Translate this page into:
A review of Viticis Fructus: botany, historical records, phytochemistry, pharmacology, toxicity, quality control, pharmacokinetics and comprehensive applications
⁎Corresponding authors at: State Key Laboratory of Component-based Chinese Medicine, Tianjin University of Traditional Chinese Medicine, Tianjin 301617, China. dkztcm@tjutcm.edu.cn (Kunze Du), Tcmcyx@tjutcm.edu.cn (Yanxu Chang)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Abstract

Abstract
Background
Viticis Fructus (also known as Manjingzi) has been used in China for more than 2000 years. It is one of the most famous traditional Chinese medicines with the main effect of dispelling wind-heat. In Asian countries, it is used in the treatment of common cold with wind-heat syndrome, swollen gums, migraines, dizziness, etc.
The aim of the review
The paper emphasizes on the botany, historical records, phytochemistry, pharmacology, toxicity, quality control, pharmacokinetics and comprehensive applications of Viticis Fructus to furnish with a scientific theoretical reference for its exploration and applications.
Materials and methods
Correlative data on Viticis Fructus were obtained from PubMed, ScienceDirect, Web of science, Embase, Scopus, Google Scholar, CNKI, WeiPu, Chinese ancient books and DuXiu academic search. The data collection ended in May 2024.
Results
The results showed 324 compounds, including terpenoids, flavonoids, lignans and others, which were isolated and identified from Viticis Fructus. In addition to treating headaches and eye pain, Viticis Fructus also had anti-inflammatory, antiplatelet activation, analgesia, antitumor and antihypertensive effects. In addition, this review summarized the botany, toxicity, counterfeit identification, pharmacokinetics and patent information of Viticis Fructus in detail. The shortcomings and feasible suggestions are put forward, which provide a basis for further research and utilization of Viticis Fructus.
Conclusion
Viticis Fructus has been used to remedy common cold with wind-heat syndrome, cephalalgia, ophthalmodynia, dizziness, etc. The pharmacological activities of the main components have been clarified and backed up the traditional applications. However, the botany, processing, pharmacological mechanism, quality control and toxicological studies of Viticis Fructus need to be further improved.
Keywords
Viticis Fructus
Phytochemistry
Pharmacology
Pharmacokinetics
Comprehensive applications
- AUC (0-t)
-
Area under the plasma concentration–time curve
- Bax
-
Bcl-2 associated X protein
- Bcl-2
-
B cell lymphoma-2 protein
- Bcl-xL
-
B-cell lymphoma extra-large
- Bim
-
Bcl-2 interacting mediator of cell death
- Caspase-3
-
Cysteinyl aspartate specific proteinase-3
- CD
-
Circular dichroism
- ChP
-
Pharmacopoeia of the People’s Republic of China
- Cmax
-
Maximum drug concentration in plasma
- CNKI
-
China National Knowledge Infrastructure
- COSY
-
Correlation spectroscopy
- CRC
-
Colorectal cancer
- CYR61
-
Cysteine-rich61
- DR5
-
Death receptor 5
- ERK
-
Extracellular regulated protein kinases
- FOXM1
-
Forkhead box protein M1
- FOXO3a
-
Forkhead box O3
- GABA
-
γ-amino butyric acid
- GC×GC–MS
-
Comprehensive two-dimensional gas chromatography hyphenated with mass spectrometry
- HCC
-
Hepatocellular carcinoma
- HDL
-
High-density lipoprotein
- HRESIMS
-
High-resolution electrospray ionization mass spectroscopy
- HRFABMS
-
High-resolution fast atom bombardment mass spectrum
- KOA
-
Knee osteoarthritis
- LC/MS
-
Liquid chromatography/mass spectrometry
- LCSLCs
-
Lung cancer stem-like cells
- LDL
-
Low density lipoprotein
- LD50
-
median lethal dose
- MAPK
-
Mitogen-activated protein kinase
- MMP-1
-
Matrix metalloproteinase-1
- MMP-2
-
Matrix metalloproteinase-2
- MMP-9
-
Matrix metalloproteinase-9
- NF-κB
-
Nuclear factor kappa-B
- NIR
-
Near Infrared
- NLRP3
-
Nucleotide oligomerization domain-like receptor protein 3
- NMR
-
Nuclear magnetic resonance
- NO
-
Nitric Oxide
- NOESY
-
Nuclear overhauser effect spectroscopy
- NPC
-
Nasopharyngeal carcinoma
- PI3K/AKT
-
Phosphatidylinositol 3-kinase/protein kinase B
- PKR
-
Protein kinase R
- PMS
-
Premenstrual syndrome
- PPY
-
Pyranopyran-1,8-dione
- ROS
-
Reactive oxygen species
- IL-1β
-
Interleukin-1β
- IL-6
-
Interleukin-6
- IR
-
Infrared ray
- SCTF
-
Shrub Chaste Tree Fruits
- TCMs
-
Traditional Chinese medicines
- Tmax
-
The time to reach maximum drug concentration
- TNF-α
-
Tumor necrosis factor-α
- UC
-
Ulcerative colitis
- HPLC-DAD
-
High-performance liquid chromatography coupled with a diode array detector
- ISSR
-
Inter simple sequence repeat
- ITS2
-
Internal transcribed spacer 2
- JNK
-
c-Jun N-terminal kinase
- K2P
-
Kimura 2-Parameter
- UHPLC-Q-Orbitrap HRMS
-
Ultra-high-performance-liquid chromatography-quadrupole-Orbitrap high resolution mass spectrometry
- UHPLC-MS
-
Ultra-High performance liquid chromatography-mass spectrometry
- UV
-
Ultraviolet.
Abbreviations
1 Introduction
Viticis Fructus (also known as Manjingzi in China) is the dry ripe fruit of Vitex rotundifolia L. (synonyms of Vitex trifolia var. simplicifolia Cham.) and Vitex trifolia L. of the Lamiaceae family. It has been used as traditional Chinese medicine in China for more than 2000 years. It is also called “Manjingshi”, “Jingzi”, “Wanjingzi”, “Baibeifeng” and so on. Viticis Fructus is initially mentioned in Shennong Bencao Jing as “Manjingshi”, which is listed among the top grade (Zhang et al., 2018). It is recorded in ancient books for treating colds, headaches, gingivitis, dizziness, ophthalmalgia and so on.
Viticis Fructus mainly contains terpenoids, flavonoids, phenolic acids and other chemical components. It is classified as the genus Vitex. Many plants in the genus Vitex have been widely studied and have multifarious pharmacological activities (Auniq et al., 2019; Hobbs, 1991; Jangwan et al., 2013). Viticis Fructus has antipyretic, analgesic, antioxidant, anti-inflammatory, antitumor, blood pressure lowering and other activities. It is not only used for the treatment of headaches, swelling and aching of gum, but also for the prevention and treatment of trigeminal neuralgia (Wen et al., 2020), atherosclerosis (Kim et al., 2020), tumors (Liu et al., 2019), premenstrual syndrome (PMS) (Ye, 2010), inflammation (Fang et al., 2019) and other diseases. Therefore, the extracts and compounds of Viticis Fructus have far-reaching medicinal value and deserve further research and development.
The research hotspots of Viticis Fructus can be explored through the frequency analysis and visual display of keywords. “V. rotundifolia”, “apoptosis”, “casticin” and “Viticis Fructus” are keywords with high frequency and the average publication year of Viticis Fructus keywords is shown in Fig. 1 (the redder the color, the higher the popularity). In botany, Vitex rotundifolia (V. rotundifolia) fruits are more frequently studied as Viticis Fructus. The family of Viticis Fructus has been controversial in recent years from Verbenaceae to Lamiaceae family. In phytochemistry, the main research focus on the flavonoids, terpenoids and iridoids of Viticis Fructus. Casticin is the most studied compound. In pharmacological effects, the main research direction of Viticis Fructus focuses on exploring pharmacological mechanisms by “apoptosis”, “proliferation”, “cells”, “in vitro” and other hot keywords. The pharmacological activity mainly focuses on anti-tumor research. The most of the current pharmacological experiments remain at the in vitro cell level via “cells” and “in-vitro”. In addition to the above points of view, many studies researched the leaves of V. rotundifolia or Vitex trifolia (V. trifolia). Viticis Fructus has been listed as the third level of Chinese wild traditional Chinese medicine species for crucial protection. The comprehensive development and utilization of non-medicinal parts can avoid resource waste and is conducive to the sustainable development of traditional Chinese medicines (TCMs).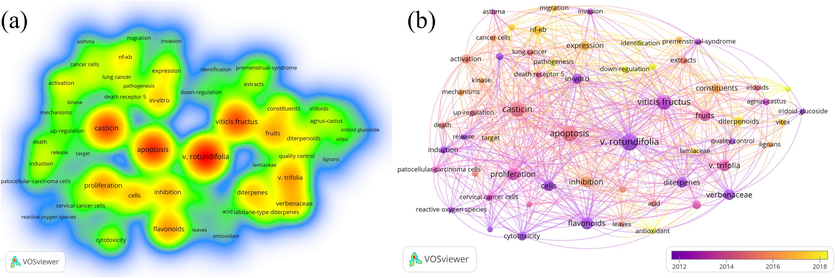
The hot word density view and average publication year of Viticis Fructus keywords.
The traditional pharmacological actions of Viticis Fructus have been supported by some modern pharmacology with the deepening of the research on Viticis Fructus, whose research direction is also gradually enriched. The plant morphology, phytochemistry and pharmacological effects have been preliminarily reviewed (Meng et al., 2023; Yan et al., 2023). However, the current summary of the chemical constituents is not comprehensive and the relevant pharmacological mechanisms are not clear enough. Thus, this review generalizes the advances on variation of the origin plant of Viticis Fructus, isolation and identification approaches of chemical constituents, counterfeit identification, toxicity studies, pharmacokinetics, patent information, clinical applications, different processing methods, changes in constituents after processing, the indications scope of various processed products and so on. In addition, the repetitive modules such as traditional applications, phytochemistry, pharmacology and so on were refined and supplemented in more detail. The corresponding chemical composition structure, pharmacological mechanism and hotspot map of Viticis Fructus research were drawn to further utilization of Viticis Fructus. A well-rounded understanding will set the basis for further studies and the development of Viticis Fructus.
2 Method
Relevant information was obtained from PubMed, ScienceDirect, Web of science, Embase, Scopus, Google Scholar, China National Knowledge Infrastructure (CNKI), WeiPu, Chinese ancient books and DuXiu academic search. The selected literature was screened by publication date (from the years 1935–––2024) and language (Chinese and English). The database was searched based on some synonyms (from https://powo.science.kew.org), such as “Viticis Fructus”, “seed of Vitex trifolia L.”, “the fruits of Vitex trifolia L.”, “the fruits of Vitex trifolia L. var. simplicifolia Cham.”, “Shrub chaste tree fruits” and “the fruits of Vitex rotundifolia”. Additionally, some data were collected from Pharmacopoeia of the People's Republic of China (ChP), Chinese classic books and official websites. The characteristics of the plant were collected from botanical database (https://www.kew.org/science, https://powo.science.kew.org, https://www.iplant.cn/foc). The traditional prescription of Viticis Fructus came from Yaozhiwang (https://www.yaozh.com). Its patent information came from CNKI, Yaozhiwang (https://www.yaozh.com) and Baiteng (https://www.baiten.cn/). CAS SciFinder (https://scifinder-n.cas.org/), PubChem (https://pubchem.ncbi.nlm.nih.gov/) and ChemSpider (https://www.chemspider.com/) were used to check the structure of the compounds in the literature. Some images were from freepik (https://www.freepik.com/).
3 Botany
3.1 Botanical origin
Viticis Fructus is considered to have two origins, namely V. rotundifolia and V. trifolia. It was found that the fruits of Vitex negundo var. cannabifolia were misused as Viticis Fructus by consulting relevant data (Zhou and Jin, 2001). The origin of Viticis Fructus has also been changing (Table S1). V. trifolia and V. negundo var. cannabifolia were not distinguished before the Northern and Southern Dynasties. It was written in Guangzhi and Guangya as “V. negundo var. cannabifolia was V. trifolia”. V. trifolia and V. negundo var. cannabifolia were distinguished by Tao Hongjing for the first time in the Bencao Jingji zhu after the Northern and Southern Dynasties (Tao, 1994). He recorded that the fruits of V. negundo var. cannabifolia were larger than Viticis Fructus. However, the statement was contrary to the present findings. In the Tang and Song Dynasties, there were two main changes. (1) In Xinxiu Bencao, the plant morphology was described in detail. It was consistent with the current statement that Viticis Fructus were larger than the fruits of V. negundo var. cannabifolia (Su, 1981). (2) In the Song Dynasty, the main difference between V. trifolia and V. negundo var. cannabifolia was whether it was a trailing plant, which was recorded in Bencao Yanyi (Kou, 1985). In the Ming Dynasty, it was listed and recorded in Bencao Gangmu: “Its branches were small and weak as V. trifolia, so it was called manjing (trailing plant)”. It could be found from the picture attached to the book that it was ternate compound leaves, which was in line with the characteristics of V. trifolia (Li, 2011). The mainstream view has been that V. rotundifolia and V. trifolia were the origin plants of Viticis Fructus since 1961 (Zhang et al., 2019).
Nowadays, the family and variety of Viticis Fructus are widely discussed. Viticis Fructus is stipulated as Verbenaceae family by the ChP (Chinese Pharmacopoeia, 2020) and Flora of China (https://www.iplant.cn/foc) according to relevant information. However, it is classified as Lamiaceae family by some botanical-related websites, such as Plants of the World Online database (https://powo.science.kew.org). The family of Viticis Fructus is uneven in the literature. In addition, the investigation on whether V. rotundifolia belongs to one of the varieties of V. trifolia also needs to be further conducted. Now V. rotundifolia is considered to be one of the varieties of V. trifolia in some standardized works of taxonomy and pharmacy in China, such as ChP and Flora of China. The Flora of China (English edition), Flora of Australia and Royal Botanic Gardens of the United Kingdom have listed V. rotundifolia as an independent species of Vitex (Sun, 2018).
3.2 Botanical taxonomy
Viticis Fructus belongs to the Lamiaceae family, mainly grown in Borneo, China, Thailand and Western Australia (https://powo.science.kew.org/). According to the Flora of China (English edition), there are three varieties of V. trifolia, namely Vitex trifolia var. taihangensis, Vitex trifolia var. trifolia and Vitex trifolia var. subtrisecta (Table S2). However, V. rotundifolia was also considered to be one of the varieties of V. trifolia in the ChP, called Vitex trifolia var. simplicifolia Cham.
3.3 Botanical description
The two origin plants of Viticis Fructus are clearly distinguished in the plant morphology. The leaves of V. trifolia are simple leaves and the leaves of V. rotundifolia are ternate compound leaves, but their fruits are very similar in morphology (Yang et al., 2023). It is spherical with a diameter of 4–6 mm and has four chambers each with one seed. The surface is gray-black or black-brown with gray powder cream-like fuzziness and has 4 longitudinal shallow ditches. Its base has gray-white calyx and short fruit stalk.
3.4 Botanical distribution
V. Rotundifolia grows in open sandy areas, usually near the sea. V. Rotundifolia is distributed in the north-central and southeast of China, such as Jiangxi and Shandong province. Besides, V. Rotundifolia is widely cultivated in Borneo, India, Japan, Korea, Vanuatu, Vietnam, Western Australia and other places. V. Rotundifolia is different from V. trifolia, which grows in plains, river beaches, sparse forests and villages. Furthermore, V. Trifolia spreads over the north-central, south-central and southeast of China. V. Trifolia is widely cultivated in Afghanistan, Algeria, Assam, Bangladesh, Bismarck Archipelago and so on (https://powo.science.kew.org/). They are all shrubs or trees that grow mainly in moist tropical communities
The origin plants of Viticis Fructus are currently unified as V. rotundifolia and V. trifolia. But the family of Viticis Fructus and varieties of V. trifolia should be unified. In addition, although the plant morphology of the two origin plants is easy to distinguish, their fruits are very similar in morphology.
4 Historical records
4.1 Traditional medicinal applications
Viticis Fructus is pungent, bitter taste with a cold character, which enters the bladder, liver and stomach meridian (Chinese Pharmacopoeia, 2020). It was used for treating colds, headaches, gingivitis, dizziness, ophthalmalgia, etc. Recent studies have discovered that Viticis Fructus exerted preventive and therapeutic effects on various diseases, including trigeminal neuralgia (Wen et al., 2020), senile cataracts (Sun and Yang, 2010), arthritis (Chu et al., 2020) and inflammation (Fang et al., 2019). These pharmacological activities were matched with headaches, dizziness, blurred vision, rheumatism and anti-inflammation. In addition, Viticis Fructus had an effect on relieving or treating diseases, such as supraorbital neuralgia (Li, 1998) and neurovascular headache (Hu and Yang, 2016) in clinical.
Viticis Fructus has been employed in clinical practice for more than 2000 years as TCM. The main traditional effects recorded in different books were to dispel wind-heat (treating fever, chills, cough, thirst and other wind-heat syndromes), treat rheumatism, headache and promote hair growth and so forth (Table S3). In the Qin-Han Dynasties, Viticis Fructus was first recorded in Shennong Bencao Jing and listed as one of the top grades. It was employed to treat rheumatism, tapeworm parasites and had healthy effects of improving eyesight, firming teeth and anti-aging (Zhang et al., 2018). In the Southern and Northern Dynasties, it was applied to treat headaches, intracranial tinnitus and benefit qi (benefiting vital energy) as recorded in Mingyi Bielu (Tao, 1986). During the Five Dynasties and Ten States period, it had therapeutic effects on eye swelling, itching and ulcerous eyelid margin and promoted hair growth. In the Tang Dynasty, its efficacy had been clarified to relieve the symptoms of headaches and promote hair growth (Sun, 1982; Zhen, 1983). In the Song Dynasty, the usage of Viticis Fructus in the treatments of head and face wind (symptoms of head and face sweating, headaches, dizziness, etc) was more frequent (Wang, 1958b). The treatment of intermittent headaches by Viticis Fructus was introduced in Danxi Xinfa in the Yuan Dynasty. The description of its efficacy in Shennong Bencao Jing was reaffirmed in Bencao Gangmu (Li, 2011). In the Qing Dynasty, the functions of Viticis Fructus were emphasized by many Chinese ancient books, such as dispelling cold-dampness syndrome (treating rheumatism), removing headaches and improving visual acuity. Viticis Fructus was recorded in Depei Bencao, which could dispel cold-dampness, cure headaches, relieve eye pain and treat damp arthralgia, intracranial tinnitus and toothaches. Additionally, it was devoted to dispelling wind, treating solar wind headaches (migraine), vertigo and eye pain according to Bencao Shugou Yuan (Yang, 1958).
Furthermore, Viticis Fructus was also used in conjunction with other herbs to exert curative effects (Table S4). There are two classic prescriptions for Viticis Fructus, which are Qiang-Huo-Sheng-Shi Decoction and Qing-Shang-Juan-Tong Decoction. Qiang-Huo-Sheng-Shi Decoction derived from Neiwai Shangbian Huolun of Li DongYuan. It was combined with Notopterygii Rhizoma et Radix, Chuanxiong Rhizoma and so on (Li, 1959; Yan et al., 2022). Its effect was to dispel wind, eliminate dampness and relieve pain (Hu et al., 2022). It was commonly used to treat rheumatoid arthritis, bone hyperplasia, ankylosing spondylitis, etc. Notopterygii Rhizoma et Radix and Angelicae Pubescentis Radix in the prescription could dispel wind-dampness and dredge joints. Their combination had favorable effects in treating rheumatism around the body and relieving arthralgia, which were sovereign drugs (playing a major role in the treatment of main syndromes or main symptoms in prescriptions). Saposhnikoviae Radix cured pain and Chuanxiong Rhizoma could not only evacuate the wind evil around the body, but also could promote blood and qi circulation to alleviate body pain. They were used to minister drugs (assisting the sovereign drugs to cure the main symptoms) and helped the sovereign drugs to disperse evil and relieve pain. Ligustici Rhizoma et Radix evacuated wind-dampness and relieved headaches, which was the assistant drug (assisting the sovereign and minister drugs to treat concurrent syndromes and secondary symptoms). Glycyrrhizae Radix et Rhizoma mitigated the nature of various herbs and reconciled medicines as assistant drugs. The compatibility of multiple herbs could dispel wind-dampness and relieve pain (Zhang et al., 2023). Viticis Fructus was an assistant drug in the prescription and mainly played the role of dispelling wind and relieving pain. Pharmacological research has supported its therapeutic roles (Chu et al., 2020; Li et al., 2020b). The other is the Qing-Shang-Juan-Tong Decoction recorded in Shoushi Baoyuan of Gong TingXian. The prescription included Radix Angelicae Sinensis, Radix Angelicae Dahuricae, Viticis Fructus, etc (Gong, 1999). It was mainly used to treat intractable pain and trigeminal neuralgia. This effect had also been supported by pharmacological studies (Wen et al., 2020; Yu et al., 2021).
Viticis Fructus could cure colds, headaches, migraines and neuralgia in Korea (Kim, et al., 2012). In India, it could be employed to improve symptoms of amenorrhoea, liver disease, rheumatic pain and other disorders (Meng et al., 2023). In Japan, it had therapeutic effects on colds, headaches, migraine and eye pain (Yan et al., 2023). Furthermore, many countries not only used the fruits of V. trifolia and V. rotundifolia to treat diseases, but also their other parts could be utilized for various illnesses. For example, Samoans had applied V. trifolia to relieve sprains and rheumatic pain. In Tonga, its efficacy had been clarified to cure oral infections and inflammation. In Papua New Guinea and New Caledonia, its stems and leaves could remedy dysentery (Kamal et al., 2022). In India, the flowers and leaves of V. trifolia were employed for fever and alopecia, respectively (Yan et al., 2023). In European herbal medicine, V. rotundifolia was used to relieve various diseases associated with women (Azizul et al. 2022). It is listed as a protected plant in China and Japan, so the development and utilization of multiple parts can avoid waste of resources and is conducive to sustainable development.
4.2 Processing
There are mainly two specifications for clinical use according to the 2020 edition of ChP, namely crude and stir-fry processed Viticis Fructus. The processing not only could remove non-medicinal parts, but also could moderate the nature of Viticis Fructus (Wang et al., 2010). Various processing methods were recorded in ancient books, such as crude, stir-frying (micro-fried, stir-frying coke, stir-frying char, stir-frying liquor) and steaming with liquor (Fig. S1, Table S5). The crude Viticis Fructus was first recorded with “the persistent sepal removal” both in ShengJi ZongLu and Taiping Huimin Heji Jufang in Song Dynasty (Hejiju, 1985; Zhao, 1982). Viticis Fructus had been emphasized to the removal of non-medicinal parts and crushed before use in many works. Its stir-fry processed products had micro-fried, stir-frying char and so forth. The two methods of Viticis Fructus recorded in Boji Fang were “washed Viticis Fructus, baked with mild fire then crushed” (Wang, 1958a). The other was “washed and then fried”. According to the records of Danxi Xingfa, it was requested that should be “fried to black” (Zhu, 1956). “It was crushed, washed with liquor, fried and decocted” in Yizong Cuiyan (Luo, 1982). Bencao Tongxuan recorded that “the persistent calyx removal, fried with liquor and crushed” (Li, 2015). Micro-fried Viticis Fructus was the most widely used among them. Furthermore, Viticis Fructus also had a liquor steaming processing method. Traditional Chinese medicine believed that the quality of Viticis Fructus was light and the smell was mild. It moved upward to treat head and face wind. The liquor stir-frying method had contributed to treating headaches by Viticis Fructus (Yin et al., 2019). Liquor steaming was first recorded in Leigong Paozhi Lun (Lei, 1986). Viticis Fructus should be removed the persistent calyx and pedicle. After soaking in liquor for ten days, it was steamed for several hours and dried in the sun. Besides, it could also be prepared by boiling method, the steaming and frying method. It was recorded in Taiping Shenghui Fang that the ratio of medicine to liquor is 1:5. It was boiled in the liquor and dried in the sun (Wang, 1958b). However, the boiling method and the steaming and frying method were less used.
Viticis Fructus and its stir-fried products are commonly used in clinics. Their chemical constituents also changed during processing. First, whether the crude and fried products are crushed is particularly critical. The content of extract significantly increased after crushing compared with uncrushed Viticis Fructus (Wang et al., 2017a). It was explained that the scientific nature of the use after crushing was emphasized in ancient books. Secondly, the volatile oil and flavonoids in Viticis Fructus changed differently during processing. For example, the total volatile oil content in Viticis Fructus decreased after thermal processing (Wang et al., 2017b). However, the flavonoids with high melting points were not easily destroyed (Wang et al., 2010). Other studies had also displayed that the content of total flavonoids was stir-frying coke > stir-frying charcoal > slightly stir-frying = crude product (Guo et al., 2005; Zhang et al., 2003). Furthermore, the casticin content of each processed product was determined, crude Viticis Fructus < stir-frying Viticis Fructus < stir-frying Viticis Fructus with liquor < baking Viticis Fructus with liquor (Xu et al., 2020).
