Translate this page into:
Biotransformed bear bile powder alleviates diet-induced nonalcoholic fatty liver disease in mice by regulating the gut microbiota and reversing lipid metabolism
⁎Corresponding authors. nsi@icmm.ac.cn (Nan Si), blbian@icmm.ac.cn (Baolin Bian), hyzhao@icmm.ac.cn (Haiyu Zhao)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Background
The pathogenesis of metabolic syndrome was strongly associated with compromised metabolism homeostasis and gut microbiota imbalance. NAFLD is a progressive metabolic liver disease for which effective interventions are lacking. Bile acids exhibited appreciable metabolic regulatory effects and selective antimicrobial activity.
Aim of the study
This study was designed to investigate the effect of BBBP, which mainly contained bile acids, on NAFLD from the perspectives of gut microbiota and metabolomics.
Materials and methods
The present study was initiated on the anti-NAFLD effect of BBBP in HFD-fed mice. The efficacy of BBBP was evaluated by mice phenotypes, liver histopathological analysis and serum lipid and glucose levels. The activation of bile acid receptors such as Nr1h4, Nr1i2 and S1pr2 were detected by qRT-PCR analysis. Subsequently, untargeted metabolomics coupled with microbiomics was used to explore the mechanism of BBBP against NAFLD. Human L02 hepatocytes induced by OA and PA were used to investigate the effect of GABA on reducing lipid accumulation in vitro.
Results
BBBP significantly and dose-dependently alleviated the obese phenotype, lipid accumulation and liver injury in mice subjected to 18 weeks HFD diet. Untargeted metabolomics and microbiomics analysis revealed that BBBP could alleviate the disturbance of lipid and amino acid metabolism and the imbalance of gut microbiota. Furthermore, BBBP oral gavage activated liver bile acid receptors, as indicated by elevated mRNA levels of Nr1h4, Nr1i2 and S1pr2. Surprisingly, we determined that BBBP, which mainly contained bile acids that possessed antimicrobial activity, could promote the growth of Lactobacillus. Correlation analysis showed a remarkable correlation between Lactobacillus and endogenous metabolites such as valine, serine, glutamine, et al. Among them, GABA which could be produced by Lactobacillus significantly reduced the lipid accumulation in L02 cells.
Conclusions
The role of BBBP in regulating lipid metabolism might be achieved by activating bile acid receptors, or partially by promoting the levels of Lactobacillus and its metabolites such as GABA. Our study provided evidence that BBBP could be a novel therapeutic candidate for the treatment of NAFLD.
Keywords
Biotransformed bear bile powder
Nonalcoholic fatty liver disease
Gamma-aminobutyric acid
Metabolomics
Gut microbiota
L02 cells
Nomenclature
- AAAs
-
aromatic amino acids
- ALT
-
alanine transaminase
- AST
-
aspartate transaminase
- BBBP
-
biotransformed bear bile powder
- BCAAs
-
branched-chain amino acids
- CA
-
cholic acid
- CCK-8
-
cell counting kit-8
- CDCA
-
chenodeoxycholic acid
- DCA
-
deoxycholic acid
- FC
-
fold change
- FXR
-
farnesoid X receptor
- GABA
-
gamma-aminobutyric acid
- HDL-C
-
high-density lipoprotein cholesterol
- HFD
-
high-fat diet
- LDL-C
-
low-density lipoprotein cholesterol
- NAFLD
-
nonalcoholic fatty liver disease
- OA
-
oil acid
- OPLS-DA
-
orthogonal partial least squares discriminant analysis
- PA
-
palmitic acid
- PCA
-
principal component analysis
- PLS-DA
-
partial least squares discriminant analysis
- PXR
-
pregnane X receptor
- QC
-
quality control
- qRT-PCR
-
quantitative real‐time PCR
- S1pr2
-
sphingosine-1-phosphate receptor 2
- SD
-
standard deviation
- TBA
-
total bile acids
- TC
-
total cholesterol
- TCA
-
taurocholate acid
- TCDCA
-
taurochenodeoxycholic acid
- TG
-
triglyceride
- TUDCA
-
tauroursodeoxycholic acid
- VIP
-
variable importance in the projection
- UDCA
-
urosodeoxycholic acid
Abbreviations
1 Introduction
NAFLD usually originates from fat accumulation in the liver and uncontrollably develops into the more severe stages such as non-alcoholic steatohepatitis, cirrhosis and hepatocellular carcinoma (Steinacher et al., 2017). The prevalence rate of NAFLD was strikingly increasing and a recent large nationwide cohort study showed significantly increased overall mortality with all NAFLD histological stages including steatosis (Simon et al., 2021). NAFLD has become one of the most serious healthcare burdens in the 21st century and steatosis can no longer be ignored as ‘‘benign and an incidental finding” (Ni et al., 2023; Shi et al., 2022).
From a mechanistic perspective, disease progression in NAFLD occurs against a complex backdrop of metabolic and endocrine derangements (Lade et al., 2014). The disruption of hepatic lipid metabolism homeostasis was considered one of the key events in the pathogenesis of NAFLD (Li et al., 2021). An association between circulating amino acids and fatty liver disease has also been found in several clinical and animal studies (Lynch and Adams, 2014). The levels of amino acids, in particular, BCAAs (leucine, isoleucine, and valine) and AAAs (tyrosine and phenylalanine) have been altered in NAFLD patients (Fischer et al., 1976; Michitaka et al., 2010). Moreover, multiple studies have found that the imbalance of gut microbiota may also be one of several crucial factors in the pathogenesis of NAFLD (Jiang et al., 2015) based on animal models or clinical trials. Individuals with fatty liver disease have a substantially altered gut microbiota, including Ruminococcus which was positively correlated with the severity of liver fibrosis (Boursier et al., 2016). Besides, the detrimental metabolites (ethanol, saturated fatty acids, polyamines, hydrogen sulfide, AAAs and so on) of gut microbiota not only directly damage the intestinal barrier, but also lead to the occurrence of a series of liver diseases (Hu et al., 2020; Aron-Wisnewsky et al., 2020a). For example, ethanol produced by Escherichia coli activated the nuclear factor-κB signaling pathway, which might lead to liver inflammation and steatohepatitis (Nair et al., 2001; Rao et al., 2004). Probiotic treatment such as cidomycin (Wu et al., 2008) and rifaximin (Ponziani et al., 2017) could effectively inhibit the overgrowth of gut microbiota and their harmful metabolites, thereby alleviating liver inflammation and fibrosis. Notably, gut microbiota has been recognized as the novel therapeutic target for NAFLD (Hou et al., 2022). Unfortunately, despite past decades of research, no FDA-approved drugs can be used to effectively treat NAFLD. Thus, effective improvement and treatment strategies for NAFLD are urgently needed.
Current scientific strategies for discovering treatment against NAFLD were usually focused on a single target. Considering that the pathogenesis of NAFLD is complex and heterogeneous, pharmacological combination therapy will be favored in the future (Xu et al., 2022). BBBP which contained multiple bile acid compounds was produced by biological transformation of the chicken bile powder in vitro. Two key enzymes 7α -HSDHs and 7β -HSDHs that could convert TCDCA into TUDCA played a crucial role in the efficient biological transformation process (Ji et al., 2018; Song et al., 2017). Studies have shown that BBBP possesses profound liver protection effects, such as anti-cholestasis, anti-hepatic fibrosis and anti-acute liver injury effects (Wu et al., 2020; Zheng et al., 2019; Cai et al., 2022). More importantly, our previous research revealed that BBBP could ameliorate non-alcoholic steatohepatitis induced by a combination of 20 weeks of HFD and high-sugar drinking water through the regulation of the arginine biosynthesis (Jiang et al., 2023a). Based on the liquid chromatography-tandem mass spectrometry method, we identified 29 bile acid compounds in BBBP, such as CA, TCA, UDCA, CDCA, DCA, TUDCA, TCDCA and et al. The results of the quantitative analysis indicated that TUDCA, TCDCA, and TCA contents in BBBP were the highest, with a total content exceeding 60% (Jiang et al., 2023a). Bile acids have several important functions as they can activate multiple receptors to assist lipid digestion and absorption once they are released into the intestine (Masoodi et al., 2021). Meanwhile, bile acids appeared to play a major role in the regulation of the gut microbiome. Recent evidence suggested bile acids could control the overgrowth and composition of the microbiota and then protect the liver (Clarke et al., 2012). Thus, we hypothesized that BBBP oral gavage was probably effective in preventing the development of NAFLD.
Accumulated evidence suggested that the composition of gut microbiota varied with the different health conditions of the host (Fan and Pedersen, 2021; Rothschild et al., 2018). Once the homeostasis of gut microbiota is broken, disease will inevitably arise (Ahrodia et al., 2022). 16S rRNA gene sequencing has been regarded as a quick and accurate technique for investigating the characteristics of gut microbiota under different treatments (Regueira-Iglesias et al., 2023). As the building blocks of cellular function, metabolites were involved in enzyme-catalyzed chemical reactions and played an important role in linking genotypes and phenotypes (Schrimpe-Rutledge et al., 2016). Metabolomics could detect the subtle alterations of metabolites, providing important insights for in-depth analysis of the pathogenesis (Schrimpe-Rutledge et al., 2016). An overwhelming number of reports demonstrated that joint analyses of high throughput data were essential for understanding the molecular mechanism of common chronic metabolic disorders such as NAFLD (Perakakis et al., 2020).
In this study, we confirmed the anti-NAFLD effect of BBBP in HFD-fed mice. Mechanically, multi-omics profiling revealed that BBBP significantly improved the homeostasis of lipid and amino acid metabolism. More importantly, the regulatory effect of BBBP on lipid metabolism was not only attributed to the activation of bile acid receptors but also related to the regulation of gut microbiota and its metabolites such as GABA, at least partially. These data provided novel therapeutic strategies for NALFD by modulating the gut microbiota and host metabolism. Our findings provided experimental evidence to support BBBP as a potential therapeutic option for NAFLD.
2 Materials and methods
2.1 Chemicals and biological materials
BBBP was obtained from Chongqing Jize Biotechnology Co., Ltd (Chongqing, China). Pioglitazone was purchased from Takeda Pharmaceutical Company Limited (Tokyo, Japan). GABA was purchased from Solarbio Science & Technology (Beijing, China). The high-fat animal diets (D12492, 60% kcal fat) were purchased from Research Diets Inc. (NJ, USA). MS-grade formic acid and acetonitrile were purchased from Thermo Fisher Scientific (MA, USA).
