Translate this page into:
Adsorption of PO43-, Cd(II), Pb(II), Cu(II), As(III), and As(V) using a carbonised Mn-based metal–organic framework
⁎Corresponding authors. Zhangshuhua@gdupt.edu.cn (Shu-Hua Zhang), liningmmc@gdupt.edu.cn (Ning Li)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Abstract
A metal–organic framework, Mn-MOF ([Mn(DMF)2(H2DHTPA)]n) was synthesized. Mn-MOF-500 was prepared using Mn-MOF through 500 °C carbonization. The BET surface area of Mn-MOF-500 is 11.83 m2/g. The qmax of Mn-MOF-500 on PbⅡ, AsV, AsⅢ are 356.2, 166.1, 51.3 mg/g, respectively. The qmax of Mn-MOF-500 on PO43-, CdⅡ, CuⅡ are 126.9, 88.5, 146.9 mg/g, respectively.
Abstract
Metal–organic frameworks (MOFs) are adsorbents suitable for water treatment. However, they are often water soluble and poorly processible, limiting their industrial applications. This study aims to develop a carbonized MOFs with high adsorption performance based on MOFs. Here, we report the production of Mn-MOF [Mn(DMF)2(H2DHTPA)]n (where DMF is N,N-dimethylformamide and H4DHTPA is 2,5-dihydroxyterephthalic acid) via a solvothermal method. This MOF shows good adsorption performance for various ions but dissolves easily in water. However, Mn-MOF-500 which was obtained by carbonising Mn-MOF at 500 °C shows high water stability. Thus, the adsorption properties of Mn-MOF-500 were investigated, revealing adsorption capacities for PO43-, Cd(II), Pb(II), Cu(II), As(III), and As(V) of 126.66, 88.46, 356.21, 146.92, 51.28, and 166.10 mg/g, respectively. Based on the adsorption isotherms and the adsorption kinetics, the adsorption of PO43-, Cd(II), and As(III) occurs via monolayer chemisorption, whereas that of Pb(II), Cu(II), and As(V) occurs via monolayer chemisorption and physisorption in pores. X-ray diffractometry and X-ray photoelectron spectroscopy analysis showed that P, Cd(II), Pb(II), Cu(II), As(III), and As(V) were adsorbed on Mn-MOF-500 as PO43-, Cd(OH)2, PbO and Pb(OH)2, Cu(OH)2, Mn3(AsO3)2, and MnHAsO4, respectively.
Keywords
Mn-MOF-500
Adsorption
PO43-
Heavy metal ions
1 Introduction
Increasing industrial development has resulted in environmental problems, such as the production of acidic wastewater from mining, and this is a global problem (Krupskaya and Zvereva, 2014). The primary methods for removing contaminants from acid mine wastewater are physical, chemical, biological, and microbial methods, which include precipitation, adsorption, ion exchange, electrochemical treatment technology, and biochemical methods (Ríos, et al., 2008; Yilmaz, et al., 2005; Vymazal, 2007; Türker, et al., 2014; Fosso-Kankeu, et al., 2011; Vignesh et al., 2014). Adsorption can occur via chemical and physical processes, and this method has been favoured by researchers for wastewater treatment because of its simplicity, efficiency, and low cost (Yilmaz, et al., 2005; Zhang, et al. 2021c; Zhang, et al. 2023). As a result, the development of adsorbents is a hot topic. Metal–organic framework materials (MOFs) are relatively new adsorbents showing high chemical and physical stability, high specific surface areas, a large number of active sites, and adjustable pore sizes, thus having advantages over activated carbon, zeolite, and minerals such as titanium dioxide and other traditional materials (Li, et al., 1999; Chen, et al., 2019; An, et al., 2018; Bhadra, et al., 2017; Li, et al., 2018; Oveisi, et al., 2018; Song, and Jhung, 2017; Zhang, et al., 2022; Wang, et al., 2021b; Zhang, et al., 2021c; Yu, et al., 2020; Wu, et al., 2019b; Dutta, et al., 2021; Liu, et al., 2020a; Zhang, et al. 2023). For example, Liu, et al., reported Zr-modified chitosan beads having a high adsorption capacity for phosphate ions, reaching 60.60 mg/g (Liu and Zhang, 2015); Veenhuyzen, et al., reported a waste activated sewage sludge (WAS) having an adsorption capacity for Pb(II) of 307 mg/g (Veenhuyzen, et al., 2015); and Zhou et al., reported a ZSM-5 zeolite supporting sulfide nanoscale zero-valent iron (S-nZVI@ZSM-5) having an adsorption capacity for As (V) of 161.66 mg/g (Zhou, et al., 2021).
Many MOF-based adsorbents have been reported in recent years, however, most MOF materials are only used for adsorption treatment of one pollution factor (Zhang, et al., 2021c; Yu, et al., 2019) while the adsorbents studies of the MOFs have not been comprehensive. We have found that MOFs have high adsorption capacities for various pollutants (Wang, et al., 2021b; Zhang, et al. 2022a). Then, since some of the materials were synthesized with water as the solution, there was a phenomenon of decomposition in the aqueous solution, which was not conducive to being used as a water adsorption material.
In recently years, Mn-base MOFs have gotten a lot of attentions (Mbonu and Abiola, 2021; Chao, et al., 2020; Xu, et al., 2013), because the Mn-MOFs can be used as electrocatalysis, adsorbent, magnetic materials, et al.
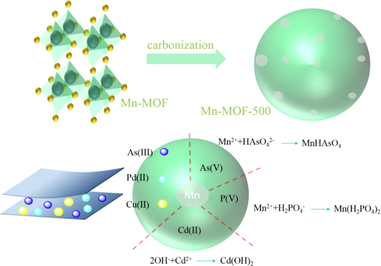
In this study, to overcome this disadvantage, a Mn-based MOF sorbent was carbonized at a high temperature to yield a carbon-based MOF composite material, and its adsorption performance was studied. Specifically, our MOF, denoted Mn-MOF ([Mn(DMF)2(H2DHTPA)]n), where DMF is N,N-dimethylformamide and H4DHTPA is 2,5-dihydroxyterephthalic acid, was synthesized by the solvothermal method. Carbonising Mn-MOF at 500℃ yielded Mn-MOF-500, which showed high water stability and good adsorption performance. The Mn-MOF-500 was used for the adsorption of phosphate and heavy metal ions (Cd(II), Pb(II), Cu(II), As(III), and As(V)) from simulated acid mine wastewater. The adsorption properties were analysed by adsorption experiments, and the adsorption mechanism was characterized by powder X-ray diffractometry (PXRD), Fourier transform infrared (FT-IR) spectrometry, X-ray photoelectron spectroscopy (XPS), and scanning electron microscopy (SEM).
2 Experiments and methods
2.1 Synthesis of Mn-MOF and Mn-MOF-n (n = 300, 400, 500, 600, 700, and 800 °C)
A mixture of H4DHTPA (5 mmol, 0.990 g), 1,3-di(4-pyridyl)propane (5 mmol, 0.990 g), MnCl2·4H2O (10 mmol, 1.979 g), and DMF (100 mL) was stirred for 30 min, and the pH was adjusted to 6 by addition of triethylamine. Then, the mixture was sealed in a 200-mL Teflon-lined stainless-steel vessel and heated at 150 °C for 48 h in an oven and slowly cooled to room temperature. Colorless block-shaped crystals were collected, washed with DMF, and dried in air. Phase pure crystals of Mn-MOF were obtained by manual separation (yield: 0.814 g, ca. 82.20 % based on H4DHTPA). Anal. Calcd. For Mn-MOF: C14H18MnN2O8 (Mr = 396.94), calcd.: C, 42.32; H, 4.53; N, 7.06%. Found: C, 42.26; H, 4.61; N, 7.15%. FT-IR data for Mn-MOF (Fig. S1, KBr, cm−1): 3384.2(vs), 2935.9(s), 1646.1(s), 1590.7(s), 1446.0(s), 1236.9(s), 1105.5(s), 781.5(s), 1491.2(m), 1387.5(m), 915.9(m), 860.7(m), 614.6 (m).
