Translate this page into:
A comprehensive review of in vitro precursor feeding strategies for the overproduction of high-value plant secondary metabolites
⁎Corresponding author at: Department of Biology, Institut Teknologi Sepuluh Nopember, Kampus ITS Sukolilo, 60111 Surabaya, Indonesia. nuruljadid@bio.its.ac.id (Nurul Jadid)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Plant tissue culture techniques have revolutionized various aspects of modern agriculture, horticulture, and pharmaceutical industries. This method contributes to enhancing the production of plant secondary metabolites, which have noteworthy applications in pharmaceuticals, agriculture, and industry. Presently, over 50,000 plant metabolites have been identified and categorized into three primary groups: terpenoids, flavonoids, and alkaloids. Numerous studies have elucidated the metabolic pathways involved in the biosynthesis of plant compounds to better understand the metabolic flux, with the aim of identifying and engineering the production of valuable constituents. In vitro precursor feeding is one phytochemical strategy that helps increase the accumulation of plant compounds. Therefore, this review focuses on investigating the application of in vitro precursor feedings to the overproduction of a high-value phytochemicals. Moreover, it evaluates the influence of type and concentration of precursors, plant species and culture conditions as well as the phytochemical products providing insights into optimization strategies. Additionally, the potential application of accumulated terpenoids, flavonoids, and alkaloid-derived compounds for agricultural, pharmaceutical, and industrial purposes are discussed. Finally, perspective challenges and limitations related to in vitro precursor feeding strategy are addressed, including production stability, cytotoxicity effect, and uptake efficiency. The overall data presented might serve as an up-to-date report on the application of in vitro precursor feedings for enhancing plant secondary metabolite production.
Keywords
Metabolic pathway
Phytochemical
In vitro precursor feeding
Pharmaceuticals
Plant secondary metabolites
1 Introduction
Plants are renewable sources, providing materials (biomass) and phytochemicals (both primary and secondary metabolites) (Guerriero et al., 2018). Moreover, plants serve as bio-factories, manufacturing human nutrition (Jadid et al., 2018). Diverse plant secondary metabolites (PSMs) have been elucidated including terpenoids, flavonoids, and alkaloid-derived compounds (Dasari et al., 2020). The PSMs are beneficial for medicinal purposes, food additives, flavorings, agriculture and other industrial ingredients. As an example, numerous ethnobotanical studies have indicated that tropical plants could be explored as alternative reservoirs for the production of medicinal drugs, owing to their varied plant secondary metabolites (PSMs) (Jadid et al., 2023). In addition, these organic substances also self-guard the plants from many adverse environmental conditions, including protecting the plants from pathogenic microorganisms, insects, and herbivore attacks. Moreover, in some cases, they protect the ecological equilibrium in nature by facilitating plant-animal pollinator contact (Slavković and Bendahmane, 2023).
Rigorous use of crop products creates a large gap between demand and availability. Moreover, PSM are available in small quantities, and harvesting at current demand scales may be environmentally unpleasant and impractical (Marchev et al., 2020; Wilson and Roberts, 2012). Furthermore, the economic impracticality of chemical synthesis for plant secondary metabolites is frequently attributed to their structural complexity (Dziggel et al., 2017; Staniek et al., 2014). Hence, finding alternatives to sourcing plant materials from natural reserves, as well as suitable systems and approaches are necessary. Plant cell cultures offer a great advantage to address these challenges, especially for propagating high commercial or medicinal crops (Fig. 1) (Jadid et al., 2024a). However, the widespread adoption of plant cell culture in commercial settings is constrained, and only a tiny fraction of secondary metabolites can be produced. A few key reasons include inadequate yields, instability in biosynthesis, and challenges in scaling up production (Sevón and Oksman-Caldentey, 2002). Notably, the prevalent issue is the inefficiency of the biosystem in producing high concentrations of the desired compounds (Qu et al., 2011). This predicament arises from the empirical approach in selecting cultures with high and consistent yields and limited information on the PSM biosynthetic pathways (Isah et al., 2018).
In vitro culture of medicinal and industrial crops. 1) Efficient shoot proliferation and callus induction in Gynura pseudochina in vitro culture (Anjalani et al. (2024), 2) callus and organogenesis of Stevia rebaudiana by Jadid et al., (2024a). 3) Callus culture and organogenesis in Pogostemon cablin by Jadid et al. (2024c) after being cultured in an optimized medium supplemented with plant growth regulators (PGR) and elicitor methyl jasmonate, resulting in enhanced growth, increased metabolic gene expression, and changes in phytochemical composition.
A good biosystem in plant cell culture for boosting the PSMs could be seen in how the system consistently ensures substantial amounts of the desired compounds. Rigorous selection of productive plant cell types and careful control of culture conditions can lead to higher levels of specific products within cultured cells. Numerous approaches have been employed to stimulate biosynthetic activities, focusing on optimizing the production of phytochemicals (Rao and Ravishankar, 2002). Typically, culture cell systems amass significant quantities of secondary compounds under specific circumstances. Over time, diverse strategies have emerged to augment PSMs production using plant cell culture, including selecting high-productive cell lines (Tripathi et al., 2019). Moreover, many studies revealed that enhancing secondary metabolites could be achieved by elicitation using both abiotic (environmental stress or chemical compounds) (Anjalani et al., 2024) and living organism-based elicitors (Tothong et al., 2023). Recently, the use of metal-based nanoparticles has also been applied to the plant in vitro medium to induce the synthesis of commercially important substances. Previous work has been demonstrated that plants develop a fascinating physiological and molecular strategies to cope with adverse environmental conditions, including the over-production of secondary metabolites (Jadid et al., 2017). However, the environmental inducers, in some cases, might also alter the genetic profile of the plants.
Another technique involves the addition of precursor feeding of the targeted PSM into the in vitro culture medium. Precursor feeding refers to supplementation of the primary building block that initiates the biosynthetic pathway leading to the production of specific plant metabolites. Generally, precursors are derived from primary metabolites including amino acids, fatty acids, sugars or other organic acids. This in vitro supplementation can result in higher yields of the desired end products (Hussain et al., 2012; Rao and Ravishankar, 2002). A critical aspect of this method involves a comprehensive exploration of the entire biosynthetic pathway. This inclusive approach considers multiple examples that might influence the production of the compound (Espinosa-Leal et al., 2018). Particularly, this method is advantageous when the precursors are cost-effective (Namdeo et al., 2007). Many studies, including those by Marchev et al., Dasari et al., Skrzypczak-Pietraszek et al., Guerriero et al., and Kundu et al., have effectively isolated specific compounds using the precursor feeding approach (Dasari et al., 2020; Guerriero et al., 2018; Kundu et al., 2018; Marchev et al., 2020; Skrzypczak-Pietraszek et al., 2018).
In this review, we emphasize the precursor feeding method as an advanced approach for augmenting the biosynthesis of PSMs through in vitro plant systems. Our review gathered some elements supporting the biosystem, including the plant species, plant organs used in the culture system, methods, target compounds, and precursors used in the experiments. This review also briefly describes the potential applications of PSMs in agriculture, pharmacy, and industries. Finally, we discussed precursor feeding methods' future prospects and limitations, including production stability, cytotoxicity effect, and uptake efficiency. This review offers an overview of the current application of precursor feeding techniques via in vitro culture for augmenting the production of PSMs. This review also serves as valuable information for future research direction on the synthesis of commercial plant metabolites in vitro using combinatorial strategies with biotic and abiotic elicitors.
2 In vitro precursor feeding
2.1 Precursor feeding method and common pathways in plant
The core idea behind the in vitro precursor feeding involves incorporating intermediate metabolites in the bioactive molecule synthesis at the beginning or during the plant cell cultivation process. This inclusion acts as extra substrates, effectively enhancing the yield of metabolites within plant tissues, cells, and organs undergoing cultivation (Isah et al., 2018; Rasche et al., 2016). The term “precursor” pertains to substances originating externally or internally that can be changed into secondary compounds by in vitro cell cultivation via biosynthesis pathways. Generally, the levels of these compounds are comparatively lower in plant cell cultures when compared to differentiated plant tissues. This discrepancy elucidates the correlation between cellular development and increased production of pharmaceutical-based natural products in various micropropagation methods involving plant tissues and organs (Constabel and Kurz, 1999). The use of transgenic cell lines and the incorporation of these compounds into the culture medium show promise as strategies to expedite the synthesis of molecules in plant cell cultures (Namdeo et al., 2007).
It is essential to ascertain the production rates and the kinetics of biosynthetic precursor utilization while attempting to increase pharmaceutical output by choosing a precursor molecule. Identifying the best conditions for these processes is also imperative (Isah et al., 2018). Critical considerations in choosing and introducing a precursor molecule into the system encompass the timing of its introduction, the concentration at which it is added, and its seamless integration into the intended biosynthetic pathway (Jackson & Attalla, 2010). Furthermore, it is crucial to consider the potential impact of feedback inhibition when determining the precise quantity to modify the culture medium for plant tissues, cells, and organs.
Shikimic acid, phenylpropanoid, and mevalonic acid pathways represent extensive biosynthesis routes frequently investigated in plant cell culture research to enhance the production of phytochemicals using the precursor feeding method. The shikimic acid pathway is particularly significant for synthesizing a range of essential aromatic compounds, including vitamins, amino acids, and other phytochemicals (Cheynier et al., 2013). It is pivotal in primary and secondary plant metabolisms (Macheroux et al., 1999). The initiation of this pathway involves the condensation of two compounds of phosphoenolpyruvate (PEP) and one molecule of erythrose-4-phosphate (E4P), both are derived from glycolysis and the pentose phosphate pathways. Following this, the pathway advances through a sequence of enzymatic reactions, yielding a crucial intermediate molecule known as shikimic acid. The ultimate step in the pathway culminates in the production of chorismate (Dewick, 2002). It acts as a branching point in the pathway and provides the precursor for various important biochemicals (see Fig. 2) (Mesnage et al., 2021). Numerous studies employing precursor feeding methods utilize intermediate compounds from this pathway as supplementary precursors. For instance, shikimic acid is added to enhance the production of 3-O-glucosyl-resveratrol and 4-(3,5-dihydroxyphenyl)-phenol from V. vinifera (Riedel et al., 2012), shikimate is introduced to increase the production of sanguinarine from P. somniferum (Verma et al., 2014a), and it is used to produce vincamine from V. minor (Verma et al., 2014b) (Fig. 2).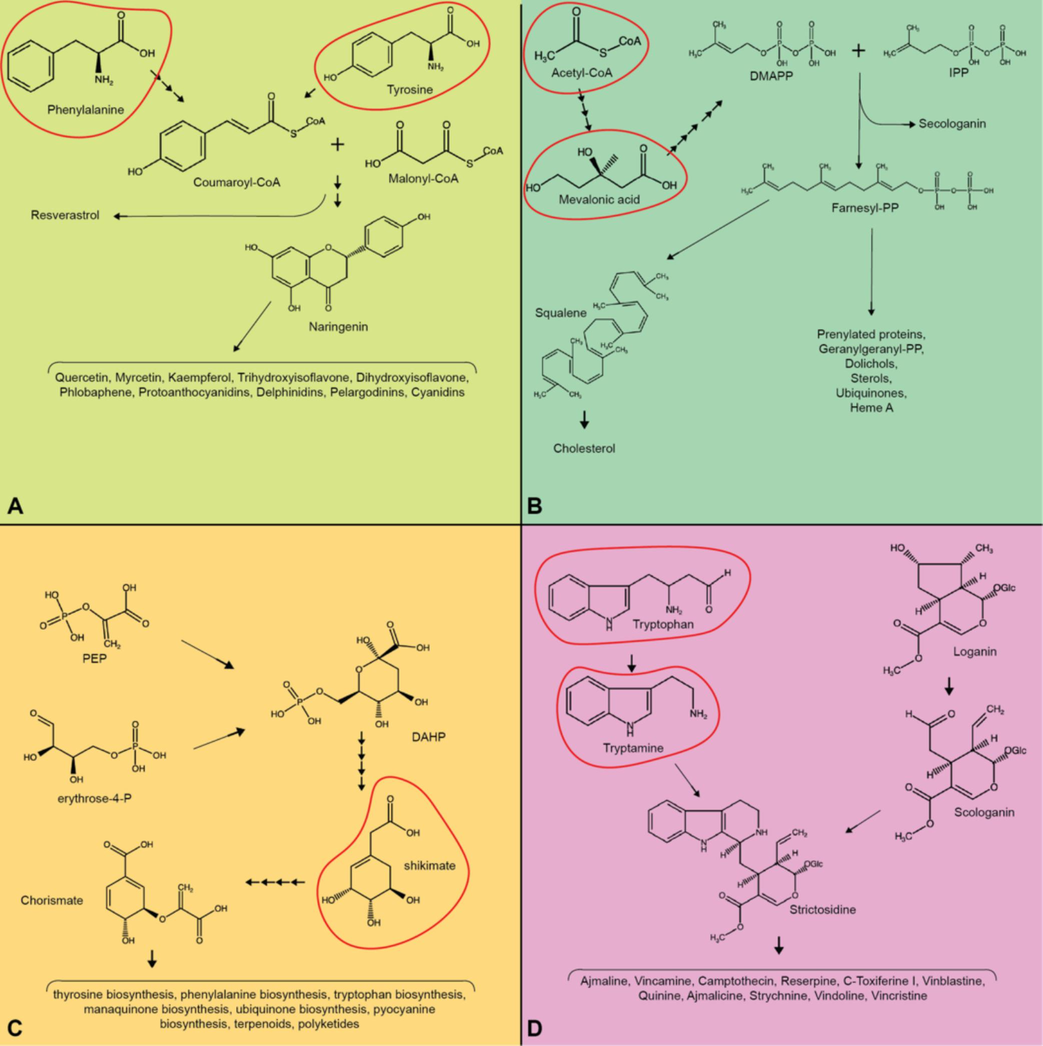
Common precursors (red circles) are used in precursor feeding method. (A) Phenylalanine and tyrosine are potential amino acids that could be used in flavonoid-derived compounds (B) Acetyl-CoA and mevalonic acid are organic acid precursors in the biosynthesis of isoprenoids (C) The shikimate serves as precursors in the shikimate-derived compounds. (D) Tryptophan and tryptamine serve as precursors in the biosynthesis of strictosidine-derived compounds.
