Translate this page into:
Fabrication of a novel electrochemical sensor based on tin disulfide/multi-walled carbon nanotunbes-modified electrode for rutin determination in natural vegetation
⁎Corresponding authors. 67749801@qq.com (Chunxia Yao), siwenshuai021@163.com (Wenshuai Si), zphuang@tongji.edu.cn (Zhipeng Huang)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
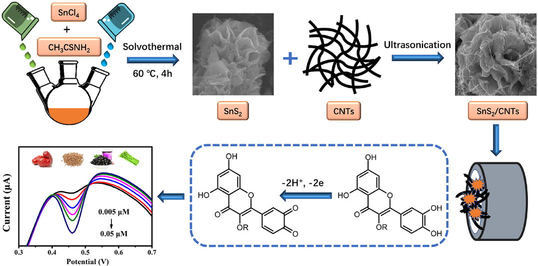
Abstract
A tin disulfide and multi-walled carbon nanotube (SnS2/CNTs) electrochemical sensor was constructed for the sensitive and selective determination of rutin in plants. Tin disulfide nanoflowers with various particle sizes were prepared by controlling the reaction time and composited with multi-walled CNTs. The morphology, crystal structure, and chemical composition of these SnS2/CNTs composites were characterized using XRD, XPS, and SEM-EDS. Results illustrated that the SnS2/CNTs had a large specific surface area, good conductivity, and remarkable electrocatalytic performance. The pH of the buffer solution, the scanning rate, and the amount of modified material were also optimized for the rapid detection of rutin. A 2-electron-2-proton mechanism, involving a few rapid and consecutive stages, was speculated to occur during rutin oxidation, based on the observed slope of -53 mV/pH. There was an appreciable linear relationship between the reductive peak current from DPV and the rutin concentration, ranging from 0.005-0.05 µmol/L and 0.1-6 µmol/L, with a detection limit of 0.22 nmol/L (S/N = 3). The sensor also demonstrated good selectivity, excellent sensitivity, and reproducibility when analyzing rutin in real plant samples, with satisfactory recovery, and was also highly consistent with results of HPLC, and thus could be used to evaluate the medicinal value of natural vegetation.
Keywords
Tin disulfide
Multi-walled carbon nanotubes
Electrochemical sensor
Rutin
1 Introduction
Rutin (vitamin P) is widely found in natural vegetation such as buckwheat, red date, and wolfberry (Al-Harbi et al., 2019; Liu et al., 2017a), and possesses several medical applications such as treating bleeding, digestive system problems, and high blood pressure (Shi et al., 2022; Sghaier et al., 2016). Therefore, quantifying the content of rutin in natural vegetation is essential to assessing and developing their medicinal value. Various analytical methods have been proposed for the qualitative and quantitative analysis of rutin, such as high-performance liquid chromatography (Kalinová et al., 2018), colorimetry (Jjgyasa, 2018), fluorescence spectrometry (Sasikumar et al., 2021), and capillary electrophoresis (Wang et al., 2016). Among these, the electrochemical method has the advantages of a fast response, low cost, simple equipment, and high sensitivity, making it suitable for the rapid detection of trace rutin in natural vegetation and evaluating the medicinal value of plants (Liang et al., 2022; Xie et al., 2022; Incebay et al., 2022; Saylakcı et al., 2021; İncebay et al., 2017; Kilic et al., 2020).
Suitable electrode materials are key to the efficient detection of rutin using electrochemical sensors, and transition metal sulfides have attracted significant attention due to their excellent physical and chemical properties (Liang et al., 2019; Diao et al., 2020; Kubendhiran et al., 2018a; Sakthivel et al., 2019). In particular, tin disulfide (SnS2) has a unique layered structure as well as favorable optical, electrical, and electrochemical properties. It has thus been widely used in sensors and lithium-ion batteries (Xu et al., 2014; Sakthivel et al., 2019; Wang et al., 2013). SnS2 materials possess various morphologies, such as nanoparticles (Jiang et al., 2016), nanosheets (Mondal et al., 2021), nanoribbons (Liu et al., 2017b), and nanoflowers (Yin et al., 2019). Of these, it has been reported that application of the hierarchical nanoflower structure of SnS2 as an electrode material can effectively improve electrochemical performance. The large specific surface area of SnS2 promotes a fast rate of electron transfer, making it an excellent choice for the preparation of modified electrodes.
However, because SnS2 nanoparticles are prone to aggregation, which results in a reduced the electron transfer rate and affects the electrochemical performance, their use in electrode modification materials can be limited (Sakthivel et al., 2019). To overcome such issues, a combination of carbon nanotubes and other nanomaterials are often added when fabricating electrochemical sensors. The presence of carbon nanomaterials can not only retard aggregation and improve the structural stability of SnS2 nanoflowers, but also improve the conductivity and sensitivity of the sensor (Pham et al., 2017; Clancy et al., 2018; Xue et al., 2016; Wang et al., 2021). As a result, integrating multi-walled carbon nanotubes and SnS2 to create synergistic effects can improve selectivity and activity in electrochemical sensors.
Based on previous reports and to the best of our knowledge, there have been no reports on the use of tin disulfide nanomaterials and multi-walled carbon nanotubes with high selectivity and sensitivity for rutin detection in natural vegetation to evaluate the medicinal value of plants. In this work, three types of SnS2 nanomaterials with different particle sizes were prepared, and their structure, crystal phase, and morphology were characterized by X-ray diffraction (XRD), X-ray photoelectron spectroscopy (XPS), and scanning electron microscopy (SEM). In addition, cyclic voltammetry (CV) and differential pulse voltammetry (DPV) were utilized to optimize other aspects of electrochemical detection, including the pH of the buffer solution, the scanning rate, and the amount of modified material by an SnS2/CNTs electrochemical sensor, to realize the enhanced sensitivity and efficient electrochemical detection of rutin in the presence of common interferents. Subsequently, the viability of rutin detection was demonstrated using plant samples, which proved the feasibility of the SnS2/CNTs electrochemical sensor.
2 Experimental section
2.1 Materials and reagents
Multi-walled CNTs were purchased from Adamas Co. ltd. Rutin, thioacetamide, anhydrous stannic chloride, ethanol, potassium ferricyanide, potassium hexacyanoferrate (II), sodium dihydrogen phosphate, dibasic sodium phosphate, potassium chloride, nitric acid, sodium hydroxide, and acetone were purchased from Sigma-Aldrich Chemical Co., China. All chemicals were analytically pure and used without further purification. The phosphate buffer solution (PBS, 0.1 M) was composed of NaH2PO4 and Na2HPO4 mixed with distilled water.
2.2 Instruments and measurements
A CHI 660E electrochemical workstation was used to conduct the electrochemical experiment in a voltametric cell (10 mL) at room temperature. The three-electrode cell consisted of an SnS2/CNT-modified glassy carbon electrode (GCE) as the working electrode, a platinum wire as the auxiliary electrode, and a saturated calomel electrode (SCE) as the reference electrode. The above instruments were purchased from Chenhua Instruments Co., China. XRD was measured using a max-2600PC via ceramic-monochromatic Cu K, operating at 45 kV and 20 mA, with a scanning rate of 5° per min in 2θ and a scanning range from 20 to 65°. XPS was performed for elemental analysis with a 60 W monochromatic Mg Kα source. The binding energy was calibrated based on the carbon 1 s peak at 284.6 eV. SEM images were obtained using a Hitachi Regulus 8100, and the energy dispersive spectrometer (EDS) spectra were recorded using an EDAX EDS Element. The TEM images, selective area electron diffraction (SAED) patterns, and low-magnification scanning TEM (STEM) images were obtained using a Titan Cubed Themis G2 300 (FEI) instrument.
