Translate this page into:
Wnt/GSK3β/β-catenin signaling pathway regulates calycosin-mediated anticancer effects in glioblastoma multiforme cells
⁎Corresponding author at: The Second Hospital of HeBei Medical University, No. 215, Heping, West Road, Xinhua District, Shijiazhuang 050011, China. songjianok@sohu.com (Jian Song)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Calycosin, an O-methylated isoflavone, has been widely reported to induce anticancer activity in different cancer cells in vitro. Nonetheless, the associated mechanism of calycosin in glioblastoma multiforme cells (U87) still remains unknown. To explore the anticancer effects, the apoptotic mechanism of calycosin via Wnt/GSK3β/β-catenin signaling was explored in U87 cells. Different assays including: cytotoxicity, free radical determination, SOD and CAT activity, GSH content, qPCR, mitochondrial membrane potential, caspase activity, and western blotting assays were performed. It was shown that calycosin mitigated cell viability in U87 cells, whereas it showed no apparent effect on BV2 microglial cells. Calycosin triggered apoptosis via upregulating the mitochondria-associated caspase pathway in U87 cells. Calycosin induced the reduction of the mitochondrial membrane potential, overexpression of Bax, downexpression of Bcl-2, and activation of caspase-9 and caspase-3. Calycosin-stimulated apoptosis was associated with the upregulation of free radical scavenging through the modulation of antioxidant enzymes, such as SOD and CAT as well as the level of GSH. The apoptotic activity of calycosin was mediated by suppression of pGSK-3βser9, β-catenin, and c-Myc at protein level. The present study suggested that calycosin triggers U87 cell death through an antioxidant effect mediated by Wnt/GSK3β/β-catenin signaling pathway.
Keywords
Glioblastoma multiforme cells
Calycosin
Anticancer
Antioxidant
Wnt/GSK3β/β-catenin signaling pathway
1 Introduction
Glioblastoma multiforme is known as the most common group of primary brain tumors, and malignant gliomas are the deadliest type of adult brain tumors (Urbańska et al., 2014). Glioblastoma accounts for more than 50 % of all glioma types (Urbańska et al., 2014). The overall incidence of tumors is 2–3 people in a hundred thousand, and 20 % of all intracranial tumors account for over sixty years and is more common in men (Urbańska et al., 2014). In cell studies, glioblastoma multiforme cells (U87) show stare shape morphology with different sizes (Jaworski et al., 2013). They have a high proliferation rate which increases the access of tumor cells to different areas of the brain (Jara et al., 2021). This type of tumor has a poor prognosis with clinical characteristics such as cognitive impairment, personality changes, headache, loss of sensation, and lack of balance in walking (Hu et al., 2021; Shah and Kochar, 2018). Despite increasing our knowledge in understanding the pathophysiology of glioblastoma and advances in regarding treatment, cancer continues to be incurable, because despite conventional therapies involving removal of the tumor with surgery followed by radiation therapy plus drug treatment, patients usually die within 12 to 15 months after surgery. One of the potential treatments of glioma cell could be induction of cytotoxicity and apoptosis to prohibit the proliferation of cancer cells (Afshari et al., 2019; Zhu et al., 2019). Therefore, the need for new therapies or drugs is considered absolutely necessary. Natural products derived from plants are considered as valuable sources of modern medicinal raw materials (Musthafa et al., 2021 Jan; Shah et al., 2020; Gezici and Şekeroğlu, 2019).
Plant-derived natural products include paclitaxel, etoposide, vinblastine, and vincristine, all of which have been shown to treat a variety of cancers (Gezici and Şekeroğlu, 2019).
Radix astragali is a plant used in Chinese traditional medicine with potential anticancer (Zhuang et al., 2019; Yang et al., 2021), immunomodulatory (Chen et al., 2020), and diuretic properties (Liu et al., 2013).
Calycosin, an O-methylated isoflavone (C16H12O5), is a phytoestrogenic compound in the dry root extract of Radix astragali (Gao et al., 2014; Gong et al., 2021). Several studies have shown that calycosin has antioxidant, antimicrobial, antiviral, antifungal, anticancer, and anti-diabetic properties (Gao et al., 2014; Gong et al., 2021; Deng et al., 2020).
Previous studies have shown that calycosin triggers apoptosis in several cancer cell lines such as cervical cancer cell (Zhang et al., 2020), gastric cancer cell (Zhang et al., 2021), osteosarcoma cell line (Tian et al., 2020), glioma cancer cell (Ni et al., 2019), and so on. Although calycosin exerts apoptosis in colorectal cancer cell, breast cancer cell, cervical cancer cell, and osteosarcoma cell line through regulation of ERβ/miR-17 (Chen et al., 2015), WDR7-7-GPR30 (Tian et al., 2017), miR-375 (Zhang et al., 2020), and ERβ‑mediated PI3K/Akt (Tian et al., 2020) signaling pathways, respectively, its anticancer mechanism still remains unknown in glioblastoma cancer cells. Thus, in the present investigation, the role of Wnt/GSK3β/β-catenin signaling pathway was explored in calycosin-triggered apoptosis in U87 glioblastoma multiforme cells using cytotoxicity, fluorimeter, qPCR, and western blotting assays.