The phenylpropanoid metabolic pathway produces diverse phytochemicals, relying on the products generated in the shikimate pathway (Vogt, 2010). Phenylalanine acts as the starting point for phenylpropanoid biosynthesis. The core of the phenylpropanoid pathway involves specific enzymatic steps, including (i) phenylalanine deamination resulting in trans-cinnamic acid, (ii) hydroxylation of trans-cinnamic acid to 4-coumarate, and (iii) conversion of 4-coumarate to 4-coumaroyl-CoA (Biała and Jasiński, 2018). Coumaroyl-CoA is a crucial intermediate in the phenylpropanoid pathway, serving as a precursor for various compounds, including stilbene, monolignol, isoflavonoid, and coumarins biosynthesis (Buchanan et al., 2015). In the context of the precursor feeding method, many studies employ phenylalanine as an additional substrate in the initial stages of this pathway. For example, it is used in the production of wedelolactone from S. calendulacea (Kundu et al., 2018), daidzein from P. corylifolia (Shinde et al., 2009), quercetin from I. tinctoria (Miceli et al., 2023), and silymarin from S. marianum (Firouzi et al., 2013) (Fig. 2).
In living organisms, the mevalonate pathway is predominantly present in eukaryotes and primarily occurs within the cytoplasm. Initially, two molecules of acetyl CoA are enzymatically transformed to create 3-hydroxy-3-methyl-glutaryl-CoA. This compound is then transformed into mevalonic acid (MVA), and subsequently, isopentenyl diphosphate (IPP) is produced. A portion of IPP undergoes isomerization resulting in the formation of dimethylallyl diphosphate (DMAPP) (Xiao and Zhong, 2016). Ultimately, the remaining IPP and DMAPP are utilized by polyisoprene pyrophosphate synthase to generate precursors for triterpenes, sesquiterpenes, and steroids. In the context of the precursor feeding method, numerous studies employ mevalonic acid, an intermediate compound from this pathway, to enhance the production of various phytochemicals. For example, mevalonic acid is used in the production of whitanolide A from W. somnifera (Sivanandhan et al., 2014), curcubitacin E from C. colocynthis (Dasari et al., 2020), artemisinin from A. annua (Baldi and Dixit, 2008), and bacoside A from B. monnieri (Hegazi et al., 2017) (Fig. 2).
2.2 Current reports on precursor feeding strategy in plant in vitro cultures
Many studies report the use of in vitro precursor feeding to promote plant secondary metabolite synthesis. The supplementation of precursors into the culture medium is commonly employed in callus and cell suspension culture. We have updated the information provided by Namdeo et al. (2007) in their previous review. We collected the data from 2007 to 2024, we found that out of 56 studies, 48 were conducted on medicinal plants (Table 1). Meanwhile, 4 of the studies focused on industrial plants such as Vitis vinifera, Isatis tinctoria, Papaver somniferum, and Argania spinosa (Riedel et al., 2012; Miceli et al., 2023; Verma et al., 2014a; Hegazi et al., 2020). The remaining 4 studies used ornamental plants as research subjects, including Solenostemon scutellarioides, Dionaea muscipula, Drosera capensis, Aronia melanocarpa (Michx.) Elliott, Aronia arbutifolia (L.) Pers., and Dendrobium fimbriatum (Dewanjee et al., 2014; Królicka et al., 2008; Szopa et al., 2020; Paul and Kumaria, 2020) (Suppl. 2).
No.
Compound Class
Plant Species
Explant
Culture Media
Precursors
Products
References
1
Steroidal lactones
Withania somnifera
root derived callus
30 ml of MS + 1 mg/l picloram, 0.5 mg/l KN, 200 mg/l L-glutamine and 5 % sucrose
cholesterol,
mevalonic acid,
squalenewithanolide A,
withanolide B, withaferin, withanone,
deoxy withanstramonolide, withanoside I, withanoside V(Sivanandhan et al., 2014)
2
Ecdysteroid
Achyranthes aspera
seed derived callus
MS + 1 mg/l 2, 4-dichlorophenoxyacetic acid and 1 mg/l α-naphthaleneacetic acid
cholesterol, 7-dehydrocholesterol,
20-hydroxyecdysone
(John et al., 2018)
3
Polyprenylated acylphloroglucinol
Hypericum perforatum
shoot
20 ml MS + 0.1 mg/l BA, 30 g/l sucrose, 0.1 g/l myo-inositol and 0.1 g/l ascorbic acid
l-isoleucine, l-threonine, l-valine
hyperforin, adhyperforin
(Karppinen et al., 2007)
4
Phytosteroid
Digitalis purpurea L.
nodal, internodal, and leaf
MS + 7.5 μM BA, and MS + 15 μM IAA
progesterone, cholesterol, and squalene
cardiotonic glycosides, digitoxin, and digoxin
(Patil et al., 2013)
5
Flavonoids and phenolic acids.
Vitex agnus castus L.
shoots
MS + BAP 1 mg/L; NAA 0.5 mg/L; GA3 0.25 mg/L and BAP 2 mg/L;NAA 0.5 mg/L.
L-phenylalanine
neochlorogenic acid, p-coumaric acid, rutin, caffeic acid, cinaroside,
(Skrzypczak-Pietraszek et al., 2018)
6
Terpenoid
Citrullus colocynthis
leaves, shoot apex and nodal callus
MS + 2.0 mg/L BAP + 0.5 mg/L NAA
squalene, mevalonic acid
cucurbitacin E
(Dasari et al., 2020)
7
Terpenoid
Artemisia annua
seed derived callus
50 ml MS + 3 % (w/v) sucrose + 0.1 mg/l NAA + 0.1 mg/l Kn.
mevalonic acid lactone
Artemisinin
(Baldi and Dixit, 2008)
8
Coumestan
Sphagneticola calendulacea
shoot derived HR
½ MS + 100 μM SA
phenylalanine
wedelolactone
(Kundu et al., 2018)
9
Lignan
Larrea divaricata
leaves derived callus
MS + 9 μM 2,4-D + 5 μM BA
l-phenylalanine, cinnamic acid, ferulic acid, and sinapic acid
nordihydroguaiaretic acid
(Palacio et al., 2011)
10
Terpenoid
Bacopa monnieri
aerial parts
MS (liquid)
l-alanine and l-phenylalanine
saponin glycosides
(Watcharatanon et al., 2019)
11
Ester
Rhodiola rosea
seeds
MS + 25 g/L sucrose + 6.5 g/L agar
cinnamyl alcohol, cinnamaldehyde
Cinnamyl alcohol glycosides
(Javid et al., 2021)
12
Stilbenes
Morus alba L.
root
MS + 1 mg/L NAA
L-tyrosine
mulberroside A, oxyresveratrol, and resveratrol
(Inyai et al., 2021)
13
Phenol
Vitis vinifera
callus
25 ml of B5VIT basal medium
shikimic acid, phenylalanine
3-O-glucosyl-resveratrol and 4-(3,5-dihydroxyphenyl)-phenol
(Riedel et al., 2012)
14
Isoflavones
Psoralea corylifolia
stem derived hairy root
50 mL MS
phenylalanine
daidzein, genistein
(Shinde et al., 2009)
15
Flavonoids
Isatis tinctoria
shoot
MS + 1.0/1.0 mg/L BAP/NAA
L-Phenylalanine and L-Tyrosine
apigetrine, quercetin, apigenin, quercitrin
(Miceli et al., 2023)
16
Amino acid
Mucuna pruriens
seed derived callus
50 mL MS + 3 % sucrose + 0.5 mg/L picloram
tyrosine, phenylalanine
L-DOPA
(Rakesh and Praveen, 2022)
17
Alkaloids
Solanum lyratum
seed derived callus
30 ml MS + 0.75 mg/l 2,4-D + 3 % sucrose
cholesterol, stigmasterol
α-solanine, solanidine, and solasodine
(Lee et al., 2007)
18
Naphthoquinone
Drosera burmannii Vahl and Drosera indica L.
whole plantlets
30 mL MS
sodium acetate
plumbagin
(Boonsnongcheep et al., 2019)
19
Alkaloids
Rauwolfia serpentina
nodal segments and shoot
Hoagland solution
tryptamine
reserpine
(Panwar and Guru, 2015)
20
Caffeic acid ester
Solenostemon scutellarioides
whole plantlets
MS
L −phenylalanine, L −tyrosine
rosmarinic
(Dewanjee et al., 2014)
21
Flavonoids
Silybum marianum
whole plantlets
50 mL MS + 3 g./L picloram + 0.4 g/L kinetin
phenylalanine
silymarin
(Firouzi et al., 2013)
22
Terpenoid
Centella asiatica
leaf and petiole derived HR
100 mL MS + 50 g/L sucrose
squalene and pyruvic acid
madecassoside, asiaticoside, madecassic acid, asiatic acid
(Baek et al., 2020)
23
Alkaloids
Rauwolfia tetraphylla L.
leaf, stem, root derived callus
MS + 2, 4-D (2.0 mg/L)
Tryptophan
reserpine
(Rohela et al., 2021)
24
Alkaloids
Papaver bracteatum
cell suspension
50 mL MS liquid
L-tyrosine
thebaine
(Zare et al., 2014)
25
Phenolic
Decalepis hamiltonii
leaf derived callus
MS + BA + Kn + NAA + 2.4 D
ferulic acid
vanillin, 2H4MB, vanillic acid
(Matam et al., 2017)
26
Anthocyanins
Panax sikkimensis
root derived callus
MS + 3 % sucrose, 0.01 % myoinositol, 0.33 μM thiamine HCL, 2.5 μM pyridoxine hydrochloride, 4.0 μM nicotinic acid, 5.4 μM NAA, and 1.2 μM Kinetin.
phenylalanine
anthocyanins content
(Biswas et al., 2020)
27
Furanocoumarins
Psoralea corylifolia L.