2.3 Synthesis of tin disulfide/multi-walled carbon nanotube composites
Anhydrous tin tetrachloride (0.05 M) and thioacetamide (0.15 M) were dissolved in anhydrous ethanol (240 mL), and ultrasonically treated for 2 min to obtain a clear solution. The solution was heated for 4 h, 6 h, and 8 h at 60 °C to obtain 4h-SnS2, 6h-SnS2, and 8 h-SnS2 samples, respectively, which were cooled down, centrifuged for 10 min, and then washed with deionized water and ethanol. Finally, all samples were dried in a vacuum furnace at 60 °C to obtain dry brownish-yellow solids. SnS2/CNT composites were prepared by dispersing carboxylated multi-walled carbon nanotubes and tin disulfide (ratio = 1:1) in ethanol.
2.4 Fabrication of 4h-SnS2/CNTs-modified electrode
The 0.05 μm and 0.3 μm alumina slurries were placed on suede and dissolved with twice-distilled water to form a suspension, and then the glassy carbon electrode was carefully polished for 3 min. The electrode surface was cleaned after immersion in ethanol, acetone, and twice-distilled water for ultrasonic oscillation cleaning for 5 min. Finally, a bright mirror was obtained. Next, 5 μL of the 4 h-SnS2/CNTs composite with ethanol was dropped onto the electrode surface and dried under an infrared lamp to obtain the 4h-SnS2/CNTs modified electrode, which was gently rinsed with twice-distilled water.
2.5 Optimization of the experimental conditions
The experimental conditions of the SnS2/CNT electrodes, including the pH of the buffer solution, the amount of modified material, and the scanning rate, were optimized to determine the rapid detection of rutin.
First, the 4h-SnS2/CNTs/GCE surface was immersed in 0.05 mol/L PBS at different pH values (pH 2.3, 3, 4, 5, and 6), in which the concentration of rutin was 5.0 μmol/L. Then, the immersed electrode was applied by CV, which was performed in the range of 0.1 V to 0.8 V at a scan rate of 100 mV and sample interval of 0.001 V.
Second, the 4h-SnS2/CNTs composite with different modification amounts (1 μL, 2 μL, 3 μL, 4 μL, 5 μL, and 6 μL) was dropped onto the electrode surface and dried under an infrared lamp, which was gently rinsed with twice-distilled water. The modified electrode was immersed in 5.0 μmol/L rutin solution, 0.05 mol/L PBS, and pH of 3.0. CV was recorded on the electrodes in the potential range between 0.1 and 0.8 V at a scan rate of 100 mV/s.
Thirdly, the CV of 5.0 μmol/L of rutin at different scanning rates (40 mV/s, 60 mV/s, 80 mV/s, 100 mV/s, 120 mV/s, 140 mV/s, and 160 mV/s) was recorded. Then, the reproducibility test was performed using three different GC electrodes under the exact same conditions. Repeatability and stability tests were also performed multiple times using the same electrode for rutin detection.
2.6 Preparation of plant samples before determination
The feasibility of the sensor was ascertained using 9 different natural plant samples including red date, black fruit wolfberry, asparagus, wolfberry, grape peel, tangerine, eggplant peel, buckwheat and apples. The plant samples were prepared for analysis according to reference (Liao et al., 2015). To prepare the samples for testing, the stems and soil of the fresh vegetables were removed for later use. All samples were crushed, freeze-dried, and passed through a 40–60 mesh sieve. Then, 0.5 g of sample (accurate to 0.0001 g) was added to 70 % ethanol (12.5 mL) and then sonicated for 15 min (150 W, 20 ℃, 60 ∼ 62 KHz). After sonication, the samples were centrifuged for 5 min, and finally, the supernatant was filtered with a 0.45 μm filter membrane for later use.
3 Results and discussion
3.1 Characterization of the SnS2/CNTs composites by XRD, XPS, SEM-EDS, and TEM
SnS2 nanoparticles were prepared by the chemical bath deposition method, and their sizes were controlled by controlling reaction times. The crystal phases of the SnS2/CNTs composites and SnS2 nanoparticles were characterized by XRD (Fig. 1). In comparison to the standard card (JCPDS 23–667), the peak height, peak shape, and peak angle of 4h-SnS2, 6h-SnS2, 8 h-SnS2 nanoparticles were the same, and their characteristic diffraction peaks coincided. It was also found that in these materials, longer reaction times resulted in smaller half peak widths and larger grain sizes. Interestingly, compared to the XRD patterns observed in the CNTs, the diffraction peaks located at the 2θ angle of 25.8° were regenerated in the SnS2/CNTs, which represented the (0 0 2) diffraction planes (Sakthivel et al., 2019) according to the periodicity between the graphene layers (Reznik et al., 1995). These peaks confirmed successful composite formation. In addition, in order to study the interaction between SnS2 nanoparticles and CNTs, zeta potential was measured. Zeta potential is related to the surface charge and electric double layer of nanoparticles, which can be used to explain functional group performance, adsorption/desorption phenomena, chemical surface modification or stability. The potentials of SnS2 particles and CNTs are (1.29 ± 0.58) mv and (-2.86 ± 0.92) mV respectively. Therefore, they could combine through electrostatic attraction.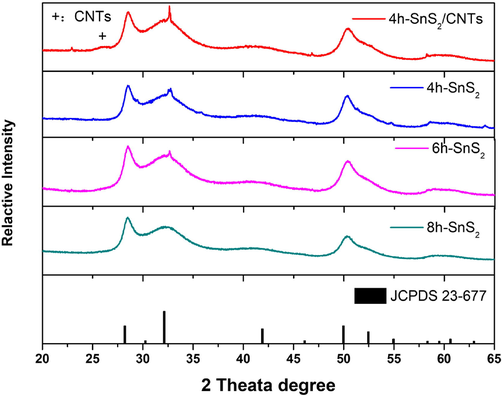
XRD patterns of the SnS2/CNTs and SnS2 nanoflowers according to the standard card (JCPDS 23–667).
