Translate this page into:
Analysis of the greenness to determine risperidone in commercial products using a green stability-indicating HPTLC method
⁎Corresponding author. p.alam@psau.edu.sa (Prawez Alam)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Abstract
The purpose of this work was to develop and verify a green stability-indicating “high-performance thin-layer chromatographic (HPTLC)” technique to measure risperidone (RISP). The ternary mixture of ethanol, ethyl acetate, and ammonia (70:20:10 v/v/v) was the green mobile phase that was employed. At 281 nm, the detection of RISP was performed. The suggested method's greener profile was produced using the analytical eco-scale (AES), ChlorTox, and analytical GREENness (AGREE) methodologies. It was then contrasted with RISP analysis published HPTLC methods. The proposed methodology for measuring RISP in the 50–1400 ng/band range showed the linearity. Furthermore, the RISP measurement methodology was green, robust, precise (% RSD = 0.80–1.02), sensitive (LOD = 1.86 ng/band and LOQ = 5.60 ng/band), and accurate (% recoveries = 98.23–101.80). The AES, total ChlorTox, and AGREE scales for the suggested technique were found to be 83, 1.26 g, and 0.75, indicating an excellent greener profile. The current methodology's AES and total ChlorTox were significantly higher than those of previously published HPTLC RISP analysis methods. RISP was found to be highly stable under acid, base, and thermal stress conditions, while it was found to be most unstable under oxidative degradation conditions. Additionally, the stability-indicating aspect of the suggested technique was able to identify RISP in the presence of its degradation products. It was discovered that commercial tablet brands A and B had 98.12 and 99.62 % of RISP, respectively. The results of this investigation suggested that RISP in commercial formulations might be regularly analyzed utilizing the suggested green technology.
Keywords
Green HPTLC
Greenness tools
Risperidone
Stability-indicating method
1 Introduction
Risperidone (RISP) is an approved antipsychotic drug, which comes under benzisoxazole class of an atypical neuroleptic medicines (Kumar et al., 2008; Kumar et al., 2009). The molecular structure of RISP is displayed in supplementary Fig. 1 (Figure S1) (Shukla et al., 2009; Shakeel et al., 2014). It is a potent medication that has been recommended for the treatment of Tourette syndrome, schizophrenia, bipolar mania, and psychotic depression (Bishop and Pavuluri, 2008; Bhat et al., 2023). At therapeutic levels, it has a low propensity to cause extrapyramidal side effects and selectively antagonizes dopamine and serotonin receptors (McDougle et al., 2005; Bhat et al., 2023). As a result, it shows fewer side effects than other antipsychotic medicines (McDougle et al., 2005). It is commercially available in form of conventional tablets, disintegrating tablets, and oral liquids (Kumar et al., 2008). It was discovered to be soluble in transcutol and weakly soluble in water (H2O) (Shakeel et al., 2014). Given that RISP is present in various marketed formulations, it is essential to examine both the qualitative and quantitative aspects of its presence in these products.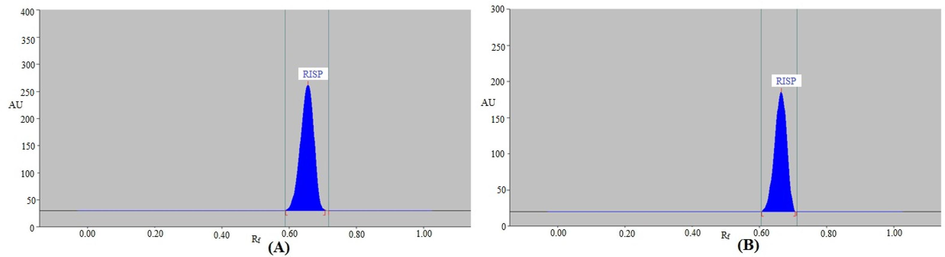
Representative chromatograms of (A) standard risperidone (RISP) and (B) marketed products for the current method.
RISP in biological materials and pharmaceutical products can be measured using a number of reported analytical techniques. According to reports, RISP in bulk form and pharmaceutical products can be determined using a few spectrophotometric techniques (Hasan et al., 2011; Kutty et al., 2013). Numerous “high-performance liquid chromatography (HPLC)” methods have been developed and validated to analyze RIPS in pure and dosage forms (Dedania et al., 2011; Svirskis et al., 2011; Ashour and Kattan, 2013; Mennickent et al., 2018; Kokane et al., 2019; Raslan et al., 2021; Yusul et al., 2022). To evaluate the photostability of RISP in tablet dosage forms, an HPLC approach has also been developed (Fujisawa et al., 2018). A green HPLC method using Corona charged aerosol detector was used to measure RISP and its related impurities in pharmaceutical products (Maljuric et al., 2020). Additionally, a number of HPLC approaches were developed and validated to measure RISP in the plasma and serum of rats and human participants (Aravagiri et al., 1993; Nagasaki et al., 1999; Schatz and Saria, 2000; Mennickent et al., 2019; Liu et al., 2020; Oloyede et al., 2022; Feyzi et al., 2023). The detection of RISP and its metabolites in human plasma samples as well as the evaluation of therapeutic drug monitoring (TDM) have both been addressed by a variety of liquid chromatography-tandem mass spectrometry (LC-MS)/MS methods that have been developed and validated (Remmerie et al., 2003; Huang et al., 2008; Koller et al., 2019; Alcantara et al., 2020). Additionally, RISP and its metabolites were identified in rat dried blood spots and dried plasma spots using an LC-MS/MS technique (Challa et al., 2020). Rat whole blood and urine samples were simultaneously evaluated for the presence of RISP, haloperidol, and chlorprothixene using the LC-MS/MS technique (Yu et al., 2020). The detection of RISP in human serum samples when combined with other antipsychotic medications and TDM assessment have also been reported using a variety of LC-MS/MS techniques (Ruan et al., 2018; Tonooka et al., 2018; Cao et al., 2020). Some ultra-performance LC (UPLC) methods have been developed and validated to determine RISP and its related impurities in pharmaceutical products (Nejedly et al., 2014; Magar et al., 2020; Pawar and Pandita, 2022). In order to identify RISP and its metabolites in rat plasma samples, a UPLC-MS/MS approach has also been reported (Rub et al., 2020). The spectrofluorometric measurement of RISP in spiked human plasma samples and pharmaceutical products was performed using guava fruit-based quantum dots (Alaqel et al., 2023). RISP can be found in human urine samples at the picogram level using a simple flow-injection chemiluminescence approach, according to a report (Xie et al., 2006). To ascertain RISP in pharmaceutical products, a capillary zone electrophoresis (CZE) technique has been created and verified (Nagase et al., 2014). A CZE method has also been used to determine RISP and its major metabolite, 9-hydroxy-RISP in beagle dog plasma samples (Fan et al., 2020). To ascertain RISP in bulk forms and commercialized products, numerous “high-performance thin-layer chromatography (HPTLC)” techniques have also been developed and verified (El-Sherif et al., 2005; Maslanka et al., 2009; Devala et al., 2010; Patel et al., 2010; Singh et al., 2017). Some HPTLC methods have also been used to measure RISP in combination with other antidepressants (Petruczynik et al., 2008; Patel et al., 2009; Patel et al., 2015). Additionally, RISP has been measured using an HPTLC technique in human plasma and urine samples in combination with other antidepressants (Maslanka et al., 2011).