The pharmacological effects of processed products showed specific differences. Crude Viticis Fructus was often used for treating wind-heat headaches, red eyes, swelling and pains, while stir-fried Viticis Fructus was chiefly used for the treatment of deafness, rheumatic arthralgia and migraine (Wang et al., 2017b). The crushed crude product was suitable for the evacuation of wind-heat. The mixed frying of 10 % yellow rice liquor and Viticis Fructus was applied for analgesia (Jin et al., 2000). It was advisable to use stir-fried carbon or stir-frying Viticis Fructus with liquor to reduce blood pressure (Diao (2018)). However, there were some bifurcations in analgesic research. Some scholars regarded the analgesic effect of fried products were more potent than that of crude products. The intensity of the analgesic effect was as follows: stir-frying coke > micro-fried products > stir-frying char > crude products (Sun et al., 1997). Other scholars believed that the liquor products were more effective than the crude products, because liquor and Viticis Fructus had compatible medicinal properties (Jin et al., 2000). It had been advocated that the crude products analgesic effect was more vital than fried products (Liu, 2005). Another point of view was that the analgesic effect of crude Viticis Fructus is strong. Its analgesic effect was reduced after stir-frying and processing with liquor does not improve its analgesic effect. Crude Viticis Fructus should be used for analgesic (Gong and Wang, 2012).
Viticis Fructus has a long history as TCM application. It can cure headaches, eye diseases, head and face wind, rheumatism, intracranial tinnitus and promote hair growth according to ancient books. Furthermore, the processing products of Viticis Fructus are mainly divided into crude, stir-frying and liquor products according to the records of ancient books. However, the primary clinical applications at present are crude Viticis Fructus and micro-fried Viticis Fructus, and more attention should be paid to other processed products. Because the types and contents of flavonoids, volatile oils and other components have been altered during the processing process, diverse processed products are appropriate for different ailments. Therefore, it is paramount to select the best processed products for disease treatment.
5 Phytochemistry
The chemical composition database of Viticis Fructus was systematically and comprehensively established and 324 compounds were isolated and identified (Table 1). It contains terpenoids, flavonoids, phenolic acids and others, of which terpenoids and flavonoids as the main compounds accounting for 57.7 % and 13.8 % (Fig. S2a), respectively. In addition, there were some differences in the 324 compounds isolated and identified in the two origin plants, of which 155 compounds could be isolated and identified in both origin plants (Fig. S2b).
Number
Compounds
Molecular formula
Molecular weight
CAS No.
Plant
Identification method
References
Terpenoids
1
viterotulin C
C24H38O6
422.27
N/A
V. rotundifolia/V. trifolia
NMR
(Fang et al., 2019)
2
vitexilactone D
C22H34O5
378.24
N/A
V. rotundifolia
NMR
(Fang et al., 2019)
3
vitexilactone
C22H34O5
378.24
61263–49-8
V. rotundifolia/V. trifolia
NMR
(Fang et al., 2019)
4
rotundifuran
C22H34O4
362.50
50656–65-0
V. rotundifolia/V. trifolia
NMR
(Fang et al., 2019)
5
vitetrifolin B
C22H34O4
362.50
329763–47-5
V. rotundifolia/V. trifolia
NMR
(Fang et al., 2019)
6
viterotulin B
C22H34O5
378.24
1469986–05-7
V. rotundifolia
NMR
(Fang et al., 2019)
7
vitetrifolin D
C24H38O5
406.60
351427–18-4
V. rotundifolia/V. trifolia
NMR
(Fang et al., 2019)
8
(rel 5S,6R,8R,9R,10S)-6-acetoxy-9-hydroxy-13(14)-labden-16,15-olide
C22H34O5
378.24
N/A
V. rotundifolia/V. trifolia
UV, NMR, CD, MS
(Li et al., 2005b)
9
viteagnusin I
C22H34O6
394.50
1345994–66-2
V. rotundifolia
UV, NMR, CD, MS
(Lee et al., 2013)
10
vitetrifolin E
C22H36O4
364.50
372967–06-1
V. rotundifolia/V. trifolia
UV, NMR, CD, MS
(Lee et al., 2013)
11
vitetrifolin F
C22H36O4
364.50
372967–07-2
V. rotundifolia/V. trifolia
UV, NMR, CD, MS
(Lee et al., 2013)
Number
Compounds
Molecular formula
Molecular weight
CAS No.
Plant
Identification method
References
12
vitetrifolin H
C22H34O4
362.25
1202522–21-1
V. rotundifolia/V. trifolia
HRESIMS, NMR
(Djimabi et al., 2021)
13
vitetrifolin G
C20H32O2
304.50
372967–08-3
V. rotundifolia
UV, NMR, CD, MS
(Lee et al., 2013)
14
9,13-epoxy-16-nor-labda-13E-en-15-al
C19H30O2
290.22
180628–06-2
V. rotundifolia/V. trifolia
UV, NMR, CD, MS
(Lee et al., 2013)
15
13-epi-2-oxokolavelool
C20H32O2
304.24
221466–41-7
V. rotundifolia
UV, NMR, CD, MS
(Lee et al., 2013)
16
isolophanthin A
C20H30O2
302.50
1370511–54-8
V. rotundifolia
UV, NMR, CD, MS
(Lee et al., 2013)
17
vitedoin B
C19H30O4
322.40
819861–42-2
V. rotundifolia
UV, NMR, CD, MS
(Lee et al., 2013)
18
viteagnusin F
C23H38O7
426.50
1206489–93-1
V. rotundifolia
UV, NMR, CD, MS
(Lee et al., 2013)
19
viteagnusin G
C23H38O7
426.50
1206489–94-2
V. rotundifolia
UV, NMR, CD, MS
(Lee et al., 2013)
20
viterotulin A
C20H32O3
320.24
1423125–18-1
V. rotundifolia
UV, NMR, CD, MS
(Lee et al., 2013)
21
(rel 3S,5S,8R,9R,10S)-3,9-dihydroxy-13(14)-labden-16,15-olide
C20H32O4
336.23
1467744–96-2
V. rotundifolia
UV, NMR, CD, MS
(Lee et al., 2013)
22
viterotulin D
C24H38O6
422.27
N/A
V. trifolia
HRESIMS, NMR
(Djimabi et al., 2021)
Number
Compounds
Molecular formula
Molecular weight
CAS No.
Plant
Identification method
References
23
15,16-epoxy-9-hydroxylabda-13(16),14-diene
C20H32O2
304.24
N/A
V. trifolia
HRESIMS, NMR
(Djimabi et al., 2021)
24
prevetexilactone
C22H34O5
378.24
2730961–28-9
V. trifolia
HRESIMS, NMR
(Djimabi et al., 2021)
25
trisnor-γ-lactone
C19H30O4
322.21
N/A
V. rotundifolia/V. trifolia
HRESIMS, NMR
(Djimabi et al., 2021)
26
vitexifolin D
C19H30O4
322.40
351427–21-9
V. rotundifolia/V. trifolia
HRESIMS, NMR
(Djimabi et al., 2021)
27
13-hydroxy-5(10),14-halimadien-6-one
C20H32O2
304.24
N/A
V. trifolia
HRESIMS, NMR
(Djimabi et al., 2021)
28
abietatrien-3β-ol
C20H30O
286.23
N/A
V. rotundifolia/V. trifolia
HRESIMS, NMR
(Djimabi et al., 2021)
29
3β-acetoxyabieta-8,11,13-trien-12-ol
C22H32O3
344.24
N/A
V. trifolia
HRESIMS, NMR
(Djimabi et al., 2021)
30
helipterol
C20H34O
290.26
120852–66-6
V. trifolia
HRESIMS, NMR
(Djimabi et al., 2021)
31
vitetrifolin I
C20H34O3
322.25
1202522–22-2
V. trifolia
MS, NMR, IR
(Wu et al., 2009)
32
6-acetoxy-9-hydroxy-13(14)-labdane-16,15-olide
C22H34O5
378.24
329976–53-6
V. trifolia
MS, NMR, IR
(Wu et al., 2009)
33
previtexilactone
C22H34O5
378.50
106894–28-4
V. trifolia
MS, NMR, IR
(Wu et al., 2009)
Number
Compounds
Molecular formula
Molecular weight
CAS No.
Plant
Identification method
References
34
6-acetoxy-9,13;15,16-diepoxy-15-methoxylabdane
C23H38O5
394.27
248925–24-8
V. trifolia
MS, NMR, IR
(Wu et al., 2009)
35
vitetrifolin A
C20H32O3
320.24
329763–38-4
V. trifolia
X-ray crystallographic analysis, NMR, MS
(Ono et al., 2000)
36
vitetrifolin C
C22H32O4
360.23
329763–48-6
V. trifolia
NMR, MS
(Ono et al., 2000)
37
dihydrosolidagenone
C20H30O3
318.22
N/A
V. trifolia
NMR, MS
(Ono et al., 2000)
38
vitrifolin B
C22H36O5
380.26
1681015–93-9
V. trifolia
HRESIMS, IR, NMR
(Wang et al., 2014)
39
vitexlactam A
C22H35NO4
377.26
459167–05-6
V. trifolia
HRESIMS, IR, NMR
(Wang et al., 2014)
40
vitextrifolin A
C24H40O6
424.28
1418297–87-6
V. trifolia
COSY-NMR, HRESIMS, IR, NOESY-NMR
(Zheng et al., 2013)
41
vitextrifolin B
C24H40O6
424.28
1418297–88-7
V. trifolia
COSY-NMR, HRESIMS, IR, NOESY-NMR
(Zheng et al., 2013)
42
vitextrifolin C
C22H32O4
360.23
1418297–89-8
V. trifolia
COSY-NMR, HRESIMS, IR, NOESY-NMR
(Zheng et al., 2013)
43
vitextrifolin D
C20H30O3
318.22
1418297–90-1
V. trifolia
COSY-NMR, HRESIMS, IR, NOESY-NMR
(Zheng et al., 2013)
Number
Compounds
Molecular formula
Molecular weight
CAS No.
Plant
Identification method
References
44
vitextrifolin E
C20H30O3
318.22
1418297–91-2
V. trifolia
COSY-NMR, HRESIMS, IR, NOESY-NMR
(Zheng et al., 2013)
45
vitextrifolin F
C20H32O4
336.23
1418297–92-3
V. trifolia
COSY-NMR, HRESIMS, IR, NOESY-NMR
(Zheng et al., 2013)
46
vitextrifolin G
C20H30O3
318.22
1418297–93-4
V. trifolia
COSY-NMR, HRESIMS, IR, NOESY-NMR
(Zheng et al., 2013)
47
isoambreinolide
C17H28O2
264.21
18676–08-9
V. trifolia
HRFABMS, IR, UV, NMR
(Kiuchi et al., 2004)
48
(3S,5S,6S,8R,9R,10S)-3,6,9-trihydroxy-13(14) labdean-16,15-olide 3-O-β-D-glucopyranoside
C26H42O10
514.31
N/A
V. trifolia
HRESIMS, IR, UV, NMR
(Bao et al., 2018)
49
viteagnuside A
C26H42O9
498.60
1401711–97-4
V. rotundifolia/V. trifolia
HRESIMS, IR, UV, NMR
(Bao et al., 2018)
Number
Compounds
Molecular formula
Molecular weight
CAS No.
Plant
Identification method
References
50
vitrifolin A
C19H28O5
336.42
1469746–48-2
V. rotundifolia
HRESIMS, IR, NMR
(Zhang et al., 2013)
51
ent-2-oxo-15,16,19-trihydroxy-pimar-8 (14)-ene
C20H32O4
336.50
N/A
V. rotundifolia
MS, NMR
(Chen et al., 2018)
52
leucasin A
C28H46O11
558.30
1423779–98-9
V. rotundifolia
HRESIMS, IR, UV, NMR
(Zhao et al., 2017)
53
leucasin B
C28H46O11
558.30
1423779–99-0
V. rotundifolia
HRESIMS, IR, UV, NMR
(Zhao et al., 2017)
54
(rel 5S,6R,8R,9R,10S,13R,15R)-6-acetoxy-9,13;15,16-diepoxy-15-methoxylabdane
C23H38O5
394.27
N/A
V. rotundifolia
NMR, X-ray crystallographic analysis
(Ono et al., 1999)
55
(rel 5S,6R,8R,9R,10S,13R,15S)-6-acetoxy-9,13;15,16-diepoxy-15-methoxylabdane
C23H38O5
394.27
N/A
V. rotundifolia
NMR, X-ray crystallographic analysis
(Ono et al., 1999)
Number
Compounds
Molecular formula
Molecular weight
CAS No.
Plant
Identification method
References
56
(rel 5S,6R,8R,9R,10S,13S,15S)-6-acetoxy-9,13;15,16-diepoxy-15-methoxylabdane
C23H38O5
394.27
N/A
V. rotundifolia
NMR, X-ray crystallographic analysis
(Ono et al., 1999)
57
(rel 5S,6R,8R,9R,10S,13S,15R)-6-acetoxy-9,13;15,16-diepoxy-15-methoxylabdane
C23H38O5
394.27
N/A
V. rotundifolia
NMR, X-ray crystallographic analysis
(Ono et al., 1999)
58
(rel 5S,6R,8R,9R,10S,13S,15S,16R)-6-acetoxy-9,13;15–16-diepoxy-15,16-methoxylabdane
C24H40O6
424.28
N/A
V. rotundifolia
NMR, X-ray crystallographic analysis
(Ono et al., 1999)
Number
Compounds
Molecular formula
Molecular weight
CAS No.
Plant
Identification method
References
59
(rel 5S,6R,8R,9R,10S,13S,15R,16R)-6-acetoxy-9,13;15–16-diepoxy-15,16-dimethoxylabdane
C24H40O6
424.28
N/A
V. rotundifolia
NMR, X-ray crystallographic analysis
(Ono et al., 1999)
60
(rel 5S,6R,8R,9R,10S,13S,15S,16S)-6-acetoxy-9,13;15–16-diepoxy-15,16-dimethoxylabdane
C24H40O6
424.28
N/A
V. rotundifolia
NMR, X-ray crystallographic analysis
(Ono et al., 1999)
Number
Compounds
Molecular formula
Molecular weight
CAS No.
Plant
Identification method
References
61
(rel 5S,6R,8R,9R,10S,13S,15R,16S)-6-acetoxy-9,13;15–16-diepoxy-15,16-dimethoxylabdane
C24H40O6
424.28
N/A
V. rotundifolia
NMR, X-ray crystallographic analysis
(Ono et al., 1999)
62
ferruginol
C20H30O
286.50
514–62-5
V. rotundifolia
NMR, X-ray crystallographic analysis
(Ono et al., 1999)
63
vitexifolin A
C20H34O
290.50
351427–17-3
V. rotundifolia
HRFABMS, NMR
(Ono et al., 2002)
64
vitexifolin B
C20H36O3
324.50
2730967–87-8
V. rotundifolia
HRFABMS, NMR
(Ono et al., 2002)
65
vitexifolin C
C20H28O
284.40
351427–23-1
V. rotundifolia
HRFABMS, NMR
(Ono et al., 2002)
66
vitexifolin E
C20H32O4
336.50
351427–22-0
V. rotundifolia
HRFABMS, NMR
(Ono et al., 2002)
67
manool
C20H34O
290.50
596–85-0
V. rotundifolia
HRFABMS, NMR
(Ono et al., 2002)
68
vitexfolin A
C25H28O11
504.50
N/A
V. rotundifolia
MS, NMR, UV
(Okuyama and Yamazaki, 1998)
69
vitexfolin C
C23H26O10
462.40
N/A
V. rotundifolia
MS, NMR, UV
(Okuyama and Yamazaki, 1998)
Number
Compounds
Molecular formula
Molecular weight
CAS No.
Plant
Identification method
References
70
(rel 5S,6R,8R,9R,10S)-6-acetoxy-9-hydroxy-13(14)-labden-16,15-olide
C22H34O5
378.24
N/A
V. rotundifolia
MS, NMR
(Ono et al., 2001)
71
(rel 5S,6S,8R,9R,10S)-6-acetoxy-9-hydroxy-13(14)-labden-16,15-olide
C22H34O5
378.24
N/A
V. rotundifolia
MS, NMR
(Ono et al., 2001)
72
(rel 5S,6R,8R,9R,10S)-6-acetoxy-9-hydroxy-15-methoxy-13(14)-labden-16,15-olide
C23H36O6
408.25
N/A
V. rotundifolia
MS, NMR
(Ono et al., 2001)
Number
Compounds
Molecular formula
Molecular weight
CAS No.
Plant
Identification method
References
73
(rel 5S,6R,8R,9R,10S,13S,16S)-6-acetoxy-9,13-epoxy-16-methoxy-labdan-15,16-olide
C23H36O6
408.25
N/A
V. rotundifolia
MS, NMR
(Ono et al., 2001)
74
(rel 5S,6R,8R,9R,10S,13R,16S)-6-acetoxy-9,13-epoxy-16-methoxy-labdan-15,16-olide
C23H36O6
408.25
N/A
V. rotundifolia
MS, NMR
(Ono et al., 2001)
75
(rel 5S,6R,8R,9R,10S,13S)-6-acetoxy-9,13-epoxy-15-methoxy-labdan-16,15-olide
C23H36O6
408.25
N/A
V. rotundifolia
MS, NMR
(Ono et al., 2001)
Number
Compounds
Molecular formula
Molecular weight
CAS No.
Plant
Identification method
References
76
(rel 5S,6R,8R,9R,10S,13R)-6-acetoxy-9,13-epoxy-15-methoxy-labdan-16,15-olide
C23H36O6
408.25
N/A
V. rotundifolia
MS, NMR
(Ono et al., 2001)
77
(rel 5S,8R,9R,10S,13S,15S,16R)-9,13;15,16-Diepoxy-15,16-dimethoxy-labdane
C24H40O6
424.28
N/A
V. rotundifolia
MS, NMR
(Ono et al., 2001)
78
(rel 5S,8R,9R,10S,13S,15R,16S)-9,13;15,16-Diepoxy-15,16-dimethoxy-labdane
C24H40O6
424.28
N/A
V. rotundifolia
MS, NMR
(Ono et al., 2001)
Number
Compounds
Molecular formula
Molecular weight
CAS No.
Plant
Identification method
References
79
(rel 5S,8R,9R,10S,13S,15R,16R)-9,13;15,16-Diepoxy-15,16-dimethoxylabdane
C24H40O6
424.28
N/A
V. rotundifolia
MS, NMR
(Ono et al., 2001)
80
negundol
C22H34O6
394.24
1421609–79-1
V. trifolia
MS, NMR, IR, UV
(Zhu, 2013)
81
deacetyl vitexilactone
C20H32O4
336.23
885069–79-4
V. trifolia
MS, NMR, IR, UV
(Zhu, 2013)
82
6β-acetoxyl-9α,16-dihydroxy-13(14)-labden-15,16-olide
C22H34O6
394.24
1345994–66-2
V. trifolia
MS, NMR, IR, UV
(Zhu, 2013)
83
vitexilactone B
C22H34O5
378.24
1276541–12-8
V. rotundifolia
NMR, IR, HRESIMS
(Yin, 2015)
84
viterofolin A
C20H34O3
322.25
N/A
V. rotundifolia
NMR, IR, HRESIMS
(Yin, 2015)
85
viterofolin B
C20H34O3
322.25
N/A
V. rotundifolia
NMR, IR, HRESIMS
(Oh et al., 2024)
86
viterofolin C
C20H32O2
304.24
N/A
V. rotundifolia
NMR, IR, HRESIMS
(Yin, 2015)
87
viterofolin D
C20H34O2
306.26
N/A
V. rotundifolia
NMR, IR, HRESIMS
(Yin, 2015)
88
viterofolin E
C20H34O2
306.26
N/A
V. rotundifolia
NMR, IR, HRESIMS
(Yin, 2015)
89
viterofolin F
C21H34O2
318.26
N/A
V. rotundifolia
NMR, IR, HRESIMS
(Yin, 2015)
Number
Compounds
Molecular formula
Molecular weight
CAS No.
Plant
Identification method
References
90
viterofolin G
C21H34O2
318.26
N/A
V. rotundifolia
NMR, IR, HRESIMS
(Yin, 2015)
91
viterofolin H
C20H34O2
306.26
N/A
V. rotundifolia
NMR, IR, HRESIMS
(Yin, 2015)
92
viterofolin I
C24H38O6
422.27
N/A
V. rotundifolia
NMR, IR, HRESIMS
(Yin, 2015)
93
viterofolin J
C20H32O3
320.24
N/A
V. rotundifolia
NMR, IR, HRESIMS
(Yin, 2015)
94
viterofolin K
C20H32O2
304.24
N/A
V. rotundifolia
NMR, IR, HRESIMS
(Yin, 2015)
95
sclareol
C20H36O2
308.50
515–03-7
V. rotundifolia/V. trifolia
MS, NMR
(Gu, 2007)
96
vitexoid
C10H16O3
184.11
1202522–23-3
V. trifolia
HRESIMS, NMR
(Djimabi et al., 2021)
97
(1R,2R,4S)-2-endo-hydroxy-1,8-cineole-β-D-glucopyranoside
C16H28O7
332.18
155836–26-3
V. rotundifolia
NMR, MS
(Wu et al., 2010)
98
pedicularis-lactone
C9H12O4
184.07
N/A
V. rotundifolia/V. trifolia
NMR
(Yu et al., 2021)
99
viteoid I
C9H12O4
184.07
193969–04-9
V. rotundifolia/V. trifolia
NMR
(Yu et al., 2021)
100
viteoid II
C9H12O4
184.07
193969–06-1
V. rotundifolia/V. trifolia
NMR
(Yu et al., 2021)
101
iridolactone
C9H12O4
184.07
138913–55-0
V. rotundifolia/V. trifolia
NMR
(Yu et al., 2021)
102
eucommiol
C9H16O4
188.10
55930–44-4
V. trifolia
HRESIMS, IR, UV, NMR
(Gu et al., 2008)
Number
Compounds
Molecular formula
Molecular weight
CAS No.
Plant
Identification method
References
103
(1S,5S,6R,9R)-10-O-p-hydroxybenzoyl-5,6β-dihydroxyiridoid 1-O-β-D-glucopyranoside
C22H26O12
482.14
N/A
V. trifolia
HRESIMS, IR, UV, NMR
(Bao et al., 2018)
104
10-O-vanilloylaucubin
C23H28O12
496.50
193969–08-3
V. rotundifolia/V. trifolia
HRESIMS, IR, UV, NMR
(Bao et al., 2018)
105
agnuside
C22H26O11
466.40
11027–63-7
V. rotundifolia/V. trifolia
HRESIMS, IR, UV, NMR
(Bao et al., 2018)
106
nishindaside
C23H30O12
498.17
88204–92-6
V. trifolia
HRESIMS, IR, UV, NMR
(Bao et al., 2018)
107
3-normal-butyl-nishindaside
C26H36O12
540.22
N/A
V. trifolia
HRESIMS, IR, UV, NMR
(Bao et al., 2018)
108
3-normal-butyl-isonishindaside
C26H36O12
540.22
N/A
V. trifolia
HRESIMS, IR, UV, NMR
(Bao et al., 2018)
109
1-oxo-eucommiol
C9H14O5
202.08
N/A
V. trifolia
NMR, HRFABMS, IR,
(Ono et al., 1997)
Number
Compounds
Molecular formula
Molecular weight
CAS No.
Plant
Identification method
References
110
(7R,8S)-dihydrodehydrodiconiferyl alcohol 9-O-β-D-glucopyranoside
C26H34O11
522.50
N/A
V. rotundifolia
UHPLC-Q-Orbitrap HRMS
(Zhang et al., 2021)
111
6′,10-di-O-(4-hydroxybenzoyl) aucubin
C29H30O13
586.17
N/A
V. rotundifolia
HRESIMS, IR, UV, NMR
(Zhao et al., 2017)
112
4,10-aromadendranediol
C15H26O2
238.37
70051–38-6
V. rotundifolia
MS, NMR
(Xu et al., 2019)
113
spathulenol
C15H24O
220.35
6750–60-3
V. trifolia
MS, NMR
(Gu, 2007)
114
ent-4α,10β-dihydroxyaromadendrane
C15H26O2
238.19
N/A
V. rotundifolia/V. trifolia
NMR, IR, HRESIMS
(Yin, 2015)
115
3β-hydroxy-30-al-urs-12-en-28-oic acid
C30H46O4
470.68
N/A
V. rotundifolia
IR, NMR, MS
(Huang et al., 2016)
116
ursolic acid
C30H48O3
456.70
77–52-1
V. rotundifolia
IR, NMR, MS
(Huang et al., 2016)
117
taraxerol
C30H50O
426.72
127–22-0
V. rotundifolia
IR, NMR, MS
(Huang et al., 2016)
Number
Compounds
Molecular formula
Molecular weight
CAS No.