2.2 Animal experiments
Male 4-week-old C57BL/6 mice were purchased from the Vital River Laboratory Animal Technology, Co., Ltd. (Beijing, China). Mice were housed under pathogen-free conditions and supplied ad libitum with sterilized feed and pure water. After 7 days of acclimation, mice were randomly divided into a control group (n = 9) and a model group (n = 36). The control group was fed a normal diet for 10 weeks, whereas the other group was fed an HFD for 10 weeks. Then, the mice fed with HFD were randomly divided into 4 groups (the model, positive, BBBPL and BBBPH group). Concurrent with the HFD, the mice were given BBBP by gavage daily in the BBBPL (75 mg/kg, n = 9), BBBPH (150 mg/kg, n = 9) group and were orally administered pioglitazone (30 mg/kg, n = 9) and pure water (0.1 mL/10 g, n = 9) daily in the positive group and model group for 8 weeks, respectively. All animal experiments were performed following the Guide for the Care and Use of Laboratory Animals. The animal experiments were approved by the Institute of Chinese Materia Medica, China Academy of Chinese Medical Sciences (approval certificate number: 2022B074).
2.3 Pharmacodynamic indicators analysis
2.3.1 Body weight and liver index indicators
The body weight was measured weekly during the experiment. After 19 weeks, mice were sacrificed. Then the blood samples and liver tissues were collected. According to the manufacturer’s instructions, the levels of blood glucose, serum TC, TG, HDL-C, LDL-C, ALT, AST and TBA were determined using a BX3010 automatic biochemical analyzer (Sysmex, Kobe, Japan). Besides, the whole livers were weighed and the liver index was calculated according to the formula (liver index = liver wet weight/body weight × 100%).
2.3.2 Tissue histopathological analysis
Experimental manipulations followed the study conducted by Jiang et al. (2023a). Briefly, the liver tissue was embedded in paraffin and routinely sectioned and stained with hematoxylin-eosin to observe the inflammation status of the liver section. Meanwhile, the hepatic steatosis was evaluated by Oil Red O staining of liver sections that were stained with 0.5% Oil Red O solution in 60% isopropanol. The pathological changes of the liver tissue were imaged using an inversion microscope (Nikon E 100, Tokyo, Japan).
2.4 L02 cell culture and in vitro cell model of lipid accumulation
Human L02 hepatocytes were obtained from the Chinese Academy of Science (Shanghai, China). L02 cells were maintained in Dulbecco’s modified Eagle’s medium (high glucose) supplemented with 10% fetal bovine serum and 1% penicillin–streptomycin at 37 ℃ in a CO2 incubator. To establish the in vitro cell model of lipid accumulation, L02 cells were treated with 0.5 mM PA and 1 mM OA for 24 h as previously described (Wang et al., 2021).
2.5 GABA treatment in the lipid accumulation cell model
The cell viability after GABA treatment was assessed by the CCK-8 assay (Dojindo Molecular Technologies, Inc., Kumamoto, Japan) according to the manufacturer’s instructions. To investigate whether GABA could reduce lipid accumulation, the L02 cells were pretreated with various concentrations of GABA (GABAH 80 µmol/L, GABAM 40 µmol/L, GABAL 20 µmol/L) for 24 h. Then the cells were incubated with 0.5 mM PA and 1 mM OA for 24 h (Jiang et al., 2022). Oil red O staining was performed for intracellular lipid accumulation examination. The TG level was determined using a BX3010 automatic biochemical analyzer (Sysmex, Kobe, Japan).
2.6 Gut microbiota diversity analysis
At the end of the experimental period, mouse faces were collected and then the 16S rRNA gene sequences of collected fecal samples were detected by Shanghai Major Biomedical Technology Co., Ltd. (Shanghai, China). RNA extraction, PCR amplification, product purification and quantification, and library preparation and sequencing were performed as previously described (Cheng et al., 2022). The V4 region of the 16S rRNA genes using 338F (5′-209 ACTCCTACGGGAGGCAGCA-3′) and 806R (5′-GGACTACHVGGGTWTCTAAT-3′) primers. Data processing and analysis were performed on the online platform of Majorbio (http://www.majorbio.com/).
2.7 Untargeted metabolomics based on LC-MS analysis
2.7.1 Serum sample preparation
50 μL serum was added with 190 μL cold methanol in the centrifuge tube. The mixture was vortexed for 3 min. 380 µL dichloromethane was added and the mixture was vortexed. Then 120 µL water was added and vortexed for 10 s to form a two-phase system. Both the resulting lower organic phase and upper aqueous phase were collected for analysis. In addition, 10 μL of each sample was mixed to obtain a QC sample.
2.7.2 Data acquisition for untargeted metabolomics
Serum untargeted metabolomics was conducted using UPLC-ESI-Orbitrap MS system (Ultimate 3000, Thermo Fisher Scientific, MA, USA). The separations of polar and non-polar metabolites were achieved on a UPLC BEH Amide column (2.1 mm × 100 mm, 1.7 μm, Waters, MA, USA) and UPLC HSS T3 column (2.1 mm × 100 mm, 1.8 μm, Waters, MA, USA), respectively. According to the established methods (Jiang et al., 2023a), the detailed methods of chromatographic conditions and the parameters of the mass spectrometer were provided in the Supplementary Information.
2.7.3 Serum metabolome data processing
As previously described (Jiang et al., 2023b), the acquired raw UPLC-MS/MS data files were imported to into the Progenesis QI software (Waters, MA, USA) to obtain reliable datasets. Then, the SIMCA-P 14.0 software (Umetrics, Sweden) was used to perform the multivariate statistical analysis. The PCA, PLS-DA, and OPLS-DA were subsequently used to assess the overall difference between groups. Metabolites with VIP ≥ 1.5, FC ≥ 2 and P<0.05 were screened as the differential metabolites. For the identification of differential metabolites, the human metabolome database (https://hmdb.ca/) was used. Finally, the KEGG pathway enrichment analysis of differential metabolites was analyzed by Metaboanalyst 5.0 (https://www.metaboanalyst.ca/).
2.8 qRT-PCR analysis
qRT-PCR analysis was performed according to the established methods (Jiang et al., 2023a). Briefly, liver total RNA was extracted using E.Z.N.A. Total RNA Kit (Omega Bio-Tek, GA, USA). Following being reverse-transcribed with the ReverTra Ace® qPCR RT Master Mix with gDNA Remover (TOYOBO, Osaka, Japan), ABI Prism 7900 HT Real-Time PCR System (Thermo Fisher Scientific, MA, USA) was used to perform the qRT-PCR with an SYBR Green Master Mix (TransGen Biotech, Beijing, China). Genes expression was evaluated using the 2−ΔΔCT method, followed by being normalized to β-actin. The primers were listed in Table 1.
Gene
Forward
Reverse
S1pr2
5ʹ-ATGGGCGGCTTATACTCAGAG-3ʹ
5ʹ-GCGCAGCACAAGATGATGAT -3ʹ
Nr1h4
5ʹ-CGCGCTCACCTGTGAGGGCTGC-3ʹ
5ʹ-CCATCACGCAGTTGCCCCCGTTC-3ʹ
Nr1i2
5ʹ-GATGGAGGTCTTCAAATCTGCC-3ʹ
5ʹ-CAGCCGGACATTGCGTTTC-3ʹ
β-Actin
5ʹ-AGAGGGAAATCGTGCGTGAC-3ʹ
5ʹ-CAATAGTGATGACCTGGCCGT-3ʹ
2.9 Correlation analysis
The correlation among gut microbiota, metabolites, and lipid index was performed by Spearman correlation analysis (https://cloud.oebiotech.com/). The visual network was constructed and modified by the Cytoscape software.
2.10 Statistical analysis
Statistical analysis was determined using SPSS software based on one-way ANOVA (comparison among multiple groups). The experiment data were shown as mean ± SD. Plotting was performed using GraphPad Prism 8. Statistical significance was indicated as P < 0.05.
3 Results
3.1 BBBP attenuated HFD-induced obese phenotypes
To determine the protective effects of BBBP on NAFLD, C57BL/6 mice were subjected to 18 weeks of HFD with or without BBBP supplementation. The details for the experimental design were displayed in Fig. 1A. As anticipated, compared to the normal diet, HFD led to a significant body weight and liver weight gain in mice (P < 0.05). After 8 weeks of treatment, visual observation found that mice in the BBBP treatment group were thinner than those in the model group. Moreover, the body weight, liver weight and liver index in the BBBPL and BBBPH group mice were significantly lower than those in the model group (P < 0.5, Fig. 1B-D).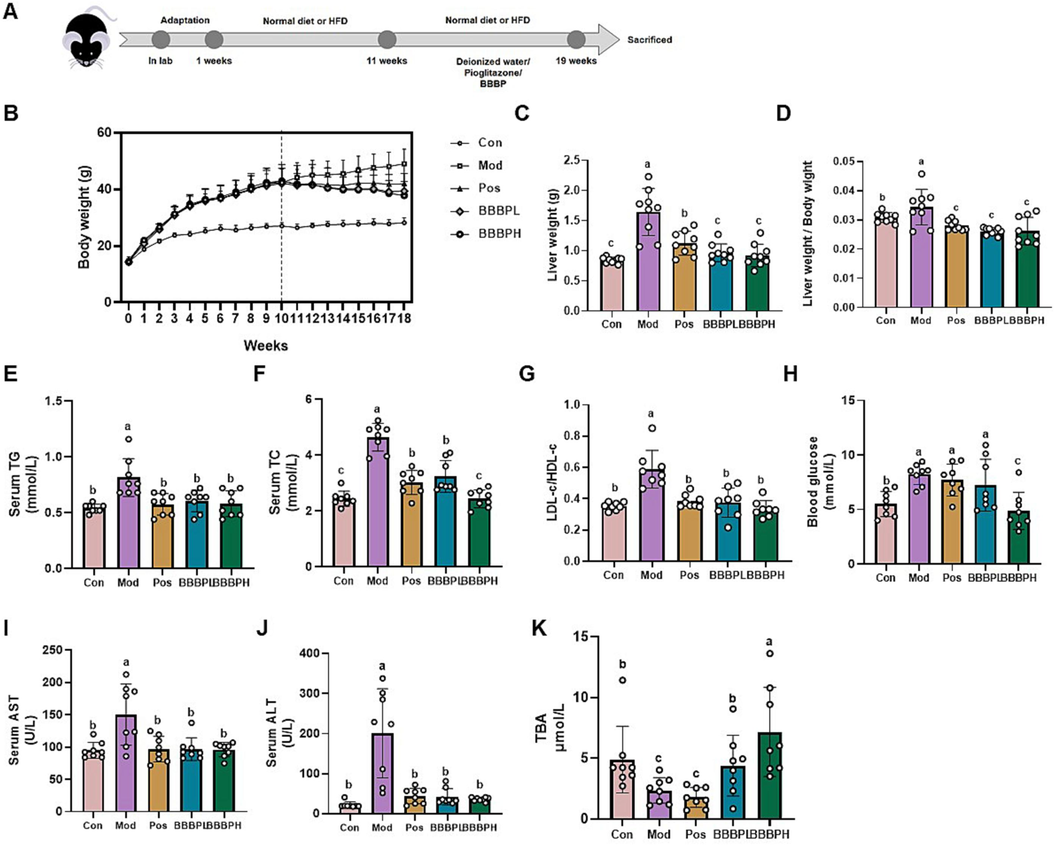
Effect of BBBP on obesity phenotypes and serum biochemical indicators in mice induced by HFD. (A) Animal experiment design. (B–D) The body weight, liver weight and liver index of mice in different groups (n = 9 per group). (E–G) Serum TG (E), TC (F), LDL-C/HDL-C ratio (G), glucose (H), AST (I), ALT (J) and TBA (K) levels (n = 8 per group). Different letters (a, b, c) represent statistically significant differences among groups (P < 0.05).