There are numerous analytical methodologies for RISP analysis in pharmaceutical products and variety of biological samples, such as blood, plasma, serum, and urine. However, there is a paucity of information regarding green HPTLC methods for RISP determination in pharmaceutical or biological materials in the literature. Furthermore, for the majority of the stated RISP analysis methods, the greener profile was not reported. HPTLC techniques offer various advantages compared to conventional liquid chromatography techniques for drug analysis (El-Yazbi et al., 2021; Foudah et al., 2021). Currently, pharmaceutical products and medications are subjected to green analysis using HPTLC techniques (El-Yazbi et al., 2021; Ragab et al., 2023). Using environmentally safe alternatives to decrease the harmful effects of hazardous solvents on the environment is one of the twelve principles of “green analytical chemistry (GAC)” (Galuszka et al., 2013). Over the past few decades, the use of ecologically friendly or greener solvents has increased exponentially, according to a literature search (Abdelrahman et al., 2020; Alam et al., 2021; Foudah et al., 2021). A few of the many green analytical strategies that have been discussed in the literature to assess the greenness and eco-friendliness of analytical methodologies are the National Environmental Method Index (NEMI) (Keith et al., 2005), Environmental Assessment Tool (EAT) (Gaber et al., 2011), Analytical Method Volume Intensity (AMVI) (Hartman et al., 2011), Analytical Eco-Scale (AES) (Galuszka et al., 2012), Green Analytical Procedure Index (GAPI) (Plotka-Wasylka, 2018), Analytical Method GREENness Score (AMGS) (Hicks et al., 2019), Red, Green, and Blue (RGB) (Nowak and Koscielniak, 2019), the Analytical GREENness (AGREE) (Pena-Pereira et al., 2020), and ChlorTox (Nowak et al., 2023). Three different tools—AES (Galuszka et al., 2012), AGREE (Pena-Pereira et al., 2020), and ChlorTox (Nowak et al., 2023) approaches—were used in this study to evaluate the greener profile of the current method. The proposed method aims to develop and evaluate a green normal-phase HPTLC strategy for RISP analysis in commercial tablets based on the aforementioned data and observations. The suggested method for RISP analysis was validated by “The International Council for Harmonization (ICH)-Q2-R1” criteria (ICH, 2005).
2 Experimental section
2.1 Materials
The ammonia (NH3) solution (25 % for HPLC) and working standard of RISP were supplied by “Sigma Aldrich (St. Louis, MO, USA)”. “E-Merck (Darmstadt, Germany)” provided the LC-grade solvents, which included ethanol (EtOH), acetone (Ace), and ethyl acetate (EA). Using a “Milli-Q® (Milli-Q, Lyon, France)” device, LC-grade H2O was produced. The commercial RISP tablets, brand A (formulation A) and brand B (formulation B), each containing 2 mg of RISP, were supplied by pharmaceutical stores in “Riyadh, Saudi Arabia”. Rest of the chemicals/reagents have analytical grades.
2.2 Chromatography
RISP in marketed products was analyzed by an “HPTLC CAMAG TLC system (CAMAG, Muttenz, Switzerland)”. A “CAMAG Automatic TLC Sampler 4 (ATS4) Sample Applicator (CAMAG, Geneva, Switzerland)” was utilized to spot samples in the form of bands (6 mm). To measure RISP, the “NP-60F254S TLC plates (E-Merck, Darmstadt, Germany)” were used as the stationary phase. The sample applicator was filled with the “CAMAG microliter Syringe (Hamilton, Bonaduz, Switzerland)”. For every analysis, the RISP application rate was fixed at 150 nL/s. The TLC plates were placed inside an “automated development chamber 2 (ADC2) (CAMAG, Muttenz, Switzerland)” at a distance of 8 cm. The EtOH-EA-NH3 (70:20:10 v/v/v) was used as the green solvent system. For thirty minutes at an ambient temperature (22 °C), the development chamber was completely filled with the vapors from the green solvent system. At a wavelength of 281 nm, RISP was detected. The scanning speed was kept at 20 mm/s, and the slit diameter was fixed at 4 x 0.45 mm2. For each analysis, three or six replications were utilized. The “WinCAT's (version 1.4.3.6336, CAMAG, Muttenz, Switzerland)” software was utilized to decode the results and data.
2.3 RISP calibration plot
The RISP stock solution with a concentration of 100 µg/mL was obtained by dissolving 10 mg of RISP in 100 mL of the green solvent system. In order to get RISP concentrations in the range of 50–1400 ng/band, this stock solution was serially diluted. On normal-phase TLC plates, 10 µL of each RISP solution was spotted, and the required TLC response was recorded. The RISP calibration curve was made using a graph of the measured TLC response vs. RISP concentrations. For each of these solutions and experiments, six replicates (n = 6) were used.
2.4 Sample preparation for RISP analysis in marketed tablets
Twenty-five commercial tablets with the brands A (formulation A) and B (formulation B), each containing 2 mg of RISP, were weighed to determine their average mass. Both brands' tablets were ground to a uniform powder. The average mass of the fine powder from formulations A and B was mixed with 10 mL of the green solvent system. Then, 1 mL of formulations A and B was diluted using 10 mL of the green solvent system. After 30 min of sonication at 25 °C, the solutions of both formulations were filtered to remove any insoluble elements. Using the proposed method, the obtained samples of both formulations were checked for RISP contents.
2.5 Validation studies
The proposed RISP analytical method was validated using ICH-Q2-R1 protocols (ICH, 2005) for several parameters, as detailed below. The ICH-Q2-R1 guidelines are used for the evaluation of pharmaceutical assays. These guidelines are necessary for the registration of analytical procedures in EC, Japan, and USA. According to these guidelines, the analytical method should be validated for accuracy, precision, repeatability, specificity, robustness, LOD, LOQ, and linear range (ICH, 2005).
2.5.1 System acceptability
To determine the system's acceptability for the proposed method of RISP analysis, the estimation of “retardation factor (Rf), asymmetry factor (As), and theoretical plates number per meter (N/m)” was performed. The values of “Rf, As, and N/m” were obtained utilizing their reported equations (Foudah et al., 2021).
2.5.2 Linearity
RISP linearity was determined by plotting the observed TLC response vs. RISP concentrations. Using six replicates (n = 6), the linearity of the RISP analytical approach in the 50–1400 ng/band range was investigated.
2.5.3 Accuracy
Using the spiking/standard addition methodology, the intra-assay and inter-assay accuracy of the suggested RISP analytical approach was assessed in terms of % recoveries (ICH, 2005). To establish low quality-control (LQC) levels of RISP of 450 ng/band, moderate quality-control (MQC) levels of 600 ng/band, and high quality-control (HQC) levels of 750 ng/band, additional 50, 100, and 150 % RISP concentrations were spiked into the previously measured RISP solution (300 ng/band). The previously measured RISP QC solutions were subjected to a second study that same day in order to assess intra-day accuracy. The same QC solutions were reevaluated over a three-day period in order to gauge inter-day accuracy. The percentage recovery was computed for both accuracies and each QC level. We measured both accuracies with six replicates (n = 6).
2.5.4 Precision
The current RISP analysis method's precision was determined in terms of repeatability (intra-assay precision) and inter-assay (intermediate) precision. It was feasible to assess newly created RISP solutions at previously established QC levels on the same day. Thus, it was possible to establish the RISP intra-assay precision (repeatability). Measurements of newly prepared RISP solution at previously established QC levels were done over a three-day period in order to evaluate the RISP inter-assay precision. Six replicates (n = 6) were employed in order to assess both precisions. The precisions were given as a percentage of relative standard deviation (% RSD).
