Translate this page into:
Chemical diversity and biological activities of marine-derived sulphur containing alkaloids: A comprehensive update
⁎Corresponding author at: School of Pharmacy, Shaanxi University of Chinese Medicine, Xianyang, Shaanxi Provance of China 712046. zhangnatprod@163.com (Dongdong Zhang)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
Objectives
The ocean is a huge ecosystem with diverse marine life. Scientists have found a large number of natural products with unique structural features and excellent biological activity from these organisms. Marine-derived sulphur-containing alkaloids are a significant family of natural products with diverse structures and bioactivities. In this paper, the chemical and biological diversity of 972 sulfur-containing alkaloids derived from marine organisms reported from 1982 to 2022 were reviewed, and the structure–activity relationship was briefly analyzed, in order to provide reference for the discovery, synthesis, biological activity research and drug development of such compounds.
Key findings
A total of 972 marine-derived sulphur-containing alkaloids have been collected. Among them, 80.36% of sulphur-containing alkaloids are from marine sponges, fungi, tunicates and bacteria. Moreover, cytotoxicity is their most significant property, About 1/3 sulphur-containing organisms are reported to be cytotoxic. Aming them, discorhabdins, curacins, tanjungides, leptosins, and latrunculins exhibit better cytotoxicity. In addition, the structure–activity relationships of the cytotoxicity of these compounds have been summarized for further investigation.
Summary
In this paper, the chemical and bioactivity diversity of marine-derived sulphur-containing alkaloids were reviewed, which are a significant family of natural products with diverse structures and bioactivities. 972 sulphur-containing alkaloids were obtained from marine algae, sponges, cnidarians, tunicates, echinoderms, molluscs, bryozoans, dinoflagellates, cyanobacteria, bacteria and fungi, which possessed a wide spectrum of pharmacology including cytotoxicity, antibacterial, antifungal, antimitotic, antiviral, and other activities.
Keywords
Sulphur-containing alkaloids
Marine organisms
Chemical diversity
Bioactivity diversity
Cytotoxicity
1 Introduction
Natural medicines found from terrestrial plant and animal resources have been widely used in the clinical treatment of various diseases. However, with continuous exploitation, it has become increasingly difficult to develop drugs from terrestrial resources. Therefore, researchers have started to work on finding new sources of drugs from the ocean. (Lu et al., 2021).
The oceans are extremely rich in biological resources, including large numbers of fish, shrimps, crabs and many lower species such as molluscs, corals and seaweeds. Together, these organisms maintain the balance and stability of the marine ecosystem. (Seipp et al., 2021). It is worth noting that the marine environment has extreme living conditions such as high pressure, high salinity, hypoxia and low light. As a result, marine organisms often produce unique and active secondary metabolites, giving them an edge in the competition for limited resources. (Shang et al., 2018).
Pharmacological studies have shown that marine natural products (MNPs) have great potential in the treatment of various diseases. These results have stimulated research and development of marine organisms. After decades of in-depth studies, a large number of active ingredients have been found. (Lu et al., 2021). To date, 11 marine-derived drugs have successfully reached the market. For example, cytarabine (Cytosar-U®), ET-743 (Yondelis®), eribulin mesylate (Halaven®) and the antibody-drug conjugates (ADCs) brentuximab (Adcetris®) and polatumumab (Polivy®) have been used to treat cancer. Lovaza®, Vascepa® and Epanova® are used to treat hypertriglyceridemia. (Liang et al., 2019). In addition, 23 compounds are in various stages of clinical development. For example, the combination therapy of prambulin and docetaxel is currently in phase III clinical trials for the treatment of non-small cell lung cancer and the prevention of chemotherapy-induced neutropenia. lurbinectedin is in phase II/III clinical trials for the treatment of BRCA1/2-mutated breast cancer and small cell lung cancer. In addition, tetrodotoxin (Tectin), an alkaloid derived from the tetrodotoxin liver, is in phase III clinical trials for the treatment of severe pain. (Jiménez, 2018).
Among the many MNPs, sulphur-containing alkaloids are important natural marine products with good bioactivity. As shown in Fig. 1, about 972 sulphur-containing alkaloids have been isolated from marine organisms from 1982. (marine fungi have become an important source of sulphur-containing alkaloids in recent 10 years, Fig. 2). The sulphur-containing alkaloids displayed a variety of biological activities such as cytotoxicity, anti-proliferation, anti-virus, anti-inflammatory and antioxidant, as listed in Table 14 (Supporting material) (Berman et al., 1999; Du et al., 2012; Goey et al., 2016; Guzmán et al., 2009; Harris et al., 2018; Jeong et al., 2003; Johnson et al., 1999; Jun et al., 2007; Lam et al., 2020; Lee et al., 2016; Li et al., 2021; Machihara and Namba, 2020; Merrouche et al., 2020; Morgan et al., 2010, 2015; Oluwabusola et al., 2022; Reid et al., 1996; Salam et al., 2013; Susana and Salvador-Reyes, 2022; Wang et al., 2022; Zhao et al., 2019). Of them, ecteinascidin 743 (yondelis) has become the first modern marine drug to treat advanced soft tissue tumors (Menchaca et al., 2003). Thiomarinols have excellent antibacterial activity and can even be effective against methicillin-resistant Staphylococcus aureus (MRSA) (Shiozawa et al., 1995). Somocystinamide A (601) shows strong cytotoxicity to Jurkat and CEM cells with IC50 values of 3 and 14 nM, respectively (Wrasidlo et al., 2008).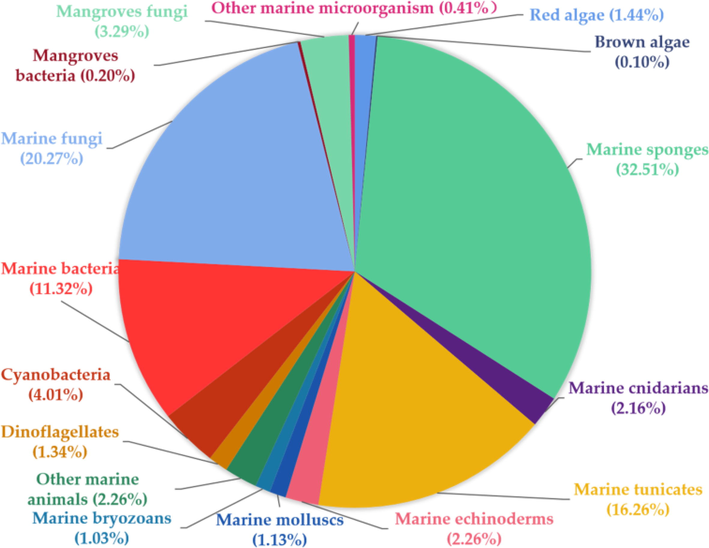
The percentage of sulphur-containing alkaloids from diverse marine organisms.
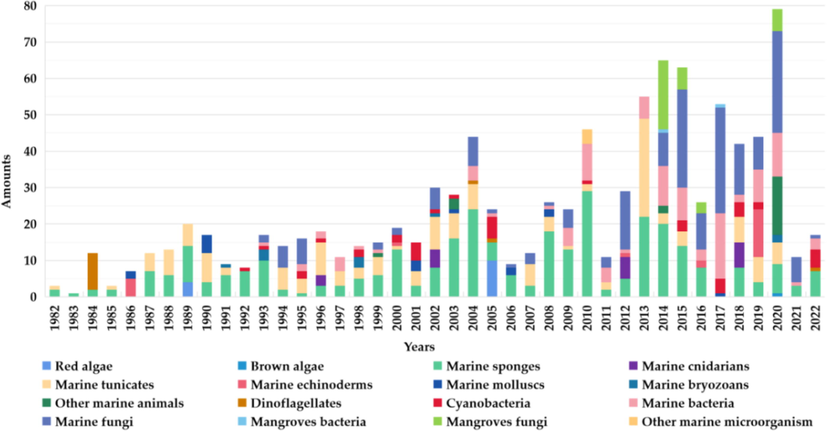
All sulphur-containing alkaloids by source/year, n = 972.
In this study, we comprehensively summarized the chemistry and biological activity of sulphur-containing alkaloids in 459 publications and provided a brief analysis of the active conformational relationships between their structure and biological activity. This will help us to provide a reference for the discovery, synthesis and biological activity studies of this class of compounds and for drug discovery and development.