cotyledon derived callus
MS + 10 µM BA + 5 µM IBA
umbelliferone, cinnamic acid, NADPH
psoralen
(Mohammadparast et al., 2015)
28
Alkaloid
Mitragyna speciosa
petiole and leaves derived callus
25 mL of liquid WPM
Tryptophan, loganin
mitragynine
(Mohamad Zuldin et al., 2013)
29
Anthraquinones
Cassia angustifolia Vahl
leaves, nodes, roots
MS + NAA + IBA + 3 % (w/v) sucrose
α-keto glutaric acid and pyruvic acid
sennoside A and B
(Chetri et al., 2016)
30
Terpenoid
Bacopa monnieri
leave derived callus
MS + 9 µM 2,4-D and 2.3 µM KIN
mevalonic acid
Bacoside A
(G. Hegazi et al., 2017)
31
Phenylpropanoid glycoside
Rhodiola imbricata
leaf and root cells suspension
MS + 0.5 mg/L TDZ + 1 mg/L NAA
tyrosol
salidroside
(Rattan et al., 2022)
32
Phenylpropanoids
Spilanthes acmella Murr.
leaf derived callus
MS + 15 µM BA + 5 µM 2,4-D
casein hydrolysate, L-phenylalanine
scopoletin
(Abyari et al., 2016)
33
Amino acid
Hybanthus enneaspermus (L.)
leaf derived HR
MS + 300 mg/L of cefotaxime
L-tyrosine
L-DOPA
(Sathish et al., 2023)
34
Phenolic
Dionaea muscipula,Drosera capensis
whole planlets
modified ½ MS + 2 % sucrose
l-phenylalanine, trans-cinnamic acid
naphthoquinones, quercetin, myricetin
(Królicka et al., 2008)
35
Alkaloid
Papaver somniferum
shoot derived callus
MS (aq) + 1 ppm 2,4-D
shikimate
sanguinarine
(Verma et al., 2014a)
36
Vitamins
Argania spinosa
leaf derived callus
100 ml MS + 4.5 µM 2,4-D + 5 µM NAA
tyrosine
α-tocopherol
(Hegazi et al., 2020)
37
Flavonoids
Silybum marianum
root derived callus
50 ml MS + 4.55 μM 2,4D + 4.44 μM BA
L-phenylalanine
Silymarin
(Hassanen et al., 2021)
38
Stilbenoid
Morus alba L.
leaf derived callus
30 ml MS + 0.1 mg/lTDZ + 1 mg/l NAA
L-phenylalanine, L-tyrosine
mulberroside A
(Pongkitwitoon et al., 2020)
39
Terpenoid
Picrorhiza kurroa
shoots
MS + 3 mg/L indole-3-butyric acid + 1 mg/L kinetin
cinnamic acid (CA) and catalpol (CAT)
picroside-I (P-I)
(Kumar et al., 2016)
40
Flavonoid
Cassia occidentalis L.
cotyledon derived callus
MS + 2.4D + Kin + NAA
Phenylalanine, Methionine
Rotenoids
(Vats and Kamal, 2014)
41
Phenolic compounds
Aronia melanocarpa (Michx.) Elliott, Aronia arbutifolia (L.) Pers.
shoot
90 mL MS
phenylalanine, cinnamic acid, benzoic acid, caffeic acid
neochlorogenic, chlorogenic, cryptochlorogenic, isochlorogenic, rosmarinic acids, and syringic
(Szopa et al., 2020)
42
Terpenoid
Bacopa floribunda
shoots and roots
MS + 2.0 mg/l BAP + 2.0 mg/l KIN and MS + 0.5 mg/l IAA + 0.5 mg/l IBA + 1.0 mg/l NAA
squalene
bacoside A3, bacopaside X, bacopaside II, and bacosaponin C
(Otari et al., 2023)
43
Phenolic compounds
Arnebia euchroma
bud derived callus
MSA + 1 mg/L kinetin and 0.3 mg/L IAA
L-phenylalanine
naphthoquinones
(Sykłowska-Baranek et al., 2012)
44
Alkaloid
Catharanthus roseus
leaf derived callus
MS + 1 mg/l kin
L-tryptophane; L-glutamine; L-asparagine; L-cystine and L-arginine
vinblastine and vincristine
(Taha et al., 2009)
45
Amino acid
Mucuna pruriens
leaf derived callus
20 ml MS + BAP 0.88 μM + NAA 11.41 μM
L-tyrosine
L-Dopa
(Raghavendra et al., 2011)
46
Phenolic, alkaloid, flavonoid, and tannins
Dendrobium fimbriatum
protocorm-like bodies
MS + 3 % sucrose + 0.7 % agar + BAP + Picloram
caffeic acid, ferulic acid and p-coumaric acid
phenolic, alkaloid, flavonoid compounds
(Paul and Kumaria, 2020)
47
Alkaloids
Rauwolfia
tetraphylla L.
leaf, stem, and root derived callus
MS + 2, 4-D2.0 mg/L
tryptophan
reserpine
(Rohela et al., 2021)
48
Phenylpropanoid
Rhodiola rosea L.
leaf derived callus
MS + 4.5 g/l agar + 30 g/l sucrose + 1 mg/l NAA + 0.5 mg/l BAP
tyramine, 4-hydroxyphenylpyruvate and tyrosol
salidroside
(Mirmazloum et al., 2019)
49
Flavonoid
Moringa oleifera
leaf and stem derived callus
MS + 1.0 mg/L BAP + 1.5 mg/L IBA
Phenylalanine
Niazirin, Benzylcarbamate, Vincosamide
(Mahood et al., 2018)
50
Coumarins,glucosinolates, phenols, flavonoids
Nasturtium officinale
microshoots
MS (solid) + 3 % (w/v) sucrose + 1 mg/L BA + 1 mg/L NAA
L-phenylalanine, L-tryptophan
coumaric, ferulic acids, rutoside, and glucosinolates
(Klimek-Szczykutowicz et al., 2021)
51
Organosulfur
Allium sativum L.
crown derived callus
MS (liquid) + 0.3 mg/L 2.4 D and 0.5 mg/L kin
glutathione
allicin, ajoene, alliin, dithiin groups, and allyl sulfide groups
(Setiowati et al., 2022)
52
Indole terpenoid alkaloids (TIAs)
Catharanthus roseus L.
leaves derived callus
½ B5 medium (Gamborg's B5) + sucrose 2 % (w/v)
L-phenylalanine, L-tyrosine
vincristine, vinblastine
(Vu et al., 2022)
53
Alkaloid
Mitragyna speciosa
petioles and leaves derived callus
WPM + 4 mg/L 2,4-D
tryptophan and loganin
mitragynine
(Mohamad Zuldin et al., 2013)
54
Amino acids
Mucuna prurita
leaf derived callus
20 ml MS (liquid) + IAA (11.41 μM) and BAP (0.88 μM)
L-tyrosine
L-Dopa
(Raghavendra et al., 2018)
55
Indole alkaloids
Vinca minor
leaf derived HR
¼ Gamborg's B5 (liquid)
shikimate, tryptophan, tryptamine, loganin, and secologanin
vincamine
(Verma et al., 2014b)
56
Alkaloid
Corylus avellana L.
cotyledons derived callus
MS (liquid) + 3 % sucrose/lactose/fructose
phenylalanine and vanadyl sulfate
Paclitaxel
(Rahpeyma et al., 2015)
The type of compounds produced in these 56 studies were diverse, including phenolic compounds (14), followed by alkaloids (13), flavonoids (9), terpenoids (8), amino acids (4), phenylpropanoids (3), steroids (3), esters (2), stilbenes (2), steroidal lactones (1), polyprenylated acylphloroglucinol (1), coumestan (1), lignan (1), isoflavones (1), naphthoquinone (1), furanocoumarins (1), anthraquinones (1), and vitamins (1). As for the type of media used, 47 studies employed suspension culture with liquid medium, and 9 studies used solid agar medium in the precursor feeding process, as seen in studies by Javid et al. (2021), Panwar and Guru (2015), Dewanjee et al. (2014), Mohammadparast et al. (2015), Chetri et al. (2016), Królicka et al. (2008), Otari et al. (2023), Mirmazloum et al. (2019), and Mahood et al. (2018).
The types of precursor compounds used were also varied, with seven different categories: amino acids (9), sterols (6), phenolic compounds (9), shikimic pathway compounds (5), intermediates in metabolic pathways (4), other nitrogenous compounds (4), and other organic compounds (2) (Suppl. 1). Meanwhile, some types of explants have been employed. Callus induced from different parts of the plant was used in 32 studies, while 5 studies used the hairy root method, 18 studies used direct organ culture, and 1 study used protocorm-like bodies (Table 1). For the culture medium, 50 studies used Murashige and Skoog (MS) supplemented also with PGR, while 2 studies used WPM medium, 2 used Gamborg’s B5 medium, and 1 used Hoagland medium (Table 1). The general step by step technique used in in vitro precursor feeding is briefly described in Fig. 3.
General step by step technique used in in vitro precursor feeding and elicitor addition for enhancing plant secondary metabolites. 1–2) Plant parts commonly used as explants in culture include leaves, apical and axillary meristem shoots, cotyledons, hypocotyls, and meristematic roots. 3–4) Culture can be performed through indirect organogenesis by inducing callus growth, or directly by growing specific organs such as hairy roots, adventitious roots, and protoplasts. 5) The culture type can be done using suspension methods or growth on solid agar medium. 6–7) Precursor feeding compounds and elicitor molecules can be added to the culture medium. 8) Changes in phytochemical composition can be analyzed using spectrometry techniques and desired compounds can be isolated to obtain pure compounds.
Precursor feeding enhances the production of secondary metabolites in in vitro cultures by supplying essential building blocks directly into the plant's biosynthetic pathways (Cheng et al., 2024). This increased availability of precursors boosts the metabolic flux toward desired compounds, upregulates key biosynthetic enzymes, and alleviates bottlenecks in the pathways. As a result, there is a significant enhancement in the yield of targeted secondary metabolites (Lu et al., 2022 ). Furthermore, precursor feeding treatments can be combined with elicitor compounds. Both can have a positive effect on cell growth and the production of secondary metabolite compounds (Rakesh and Praveen, 2022).
Different precursors have distinct mechanisms for entering the plant cells. Many of them are facilitated by transporters. The AAT (Amino Acid Transporter) plays a role in introducing amino acid precursor compounds (Dong et al., 2024). The ABC (ATP-binding cassette) transporter is involved in introducing lipid-derived compounds and organic compounds that are intermediates in secondary metabolite biosynthesis pathways (Kang et al., 2011). Other precursor molecules can enter the cell through passive diffusion. In addition to serving as building blocks in phytochemical biosynthesis, precursors may also influence the expression of growth genes and hormones (Inyai et al., 2021), resulting in increased biomass growth, which correlates with a higher production of secondary metabolite compounds. For elicitors, their interaction with cell receptors triggers a defence response by activating genes encoding secondary metabolite compounds (Namdeo, 2007; Rakesh and Praveen, 2022). The combined use of precursor feeding, and elicitors can yield optimal results in the production of secondary metabolite compounds (Fig. 4).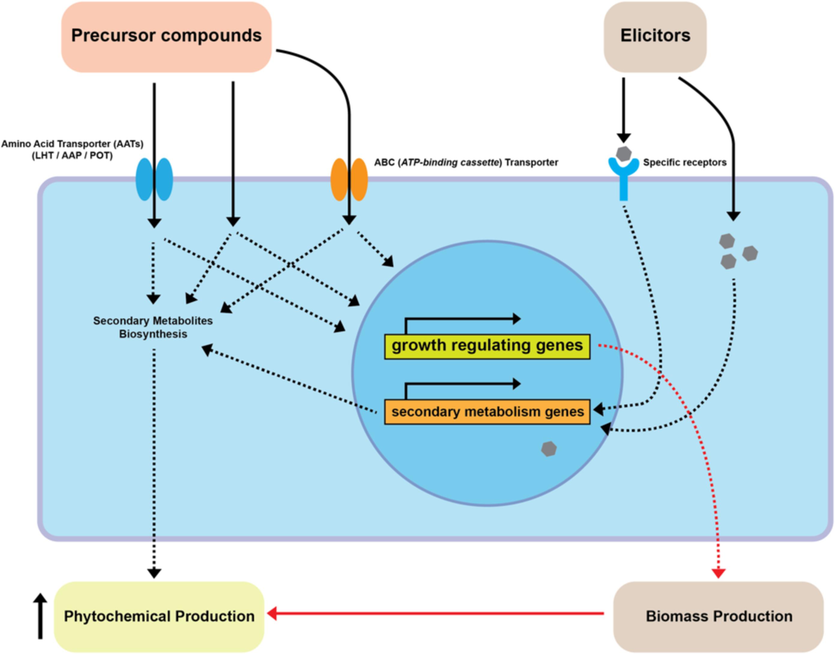
Predicted mechanism of intake and the role of precursor feeding and elicitor compounds in plant cells. Various types of precursor additive compounds can enter the cell through different pathways, including Amino Acid Transporters (AATs) such as LHT (Lysine-Histidine Transporters), AAP (Amino Acid Permease), and POT (Proton-dependent Oligopeptide Transporters). Additionally, precursors can enter the cell via ABC Transporters, facilitated diffusion, and passive diffusion. Meanwhile, elicitors interact directly with receptors or target molecules. Both applications influence the regulation of growth genes and the expression of secondary metabolite genes. The synergy between these two applications can enhance the production of desired phytochemicals.