2.5.5 Robustness
The analytical procedure's robustness is assessed to verify the method's ability to withstand slight changes in its parameters (ICH, 2005). As a result, this work determined it. By introducing small deliberate changes to the green mobile phase composition during RISP analysis, the robustness for the present method was evaluated. For this, the original EtOH-EA-NH3 (70:20:10 v/v/v) system was changed to EtOH-EA-NH3 (72:18:10 v/v/v) and EtOH-EA-NH3 (68:22:10 v/v/v), and the necessary variations in peak response and Rf values were noted at each set of conditions.
2.5.6 Sensitivity
The sensitivity of the present RISP analysis method was determined in terms of “limit of detection (LOD) and limit of quantification (LOQ)” using the standard deviation technique (ICH, 2005). The blank solution (without RISP) was evaluated in six replicates (n = 6) using the recommended methodology, and the standard deviation was computed. Using the reported formulae, the RISP's “LOD and LOQ” were then computed utilizing the standard deviation of the intercept and slope of the RISP calibration plot (ICH, 2005; Alam et al., 2022).
2.5.7 Specificity
The Rf values, UV-absorption spectra, and 3D spectra of RISP in commercial formulations A and B were compared with those of pure RISP in order to evaluate the specificity of the method for RISP analysis.
2.6 Forced degradation studies
Four stress settings—acid (HCl), base (NaOH), oxidative (H2O2), and thermal degradation conditions—were used in studies on forced degradation (ICH, 2005; Haq et al., 2023). For every degradation study, a new MQC solution of RISP (600 ng/band) was made with the green solvent system. By combining 1 mL of MQC solution with 4 mL of either 1 M HCl or 1 M NaOH, acid and base hydrolysis were performed. The green solvent system was successful in diluting acid and alkaline hydrolysis solutions. After 48 h of refluxing at 60 °C, these samples were evaluated using the suggested method for RISP breakdown (ICH, 2005).
For oxidative degradation conditions, a fresh RISP MQC sample was prepared utilizing the green solvent system. After that, 1 mL of this solution was oxidized with 4 mL of 30 % H2O2. This mixture was effectively diluted utilizing the green solvent system. The proposed approach was used to assess the degradation of RISP in this combination after refluxing it for 48 h at 60 °C (ICH, 2005).
Once the RSIP MQC sample had been adequately diluted using the green solvent system, it was placed in a hot air oven and heated to 60 °C for 48 h. This led to the MQC (600 ng/band) solution's thermal hydrolysis. Then, RISP thermal deterioration was assessed using the suggested methodology (ICH, 2005).
2.7 Application of the current methodology in the determination of RISP in marketed formulations
For the current method, the solutions of commercial products A and B were spotted onto normal-phase TLC plates. The identical experimental setup as for standard RISP was utilized to calculate RISP, and three measurements of the peak area were made. The current methodology was utilized to calculate the percentage content of RISP in commercial products A and B using the RISP calibration curve.
2.8 Greenness assessment
Three distinct tools were utilized to obtain the greenness scale of the current technique in this study: AES (Galuszka et al., 2012), AGREE (Pena-Pereira et al., 2020), and ChlorTox (Nowak et al., 2023) approaches. AES is a semi-quantitative technique that considers instruments, waste, and each step of the analytical process. It is expected that substances requiring minimal to no reagent use, low energy, and no waste will have an ideal analysis with 100 points. Penalty points are awarded and deducted from 100 in total if any of these parameters are violated (Galuszka et al., 2012). According to the ChlorTox approach, equation (1) is used to calculate the ChlorTox scale (Nowak et al., 2023):
3 Results and discussions
3.1 Method development
Many binary and ternary combinations, including EtOH-H2O, Ace-H2O, EtOH-EA, EtOH-H2O-NH3, Ace-H2O-NH3, and EtOH-EA-NH3 in varying proportions, were evaluated as the green solvent systems in order to develop the current RISP analytical approach. Since the solvents under examination—EtOH, H2O, Ace, and EA—are harmless for the environment, they are classified as green solvents (Cue and Zhang, 2009; Prat et al., 2016). Each and every solvent system was created in a saturated chamber. Figure S2 displays a representative TLC image for the commercial products, forced-degradation samples, and reference RISP samples based on the optimal solvent systems. The measured chromatographic characteristics and the constituents of different binary and ternary green solvent system mixes are shown in Table 1. The unreliable RISP signals with higher As (As = 1.32–1.38) and lower N/m (N/m = 2013–2612) were observed when different combinations, such as EtOH-H2O (50:50 v/v), EtOH-H2O (70:30 v/v), Ace-H2O (50:50 v/v), Ace-H2O (70:30 v/v), EtOH-H2O-NH3 (70:20:10 v/v/v), and Ace-H2O-NH3 (70:20:10 v/v/v), were investigated. EtOH: ethanol; H2O: water; Ace: acetone; EA: ethyl acetate; Rf: retardation factor; As: asymmetry factor; N/m: theoretical plates number per meter.
Green solvent system
As
N/m
Rf
EtOH-H2O (50:50 v/v)
1.36 ± 0.06
2315 ± 2.11
0.76 ± 0.04
EtOH-H2O (70:30 v/v)
1.34 ± 0.05
2476 ± 2.18
0.74 ± 0.03
EtOH-H2O-NH3 (70:20:10 v/v/v)
1.32 ± 0.04
2612 ± 2.31
0.72 ± 0.02
Ace-H2O (50:50 v/v)
1.38 ± 0.06
2013 ± 1.98
0.80 ± 0.04
Ace-H2O (70:30 v/v)
1.37 ± 0.05
2221 ± 2.02
0.78 ± 0.03
Ace-H2O-NH3 (70:20:10 v/v/v)
1.35 ± 0.04
2388 ± 2.13
0.76 ± 0.02
EtOH-EA (50:50 v/v)
1.24 ± 0.05
3214 ± 2.65
0.70 ± 0.03
EtOH-EA (70:30 v/v)
1.18 ± 0.04
3422 ± 2.74
0.68 ± 0.02
EtOH-EA-NH3 (70:20:10 v/v/v)
1.09 ± 0.03
4784 ± 2.90
0.66 ± 0.01
The RISP chromatographic signals were slightly improved with low As (As = 1.09–1.24) and high N/m values (N/m = 3214–4784) when different combinations of EtOH-EA (50:50 v/v), EtOH-EA (70:30 v/v), and EtOH-EA-NH3 (70:20:10 v/v/v) were tested (Table 1). Out of all these combinations, the green EtOH-EA-NH3 (70:20:10 v/v/v) solvent system was the most distinct, showing a distinct and continuous RISP signal at Rf = 0.66 ± 0.01 (Fig. 1A). Furthermore, it was found that RISP had a 1.09 As value, which is an appropriate value for RISP analysis. So EtOH-EA-NH3 (70:20:10 v/v/v) has been selected as the most ecologically friendly solvent system for the suggested RISP analysis procedure. For the selection of an optimum wavelengths, the RISP spectral bands were recorded in absorbance mode at 200–400 nm range. It was discovered that 281 nm had the highest HPTLC response. As a consequence, the 281 nm was selected as an optimum wavelength for the entire RISP measurements.
3.2 Validation studies
As shown below, the ICH-Q2-R1 recommendations (ICH, 2005) were used to establish a number of validation variables for RISP analysis:
3.2.1 System acceptability
The system acceptability parameters for the proposed methodology were derived using their standard formulae. The “Rf, As, and N/m” for the proposed technique's RISP analysis were computed to be 0.66 ± 0.01, 1.09 ± 0.03, and 4784 ± 2.94, respectively, and were found to be suitable for RISP analysis.