3.2 BBBP alleviated serum lipid and glucose levels
As shown in Fig. 1E, F, the serum lipid levels, including TG and TC, were significantly elevated in HFD-fed mice as compared to the normal-diet-fed mice. And the LDL-C/ HDL-C ratio which was positively correlated with the severity of hepatic steatosis was also increased (Tutunchi et al., 2021; Nobili et al., 2010, Fig. 1G). However, the above indicators were significantly decreased by BBBP supplementation (P < 0.05). Meanwhile, mice treated with BBBP presented lower levels of fasting blood glucose than those in the model group (Fig. 1H). Moreover, BBBP significantly reduced HFD-mediated upregulation of serum ALT and AST levels (Fig. 1I, J). High doses of BBBP could significantly increase serum TBA levels (P < 0.05), while the elevated TBA effect of low-dose BBBP was minimal (Fig. 1K, P > 0.05). Notably, high doses of BBBPH had a better protective effect on NAFLD than pioglitazone.
3.3 BBBP activated bile acid receptors and attenuated hepatic steatosis
One of the characteristic manifestations of NAFLD was abnormal hepatic lipid accumulation. As shown in Fig. 2A, B, the results of H&E staining showed that there were more infiltrated inflammatory cells and visible fat vacuoles in the model group. Oil red O staining presented that there were a large number of lipid droplets stained by red in the liver of mice induced by HFD. BBBP treatment remarkably attenuated HFD-induced hepatic ballooning, inflammatory infiltration and lipid accumulation. Cell surface receptors (S1pr2 et al.) and nuclear receptors (PXR (Nr1h4), FXR (Nr1i2), et al.) have been regarded as bile acid receptors and play important roles in the regulation of liver lipid metabolism. The results of qRT-PCR analysis showed BBBP directly activated bile acid receptors, as indicated by elevated mRNA levels of Nr1h4, Nr1i2 and S1pr2 (Fig. 2C-E).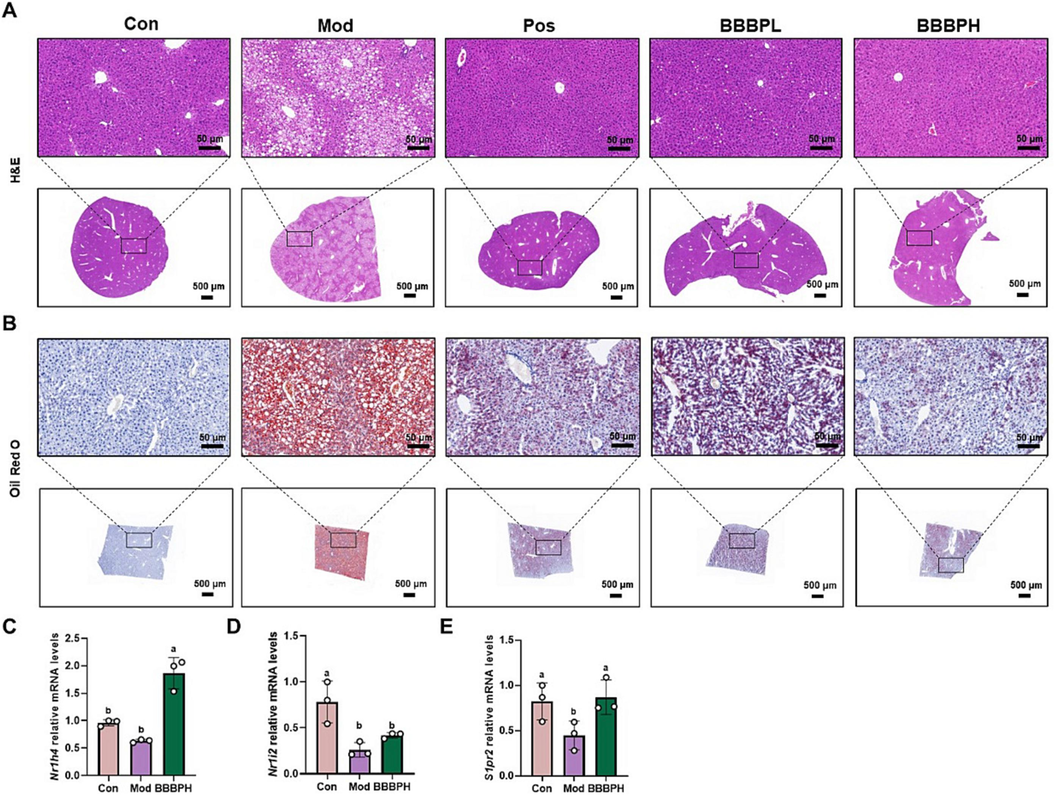
BBBP alleviated hepatic steatosis in HFD-induced NAFLD. (A, B) Representative morphology of liver H&E (A) and Oil Red O staining (B). (C–E) The mRNA levels of Nr1h4 (C), Nr1i2 (D) and S1pr2 (E) in mice liver (n = 3 per group). Different letters (a, b) represent statistically significant differences among groups (P < 0.05).
3.4 BBBP attenuated in vivo metabolic disturbances induced by HFD
Serum samples were analyzed by LC-MS-based untargeted metabolomics. The heatmap showed a correlation between samples (Fig. 3A-D). In PCA analysis, QC samples were closely clustered among all samples, indicating that the method and the results were reliable (Fig. 3E-H). In the positive and negative ion modes, a distinct clustering of serum samples was observed between the control and model groups, and the BBBPH groups tended to approach the control group in the PCA plots (Fig. 3E-H), implying a marked metabolic disorder in HFD fed mice, which were significantly ameliorated after BBBP treatment. Furthermore, the same results were obtained in the PLS-DA score plots (Fig. 3I-L). The model parameters of permutation analysis showed an upward trend, indicating that the models were stable and accurately predictive (Fig. 3M-P). Subsequently, 39 differential metabolites were screened by VIP > 1, P < 0.05 and FC ≥ 1.5 (Table 2). Then they were identified using the HMDB database, and the KEGG pathway enrichment analysis of differential metabolites illustrated that lipid and amino acid metabolism such as biosynthesis of unsaturated fatty acids, histidine metabolism, arginine biosynthesis and et al. might play a crucial role in BBBP preventing NAFLD (Fig. 4A, B). Note: *P < 0.05, **P < 0.01, ***P < 0.001 vs. control group; #P < 0.05, ##P < 0.01, ###P < 0.001 vs. model group. ↑ indicated a relative increase, while ↓ indicated a relative decrease.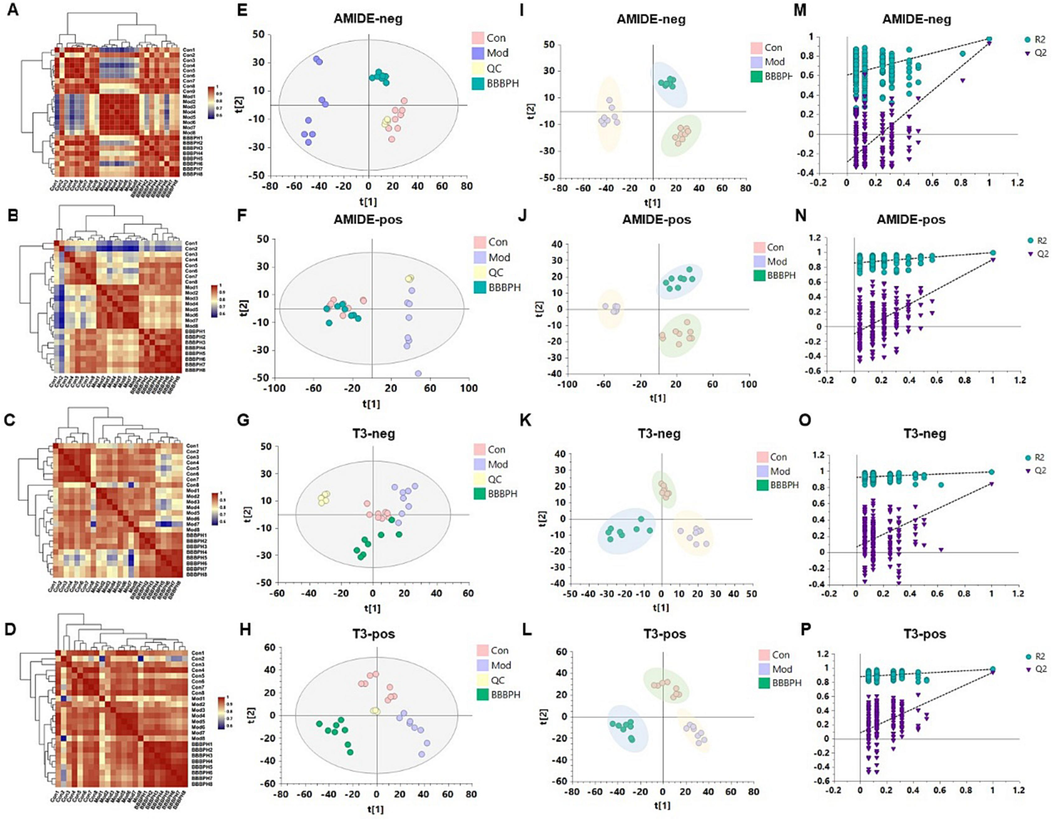
Characterization of serum metabolites in different groups of mice. (A–D) Heatmap of sample correlation (A Metabolites separated by AMIDE columns in negative ion mode; B Metabolites separated by AMIDE columns in positive ion mode; C Metabolites separated by T3 columns in negative ion mode; D Metabolites separated by T3 columns in positive ion mode). (E–H) PCA score plots of serum samples from different groups. (I–L) PLS-DA score and (M−P) the validation plots.