Mn-MOF-n (n = 300, 400, 500, 600, 700, and 800 °C). Mn-MOF-n were obtained by carbonising the Mn-MOF (1 g) in a tube furnace under oxygen conditions at the corresponding temperatures (n = 300, 400, 500, 600, 700, and 800 °C) for 240 min and allowing the samples to cool to room temperature.
2.2 Adsorption experiments and characterization
Relevant adsorption experiments, characterisation methods, and equations are shown in the Supporting Material, Section 1.2.
2.3 Single-crystal X-ray crystallography
Single-crystal X-ray diffraction data of Mn-MOF was collected on an Agilent G8910A CCD diffractometer with graphite monochromated Mo-Kα radiation (λ = 0.71073 Å) at 20 ± 1 °C in ω-θ scan mode. Raw frame data were integrated using SAINT, and the structure was solved using direct methods with SHELXT (Sheldrick, 2015) and refined by full-matrix least-squares on F2 using SHELXL-2018 within the OLEX-2 GUI (Dolomanov, et al., 2009). An empirical absorption correction was applied with spherical harmonics, as implemented in the SCALE3 ABSPACK scaling algorithm. All non-hydrogen atoms were refined anisotropically, and all hydrogen atoms were positioned geometrically and refined using the riding model. Calculations and visualisation were achieved using SHELXTL (Sheldrick, 2015). The crystallographic details are provided in Table S1. Selected bond lengths and angles for Mn-MOF are listed in Table S2.
3 Results and discussion
3.1 Crystal structure of [Mn(DMF)2(H2DHTPA)]n (Mn-MOF)
The reaction of H2DHTPA, 1,3-di(4-pyridyl)propane, MnCl2·4H2O, and DMF (100 mL) at 150 °C for 48 h, the Mn-MOF was synthesized (Scheme 1). The 1,3-di(4-pyridyl)propane ligand does not participate in the coordination.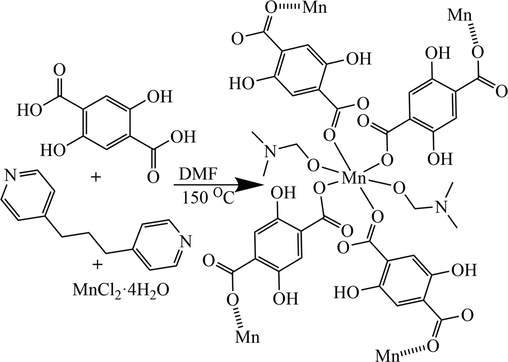
Synthetic routes for Mn-MOF.
The single-crystal X-ray diffraction analysis revealed that Mn-MOF crystallised in the monoclinic crystal system and the I2/a space group (Fig. 1, Table S1), and the asymmetric unit contained one crystallographically independent Mn(II) ion, one H2DHTPA ligand, and two DMF ligands, as shown in Fig. 1. Mn1 is six-coordinated by four oxygen (O) atoms from the carboxyl group of the four different H2DHTPA ligands (Mn1 − O3, 2.146(2) Å; Mn1 − O2B, 2.169(2) Å; symmetry code: (B) × -0.5, y + 0.5, z + 0.5.) and two O atoms from the two different DMF molecules (Mn1 − O1, 2.187(2) Å). Thus, Mn1 forms a slightly distorted octahedral geometry, whereas the cis-O-Mn-O bond angles are 180° (O1-Mn1-O1A; O3-Mn1-O3A; O2B-Mn1-O2C, 180°, symmetry codes: (A) 0.5 − x, y + 1, − z; (C) 2 − x, 1.5 + y, 0.5 − z.). The Mn-MOF forms a dinuclear unit through double carboxyl group bridges, which further form a 1D chain through carboxyl group bridge expansion (Fig. S2). The 1D chain constructed a 3D network through η1:η1:η1:η1-µ4-H2DHTPA bridge ligands (Fig. S3). Interestingly, there are strong intramolecular O-H···O hydrogen bonds (O4···O3, 2.586(3) Å), and the porosity of Mn-MOF, as calculated by Platon, is 53.6% (based on the omission of the DMF molecules), and a one-dimensional rhombus-shaped channel can be seen in the direction of the a-axis; the channel size is 15.288(1) × 11.986(2) Å 2 (Fig. 2). Herein, the distance of 15.288(1) Å is Mn1···Mn1i distance (symmetry code: (i) × − 1, y – 1, z.) while the distance of 11.986(2) Å is O2···O2ii distance (symmetry code: (ii) −0.5 − x, y, 1 – z.). In the one-dimensional rhombus-shaped channel, manganese atoms are not saturated in coordination and there are uncoordinated hydroxyl oxygen atoms.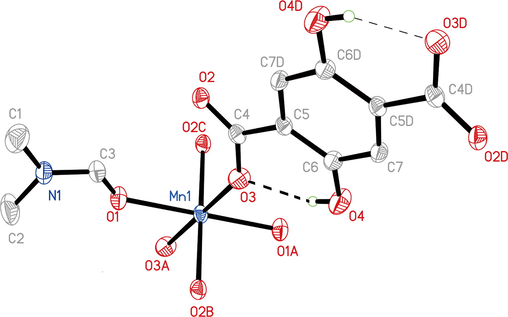
Molecule structure of Mn-MOF.
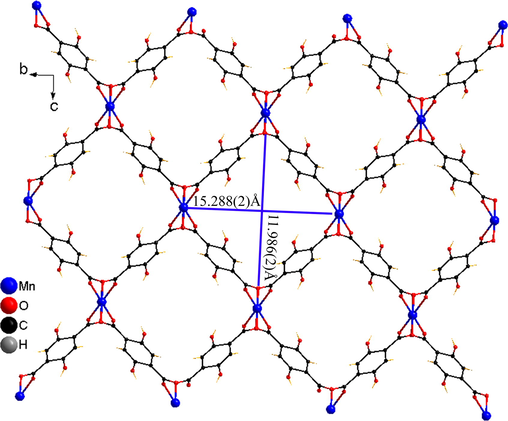
3D network of Mn-MOF, omitted the DMF molecules.
3.2 Selection of optimum carbonization temperature
As shown in Table 1, Mn-MOF showed good adsorption for PO43-, Cd(II), Pb(II), Cu(II), As(III), and As(V) ions. For Mn-MOF-n, the adsorption behaviors for the six ions were different. The adsorption capacities of Mn-MOF-n for the ions increased with the increase of carbonisation temperature, but, after carbonisation 500 °C, the adsorption capacity decreased. Thus, the optimal carbonisation temperature was 500 °C, and Mn-MOF-500 was used for subsequent studies.