Plant
Identification method
References
118
taraxerone
C30H48O
424.70
514–07-8
V. rotundifolia
IR, NMR, MS
(Huang et al., 2016)
119
α-amyrin
C30H50O
426.72
638–95-9
V. rotundifolia
IR, NMR, MS
(Huang et al., 2016)
120
β-amyrin
C30H50O
426.72
559–70-6
V. rotundifolia
IR, NMR, MS
(Huang et al., 2016)
121
lupeol
C30H50O
426.70
545–47-1
V. rotundifolia
IR, NMR, MS
(Huang et al., 2016)
122
betulinic acid
C30H48O3
456.70
472–15-1
V. rotundifolia
IR, NMR, MS
(Huang et al., 2016)
123
tormentic acid
C30H48O5
488.70
13850–16-3
V. rotundifolia
MS, NMR
(Chen et al., 2018)
124
2α,3β,23-trihydroxyolean-12-en-28-oic acid
C30H48O5
488.70
102519–34-6
V. rotundifolia
MS, NMR
(Chen et al., 2018)
125
dammarenediol-I 3S-O-β-glucopyranoside
C36H62O7
606.45
N/A
V. rotundifolia
FAB-MS, NMR
(Ono et al., 1998a)
126
arjunglucoside
C36H58O11
666.80
62319–70-4
V. rotundifolia
HRESIMS, IR, UV, NMR
(Zhao et al., 2017)
127
nigaichigoside F1
C36H58O11
666.80
95262–48-9
V. rotundifolia
HRESIMS, IR, UV, NMR
(Zhao et al., 2017)
128
3-oxotaraxer-14-en-30-al
C30H46O2
438.35
1527520–76-8
V. rotundifolia
NMR, IR, HRESIMS
(Huang et al., 2013)
129
3-epiursolic acid
C30H48O3
456.70
989–30-0
V. rotundifolia
NMR, IR, HRESIMS
(Huang et al., 2013)
Number
Compounds
Molecular formula
Molecular weight
CAS No.
Plant
Identification method
References
130
2α,3β-dihydroxyurs-12-en-28-oic acid
C30H48O4
472.36
4547–24-4
V. rotundifolia
NMR, IR, HRESIMS
(Huang et al., 2013)
131
β-daucosterol
C35H60O6
576.85
474–58-8
V. rotundifolia
NMR, IR, HRESIMS
(Huang et al., 2013)
132
oleanolic acid
C30H48O3
456.70
508–02-1
V. trifolia
MS, NMR, IR, UV
(Zhu, 2013)
133
maslinic acid
C30H48O4
472.70
4373–41-5
V. trifolia
MS, NMR, IR, UV
(Zhu, 2013)
134
betulinaldehyde
C30H48O2
440.70
13159–28-9
V. trifolia
MS, NMR
(Gu, 2007)
135
8-hydroxycolumbin
C20H22O7
374.39
104513–87-3
V. rotundifolia
UHPLC-Q-Orbitrap HRMS
(Zhang et al., 2021)
136
atractylenolide Ⅱ
C15H20O2
232.32
73069–14-4
V. rotundifolia
UHPLC-Q-Orbitrap HRMS
(Zhang et al., 2021)
137
neoandrographolide
C26H40O8
480.59
27215–14-1
V. rotundifolia
UHPLC-Q-Orbitrap HRMS
(Zhang et al., 2021)
138
atractylenolide Ⅰ
C15H18O2
230.30
73069–13-3
V. rotundifolia
UHPLC-Q-Orbitrap HRMS
(Zhang et al., 2021)
139
2α,19α-dihydroxyur-3-oxo-urs-12-en-28-oic acid
C30H46O5
486.68
176983–21-4
V. rotundifolia
UHPLC-Q-Orbitrap HRMS
(Zhang et al., 2021)
140
euscaphic acid
C30H48O5
488.70
53155–25-2
V. rotundifolia
UHPLC-Q-Orbitrap HRMS
(Zhang et al., 2021)
141
enoxolone
C30H46O4
470.68
471–53-4
V. rotundifolia
UHPLC-Q-Orbitrap HRMS
(Zhang et al., 2021)
142
2α-hydroxyursolic acid
C30H48O4
472.70
52213–27-1
V. rotundifolia
UHPLC-Q-Orbitrap HRMS
(Zhang et al., 2021)
Number
Compounds
Molecular formula
Molecular weight
CAS No.
Plant
Identification method
References
143
viteoside A
C28H44O11
556.60
209899–63-8
V. rotundifolia
MS, NMR
(Ono et al., 1998b)
Flavonoids
144
schaftoside
C26H28O14
564.50
51938–32-0
V. rotundifolia
UHPLC-Q-Orbitrap HRMS
(Zhang et al., 2021)
145
kaempferil-3-β-D-glucopyranoside
C21H20O11
448.09
N/A
V. rotundifolia
UHPLC-Q-Orbitrap HRMS
(Zhang et al., 2021)
146
cyanidin-3-O-glucoside
C21H20O11
448.11
N/A
V. rotundifolia
UHPLC-Q-Orbitrap HRMS
(Zhang et al., 2021)
147
vitexin
C21H20O10
432.38
3681–93-4
V. rotundifolia
UHPLC-Q-Orbitrap HRMS
(Zhang et al., 2021)
148
cynaroside
C21H20O11
448.38
5373–11-5
V. rotundifolia
UHPLC-Q-Orbitrap HRMS
(Zhang et al., 2021)
149
taxifolin
C15H12O7
304.25
480–18-2
V. rotundifolia
UHPLC-Q-Orbitrap HRMS
(Zhang et al., 2021)
150
5,3′-dihydroxy-6,7,4′-trimethoxy-flavanone
C18H18O7
346.11
N/A
V. rotundifolia
UHPLC-Q-Orbitrap HRMS
(Zhang et al., 2021)
151
genistein
C15H10O5
270.24
446–72-0
V. rotundifolia
UHPLC-Q-Orbitrap HRMS
(Zhang et al., 2021)
152
luteolin
C15H10O6
286.24
491–70-3
V. rotundifolia
MS, NMR
(Chen et al., 2018)
153
quercetin
C15H10O7
302.23
117–39-5
V. rotundifolia
UHPLC-Q-Orbitrap HRMS
(Zhang et al., 2021)
154
5,3′-dihydroxy-6,7,5′-trimethoxyflavanone
C18H16O7
344.09
N/A
V. rotundifolia
UHPLC-Q-Orbitrap HRMS
(Zhang et al., 2021)
Number
Compounds
Molecular formula
Molecular weight
CAS No.
Plant
Identification method
References
155
penduletin
C18H16O7
344.32
569–80-2
V. rotundifolia
MS, NMR
(Chen et al., 2018)
156
luteolin-4′-O-glucoside
C21H20O11
448.38
6920–38-3
V. rotundifolia
MS, NMR
(Chen et al., 2018)
157
hypolaetin-7-O-β-D-glucopyranoside
C21H20O12
464.40
32455–43-9
V. rotundifolia
MS, NMR
(Chen et al., 2018)
158
swertisin
C22H22O11
462.40
6991–10-2
V. rotundifolia
MS, NMR
(Chen et al., 2018)
159
agestricin D
C18H18O7
346.30
85563–76-4
V. rotundifolia
MS, NMR
(Chen et al., 2018)
160
eupatorin
C18H16O7
344.32
855–96-9
V. rotundifolia
HRESIMS, IR, UV, NMR
(Zhao et al., 2017)
161
casticin-3′-O-β-D-glucopyranoside
C25H28O13
536.15
N/A
V. rotundifolia
HRESIMS, IR, UV, NMR
(Zhao et al., 2017)
162
casticin/vitexicarpin
C19H18O8
374.30
479–91-4
V. rotundifolia
MS, NMR
(Chen et al., 2018)
163
artemetin
C20H20O8
388.37
479–90-3
V. rotundifolia
HRFABMS, NMR
(Ono et al., 2002)
164
centaureidin
C18H16O8
360.30
17313–52-9
V. rotundifolia
HRFABMS, NMR
(Ono et al., 2002)
165
5,5′-dihydroxy-4′,6,7-trimethoxyflavanone
C18H18O6
330.11
N/A
V. rotundifolia
IR, NMR
(Yoshioka et al., 2004)
166
kaempferol
C15H10O6
286.24
520–18-3
V. rotundifolia
NMR, MS
(Wu et al., 2010)
Number
Compounds
Molecular formula
Molecular weight
CAS No.
Plant
Identification method
References
167
chrysospleol D
C18H16O8
360.32
14965–20-9
V. rotundifolia
MS, NMR
(Chen et al., 2018)
168
persicogenin
C17H16O6
316.30
28590–40-1
V. trifolia
UV, NMR, MS
(Li et al., 2005a)
169
luteolin 7-methyl ether
C16H12O6
300.26
20243–59-8
V. trifolia
MS, NMR, IR, UV
(Zhu, 2013)
170
3′,4′,5-trihydroxy-3,7-dimethoxy-flavone
C17H14O7
330.29
2068–02-2
V. trifolia
MS, NMR, IR, UV
(Zhu, 2013)
171
acacetin
C16H12O5
284.26
480–44-4
V. trifolia
MS, NMR, IR, UV
(Zhu, 2013)
172
3′,4′,7-trimethoxy-5-hydroxy-flavanone
C18H16O6
328.32
29080–58-8
V. trifolia
MS, NMR, IR, UV
(Zhu, 2013)
173
apigenin
C15H10O5
270.24
520–36-5
V. trifolia
MS, NMR, IR, UV
(Zhu, 2013)
174
4′,5-dihydroxy-3, 6,7-trimethoxy-flavone
C17H14O6
314.08
41365–32-6
V. trifolia
MS, NMR, IR, UV
(Zhu, 2013)
175
7,3′-dihydroxy-5′-methoxy-isoflavone
C16H12O5
284.26
947611–61-2
V. trifolia
MS, NMR, IR, UV
(Zhu, 2013)
176
isoorientin
C21H20O11
448.38
4261–42-1
V. rotundifolia
HRESIMS, IR, NMR
(Zhang et al., 2013)
177
oroxylin A
C16H12O5
284.26
480–11-5
V. rotundifolia/V. trifolia
IR, MS, NMR
(Xin, 2005)
Number
Compounds
Molecular formula
Molecular weight
CAS No.
Plant
Identification method
References
Phenols
178
p-hydroxybenzoic acid ethyl ester
C9H10O3
166.17
120–47-8
V. trifolia
HRESIMS, NMR
(Djimabi et al., 2021)
179
p-hydroxyacetophenone
C8H8O2
136.15
99–93-4
V. trifolia
HRESIMS, NMR
(Djimabi et al., 2021)
180
vanillin
C8H8O3
152.15
121–33-5
V. trifolia
HRESIMS, NMR
(Djimabi et al., 2021)
181
coniferaldehyde
C10H10O3
178.18
458–36-6
V. trifolia
HRESIMS, NMR
(Djimabi et al., 2021)
182
9,12,15-octadecatrienoic acid
C18H30O2
278.40
28290–79-1
V. rotundifolia
UHPLC-Q-Orbitrap HRMS
(Zhang et al., 2021)
183
protocatechuic acid
C7H6O4
154.12
99–50-3
V. rotundifolia
UHPLC-Q-Orbitrap HRMS
(Zhang et al., 2021)
184
neochlorogeinic acid
C16H18O9
354.31
906–33-2
V. rotundifolia
UHPLC-Q-Orbitrap HRMS
(Zhang et al., 2021)
185
chlorogenic acid
C16H18O9
354.31
327–97-9
V. rotundifolia
UHPLC-Q-Orbitrap HRMS
(Zhang et al., 2021)
186
hydroxybenzoic acid
C7H6O3
138.12
99–96-7
V. rotundifolia
UHPLC-Q-Orbitrap HRMS
(Zhang et al., 2021)
187
salicylic acid
C7H6O3
138.12
69–72-7
V. rotundifolia
UHPLC-Q-Orbitrap HRMS
(Zhang et al., 2021)
188
ω-hydroxypropioguaiacone
C10H12O4
196.20
2196–18-1
V. rotundifolia
UHPLC-Q-Orbitrap HRMS
(Zhang et al., 2021)
Number
Compounds
Molecular formula
Molecular weight
CAS No.
Plant
Identification method
References
189
vanillic acid
C8H8O4
168.15
121–34-6
V. rotundifolia
UHPLC-Q-Orbitrap HRMS
(Zhang et al., 2021)
190
4-p-coumaroylquinic acid
C16H18O8
338.31
93451–44-6
V. rotundifolia
UHPLC-Q-Orbitrap HRMS
(Zhang et al., 2021)
191
apocynin
C9H10O3
166.17
498–02-2
V. rotundifolia
UHPLC-Q-Orbitrap HRMS
(Zhang et al., 2021)
192
cryptochlorogenic acid
C16H18O9
354.31
905–99-7
V. rotundifolia
UHPLC-Q-Orbitrap HRMS
(Zhang et al., 2021)
193
coniferyl aldehyde
C10H10O3
178.19
20649–42-7
V. rotundifolia
UHPLC-Q-Orbitrap HRMS
(Zhang et al., 2021)
194
3,4-Di-O-caffeoylquinic acid methyl ester
C26H26O12
530.48
114637–83-1
V. rotundifolia
UHPLC-Q-Orbitrap HRMS
(Zhang et al., 2021)
195
pyrogallol
C6H6O3
126.11
87–66-1
V. rotundifolia
UHPLC-Q-Orbitrap HRMS
(Zhang et al., 2021)
196
erythro-guaiacylglycerol
C10H14O5
214.21
38916–91-5
V. rotundifolia
MS, NMR, UV
(Okuyama and Yamazaki, 1998)
197
threo-guaiacylglycerol
C10H14O5
214.08
N/A
V. rotundifolia
MS, NMR, UV
(Okuyama and Yamazaki, 1998)
198
4-hydroxybenzoic acid methyl ester
C8H8O3
152.05
99–76-3
V. rotundifolia
IR, NMR
(Yoshioka et al., 2004)
Number
Compounds
Molecular formula
Molecular weight
CAS No.
Plant
Identification method
References
199
vanillic acid methyl ester
C9H10O4
182.17
3943–74-6
V. rotundifolia
IR, NMR
(Yoshioka et al., 2004)
200
4-hydroxy benzaldehyde
C7H6O2
122.12
123–08-0
V. rotundifolia
IR, NMR
(Yoshioka et al., 2004)
201
ferulic acid
C10H10O4
194.18
1135–24-6
V. rotundifolia
IR, NMR
(Yoshioka et al., 2004)
202
docosanoic acid
C22H44O2
340.58
112–85-6
V. rotundifolia
NMR, IR, HRESIMS
(Huang et al., 2013)
203
tetracosanoic acid
C24H48O2
368.64
557–59-5
V. rotundifolia
NMR, IR, HRESIMS
(Huang et al., 2013)
204
cerotic acid
C26H52O2
396.69
506–46-7
V. rotundifolia
NMR, IR, HRESIMS
(Huang et al., 2013)
205
4-ethoxyphenol
C8H10O2
138.16
622–62-8
V. trifolia
MS, NMR, IR, UV
(Zhu, 2013)
206
1-ethoxy-4-methoxybenzene
C9H12O2
152.19
5076–72-2
V. trifolia
MS, NMR, IR, UV
(Zhu, 2013)
207
p-cresol
C7H8O
108.14
106–44-5
V. trifolia
MS, NMR, IR, UV
(Zhu, 2013)
208
3-p-tolylpropanoic acid
C10H12O2
164.20
1505–50-6
V. trifolia
MS, NMR, IR, UV
(Zhu, 2013)
209
raspberry ketone
C10H12O2
164.20
5471–51-2
V. rotundifolia
MS, NMR
(Xu et al., 2019)
Number
Compounds
Molecular formula
Molecular weight
CAS No.
Plant
Identification method
References
210
α-hydroxy acetovanillone
C9H10O4
182.06
N/A
V. rotundifolia
MS, NMR
(Xu et al., 2019)
211
phenyl β-D-glucopyranoside
C12H16O6
256.25
1464–44-4
V. rotundifolia
MS, NMR
(Xu et al., 2019)
Lignan
212
(7S,8R)-dihydrodehydrodiconiferyl alcohol
C20H24O6
360.16
28199–69-1
V. rotundifolia/V. trifolia
UV, NMR, CD, MS
(Lee et al., 2013)
213
viterolignan A
C21H24O7
388.15
1469986–06-8
V. rotundifolia
UV, NMR, CD, MS
(Lee et al., 2013)
214
viterolignan B
C22H26O7
402.17
1469986–07-9
V. rotundifolia
UV, NMR, CD, MS
(Lee et al., 2013)
215
ficusal
C18H18O6
330.30
321991–55-3
V. rotundifolia
UV, NMR, CD, MS
(Lee et al., 2013)
216
(+)-lariciresinol
C20H24O6
360.40
27003–73-2
V. rotundifolia
UV, NMR, CD, MS
(Lee et al., 2013)
217
ficusesquilignan A
C31H36O11
584.23
321991–56-4
V. rotundifolia
UV, NMR, CD, MS
(Lee et al., 2013)
218
vitrifol A
C30H34O9
538.22
1111080–82-0
V. trifolia
IR, UV, NMR, HRESIMS
(Gu et al., 2008)
219
dihydrodehydrodiconiferyl alcohol
C20H24O6
360.40
28199–69-1
V. rotundifolia
MS, NMR
(Chen et al., 2018)
220
salicifoliol
C13H14O5
250.25
125564–65-0
V. rotundifolia
MS, NMR
(Chen et al., 2018)
Number
Compounds
Molecular formula
Molecular weight
CAS No.
Plant
Identification method
References
221
(+)-sesamin
C20H18O6
354.40
607–80-7
V. trifolia
MS, NMR, IR, UV
(Zhu, 2013)
222
4-hydroxysesamin
C20H18O7
370.40
63427–86-1
V. trifolia
MS, NMR, IR, UV
(Zhu, 2013)
223
(+)-paulownin
C20H18O7
370.35
13040–46-5
V. trifolia
MS, NMR, IR, UV
(Zhu, 2013)
224
4, 8-dihydroxysesamin
C20H18O7
370.11
63398–39-0
V. trifolia
MS, NMR, IR, UV
(Zhu, 2013)
Volatile oil
225
1-vinyl-1-methyl-4-methylene-2-(2-methyl-1-propen-1-yl) cycloheptane
C15H24
204.00
N/A
V. rotundifolia
GC–MS
(Wang et al., 2017b)
226
1,3,3-trimethyl-2-(1-methylbutene-1-ene-3-carbonyl) cyclohexene
C14H22O
206.00
N/A
V. rotundifolia
GC–MS
(Wang et al., 2017b)
227
(7α-isopropenyl-4,5-dimethyl-octahydro-inden-4-yl) methanol
C15H26O
222.00
N/A
V. rotundifolia
GC–MS
(Wang et al., 2017b)
Number
Compounds
Molecular formula
Molecular weight
CAS No.
Plant
Identification method
References
228
caryophyllene oxide
C15H24O
220.35
1139–30-6
V. rotundifolia
GC–MS
(Wang et al., 2017b)
229
2,6-dimethyl-3-citronellylpyrazine
C16H26N2
246.39
N/A
V. rotundifolia
GC–MS
(Wang et al., 2017b)
230
4-(2,7,7-trimethylcyclo [3.2.0] hept-2-en-1-yl)-3-en-2-one
C14H20O
204.00
N/A
V. rotundifolia
GC–MS
(Wang et al., 2017b)
231
7-isopropyl-1,1,4α-trimethyl-1,2,3,4,4α, 9,10,10α-octahydrophenanthrene
C20H30
270.00
N/A
V. rotundifolia
GC–MS
(Wang et al., 2017b)
232
2,4α, 8, 8-tetramethyl-decahydrocyclopropane [d]-nai
C15H26
206.00
N/A
V. rotundifolia
GC–MS
(Wang et al., 2017b)
233
methyl abietate
C21H32O2
316.50
127–25-3
V. rotundifolia
GC–MS
(Wang et al., 2017b)
Number
Compounds
Molecular formula
Molecular weight
CAS No.
Plant
Identification method
References
234
9,19-cycloergost-24(28)-en-3-ol,4,14-dimethyl-acetate(3α,4α,5α)
C32H52O2
468.00
N/A
V. rotundifolia
GC–MS
(Wang et al., 2017b)
235
cedranone
C15H24O
220.35
68891–95-2
V. rotundifolia
GC–MS
(Wang et al., 2017b)
236
3α, 9β-dihydroxy-3,5α, 8-trimethyl-tricyclo [6.3.1.0 (1,5)] dodecane
C15H26O2
238.00
N/A
V. rotundifolia
GC–MS
(Wang et al., 2017b)
237
13-heptadecen-1-ol
C17H32O
252.00
56554–77-9
V. rotundifolia
GC–MS
(Wang et al., 2017b)
238
α-bisabolene
C15H24
204.00
17627–44-0
V. rotundifolia
GC–MS
(Wang et al., 2017b)
239
(−)-globulol
C15H26O
222.36
489–41-8
V. rotundifolia
GC–MS
(Wang et al., 2017b)
240
sclareol oxide
C18H30O
262.00
5153–92-4
V. rotundifolia
GC–MS
(Wang et al., 2017b)
241
(1S,2E,4S,5R,7E,11E)-2,7,11-cembratriene
C20H34O2
306.00
N/A
V. rotundifolia
GC–MS
(Wang et al., 2017b)
Number
Compounds
Molecular formula
Molecular weight
CAS No.
Plant
Identification method
References
242
3,7-dimethyl-1-acetoxy-6,11-undecene
C16H28O2
252.00
N/A
V. rotundifolia
GC–MS
(Wang et al., 2017b)
243
3,3α-epoxydicyclopenta [a, d] cyclooctan-4β-ol,9,10α-dimethyl-6-methylene-3β-isopropyl
C20H32O2
304.00
N/A
V. rotundifolia
GC–MS
(Wang et al., 2017b)
244
1-(2,8,8-trimethyl-5,6,7,8-tetrahydro-4H-cycloheptatrieno [b] furan-5-yl) ethanone
C14H20O2
220.00
N/A
V. rotundifolia
GC–MS
(Wang et al., 2017b)
245
4-(2,6,6-trimethyl-cyclohexen-1-yl)-butane-2-ol
C13H24O
196.00
N/A
V. rotundifolia
GC–MS
(Wang et al., 2017b)
Number
Compounds
Molecular formula
Molecular weight
CAS No.
Plant
Identification method
References
246
2,5,5,8α-Tetramethyloctahydro-2H-benzopyran
C13H24O
196.00
N/A
V. rotundifolia
GC–MS
(Wang et al., 2017b)
247
acetic acid,1- [2-(2,2,6-trimethyl-bicyclo [4.1.0] hept-1-yl)-ethyl]-vinyl ester
C16H26O2
250.00
N/A
V. rotundifolia
GC–MS
(Wang et al., 2017b)
248
di (2-ethyl hexyl) adipate (deha)
C22H42O4
370.00
N/A
V. rotundifolia
GC–MS
(Wang et al., 2017b)
249
9,11-dedihydrotestosterone, acetate
C21H28O3
328.00
N/A
V. rotundifolia
GC–MS
(Wang et al., 2017b)
250
(−)-epicedrol
C15H26O
222.37
19903–73-2
V. rotundifolia
GC–MS
(Wang et al., 2017b)
251
17-hydroxyandrostane-3,11-dione
C19H28O3
304.00
1010823–99-0
V. rotundifolia
GC–MS
(Wang et al., 2017b)
Number
Compounds
Molecular formula
Molecular weight
CAS No.
Plant
Identification method
References
252
cedrol
C15H26O
222.37
77–53-2
V. rotundifolia
GC–MS
(Wang et al., 2017b)
253
2,2,4-trimethyl-3-(3,8,12,16-tetramethyl-heptadeca-3,7,11,15-tetraenyl)-cyclohexanol
C30H52O
428.00
N/A
V. rotundifolia
GC–MS
(Wang et al., 2017b)
254
cyclodecacyclotetradecene,1,2,3,4,5,6,7,8,9,10,11,12,13,14,15,16,17,18,19,20-eicosahydro
C22H40
304.00
N/A
V. rotundifolia
GC–MS
(Wang et al., 2017b)
255
1,4-methanoazulen-9-one, decahydro-1,5,5,8α-tetramethyl-[1R-(lα-3αβ,4α,8αβ)]
C15H24O
220.00
N/A
V. rotundifolia
GC–MS
(Wang et al., 2017b)
Number
Compounds
Molecular formula
Molecular weight
CAS No.