3 Potential applications of plant secondary metabolites
Plant phytochemicals are typically classified into three main groups based on their biosynthetic pathways: terpenes, phenolics, and alkaloids (Bourgaud et al., 2001). These three clusters of phytochemicals have a very diverse range of applications, especially in the fields of pharmaceuticals, agriculture, the food industry, and others. Many reports explain the biological effects of phytochemical groups. This section provides a summary of the applications of plant phytochemicals.
Phenolic compounds, including flavonoids, are often recommended for inclusion in dietary supplements and nutraceuticals due to their perceived significance in the human diet. Apart from their role as antioxidants, these compounds possess a diverse range of biological capabilities (Pengfei et al., 2009). For instance, several research has shown that phenolic compounds have ability as anticancer (Tavsan and Kayali, 2019; Rosa et al., 2018), microcirculation-improving (Mastantuono et al., 2018), antihypertensive (Hou et al., 2012), hypo-lipidemic agents (Bae et al., 2014), and anti-inflammatory agents (Jang et al., 2020). They have also demonstrated potential as natural colorants (Rose et al., 2018) and active ingredients in the cosmetic industry (Chiocchio et al., 2021; Lianza et al., 2020; Shin et al., 2013).
Phenolic acids, which naturally occur in fruits and vegetables, encompass a diverse array of bioactivities, including antihyperglycemic (Sanchez et al., 2017), neuroprotective (Zaitone et al., 2019), antihypertensive (Agunloye et al., 2019), antidepressant (Barauna et al., 2018), anti-inflammatory (Zaitone et al., 2019), anticancer, and antidiarrheal (Frauches et al., 2016). In the veterinary area, tannins are employed as anthelmintic and antibacterial agents (del Carmen Acevedo-Ramirez et al., 2019; Redondo et al., 2014). Additionally, their tanning properties make phenolic acids useful in the hide industry (Mutiar et al., 2019). Nonetheless, caution is warranted when using tannins, as alongside their health-promoting attributes, certain detrimental effects have been observed (Labieniec and Gabryelak, 2003). Even though tannin-rich substances are frequently used in bovine feed, little is known about how hydrolyzable tannins affect the ruminant intestinal bacteria. It is well-recognized that several metabolites generated from hydrolyzable tannins, such as pyrogallol, have negative effects on the host animal's intestinal microorganism (Lotfi, 2020).
Terpenes are employed as medicines, food supplements, flavors, scents, biopesticides, and other things thanks to their broad spectrum of bioactivities in the cosmetic, food, cosmetic, pharmaceutical, agriculture, and perfumery (Chiocchio et al., 2021). Within the pharmaceutical industry, terpenes play a crucial role as therapeutic factors with a wide array of bioactivities. These include chemo-preventive, antifungal, anti-inflammatory, antimicrobial, antihyperglycemic, antiviral, analgesic, and antiparasitic activities (Anthoni et al., 2006; Hammer et al., 2003; Jesch and Carr, 2017). Terpenes are also utilized as excipients in the pharmaceutical industry to optimize the intake of active ingredients across the skin (Fox et al., 2011).
This family of chemicals is crucial for food, aromatherapy, and fragrances due to the unique scent of many monoterpenes, which are mostly found in aromatic plant essential oils. Monoterpenes derived from essential oils, including carvacrol, thymene, and thymol (commonly found in plants of the Lamiaceae family), possess not only aromatic properties but also a diverse array of biological activities. For example, they have the potential to treat diseases of the nervous, cardiovascular, and respiratory systems as well as act as antimicrobial and antioxidant agents (Salehi et al., 2018).
Furthermore, diterpenes of the labdane type are valuable in the industry of perfumes and serve as fixative agents in premium fragrances. Essentially, a fixative agent is a low-volatility substance that prolongs the longevity of a fragrance in perfumes, provides a lasting aroma, and enables harmonious blending with other components. Resin gathered from C. ladanifer (a member of the Cistaceae family) is a significant origin of labdanum-type diterpenes utilized as a fixing agent in perfumery (Raimundo et al., 2018).
Saponins, belonging to the subclass of terpenoids, hold immense significance in the food industry. Processed foods like desserts, ice creams, baked goods, sauces, and beverages often incorporate dispersions such as emulsions and foams. These dispersions play a vital role in defining, stabilizing, and managing the consistency and flow-related characteristics of these products. Saponins, because of their amphiphilic characteristics, have demonstrated the capability to maintain the stabilization of emulsions in food. Importantly, they exhibit lower sensitivity to factors such as ionic concentration, pH, and elevated temperatures (approximately 90 °C) in comparison to the emulsifiers presently utilized (McClements & Gumus, 2016).
Additionally, saponins demonstrated several biological actions crucial for human healthcare at modest doses (del Hierro et al., 2018; Rehan et al., 2020; Singh et al., 2017). The cholesterol-lowering, anticancer, and antiviral characteristics are the most pertinent among them (Jesch and Carr, 2017; Marrelli et al., 2016; Vinarova et al., 2015; Zhao et al., 2008). Boswellic and betulinic acids are two examples of the medicinal potential of triterpenic saponins. Betulinic acid exhibits a broad spectrum of biological functions, notably displaying potent antiviral effects (Alakurtti et al., 2006; Singh and Sharma, 2015). Another compound derived from the resin of incense trees (Banksia serrata Roxb.), boswellic acid, is utilized as an anti-inflammatory drug (Anthoni et al., 2006). Furthermore, clinical tests utilizing the gum resin from B. serrata have shown a decrease in signs among patients coping with rheumatoid arthritis and osteoarthritis (Poeckel et al., 2005, 2006).
Saponins are antibacterial substances that are effective against fungi and bacteria that attack plants (Hoagland et al., 1996; Moses et al., 2014). The probable mechanism underlying these effects is the capability of saponins to interact with sterols found in bacterial membranes, resulting in membrane disruption (Augustin et al., 2011; Sreij et al., 2019). Along with their allelopathic effects on certain plant species, saponins also have insecticidal, and molluscicidal effects (Nielsen et al., 2010; Huang et al., 2003). Saponins possess potential as natural biopesticides for agricultural applications because of their inherent characteristics and biological role in plant defense. For instance, Trdá et al. (2019) reported that a specific saponin called aescin not only exhibits antifungal properties against crop diseases but also can stimulate the immunity of B. napus and A. thaliana against fungus and bacteria, due to aescin contribution in salicylic acid-dependent resistance.
Alkaloids boast a longstanding history of utilization in medicinal applications and continue to play a crucial role in modern medicine (Newman and Cragg, 2020). An example of their significance is morphine derived from Papaver somniferum, which stands as among the most widely utilized pain relievers today. In their overview (Debnath et al., 2018), Debnath et al. listed the biological features of the major subclasses of alkaloids. Examples include the historically significant antimalarial drugs quinidine and quinine, which are produced from the bark of C. officinalis L. Another notable alkaloid is an adrenergic amine sourced from plants within the Ephedra genus (Ephedraceae family), ephedrine. Ephedrine finds application in various drug formulations, serving as respiratory dilators for individuals with asthma and allergy issues, as well to prevent low blood pressure during intrathecal anesthesia (Ma et al., 2007). Additionally, the Apocynaceae plant C. roseus (L.) is a significant source of anticancer drugs, specifically vinblastine and vincristine (Ronghe et al., 2001).
Other alkaloids have garnered attention for their potential bioactivity. For instance, a tropane alkaloid named catuabine present in the bark of T. catigua A. Juss., demonstrated effects similar to an antidepressant in a rodent depression model (Campos et al., 2005). Another compound, berberine, found in the stem bark and roots of various Berberidaceae, has displayed anti-diabetic effects in rodent models of insulin resistance induction (Turner et al., 2008). Moreover, berberine showcases a range of bioactivities including antidepressant, anti-inflammatory, antioxidant, hepatoprotective, antihypertensive, and anti-cancer features (Amritpal et al., 2010).
Additionally, certain alkaloids are vital in spices and well-known drinks because they have psychoactive characteristics, such as the caffeine found in coffee, which plays a part in social and ceremonial events (Crozier et al., 2006). Coffea arabica L. and Camelia sinensis (L.) are the two most significant biological sources. Caffeine is classified as a methylxanthine alkaloid. It finds applications in various domains, including cosmetics, analgesics, anti-cold medications, and slimming products.
Numerous alkaloids also exhibit insecticidal and fungicidal properties, aligning with their protective function (Liu et al., 2012; Yang et al., 2002). For instance, the phytopathogen Puccinia recondita is highly vulnerable to the potent fungicidal activity of the piperidine alkaloid pipernonaline. This alkaloid is purified from the hexane fraction of P. longum (Yogendra et al., 2017). P. grisea, E. graminis, B. cinerea, P. recondita, R. solani, and P. infestans, are among the phytopathogens that the Coptis japonica Makino extracts (berberine chloride, coptisine chloride, palmatine iodide, and isoquinoline alkaloids,) and its alkaloid content exhibit fungicidal activity against in an in vivo plant model (Ju-Hyun, 2005).
4 Prospects and limitations of in vitro precursor feeding methods for boosting the production of plant secondary metabolites
We have successfully compiled and analyzed the precursor feeding studies to elevate the biosynthesis of various types of PSMs via in vitro. These data unequivocally demonstrate the positive impact of incorporating diverse feeding precursors on the synthesis of secondary metabolite compounds. The strategic utilization of amino acids (organic chemicals) to the culture media, has been validated to be an effective method for augmenting the in vitro production of several therapeutic plant byproduct compounds. Notably, in Nasturtium officinale microshoot cultures, amino acid supplementation, including L-Phenylalanine and L-Tryptophan, resulted in heightened production of coumaric acid, ferulic acid, rutoside, and glucosinolates (Klimek-Szczykutowicz et al., 2021). Furthermore, the leaf callus of Hypericum perforatum, when cultivated in a growth medium enriched with 200 mM/L L-tyrosine, demonstrated a substantial enhancement in the production of L-Dopa, increasing by 36.36 folds (Raghavendra et al., 2018).
At times, the introduction of a precursor of biosynthetic to the growth media can be used to stimulate the formation of metabolites without impeding biomass accumulation. However, there are instances where the use of biosynthetic precursors alone might not lead to high yield of bioactive compounds in in vitro cultures. In such cases, employing a combination of biosynthetic precursors with enhancement techniques may offer a workaround. This approach is particularly useful when there is a scarcity of the precursor or constraints in its absorption by the cells from the culture medium, and integration into metabolic pathways (Isah et al., 2018).