Ions
Mn-MOF
300 °C
Mn-MOF400 °C
Mn-MOF500 °C
Mn-MOF600 °C
Mn-MOF700 °C
Mn-MOF800 °C
Mn-MOF
PO43-
93.08%
58.74%
66.82%
73.42%
69.54%
60.96%
53.08%
Cd(Ⅱ)
79.46%
39.72%
85.52%
93.34%
77.76%
38.32%
33.84%
Pb(Ⅱ)
98.04%
96.44%
98.21%
99.45%
95.22%
53.52%
80.81%
Cu(Ⅱ)
95.36%
48.94%
75.44%
96.91%
33.80%
13.62%
40.08%
As(III)
83.08%
9.48%
61.54%
79.16%
66.82%
39.16%
26.84%
As(V)
92.88%
39.48%
73.52%
96.88%
78.62%
57.28%
43.54%
3.3 Adsorption performance analysis of MN-MOF-500
3.3.1 Single-factor experiment of Mn-MOF-500
The pH and adsorbent dosage are important factors in adsorption experiments. Thus, the influence of these factors on the sorption of PO43-, Cd(II), Pb(II), Cu(II), As(III), and As(V) was investigated, as detailed in the Supporting Material, Section 1.3.1. The optimum pH for the adsorption of the six ions by Mn-MOF-500 was pH 5, and the optimum dosage for PO43-, Pb(II), Cu(II), and As(V) ions was 0.02 g, whereas that for Cd(II) and As(III) was 0.05 g.
3.3.2 Adsorption isotherm fitting
The adsorption mechanism and adsorption capacity were investigated using adsorption experiments. Fitting of the data to the Langmuir and Freundlich adsorption isotherms for PO43-, Cd(II), Pb(II), Cu(II), As(III), and As(V) at 288, 298, and 308 K was carried out, and the results are detailed in Table 2 and Fig. 3.
Ions
T(K)
qmaxEX*
/ (mg/g)Langmuir
Freundlich
qmax
/ (mg/g)
b
(L/mg)
R2
KF
n
R2
PO43-
288
75.69
79.38
0.06
0.99
12.86
2.90
0.91
298
93.83
98.26
0.05
0.99
14.84
2.77
0.93
308
121.30
126.88
0.05
0.99
17.82
2.63
0.94
Cd(II)
288
71.97
74.21
0.08
0.99
12.28
2.63
0.95
298
82.71
86.57
0.09
0.99
14.36
2.55
0.96
308
86.31
88.46
0.10
0.99
16.69
2.65
0.96
Pb(II)
288
262.43
257.83
0.54
0.99
95.57
5.52
0.91
298
321.3
318.07
1.25
0.98
134.60
6.01
0.87
308
357.6
356.21
2.53
0.97
165.61
6.62
0.84
Cu(II)
288
117.68
122.00
1.29
0.95
40.13
4.93
0.81
298
125.18
129.84
1.83
0.93
43.66
5.02
0.81
308
145.13
146.92
2.65
0.99
53.86
5.21
0.84
As(III)
288
37.47
38.17
0.06
0.98
6.84
3.04
0.95
298
43.11
43.55
0.24
0.99
11.06
3.68
0.93
308
50.79
51.28
0.24
0.98
12.43
3.52
0.95
As(V)
288
122.70
126.90
0.05
0.99
19.25
2.94
0.90
298
143.93
150.99
0.07
0.99
24.95
3.03
0.88
308
160.58
166.10
0.09
0.99
31.14
3.20
0.88
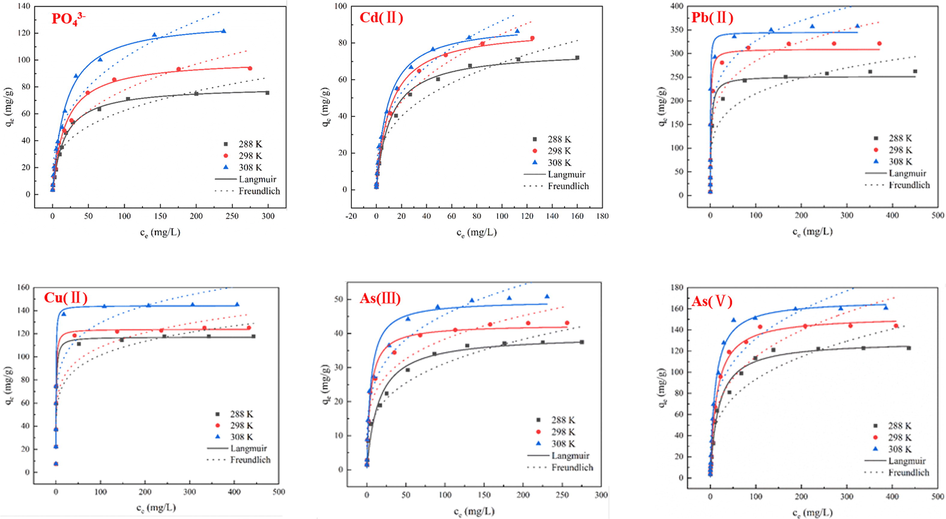
Nonlinear fitting to the Langmuir and Freundlich models of the adsorption of PO43-, Cd(II), Pb(II), Cu(II), As(III), and As(V) over Mn-MOF-500.
The coefficients of determination (R2) of the Langmuir model for PO43- adsorption were 0.99, 0.99, and 0.99 at 288, 298, and 308 K, respectively, higher than those of the Freundlich model at the same temperatures (0.91, 0.93, and 0.94, respectively). The results show that the adsorption of PO43- is monolayer adsorption, and the adsorption capacity is the same over the whole surface of the adsorbent. For the Langmuir model, the maximum adsorption capacities (qmax) of Mn-MOF-500 for PO43- at 288, 298, and 308 K were 79.38, 98.26, and 126.88 mg/L, respectively. The greatest value of qmax (126.88 mg/L) was obtained for PO43- (Liu and Zhang, 2015; Biswas et al., 2007; Xu et al., 2009; Köse and Kıvanç, 2011; Anirudhan et al., 2006).
For Cd(II), Pb(II), and Cu(II) ions, the Langmuir model fitted the data better than the Freundlich model, as shown by the R2 values (Table 2), indicating monolayer, homogenous adsorption on the surface of the material. The qmax values for Cd(II) are 72.21, 86.57, and 88.46 mg/g, respectively, The qmax values for Pb(II) are 257.83, 318.07, and 356.21 mg/g, and The qmax values for Cu(II) are 122.00, 129.84, and 146.92 mg/g at 288, 298, and 308 K, respectively. Further, the qmax values are 88.46, 356.21, and 146.92 mg/g for Cd(II), Pb(II), and Cu(II) ions, respectively, which are the largest reported values for the sorption of Cd(II) (Liu, et al., 2021; Xie, et al., 2021; Zhang, et al., 2021d; Ahmed, et al., 2020; Xu, et al., 2021), Pb(II) (Veenhuyzen, et al., 2021; Wang, et al., 2019a; Shi, et al., 2021; Wu, et al., 2019a; Wang, et al., 2021c), Cu(II) ions (Liu, et al., 2020b; He, et al., 2020; Zhang, et al., 2021a; Zhang, et al., 2021b; Jin, et al., 2021), respectively.
For the adsorption of As(III), and As(V) ions, the data fitted the Langmuir model better than the Freundlich model based on the R2 values (Table 2), indicating mainly monolayer adsorption. The qmax values of Mn-MOF-500 for As(III) were 38.17, 43.55, and 51.28 mg/g, respectively, and those for As(V) were 126.90, 150.99, and 166.10 mg/g at 288, 298, and 308 K, respectively. The qmax values were 51.28, 166.10 mg/g for As(III), As(V) ions, respectively, which are the largest reported values for the sorption of As(III) (Wang, et al., 2021a; Pervez, et al., 2021; Lin, et al., 2019; Mudzielwana, et al., 2019; Dong, et al., 2020), As(V) ions (Zhou, et al., 2021; Pervez, et al., 2021; Mahato, et al., 2021; Wang, et al., 2019b; Salih, et al., 2019), respectively.