3.2.2 Linearity
Table 2 displays the findings of the linearity evaluation for the RISP calibration plot using the current approach. For the concentration range of 50–1400 ng/band, it was discovered that the RISP calibration curve for the current technique was linear. For the suggested methodology, the RISP's determination coefficient (R2) and regression coefficient (R) were, respectively, 0.9972 and 0.9985. These results showed an outstanding relationshipt between the recorded response and the RISP concentrations. These results proved that the existing RISP analysis method is linear.
Parameters
Data
Linear range (ng/band)
50–1400
Regression equation
y = 13.911x + 1810.6
R2
0.9972
R
0.9985
Standard error of slope
0.23
Standard error of intercept
3.21
95 % confidence interval of slope
12.89–14.93
95 % confidence interval of intercept
1796.77–1824.42
LOD ± SD (ng/band)
1.86 ± 0.02
LOQ ± SD (ng/band)
5.60 ± 0.06
3.2.3 Accuracy
The present RISP analytical assay's two degrees of accuracy were achieved by using the spiking approach discussed under experimental section. Table 3 presents the resulting data of the % recovery utilizing the suggested technique. Using the suggested approach, it was determined that the intra-day% recoveries of RISP at three different QC levels ranged from 98.61 to 100.93 %. At three different QC levels, the RISP inter-day % recoveries for the suggested technique were found to range from 98.23 to 101.80 %. The outcomes demonstrated that RISP could be accurately measured using the current method.
Conc. (ng/band)
Conc. found (ng/band) ± SD
Recovery (%)
RSD (%)
Intra-day accuracy
450
454.22 ± 4.87
100.93
1.07
600
592.98 ± 5.97
98.83
1.00
750
739.64 ± 6.51
98.61
0.88
Inter-day accuracy
450
458.12 ± 5.10
101.80
1.11
600
589.41 ± 6.08
98.23
1.03
750
738.91 ± 6.61
98.52
0.89
3.2.4 Precision
The RISP analysis precision are expressed as %RSD for two degree of precisions. The results of both precisions for the current RISP detection method are displayed in Table 4. For the suggested technique, the intra-day precision RSD of RISP was derived to be between 0.80 and 0.97 %. It was observed that the inter-day precision of the current assay has an RSD of RISP ranging from 0.83 to 1.02 %. These findings showed that the RISP can be precisely determined using the current technique. SE: standard error; RSD: relative standard deviation.
Conc. (ng/band)
Intra-day precision
Inter-day precision
Conc. (ng/band) ± SD
SE
RSD (%)
Conc. (ng/band) ± SD
SE
(%) RSD
450
445.23 ± 4.35
1.77
0.97
447.58 ± 4.58
1.87
1.02
600
612.31 ± 5.76
2.35
0.94
604.28 ± 5.92
2.41
0.97
750
761.12 ± 6.14
2.50
0.80
755.64 ± 6.31
2.57
0.83
3.2.5 Robustness
The robustness of the proposed RISP analytical approach was derived by deliberately changing the percentage of the green solvent mixture. Table 5 shows the resulting data of the robustness evaluation for the proposed approach. The RISP % RSD of the current approach is calculated to be between 1.01 and 1.06 %. For the current technique, the RISP Rf values varied between 0.65 and 0.67. These outcomes proved that the present RISP analysis method is robust.
Conc. (ng/band)
Green mobile phase (EtOH-EA-NH3)
Results
Original
Used
Level
Conc. (ng/band) ± SD
RSD (%)
Rf
72:18:10
+2.0
586.31 ± 5.96
1.01
0.65
600
70:20:10
70:20:10
0.0
597.63 ± 6.16
1.03
0.66
68:22:10
−2.0
607.41 ± 6.48
1.06
0.67
3.2.6 Sensitivity
To assess the sensitivity of the current RISP analysis method, the “LOD and LOQ” were derived. The computed “LOD and LOQ” of RISP for the suggested methodology are shown in Table 2. The “LOD and LOQ” of RISP are 1.86 ± 0.02 and 5.60 ± 0.06 ng/band, respectively, based on the estimates in Table 2. The results showed how sensitive the available RISP measurement technology is.
3.2.7 Specificity
By contrasting the Rf data, UV-absorption spectrum, and 3D spectrum of RISP in commercially available products to those of standard RISP, the specificity of the current method for RISP analysis was evaluated. The superimposed UV-absorption spectrum of standard RISP and commercially available products are displayed in Fig. 2. The 3D spectrum of standard RISP and commercially available products are displayed in Figure S3.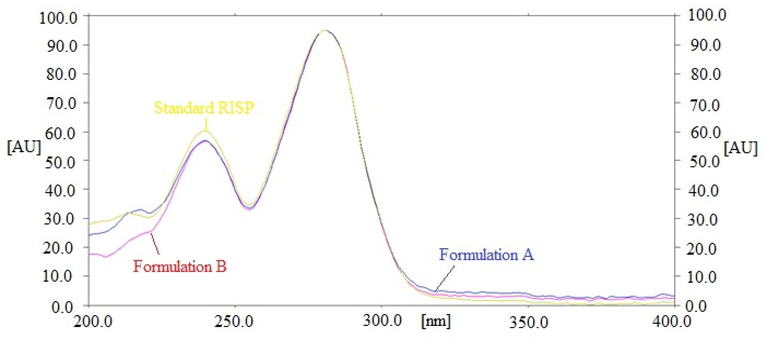
UV-absorption spectrum of reference RISP and formulations A and B.
The peak purity of pure RISP and RISP in commercial tablet brands A and B was evaluated by comparing the spectra at the peak start (S), peak apex (M), and peak end (E) positions of the spots (Youssef et al., 2012; Youssef et al., 2014). The calculated values of r (S,M) and r (M,E) of pure RISP and commercial tablets were found to be greater than 0.99, indicating the peaks’ homogeneity (Youssef et al., 2014, Shakeel et al., 2024). The UV-absorption and 3D spectra of standard and commercial tablets were same. At a wavelength of 281 nm, RISP in standard and commercially available products showed the greatest chromatographic response. Furthermore, the Rf values of pure RISP and commercial tablets were also same. Overall, the UV-absorption spectrum, 3D spectrum, Rf data, and wavelengths in standard and commercially available products were same. These results indicated the specificity of the present RISP analysis method.
3.3 Comparison of present method’s validation parameters with reported HPTLC methods
The present method for the measurement of RISP was compared with five different literature HPTLC methods in terms of linear range, accuracy, precision, LOD, and LOQ. The results of comparisons are summarized in Table 6. The linear range for five different HPTLC methods has been reported to be 2000–10000 ng/band (El-Sherif et al., 2005), 90–140 ng/band (Maslanka et al., 2009), 100–600 ng/band (Patel et al., 2010), 4000–8000 ng/band (Devala et al., 2010), and 100–700 ng/band (Singh et al., 2017), respectively. The linear range of present method of RISP analysis (50–1400 ng/band) has been found superior to all of the reported HPTLC methods. The accuracy of all reported HPTLC methods was within the magnitude of 100 ± 2 %, and hence found to be similar to the current RISP analysis method (El-Sherif et al., 2005; Maslanka et al., 2009; Devala et al., 2010; Patel et al., 2010; Singh et al., 2017). Compared to one of the documented HPTLC approaches (Maslanka et al., 2009), the current method's RISP precision was superior. However, it was similar to the remaining four HPTLC methods (El-Sherif et al., 2005; Devala et al., 2010; Patel et al., 2010; Singh et al., 2017). The LOD for five different HPTLC methods has been reported to be 249.69 ng/band (El-Sherif et al., 2005), 220 ng/band (Maslanka et al., 2009), 22.44 ng/band (Patel et al., 2010), 197.69 ng/band (Devala et al., 2010), and 46.56 ng/band (Singh et al., 2017), respectively. However, the LOQ for five different HPTLC methods has been found to be 629.85 ng/band (El-Sherif et al., 2005), 670 ng/band (Maslanka et al., 2009), 68.01 ng/band (Patel et al., 2010), 599.01 ng/band (Devala et al., 2010), and 141.11 ng/band (Singh et al., 2017), respectively. Compared to all previously published HPTLC approaches, the current method's LOD (1.86 ng/band) and LOQ (5.60 ng/band) of RISP were significantly lower. Therefore, compared to all previously published HPTLC approaches for RISP assessment, the current method is found to be far more sensitive. Overall, it has been discovered that the proposed RISP analysis approach is more linear and highly-sensitive than any of the HPTLC methods that have been previously published.