To evaluate the chemical composition, purity, integrity, and state of surface oxidation of the 4h-SnS2/CNTs composites, XPS was applied to determine the binding energy of the elements in the micro and nano materials (Kubendhiran et al., 2018a). The XPS survey scan spectra and characteristic peaks for the Sn 3d, S 2p, and C 1 s levels in the 4h-SnS2/CNT composites were as shown in Fig. 2. The peaks at 494.8 eV and 486.4 eV (Fig. 2B) correspond to the Sn 3d3/2 and Sn3d5/2 spin–orbit spin components of Sn4+ in SnS2 (Wang et al., 2017; Xu et al., 2019). In addition, the high-resolution scan of S 2p unambiguously represents two major peaks due to S 2p1/2 at 163.2 eV and S 2p3/2 at 162.0 eV, separately shown in Fig. 2C (Huang et al., 2018). As shown in Fig. 2D, the three dominant peaks of the C 1 s at 285.0 eV, 286.1 eV, and 287.0 eV could be allocated to the non-oxygenated ring carbon (C—C/C⚌C,), the carbon in the C—O bonds, and the carbon in the —COOH bond, respectively (Kubendhiran et al., 2018b; Zhou et al., 2015).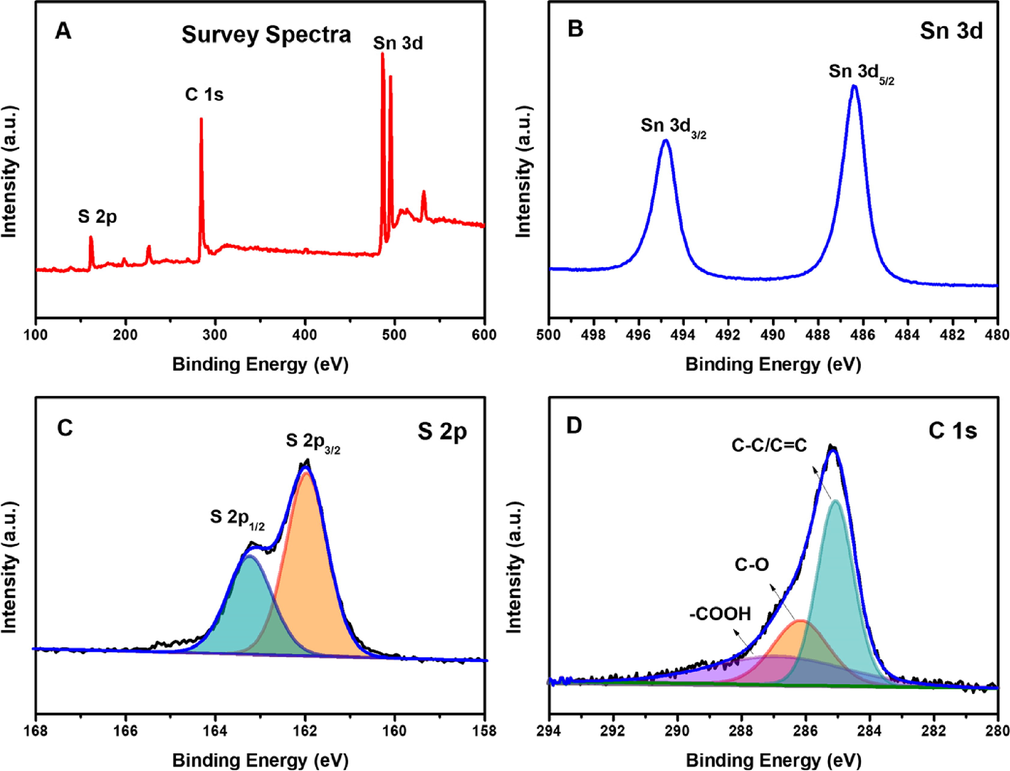
(A) XPS survey spectrum of the 4h-SnS2/CNTs; core level spectrum of (B) Sn 3d, (C) S 2p, (D) C 1 s.
The low-resolution SEM images (Fig. 3A–C) reveal that morphologically, the 4h-SnS2, 6h-SnS2, and 8 h-SnS2 nanoparticles consisted of flower-like microspheres with a smooth surface. The size of these flower-like microspheres increased with the increase of the reaction time. The SEM images of 4h-SnS2/CNTs (Fig. 3D) and 4h-SnS2 (Fig. 3A) indicated that CNTs were uniformly adsorbed on the surface of these microspheres, thus resolving the aggregation problem of CNTs and increasing the specific surface area of the modified materials. In addition, the TEM images of the nanoparticles and the nanoparticle/CNTs materials obtained to verify the size distribution of the SnS2, 4h-SnS2, 6h-SnS2, and 8 h-SnS2 nanoparticles were as shown in Fig S1. The size of the flower-like microspheres increased with increasing reaction time. Moreover, as shown in Fig. S1D, it was observed that carbon nanotubes covered the nanoflower-like SnS2 structures in the SnS2/CNTs composites. The obtained TEM images corroborated the SEM images, both confirming the formation and presence of the SnS2/CNTs composites. To further confirm that the synthesized materials were SnS2 and 4h-SnS2/CNTs, their elemental weight distributions and atomic contents were analyzed by SEM-EDS, and results were as shown in Fig. 3. The atomic ratio of tin and sulfur in these two materials was approximately 1:2, as given in Table S1, and a high proportion of carbon was found in the SnS2/CNTs, indicating the successful synthesis of both SnS2 and 4h-SnS2/CNTs.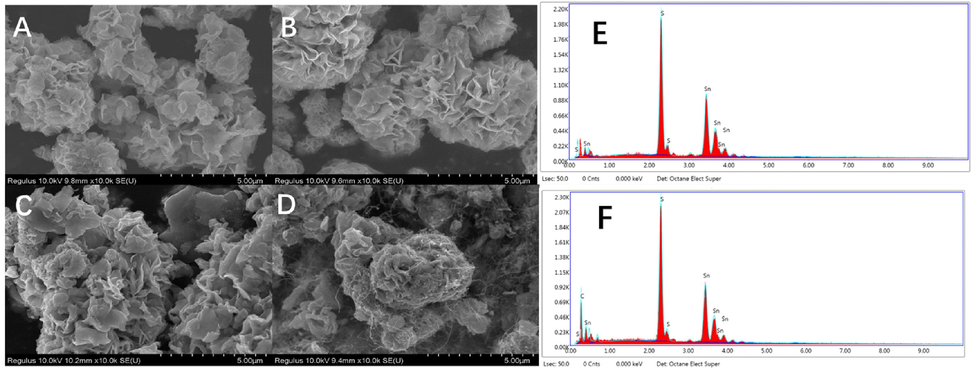
SEM images of SnS2 and SnS2/CNTs: (A) 4h-SnS2, (B) 6h-SnS2, (C) 8 h-SnS2, (D) 4h-SnS2/CNTs, and SEM-EDS spectra of (E) 4h-SnS2 and (F) 4h-SnS2/CNTs.
