Translate this page into:
Assessment of antioxidant and cytotoxic potential of silver nanoparticles synthesized from root extract of Reynoutria japonica Houtt
⁎Corresponding author. fazlikhuda@uop.edu.pk (Fazli Khuda)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
Free radicals, mostly consist of reactive oxygen species, are generated in human body by several exogenous and endogenous systems. Overproduction of free radicals is known to cause several degenerative disorders including cancer. The aim of this study is to synthesize silver nanoparticles (AgNPs) using root extract of Reynoutria japonica and to investigate its antioxidant and cytotoxic potential. AgNPs were synthesized by green approach and subsequently characterized using UV–vis spectroscopy, SEM, TEM, FTIR, XRD, EDS and DLS. The antioxidant activity was investigated using DPPH, FRAP, H2O2, and ABTṠ+ radical scavenging assays while the cytotoxic effect was assessed using different human cancer cell lines including lung (A549), liver (Hep-G2) and breast (MDA-MB-231) by MTS assay. Moreover, the specificity of NPs was assessed against two normal human cell lines e.g. alveolar and renal primary epithelial cells (HPAEpiC and HRPTEpiC). The UV–vis spectra confirmed the synthesis of AgNPs by producing a characteristic peak at 410 nm. Further analysis confirmed that AgNPs were crystalline in nature, predominantly spherical in shape, with an average width and area of 17.34 nm and 164.46 nm2, respectively. DLS analysis revealed that NPs possess a high negative zeta potential value (−28.5 mV), thus facilitating its electrostatic stabilization. AgNPs showed dose dependent antioxidant activity against DPPH, FRAP, H2O2 and ABTS with IC50 values 19.25, 22.45, 24.20 and 18.88 µg/ml, respectively. The AgNPs depicted significant cytotoxic effects against A549, Hep-G2 and MDA-MB-231 cell lines with IC50 values of 4.5, 5.1 and 3.46 µg/ml, respectively. Moreover, the NPs exhibited highest selectivity index (>2.0) for A549, Hep-G2 and MDA-MB-231, confirming its specificity towards cancer cell lines. In conclusion, AgNPs prepared from root extract of R. japonica possess strong antioxidant and cytotoxic potential which suggests that they should be investigated further in order to develop safe and effective antioxidant and/or cytotoxic formulations.
Keywords
Reynoutria japonica
Silver nanoparticles
Characterization
Antioxidant
Cytotoxic
- WHO
-
World Health Organization
- AgNPs
-
Silver nanoparticles
- NPs
-
Nanoparticles
- R. japonica
-
Reynoutria japonica
- mM
-
Millimolars
Abbreviations
1 Introduction
Cancer remains one of the most active area of research, being a major public health problem worldwide, with increasing mortality and cost (Ziyu et al., 2021; Hanène et al., 2020). According to the World Health Organization (WHO), approximately 21 million new cancer cases and 13 million cancer-related deaths are expected across the world by 2030 (García-Castillo et al., 2016). Breast cancer is one the most common cancer in females, accounting for about 11.6 % of all cancer-related deaths (Qiubing et al., 2022). In 2018, about 2.1 million new cases of breast cancer were reported, with an estimated 627,000 deaths (Bray et al., 2018; Fitzmaurice et al., 2017). Pakistan has the highest incident of this cancer in Asia, which is likely to increase in coming years. It has been reported that one in nine women are diagnosed with this cancer during their lifetime (Sohail et al., 2007). In Pakistan, 59 % of the patients are diagnosed at an advanced stage while 89 % at later stage due to several reasons such as socio-economic and cultural factors, fear of surgery, lack of awareness and belief in traditional treatments. Due to these reasons, the mortality rate in breast cancer patients is very high (Gulzar et al., 2019). Similarly, hepatocellular carcinoma is one of the fatal tumors in the world with the second highest mortality among all cancers (Huajun et al., 2021). It is the sixth most common cancer worldwide, with 5.7 % new cases annually with a survival rate of only 3–5 % (Parkin, 2006). According to literature review, about 82 % of cases occurred in developing countries including 55 % from China only (Raza et al., 2007). In Pakistan, its prevalence varies from 3.7 to 16 % where the most common causes are viral hepatitis C or B (68 and 22 %, respectively) (Munaf et al., 2014). Despite some global decline, lung cancer remains a major challenge for health policies, with over 470 thousand cases reported in Europe each year. This figure accounts for approximately 12 % of all tumors, 65 % of which occurred in males (Mireia et al., 2021). Similarly, in females it is the second most common cancer after breast cancer globally (Sarwar et al., 2017). According to the American cancer society, it was the leading cause of death in the United States in 2017. This cancer causes more deaths compared to breast, colon and prostate cancer combined (Saad et al., 2010).
Despite the therapeutic advances in mitigating cancer, the overall mortality rate is still very significant. Resistance to anticancer agents remains one of the hallmark of cancer therapy (Muhammad et al., 2021). The generation of free radicals in our body is a major reason for the development of cancer. It has been reported that under stress condition our body produces more reactive oxygen species than enzymatic antioxidants. These includes hydroxyl (·OH), superoxide anion (O2·–), perhydroxyl (HO2·) and some non-free radicals i.e·H2O2 (Duduku et al., 2011). In the absence of effective antioxidants, they facilitate the development of many diseases such as cancer, inflammation, cardiac and a variety of neurodegenerative disorders (Nisa et al., 2013). Synthetic antioxidants (butylated hydroxytoluene and hydroxyanisole) are commonly used in medicines and food industry however, they are associated with several side effects such as carcinogenesis and liver cirrhosis (Sajjad et al., 2015). Therefore, in recent years the use of natural antioxidants have got much attention. Traditional medicines are widely used since long time to treat several health problems however; large doses are frequently required to get the therapeutic outcomes.
Nanotechnology is considered an advanced field of science that encompasses the utilization of nanoparticles (NPs) for various purposes. A variety of NPs made up of different noble heavy metals such as platinum, gold, palladium and silver (Ag) are reported in literature. However, among these AgNPs are of significant importance because of their unique structure and functions. Unlike copper and gold, silver is a naturally occurring abundant metal possessing numerous biological, physical and chemical properties including electrical conductivity, surface enhanced Raman scattering and catalytic effects. The unique biochemical properties makes it the most appropriate candidate with diverse biomedical potential i.e. as antimicrobial, antiseptic, larvicidal, significance in drug delivery, cosmetic products, optoelectronics, biomedicines and food preservation. Antimicrobials containing silver ions showed more efficacy against different microbes compared to other NPs. It has been documented that silver ions react with thiol group of proteins and leads to DNA disruption. Similarly, this metal impair DNA replication due to uncoupling of electron transport from oxidative phosphorylation; inhibits enzyme and disrupts the membrane permeability. In addition, AgNPs have shown comparatively high cytotoxic potential against a variety of cell lines as compared to other NPs (Qindeel et al., 2021; Lateef et al., 2019; Verma et al., 2019; Jabeen et al., 2021).