Peak
Formula
Theoretical Mass m/z
Experimental Mass m/z
Error ppm
Identification
Ion mode
Change trend (Mod VS Con)
Change trend (BBBPH VS Mod)
1
C8H7ONNa
156.0419
156.0416
−2.724
Indoxyl
[M + Na]+
***
###
2
C4H10O2N
104.0706
104.0702
−4.373
Gamma-aminobutyric acid
[M + H]+
**
###
3
C5H12O2N
118.0862
118.0859
−3.177
L-Valine
[M + H]+
***
###
4
C10H9N2
157.0760
157.0755
−3.342
3-Indoleacetonitrile
[M + H]+
***
###
5
C4H10O2N3
132.0767
132.0762
−4.036
Creatine
[M + H]+
***
###
6
C5H12O2NS
150.0583
150.0577
−4.237
L-Methionine
[M + H]+
***
###
7
C5H5O3N4
169.0356
169.0350
−3.470
Uric acid
[M + H]+/ [M−H]-
***
###
8
C6H14O6Na
205.0682
205.0675
−3.849
Mannitol
[M + Na]+
***
###
9
C6H12O3N
146.0811
146.0806
−3.832
4-Acetamidobutanoic acid
[M + H]+
***
###
10
C4H8O4N
134.0447
134.0444
−2.717
L-Aspartic acid/D-Aspartic acid
[M + H]+
***
###
11
C5H10O4N
148.0604
148.0599
−3.339
L-Glutamic acid
[M + H]+/ [M−H]-
***
##
12
C9H18O8Na
277.0893
277.0884
−3.424
Galactosylglycerol
[M + Na]+
***
###
13
C5H8O3N
130.0498
130.0494
−3.534
Pyroglutamic acid
[M + H]+
***
##
14
C5H11O3N2
147.0764
147.0759
−3.255
L-Glutamine
[M + H]+/ [M−H]-
***
###
15
C3H8O3N
106.0498
106.0494
−4.240
D-Serine/L-Serine
[M + H]+
***
##
16
C6H14O3N3
176.1029
176.1025
−2.770
Citrulline
[M + H]+/ [M−H]-
***
###
17
C5H10N3
112.0869
112.0866
−3.336
Histamine
[M + H]+
***
###
18
C7H12O2N3
170.0924
170.0918
−3.487
3-Methylhistidine/1-Methylhistidine
[M + H]+
***
###
19
C6H12O2N
130.08625
130.0859
−2.884
Pipecolic acid/L-Pipecolic acid
[M + H]+
***
###
20
C6H15O2N2
147.1128
147.1124
−2.544
D-Lysine/L-Lysine
[M + H]+
***
###
21
C9H8ON
146.0600
146.0595
−3.563
Indole-3-carboxaldehyde
[M + H]+
***
###
22
C4H9O3N2
133.0607
133.0603
−3.823
L-Asparagine
[M + H]+
***
###
23
C4H7O2N2
115.0502
115.0498
−3.772
Dihydrouracil
[M + H]+
***
###
24
C10H10ON
160.0756
160.0751
−3.564
Indoleacetaldehyde
[M + H]+
***
###
25
C6H7O2N2
139.0502
139.0497
−3.337
Urocanic acid
[M + H]+
***
###
26
C5H3O4N4
183.0148
183.0151
1.305
5-Hydroxyisourate
[M−H]-
***
###
27
C9H10O3N
180.0655
180.0656
0.668
L-Tyrosine
[M−H]-
***
###
28
C6H10O4N
160.0604
160.0608
2.222
Aminoadipic acid
[M−H]-
***
###
29
C6H11O7
195.0499
195.0503
1.850
Galactonic acid
[M−H]-
***
###
30
C6H8O2N3
154.0611
154.0617
4.005
L-Histidine
[M−H]-
***
###
31
C21H44O7P
439.2819
439.2818
−0.197
LysoPA(0:0/18:0)/LysoPA(18:0/0:0)
[M + H]+
***
###
32
C26H54O7NPNa
546.3530
546.3496
−6.224
LysoPC(18:0/0:0)
[M + Na]+
***
###
33
C22H35O2
331.2631
331.2630
−0.383
Adrenic acid
[M−H]-
**
#
34
C22H31O2
327.2318
327.2310
−2.649
Docosahexaenoic acid
[M−H]-
***
###
35
C20H31O2
303.2318
303.2312
−2.166
Arachidonic acid
[M−H]-
***
###
36
C22H45O7NP
466.2928
466.2915
−2.757
LysoPC(14:0/0:0)
[M−H]-
***
##
37
C24H49O7NP
494.3241
494.3219
−4.482
LysoPC(16:0/0:0)
[M−H]-
***
###
38
C25H51O7NP
508.3397
508.3376
−4.359
LysoPC(17:0/0:0)
[M−H]-
***
##
39
C23H47O7NP
480.3084
480.3064
−4.363
LysoPC(15:0/0:0)
[M−H]-
**
#
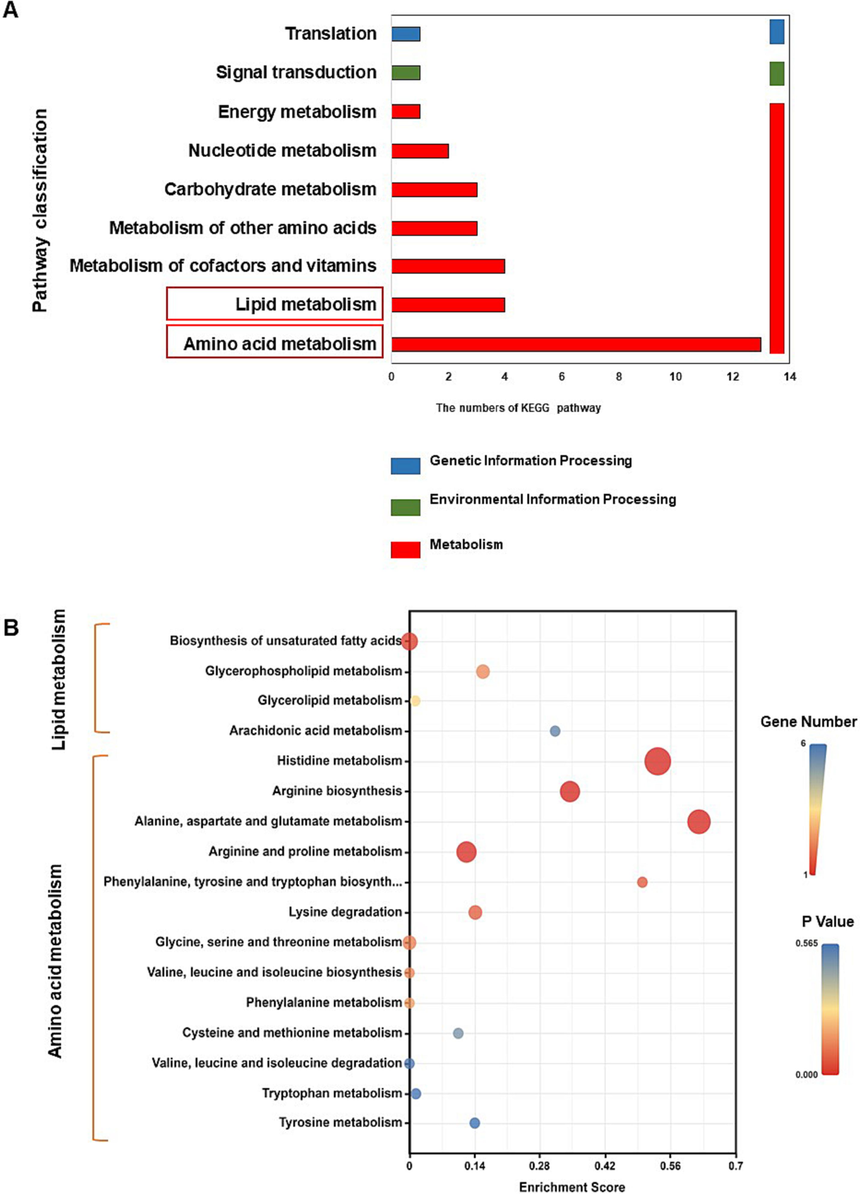
BBBP supplementation regulated serum metabolite profiles and related metabolic pathways in HFD-fed mice. (A) Secondary classification of KEGG pathway enriched with differential metabolites. (B) The lipid and amino acid metabolic pathways regulated by BBBP in mice induced by HFD.
