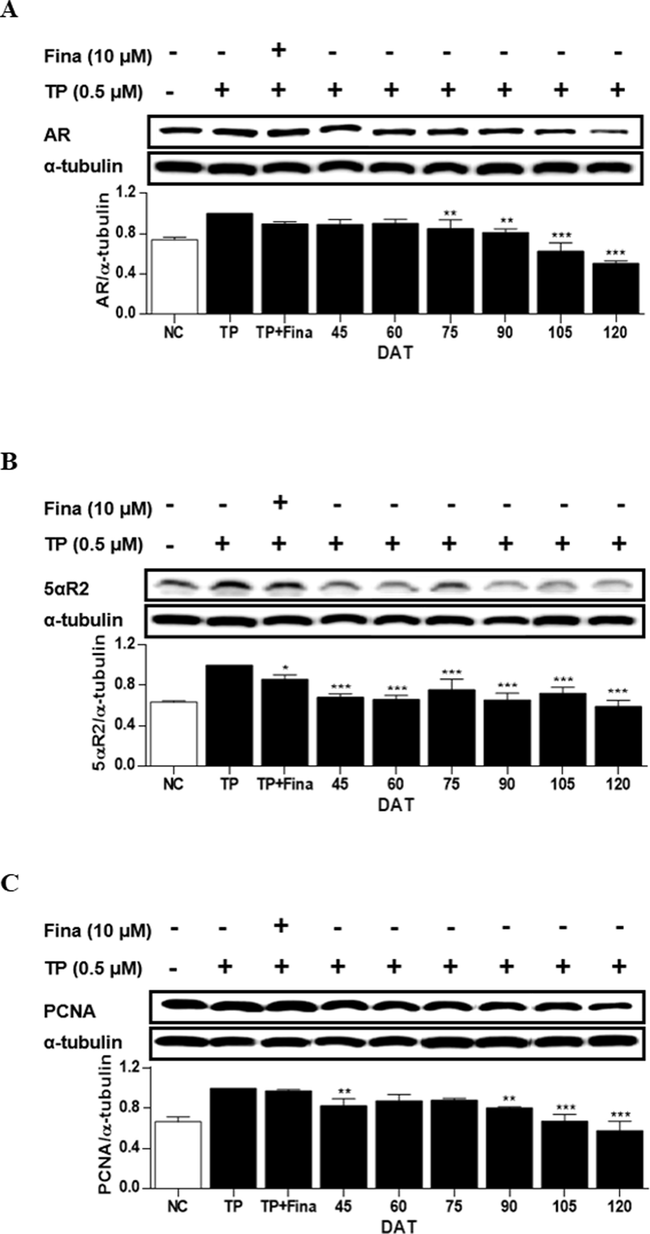
Effects of extract of S. rhombifolia stems on the expressions of AR, 5αR2, and PCNA proteins in RWPE-1 cells at different growth stages. The expression levels of AR, 5αR2, and PCNA proteins were analyzed by western blot. The relative protein levels were quantified and normalized to α-tubulin. The values shown are the mean ± S.D. of data from three experiments. NC: Normal Control, TP: Testosterone Propionate, TP + Fina: Testosterone Propionate + Finasteride, DAT: Days After Transplantation. *p < 0.05, **p < 0.01 and ***p < 0.001 vs TP.
In LNCaP cells, the treatment of cells with TP induced the expressions AR, 5αR2 and PSA by 35 %, 17 % and 49 % compared to that of non-treated cells (Fig. 6). In cells treated with extract of S. rhombifolia stems, the reduction of AR and PSA expression was shown from 90 to 120 DAT, and from 60 to 120 DAT, respectively. While the significant reduction of 5αR2 expression was only observed at 120 DAT. The activities of S. rhomibifolia on the enzymes did not entirely coincide in two in vitro systems, however, a similar trend was observed. In LNCaP cells, the period during which 5AR and PSA were suppressed by S. rhombifolia occurred after 90 DAT, while the time point at which all three enzymes, including AR, were suppressed was 120 DAT.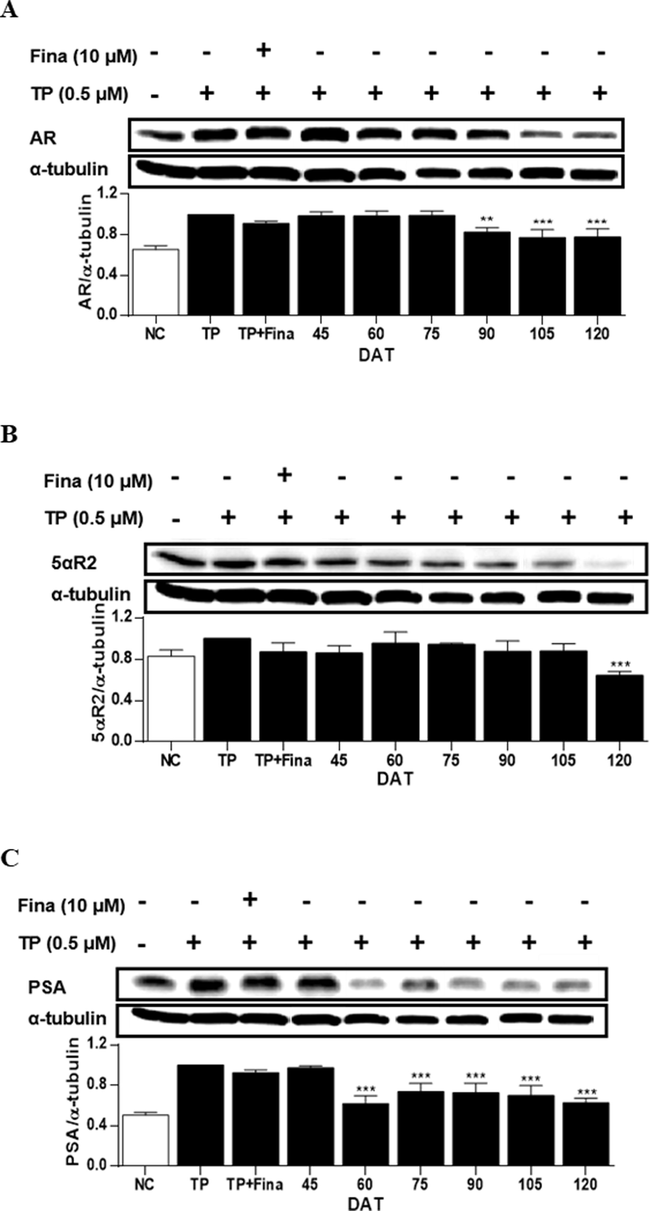