1.1 Search strategy
Comprehensive research and analysis of previously published literature were conducted for studies on the chemical and biological diversity of the marine-derived sulphur-containing alkaloids. The search was conducted using databases such as Sciencedirect, SciFinder, Medline PubMed, Google Scholar, Baidu Scholar, and CNKI by using the keywords such as marine alkaloids, marine-derived sulphur-containing alkaloids, sulphur-containing alkaloids. Furthermore, part of the analyzed studies was got by a manual search of articles in the reference lists of the included studies. The PRISMA template for determining the list of articles is displayed in Fig. 3. The chemical structures were drawn using ChemDraw Professional 20.0.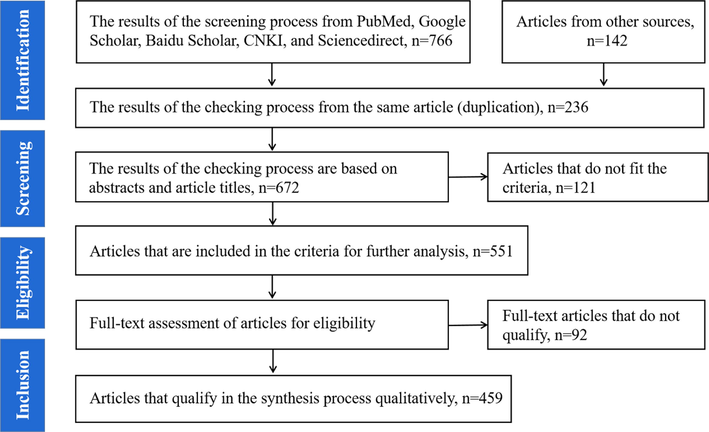
Research Data Search & Selection Flow.
1.2 Chemical diversity of Marine-Derived Sulphur-containing alkaloids
1.2.1 Marine algae
The photosynthesis of algae is an extremely important source of oxygen. At the same time, the organic matter they produce and the energy they accumulate are the basis for the survival and development of the entire marine biosphere. Therefore, marine algae are considered to be an important marine biological resource. Human exploitation of marine algal resources has a long history. In the early days, some seaweeds such as roundworms, kelp and nori were used as food. Later, seaweed was used as medicine, animal feed and fertiliser. With the development of seaweed resources, one of the most important uses of seaweed is the extraction of various seaweed extracts. For example, agar is widely used as a bacterial culture medium and carrageenan is widely used in the food industry.
Although abundant compounds were isolated from the marine algae by natural product chemists, only 15 sulphur-containing alkaloids (1–15) were reported from red algae and brown algae (Fig. 4 and Table 1). Of them, sulphur-containing alkaloids reported from the red algae are all reported from Laurencia brongniartii (Tanaka et al., 1989). In addition, it’s worth noting that these alkaloids are all indole alkaloids and compounds 4, 8–14 are special indole alkaloid dimers (El-Gamal et al., 2005).
Sulphur-containing alkaloids from marine algae.
No.
Compounds
Time
From
Location
Ref.
Marine algae
Red algae
1.
itomanindole A
1989
Laurencia brongniartii
Okinawa, Japan
(Tanaka et al., 1989)
2.
itomanindole B
1989
3.
4,6-dibromo-2-(methylthio)indole
1989
4.
3,3-bis(4,6-dibromo-t-methyIthio)indole
1989
5.
2-methylsulfinyl-3-methylthio-4,5,6-tribromoindole
2005
Ken-Ting National Park, South Taiwan
(El-Gamal et al., 2005)
6.
3-methylsulfinyl-2,4,6-tribromoindole
2005
7.
4,6-dibromo-2,3-di(methylsulfinyl)indole
2005
8.
3,3′-bis(2′-methylsulfinyl-2-methylthio-4,6,4′,6′-tetrabromo)indole
2005
9.
3,3-bis(4,6-dibromo-2-methylsulfinyl)indole
2005
10.
2,4,4′.6.6′-pentabromo-2′,3-bis(methylthio)-1,3′-bi-1H-indole
2005
Kikai Island, Japan
(Natsuki et al., 2005)
11.
2.4.4‘0.5′.6.6‘-hexabromo-2′,3-bis(methylthio)-1,3′-bi-1H-indole
2005
12.
2,4,4′,5,6,6′-hexabromno-2′,3-bis(methylthio)-1,3′-bi-1H-indole
2005
13.
2.4.4‘0.5.5′.6.6′-heptabromo-2′,3-bis(methylthio)-1,3′-bi-1H-indole
2005
14.
2.4.4′.6.6′-pentabromo-2′-methylthio-3,3′-bi-1H-indole
2005
Brown algae
15.
sargassulfamide A
2020
Sargassum naozhouense
Leizhou Peninsula, Guangdong, China
(Peng et al., 2020)
1.2.2 Marine fauna
1.2.2.1 Marine sponges
Marine sponges are the most primitive multicellular animal, which have been living in the ocean since 600 million years ago. They have developed to more than 10,000 species, accounting for 1/15 of the marine animal species. Sponges have been developed very early by ancient humans. Now they are extensive used in technology, medicine and daily life and have become an important resource for marine drug development. A total of 316 (16–331) sulphur-containing alkaloids were reported from the marine sponges (Fig. 5 and Table 2). These compounds isolated from marine sponges have various bioactivities such as antitumor, antifungal, antibacterial and enzyme inhibitory activities.
Sulphur-containing alkaloids from marine sponges.
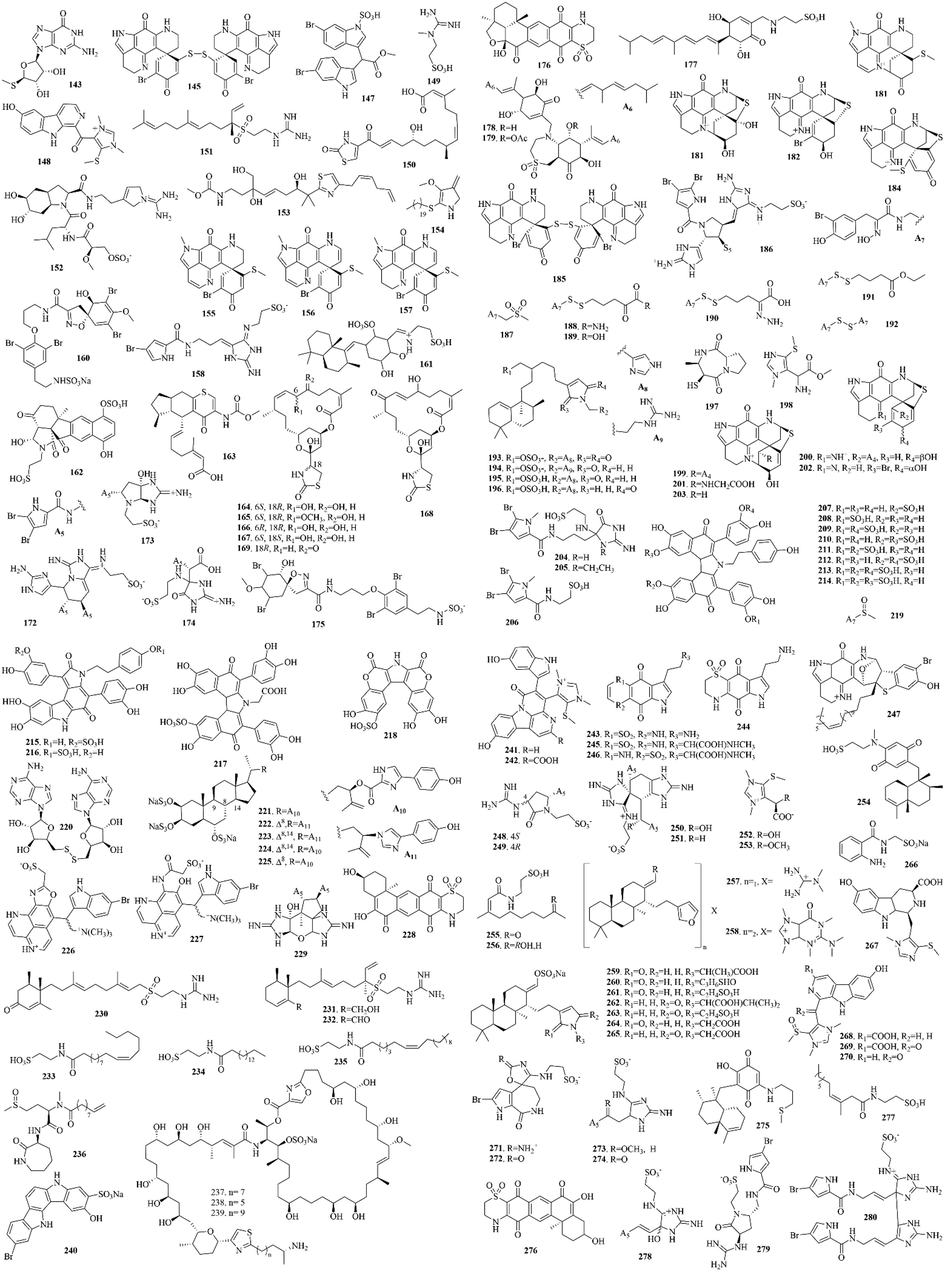
Sulphur-containing alkaloids from marine sponges.