3.5 BBBP restored the overall structure and composition of gut microbiota in mice induced by HFD
As gut microbiota played a vital role in NAFLD progression, we speculated that the anti-NAFLD efficacy of BBBP oral gavage might be related to gut microbiota in mice. Therefore, fecal 16S rRNA sequencing was performed. As shown in Fig. 5A, BBBP significantly decreased the Sobs index (observed species) compared with the model group (P < 0.05). The Simpson index reflected gut microbiota diversity. Compared with normal mice, HFD-fed mice showed a significant decrease in the Simpson index (P < 0.05), while mice treated with BBBP showed an increase in the Simpson index. The community richness was determined by the Ace and Chao index. The significantly lower Ace and Chao index were observed in the comparison between the model group and the BBBPH group (P < 0.05). We also observed that the model mice showed significantly different gut microbiota profiles from the normal mice. However, after BBBP treatment, the gut microbiota profiles of the HFD-fed mice were adjusted to a normal state (Fig. 5B-D), suggesting that BBBP treatment regulated gut microbiota structure in mice. The circos and ternary analysis confirmed that the richness of Lactobacillus, a dominant bacterium, was significantly enhanced after BBBP treatment (Fig. 5E, F).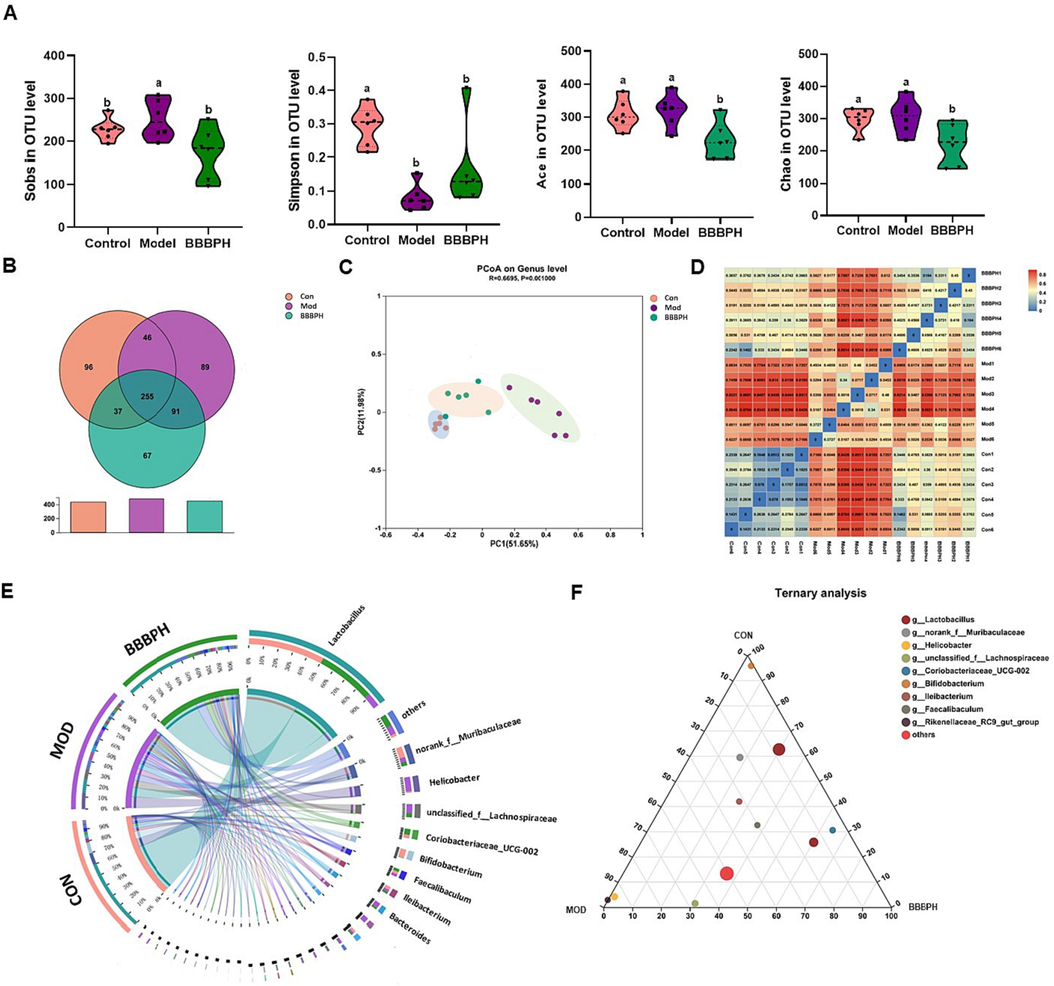
Overall gut microbiota analysis. (A) The community α-diversity analysis (Sobs, Simpson, Ace and Chao index). (B) Venn analysis based on the OUT number. (C) PCoA analysis at OTU level. (D) Heatmap of sample correlation. (E) Community circos plot of the major bacteria at the genus level. (F) Ternary analysis based on the genus level (n = 6 per group). Different letters (a, b) represent statistically significant differences among groups (P < 0.05).
3.6 BBBP regulated imbalance of gut microbiota taxonomic composition
At the phylum level (Fig. 6A), Firmicutes, Bacteroidota and Actinobacteriota were the main bacteria in the control group mice (>90%). The remarkable reduction of Firmicutes and Actinobacteriota and elevation of Bacteroidota were observed in the HFD-fed mice when compared with those in the control group (P < 0.05). BBBPH supplementation obviously increased the relative abundance of Firmicutes and Actinobacteriota and decreased the relative abundance of Bacteroidota (P < 0.05). We further identified the specific bacterial genus that responded to HFD feeding and BBBP supplementation. As shown in Fig. 6B, C, compared with the control group, the richness of Lactobacillus, Helicobacter, unclassified_f__Lachnospiraceae, Coriobacteriaceae_UCG-002, Bifidobacterium, Bacteroides, Rikenellaceae_RC9_gut_group, Desulfovibrio, Colidextribacter and Lactococcus were significantly changed in the model group (P < 0.05). After BBBP treatment, the relative abundance of these microorganisms was restored. Among them, the abundance of Lactobacillus was decreased by approximately 6-fold (P < 0.05) in the model group compared with the control group. BBBP treatment increased approximately 4-fold of its richness and reached a significant level (P < 0.05). These above results indicated that HFD greatly perturbed the mouse gut microbiota, in which Lactobacillus was largely eliminated. And BBBP showed a protective effect on Lactobacillus.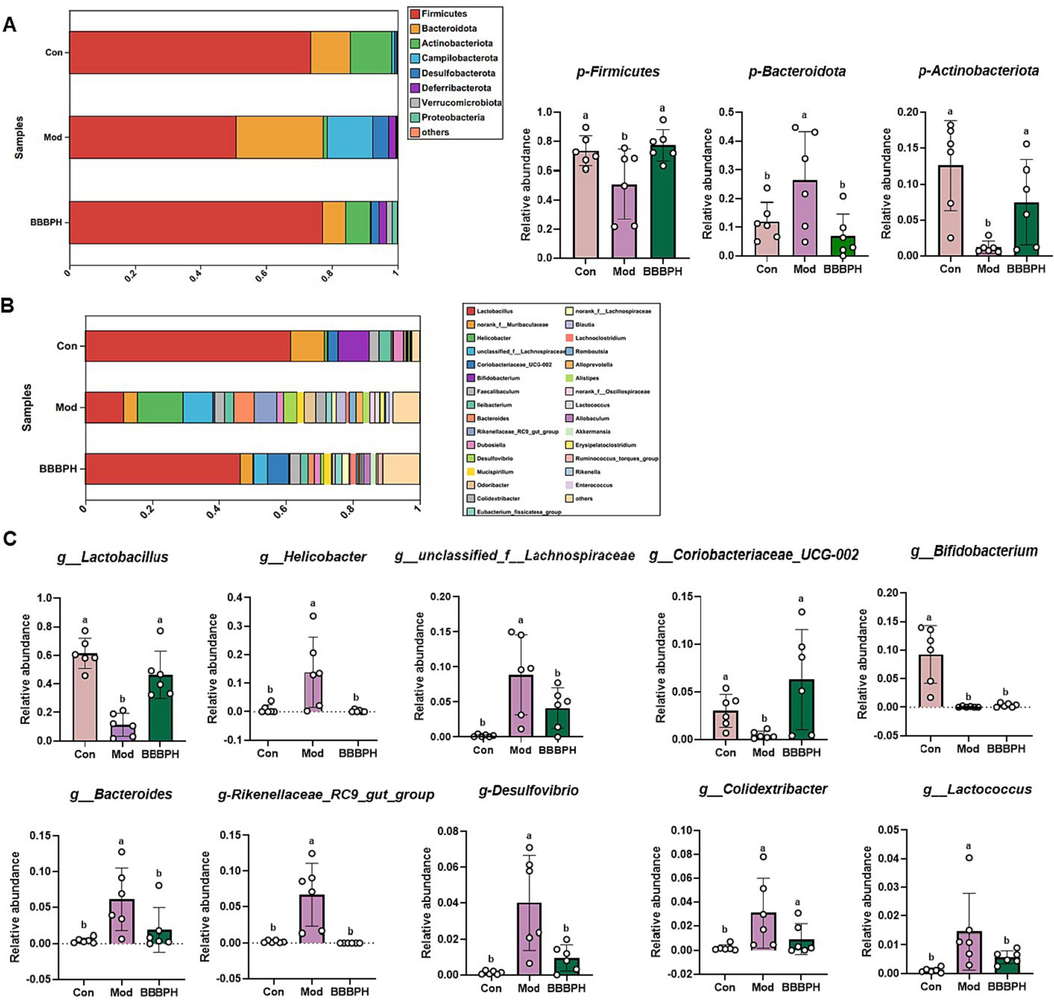
Taxonomy analysis of gut microbiota. (A) The structural composition of the gut microbiota in each group at the phylum level. (B-C) The structural composition of the gut microbiota in each group at the genus level. n = 6 per group. Different letters (a, b) represent statistically significant differences among groups (P < 0.05).