Effects of extract of S. rhombifolia stems on the expressions of AR, 5αR2, and PSA proteins in LNCaP cells at different growth stages. The expression levels of AR, 5αR2, and PSA proteins were analyzed by western blot. The relative protein levels were quantified and normalized to α-tubulin. The values shown are the mean ± S.D. of data from three experiments. NC: Normal Control, TP: Testosterone Propionate, TP + Fina: Testosterone Propionate + Finasteride, DAT: Days After Transplantation. **p < 0.01 and ***p < 0.001 vs TP.
3.6 Activities of leaves of S. rhombifolia to improve the expressions of AR, 5αR2, PCNA and PSA in testosterone propionate-activated RWPE-1 and LNCaP cells
In RWPE-1 cells, when treated with TP, the expression levels of intracellular AR, 5αR2, and PCNA were increased by 15 %, 16 %, and 23 %, respectively, compared to those of NC (Fig. 7). The expressions of these proteins were significantly reduced by the treatment with the extract of S. rhombifolia leaves at all stages of growth. The reductions in AR and 5αR2 expressions were remarkable at 105 DAT demonstrating the decreases of 29 % and 53 % compared to the TP-only treated group, respectively. The reduction in PCNA expression was significant at 90 DAT resulting in a decrease of 53 % compared to the TP-only treated group. Based on the above results, it was found that the period with the highest activity of S. rhombifolia leaves for suppressing 5αR2, AR, and PCNA in RWPE-1 cells occurred between 90 and 120 DAT.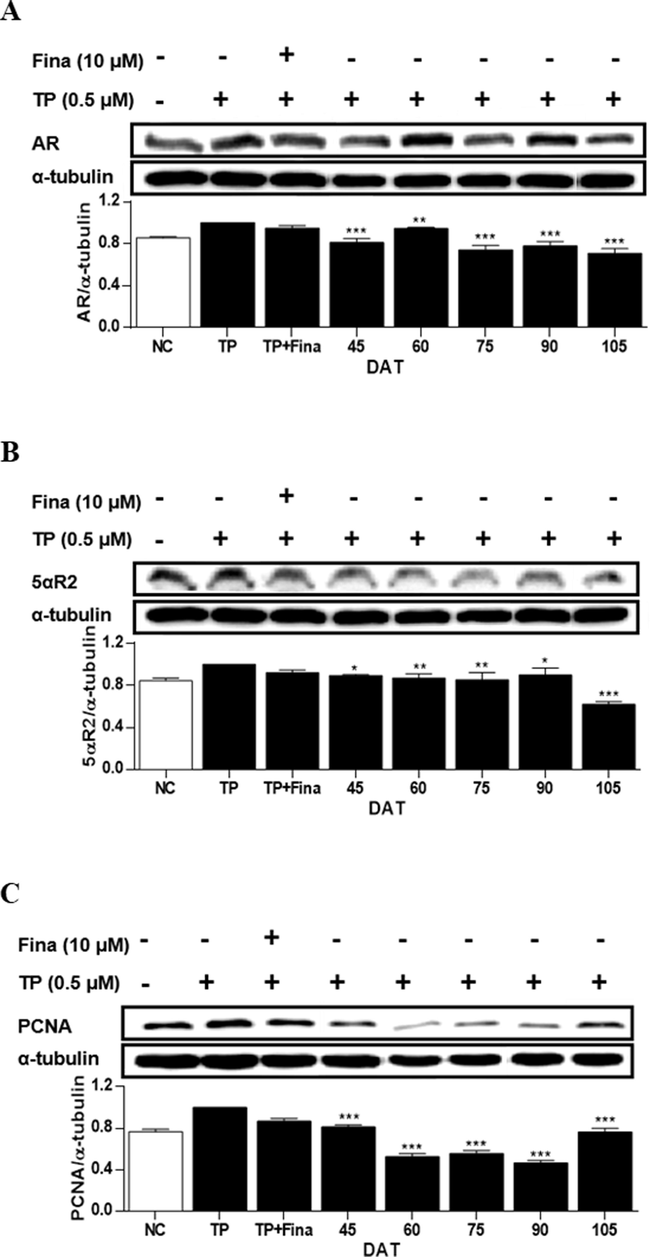
