Translate this page into:
Quality markers screening of traditional Chinese medicine prescriptions based on the multi-factor analysis strategy: Jin-Zhen oral liquid as a case
⁎Corresponding authors at: College of Pharmacy, Jinan University, Guangzhou 510632, China(Y. Yu), Jiangsu Kanion Pharmaceutical Co. Ltd. and National Key Laboratory on Technologies for Chinese Medicine Pharmaceutical Process Control and Intelligent Manufacture, Jiangsu, Lianyungang, 222001, China(W. Xiao), College of Pharmacy, Jinan University, Guangzhou 510632, China(X.-s. Yao). tyaoxs@jnu.edu.cn (Xin-sheng Yao), xw_kanion@163.com (Wei Xiao), 1018yuyang@163.com (Yang Yu)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Background
Jin-Zhen oral liquid (JZOL), a well-known traditional Chinese medicine prescription (TCMp), has extensively been used to treat acute bronchitis in children more than four hundred years in China. However, the current quality control standard of JZOL is inadequate, posing challenges for its internationalization.
Purpose
In this study, a Q-marker screening strategy based on multi-factor analysis was proposed to comprehensively evaluate anti-inflammatory Q-markers of Jin-Zhen oral liquid (JZOL).
Methods
Firstly, the chemical profile and the pharmacokinetics properties of multiple components in JZOL were characterized by UPLC-Q/TOF-MS and UPLC-QqQ-MS. By integrating the measurable and absorbed components, twenty-two components with structures accurately defined, were selected as candidate Q-markers of JZOL. Following that, a network connecting 22 components and targets closely associated with bronchitis was established. Afterwards, a multi-factor analysis mode was developed to balance the components’ multiple characteristics, and screen out the anti-inflammatory Q-markers in JZOL. Finally, the anti-inflammatory activity evaluation was conducted by LPS-induced RAW 264.7 macrophages to prove the representativeness of anti-inflammatory Q-markers in JZOL.
Results
As a result, a total of 92 components were characterized in JZOL and the pharmacokinetics properties of 11 bioactive components in vivo were further characterized. Then, an overlapping of 46 targets involved in the interactions of selected 22 candidate Q-markers and the regulation of bronchitis inflammation were collected. Subsequently, a multi-factor analysis was developed on 22 candidate anti-inflammatory Q-markers covering five factors, with the statistic KMO values of 0.645, and the P values of Bartlett's Test equal to 0.000. A total of seven ingredients (aloeemodin-8-O-β-d-glucopyranoside, baicalin, chrysin-7-O-β-d-glucuronide, oroxylin A 7-O-β-d-glucuronide, wogonoside, chrysophanol-8-O-β-d-glucopyranoside, and skullcapflavone II) were selected as Q-markers of JZOL, and the inhibitory effects of these candidate Q-markers on the secretion of inflammatory cytokes (NO, IL-6, IL-1β, and PGE2) in lipopolysaccharide (LPS)-stimulated RAW264.7 cells were evaluated and confirmed.
Conclusion
This study not only offers a fresh approach to uncovering Q-markers in the quality control research of TCMps but also identifies the suitable anti-inflammatory Q-markers for JZOL for the first time, with the potential to serve as a reference for existing quality control standards of JZOL.
Keywords
Q-markers
Jin-Zhen oral liquid (JZOL)
Acute bronchitis
Anti-inflammatory
Traditional Chinese Medicine prescriptions
Multi-factor analysis
- AUC
-
area under the concentration–time curve
- IS
-
internal standard
- LLOQ
-
Lower limits of quantification
- MRT(0-t)
-
mean resident time
- RSD
-
relative standard deviation
- RE
-
relative error
- TCM
-
traditional Chinese medicine
- Tmax
-
time to the Cmax
- KMO
-
Kaiser-Meyer-Olkin
- ULOQ
-
upper limit of quantitation
Abbreviations
1 Introduction
Traditional Chinese Medicine prescriptions (TCMps) with the characteristics of “multi-components, multi-targets, and multi-pathways” have been used to treat a variety of ailments for thousands of years (Kurniawan, Y.S., 2023; Chu et al., 2022). Quality control of TCMps is not only essential for their clinical efficacy and safety but also plays a significant role in their internationalization and modernization process (Ouyang et al., 2022; Wang et al., 2016). However, the quality of TCMps is frequently inconsistent due to various factors such as variety, origin, processing method, and the interaction of components derived from different herbs, posing a significant challenge for dependable quality control of TCMps. To address this issue, a novel concept of quality marker (Q-marker) has been proposed recently to improve the consistency of TCMps′ quality (Liu et al., 2017; Liu et al., 2016).
Q-markers are the efficacy-related essential components naturally existing in individual herbs that can be determined qualitatively and quantitatively, and closely linked to the compatibility theories of Traditional Chinese Medicine. Ideal TCMp Q-markers should possess properties including traceability, specificity, bioactivity, measurability, and compatibility contribution (Liu et al., 2021). However, screening for suitable Q-markers from hundreds and thousands of ingredients in TCMps is a significant challenge. To reveal Q-markers in TCMps, several methods have been developed, such as the metabolomics (Gao et al., 2022), chinmedomics approach (Xiong et al., 2020), network pharmacology (Liu et al., 2020), pharmacokinetic analysis (He et al., 2018), and systems biology (Li et al., 2022). Nevertheless, conventional approaches are inadequate in providing a comprehensive and intuitive assessment of the various characteristics of Q-markers. Therefore, balancing various properties and screening out pivotal Q-markers of TCMps remains a challenge and requires a comprehensive evaluation strategy.
Jin-Zhen oral liquid (JZOL) is a famous prescribed TCMps comprising eight medicines: Caprae Hircus Cornu, Scutellariae Radix, Fritillariae Ussuriensis Bulbus, Rhei Radix et Rhizoma, Gypsum Fibrosum, Bovis Calculus Artifactus, Chloriti Lapis, and Glycyrrhizae Radix et Rhizoma. Double-blind, placebo-controlled clinical trials with centralized, randomized controlled studies (Liu et al., 2014; Lu et al., 2010) have confirmed its clinical efficacy in treating acute bronchitis (Shu et al., 2018; Sun et al., 2016; Zhang & Gu, 2016), and it may exert anti-inflammatory effects by regulating the TLR4/MyD88/NF-κB signaling pathway (Li et al., 2023). The pharmacological activity may attribute to its composed medicinal herbs, including Scutellariae Radix (Zhu et al., 2023), and Rhei Radix et Rhizoma (Hu et al., 2021) which possess significant anti-inflammatory properties in various diseases. JOZL has been found to have a broad-spectrum anti-inflammatory in clinical practice, primarily utilized for treating various respiratory conditions in children (Cui et al., 2022, Yang, 2021). Accordingly, the current quality evaluation marker (The Pharmacopoea Commission of PRC, 2020) is inadequate in representing the overall efficacy of JZOL, as it fails to reflect the combined anti-inflammatory effects through multiple components present in TCMps.
In this study, an anti-inflammatory Q-marker screening strategy based on multiple factors analysis was first proposed and applied to a research example of JZOL (Fig. 1). Based on the scientific concept of Q-marker, the quality control index of JZOL should fulfill the requirements in terms of specificity, quality transferability, measurability, compatibility, and pharmacological efficacy. Therefore, we first characterized the chemical profile of JZOL using UPLC-Q/TOF-MS to ensure chemical specificity and measurability. Secondly, the pharmacokinetic (PK) properties were investigated to follow the requirement of feature transferability from in vitro to in vivo. Thirdly, the pharmacological efficacy was primarily considered based on the components and related targets. Inspired by the previous work on content analysis (Li et al., 2020a) and metabolic analysis (Zhang et al., 2020), a multi-factor mode was developed to screen out JZOL Q-markers that balanced multiple characteristics (the compatibility contribution of herbal medicines, the content, the in vivo PK characteristics, and the degree of network pharmacology). Afterwards, the mentioned factors were normalized and subjected to multi-factor analysis, with the suitability for analysis being evaluated through pre-calculation and applicability testing, and the Q-Markers was chosen based on the ranking of the F value. Finally, the activity evaluation was conducted to verify the feasibility of Q-markers in JZOL. In a summary, this study presents a fresh approach for disclosing Q-markers in TCMps and helps to find superior Q-markers of JZOL to improve its quality control measures.
The novel strategy for the discovery of Q-markers in JZOL based on “multi-factor analysis”.
2 Experiment and methods
2.1 Materials and chemicals
Liquiritigenin apioside and licorice-saponin G2 were isolated in our laboratory, and their structures were unambiguously identified by nuclear magnetic resonance (NMR) and MS methods. The other reference standards were purchased from National Institutes for Food and Drug Control., Chengdu Puruifa Medical Technological Co., Ltd (Chengdu, China), Lemeitian Co. Ltd (Chengdu, China), Shanghai yuanye Bio-Technology Co., Ltd (Shanghai, China) and Shanghai Macklin Biochemical Co., Ltd (Shanghai, China). Detailed information regarding the reference standards used in this study is presented in Table S1.
JZOL was provided by Jiangsu Kanion Pharmaceutical Co. Ltd. (No. 181026, Lianyungang, China). Scutellariae Radix, Fritillariae Ussuriensis Bulbus, Rhei Radix et Rhizoma, Bovis Calculus Artifactus, and Glycyrrhizae Radix et Rhizomawere were purchased from the Ji’an Medical Material Market (Jiangxi, China). All herbal medicines were identified by Prof. GuangXiong Zhou of Jinan University, and voucher specimens were deposited at Institute of Traditional Chinese Medicine & Natural Products, college of pharmacy, Jinan, University, Guangzhou, China.
LC-MS grade acetonitrile: Fisher Scientific, LC-MS-grade formic acid: Sigma-Aldrich, Water: Watsons. Methanol, and ethanol used for sample extraction were of analytical grade. Details of other materials are recorded in the corresponding methods.
2.2 Sample preparation
JZOL was directly evaporated with a rotary evaporator and then diluted to a concentration of 10 mg/mL. Subsequently, 2 mL of these solutions were transferred into a separate clean tube, dried under a nitrogen gas stream at room temperature, and then suspended in 2 mL of water. For the Solid-phase extraction (SPE, Vac 3 cc, 200 mg Phenomenex strata C18-E cartridges from Torrance, CA) process: the cartridge were pre-conditioned with 3 mL of methanol, followed by 3 mL of water before application. The centrifuged supernatants were loaded onto the SPE cartridges and washed with 2 mL of water. Afterward, an elution step was performed using 4 mL of methanol, and the eluted solution was collected. Supernatant aliquots of 2 µL centrifuged supernatant were then injected into the LC-MS system.
2.3 Animals and drug administration
Healthy male Sprague Dawley rats, weighing 200 ± 20 g, were purchased from the experimental animal center of Guangdong province (Guangzhou, China). All animals were randomly assigned to groups and maintained under standard condition (23 ± 2 °C and 55–60 % relative humidity). The rats were given free access to water and standard chow for a week before the experiment. Next, the drug-treated group (n = 7) was administered JZOL at the dose of 3.31 g/kg/day (equal clinical dose). Blood samples (0.3 mL) were collected from the jugular vein and placed in heparinized tubes at 0.25, 0.5, 1.0, 2.0, 4.0, 6.0, 8.0, 12.0, 24.0, 36.0, 48.0 and 72.0 h after the drug administration. After centrifuging (14,000 rpm, 4 °C) the blood sample for 10 min, the obtained plasma was stored at −80 °C.