Fitting to the Freundlich model yielded n ≥ 1 for PO43-, Cd(II), Pb(II), Cu(II), As(III), and As(V) ions at the three temperatures, indicating facile adsorption and qmax of Mn-MOF-500 on PO43-, Cd(II), Pb(II), Cu(II), As(III), and As(V) ions are 126.88, 88.46, 356.21, 146.92, 51.28, and 166.10 mg/g, respectively. Based on a comparison of these adsorption capacities with reported values, Mn-MOF-500 has excellent adsorption performance for these six ions (Table 3) (Liu and Zhang, 2015; Veenhuyzen, et al., 2021; Zhou, et al., 2021; Biswas, et al., 2007; Xu, et al., 2009; Köse and Kivanç, 2011; Anirudhan, et al., 2006; Liu, et al., 2021; Xie, et al., 2021; Zhang, et al., 2021d; Ahmed, et al., 2020; Xu, et al., 2021; Wang, et al., 2019a; Shi, et al., 2021; Wu, et al., 2019a; Wang, et al., 2021c; Liu, et al., 2020b; He, et al., 2020; Zhang, et al., 2021a; Zhang, et al., 2021b; Jin, et al., 2021; Wang, et al., 2021a; Pervez, et al., 2021; Lin, et al., 2019; Mudzielwana, et al., 2019; Dong, et al., 2020; Mahato, et al., 2021; Wang, et al., 2019b; Salih, et al., 2019).
ions
adsorbent
qm(mg/g)
Ref
PO43-
Mn-MOF-500
126.88
This work
La(III)-loaded saponified orange waste
14.00
Biswas, et al., 2007
Zr-treated chitosan beads
60.60
Liu and Zhang 2015
Modified wheat straw
22.90
Xu, et al., 2009
Calcined waste eggshell
32.60
Köse and Kivanç 2011
Banana stem (modified with DMAHPb)
23.60
Anirudhan, et al., 2006
Cd(II)
Mn-MOF-500
88.46
This work
RHBC
81.3
Liu, et al., 2021
AAM-MSM
78.1861
Xie, et al., 2021
MAC-300
73.3
Zhang, et al., 2021d
OCT-VER
69.595
Ahmed, et al., 2020
HA-Fe-PILC
32.15
Xu, et al., 2021
Pb(II)
Mn-MOF-500
356.21
This work
GO
273.0
Wang, et al., 2019a
waste activated sewage sludge
307
Veenhuyzen, et al., 2021
MnO2/PDA/Fe3O4
205.07
Shi, et al., 2021
P-MCS
151.06
Wu, et al., 2019a
BC400
124.7
Wang, et al., 2021c
Cu(II)
Mn-MOF-500
146.92
This work
A-Si/Mn-CRBC
141.76
Liu, et al., 2020b
PVA-CS-EDTA
127.81
He, et al., 2020
TSC-PVA
82.36
Zhang, et al., 2021a
CMBC600
70.42
Zhang, et al., 2021b
LESS600
65.61
Jin, et al., 2021
As(III)
Mn-MOF-500
51.28
This work
FRKF
37.4
Wang, et al., 2021a
α-FeOOH@GCA
32.19
Pervez, et al., 2021
FMLBC3
15.34
Lin, et al., 2019
SMK
2.33
Mudzielwana, et al., 2019
PSMPs
1.12
Dong, et al., 2020
As(V)
Mn-MOF-500
166.10
This work
S-nZVI@ZSM-5
161.66
Zhou, et al., 2021
ZnO/AlSBA-15
123.99
Mahato, et al., 2021
MCS/ZnO@Alg
63.69
Wang, et al., 2019b
α-FeOOH@GCA
61.91
Pervez, et al., 2021
CSD beads
44.07
Salih, et al., 2019
3.3.3 Adsorption kinetics
The data were fitted to the pseudo-first-order (PFO) and pseudo-second-order (PSO) kinetic models at 288, 298, and 308 K, and the results are shown in Table 4 and Fig. 4. Note:* qmaxEX means experimental value qmax.
Ions
T (K)
qmaxEX* (mg/g)
pseudo-first-order
pseudo-second-order
k (/min)
qe (mg/g)
R2
k [g/(mg min)]
qe (mg/g)
R2
PO43-
288
30.38
5.14
28.30
0.69
032
30.51
0.94
298
31.99
5.99
29.79
0.68
0.33
32.19
0.93
308
33.67
6.92
31.42
0.65
0.34
34.12
0.91
Cd(Ⅱ)
288
7.26
5.39
6.98
0.87
1.33
7.37
0.97
298
7.32
7.41
7.13
0.92
1.91
7.45
0.99
308
7.41
9.36
7.21
0.84
2.62
7.49
0.97
Pb(Ⅱ)
288
37.34
2.49
37.31
0.99
0.08
39.49
0.98
298
37.42
4.15
37.54
0.98
0.15
38.82
0.96
308
37.45
8.62
37.38
0.98
0.37
38.20
0.92
Cu(Ⅱ)
288
37.09
2.48
36.92
0.99
0.08
39.26
0.94
298
37.26
3.46
37.29
0.99
0.12
38.96
0.92
308
37.40
4.63
37.35
0.99
0.17
38.77
0.92
As(III)
288
7.18
3.28
6.94
0.92
0.72
7.37
0.96
298
7.29
3.61
7.01
0.89
0.82
7.44
0.96
308
7.37
5.99
7.18
0.93
1.44
7.52
0.98
As(V)
288
12.47
1.69
12.46
0.99
0.15
13.28
0.98
298
13.76
1.80
13.82
0.99
0.14
14.67
0.97
308
14.27
2.06
14.40
0.99
0.17
15.14
0.96
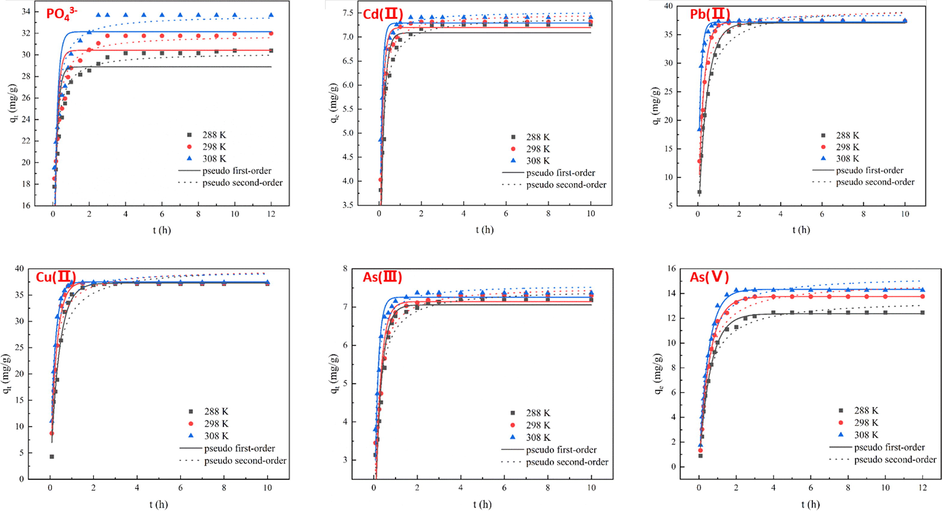
Pseudo-first-order kinetic and Pseudo-second-order kinetic nonlinear dynamics fitting of Mn-MOF-500 at 288.15 K, 298.15 K, and 308.15 K.