3.2 Electrochemical behavior
3.2.1 Electrochemical characterization of SnS2 and SnS2/CNTs-modified electrodes
To investigate the performances of the modified electrodes, bare GCE, 4h-SnS2/GCE, 6h-SnS2/GCE, 8 h-SnS2/GCE, CNTs/GCE, and 4h-SnS2/CNTs/GCE were scanned in a K3[Fe(CN)6]/K4[Fe(CN)6] solution (5 mmol/L) using cyclic voltammetry (CV). The electrochemical performances of the modified electrodes were analyzed by comparing their redox peak currents and peak potentials. As discernible in Fig. 4A, the redox peak current of Fe2+/Fe3+ pairs for bare GCE was the smallest, whereas the redox peak current of SnS2/GCE was slightly higher, indicating that tin disulfide had a catalytic effect on the redox reaction. Moreover, the peak current of 4h-SnS2/GCE was found to be the largest, followed by 6h-SnS2/GCE and 8 h-SnS2/GCE. These results support that smaller particle sizes correspond to larger specific surface areas, and thus better electron transfer ability and stronger conductivity. Bare GCE was modified with CNTs to further enhance its current response and results were as illustrated in Fig. 4B. As it was apparent that the electrochemical performance of CNTs was better than that of SnS2, 4h-SnS2 nanoparticles were combined with CNTs to form a 4h-SnS2/CNTs composite. The modified electrode based on 4h-SnS2/CNTs (4h-SnS2/CNTs/GCE) had the highest response because 4h-SnS2/CNTs had excellent electrocatalytic ability for Fe2+/Fe3+ pairs.![CV and EIS of different electrodes in 0.1 mol/L of KCl solution containing 5 mmol/L of K[Fe(CN)6]3−/4− at a scan rate of 100 mV/s: (A) GCE, 4h-SnS2/GCE, 6h-SnS2/GCE, 8h-SnS2/GCE, (B) GCE, 4h-SnS2/GCE, CNTs/GCE, 4h-SnS2/CNTs/GCE and (C) EIS of different modified electrodes recorded.](/content/184/2023/16/5/img/10.1016_j.arabjc.2023.104613-fig5.png)
CV and EIS of different electrodes in 0.1 mol/L of KCl solution containing 5 mmol/L of K[Fe(CN)6]3−/4− at a scan rate of 100 mV/s: (A) GCE, 4h-SnS2/GCE, 6h-SnS2/GCE, 8h-SnS2/GCE, (B) GCE, 4h-SnS2/GCE, CNTs/GCE, 4h-SnS2/CNTs/GCE and (C) EIS of different modified electrodes recorded.
Electrochemical impedance spectroscopy (EIS) can provide useful information regarding impedance changes on the electrode surface during the fabrication process, further verifying its results (Fig. 4C). The charge-transfer resistance was related to the electron-transfer kinetics of the redox probe ([Fe(CN)6]3−/[Fe(CN)6]4−) at the electrode interface and could be estimated from the diameter of the semicircular part of the EIS curve. Fig. 4C illustrates the typical Nyquist plots obtained from bare GCE, 8h-SnS2/GCE, 6h-SnS2/GCE, 4h-SnS2/GCE, CNTs/GCE, and 4h-SnS2/CNTs/GCE in aqueous KCl (0.1 mol/L) containing 5.0 mmol/L [Fe(CN)6]3−/[Fe(CN)6]4−. A large diameter of the semicircle part was observed for GCE. The diameter of the semicircle was reduced when the GCE surface was coated with SnS2 nanoparticles due to improved electrical conductivity. In addition, the smaller surface area facilitated electron transport, and thus the semicircle diameter of 4h-SnS2/GCE was much smaller. The semicircle diameter showed an even greater decrease in CNTs/GCE due to the excellent electron transfer ability of the carbon nanotubes. The electrochemical impedance spectrum of 4h-SnS2/CNTs/GCE had the smallest semicircle diameter, which proved that the fastest electron-transfer kinetics of [Fe(CN)6]3−/4− occurred in 4h-SnS2/CNTs/GCE rather than in the other modified electrodes. The EIS results were in full agreement with the CV results.
To understand the electrochemical behavior of each modified electrode, the electrochemically active surface area (A) of each electrode could be derived from Randles Sevcik Eq. (1) (Zhang et al., 2020; Rao et al., 2018; Prasad et al., 2017):
3.2.2 Electrochemical responses of various modified electrodes in rutin solutions
The CV curves of bare GCE and various modified electrodes in 5 μmol/L of rutin in 0.05 mol/L PBS (pH 3.0) were as shown in Fig. 5. At a scanning rate of 100 mV/s, a reduction peak was observed at 0.48 V and the electrochemical responses of the SnS2-modified electrodes to rutin were each slightly different (Fig. 5A). The response current of rutin in 4h-SnS2/GCE with the smallest particle size was found to be the largest, which suggested that it was the most suitable material for electrodes. As evident in Fig. 5B, the response currents of GCE, 4h-SnS2/GCE, CNTs/GCE, and 4h-SnS2/CNTs/GCE increased significantly. The 4h-SnS2/CNTs composite had a large specific surface area and good conductivity. Hence, faster electron transfer in the electrode was due to improved catalytic efficiency.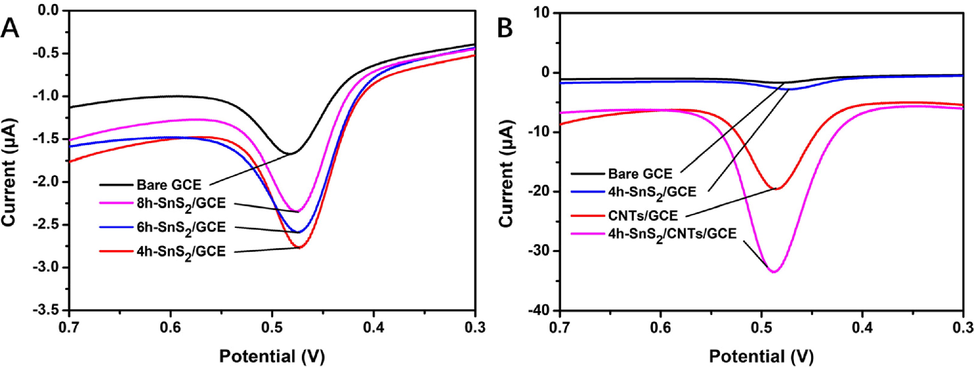
CV of different electrodes in 5 μmol/L of rutin in 0.05 mol/L PBS (pH 3.0) at a scan rate of 100 mV/s: (A) SnS2-modified electrodes and (B) modified electrodes.
3.3 Optimization of electrochemical experimental conditions
3.3.1 Effect of buffer solution pH
The pH value of the buffer solution is considered a significant influential factor that will influence on the results of electrochemical detection. Therefore, it was necessary to select the appropriate pH of PBS to optimize the sensitivity for rutin determination. As shown in Fig. 6, increase in the pH of the buffer solution moved the peak potential of rutin moved in the negative direction. A linear relationship was observed between the values of potential and pH and could be expressed as potential = -0.0530 pH + 0.6219 (R2 = 0.9947). The slope in this relationship (53 mV/pH) approximated the theoretical value of -59 mV/pH. This indicated that the proton number and electron transfer number involved in rutin oxidation on 4h-SnS2/CNTs/GCE electrodes were equal, implying that it possibly followed the 2-electron-2-proton mechanism (El Jaouhari et al. 2020). According to Scheme 1, the electrochemical oxidation mechanism of rutin involved several rapid and continuous stages, which was first triggered by electron transfer to produce phenoxy radicals in the form of four mesomeric intermediates (A-D). Due to the electronic effects of the hydroxyl groups, the structure of radical B was the most stable. However, it instantly experienced electron transfer once more, producing the carbocation, and then was dehydrated rapidly, transforming into the final product 30, 40-diquinone (E), which could be reduced to the original rutin in the reverse reaction (Zhang et al., 2015; Sun et al., 2008). Moreover, the peak current first increased and then decreased as the pH of the buffer solution increased, which indicated that the electrode reaction of rutin required the participation of protons. Rutin has four phenolic hydroxyl groups, which have difficulty reacting electrochemically due to the lack of protons under alkaline conditions. The optimal pH of buffer solution was selected to be 3 based on the pH at which the largest peak current was achieved.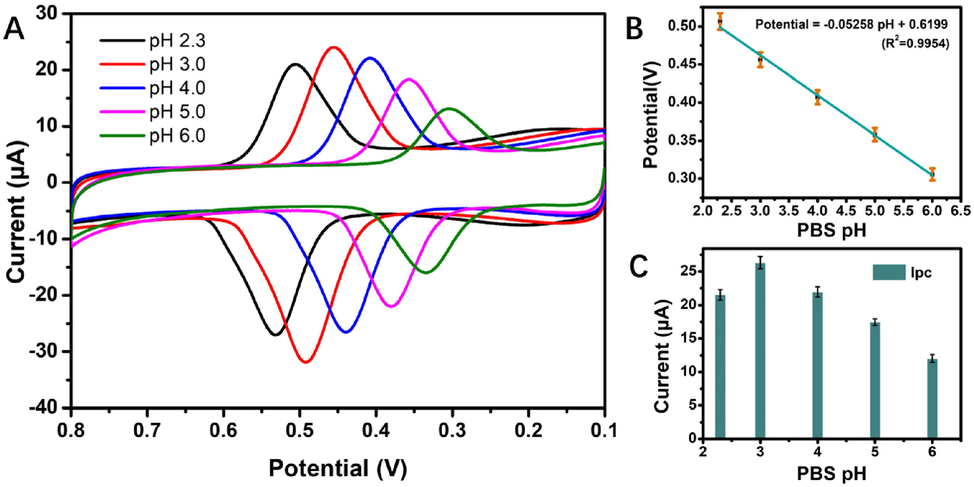
(A) CV of 5.0 μmol/L of rutin in 0.05 mol/L pH phosphate buffer solutions (pH = 2.3, 3, 4, 5, 6) at a scan rate of 100 mV/s; (B) effects of different pH values on the peak potential, and (C) effects of different pH values on the peak current.