2.4 Pretreatment of biological samples
A volum of 100 µL plasma sample was spiked with 50 µL of IS and 400 µL of 0.5 % formic acid in acetonitrile for protein precipitation. Following a 2-minute vortexing and subsequent centrifugation (14,000 rpm, 20 min), the supernatants were dried to completion using nitroge. The residue was reconstituted in 100 µL methanol and vortexed for 2 min. After centrifuged (14,000 rpm, 20 min), an aliquot of 2 µL was injected into the LC-MS system for analysis.
2.5 UPLC-Q/TOF-MS analysis
UPLC analyses: ACQUITYTM UPLC I-Class system, binary solvent system, and an automatic sample manager. Q/TOF-MS analyses: Waters SYNAPT™ G2 mass spectrometer (Waters, Manchester, UK), and an ESI interface. The detail information of optimal conditions was presented in supplementary material.
2.6 UPLC-QqQ-MS analysis
Components were detected under a Xevo TQ-XS mass spectrometer (Waters Corp., Milford, MA, USA) equipped with electrospray ionization (ESI) interface. Detailed information regarding the optimal conditions can be found in the supplementary materials. The most appropriate precursor ion, daughter ion, cone voltage, and collision energy were optimized and displayed in Table S2.
All experimental data were collected and processed by Waters Masslynx™ software 4.1 and the Quanlynx program (Waters, Milford, MA, USA).
2.7 Method validation of pharmacokinetic research
The method validation was carried out according to the 2018 Food and Drug Administration (FDA) guidelines on bioanalytical method validation in terms of selectivity, linearity, accuracy, matrix effect, precision, and stability. The detail information of method validation was presented in supplementary material.
2.8 The candidate Q-markers selection
Twenty-two components, whose structures are all well-defined by the reference standards, covering the characteristics of multiple structural types (flavonoids, triterpenoid saponins, alkaloids, bile acids, anthraquinones and others), higher content (11.56 ∼ 867.40 μg/mL), and bioavailable constituents in vivo (absorbed in blood) were selected as candidate Q-markers of JZOL based on the researches of qualitative and quantitative chemical analysis, as well as PK study. In detail, the selection of the 22 components was a comprehensive compilation, consisting of 13 components that could be measured for in vitro content, and 21 prototypes (Table S3, Fig. S1) (Table S3, Fig. S1) that were accurately identified with reference standard for in vivo verification.
2.9 Network pharmacology studies
The structures of selected candidate Q-markers in JZOL were prepared in SMILES format, and imported into the SwissTargetPrediction (https://www.swisstargetprediction.ch/) for the prediction of targets in “homo sapiens” species (Gfeller et al., 2014). The target set was created to explore potential interactions between protein targets and the compounds in JZOL. As the Drugbank database (https://www.drugbank.ca/) provides comprehensive profiles of protein targets associated with clinical symptoms (Wishart et al., 2018), the protein targets involved in the symptom of “inflammation” were systematically searched and derived as a target set of inflammation. The target sets involved in the regulation of Gypsum Fibrosum and Chloriti Lapis was respectively derived from the STITCH server (https://stitch.embl.de/) (Kuhn et al., 2014) based on the main ionic components including Ca2+, SO42-, Fe2+, Mg2+, K+, Na+ (Ikarashi et al., 2012; Chen et al., 2022). These target sets were compared and overlapped to get the common targets for further analysis (Amala et al., 2023).
The key targets involved in the interactions of 22 compounds and the symptom of “inflammation” were collected for bioinformatics analysis. The Search Tool for the Retrieval of Interacting Genes (STRING) database (https://string-db.org/) is used to input all essential targets for protein–protein interaction (PPI) prediction (Szklarczyk et al., 2021). The detail information was supported in supporting information. The regulation effect of 22 compounds of JZOL as well as the main ionic components of Gypsum Fibrosum and Chloriti Lapis were characterized by their involved key targets, while the corresponding signaling pathways were further analyzed by gene set enrichment analysis (GSEA) (version 3.0, https://software.broadinstitute.org/gsea/). The top 20 items with the most significant were derived for further analysis.
The importance of targets that involved in the interactions of 22 compounds and the symptom of “inflammation” were further evaluated by molecular docking using Glide 6.6 of Schrödinger Software Suite (Schrödinger, LLC, New York, NY, 2015). The 3D crystal structures for 46 unique targets were retrieved from the Protein Data Bank database (https://www.rcsb.org/) or the AlphaFold protein structure database (https://alphafold.ebi.ac.uk/). The target structures were initially readied by introducing hydrogen atoms, rectifying absent side chains, and conducting minimization using the Protein Preparation Wizard module. Thereafter, the binding pocket for each target was defined according to the location of the co-crystalized ligand or the best site predicted by the fpocket software.
The 3D structures of 22 compounds were generated and optimized using the LigPrep module, targeting a pH range of 7.0 ± 2.0. The standard precision (SP) protocol was utilized to conduct docking simulations involving 46 targets and 22 compounds. The best docking score for each target-compound couple in the docking runs was selected for analysis, while the docking scores for those with no binding results were set as 0 (Table S4). All other parameters were set as default values.
2.10 Development of multi-factor analysis to balance and screen Q-markers with the multiple characteristics in JZOL
A multi-factor analysis mode was developed to balance the Q-markers related multiple characteristics and to screen the Q-markers in JZOL. The candidate Q-markers with clear herbal origin were defined as king, minister, assistant, and guide based on the chemical profile characterization and TCM compatibility theory. In addition, the network pharmacology was applied to provide a bridge to link the chemical constituents in TCM prescriptions with the corresponding targets to gain comprehensive insight into the therapeutic mechanism of multi-components. Consequently, a multi-factor analysis mode was developed by SPSSPRO (https://www.spsspro.com/mydata/index) platform, and the properties of compatibility contribution (the candidate Q-markers from King, Minister, Assistant, or Guide herb are defined as 4, 3, 2, and 1, respectively), content, pharmacokinetics (Cmax and AUC0-∞), and network pharmacology (degree value) were converted into five dimensions. However, there may be missing values among multiple dimensions. For example, although some components are highly exposed in TCM prescriptions, they may not be absorbed in vivo. Hence, the supplement of the missing value follows these principles: if the candidate Q-marker cannot be absorbed in vivo, its value is filled with 0; if the candidate Q-marker is difficult to quantify accurately, its value is filled with the corresponding average; the missing value of content is filled with the minimum value of content. All the data are then standardized as 0–1 points from minimum to maximum according to Eq. (1). The maximum variance method is used to carry out factor rotation. The KMO value (>0.5) and Bartlett's test (p ≤ 0.05) are applied to judge whether factor analysis is suitable. The number of extracted factors is determined as the main common factor through the gravel map. Based on the determined common factors at all levels, the ingredients' use obtains an eigenvalue matrix, and by calculating the weight of different common factors, the composite score (Eq.2–3) is ultimately calculated for the sample and ranked.
V is the variable of corresponding characteristic data of candidate JZOL Q-marker; D is the standardized value of variables; F1-FP is the score of each common factor, which is used to calculate the overall score of total factors (F); means factor coefficient, and z is observed variable, which is calculated by a linear combination of several independent factors and a unique variable (Schreiber et al., 2021). X is the weight of different common factors.
2.11 Confirmation of Q-markers based on anti-inflammatory assays
In a humidified environment at 37 °C with 5 % CO2, RAW 264.7 cells were cultured and introduced into DMEM supplemented with 10 % fetal bovine serum (FBS, Gibco, New York, USA). Cell viability was assessed using the [3-(4,5-dimethyl-thiazol-2-yle) 2,5-diphenyltetrazolium bromide] (MTT, Aladdin, Shanghai, China) colorimetric assay. RAW 264.7 cells were incubated for 24 h in 96-well plates at a concentration of 2 × 105 cells per well. Then the components were dissolved in the medium (containing 1 µg/ml LPS) and given to the test wells. The negative control was treated with complete medium (containing 0.1 % DMSO) and the positive control was treated with 1 µg/ml LPS. After 24 h treatment under normal conditions, the supernatant was collected and analyzed by a commercial NO test kit (Nanjing Jiancheng Biological Engineering Research Institute, China). The IL-6, IL-1β, and PGE2 analysis was measured by ELISA kit (IL-6: Multisciences Biotechnology, Zhejiang, China; IL-1β: R&D system, Minneapolis, MN; PGE2: Enzo Life Sciences, Farmingdale, NY) according to the manufacturer's instructions.
Data: Mean ± SD from three independent experiments. Statistical analysis: GraphPad PRISM v7.04, ANOVA followed by Dunnett's post hoc test, P < 0.05 considered significant.
3 Results
3.1 Chemical profile of JZOL by UPLC-Q/TOF-MS analysis
The chemical constituents in JZOL were systematically investigated by searching online databases or Internet search engines (Chemical Abstracts Service (CAS) database, Massbank, Web of Science, and ChemSpider). As a result, a total of 92 compounds, including 37 flavonoids (23 flavones, 4 chalcones, 4 flavanones, 4 isoflavones, and 2 isoflavanones), 14 triterpenoid saponins, 12 alkaloids, 11 bile acids, seven diterpenoids, six anthraquinones, and five other types of compounds were identified or tentatively characterized in JZOL (Table 1; Fig. 2). Out of these, 28 peaks were further confirmed by comparison with reference standards (Fig. S2). The base peak intensity (BPI) profiles of JZOL in both negative and positive ion modes are shown in Fig. 3. The origins of the identified compounds were determined by comparing the base peak chromatograms of JZOL with those of individual single herbs (Fig. S3 and S4). Fig. 4 illustrates the detailed fragmentation and proposed fragment pathways for representative compounds. Fig. 4 displays the intricate fragmentation patterns and proposed fragment pathways of representative compounds, while additional in-depth information can be found in the Supporting Information. Note: * the compound was unambiguously identified with reference standard, F: Flavonoids, T: Triterpenoid saponins, A: Alkaloids, B: Bile acids, AN: Anthraquinones, D: Diterpenoids, O: Others.