These NPs are prepared using different techniques including chemical, physical and biological. The chemical method is less time consuming and require least labour however; it utilizes various hazardous chemicals for reduction and stabilization of synthesized NPs (Ponsanti et al., 2020). In contrast, green synthesis is an eco-friendly technique, using biological materials such as plants, algae, bacteria, and fungi as reducing and stabilizing agents. Several microalgal species have been used for the synthesis of iron, silver, palladium and gold NPs due to their high rate of intracellular metal accumulation. Extracellular synthesis of NPs is highly advantageous due to their simplicity in the purification process. Several biological applications of microorganism mediated NPs have been reported in literature (Adelere and Lateef, 2021). Similarly, enzyme-mediated synthesis of NPs is one of the recent advancements in the field of nanotechnology. Several enzymes including sulfite reductase, cellulose, laccase, ligninase and nitrate reductase have been used for the synthesis of metal NPs. These enzymes acts a natural reducing and capping agents in the formation of NPs. Enzyme based NPs have been reported for different biological activities (Adelere and Lateef, 2016). According to literature review, plant mediated synthesis of nanoparticles has been shown to produce particles with comparable shape and size to those produced through physical and chemical procedures. In addition, plants contain several phytochemicals such as phenols and flavonoids that play a vital role in reducing Ag+ (Lim et al., 2020). Several plants including Acacia nilotica, Conocarpus Lancifolius, Datura inoxia Mill, Persea Americana, Hyptis suaveolens, Annona muricata, Opuntia ficus-indica and Sambucus wightiana have been reported to fabricate AgNPs with improved biological activities (Saratale et al., 2019; Oves et al., 2022; Bagewadi et al., 2019; Adebayo et al., 2019; Lateef et al., 2020; Badmus et al., 2020; Adebayo et al., 2019; Fazli et al., 2021). Beside plants and microorganisms, animal waste materials have been used for the synthesis of AgNPs. These NPs have shown considerable potentials as antioxidant, anticoagulant and thrombolytic agents (Akintayo et al., 2020). Considering the importance of using plant extracts for green synthesis of NPs, the present study is therefore designed to prepare AgNPs, using the crude extract of Reynoutria japonica.
Reynoutria japonica Houtt. (Polygonaceae) known as Japanese knotweed is a herbaceous perennial plant, widely distributed in Asia, Japan, China and Korea (Anna et al., 2019). The first mention of R. japonica for the treatment of abdominal masses and gallstones appeared in a Chinese medicine monograph, Mingyi Bielu, which was written during China's Han dynasty (B.C. 202–A.D. 200) (T'ao, 1986). Its clinical applications were recorded in Bencao Gangmu, another Chinese herbology book that was first published in Korea in 1613. Literature review showed that this plant has been used for the treatment of various disorders including hepatitis, cancer, skin burns and inflammation (Vastano et al., 2000). In addition, it has shown antimicrobial and antioxidant activities (Chan et al., 2008; Feng et al., 2006; Kim et al., 2008). Phytochemical analysis of the plant showed the presence of physiologically active substances, such as Physcion, polydatin, Quercetin, resveratrol, citreorosein, (+)-Catechin and emodin (Chang et al., 2012; Yi et al., 2007). Resveratrol from this plant has been reported to prevent lung cancer in mice. It has also shown anticancer activity in mice bearing neuroblastoma, melanoma and ovarian cancer (Kimura et al., 2001; Guo et al., 2010). Because of its diverse biological activities, biosynthesis of silver nanoparticles from R. japonica extract will exhibit greater therapeutic benefits.
2 Material and methods
2.1 Chemicals
Chemicals and reagents used in present experiment include: silver nitrate (99 %), FRAP (99 %), DPPH (99 %), H2O2, potassium persulfate and ABTS, 1 % penicillin–streptomycin solution, Dulbecco’s Modified Eagle’s Medium supplemented with 10 % fetal bovine serum (FBS), Methanol (99 %), NaOH (98 %), 3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide (MTT) and acetate buffer. All the reagents and chemicals were purchased from Sigma-Aldrich, Germany.
2.2 Plant material
The plant was purchased from Kyung-Dong Oriental Market, Seoul, Korea and was authenticated by Dr. Mi-Jeong Ahn, Professor of Pharmacy, at College of Pharmacy, Gyeongsang National University, Korea (Specimen no. GSC-104).
2.2.1 Preparation of extract
The coarse powder (100 g) was soaked in deionized water (1 L) and subsequently heated at 45 to 55 °C for about 40 min. Following cooling, the extract was filtered through a nylon cloth and the filtrate was centrifuged (7000 rpm) for 10 min to remove any dissolved impurities. The supernatant was collected and stored at 4 °C until further use.
2.3 Nanoparticles synthesis and purification
Fixed volume of plant extract (5 ml v/v) was added to different ratios (3, 5, 10, 15, 20, 50 and100 ml v/v) of silver nitrate aqueous solution (1 mM) followed by stirring (300 rpm; 25 °C) for about 60 min. The change in color from light green to yellowish-brown indicates the formation of AgNPs, which was further confirmed by UV visible spectroscopy (350 and 800 nm). For optimum synthesis of NPs, the mixture was incubated at 37 °C for 24 hrs. The resulting NPs were subjected to centrifugation at 10,000 rpm for about 5 min. The obtained pellets were washed, three time with distilled water for removal of any free ions and unreacted moieties. Finally, the pure colloidal NPs were freeze dried and stored at 25 °C for further characterization and biological activities (Ilahi et al., 2021).
2.4 Physical characterization
The following techniques were used for the analysis of AgNPs.
2.4.1 UV–vis spectrophotometry
Initially, the synthesis of AgNPs was confirmed by UV–vis spectrophotometry (Perkin-Elmer, Lambda 35, Germany). The reduction of silver ions (Ag+) in the reaction mixture was periodically monitored by taking samples at different time interval and were scanned in 350–800 nm range.
2.4.2 SEM and TEM analysis
The surface morphologies and crystalline nature of AgNPs were confirmed using SEM (S-2500, Japan) and TEM techniques, respectively. For TEM analysis, the colloidal particles were dispersed in distilled water and a drop of this dispersion was put on a staining mat. A “carbon coated Copper-grid” was inserted into the drop (coated side upward), and after 10 min. it was removed and dried in air and finally screened using Hitachi-7650 (Japan) transmission electron microscope.
2.4.3 FTIR analysis
The possible role of different functional groups involved in the green synthesis and stabilization of AgNPs were analyzed using FTIR (Version 10.5.1, Perkin-Elmer). The spectra was recorded in 400–4000 cm−1 range. Vibrations at different modes were identified and assigned to determine functional groups present in the extract and NPs. Finally, the spectra’s were compared.
2.4.4 XRD analysis
The crystalline nature of NPs was investigated using XRD (JDX-3532 JEOL, Japan) operated at a specific voltage and current (40 kV and 30 mA, respectively) with Cu Kα radiation in θ-2θ configurations. The size of crystal was calculated from the width of XRD peaks using Debye-Scherrer’s equation.
Where D: mean crystal size; λ: X-ray wavelength; β: full-width at half maximum (FWHM); θ: diffraction angle.
2.4.5 EDX analysis
The elemental composition such as the presence of silver, carbon, oxygen and other such elements in the synthesized NPs was investigated using EDX (NOVA-450 instrument). EDX analysis is used to confirm the purity of NPs.
2.4.6 Dynamic light scattering (DLS)
The average particle size, polydispersibility index (PDI) and zeta potential of AgNPs were analysed using a Zetasizer (Malvern, UK). The sample was prepared by dispersing the NPs in distilled water filtered through 0.45 μm filter paper. Particle size and PDI were measured at room temperature with a scattering angle of 90. For zeta potential measurement, the sample was placed in a disposable zeta cell at room temperature and was measured by PALS technology. The analysis was performed in triplicate.
2.5 Stability and optimization of AgNPs synthesis
The effect of substrate concentration, pH and temperature on the biosynthesis of AgNPs were optimized. To optimize substrate concentration, fixed volume of crude extract was added to different ratios of silver nitrate solution (1:3, 1:5, 1:9, and 1:13 v/v) and maximum absorbance was determined using UV–Vis spectroscopy. Temperature optimization was conducted by mixing crude extract with silver nitrate at varying temperature (25, 40, 60 and 80 °C) and the corresponding spectra’s were obtained. The pH was optimized by dispersing AgNPs in phosphate buffer at different pH (4,6,8 and 12). Moreover, the short-term stability was assessed after 30 min, 48 and 96 hr following synthesis of AgNPs and the SPR peaks were anaysed for maximum absorbance.