Plant
Identification method
References
256
oxalic acid,2-ethylhexyl octadecyl ester
C28H54O4
454.00
N/A
V. rotundifolia
GC–MS
(Wang et al., 2017b)
257
8-propoxy cedrane
C18H32O
264.40
N/A
V. rotundifolia
GC–MS
(Wang et al., 2017b)
258
trumpet alcohol
C15H26O
222.00
N/A
V. rotundifolia
GC–MS
(Wang et al., 2017b)
259
4-(2,2,6-trimethyl-bicyclo [4.1.0] hept-1-yl)-butan-2-one
C14H24O
208.00
N/A
V. rotundifolia
GC–MS
(Wang et al., 2017b)
260
cis-2,3,4,4α, 5,6,7,8-octahydro-1,1,4α, 7-tetramethyl-1H-benzocycloheptene-7-ol
C15H26O
222.00
N/A
V. rotundifolia
GC–MS
(Wang et al., 2017b)
261
N-(N-methylformamidyl)-semithiocarbazide
C3H8N4OS
148.00
N/A
V. rotundifolia
GC–MS
(Wang et al., 2017b)
Number
Compounds
Molecular formula
Molecular weight
CAS No.
Plant
Identification method
References
262
iron, tetracarbonyl [(6,7-eta.)-3-oxabicyclo [3.2.0] hept-6-ene-2,4-dione]
C10H4FeO7
292.00
N/A
V. rotundifolia
GC–MS
(Wang et al., 2017b)
263
spiro [2.5] octane,5,5-dimethyl-4-(3-oxobutyl)
C14H24O
208.00
N/A
V. rotundifolia
GC–MS
(Wang et al., 2017b)
264
1β,4α-epoxy-2H-cyclopenta [3,4] cyclopropa [8,9] cycloundec[1,2-b] oxiren-5(6H)-one,7-(acetyloxy) decahydro-2,9,10-trihydroxy-3,6,8,8,10a-pentamethyl
C22H32O8
424.00
N/A
V. rotundifolia
GC–MS
(Wang et al., 2017b)
Number
Compounds
Molecular formula
Molecular weight
CAS No.
Plant
Identification method
References
265
valeric acid,2,6-dimethylnon-1-en-3-yn-5-yl ester
C16H26O2
250.00
N/A
V. rotundifolia
GC–MS
(Wang et al., 2017b)
266
thunbergol
C20H34O
290.00
25269–17-4
V. rotundifolia
GC–MS
(Wang et al., 2017b)
267
4,8,13-doufatriene-1,3-diol
C20H34O2
306.00
N/A
V. rotundifolia
GC–MS
(Wang et al., 2017b)
268
cholestan-3,5-diol-6-one,3-acetate
C29H48O4
460.00
N/A
V. rotundifolia
GC–MS
(Wang et al., 2017b)
269
D-homo-24-nor-17-oxachola-20,22-diene-3,7,16-trione,14,15:21,23-diepoxy-4,4,8-trimethyl-(5α,13α,14β,15β,17α)
C26H32O6
440.00
N/A
V. rotundifolia
GC–MS
(Wang et al., 2017b)
270
8-n-Hexylpentadecane
C21H44
296.57
13475–75-7
V. rotundifolia
GC–MS
(Wang et al., 2017b)
Number
Compounds
Molecular formula
Molecular weight
CAS No.
Plant
Identification method
References
271
squalene
C30H50
410.71
7683–64-9
V. rotundifolia
GC–MS
(Wang et al., 2017b)
272
heneicosane
C21H44
296.57
629–94-7
V. rotundifolia
GC–MS
(Wang et al., 2017b)
273
3-keto-N-acetyl-dihydro-pseudotomatidine
C29H47NO3
457.00
N/A
V. rotundifolia
GC–MS
(Wang et al., 2017b)
274
pentacosane
C25H52
352.68
629–99-2
V. rotundifolia
GC–MS
(Wang et al., 2017b)
275
tetrahy droactinidiolide
C11H18O2
182.26
16778–27-1
V. rotundifolia
GC–MS
(Wang et al., 2017b)
276
hentriacontane
C31H64
436.84
630–04-6
V. rotundifolia
GC–MS
(Wang et al., 2017b)
277
hexatriacontane
C36H74
506.00
630–06-8
V. rotundifolia
GC–MS
(Wang et al., 2017b)
278
tetracontane
C40H82
563.08
4181–95-7
V. rotundifolia
GC–MS
(Wang et al., 2017b)
279
(1S,2E,4S,6R,7E)-2,7,11-cembratriene
C20H34O2
306.00
N/A
V. rotundifolia
GC–MS
(Wang et al., 2017b)
280
n-tetratetracontane
C44H90
619.19
7098–22-8
V. rotundifolia
GC–MS
(Wang et al., 2017b)
281
α-thujene
C10H16
136.00
470–82-6
V. rotundifolia
GC–MS
(Chen et al., 2007)
282
Δ3-carene
C10H16
136.00
13466–78-9
V. rotundifolia
GC–MS
(Chen et al., 2007)
283
camphene
C10H16
136.234
79–92-5
V. rotundifolia
GC–MS
(Chen et al., 2007)
Number
Compounds
Molecular formula
Molecular weight
CAS No.
Plant
Identification method
References
284
β-phellandrene
C10H16
136.00
555–10-2
V. rotundifolia
GC–MS
(Chen et al., 2007)
285
sabinene
C10H16
136.234
3387–41-5
V. rotundifolia
GC–MS
(Chen et al., 2007)
286
β-pinene
C10H16
136.00
127–91-3
V. rotundifolia
GC–MS
(Chen et al., 2007)
287
m-cymene
C10H14
134.22
535–77-3
V. rotundifolia
GC–MS
(Chen et al., 2007)
288
1, 8-cineole
C10H18O
154.00
N/A
V. rotundifolia
GC–MS
(Chen et al., 2007)
289
eucalyptone
C28H38O7
486.597
172617–99-1
V. rotundifolia
GC–MS
(Luo et al., 2015)
290
β-terpineol
C10H18O
154.249
138–87-4
V. rotundifolia
GC–MS
(Luo et al., 2015)
291
(5β,9β,10α)-kaur-16-ene
C20H32
272.468
20070–61-5
V. rotundifolia
GC–MS
(Luo et al., 2015)
292
estrone
C18H22O2
270.366
53–16-7
V. rotundifolia
GC–MS
(Luo et al., 2015)
293
4-carene
C10H16
136.234
29050–33-7
V. rotundifolia
GC–MS
(Luo et al., 2015)
294
cis-vaccenic acid
C18H34O2
282.461
506–17-2
V. rotundifolia
GC–MS
(Zhang et al., 2011)
295
octadeca-9,17-dienoic acid
C18H32O2
280.445
17351–35-8
V. rotundifolia
GC–MS
(Zhang et al., 2011)
296
aromadendrene
C15H24
204.35
489–39-4
V. rotundifolia
GC–MS
(Chen et al., 2015)
297
caryophyllene oxide
C15H24O
220.35
1139–30-6
V. rotundifolia
GC–MS
(Chen et al., 2015)
298
agarospirol
C15H26O
222.366
23811–08-7
V. rotundifolia
GC–MS
(Chen et al., 2015)
Number
Compounds
Molecular formula
Molecular weight
CAS No.
Plant
Identification method
References
299
α-muurolene
C15H24
204.00
10208–80-7
V. rotundifolia
GC–MS
(Chen et al., 2007)
300
hexadecyne
C16H30
222.41
451500–33-7
V. rotundifolia
GC–MS
(Chen et al., 2015)
301
α-caryophyllene
C15H24
204.35
6753–98-6
V. rotundifolia
GC–MS
(Chen et al., 2015)
302
eudesmol
C15H28O
224.38
51317–08-9
V. rotundifolia
GC–MS
(Chen et al., 2015)
303
limonene oxide
C10H16O
152.23
203719–54-4
V. rotundifolia
GC–MS
(Chen et al., 2015)
304
3,5-di-tert-butylphenol
C14H22O
206.32
1138–52-9
V. rotundifolia
GC–MS
(Chen et al., 2015)
Other compounds
305
stigmast-4-ene-3,6-dione
C29H46O2
426.70
23670–94-2
V. trifolia
HRESIMS, NMR
(Djimabi et al., 2021)
306
6β-hydroxystigmast-4-en-3-one
C29H48O2
428.69
36450–02-9
V. trifolia
HRESIMS, NMR
(Djimabi et al., 2021)
307
β-sitosterol
C29H50O
414.71
83–46-5
V. trifolia
HRESIMS, NMR
(Djimabi et al., 2021)
308
stigmast-4-en-6b-ol-3-one
C29H48O2
428.00
N/A
V. trifolia
HRESIMS, IR, UV, NMR
(Gu et al., 2008)
309
7-oxositosterol
C29H48O2
428.37
2034–74-4
V. trifolia
MS, NMR, IR, UV
(Zhu, 2013)
310
karakoline
C22H35NO4
377.52
39089–30-0
V. rotundifolia
UHPLC-Q-Orbitrap HRMS
(Zhang et al., 2021)
Number
Compounds
Molecular formula
Molecular weight
CAS No.
Plant
Identification method
References
311
panaxydol
C17H24O2
260.37
72800–72-7
V. rotundifolia
UHPLC-Q-Orbitrap HRMS
(Zhang et al., 2021)
312
coronaric acid
C18H32O3
296.40
6814–52-4
V. rotundifolia
UHPLC-Q-Orbitrap HRMS
(Zhang et al., 2021)
313
ricinolic acid
C18H34O3
298.50
141–22-0
V. rotundifolia
UHPLC-Q-Orbitrap HRMS
(Zhang et al., 2021)
314
sucrose
C12H22O11
342.12
57–50-1
V. rotundifolia
MS, NMR, IR, UV
(Zhu, 2013)
315
physcion
C16H12O5
284.26
521–61-9
V. rotundifolia
HPLC, ESIMS, NMR
(Guan et al., 2010)
316
stearic acid
C18H36O2
284.48
57–11-4
V. rotundifolia
GC–MS
(Hu et al., 2007)
317
pyranopyran-1,8-dione
C8H4O4
164.01
N/A
V. trifolia
HRESIMS, IR, UV, NMR
(Lee et al., 2017)
318
thymylisobutyrate
C14H20O2
220.31
5451–67-2
V. rotundifolia
UHPLC-Q-Orbitrap HRMS
(Zhang et al., 2021)
319
dihydrodehydrodiconiferylalcohol-β-D-(2′-O-p-hydroxybenzoyl) glucoside
C33H38O13
642.23
N/A
V. rotundifolia
MS, NMR, UV
(Okuyama and Yamazaki, 1998)
320
vanilloyl-β-D-(2′-O-p-hydroxybenzoyl) glucoside
C21H22O11
450.12
N/A
V. rotundifolia
MS, NMR, UV
(Okuyama and Yamazaki, 1998)
Number
Compounds
Molecular formula
Molecular weight
CAS No.
Plant
Identification method
References
321
(E)-3,3′-dimethoxy-4,4′-dihydroxystilbene
C16H16O4
272.10
7329–69-3
V. rotundifolia
MS, NMR
(Xu et al., 2019)
322
(8R)-evofolin B
C17H18O6
318.32
N/A
V. rotundifolia
MS, NMR
(Xu et al., 2019)
323
l-picein
C14H18O7
298.29
530–14-3
V. rotundifolia
MS, NMR
(Xu et al., 2019)
324
4-(4′-hydroxyphenyl)-2-butanone-4′-O-β-D-glucopyranoside
C17H24O7
340.15
N/A
V. rotundifolia
MS, NMR
(Xu et al., 2019)
5.1 Terpenoids
Terpenoids account for the largest proportion, mainly diterpenoids and iridoids (1–143) (Fig. 2, Fig. 3 and Fig. 4). Labdane-type diterpenoids vitexilactone (3), rotundifuran (4) and halimane-type diterpene vitetrifolin D (7) were isolated and purified from the ethyl acetate fraction of Viticis Fructus (Fang et al., 2019). Iridoids are also a class of special monoterpenoids composed of five-membered carbon rings and six-membered oxygen heterocycles. They mainly exist in the form of glycosides in Viticis Fructus. Two iridoids were isolated and identified from 80 % ethanol extract: 10-O-vanilloylaucubin (104) and agnuside (150) (Bao et al., 2018).
Structures of terpenoids (1–85).
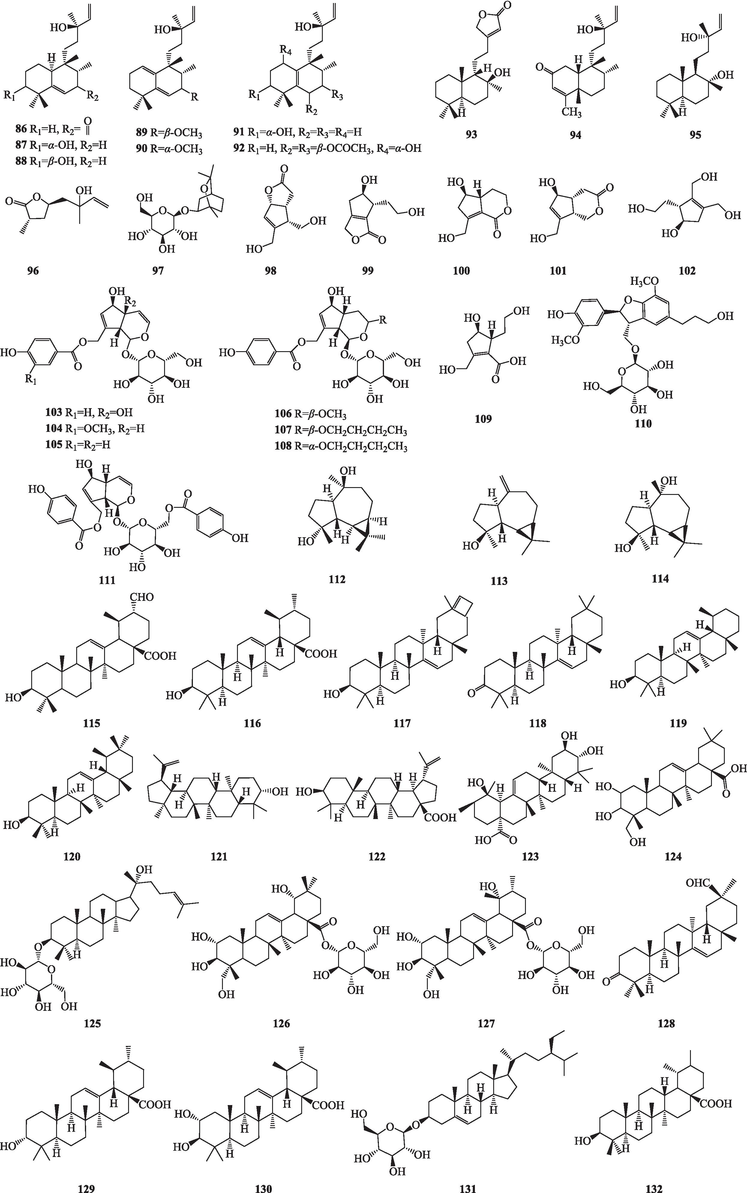
Structures of terpenoids (86–132).
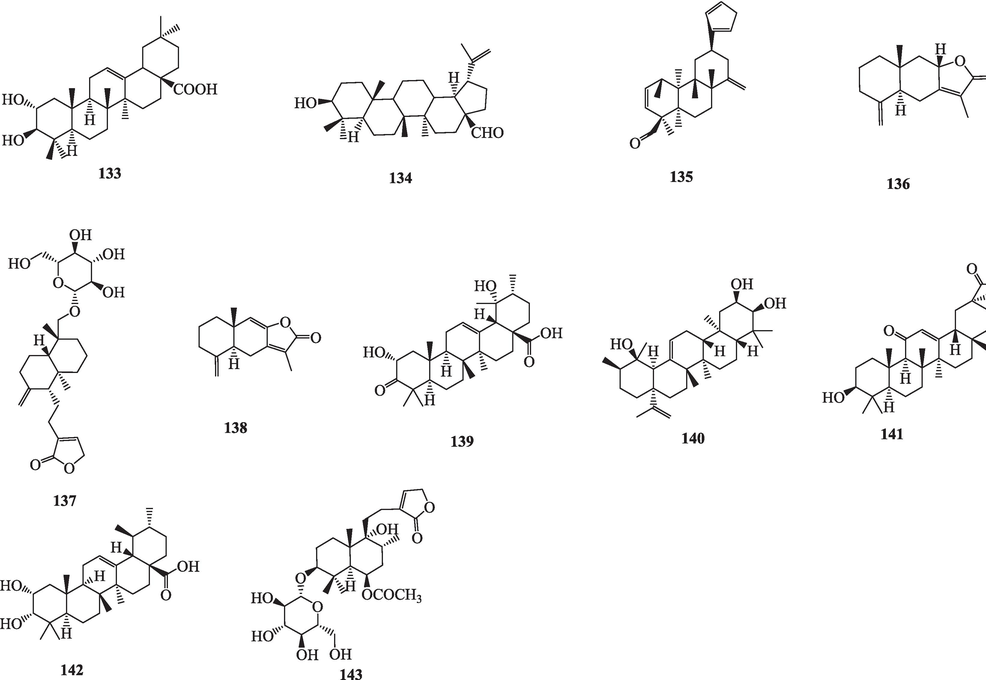
Structures of terpenoids (133–143).
5.2 Flavonoids
Flavonoids are typical compounds in Viticis Fructus (144–177) (Fig. 5). Casticin is the only index component of Viticis Fructus in ChP, which is used to measure and evaluate the quality of Viticis Fructus. The flavonoids in Viticis Fructus are mainly divided into flavonoids, isoflavones, dihydroflavones and anthocyanins, among which flavonoids are the main compounds. Luteolin (152) was identified from cold soak of 95 % ethanol. Three flavonoids were isolated from chloroform fraction of Viticis Fructus: luteolin-4′-O-glucoside (156), casticin (162) and apigenin (173) (Chen et al., 2018). In addition, flavonoids in Viticis Fructus were recognized by ultra-high-performance-liquid chromatography-quadrupole-Orbitrap high-resolution mass spectrometry (UHPLC-Q-Orbitrap HRMS), including taxifolin (149), genistein (151), quercetin (153) (Zhang et al., 2021).
Structures of flavonoids (144–177) and phenolic acids (178–211).
5.3 Phenolic acids
Viticis Fructus also includes phenolic acids (178–211) (Fig. 5), such as protocatechuic acid (183), neochlorogenic acid (184), chlorogenic acid (185), hydroxybenzoic acid (186), vanillic acid (189), ferulic acid (201) and so on (Yoshioka et al., 2004; Zhang et al., 2021). V. trifolia fruits were extracted with ethanol to obtain a crude extract and then further extracted with ethyl acetate. The ethyl acetate fraction was eluted with a mixture of petroleum ether and acetone, and the compounds (179–181) were successfully isolated and identified in the fraction (Djimabi et al., 2021).
5.4 Lignans
Thirteen lignan compounds were isolated from Viticis Fructus, which were divided into diepoxy lignans, neolignans and other types (212–224) (Fig. 6). Viticis Fructus extract was carried out repeatedly by column chromatography using silica gel, RP-18 and MCI gel to obtain viterolignan A (213), viterolignan B (214), ficusal (215) and so on (Lee et al., 2013).
Structures of lignan (212–224) and others (305–324).
5.5 Volatile oil
Fresh Viticis Fructus contains massive volatile oil. The crude and processed products of Viticis Fructus were extracted by continuous reflux in soxhlet extractors and then identified by GC–MS. A total of 56 compounds (225–280) were identified, such as hentriacontane (276), hexatriacontane (277), tetracontane (278) (Wang et al., 2017b). In another study, the volatile oil was extracted by steam distillation for 6 h and 9 compounds (281–288, 299) were identified by GC–MS (Chen et al., 2007).
5.6 Other compounds
In addition to the above compounds, steroids, anthraquinones, sugars, fatty acids and alkaloids were also isolated from Viticis Fructus (Fig. 6). Three compounds (305–307) were identified by nuclear magnetic resonance (NMR) from Viticis Fructus extracted with 95 % ethanol (Djimabi et al., 2021). Five compounds such as karakoline (310) were identified from the extract of Viticis Fructus by UHPLC-Q-Orbitrap HRMS (Zhang et al., 2021). It may still contain other compounds in Viticis Fructus, which gives a direction for further research.
Various compounds had been isolated and identified from Viticis Fructus, most of which were from the fruits of V. rotundifolia. The main chemical constituents were terpenoids and flavonoids. Terpenoids had anti-tumor activity and most of terpenoids were diterpenes and iridoids in structure. Their structure–activity relationship should be further elucidated. Casticin had good activity in tumors, inflammation, oxidative damage and other ailments. Some studies revealed a difference in the casticin content of V. rotundifolia and V. trifolia fruits, which was possible to use as quality markers. Therefore, the extraction process optimization and batch preparation of casticin should be focused on. Moreover, the information on many compounds is not complete, such as CAS number. It is necessary to further improve the compound information to avoid the misuse of synonyms.
6 Pharmacological activities
Viticis Fructus was mainly used for treating colds, headaches, eye pain, swelling, aching of gum and dizziness according to ancient records. With the progress and development of pharmacology, it could also be used to prevent cervical cancer (Zeng et al., 2012), prostate cancer (Meng et al., 2012) and other cancers, as well as anti-inflammatory (Fang et al., 2019), antioxidant (Le et al., 2022), PMS (Ye, 2010) (Table 2). Additionally, V. rotundifolia fruits were more widely used in the treatment of various diseases than V. trifolia fruits.
Activity
Compounds or extracts
Dose
Model/ Animal/Cell lines
Result/Mechanism
Reference
Antitumor-breast cancer effects
vitexilactone, (rel 5S,6R,8R,9R,10S)-6-acetoxy-9-hydroxy-13(14)-labden-16,15-olide, rotundifuran, vitetrifolin D and vitetrifolin E
100 μg/mL
mouse breast cancer thermosensitive stFTZ10 cell line
Inhibited the proliferation of tsFT210 cells through arresting the cell cycle at the G2/M phase and inducing apoptosis.
(Li et al., 2005a)
Antitumor-breast cancer effects
casticin
0.5 µM
MDA-MB-231 and MCF-7 cell lines
FOXO3a was a fatal mediator of casticin inhibiting breast cancer cell apoptosis.
(Liu et al., 2014b)
Antitumor-breast cancer effects
casticin
0.25 and 0.5 μM
human breast cancer cell line MDA-MB-231 and mouse breast cancer cell line 4 T1
Inhibited the migration and invasion of breast cancer cells.
(Fan et al., 2018)
Antitumor-breast cancer effects
casticin
0.5–2 μM
MCF-7 sub-lines MN1 and MDD2
Led to apoptosis.
(Haïdara et al., 2006)
Antitumor-cervical cancer effects
casticin
0.5, 1, 2 and 4 μM
human cervical cancer cell lines HeLa, CasKi, and SiHa
Inhibit cell apoptosis.
(Zeng et al., 2012)
Antitumor-cervical cancer effects
RTF
8 and 16 µM. 10 and 40 mg/kg/day
HeLa, SiHa cells and BALB/C nude mice
It is related to ROS-induced mitochondrial-dependent apoptosis.
(Gong et al., 2021)
Activity
Compounds or extracts
Dose
Model/ Animal/Cell lines
Result/Mechanism
Reference
Antitumor-cervical cancer effects
casticin
2 and 4 µM
human cervical cancer cell lines
Induced apoptosis of cervical cancer cells by ROS-mediated mitochondrial pathway
(Chen et al., 2011)
Antitumor-cervical cancer effects
rotundifuran
8 and 16 µM
HeLa and SiHa cells
Inhibited the proliferation and induce apoptosis of cervical cancer cells.
(Shen, 2020)
Antitumor-lung cancer effects
total flavonoids of Viticis Fructus
0.5, 1 and 2 µg/mL
lung cancer NCI-H446 cell line
Inhibited the characteristics of LCSLCs.
(Cao et al., 2014)
Antitumor-lung cancer effects
casticin
1, 5 and 10 μM
human lung cancer A549 cell line, were injected subcutaneously into the back of BALB/C-nude mice.
Inhibited the self-renewal and invasion of lung cancer stem-like cells.
(Liu et al., 2014a)
Antitumor-liver cancer effects
casticin
0.1, 0.3, 1, 3 and 10μM
MHCC97 cell line
Arrested self-renewal of MHCC97 liver cancer stem cells.
(He et al., 2014)
Antitumor-liver cancer effects
casticin
1, 3, 10, 30 and 100 μM
MHCC97H, SK-Hep-1, L02 cell Lines and male BALB/c-nude mice.