No
tR
Selected ion
Elemental Composition
Measured mass
Calculated mass
Mass error
Fragmentations (m/z)
Identification or tentative characterization
Type
Sourcea
1
0.63
[M + Na]+
C12H22O11
365.1060
365.1060
0.0
707.2219 [2 M + Na]+; 365.1060 [M + Na]+; 203.0529 [M + Na-C6H10O5]+
sucrose
O
FL
2*
1.10
[M−H]-
C7H6O5
169.0136
169.0137
−0.6
169.0136 [M−H]-; 125.0242 [M−H−CO2]-
gallic acid
O
DH
3
2.07
[M + H]+
C11H9NO2
188.0713
188.0712
0.5
188.0713 [M + H]+; 170.0604 [M + H-H2O]+; 146.0606 [M + H-C2H2O]+
2-hydroxy-3-naphthamide
O
PBM
4
2.19
[M−H]-
C9H10O3
165.0554
165.0552
1.2
165.0554 [M−H]-; 121.0654 [M−H−CO2]-
paeonol
O
GC
5
2.50
[M + H]+
C27H45NO6
480.3326
480.3325
0.2
480.3326 [M + H]+; 462.3099 [M + H-H2O]+; 444.2100 [M + H-2H2O]+
pingbeimine B
A
PBM
6
2.54
[M + H]+
C27H45NO5
464.3370
464.3376
−1.3
464.3370 [M + H-H2O]+; 446.3199 [M + H-H2O]+; 428.3100 [M + H-2H2O] +
pingpeimine A
A
PBM
7
2.57
[M−H]-
C21H22O9
417.1183
417.1186
−0.3
417.1183 [M−H]-; 255.0665 [M−H−Glc]-; 135.0080[1,3A0]-; 119.0503 [1,3B0]-
neoisoliquiritin
F
GC
8
2.65
[M + H]+
C27H41NO5
460.3065
460.3063
0.2
460.3065 [M + H]+; 442.2870 [M + H-H2O]+; 424.0822 [M + H-2H2O]+
15β-hydroxy-23-isopengbeisine B or verdine
A
PBM
9
2.74
[M + H]+
C27H43NO6
478.3171
478.3169
0.4
478.3171 [M + H]+; 460.3003 [M + H-H2O]+; 442.2882 [M + H-2H2O]+
pingbeimine C
A
PBM
10
3.15
[M−H]-
C26H28O14
563.1403
563.1401
0.4
563.1403 [M−H]-; 473.1078 [M−H−C3H6O3]-; 443.1001 [M−H−C3H6O3−CH2O]-; 383.0754 [M−H−2C3H6O3]-; 353.0668 [M−H−2C3H6O3−CH2O]-
schaftoside
F
GC
11
3.19
[M−H]-
C26H28O14
563.1403
563.1401
0.4
563.1403 [M−H]-; 473.1065 [M−H−C3H6O3]-; 443.0986 [M−H−C3H6O3−CH2O]-; 383.0779 [M−H−2C3H6O3]-; 353.0668 [M−H−2C3H6O3−CH2O]-
isoschaftoside
F
GC
12
3.59
[M + H]+
C33H51NO8
590.3694
590.3693
0.2
590.3694 [M + H]+; 572.3615 [M + H-H2O]+
(20R,25R)–23,26-epimino-3β-hydroxy-5α-cholest-23(N)-ene-6,22-dione-3-O-β-d-glucopyranoside
A
PBM
13*
3.65
[M−H]-
C21H20O10
431.0970
431.0978
−1.9
431.0970 [M−H]-; 269.0445 [M−H- Glu]-
aloeemodin-8-O-β-D-glucopyranoside
AN
DH
14
3.66
[M−H]-
C26H30O13
549.1605
549.1608
−0.4
549.1605 [M−H]-; 417.1778 [M−H−C5H8O4]-; 255.0655 [M−H−C5H8O4−Glu]-
isoliquiritin apioside
F
GC
15*
3.75
[M−H]-
C26H30O13
549.1617
549.1608
1.6
549.1617 [M−H]-; 417.1776 [M−H−C5H8O4]-; 255.0635 [M−H−C5H8O4−Glu]-
liquiritigenin apioside
F
GC
16*
3.80
[M−H]-
C21H22O9
417.1182
417.1186
−1.0
417.1182 [M−H]-; 255.0658 [M−H−Glc]-;135.0086 [1,3A0]-;119.0497 [1,3B0]-
liquiritin
F
GC
17
4.04
[M + H]+
C27H38NO3
424.2852
424.2860
−0.8
424.2852 [M + H]+; 406.0642 [M + H-H2O] +
ussuriedine
A
PMB
18
4.12
[M−H]-
C26H28O13
547.1466
547.1452
2.6
547.1466 [M−H]-; 457.1129 [M−H−C3H6O3]-; 427.1027 [M−H−C3H6O3−CH2O]-; 367.0843 [M−H−2C3H6O3 ]-; 337.0788 [M−H−2C3H6O3−CH2O]-
chrysin6-C-β-d-glucopyranosyl-8-C-α-l-arabinopyranoside
F
HQ
19
4.22
[M−H]-
C26H28O13
547.1458
547.1452
1.1
547.1458 [M−H]-; 457.1130 [M−H−C3H6O3]-; 427.1030 [M−H−C3H6O3−CH2O]-; 367.0817 [M−H−2C3H6O3 ]-; 337.0702 [M−H−2C3H6O3−CH2O]-
chyrsin-6-C-α-l-arabinopyranosyl-8-C-β-d-glucopyranoside
F
HQ
20
4.37
[M + H]+
C33H54NO8
592.3845
592.3847
−0.2
592.3845 [M + H]+; 574.3664 [M + H-H2O]+
zhebeinone-3-β-d-glucoside
A
PMB
21
4.57
[M−H]-
C22H20O12
475.0876
475.0877
−0.2
475.0876 [M−H]-; 299.0545 [M−H−GluA]-
diosmetin-7-O-β-d-glucuronide
F
HQ
22
4.66
[M−H]-
C21H20O9
415.1029
415.1029
0.0
415.1029 [M−H]-; 253.0434[M-H-Glu]-;239.0428[M-H-Glu-CH2]- 225.0550[M-H-Glu-CO]-
pulmatin
AN
DH
23*
4.75
[M + H]+
C27H41NO3
428.3167
428.3165
0.2
428.3167 [M + H]+; 410.3097 [M + H-H2O]+; 393.2801[M + H-H2O-NH3]+
peimisine
A
PBM
24
4.97
[M + H]+
C27H45NO3
432.3467
432.3478
−2.5
432.3467 [M + H]+; 414.3371 [M + H-H2O]+
verticine
A
PBM
25
5.12
[M−H]-
C22H20O12
475.0874
475.0877
−0.6
475.0874 [M−H]-; 299.0540 [M−H−GluA]-; 284.0377 [M−H−GluA−CH3]-
hispidalin-7-O-β-d-glucuronide
F
HQ
26
5.38
[M−H]-
C26H30O13
549.1608
549.1608
0.0
549.1608 [M−H]-; 417.1788 [M−H−Api]-; 255.0656 [M−H−Api−Glc]-
liguiritigenin-7-O-β-d-apiosyl-4′-O-β-d-glucoside
F
GC
27*
5.45
[M + H]+
C27H43NO3
430.3319
430.3321
−0.5
430.3319 [M + H] +; 412.3211 [M + H-H2O]+; 396.2971 [M + H-H2O-CH4]+
peiminine
A
PBM
28
5.55
[M + H]+
C32H52NO8
578.3701
578.3693
0.8
578.3701 [M + H]+; 560.3484 [M + H-H2O]+; 164.1428 [M + H-C21H34O8]+
pingbeinone-3-O-β-d-glucoside
A
PBM
29*
5.69
[M−H]-
C21H22O9
417.1180
417.1186
−1.4
417.1180 [M−H]-; 255.0657 [M−H−Glc]-; 135.0086 [1,3A0]-; 119.0508 [1,3B0]-
isoliquiritin
F
GC
30*
6.10
[M−H]-
C21H18O11
445.0770
445.0771
−0.2
891.1619 [2 M−H]-;445.0770 [M−H]-; 269.0447 [M−H−GluA]-; 251.0341 [M−H−GluA−H2O]-; 241.0508 [M−H−GluA−CO]-; 223.0401 [M−H−GluA−H2O−CO]-; 195.0452 [M−H−GluA−H2O−2CO]-; 175.0249[M-H-C15H10O5]-; 113.0236 [M−H- C16H12O8]-
baicalin
F
HQ
31
6.20
[M−H]-
C21H18O11
445.0775
445.0771
0.9
445.0771 [M−H]-; 269.0450 [M−H−GluA]-; 251.0342 [M−H−GluA−H2O]-; 223.0353 [M−H−GluA−H2O−CO]-;195.0462 [M−H−GluA−H2O−2CO]-
baicalein-6-O-β-d-glucuronide
F
HQ
32*
6.24
[M−H]-
C15H12O4
255.0657
255.0657
0.0
255.0657 [M−H]-; 135.0080[1,3A0]-; 119.0503 [1,3B0]-
liquiritigenin
F
GC
33
6.30
[M−H]-
C22H20O11
459.0930
459.0927
0.7
459.0930 [M−H]-; 269.0460 [M−H−methylgluA]-; 251.0334 [M−H−methylgluA−H2O]-; 241.0487 [M−H−methylgluA−CO]-; 223.0398 [M−H−methylgluA−H2O−CO]-; 197.0571 [M−H−methylgluA−CO−CO2]-;
baicalein-7-O-glucuronide-methyl ester
F
HQ
34
6.60
[M + H]+
C26H42NO3
416.3155
416.3175
−2.0
416.3155 [M + H]+; 398.3002[M + H-H2O]+; 163.1315 [M + H-C15H25O3] +;
pingbeinone
A
PBM
35
7.13
[M−H]-
C21H18O11
445.0762
445.0771
−2.0
445.0762 [M + H] +; 269.0456 [M−H−GluA]-; 251.0352 [M−H−GluA−H2O]-; 241.0459 [M−H−GluA−CO]-; 223.0352 [M−H−GluA−H2O−CO]-; 195.0460 [M−H−GluA−H2O−2CO]-
apigenin-7-O-β-d-glucronide
F
HQ
36
7.33
[M−H]-
C21H18O11
445.0769
445.0771
−0.4
445.0769 [M−H]-; 269.0473[M-H-GluA]-
norwogonin-7-O-β-d-glucuronide
F
HQ
37
7.48
[M−H]-
C22H20O12
475.0871
475.0877
−1.3
475.0871 [M−H]-; 299.0540 [M−H−GluA]-; 284.0377[M-H-GluA-CH3]-
scutevurin-7-O-β-d-glucuronide
F
HQ
38*
7.73
[M−H]-
C21H18O10
429.0826
429.0822
0.9
429.0826 [M−H]-; 253.0500 [M−H−GluA]-; 209.0613 [M−H−GluA−CO2]-; 151.0046 [1,3A0]-
chrysin 7-O-β-d-glucuronide
F
HQ
39*
7.79
[M−H]-
C22H20O11
459.0935
459.0927
1.7
919.1959 [2 M−H]-; 459.0935 [M−H]-; 283.0612 [M−H−GluA]-; 268.0377 [M−H−GluA−CH3]-; 240.0412 [M−H−GluA−CH3−CO]-
oroxylin A 7-O-β-d-glucuronide
F
HQ
40*
7.83
[M−H]-
C21H20O9
415.1041
415.1029
2.9
415.1041 [M−H]-; 295.0607 [M−H−C4H8O4]-; 277.0515 [M−H−C4H8O4−H2O]-; 253.0503 [M−H−Glu]-; 225.0560 [M−H−Glu−CO]-
chrysophanol-1-O-β-d-glucopyranoside
AN
DH
41
7.98
[M−H]-
C22H20O12
475.0877
475.0877
0.0
475.0877 [M−H]-; 284.0377 [M−H−methylester]-
scutellarin methyl ester
F
HQ
42
8.05
[M−H]-
C21H20O10
431.0986
431.0978
1.9
431.0986 [M−H]-; 269.0442 [M−H−Glu]-; 151.0028 [1,3A0]-
apigenin 7-O-β-d-glucoside
F
GC
43*
8.08
[M−H]-
C21H20O10
431.0976
431.0978
−0.5
431.0976 [M−H]-; 269.0445 [M−H−Glu]-; 241.0506 [M−H−Glu−CO]-; 223.0416 [M−H−Glu−CO−H2O]-
baicalin 7-O-β-d-glucoside
F
HQ
44
8.15
[M−H]-
C21H18O11
445.0763
445.0771
−1.8
445.0763 [M−H]-; 283.0605 [M−H−Glu]-; 239.0341 [M−H−Glu−CO2]-; 211.0388 [M−H−Glu−CO2−CO]-; 183.0446 [M−H−Glu−CO2−2CO]-
rhein-8-O-β-d-glucoside
AN
DH
45*
8.22
[M−H]-
C21H20O9
415.1025
415.1029
−1.0