3.7 Correlation analysis of differential metabolites, gut microbiota, and pharmacodynamic indicators
The above results indicated that BBBP alleviated the gut dysbiosis imbalance and metabolic disorder caused by HFD. Gut microbiota and its metabolites were also associated with metabolic diseases such as hyperlipidemia (Liu et al., 2023). The visual network showed a strong correlation among gut microbiota, metabolites, and lipid index (Fig. 7A). To investigate the potential role of Lactobacillus in the anti-NAFLD mechanism of BBBP, Spearman correlation analysis was performed (Fig. 7B). Lactobacillus showed a significant correlation with serum TG, TC, LDL-C and HDL-C levels (P < 0.05). Spearman correlation analysis also demonstrated the positive correlations of Lactobacillus with citrulline, L-glutamine, L-glutamic acid, et al. and the inverse correlations of Lactobacillus with arachidonic acid, LysoPC(16:0/0:0), LysoPC(17:0/0:0), et al (Fig. 7C). Further scatter plots showed that Lactobacillus abundance was negatively associated with serum TG, TC, LDL-C, HDL-C levels and positively associated with serum GABA level (Fig. 7D).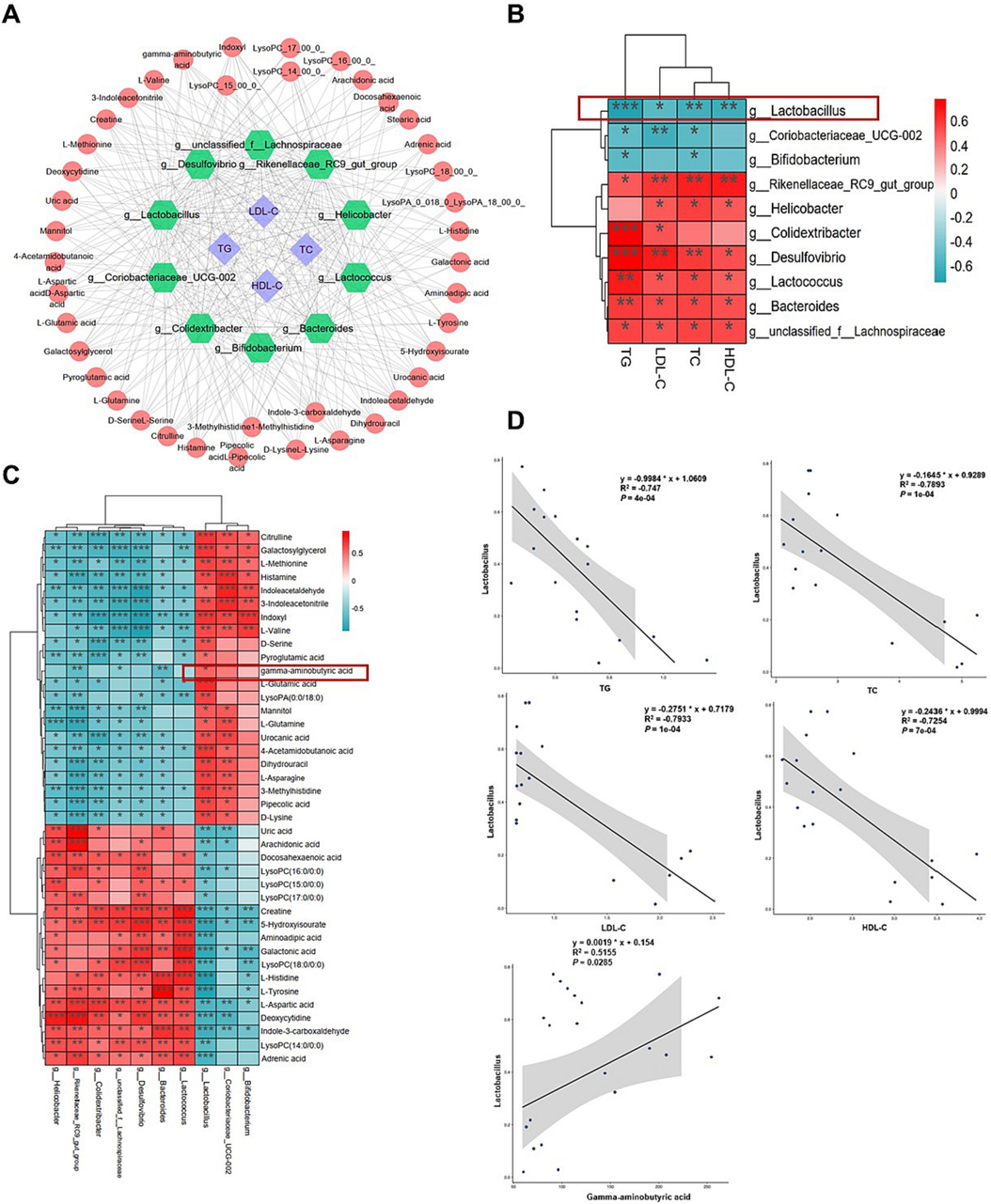
Correlation analysis. (A) Spearman correlation between lipid index, gut microbiota and serum metabolites. (B) Spearman correlation between lipid index and the differential gut microbiota. (C) Spearman correlation between the differential gut microbiota and metabolites. (D) Scatter plots of Lactobacillus with serum TG, TC, LDL-C, HDL-C and GABA levels. *P < 0.05, **P < 0.01, ***P < 0.001.
3.8 GABA inhibited the lipid accumulation in L02 cells
The abundance of probiotics Lactobacillus and GABA levels were significantly elevated after BBBP treatment. Several lines of evidence have shown that GABA which can produced by Lactobacillus has a significant impact on systemic metabolic health (Sarasa et al., 2020). Therefore, GABA was cultivated to evaluate its health-promoting effect in vitro. The viability of L02 cells treated with GABA showed a slight decline at the high dose of GABA (100 μmol/L) (Fig. 8A). As shown in Fig. 8B-F, lipid accumulation in L02 cells treated with PA and OA was significantly higher than that in the control group (P < 0.05). GABA treatment decreased the lipid accumulation in steatosis L02 cells.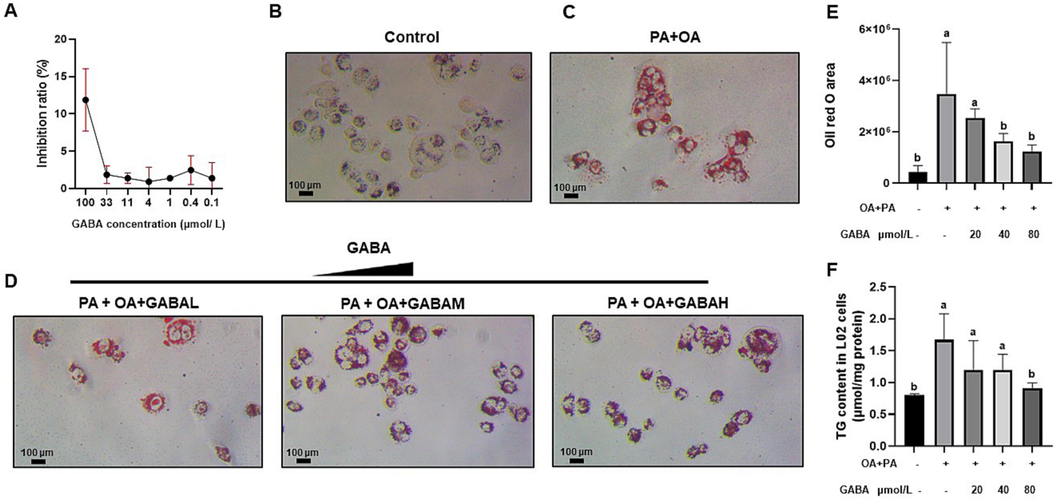
BBBP reduced lipid droplet accumulation in L02 cells. (A) The effect of BBBP treatment on L02 cell viability. (B–E) Oil Red O staining of L02 cells in the different groups. (F) Intracellular triglyceride content in L02 cells. Different letters (a, b) represent statistically significant differences among groups (P < 0.05).
4 Discussion
As a critical metabolic syndrome, NAFLD is characterized by aberrant accumulation of lipids in hepatocytes. The high prevalence of NAFLD is linked to overnutrition and consumption of highly processed foods. Obesogenic diets similar to modern human consumption could induce fatty liver that replicated the pathological features of humans (Eng and Estall, 2021). Thus, in this study, HFD was fed to mice for 18 weeks to establish a NAFLD model. NAFLD mice presented a distinct obese phenotype, abnormal liver pathology and serum biochemical indicators. Intriguingly, these changes were significantly reversed by BBBP supplementation. Chemical research revealed that BBBP mainly contained bile acids which played critical roles in regulating lipid and glucose homeostasis by activating bile acids receptors such as cell surface and nuclear receptors (Fiorucci et al., 2020; Lin and Kohli, 2018). As expected, the results of our research indicated that bile acid receptors in the liver were activated after BBBP treatment, as indicated by elevated mRNA levels of S1pr2, Nr1h4 and Nr1i2. Meanwhile, the high doses of BBBP appeared to be stronger than pioglitazone, a PPARγ agonist, in resisting NAFLD. Therefore, BBBP might be a candidate drug for NAFLD treatment.
Furthermore, we systemically evaluated the specific effects of BBBP on the metabolic spectrum of HFD-fed mice. The metabolic profiles were remarkably different among mice in the control, model and BBBPH group. It was also found that the differential metabolites were mainly involved in lipid metabolism and amino acid metabolism. Given that bile acids were well known for their role in fat absorption (Chow et al., 2017), it was not surprising that BBBP exhibited the ability to regulate lipid metabolism. Besides, there was increasing evidence that serum concentrations of amino acids were altered in NAFLD patients and animal models (Gaggini et al., 2018; Tricò et al., 2021). In the present study, serum levels of valine, serine, glutamine, et al. were altered in HFD-fed mice and then BBBP treatment reversed the abnormal changes in the aforementioned amino acids. As one of the BCAAs, valine could modulate liver glucose metabolism while this action could be disrupted by HFD-induced insulin resistance (Arrieta-Cruz et al., 2016). It has been reported that the reduced levels of serine and glutamine involved in glutathione synthesis were closely associated with the development of fatty liver disease (Sim et al., 2020). Serine and glutamine supplementation decreased hepatic steatosis in humans and reduced the degree of oxidative stress in mice, respectively (Mardinoglu et al., 2017; Lin et al., 2014). These findings were not astonishing because most proteins and amino acids were metabolized in the liver. Various drugs such as BPTES (glutaminase inhibitor), ornithine phenylacetate (ammonia-lowering drug) and et al. targeting abnormal amino acid metabolism are being tested for NAFLD (Rojas et al., 2022; Du et al., 2018). Our results suggested that BBBP coordinately modulated both lipid metabolism and amino acid metabolism and subsequently protected mice from NAFLD.