2.6 Antioxidant assay
2.6.1 DPPḢ assay
The antioxidant potential of AgNPs from R. japonica extract was evaluated using a reported method (Sagar and Sing, 2011). Different concentrations (62.5, 125, 250, 500 and 1000 μg/ml) of AgNPs and ascorbic acid were prepared in methanol. Similarly, DPPH solution (0.3 mM) was also prepared in the same solvent. Thereafter, from each working solution 100 µl was mixed with 1 ml of the reagent solution in separate test tubes and vortexed thoroughly. The reaction mixture was incubated in dark place for 15 min. For control, 2 ml of methanol was added instead of AgNPs and simultaneously run with the test samples. Finally, the maximum absorbance was measured at 517 nm and the percentage radical scavenging activity of AgNPs and extract were calculated using the formula.
Where SAbs and CAbs represents the absorbance of standard/test samples and control respectively.
2.6.2 FRAP assay
The assay was conducted following the method of Iris and Strain (Iris and Strain, 1999). This assay measures the antioxidant potential in test samples via reduction of ferric ion (Fe3+) to ferrous ion (Fe2+); the later forms a complex with 2,4,6-tripyndyl-s-triazine (TPTZ), that can be quantified at 593 nm using colorimetric techniques. In brief, acetate buffer (300 mM; pH 3.5) was freshly prepared. Then a mixture of 10 mM TPTZ (0.25 ml) in 40 mM HCL, 20 mM FeCl3 (0.25 ml) and acetate buffer in the ratio of (1:1:10 v/v) was prepared for FRAP reagent. The assay was conducted using a 96-well plate. The FRAP reagent (170 µl) and sample solutions (20 µl) was added to each well, mixed in dark at ambient temperature and allowed to incubate for 30 min. Finally, the maximum absorbance was measured at 593 nm using a microplate reader.
2.6.3 Hydrogen peroxide scavenging assay
Different concentrations of tested samples and H2O2 (2 mM) was mixed (1:1 v/v) and incubated for 20 min at ambient temperature. Following this, the sample solutions and H2O2 was mixed with methanol (10 µl) and FOX reagent (0.9 ml). This mixture was thoroughly mixed using a vertex mixer and subsequently incubated at 25 °C. The maximum absorbance of the formed complex was measured at 560 nm. The percent H2O2 scavenging activity was then determined and compared to sodium pyruvate (standard) (Chandan et al., 2014).
2.6.4 ABTS scavenging assay
The ABTS·+ radicals was produced by the reaction between potassium persulfate (2.5 mM) and ABTS (7 mM) in 1:1 (v/v) ratio and subsequently stored away from light at 25 °C for 10–15 hr before use. The reagent solution was diluted with methyl alcohol until an absorbance of 0.700 was achieved at 734 nm. Silver NPs and/or extract (5 µl) was mixed with ABTS solution (4 ml) and after 30 min, the absorbance was measured. All the anti-oxidant assays were performed in triplicate (Chandan et al., 2014). Finally, the percent radical scavenging activity was determined.
2.7 Cytotoxicity assay
The MTS assay was used to investigate the cytotoxic potential of R. japonica mediated AgNPs against various cell lines including hepatocellular carcinoma (Hep-G2), lung adenocarcinoma (A549) and breast adenocarcinoma cells (MDA-MB-231) as well as two normal cell lines i.e. renal and alveolar primary epithelial cells (HPAEpiC and HRPTEpiC) (Fazli et al., 2022). In this assay the soluble MTT salt, 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide is reduced to an insoluble colored formazan product which is subsequently measured using a spectrophometer. The level of MTT salt indicates the normal function of mitochondrial dehydrogenase enzymes which in turn represents the number of viable cells.
Briefly, the cells treated with AgNPs and plant extract were incubated (37 °C; 48 h; 5 % CO2). The MTS reagent (25 µl) in phosphate buffer solution was added to each well and incubated for 30 min at 37 °C The insoluble formazan crystals were collected and solubilized in DMSO (100 μl) and the absorbance was measured at 490 nm using a microplate reader. The cells treated with sample were compared with controls. The MTT salt was reduced to colored formazan by cellular enzymes in viable cells only. Therefore, the amount of formed formazan dye directly correlates with the number of viable cells in culture media which is measured as function of total absorbance. The % cell viability was calculated using the following equation.
To find out the specificity of the NPs for cancer cell lines, their selectivity index was determined. It was calculated by dividing the IC50 values of the normal cells with the IC50 values for cancer cell lines. A value of two or more indicated high specificity.
2.8 Statistical analysis
The analytical determinations were performed in triplicate. One-way ANOVA (GraphPad Software Inc., San Diego, California, USA) was used for data analysis. The data were presented as mean ± SEM (n = 5).
3 Results and discussion
3.1 Synthesis of AgNPs
The addition of 1 mM silver nitrate solution changed the color of the extract from light green to yellowish-brown. The rapid synthesis of NPs shows the presence of high levels of phenolics and flavonoids which are predominately involved in the reduction of Ag to AgNPs. This method is comparatively safe, rapid and do not involve the use of hazardous chemical substances.
3.2 Characterization of AgNPs
3.2.1 Physical characterization
Initially, AgNPs were characterized with color change resulted from the reduction of Ag+ by the green extract. As the reduction of Ag+ increased, an intense color was produced that become stable upon the completion of reduction process. In present study, the color of NPs solution changed from light green to yellowish-brown. This color change was observed within 30 min to 1 hr. No such change in color was observed up to 24 hrs following incubation. The short duration for completion of biochemical reduction indicates the presence of high levels of phenolics and flavonoids that could be involved in the reduction of Ag to AgNPs (Yugal et al., 2017).
3.2.2 UV–vis spectrophotometry
This reduction was further investigated using UV–vis spectroscopy which is a valuable technique used to detect the characteristics surface plasmon resonance (SPR) pattern of the metallic NPs. Fig. 1A depicts the UV–visible spectrum for AgNPs at 410 nm, a characteristic band for Ag. The absence of any other peaks at this range confirms the synthesis of AgNPs. The observed value are well within the range reported for AgNPs (Yugal et al., 2017).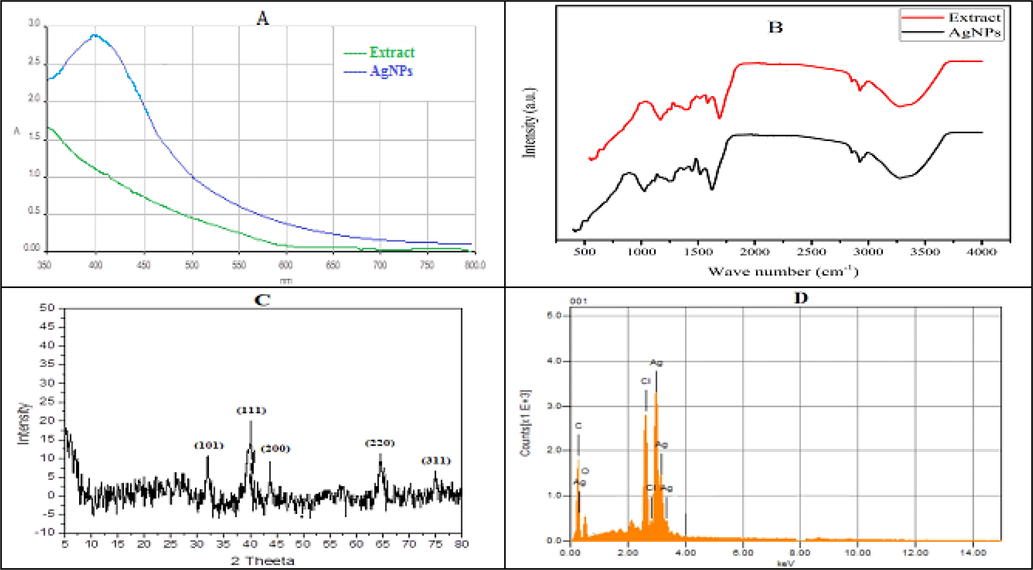
(A) UV–visible spectra (B) FT-IR spectra (C) XRD pattern and (D) EDX spectra of AgNPs.