415.1025 [M−H]-; 295.0605 [M−H−C4H8O4]-; 277.0512 [M−H−C4H8O4−H2O]-; 253.0507 [M−H−Glu]-; 225.0550 [M−H−Glu−CO]-
chrysophanol-8-O-β-d-glucopyranoside
AN
DH
46*
8.31
[M−H]-
C22H20O11
459.0934
459.0927
1.5
919.1932 [2 M−H]-; 459.0934 [M−H]-; 283.0599 [M−H−GluA]-; 268.0378 [M−H−GluA−CH3]-;240.0417 [M−H−GluA−CH3−CO]-; 175.0248 [M−H−C16H12O5]-; 163.0035 [M−H−C14H16O7]-; 113.0241 [M−H−C16H12O5−CO2−H2O]-
wogonoside
F
HQ
47
8.48
[M−H]-
C22H22O11
461.1084
461.1084
0.0
461.1084 [M−H]-; 315.0685 [M−H−C9H7O2]-; 313.0798 [M−H−C9H7O2−2H]-
2-O-cinnamoyl-glucogallin
O
DH
48
8.76
[M−H]-
C44H64O19
895.3960
895.3964
−0.3
895.3960 [M−H]-; 837.3891 [M−H−C2H2O2]-
22β-acetoxyl licorice saponin G2
T
GC
49
9.00
[M−H]-
C44H70O23
965.4227
965.4230
−0.3
965.4230 [M−H]-; 803.3706 [M−H−Glc]-;641.3164 [M−H−2Glc]-
rebaudioside A
D
FL
50*
9.08
[M−H]-
C38H60O18
803.3707
803.3701
0.6
803.3707 [M−H]-; 641.3176 [M−H−Glc]-; 479.2609 [M−H−2Glc]-
stevioside
D
FL
51
9.33
[M−H]-
C26H45NO7S
514.2843
514.2838
1.0
514.2843 [M−H]-
taurocholic acid
B
RGNH
52
9.40
[M−H]-
C16H12O5
935.4132
935.4124
0.9
935.4132 [M−H]-; 283.0602 [M−H−3Glc−Xyl]-
rebaudioside B xyloside
D
FL
53
9.52
[M−H]-
C44H70O22
949.4291
949.4280
1.2
949.4291 [M−H]-; 787.3871 [M−H−Glc]-; 625.3272 [M−H−2Glc]-
rebaudioside C
D
FL
54*
9.69
[M−H]-
C15H12O4
255.0649
255.0657
−3.1
255.0649 [M−H]-; 119.0503 [1,3B0]-
isoliquiritigenin
F
GC
55
9.74
[M−H]-
C38H60O18
803.3693
803.3701
−1.0
849.3749 [M−H + HCOOH]-; 803.3693 [M−H]-; 641.3174 [M−H−Glc]-; 479.2610 [M−H−2Glc]-
rebaudioside G
D
FL
56
9.77
[M−H]-
C42H62O17
837.3904
837.3909
−0.6
837.3904 [M−H]-; 485.3302 [M−H−2GulA]-; 351.0554 [M−H−C30H46O5]-; 193.0357 [M−H−C36H52O10]-
licorice-saponin P2
T
GC
57
10.07
[M + H]+
C16H12O4
269.0816
269.0814
0.7
269.0816 [M + H]+
formononetin
F
GC
58
10.42
[M−H]-
C16H10O6
297.0400
297.0399
0.3
297.0400 [M−H]-; 253.0497 [M−H−CO2]-
glyzaglabrin
F
GC
59
10.47
[M−H]-
C42H60O16
819.3802
819.3803
−0.1
819.3802 [M−H]-; 351.0555 [M−H−C30H44O4]-
licorice-saponin E2
T
GC
60*
10.49
[M−H]-
C42H62O17
837.3914
837.3909
0.6
837.3914 [M−H]-; 351.0561 [M−H−C30H46O5]-
licorice-saponin G2
T
GC
61
10.60
[M−H]-
C32H50O13
641.3170
641.3173
−0.5
641.3170 [M−H]-; 479.2609 [M−H−Glc]-
steviobioside
D
FL
62
10.72
[M + H]+
C26H43NO6
466.3169
466.3169
0.0
931.6238 [2 M + H]+; 466.3169 [M + H]+; 448.3058 [M + H-H2O]+; 430.2952 [M + H-2H2O]+; 412.2847 [M + H-3H2O]+; 337.2531 [M + H-3H2O-C2H5NO2]+
glycocholic acid
B
RGNH
63
10.77
[M−H]-
C42H62O17
837.3921
837.3909
1.4
837.3921 [M−H]-
uralsaponin U
T
GC
64
10.82
[M−H]-
C38H60O17
787.3761
787.3752
1.1
787.3761 [M−H]-; 625.3240 [M−H−Glc]-
dulcosideA
D
FL
65
10.96
[M−H]-
C26H45NO6S
498.2889
498.2889
0.0
498.2889 [M−H]-
taurodeoxycholic acid
B
RGNH
66
11.10
[M + H]+
C42H62O16
823.4115
823.4116
−0.5
823.4115 [M + H]+; 647.3783 [M + H-GluA]+; 453.3377 [M + H-2GluA-H2O]+
licorice-saponin K2
T
GC
67*
11.16
[M + H]+
C42H62O16
823.4118
823.4116
0.2
823.4118 [M + H]+; 805.3953 [M + H-H2O]+; 647.3783 [M + H-GluA]+; 471.3643 [M + H-2GluA]+; 453.3363 [M + H-2GluA-H2O]+; 435.3257 [M + H-2GluA-2H2O]+; 407.3294 [M + H-2GluA-2H2O-CO]+; 285.2221 [ M + H-2GluA-2H2O-CO-C9H14]+; 189.1645 [ M + H-2GluA-2H2O-CO-C9H14-C5H8O]+
glycyrrhizic acid
T
GC
68*
11.25
[M−H]-
C15H8O6
283.0239
283.0243
−0.2
283.0239 [M−H]-; 239.0344 [M−H−CO2]-; 211.0388 [M−H−CO2−CO]-; 183.0446 [M−H−CO2−2CO]-
rhein
AN
DH
69
11.27
[M−H]-
C26H45NO6S
498.2898
498.2889
1.8
498.2898 [M−H]-
taurochenodeoxycholic acid
B
RGNH
70*
11.32
[M−H]-
C16H12O5
283.0608
283.0606
0.2
283.0606 [M−H]-; 268.0365[M-H-CH3]-;240.0413[M-H-CH3-CO]-; 239.0336[M-H-CH4-CO]-;212.04553[M-H-CH3-CO-H2O]-
wogonin
F
HQ
71
11.40
[M−H]-
C42H64O15
807.4149
807.4167
−1.6
807.4149 [M−H]-; 351.0558 [M−H−C30H48O3]-; 193.0343 [M−H−C36H54O8]-
licorice-saponin B2
T
GC
72
11.46
[M−H]-
C15H10O4
253.0493
253.0501
0.3
253.0493 [M−H]-; 225.0565 [M−H−CO]-; 209.0600[M-H-CO2]-; 181.0699 [M−H−CO−CO2]-; 153.0745[M-H-2CO-CO2]-
chrysin
F
HQ
73
11.60
[M−H]-
C24H40O5
407.2793
407.2797
−1.0
407.2793 [M−H]-; 389.2767 [M−H−H2O]-; 371.2608 [M−H−2H2O]-; 353.2482 [M−H−3H2O]-; 345.2792 [M−H−CO2−H2O]-
allocholic acid
B
RGNH
74*
11.77
[M−H]-
C19H18O8
373.0931
373.0923
2.1
373.0931 [M−H]-; 358.0697 [M−H−CH3]-; 343.0466 [M−H−2CH3]-; 328.0220 [M−H−3CH3]-; 313.0011 [M−H−4CH3]-
skullcapflavone II
F
HQ
75*
11.85
[M + H]+
C42H62O16
823.4099
823.4116
−2.1
823.4009 [M + H]+; 647.3802 [M + H-GluA]+; 453.3364 [M + H-2GluA-H2O]+; 263.1619 [M + H-2GluA-H2O-C14H22]+; 191.1812 [M + H-2GluA-H2O-C16H22O3]+
(18β, 20α)-glycyrrhizic acid
T
GC
76
12.05
[M−H]-
C24H40O5
407.2795
407.2797
−0.5
407.2795 [M−H]-; 389.2698 [M−H−H2O]-; 371.2572 [M−H−2H2O]-
hyocholic acid
B
RGNH
77
12.08
[M + H]+
C42H62O16
823.4100
823.4116
−1.9
823.4100 [M + H]+; 647.3790 [M + H-GluA]+; 453.3354 [M + H-2GluA-H2O]+
uralsaponin B
T
GC
78*
12.19
[M−H]-
C24H40O5
407.2794
407.2797
−0.7
815.5669[2 M−H]-; 407.2794 [M−H]-; 389.2696 [M−H−H2O]-; 371.2601 [M−H−2H2O]-; 353.2473 [M−H−3H2O]-; 345.2807 [M−H−CO2−H2O]-; 343.2636 [M−H−2H−CO2−H2O]-; 327.2683 [M−H−CO2−2H2O]-; 325.2518 [M−H−2H−CO2−2H2O]-; 289.2173 [M−H2O−C5H8O2]-
cholic acid
B
RGNH
79*
12.46
[M−H]-
C24H40O4
391.2850
391.2848
0.5
783.5778 [2 M−H]-; 391.2850 [M−H]-;373.2743 [M−H−H2O]-; 355.0060 [M−H−2H2O]-; 328.9925 [M−H−CO2−H2O]-
hyodeoxycholic acid
B
RGNH
80
12.56
[M−H]-
C42H64O16
823.4114
823.4116
−0.2
823.4114 [M−H]-; 351.0561 [2GluA]-
uralsaponin C
T
GC
81
12.68
[M−H]-
C21H24O5
355.1548
355.1545
0.8
711.3177 [2 M−H]-; 355.1548 [M−H]-
glyasperin C
F
GC
82
12.82
[M + H]+
C26H43NO5
450.3228
450.3219
0.2
450.3228 [M + H]+; 414.3009 [M + H-2H2O]+; 339.2680 [M + H-2H2O-C2H5NO2]+
glycohyodeoxycholic acid
B
RGNH
83
13.16
[M−H]-
C20H18O6
353.1024
353.1025
−0.3
353.1024 [M−H]-
licoflavonol
F
GC
84
13.24
[M−H]-
C42H62O16
821.3979
821.3960
2.3
821.3979 [M−H]-; 351.0574 [M−H−C30H46O4]-; 193.0386 [M−H−C36H52O9]-
licorice-saponin H2
T
GC
85
13.51
[M−H]-
C42H64O16
823.4111
823.4116
−0.6
823.4111 [M−H]-; 351.0869 [M−H−C30H48O4]-
licorice-saponin J2
T
GC
86
13.54
[M−H]-
C22H22O6
381.1330
381.1336
−0.8
381.1330 [M−H]-; 351.0880 [M−H−CH2O]-; 323.0558 [M−H−CH2O−CO]-
licoricone
F
GC
87
13.65
[M−H]-
C15H10O5
269.0446
269.0450
0.3
269.0446 [M−H]-; 251.0382 [M−H−H2O]-; 241.0497 [M−H−CO]-; 225.0540 [M−H−CO2]-; 197.0602 [M−H−CO2−CO]-; 195.0467 [M−H−2CO−H2O]-
baicalein
F
HQ
88*
14.27
[M−H]-
C24H40O4
391.2848
391.2848
0.0
783.5778 [2 M−H]-; 391.2848 [M−H]-; 373.2743[M-H-H2O]-; 355.0060 [M−H−2H2O]-; 328.9925[M-H-CO2-H2O]-
chenodeoxycholic acid
B
RGNH
89
14.37
[M−H]-
C20H16O6
351.0867
351.0869
−0.6
351.0867 [M−H]-
licoisoflavone B
F
GC
90*
14.44
[M−H]-
C24H40O4
391.2851
391.2848
0.8
783.5765 [2 M−H]-; 391.2851 [M−H]-; 373.2748 [M−H−H2O]-; 355.0062 [M−H−2H2O]-; 328.9931 [M−H−CO2−H2O]-
deoxycholic acid
B
RGNH
91
14.89
[M−H]-
C22H26O5
369.1700
369.1702
−0.2
369.1700 [M−H]-; 341.1080 [M−H−CO]-
glyasperin D
F
GC
92*
15.46
[M−H]-
C30H46O4
469.3325
469.3318
0.7
469.3325 [M−H]-; 433.2961 [M−H−2H2O]-
glycyrrhetinic acid
T
GC
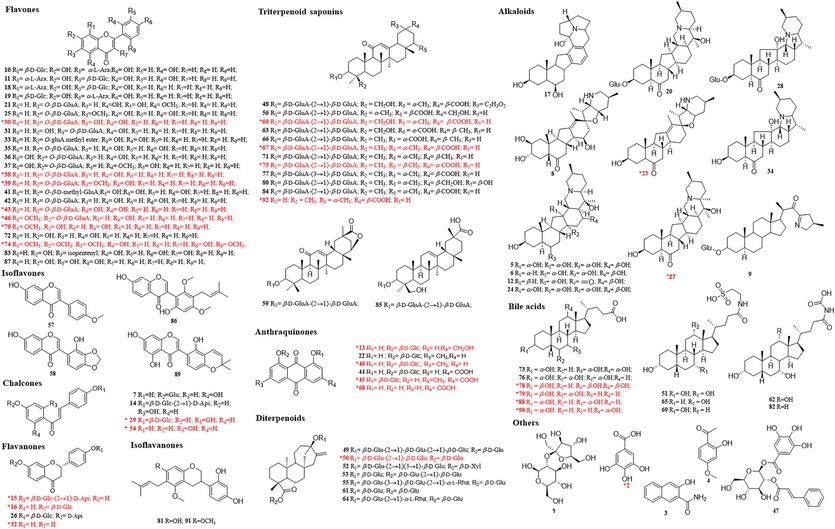
Chemical structures of components identified or characterized in JZOL (red color represented the confirmed compounds by comparison with reference standards). (For interpretation of the references to color in this figure legend, the reader is referred to the web version of this article.)