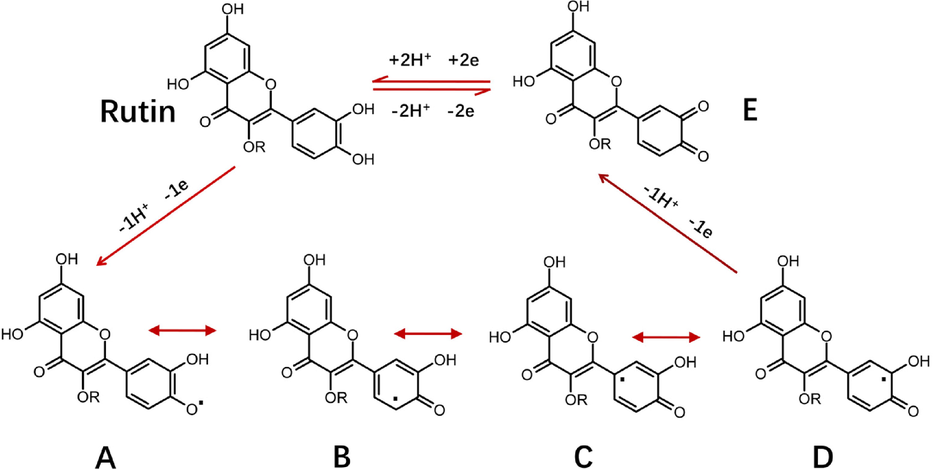
Electrochemical oxidation mechanism of rutin.
3.3.2 Effect of 4h-SnS2/CNTs dosage
The peak current of rutin will also be affected by the extent of modification to a composite. Generally, the larger the specific surface area of the electrode material, the higher the number of active sites in the electrode. However, when a high amount of modified material results in an overly thick modified film, electron transfer in the electrode is hindered and the electrochemical response is reduced. Therefore, it is necessary to control the dosage of 4h-SnS2/CNTs composite to improve its sensitivity to rutin (Fig. S2). When 5 μL of the modified materials was used, 4h-SnS2/CNTs/GCE had the highest response to rutin. However, its electrochemical response decreased as the thickness of the modified layer increased due to the restriction to the ionic transfer rate with excessive materials. Therefore, 5.0 μL of 4h-SnS2/CNTs nanocomposites was considered the optimal amount to promote electrochemical activity with extensive active sites and edges.
3.3.3 Effects of scanning rate and the mechanism of electro surface reaction
An increase in scanning rate from 40 mV/s to 180 mV/s resulted in not only the substantial enhancement of the oxidation and reduction peak current of rutin (Fig. 7), but also a linear movement towards the negative potential in the oxidation and reduction peak current, demonstrating that the surface of 4h-SnS2/CNTs/GCE had improved adsorption capacity and electron transfer kinetics. The linear relationship between the anodic peak current (Ipa) and cathodic peak current (Ipc) could be expressed as Ipa (μA) = -0.2567ν (mV/s) - 1.6664 (R2 = 0.9992) and Ipc (μA) = 0.2181ν (mV/s) + 0.3506 (R2 = 0.9998). These results indicated that the electrochemical behavior of rutin on 4h-SnS2/CNTs/GCE exhibited adsorption-controlled electron transfer without any fouling effect (El Jaouhari et al. 2020; Yao et al., 2019; Chen et al. 2019). As the scanning rate decreased, the current response of rutin decreased and the sensitivity worsened. In contrast, as the scanning rate increased, the current response and sensitivity both increased, whereas reversibility of the redox reaction decreased. Therefore, the optimal scanning rate was determined to be 100 mV/s.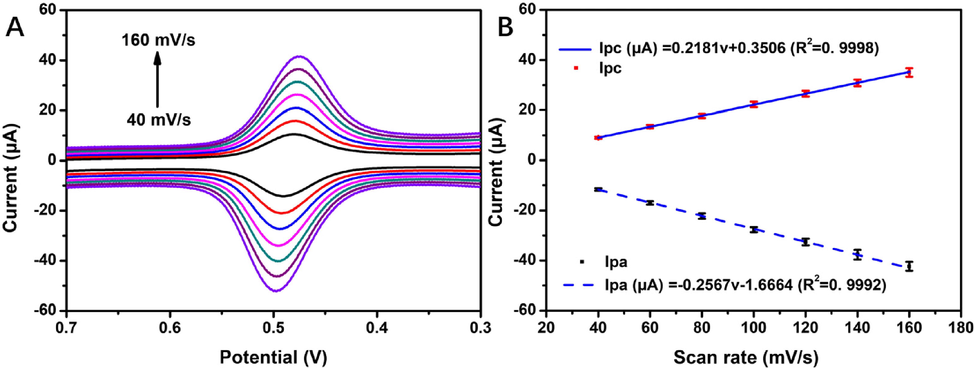
(A) CV of 5.0 μmol/L of rutin in 0.05 mol/L PBS (pH 3.0) at different scanning rates (40 mV/s, 60 mV/s, 80 mV/s, 100 mV/s, 120 mV/s, 140 mV/s, and 160 mV/s); (B) linear relationship between the scanning rate and response peak current.