For the adsorption of PO43-, the PSO model fitted the data best: R2 values are 0.94, 0.93, and 0.91 at 288, 298, and 308 K, respectively. Thus, the adsorption rate is proportional to the square of the concentration, and the adsorption process is based on chemisorption.
For Cd(II), Pb(II), and Cu(II) ions, adsorption by Mn-MOF-500 can be divided into three stages: fast, slow, and equilibrium adsorption stages. With increase in temperature, the adsorption rates for Cd(II), Pb(II), and Cu(II) ions also accelerated, and the time to reach saturation was reduced. For Cd(II) adsorption, the PSO model fitted the data best, suggesting chemisorption. For Pb(II) and Cu(II) ions, the PFO model fitted best. The R2 for the PSO was greater than 0.9, indicating that the adsorption of Pb(II) and Cu(II) ions by Mn-MOF-500 is mainly physisorption supplemented by chemisorption.
For As(III), the fitting to the PSO model was better than that to the PFO model, and the equilibrium adsorption (qe) value calculated by the PSO kinetic model is close to the experimental qmaxEX, indicating that the adsorption of As(III) is chemisorption.
For As(V), the PFO model fitted best, yielding R2 values are 0.99, 0.99, and 0.99 at 288, 298, and 308 K, respectively, whereas those for the PSO model were 0.98, 0.97, and 0.96 at 288, 298, and 308 K, respectively. The fitting coefficients for the two kinetic models are both higher 0.95, suggesting a mix of physi- and chemisorptions.
3.3.4 Desorption and regeneration experiments
As shown in Fig. S4, the sorbent showed good recyclability for the sorption of Cd(II), Pb (II), and Cu(II) ions. After four cycles, the sorption capacities of Cd(II), Pb(II), and Cu(II) ions were 78.97%, 82.7%, and 79.02%, respectively. For PO43-, As(III), and As(V) ions, the sorption performance was not good on cycling, suggesting their poor desorption. Therefore, future experiments, investigation of desorption agents is required.
3.4 Effect of carbonisation
Water solubility analysis (see Supporting Material, Section 1.2.5 for experimental steps) showed that Mn-MOF was soluble in water, suggesting that it contains many hydrophilic groups and is unsuitable for wastewater treatment. In contrast, Mn-MOF-500 showed good water stability, indicating that carbonisation converts the Mn-MOF to a water-stable material.
The PXRD patterns of Mn-MOF and Mn-MOF-500 are shown in Fig. 5. The experimental pattern is consistent with the database pattern, indicating high purity and crystallinity. In contrast, the PXRD patterns of Mn-MOF-500 contain peak shifts, indicating a reduction in crystallinity on carbonisation.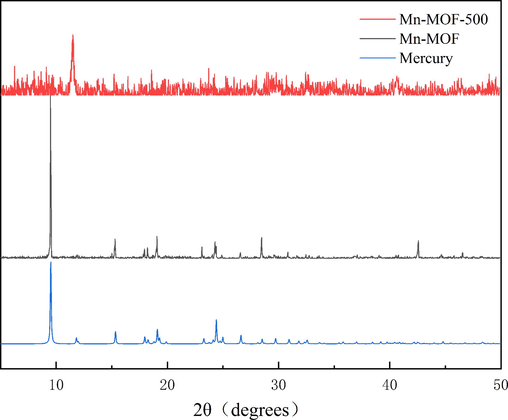
XRD image of Mn-MOF and Mn-MOF-500.
The Brunauer–Emmett–Teller (BET) nitrogen adsorption–desorption isotherm results are shown in Fig. S5. The BET specific surface area of Mn-MOF is 0.82 m2/g, the average pore size is 34.82 nm, and the average mesopore diameter is 5.36 nm. After carbonisation, these values were 11.83 m2/g, 25.15 nm, and 34.08 nm, respectively. Compared with that of Mn-MOF, the BET specific surface area of Mn-MOF-500 is 14 times larger than that of Mn-MOF. Although the average pore size is decreased, the average mesoporous diameter of Mn-MOF-500 is six times larger than that of Mn-MOF, indicating that high-temperature carbonisation modification can increase the specific surface area and mesopore size, which is conducive to enhancing the adsorption performance of Mn-MOF-500.
The thermogravimetric analysis (TGA) results are shown in Fig. S6. At 200 °C, the weight loss rate of Mn-MOF increased. At 650 °C, the weight of Mn-MOF was only 30% of the original weight. Thus, decomposition starts at 200 °C, probably as a result of the loss of DMF molecules. The boiling point of DMF is 153 °C, but the loss at 200 °C is likely a result of the interaction between Mn(II) ions and DMF molecules. At 258 °C, the weight loss of the Mn-MOF was 36.2% (theoretical weight loss of 36.8% based on the loss of two coordination DMF molecules). In contrast, Mn-MOF-500 showed greater thermal stability, indicating that no coordinated DMF molecules are present. Therefore, the TGA results indicate that carbonisation damaged the structure of Mn-MOF.
SEM imaging was carried out using a JMS-7900 scanning electron microscope, as shown in Fig. 6a and b. Comparative analysis of the morphology of Mn-MOF and Mn-MOF-500 shows that the former is composed of regular tetrahedral particles before carbonisation. In contrast, Mn-MOF-500 is fluffy and porous. In addition, the colour changed (Fig. 6c and d), and the single crystalline structure was lost, further indicating that carbonisation destroys the structure of Mn-MOF.
SEM image of Mn-MOF(a) and Mn-MOF-500(b), Solid sample drawing of Mn-MOF(c) and Mn-MOF-500(d).
The XPS results are shown in Fig. 7 and show that the elemental contents were not changed by carbonation.
3.5 Effects of ion adsorption
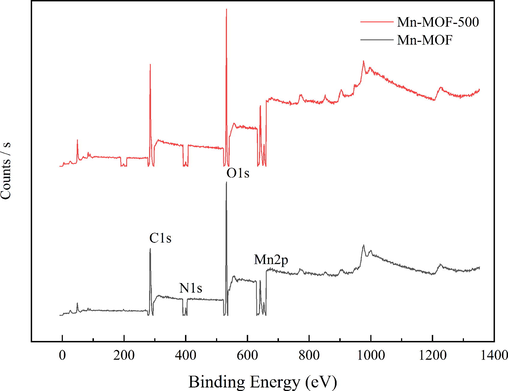
- Full XPS spectra of Mn-MOF and Mn-MOF-500.
Mn-MOF-500, Mn-MOF-500-P, Mn-MOF-500-Cd(II), Mn-MOF-500-Pb(II), Mn-MOF-500- Cu(II), Mn-MOF-500-As(III), and Mn-MOF-500-As(V) were characterised by PXRD, and the results are shown in Fig. S7. After adsorption, the main peaks corresponding to the Mn-MOF-500 remain, indicating that adsorption does not alter the structure. Jade 6 was used to analyse the PXRD data.