To achieve the necessary productivity, it is essential to employ various augmentation strategies. One of the most frequently utilized methods is synergistic precursor and elicitor application. This strategy has efficiently proven in boosting the accumulation of PSMs. For instance, in callus cultures originating from leaves of Cassia augustifolia, combining elicitation with precursor addition yielded in a notable increase in the production of sennoside A and B (Chetri et al., 2016). Similarly, the incorporation of both elicitation and precursor feeding into cell suspension cultivation of Mucuna pruriens resulted in a noteworthy increase in the production of L-Dopa (Raghavendra et al., 2011). Notably, in this context, precursor feeding demonstrated greater success in boosting production compared to elicitation.
Given that many of the biochemical processes involved are cross-linked in cells, in certain cases a precursor might demonstrate an impact on the creation of more than one pathway of certain biochemical. For instances, Glutathione addition in the culture media of Allium sativum L. promoted allyl sulfide group (allyl methyl disulfide; allyl propyl disulfide; 1-propenyl allyl disulfide; 2-propenyls-1-propenyl disulfide; allyl trisulfide; diallyl heptasulfide; allyl methyl trisulfide;) and allicin, alliin, dithiin, ajoene, groups (3-vinyl 1,2-dithiin; 2-vinyl 1,3-dithiin) (Setiowati et al., 2022). The synthesis of asiatic acid, madecassoside, asiaticoside, and madecassic acid was enhanced by adding squalene to the Centella asiatica calli culture media (Baek et al., 2020). In cell cultures of Solanum lyratum, the production of insolasodine, solasonidine, and solanine was significantly boosted when exogenous sterols like cholesterol, stigmasterol, or mixed sterols were introduced (Lee et al., 2007). However, there was no influence on the growth of biomass. Precursor feeding strategy in plant in-vitro culture hold promising prospects and provide a variety of possible uses and advantages. These techniques have a lot of promise for several companies and fields of study. The following are some significant prospects of the precursor feeding application:
4.1 Enhanced phytochemical production
Combined in vitro plant propagation and precursor feeding enables the targeted and regulated augmentation of secondary metabolite synthesis. This is especially useful for getting larger yields of certain bioactive compounds, such as phenolics, flavonoids, and alkaloids that exhibit drug-related applications, nutraceutical, and industrial industries. Numerous studies previously indicated that adding precursors to the growth medium yields in a rise in the formation of secondary metabolite chemicals. The production of mulberroside A compounds with addition of L-Phenylalanine and L-Tyrosine, the increase in L-Dopa compounds with L-Tyrosine precursors, and the production of Silymarin compounds with L-Phenylalanine precursors are a few examples (Hassanen et al., 2021; Pongkitwitoon et al., 2020; Raghavendra et al., 2018).
4.2 Applications in pharmacology
Many secondary metabolites made by plants because of feeding on precursors have pharmacological characteristics. These metabolites can be further isolated and used as a source of chemicals for the synthesis of new medications or for the creation of innovative pharmaceuticals. A regulated method to maximize the generation of these beneficial chemicals is precursor feeding. Most of the secondary metabolite compounds are intended for application as medicines. Examples of applications of precursor feeding to increase compound production are vincristine, vinblastine, Paclitaxel, reserpine, silymarin, and salidroside, which have implications for cancer therapy drugs (vincristine and vinblastine), chemotherapy agents, hypertension, hepatoprotective, nerve protection (Mirmazloum et al., 2019; Rahpeyma et al., 2015; Rohela et al., 2021; Vu et al., 2022).
4.3 Nutraceutical and functional food development
By feeding precursors, secondary metabolites can be used to optimize the nutritional and functional qualities of food items. Bioactive ingredients can be added to functional meals or dietary supplements to promote health and wellness while addressing a variety of medical issues. An example of a compound that is useful in the food and beverage industry is 2-hydroxy-4-methoxybenzaldehyde (2H4MB) with the addition of ferulic acid as a precursor (Matam et al., 2017).
4.4 Crop improvement and stress tolerance
Utilizing precursor feeding for enhancing the synthesis of plant phytochemicals involved in stress response can aid plants development, increasing their tolerance to environmental stressors and pests. This has potential implications for improving crop resilience and productivity, especially to faceclimate change and changing environmental conditions. Rotenoids and solanine are examples of secondary metabolite compounds that can be utilized as fungicides and pesticides. These compounds can be produced by adding feeding precursors such as phenylalanine and methionine and exogenous sterols (cholesterol, stigmasterol) respectively (Lee et al., 2007; Vu et al., 2022).
4.5 Flavour and fragrance industries
Specific phytochemicals are needed for the industries of flavour and fragrance. The creation of certain compounds contributing to the flavour and scent of various goods, such as perfumes, essential oils, drinks, and food products, can be optimized using precursor feeding. An example of the application of precursor feeding is the vanillin compounds, 2H4MB, and Vanillic acid production, coumarins with ferulic acid, L-phenylalanine, and L-tyrosine as the precursors (Klimek-Szczykutowicz et al., 2021; Matam et al., 2017).
4.6 Bioremediation and environmental applications
The capacity to detoxify or break down contaminants exists in several secondary metabolites. These metabolites, which might be used in phytoremediation or bioremediation techniques to clean up polluted settings, can be stimulated by precursor feeding in plant cultures. For example, bacteria that can degrade Polychlorinated Biphenyls (PCBs) utilize coumarin and myricetin as carbon sources (Klimek-Szczykutowicz et al., 2021; Singer et al., 2003). In these cases, myricetin demonstrated the highest efficacy in PCB degradation. Burkholderia cepacia LB400, for instance, was able to degrade 16 out of 19 tested congeners using myricetin, while coumarin led to the degradation of 13 congeners by Corynebacterium sp. MB1, surpassing the efficacy of PCB degradation (Donnelly et al., 1994).
4.7 Customized metabolite profiles
The reaction mixture and concentration of the precursors can be changed to create specific metabolite profiles using precursor feeding techniques. The focused creation of metabolites following industrial, or research requirements is made possible by this level of customization. for example, according to Kiong et al., (Kiong et al., 2005), the addition of Farnesyl pyrophosphate (FPP), squalene, leucine, and Isopentenyl pyrophosphate (IPP) into Centella asiatica cell culture medium, can enhance triterpenoids productions.
Precursor feeding techniques in plant in-vitro culture provide a lot of benefits, but they also have certain drawbacks and difficulties that need to be considered before using them. The primary limitations of the precursor feeding strategy employed in plant in vitro culture are as follows:
The metabolic processes involved in the synthesis of phytochemicals are often complicated and may vary among different plant species, organs, and even individual plants. Comparing plant metabolic networks to those of other living things, they are considerably complex. This is caused by several interconnected characteristics of plant life, including its sessileness, ectothermic, and autotrophic nature, as well as its extensive chemical repertory and high levels of subcellular compartmentation (Allen et al., 2009). Moreover, the required secondary metabolites may not always be produced in high or consistent quantities because of precursor feeding (Palacio et al., 2011; Srivastava & Srivastava, 2014). The production and quality of the phytochemicals are affected by a broad spectrum of elements, including the stage of plant development, culture conditions, and genetic diversity (Verma and Shukla, 2015). Therefore, studies of gene expressions involved in secondary metabolite biosynthesis are also important (Jadid et al., 2016). Due to this intricacy and diversity, precursor feeding may not always provide predictable results.
Additionally, secondary metabolite biosynthesis pathways and the involved enzymes are poorly understood. The accurate implementation of precursor feeding techniques, and the optimization of results may be hampered by this lack of understanding. Metabolic pathway studies use a combination of several approaches such as: metabolic and bioinformatics pathway databases (e.g., metaCyc, cathaCyc, and KEGG), metabolite identification (GC–MS, HPLC–MS, and NMR), RNA-seq, Metabolic Flux Analysis (MFA), and structural interaction study of several enzymes implying in secondary metabolite pathways can help in studying phytochemicals pathways (Caspi et al., 2020; Jadid et al., 2024b; Marguerat and Bähler, 2010; Shih and Morgan, 2020; P. Zhang et al., 2005).
The production of a variety of phytochemicals by plant cells in response to precursor feeding might make it difficult to target the biosynthesis of a particular molecule. The targeted metabolite may not be a single reaction, resulting in a variety of chemicals in the culture. Several strategies such as optimizing co-culture condition, substrate channeling, CRISPR-Cas9 genome editing, and inhibiting undesired pathways with artificial microRNA, can be utilized for enhancing the specificity of precursor feeding (Endo et al., 2019; Hidalgo et al., 2017; Marchev et al., 2020; Y. Zhang and Fernie, 2021).
Using high precursor concentrations can be perilous for plant cells cultivated in vitro, potentially leading to cell death or reduced growth rates. Striking a delicate balance is crucial to optimize metabolite synthesis while mitigating potential cell damage caused by high precursor concentrations. An illustration of this intricate balance is evident in the use of sodium acetate as a precursor for Azadirachtin production in the hairy root culture of Azadirachta indica. Studies suggest that the inclusion of sodium acetate in the medium can potentially damage plant cells and trigger the release of oligogalacturonides from the plant cell (Srivastava and Srivastava, 2014). This signaling can subsequently trigger an upregulation in phytoalexin production, which are defensive-related secondary metabolites (Davis et al., 1986). In a different investigation by Palacio et al., (2011), focusing on nordihydroguaiaretic acid (NDGA) production from Larrea divaricata, the supplementation of l-phenylalanine at varying concentrations (3, 1, and 0.5 mM) resulted in a notable increase in NDGA levels, reaching of up to 190.53 ± 19.50, 285.23 ± 28.44, and 301.35 ± 1.19 μg/g DW, respectively. However, the introduction of 0.5 μM cinnamic acid promoted cell propagation but did not influence NDGA accumulation. On the other hand, when cinnamic acid (at 1 and 1.5 μM), ferulic acid (at 0.1, 0.5, and 1 mM), and sinapic acid (at 0.1, 0.5, and 1 mM) was supplemented, the medium became excessively toxic, leading to suppression of phenylpropanoid and NDGA production because of the high toxicity levels.
The cost corelated with obtaining and incorporating purified precursors into the culture medium can be relatively high, especially for specialized or rare precursors. This cost factor can significantly impact the economic feasibility and scalability of precursor feeding methods, particularly for mass production. To optimize the production of specific compounds in the culture system, it is essential to establish procedures that maximize the use of a precursor, especially when it is the most cost-effective byproduct of other processes (Isah et al., 2018). In modern scale-up production studies, there is a notable shift towards focusing on cell suspension cultures rather than differentiated tissues such as hairy roots and somatic embryos, which are used less frequently (Ferrie, 2010; Ziv, 2010). This change is probably attributed to the benefits of cell suspension cultures in comparison to tissue and organ cultures, particularly in terms of simplicity, predictability, and ease of extracting metabolites from the biomass or growth medium (Park and Paek, 2014). Furthermore, the biosynthesis of plant chemicals can be scaled up by employing a combination of various methods, including cell permeabilization, immobilization, and elicitation (Choi et al., 1995).
Beside of many advantageous of the in vitro precursor feeding for accelerating the quantity of PSMs, these techniques also genetic instability of the plant and production of the targeted compounds as well as cytotoxicity. Genetic instability in tissue cultures over extended periods often detrimental. Plant cells grown in vitro are often subject to somaclonal variation, leading to genetic mutations and altered metabolic profiles, reducing the reliability and consistency of metabolite production (Kang et al., 2011). Additionally, precursor uptake and transport can be also inefficient due to mechanic barriers of the plant like cell walls or the absence of specific transporters. This will end up with an inefficient uptake of the precursors into the cells. Moreover, high concentrations of precursors can induce stress or cytotoxicity, leading to cellular damage or abnormal metabolic responses. Therefore, suitable concentration of the precursors, re-checking the metabolic profile and additional genetic fidelity confirmation using molecular markers such as random Amplified Polymorphic DNA (RAPD), inter-simple sequence repeat (ISSR) and start codon targeted (SCoT) should also be performed (Andriyani and Jadid, 2021).