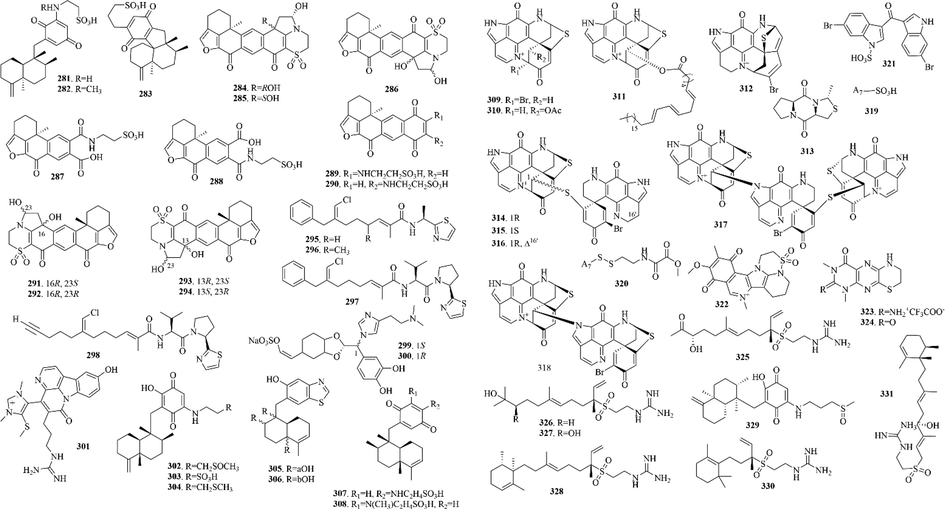
Sulphur-containing alkaloids from marine sponges.
No.
Compounds
Time
From
Location
Ref.
Marine fauna
Marine sponges
16
latrunculin A
1982
Latruncularia magnifica
Red Sea
(Spector et al., 1983)
17
latrunculin B
1982
18
latrunculin C
1985
(Kashman et al., 1985)
19
latrunculin D
1985
20
agelasidine B
1984
Agefas nakamurai
Okinawa, Japan
(Nakamura et al., 1985)
21
agelasidine C
1984
22
prianosin A
1987
Prianos melanos
(Kobayashi et al., 1987)
23
psammaplin A
1987
Psammaplvsilla sp.
Tonga
(Quiñoà and Crews, 1987)
24
(E,Z)-isomer of psammaplin A
1987
unidentified sponge
Guam, U.S.A.
(Arabshahi and Schmitz, 1987)
25
bisaprasin
1987
Thorectopsamma xana
(Rodriguez et al., 1987)
26
mycothiazole
1988
Spongia mycofijiensis
Vanuatu
(Crews et al., 1988),(Sugiyama et al., 2003)
27
dercitin
1989
Dercitus sp.
Bahamas
(Burres et al., 1989)
28
prianosin B
1988
Prianos melanos
Motobu Peninsula, Okinawa, Japan
(Cheng et al., 1988)
29
prianosin C
1988
30
prianosin D(discorhabdin D)
1988
31
discorhabdin A
1988
Latrunculia sp.
New Zealand
(Perry et al., 1988)
32
discorhabdin B
1988
33
adociaquinone A
1987
Adocia sp.
Truk Lagoon
(Schmitz and Bloor, 1988)
34
adociaquinone B
1987
35
3-ketoadociaquinone A
1987
36
agelasidine A
1983
Agelas sp.
Okinawa, Japan
(Nakamura et al., 1983)
37
6,7-epoxy-latrunculin A
1989
Latruncularia magnifica
Red Sea
(Blasberger et al., 1989)
38
latrunculin M
1989
39
corallistine
1989
Corallistes fulvodesmus
New Caledonia
(Debitus et al., 1989)
40
batzelline A
1989
Batzella sp.
Bahamas
(Sakemi et al., 1989)
41
batzelline B
1989
42
cyclodercitin
1989
Dercitus sp.
(Gunawardana et al., 1989)
43
nordercitin
1989
Stelletta sp.
44
dercitamine
1989
45
dercitamide
1989
46
isobatzelline A
1990
Batzella sp.
Caribbean
(Sun et al., 1990)
47
isobatzelline B
1990
48
isobatzelline D
1990
49
neamphine
1991
Neamphius huxleyi
Papua New Guinea
(de Silva et al., 1991)
50
psammaplin B
1991
Psammaplysilla purpurea
–
(Jiménez and Crews, 1991)
51
psammaplin C
1991
52
psammaplin D
1991
53
prepsammaplin A
1991
54
phloeodictine B
1992
Phloeodictyon sp.
New Caledonian
(Kourany-Lefoll et al., 1992)
55
stellettamine
1992
Stelletta sp.
–
(Gunawardana et al., 1992)
56
(9E)-clathridine 9-N-(2-sulfoethyl)-imine
1992
Leucetta microraphis
pohnpei
(He et al., 1992)
57
melemeleone A
1992
Dysidea avara
Solomon Islands
(Alvi et al., 1992)
58
melemeleone B
1992
59
(−)-agelasidine C
1992
Agelas clathrodes
Puerto Rico
(Morales and Rodríguez, 1992)
60
(−)-agelasidine D
1992
61
dysideathiazole
1993
Dysidea herbacea
Pohnpei and Palau
(Unson et al., 1993)
62
N-methyldysideathiazole
1993
63
l0-dechloro-N-methyldysideathiazole
1993
64
10-dechlorodysideathiazole
1993
65
9,l0-adechloro-N-methyldysideathiazo
1993
66
potent aldose reductase inhibitor la
1993
Dictyodendrilla sp.
Kagoshima, Japan
(Sato et al., 1993)
67
potent aldose reductase inhibitor lb
1993
68
potent aldose reductase inhibitor 2a
1993
69
makaluvamine F
1993
Zyzzya fuliginosa
Fijian
(Radisky et al., 1993)
70
34-O-sulfatobastadin-13
1993
Zanthella sp.
Great Barrier Reef
(Gulavita et al., 1993)
71
mauritamide A
1994
Agelas mauritiana
Fijian
(Jiménez and Crews, 1994)
72
6-(p-hydroxyphenyl)–2H-3,4-dihydro-1,1-dioxo-1,4-thiazine
1994
Anchinoe tenacior
Mediterranean
(Casapullo et al., 1994)
73
herbamide A
1995
Dysidea herbacea
Papua New Guinea
(Clark and Crews, 1995)
74
latrunculin S
1996
Fasciospongia rimosa
Okinawa, Japan
(Tanaka et al., 1996)
75
hyrtiomanzamine
1996
Hyrtios erecta
Red Sea
(Bourguet-Kondracki et al., 1996)
76
sagitol
1996
Oceanapia sagittaria
Palau
(Salomon and Faulkner, 1996)
77
5,5-dichloro-4-methyl-2-[methyl(4,4-dichloro-3-methyl-1-oxobutyl)amino]-N-(thiazol-2-ylmethyl)pentanamide
1997
Dysidea herbacea
southern Great Barrier Reef
(Dumdei et al., 1997)
78
tauroacidin A
1997
Hymeniacidon sp.
Okinawa, Japan
(Kobayashi et al., 1997)
79
tauroacidin B
1997
80
thiomycalolide A
1998
Mycale sp.
Japan
(Matsunaga et al., 1998)
81
thiomycalolide B
1998
82
kuanoniamine C
1998
Oceanapia sp.
Truk, Micronesia.