Based on the peaks at 14.676°, 11.711°, 11.218°, 11.393°, 11.445°, and 11.081°, PO43-, Cd(OH)2, PbO and Pb(OH)2, Cu(OH)2, Mn3(AsO4)2, and MnHAsO4 were present. In addition, the FTIR spectra of pristine Mn-MOF-500 and those after adsorption are shown in Fig. S8. There are no obvious changes before and after adsorption, suggesting that Mn-MOF-500 is stable and recyclable. Next, XPS was used to analyse the samples after adsorption, as shown in Fig. 8. For P, Cd(II), Pb(II), Cu(II), As(III), and As(V), the P2p peak at 133.92 eV indicates PO43-; Cd 3d5/2 and Cd 3d3/2 peaks at 405.63 and 412.36 eV, respectively, indicate Cd(OH)2; Pb 4f7/2 and Pb 4f5/2 peaks at 139.08 and 143.94 eV, indicate PbO and Pb(OH)2, respectively; Cu 2p3/2 peak at 934.79 eV indicate Cu(OH)2; As 3d peak at 48.80 eV indicates Mn3(AsO3)2, and As 3d peaks at 45.10 and 48.75 eV, respectively, indicate MnHAsO4. The SEM images of the materials after adsorption are shown in Fig. S9, revealing few changes to the structure after adsorption, suggesting the suitability of this material for industrial wastewater treatment.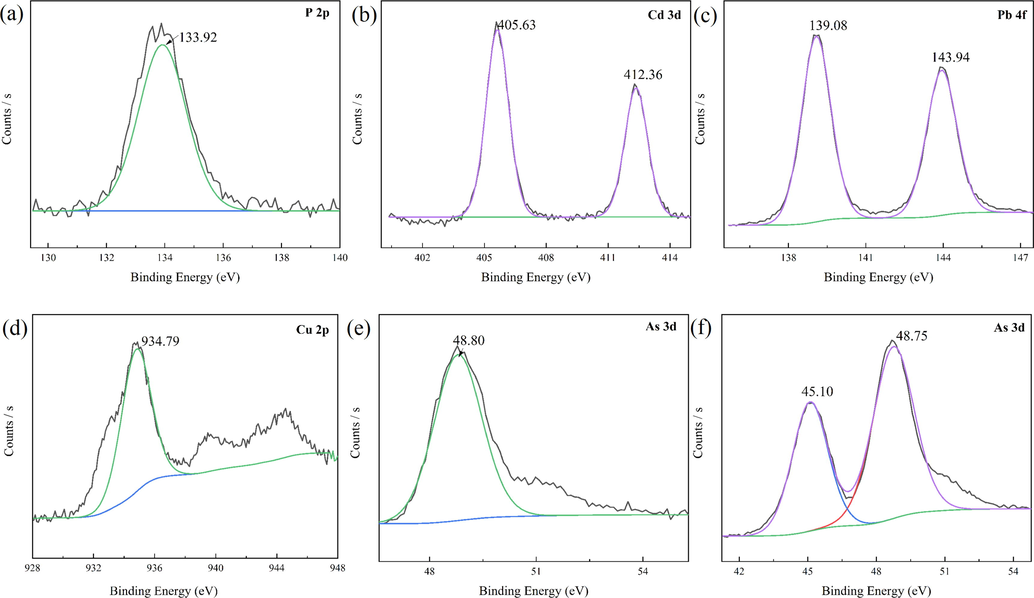
Fine spectrum of PO43-, Cd(II), Pb(II), Cu(II), As(III), and As(V) after adsorption by Mn-MOF-500.
3.6 Analysis of Mn-MOF-500 adsorption mechanism
The adsorption mechanism is shown in Fig. 9. Adsorption performance and mechanism of Mn-MOF-500 on PO43-: the research shown that the maximum adsorption capacity of Mn-MOF-500 on PO43 - was 126.66 mg/g, and its adsorption process conforms to Langmuir model and quasi second-order kinetic model. This adsorption process was single-molecule layer chemical adsorption. Dynamic adsorption experimental analysis shown that Mn-MOF-500 had strong adsorption persistence on PO43-, which was conducive to later industrial promotion and research. It was found that P was adsorbed on Mn-MOF-500 in the form of PO43- by XRD, IR, XPS and SEM.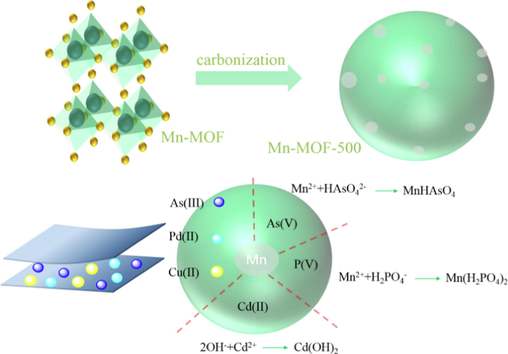
Mn-MOF-500 adsorption mechanism.
Adsorption performance and mechanism of Mn-MOF-500 on Cd(Ⅱ), Pb(Ⅱ) and Cu(Ⅱ): the study shown that the maximum adsorption capacity of Mn-MOF-500 on Cd(Ⅱ), Pb(Ⅱ) and Cu(Ⅱ) were 88.46, 356.21 and 146.92 mg/g, respectively. The isothermal adsorption experiments of Cd (II), Pb (II) and Cu (II) by Mn-MOF-500 and the fitting degree of Langmuir model were higher than that of Freundlich model. In the kinetic experiments, the adsorption process of Cd (II) by Mn-MOF-500 only conformed to the quasi second-order kinetic model, and the adsorption process of Pb (II) and Cu (II) by Mn-MOF-500 conformed to both the quasi first-order kinetic model and the quasi second-order kinetic model, indicating that the adsorption mechanism of Cd(II) by Mn-MOF-500 was monolayer chemisorption, The adsorption of Pb (Ⅱ) and Cu (Ⅱ) by Mn-MOF-500 were the result of the interaction of monolayer chemisorption and pore physical adsorption. According to the experimental data of adsorption and desorption, Mn-MOF-500 shown good cyclic regeneration for Cd (Ⅱ), Pb (Ⅱ) and Cu (Ⅱ). It was found that Cd (Ⅱ), Pb (Ⅱ) and Cu (Ⅱ) were adsorbed on Mn-MOF-500 in the form of Cd(OH) 2, PbO, Pb(OH)2 and Cu(OH)2 respectively by XRD, IR, XPS and SEM.
Adsorption performance and mechanism of Mn-MOF-500 for As(III), and As(V): the study shown that the maximum adsorption capacity of Mn-MOF-500 for As(III), and As(V) was 51.28 and 166.10 mg/g respectively. The adsorption process of Mn-MOF-500 on As(III), and As(V) was higher that of Langmuir model than Freundlich model. The adsorption process of Mn-MOF-500 on As (Ⅲ) only conforms to the quasi second-order kinetic model. The adsorption process of Mn-MOF-500 on As(V) conformed to both the quasi first-order kinetic model and the quasi second-order kinetic model, indicating that the adsorption of Mn-MOF-500 on As(III) was a single molecular layer chemisorption. The adsorption of As(Ⅴ) on Mn-MOF-500 has both monolayer chemisorption and pore physical adsorption. It was found that As(III), and As(V) were adsorbed on Mn-MOF-500 in the form of Mn3(AsO3)2 and MnHAsO4 by XRD, IR, XPS and SEM.
4 Conclusion
A water-soluble metal–organic framework material, Mn-MOF, was prepared using a solvothermal method. To reduce water solubility, direct carbonisation was used. The optimal carbonisation temperature was found to be 500 °C. Subsequently, Mn-MOF and Mn-MOF-500 were characterised using XRD, BET, XPS, and other methods, revealing that carbonisation destroyed the internal structure of Mn-MOF but did not affect the constituent elements. After carbonisation, Mn-MOF-500 had increased stability in water, the BET specific surface was increased 14 times to 11.83 m2/ g, and the size of the mesopores was expanded by six times to 34.08 nm, both of which are conducive to enhancing adsorption performance.