3.4 Linearity, interference, reproducibility, and stability
To explore the electrochemical performance of 4h-SnS2/CNTs/GCE, DPV was applied to determine the concentration of rutin in optimal experimental conditions, with results as shown in Fig. 8. The optimal concentrations of rutin were 0.005-0.05 μmol/L and 0.1-6.0 μmol/L. A linear relationship was observed between the Ipa and concentration (C) of 4h-SnS2/CNTs/GCE. At low concentrations, this relationship could be expressed as the equation Ipa (μA) = -37.85C (μmol/L) - 0.0162 (R2 = 0.9947), and at high concentrations, as Ipa (μA) = -20.24C (μmol/L) - 0.9073 (R2 = 0.9857). According to the signal measured in the blank solution, which was determined using the SnS2/CNTs electrochemical sensor measuring 6 times in parallel, the current was about 0.019 μA (Fig. S3). Based on the equation Ipa (μA) = -37.85C (μmol/L) - 0.0162 (R2 = 0.9947) as well as the relationship between the current signal of lowest detection limit and the noise signal (S/N = 3), the limit of detection (LOD) was calculated to be 0.22 nmol/L.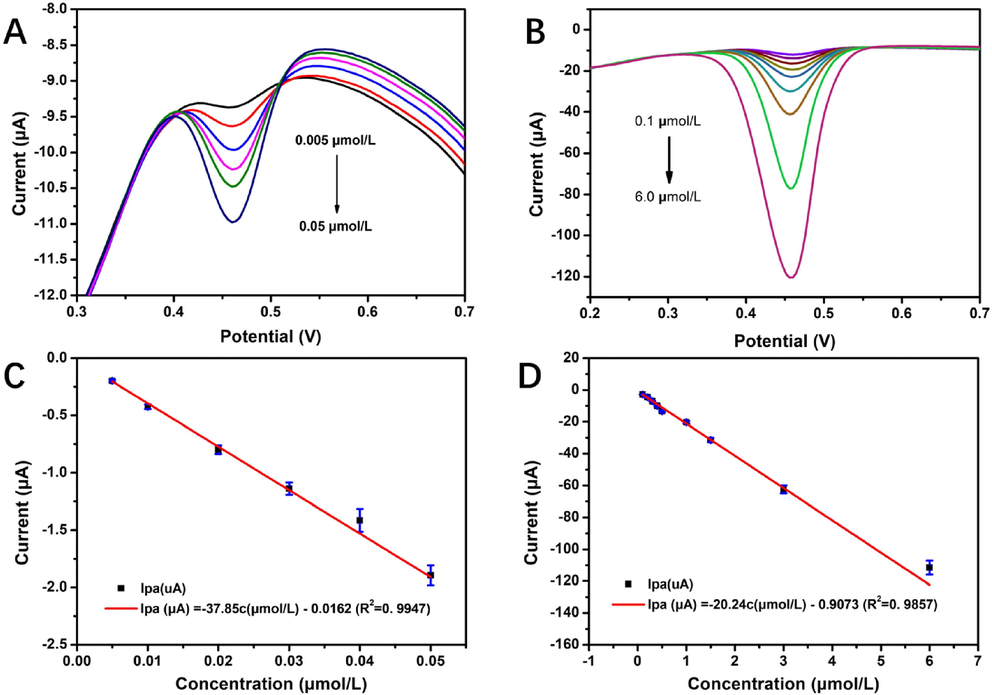
DPV curves in 0.05 mol/L PBS (pH 3.0) at a scan rate of 100 mV/s for rutin concentrations of (A) 0.005–0.050 µmol/L and (B) 0.1–6.0 µmol/L, (C) standard curve in the low concentration range of rutin (0.005–0.050 μmol/L), and (D) standard curve in the high concentration range of rutin (0.1–6.0 μmol/L).
It was also found that compared to other common methods of rutin detection, the electrochemical method used in this experiment had the lowest detection limit and the highest sensitivity (Table 1). Therefore, the proposed electrochemical method was fast, simple, and sensitive for rutin determination.
Detection method
Innovation
LOD (mol/L)
Reference
Capillary electrophoresis
Response surface methodology
4.97 × 10−6
Martí et al., 2017
Photodiode array detector.
5.45 × 10−7
Memon et al., 2017
Fluorescence spectrometry
Cysteine-stabilized copper nanoclusters
21 × 10−8
Wang et al., 2016
MoS2 quantum dots
3.50 × 10−7
Sun et al., 2020
High-performance liquid chromatography
UHPLC/ESI-Q-TOF-MS/MS
1.35 × 10−7
Ahmad et al., 2016
Modified QuEChERS method
1.20 × 10−7
Rotta et al., 2019
Electrochemical method
Cu2O-Au/nitrogen-doped graphene nanocomposites
3 × 10−8
Li et al., 2017
rGO-InTAPc/GCE
2 × 10−9
Shi et al., 2022
SnS2/CNTs/GCE sensor
1.67 × 10−9
This report
In real sample analysis, the interference of unknown substances is inevitable, thus, anti-interference is an important characteristic of modified electrodes. Ions and molecules that might interfere with the experiment were added to measure the electrochemical response of 4h-SnS2/CNTs/GCE. The interference of the 4h-SnS2/CNTs modified electrode was explored in solutions of 5 μmol/L rutin, with the presence of ions (50-fold concentration) or organic substances (10-fold concentration). As shown in Fig. 9, I0 and I were the Ipa of rutin in 5 μmol/L solutions before and after adding the interferents, respectively. The results suggested that the interferents had no significant impact on the determination of rutin by 4h-SnS2/CNTs/GCE, which indicated good selectivity for rutin in the electrodes.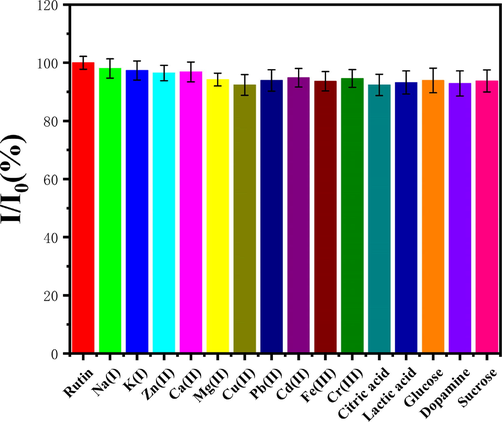
Selectivity of 4h-SnS2/CNTs/GCE toward rutin.
The storage stability test was investigated by DPV in a solution of 5.0 μM rutin. Notably, after 10 days of storage, the peak current was reduced by 8.2 %. Furthermore, the repeatability was assessed using the same 4h-SnS2/CNTs/GCE after 6 successive tests (RSD ≤ 3.83 %), and reproducibility was calculated 6 times using 6 different 4h-SnS2/CNTs/GC (RSD ≤ 4.18 %). The above results verified that the sensor has reliable electrochemical performance.
3.5 Analysis of real samples
After the synthesis and optimization of 4h-SnS2/CNTs, a 4h-SnS2/CNTs/GCE sensor was fabricated to detect rutin in medicine samples. This sensor was applied to detect trace levels of rutin in real samples. Twenty tablets of rutin were weighed to be 1.8481 g in total, and the average weight of each was 92.41 mg. The content of rutin in 1 g tablets was 0.2164 g, and 1.099 μmol/L of rutin solution was prepared from a small amount of powder. The peak current response for 4h-SnS2/CNTs/GCE was calculated, and the average rutin concentration was 1.06 μmol/L (RSD ≤ 3.58 %). When the tablets were spiked with three levels of rutin (5.00 μM, 10.00 μM, and 15.00 μM), the recoveries ranged between 96.6 % and 102.3 % (RSD ≤ 3.5 %, n = 5) (Table 2). Therefore, the 4h-SnS2/CNTs/GCE delivered good analytical performance in terms of sensitivity, precision, and accuracy for rutin detection in medicine samples.