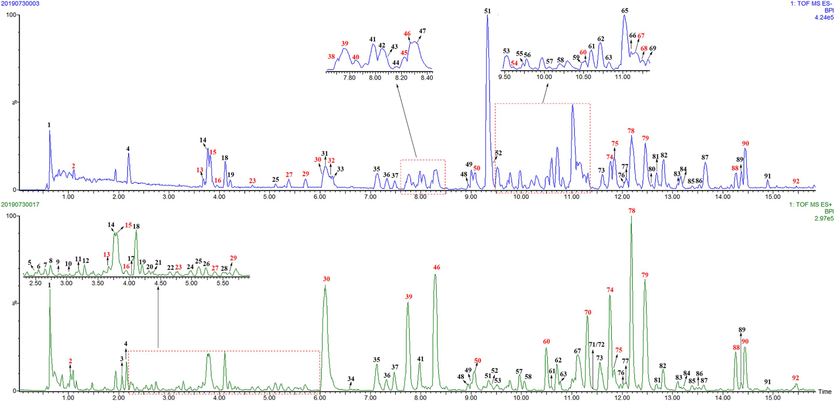
The basic peak ions chromatography of JZOL detected by UPLC-Q/TOF-MS (red color represented the confirmed compounds by comparison with reference standards). (For interpretation of the references to color in this figure legend, the reader is referred to the web version of this article.)
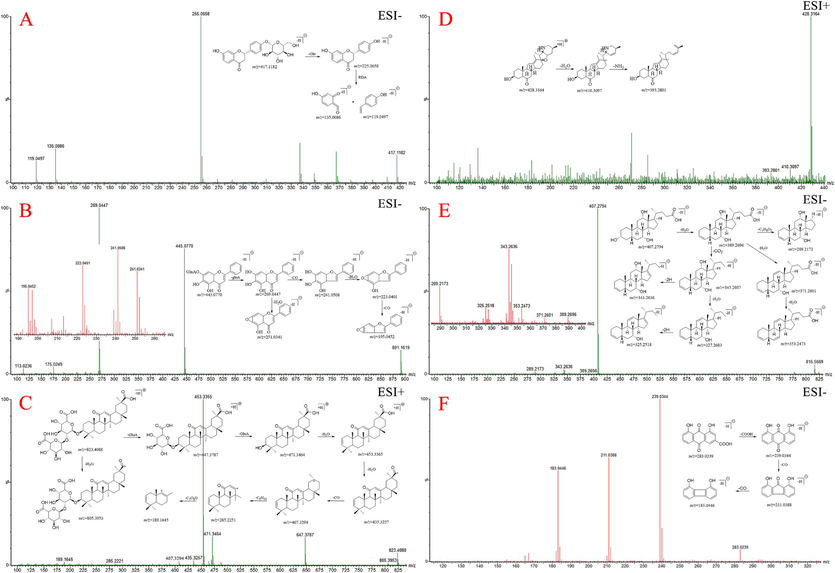
The detailed fragmentation and proposed fragment pathways of compounds (A) 16-liquiritin; (B) 30-baicalin; (C) 67-glycyrrhizic acid; (D) 23-peimisine; (E) 78- cholic acid; (F) 68-rhein.
3.2 Method validation of pharmacokinetic research
Based on the analysis of exogenous substances by JZOL in rats, 21 accurately identified prototype components were detected in rat plasma (Zhang et al., 2022, Table S3), 11 of which showed high exposure and covered different structural types of the compounds. These compounds included baicalin (flavonoids), wogonoside (flavonoids), wogonin (flavonoids) and oyoxylin A-7-O-β-d-gluA (flavonoids) from Scutellariae Radix, liquiritin (flavonoids), liquiritigenin (flavonoids), isoliquiritigenin (flavonoids), isoliquiritin (flavonoids), glycyrrhizic acid (triterpenoid saponins) from Glycyrrhizae Radix et Rhizoma, rhein from Rhei Radix et Rhizoma, and peimisine (alkaloids) from Fritillariae Ussuriensis Bulbus.
A set of method validation tests of 11 analytes (Fig. S5) were conducted in the matrix, including specificity and selectivity, standard curve and linear range, precision and accuracy, extraction recovery and matrix effect, and stability. Specificity testing demonstrated no or negligible chromatographic interference to the analytes (Fig. S6). All analytes exhibited good linearity (r ≥ 0.99) within the test ranges (Table S5), and both the accuracy (RE: 3.33 %-8.68 %) and precision (RSD: −2.32 %-2.16 %) of the LLOQ met the requirements of the guidelines (Table S6), indicating that the established method was sufficiently sensitive for quantifying the 11 analytes in rat plasma. As shown in Table S7, the values of intra- and inter-day precision at three concentrations (LQC, MQC, HQC) were less than 15 %. And the accuracy (RE%) of 11 analytes ranged between −10.16 % to 14.05 % for intra-day and −7.51 %–10.01 % for inter-day, demonstrating satisfactory precision and accuracy for pharmacokinetic studies. The recoveries of the analytes varied from 84.79 % to 107.94 % (glycyrrhizic acid: 10.79 %) and the recovery of IS was 100.74 %, illustrating consistent recovery and precision (Table S8). The matrix effects of the analytes ranged from 85.46 % to 111.97 % with RSDs within 8.94 %, indicating no remarkable matrix effect in rat plasma (Table S8). The carry-over effect was negligible in this method, as insignificant peaks were observed in each channel of the blank plasma sample after ULOQ (Fig. S7). The RSD values of stability under different conditions (free-thaw cycles, room temperature for 8 h, −80 °C for 2 weeks, MS Auto–sampler for 24 h) were less than 12.54 % (Table S9), indicating that the analytes remained stable under analytical conditions. The summarized results prove the validity of the developed UPLC-MS/MS method for the pharmacokinetic analysis of 11 ingredients in rat plasma.
3.3 Pharmacokinetic studies
The validated UPLC-MS/MS method for analyzing 11 components in rat plasma was applied for the pharmacokinetic study following JZOL oral administration. The mean plasma concentration–time profiles of the 11 detected prototypes are shown in Fig. 5. Statistical analysis of the pharmacokinetic parameters was performed with WinNonlin 6.3 software, and the corresponding estimated pharmacokinetic parameters (t1/2, Tmax, Cmax, AUC0-∞, AUC0-t, MRT0-∞, MRT0-t) are given in Table 2.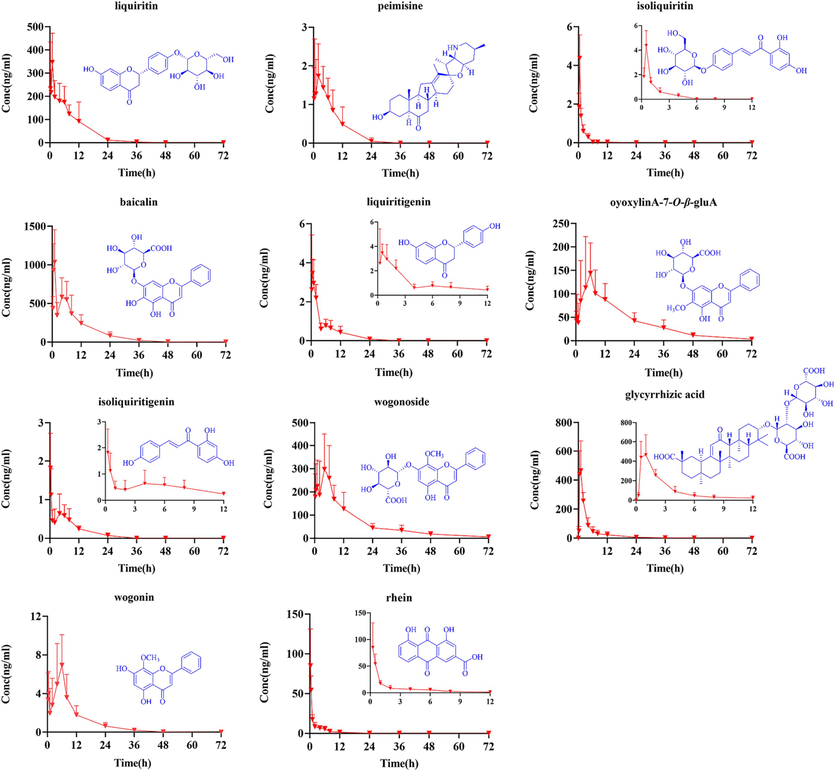
Comparison of serum concentration–time curves of 11 analytes in JZOL (n = 7).