Analytical method
Linear range (ng/band)
Accuracy (% recovery)
Precision (% RSD)
LOD (ng/band)
LOQ (ng/band)
Ref.
HPTLC
2000–10000
100.08
1.17
249.69
629.85
(El-Sherif et al., 2005)
HPTLC
90–140
98.20–100.82
1.87–2.56
220
670
(Maslanka et al., 2009)
HPTLC
100–600
99.26–100.22
0.45–1.34
22.44
68.01
(Patel et al., 2010)
HPTLC
4000–8000
98.32–99.27
0.26–0.51
197.69
599.08
(Devala et al., 2010)
HPTLC
100–700
99.08–99.95
0.89–1.95
46.56
141.11
(Singh et al., 2017)
HPTLC
50–1400
98.23–101.80
0.80–1.02
1.86
5.60
Present work
3.4 Forced degradation studies
Under four different stress situations, the present methodology's forced degradation was examined. The results are shown in Table 7 and data are plotted in Fig. 3. It was discovered that RISP did not degrade since it remained at 100.00 % under the acid-, base-, and thermal-degradation stress settings (Table 7). Consequently, it was deduced that RISP exhibited remarkable stability in the face of stressors conditions such as acid, base, and heat degradation. It was discovered that the Rf value of RISP remained constant (Rf = 0.66) under the acid (Fig. 3A), base (Fig. 3B), and thermal-degradation (Fig. 3D) stress settings. Under oxidative degradation condition, RISP remained at 40.82 %, while 59.18 % was decomposed after stress setting (Table 7 and Fig. 3C). As a result, it was observed that RISP was sufficiently unstable at oxidative settings. HPTLC signals 1, 2, 3, and 4 in Fig. 3C, which stand in for the breakdown product signals, were separated by Rf values of 0.19, 0.35, 0.52, and 0.54, respectively. The Rf value for RISP under oxidative degradation remained constant at 0.66. The maximal RISP breakdown was discovered during an oxidative degradation test using the current method. These findings all pointed to the possibility that RISP may be detected with the current method even in the presence of its breakdown components. These outcomes proved the current method's selectivity and stability-indicating qualities.
Stress setting
Number of degradation products (Rf)
RISP Rf
RISP remained (ng/band)
RISP recovered (%)
1 M HCl
0
0.66
600.00
100.0 ± 0.00
1 M NaOH
0
0.66
600.00
100.00 ± 0.00
30 % H2O2
4 (0.19, 0.35, 0.52, 0.54)
0.66
244.92
40.82 ± 1.77
Thermal
0
0.66
600.00
100.00 ± 0.00
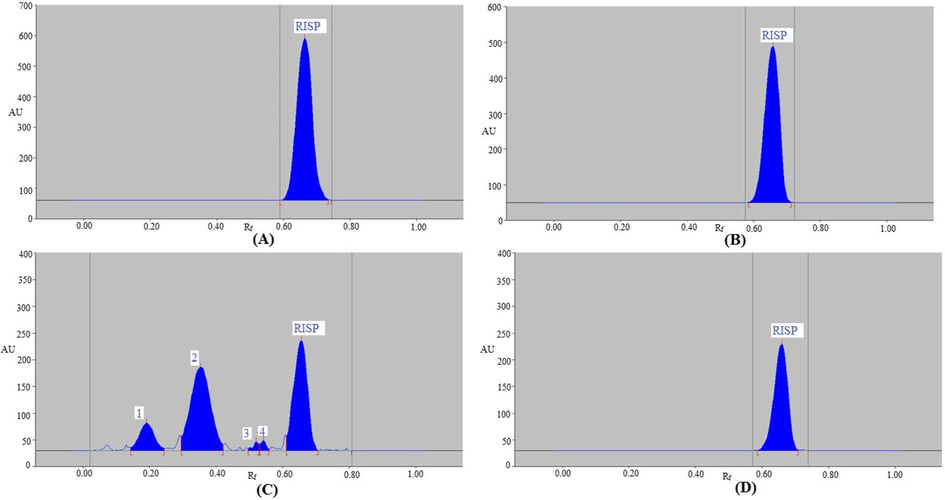
Representative spectrodensitograms of RISP recorded under (A) acid (B) base, (C) oxidative, and (D) thermal stress degradation conditions of RISP.
3.5 Application of the present technique in RISP assay in commercial formulations
In place of conventional liquid chromatographic techniques, the current technology was used to analyze RISP in commercial formulations A and B. By comparing the TLC band at Rf = 0.66 ± 0.01 for RISP with standard RISP using the suggested method, the chromatogram of RISP from the marketed tablets was verified. The chromatograms of RISP in both formulations were identical to the reference RISP when assessed using the suggested methodology. The lack of excipient peaks in both formulations indicated that there was no interaction between the formulations' ingredients and RISP (Fig. 1B). Using the RISP calibration curve, the amount of RISP in formulations A and B was determined. Formulations A and B had 98.12 ± 0.83 % and 99.62 ± 1.11 % of RISP, respectively, according to the current technique. The mean % RISP content in commercial tablets has been reported as 99.50 ± 0.46 % (Devala et al., 2010). The mean % RISP content in commercial tablets was found to be 99.64 ± 0.19 % by another report (Singh et al., 2017). The outcomes of the present RISP analysis method in commercial tablets were compared with previously reported HPTLC methods using the Student’s t-test and the variance ratio F-test (Devala et al., 2010; Singh et al., 2017). The obtained t and F values did not surpass their theoretical values, indicating that there were no appreciable variations in the accuracy and precision of the compared methods. These results suggested that the existing approach was suitable for the RISP pharmaceutical analysis.
3.6 Greenness assessment
For the evaluation of the analytical procedure's greenness, numerous green analysis methods such as NEMI (Keith et al., 2005), EAT (Gaber et al., 2011), AMVI (Hartman et al., 2011), AES (Galuszka et al., 2012), GAPI (Plotka-Wasylka, 2018), AMGS (Hicks et al., 2019), RGB (Nowak and Koscielniak, 2019), AGREE (Pena-Pereira et al., 2020), and ChlorTox (Nowak et al., 2023) have been developed. The current approach's greenness was evaluated using three distinct methods in this work: AES, ChlorTox, and AGREE methodologies. Table 8 presents the AES scales with penalty points for the current method compared to published HPTLC methods. The suggested method's AES scale, which was calculated to be 83, showed a very good green profile. The results of El-Sherif et al. (2005), Maslanka et al. (2009), Devala et al. (2010), Patel et al. (2010), and Singh et al. (2017) showed that the AES scales for five distinct literature HPTLC approaches ranged from 59 to 75. The AES results showed that compared to previously published HPTLC methods, the current technology was significantly more environmentally friendly.