Samples
Rutin spiked (μM)
Rutin found (μM)
Recovery (%)
RSD
(n = 5, %)
Rutin tablets
0.00
1.06
–
3.58
5.00
6.83
96.6
3.29
10.00
11.23
102.3
3.41
15.00
16.18
101.2
3.19
Additionally, the practicability of the 4h-SnS2/CNTs/GCE sensor was explored by analysis of nine different natural plants prepared as described in section 2.6. The DPV and HPLC results under optimal conditions were as presented in Table 3. Noticeably, results from DPV and HPLC analyses were in strong agreement. This validated that the 4h-SnS2/CNTs/GCE sensor had excellent analytical performance in terms of precision and could be applied the quantification of rutin in plant samples.
Samples
Average concentration (μM)
DPV
HPLC
Red date
98.71 ± 4.73
101.23
Black fruit wolfberry
272.82 ± 18.63
280.09
Asparagus
676.29 ± 19.15
673.85
Wolfberry
430.63 ± 22.91
422.26
Grape peel
291.96 ± 6.05
293.69
Tangerine
217.50 ± 5.87
215.23
Eggplant peel
268.94 ± 13.38
280.09
Buckwheat
53.13 ± 1.48
55.69
Apple
55.42 ± 3.47
57.98
4 Conclusion
In summary, a rapid and selective electrochemical sensor was proposed and synthesized in this study for the determination of rutin concentration. Nanoflower-like structured SnS2 was successfully synthesized and composited with multi-walled carbon nanotubes via simple sono-chemical treatments. The prepared SnS2/CNTs modified electrode was then verified to possess higher effectiveness in the electrochemical determination of rutin, exhibiting a good linear range from 0.005 - 0.05 μmol/L and 0.1–6.0 μmol/L with an LOD value of 0.22 nmol/L (S/N = 3). In contrast to previous methods, the proposed sensor is characterized by good anti-interference properties, superior reproducibility and repeatability, and long-term stability. Moreover, the fabricated sensor was successfully applied to determine rutin not only in commercially available rutin tablets, but also in natural vegetation to evaluate the medicinal value of plants. Therefore, this strategy could be further applied to the development of sensing devices for analyze different substances.
CRediT authorship contribution statement
Ying Liang: Conceptualization, Methodology, Investigation, Formal analysis, Validation, Visualization, Writing – original draft, Funding acquisition. Lingyu Zhang: Formal analysis, Investigation, Validation. Hongmei Wang: Formal analysis, Investigation, Validation. Xinru Cai: Formal analysis, Investigation, Validation. Li Zhang: Formal analysis, Investigation, Validation. Yixin Xu: Formal analysis, Investigation, Validation. Chunxia Yao: Conceptualization, Methodology, Investigation, Formal analysis, Validation, Visualization, Funding acquisition. Wenshuai Si: Conceptualization, Resources, Supervision, Methodology, Project administration. Zhipeng Huang: Conceptualization, Resources, Supervision, Methodology, Project administration. Guoyue Shi: Conceptualization, Resources, Supervision, Methodology, Project administration.
Funding
This work was supported by the Foundation of Shanghai Municipal Commission of Science & Technology Commission (20392002200) and the Fundamental Research Funds of Shanghai University of Medicine & Health Sciences (HXXM-20-07-004, SSF-21-07-001).
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Quantification of rutin in rat's brain by UHPLC/ESI-Q-TOF-MS/MS after intranasal administration of rutin loaded chitosan nanoparticles. EXCLI J.. 2016;15:518.
- [CrossRef] [Google Scholar]
- Rutin inhibits carfilzomib-induced oxidative stress and inflammation via the NOS-mediated NF-κB signaling pathway. Inflammopharmacology. 2019;27:817-827.
- [CrossRef] [Google Scholar]
- Facile fabrication of electrochemical sensor based on novel core-shell PPy@ZIF-8 structures: enhanced charge collection for quercetin in human plasma samples. Sens. Actuators B. 2019;290:434-442.
- [CrossRef] [Google Scholar]
- Charged carbon nanomaterials: redox chemistries of fullerenes, carbon nanotubes, and graphenes. Chem. Rev.. 2018;118:7363-7408.
- [CrossRef] [Google Scholar]
- Vertical heterostructure of SnS–MoS2 synthesized by sulfur-preloaded chemical vapor deposition. ACS Appl. Mater. Interfaces. 2020;12(6):7423-7431.
- [CrossRef] [Google Scholar]
- One-step hydrothermal synthesis of a CoS2@MoS2 nanocomposite for high-performance supercapacitors. J. Alloy. Compd.. 2018;742:844-851.
- [CrossRef] [Google Scholar]
- Electrochemical determination of indigo carmine in food and water samples using a novel platform based on chiral amine-bis (phenolate) boron complex. Dyes Pigm.. 2022;197:109921
- [CrossRef] [Google Scholar]
- Effect of different copper salts on the electrochemical determination of Cu (II) by the application of the graphene oxide-modified glassy carbon electrod. Surf. Interfaces. 2017;9:160-166.
- [CrossRef] [Google Scholar]
- Enhanced molecular imprinted electrochemical sensor based on zeolitic imidazolate framework/reduced graphene oxide for highly recognition of rutin. Anal. Chim. Acta. 2020;1106:103-114.
- [CrossRef] [Google Scholar]
- Ultrasmall SnS2 nanoparticles anchored on well-distributed nitrogen-doped graphene sheets for Li-ion and Na-ion batteries. J. Mater. Chem. A. 2016;4(27):10719-10726.
- [CrossRef] [Google Scholar]
- Bio-polyphenols promoted green synthesis of silver nanoparticles for facile and ultra-sensitive colorimetric detection of melamine in milk. Biosens. Bioelectron.. 2018;120(30):153-159.
- [CrossRef] [Google Scholar]
- Contribution to the study of rutin stability in the achenes of Tartary buckwheat (Fagopyrum tataricum) Food Chem.. 2018;258:314-320.
- [CrossRef] [Google Scholar]
- Synthesis and electrochemical investigation of chiral amine bis (phenolate)-boron complexes: in vitro antibacterial activity screening of boron compounds. Inorg. Chim. Acta. 2020;510:119777
- [CrossRef] [Google Scholar]
- Innovative strategy based on a novel carbon-black− β-cyclodextrin nanocomposite for the simultaneous determination of the anticancer drug flutamide and the environmental pollutant 4-nitrophenol. Anal. Chem.. 2018;90(10):6283-6291.
- [CrossRef] [Google Scholar]
- Electrochemical co-preparation of cobalt sulfide/reduced graphene oxide composite for electrocatalytic activity and determination of H2O2 in biological samples. J. Colloid Interface Sci.. 2018;509:153-162.
- [CrossRef] [Google Scholar]
- A facile and green fabrication of Cu2O-Au/NG nanocomposites for sensitive electrochemical determination of rutin. J. Electroanal. Chem.. 2017;786:20-27.
- [CrossRef] [Google Scholar]
- Vertical stacking of copper sulfide nanoparticles and molybdenum sulfide nanosheets for enhanced nonlinear absorption. ACS Appl. Mater. Interfaces. 2019;11(39):35835-35844.