2 Materials and methods
2.1 Cell culture
Human malignant glioblastoma multiforme (U87) cells and BV2 microglial cells were obtained from the American Type Culture Collection (Rockville, MD, USA) and cultured in Dulbecco's modified Eagle's medium (DMEM) supplemented with heat-inactivated fetal calf serum (FCS, 10 %), and antibiotics and maintained at 37 °C with 5 % CO2.
2.2 Cell viability
The influence of calycosin on the viability of cells was explored employing the MTT analysis. Cells were incubated with calycosin at various concentrations in the range of 1–100 µg/ml for 24 h. 30 μl MTT stock solution (4 mg/ml) was then added to each well and incubated for 4 h followed by removal of the media. Finally, formazan crystals were dissolved in 150 μl DMSO and the absorbance of the samples was read at 570 nm using a scanning multi-well spectrophotometer.
2.3 Detection of superoxide radical
Formation of superoxide radical in U87 cells was quantified by dihydroethidium (DHE) staining, as reported in the literature (Heikkilä et al., 2010). Briefly, the cells (1 × 106) were incubated with IC50 concentration of calycosin for 24 h, collected, washed, and resuspended in reaction mixture (1 ml) supplemented Dihydroethidium (DHE, 10 μM). Finally, the fluorescence intensity of the probes was determined by microplate fluorimeter (Synergy Hybrid Microplate Reader, BioTek).
2.4 Detection of hydroxyl radical
Hydroxyphenyl fluorescein (HPF), as a cell-permeable fluorescent probe was used to specifically determine the hydroxyl radical formation (Zunino et al., 2007). The cells (1 × 106) treated with IC50 concentration of calycosin for 24 h were added by 10 μM HPF for an additional 2 h at 28 °C. After further washing, fluorescence of the probes was determined by microplate fluorimeter (Synergy Hybrid Microplate Reader, BioTek).
2.5 Intracellular and mitochondrial ROS measurements
Quantification of intracellular and mitochondrial ROS generation was carried out by using 2,7 dichlorodihydrofluorescein diacetate (DCFH2-DA) and dihydrorhodamine (DHR)-123 probes as reported previously (Kang et al., 2017). Briefly, after treating cells with IC50 concentration of calycosin for 24 h, 30 μM DCFH2-DA or DHR-123 was added to cells for an additional 40 min at 37 °C. After washing, the fluorescence signal of the probe was detected using a microplate fluorimeter (Synergy Hybrid Microplate Reader, BioTek).
2.6 Measurement of superoxide dismutase (SOD) activity
After trading with IC50 concentration of calycosin, the cells were washed, scraped, suspended in 10 mM phosphate buffer (pH 7.5), lysed, centrifuged, and the protein content of the supernatant was explored. SOD activity was then determined by assessing the oxidation inhibition of epinephrine at 480 nm using a UV/VIS spectrophotometer as determined previously (Carrillo et al., 1991).
2.7 Measurement of catalase (CAT) activity
After preparing the samples as described above, 40 μg of protein were added to the phosphate buffer (PBS) (50 mM, pH 7.0), supplemented with H2O2 (100 mM). After 2 min of incubation, absorbance was read at 240 nm for a period of 5 min based on the previous report (Carrillo et al., 1991).
2.8 Detection of glutathione (GSH) level
The intracellular GSH level was quantified with CMAC as a GSH-sensitive fluorescence probe. After treating, the cells were treated with CMAC (10 μM) for 30 min and CMAC fluorescence intensity was analyzed with a microplate fluorimeter (Synergy Hybrid Microplate Reader, BioTek) (Tauskela et al., 2000).
2.9 Determination of apoptosis by Annexin-V
The level of apoptosis was explored by Annexin-V FITC as a sensitive fluorescence probe. After treating, the cells were added by relevant buffers and 50 nM Annexin-V-FITC for 10 min in the dark based on the kit manufacturer’s protocols (ab14085). The fluorescence signal of the probe was detected using a microplate fluorimeter (Synergy Hybrid Microplate Reader, BioTek) with exciation and emission at 480 and 532 nm, respectively.
2.10 Quantitative real-time PCR (qPCR) assay
After treatment with IC50 concentration of calycosin, the cells were lysed with TRIzol reagent (Invitrogen) to extract total RNA, where its purity was explored using nanodrop (ND-1000; Thermo Scientific) at an absorbance of 260 nm. RNA samples were stored at − 20 °C. cDNA was then synthesized by the Reverse Transcription Kit (Invitrogen). qPCR assay was done for Bax, Fw: TCTGACGGCAACTTCAACTG, Rev, TTGAGGAGTCTCACCCAACC and Bcl2, Fw: GGATGCCTTTGTGGAACTGT, Rev: AGCCTGCAGCTTTGTTTCAT using applied biosystems (USA) according to the manufacturer’s protocols (Alarifi et al., 2017; Livak and Schmittgen, 2001). The analysis and the procedures were done based on the previous studies. GAPDH was also used as the housekeeping gene.