Suppressed the stemness of HCC cells.
(Li et al., 2020a)
Antitumor-liver cancer effects
casticin
2.5, 5 and 10 μM
Hep G2 (p53 wild-type) and PLC/PRF/5 (p53 mutant) cells.
Induced growth inhibition and cell cycle arrest.
(He et al., 2013)
Activity
Compounds or extracts
Dose
Model/Diseases
Result/Mechanism
Reference
Antitumor-liver cancer effects
casticin
3, 10 and 30μM
PLC/PRF/5 (p53 mutant) and Hep G2 (p53 wild type) human HCC cells.
Glutathione depletion and DR5 upregulation.
(Yang et al., 2011)
Antitumor-colorectal cancer effects
casticin
40 μM
colo 205 human colon cancer cells
Induced apoptosis in human CRC cells.
(Shang et al., 2017)
Antitumor-colorectal cancer effects
casticin
1 and 3μM
human colon cancer HT-29, HCT-116 and SW480 cell lines.
Caused apoptosis via regulating anti-apoptotic proteins and DR5 from colon cancer cells.
(Tang et al., 2013)
Antitumor-glioma effects
casticin
10, 20 µM
human glioma cells U251, U87 and U373
Prevented the growth of human glioma cells by mitosis arrest.
(Liu et al., 2013)
Antitumor-gastric cancer effects
casticin
1μM
human gastric cancer cells BGC-823, SGC-7901 and MGC-803
Enhanced trail-induced apoptosis via down-regulating cell survival protein and up-regulating DR5 receptor.
(Zhou et al., 2013)
Antitumor-prostate carcinoma effects
casticin
10, 30 and 50 μM
human prostate cancer PC-3 cell line
Induced apoptosis of human prostate cancer cells by arresting G2/M phase.
(Meng et al., 2012)
Activity
Compounds or extracts
Dose
Model/Diseases
Result/Mechanism
Reference
Antitumor-leukaemia effects
rotundifuran
25 and 50 µM
human myeloid leukaemia HL-60 cells
Killed HL-60 cells by the activation of the apoptosis mechanism.
(Ko et al., 2001)
Antitumor-leukemia effects
casticin
0.1, 0.2 and 0.4 mg/kg
male BALB/c mice were peritoneally injected with WEHI-3 leukemia cells
Potentiated immune response.
(Lai et al., 2019)
Antitumor-melanoma effects
casticin
0.25–5 μM
B16F10 cell line
Attenuated the proliferation, migration and invasion of cells.
(Shih et al., 2017)
Antitumor-leukemia effects
2′,3′,5-trihydroxy-3,6,7-trimethoxyflavone, vitexicarpin and artemetin
N/A
HL-60 cells
Suppressed the proliferation of human myeloid leukemia cells by inducing apoptosis.
(Ko et al., 2000)
Antitumor effects
persicogenin, artemetin, luteolin, penduletin, vitexicarpin
100, 50, 25, 6.25 and 1 μg/mL
tsFT210 and K562 cells
Inhibited cell cycle progression of G2/M and induced apoptosis in mammalian cancer cells.
(Li et al., 2005a)
Antitumor effects
vitetrifolin H, vitetrifolin I and vitexoid
25–50 μM
tsFT210 cells
Inhibited the proliferation of tsFT210 cells.
(Wu et al., 2009)
Anti-inflammatory effects
casticin
N/A
human neutrophils
Inhibited the chemotaxis of human neutrophils.
(Ahmad et al., 2010)
Activity
Compounds or extracts
Dose
Model/Diseases
Result/Mechanism
Reference
An-inflammation effects
casticin
0.3, 1, 3 and 10 μM
lipopolysaccharide-stimulated RAW264.7 cells
The anti-inflammatory activity suppressed the expression of COX-2 and iNOS by blocking NF-κB, MAPK and Akt pathways.
(Liou et al., 2014)
An-inflammation effects
casticin
5–100 nM
human umbilical vein endothelial cells
Alleviated vascular inflammation.
(Lee et al., 2012)
Anti-inflammation effects
viterotulin C, vitexilactone D, vitexilactone, rotundifuran, vitetrifolin B, viterotulin B, vitetrifolin D
50 μM
HEK293 cells
Activation of NF-κB
(Fang et al., 2019)
An-inflammation-asthma effects
casticin
5 and 10 mg/kg
ovalbumin −induced asthma in female BALB/c mice
Improved pathological changes by inhibiting the expression of T helper 2 cell cytokines in asthmatic mice.
(Liou et al., 2018)
Anti-inflammation-UC effects
casticin
5, 10 and 20 mg/kg
RAW264.7 cell line and dextran sulfate sodium‐induced colitis in C57BL/6 mice
Prevented UC in mice by suppressing NF-κB and ROS signaling pathways.
(Ma et al., 2018)
Activity
Compounds or extracts
Dose
Model/Diseases
Result/Mechanism
Reference
Anti-inflammation-KOA effects
casticin
0.2 mg/kg/day
monoiodoacetic acid-induced knee osteoarthritis in mice and an inflammatory model in primary synovial fibroblasts induced by lipopolysaccharide
Treated KOA via suppressing HIF-1α/NLRP3 inflammasome signaling.
(Li et al., 2020b)
Anti-inflammation effects
casticin
animal experiment:10 mg/kg. cell experiment:10, 20 and 30 μM
surgery destabilizing the medial meniscus was performed on the right knees of mice and IL-1β-induced inflammation in mice
Ameliorated osteoarthritis-related cartilage degeneration.
(Chu et al., 2020)
Antioxidation effects
orientin and 3,4-di-O-caffeoylquinic acid
0.1–100 μg/mL
DPPH
Significant antioxidant activity.
(Kim, 2009)
Antioxidation effects
vitrifolin A
1, 5, 10, 20, 40 and 60 µM
lipopolysaccharide-activated mouse macrophages
Vitrifolin A had a moderate inhibitory activity on lipopolysaccharide-activated rat macrophages.
(Zhang et al., 2013)
Activity
Compounds or extracts
Dose
Model/Diseases
Result/Mechanism
Reference
Antioxidation effects
ferruginol
N/A
N/A
Compared with the standard antioxidant, ferruginol showed more potent antioxidant activity.
(Ono et al., 1999)
Antioxidation effects
methanol extract
N/A
ferric thiocyanate method
They could exhibit stronger antioxidative activity than 3-tert-butyl-4hhydroxyanisole.
(Ono et al., 1998b)
Antioxidation effects
Viticis Fructus extract
300, 150 and 75 mg/kg
mice neck hypodermic injection D-galactose
The ethanol extract of Viticis Fructus had good anti-aging and antioxidant effects.
(Yin et al., 2016)
PMS effects
rotundifuran and casticin
rotundifuran and casticin:40 mg/kg/d, 10 mg/kg/d.
diethylstilbestrol induction female SD rats
Improved the related pathological changes of PMS.
(Ye, 2010)
Cardiovascular Disease-hyperlipidemia effects
viterofolin H, (5S, 6R,
8R, 9R, 10S)-6-acetoxy-9-hydroxy-13 (14)-labden-16,15-olide and previtexilactoneN/A
HepG2 cells
Viterofolin H, previtexilactone and (5S, 6R,8R, 9R, 1 0S)-6-acetoxy-9-hydroxy-13 (14)-labden-16,15-olide had moderate activity to promote LDL uptake.
(Wang et al., 2018)
Activity
Compounds or extracts
Dose
Model/Diseases
Result/Mechanism
Reference
Cardiovascular Disease-antiplatelet activation effects
casticin
1 and 2μM
blood of healthy volunteers aged 18–26 years
Inhibited platelet activation.
(Xiong et al., 2022)
Cardiovascular Disease-decrease blood pressure effects
alcohol extract of Viticis Fructus
N/A
cat
The alcohol extract of Viticis Fructus had noticeable antihypertensive effect and maintained for a long time.
(Guan, 2011)
Cardiovascular disease-antiatherosclerosis effects
ethanol extract of V. rotundifolia, casticin and luteolin
extract:10 and 100 µg/mL
casticin and luteolin: 10, 20 and 40 µM.young and healthy male volunteers
Inhibited LDL and HDL oxidation.
(Kim et al., 2020)
Other-analgesia effects
Viticis Fructus methanolic extract
1.75 and 7 g/kg
nitroglycerin-induced migraine in mice
Suppressed hyperalgesia of the trigeminovascular system.
(Wen et al., 2020)
Activity
Compounds or extracts
Dose
Model/Diseases
Result/Mechanism
Reference
Other-analgesia effects
pedicularis-lactone, viteoid I and viteoid II
15 mg/kg
paclitaxel-induced mechanical allodynia in C57BL/6NCr mice and LY-PPB6 cell line
Inhibited paclitaxel-induced heterologous pain in mice.
(Yu et al., 2021)
Other-analgesia effects
vitexfolin A, agnuside, 10-O-vanilloylaucubin, dihydrodehydrodiconiferylalcohol-β-D-(2′-O-p-hydroxybenzoyl) glucoside
15, 50, 25 and 50 mg/kg
acetic acid-induced writhing method in mice
Alleviated the writhing symptoms.
(Okuyama and Yamazaki, 1998)
Other-analgesia effects
Viticis Fructus extract
1.17 g/kg
0.6% acetic acid-induced pain in mice
Total flavonoids and volatile oil were analgesic active ingredients.
(Sun et al., 1997)
Other-aldose reductase activity effects
ether extract
N/A
enzyme reactions
Inhibited the enzyme activity.
(Shin et al., 1994)
Other-antiasthmatic effects
the water decoction and petroleum ether extract of Viticis Fructus
N/A
isolated tracheal volume measurement method
The water decoction and petroleum ether extract of Viticis Fructus had anti-asthmatic effect.
(Liu, 2002)
Activity
Compounds or extracts
Dose
Model/Diseases
Result/Mechanism
Reference
Other-promote sleep effects
Viticis Fructus water extract
8 g/kg
rat
The effect of V. rotundifolia fruit on promoting sleep was stronger than that of V. trifolia fruit.
(Zhong et al., 1996)
Other-pulmonary fibrosis effects
Viticis Fructus formula granules
0.025 g/mL
pulmonary fibrosis induced by bleomycin in rat
Viticis Fructus could intervene pulmonary fibrosis.
(Tian, 2017)
Other-osteoporosis effects
casticin
0.125, 0.25 and 0.5 μM
RAW 264.7 cell
Inhibited the differentiation of RAW 264.7 cells into osteoclasts.
(Huang, 2022)
6.1 Antitumor
The extract and its main compounds of Viticis Fructus had different degrees of inhibitory and preventive effects on cervical cancer, nasopharyngeal carcinoma, lung cancer, breast cancer and other cancers (Fig. 7, Fig. 8 and Fig. 9). Helianane diterpenoids from Viticis Fructus slowed down cell division in human chronic myeloid leukemia K562 cells and mouse breast cancer tsFT210 cells, resulting in apoptosis of cancer cells (Li et al. 2005b). The flavonoids of Viticis Fructus, such as persicogenin, artemetin and luteolin, restrained G2/M of cell cycle progression and induced mammalian cancer cell death (Li et al., 2005a). However, the anti-tumor effect of Viticis Fructus was mostly in vitro tests and was rarely reported in vivo tests. The effect should be further evaluated in combination with clinical research.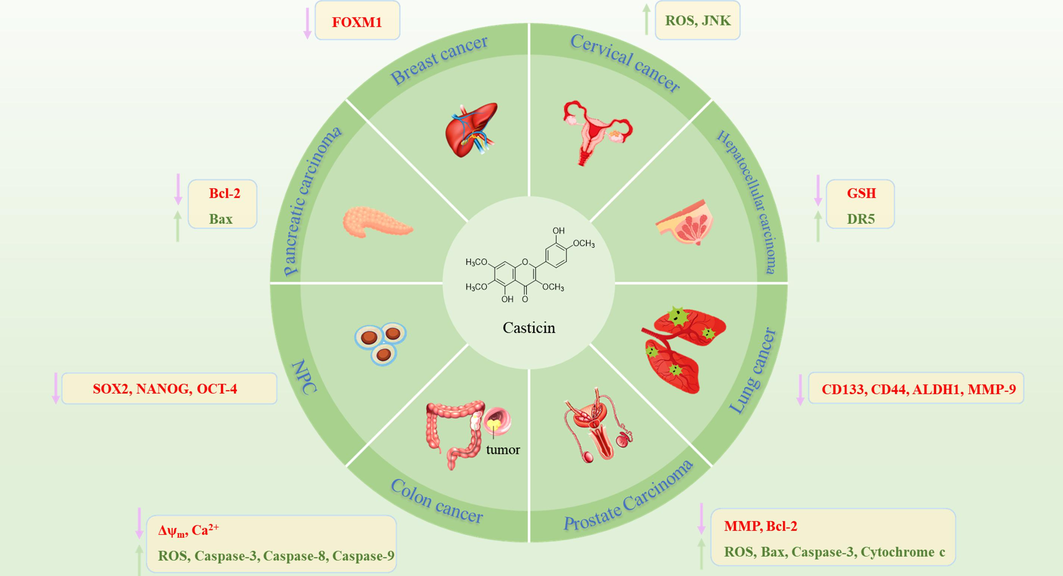
Antitumor activity of casticin.
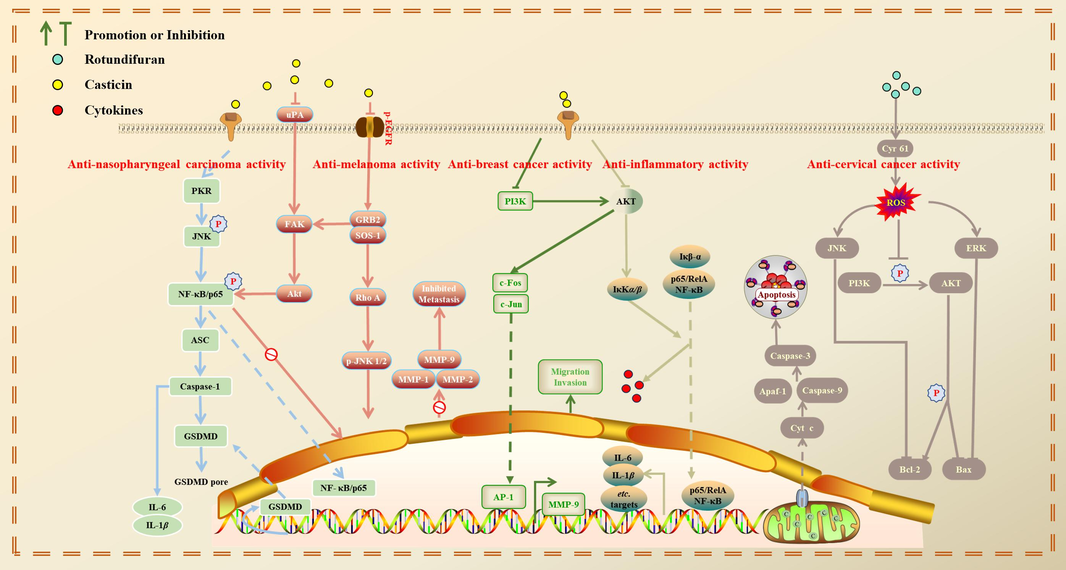
The molecular mechanism of the active components of Viticis Fructus on anti-nasopharyngeal cancer, anti-cervical cancer, anti-breast cancer and anti-inflammatory.
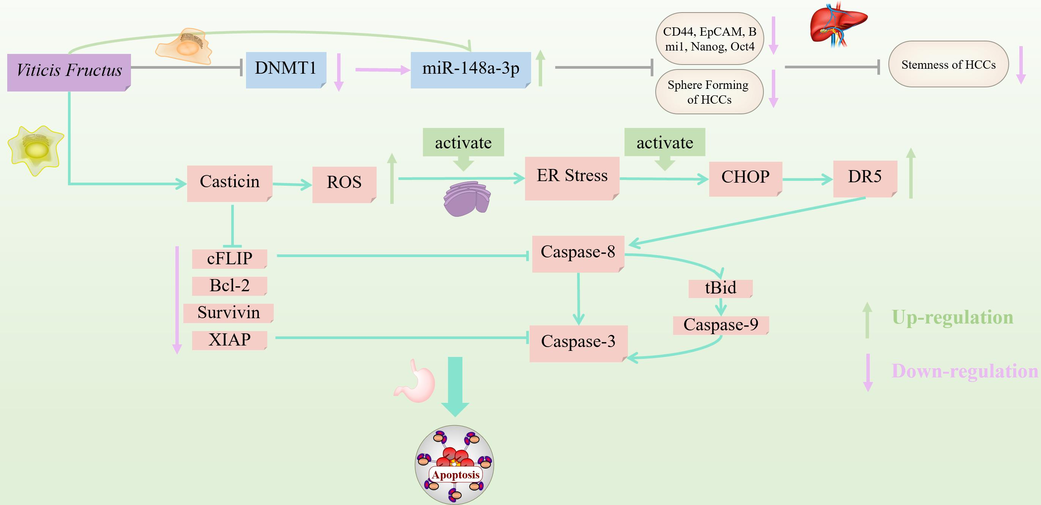
Changes of biological indexes of anti-hepatocarcinoma and anti-gastric cancer of Viticis Fructus.
6.1.1 Breast cancer
Breast cancer cell multiplication in rats was successfully halted by the acetic acid extract of Viticis Fructus. The acetic acid extract of Viticis Fructus had a good inhibitory effect on the proliferation of breast cancer cells in rats (Guan, 2011). Casticin was of great significance for the development of breast cancer treatment drugs (Li et al., 2005a). There were three main conclusions about the therapeutic effect of casticin on breast cancer. (1) Casticin could act directly on cyclin A. Finally, the down-regulation of the anti-apoptotic protein B cell lymphoma-2 protein (Bcl-2) led to apoptosis (Haïdara et al., 2006). (2) Forkhead box O3 (FOXO3a) was a crucial mediator for casticin inducing apoptosis of breast cancer cells (Liu, 2014). (3) It inhibited the expression of matrix metalloproteinase-9 (MMP-9) via curbing phosphatidylinositol 3-kinase/protein kinase B (PI3K/AKT) signaling pathway, preventing breast cancer cells from migration and invasion (Fan et al., 2018). Therefore, Viticis Fructus could prevent and treat breast cancer by blocking G2/M cell growth, acting on FOXO3a target and PI3K/AKT pathways.
6.1.2 Cervical cancer
The PI3K/AKT pathway is a key pathway for the treatment of cervical cancer by Viticis Fructus. The hot water extract of Viticis Fructus exhibited inhibition on the cervical cancer cells (Jiangsu New Medical College, 1977). Rotundifuran and casticin had been proven effective in treating cervical cancer (Chen et al., 2011). It was found that rotundifuran dramatically repressed the propagation and invoked apoptosis of cervical cancer cells. The mechanism was that it bound to cysteine-rich61 (CYR61) protein to promote the expression of CYR61, inhibited PI3K/AKT and extracellular regulated protein kinases (ERK) pathways, activated the c-Jun N-terminal kinase (JNK) pathway, up-regulated Bcl-2 interacting mediator of cell death (Bim) and Bcl-2 Associated X Protein (Bax), down-regulated Bcl-2 and B-cell lymphoma extra-large (Bcl-xL), thereby enabled activation of the mitochondrial apoptosis pathway and causing cell death (Shen, 2020). Rotundifuran inhibition of cervical cancer cell proliferation was related to mitochondrial apoptosis. Reactive oxygen species (ROS) invoked mitochondrial-dependent apoptosis via mitogen-activated protein kinase (MAPK) and PI3K/AKT (Gong et al., 2021). ROS and JNK could be regulated by casticin in human cervical cancer cell lines, thereby increasing intracellular ROS, promoting the expression of C-Jun protein and phosphorylated JNK to induce apoptosis (Zeng et al., 2012).
6.1.3 Lung cancer
It has been found that reducing the phosphorylation level of AKT is the main way for Viticis Fructus to alleviate lung cancer by numerous studies. The lung cancer stem-like cells (LCSLCs) of the NCI-H446 cell line could be restrained in terms of their function and characteristics by the total flavonoids of Viticis Fructus. The mechanism of action involved the reduction of AKT phosphorylation level in LCSLCs, thereby inhibiting stem cell markers (CD133, CD44 and ALDH 1), self-renewal transcription factors (Bmil, Nanog and Oct4), invasion-related factors (Twist 1 and Snail 1) and protein expression (Cao et al., 2014). Another experiment also showed that down-regulation of p-AKT inhibited the invasion and self-renewal of LCSLCs in A549 cells, which had a certain delaying effect on lung cancer cells (Liu et al., 2014a).
6.1.4 Liver cancer
The extract of Viticis Fructus (the fruits of V. rotundifolia) had some preventive action on liver cancer cell H22. The mice tumor weight in the group receiving high-dose treatment markedly showed less than that of the control group (normal saline treatment) (Yan et al., 2023). Moreover, Viticis Fructus induced apoptosis by reducing glutathione content in human hepatocellular carcinoma (HCC) cells, up-regulating death receptor 5 (DR5) and then activating cysteinyl aspartate specific proteinase-3, -8 and -9 (caspase-3, -8 and-9) (Yang et al., 2011). It could cause forkhead box protein M1 (FOXM1) inactivation in HCC cells by inhibiting FOXO3a phosphorylation to induce growth repression and cell cycle stagnation (He et al., 2013). With the deepening of research, it had been found that casticin could effectively eradicate liver cancer stem cells through the β-catenin target (He et al., 2014). It could additionally suppress the stemness characteristics of HCC cells via interfering with the mutual negative adjustment between miR-148a-3p and DNA methyltransferase 1 (Li et al., 2020a).
6.1.5 Colorectal cancer
Viticis Fructus could potentially become a candidate herb for developing chemoprevention or therapeutic drugs for human colorectal cancer (CRC). The ethanol extracts of V. rotundifolia fruits inhibited the proliferation of human CRC cells through down-regulating Cyclin D1 and cyclin-dependent kinases-4 (CDK4) (Yan et al., 2023). Casticin had significant proliferation inhibitory activity on HCT116 (human CRC cells) (Ono et al., 2002). It could enhance TRAIL-induced apoptosis in human CRC cells. On the one hand, it could down-regulate cell survival proteins, including X-chromosome-linked Inhibitor of apoptosis (IAP), cellular IAP1 and so forth. Furthermore, it could also up-regulate the expression of Bax and induce DR5 (Tang et al., 2013). It could also induce apoptosis in human Colo 205 (CRC cells) by activating caspase and/or mitochondrial-dependent signaling cascades, ROS accumulation and altering the expression of related genes (Shang et al., 2017).
6.1.6 Nasopharyngeal carcinoma
Viticis Fructus decoction could be used to treat nasopharyngeal carcinoma (Li, 2003). Casticin suppressed the expansion of nasopharyngeal carcinoma (NPC) (Jiang et al., 2020). It inhibited the growth of NPC by targeting phosphoinositide 3-kinase (Liu et al., 2019). Inhibition of 5-8F cell (human NPC cell lines) proliferation via triggering cell cycle stagnation and pyroptosis, protein kinase R (PKR) /JNK axis was pivotal in caspase-1 inflammasome and the release of inflammatory cytokines (Jiang et al., 2020).
6.1.7 Other cancers
It was also confirmed that Viticis Fructus had the potential to treat pancreatic cancer, glioma, gastric cancer, prostate carcinoma, melanoma, leukemia, etc. Viticis Fructus had significant inhibitory activity on PC-12 proliferation (human lung cancer cells) (Ono et al., 2002). It greatly repressed the proliferation of PANC-1 (pancreatic carcinoma cells), mainly by arresting G2/M of the cell cycle, regulating the proportion of Bax/Bcl-2 and triggering apoptosis by activating caspase-3 (Ding et al., 2012; Huang et al., 2013). Casticin in Viticis Fructus suppressed U251 (human glioma cells) in a dose-dependent manner, which could control the polymerization of tubulin in U251 cells, block cells in G2/M phase and had a p53 and Caspase-3-dependent effect on apoptosis (Liu et al., 2013). Casticin could enhance the apoptosis of gastric cancer cell lines through endoplasmic reticulum stress. The mechanism involved the down-regulation of cell survival protein and the up-regulation of the DR5 (Zhou et al., 2013). Another study confirmed that the active compounds in Viticis Fructus were a potential leading drug in the therapy of prostate carcinoma. Casticin induced apoptosis of PC-3 (prostate cancer cells). The mechanism involved blocking G2/M phase, increasing intracellular ROS, decreasing membrane potential of mitochondria, freeing cytochrome C, initiating Caspase-3, up-regulating pro-apoptotic protein Bax, down-regulating anti-apoptotic protein Bcl-2 and intracellular cyclins Cyclin B1 and CDK1 (Meng et al., 2012). Rotundifuran, 2′, 3′, 5-trihydroxy-3, 6, 7-trimethoxyflavone, casticin and artemetin could inhibit the multiplication of HL-60 cells (human myeloid leukemia) (Ko et al., 2000; Ko et al., 2001). Furthermore, casticin has been proven in vivo experiments that could promote the immune response in leukemia mice, thereby increasing the survival rate of leukemia mice (Lai et al., 2019). In B16F10 cancer cells (mouse skin melanoma cells), casticin arrested the expression of the p-EGFR, AKT and Nuclear factor kappa-B (NF-κB) pathways, which led to the suppression of MMP-9, MMP-2 and MMP-1(Shih et al., 2017).