Name
Tmax (h)
t1/2 (h)
Cmax(ng/mL)
AUC0-t (ng*h*mL−1)
AUC0-∞(ng*h*mL−1)
MRT0-t (h)
MRT0-∞ (h)
liquiritin
2.54 ± 2.44
4.16 ± 1.92
138.02 ± 40.22
1086.90 ± 344.28
1111.93 ± 343.32
6.87 ± 1.88
7.52 ± 1.80
peimisine
5.37 ± 2.92
38.29 ± 32.94
1.59 ± 0.46
18.36 ± 2.28
35.16 ± 16.40
13.62 ± 3.78
53.67 ± 42.07
isoliquiritin
0.50 ± 0.00
1.39 ± 0.27
4.59 ± 1.33
4.72 ± 0.87
4.78 ± 0.87
1.39 ± 0.25
1.51 ± 0.23
baicalin
4.33 ± 0.82
11.15 ± 5.40
1134.15 ± 550.36
9650.72 ± 5013.90
10596.89 ± 5194.60
12.93 ± 3.37
17.47 ± 8.83
liquiritigenin
0.75 ± 0.27
7.28 ± 2.09
4.05 ± 0.75
14.79 ± 3.49
17.27 ± 5.78
5.56 ± 1.14
8.61 ± 2.55
oyoxylinA-7-O-β-d-gluA
9.37 ± 8.11
50.93 ± 35.79
98.97 ± 63.50
2164.23 ± 1398.77
3531.70 ± 2526.75
25.78 ± 5.50
70.06 ± 44.04
wogonoside
5.37 ± 3.86
15.85 ± 7.13
343.88 ± 223.12
3685.52 ± 869.65
4011.55 ± 1089.60
18.02 ± 5.03
23.87 ± 9.00
isoliquiritigenin
0.37 ± 0.14
8.80 ± 6.26
2.01 ± 0.84
8.10 ± 2.02
9.69 ± 3.19
7.69 ± 0.87
12.70 ± 7.13
glycyrrhizic acid
1.92 ± 1.71
9.27 ± 2.60
528.59 ± 109.52
2201.08 ± 1040.42
2323.70 ± 1141.28
5.93 ± 1.66
7.92 ± 3.16
wogonin
7.95 ± 2.54
5.32 ± 0.62
8.73 ± 2.74
72.45 ± 24.00
72.68 ± 24.00
9.91 ± 1.04
10.07 ± 1.04
rhein
0.42 ± 0.13
9.86 ± 2.73
18.98 ± 7.19
87.12 ± 46.50
89.85 ± 46.70
9.38 ± 3.01
11.70 ± 4.10
According to the pharmacokinetic profiles of the 11 ingredients from JZOL, the t1/2 value was in the range of 1.39–50.93 h. Liquiritin, wogonoside, oroxylin A 7-O-β-d-glucuronide and baicalin were rapidly absorbed, with high Cmax values (138.02 ± 40.22, 343.88 ± 223.12, 98.97 ± 63.50 and 1134.15 ± 550.36 ng/mL) and high AUC0-∞ values (1086.90 ± 344.28, 3685.52 ± 869.65, 2164.23 ± 1398.77 and 9650.72 ± 5013.90 ng*h*mL−1), indicating high exposure and peak concentrations in blood. The Cmax values of wogonin, isoliquiritigenin, liquiritigenin, and isoliquiritigenin ranged from 1.67 to 8.73 ng/mL. Among them, isoliquiritigenin and liquiritigenin exhibited two peaks in the pharmacokinetic profile.
Rhein derived from Rhei Radix et Rhizoma demonstrated rapid but poor absorption from the gastrointestinal tract (Tmax = 0.42 ± 0.13 h, t1/2 = 9.86 ± 2.73 h, Cmax = 18.98 ± 7.19 ng/mL, AUC0-∞=89.85 ± 46.70 ng*h*mL−1, MRT0-∞=11.70 ± 4.10 h). In this research, the peak plasma concentration (Cmax) of glycyrrhizic acid was 528.59 ± 109.52 ng/mL, and its AUC0-t reached 2201.08 ± 1040.42. Peimisine had a longer peak time (Tmax = 5.37 ± 2.92 h), a lower peak value (Cmax = 1.59 ± 0.46 ng/mL), and a relatively longer in vivo residence time (MRT0-∞=53.67 ± 42.07 h).
3.4 Network pharmacology
By integrating 13 candidate components selected by chemical profile with 21 components selected by metabolite profile and PK profile, 22 candidate Q-markers were selected. As shown in Fig. 6A, an overlap of 46 targets was simultaneously involved in the interactions with 22 Q-marker candidates and the regulation of inflammation, suggesting that these targets formed a network target group (Wang et al., 2022) for the anti-inflammatory effect of JZOL. To reveal the relationship among these targets, the protein–protein interaction (PPI) network was constructed as shown in Fig. 6B and sequent subnetwork extraction showed that 12 targets in Mode 1 (BCL2L1, MAPK8, PPARG, MAPK14, HSP90AA1, IKBKB, NOS2, PTGS2, NFKB1, CASP1, GAPDH, TLR9) have had the most significant interaction association and were likely to be the core of the network target group. The further pharmacological network analysis (Fig. 6C) also suggests that these targets from a broad interaction with all 22 compounds, with targets like PTGS2 and CASP1 potentially playing a more important role by interacting with more compounds than other targets. The further signaling pathway analysis also suggested that “signaling by interleukins” and “cytokine signaling in immune system” were the top two significant pathways (Fig. 6D) and were mainly associated with most of the targets in Mode 1 (Fig. S7). As for the regulatory effect of Gypsum Fibrosum and Chloriti Lapi, it’s indicated that nine targets (CXCR4, HPSE, LTB4R, PLA2G4A, PRKCA, PRKCQ, PRKCG, PTAFR, TRPV1) among the network target group (Fig. 6A) were involved in the interaction with the ionic composition in Gypsum Fibrosum and Chloriti Lapis. All nine targets, except HPSE, were recognized for Gypsum Fibrosum and Chloriti Lapis, probably due to the common ionic composition of Ca2+. These targets mainly played an auxiliary role by affecting pathways such as “signaling by GPCR” (Fig. S8), which is consistent with the Chinese medicine theory that Gypsum Fibrosum and Chloriti Lapis mainly assist other herbs in exerting their function.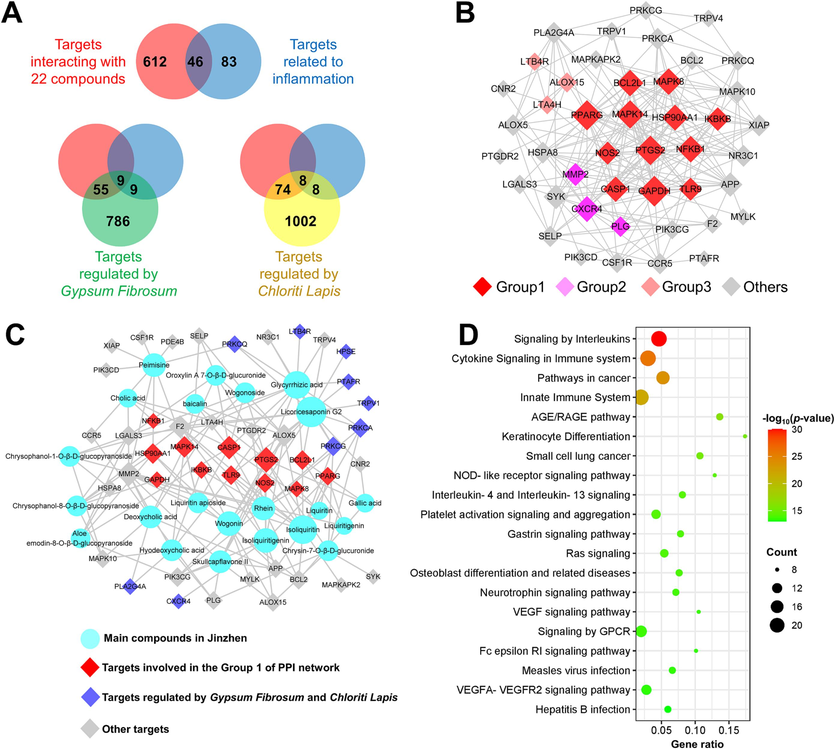
The potential regulation effect of 22 Q-marker candidates as well as Gypsum Fibrosum and Chloriti Lapis in JZOL via the network pharmacology analysis. (A) The Venn diagram for the discovery of 46 key targets from different target sets, namely targets interacting with compounds, targets related to inflammation, targets regulated by G. Fibrosum and targets regulated by C. Lapis. (B) The main target interaction mode recognized by the protein–protein interaction analysis. (C) The pharmacological network involved in the 22 Q-marker candidates in JZOL and ions in G. Fibrosum and C. Lapis. (D) The top 20 significant signaling pathways by GSEA analysis.
The binding affinities between 46 targets in the network target group and 22 Q-marker candidates of JZOL were evaluated further through molecular docking, as shown in Fig. 7A. It can be seen that all compounds, except Glycyrrhizic acid and Licoricesaponin G2, formed a direct interaction with the network target group. These two compounds might exert indirect regulation with the targets related to Gypsum Fibrosum and Chloriti Lapis, as shown in Fig. 6C. Based on the cutoff of binding affinities, the Q-marker candidates showed different levels of importance in targeting the network target group, as indicated by the statistics of target numbers (Fig. 7B).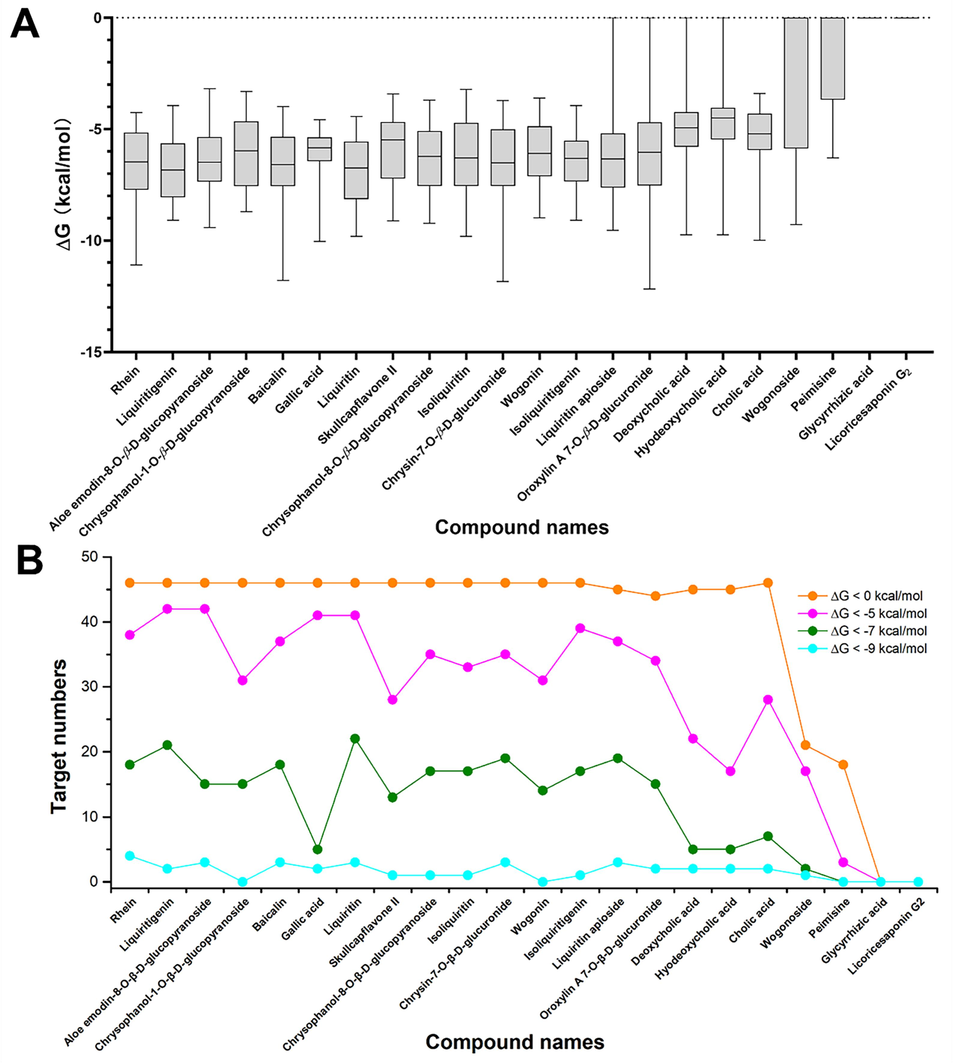
The affinity evaluation between 22 Q-marker candidates and 46 key targets by molecular docking. (A) The affinity (ΔG) distribution for 22 Q-marker candidates when interacting 46 key targets were evaluated by boxplot. (B) The importance of candidates analyzed by the number of interacting targets with different affinity cutoffs (0, −5, −7, −9 kcal/mol).