The maximum adsorption capacity of Mn-MOF-500 for PO43-, Cd(II), Pb(II), Cu(II), As(III), and As(V) was 126.66, 88.46, 356.21, 146.92, 51.28 and 166.10 mg/g, respectively. Compared with published research results, The maximum adsorption capacity of Mn-MOF-500 for PO43-, Cd(II), Pb(II), Cu(II), As(III), and As(V) are higher than the vast majority of reported adsorption materials. Further, XRD, IR, XPS, and SEM analysis revealed that PO43-, Cd (II), Pb (II), Cu (II), As(III), and As(V) were adsorbed on Mn-MOF-500 in the form of PO43-, Cd(OH)2, PbO, Pb(OH)2, Cu(OH)2, Mn3(AsO3)2, and MnHAsO4, respectively.
Adsorption for phosphorous, Cd(II), Pb(II), Cu(II), As(III), and As(V) were consistent with the Langmuir model. In addition, the fitting of the kinetic data for phosphorous, Cd(II), Pb(II), and Cu(II) conformed to pseudo-second-order kinetic model, pseudo second-order kinetic model, quasi first-order kinetic model and the pseudo first-order kinetic model, respectively. indicating that phosphorous, Cd(II), Pb(II) and Cu(II) were adsorbed by monolayer chemisorptions, monolayer chemisorptions, mainly physisorption and mainly physisorption, respectively. In addition, the adsorption of As(III) conforms to the pseudo-second-order kinetic model, whereas that of As(V) conforms to both the pseudo-first-order kinetic model and the pseudo-second-order kinetic model, indicating that As(III) was adsorbed by monolayer chemisorption, whereas As(V) is adsorbed by both monolayer chemisorption and physisorption in the pores. Recycling experiments with Cd(II), Pb(II), and Cu(II) revealed the good recycling performance of this material.
Authors contributions
Investigation: Zhao Chen has completed the synthesis of complexes Mn-MOF and Mn-MOF-n (300, 400, 500, 600, 700, 800 °C), elemental analysis, IR and single-crystal structures analysis of complex Mn-MOF, and Adsorption experiment and characterization, Analysis of Mn-MOF-500 adsorption mechanism. Yating Chen has completed the refine the structure of Mn-MOF, Selection of optimum modification temperature, Adsorption performance analysis of Mn-MOF-500, Comparative analysis of carbonization before and after, Characterization of Mn-MOF-500 before and after adsorption.
CRediT authorship contribution statement
Yating Chen: . Zhao Chen: . Lingyu Yuan: . Yu Xiao: Resources, Funding acquisition. Shu-Hua Zhang: Writing – review & editing, Supervision, Resources, Project administration, Funding acquisition. Ning Li: Writing – original draft, Supervision, Project administration.
Acknowledgments
This work was supported by the Nature Science Foundation of China (No. 21861014), and Projects of Talents Recruitment of Guangdong University of Petrochemical Technology (2020rc033, 2020rc035).
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Enhanced simultaneous adsorption of Cd (II) and Pb (II) on octylamine functionalized vermiculite. Colloid. Surf. A.. 2020;604:125285
- [Google Scholar]
- Adsorptive removal of wide range of pharmaceutical and personal care products from water by using metal azolate framework-6-derived porous carbon. Chem. Eng. J.. 2018;343:447-454.
- [Google Scholar]
- Phosphate removal from wastewaters using a weak anion exchanger prepared from a lignocellulosic residue. Environ. Sci. Technol.. 2006;40:2740-2745.
- [Google Scholar]
- Adsorptive removal of ibuprofen and diclofenac from water using metal-organic framework-derived porous carbon. Chem. Eng. J.. 2017;314:50-58.
- [Google Scholar]
- The adsorption of phosphate from an aquatic environment using metal-loaded orange waste. J. Colloid Interf. Sci.. 2007;312:214-223.
- [Google Scholar]
- Pristine S, N-containing Mn-based metal organic framework nanorods enable efficient oxygen reduction electrocatalysis. Dalton Trans.. 2020;49:4336-4342.
- [Google Scholar]
- Reticular access to highly porous acs-MOFs with rigid trigonal prismatic linkers for water sorption. J. am. Chem. Soc.. 2019;141:2900-2905.
- [Google Scholar]
- OLEX2: a complete structure solution, refinement and analysis program. J. Appl. Crystallogr.. 2009;42(2):339-341.
- [Google Scholar]
- As (III) adsorption onto different-sized polystyrene microplastic particles and its mechanism. Chemosphere. 2020;239:124792
- [Google Scholar]
- Multicomponent isoreticular metal-organic frameworks: principles, current status and challenges. Coordin. Chem. Rev.. 2021;445:214074
- [Google Scholar]
- Prediction of metal-adsorption behaviour in the remediation of water contamination using indigenous microorganisms. J. Environ. Manage.. 2011;92:2786-2793.
- [Google Scholar]
- Efficient adsorption of Cu (II) from aqueous solutions by acid-resistant and recyclable ethylenediamine tetraacetic acid-grafted polyvinyl alcohol/chitosan beads. J Mol. Liq.. 2020;316:113856
- [Google Scholar]
- Characterization of biochars derived from various spent mushroom substrates and evaluation of their adsorption performance of Cu (II) ions from aqueous solution. Environ. Res.. 2021;196:110323
- [Google Scholar]
- Adsorption of phosphate from aqueous solutions using calcined waste eggshell. Chem. Eng. J.. 2011;178:34-39.
- [Google Scholar]
- Bioaccumulation of heavy metals with environmental objects and assessment of health risks (the former mining enterprise Khingansky GOK as an example) Russ. J. Gen. Chem+.. 2014;84:2542-2544.
- [Google Scholar]
- Design and synthesis of an exceptionally stable and highly porous metal-organic framework. Nature. 1999;402:276-279.
- [Google Scholar]
- Three coordination compounds based on tris (1-imidazolyl) benzene: Hydrothermal synthesis, crystal structure and adsorption performances toward organic dyes. Polyhedron. 2018;139:89-97.
- [Google Scholar]
- Synthesis and adsorption of Fe-Mn-La- impregnated biochar composite as an adsorbent for As(III) removal from aqueous solutions. Environ. Pollut.. 2019;247:128-135.
- [Google Scholar]
- Recent developments in luminescent coordination polymers: Designing strategies, sensing application and theoretical evidences. Coordin. Chem. Rev.. 2020;406:213145
- [Google Scholar]
- Capacity and potential mechanisms of Cd (II) adsorption from aqueous solution by blue algae-derived biochars. Sci. Total. Environ.. 2021;767:145447
- [Google Scholar]
- Modification of calcium-rich biochar by loading Si/Mn binary oxide after NaOH activation and its adsorption mechanisms for removal of Cu (II) from aqueous solution. Colloid. Surface. A.. 2020;601:124960
- [Google Scholar]
- Removal of phosphate anions using the modified chitosan beads: adsorption kinetic, isotherm and mechanism studies. Powder. Technol.. 2015;277:112-119.
- [Google Scholar]
- Rapid adsorption of As (V) from aqueous solution by ZnO embedded in mesoporous aluminosilicate nanocomposite adsorbent: parameter optimization, kinetic, and isotherms studies. Surf. Interfaces.. 2021;23:100636
- [Google Scholar]
- Mbonu I.J., Abiola O.K., 2021. Adsorption of Nitrogen on Mn(II) Metal-Organic Framework Nanoparticles JOTCSA. 8(3) 941-952.
- Performance evaluation of surfactant modified kaolin clay in As (III) and As (V) adsorption from groundwater: adsorption kinetics, isotherms and thermodynamics. Heliyon. 2019;5:e02756.
- [Google Scholar]
- MIL-Ti metal-organic frameworks (MOFs) nanomaterials as superior adsorbents: Synthesis and ultrasound-aided dye adsorption from multicomponent wastewater systems. J. Hazard. Mater.. 2018;347:123-140.