5 Conclusion
In summary, precursor feeding stands as a promising in-vitro culture technique, offering a scalable and sustainable approach for producing secondary metabolites without relying on traditional agricultural processes. It provides a flexible and effective strategy for controlling and enhancing the production of valuable phytochemicals. These applications present intriguing subjects for study and advancement in contemporary plant biotechnology, with far-reaching implications for various industries and research domains. To entirely harness the potential of precursor feeding strategies and optimize the formation of phytochemicals in plant in-vitro cultures, a deep understanding of the involved constraints is required. Enhancing the efficacy and efficiency of secondary metabolite synthesis necessitates focused attention on these aspects. Integration between precursor feeding and cutting-edge biotechnological methods like synthetic biology and metabolic engineering shows potential for substantially enhancing the precision and efficiency of phytochemical synthesis in plant cultures. However, it's important to acknowledge the existing limitations that affect the production of PSMs through this method including potential genetic instability, cytotoxicity and altered metabolic profile. Therefore, additional molecular-based approaches including RAPD, ISSR and ScoT as well as metabolomic assessment should also performed to complement the results of the study. Finally, continued research and progress in biotechnology have the potential to surmount these challenges, offering opportunities to enhance the application of precursor feeding for optimized phytochemical synthesis in plants.
CRediT authorship contribution statement
Muhammad Rifqi Nur Ramadani: Data curation, Visualization, Writing – original draft. Nurul Jadid: Conceptualization, Funding acquisition, Supervision, Writing – original draft, Writing – review & editing.
Acknowledgments
The authors gratefully acknowledge the Ministry of education, culture, research and technology of the Republic of Indonesia for financially supporting this project through fundamental research scheme No. 1770/PKS/ITS/2024.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Enhanced accumulation of scopoletin in cell suspension culture of Spilanthes acmella Murr. using precursor feeding. Braz. Arch. Biol. Technol.. 2016;59
- [CrossRef] [Google Scholar]
- Cardio-protective and antioxidant properties of caffeic acid and chlorogenic acid: mechanistic role of angiotensin converting enzyme, cholinesterase and arginase activities in cyclosporine induced hypertensive rats. Biomed. Pharmacotherapy. 2019;109:450-458.
- [CrossRef] [Google Scholar]
- Pharmacological properties of the ubiquitous natural product betulin. Eur. J. Pharm. Sci.. 2006;29(1):1-13.
- [CrossRef] [Google Scholar]
- Metabolic flux analysis in plants: coping with complexity. Plant Cell Environ.. 2009;32(9):1241-1257.
- [CrossRef] [Google Scholar]
- Berberine: alkaloid with wide spectrum of pharmacological activities. J. Nat. Prod. (India). 2010;3:64-75.
- [Google Scholar]
- Genetic diversity and morphological responses of Capsicum annuum varieties under aluminum stress. Biodiversitas J. Biol. Div.. 2021;22(5)
- [CrossRef] [Google Scholar]
- Methyl jasmonate stimulates growth and upregulates the expression of Phenylalanine Ammonia-Lyase (PAL) gene in Gynura pseudochina in vitro micropropagation. Biodiversitas J. Biol. Div.. 2024;25(5)
- [CrossRef] [Google Scholar]
- Mechanisms underlying the anti-inflammatory actions of boswellic acid derivatives in experimental colitis. American J. Physiol.-Gastrointestinal Liver Physiol.. 2006;290(6):G1131-G1137.
- [CrossRef] [Google Scholar]
- Molecular activities, biosynthesis and evolution of triterpenoid saponins. Phytochemistry. 2011;72(6):435-457.
- [CrossRef] [Google Scholar]
- Quercetin-rich onion peel extract suppresses adipogenesis by down-regulating adipogenic transcription factors and gene expression in 3T3-L1 adipocytes. J. Sci. Food Agric.. 2014;94(13):2655-2660.
- [CrossRef] [Google Scholar]
- Enhanced biosynthesis of triterpenoids in Centella asiatica hairy root culture by precursor feeding and elicitation. Plant Biotechnol. Reports. 2020;14:45-53.
- [CrossRef] [Google Scholar]
- Yield enhancement strategies for artemisinin production by suspension cultures of Artemisia annua. Bioresour. Technol.. 2008;99(11):4609-4614.
- [CrossRef] [Google Scholar]
- Antioxidant and antidepressant-like effects of Eugenia catharinensis D. Legrand in an animal model of depression induced by corticosterone. Metab. Brain Dis.. 2018;33:1985-1994.
- [CrossRef] [Google Scholar]
- The phenylpropanoid case–it is transport that matters. Front. Plant Sci.. 2018;9:1610.
- [CrossRef] [Google Scholar]
- Elicitation and phenylalanine precursor feeding based modulation of in vitro anthocyanin production, enzyme activity and gene expression in an Indian ginseng congener-Panax sikkimensis Ban. Ind. Crop. Prod.. 2020;145:111986
- [CrossRef] [Google Scholar]
- Artificial color light sources and precursor feeding enhance plumbagin production of the carnivorous plants Drosera burmannii and Drosera indica. J. Photochem. Photobiol. B Biol.. 2019;199:111628
- [CrossRef] [Google Scholar]
- Production of plant secondary metabolites: a historical perspective. Plant Sci.. 2001;161(5):839-851.
- [CrossRef] [Google Scholar]
- Biochemistry and Molecular Biology of Plants. John wiley & sons; 2015.
- Antidepressant-like effects of Trichilia catigua (Catuaba) extract: evidence for dopaminergic-mediated mechanisms. Psychopharmacology. 2005;182:45-53.
- [CrossRef] [Google Scholar]
- The MetaCyc database of metabolic pathways and enzymes-a 2019 update. Nucleic Acids Res.. 2020;48(D1):D445-D453.
- [CrossRef] [Google Scholar]
- Bisphenol F and bisphenol S induce metabolic perturbations in human ovarian granulosa cells. Arabian Journal of Chemistry. 2024;17(9):105904.
- [CrossRef] [Google Scholar]
- Marked enhancement of sennoside bioactive compounds through precursor feeding in Cassia angustifolia Vahl and cloning of isochorismate synthase gene involved in its biosynthesis. Plant Cell Tissue Organ Culture (PCTOC). 2016;124:431-446.
- [CrossRef] [Google Scholar]
- Plant phenolics: recent advances on their biosynthesis, genetics, and ecophysiology. Plant Physiol. Biochem.. 2013;72:1-20.
- [CrossRef] [Google Scholar]
- Plant secondary metabolites: an opportunity for circular economy. Molecules. 2021;26(2):495.
- [CrossRef] [Google Scholar]
- Enhancement of secondary metabolite production by immobilized Gossypium arboreum cells. Biotechnol. Prog.. 1995;11(3):306-311.
- [CrossRef] [Google Scholar]
- Cell differentiation and secondary metabolite production. Morphogenesis Plant Tissue Cult. 1999:463-501.
- [CrossRef] [Google Scholar]
- Plant secondary metabolites. In: Occurrence, Structure and Role in the Human Diet. Blackwell–Publishers; 2006.
- [Google Scholar]
- Enhancement of production of pharmaceutically important anti-cancerous compound; cucurbitacin E via elicitation and precursor feeding of in vitro culture of Citrullus colocynthis (L.) Schard. Vegetos. 2020;33:323-334.
- [CrossRef] [Google Scholar]
- Several biotic and abiotic elicitors act synergistically in the induction of phytoalexin accumulation in soybean. Plant Mol. Biol.. 1986;6(1):23-32.
- [CrossRef] [Google Scholar]
- Role of plant alkaloids on human health: a review of biological activities. Mater. Today Chem.. 2018;9:56-72.
- [CrossRef] [Google Scholar]
- Anthelmintic effect and tissue alterations induced in vitro by hydrolysable tannins on the adult stage of the gastrointestinal nematode Haemonchus contortus. Vet. Parasitol.. 2019;266:1-6.
- [CrossRef] [Google Scholar]
- The gastrointestinal behavior of saponins and its significance for their bioavailability and bioactivities. J. Funct. Foods. 2018;40:484-497.
- [CrossRef] [Google Scholar]
- Enhanced rosmarinic acid biosynthesis in Solenostemon scutellarioides culture: a precursor-feeding strategy. Nat. Prod. Res.. 2014;28(20):1691-1698.
- [CrossRef] [Google Scholar]
- Medicinal Natural Products: A Biosynthetic Approach. John Wiley & Sons; 2002.
- Improvement of plant quality by amino acid transporters: a comprehensive review. Plant Physiol. Biochem. 2024
- [CrossRef] [Google Scholar]
- Growth of PCB-degrading bacteria on compounds from photosynthetic plants. Chemosphere. 1994;28(5):981-988.
- [CrossRef] [Google Scholar]
- Tools of pathway reconstruction and production of economically relevant plant secondary metabolites in recombinant microorganisms. Biotechnol. J.. 2017;12(1):1600145.
- [CrossRef] [Google Scholar]
- A novel approach to carotenoid accumulation in rice callus by mimicking the cauliflower Orange mutation via genome editing. Rice. 2019;12(1):1-5.
- [CrossRef] [Google Scholar]
- In vitro plant tissue culture: means for production of biological active compounds. Planta. 2018;248:1-18.
- [CrossRef] [Google Scholar]
- Protocols for In Vitro Cultures and Secondary Metabolite Analysis of Aromatic and Medicinal Plants. Oxford University Press; 2010.
- Enhancement of silymarin production in cell culture of Silybum marianum (L) Gaertn by elicitation and precursor feeding. J. Herbs Spices Med. Plants. 2013;19(3):262-274.
- [CrossRef] [Google Scholar]
- Transdermal drug delivery enhancement by compounds of natural origin. Molecules. 2011;16(12):10507-10540.
- [CrossRef] [Google Scholar]
- Brazilian myrtaceae fruits: a review of anticancer proprieties. J. Pharm. Res. Int. 2016;12:1-15.
- [CrossRef] [Google Scholar]
- Production of plant secondary metabolites: examples, tips and suggestions for biotechnologists. Genes. 2018;9(6):309.
- [CrossRef] [Google Scholar]
- Antifungal activity of the components of Melaleuca alternifolia (tea tree) oil. J. Appl. Microbiol.. 2003;95(4):853-860.
- [CrossRef] [Google Scholar]
- Silymarin accumulation in Silybum marianum suspension culture via precursor feeding. Catrina: Int. J. Environ. Sci.. 2021;24(1):15-23.
- [CrossRef] [Google Scholar]
- Improving α-tocopherol accumulation in Argania spinosa suspension cultures by precursor and nanoparticles feeding. Plant Arch. 2020;20:2431-2437.
- [Google Scholar]
- Enhancing in vitro production of Bacoside a from Bacopa monnieri using precursor and elicitors feeding. J. Basic Appl. Sci. Res.. 2017;7(9):27-35.
- [Google Scholar]
- Tailoring tobacco hairy root metabolism for the production of stilbenes. Sci. Rep.. 2017;7(1):17976.
- [CrossRef] [Google Scholar]
- Studies of the phytotoxicity of saponins on weed and crop plants. Saponins Used Food Agric. 1996:57-73.
- [CrossRef] [Google Scholar]
- Effect of the flavonoid fraction of Lithocarpus polystachyus Rehd. on spontaneously hypertensive and normotensive rats. J. Ethnopharmacol.. 2012;143(2):441-447.
- [CrossRef] [Google Scholar]
- Current approaches toward production of secondary plant metabolites. J. Pharm. Bioallied Sci.. 2012;4(1):10.
- [CrossRef] [Google Scholar]
- Improvement of stilbene production by mulberry Morus alba root culture via precursor feeding and co-elicitation. Bioprocess Biosyst. Eng.. 2021;44:653-660.
- [CrossRef] [Google Scholar]
- Secondary metabolism of pharmaceuticals in the plant in vitro cultures: strategies, approaches, and limitations to achieving higher yield. Plant Cell Tissue Organ Culture (PCTOC). 2018;132:239-265.
- [CrossRef] [Google Scholar]
- N-Nitrosopiperazines form at high pH in post-combustion capture solutions containing piperazine: a low-energy collisional behaviour study. Rapid Commun. Mass Spectrom.. 2010;24(24):3567-3577.