3.5 Candidate Q-markers prediction based on multi-factor analysis
A table filled with five factors corresponding to 22 selected components was input to the SPSSPRO website for multi-factor analysis. Firstly, through pre-computation and applicability test, the statistic Kaiser-Meyer-Olkin (KMO) values were 0.645, which were greater than 0.6, and the p values of Bartlett's Test were equal to 0.000, reaching a very significant level. This indicates that the 22 candidate Q-markers covering five factors were suitable for factor analysis (Table S10, S11). Based on the result of the explanation rate of cumulative variance after rotation, the common factors were determined as three, which explained the information of 97.1 % of the original data (X1 = 56.11 %, X2 = 20.92 %, and X3 = 20.11 %). The three common factors were mainly attributed to Cmax (PK result), compatibility, and network pharmacology, indicating their importance in distinguishing candidate Q-markers. According to Eq (2)–(3), the F value for each candidate Q-marker was then calculated between −0.70 to 2.87, and component 30 (baicalin, F = 2.87), component 39 (oroxylin A 7-O-β-d-glucuronide, F = 0.44), and component 38 (chrysin-7-O-β-d-glucuronide, F = 0.41) were the top three ingredients (Table 3). It is worth noting that the F score for baicalin is almost five times that of the second chemical, preliminarily proving the importance of baicalin in JZOL, as stated in the 2020 edition of the Pharmacopoeia of the People's Republic of China.
Rank
Name
F value
Compatibility (min–max standardized)
System pharmacology (min–max standardized)
Cmax (min–max standardized)
Content (min–max standardized)
AUC0-∞ (min–max standardized)
1
Baicalin
2.87
1.00
0.82
1.00
1.00
1.00
2
Oroxylin A 7-O-β-d-glucuronide
0.46
1.00
0.68
0.09
0.09
0.33
3
Chrysin-7-O-β-d-glucuronide
0.41
1.00
0.86
0.18
0.01
0.19
4
Wogonoside
0.38
1.00
0.09
0.30
0.21
0.38
5
Skullcapflavone II
0.20
1.00
0.59
0.18
0.00
0.19
6
Chrysopyranol-8-O-β-d-glucopyranoside
0.06
0.50
0.77
0.18
0.01
0.19
7
Aloeemodin-8-O-β-d-glucopyranoside
0.02
0.50
0.68
0.18
0.03
0.19
8
Hyodeoxycholic acid
−0.03
0.50
0.23
0.18
0.26
0.19
9
Liquiritin
−0.09
0.00
1.00
0.12
0.12
0.10
10
Wogonin
−0.10
1.00
0.64
0.01
0.00
0.01
11
Cholic acid
−0.10
0.50
0.32
0.18
0.13
0.19
12
Glycyrrhizic
−0.13
0.00
0.00
0.47
0.35
0.22
13
Liquirtin apioside
−0.15
0.00
0.86
0.18
0.00
0.19
14
Gallic acid
−0.18
0.50
0.23
0.18
0.11
0.19
15
Rhein
−0.24
0.50
0.82
0.02
0.00
0.01
16
Deoxycholic acid
−0.30
0.50
0.23
0.18
0.00
0.19
17
Chrysopyranol-1-O-β-d-glucopyranoside
−0.35
0.50
0.68
0.00
0.00
0.00
18
Liquiritigenin
−0.41
0.00
0.95
0.00
0.02
0.00
19
Peimisine
−0.52
1.00
0.00
0.00
0.00
0.00
20
Isoliquiritin
−0.55
0.00
0.77
0.00
0.00
0.00
21
Isoliquiritigenin
−0.55
0.00
0.77
0.00
0.00
0.00
22
Licorice-saponin G2
−0.71
0.00
0.00
0.18
0.00
0.19
Based on the results of the multi-factor analysis, seven components (aloeemodin-8-O-β-d-glucopyranoside, baicalin, chrysin-7-O-β-d-glucuronide, oroxylin A 7-O-β-d-glucuronide, wogonoside, chrysophanol-8-O-β-d-glucopyranoside, skullcapflavone II) were selected as candidate Q-markers of JZOL with the F > 0.
3.6 Activity evaluation
The anti-inflammatory activities of the above seven candidate Q-markers were further verified through anti-inflammatory assays, with two chemicals (16 and 79) with F<0 selected as negative controls. Firstly, all chemicals were screened at respective maximum non-toxic concentrations. The concentrations of compounds 13, 16, 30, 38, 39, 45, 46, 74 and 79 were 50, 100, 50, 3.13, 50, 12.5, 3.13, 12.5, and 3.13 µg/ml, respectively. As shown in Fig. 8, baicalin (30), oroxylin A 7-O-β-d-glucuronide (39) and wogonoside (46) showed significant anti-inflammatory activities in inhibiting the production of inflammatory factors (NO, IL-6, IL-1β, and PGE2) in LPS-induced RAW264.7 cells. Compounds 13 and 38 could effectively reduce the release of NO and PGE2; compound 45 has a significant effect on reducing the release of IL-1β and PGE2 at a lower concentration range, and compound 74 showed substantial reduction in the release of IL-6 and NO. Although compounds 16 and 79 showed no obvious therapeutic effect on the constructed cell model and evaluation indicators in this experiment, it further demonstrated that the rationality of the strategy used in this study in balancing the various properties of the Q-markers simultaneously and in screening out suitable anti-inflammatory Q-markers from hundreds and thousands of ingredients in TCMps.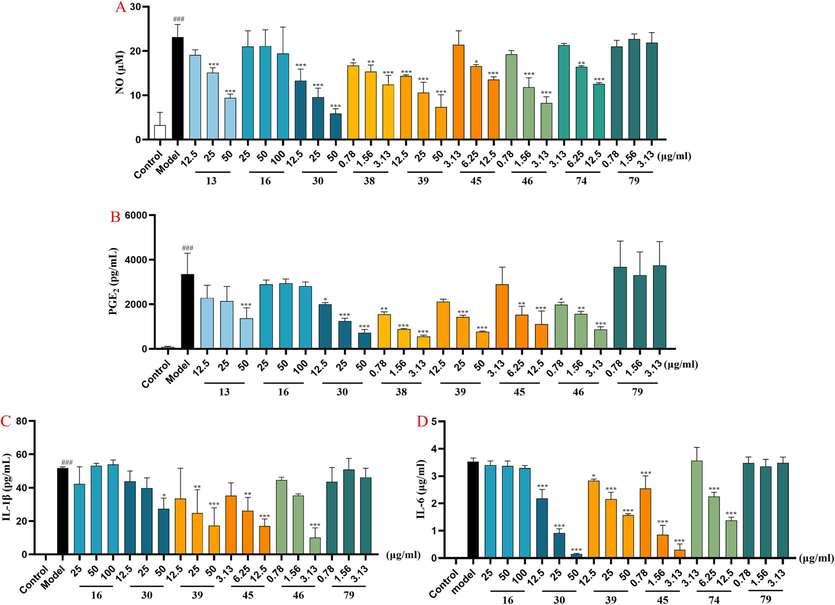
The anti-inflammatory activity evaluation of compounds 13, 16, 30, 38, 39, 45, 46, 74 and 79 with the LPS- induced RAW264.7 cell model (‾x ± s, n = 3). (A) statistical analysis of inhibition on NO production, (B) statistical analysis of inhibition on PGE2 production, (C) tatistical analysis of inhibition on IL-1β production, (D) statistical analysis of inhibition on IL-6 production. Cells were exposed to three different administration concentrations (low, medium, high) of compounds and LPS. ###p < 0.001 vs. control group and ***p < 0.001, **p < 0.01, *p < 0.05 vs. LPS-treated group.
4 Discussion
The “multi-component, multi-target and multi-pathway” feature of TCMps poses significant challenges in achieving dependable quality control, which is a crucial factor restricting the modernization process of traditional Chinese medicine (Yang et al., 2017, He et al., 2018). To enhance the quality standards, it is necessary to identify Q-markers that can represent the entire formula and establish scientific methods for evaluation. In this study, we focus on JZOL as an example and delve into the discovery of quality markers for TCMps. JZOL is a Chinese herbal formula used for treating acute bronchitis in children for hundreds of years. According to the ‘‘Pharmacopoeia of the People’s Republic of China in 2020’’, the quality standard for JZOL is to control the content of baicalin not lower than 0.25 mg/ml. However, a single-component standard is insufficient for Chinese herbal formulas due to their multiple components, targets, and mechanisms of action. Currently, research on JZOL mainly focuses on the identification of active substances and the exploration of its mechanisms of action, with limited studies on improving quality control standards. Therefore, this study proposes a comprehensive multi-factor analysis strategy by conducting systematic investigations into the chemical composition, pharmacokinetics, and network pharmacology of JZOL. The goal is to screen representative anti-inflammatory quality markers for JZOL and validate their reliability through anti-inflammatory activity evaluation.
In our research, UPLC-Q/TOF-MS enables precise JZOL chemical identification with high resolution and accurate mass measurement, while UPLC-QqQ-MS facilitates pharmacokinetic profile characterization with high sensitivity and selective quantification. However, the animal-derived and mineral drugs were fall to detecte by MS technology. We conducted qualitative and quantitative analysis of the amino acid components in JZOL to elucidate the potential contribution of animal drugs in the formulation in our previous research (Li et al., 2020b). In our current research, network pharmacology was employed to identify key targets related to mineral drugs and their interactions with the screened components, aiming to elucidate the mechanism of action of mineral drugs in JZOL. Furthermore, a novel “multi-factor analysis” strategy was introduced to address the Q-marker screening successfully. The strategy was applied to determine anti-inflammatory Q-markers of JZOL by integrating chemical profile characterization, PK research, and network pharmacology research. Ultimately, seven components were selected as anti-inflammatory Q-markers of JZOL, namely aloeemodin-8-O-β-d -glucopyranoside (13), baicalin (30), chrysin-7-O-β- d -glucuronide (38), oroxylin A 7-O-β- d -glucuronide (39), wogonoside (46), chrysophanol-8-O-β- d -glucopyranoside (45), and skullcapflavone II (74). The anti-inflammatory activity of the seven Q-markers was further verified to confirm the rationality. Furthermore, the result showed that baicalin was superior to other components in reducing productions of NO, IL-6, IL-1β and PGE2, which consisted with the finding of previous studies that polymethoxy flavones have greater anti-inflammatory activity (Wang et al., 2014). Therefore, baicalin was determined as the most important Q-markers of JZOL, corresponding to the current quantitative indicator of JZOL recorded by ‘‘Pharmacopoeia of the People’s Republic of China in 2020’’.