6.2 Anti-inflammation
The anti-inflammatory activity of Viticis Fructus mainly involves NF-κB, MAPK and AKT signaling pathways (Fig. 8). There was a certain gap in the anti-inflammatory activity of different extraction parts of Viticis Fructus, among which the n-butanol fraction had better activity. It was speculated that terpenoids and flavonoids, such as Viteagnusin I, Vitetrifolin D and others, inhibited the production of nitric oxide (NO) (Yan et al., 2023). Viterotulin C, Vitexilactone and Rotundifuran had anti-inflammatory activity and suppressed tumor necrosis factor-α (TNF-α)-induced NF-κB activation. The inhibition rate was 42.52 % − 68.86 % at a concentration of 50 μM (Fang et al., 2019). Casticin could repress the chemotaxis of cultured human neutrophils and produce anti-inflammatory effects (Ahmad et al., 2010). It attenuated the generation of pro-inflammatory cytokines interleukin-1β (IL-1β), interleukin-6 (IL-6) and TNF-α by blocking NF-κB, AKT and MAPK signaling pathways, thereby inhibiting NO and Prostaglandin E2 levels and exerting anti-inflammatory activity (Liou et al., 2014).
Viticis Fructus played a therapeutic role in many diseases through anti-inflammation, such as systemic anaphylaxis reaction, allergic asthma, ulcerative colitis (UC), knee osteoarthritis (KOA), vascular inflammation, etc. Its extract and monomer components had been proven to have certain anti-inflammatory activities. The water extract (the fruits of V. rotundifolia) and pyranopyran-1,8-dione (PPY) had protective effects on systemic anaphylaxis reaction, pulmonary inflammation and asthma. They restrained secretion of inflammatory factors by declining the activation of ERK1/2 and NF-κB signaling pathways, such as TNF-α and IL-6 (Yan et al., 2023). Another study found that casticin was a prominent immunomodulator that improved the pathology by inhibiting the expression of T-helper two-cell cytokines in asthmatic mice (Liou et al., 2018). Casticin was also a potential remedy for KOA, particularly for fibrosis of synovial membrane. It reduced the release of inflammatory mediators and the elevation of fibrosis markers induced by monoiodoacetic acid/lipopolysaccharide through suppressing the stimulation of nucleotide oligomerization domain-like receptor protein 3 (NLRP3) inflammasome (Chu et al., 2020). In addition, casticin was able to repress ROS-mediated NF-κB pathway to achieve marked relief of cartilage degeneration associated with experimental osteoarthritis (Chu et al., 2020). The protective effect of casticin on dextran sulfate sodium-induced UC was achieved by increasing the expression of antioxidant enzymes peroxiredoxin three and Mn-containing superoxide dismutase, and reducing the generation of proinflammatory cytokines via inhibiting AKT pathway (Ma et al., 2018). Casticin could markedly decrease vascular inflammation by inhibiting the NF-κB pathway of vascular endothelium cells (Lee et al., 2012).
6.3 Antioxidation
Viticis Fructus was discovered good antioxidant activity and individual compounds were even better than vitamin C in antioxidant activity. The compounds (such as vanillic acid and taxifolin) in the methanol extract of V. rotundifolia fruits showed more potent antioxidant action over 3-tert-butyl-4-hydroxyanisole (Ono et al., 1998b). The scavenging ability of alkaloids, total flavonoids and volatile oil on hydroxyl radicals and superoxide radicals displayed a significant dose–effect relationship with their concentrations (Yan et al., 2023). Ferruginol and vitrifolin A had certain antioxidant activity (Ono et al., 1999; Zhang et al., 2013). The superoxide quenching activity of orientin and 3, 4-di-O-caffeoylquinic acid was more powerful than that of vitamin C (Kim, 2009). In addition, it was found that 3,4-dihydroxybenzaldehyde, chlorogenic acid and orientin showed strong DPPH free radical scavenging ability (Le et al., 2022).
6.4 Premenstrual syndrome
The pathogenesis of PMS was generally believed to be the result of a combination of psychosocial factors and endocrine regulation, such as prolactin, ovarian hormones and brain neurotransmitter imbalance (Ye, 2010). Pharmacological studies had found that ethanol extract of V. rotundifolia fruits was used to alleviate PMS regulating prolactin levels, estrogen and progesterone levels (Hu, 2007). First, directly reduced the excessive estrogen content in the body so that the estradiol/progesterone ratio decreased. Casticin could reduce the content of estradiol in serum. At the same time, rotundifuran could increase serum progesterone content. Second, Decreased the content of β-endorphin in the hypothalamus. Third, Casticin could significantly reduce the content of prolactin (Ye, 2010).
6.5 Cardiovascular protection
Viticis Fructus had certain therapeutic effects on some cardiovascular diseases, such as hypertension, hyperlipidemia and coagulopathy. Viticis Fructus had an antihypertensive effect. The blood pressure of cats decreased significantly after administration of Viticis Fructus decoction and Viticis Fructus ethanol extract (Guan, 2011). Its extract could significantly prolong the time of bovine thrombin condensing human fibrinogen in vitro, indicating that it had a strong anticoagulant effect (Ou et al., 1987). Casticin could inhibit platelet aggregation, release, adhesion and plaque retraction, which was achieved by inhibiting PI3K/Akt signaling pathway (Xiong et al., 2022). Furthermore, V. rotundifolia fruits extract had strong activities toward the oxidation of low-density-lipoprotein (LDL) and high-density lipoprotein (HDL) (Kim et al., 2020). Viterofolin H, previtexilactone and other compounds indicated moderate activities in facilitating LDL uptake. These compounds showed certain anti-hyperlipidemic activity (Wang et al., 2019a).
6.6 Other activities
In addition to the above effects, Viticis Fructus can also be used for analgesic, antipyretic, expectorant, asthma, whitening, antibacterial, anti-fatigue, etc. It is a TCM with an analgesic effect. The methanol extract of V. trifolia fruits could effectively relieve migraines, among which the flavonoid fraction has a good analgesic response inhibition rate. Pedicularis-lactone, viteoid I and viteoid II significantly suppressed paclitaxel-induced mechanical allodynia. This is related to the levels of 5-hydroxytryptamine and γ-aminobutyric acid (GABA) increased, and the decrease of plasma calcitonin gene-related peptide and substance P levels (Yan et al., 2023). Crude and processed V. rotundifolia fruits products had an apparent antipyretic effect (Yan et al., 2023). It had expectorant and antiasthmatic effects. An obvious expectorant effect could be observed by phenol red excretion method in mice after oral administration of water decoction and alcohol extract of Viticis Fructus. The water decoction and petroleum ether extract of Viticis Fructus could relax the isolated guinea pig tracheal specimens. It could slow histamine-induced tracheal contraction (Liu, 2002). This was mutually supportive with its traditional efficacy in treating wind-heat colds (upper respiratory tract infections). Aldolase role of the rat lens was strongly inhibited by ether extract of V. rotundifolia fruits (Shin et al., 1994). Viticis Fructus water extract could also promote sleep in mice (Zhong et al., 1996). Viticis Fructus showed an effect against tyrosinase, indicating that it was able to inhibit the formation of melanin (Wang, 2003). Viticis Fructus water decoction had a moderate antibacterial effect on staphylococcus epidermidis in vitro and had moderate antibacterial effect on bacillus subtilis (Guan, 2011). Viticis Fructus could also interfere with pulmonary fibrosis by inhibiting apoptosis and improving excessive angiogenesis (Tian, 2017). Viticis Fructus could be used as a potential anti-menopausal osteoporosis drug. Within the safe range of casticin, it could inhibit the differentiation of RAW 264.7 cells into osteoclasts through the NF-κB/BCL-2 signaling pathway (Huang, 2022).
Viticis Fructus can trigger the apoptosis of various tumors and cancer cells to prevent and treat cancer with respect to antitumor activity. Many studies have manifested that Viticis Fructus induced apoptosis by regulating JNK, NF-κB, PI3K/AKT, ERK and other pathways. The hot water extract, ethanol extract, terpenoids and flavonoids of Viticis Fructus are the main components of its function. Among them, casticin can modulate key targets and pathways of breast cancer, cervical cancer, nasopharyngeal carcinoma and other cancers. It needs further research and may be used as a potential new drug to inhibit cancer cells. The anti-inflammatory properties of various Viticis Fructus fractions varied to some extent in terms of anti-inflammatory effects. The optimal extraction solvent should be selected to ensure better anti-inflammatory activity of Viticis Fructus. It could prevent the formation of anti-inflammatory proteins by modulating the NF-κB, AKT and MAPK signaling pathways, thereby reducing inflammation and curing various illnesses. However, the mechanism of many diseases was still unclear, such as systemic allergic reactions. In addition, the anti-rheumatic effect of Viticis Fructus was mentioned in many books such as Bencao Gangmu and Shennong Bencao Jing, which was mutually corroborated with pharmacological research, but the specific action mechanism should be further studied. Flavonoids and terpenoids in Viticis Fructus manifested favorable antioxidant activity in vitro. Oxidative-related diseases should be validated at the cellular and animal levels for better clinical application of Viticis Fructus. In the area of female diseases, Viticis Fructus had a good preventive and therapeutic effect on female illness by regulating hormone levels such as prolactin, estrogen and progesterone. In addition to improving the symptoms of PMS, it could also be used to relieve hyperprolactinemia and menopausal symptoms (Yan et al., 2023). The mechanism of Viticis Fructus in cardiovascular disease is still unclear. Further experiments in vivo are needed to prove its efficacy and clarify the corresponding mechanism of action. This can expand its use scope and provide a new direction for the treatment of cardiovascular.
7 Toxicity
Adverse reactions and toxins of TCM are issues that need constant attention to ensure the safety of TCM. Toxicological experiments were performed on manjing oral liquid with Viticis Fructus as the primary raw material, including median lethal dose (LD50), mutagenicity test (mouse bone marrow micronucleus test, ames test and mouse sperm deformity test). It emerged that the LD50 exceeded 20 g/kg, demonstrating non-toxicity. Negative results of three mutagenicity tests demonstrated the absence of mutagenic effects (Zhang et al., 2000). Continuous administration of aqueous extracts of Viticis Fructus and Scrophulariae Radix (50 or 100 mg/kg) had no systemic toxicity in normal mice (Kim and Ma, 2019). The mice survived after administration of the ethanol extract and aqueous extract of Viticis Fructus with exceeding the clinical dose, which implied a low level of toxicity (Wang et al., 2008). The impact of varying Viticis Fructus volatile oil concentrations on the survival rate of HaCaT cells (Human immortalized keratinocytes) was approximately concentration-dependent. Viticis Fructus essential oil (285.10 μg/mL) had a half-inhibitory concentration that was above that of azone (17.08 μg/mL), a positive penetration enhancer. It showed that Viticis Fructus volatile oil has low irritation to the skin. Only excessive use can damage skin cells. 0.5 %, 1 % and 2 % Viticis Fructus essential oil had no irritation symptoms such as erythema and edema on the intact skin of guinea pigs after repeated administration (Liang, 2019).
Viticis Fructus has a low degree of toxicity and it rarely causes harm to the body unless improperly used. However, although studies have indicated that Viticis Fructus has a low toxic level, its toxicological research is relatively weak. Toxicological studies on mice only are one-sided. Toxicological research should be carried out on different animals and different doses should be explored to ascertain the toxic range. In addition, the toxic reactions and toxicity mechanism of Viticis Fructus have not been elucidated.
8 Quality control
Quality control is the basis for ensuring the stability and safety applications of TCM. Therefore, different scientific and technological means were applied to evaluate and control the quality of TCM. The quality control of Viticis Fructus covered the authentication of TCM, physical and chemical identification, microscopic identification, impurities, moisture, total ash, extract and content determination. Presently, the main methods for evaluating the quality of Viticis Fructus were ultraviolet–visible (UV) spectrometry, inter simple sequence repeat (ISSR), Near Infrared (NIR), Ultra-performance liquid chromatography-mass spectrometry (UPLC-MS) analysis, UPLC-Orbitrap-MS, comprehensive two-dimensional gas chromatography hyphenated with mass spectrometry (GC × GC–MS), liquid chromatography/mass spectrometry (LC/MS) analysis and high-performance liquid chromatography coupled with a diode array detector (HPLC-DAD). However, the research on the quality control of Viticis Fructus is relatively simple, mostly for content determination and counterfeit identification. There is an urgent need to improve the relevant contents of quality control for Viticis Fructus and then comprehensively evaluate the quality Viticis Fructus.
The quality control of different origin plants, processed products and batches of Viticis Fructus was carried out by content determination. Viticis Fructus chemical constituents before and after processing were distinguished by NIR, HPLC and UPLC-MS analysis. Forty-two of the detected compounds underwent considerable alteration during processing and sixteen chemical constituents were picked as distinctive markers. Crude and processed Viticis Fructus could be successfully identified using these techniques (Diao, 2018). The contents of casticin and agnuside were evidently varied in the two origin plants by establishing the HPLC fingerprint of Viticis Fructus. The content of casticin in V. rotundifolia fruits was higher, while the content of agnuside in V. trifolia fruits was higher. There was no significant difference in the content of isoorientin and p-hydroxybenzoic acid (Yang et al., 2023). The comparative study of Viticis Fructus and the fruits of Vitex negundo was carried out by UPLC characteristics chromatogram combined with multi-index content determination. Compared with the fruits of Vitex negundo, orientin and isovitexin were not detected in Viticis Fructus (Li et al., 2023). It is worth mentioning that the significant polarity differences among the chemical constituents of Viticis Fructus make simultaneous extraction challenging. Using the Beta/ZSM-22 Zeolites-Based-Mixed Matrix Solid-Phase Dispersion approach, eight chemicals (agnuside, DHTMF, vanillin, etc) from Viticis Fructus were extracted and measured concurrently. Compared to the conventional approaches, achieving the highest extraction rate is simple (He et al., 2019).
The adulteration of Viticis Fructus often appeared in the market, which in the market circulation are the fruits of Vitex negundo, Euphorbiae Semen, Litseae Fructus and so on. Their appearance traits are very similar and difficult to distinguish (Zhou and Jin, 2001). Based on the differences in the maximum intraspecific Kimura 2-Parameter (K2P) genetic distance of Viticis Fructus and its adulterants, the Internal Transcribed Spacer 2 (ITS2) sequence method was used to successfully distinguish the adulteration. The maximum intraspecific K2P genetic distance of Viticis Fructus based on ITS2 sequence was less than the minimum interspecific K2P genetic distance with adulterants (Zhang et al., 2014). Shrub Chaste Tree Fruits (SCTF, synonyms of Viticis Fructus)-specific marker compounds (3-O-trans-feruloyl tormentic acid) were identified by LC/MS metabolic analysis, then accurately identified their origin plant and clearly distinguished between SCTF and Agnus Castus fruit (Yahagi et al., 2016). In addition, the stereoscope and scanning electron microscope were adopted to compare the microscopic and ultramicroscopic characteristics of the peel surface of Viticis Fructus and its adulterants. It was found that the surface ultrastructure of Viticis Fructus and its adulterants were significantly different, which could be used as a basis for identification. Its surface was reticular, while the surface of the counterfeit showed other traits (Cai et al., 2017). UPLC-Orbitrap-MS and GC × GC–MS were used to select betulinic acid, myricetin and volatile 4-(2,2,6-trimethyl-bicyclo [4.1.0] hept-1-yl)-butane-2-one as specific markers to successfully distinguish Viticis Fructus and its adulterants (Vitex cannabifolia fruits, Vitex negundo fruits, Piper cubeba fruits, Euphorbia lathyris seeds and Vaccinium bracteatum fruits) (Li et al., 2020c). Moreover, the HPLC fingerprinting and ISSR molecular markers methods were built to study the relationship between the intraspecific variation and the chemical composition diversity of Vitex negundo var. heterophylla. It was found that the influence of different genetic backgrounds cannot be ignored. Combined with chemical and genetic diversity, chemical type selection could be carried out for different efficacies of Viticis Fructus to achieve quality control (Hu et al., 2010).
The quality control of Viticis Fructus primarily centered on content determination and counterfeit identification. There are few studies on the identification of the two origin plants of Viticis Fructus, but the fruits of V. rotundifolia are more used than the fruits of V. trifolia. There is a big gap between the two origin plants in the content of compounds such as casticin, agnuside and other compounds. In addition, there are many counterfeits and a simpler method for identifying counterfeits should be developed.
9 Pharmacokinetics
The active ingredients of TCM are in a dynamic process of change after intake into the human body (Ma et al., 2023). Studying the process of drug concentration in the blood over time is indispensable for interpreting the scientific connotation of TCM. Pharmacokinetics is a powerful tool for promoting the scientific development of TCM. The pharmacokinetic studies of Viticis Fructus in various aspects are also constantly enriched, providing the scientific basis for the development and utilization of Viticis Fructus.
The main pharmacokinetics of Viticis Fructus include the comparison of water extract and alcohol extract of Viticis Fructus, the comparison of oral and intravenous injections of casticin, the comparison of different processed Viticis Fructus and the metabolic process of casticin in Viticis Fructus. UHPLC-MS/MS was used to analyze and determine agnuside, casticin, luteolin and 10-O-vanillyl eucommoside in rat plasma after oral administration. It was successfully used to evaluate the pharmacokinetic characteristics of four active compounds in water extract and ethanol extract of Viticis Fructus in rat plasma. Agnuside in the water and alcohol extracts showed a large difference and the water extract was absorbed faster. 10-O-vanilloylaucubin was not detected in rats after oral administration of alcohol extract. Its absorption in aqueous extract is faster, but the absorption concentration in vivo was still at a low level. The absorption of luteolin in the two extracts was poor. The maximum drug concentration in plasma (Cmax), the time to reach maximum drug concentration (Tmax) and area under the plasma concentration–time curve (AUC (0-t)) of water extraction were 6.37 ± 3.61 ng/mL, 0.25 ± 0.13 h and 5.14 ± 1.85 ng/mL*h, respectively. The Cmax, Tmax and AUC (0-t) of alcohol extraction were 8.14 ± 5.45 ng/mL, 0.19 ± 0.17 h and 11.5 ± 3.76 ng/mL*h, respectively. The content of casticin in the two extracts was so low that the relevant parameters could not be calculated (Chen et al., 2021). LC-MS was used to quantify casticin content in rat plasma following oral and intravenous injections. The Cmax, Tmax and AUC (0-t) were 287.06 ± 40.68 ng/mL, 43.83 ± 1.47 min and 18,652.72 ± 4030.88 ng/mL*h, respectively; for taking orally casticin 400 mg/kg to rats. The Cmax, Tmax and AUC (0-t) were 12,737.82 ± 7243.88 ng/mL, 0 min and 41,225.92 ± 1403.37 ng/mL*h, respectively, for intravenous administration of 50 mg/kg of casticin. It was rapidly distributed and eliminated in rats because casticin is a polyhydroxy flavone that is easily hydroxylated in plasma resulting in a short half-life (Xu et al., 2012). In another study, the pharmacokinetic effects of casticin and isoorientin in rats before and after processing Viticis Fructus showed no clear distinction between the crude and processed Viticis Fructus after oral administration. The Tmax and Cmax of casticin were about 5 min and 20 μg/L, respectively. The Tmax of isoorientin was 1.50 ± 0.39 h and 1.75 ± 0.39 h, respectively (Yu, 2019). In addition, the metabolic process of casticin in vivo was also studied. 25 metabolites and main metabolic pathways were speculated. One is demethylation process, the other is methylation, sulfation and glucuronidation process (Zhu, 2013).
The pharmacokinetic study of Viticis Fructus focused on healthy rats. Other animal models should be added, such as dogs, rabbits and even healthy volunteers. In addition, the disease models should be established to compare with the normal group, reflecting the changes of Viticis Fructus metabolism in the disease state and promoting the better application of Viticis Fructus in clinical practice. There had been some investigating on absorption and metabolism of Viticis Fructus, but little was known about its distribution or excretion. It is crucial to gaze at the distribution and excretion kinetics of Viticis Fructus in vivo.
10 Comprehensive applications
10.1 Clinical application
Viticis Fructus is a commonly used TCM in clinics. It is used for treating various diseases combined with other herbs, such as nervous system disorders, eye diseases, otolaryngology diseases, digestive system diseases and so on (Fig. 10).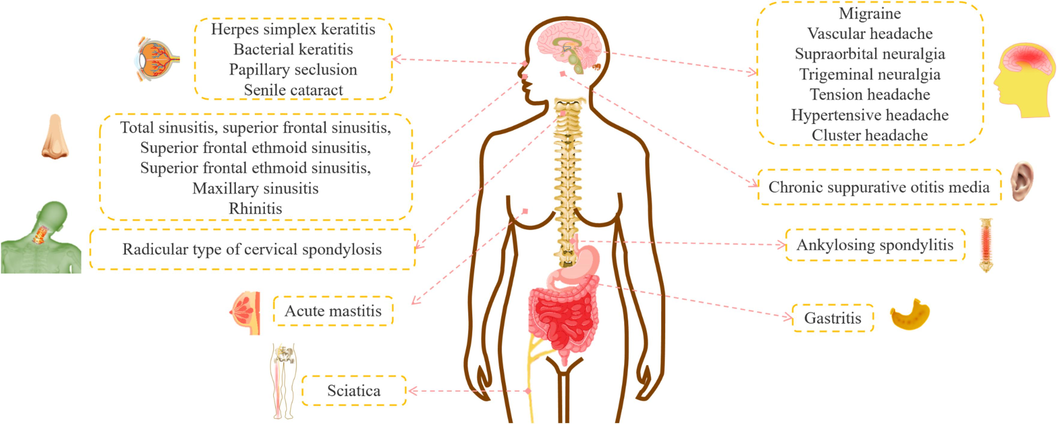
The application of prescriptions containing Viticis Fructus in clinical diseases.
In the nervous system disorders, it was recorded in the ancient book that all headaches could be treated with Viticis Fructus., regardless of the left and right sides (Gong, 1999; Tian and Shi, 2022). It combined with other TCM in the treatment of headaches and curative effect was favorable. For example, Viticis Fructus combined with Bupleuri Radix, Chuanxiong Rhizome and Chrysanthemi Flos to treat migraine, played a role in clearing heat and relieving pain (Zhao, 2018; Niu et al., 2005). Neurovascular headache, tension headache, hypertension-related headache, trigeminal migraine, migraines and cluster headache could be treated with Chrysanthemi Flos, Notopterygii Rhizome et Radix, Scutellariae Radix, etc (Hu and Yang, 2016). One hundred and twenty patients with migraine were cured with Viticis Fructus Tou-Feng Decoction (Chrysanthemi Flos, Uncariae Ramulus Cum Uncis, Menthae Haplocalycis Herba, etc) for 10 days (twice a day). Headache symptoms disappeared in 92 patients. The degree of headache attack in 26 patients was significantly anesis and the frequency decreased. The total effective rate was 98.3 %. The difference was significant compared with the total effective rate of 88.3 % in the control group (intravenous ligustrozine hydrochloride injection, 60 volunteers). In addition, the recurrence rate was lower in the treatment group (10.87 %) than in the control group (32.43 %). Compared with the two groups, the treatment group was significantly better than the control group. Viticis Fructus mainly played an analgesic role in the prescription (Xu, 2007). Clinically, Viticis Fructus could be used alone to treat sciatica (Wang, 2001) and trigeminal neuralgia (Li, 1998). Fifty-six patients with sciatica were treated with Viticis Fructus juice twice a day for 21 days. The total effective rate was 98.2 %. Primary sciatica was mostly related to rheumatism, cold and other reasons. Viticis Fructus combined with liquor had a fine effect of expelling wind, removing dampness and cold-dispelling effect, so the effect is obvious (Wang, 2001).