2.11 Detection of mitochondrial membrane potential
Mitochondrial membrane potential was detected based on using JC-1 as a fluorescence probe. After treating, the cells were added by JC-1 (200 nM) and incubated for 20 min at 37 °C. After washing with PBS, the fluorescence signal of the probe was detected using a microplate fluorimeter (Synergy Hybrid Microplate Reader, BioTek).
2.12 Caspase assay
Caspase-9 (ab65608) and caspase-9 (39401) activities were assessed based on the kit manufacturer’s protocols. Briefly, the protein samples were added to the wells, followed by addition of reaction buffer and relevant substrate and incubation for 60–120 min at 37 °C. The OD of samples were read at 450 nm using a scanning multi-well spectrophotometer.
2.13 Western blot analysis
Whole cell lysates from the U87 cells incubated with calycosin were prepared using a lysis buffer based on the previous study (Park et al., 2013). After treating and sample proration as described above either, 40 μg of proteins were boiled, electrophoresed (10 % SDS-polyacrylamide gel), transferred onto nitrocellulose membranes, incubated with primary antibodies (1:1000, Santa Cruz Biotechnology), and incubated with secondary anti-immunoglobulin-G-horseradish peroxidase conjugates (1:2000) (Pierce, Rockford, USA). Protein bands were then detected employing an increased chemiluminescence western blotting detection kit (Amersham, Little Chalfont, UK).
2.14 Statistical analysis
All assays were carried out in triplicate and data represent the mean ± standard deviation (SD) of the mean (SEM). The outcomes were reported based on the analysis of variance (ANOVA) using Tukey's test to determine differences. P < 0.05 was considered significant.
3 Results
3.1 Calycosin selectively mitigates the growth of U87 cells
The effect of calycosin on the proliferation of U87 cells was assessed at 24 h employing the MTT assay, and IC50 value was quantified. At 48 h, calycosin showed cytotoxic effects in a concentration-dependent manner. As depicted in Fig. 1a, cell viability was reduced by calycosin as follows: 96.84 ± 13.43 % at 1 μg/ml, 93.81 ± 13.47 % at 5 μg/ml, 75.47 ± 10.47 % at 10 μg/ml, 65.86 ± 12.79 % at 20 μg/ml, 42.27 ± 4.30 % at 50 μg/ml, and 34.94 ± 4.66 % at 100 μg/ml. Among the studied concentrations, the IC50 concentration was determined to be around 43.71 μg/ml and this concentration of calycosin was used for further experiments. The cytotoxicity of calycosin in normal microglial cells (BV2) was also explored. As exhibited in Fig. 1b, the viability of calycosin-incubated BV2 cells was higher than that of calycosin-incubated U87 cells. Since calycosin is known as a potential antioxidant small molecule, the influence of known antioxidant compounds including vitamin C (Vit. C) and N-acetylcysteine (NAC) on U87 cell viability was explored. As displayed in Fig. 1c, Vit. C and NAC, decreased U87 cell viability compared with that in calycosin-treated cells.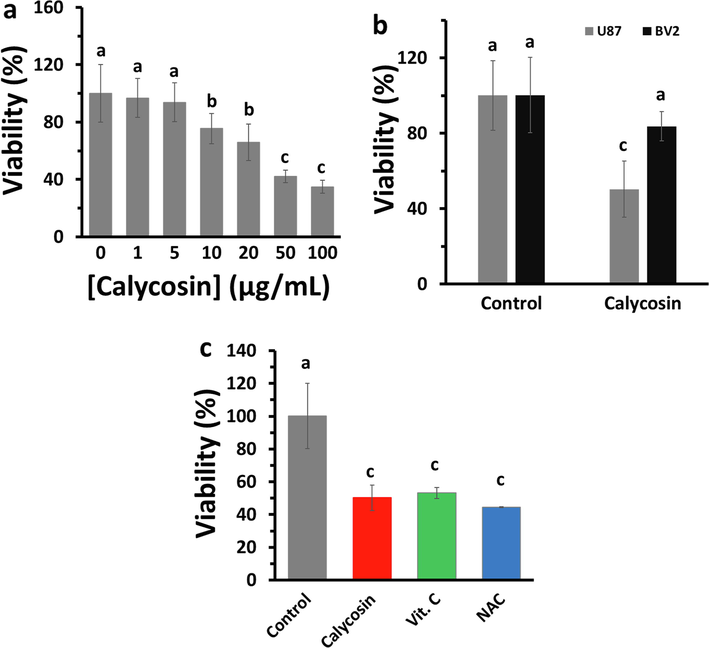
Cytotoxic influences of calycosin on cells. (a) U87 cells were treated with various concentrations of calycosin for 24 h, and cell viability was determined by the MTT assay to explore IC50 value. (b) The cell viability of calycosin-incubated (IC50: 43.71 μg/ml) U87 and BV2 cells were explored at 24 h employing the MTT assay. (c) The cell viability of vitamin c (Vit. C) and N-acetylcysteine (NAC) as antioxidant compounds in U87 with similar concentration of 43.71 μg/ml was determined using the MTT assay. a, b, c letters represent the significance differences at aP < 0.05, bP < 0.01, cP < 0.001 relative to control.