Although the “multi-factor analysis” strategy represents a promising approach to discovering Q-markers in quality control research of TCMps, the study still have some limitations. Firstly, the selected factors have a greater impact on the screening of Q-markers, and the research results could not screen out the components that exist in animal drugs (Caprae Hircus Cornu). Secondly, the visualization of the method and the obtained results are insufficient. Furthermore, our research mainly focuses on the anti-inflammatory activities, the result presented the anti-inflammatory Q-markers. Other ingredients of JZOL may exert therapeutic effects through other pathways of activation: liquiritin mediated the antitussive effects through dual inhibition of TRPV1 and TRPA1 channels (Liu et al., 2020). And hyodeoxycholic acid may serve efforts in antipyretic of JZOL (Zhang et al., 2016).
5 Conclusion
In this study, a “multi-factor analysis” strategy was conducted to analyze the compatibility contribution of herbal medicines, the content, the in vivo PK characteristics, and the degree of network pharmacology of the components, aiming to select suitable anti-inflammatory Q-markers of JZOL. As a result, a total of seven ingredients (aloeemodin-8-O-β-d-glucopyranoside, baicalin, chrysin-7-O-β-d-glucuronide, oroxylin A 7-O-β-d-glucuronide, wogonoside, chrysophanol-8-O-β-d-glucopyranoside, and skullcapflavone II) with better anti-inflammatory activity were selected as anti-inflammatory Q-markers of JZOL. This new strategy provides new insights for the discovery of quality markers in TCMps and lays a solid foundation for their quality control improvement.
CRediT authorship contribution statement
Ling-xian Liu: Conceptualization, Methodology, Software, Formal analysis, Investigation, Data curation, Writing – original draft, Visualization. Hai-bo Li: Conceptualization, Methodology, Software, Formal analysis, Investigation, Data curation, Writing – original draft, Visualization. Jia-ying Zhang: Investigation, Formal analysis, Data curation. Dan-feng Shi: Formal analysis, Data curation, Writing – original draft. Zhen-zhong Wang: Supervision, Funding acquisition, Project administration. Xin-sheng Yao: Supervision, Funding acquisition, Project administration. Wei Xiao: Conceptualization, Supervision, Methodology, Writing – review & editing, Project administration. Yang Yu: Conceptualization, Supervision, Methodology, Writing – review & editing, Project administration.
Acknowledgments
This work was supported by the Programs Foundation for Leading Talents in National Administration of Traditional Chinese Medicine of China ‘‘Qihuang scholars” Project. The authors are grateful to State Key Laboratory of New-Tech for Chinese Medicine Pharmaceutical Process, Jiangsu Kanion Pharmaceutical Co. Ltd. for their assistance of activity test. The network pharmacology analysis and molecular docking in this work were supported by the high-performance computing platform of Jinan University.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- W.N. Amala R.A.Z. Aziz N. Rohmah H.N. Imtiyaz M.D.I. Iskandar E.R. Purnama In silico Exploration on The Potency of Basil (Ocimum basilicum) as an Anti-Aging Skin Agent Bioactivities. https:// 2023 doi.org/10.47352/bioactivities.2963-654X.193.
- Progress in ICP-MS analysis of minerals and heavy metals in traditional medicine. amage. Front. Pharmacol.. 2022;13
- [CrossRef] [Google Scholar]
- Identifying quality markers of Mailuoshutong pill against thromboangiitis obliterans based on chinmedomics strategy. Phytomedicine. 2022;104:154313
- [CrossRef] [Google Scholar]
- Efficacy, safety and mechanism of Jinzhen oral liquid in the treatment of acute bronchitis in children: A randomized, double-blind, multicenter clinical trial protocol. Front. Pharmacol.. 2022;13
- [CrossRef] [Google Scholar]
- Discovery of processing-associated Q-marker of carbonized traditional Chinese medicine: An integrated strategy of metabolomics, systems pharmacology and in vivo high-throughput screening model. Phytomedicine. 2022;102:154152
- [CrossRef] [Google Scholar]
- SwissTarget Prediction: a web server for target prediction of bioactive small molecules. Nucleic Acids Res.. 2014;42(W1):W32-W38.
- [CrossRef] [Google Scholar]
- Discovery and identification of quality markers of Chinese medicine based on pharmacokinetic analysis. Phytomedicine. 2018;44:182-186.
- [CrossRef] [Google Scholar]
- Assessment of the anti-inflammatory effects of three rhubarb anthraquinones in LPS-Stimulated RAW264. 7 macrophages using a pharmacodynamic model and evaluation of the structure-activity relationships. J. Ethnopharmacol.. 2021;273:114027
- [CrossRef] [Google Scholar]
- Gypsum fibrosum and its major component CaSO4 increase cutaneous aquaporin-3 expression levels. J. Ethnopharmacol.. 2012;139(2):409-413.
- [CrossRef] [Google Scholar]
- STITCH 4: integration of protein-chemical interactions with user data. Nucleic Acids Res.. 2014;42(D1):D401-D407.
- [CrossRef] [Google Scholar]
- Y.S. Kurniawan T. Indriani H. Amrulloh L.C. Adi A.C. Imawan K.T.A. Priyangga E. Yudha The Journey of Natural Products: From Isolation Stage to Drug’s Approval in Clinical Trials Bioactivities. https:// 2023 doi.org/10.47352/bioactivities.2963-654X.190.
- Jinzhen Oral Liquid alleviates lipopolysaccharide-induced acute lung injury through modulating TLR4/MyD88/NF-κB pathwa. Phytomedicine. 2023;114:154744
- [CrossRef] [Google Scholar]
- A novel strategy by integrating chemical profiling, molecular networking, chemical isolation, and activity evaluation to target isolation of potential anti-ACE2 candidates in Forsythiae Fructus. Phytomedicine. 2022;96:153888
- [CrossRef] [Google Scholar]
- Overall quality control of Jinzhen Oral Liquid based on HPLC-UVD-ELSD fingerprint and simultaneous determination of 13 main representative components. Chin. Tradit. Herb Drugs. 2020;51(22):5737-5747.
- [CrossRef] [Google Scholar]
- Quality control of Jinzhen oral liquid based on amino acids fingerprint and simultaneous determination of 29 amino adcids. Chin. Tradit. Herb Drugs. 2020;51(23):5972-5979.
- [CrossRef] [Google Scholar]
- A new concept on quality marker of Chinese materia medica: Quality control for Chinese medicinal products. Chin. Tradit. Herb Drugs. 2016;47(9):1443-1457.
- [CrossRef] [Google Scholar]
- A new concept on quality marker for quality assessment and process control of Chinese medicines. Chin. Herb Med.. 2017;9(1):3-13.
- [CrossRef] [Google Scholar]
- Liquiritin, a novel inhibitor of TRPV1 and TRPA1, protects against LPS-induced acute lung injury. Cell Calcium. 2020;88:102198
- [CrossRef] [Google Scholar]
- Network pharmacology and pharmacokinetics integrated strategy to investigate the pharmacological mechanism of Xianglian pill on ulcerative colitis. Phytomedicine. 2021;82:153458
- [CrossRef] [Google Scholar]
- Therapeutic effect of Jinzhen oral liquid for hand foot and mouth disease: a randomized, multi-center, double-blind, placebo-controlled trial. PLoS One. 2014;9(4):e94466.
- [Google Scholar]
- A prospective multicenter randomized controlled study on the efficacy and safety of Jin-Zhen oral solution. Chin. J. Pract. Pediatrics. 2010;25(5):383-387.
- [Google Scholar]
- Shengmai San-derived Compound Prescriptions: a review on Chemical constituents, Pharmacokinetic studies, Quality control, and Pharmacological properties. Phytomedicine. 2022;107:154433
- [CrossRef] [Google Scholar]
- Issues and recommendations for exploratory factor analysis and principal component analysis. Res. Soc. Admin. Pharm.. 2021;17(5):1004-1011.
- [CrossRef] [Google Scholar]
- Clinical effect of Jinzhen oral liquid on bronchitis in children. Guizhou Med. J.. 2018;42(1):48-50.
- [CrossRef] [Google Scholar]
- Jinzhen oral liquid in the treatment of 43 children with acute bronchitis. Chin. Tradit. Herb Drugs. 2016;29(5):12-14.
- [CrossRef] [Google Scholar]
- The STRING database in 2021: customizable protein-protein networks, and functional characterization of user-uploaded gene/measurement sets. Nucleic Acids Res.. 2021;49(18):10800.
- [CrossRef] [Google Scholar]
- The Pharmacopoea Commission of PRC, 2020. The Pharmacopoeia of the People’s Republic of China, part 1. China Medical Science and Technology Press, Beijing, p. 1164-1165.
- Identification of the effective constituents for anti-inflammatory activity of Ju-Zhi-Jiang-Tang, an ancient traditional Chinese medicine formula. J. Chromato. A. 2014;1348:105-124.
- [CrossRef] [Google Scholar]
- Current status of standardization of traditional Chinese medicine in China. Evid. Based Complement Altern. Med.. 2016;2016:9123103.
- [CrossRef] [Google Scholar]
- Traditional Chinese medicine network pharmacology: development in new era under guidance of network pharmacology evaluation method guidance. China J. Chin. Mater. Med.. 2022;47(1):7-17.
- [CrossRef] [Google Scholar]
- DrugBank 5.0: a major update to the DrugBank database for 2018. Nucleic Acids Res.. 2018;46(D1):D1074-D1082.
- [CrossRef] [Google Scholar]
- Discovery of quality-marker ingredients of Panax quinquefolius driven by high-throughput chinmedomics approach. Phytomedicine. 2020;74:152928
- [CrossRef] [Google Scholar]
- Yang, Z.H., Wang, B., Ma, Q., Wang, L., Lin, Y.X., Yan, H.F., Fan, Z.X., Chen, H.J., Ge, Z., Zhu, F., Wang, H.,J., Zhang, B.N., Sun, H.D., Feng, L.M., 2021. Corrigendum: potential Mechanisms of Action of Chinese Patent Medicines for COVID-19: a Review. Front. Pharmacol. 12, 770125. https://doi.org/10.3389/fphar.2021.668407.
- Approaches to establish Q-markers for the quality standards of traditional Chinese medicines. Acta Pharm. Sin. B. 2017;7(4):439-446.
- [CrossRef] [Google Scholar]
- Jinzhen oral liquid in the treatment of 50 cases of acute bronchitis. World Latest Med. Inform.. 2016;16(60):156-163.
- [CrossRef] [Google Scholar]
- Characterization the xenobiotics of Jinzhen Oral Liquid in rats based on UPLC-Q/TOF-MS. Chin. Tradit. Herb Drugs. 2022;53(10):2956-2967.
- [CrossRef] [Google Scholar]
- Pharmacokinetic-pharmacodynamic modeling to study the antipyretic effect of Qingkailing injection on pyrexia model rats. Molecules. 2016;21(3):317.
- [CrossRef] [Google Scholar]
- Anti-inflammatory effects of Scutellaria baicalensis water extract in LPS-induced THP-1 Macrophages through metabolomics study. Ara. J. Chem.. 2023;16(3):104507
- [CrossRef] [Google Scholar]
Appendix A
Supplementary material
Supplementary data to this article can be found online at https://doi.org/10.1016/j.arabjc.2023.105433.
Appendix A
Supplementary material
The following are the Supplementary data to this article:Supplementary data 1
Supplementary data 1