Effects of extract of S. rhombifolia leaves on the expressions of AR, 5αR2, and PCNA proteins in RWPE-1 cells at different growth stages. The expression levels of AR, 5αR2, and PCNA proteins were analyzed by western blot. The relative protein levels were quantified and normalized to α-tubulin. The values shown are the mean ± S.D. of data from three experiments. NC: Normal Control, TP: Testosterone Propionate, TP + Fina: Testosterone Propionate + Finasteride, DAT: Days After Transplantation. *p < 0.05, **p < 0.01 and ***p < 0.001 vs TP.
In the TP-treated LNCaP cells, the expressions of AR, 5αR2, and PSA increased by 31 %, 93 %, and 58 %, respectively, compared to NC (Fig. 8). Conversely, treatment of cells with the extract of S. rhombifolia leaves resulted in a reduction of AR, 5αR2, and PSA expressions at all stages of plant growth. The most notable decrease in AR and 5αR2 expressions was observed at 105 DAT, whereas the most significant reduction in PSA expression was found at 45 DAT. Therefore, the period with the highest suppressive activity of S. rhombifolia on these proteins associated to BPH in LNCaP cells was identified to be at 105 DAT.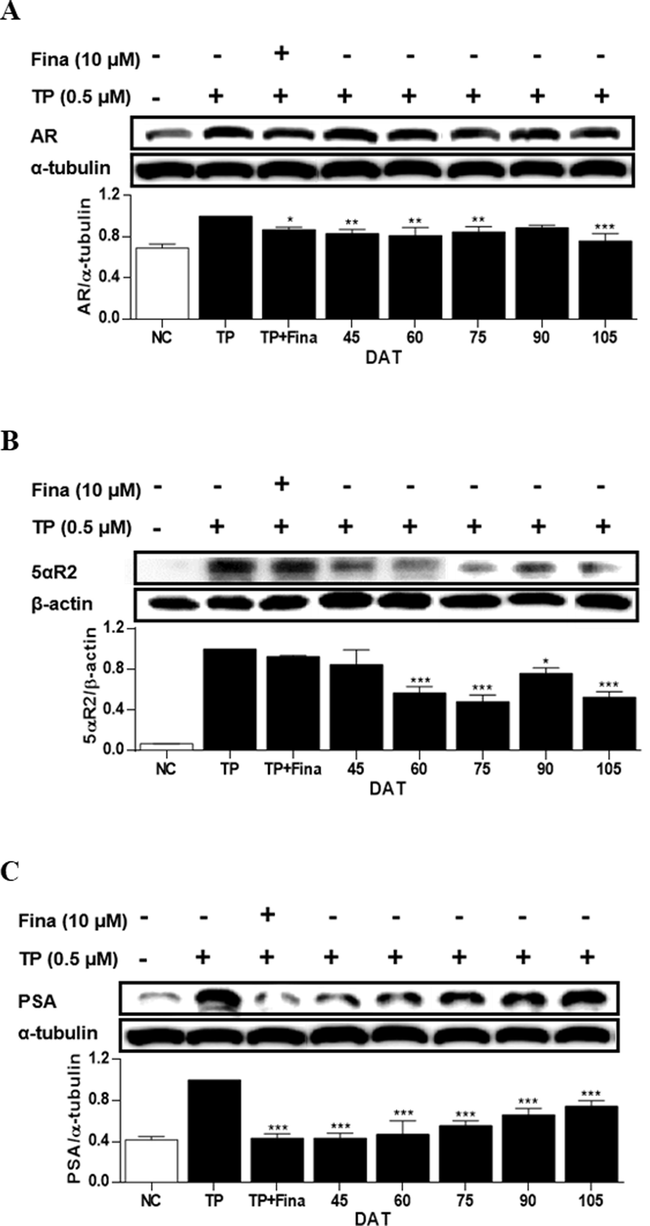
Effects of extract of S. rhombifolia leaves on the expressions of AR, 5αR2 and PSA proteins in LNCaP cells at different growth stages. The expression levels of AR, 5αR2, and PSA proteins were analyzed by western blot. The relative protein levels were quantified and normalized to α-tubulin and β-actin. The values shown are the mean ± S.D. of data from three experiments. NC: Normal Control, TP: Testosterone Propionate, TP + Fina: Testosterone Propionate + Finasteride, DAT: Days After Transplantation. *p < 0.05, **p < 0.01 and ***p < 0.001 vs TP.