In Otorhinolaryngological diseases, 110 patients with chronic suppurative otitis media were treated with Viticis Fructus Decoction (Cimicifugae Rhizoma, Peucedani Radix, Mori Cortex, etc) and Hong-Mian powder. Thirty-eight cases were treated with Western medicine (antibiotics and steroid hormones). The curative effect of TCM group was significantly better than that of the Western medicine group. The cure rate was better in the Chinese medicine group (89.1 %) compared with the Western medicine group (86.8 %). And the recurrence rate of the Chinese medicine group (1.8 %) was significantly lower than that of the Chinese medicine group (31.6 %) (Guo and Wang, 2003). Viticis Fructus, Eupatorii Herba, Angelicae Dahuricae Radix and Xanthii Fructus were applied clinically for the treatment of rhinitis, sinusitis, ethmoid sinusitis and maxillary sinusitis (Guo, 2006). It was worth mentioning that Viticis Fructus also had a good therapeutic effect on gastritis. Four groups were set up, namely superficial gastritis with Weishu No.1 (Angelicae sinensis radix, Paeoniae radix alba and so on) plus Viticis Fructus 20–30 g, atrophic gastritis with Weishu No.2 (Lilii bulbus, Codonopsis radix) plus Viticis Fructus 20–30 g and the control groups without the addition of Viticis Fructus. Patients were monitored and had a repeat gastroscopy after 2–3 sessions (2–3 months). The heartburn, distension and pain disappeared on average 14.3 days in the treatment group and the control group 26.1 days on average. Gastroscopy results in patients with gastritis displayed an efficacy rate of 91.7 % in the treatment group and the control group was 74 %. Viticis Fructus played an analgesic and anti-inflammatory role in the prescription (Huang and Wen, 2000). Combined external application of Viticis Fructus powder and yellow wine had a good therapeutic effect on 19 patients with acute mastitis. The clinical symptoms of 17 patients disappeared and the total number of white blood cells and neutrophils in blood tests were normal. The symptoms and signs of two patients were significantly reduced. The total number of white blood cells and neutrophils was close to normal (Xiang, 1999). In addition, it could also treat eye diseases and rheumatism. Viticis Fructus prescription could cure herpes simplex keratitis, bacterial keratitis and papillary closed keratitis. It plays the role of heat dissipation, dispelling wind and relieving pain (Jin, 2018). Qiang-Huo-Sheng-Shi Decoction containing Viticis Fructus could improve symptoms of knee osteoarthritis (Wang, 2021), ankylosing spondylitis (Yang et al., 2021) and lower back pain (Lin, 2019) in clinical practice. It helped to relieve pain of patients.
10.2 Other applications
Viticis Fructus is used in cosmetics, healthcare products and daily necessities (Table S6). For example, Viticis Fructus could be made into medicine pillows, health tea, health liquor, beverages and other health care products. It can be applied to lowering blood pressure, lowering blood lipid, lowering blood sugar, improving menopausal syndrome, hair growth, benefits qi and so on (Gao et al., 2022; Park, 1999; Shi, 2016). In cosmetics and daily necessities, casticin, as a sunscreen ingredient in cosmetics, could reduce the use of current chemical sunscreens and play a beneficial role in synergistic sunscreen effects (Wang et al., 2019b). Longanae Arillus and Viticis Fructus extract had excellent effects on promoting skin elasticity, relieving wrinkles, skin moisturizing and relieving skin problems (Kim et al., 2018). In addition, toothpaste containing Viticis Fructus had significant antibacterial, anti-inflammatory and analgesic effects, which could fundamentally reduce oral problems such as swelling, aching and bleeding of gum (Mu and Liu, 2017). There were two kinds of head care products containing Viticis Fructus. One kind of hair shampoo had an antipruritic effect and the other kind of plant hair dye enhanced the dyeing effect without any toxic or side effects (Yin, 2019; Jiang, 2016). The volatile oil of Viticis Fructus has a strong aroma and could be used as material for blending flavors. The combination of Viticis Fructus and camphor is an efficient aromatic repellent (Xin et al., 2004).
The clinical effect of Viticis Fructus is worthy of recognition, which can be applied to treat migraine, neurovascular headache, tension headache, gastritis, acute mastitis and so on, and achieve splendid therapeutic effects. However, the detection techniques currently in use are gastroscopy and blood sample detection, which is not enough. It should be combined with more advanced medical instruments and technologies to evaluate the indicators of Viticis Fructus in the treatment of various diseases and to reflect the therapeutic effect more comprehensively and accurately. Currently, Viticis Fructus is utilized finitely. We ought to focus on its application in different domains to broaden the use of Viticis Fructus and maybe discover novel approaches to treating intricate illnesses.
10.3 Patent information
Patents could reflect the development status of hot frontier technology and developmental trend of a field to a certain extent. It was closely related to all aspects of product production and was the carrier of scientific research achievements transformation (Chen et al., 2023). More than 2,000 results were obtained by searching on the Baiten Patent Search Platform, Yaozhi Network and CNKI. Baiten patent retrieval platform analyzed the patent-related situation of Viticis Fructus. The main technical field of Viticis Fructus patent was necessary for human life, chemistry, metallurgy, operations, transport, physical, textile and papermaking. The necessities for human life account for the largest proportion among them. Viticis Fructus was most widely studied from 2014 to 2018, among which the application and publication peak in 2015 were 445 and 415 respectively (Fig. S3). The main areas of patent applications for Viticis Fructus were in China and a few other countries were distributed. In China, the patent applications of Viticis Fructus were concentrated in Shandong Province, Anhui Province, Jiangsu Province and Guangxi Province. Patent applications include cosmetics, daily products, health products, medical use and technical methods (Table S6). Cosmetics, daily products and health products are discussed in application in other fields above. It can also be used for antibacterial, antidepressant, treatment of depression and so on in terms of medical use. Patent applications for technical methods focus on quality control and extraction separation, such as component quantification, fingerprint establishment, extraction and separation of total flavonoids.
The patent applications of Viticis Fructus are concentrated in the fields of necessary for human life, chemistry, metallurgy, operations, transport, physical, textile and papermaking in terms of patents. Viticis Fructus is listed as the top grade in the Shennong Bencao Jing and is considered to have favorable tonic, health care and therapeutic properties. In addition, it can be used as food in some countries. Its edible value can be studied more to promote the better use of Viticis Fructus.
11 Conclusion and future perspectives
Viticis Fructus was a commonly used Chinese herbal medicine with a large market demand (Gong, 2016). It has apparent curative effect, low toxins and its plants could be used for coastal sand control afforestation (Liu, 2015). This paper summarizes the botany, traditional use, phytochemistry, pharmacological activity, quality control, pharmacokinetics, comprehensive application and toxicity of Viticis Fructus. In phytochemistry, the established database of chemical constituents of Viticis Fructus contains 324 compounds, among which terpenoids and flavonoids serve as their main representative compounds. Pharmacology has proven that Viticis Fructus could prevent and treat cancer, inflammation, PMS, hyperlipidemia, oxidative damage, fever, swollen gums, etc. In terms of quality control, the current research focused on the content determination of Viticis Fructus and the identification of adulterants. The 2020 edition of ChP recorded that the content of casticin was not less than 0.030 %. Nevertheless, there are still some gaps in the study of Viticis Fructus and further research is required to comprehend and utilize Viticis Fructus more effectively.
First, whether Viticis Fructus to Verbenaceae and whether the origin plant of V. rotundifolia belongs to an independent species needs further study and improvement. Secondly, V. rotundifolia and V. trifolia fruits are very similar in appearance and it is difficult to distinguish them effectively at present. It can be found that the applications of V. rotundifolia fruits are more extensive from Fig. 1 and the actual situation. Therefore, it is indispensable to distinguish V. rotundifolia and V. trifolia fruits. Thirdly, the research on processing methods is relatively weak. On the one hand, the processing research of Viticis Fructus focuses on the total components and individual pharmacological effects. There are few studies on the changes of specific active compounds before and after processing. The effects mainly involve the comparison of individual pharmacological effects, such as analgesic and cold before and after processing. There are differences in analgesia as previously discussed and further research is needed. On the other hand, the processing of Viticis Fructus is still mainly the stir-frying. However, the frying process of Viticis Fructus has no objective frying process parameters and the quality is difficult to guarantee. Fourthly, most of the studies on the antitumor activity of Viticis Fructus are limited to in vitro and the efficacy in vivo remains to be confirmed. Fifthly, the toxicology of Viticis Fructus is relatively weak. Its toxic reaction and toxic mechanism have not been elucidated. Sixthly, the pharmacokinetic study of Viticis Fructus is not comprehensive. The tissue distribution and excretion kinetics of Viticis Fructus are still vacant. Seventhly, ChP only stipulates casticin as the quality evaluation index of Viticis Fructus. However, the content of a single component does not fully represent the overall quality. Eighthly, Modern pharmacology mainly concentrates on the study of casticin. There are many compounds isolated and identified from Viticis Fructus, but only fewer compounds have been studied, such as rotundifuran, vitetrifolin H, artemetin, etc. Their pharmacological activity, active site and action mechanism need to be elucidated. Lastly, Viticis Fructus can be devoted to treat blurred vision, rheumatism, headache, toothache and other diseases according to ancient books. In the Shennong Bencao Jing and Bencao Gangmu, it has been repeatedly emphasized that Viticis Fructus can dispel rheumatism. It is also used in the classic prescription Qiang-Huo-Sheng-Shi Decoction. However, the action mechanism of arthritis is still unclear. In addition, rheumatism is divided into many types and the current experiment is limited to KOA model.
According to the above deficiencies raised, the following views are put forward. The family of Viticis Fructus and varieties of V. trifolia should be unified to promote the mutual development and utilization in varieties and families. The genetic variation patterns and evolutionary history of V. rotundifolia and V. trifolia were revealed by using modern technologies such as nuclear gene sequencing and chloroplast gene sequencing, which laid a foundation for solving the problems of taxonomy and genetic relationship between the two species. Accurate, stable and reliable analytical methods should be established to discriminate these two origin plants and ensure the medication quality. The difference in composition and curative effect of different processed products needs to be further studied in vivo and in vitro. Reliable and stable quality markers should be found to distinguish different processed products via metabolomics, cluster analysis and other technical means. The analgesic effect was accurately and reliably evaluated by establishing several analgesic models, such as acetic acid writhing test, hot plate test and formalin test. Moreover, there are numerous records of liquor-processed Viticis Fructus in ancient processing. Therefore, more attention can be paid to liquor-processed Viticis Fructus. The fried products of Viticis Fructus need the provision for uniform frying standards, such as frying time, frying fire and other related parameters. Anti-tumor animal models should be established in vivo. The targets, pathways and related mechanisms are explained from multiple levels of genes and proteins through advanced technologies such as transcriptomics, proteomics, etc. Different doses of Viticis Fructus can be injected or intragastrical administered to explore its toxic mechanism. Quality markers should be explored and multiple components together reflect the overall quality of Viticis Fructus. It should conduct a systematic, in-depth study of the therapeutic material basis and complement the mechanism of action, pathway and target, in order to better exploit Viticis Fructus. A variety of rheumatism models such as rheumatoid arthritis, ankylosing spondylitis and osteoarthritis should be established to determine the type of rheumatism best treated by Viticis Fructus and to study its action mechanism.
Consequently, this article makes a comprehensive review from botany, historical records, phytochemistry, pharmacology, toxicity, quality control, pharmacokinetics and comprehensive applications of Viticis Fructus. At present, the application of Viticis Fructus still has certain limitations. Further research and discovery are needed to explore the scientific connotation of Viticis Fructus.
CRediT authorship contribution statement
Xue Meng: Writing – review & editing, Writing – original draft, Visualization, Validation, Software, Methodology, Investigation, Formal analysis, Data curation, Conceptualization. Yang Liu: Writing – review & editing, Visualization, Software, Methodology, Data curation, Conceptualization. Suyi Liu: Writing – review & editing, Investigation, Data curation, Conceptualization. Qianqian Zhang: Visualization, Project administration, Formal analysis, Conceptualization. Kunze Du: Writing – review & editing, Formal analysis, Data curation. Omachi Daniel Ogaji: Resources, Investigation, Formal analysis. Lirong Wang: Resources, Investigation, Data curation. Xingyue Jin: Resources, Formal analysis, Conceptualization. Jin Li: Investigation, Data curation, Conceptualization. Yanxu Chang: Writing – review & editing, Supervision, Project administration, Funding acquisition, Conceptualization.
Acknowledgement
This study was supported by the National Natural Science Foundation of China (82474073) and Science and Technology Program of Tianjin of China [23ZYJDSS00010].
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Biological activities of a new compound isolated from the aerial parts of Vitex agnus castus L. Afr. J. Biotechnol.. 2010;09(53):9063-9069.
- [Google Scholar]
- Assessment of anti-nociceptive and anthelmintic activities of Vitex Peduncularis Wall. leaves and in silico molecular docking, ADME/T, and PASS prediction studies of its isolated compounds. J. Complementary Med. Res.. 2019;10(04):170.
- [Google Scholar]
- The coastal medicinal plant Vitex rotundifolia: A mini-review on its bioactive compounds and pharmacological activity. Tradit. Med. Res. 2021;6(02):11.
- [CrossRef] [Google Scholar]
- Terpenoids from Vitex trifolia and their anti-inflammatory activities. J Nat Med. 2018;72(02):570-575.
- [CrossRef] [Google Scholar]
- Cai, Z.Q., Qu, W.S., Gao, S., Ma, H.Y., 2017. Identification of Viticis Fructus, Cuscutae Semen and Its Adulterants. Guangdong Chem. Ind. 44(02), 49, 36.
- Inhibition of Viticis Fructus total flavonoids on self-renewal of lung cancer stem cells derived from NCI-H446 cell line. Chin. Tradit. Herb Drugs.. 2014;45(09):1284-1287.
- [CrossRef] [Google Scholar]
- Induction of apoptosis by casticin in cervical cancer cells through reactive oxygen species-mediated mitochondrial signaling pathways. Oncol. Rep.. 2011;26(05):1287-1294.
- [CrossRef] [Google Scholar]
- Chen, H.Y., Tu, L.F., Xiao, C.R., Luo, Y.M., 2018. Chemical constituents from fruits of Vitex trifolia var. simplicifolia. China J. Chin. Mater. Med. 43(18), 3694-3700. https://doi.org/ 10.19540/j.cnki.cjcmm.20180625.002.
- Chen, X.H., Wang, X.Y., Ma, L., Fang, S.M., Chang, Y.X., 2021. The network pharmacology integrated with pharmacokinetics to clarify the pharmacological mechanism of absorbed components from Viticis Fructus extract. J. Ethnopharmacol(07), 114336. https://doi.org/ 10.1016/j.jep.2021.114336.
- Chen, Z.X., Wu, L.Z., Li, Y., Hu, X., You, S., J., Yu, S.H., Yuan, C., Yang, S.B., Yang, Y., Yu, F.L., 2023. Analysis on food patent pattern of Amomum tsaoko based on Baiten patent database. Sci. Technol. Food Ind., 01-11. https://doi.org/10.13386/j.issn1002-0306.2022110164.
- Comparison of chemical constituents of the essential oil from Fructus Viticis during different harvesting time. Chem. Ind. for. Prod.. 2007;06:99-104.
- [Google Scholar]
- Analysis of volatile oil components of Viticis Fructus from different producing areas. J. Jiangxi Tradit. Chin. Med.. 2015;46(07):67-70.
- [Google Scholar]
- Pharmacopoeia of the People's Republic of China, 2020ed. China: Chinese Pharmacopoeia Commission; 2020.
- Chu, J., Yan, B., Zhang, J., Peng, L.l., Ao, X., Zheng, Z.Y., Jiang, T., Zhang, Z.M., 2020. Casticin attenuates osteoarthritis-related cartilage degeneration by inhibiting the ROS-mediated NF-κB signaling pathway in vitro and in vivo. Inflammation. 43, 810-820. https://doi.org/10.1007/s10753-019-01167-y.
- Dictionary of traditional Chinese pharmacy. Shanghai: Shanghai people's publishing house; 1977.
- Diao, J., Y., Xu, C., Zheng, H.T., He, S.Y., Wang, S.M., 2018. An integrated strategy to qualitatively differentiate components of raw and processed Viticis Fructus based on NIR, HPLC and UPLC-MS analysis. Planta Med. 84(17), 1280-1291. https://doi.org/ 10.1055/a-0639-5450.
- Casticin induces apoptosis and mitotic arrest in pancreatic carcinoma PANC-1 cells. Afr. J. Pharm. Pharmacol. 2012;6(06):412-418.
- [CrossRef] [Google Scholar]
- Chemical constituents from the fruits of Vitex trifolia L. (Verbenaceae) and their chemotaxonomic significance. Biochem. Syst. Ecol.. 2021;158
- [CrossRef] [Google Scholar]
- Fan, L., Zhang, Y., Zhou, Q.H., Liu, Y., Gong, B.L., Lü, J.Y., Zhu, H., Zhu, G.J., Xu, Y.P., Huang, G.G., 2018. Casticin inhibits breast cancer cell migration and invasion by down-regulation of PI3K/Akt signaling pathway. Biosci. Rep. 38(06). https://doi.org/10.1042 /BSR20180738.
- Fang, S.M., Liu, R., Li, L., Yao, J.L., Liu, E.W., Fan, G.W., Zhang, H., Gao, X.M., 2019. Anti-inflammatory diterpenes from the fruits of Vitex trifolia L. var. simplicifolia Cham. J. Asian Nat. Prod. Res. 21(10), 985-991. https://doi.org/10.1080/10286020.2018. 1482881.
- Gao, B.X., Zheng, D.H., Huang, Z.T., Piao, J.S., Lu, Z., 2022. Composition for preventing ameliorating or treating andropause syndrome comprising Vitex rotundifolia extract as effective component.
- Shoushi Baoyuan. Tianjing: Tianjin Science and Technology Press; 1999.
- Analysis of market price trend of Viticis Fructus. Spec. Econ. Anim. Plants.. 2016;19(09):22.
- [Google Scholar]
- The Cyr61 is a potential target for rotundifuran, a natural labdane-type diterpene from Vitex trifolia L., to trigger apoptosis of cervical cancer cells. Oxid. Med. Cell. Longev.. 2021;2021:01-15.
- [CrossRef] [Google Scholar]
- Discussion on processing method of analgesic effect of Viticis Fructus. Chin. J. Mod. Drug Appl.. 2012;6(04):134-135.
- [CrossRef] [Google Scholar]
- Studies on chemistry and anti-HIV components of five medicinal plants. Kunming Institute of Botany Cas. 2007
- [Google Scholar]
- Gu, Q., Zhang, X.M., Zhou, J., Qiu, S.X., Chen, J.J., 2008. One new dihydrobenzofuran lignan from Vitex trifolia. J Asian Nat Prod Res 10(5-6), 499-502. https://doi.org/ 10.1080/10286020801967359.
- Preparative isolation and purification of the active components from Viticis Fructus by high-speed. Chin. J. Chromatogr.. 2010;28(11)
- [CrossRef] [Google Scholar]
- Guan, R.J., 2011. Studies on the chemical constituents and dtermination method of Viticis Fructus. Shandong University of Traditional Chinese Medicine.
- Guo, C.Q., Cheng, L.F., Zhao, Z.H., Liu, W.L., 2005. Determination of total flavonoids content in Viticis Fructus and its different degrees of fried products. Modern. Tradit. Chin. Med. Mater. Medica-World Sci. Technol.(05), 30-32, 29, 83-84. https://doi.org/ 10.11842/wst.2005.5.[sequence].
- 110 cases of chronic suppurative otitis media treated with Viticis Fructus Decoction and Hongmian Powder. Hebei J. Tradit. Chin. Med.. 2003;07:508.
- [CrossRef] [Google Scholar]
- Guo, G.F., 2006. Preparation and clinical application of Biedele. China's Naturopathy(10), 23-24. https://doi.org/10.19621/j.cnki.11-3555/r.2006.10.023.
- Haïdara, K., Zamir, L., Shi, Q.W., Batist, G., 2006. The flavonoid casticin has multiple mechanisms of tumor cytotoxicity action. Cancer Lett. (N. Y., NY, U. S.) 242(02), 180-190. https://doi.org/10.1016/j.canlet.2005.11.017.
- Casticin inhibits self-renewal of liver cancer stem cells from the MHCC97 cell line. Oncol. Lett.. 2014;07(06):2023-2028.
- [CrossRef] [Google Scholar]
- A Beta/ZSM-22 Zeolites-Based-Mixed Matrix Solid-Phase Dispersion Method for the Simultaneous Extraction and Determination of Eight Compounds with Different Polarities in Viticis Fructus by High-Performance Liquid Chromatography. Molecules.. 2019;24(19):3423.
- [CrossRef] [Google Scholar]
- Casticin induces growth suppression and cell cycle arrest through activation of FOXO3a in hepatocellular carcinoma. Oncol. Rep.. 2013;29(01):103-108.
- [CrossRef] [Google Scholar]
- Taiping Huimin Hejiju Fang. Beijing: People's medical publishing house; 1985.
- Hobbs, C., 1991. The chaste tree: Vitex agnus castus. Pharm. Hist. 33(01), 19-24. http://www. jstor.org/stable/41111358.
- Association between chemical and genetic variation of medicinal plant Vitex rotundifolia and the mechanism of Viticis Fructus for anti-PMS effect. The Second Military Medical University; 2007.
- Estrogen-like activity of volatile components from Vitex rotundifolia L. Indian J. Med. Res.. 2007;126(01):68-72.
- [Google Scholar]
- Qiang-Huo-Sheng-Shi decoction exerts anti-inflammatory and analgesic via MAPKs/CREB signaling pathway. J. Ethnopharmacol.. 2022;284:114776
- [CrossRef] [Google Scholar]
- Qing-Shang-Juan-Tong Decoction in the treatment of 62 cases of headache. Shanxi J. Tradit. Chin. Med.. 2016;37(07):818-819.
- [CrossRef] [Google Scholar]
- Association between chemical and genetic variation of Vitex rotundifolia populations from different locations in China: its implication for quality control of medicinal plants. Biomed. Chromatogr.. 2010;21(09):967-975.
- [CrossRef] [Google Scholar]
- Huang, Y.G., Wen, G.G., 2000. Viticis Fructus is a good medicine for gastritis. J. Tradit. Chin. Med. (12), 712-713. https://doi.org/ 10.13288/j.11-2166/r.2000.12.004.
- A new taraxastane-type triterpene from Vitex trifolia var. simplicifolia. Helv. Chim. Acta. 2013;96(11):2040-2045.
- [CrossRef] [Google Scholar]
- A new ursane triterpenoid possessing cytotoxicity from the fruits of Vitex trifolia var. simplicifolia. Chem. Nat. Compd.. 2016;52(04):660-663.
- [CrossRef] [Google Scholar]
- A. Huang Casticin inhibits osteoclast formation through NF-κB/BCL-2 signal pathway 2022 NanChang University.
- Chemical constituents of ethanol extract of leaves and molluscicidal activity of crude extracts from Vitex trifolia Linn. Herba Pol.. 2013;59(04):19-32.
- [CrossRef] [Google Scholar]
- Casticin elicits inflammasome-induced pyroptosis through activating PKR/JNK/NF-κB signal in 5–8F cells. Biomed. Pharmacother.. 2020;123:109576
- [CrossRef] [Google Scholar]
- Jiang, B., 2016. A health-preserving herbal hair dye.
- Three cases of Xin-Zhi-Chai-lian Decoction in the treatment of external eye disease. J. Zhejiang Univ. Tradit. Chin. Med.. 2018;53(11):850.
- [Google Scholar]
- Preliminary probe into the processing of Viticis Fructus. Lishizhen Med. Mater. Med. Res.. 2000;06:503-504.
- [CrossRef] [Google Scholar]
- Traditional Medicinal Uses, Phytochemistry, Biological Properties, and Health Applications of Vitex sp. Plants.. 2022;11:1944.
- [CrossRef] [Google Scholar]
- Antioxidative constituents from the twigs of Vitex rotundifolia. Biomol. Ther.. 2009;17(04):412-417.
- [CrossRef] [Google Scholar]
- Kim, Byunghyun, Roh, Hyosun, Hong, Young, C., Ahn, Je, Y., Hwang, Jin, S., Jin, Hyun, M., 2018. Cosmetic composition comprising extract of medicinal herbs as active ingredient.
- Anti-atherosclerotic activity of (3R)-5-hydroxymellein from an endophytic fungus Neofusicoccum parvum JS-0968 derived from Vitex rotundifolia through the inhibition of lipoproteins oxidation and foam cell formation. Biomolecules.. 2020;10(05):715.
- [CrossRef] [Google Scholar]
- SRVF, a novel herbal formula including scrophulariae radix and viticis fructus, disrupts focal adhesion and causes detachment- induced apoptosis in malignant cancer cells open. Sci. Rep.. 2019;07(01):12756.
- [CrossRef] [Google Scholar]
- Cytotoxic compounds from the fruits of Vitex rotundifolia against human cancer cell lines. J. Korean Soc. Appl. Biol. Chem.. 2012;55(03):433-437.
- [CrossRef] [Google Scholar]
- New norditerpenoids with trypanocidal activity from Vitex trifolia. Chem. Pharm. Bull.. 2004;52(12):1492-1494.