3.2 Calycosin displays ROS scavenging properties
To examine the ROS scavenging properties of calycosin, the generation of superoxide and hydroxyl radical was determined. The superoxide radical and the hydroxyl radical produced in cells were determined via microplate fluorimeter. The outcomes showed that the superoxide radical signal was 828.66 ± 59.96 in the control sample, however, the superoxide radical signal was reduced to 387.66 ± 33.00 (Fig. 2a) in the calycosin-incubated sample. The hydroxyl radical signal was 641.33 ± 53.40 in the control group, however, the hydroxyl radical signal decreased to 268.66 ± 31.34 in the calycosin-incubated sample (Fig. 2b).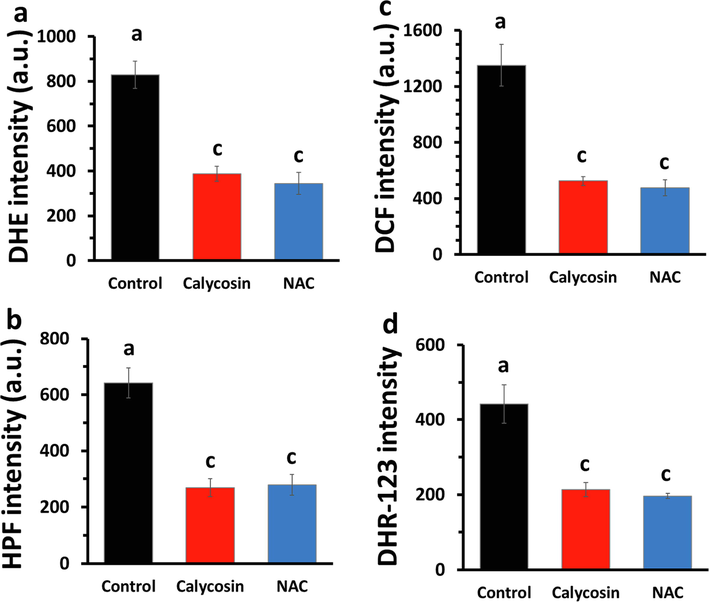
ROS scavenging effect of calycosin (IC50: 43.71 μg/ml) in U87 cells after 24 h. (a) Superoxide radical scavenging assay. (b) Hydroxyl radical scavenging assay. (c) Intracellular ROS. (d) Mitochondrial ROS. All assays were done using microplate fluorimeter analyzing the relevant fluorescence probes. a, c letters represent the significance differences at aP < 0.05, cP < 0.001 relative to control.
The scavenging effect of calycosin on intracellular ROS in U87 cells was analyzed. As depicted in Fig. 2c, IC50 concentration of calycosin significantly reduced the intensity of the DCF signal relative to that in the control sample. To assess the scavenging properties of calycosin on mitochondrial ROS in U87 cells, the DHR 123 fluorescence probe assay was explored to analyze mitochondrial ROS in cells incubated with or without calycosin. The data showed that calycosin treatment reduced the level of mitochondrial ROS relative to that in control cells (Fig. 2d). Consistently, in all experiments, NAC as an antioxidant agent, apparently decreased the intensity of the oxidant agents compared with those in calycosin-treated cells (Fig. 2).
3.3 Effects of calycosin on antioxidant enzymes and GSH levels in U87 cells
Analysis of the influence of calycosin on the activity of antioxidant enzymes including SOD and CAT showed that calysocin enhanced the levels of SOD and CAT (Fig. 3a) activity. Indeed, the activity of SOD and CAT increased upon treatment of U87 cells with IC50 concentration of calycosin for 24 h (Fig. 3a). As shown in Fig. 3a, NAC also increased the activity of SOD and CAT compared with that in calycosin-treated cells.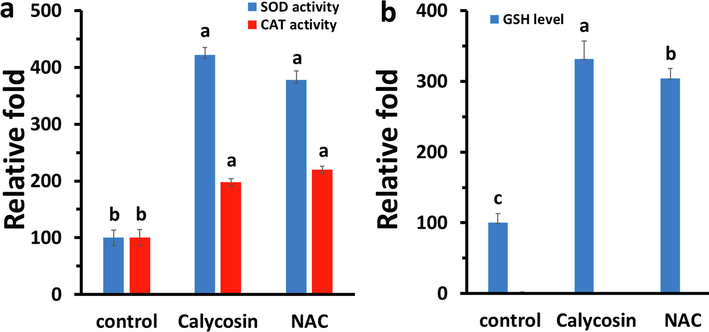
Effects of calycosin (IC50: 43.71 μg/ml) on antioxidant enzymes and cellular GSH content after 24 h. (a) Relative SOD and CAT activity. (b) Relative GSH content as determined by microplate fluorimeter. a, b, c letters represent the significance differences at aP < 0.05, bP < 0.01, cP < 0.001 relative to control.