- [CrossRef] [Google Scholar]
- Reverse transcription-PCR analysis of geranylgeranyl diphosphate synthase (JcGGPPS) in Jatropha curcas L. and in silico analysis of Casbene Synthase (JcCS) among Euphorbiaceae. AIP Conf. Proc.. 2016;1744(1)
- [CrossRef] [Google Scholar]
- Growth and physiological responses of some Capsicum frutescens varieties to copper stress. AIP Conf. Proc.. 2017;1854:020018
- [CrossRef] [Google Scholar]
- Proximate composition, nutritional values and phytochemical screening of Piper retrofractum vahl. fruits. Asian Pac. J. Trop. Biomed.. 2018;7(1):37-43.
- [CrossRef] [Google Scholar]
- The medicinal Umbelliferae plant Fennel (Foeniculum vulgare Mill.): cultivation, traditional uses, phytopharmacological properties, and application in animal husbandry. Arab. J. Chem. 2023
- [CrossRef] [Google Scholar]
- In vitro propagation of Indonesian stevia (Stevia rebaudiana) genotype using axenic nodal segments. BMC Res. Notes. 2024;17(45)
- [CrossRef] [Google Scholar]
- In silico characterization of GbPAL, GbCHS, GbDFR and GbANS structural genes involved in the biosynthesis of flavonoids in Gynura bicolor DC. S. Afr. J. Bot.. 2024;165:428-442.
- [CrossRef] [Google Scholar]
- Exogenous methyl jasmonate (MeJA) altered phytochemical composition and enhanced the expression of PatAACT gene of in vitro culture-derived patchouli var. Sidikalang (Pogostemon cablin Benth.) J. King Saud Univ.-Sci. 2024
- [CrossRef] [Google Scholar]
- Inhibitory effects of myricetin on lipopolysaccharide-induced neuroinflammation. Brain Sci.. 2020;10(1):32.
- [CrossRef] [Google Scholar]
- Enhancing the accumulation of rosavins in Rhodiola rosea L. plants grown in vitro by precursor feeding. Agronomy. 2021;11(12):2531.
- [CrossRef] [Google Scholar]
- Food ingredients that inhibit cholesterol absorption. Prevent. Nutr. Food Sci.. 2017;22(2):67.
- [CrossRef] [Google Scholar]
- Effect of elicitation and precursor feeding on accumulation of 20-hydroxyecdysone in Achyranthes aspera Linn. cell suspension cultures. Physiol. Mol. Biol. Plants. 2018;24:275-284.
- [CrossRef] [Google Scholar]
- In vivo antifungal effects of Coptis japonica root-derived isoquinoline alkaloids against phytopathogenic fungi. J. Microbiol. Biotechnol.. 2005;15(6):1402-1407.
- [Google Scholar]
- Plant ABC transporters. Arabidopsis Book/American Soc. Plant Biol.. 2011;9
- [CrossRef] [Google Scholar]
- Biosynthesis of hyperforin and adhyperforin from amino acid precursors in shoot cultures of Hypericum perforatum. Phytochemistry. 2007;68(7):1038-1045.
- [CrossRef] [Google Scholar]
- Effects of precursor supplementation on the production of triterpenes by Centella asiatica callus culture. Pak. J. Biol. Sci.. 2005;8(8):1160-1169.
- [CrossRef] [Google Scholar]
- Precursor-boosted production of Metabolites in Nasturtium officinale microshoots grown in Plantform Bioreactors, and antioxidant and antimicrobial activities of Biomass extracts. Molecules. 2021;26(15):4660.
- [CrossRef] [Google Scholar]
- Stimulation of antibacterial naphthoquinones and flavonoids accumulation in carnivorous plants grown in vitro by addition of elicitors. Enzyme Microb. Technol.. 2008;42(3):216-221.
- [CrossRef] [Google Scholar]
- Exogenous feeding of immediate precursors reveals synergistic effect on picroside-I biosynthesis in shoot cultures of Picrorhiza kurroa Royle ex Benth. Sci. Rep.. 2016;6(1):29750.
- [CrossRef] [Google Scholar]
- Development of transgenic hairy roots and augmentation of secondary metabolites by precursor feeding in Sphagneticola calendulacea (L.) Pruski. Ind. Crop. Prod.. 2018;121:206-215.
- [CrossRef] [Google Scholar]
- Effects of tannins on Chinese hamster cell line B14. Mutat. Res./Genet. Toxicol. Environ. Mutagenesis. 2003;539(1–2):127-135.
- [CrossRef] [Google Scholar]
- Precursor-feeding strategy for the production of solanine, solanidine and solasodine by a cell culture of Solanum lyratum. Process Biochem.. 2007;42(5):899-903.
- [CrossRef] [Google Scholar]
- Screening of ninety herbal products of commercial interest as potential ingredients for phytocosmetics. J. Enzyme Inhib. Med. Chem.. 2020;35(1):1287-1291.
- [CrossRef] [Google Scholar]
- Mosquito larvicidal activity of alkaloids and limonoids derived from Evodia rutaecarpa unripe fruits against Aedes albopictus (Diptera: Culicidae) Parasitol. Res.. 2012;111:991-996.
- [CrossRef] [Google Scholar]
- A commentary on methodological aspects of hydrolysable tannins metabolism in ruminant: A perspective view. Lett. Appl. Microbiol.. 2020;71(5):466-478.
- [CrossRef] [Google Scholar]
- Met1-specific motifs conserved in OTUB subfamily of green plants enable rice OTUB1 to hydrolyse Met1 ubiquitin chains. Nature Communications. 2022;13(1):4672.
- [CrossRef] [Google Scholar]
- Pharmacological effects of ephedrine alkaloids on human α1-and α2-adrenergic receptor subtypes. J. Pharmacol. Exp. Ther.. 2007;322(1):214-221.
- [CrossRef] [Google Scholar]
- A unique reaction in a common pathway: mechanism and function of chorismate synthase in the shikimate pathway. Planta. 1999;207:325-334.
- [CrossRef] [Google Scholar]
- Improvement of alkaloids yield using phenylalanine as a precursor supplemented to Morina oleifera L. callus cultures. Biochem. Cell. Arch.. 2018;18
- [Google Scholar]
- Green (cell) factories for advanced production of plant secondary metabolites. Crit. Rev. Biotechnol.. 2020;40(4):443-458.
- [CrossRef] [Google Scholar]
- RNA-seq: from technology to biology. Cell. Mol. Life Sci.. 2010;67:569-579.
- [CrossRef] [Google Scholar]
- Effects of saponins on lipid metabolism: A review of potential health benefits in the treatment of obesity. Molecules. 2016;21(10):1404.
- [CrossRef] [Google Scholar]
- Rat pial microvascular changes during cerebral blood flow decrease and recovery: Effects of cyanidin administration. Front. Physiol.. 2018;9:540.
- [CrossRef] [Google Scholar]
- Enhanced production of vanillin flavour metabolites by precursor feeding in cell suspension cultures of Decalepis hamiltonii Wight & Arn., in shake flask culture. 3 Biotech. 2017;7:1-9.
- [CrossRef] [Google Scholar]
- Natural emulsifiers—Biosurfactants, phospholipids, biopolymers, and colloidal particles: Molecular and physicochemical basis of functional performance. Adv. Colloid Interface Sci.. 2016;234:3-26.
- [CrossRef] [Google Scholar]
- Use of shotgun metagenomics and metabolomics to evaluate the impact of glyphosate or Roundup MON 52276 on the gut microbiota and serum metabolome of Sprague-Dawley rats. Environ. Health Perspect.. 2021;129(1):17005.
- [CrossRef] [Google Scholar]
- Improvement in the biosynthesis of antioxidant-active metabolites in in vitro cultures of isatis tinctoria (Brassicaceae) by biotechnological methods/elicitation and precursor feeding. Antioxidants. 2023;12(5):1111.
- [CrossRef] [Google Scholar]
- Identification of a novel UDP-glycosyltransferase gene from Rhodiola rosea and its expression during biotransformation of upstream precursors in callus culture. Int. J. Biol. Macromol.. 2019;136:847-858.
- [CrossRef] [Google Scholar]
- Induction and analysis of the alkaloid mitragynine content of a Mitragyna speciosa suspension culture system upon elicitation and precursor feeding. Sci. World J.. 2013;2013
- [CrossRef] [Google Scholar]
- In vitro enhancement of psoralen as an important anticancer compound in Psoralea corylifolia through precursor feeding. Pharm. Biol.. 2015;53(5):735-738.
- [CrossRef] [Google Scholar]
- Metabolic and functional diversity of saponins, biosynthetic intermediates and semi-synthetic derivatives. Crit. Rev. Biochem. Mol. Biol.. 2014;49(6):439-462.
- [CrossRef] [Google Scholar]
- Quality of leather using vegetable tannins extract of Acacia mangium bark from waste of industrial plantation. IOP Conference Ser.: Earth Environ. Sci.. 2019;327(1):12012.
- [CrossRef] [Google Scholar]
- Phcog Rev.: review article precursor feeding for enhanced production of Secondary metabolites: a review. Pharmacogn Rev. 2007;1:227-231.
- [Google Scholar]
- Natural products as sources of new drugs over the nearly four decades from 01/1981 to 09/2019. J. Nat. Prod.. 2020;83(3):770-803.
- [CrossRef] [Google Scholar]
- Resistance in the plant, Barbarea vulgaris, and counter-adaptations in flea beetles mediated by saponins. J. Chem. Ecol.. 2010;36:277-285.
- [CrossRef] [Google Scholar]
- Micropropagation and elicited production of triterpenoid saponin glycosides and stigmasterol via precursor and elicitor feeding in Bacopa floribunda (R. Br.) Wettst.—a potential nootropic herb. Front. Plant Sci.. 2023;14
- [CrossRef] [Google Scholar]
- Phenolic compound production by Larrea divaricata Cav. plant cell cultures and effect of precursor feeding. Process Biochem.. 2011;46(1):418-422.
- [CrossRef] [Google Scholar]
- Stimulation of reserpine production in the whole plant culture of Rauwolfia serpentina L. by elicitors and precursor feeding. J. Plant Biochem. Biotechnol.. 2015;24:49-55.
- [CrossRef] [Google Scholar]
- Bioreactor culture of shoots and somatic embryos of medicinal plants for production of bioactive compounds. Prod. Biomass Bioactive Compounds Bioreactor Technol. 2014:337-368.
- [CrossRef] [Google Scholar]
- In vitro propagation and production of cardiotonic glycosides in shoot cultures of Digitalis purpurea L. by elicitation and precursor feeding. Appl. Microbiol. Biotechnol.. 2013;97:2379-2393.
- [CrossRef] [Google Scholar]
- Precursor-induced bioaccumulation of secondary metabolites and antioxidant activity in suspension cultures of Dendrobium fimbriatum, an orchid of therapeutic importance. S. Afr. J. Bot.. 2020;135:137-143.
- [CrossRef] [Google Scholar]
- Antioxidant properties of isolated isorhamnetin from the sea buckthorn marc. Plant Foods Hum. Nutr.. 2009;64:141-145.
- [CrossRef] [Google Scholar]
- Induction of central signalling pathways and select functional effects in human platelets by β-boswellic acid. Br. J. Pharmacol.. 2005;146(4):514-524.
- [CrossRef] [Google Scholar]
- 3-O-acetyl-11-keto-boswellic acid decreases basal intracellular Ca2+ levels and inhibits agonist-induced Ca2+ mobilization and mitogen-activated protein kinase activation in human monocytic cells. J. Pharmacol. Exp. Ther.. 2006;316(1):224-232.
- [CrossRef] [Google Scholar]
- Combined UV-C irradiation and precursor feeding enhances mulberroside A production in Morus alba L. cell suspension cultures. ScienceAsia. 2020;46(6)
- [CrossRef] [Google Scholar]
- A combination of elicitation and precursor feeding leads to increased anthocyanin synthesis in cell suspension cultures of Vitis vinifera. Plant Cell Tissue Organ Culture (PCTOC). 2011;107:261-269.
- [CrossRef] [Google Scholar]
- Elicitors and precursor induced effect on L-Dopa production in suspension cultures of Mucuna pruriens L. Front. Life Sci.. 2011;5(3–4):127-133.