- [Google Scholar]
- A bifunctional α-FeOOH@ GCA nanocomposite for enhanced adsorption of arsenic and photo Fenton-like catalytic conversion of As (III) Environ. Technol. Inno.. 2021;22:101437
- [Google Scholar]
- Removal of heavy metals from acid mine drainage (AMD) using coal fly ash, natural clinker and synthetic zeolites. J Hazard. Mater.. 2008;156:23-35.
- [Google Scholar]
- Competitive adsorption of As (III) and As (V) onto chitosan/diatomaceous earth adsorbent. J. Environ. Chem. Eng.. 2019;7:103407
- [Google Scholar]
- SHELXT–Integrated space-group and crystal-structure determination. Acta. Crystallogr. A.. 2015;71:3-8.
- [Google Scholar]
- High saturation magnetization MnO2/PDA/Fe3O4 fibers for efficient Pb (II) adsorption and rapid magnetic separation. Appl. Surf. Sci.. 2021;541:148379
- [Google Scholar]
- Adsorption of pharmaceuticals and personal care products over metal-organic frameworks functionalized with hydroxyl groups: quantitative analyses of H-bonding in adsorption. Chem. Eng. J.. 2017;322:366-374.
- [Google Scholar]
- Constructed wetlands as green tools for management of boron mine wastewater. Int. J. Phytoremediat.. 2014;16:537-553.
- [Google Scholar]
- High capacity Pb (II) adsorption characteristics onto raw-and chemically activated waste activated sludge. J. Hazard. Mater.. 2021;416:125943
- [Google Scholar]
- Fabrication of CdS and CuWO4 modified TiO2 nanoparticles and its photocatalytic activity under visible light irradiation. J. Ind. Eng. Chem.. 2014;20:435-443.
- [Google Scholar]
- Removal of nutrients in various types of constructed wetlands. Sci. Total. Environ.. 2007;380:48-65.
- [Google Scholar]
- Exploration of the adsorption performance and mechanism of zeolitic imidazolate framework-8@ graphene oxide for Pb (II) and 1-naphthylamine from aqueous solution. J. Colloid. Interf. Sci.. 2019;542:410-420.
- [Google Scholar]
- Fabrication of chitosan-based MCS/ZnO@ Alg gel microspheres for efficient adsorption of As (V) Int. J. Biol. Macromol.. 2019;139:886-895.
- [Google Scholar]
- Effects of cotton straw-derived biochar under different pyrolysis conditions on Pb (II) adsorption properties in aqueous solutions. J. Anal. Appl. Pyrol.. 2021;157:105214
- [Google Scholar]
- Adsorption of As (III) and As (V) by using the Fenton reaction modified kapok fiber. J. Environ. Chem. Eng.. 2021;9:105918
- [Google Scholar]
- A study of GUPT-2, a water-stable zinc-based metal–organic framework as a highly selective and sensitive fluorescent sensor in the detection of Al3+ and Fe3+ ions. CrstEngComm. 2021;23:4059-4068.
- [Google Scholar]
- Bi-Microporous Metal-Organic Frameworks with Cubane [M4(OH)4] (M= Ni, Co) Clusters and Pore-Space Partition for Electrocatalytic Methanol Oxidation Reaction. Angew. Chem.. 2019;131:12313-12317.
- [Google Scholar]
- Phosphorylated chitosan/CoFe2O4 composite for the efficient removal of Pb (II) and Cd (II) from aqueous solution: adsorption performance and mechanism studies. J. Mol. Liq.. 2019;277:181-188.
- [Google Scholar]
- Mechanically activated starch magnetic microspheres for Cd (II) adsorption from aqueous solution. Chinese. J. Chem. Eng.. 2021;33:40-49.
- [Google Scholar]
- Adsorption of phosphate from aqueous solutions onto modified wheat residue: characteristics, kinetic and column studies. Colloid. Surface. B.. 2009;70:46-52.
- [Google Scholar]
- Cd (II) and Pb (II) absorbed on humic acid-iron-pillared bentonite: kinetics, thermodynamics and mechanism of adsorption. Colloid. Surface. A.. 2021;612:126005
- [Google Scholar]
- Novel Mn(II)-Based Metal−Organic Frameworks Isolated in Ionic Liquids. Cryst. Growth Des.. 2013;13:1260-1266.
- [Google Scholar]
- Adsorption of boron from boron- containing wastewaters by ion exchange in a continuous reactor. J. Hazard. Mater.. 2005;117:221-226.
- [Google Scholar]
- Highly efficient and facile removal of Pb2+ from water by using a negatively charged azoxy-functionalized metal–organic framework. Cryst. Growth. Des.. 2020;20:5251-5260.
- [Google Scholar]
- Study of the dynamic adsorption and the effect of the presence of different cations and anions on the adsorption of As (V) on GUT-3. Appl. Organomet. Chem.. 2021;35:e6289.
- [Google Scholar]
- Zn Metal-Organic Framework with High Stability and Sorption Selectivity for CO2. Organometallics. 2022;41:829-835.
- [Google Scholar]
- Studies on the removal of phosphate in water through adsorption using a novel Zn-MOF and its derived materials. Arab. J. Chem.. 2022;15(8):103955.
- [Google Scholar]
- Highly efficient and facile removal of As(V) from water by using Pb-MOF with higher stable and fluorescence. Appl Organomet Chem.. 2023;37:e7066.
- [Google Scholar]
- Adsorption of Pb (II) and Cd (II) by magnetic activated carbon and its mechanism. Sci. Total. Environ.. 2021;757:143910
- [Google Scholar]
- Preparation of thiosemicarbazide-modified polyvinyl alcohol and its selective adsorption of Cu (II) Colloid. Interfac. Sci.. 2021;43:100377
- [Google Scholar]
- Characteristics, adsorption behaviors, Cu (II) adsorption mechanisms by cow manure biochar derived at various pyrolysis temperatures. Bioresource. Technol.. 2021;331:125013
- [Google Scholar]
- Simultaneous adsorption of As (V) and Cr (VI) by zeolite supporting sulfide nanoscale zero-valent iron: Competitive reaction, affinity and removal mechanism. J. Mol. Liq.. 2021;338:116619
- [Google Scholar]
Appendix A
Supplementary data
Supplementary data to this article can be found online at https://doi.org/10.1016/j.arabjc.2023.104950.
Appendix A
Supplementary data
Crystallographic data (excluding structure factors) for the structures in this paper have been deposited with the Cambridge Crystallographic Data Centre as supplementary publication CCDC 2165264 for Mn-MOF. Copies of the data can be obtained free of charge on application to CCDC, 12 Union Road, Cambridge CB2 1EZ, UK (fax: 44 1223 336033, e-mail: deposit@ccdc.cam.ac.uk). Electronic supplementary information (ESI) available: IR of Mn-MOF and Mn-MOF-500; 1D chain of the Mn-MOF; 3D network of Mn-MOF; Effect of regeneration times on Cd(II), Pb(II), Cu(II) removal rate; BET desorption curve of Mn-MOF (a), Mn-MOF-500 (b); TG curves; IR curves; SEM image; Effect of Ph; Effect of dosage; Crystallographic Data for Mn-MOF; Selected bond lengths (Å) and angles (°) for Mn-MOF. Materials and physical measurements; Adsorption Experimental and Characterization; Analysis of experimental results. The following are the Supplementary data to this article:Supplementary data 1
Supplementary data 1
Supplementary data 2
Supplementary data 2
Supplementary data 3
Supplementary data 3