GSH is known as one of the most crucial intracellular antioxidants, and exploration of alterations in the level of GSH can be used as a potential method to analyze the oxidative stress within cells (Tauskela et al., 2000). The GSH-activated fluorescent probe, CMAC, was used as a sensitive probe to explore the level of intracellular GSH (Tauskela et al., 2000). As depicted in Fig. 3b, cells incubated with calycosin at 43.71 μg/ml and labeled with CMAC demonstrated higher fluorescence intensity of cellular GSH than control sample assessed using microplate fluorimeter. This pattern was also comparable with NAC, as a potential antioxidant.
3.4 Calycosin triggers apoptosis by the mitochondrial-mediated pathway
To explore the cytotoxic effect of calycosin via apoptosis, fluorescence, expression of Bax and Bcl-2 mRNA, mitochondrial membrane potential, and caspase activity assays were done. The Annexin-V FITC data in Fig. 4a showed that calycosin and NAC-incubated cells had significant increase in the Annexin-V FITC fluorescence intensity relative to control cells, which is a feature of apoptosis. In addition, the effect of calycosin on apoptosis in U87 cells was confirmed by qPCR analysis of the antiapoptotic and proapoptotic proteins, Bcl-2 and Bax, respectively. As shown in Fig. 4b, calycosin upregulated the expression of Bax mRNA but downregulated the expression of Bcl-2 mRNA. As shown in Fig. 4c, calycosin caused the loss of mitochondrial membrane potential. The level of caspase-9 activity was examined because this enzyme is activated during mitochondrial membrane disruption (Kang et al., 2017). As shown in Fig. 4d, the levels of caspase-9 and caspase-3 activities were higher in the calycosin and NAC-treated cells than those in the control sample.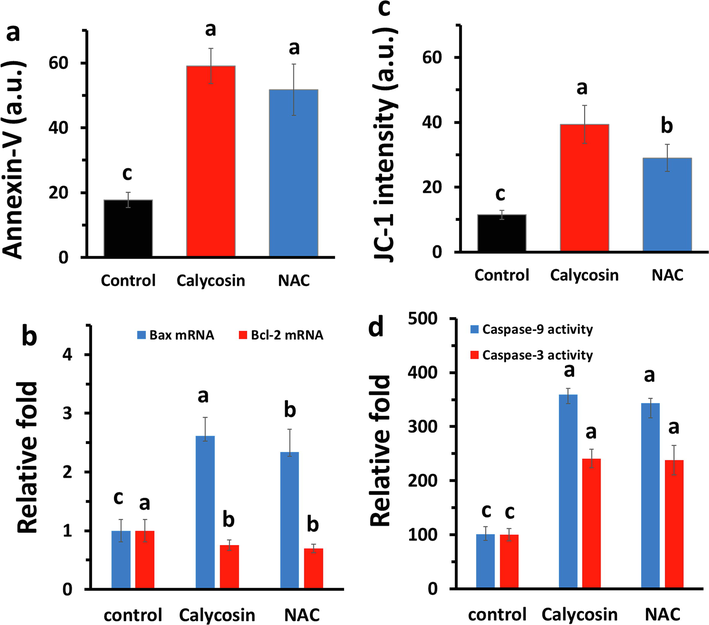
Effects of calycosin (IC50: 43.71 μg/ml) on mitochondria-mediated apoptosis in U87 cells after 24 h. (a) Annexin-V FITC assay was done by microplate fluorimeter. (b) qPCR assay for exploring the expression of Bax mRNA and Bcl-2 mRNA. (c) Determination of membrane potential by associated probe (JC-I dye) on a microplate fluorimeter. (d) Relative caspase-9 and caspase-3 activity as carried out by methods described. a, b, c letters represent the significance differences at aP < 0.05, bP < 0.01, cP < 0.001 relative to control.
It was seen that the data obtained for calycosin were almost comparable with NAC, as a potential antioxidant.
3.5 Effect of calycosin on the Wnt/GSK3β/β-catenin signaling pathway
Glycogen synthase kinase 3 (GSK3) as a modulator of glycogen pathway plays some major roles in protein metabolism, cell growth, cell differentiation and dynamics, cell viability, and apoptosis (Park et al., 2013). GSK3 is also involved in the modulation of Wnt/β-catenin signaling process (Park et al., 2013).