(Eder et al., 1998)
83
kuanoniamine D
1998
84
N-deacetylkuanoniamine C
1998
85
the methylthio derivative isobatzelline B
1990
Batzella sp.
Caribbean
(Sun et al., 1990)
86
discorhabdin Q
1999
Latrunculia purpurea, Zyzzya massalis, Zyzzya fuliginosa, and Zyzzya spp.
Assail Bank, between North Island and the Wallab Group, Australia,
(Dijoux et al., 1999)
87
echinosulfonic acid A
1999
Echinodictyum sp.
Great Australian Bight, Southern Australian
(Ovenden and Capon, 1999),(Neupane et al., 2020)
88
echinosulfonic acid B
1999
89
echinosulfonic acid C
1999
90
S1319
1999
Dysidea sp.
Okinawa, Japan
(Suzuki et al., 1999)
91
penarolide sulfate A1
2000
Penares sp.
Japan
(Nakao et al., 2000)
92
penarolide sulfate A2
2000
93
pateamine
1991
Mycale sp.
New Zealand
(Northcote et al., 1991)
94
(−)-neodysidenin
2000
Dysidea herbacea
Great Barrier Reef
(MacMillan et al., 2000)
95
taurodispacamide A
2000
Agelas oroides
The Bay of Naples
(Fattorusso and Taglialatela-Scafati, 2000)
96
discorhabdin R
2000
Latrunculia sp.
the central Prydz channel of Prydz Bay, Antarctica
(Ford and Capon, 2000)
Negombata sp.
Victoria, Port Campbell
97
ianthesine C
2000
Ianthella sp.
Australian
(Okamoto et al., 2000)
98
ianthesine D
2000
99
psammaplin A1
2000
Aplysinella rhax
Pohnpei and Palau
(Shin et al., 2000)
100
psammaplin A2
2000
101
aplysinellin A
2000
102
aplysinellin B
2000
103
psammaplin A 11′-sulfate
2000
Aplysinella rhax
Great Barrier Reef
(Pham et al., 2000)
104
bisaprasin 11′-sulfate
2000
105
wondonin A
2001
Poecillastra wondoensisand Japsis sp.
Keomun Island, Korea
(Shin et al., 2001)
106
wondonin B
2001
107
microxine
2001
Microxina sp.
Cape Jaffa, Australian
(Killday et al., 2001)
108
irciniamine
2002
Ircinia sp.
Ehime Prefecture, Japan
(Kuramoto et al., 2002)
109
ancorinolate A
2002
Ancorina sp.
Chatham Island,New Zealand
(Meragelman et al., 2002)
110
ancorinolate B
2002
111
bis-ancorinolate B
2002
112
ancorinazole
2002
113
psammaplin K
2002
Aplysinella rhax
Fijian
(Tabudravu et al., 2002)
114
psammaplin L
2002
115
cribronic acid
2003
Cribrochalina olemda
Palau
(Sakai et al., 2003)
116
(2S,4S)-4-sulfooxypiperidine-2-carboxylic acid
2003
Stylotella aurantium, andAxinella carteri
Yap State, Micronesia
117
dictyodendrin A
2003
Dictyodendrilla verongiformis
Nagashima Island, Japan
(Warabi et al., 2003)
118
dictyodendrin B
2003
119
dictyodendrin C
2003
120
dictyodendrin D
2003
121
dictyodendrin E
2003
122
penasulfate A
2004
Penares sp.
Hachijo-jima Island,Tokyo, Japan
(Nakao et al., 2004)
123
spongiacysteine
2004
Spongia sp.
Tateyama beach,Chiba Prefecture, Japan
(Kobayashi et al., 2004)
124
dragmacidonamine A
2004
Dragmacidon sp.
Adaman Islands, India
(Pedpradab et al., 2004)
125
dragmacidonamine B
2004
126
1-methoxydiscorhabdin D
2004
Latrunculia bellae
Thunderbolt Reef,Algoa Bay, South Africa
(Antunes et al., 2004)
127
1-aminodiscorhabdin D
2004
128
discorhabdin G*
2004
129
discorhabdin N
2004
130
discorhabdin H
2004
Strongylodesma algoaensis
131
discorhabdin I
2004
Latrunculia brevis
Tierra del Fuego, Patagonia, Argentina
(Reyes et al., 2004)
132
discorhabdin L
2004
133
cribrostatin 7
2004
Petrosia sp. PC00-11–149
Kalampisauan Island, Philippines
(Sandoval et al., 2004)
134
bisdemethylaaptamine-9-O-sulfate
2004
Aaptos sp.
Bunaken Island, Indonesian
(Herlt et al., 2004)
135
nagelamide H
2004
Agelas sp.
Seragaki Beach, Okinawan
(Endo et al., 2004)
136
schulzeine A
2004
Penares schulzei
Hachijo-kojima Island, Japan
(Takada et al., 2004)
137
schulzeine B
2004
138
schulzeine C
2004
139
1-O-sulfatohemibastadin-1
2004
Ianthella basta
Mangilao, Guam, U.S.A.
(Masuno et al., 2004)
140
1-O-sulfatohemibastadin-2
2004
141
34-O-sulfatobastadin-9
2004
142
32-O-sulfatobastadin-13
2004
143
hamiguanosinol
2004
Mediterranean hamigera
Elba, Mediterranean Sea
(Hassan et al., 2004),(Jamison et al., 2014)
144
3-ketoadociaquinone B
2005
Xestospongia sp.
Indonesia, Sulawesi
(Cao et al., 2005)
145
discorhabdin W
2005
Latrunculia sp.
New Zealand
(Lang et al., 2005)
146
discorhabdin G*/I
2005
147
echinosulfonic acid D
2005
Psammoclemma sp.
New Caledonia
(Rubnov et al., 2005), (Neupane et al., 2020)
148
gesashidine A
2005
An unidentified member of the Thorectidae family
Okinawan
(Iinuma et al., 2005)
149
halichondria sulfonic acid
2006
Halichondria rugosa
South China Sea
(Jin et al., 2006)
150
latrunculin T
2006
Negombata magnifica
Red Sea (near Egypt)
(El Sayed et al., 2006)
151
(−)-agelasidine A
2006
Agelas clathrodes
Curaçao, Caribbean sea
(Medeiros et al., 2006)
152
dysinosin A
2002
a New Genus and Species of Sponge of Dysideidae
Lizard Island, North Queensland, Australia
(Carroll et al., 2002)
153
mycothiazole-4,19-diol
2006
Cacospongia mycofijiensis
Vanuatu
(Sonnenschein et al., 2006)
154
ircinamine B
2006
Dactylia sp.
Cape Sada, Japan
(Sato et al., 2006)
155
discorhabdin S
2003
Batzella sp.
Bimini, Bahamas
(Gunasekera et al., 2003)
156
discorhabdin T
2003
157
discorhabdin U
2003
158
2-debromotaurodispacamide A
2006
Axinella verrucosa
Corsica, France
(Aiello et al., 2006)
159
oxalatrunculin B
2007
Negombata corticata
Red Sea (near Egypt)
(Ahmed et al., 2007)
160
araplysillin-N9-sulfamate
2007
Aplysina fulva
Key Largo, Florida
(Rogers and Molinski, 2007)
161
siphonodictyals B1
2007
Aka coralliphagum
San Salvador, Bahamas
(Grube et al., 2007)
162
exiguaquinol
2008
Neopetrosia exigua
Queensland, Australia
(de Almeida Leone et al., 2008)
163
CTP-431
2008
Cacospongia mycofijiensis
Beqa Lagoon, Fiji
(Johnson et al., 2008)
164
latrunculol A
2008
(Amagata et al., 2008)
165
latrunculol B
2008
166
latrunculol C
2008
167
18-epi-latrunculol A
2008
168
latrunculone A
2008
169
latrunculone B
2008
170
16-epi-latrunculin B
2004
Latruncularia magnifica
Red Sea
171
15-methoxylatrunculin B
2004
172
nagelamide K
2008
Agelas sp.
Seragaki, Okinawa, Japan
(Araki et al., 2008)
173
nagelamide M
2008
(Kubota et al., 2008)
174
nagelamide N
2008
175
ianthesine E
2008
Pseudoceratina sp.
Swain Reefs, Australia
(Kalaitzis et al., 2008)
176
alisiaquinone C
2008
An unidentified sponge
New Caledonia
(Desoubzdanne et al., 2008)
177
phorbasin D
2008
Phorbas sp.