3.2.3 SEM and TEM analysis
SEM and TEM analysis were conducted to investigate the surface and inner morphological features of AgNPs. SEM results confirmed the formation of heterogeneously dispersed AgNPs (Fig. 2). TEM analysis revealed different shapes of AgNPs such as cubic, hexagonal, trigonal and polygonal with an average area of 164.46 nm2 and width of 17.34 nm (Fig. 3). The formation of poly-dispersed particles may be due to the early and later stages of nucleation.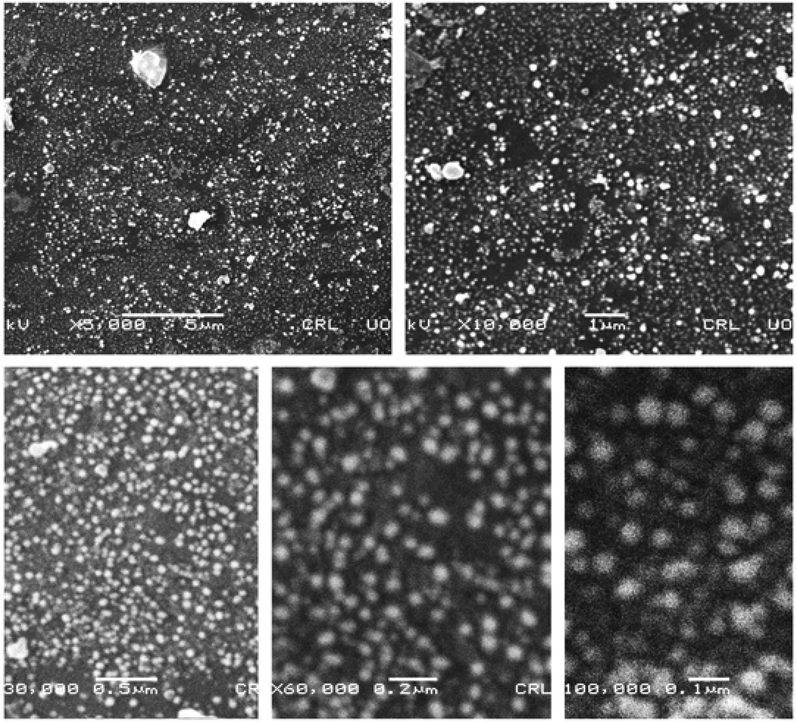
SEM images of silver nanoparticles at various resolutions.

TEM images of silver nanoparticles (A: 50 nm; B: 10 nm; C: 20 nm; D: 5 nm).
3.2.4 FTIR analysis
FTIR spectra was used to identify different functional groups involved in the synthesis of AgNPs (Fig. 1B). The spectra of the extract and AgNPs showed different stretching vibration pattern of the functional groups. The broader band at 3315 cm−1 are due to the stretching vibration of OH group while the bands at 2933, 1618, 1376 and 1022 cm−1 are assigned to C—H, C⚌O, N—O and C—N groups, respectively. According to literature review, functional groups responsible for the reduction and stabilization of synthesized AgNPs includes but not limited to phenolic, alcoholic and carboxylic acid.
3.2.5 XRD analysis
The XRD analysis showed diffraction peaks at 2θ values of 32.5, 38.2, 44.5, 64.6, and 77.3°which were assigned to the planes (1 1 1), (2 0 0), (2 2 0), and (3 1 1) facet of Ag crystal, respectively. The diffractogram (Fig. 1C) was compared to JCPDS card: silver file number 04–0783. The average size of AgNPs was calculated using Scherrer’s equation and was found to be 20.5 nm.
3.2.6 EDX analysis
The EDX analysis was conducted to confirm the presence of elemental silver. Fig. 1D carries a prominent Ag-peak that shows the presence of silver in the sample. The spectrum also showed peaks for other elements such as O and C that may be due to capped biomolecules on the surface of silver NPs. The spectrum did not showed any other peak, which confirm the high purity of synthesized NPs.
3.2.7 Dynamic light scattering
The size and zeta potential of the AgNPs was determined using Zetasizer. By studying the DLS, the average size of AgNPs reveals a narrow size distribution (approx. 35–100 nm) (Fig. 4). These results are in concurrence with those of the TEM images. Similarly, the analysis showed the presence of AgNPs with low PdI (0.185). Moreover, the apparent zeta potential recorded was − 28.5 mV. The negative zeta potential of AgNPs may be due to the capping of particles by the –OH groups of different biomolecules. According to literature review, particles with zeta potential above + 20 mV or below − 20 mV are considered stable. The obtained value of zeta potential showed that AgNPs remained highly stable and dispersed even after 96 hrs of its synthesis. This may also be the reason for producing particles with a very narrow size distribution index.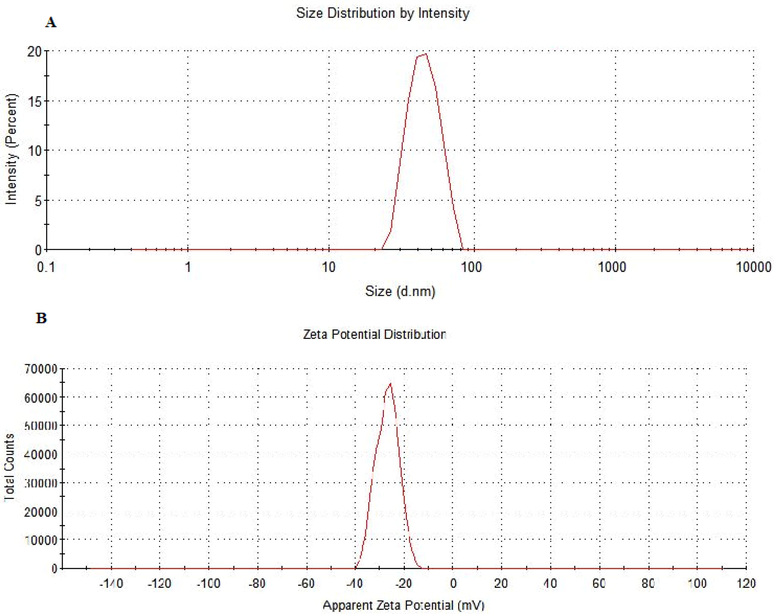
DLS spectra of (A) hydrodynamic size distribution and (B) Zeta potential (mV) of synthesized silver nanoparticles.