3.7 Activities of flowers of S. rhombifolia to improve the expressions of AR, 5αR2, PCNA and PSA in testosterone propionate-activated RWPE-1 and LNCaP cells
In the TP-treated RWPE-1 cells, the treatment with the extract of S. rhombifolia flowers significantly reduced the expressions of AR, 5αR2 and PCNA proteins (Fig. 9). The expressions of AR, 5αR2, and PCNA were decreased by S. rhombifolia flowers by 7 %, 17 %, and 20 %, respectively, compared to the TP-only treated group.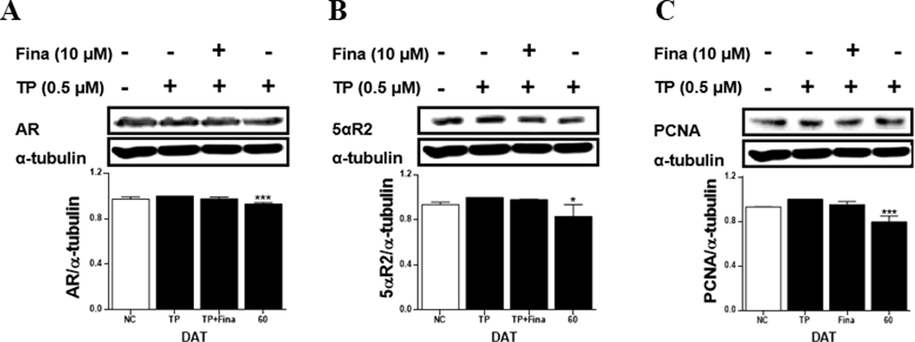
Effects of extract of S. rhombifolia flowers on the expressions of AR, 5αR2, and PCNA proteins in RWPE-1 cells at different growth stages. The expression levels of AR, 5αR2, and PCNA proteins were analyzed by western blot. The relative protein levels were quantified and normalized to α-tubulin. The values shown are the mean ± S.D. of data from three experiments. NC: Normal Control, TP: Testosterone Propionate, TP + Fina: Testosterone Propionate + Finasteride, DAT: Days After Transplantation. *p < 0.05 and ***p < 0.001 vs TP.
In the TP-treated LNCaP cells, the extract of S. rhombifolia flowers reduced the levels of AR, 5αR2, and PSA proteins (Fig. 10). The reduction in AR, 5αR2, and PSA expressions by S. rhombifolia flowers was 18 %, 26 %, and 11 %, respectively, compared to the TP-only treated group.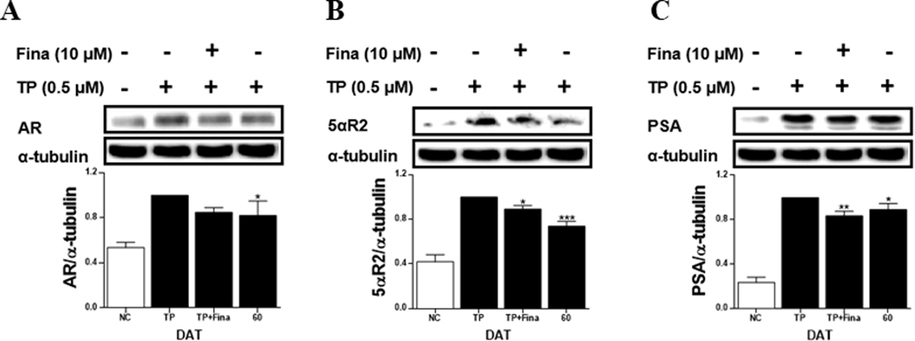
Effects of extract of S. rhombifolia flowers on the expressions of AR, 5αR2 and PSA proteins in LNCaP cells at different growth stages. The expression levels of AR, 5αR2, and PSA proteins were analyzed by western blot. The relative protein levels were quantified and normalized to α-tubulin. The values shown are the mean ± S.D. of data from three experiments. NC: Normal Control, TP: Testosterone Propionate, TP + Fina: Testosterone Propionate + Finasteride, DAT: Days After Transplantation. *p < 0.05, **p < 0.01 and ***p < 0.001 vs TP.