Great Australian Bight, South Australia
(Zhang and Capon, 2008)
178
phorbasin E
2008
179
phorbasin F
2008
180
(+)-debromodiscorhabdin A
2009
Higginsia sp.
South Australia
(El-Naggar and Capon, 2009)
181
(+)-discorhabdin X
2009
182
(−)-dihydrodiscorhabdin A
2009
183
(+)-Dihydrodiscorhabdin L
2009
Spongosorites sp.
184
(6R,8S)-1-thiomethyldiscorhabdin G*/I
2009
Latrunculia wellingtonesis
Wellington, New Zealand
(Grkovic and Copp, 2009)
185
16a,17a-dehydrodiscorhabdin W
2009
186
nagelamide Q
2009
Agelas sp.
Okinawan, Japan
(Araki et al., 2009)
187
psammaplin I
2003
Pseudoceratina purpurea
Papua New Guinea
(Piña et al., 2003)
188
psammaplin E
2003
189
psammaplin F
2003
190
psammaplin G
2003
191
psammaplin H
2003
192
psammaplin J
2003
193
19-oxofasciospongine A
2009
Fasciospongia sp.
Palau
(Yao et al., 2009)
194
fasciospongine C
2009
195
fasciospongine A
2009
196
fasciospongine B
2009
197
callyspongine
2010
Callyspongia sp.
South China Sea(Hainan island)
(Huang et al., 2010)
198
dysideanin A
2010
Dysidea sp.
Lingshui County, Hainan, China
(Ren et al., 2010)
199
(+)-discorhabdin H2
2010
Latrunculia fiordensi
New Zealand
(Grkovic et al., 2010)
200
(−)-discorhabdin K2
2010
201
(−)-discorhabdin N
2010
Latrunculia bellae
202
dihydrodiscorhabdin B
2010
Latrunculia sp.
Aleutian Islands, U.S.A.
(Na et al., 2010)
203
(−)-3-dihydrodiscorhabdin D
2010
Sceptrella sp.
Gageodo, Korea
(Jeon et al., 2010)
204
mauritamide B
2010
Agelas linnaei
Peniki East island, ThoU.S.A. nd Islands, Indonesia
(Hertiani et al., 2010)
205
mauritamide C
2010
206
mauritamide D
2010
207
baculiferin A
2010
Iotrochota baculifera
Hainan island,South China Sea
(Fan et al., 2010)
208
baculiferin B
2010
209
baculiferin C
2010
210
baculiferin D
2010
211
baculiferin E
2010
212
baculiferin F
2010
213
baculiferin G
2010
214
baculiferin H
2010
215
baculiferin I
2010
216
baculiferin J
2010
217
baculiferin M
2010
218
baculiferin O
2010
219
psammaplin N
2010
Aplysinella rhax
Inner Gneerings Reef, Queensland, Australia
(Graham et al., 2010)
220
9-(5′-deoxy-5′-thio-β-d-xylofuranosyl)adenine disulfide
2010
Trachycladus laevispirulifer
Great Australian Bight, South Australian
(Peng et al., 2010)
221
amaranzole B
2010
Phorbas amaranthus
Dry Reef Rocks, Key Largo, Florida
(Morinaka et al., 2010)
222
amaranzole C
2010
223
amaranzole D
2010
224
amaranzole E
2010
225
amaranzole F
2010
226
nakijinamine C
2011
Suberites sp.
Unten Port, Okinawa, Japan
(Takahashi et al., 2011)
227
nakijinamine D
2011
228
xestosaprol N
2012
Xestospongia sp.
Weno island, Chuuk State, Federated States of Micronesia
(Lee et al., 2012)
229
14-O-sulfate massadine
2012
Axinella sp.
Great Australian Bight
(Zhang et al., 2012)
230
(+)-2-oxo-agelasidine C
2012
Agelas mauritiana
Yongxing island,South China Sea
(Yang et al., 2012)
231
(−)-agelasidine E
2012
Agelas citrina
Bahamas
(Stout et al., 2012)
232
(−)-agelasidine F
2012
233
2-heptadec-11-enamidoethanesulfonic acid
2013
Axinella sp.
Hainan island,South China Sea
(Huang et al., 2013)
234
2-palmitamidoethanesulfonic acid
2013
235
2-octadec-7-enamidoethanesulfonic
2013
236
ciliatamide D
2013
Stelletta sp.
Oshimashinsone, Japan
(Imae et al., 2013),(Takada et al., 2017)
237
theonezolide A
2013
Theonella sp.
Okinawa, Japan
(Nozawa et al., 2013)
238
theonezolide B
2013
239
theonezolide C
2013
240
catechol sulfonate
2013
Asteropus sp.
Ocean Cay, Bahamas
(Russell et al., 2013)
241
hyrtimomine D
2013
Hyrtios sp.
Kerama island, Okinawa, Japan
(Tanaka et al., 2013c)
242
hyrtimomine E
2013
243
thiaplakortone A
2013
Plakortis lita
Tydeman Reef,Queensland, Australia
(Davis et al., 2013)
244
thiaplakortone B
2013
245
thiaplakortone C
2013
246
thiaplakortone D
2013
247
atkamine A
2013
Latrunculia sp.
Aleutian island, Alaska, U.S.A.
(Zou and Hamann, 2013)
248
nagelamide U
2013
Agelas sp.
Kerama islands,Okinawa, Japan
(Tanaka et al., 2013a)
249
nagelamide V
2013
250
nagelamide Y
2013
(Tanaka et al., 2013b)
251
nagelamide Z
2013
252
reticulatin A
2013
Hyrtios reticulatus
N. Sulawesi, Indonesia
(Imada et al., 2013)
253
reticulatin B
2013
254
N-methylmelemeleone-A
2013
Dysidea avara
Fethiye, Turkey
(Hamed et al., 2013)
255
deacyl irciniasulfonic acid C
2014
Coscinoderma sp.
Weno island, Chuuk State, Micronesia
(Kim et al., 2014a)
256
sodium deacyl irciniasulfonate D
2014
257
N,N-dimethylguanidium salt
2014
258
N,N-dimethyl-1,3-dimethylherbipoline salt
2014
259
coscinolactam C
2014
260
coscinolactam D
2014
261
coscinolactam E
2014
262
coscinolactam F
2014
263
coscinolactam G
2014
264
coscinolactam A
2009
Coscinoderma mathewsi
Vangunu Island,Solomon Islands
(De Marino et al., 2009)
265
coscinolactam B
2009
266
glassponsine
2014
Anoxycalyx joubini
Trawled, E. Weddell Sea, Antarctica
(Carbone et al., 2014)
267
hainanerectamine C
2014
Hyrtios erecta
Lingshui Bay, China
(He et al., 2014)
268
hyrtimomine H
2014
Hyrtios sp.
Kerama islands,Okinawa, Japan
(Tanaka et al., 2014)
269
hyrtimomine J
2014
270
hyrtimomine K
2014
271
callyspongisine A
2014
Callyspongia sp.
Great Australian Bight
(Plisson et al., 2014)
272
callyspongisine B
2014
273
tauroacidin C
2014
Agelas sp.
Kerama islands,Okinawa, Japan
(Kusama et al., 2014)
274
tauroacidin D
2014
275
5-epi-nakijiquinone U
2014
Dactylospongia metachromia
Ambon, Indonesia
(Daletos et al., 2014)
276
xestosaprol O
2014
Xestospongia vansoesti
Palawan island,Philippines
(Centko et al., 2014)
277
2-(3-methyl-dec-3-enamido)ethanesulfonic Acid
2015
Callyspongia sp.
Hainan island, China
(Huang et al., 2015)
278
tauroacidin E
2015
Agelas sp.
Kerama island, Okinawa, Japan
(Kusama et al., 2015)
279
2-debromonagelamide U
2015
(Kenta Nakamura, 2015)
280
citrinamine B
2015
Agelas citrina
San Salvador, Bahamas
(Cychon et al., 2015)
281
melemeleone C
2015
Dysidea sp.
Chuuk island, Federated States of Micronesia
(Kim et al., 2015)
282
melemeleone D
2015
283
cycloaurenone A
2015
284
xestoadociaminal A
2015
Xestospongia sp.