3.3 Optimization of AgNPs
Fixed volume of crude extract was mixed with different ratios of silver nitrate solution (1:3, 1:5, 1:9, and 1:13 v/v). Optimum synthesis occurred at 1:5 v/v (Fig. 5A). The synthesis of AgNPs was also investigated at different pH (Fig. 5B). The synthesized NPs showed maximum absorption at acidic pH while at basic pH the SPR peaks gradually diminished. The stability of AgNPs was also studied at different temperature ranging from 25, 40, 60 and 80 °C Maximum absorbance was observed at 80 °C (Fig. 5C). Similarly, the short-term stability was assessed after 30 min, 48 and 96 hrs by storing the AgNPs in a dark place. An increase in maximum absorbance was observed with the passage of time, which was also noticed with naked eye as darkening of the solution. Maximum absorbance was observed after 96 hrs of synthesis (Fig. 5D).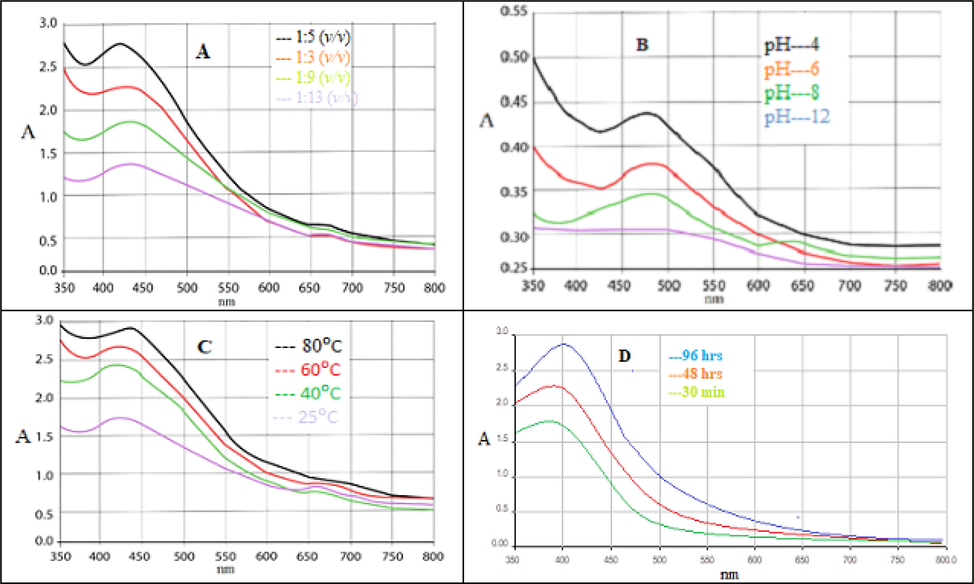
(A) UV–Vis absorption spectra of silver nanoparticles at different concentration of silver salt; (B) pH; (C) temperature and (D) at different time interval.
3.4 Antioxidant activity
Antioxidants play a significant rule in the prevention of various human diseases. Herbal remedies with antioxidant potential may serve as hydrogen donors, free radical scavengers and single oxygen quenchers, thereby protecting the body from a variety of degenerative diseases including cancer. These damaging reactive species are generated during cellular metabolism that are responsible for initiating a state of oxidative stress. Subsequently, it leads to the pathogenesis of several diseases by damaging DNA, lipids and proteins. Polyphenols are the most abundant compounds present in plants with high potential for antioxidant activity. The most common phenolic compounds are flavonoids, tannins, phenolic acids and lignins. These compounds possess significant redox properties; allow them to act as potential reducing agents (Ankita et al., 2015). According to literature review, plant mediated AgNPs have shown significant antioxidant activities against DPPH, FRAP, H2O2 and ABTS scavenging assays (Adebayo et al., 2019); Badmus et al., 2020).
3.4.1 DPPH assay
In present study, root extract of R. japonica was investigated for their antioxidant potential using different in vitro models. The IC50 values and % free radical scavenging activity of the extract and NPs are summarized in Table 1.. Silver NPs caused 10.45, 21.58, 30.65, 51.75 and 67.50 % inhibition at 62.5, 125, 250, 500 and 1000 µg/ml concentrations, respectively. Crude extract showed much lower activity at the same concentrations as evident from the respective IC50 values. The IC50 values for crude extract, NPs and standard were 27.80, 19.25 and 16.98 µg/ml, respectively. It has been documented that DPPH free radical scavenging effect occur due to the neutralization of these radicals by different phytochemicals mainly by the transfer of hydrogen and/or an electron. The results showed that silver NPs exhibited significant antioxidant activity compared to crude extract in a dose dependent manner therefore, it may be considered as an effective and safe alternative herbal therapy for the mitigation of a variety of degenerative disorders. Adebayo et al. (Adebayo et al, 2109) reported DPPH free radical scavenging activities of Persea americana fruit feel extract mediated AgNPs as 57.82–63.25 %. The activity may be assigned to the presence of biomolecules adhere at the surface of AgNPs.
Concentration (µg/ ml)
DPPH Scavenging activity (%)
Extract
AgNPs
Ascorbic acid
62.5
–
10.45 ± 0.32
18.75 ±
125
9.25 ± 0.85
21.58 ± 0.98
28.80 ± 1.25
250
24.76 ± 1.55
39.65 ± 2.34
46.75 ± 2.80
500
36.42 ± 2.11
51.75 ± 3.10
61.88 ± 4.45
1000
48.90 ± 4.45
67.50 ± 5.59
82.65 ± 6.70
IC50
27.80
19.25
16.98
3.4.2 FRAP assay
The results of this assay are summarized in Table 2. Crude extract, silver NPs and standard revealed 42, 56 and 78 % inhibition at 1000 µg/ml, respectively. The respective IC50 values were 29.45, 22.45 and 17.46 µg/ml, respectively. The results showed that there is a concentration dependent radical scavenging activity, the maximum activity being observed at 1000 µg/ml. This assay is based on the ability of antioxidants to facilitate the reduction of Fe3+ to Fe2+ ions in the presence of TPTZ, producing Fe2+-TPTZ complex (intense blue) that show maximum absorption at 593 nm. The decrease in maximum absorption has direct correlation with the antioxidant contents (Zahra et al., 2015). It has been reported that R. japonica contain phenolic compounds and has been widely used in traditional medicines for the treatment of various ailments including cancer, inflammation and a number of pathological conditions. Results of the study suggests that AgNPs have strong antioxidant activity due to the presence of bioactive compounds most probably phenolics on their surface. Therefore, we suggests that suitable formulation of AgNPs might be used in the treatment of mentioned human diseases.
Concentration (µg/ ml)
FRAP Scavenging activity (%)
Extract
AgNPs
Ascorbic acid
62.5
3.20 ± 0.25
9.80 ± 0.75
21.25 ± 1.12
125
7.65 ± 0.70
18.78 ± 0.56
34.67 ± 1.89
250
18.90 ± 1.25
32.56 ± 3.45
46.28 ± 3.36
500
29.75 ± 1.87
43.92 ± 5.10
59.73 ± 5.90
1000
42.56 ± 3.11
56.36 ± 7.80
78.80 ± 7.78
IC50
29.45
22.45
17.46
3.4.3 Hydrogen per oxide scavenging assay
Results of this assay are presented in Table 3. AgNPs showed considerable hydrogen peroxide scavenging activity compared to sodium pyruvate. Crude extract, AgNPs and standard revealed 34, 50 and 77 % inhibition at the highest concentration (1000 µg/ml) with IC50 values 33.66, 24.20 and 18.80 µg/ml, respectively. Hydrogen peroxide is a weak oxidizing agent, responsible for inactivation of several enzymes via oxidation of essential thiol (-SH) groups. It can rapidly cross the cell membrane and has the ability to react Cu2+ and Fe2+ ions to form – OH radicals that in turn initiate cell damage in vivo. Scavenging this radical remained an important antioxidant activity for the protection of living cell and mitigating diseases initiated by free radicals (Giovanni et al., 2022).