It was shown that calycosin reduced the expression of phosphorylated GSK-3β which is the inactivated form of GSK-3β. In fact, phosphorylation of GSK-3β at serine (Ser9) can result in destabilization of β-catenin and block the transcription of target proteins like c-Myc. The vital components play a key role in Wnt signaling pathway are GSK-3β, β-catenin and c-Myc. Here we observed that calycosin resulted in the phosphorylation of GSK-3β (ser-9), which led to reduction in the expressions of β-catenin in U87 cells (Fig. 5). Also, the downstream factors of β-catenin, c-Myc, was downregulated in calycosin-treated U87cells.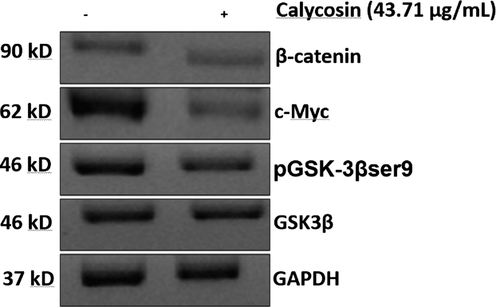
Effects of calycosin (IC50: 43.71 μg/ml) on the Wnt/GSK3β/β-catenin signaling pathway in U87 cells after 24 h. western blot analysis of cell lysate as described in 2–14 section.
This data indicated that calycosin-triggered anticancer effects are modulated through the Wnt/GSK3β/β-catenin signaling pathway.
Indeed, the protein levels of the Wnt/β-catenin signaling pathway related proteins (GSK-3β, β-catenin and c-Myc) showed that the levels of these proteins were downregulated in calycosin-treated glioma cells.
4 Discussion
Oxidative stress is a phenomenon that occurs at the cellular level and can worsen the condition of healthy cells and lead to their death (Poillet-Perez et al., 2015). Cancer cells often use this phenomenon to its advantage, but can oxidative stress, which is a major cause of cancer, be used against the disease? ROS are substances that are produced naturally after the process of oxygen metabolism. These species usually play an important role in regulating biological function (homeostasis) as well as in cell signaling. But as long as ROS reach abnormal levels, they cause oxidative stress, which in turn leads to cellular aging and worsening of the condition. (Poillet-Perez et al., 2015) Unlike healthy cells, cancer cells need a much higher level of ROS, which allows them to grow and spread rapidly (Kang et al., 2015). In cancer cells, an increase in ROS causes by increased metabolic activity, mitochondrial dysfunction, peroxisome activity, increased receptor-mediated signaling, activation of oncogenes, increased activity of oxidases, cyclooxygenases, lipoxygenases, and thymidine phosphorylase (Storz, 2005). Expression of transformation-related genes such as Bcr–Abl, Ras, and c-Myc oncogenes induces ROS production (Lu et al., 2007), which is essential for the tumorigenic function of some of these oncogenes. K-Ras oncogenic mutations increase the amount of cellular ROS and stimulate cell proliferation by affecting the activity of the protein kinase A pathway and increasing c-AMP (Shirazi et al., 2020). Increased expression of c-Myc oncogene also induces DNA damage, p53 activation and increased ROS levels (Vafa et al., 2002). Also, The increased oxidative stress presents in cancer cells is caused not only by ROS overproduction but also from inactivation of antioxidant enzymes and compounds (Lu et al., 2007). Calycosin has shown anticancer activity associated with its role as inhibition of migration and invasion of cancer cells (Li et al., 2017) and the induction of cell cycle arrest and apoptosis (Liu et al., 2021).
It has been also shown that calycosin induces anticancer effects through a ROS-mediated signaling pathway in hepatocellular carcinoma cells (Liu et al., 2021) and gastric cancer cell (Zhang et al., 2021).
The Wnt family is secreted glycoproteins with several enriched and protected cysteine fragments that are involved in a variety of processes such as embryonic induction, cell proliferation, and cell signaling (Barker and Clevers, 2000). Wnt signals are preceded as a result of the binding of Wnt proteins to Frizzled related proteins. So far, 19 types of Wnt protein and 10 types of Frizzled receptors have been identified, which has led to the complexity of the Wnt signaling pathway (Barker and Clevers, 2000). The Wnt signaling pathway is induced through three intracellular pathways, which include the 1-main Wnt / β-catenin pathway, 2- Wnt / polarity pathway, and the 3-pathway Wnt / Ca2+ pathway. In the Wnt / β-catenin pathway, binding of Wnt to a receptor induces hyperphosphorylation of cytoplasmic protein called dishevelled and leads to inhibition of serine-threonine kinase called as GSK3β (Barker and Clevers, 2000). This eventually leads to the release and stabilization of catenin-β, resulting in its increase in the cytoplasm of the cell. β-catenin enters the nucleus and interacts with transcription factors including Lef / Tcf and regulates the expression of target genes including cyclin D1 and c-Myc (Barker and Clevers, 2000).