Manado, N. Sulawesi, Indonesia
(He et al., 2015)
285
xestoadociaminal B
2015
286
xestoadociaminal C/D
2015
287
xestoadociaquinone A
2015
288
xestoadociaquinone B
2015
289
seadociaquinone A
2015
290
seadociaquinone B
2015
291
petroquinone I
2016
Petrosia alfiani
Ti Toi, N. Sulawesi, Indonesia
(Tanokashira et al., 2016)
292
petroquinone J
2016
293
petroquinone K
2016
294
petroquinone L
2016
295
conulothiazole A
2016
Smenospongia conulosa
Little Inagua island, Bahamas
(Esposito et al., 2016)
296
conulothiazole B
2016
297
smenothiazole A
2016
298
smenothiazole B
2016
299
(–)-isowondonin A
2008
Poecillastra wondoensis
Keomun Island, Korea
(Chang et al., 2008)
300
(–)-isowondonin B
2008
301
ishigadine A
2018
Hyrtios sp.
Ishigaki island, Okinawa, Japan
(Takahashi et al., 2018)
302
langcoquinone D
2018
Spongia sp.
Son Cha, Lang Co, Tha Thien-Hue City, Vietnam
(Ito et al., 2018)
303
langcoquinone E
2018
304
langcoquinone B
2018
305
dactylospongin A
2018
Dactylospongia sp.
Xisha island,South China Sea
(Li et al., 2018)
306
dactylospongin B
2018
307
ent-melemeleone B
2018
308
melemeleone E
2018
309
(−)-2-bromo-discorhabdin D
2019
Latrunculia biformis
Dredge,Southern Weddell Sea, Antarctica
(Li et al., 2019)
310
(−)-1-acetyl-discorhabdin L
2019
311
(+)-1-octacosatrienoyl-discorhabdin L
2019
312
aleutianamine
2019
Latrunculia austini
Aleutian Islands, Alaska, U.S.A.
(Zou et al., 2019)
313
tedanizaine A
2020
Tedania sp.
Zhanjiang, Guangdong, China
(Zhang et al., 2020b)
314
(−)-(1S,2R,6R,8S,6′S)-discorhabdin B dimer
2020
Latrunculia biformis
Dredge,Southern Weddell Sea, Antarctica
(Li et al., 2020b)
315
(−)-(1R,2R,6R,8S,6′S)-16′,17′-dehydrodiscorhabdin B dimer
2020
316
(−)-(1R,2R,6R,8S,6′S)-discorhabdin B dimer
2020
317
(−)-tridiscorhabdin
2020
Dredge,Southern Weddell Sea, Antarctica
(Li et al., 2020c)
318
(−)-didiscorhabdin
2020
319
psammaplin O
2020
Aplysinella rhax
Wainunu, Bua, Fiji island
(Oluwabusola et al., 2020)
320
psammaplin P
2020
321
echinosulfone A
1999
Echinodictyum sp.
Great Australian Bight, Southern Australian
(Ovenden and Capon, 1999)
322
neopetrothiazide
2021
Neopetrosia sp.
Helen Reef, Southwest Islands, Palau
(Wang et al., 2021)
323
tedaniophorbasin A
2021
Tedaniophorbas ceratosis
northern New South Wales, Australia.
(Hiranrat et al., 2021)
324
tedaniophorbasin B
2021
325
agelasidine G
2022
Agelas nakamurai
Orchid Island, Taiwan
(Lin et al., 2022)
326
agelasidine H
2022
327
agelasidine I
2022
328
isoagelasidine B
2022
329
24-methylsulfinyllancoquinone B
2022
Spongia pertusa
South China Sea
(Tang et al., 2022)
330
cyclohexylagelasidine A
2022
Agelas nakamurai
Orchid Island, Taiwan
(Fu et al., 2022)
331
(+)-12-hydroxyagelasidine C
2022
Agelas citrina
Cozumel Island, Mexico
(Pech-Puch et al., 2022)
Among them, discorhabdins, psammaplins and latrunculins are the three noteworthy chemical components. (1) The discorhabdin alkaloids, which have a unique structure with azacarbocyclic spirocyclohexanone and pyrroloiminoquinone units, usually have cytotoxicity against a variety of tumor cells. And when they have a sulfur-containing six-membered ring, discorhabdin alkaloids often have good cytotoxicity (Antunes et al., 2004). Notably, dimers (Lang et al., 2005) and trimers (Li et al., 2020c) of discorhabdin alkaloids, which were reported in recent years, also have good cytotoxicity. (2) Psammaplins are bromotyrosine derivatives with oxime groups and carbon–sulfur bonds. Among them, psammaplin A (23) is the first identified symmetrical bromotyrosine-derived disulfide dimer, which has a broad bioactive spectrum, especially in terms of antimicrobial and antiproliferative activities (Quiñoà and Crews, 1987). (3) Latrunculins, toxins from the red sea sponge Latrunculia magnifica, are concerned as a kind of F-actin-severing compound. Of which, latrunculin A (16) is the most widely used reagent to depolymerize actin filaments in experiments on live cells (Spector et al., 1983).
1.2.2.2 Marine cnidarians
Cnidarians are the most primitive metazoan, which can be divided into three classes: Hydra, Aquarius and Corallus. And with growing bioprospecting efforts and the screening of previously unexplored marine habitats, the phylum cnidarians have been a large, diverse and ecologically important group of marine invertebrates that includes over 11,000 extant species (Rocha et al., 2011).
A total of 21 (332–352) sulphur-containing alkaloids were reported from the marine cnidarians (Fig. 6 and Table 3). Among them, tridentatols E-H are sodium sulfate salt of tridentatols A-D. When potential predators appear, Tridentata marginata will rapidly convert tridentatols E-H to tridentatols A-D, which are nonprotein venom produced by cnidarian nematocysts, and repel the potential predators (Lindquist, 2002).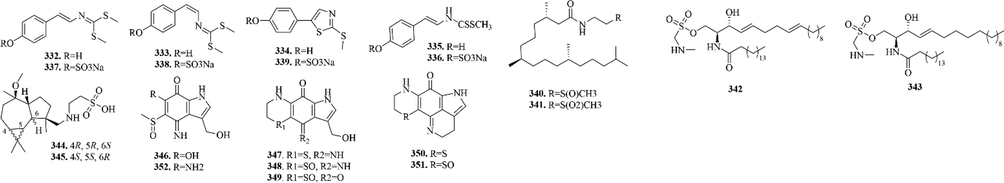
Sulphur-containing alkaloids from marine cnidarians.
No.
Compounds
Time
From
Location
Ref.
Marine cnidarians
332
tridentatol A
1996
Tridentata marginata
Morehead City,North Carolina, USA.
(Lindquist et al., 1996)
333
tridentatol B
1996
334
tridentatol C
1996
335
tridentatol D
2002
(Lindquist, 2002)
336
tridentatol E
2002
337
tridentatol F
2002
338
tridentatol G
2002
339
tridentatol H
2002
340
sinulasulfoxide
2012
Sinularia sp.
Manado, North Sulawesi, Indonesia
(Putra et al., 2012)
341
sinulasulfone
2012
342
palyosulfonoceramide A
2012
Palythoa caribaeorum andProtopalythoa variabilis
Paracuru beach,Fortaleza, Brazil
(Almeida et al., 2012)
343
palyosulfonoceramide B
2012
344
(+)-4β-N-methenetauryl-10β-methoxy1β,5α,6β,7β-aromadendrane
2012
Melitodes squamata
Sanya, Hainan,South China Sea
(Huang et al., 2012)
345
(−)-4β-N-methenetauryl-10β-methoxy-1β,5β,6α,7α-aromadendrane
2012
346
macrophilone B
2018
Macrorhynchia philippina
Northwestern Australia
(Yan et al., 2018)
347
macrophilone C
2018
348
macrophilone D
2018
349
macrophilone E
2018
350
macrophilone F
2018
351
macrophilone G
2018
352
macrophilone A
2018
1.2.2.3 Marine tunicates
Tunicates, which distribute in the world's major seas, are soft-bodied solitary or colonial sessile small marine organisms belonging to the family Ascidiacea under the subphylum Urochordata, phylum Chordata. There’re more than 2,800 species of tunicate species, which are divided into three classes: Ascidiacea, Thaliacea and Appendicularia. Tunicates will lose the notochord and post-anal tail; thus, these organisms are often referred to as the “evolutionary connecting link” between invertebrates and chordates (Ramesh et al., 2021).