Concentration (µg/ ml)
H2O2 Scavenging activity (%)
Extract
AgNPs
Sodium pyruvate
62.5
3.55 ± 0.30
8.45 ± 0.55
15.78 ± 0.80
125
7.12 ± 0.25
11.90 ± 0.55
22.56 ± 1.25
250
11.45 ± 0.65
21.56 ± 2.66
38.29 ± 2.56
500
19.80 ± 1.0
34.56 ± 4.45
55.39 ± 5.89
1000
34.35 ± 3.78
50.0 ± 6.90
77.80 ± 6.88
IC50
33.66
24.20
18.80
3.4.4 ABTS scavenging assay
Table 4 represents the ABTS radical scavenging effects of the test samples. The % inhibition values observed at 1000 µg/ ml for crude extract, AgNPs and standard were 61, 74 and 80 %, respectively. The corresponding IC50 values were 25.45, 18.88 and 15.34 µg/ ml, respectively. All the test samples exhibited dose dependent activity. ABTS is a stable radical cation capable of donating hydrogen ions. It has been reported that phenolics have more ability to quench ABTS free radicals (Nataraj et al., 2013; Oana et al., 2020). The present results revealed that beside silver NPs, crude extract demonstrated strong antioxidant activity compared to standard. Since, the extract has the potential to scavenge free radicals, and thereby prevent lipid oxidation it could serve as potential nutraceutical. Similarly, silver NPs showed improved antioxidant effects compared to crude extract therefore; it could be used to prepare suitable formulation for the safe and effective treatment of several diseases. Previous studies have reported the antioxidant potential of plant mediated AgNPs using Annona muricata, Persea americana and Opuntia ficus-indica extracts as good scavengers of free radicals. These NPs have comparatively many advantages over synthetic antioxidants including improved bioavailability, stability and targeted delivery (Badmus et al., 2020; Adebayo et al., 2019).
Concentration (µg/ ml)
ABTS radical Scavenging activity (%)
Extract
AgNPs
Ascorbic acid
62.5
7.35 ± 0.25
19.60 ± 0.80
24. 55 ± 1.10
125
19.56 ± 1.0
25.58 ± 2.34
29.25 ± 1.80
250
28.32 ± 1.88
41.60 ± 3.30
45.50 ± 3.30
500
42.67 ± 3.80
58.10 ± 5.38
63.78 ± 7.75
1000
61.70 ± 6.35
74.48 ± 7.12
80.58 ± 8.80
IC50
25.45
18.88
15.34
3.5 Cytotoxicity assay
In this study, the cytotoxic potential of R. japonica root extract and its NPs were evaluated against different human cancer cell lines including A549, MDA-MB-231 and Hep-G2 using MTS assay. Furthermore, the safety profile of the test samples were investigated against two normal human cell lines e.g. HPAEpiC and HRPTEpiC. Silver NPs have encouraging applications in mitigating wound healing, breast, skin, lung and liver cancers (Oana et al., 2020). To validate and support the development of anticancer formulations, the synthesized silver NPs were investigated for their cytotoxic potential against the mentioned cell lines. In present study, the % cell viability treated with different concentrations of extract and NPs were determined (Figure S1-S3) and the corresponding IC50 values were calculated (Fig. 6).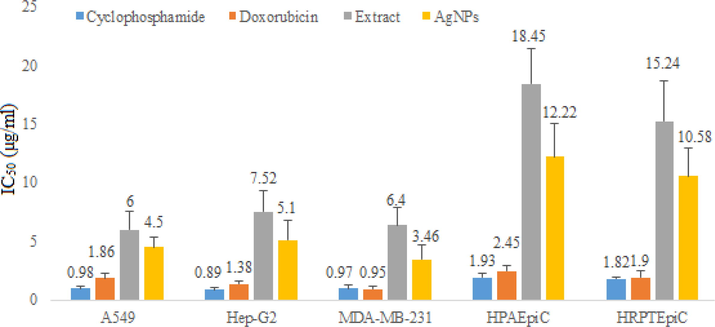
IC50 values of extract and silver nanoparticles against different cell lines using cyclophosphamide and doxorubicin as positive and DMSO as negative control.
Silver NPs demonstrated significant cytotoxic activity against A549, Hep-G2 and MDA-MB-231 cell lines with IC50 values 4.50, 5.15 and 3.46 µg/ml, respectively, compared to standards cyclophosphamide (IC50 values 0.98, 0.89 and 0.97 µg/ml, respectively) and doxorubicin (IC50 values 1.86, 1.38 and 0.95, µg/ml, respectively). On the other hand, crude methanol extract showed relatively week cytotoxic activity against the mentioned cell lines with IC50 values 6, 7.52 and 6.45 µg/ml, respectively. In order to assess the safety profile of the test samples, they were tested against two normal human cell lines. The percent cell viability values are depicted in figures S4 & S5. The corresponding images for different cell lines are depicted in figures S6-S9. The IC50 values of the extract and NPs were 18.45 and 12.22 µg/ml, respectively against HPAEpiC and 15.24 and 10.58 µg/ml, respectively against HRPTEpiC. The results showed that IC50 values remained high against the normal human cell lines that preliminary confirms its safety against normal cell. In order to further elaborate the selectivity of the test samples, the respective selectivity index values were determined (Figs. 7 & 8).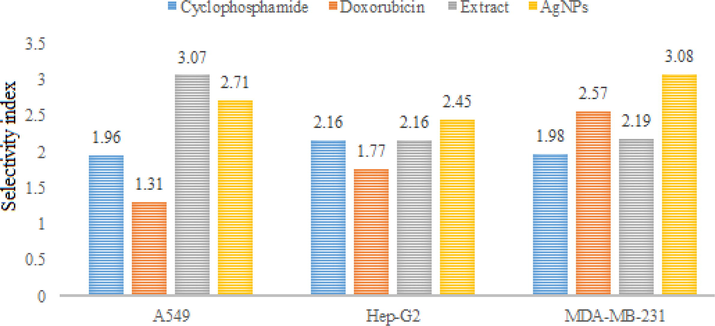
Selectivity indices of standards, extract and silver nanoparticles against HPAEpiC.
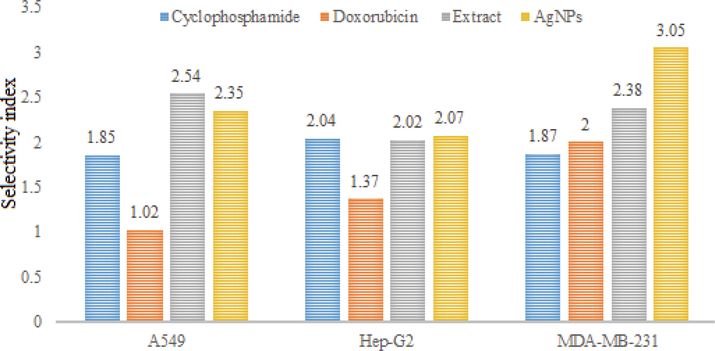
Selectivity indices of standards, extract and silver nanoparticles against HRPTEpiC.
Crude extract and silver NPs exhibited high selectivity index values for A549, Hep-G2 and MDA-MB-231. The SI values of crude extract for the mentioned cell lines were 3.07, 2.16 and 2.19, respectively when investigated against HPAEpiC. The corresponding SI values of silver NPs for the same cell lines were 2.17, 2.45 and 3.08, respectively. SI values of two or more indicated high specificity. Thus, the results suggest that R. japonica extract and its NPs exhibit strong anti-proliferative effects without affecting the normal cells. The possible mechanism(s) for the said activity many include but not limited to ROS generation and activation of several cellular metabolic pathways e.g. tumor necrosis factor-alpha (TNF-α) and nuclear factor kB (NF- kB) which ultimately triggers apoptosis and cell death. Plant mediated AgNPs have shown significant cytotoxic activity against different cell lines including Hep-G2 (liver cancer), PC3 (human prostate cancer), HeLa (cervical cancer), A549 (lung cancer), BxPC-3 (pancreas cancer) and MCF-7 (human breast cancer). AgNPs synthesized from mint extract have shown promising activity against HCT116 colon cancer cell lines; the author attributed the cytotoxic activity to the delayed cell division in G1 phase. In another study, AgNPs from leaf extract of Piper nigrum showed significant activity against MCF-7 and Hella cancer cell lines. Significant disruption in plasma membrane integrity and inhibition in cell growth was found in the studied cells (Jabeen et al., 2021). Similarly, the cytotoxicity of AgNPs can be attributed to; formation of stable S-Ag bond with thiol group of specific enzymes in cell membrane and its deactivation; denaturation of DNA by breaking H-bonds between nitrogen base of DNA; production of free radicals and ATP leakage (Patil and Kumbhar 2017; Shahzadi et al., 2022; Ragupathi et al., 2016). In last few years, a number of potent cytotoxic drugs have been developed however, the emergence of resistance and side effects associated with these drugs demand for search of safe and effective cytotoxic agents. Present study strongly revealed the cytotoxic potential of synthesized AgNPs. This activity may be due to the synergistic effect of nano sized silver and bioactive molecules attached on their surface. However, we suggests further studies to find out the exact mechanism for cytotoxic activity associated with these nanoparticles.