4 Discussion
BPH is one of the most common urinary conditions in elderly men, occurring when the prostate around the urethra abnormally grows and blocks urine output. It has been reported that BPH often lead to LUTS, which significantly negative impact on patients' quality of life (Welch et al., 2002). In BPH patients, higher levels of DHT were observed compared to normal people (Parsons et al., 2010). T is the precursor of DHT, predominantly synthesized in the Leydig cells of the testes in response to the hormone signaling from the hypothalamic-pituitary axis. T produced in the testes migrates to the prostate and is converted to DHT by the enzyme 5αR (Roehrborn, 2008). Two isoenzymes of 5αR exist in the body, 5αR1 mainly expressed in in epithelial cells (skin, liver) and 5αR2 expressed in stromal cells (mainly prostate) (Carson Ⅲ et al., 2003; Bartsch et al., 2002; Kaefer et al., 1996). Finasteride and dutasteride are 5αR inhibitors. Finasteride selectively inhibits the 5αR2 isozyme, whereas dutasteride is a non-selective inhibitor of 5αR1 and 5αR2. Finasteride reduced serum DHT levels by 70–90 %, while dutasteride can effectively decrease serum DHT levels to nearly undetectable levels. Nevertheless, these drugs are associated with adverse effects, including erectile dysfunction, decreased libido, and ejaculation disorder, as documented in studies by Sarma et al. (2012) and Yoo et al. (2008). Hence, there exists a necessity to advance the development of novel compounds demonstrating superior efficacy in alleviating BPH while mitigating the adverse effects associated with current 5α-reductase inhibitors. Notably, recent studies have highlighted the promising potential of natural products exhibiting remarkable efficacy in ameliorating BPH symptoms. The extract of medicinal plants or derived compounds suppressed 5αR and regulated the BPH-associated biomarkers in vitro and in vivo. Park et al., 2022 reported that ellagic acid significantly reduced the prostate size, serum level of DHT, and the protein levels of AR, 5αR2, and PSA in TP-induced rat models. Deng et al., 2019 reported in vitro investigations that revealed significant anti-BPH effects of E. angustifolium. Subsequent in vivo studies demonstrated that E. angustifolium exerted therapeutic effects against TP-induced BPH in Sprague-Dawley rats by reducing androgen levels, suppressing NF-κB expression, and ultimately mitigating inflammatory responses and oxidative stress. The anti-BPH activities of single compounds derived from natural products have been also reported. Pyridinium alkaloids, Voratin A-C isolated from Marine dinoflagellate Effrenium voratum decreased the expressions of AR, 5αR2 and PSA in TP-activated LNCaP cells (Lee et al., 2022). Recently, Kaempferol has been shown to exhibit androgenic-like activity, acting as a selective androgen receptor modulator implicated in androgen-related BPH development. This was evidenced through molecular docking, cellular immunofluorescence staining, chromatin immunoprecipitation, and dual luciferase reporter assay (Wang et al., 2021). In our efforts to search for new bioactive candidates from natural products to improve BPH, it was found that the methanolic extract of S. rhombifolia stems and leaves significantly decreased the expressions of 5αR2 and AR in prostate cells activated with TP.
Primary metabolites carry out essential functions such as photosynthesis and respiration, crucial for the species’ survival. In contrast, secondary metabolites are organic molecules unrelated to the growth and development process of organism (Agostini et al., 2012). The chemical composition of secondary metabolites is significantly influenced by environmental stressors such as temperature, carbon dioxide levels, light exposure, and soil conditions (Pant et al., 2021). For instance, in various growth stages of Physalis peruviana, changes in carotenoid compositions were observed. The highest carotenoid content was noted during the half-ripe stage, with zeaxanthin levels peaking during the yellowing phase, and neoxanthin and lutein levels reaching their highest during the green phase (Yu et al., 2019). Similarly, a comparative HPLC analysis of alkaloid content in Catharanthus roseus leaves collected at different growth stages was reported. It was observed that bisindole alkaloids increased while monoindole alkaloids decreased, as the leaves aged and the plants matured (Pan et al., 2016). The influence of environmental factors on secondary metabolite content is also evident in plants of the Sida genus. Verma et al. (2007) reported variations in the content of total alkaloids and phenols in Sida cordifolia according to the season and growth stage. The maximum accumulation of alkaloids and phenols occurred in April-June for S. cordifolia.