- [CrossRef] [Google Scholar]
- A novel molecularly imprinted polymer composite based on polyaniline nanoparticles as sensitive sensors for parathion detection in the field. Food Control. 2022;133:108638
- [CrossRef] [Google Scholar]
- New method to enhance the extraction yield of rutin from Sophora japonica using a novel ultrasonic extraction system by determining optimum ultrasonic frequency. Ultrason. Sonochem.. 2015;27:110-116.
- [CrossRef] [Google Scholar]
- Strain effect on SnS2 nanoribbons: robust direct bandgap of zigzag-edge and sensitive indirect semiconductor with armchair-edge states. Superlattice. Microst.. 2017;111:480-486.
- [CrossRef] [Google Scholar]
- Sensitive electrochemical detection of rutin and isoquercitrin based on SH-β-cyclodextrin functionalized graphene-palladium nanoparticles. Biosens. Bioelectron.. 2017;89(12):444-452.
- [CrossRef] [Google Scholar]
- Simultaneous determination of main phenolic acids and flavonoids in tomato by micellar electrokinetic capillary electrophoresis. Food Chem.. 2017;221:439-446.
- [CrossRef] [Google Scholar]
- Simultaneous determination of quercetin, rutin, naringin, and naringenin in different fruits by capillary zone electrophoresis. Food Anal. Methods. 2017;10(1):83-91.
- [CrossRef] [Google Scholar]
- Facile transfer of excited electrons in Au/SnS2 nanosheets for efficient solar-driven selective organic transformations. Appl Catal B. 2021;286:119927
- [CrossRef] [Google Scholar]
- Properties of functionalized carbon nanotubes and their interaction with a metallic substrate investigated by scanning tunneling microscopy. J. Phys. Chem. C. 2017;121(43):24264-24271.
- [CrossRef] [Google Scholar]
- Synthesis of novel monomeric graphene quantum dots and corresponding nanocomposite with molecularly imprinted polymer for electrochemical detection of an anticancerous ifosfamide drug. Biosens. Bioelectron.. 2017;94:1-9.
- [CrossRef] [Google Scholar]
- Nitrogen-doped carbon nanosheet frameworks decorated with Fe and molecularly imprinted polymer for simultaneous detection of mebendazole and catechol. Chem. Eng. J.. 2018;338:478-487.
- [CrossRef] [Google Scholar]
- X-ray powder diffraction from carbon nanotubes and nanoparticles. Phys. Rev. B. 1995;52(1):116.
- [CrossRef] [Google Scholar]
- Determination of phenolic compounds and antioxidant activity in passion fruit pulp (Passiflora spp.) using a modified QuEChERS method and UHPLC-MS/MS. LWT. 2019;100:397-403.
- [CrossRef] [Google Scholar]
- Rational design and facile synthesis of binary metal sulfides VS2-SnS2 hybrid with functionalized multiwalled carbon nanotube for the selective detection of neurotransmitter dopamine. Anal. Chim. Acta. 2019;1071:98-108.
- [CrossRef] [Google Scholar]
- Water-soluble luminescent copper nanoclusters as a fluorescent quenching probe for the detection of rutin and quercetin based on the inner filter effect. Luminescence. 2021;36(2):326-335.
- [CrossRef] [Google Scholar]
- An electrochemical platform of tannic acid and carbon nanotubes for the sensitive determination of the antipsychotic medication clozapine in pharmaceutical and biological samples. J. Electroanal. Chem.. 2021;898:115638
- [CrossRef] [Google Scholar]
- Rutin inhibits proliferation, attenuates superoxide production and decreases adhesion and migration of human cancerous cells. Biomed. Pharmacother.. 2016;84:1972-1978.
- [CrossRef] [Google Scholar]
- Soluble tetraaminophthalocyanines indium functionalized graphene platforms for rapid and ultra-sensitive determination of rutin in Tartary buckwheat tea. Food Control. 2022;132:108550
- [CrossRef] [Google Scholar]
- MoS2 quantum dots as a specific fluorescence sensor for selection of rutin and for temperature sensing. Luminescence. 2020;35(8):1416-1423.
- [CrossRef] [Google Scholar]
- Electrochemical behavior and determination of rutin on a pyridinium-based ionic liquid modified carbon paste electrode. J. Pharm. Biomed. Anal.. 2008;48(5):1326-1331.
- [CrossRef] [Google Scholar]
- CTAB-assisted synthesis of single-layer MoS2-graphene composites as anode materials of Li-ion batteries. J. Mater. Chem. A. 2013;1(6):2202-2210.
- [CrossRef] [Google Scholar]
- Magnetic tetraethylenepentamine modified multi-walled carbon nanotubes as matrix clean-up materials for organophosphorus pesticide residues analysis in cucumber. Food Control. 2021;124:107904
- [CrossRef] [Google Scholar]
- Separation and determination of flavonoids in three traditional chinese medicines by capillary electrophoresis with amperometric detection. J. Sep. Sci.. 2016;39(7):1357-1362.
- [CrossRef] [Google Scholar]
- Synthesis of a hierarchical SnS2 nanostructure for efficient adsorption of Rhodamine B dye. J. Colloid Interface Sci.. 2017;507:225-233.
- [CrossRef] [Google Scholar]
- Recent advances in aptamer-based optical and electrochemical biosensors for detection of pesticides and veterinary drugs. Food Control. 2022;131:108399
- [CrossRef] [Google Scholar]
- Nanostructured transition metal sulfides for lithium ion batteries: Progress and challenges. Nano Today. 2014;9(5):604-630.
- [CrossRef] [Google Scholar]
- Large-scale growth and field-effect transistors electrical engineering of atomic-layer SnS2. Small. 2019;15(46):1904116.
- [CrossRef] [Google Scholar]
- Polypiperazine-amide nanofiltration membrane modified by different functionalized multiwalled carbon nanotubes (MWCNTS) ACS Appl. Mater. Interfaces. 2016;8(29):19135-19144.
- [CrossRef] [Google Scholar]
- Experimental and theoretical studies of a novel electrochemical sensor based on molecularly imprinted polymer and B, N, F-CQDs/AgNPs for enhanced specific identification and dual signal amplification in highly selective and ultra-trace bisphenol S determination in plastic products. Anal. Chim. Acta. 2019;1066:36-48.
- [CrossRef] [Google Scholar]
- Construction of nanoflower SnS2 anchored on g-C3N4 nanosheets composite as highly efficient anode for lithium ion batteries. Electrochim. Acta. 2019;293:408-418.
- [CrossRef] [Google Scholar]
- Synthesis and characterization of PEDOT-MC decorated AgNPs for voltammetric detection of rutin in real samples. J. Electroanal. Chem.. 2020;877:114632
- [CrossRef] [Google Scholar]
- Poly (3, 4-ethylenedioxythiophene) nanorods grown on graphene oxide sheets as electrochemical sensing platform for rutin. J. Electroanal. Chem.. 2015;739:66-72.
- [CrossRef] [Google Scholar]
- Preparation of a reduced graphene oxide/zirconia nanocomposite and its application as a novel lubricant oil additive. RSC Adv.. 2015;5(111):91802-91812.
- [CrossRef] [Google Scholar]
Appendix A
Supplementary material
Supplementary data to this article can be found online at https://doi.org/10.1016/j.arabjc.2023.104613.
Appendix A
Supplementary material
The following are the Supplementary data to this article:Supplementary data 1
Supplementary data 1