4 Conclusion
Herein, a simple and ecofriendly method was adopted for the synthesis of silver NPs using R. japonica root extract, which produced spherical shaped NPs without using any harmful reducing or capping agents. The role of phytochemicals in the synthesis of AgNPs was confirmed by FTIR studies. The AgNPs exhibited strong antioxidant potential through DPPH, FRAP, H2O2 and ABTS radical scavenging assays. This activity is believed to be due to the adsorption of phenolics and flavonoids on the surface of AgNPs. Moreover, AgNPs showed significant cytotoxic effects against the selected cancer cell lines. This activity is believed to be due to the small size and improved bioavailability of AgNPs as compared to crude extract. In addition, the NPs remained highly selective for the mentioned cell lines as evident from its selectivity index values. Current study provides an insight into the usage of R. japonica root extract as a potential natural source of antioxidant and could have great importance as therapeutic agent in ameliorating or preventing oxidative stress related diseases such as cancer. However, further studies are needed to confirm the role of R. japonica mediated AgNPs in mitigating the mentioned type of cancers and to validated its proper mechanism.
Acknowledgements
The authors wish to thanks the Researchers Support Project Number (RSP-2021/110) at King Saud University, Riyadh, Saudi Arabia, for financial support.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Antimicrobial and antioxidant activity of silver, gold and silver-gold alloy nanoparticles phytosynthesized using extract of Opuntia ficus-indica. Rev. Adv. Mater. Sci.. 2019;58:313-326.
- [CrossRef] [Google Scholar]
- Biosynthesis of silver, gold and silver–gold alloy nanoparticles using Persea americana fruit peel aqueous extract for their biomedical properties. Nanotechnol. Environ. Eng.. 2019;4:13.
- [CrossRef] [Google Scholar]
- Adelere, I.A., Lateef, A., 2021. Microalgal Nanobiotechnology and Its Applications—A Brief Overview. In: Microbial Nanobiotechnology: Principles and Applications. Springer Nature Singapore Pte. Ltd. ISBN 978-981-334-777-9 Pp. 233-255. https://doi.org/10.1007/978-981-33-4777-9_8.
- A novel approach to the green synthesis of metallic nanoparticles: the use of agro-wastes, enzymes, and pigments. Nanotechnol. Rev.. 2016;5(6):567-587.
- [CrossRef] [Google Scholar]
- Synthesis, bioactivities and cytogenotoxicity of animal fur-mediated silver nanoparticles. IOP Conf. Ser.: Mater. Sci. Eng.. 2020;805:012041.
- [Google Scholar]
- In vitro antioxidant properties and total phenolic and flavonoid contents of Rumex vesicaius. Int. J. Pharm. Pharm. Sci.. 2015;7:7.
- [Google Scholar]
- Impact of effective microorganisms and manure on the chemical properties of japanese knotweed leaves and their use in the production of functional foods. Pak. J. Agri. Sci.. 2019;56:313-320.
- [CrossRef] [Google Scholar]
- Photo-assisted bio-fabrication of silver nanoparticles using Annona muricata leaf extract: exploring the antioxidant, anti-diabetic, antimicrobial, and cytotoxic activities. Heliyon. 2020;6(11):EO5413.
- [CrossRef] [Google Scholar]
- Biochemical and enzyme inhibitory attributes of methanolic leaf extract of Datura inoxia Mill. Environ. Sustain.. 2019;2:75-87.
- [CrossRef] [Google Scholar]
- Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin.. 2018;68(6):394-424.
- [CrossRef] [Google Scholar]
- Antioxidant activity of chinese medicinal herbs. Pharm. Biol.. 2008;46:587-595.
- [CrossRef] [Google Scholar]
- Antioxidant Activities and Phenolic Content of Piper wallichii (Miq.) Hand.-Mazz. Int. J. Food Prop.. 2014;17:309-320.
- [CrossRef] [Google Scholar]
- Transcriptional repression of TWIST1 gene by Prospero-related homeobox 1 inhibits invasiveness of hepatocellular carcinoma cells. Lett.. 2012;586:3746-3752.
- [CrossRef] [Google Scholar]
- A review of the antioxidant potential of medicinal plant species. Food Bioprod. Process.. 2011;89:217-233.
- [CrossRef] [Google Scholar]
- Synthesis of gold nanoparticles using Sambucus wightiana extract and investigation of its antimicrobial, anti-inflammatory, antioxidant and analgesic activities. Arab. J. Chem.. 2021;14:103343
- [CrossRef] [Google Scholar]
- Screening of Rhamnus Purpurea (Edgew.) leaves for antimicrobial, antioxidant, and cytotoxic potential. ACS Omega 20222c03094
- [CrossRef] [Google Scholar]
- Studies on active substance of anticancer effect in Polygonum cuspidatum. Chin. Med. Mater.. 2006;29(7):689-691. PMID: 17059010
- [Google Scholar]
- Global, regional, and national cancer incidence, mortality, years of life lost, years lived with disability, and disability-adjusted life-years for 32 cancer groups, 1990 to 2015: a systematic analysis for the global burden of disease study. JAMA Oncol.. 2017;3(4):524-548.
- [CrossRef] [Google Scholar]
- Microbiota dysbiosis: a new piece in the understanding of the carcinogenesis puzzle. J. Med. Microbiol.. 2016;65:1347-1362.
- [CrossRef] [Google Scholar]
- Free radical properties, source and targets, antioxidant consumption and health. Oxygen. 2022;2:48-78.
- [CrossRef] [Google Scholar]
- Identifying the reasons for delayed presentation of Pakistani breast cancer patients at a tertiary care hospital. Cancer Manag. Res.. 2019;11:1087-1096.
- [CrossRef] [Google Scholar]
- Anticancer activity and molecular mechanism of resveratrol-bovine serum albumin nanoparticles on subcutaneously implanted human primary ovarian carcinoma cells in nude mice. Cancer Biother. Radiopharm.. 2010;25:471-477.
- [CrossRef] [Google Scholar]
- Antiproliferative activity of green, black tea and olive leaves polyphenols subjected to biosorption and in vitro gastrointestinal digestion in Caco-2 cells. Food Res. Int.. 2020;136:109317
- [CrossRef] [Google Scholar]
- Circ_0078710 promotes the development of liver cancer by upregulating TXNDC5 via miR-431-5p. Ann. Hepatol. 2021100551
- [CrossRef] [Google Scholar]
- Synthesis of silver nanoparticles using root extract of Duchesnea indica and assessment of its biological activities. Arab. J. Chem.. 2021;14(5):103110
- [CrossRef] [Google Scholar]
- Simultaneous measurement of total antioxidant power and ascorbic acid concentration. Methods Enzymol.. 1999;299:15-27.