Reagents/instruments/waste
Penalty points
HPTLC (El-Sherif et al., 2005)
HPTLC (Maslanka et al., 2009)
HPTLC (Patel et al., 2010)
HPTLC (Devala et al., 2010)
HPTLC (Singh et al., 2017)
Present method
EtOH
–
–
–
4
–
4
EA
–
–
4
–
–
4
NH3 (25 %, HPLC)
–
–
–
–
–
6
Methanol
18
12
18
18
18
–
n-Butanol
–
12
–
–
–
–
Acetic acid
–
4
–
–
–
–
Water
–
0
–
–
–
–
Dichloromethane
–
–
–
2
–
–
Triethylamine
–
–
–
6
12
–
Acetonitrile
8
–
–
–
8
–
Propanol
6
–
–
–
–
–
Triethanolamine
6
–
–
–
–
–
Instruments
0
0
0
0
0
0
Waste
3
3
3
3
3
3
Total penalty points
41
31
25
33
41
17
AES scale
59
69
75
67
59
83
The results of total ChlorTox scale and the individual greener solvent ChlorTox scale for the proposed approach are shown in Table 9 along with a comparison to published HPTLC methods. It was predicted that the proposed method's total ChlorTox scale would be 1.26 g, making it both safer and more environmentally beneficial (Nowak et al., 2023). Five distinct literature HPTLC methods were expected to have total ChlorTox scales ranging from 1.93 to 2.14 g, suggesting that all of the published procedures were both unsafe and less green (El-Sherif et al., 2005; Maslanka et al., 2009; Devala et al., 2010; Patel et al., 2010; Singh et al., 2017). The current method is safer and more environmentally friendly than any of the previously published HPTLC methods, according to the ChlorTox data.
Stage
Solvent/reagent
Relative hazard (CHsub/CHCHCl3)
msub (mg)
ChlorTox (g)
Total ChlorTox (g)
Ref.
Sample preparation
EtOH
0.26
1400
0.36
EA
0.34
400
0.13
NH3
0.73
200
0.14
HPTLC analysis
EtOH
0.26
1400
0.36
EA
0.34
400
0.13
NH3
0.73
200
0.14
1.26
Present method
Sample preparation
Methanol
0.56
2000
1.12
HPTLC analysis
Acetonitrile
0.39
1619
0.63
Methanol
0.56
229
0.13
Propanol
0.39
114
0.05
1.93
(El-Sherif et al., 2005)
Sample preparation
Methanol
0.56
2000
1.12
HPTLC analysis
n-Butanol
0.60
1200
0.72
Acetic acid
0.43
300
0.13
1.97
(Maslanka et al., 2009)
Sample preparation
Methanol
0.56
2000
1.12
HPTLC analysis
Methanol
0.56
1600
0.89
EA
0.34
400
0.13
2.14
(Patel et al., 2010)
Sample preparation
Methanol
0.56
2000
1.12
HPTLC analysis
Dichlorometane
0.48
797.34
0.38
Methanol
0.56
797.34
0.45
EtOH
0.26
398.67
0.10
Triethylamine
0.78
6.65
0.00
2.05
(Devala et al., 2010)
Sample preparation
Methanol
0.56
2000
1.12
HPTLC analysis
Acetonitrile
0.39
1923
0.75
Triethylamine
0.78
77
0.06
1.93
(Singh et al., 2017)
Since AGREE incorporates all 12 GAC criteria, this approach is the most used quantitative method for evaluating greenness (Pena-Pereira et al., 2020). According to Pena-Pereira et al., (2020), an AGREE score of 0.75 or higher denoted an excellent greenness, 0.75 but greater than 0.50 indicated adequate greenness, and 0.50 or lower indicated inadequate greenness. Fig. 4 summarizes the recorded total AGREE scale using the 12 GAC criteria. The suggested HPTLC method's overall AGREE scale was recorded to be 0.75. The AGREE results once again illustrated the excellent green characteristics of the current approach. The results of all greenness approaches demonstrated that the current method for measuring RISP in commercially available tablets had an excellent greener profile when compared to published HPTLC methods (El-Sherif et al., 2005; Maslanka et al., 2009; Devala et al., 2010; Patel et al., 2010; Singh et al., 2017).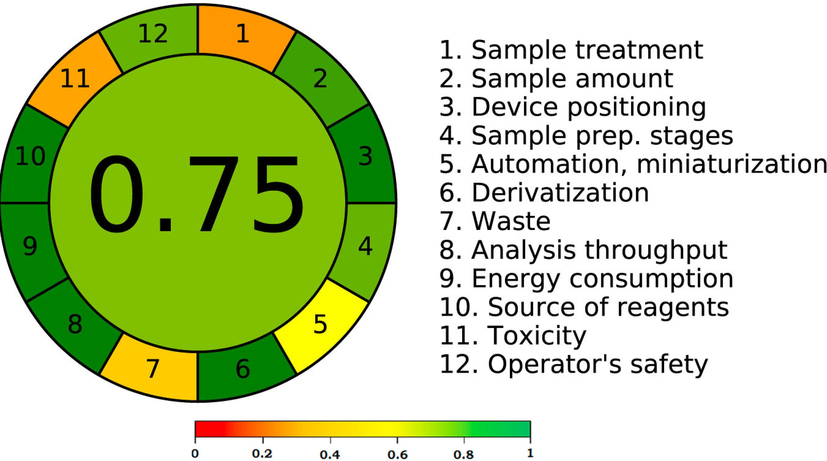
Analytical GREEnness (AGREE) scale for the present analytical method of RISP analysis.
4 Conclusions
A green normal-phase HPTLC technique for the quantitative analysis of RISP in commercially available products was developed and validated. It was discovered that the suggested RISP analytical technique for RISP estimation was simple, accurate, robust, sensitive, selective, stability-indicating, and environmentally friendly. In compared to previously published HPTLC methods for the determination of RISP, the findings of the AES and ChlorTox analysis demonstrated that the current method had outstanding greener features. RISP was shown to be exceedingly stable under conditions of acid, basic, and heat degradation; nevertheless, it was found to be extremely unstable under conditions of oxidative hydrolysis. The potential of the suggested approach to quantify RISP in the presence of its breakdown compounds suggested that the proposed methodology was selective and stability-indicating. Furthermore, it was efficiently utilized for the quantitative measurement of RISP in commercially available products. Overall, it has been discovered that the current method is safer, greener, more accurate, linear, and sensitive than previously published HPTLC methods. The suggested method could be used to analyze RISP quantitatively in various pharmaceutical dosage forms where RISP is one of the ingredients.
Author contribution statement
Prawez Alam: Conceptualization, Methodology, Investigation, Project administration, Supervision, Funding acquisition. Faiyaz Shakeel: Conceptualization, Investigation, Formal Analysis, Data curation, Software, Funding acquisition, Writing – original draft. Sultan Alshehri: Software, Validation, Visualization, Writing – review & editing. Ali Alhaiti: Validation, Formal analysis, Resources, Writing – review & editing. Mohammed H. Alqarni: Methodology, Investigation, Validation, Writing – review & editing. Ahmed I. Foudah: Methodology, Investigation, Writing – review & editing. Tariq M. Aljarba: Methodology, Formal analysis, Validation, Writing – review & editing.