Polyphenolic antioxidants are known as potential ROS scavengers and regulators of different signaling pathways (Gupta et al., 2014). Calycosin is an isoflavonoid that contains three aromatic rings. The ROS-mediated tumor suppressor activities of calycosin were found to be mediated in part by the regulation of Wnt/GSK3β/β-catenin signaling pathway, which in turn this process have been shown to regulate the expression of specific oncogenes, which may be a possible reason for selective anticancer effects of calycosin. In agreement with this study, it has been also shown that calycosin depicts tumor suppressor activities through upregulation of antioxidant enzymes, reduction of ROS, and induction of apoptosis (Kang et al., 2017).
Calycosin has a unique structure due to the presence of the methyl group. Indeed, despite a number of potential functions of flavonoids, medical applications have not been fully successful when explorations were extended to the in vivo, especially in humans. Weak stability and low bioavailability of dietary polyphenols are the main drawbacks behind these concerns (Koirala et al., 2016). Researches have shown that the methylation of flavonoids may enhance their potential as pharmaceutical compounds causing promising implementations (Koirala et al., 2016). Indeed, conjugation of calycosin with other compounds can control its potency to induce several protective effects through different signaling pathways (Jian et al., 2015).
5 Conclusion
The present study revealed that calycosin triggered apoptosis in U87 cells through a ROS-mediated mitochondrion-associated caspase activation pathway via Wnt/GSK3β/β-catenin mechanism. We used several approaches to determine the ROS scavenging and anticancer effects of calycosin, and the results indicated that calycosin has potential antioxidant and anticancer activities against U87 cells. Taken together, the outcomes of the present report indicate that calycosin-mediated apoptosis is linked with the antioxidant features of calycosin.
Conflicts of interest
None.
Acknowledgement
This study was supported by Hebei Natural Science Foundation (NO. H2019206710).
References
- Auraptene-induced cytotoxicity mechanisms in human malignant glioblastoma (U87) cells: role of reactive oxygen species (ROS) EXCLI journal.. 2019;18:576.
- [Google Scholar]
- Regulation of apoptosis through bcl-2/bax proteins expression and DNA damage by nano-sized gadolinium oxide. International journal of nanomedicine.. 2017;12:4541.
- [Google Scholar]
- (−) Deprenyl induces activities of both superoxide dismutase and catalase but not of glutathione peroxidase in the striatum of young male rats. Life sciences.. 1991;48(6):517-521.
- [Google Scholar]
- Astragali Radix (Huangqi): a promising edible immunomodulatory herbal medicine. Journal of ethnopharmacology.. 2020;10(258):112895
- [Google Scholar]
- Calycosin induces apoptosis by the regulation of ERβ/miR-17 signaling pathway in human colorectal cancer cells. Food & function.. 2015;6(9):3091-3097.
- [Google Scholar]
- Calycosin: a Review of its Pharmacological Effects and Application Prospects. Expert Review of Anti-infective Therapy.. 2020;22:1-5.
- [Google Scholar]
- Pharmaceutical properties of calycosin, the major bioactive isoflavonoid in the dry root extract of Radix astragali. Pharmaceutical biology.. 2014;52(9):1217-1222.
- [Google Scholar]
- Gezici, S, Şekeroğlu, N. 2019. Current perspectives in the application of medicinal plants against cancer: novel therapeutic agents. Anti-Cancer Agents in Medicinal Chemistry (Formerly Current Medicinal Chemistry-Anti-Cancer Agents). 19(1), 101-11.
- Pharmaceutical Values of Calycosin: One Type of Flavonoid Isolated from Astragalus. Evidence-Based Complementary and Alternative Medicine.. 2021;7:2021.
- [Google Scholar]
- Oxidative stress and antioxidants in disease and cancer: a review. Asian Pacific journal of cancer prevention.. 2014;15(11):4405-4409.
- [Google Scholar]
- Cytotoxicity study of ordered mesoporous silica MCM-41 and SBA-15 microparticles on Caco-2 cells. European Journal of Pharmaceutics and Biopharmaceutics.. 2010;74(3):483-494.
- [Google Scholar]
- Molecular and clinical characterization of UBE2S in glioma as a biomarker for poor prognosis and resistance to chemo-radiotherapy. Frontiers in oncology. 2021:11.
- [Google Scholar]
- Vitamin C deficient reduces proliferation in a human periventricular tumor stem cell-derived glioblastoma model. Journal of Cellular Physiology.. 2021;236(8):5801-5817.
- [Google Scholar]
- Comparison of tumour morphology and structure from U87 and U118 glioma cells cultured on chicken embryo chorioallantoic membrane. Bull Vet Inst Pulawy.. 2013;57(4):593-598.
- [Google Scholar]
- Calycosin-7-O-β-d-glucopyranoside stimulates osteoblast differentiation through regulating the BMP/WNT signaling pathways. Acta Pharmaceutica Sinica B.. 2015;5(5):454-460.
- [Google Scholar]
- ROS and energy metabolism in cancer cells: alliance for fast growth. Archives of pharmacal research.. 2015;38(3):338-345.