- [Google Scholar]
- Application of green synthesized silver nanoparticles in cancer treatment—a critical review. Mater. Res. Express. 2021;8:092001
- [Google Scholar]
- Protective effect of polygoni cuspidati radix and emodin on Vibrio vulnificus cytotoxicity and infection. J. Microbiol.. 2008;46:737-743.
- [CrossRef] [Google Scholar]
- Resveratrol isolated from Polygonum cuspidatum root prevents tumor growth and metastasis to lung and tumor-induced neovascularization in lewis lung carcinoma-bearing mice. J. Nutr.. 2001;131:1844-1849.
- [CrossRef] [Google Scholar]
- Biomedical applications of green synthesized-metallic nanoparticles: a review. PAJOLS. 2019;3:157-182.
- [CrossRef] [Google Scholar]
- Facile synthesis of silver nanoparticles using leaf extract of Hyptis suaveolens (L.) Poit for environmental and biomedical applications. IOP Conf. Ser.: Mater. Sci. Eng.. 2020;805:012042
- [Google Scholar]
- In situ syntheses of silver nanoparticles inside silver citrate nanorods via catalytic nanoconfinement effect. Colloids Surf., A Physicochem. Eng. Asp.. 2020;605:125343
- [CrossRef] [Google Scholar]
- Health and economic impact at a population level of both primary and secondary preventive lung cancer interventions: a model-based cost-effectiveness analysis. Lung Cancer.. 2021;159:153-161.
- [CrossRef] [Google Scholar]
- Gut microbiota targeted nanomedicine for cancer therapy: challenges and future considerations. Trends Food Sci. Technol.. 2021;107:240-251.
- [CrossRef] [Google Scholar]
- Comparison of viral hepatitis-associated hepatocellular carcinoma due to HBV and HCV - cohort from liver clinics in Pakistan. Asian Pac. J. Cancer Prev.. 2014;15:7563-7567.
- [CrossRef] [Google Scholar]
- Antioxidant activity and free radical scavenging capacity of phenolic extracts from Helicteres isora L. and Ceiba pentandra L. J. Food Sci. Technol.. 2013;50(4):687-695.
- [CrossRef] [Google Scholar]
- Antimicrobial and antioxidant activities of alcoholic extracts of Rumex dentatus L. Microb. Pathog.. 2013;57:17-20.
- [CrossRef] [Google Scholar]
- An updated review on silver nanoparticles in biomedicine. Nanomaterials. 2020;10:2318.
- [CrossRef] [Google Scholar]
- Green synthesis of silver nanoparticles by Conocarpus Lancifolius plant extract and their antimicrobial and anticancer activities. Saudi. J. Biol. Sci.. 2022;29(1):460-471.
- [CrossRef] [Google Scholar]
- The global health burden of infection-associated cancers in the year 2002. Int. J. Cancer. 2006;118(12):3030-3044.
- [CrossRef] [Google Scholar]
- Antioxidant, antibacterial and cytotoxic potential of silver nanoparticles synthesized using terpenes rich extract of Lantana camara L. leaves. Biochem. Biophys. Rep.. 2017;10:76-81.
- [CrossRef] [Google Scholar]
- A flower shapegreen synthesis and characterization of silver nanoparticles (AgNPs) with different starch as a reducing agent. J. Mater. Res.. 2020;9:11003-11012.
- [CrossRef] [Google Scholar]
- New insights into the application of nanoghosts as theranostic tools with an emphasis on cardiovascular diseases. J. Nanopart. Res.. 2021;23:254.
- [CrossRef] [Google Scholar]
- Natural exosome-like nanovesicles from edible tea flowers suppress metastatic breast cancer via ROS generation and microbiota modulation. Acta Pharm. Sin. B.. 2022;12(2):907-923.
- [CrossRef] [Google Scholar]
- Preparation and physicochemical characterization of Ag nanorods phytosynthesis by the Petroselinum crispum plant extract. Adv. Sci. Eng. Med. 2016;8(11):862-867.
- [CrossRef] [Google Scholar]
- Worldwide variation in the relative importance of hepatitis B and hepatitis C viruses in hepatocellular carcinoma: a systematic review. Br. J. Cancer. 2007;96(7):1127-1134.
- [CrossRef] [Google Scholar]
- Lung carcinoma: its profile and changing trends. J. Ayub. Med. Coll. Abbottabad. 2010;22:3.116-119. PMID: 22338434
- [Google Scholar]
- Genesis and development of DPPH method of antioxidant assay. J. Food Sci. Technol.. 2011;48:412-422.
- [CrossRef] [Google Scholar]
- Sajjad, A., Farhat, U., Muhammad, A., Abdul, S., Muhammad, I., 2015. Antioxidant and anticholinesterase investigations of Rumex hastatus D. Don: potential effectiveness in oxidative stress and neurological disorders. Biol. Res. 26, 48, 1, 20. DOI: 10.1186/s40659-015-0010-2
- Phyto-fabrication of silver nanoparticles by Acacia nilotica leaves: Investigating their antineoplastic, free radical scavenging potential and application in H2O2 sensing. J. Taiwan. Inst. Chem. Eng.. 2019;99:239-249.
- [CrossRef] [Google Scholar]
- Cancer prevalence, incidence and mortality rates in Pakistan in 2012. Cogent Med.. 2017;4:1288773.
- [CrossRef] [Google Scholar]
- Antioxidant, cytotoxic, and antimicrobial potential of silver nanoparticles synthesized using Tradescantia pallida extract. Front. Bioeng. Biotechnol.. 2022;10:907551
- [CrossRef] [Google Scholar]
- Breast cancer in Pakistan-awareness and early detection. J. Coll. Phys. Surg. Pak.. 2007;17(12):711-712.
- [Google Scholar]
- T'ao, 1986. Mingyi Bielu: Jijiao Ben. Renmin Weisheng Chubanshe, Beijing Pharmacopoeia, 2000. Pharmacopoeia of the People's Republic of China. Beijing.
- Isolation and identification of stilbenes in two varieties of Polygonum cuspidatum. J. Agric. Food Chem.. 2000;48:253-256.
- [CrossRef] [Google Scholar]
- Verma, D.K., Goyal, M.R., Suleria, H.A.R. (Eds.). 2019. Green Synthesis of Silver (Ag), Gold (Au), and Silver–Gold (Ag–Au) Alloy Nanoparticles: A Review on Recent Advances, Trends, and Biomedical Applications. Apple Academic Press Inc. /CRC Press, Taylor and Francis Group, Oakville, Ontario, Canada. ISBN 978-1-77188-764-9. Pp. 3-89. https://doi.org/10.1201/9780429425660-1.
- Analysis of Rhizoma Polygoni Cuspidati by HPLC and HPLC-ESI/MS. Phytochem. Anal.. 2007;18:387-392.
- [CrossRef] [Google Scholar]
- Antimicrobial, antioxidant and cytotoxic activity of silver nanoparticles synthesized by leaf extract of Erythrina suberosa (Roxb.) Front. Mol. Biosci.. 2017;17, 4:14.
- [CrossRef] [Google Scholar]
- Zahra, S., Jafar, V., Omid, A.S., Maryam, A., 2015. Antioxidant activity and total phenolic content of Boerhavia elegans (choisy) grown in Baluchestan, Iran. Avicenna J Phytomed. 5, 1, 1–9. PMCID: PMC4352527.
- Human gut-microbiome interplay: Analysis of clinical studies for the emerging roles of diagnostic microbiology in inflammation, oncogenesis and cancer management. Infect. Genet. Evol.. 2021;93:104946
- [CrossRef] [Google Scholar]
Appendix A
Supplementary material
Supplementary data to this article can be found online at https://doi.org/10.1016/j.arabjc.2022.104327.
Appendix A
Supplementary material
The following are the Supplementary data to this article:Supplementary data 1
Supplementary data 1