In this study, we attempted to evaluate the variations in the activity of S. rhombifolia to improve BPH and the changes in the content of major secondary metabolites according to its growth stages. For this, the plant growth characteristics such as plant height, stem diameter, leaf length, leaf width, and number of leaves were examined at intervals of approximately 15 days for 10 weeks. The contents of major constituents of S. rhombifolia, 20HE and E were analyzed through LC-MS/MS analysis. Also, the activities regulating the expressions of AR, 5αR2, PCNA and PSA, biomarkers of BPH, were measured in TP-activated RWPE-1 and LNCaP cells. A study on the growth characteristics of S. rhombifolia revealed that both the height and stem diameter increased rapidly up to 52 DAT, followed by a more gradual growth rate thereafter. Similarly, the length and width of S. rhombifolia leaves exhibited rapid growth up to 30 DAT, with a subsequent slowing of the growth rate. Overall, it was found that all growth factors exhibited rapid growth up to 52 DAT, after which there was a noticeable deceleration in their growth rates. Adjanohoun et al. (1996) reported that S. rhombifolia grows to a height of 50–120 cm. In the present study, the height of S. rhombifolia reached approximately 86 cm by 75 DAT, with no significant increase observed thereafter, up to 120 DAT. In the case of stems, a correlation between the dry yield and the content of major compounds, 20HE and E was observed at different growth stages. The dry yield of the stems increased until 75 DAT, followed by a sharp decline between 75 and 90 DAT, reaching its lowest point at 90 DAT. Subsequently, the yield increased again, reaching 69.28 % at 120 DAT. Consistently, the contents of 20HE and E in the stems followed a similar trend. After 90 DAT, S. rhombifolia exhibited distinct semi-woody characteristics, which are presumed to be related to the changes in dry yield and major compound content observed at 90 DAT. Throughout the entire growth period, the average content of 20HE and E in the stems was 5.01 µg/g and 0.96 µg/g, respectively, and 2.28 µg/g and 0.37 µg/g in the leaves, indicating that the concentration of 20HE in the stems was approximately 5.23 times higher than that of E. Notably, the content of 20HE and E in the stems of S. rhombifolia consistently exceeded that in the leaves across all growth stages. These findings are consistent with those reported by Avula et al. (2008), which demonstrated that 20HE levels were approximately 3.48 times higher than E in S. rhombifolia based on LC-UV analysis. The activities of S. rhombifolia stems and leaves to improve BPH were evaluated in TP-activated RWPE-1 and LNCaP cells. The expression levels of AR, 5aR2, PCNA and PSA induced by TP were effectively attenuated by S. rhombifolia stems or leaves. 5αR2, an isozyme of 5αR, was primarily expressed in prostate tissue, where it converts T into DHT (Pais, 2010). The converted DHT is reported to be a factor inducing BPH by stimulating cell proliferation through its binding to AR (Nicholson et al., 2011). PCNA plays a crucial role in regulating cell cycles, including DNA replication and cell division (Strzalka et al., 2011). It has been reported that PCNA contributes to prostate hypertrophy by promoting the proliferation of prostate cells (Zhong et al., 2008). PSA is synthesized in the epithelial cells of the prostate and is commonly used as an indicator for diagnosing prostate cancer (Chung et al., 1996). Studies suggest that PSA levels increase with prostate enlargement compared to normal prostate sizes (Lee et al., 2008). The activities of S. rhombifolia stems and leaves in regulating AR, 5αR2, PCNA, and PSA expression varied according to the growth stage of S. rhombifolia. Particularly noteworthy, extract from S. rhombifolia stems significantly inhibited the expressions of AR, 5αR2, and PCNA after 90 DAT in RWPE-1 cells. Moreover, these extracts significantly inhibited the expression of 5αR2 and PSA after 90 DAT in LNCaP cells. Extracts from S. rhombifolia leaves exhibited inhibitory activity on AR, 5αR2, PCNA and PSA across all growth stages, with significant activity observed after 60 DAT, except for AR expression in both RWPE-1 and LNCaP cells. Based on these results, it was found that the extract of S. rhombifolia stems exhibited distinct variations in bioactivity depending on the growth stage, whereas the extract of leaves showed relatively consistent bioactivity throughout the entire growth period.
5 Conclusions
Over a 120-day period following transplantation, an evaluation of the growth characteristics of S. rhombifolia, the content of major compounds (20HE and E), and its regulatory activity on BPH-associated biomarkers was conducted. It was notably found that the height and stem diameter of S. rhombifolia increased until 52 DAT, after which the growth rate slowed. Additionally, the dry yield and compound content in the stems reached their lowest point at 90 DAT, followed by a sharp increase until 120 DAT. Consistent with these changes, the inhibitory activities of the stem extract on AR, 5αR2, and PCNA in RWPE-1 cells, as well as on 5αR2 and PSA in LNCaP cells, increased significantly after 90 DAT. Based on these results, it is suggested that the appropriate period to collect S. rhombifolia as an active substance to improve BPH might be after 90 DAT, considering the yield of the raw materials, the contents of metabolites, and the bioactivities.
CRediT authorship contribution statement
Min Gyu Park: Writing – original draft, Investigation, Formal analysis. Yun Na Kim: Investigation, Formal analysis. Jae sun Lee: Formal analysis. Yu Jung Kim: Formal analysis. Soo-Yong Kim: Resources. Sangho Choi: Resources. Min Hye Yang: Investigation, Methodology. Bong-Oh Kwon: Funding acquisition, Project administration, Writing – review & editing. Jung-Rae Rho: Methodology, Data curation, Supervision. Eun Ju Jeong: Writing – review & editing, Writing – original draft, Data curation, Conceptualization, Project administration, Supervision.