- [CrossRef] [Google Scholar]
- Differential effects of elicitor and precursor feeding on the enzyme tyrosine hydroxylase and their influence on the production of L-Dopa in the cell suspension cultures of Mucuna prurita H. J. Pharmacognosy Phytochem.. 2018;7(4):179-185.
- [Google Scholar]
- Paclitaxel production is enhanced in suspension-cultured hazel (Corylus avellana L.) cells by using a combination of sugar, precursor, and elicitor. Eng. Life Sci.. 2015;15(2):234-242.
- [CrossRef] [Google Scholar]
- Neglected Mediterranean plant species are valuable resources: the example of Cistus ladanifer. Planta. 2018;248:1351-1364.
- [CrossRef] [Google Scholar]
- Elicitor and precursor-induced approaches to enhance the in vitro production of L-DOPA from cell cultures of Mucuna pruriens. Ind. Crop. Prod.. 2022;188:115735
- [CrossRef] [Google Scholar]
- Plant cell cultures: chemical factories of secondary metabolites. Biotechnol. Adv.. 2002;20(2):101-153.
- [CrossRef] [Google Scholar]
- More for less: improving the biomass yield of a pear cell suspension culture by design of experiments. Sci. Rep.. 2016;6(1):23371.
- [CrossRef] [Google Scholar]
- Enhanced production of phenylethanoids mediated through synergistic approach of precursor feeding and light regime in cell suspension culture of Rhodiola imbricata (Edgew.) Appl. Biochem. Biotechnol.. 2022;194(7):3242-3260.
- [CrossRef] [Google Scholar]
- Perspectives in the use of tannins as alternative to antimicrobial growth promoter factors in poultry. Front. Microbiol.. 2014;5:118.
- [CrossRef] [Google Scholar]
- Structural diversity, natural sources, and pharmacological potential of plant-based saponins with special focus on anticancer activity: a review. Med. Chem. Res.. 2020;29(10):1707-1722.
- [CrossRef] [Google Scholar]
- Elicitation and precursor feeding influence phenolic acids composition in Vitis vinifera suspension culture. Afr. J. Biotechnol.. 2012;11(12):3000-3008.
- [CrossRef] [Google Scholar]
- High performance liquid chromatography based quantification of reserpine in Rauwolfia tetraphylla L. and enhanced production through precursor feeding. Acta Chromatogr.. 2021;34(2):120-129.
- [CrossRef] [Google Scholar]
- Remission induction therapy for childhood acute lymphoblastic leukaemia: clinical and cellular pharmacology of vincristine, corticosteroids, L-asparaginase and anthracyclines. Cancer Treatment Rev.. 2001;27(6):327-337.
- [CrossRef] [Google Scholar]
- Pharmacokinetic, antiproliferative and apoptotic effects of phenolic acids in human colon adenocarcinoma cells using in vitro and in silico approaches. Molecules. 2018;23(10):2569.
- [CrossRef] [Google Scholar]
- Application of anthocyanins from blackcurrant (Ribes nigrum L.) fruit waste as renewable hair dyes. J. Agric. Food Chem.. 2018;66(26):6790-6798.
- [CrossRef] [Google Scholar]
- Thymol, thyme, and other plant sources: Health and potential uses. Phytother. Res.. 2018;32(9):1688-1706.
- [CrossRef] [Google Scholar]
- Potential of the chlorogenic acid as multitarget agent: Insulin-secretagogue and PPAR α/γ dual agonist. Biomed. Pharmacother.. 2017;94:169-175.
- [CrossRef] [Google Scholar]
- Precursor feeding enhances L-Dopa production in hairy root culture of Hybanthus enneaspermus (L.) F. Muell. Biologia. 2023;78(3):913-923.
- [CrossRef] [Google Scholar]
- Enhanced production of organosulfur bioactive compounds in cell suspension culture of single garlic (Allium sativum L.) using precursor feeding. Jordan J. Biol. Sci.. 2022;15(2)
- [CrossRef] [Google Scholar]
- Agrobacterium rhizogenes-mediated transformation: root cultures as a source of alkaloids. Planta Med.. 2002;68(10):859-868.
- [CrossRef] [Google Scholar]
- Metabolic flux analysis of secondary metabolism in plants. Metab. Eng. Commun.. 2020;10
- [CrossRef] [Google Scholar]
- Antagonizing effects and mechanisms of afzelin against UVB-induced cell damage. PLoS One. 2013;8(4)
- [CrossRef] [Google Scholar]
- Enhanced production of phytoestrogenic isoflavones from hairy root cultures of Psoralea corylifolia L. using elicitation and precursor feeding. Biotechnol. Bioprocess Eng.. 2009;14:288-294.
- [CrossRef] [Google Scholar]
- Secondary plant metabolites in phytoremediation and biotransformation. Trends Biotechnol.. 2003;21(3):123-130.
- [CrossRef] [Google Scholar]
- Plant terpenes: defense responses, phylogenetic analysis, regulation and clinical applications. 3 Biotech. 2015;5(2):129-151.
- [CrossRef] [Google Scholar]
- Saponins in pulses and their health promoting activities: a review. Food Chem.. 2017;233:540-549.
- [CrossRef] [Google Scholar]
- Enhanced biosynthesis of withanolides by elicitation and precursor feeding in cell suspension culture of Withania somnifera (L.) Dunal in shake-flask culture and bioreactor. PLoS One. 2014;9(8):e104005.
- [Google Scholar]
- Enhanced production of the pharmaceutically important polyphenolic compounds in Vitex agnus castus L. shoot cultures by precursor feeding strategy. Eng. Life Sci.. 2018;18(5):287-297.
- [CrossRef] [Google Scholar]
- Floral phytochemistry: impact of volatile organic compounds and nectar secondary metabolites on pollinator behavior and health. Chem. Biodivers.. 2023;20(4)
- [CrossRef] [Google Scholar]
- Aescin-cholesterol complexes in DMPC model membranes: a DSC and temperature-dependent scattering study. Sci. Rep.. 2019;9(1):5542.
- [CrossRef] [Google Scholar]
- Effect of elicitors and precursors on azadirachtin production in hairy root culture of Azadirachta indica. Appl. Biochem. Biotechnol.. 2014;172:2286-2297.
- [CrossRef] [Google Scholar]
- Natural products–learning chemistry from plants. Biotechnology journal. 2014;9(3):326-336.
- [CrossRef] [Google Scholar]
- Effect of l-phenylalanine on PAL activity and production of naphthoquinone pigments in suspension cultures of Arnebia euchroma (Royle) Johnst. In Vitro Cell. Dev. Biol.-Plant. 2012;48:555-564.
- [CrossRef] [Google Scholar]
- The effect of feeding culture media with biogenetic precursors on high production of depsides in agitated shoot cultures of black and red aronias. Plant Cell, Tissue Organ Culture (PCTOC). 2020;142(2):379-399.
- [CrossRef] [Google Scholar]
- In vitro studies on Egyptian Catharanthus roseus (L.) G. Don. IV: manipulation of some amino acids as precursors for enhanced of indole alkaloids production in suspension cultures. Australian J. Basic Appl. Sci.. 2009;3(4):3137-3144.
- [Google Scholar]
- Flavonoids showed anticancer effects on the ovarian cancer cells: involvement of reactive oxygen species, apoptosis, cell cycle and invasion. Biomed. Pharmacother.. 2019;116:109004
- [CrossRef] [Google Scholar]
- Integration of phenylalanine precursor feeding and elicitor treatment in concert with the cellulase-assisted extraction approach to improve stilbene compound production. Plant Cell, Tissue and Organ Culture (PCTOC). 2023;155(3):665-679.
- [CrossRef] [Google Scholar]
- Dual mode of the saponin aescin in plant protection: antifungal agent and plant defense elicitor. Front. Plant Sci.. 2019;10:1448.
- [CrossRef] [Google Scholar]
- Plant tissue culture technology: sustainable option for mining high value pharmaceutical compounds. Int. J. Curr. Microbiol. App. Sci.. 2019;8(2):102-110.
- [CrossRef] [Google Scholar]
- Berberine and its more biologically available derivative, dihydroberberine, inhibit mitochondrial respiratory complex I: a mechanism for the action of berberine to activate AMP-activated protein kinase and improve insulin action. Diabetes. 2008;57(5):1414-1418.
- [CrossRef] [Google Scholar]
- Cassia occidentalis L. (a new source of rotenoids): its in vitro regulation by feeding precursors and larvicidal efficacy. Plant Cell Tissue Organ Culture (PCTOC). 2014;116:403-409.
- [CrossRef] [Google Scholar]
- Improved sanguinarine production via biotic and abiotic elicitations and precursor feeding in cell suspensions of latex-less variety of Papaver somniferum with their gene expression studies and upscaling in bioreactor. Protoplasma. 2014;251:1359-1371.
- [CrossRef] [Google Scholar]
- Regulation of vincamine biosynthesis and associated growth promoting effects through abiotic elicitation, cyclooxygenase inhibition, and precursor feeding of bioreactor grown Vinca minor hairy roots. Appl. Biochem. Biotechnol.. 2014;173:663-672.
- [CrossRef] [Google Scholar]
- Impact of various factors responsible for fluctuation in plant secondary metabolites. J. Appl. Res. Med. Aromat. Plants. 2015;2(4):105-113.
- [CrossRef] [Google Scholar]
- Lowering of cholesterol bioaccessibility and serum concentrations by saponins: in vitro and in vivo studies. Food Funct.. 2015;6(2):501-512.
- [CrossRef] [Google Scholar]
- In vitro growth and content of vincristine and vinblastine of Catharanthus roseus L. hairy roots in response to precursors and elicitors. Plant Sci. Today. 2022;9(1):21-28.
- [CrossRef] [Google Scholar]
- Improved triterpenoid saponin glycosides accumulation in in vitro culture of Bacopa monnieri (L.) Wettst with precursor feeding and LED light exposure. Ind. Crop. Prod.. 2019;134:303-308.
- [CrossRef] [Google Scholar]
- Recent advances towards development and commercialization of plant cell culture processes for the synthesis of biomolecules. Plant Biotechnol. J.. 2012;10(3):249-268.
- [CrossRef] [Google Scholar]
- Production of useful terpenoids by higher-fungus cell factory and synthetic biology approaches. Trends Biotechnol.. 2016;34(3):242-255.
- [CrossRef] [Google Scholar]
- A piperidine amide extracted from Piper longum L. fruit shows activity against Aedes aegypti mosquito larvae. J. Agric. Food Chem.. 2002;50(13):3765-3767.
- [CrossRef] [Google Scholar]
- StWRKY8 transcription factor regulates benzylisoquinoline alkaloid pathway in potato conferring resistance to late blight. Plant Sci.. 2017;256:208-216.
- [CrossRef] [Google Scholar]
- Caffeic acid improves locomotor activity and lessens inflammatory burden in a mouse model of rotenone-induced nigral neurodegeneration: Relevance to Parkinson’s disease therapy. Pharmacol. Rep.. 2019;71:32-41.
- [CrossRef] [Google Scholar]
- Enhanced thebaine production in Papaver bracteatum cell suspension culture by combination of elicitation and precursor feeding. Nat. Prod. Res.. 2014;28(10):711-717.
- [CrossRef] [Google Scholar]
- Metabolons, enzyme–enzyme assemblies that mediate substrate channeling, and their roles in plant metabolism. Plant Commun.. 2021;2(1)
- [CrossRef] [Google Scholar]
- MetaCyc and AraCyc. Metabolic pathway databases for plant research. Plant Physiol.. 2005;138(1):27-37.
- [CrossRef] [Google Scholar]
- Anti-hepatitis B virus activities of triterpenoid saponin compound from Potentilla anserine L. Phytomedicine. 2008;15(4):253-258.
- [CrossRef] [Google Scholar]
- Bioreactor technology for plant micropropagation. Janick. J. Editor. Horticulture Rev.. 2010;24
- [Google Scholar]
Appendix A
Supplementary data
Supplementary data to this article can be found online at https://doi.org/10.1016/j.arabjc.2024.106018.
Appendix A
Supplementary data
The following are the Supplementary data to this article:Supplementary Data 1
Supplementary Data 1