158 (353–510) sulphur-containing alkaloids were reported from the marine tunicates (Fig. 7 and Table 4). Among them, eudistomins and ecteinascidins are noteworthy chemical components. (1) Eudistomins attract the attention of scientists because of their good antiviral activity. Subsequent studies have found that eudistomins have the strongest anti-tumor activity when they contain a 1,3,7-oxathiazepine ring. For example, eudistomins C and E, which contain a 1,3,7-oxathiazepine ring, are potent antiviral against RNA viruses (Coxsackie A-21 virus and equine rhinovirus) as well as DNA viruses (HSV-1, HSV-2, and Vaccinia virus). Besides the substituents on the pyridine ring of the β-carboline, the substituents (Br and/or OH) and their positions on the benzenoid ring of the β-carboline may influence the antiviral activity of eudistomins; the order of antiviral activity observed is E (5-Br, 6-OH) > C (6-OH, 7-Br) > L (6-Br). But acetylation of the phenol and primary amine functions of eudistomin C affected a 100-fold reduction in activity (Blunt et al., 1987). (2) Ecteinascidins, a kind of sulphur-containing alkaloids, are marine natural products with potent antitumor activity. These compounds have been a lot of synthetic research and structural modification. Of them, ecteinascidin 743 (yondelis) has been approved by the European Union in October 2007 for the treatment of advanced soft tissue tumors, which became the first modern marine drug (Menchaca et al., 2003).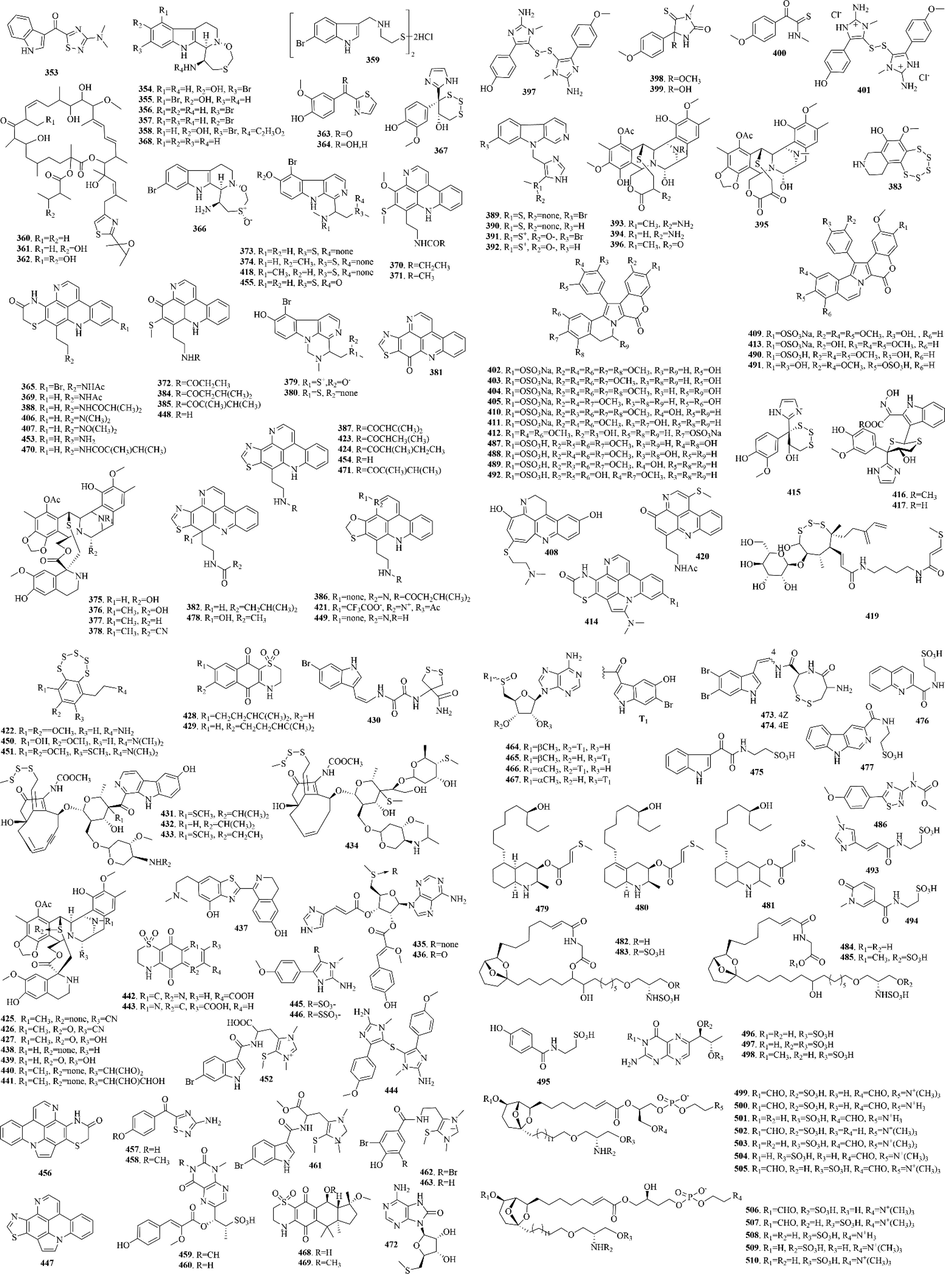
Sulphur-containing alkaloids from marine tunicates.
No.
Compounds
Time
From
Location
Ref.
Marine tunicates
353
dendrodoine
1982
Dendrodoa grossularia
–
(Heitz et al., 1980)
354
eudistomin C
1987
Eudistoma olivaceum
Caribbean
(Rinehart et al., 1987),(Blunt et al., 1987)
355
eudistomin E
1987
356
eudistomin K
1987
357
eudistomin L
1987
358
eudistomin F
1987
359
citorellamine
1985
Polycitorella mariae
Suva, Fiji
(Roll and Ireland, 1985),(Moriarty et al., 1987)
360
patellazole A
1988
Lissoclinum patella
Palau
(Zabriskie et al., 1988)
361
patellazole B
1988
362
patellazole C
1988
363
(4-hydroxy-3-methoxyphenyl)(thiazol-2-yl)methanone
1988
Aplydium pliciferum
Australian
(Arabshahi and Schmitz, 1988)
364
4-(hydroxy(thiazol-2-yl)methyl)-2-methoxyphenol
1988
365
shermilamine A
1988
Trididemnum sp.
Pago Bay, Guam
(Cooray et al., 1988)
366
eudistomin K sulfoxide
1988
Ritterella sigillinoides
New Zealand
(Lake et al., 1988)
367
cis-5-hydroxy-4-(4′-hydroxy-3′-methoxyphenyl)-4-(2″-imidazolyl)-1,2,3-trithiane
1989
Aplidium sp.
(Copp et al., 1989)
368
debromoeudistomin K
1989
Ritterella sigillinoides
(Lake et al., 1989)
369
shermilamine B
1989
Trididemnum sp.
Pago Bay, Guam
(Carroll et al., 1989)
370
varamine A
1989
Lissoclinum vareau
Yasawa island chain, Fiji
(Molinski and Ireland, 1989)
371
varamine B
1989
372
diplamine
1989
Diplosomra sp.
Fiji
(Charyulu et al., 1989)
373
eudistomidin C
1990
Eudistoma glaucus
Ie Island, Okinawan,Japan
(Kobayashi et al., 1990)
374
6-O-methyleudistomidin C
1990
375
ecteinascidin 729
1990
Ecteinascidia turbinata
Caribbean
(Menchaca et al., 2003)
376
ecteinascidin 743
1990
377
ecteinascidin 745
1990
378
ecteinascidin 770
1990
379
eudistomidin E
1991
Eudistoma glaucus
Ie Island, Okinawan,Japan
(Murata et al., 1991)
380
eudistomidin F
1991
381
kuanoniamine A
1990
an unidentified Micronesian tunicate
Mante Channel, Pohnpei,Micronesia
(Carroll and Scheuer, 1990)
382
kuanoniamine B
1990
383
lissoclinotoxin B
1994
Lissoclinum perforatum
Northern Brittany, France
(Litaudon et al., 1994)
384
lissoclin A
1994
Lissoclinum sp.
Great Barrier Reef
(Searle and Molinski, 1994)
385
lissoclin B
1994
386
benzo-1,3-oxathiazoline
1994
387
dehydrokuanoniamine B
1994
Cystodytes sp.