Accumulating evidence revealed that the alteration of gut microbiota composition could disturb the hepatic carbohydrate and lipid metabolism in the liver, thus resulting in the development of NAFLD (Ni et al., 2020; Aron-Wisnewsky et al., 2020b). Given that bile acids impacted microbiome community structure and function, we speculated that BBBP administered orally might exert therapeutic effects by acting on the gut microbiome in mice. Studies have assessed the effect of HFD on the growth of different gut microbiota, with different results depending on the microbe (Bibbò et al., 2016). For example, the prevalence of Bacteroides was positively correlated with the HFD, while Firmicutes showed an opposite correlation (Bibbò et al., 2016). Consistent with our results, 18 weeks of HFD feeding caused a significant increase in the abundance of Bacteroides and BBBP could effectively restore the abundance of Bacteroides altered by HFD. Furthermore, we found that the levels of 10 genera deviated from the normal state and the abundance was reversed after BBBP treatment. Of note, Lactobacillus with the highest abundance had the most significant changes. Although bile acids exhibited antimicrobial activity by disrupting bacterial cell membranes (Kurdi et al., 2006), our results suggested that BBBP could promote the growth of Lactobacillus. As probiotics, the promising role of Lactobacillus strains in improving NAFLD has been confirmed. For example, Lactobacillus salivarius SNK-6 has been found to regulate liver lipid metabolism via the miR-130a-5p/MBOAT2 pathway (Vallianou et al., 2021). Correlation analysis of this study found that the changes of Lactobacillus were negatively correlated with the serum TG, TC, LDL-C and HDL-C levels. Thus, the above results suggested that HFD-induced microbial changes were recovered by BBBP treatment and there were certain bacteria such as Lactobacillus specifically in response to BBBP.
The gut microbiota dysbiosis led to microbiome-derived metabolite alteration which was related to the pathogenesis of NAFLD (Hoozemans et al., 2021). Indeed, we found a strong correlation between Lactobacillus and serum differential metabolites. Similar results from another study further confirmed this notion, showing that there was a causal relationship between blood metabolites and gut microbiome (Liu et al., 2022). Among the differential metabolites in serum, GABA which was significantly positively correlated with Lactobacillus caught our attention. GABA, a non-protein amino acid, was produced primarily from the irreversible α-decarboxylation of L-glutamate by the enzyme glutamate decarboxylase (Sarasa et al., 2020; Barrett et al., 2012). As an inhibitory neurotransmitter in the central nervous system of animals, it was found predominantly in the brain and also possessed lipid-lowering effects (Kim and Yoon, 2023). Moreover, intestinally derived strains of Lactobacillus could also produce GABA (Tette et al., 2022; Wu and Shah, 2018; Cui et al., 2020; Banerjee et al., 2021; Barrett et al., 2012). Thus, our study hypothesized that the effect of BBBP on lipid metabolism could be, at least partially, attributed to the modulation of Lactobacillus and its metabolites such as GABA. And this hypothesis was confirmed by the effect of GABA on reducing lipid accumulation in L02 cells. Our results suggested the intracellular TG contents and lipid deposition were decreased after GABA treatment. Evidence has shown GABA could regulate the mRNA expression of TG breakdown enzymes such as adipose triglyceride lipase and lipoprotein lipase (Xie et al., 2014). Besides, recent studies suggested that the effect of GABA in obesity rodent models induced by HFD was attributed to the promotion of lipolysis and fatty acid β-oxidation (Xie et al., 2014; Chen et al., 2022). Together, the possible mechanism of BBBP treatment for NAFLD was shown in Fig. 9. However, whether other metabolites and bacteria have substantial roles in BBBP against NAFLD remains to be established. In summary, these findings provided novel evidence that BBBP might serve as a new candidate for the treatment of NAFLD.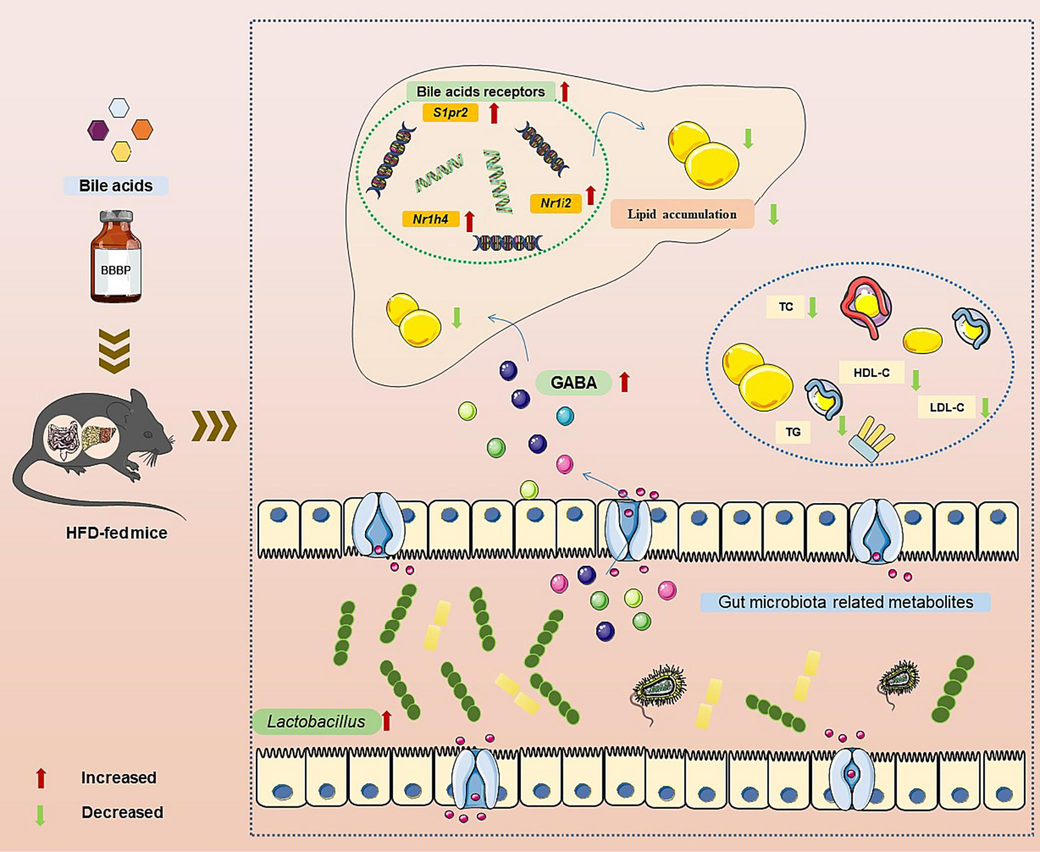
The possible mechanism of BBBP preventing NAFLD from the perspectives of gut microbiota and metabolomics.
5 Conclusion
The current study systemically demonstrated that BBBP significantly ameliorated obese phenotype and hepatic steatosis in NAFLD mice induced by HFD. Multi-omics results indicated that BBBP could alleviate lipid accumulation by activating bile acid receptors and broadly regulating gut microbiota and its metabolites, such as GABA. Taken together, our research indicated that BBBP holds great potential as a cure for NAFLD induced by HFD.
Funding
The work was supported by the CACMS Innovation Fund (CI2021A04502) and the National Administration of Traditional Chinese Medicine Young Qihuang Scholar Project, China.
CRediT authorship contribution statement
Shan Jiang: Software, Methodology, Data curation, Writing – original draft, Investigation, Writing – review & editing. Xiaolu Wei: Software, Investigation. Hongjie Wang: Investigation. Yanyan Zhou: Investigation. Lihua Chen: Investigation. Lianmei Wang: Investigation. Feng Wang: Investigation. Qilong Wu: Investigation. Nan Si: Conceptualization, Supervision, Resources, Funding acquisition. Baolin Bian: Conceptualization, Writing – review & editing, Supervision, Resources, Funding acquisition. Haiyu Zhao: Conceptualization, Writing – review & editing, Supervision, Resources, Funding acquisition.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Structure, functions, and diversity of the healthy human microbiome. Prog. Mol. Biol. Transl. Sci.. 2022;191(1):53-82.
- [Google Scholar]
- Gut microbiota and human NAFLD: disentangling microbial signatures from metabolic disorders. Nat. Rev. Gastroenterol. Hepatol.. 2020;17(5):279-297.
- [Google Scholar]
- Nonalcoholic fatty liver disease: modulating gut microbiota to improve severity? Gastroenterology.. 2020;158(7):1881-1898.
- [Google Scholar]
- Suppression of endogenous glucose production by isoleucine and valine and impact of diet composition. Nutrients.. 2016;8(2):79.
- [Google Scholar]
- Transcriptomics reveal different metabolic strategies for acid resistance and gamma-aminobutyric acid (GABA) production in select Levilactobacillus brevis strains. Microb. Cell Fact.. 2021;20(1):173.
- [Google Scholar]
- γ-Aminobutyric acid production by culturable bacteria from the human intestine. J. Appl. Microbiol.. 2012;113(2):411-417.
- [Google Scholar]
- The role of diet on gut microbiota composition. Eur. Rev. Med. Pharmacol. Sci.. 2016;20(22):4742-4749.
- [Google Scholar]
- The severity of nonalcoholic fatty liver disease is associated with gut dysbiosis and shift in the metabolic function of the gut microbiota. Hepatology.. 2016;63(3):764-775.
- [Google Scholar]
- Cultured bear bile powder ameliorates acute liver injury in cholestatic mice via inhibition of hepatic inflammation and apoptosis. J. Ethnopharmacol.. 2022;284:114829
- [Google Scholar]
- Dietary γ-aminobutyric acid supplementation inhibits high-fat diet-induced hepatic steatosis via modulating gut microbiota in broilers. Microorganisms.. 2022;10(7):1281.
- [Google Scholar]
- Fecal 16S rRNA sequencing and multi-compartment metabolomics revealed gut microbiota and metabolites interactions in APP/PS1 mice. Comput. Biol. Med.. 2022;151(Pt A):106312
- [Google Scholar]
- The role of bile acids in nonalcoholic fatty liver disease and nonalcoholic steatohepatitis. Mol. Aspects Med.. 2017;56:34-44.
- [Google Scholar]
- The gut microbiota and its relationship to diet and obesity: new insights. Gut. Microbes.. 2012;3(3):186-202.
- [Google Scholar]
- Production of gamma-aminobutyric acid from lactic acid bacteria: a systematic review. Int. J. Mol. Sci.. 2020;21(3):995.
- [Google Scholar]
- Hedgehog-YAP signaling pathway regulates glutaminolysis to control activation of hepatic stellate cells. Gastroenterology.. 2018;154(5):1465-1479.e13.
- [Google Scholar]
- Diet-induced models of non-alcoholic fatty liver disease: food for thought on sugar, fat, and cholesterol. Cells.. 2021;10(7):1805.