- [CrossRef] [Google Scholar]
- Polymethoxyflavonoids from Vitex rotundifolia inhibit proliferation by inducing apoptosis in human myeloid leukemia cells. FCT. 2000;38(10):861-865.
- [CrossRef] [Google Scholar]
- Rotundifuran, a labdane type diterpene from Vitex rotundifolia, induces apoptosis in human myeloid leukaemia cells. PTR. 2001;15(06):535-537.
- [CrossRef] [Google Scholar]
- Bencao Yanyi. Ltd, Beijing: Zhonghua Book Co.; 1985.
- Casticin promotes immune responses, enhances macrophage and NK cell activities, and increases survival rates of leukemia BALB/c mice. Am. J. Chin. Med.. 2019;47(01):223-236.
- [CrossRef] [Google Scholar]
- Analysis of antioxidant phytochemicals and anti-inflammatory effect from Vitex rotundifolia L.f. Antioxidants.. 2022;11(03):454.
- [CrossRef] [Google Scholar]
- Pyranopyran-1, 8-dione, an active compound from Vitices Fructus, attenuates cigarette-smoke induced lung inflammation in mice. Int J Mol Sci.. 2017;18(7):1602.
- [CrossRef] [Google Scholar]
- Vascular protective role of vitexicarpin isolated from Vitex rotundifolia in human umbilical vein endothelial cells. Inflammation. 2012;35:584-593.
- [CrossRef] [Google Scholar]
- Anti-inflammatory constituents from the fruits of Vitex rotundifolia. Bioorg. Med. Chem. Lett.. 2013;23(21):6010-6014.
- [CrossRef] [Google Scholar]
- Lei, X., 1986. Leigong Paozhi Lun, Shanghai university of Chinese medicine press.
- Neiwai Shangbian Huolun. Beijing: People's Medical Publishing House; 1959.
- Studies on the anticancer constituents of Vitex trifolia L., a taditional chinese medicine. Shenyang Pharmaceutical University; 2003.
- Bencao Gangmu. Beijing: Huaxia Publishing House; 2011.
- Bencao Tongxuan. Beijing: China traditional Chinese medicine publishing house; 2015.
- Labdane-type diterpenes as new cell cycle inhibitors and apoptosis inducers from Vitex trifolia L. J. Asian Nat. Prod. Res.. 2005;07(02):95-105.
- [CrossRef] [Google Scholar]
- Li, W.X., Cui, C.B., Cai, B., Wang, H.Y., Yao, X.S., 2005a. Flavonoids from Vitex trifolia L. inhibit cell cycle progression at G2/M phase and induce apoptosis in mammalian cancer cells. J. Asian Nat. Prod. Res. 07(04), 615-626. https://doi.org/10.1080/10286020 310001625085.
- Study on characteristic chromatogram and content determination of phenols and flavonoids of Viticis Negundo Fructus and Viticis Fructus. Nat. Prod. Res. Dev. 2023:01-13.
- [CrossRef] [Google Scholar]
- Casticin suppresses monoiodoacetic acid-induced knee osteoarthritis through inhibiting HIF-1α/NLRP3 inflammasome signaling. Int. Immunopharmacol.. 2020;86:106745
- [CrossRef] [Google Scholar]
- Casticin inhibits stemness of hepatocellular carcinoma cells via disrupting the reciprocal negative regulation between DNMT1 and miR-148a-3p. Toxicol. Appl. Pharmacol.. 2020;396:114998
- [CrossRef] [Google Scholar]
- Comprehensive identification of Vitex trifolia fruit and its five adulterants by comparison of micromorphological, microscopic characteristics and chemical profiles. Microsc. Res. Tech.. 2020;83(12):1530-1543.
- [CrossRef] [Google Scholar]
- Li, K.M., 1998. Treatment of rhinitis and supraorbital neuralgia by Viticis Fructus. J. Yunnan. Univ. Tradit. Chin. Med.(03), 41. https://doi.org/10.19288/j.cnki.issn.1000-2723.1998. 03.015.
- Percutaneous permeability enhancement of essential oil from Fructus Viticis and essential oil from ramulus cinnamomi. Guangdong Pharmaceutical University; 2019.
- Treating 60 cases of lumbago with the Qiang-Huo-Sheng-Shi Decoction. Clin. J. Chin. Med.. 2019;11(01):69-70.
- [Google Scholar]
- Casticin inhibits COX-2 and iNOS expression via suppression of NF-κB and MAPK signaling in lipopolysaccharide-stimulated mouse macrophages. J. Ethnopharmacol.. 2014;158:310-316.
- [CrossRef] [Google Scholar]
- Protective effects of casticin from Vitex trifolia alleviate eosinophilic airway inflammation and oxidative stress in a murine asthma model. Front. Pharmacol.. 2018;09:635.
- [CrossRef] [Google Scholar]
- Study on the effective components and quality of Viticis Fructus. Heilongjiang University of Chinese Medicine; 2002.
- Study on different processing techniques of Fructus Arctii and Viticis Fructus. Hunan University of Chinese Medicine; 2005.
- Study on antioxidant activity of total volatile oil from Vitex trifolia L.in vitro. Anti-Infect. Pharm.. 2014;11(02):119-121.
- [CrossRef] [Google Scholar]
- Liu, F., Cao, X.Z., Liu, Z.H., Guo, H., Ren, K.Q., Quan, M.F., Zhou, Y.F., Xiang, H.L., Cao, J.G., 2014. Casticin suppresses self-renewal and invasion of lung cancer stem-like cells from A549 cells through down-regulation of pAkt. ABBS. 46(01), 15-21. https://doi.org/ 10.1093/abbs/gmt123.
- Liu, L.P., Cao, X.C., Liu, F., Quan, M., F,, Sheng, X.F., Ren, K.Q., 2014. Casticin induces breast cancer cell apoptosis by inhibiting the expression of forkhead box protein M1. Oncol. Lett. 07(05), 1711-1717. https://doi.org/10.3892/ol.2014.1911.
- Casticin induces human glioma cell death through apoptosis and mitotic arrest. Cell Physiol. Biochem.. 2013;31(06):805-814.
- [CrossRef] [Google Scholar]
- Casticin inhibits nasopharyngeal carcinoma growth by targeting phosphoinositide 3-kinase. Cancer Cell Int.. 2019;19:01-16.
- [CrossRef] [Google Scholar]
- Yizong Cuiyan. Taibei: Xinwenfeng publishing company; 1982.
- Effect of Maturity on quality of Viticis Fructus in Jiangxi. Chin. J. Exp. Tradit. Med. Formulae. 2015;21(16):23-27.
- [CrossRef] [Google Scholar]
- Ma, L., Wei, Q.Q., Chang, Z.J., Qi, J.S., Liu, X.Y., Zhao, M., 2023. Research progress on the pharmacological mechanism of chinese herbal additives and their effects on reproductive performance of ruminants. Feed Ind. (Shenyang, China), 01-16. https://doi.org/ b10.13302/j.cnki.fi.2023.06.008.
- Casticin prevents DSS induced ulcerative colitis in mice through inhibitions of NF-κB pathway and ROS signaling. PTR. 2018;32(09):1770-1783.
- [CrossRef] [Google Scholar]
- Traditional use, phytochemistry and pharmacology of Viticis Fructus. Heliyon.. 2023;09(09):e19144.
- [Google Scholar]
- Vitexicarpin induces apoptosis in human prostate carcinoma PC-3 cells through G2/M phase arrest. Asian Pac. J. Cancer Prev.. 2012;13(12):6369-6374.
- [CrossRef] [Google Scholar]
- Mu, K.Y., Liu, D.S., 2017. A toothpaste containing Viticis Fructus composition.
- Xiong-Ju-Shang-Qing Pills combined with psychological counseling in the treatment of migraine in 52 cases. Clin. J. Tradit. Chin. Med.. 2005;01:40.
- [CrossRef] [Google Scholar]
- Isolation of new diterpenoids from the halophyte, Vitex rotundifolia, and their antioxidant and anti-inflammatory activities. Chem. Biodiversity 2024:e202301115-e.
- [CrossRef] [Google Scholar]
- Pharmacologically active components of Viticis Fructus (Vitex rotundifolia) II. The components having analgesic effects. Chem. Pharm. Bull.. 1998;46(06):655.
- [CrossRef] [Google Scholar]
- Ono, M., Ito, Y., Kubo, S., Nohara, T., 1997. Two new iridoids from Viticis trifoliae Fructus (fruit of Vitex rotundifolia L.). Chem. Pharm. Bull. 45, 1094-1096. https://doi.org/ 10.1248/cpb.45.1094.
- Ono, M., Yamamoto, M., Masuoka, C., Ito, Y., Yamashita, M., Nohara, T., 1999. Diterpenes from the fruits of Vitex rotundifolia. J. Nat. Prod. 62(11), 1532-1537. https://doi.org/ 10.1016/S0031-9422(00)00214-4.
- Ono, Yamamoto, Yanaka, Ito, Nohara, 2001. Ten new labdane-type diterpenes from the fruit of Vitex rotundifolia. Chem. Pharm. Bull. 49(01), 82-86. https://doi.org/10.1248/ cpb.49.82.
- Ono, M., Yanaka, T., Yamamoto, M., Ito, Y., Nohara, T., 2002. New diterpenes and norditerpenes from the fruits of Vitex rotundifolia. J Nat Prod 65(04), 537-541. https://doi.org/ 10.1021/np0105331.
- A labdane diterpene glycoside from fruit of Vitex rotundifolia. Phytochemistry.. 1998;48(01):207-209.
- [CrossRef] [Google Scholar]
- Antioxidative constituents from Viticis trifoliae Fructus (fruit of Vitex rotundifolia L.) Food Sci. Technol. Int., Tokyo. 1998;04(01):09-13.
- [CrossRef] [Google Scholar]
- Diterpenoids from the fruits of Vitex trifolia. Phytochemistry. 2000;55(08):873-877.
- [CrossRef] [Google Scholar]
- Experimental observation on antithrombin effect of 126 kinds of traditional Chinese medicine. Chin. Tradit. Herb Drugs. 1987;18(04):21.
- [Google Scholar]
- Park, Hyo, Suk, 1999. Topical liquid composition for promoting hari frowth and process of manufacturing thereof.
- Shang, H.S., Liu, J.Y., Lu, H.F., Chiang, H.S., Lin, C.H., Chen, A., Lin, Y.F., Chung, J.G., 2017. Casticin induced apoptotic cell death and altered associated gene expression in human colon cancer colo 205 cells. Environ. Toxicol. 32(08), 2041-2052. https://doi.org/ 10.1002/tox.22381.
- Shen, Y.L., 2020. Study on the molecular mechanism of apoptosis induced by rotundifuran in cervical cancer cells Shanghai University of Traditional Chinese Medicine.
- Shi, Y., 2016. A kind of Viticis Fructus Bu-Pi-Yi-Shen granule beverage and its preparation method.
- Casticin impairs cell migration and invasion of mouse melanoma B16F10 cells via PI3K/AKT and NF-κB signaling pathways. Environ. Toxicol.. 2017;32(09):2097-2112.
- [CrossRef] [Google Scholar]
- Isolation of an aldose reductase inhibitor from the fruits of Vitex rotundifolia: Part 2 in the series “Studies on the inhibitory effects of medicinal plant constituents on cataract formation”. Phytomedicine.. 1994;01(02):145-147.
- [Google Scholar]
- Study on pharmacodynamics of processing Viticis Fructus. Chin. Tradit. Herb. 1997;Drugs(01):32-34.
- [CrossRef] [Google Scholar]
- Clinical observation on treating 50 cases of early senile cataract in the integrative medicine. J. Zhejiang Univ. Tradit. Chin. Med.. 2010;45(08):594.
- [CrossRef] [Google Scholar]
- Sun, S.M., 1982. Qianjin Yifang People's Medical Publishing House, Beijing.
- Sun, Y.Q., 2018. Study on the genetic relationship and quality evaluation of the original plants of Chinese medicine Viticis Fructus. Liaoning University of Traditional Chinese Medicine.
- Casticin, a flavonoid, potentiates TRAIL-induced apoptosis through modulation of anti-apoptotic proteins and death receptor 5 in colon cancer cells. Oncol. Rep.. 2013;29(02):474-480.
- [CrossRef] [Google Scholar]
- Bencao Jingji Zhu. Beijing: People's Medical Publishing House; 1994.
- Tao, H.J., 1986. Mingyi Bielu People's Medical Publishing House, Beijing.
- Tian, T., Shi, X.D., 2022. Experience of Qing-Shang-Juan-Tong Decoction in treating headache. China J. Tradit. Chin. Med. Pharm. (2005-) 37(04), 2112-2114.
- Tian, W.L., 2017. The effect of Viticis Fructus/Inulae Flos on apoptosis and angiogenesis in IPF rats. Beijing university of Traditional Chinese Medicine.
- Boji Fang. Beijing: the commercial press; 1958.
- Taiping Shenghui Fang. Beijing: People's health publishing house; 1958.
- Treating 56 cases of sciatica with Viticis Fructus. J. Hebei. Tradit. Chin. Med. Pharmacol.. 2001;04:24.
- [CrossRef] [Google Scholar]
- Study on quality and fingerprint of Viticis Fructus. Heilongjiang University of Traditional Chinese Medicine; 2003.
- Clinical observation on modified Qiang-Huo-Sheng-Shi Decoction in the treatment of knee osteoarthriti. Guangming J. Chin. Med.. 2021;36(23):3937-3940.
- [CrossRef] [Google Scholar]
- Wang, X.Q., Zhang, T., Zheng, B., Xie, W.D., Shen, T., 2014. Labdane-type diterpenoids from the fruits of Vitex rotundifolia. Bull. Korean Chem. Soc. 35(02), 672-674. http:// dx.doi.org/10.5012/bkcs.2014.35.2.672.
- Wang, T., Tang, L.Y., Guo, R.X., Xu, M.Y., Yu, X.K., Wang, Z.J., 2017a. Influence of processing on the dissolution rates of flavonoids in Viticis Fructus. Chin. J. Pharmacovigilance 14(04), 215-217, 241.
- Wang, T., Wu, H.W., Guo, R.X., Xu, M.Y., Yu, X.K., Tang, L.Y., Wang, Z.J., 2017b. Influence of processing on volatile components in Viticis Fructus by GC-MS. Chin. J. Exp. Tradit. Med. Formulae 23(19), 34-39. https://doi.org/10.13422/j.cnki.syfjx. 2017190034.
- Wang, W.Q., Yin, Y.P., Jun, L., Xuan, L., J,, 2018. Halimane-type diterpenoids from Vitex rotundifolia and their anti-hyperlipidemia activities. Phytochemistry 146, 56-62. https://doi.org/10.1016/j.phytochem.2017.12.002.
- Wang, L., Zhang, J.L., Jiang, L.G., Li, X.H., Bi, Y.X., Xi, C.H., 2019b. Application of vitexicarpin as sunscreen ingredient in cosmetics.
- Research progress on chemistry, pharmacology and processing of Viticis Fructus. Acta Chin. Med. Pharmacol.. 2008;01:69-71.
- [CrossRef] [Google Scholar]
- Effect of recipes for tonifying kidney and replenishing Qi on learning and memory-related genes in aged rats. Chin. J. Exp. Tradit. Med. Formulae.. 2019;25(06):15-22.
- [Google Scholar]
- Study on processing standardization of Viticis Fructus. Chin. Tradit. Pat. Med.. 2010;32(03):515-517.
- [Google Scholar]
- Viticis Fructus methanolic extract attenuates trigeminal hyperalgesia in migraine by regulating injury signal transmission. Exp. Ther. Med.. 2020;19(01):85-94.
- [CrossRef] [Google Scholar]
- Wu, J., Zhou, T., Zhang, S.w., Zhang, X.H., Xuan, L.J., 2009. Cytotoxic terpenoids from the fruits of Vitex trifolia L. Planta Med. 75(04), 367-370. https://doi.org/10.1055/s-0028-1112211.
- Wu, C., Zhang, J., Yin, Z.Q., 2010. Chemical constituents of Viticis Fructus. Chin. J. Pharm. Biotechnol. 17(06), 504-507, 564. https://doi.org/10.19526/j.cnki.1005-8915.2010.06. 007.
- Treatment of 19 cases of acute mastitis with Viticis Fructus. Hunan J. Tradit. Chin. Med.. 1999;03:50.
- [Google Scholar]
- The chemical constituents of anti-PMS activity, intraspecific variation and quality evaluation of Viticis Fructus. The Second Military Medical University; 2005.
- Xin, H.L., Wu, C.H., Zhang, Q.Y., Zheng, H.C., 2004. The distribution, development prospect and resource protection of Vitex trifolia L.plants, The 4th Natl. Symp. Pharm. Bot. Bot. Med. Nanjing China, 04.
- Study on antiplatelet activation of vitexicarpin via phosphatidylinositol-3 kinase/protein kinase B signaling pathway. Chin. J. Clin. Pharmacol.. 2022;38(18):2133-2137.
- [CrossRef] [Google Scholar]
- Clinical observation on the treatment of migraine with Man-Jing-Zi-Tou-Feng Decoction. Med. J. Chin. People's Health.. 2007;20:862-863.
- [CrossRef] [Google Scholar]
- Chemical constituents of Viticis Fructus. J. Int. Pharm. Res.. 2019;46(11):848-854.
- [CrossRef] [Google Scholar]
- Effects of different processing methods on casticin. Xinjiang J. Tradit. Chin. Med.. 2020;38(01):41-42.
- [Google Scholar]
- Quantitative determination and pharmacokinetic study of casticin in rat plasma by liquid chromatography-mass spectrometry. J. Pharm. Biomed. Anal.. 2012;61:242-246.
- [CrossRef] [Google Scholar]
- Determination and identification of a specific marker compound for discriminating Shrub Chaste Tree Fruit from Agnus Castus Fruit based on LC/MS metabolic analysis. Chem. Pharm. Bull.. 2016;64(04):305-310.
- [CrossRef] [Google Scholar]
- Fingerprint of classical prescription Qiang-Huo-Sheng-Shi decoction and predictive analysis of efficacy-related substances. Chin. J. Pharm. Anal.. 2022;42(04):618-629.
- [CrossRef] [Google Scholar]
- Vitex rotundifolia L. f. and Vitex trifolia L.: A review on their traditional medicine, phytochemistry, pharmacology. J.. 2023;Ethnopharmacol:116273
- [CrossRef] [Google Scholar]
- Bencao Shugou Yuan. Beijing: Science and technology health publishing house; 1958.
- Yang, X., Y., Gao, P.Y., Chen, X.X., Wang, L.X., Jiang, T., Wu, T., Chen, Y.Y., Yue, C.Y., Wu, H.W., Tang, L.Y., Wang, Z.J., 2023. Comparison of HPLC fingerprint and determination of main components of Viticis Fructus from different species. China J. Chin. Mater. Med., 01-11. https://doi.org/10.19540/j.cnki.cjcmm.20230104.201.
- Effect of modified modified Qiang-Huo-Sheng-Shi decoction on spinal function and acute phase reactants in patients with ankylosing spondylitis. J. Pract. Med.. 2021;37(18):2423-2427.
- [CrossRef] [Google Scholar]
- Casticin-induced apoptosis involves death receptor 5 upregulation in hepatocellular carcinoma cells. World J. Gastroenterol.. 2011;17(38):4298.
- [CrossRef] [Google Scholar]
- Ye, Q., 2010. Integrative anti-PMS effect of active constituents in Viticis Fructus. The Second Military Medical University.
- The anti-aging and antioxidant effects of alcohol abstracts from Vitex trifolia L. on peanut oils. J. Chin. Cereals Oils Assoc.. 2016;31(09):91-94.
- [CrossRef] [Google Scholar]
- Study on the ancient processing technology of Viticis Fructus. Xinjiang J. Tradit. Chin. Med.. 2019;37(03):50-52.
- [Google Scholar]
- Y.P. Yin Chemical constituents of Vitex rotundifolia and cassia mimosoides 2015 NanChang University.
- Yin, S.C., 2019. A hair shampoo.
- Phenolic compounds and flavonoids as plant growth regulators from fruit and leaf of Vitex rotundifolia. Z. Naturforsch c. J. Biosci.. 2004;59(7–8):509-514.
- [CrossRef] [Google Scholar]
- Effects of processing on pharmacokinetic of four semen chinese medicine. Sci: China Acad. Chin. Med; 2019.
- Iridoids isolated from Viticis Fructus inhibit paclitaxel-induced mechanical allodynia in mice. J. Nat. Med.. 2021;75(01):48-55.
- [CrossRef] [Google Scholar]
- Zeng, F.X., Tian, L., Liu, F., Cao, J.G., Quan, M.F., Sheng, X.F., 2012. Induction of apoptosis by casticin in cervical cancer cells: reactive oxygen species-dependent sustained activation of Jun N-terminal kinase. ABBS 44(05), 442-449. https://doi.org/ 10.1093/abbs/gms013.
- Zhang, J.H., Chen, D.J., Yu, H.Y., 2003. Experimental study on total flavonoids of Viticis Fructus and its processed products. Inf. Tradit. Chin. Med.(04), 27. https://doi.org/ 10.3969/j.issn.1002-2406.2003.04.013.
- Zhang, P., Wang, Q.J., Xu, D.K., Jiang, X.F., Li, B.B., 2023. Modified Qiang-Huo-Sheng-Shi-Tang combined with ozone injection and radiofrequency thermocoagulation target puncture in treatment of cervical spondylotic radiculopathy with wind-cold blockage type. Chin. J. Exp. Tradit. Med. Formulae 29(01), 91-96. https://doi.org/10.13422/ j.cnki.syfjx.202201729.
- Chemical Constituents from the Fruits of Vitex trifolia var. simplicifolia by UHPLC-Q-Orbitrap HRMS. J. Chin. Med. Mater.. 2021;44(05):1142-1148.
- [CrossRef] [Google Scholar]
- Analysis of supercritical carbon dioxide extract of Viticis Fructus by gas chromatography-mass spectrometry. Lishizhen Med. Mater. Med. Res.. 2011;22(02):387-388.
- [CrossRef] [Google Scholar]
- Herbal textual research on Chinese medicine “Manjingzi” (Viticis Fructus) China. J. Chin. Mater. Med.. 2019;44(24):5503-5507.
- [CrossRef] [Google Scholar]
- Study on the toxicity of Manjing Oral Liquid. J. Jiangxi Univ. Chin. Med.. 2000;04:183-184.
- [Google Scholar]
- Identification of Viticis Fructus and its adulterants by ITS2 sequence. Modern. Tradit. Chin. Med. Mater. Medica-World Sci. Technol.. 2014;16(11):2366-2370.
- [CrossRef] [Google Scholar]
- Vitrifolin A: A norlabdane diterpenoid from the fruits of Vitex trifolia Linn. var. simplicifolia Cham. J. Chin. Chem. Soc. (taipei, Taiwan). 2013;60(05):542-545.
- [CrossRef] [Google Scholar]
- Shennong Bencao Jing. Nanning: Guangxi Science&Technology Publishing; 2018.
- Shengji Zonglu. Beijing: People's Medical Publishing House; 1982.
- A new iridoid glycoside from the fruits of Vitex rotundifolia. Nat. Prod. Res.. 2017;31(21):2491-2496.
- [CrossRef] [Google Scholar]
- Zhao, G.R., 2018. Clinical observation on 40 cases of migraine treated by syndrome differentiation. J. Practi. Tradit. Chin. Internal Med. 32(09), 22-24. https://doi.org/ 10.13729/j.issn.1671-7813.z20180049.
- Yaoxing Lun. Wuhu: Wannan Medical College; 1983.
- Labdane-type diterpenoids from the fruits of Vitex trifolia. J. Nat. Prod.. 2013;76(02):287-291.
- [CrossRef] [Google Scholar]
- Comparison of pharmacological activities of fruits of Vitex rotundifolia, fruits of Vitex trifolia, fruits of Vitex negundo and fruits of Vitex negundo L. var. cannabifolia. Pharmacol. Clin. Chin. Mater. Med.. 1996;01:37-39.
- [Google Scholar]
- Identification of the authenticity of Viticis Fructus. Lishizhen Med. Mater. Med. Res.. 2001;01:50.
- [Google Scholar]
- Casticin potentiates TRAIL-induced apoptosis of gastric cancer cells through endoplasmic reticulum stress. PLoS One.. 2013;08(03):e58855.
- [Google Scholar]
- Danxi Xinfa. Shanghai: Shanghai scientific and technical publishers; 1956.
- Chemical constituents from fruits of Vitex trifolia L. and metabolism of casticin. The Second Military Medical University; 2013.
Appendix A
Supplementary data
Supplementary data to this article can be found online at https://doi.org/10.1016/j.arabjc.2024.106008.
Appendix A
Supplementary data
The following are the Supplementary data to this article:Supplementary Data 1
Supplementary Data 1