Acknowledgements
This research was supported by National Research Foundation of Korea (NRF) grants funded by the Ministry of Science, ICT and Future Planning (Grant Number NRF- 2023R1A2C1007448), and funded by a grant from “Development of living shoreline technology based on blue carbon science toward climate change adaption” of the Korea Institute of Marine Science & Technology Promotion (KIMST) funded by the Ministry of Ocean and Fisheries (KIMST-20220526).
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Kom, J., Keita, A., Mbeukam, T., Mbi, C. N., Mbiele, A. L., Mbome, I. L., Mubiru, N. K., Nancy, W. L., Nkongmeneck, A. B., Tabie, B., Sofowora, A., Tamze, V., Wirmum, C. K., 1996. Traditional Medicine and Pharmacopoeia: Contribution to Ethnobotanical and Floristic Studies in Cameroon. OUA/STRC: Lagos, 301.
- Anti-hyperglycemic, antioxidant, and anti-inflammatory activities of extracts and metabolites from Sida acuta and Sida rhombifolia. Química Nova.. 2017;40:176-181.
- [CrossRef] [Google Scholar]
- Quantitative determination of ecdysteroids in Sida rhombifolia L. and various other Sida species using LC-UV, and their anatomical characterization. Nat. Prod. Commun.. 2008;3(5):1934578X0800300508
- [CrossRef] [Google Scholar]
- Dihydrotestosterone and the concept of 5α-reductase inhibition in human benign prostatic hyperplasia. World J. Urol.. 2002;19:413-425.
- [CrossRef] [Google Scholar]
- The development of human benign prostatic hyperplasia with age. J. Urol.. 1984;132(3):474-479.
- [CrossRef] [Google Scholar]
- Medicinal plants: Influence of environmental factors on the content of secondary metabolites. Medicinal Plants and Environmental Challenges.. 2017;259–277
- [CrossRef] [Google Scholar]
- First record of Sida rhombifolia L. (Malvaceae) for Italian flora: taxonomical and ecological investigation. Acta Bot. Croat.. 2022;81(2):159-167.
- [CrossRef] [Google Scholar]
- The role of dihydrotestosterone in benign prostatic hyperplasia. Urology. 2003;61(4):2-7.
- [CrossRef] [Google Scholar]
- Chaves, O. S., Gomes, R. A., Tomaz, A. C. D. A., Fernandes, M. G., Das Graças Mendes Junior, L., de Fátima Agra, M., de Fátima Vanderlei de Souza, M. 2013. Secondary Metabolites from Sida rhombifolia L. (Malvaceae) and the Vasorelaxant Activity of Cryptolepinone. Molecules. 18(3), 2769-2777. doi: 10.3390/molecules18032769.
- Alkaloids and Phenolic Compounds from Sida rhombifolia L. (Malvaceae) and Vasorelaxant Activity of Two Indoquinoline Alkaloids. Molecules. 2017;22(1):94.
- [CrossRef] [Google Scholar]
- The Relationship of Cellular Composition of Benign Prostatic Hyperplasia and Prostate-Specific Antigen (PSA) Levels. Korean J. Urol. 1996:1093-1096.
- [Google Scholar]
- Evaluation of the therapeutic effect against benign prostatic hyperplasia and the active constituents from Epilobium angustifolium L. J. Ethnopharmacol.. 2019;232:1-10.
- [CrossRef] [Google Scholar]
- Hepatoprotective activity of Sida rhombifolia ssp. retusa against thioacetamide and allyl alcohol intoxication in rats. PhOL.. 2006;3:259-266. 5 Jun 2024
- [Google Scholar]
- Antihyperglycemic activity of root bark of Polyalthia longifolia var. pendula and aerial parts of Sida rhombifolia Linn. and its relationship with antioxidant property. Asian J. Chem.. 2011;23(1):141. 5 Jun 2024
- [Google Scholar]
- Effect of Natural products isolated from three species of Sida on some Gram positive and Gram negative bacteria. J. Indian Chem. Soc.. 1988;65:74-76.
- [Google Scholar]
- Anti-arthritic activity of various extracts of Sida rhombifolia aerial parts. Nat. Prod. Res.. 2009;23(8):689-695.
- [CrossRef] [Google Scholar]
- Ecdysteroid Glycosides from Sida rhombifolia L. Chem. Biodiversity.. 2007;4(9):2225-2230.
- [CrossRef] [Google Scholar]
- The effects of Sida rhombifoia L. on benign prostatic hyperplasia in testosterone propionate-induced animal model and its active constituents. Planta Med.. 2019;85(18):P-392.
- [CrossRef] [Google Scholar]
- Characterization of type I 5α-reductase activity in DU145 human prostatic adenocarcinoma cells. J. Steroid Biochem. Mol. Biol.. 1996;58(2):195-205.
- [CrossRef] [Google Scholar]
- The Effect of Dutasteride on Serum Prostate-specific Antigen (PSA) in Patients with Benign Prostate Hypertrophy. Int Neurourology J.. 2008;12(1):42-47. accessed 5 Jun 2024
- [Google Scholar]
- Voratins A–C: Pyridinium Alkaloids from the Marine Dinoflagellate Effrenium voratum with Inhibitory Effects on Biomarkers for Benign Prostatic Hyperplasia. J. Nat. Prod.. 2022;85(6):1495-1502.