- [Google Scholar]
- Gut microbiota in human metabolic health and disease. Nat. Rev. Microbiol.. 2021;19(1):55-71.
- [Google Scholar]
- Bile acid modulators for the treatment of nonalcoholic steatohepatitis (NASH) Exp. Opin. Invest. Drugs.. 2020;29(6):623-632.
- [Google Scholar]
- The effect of normalization of plasma amino acids on hepatic encephalopathy in man. Surgery.. 1976;80(1):77-91.
- [Google Scholar]
- Altered amino acid concentrations in NAFLD: impact of obesity and insulin resistance. Hepatology.. 2018;67(1):145-158.
- [Google Scholar]
- Gut Microbiome and metabolites in patients with NAFLD and after bariatric surgery: a comprehensive review. Metabolites.. 2021;11(6):353.
- [Google Scholar]
- Alteration of fecal microbiome and metabolome by mung bean coat improves diet-induced non-alcoholic fatty liver disease in mice. Food Sci. Hum. Well.. 2022;11(5):1259-1272.
- [Google Scholar]
- Intestinal microbiome and NAFLD: molecular insights and therapeutic perspectives. J. Gastroenterol.. 2020;55(2):142-158.
- [Google Scholar]
- Co-immobilised 7α- and 7β-HSDH as recyclable biocatalyst: high-performance production of TUDCA from waste chicken bile. RSC Adv.. 2018;8(60):34192-34201.
- [Google Scholar]
- Biotransformed bear bile powder ameliorates diet-induced nonalcoholic steatohepatitis in mice through modulating arginine biosynthesis via FXR/PXR-PI3K-AKT-NOS3 axis. Biomed. Pharmacother.. 2023;168:115640
- [Google Scholar]
- The immediate adverse drug reactions induced by ShenMai Injection are mediated by thymus-derived T cells and associated with RhoA/ROCK signaling pathway. Front Immunol.. 2023;14:1135701.
- [Google Scholar]
- Dysbiosis gut microbiota associated with inflammation and impaired mucosal immune function in intestine of humans with non-alcoholic fatty liver disease. Sci. Rep.. 2015;5:8096.
- [Google Scholar]
- Targeting mitochondrial ROS-mediated ferroptosis by quercetin alleviates high-fat diet-induced hepatic lipotoxicity. Front Pharmacol.. 2022;13:876550
- [Google Scholar]
- Gamma-aminobutyric acid signaling in damage response, metabolism, and disease. Int. J. Mol. Sci.. 2023;24(5):4584.
- [Google Scholar]
- Mechanism of growth inhibition by free bile acids in lactobacilli and bifidobacteria. J. Bacteriol.. 2006;188(5):1979-1986.
- [Google Scholar]
- Contributions of metabolic dysregulation and inflammation to nonalcoholic steatohepatitis, hepatic fibrosis, and cancer. Curr. Opin. Oncol.. 2014;26(1):100-107.
- [Google Scholar]
- Integrative lipidomic and transcriptomic study unravels the therapeutic effects of saikosaponins A and D on non-alcoholic fatty liver disease. Acta Pharm. Sin. B.. 2021;11(11):3527-3541.
- [Google Scholar]
- Effects of glutamine on oxidative stress and nuclear factor-κB expression in the livers of rats with nonalcoholic fatty liver disease. Exp. Ther. Med.. 2014;7(2):365-370.
- [Google Scholar]
- Bile acid metabolism and signaling: potential therapeutic target for nonalcoholic fatty liver disease. Clin. Transl. Gastroenterol.. 2018;9(6):164.
- [Google Scholar]
- Lotus seed resistant starch and sodium lactate regulate small intestinal microflora and metabolite to reduce blood lipid. Int. J. Biol. Macromol.. 2023;233:123553
- [Google Scholar]
- Mendelian randomization analyses support causal relationships between blood metabolites and the gut microbiome. Nat. Genet.. 2022;54(1):52-61.
- [Google Scholar]
- Branched-chain amino acids in metabolic signalling and insulin resistance. Nat. Rev. Endocrinol.. 2014;10(12):723-736.
- [Google Scholar]
- Personal model-assisted identification of NAD+ and glutathione metabolism as intervention target in NAFLD. Mol. Syst. Biol.. 2017;13(3):916.
- [Google Scholar]
- Metabolomics and lipidomics in NAFLD: biomarkers and non-invasive diagnostic tests. Nat. Rev. Gastroenterol. Hepatol.. 2021;18(12):835-856.
- [Google Scholar]
- Amino acid imbalance in patients with chronic liver diseases. Hepatol. Res.. 2010;40(4):393-398.
- [Google Scholar]
- Obesity and female gender increase breath ethanol concentration: potential implications for the pathogenesis of nonalcoholic steatohepatitis. Am. J. Gastroenterol.. 2001;96(4):1200-1204.
- [Google Scholar]
- The gut microbiota and its metabolites, novel targets for treating and preventing non-alcoholic fatty liver disease. Mol. Nutr. Food Res.. 2020;64(17):e2000375.
- [Google Scholar]
- Resistant starch decreases intrahepatic triglycerides in patients with NAFLD via gut microbiome alterations. Cell Metab.. 2023;35(9):1530-1547.
- [Google Scholar]
- Severity of liver injury and atherogenic lipid profile in children with nonalcoholic fatty liver disease. Pediatr. Res.. 2010;67(6):665-670.
- [Google Scholar]
- The role of omics in the pathophysiology, diagnosis and treatment of non-alcoholic fatty liver disease. Metabolism.. 2020;111S:154320
- [Google Scholar]
- Eubiotic properties of rifaximin: disruption of the traditional concepts in gut microbiota modulation. World J. Gastroenterol.. 2017;23(25):4491-4499.
- [Google Scholar]
- Recent advances in alcoholic liver disease I. Role of intestinal permeability and endotoxemia in alcoholic liver disease. Am. J. Physiol. Gastrointest. Liver Physiol.. 2004;286(6):G881-G884.
- [Google Scholar]
- Critical review of 16S rRNA gene sequencing workflow in microbiome studies: from primer selection to advanced data analysis. Mol. Oral. Microbiol.. 2023;38(5):347-399.
- [Google Scholar]
- Glutaminolysis-ammonia-urea cycle axis, non-alcoholic fatty liver disease progression and development of novel therapies. J. Clin. Transl. Hepatol.. 2022;10(2):356-362.
- [Google Scholar]
- Environment dominates over host genetics in shaping human gut microbiota. Nature.. 2018;555(7695):210-215.
- [Google Scholar]
- A brief review on the non-protein amino acid, Gamma-amino Butyric Acid (GABA): its production and role in microbes. Curr. Microbiol.. 2020;77(4):534-544.
- [Google Scholar]
- Untargeted metabolomics strategies-challenges and emerging directions. J. Am. Soc. Mass. Spectrom.. 2016;27(12):1897-1905.
- [Google Scholar]
- Structural insights into amelioration effects of quercetin and its glycoside derivatives on NAFLD in mice by modulating the gut microbiota and host metabolism. J. Agric. Food Chem.. 2022;70(46):14732-14743.
- [Google Scholar]
- Downregulation of PHGDH expression and hepatic serine level contribute to the development of fatty liver disease. Metabolism.. 2020;102:154000
- [Google Scholar]
- Mortality in biopsy-confirmed nonalcoholic fatty liver disease: results from a nationwide cohort. Gut.. 2021;70:1375-1382.
- [Google Scholar]
- Discovery of tauroursodeoxycholic acid biotransformation enzymes from the gut microbiome of black bears using metagenomics. Sci. Rep.. 2017;7:45495.
- [Google Scholar]
- Therapeutic mechanisms of bile acids and nor-ursodeoxycholic acid in non-alcoholic fatty liver disease. Dig. Dis.. 2017;35(3):282-287.
- [Google Scholar]
- Therapeutic anti-depressant potential of microbial GABA produced by lactobacillus rhamnosus strains for GABAergic signaling restoration and inhibition of addiction-induced HPA axis hyperactivity. Curr. Issues Mol. Biol.. 2022;44(4):1434-1451.
- [Google Scholar]
- Protein and amino acids in nonalcoholic fatty liver disease. Curr. Opin. Clin. Nutr. Metab. Care.. 2021;24(1):96-101.
- [Google Scholar]
- The association of the steatosis severity, NAFLD fibrosis score and FIB-4 index with atherogenic dyslipidaemia in adult patients with NAFLD: A cross-sectional study. Int. J. Clin. Pract.. 2021;75(6):e14131.
- [Google Scholar]
- Understanding the role of the gut microbiome and microbial metabolites in non-alcoholic fatty liver disease: current evidence and perspectives. Biomolecules.. 2021;12(1):56.
- [Google Scholar]
- Hepatic regulator of G protein signaling 5 ameliorates nonalcoholic fatty liver disease by suppressing transforming growth factor beta-activated kinase 1-c-Jun-N-terminal kinase/p38 signaling. Hepatology.. 2021;73(1):104-125.
- [Google Scholar]
- Metabolomics research on the hepatoprotective effect of cultured bear bile powder in α-naphthylisothiocyanate-induced cholestatic mice. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci.. 2020;1153:122269
- [Google Scholar]
- Restoration of GABA production machinery in Lactobacillus brevis by accessible carbohydrates, anaerobiosis and early acidification. Food Microbiol.. 2018;69:151-158.
- [Google Scholar]
- Small intestinal bacteria overgrowth decreases small intestinal motility in the NASH rats. World J. Gastroenterol.. 2008;14(2):313-317.
- [Google Scholar]
- Gamma-aminobutyric acid improves oxidative stress and function of the thyroid in high-fat diet fed mice. J. Funct. Foods.. 2014;8(1):76-86.
- [Google Scholar]
- Targeted therapeutics and novel signaling pathways in non-alcohol-associated fatty liver/steatohepatitis (NAFL/NASH) Signal Transduct. Target Ther.. 2022;7(1):287.
- [Google Scholar]
- Protective effect of cultured bear bile powder against dimethylnitrosamine-induced hepatic fibrosis in rats. Biomed. Pharmacother.. 2019;112:108701
- [Google Scholar]
Appendix A
Supplementary material
Supplementary data to this article can be found online at https://doi.org/10.1016/j.arabjc.2023.105578.
Appendix A
Supplementary material
The following are the Supplementary data to this article:Supplementary Data 1
Supplementary Data 1