- [Google Scholar]
- Calycosin induces apoptotic cell death via antioxidant activity in human colon cancer cells. International journal of oncology.. 2017;51(4):1169-1178.
- [Google Scholar]
- Methylation of flavonoids: Chemical structures, bioactivities, progress and perspectives for biotechnological production. Enzyme and microbial technology.. 2016;1(86):103-116.
- [Google Scholar]
- Calycosin inhibits the migration and invasion of human breast cancer cells by down-regulation of Foxp3 expression. Cellular Physiology and Biochemistry.. 2017;44(5):1775-1784.
- [Google Scholar]
- Spectrum-effect relationship in diuretic effect of Astragali Radix. Chin. J. Mod. Appl. Pharm.. 2013;30:491-495.
- [Google Scholar]
- Calycosin induces mitochondrial-dependent apoptosis and cell cycle arrest, and inhibits cell migration through a ROS-mediated signaling pathway in HepG2 hepatocellular carcinoma cells. Toxicology in Vitro.. 2021;1(70):105052
- [Google Scholar]
- Analysis of relative gene expression data using real time quantitative PCR and the (2CT) method. Methods.. 2001;25(4):402-408.
- [Google Scholar]
- Models of reactive oxygen species in cancer. Drug Discovery Today: Disease Models.. 2007;4(2):67-73.
- [Google Scholar]
- Gallic acid synergistically enhances the apoptotic ability of Abutilon indicum Linn. Stem fraction inhuman U87 glioblastoma cells. Materials Today: Proceedings.. 2021 Jan;1(40):S216-S223.
- [Google Scholar]
- In vitro and in vivo study on glioma treatment enhancement by combining temozolomide with calycosin and formononetin. Journal of ethnopharmacology.. 2019;5(242):111699
- [Google Scholar]
- Inhibition of Wnt/β-catenin signaling mediates ursolic acid-induced apoptosis in PC-3 prostate cancer cells. Pharmacological reports.. 2013;65(5):1366-1374.
- [Google Scholar]
- Interplay between ROS and autophagy in cancer cells, from tumor initiation to cancer therapy. Redox biology.. 2015;1(4):184-192.
- [Google Scholar]
- Brain cancer: implication to disease, therapeutic strategies and tumor targeted drug delivery approaches. Recent patents on anti-cancer drug discovery.. 2018;13(1):70-85.
- [Google Scholar]
- Current progress of phytomedicine in glioblastoma therapy. Current Medical Science.. 2020;40(6):1067-1074.
- [Google Scholar]
- Activating KRAS, NRAS, and BRAF mutants enhance proteasome capacity and reduce endoplasmic reticulum stress in multiple myeloma. Proceedings of the National Academy of Sciences. 2020;117(33):20004-20014.
- [Google Scholar]
- Reactive oxygen species in tumor progression. Front Biosci.. 2005;10(1–3):1881-1896.
- [Google Scholar]
- Evaluation of glutathione-sensitive fluorescent dyes in cortical culture. Glia.. 2000;30(4):329-341.
- [Google Scholar]
- Calycosin inhibits the in vitro and in vivo growth of breast cancer cells through WDR7-7-GPR30 Signaling. Journal of Experimental & Clinical Cancer Research.. 2017;36(1):1-3.
- [Google Scholar]
- Calycosin induces apoptosis in osteosarcoma cell line via ERβ–mediated PI3K/Akt signaling pathways. Molecular medicine reports.. 2020;21(6):2349-2356.
- [Google Scholar]
- c-Myc can induce DNA damage, increase reactive oxygen species, and mitigate p53 function: a mechanism for oncogene-induced genetic instability. Molecular cell.. 2002;9(5):1031-1044.
- [Google Scholar]
- An integrated strategy for effective-component discovery of Astragali Radix in the treatment of lung cancer. Frontiers in pharmacology.. 2021;14(11):2116.
- [Google Scholar]
- Calycosin inhibits viability, induces apoptosis, and suppresses invasion of cervical cancer cells by upregulating tumor suppressor miR-375. Archives of Biochemistry and Biophysics.. 2020;30(691):108478
- [Google Scholar]
- Calycosin Induces Gastric Cancer Cell Apoptosis via the ROS-Mediated MAPK/STAT3/NF-κB Pathway. OncoTargets and therapy.. 2021;14:2505.
- [Google Scholar]
- Lovastatin enhances cytotoxicity of temozolomide via impairing autophagic flux in glioblastoma cells. BioMed research international.. 2019;23:2019.
- [Google Scholar]
- Molecular mechanism of ligustri lucidi fructus-astragali radix for anti-cancer based on network pharmacology. Chinese Journal of Experimental Traditional Medical Formulae. 2019:195-202.
- [Google Scholar]
- Parthenolide induces significant apoptosis and production of reactive oxygen species in high-risk pre-B leukemia cells. Cancer letters.. 2007;254(1):119-127.
- [Google Scholar]