Acknowledgments
“Authors are thankful to the Researchers Supporting Project number (RSPD2024R1040), King Saud University, Riyadh, Saudi Arabia for supporting this work”. “The authors also thank Prince Sattam bin Abdulaziz University for supporting this work via project number (PSAU/2024/R/1445)”. “Sultan Alshehri would like to express sincere gratitude to AlMaarefa University, Riyadh, Saudi Arabia for providing funding to conduct this research”.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Determination of the abused intravenously administered madness drops (tropicamide) by liquid chromatography in rat plasma; an application to pharmacokinetic study and greenness profile assessment. Microchem. J.. 2020;159:E105582
- [Google Scholar]
- Quantitative analysis of cabozantinib in pharmaceutical dosage forms using green RP-HPTLC and green NP-HPTLC methods: a comparative evaluation. Sus. Chem. Pharm.. 2021;21:E100413
- [Google Scholar]
- Simultaneous determination of caffeine and paracetamol in commercial formulations using greener normal-phase and reversed-phase HPTLC methods: a contrast of validation parameters. Molecules. 2022;27:E405.
- [Google Scholar]
- Guava-fruit based synthesis of carbon quantum dots for spectrofluorometric quantitative analysis of risperidone in spiked human plasma and pharmaceutical dosage forms. RSC Adv.. 2023;13:17765-17774.
- [Google Scholar]
- A fast DLLME-LC-MS/MS method for risperidone and its metabolite 9-hydroxyrisperidone determination in plasma samples for therapeutic drug monitoring of patients. Microchem. J.. 2020;156:E104894
- [Google Scholar]
- Determination of risperidone in plasma by high-performance liquid chromatography with electrochemical detection: application to therapeutic drug monitoring in schizophrenic patients. J. Pharm. Sci.. 1993;82:447-449.
- [Google Scholar]
- Sensitive method for the quantitative determination of risperidone in tablet dosage form by high-performance liquid chromatography using chlordiazepoxide as internal standard. Int. J. Biomed. Sci.. 2013;9:91-97.
- [Google Scholar]
- Neuropharmacological effect of risperidone: from chemistry to medicine. Chem. Biol. Interact.. 2023;369:E110296
- [Google Scholar]
- Review of risperidone for the treatment of pediatric and adolescent bipolar disorder and schizophrenia. Neuropsychiatr. Dis. Treat.. 2008;4:55-68.
- [Google Scholar]
- A simple and rapid LC-MS/MS method for the simultaneous determination of eight antipsychotics in human serum, and its application to therapeutic drug monitoring. J. Chromatogr. b.. 2020;1147:E122129.
- [Google Scholar]
- Bioassays studies of risperidone and its active metabolite in rat dried blood spots and dried plasma spots using LC-ESI-MS/MS: comparison of their pharmacokinetic profiles. Sep. Sci. plus. 2020;3:444-450.
- [Google Scholar]
- Green process chemistry in the pharmaceutical industry. Green Chem. Lett. Rev.. 2009;2:193-211.
- [Google Scholar]
- Stability-indicating HPLC determination of risperidone in bulk drug and pharmaceutical formulations. Int. J. Anal. Chem.. 2011;2011:E124917
- [Google Scholar]
- Development and validation of TLC-densitometry method for the estimation of antipsychotic drug in bulk and tablet formulation. Int. J. ChemTech. Res.. 2010;2:2063-2069.
- [Google Scholar]
- High performance liquid chromatographic and thin layer densitometric methods for the determination of risperidone in the presence of its degradation products in bulk powder and in tablets. J. Pharm. Biomed. Anal.. 2005;36:975-981.
- [Google Scholar]
- Greenness assessment of a stability indicating simple inexpensive high performance thin layer chromatography-dual wavelength method for simultaneous determination of mometasone furoate and salicylic acid in complex matrix using analytical eco scale. J. Planar Chromatogr.. 2021;34:455-466.
- [Google Scholar]
- Field-amplified sample injection in capillary zone electrophoresis for the pharmacokinetic research of trace risperidone and its major metabolite 9-hydroxyrisperidone in beagle dogs. J. Sep. Sci.. 2020;43:3555-3564.
- [Google Scholar]
- Dispersive solid-phase extraction of risperidone from plasma samples using graphene oxide aerogels and determination with liquid chromatography. J. Sep. Sci.. 2023;46:E2201028
- [Google Scholar]
- A rapid and sensitive stability-indicating green RP-HPTLC method for the quantitation of flibanserin compared to green NP-HPTLC method: Validation studies and greenness assessment. Microchem J.. 2021;164:E105960
- [Google Scholar]
- Photostability of risperidone in tablets. KONA Powder Particle. J.. 2018;35:209-215.
- [Google Scholar]
- HPLC-EAT (Environmental Assessment Tool): a tool for profiling safety, health and environmental impacts of liquid chromatography methods. Green Chem.. 2011;13:2021-2025.
- [Google Scholar]
- Analytical eco-scale for assessing the greenness of analytical procedures. Trends Anal. Chem.. 2012;37:61-72.
- [Google Scholar]
- The 12 principles of green analytical chemistry and the significance mnemonic of green analytical practices. Trends Anal. Chem.. 2013;50:78-84.
- [Google Scholar]
- Environmentally friendly stability-indicating HPLC method for the determination of isotretinoin in commercial products and solubility samples. Heliyon. 2023;9:E18405.
- [Google Scholar]
- Analytical method volume intensity (AMVI): a green chemistry metric for HPLC methodology in the pharmaceutical industry. Green Chem.. 2011;13:934-939.
- [Google Scholar]
- Development and validation of spectrophotometric method for the determination of risperidone in bulk drug and pharmaceutical formulation. Int. J. Pharm. Sci. Res.. 2011;2:378-382.
- [Google Scholar]
- Making the move towards modernized greener separations: introduction of the analytical method GREEnness score (AMGS) calculator. Green Chem.. 2019;21:1816-1826.
- [Google Scholar]
- Determination of risperidone in human plasma by HPLC-MS/MS and its application to a pharmacokinetic study in Chinese volunteers. J. Zhejiang Uni. Sci. b.. 2008;9:1142-2120.
- [Google Scholar]
- International conference on harmonization (ICH), Q2 (R1): validation of analytical procedures–text and methodology, Geneva, Switzerland, 2005.
- An introduction to the national environmental methods index. Env. Sci. Technol.. 2005;39:173A-A176.
- [Google Scholar]
- RP-HPLC method development and validation for estimation of risperidone in bulk & dosage forms. Int. J. Pharm. Sci. Rev. Res.. 2019;54:429-432.
- [Google Scholar]
- Simultnaeous determination of six antipsychotics, two of their metabolites and caffeine in human plasma by LC-MS/MS using a phospholipid-removal microelution-solid phase extraction method for sample preparation. Talanta. 2019;198:159-168.
- [Google Scholar]
- Intranasal nanoemulsion based brain targeting drug delivery system of risperidone. Int. J. Pharm.. 2008;358:285-291.
- [Google Scholar]
- Formulation and characterization of nanoemulsion-based drug delivery system of risperidone. Drug Dev. Ind. Pharm.. 2009;35:387-395.
- [Google Scholar]
- Development and validation of spectrophotometric method for the determination of risperidone in pure and pharmaceutical formulation using MBTH reagent. Res. J. Pharm. Technol.. 2013;6:388-391.
- [Google Scholar]
- Determination of risperidone and 9-hydroxyrisperidone in human serum by heart-cutting isocratic two-dimensional liquid chromatography. Anal. Lett.. 2020;53:2002-2019.
- [Google Scholar]
- Determination of impurities of risperidone API by ultea performance liquid chromatography (UPLC) Rasayan J. Chem.. 2020;13:940-948.