- [CrossRef] [Google Scholar]
- Epidemiology of clinical benign prostatic hyperplasia. Asian J. Urol.. 2017;4(3):148-151.
- [CrossRef] [Google Scholar]
- Androgens and estrogens in benign prostatic hyperplasia: past, present and future. Differentiation. 2011;82(4–5):184-199.
- [CrossRef] [Google Scholar]
- Potency of a novel saw palmetto ethanol extract, SPET-085, for inhibition of 5α-reductase II. Adv. Ther.. 2010;27:555-563.
- [CrossRef] [Google Scholar]
- A simple and rapid HPLC-DAD method for simultaneously monitoring the accumulation of alkaloids and precursors in different parts and different developmental stages of Catharanthus roseus plants. J. Chromatogr. B. 2016;1014:10-16.
- [CrossRef] [Google Scholar]
- The influence of environmental conditions on secondary metabolites in medicinal plants: A literature review. Chem. Biodiv.. 2021;18(11)
- [CrossRef] [Google Scholar]
- Ellagic acid improves benign prostate hyperplasia by regulating androgen signaling and STAT3. Cell Death Dis.. 2022;13(6):554.
- [CrossRef] [Google Scholar]
- Park, M. G. 2023. Variations in metabolites content and bioactivity according to the growth stages of sida rhombifolia. Thesis, Gyeongsang National University, 2023.
- Prospective study of serum dihydrotestosterone and subsequent risk of benign prostatic hyperplasia in community dwelling men: the rancho bernardo study. The Urol.. 2010;184(3):1040-1044.
- [CrossRef] [Google Scholar]
- Prakash, A., Varma, R. K., Ghosal, 1981. Alkaloid Constituents of Sida acuta, S. humilis, S. rhombifolia and S. spinos S. Planta Medica. 43(12), 384-388. doi: 10.1055/s-2007-971529.
- Systematic review and meta-analysis of prostatic artery embolisation for lower urinary tract symptoms related to benign prostatic hyperplasia. Clin. Radiol.. 2017;72(1):16-22.
- [CrossRef] [Google Scholar]
- Pathology of benign prostatic hyperplasia. Int. J. Impotence Res.. 2008;20(3):S11-S18.
- [CrossRef] [Google Scholar]
- Benign prostatic hyperplasia and lower urinary tract symptoms. N. Engl. J. Med.. 2012;367(3):248-257.
- [CrossRef] [Google Scholar]
- Proliferating cell nuclear antigen (PCNA): a key factor in DNA replication and cell cycle regulation. Ann. Bot.. 2011;107(7):1127-1140.
- [CrossRef] [Google Scholar]
- Verma, V. A. N. D. A. N. A., Kasera, P. K. 2007. Variations in secondary metabolites in some arid zone medicinal plants in relation to season and plant growth. Indian J. Plant Physiol. 12(2), 203. doi: 10.5555/20073254617.
- Structural characterization and identification of ecdysteroids from Sida rhombifolia L. in positive electrospray ionization by tandem mass spectrometry. Rapid Commun. Mass Spectrom.. 2008;22(16):2413-2422.
- [CrossRef] [Google Scholar]
- Establishment of a Novel Model for Studying the Effects of Extracts of Chinese Herb Medicine on Human Type II 5α-Reductase in Vitro. Yakugaku Zasshi. 2010;130(9):1207-1214.
- [CrossRef] [Google Scholar]
- Kaempferol inhibits benign prostatic hyperplasia by resisting the action of androgen. Eur. J. Pharmacol.. 2021;907:174251
- [CrossRef] [Google Scholar]
- Quality-of-life impact of lower urinary tract symptom severity: results from the Health Professionals Follow-up Study. Urology. 2002;59(2):245-250.
- [CrossRef] [Google Scholar]
- Evaluation of antibacterial activities of compounds isolated from Sida rhombifolia Linn. (malvaceae). Nat Prod Chem Res.. 2012;1(101):2.
- [CrossRef] [Google Scholar]
- The restudying of naturalized plants in Jeju Island. Korean J. Plant Res.. 2005;18(2):325-336. 5 Jun 2024
- [Google Scholar]
- Shape-up LUTS/BPH guideline in Korea: Medical treatment. J Korean Med Assoc.. 2008;51(7):672-674.
- [CrossRef] [Google Scholar]
- Metabolomic profiling of carotenoid constituents in Physalis peruviana during different growth stages by LC‐MS/MS Technology. J. Food Sci.. 2019;84(12):3608-3613.
- [CrossRef] [Google Scholar]
- Ki-67 and PCNA expression in prostate cancer and benign prostatic hyperplasia. Clin. Invest. Med.. 2008;31(1):E8-E15.
- [CrossRef] [Google Scholar]