- [Google Scholar]
- A new strategy for development of eco-friendly RP-HPLC method using corona charged aerosol detector and its application for simultaneous analysis of risperidone and its related impurities. Microchem. J.. 2020;153:E104394
- [Google Scholar]
- Determination of risperidone in tablets in the presence of its degradation products and placebo-derived constituents. Acta Pol. Pharm.. 2009;66:461-470.
- [Google Scholar]
- Identification and determination of compounds belonging to the group of OUN pharmaceutical agents by thin-layer chromatography with densitometric detection in biological material. Acta Chromatogr.. 2011;23:247-266.
- [Google Scholar]
- Risperidone for the core symptom domains of autism: results from the study by the autism network of the research units on pediatric psychopharmacology. Am. J. Psychiatr.. 2005;162:1142-1148.
- [Google Scholar]
- Stability-indicating HPLC method for quantification of risperidone in tablets. J. Chil. Chem. Soc.. 2018;63:4150-4154.
- [Google Scholar]
- Simplified liquid-liquid extraction method coupled with LC/DAD by quantitative determination of risperidone in human serum. J. Chil. Chem. Soc.. 2019;64:4265-4267.
- [Google Scholar]
- Determination of risperidone and 9-hydroxyrisperidone in human plasma by high-performance liquid chromatography: application to therapeutic drug monitoring in Japanese patients with schizophrenia. J. Pharm. Biomed. Anal.. 1999;19:595-601.
- [Google Scholar]
- Development and validation of a capillary zone electrophoresis method for the quantitative determination of atypical antipsychotic risperidone in pharmaceutical dosage forms. Curr. Anal. Chem.. 2014;10:205-215.
- [Google Scholar]
- Development and validation of rapid UHPLC method for determination of risperidone and its impurities in bulk powder and tablets. Int. J. Res. Pharm. Chem.. 2014;4:261-266.
- [Google Scholar]
- What color is your method? adaptation of the RGB additive color model to analytical method evaluation. Anal. Chem.. 2019;91:10343-10352.
- [Google Scholar]
- How to evaluate methods used in chemical laboratories in terms of the total chemical risk? a ChlorTox Scale. Green Anal. Chem.. 2023;5:E100056
- [Google Scholar]
- Development and validation of a reverse phase by high performance liquid chromatographic method, using standard addition calibration, for determination of risperidone in human plasma. J. Pharm. Bioresours.. 2022;19:9-15.
- [Google Scholar]
- TLC determination of amitriptyline HCl, trifluoperazine HCl, risperidone and alprazolam in pharmaceutical products. Chromatographia. 2009;69:393-396.
- [Google Scholar]
- HPTLC method development and validation for analysis of risperidone in formulations, and in-vitro release study. Acta Chromatogr.. 2010;22:549-567.
- [Google Scholar]
- Stability-indicating HPTLC method for simultaneous determination of risperidone and trihexyphenidyl hydrochloride from their combination drug products. Afr. J. Pharm. Biol. Med. Sci.. 2015;2:120-131.
- [Google Scholar]
- Application of the “Method Operable Design Region” (MODR) approach for the development of a UHPLC method for the assay and purity determination of risperidone using drug substance and other formulations. Biomed. Chromatogr.. 2022;36:E5433
- [Google Scholar]
- AGREE-Analytical GREEnness metric approach and software. Anal. Chem.. 2020;92:10076-10082.
- [Google Scholar]
- Analysis of selected anti-depressive drugs by high performance thin-layer chromatography. J. Liq. Chromatogr. Relat. Technol.. 2008;31:1913-1924.
- [Google Scholar]
- A new tool for the evaluation of the analytical procedure: green analytical procedure index. Talanta. 2018;181:204-209.
- [Google Scholar]
- CHEM21 selection guide of classical- and less-classical solvents. Green Chem.. 2016;18:288-296.
- [Google Scholar]
- An ecofriendly multi-analyte high performance thin layer chromatographic densitometric determination of amoxicillin, metronidazole, and famotidine in their ternary mixtures and simulated gastric juice: a promising protocol for eradicating Helicobacter pylori. J. Sep. Sci.. 2023;46:E2200951
- [Google Scholar]
- Development of a novel method for determination of risperidone in pharmaceutical products and its quality control application. Acta Sci. Pharm. Sci.. 2021;5:101-109.
- [Google Scholar]
- Validated method for the determination of risperidone and 9-hydroxyrisperidone in human plasma by liquid chromatography-tandem mass spectrometry. J. Chromatogr. b.. 2003;783:461-472.
- [Google Scholar]
- Quantitative determination of risperidone, paliperidone and olanzapine in human serum liquid chromatography-tandem mass spectrometry coupled with on-line solid-phase extraction. Biomed. Chromatogr.. 2018;32:E4209
- [Google Scholar]
- Systematic development of a bioanalytical UPLC-MS/MS method for estimation of risperidone and its active metabolite in long-acting microsphere formulation in rat plasma. J. Chromatogr. b.. 2020;1160:E122433
- [Google Scholar]
- Simultaneous determination of paroxetine, risperidone and 9-hydroxyrisperidone in human plasma by high-performance liquid chromatography with coulometric detection. Pharmacology. 2000;60:51-56.
- [Google Scholar]
- Solubility of antipsychotic drug risperidone in Transcutol + water co-solvent mixtures at 298.15 to 333.15 K. J. Mol. Liq.. 2014;191:68-72.
- [Google Scholar]
- A greener RP-HPTLC-densitometry method for the quantification of apremilast in nanoformulations and commercial tablets: Greenness assessment by analytical eco-scale, ChlorTox, and AGREE methods. Arabian J. Chem.. 2024;17:E105571.
- [Google Scholar]
- Preparation and in-vitro characterization of Risperidone-cyclodextrin inclusion complexes as a potential injectable product. DARU J. Pharm. Sci.. 2009;17:226-235.
- [Google Scholar]
- Method development and validation of stability indicating assay for risperidone in solid dosage form by using HPTLC. Asian J. Chem.. 2017;29:99-103.
- [Google Scholar]
- A stability-indicating HPLC method for the determination of electrochemically controlled release of risperidone. J. Chromatogr. Sci.. 2011;49:780-785.
- [Google Scholar]
- Sensitive liquid chromatography/tandem mass spectrometry method for the simultaneous determination of risperidone, olanzapine, quetiapine, clozapine, ziprasidone, perospirone, aripiprazole and blonanserin in human serum. Am. J. Anal. Chem.. 2018;9:88-97.
- [Google Scholar]
- Determination of risperidone at pictogram level in human urine by luminol-H2O2 chemiluminescence. Chem. Pap.. 2006;60:288-292.
- [Google Scholar]
- Validated HPTLC method for simultaneous determination of loratadine and desloratadine in presence of co formulated drug. J. Planer Chromatogr.. 2012;25:456-462.
- [Google Scholar]
- Development and validation of a high-performance thin-layer chromatographic method for the assay of ternary mixtures containing cetirizine dihydrochloride in pharmaceutical dosage forms. J. Planer Chromatogr.. 2014;27:58-65.
- [Google Scholar]
- Electromembrane extraction of chlorprothixene, haloperidol and risperidone from whole blood and urine. J. Chromatogr. a.. 2020;1629:E461480
- [Google Scholar]
- Condition optimization for the analysis of risperidone and 9-OH-risperiidone by high-performance liquid chromatography. Med. Lab. Technol. J.. 2022;8:12-19.
- [Google Scholar]
Appendix A
Supplementary data
Supplementary data to this article can be found online at https://doi.org/10.1016/j.arabjc.2024.105693.
Appendix A
Supplementary data
The following are the Supplementary data to this article:Supplementary data 1
Supplementary data 1