Fiji
(McDonald et al., 1994)
388
shermilamine C
1994
389
didemnoline A
1995
Didemnum sp.
Rota, Northern Mariana Islands
(Schumacher and Davidson, 1995)
390
didemnoline B
1995
391
didemnoline C
1995
392
didemnoline D
1995
393
ecteinascidin 597
1996
Ecteinascidia turbinata
Caribbean
(Sakai et al., 1996)
394
ecteinascidin 583
1996
395
ecteinascidin 594
1996
396
ecteinascidin 596
1996
397
polycarpine
1996
Polycarpa clavataPolycarpa aurata
Western AustraliaChuuk, Federated States of Micronesia
(Kang and Fenical, 1996),(Abas et al., 1996)
398
4-methoxy-4-(4-methoxyphenyl)-1-methyl-5-thioxoimidazolidin-2-one
1996
399
4-hydroxy-4-(4-methoxyphenyl)-1-methyl-5-thioxoimidazolidin-2-one
1996
Polycarpa clavata
Western Australia
(Kang and Fenical, 1996)
400
N-methyl-(4-methoxyphenyl)-2-oxothioacetamide
1996
Polycarpa aurata
Chuuk, Federated States of Micronesia
(Abas et al., 1996)
401
polycarpine dihydrochloride
1996
402
the 20-sulfate of lamellarins T
1997
An unidentified ascidian
Arabian Sea (near India)
(Reddy et al., 1997)
403
the 20-sulfate of lamellarins U
1997
404
the 20-sulfate of lamellarins V
1997
405
the 20-sulfate of lamellarins Y
1997
406
shermilamine D
1998
Cystodytes violatinctus
Mayotte lagoon, ComorosIslands, Madagascar
(Koren-Goldshlager et al., 1998)
407
shermilamine E
1998
408
tintamine
1998
409
the 20-sulfates of lamellarin B
1999
Didemnum chartaceum
Great Barrier Reef
(Davis et al., 1999)
410
the 20-sulfates of lamellarin C
1999
411
the 20-sulfates of lamellarin L
1999
412
the 20-sulfates of lamellarin G
1999
413
lamellarin α 20-sulfate
1999
an unidentified ascidian
Arabian Sea(near Trivandrum, India)
(Reddy et al., 1999)
414
cycloshermilamine D
2000
Cystodytes violatinctus
Mayotte lagoon, ComorosIslands, Madagascar
(Koren-Goldshlager et al., 2000)
415
(−)-enantiomer
2001
Hypsistozoa fasmeriana
New Zealand
(Pearce et al., 2001)
416
fasmerianamine A
2001
417
fasmerianamine B
2001
418
14-methyleudistomidin C
2001
Eudistoma gilboverde
Sias Tunnel, Palau
(Rashid et al., 2001)
419
(2E,4′R,5′S,6′R,7′R,8′S,2′'''E)-3-{8′-hydroxy-4′,6′-dimethyl-4′-(3′'-methylenepent-4′'-enyl)-7′-(-L-mannopyranosyloxy)-[1′,2′,3′]-trithiocan-5′-yl}-N-[4′''-(3′'''-methylsulfanylacryloylamino)-butyl]aerylamide
2002
Perophora viridis
Atlantic coast (near North Carolina)
(Řezanka and Dembitsky, 2002)
420
isodiplamine
2002
Lissoclinum notti
Leigh Harbour, Northland, New Zealand
(Appleton et al., 2002)
421
lissoclinidine
2002
422
varacin
2002
423
kuanoniamine E
2002
an unidentified Singaporean ascidian
Pulau Subar Laut,Singapore
(Nilar et al., 2002)
424
kuanoniamine F
2002
425
ecteinascidin 770
2002
Ecteinascidia thurstoni
Phuket Island, Thai
(Suwanborirux et al., 2002)
426
ecteinascidin 786
2002
427
ecteinascidin 759B
2002
428
conicaquinone A
2003
Aplidium conicum
Capo Caccia, Alghero, Italy
(Aiello et al., 2003)
429
conicaquinone B
2003
430
kottamide E
2003
Pycnoclavella kottae
New Zealand
(Appleton and Copp, 2003)
431
shishijimicin A
2003
Didemnum proliferum
South Japan
(Oku et al., 2003)
432
shishijimicin B
2003
433
shishijimicin C
2003
434
namenamicin
2003
435
methylthioadenosine
2004
Atriolum robustum
Heron Islands, Wistari Reef, Great Barrier Reef
(Kehraus et al., 2004)
436
methylsulfinyladenosine
2004
437
violatinctamine
2004
Cystodytes cf. violatinctus
Kenya
(Chill et al., 2004)
438
ecteinascidin 731
2004
Ecteinascidia turbinata
Caribbean
(Blunt et al., 2006)
439
ecteinascidin 745b
2004
440
ecteinascidin 808
2004
441
ecteinascidin 815
2004
442
ascidiathiazone A
2007
Aplidium sp.
Tom Bowling Bay,Northland, New Zealand
(Pearce et al., 2007)
443
ascidiathiazone B
2007
444
polycarpaurine A
2007
Polycarpa aurata
Lembeh Strait, Indonesia
(Wang et al., 2007)
445
polycarpaurine B
2007
446
polycarpaurine C
2007
447
nordehydrocyclodercitin
2007
Aplidium sp.
Arab Reef, Australia
(Agrawal and Bowden, 2007)
448
diplamine B
2008
Lissoclinum cf. badium
Port Moresby,Papua New Guinea
(Clement et al., 2008)
449
lissoclinidine B
2008
450
isolissoclinotoxin B
2008
451
N,N-dimethyl-5-methylvaracin
2008
452
leptoclinidamine C
2009
Leptoclinides durus
Heron island, Queensland, Australia
(Carroll and Avery, 2009)
453
N-deacetylshermilamine B
2010
Cystodytes dellechiajei
Catalonia, Spain
(Bontemps et al., 2010)
454
N-deacetylkuanoniamine D
2010
455
eudistomidin J
2011
Eudistoma glaucus
Ie island, Okinawa, Japan
(Suzuki et al., 2011)
456
13-didemethylaminoshermilamine D
2011
Cystodytes dellechiajei
Catalonia, Spain
(Bry et al., 2011)
457
polycarpathiamine A
2013
Polycarpa aurata
Ambon, Indonesia
(Pham et al., 2013)
458
polycarpathiamine B
2013
459
duramidine A
2013
Leptoclinides durus
Swains Reef, Great Barrier Reef
(Rudolph et al., 2013)
460
duramidine C
2013
461
leptoclinidamine D
2013
462
leptoclinidamine E
2013
463
leptoclinidamine F
2013
464
momusine A
2013
Herdmania momus
Jeju island, Korea
(Li et al., 2013)
465
momusine B
2013
466
momusine C
2013
467
momusine D
2013
468
conthiaquinone A
2013
Aplidium conicum
Porto Cesareo, Lecce, Italy
(Menna et al., 2013)
469
conthiaquinone B
2013
470
shermilamine F
2013
Cystodytes violatinctus
Solomon islands
(Bontemps et al., 2013)
471
dehydrokuanoniamine F
2013
472
salvadenosine
2014
Didemnum sp.
Little San Salvador island, Bahamas
(Jamison et al., 2014)
473
tanjungide A
2014
Diazona cf formosa
East Timor
(Murcia et al., 2014)
474
tanjungide B
2014
475
stolonine A
2015
Cnemidocarpa stolonifera
Peel island, Australia
(Tran et al., 2015)
476
stolonine B
2015
477
stolonine C
2015
478
sagitol D
2015
an unidentified Vietnamese ascidian
PhuQuok, Vietnam
(Utkina, 2015)
479
lepadin I
2018
Didemnum sp.
Stirrup Cay, Bahamas
(Ómarsdóttir et al., 2018)
480
lepadin J
2018
481
lepadin K
2018
482
siladenoserinol M
2018
Didemnum sp.
Siladen, North Sulawesi, Indonesia
(Torii et al., 2018)
483
siladenoserinol N
2018
484
siladenoserinol O
2018
485
siladenoserinol P
2018
486
polyaurine B
2019
Polycarpa aurata
(Casertano et al., 2019)
487
lamellarin K-20-sulfate
2019
Didemnum ternerratum
Eua, Kingdom of Tonga
(Bracegirdle et al., 2019)
488
lamellarin E-20-sulfate
2019
489
lamellarin A3-20-sulfate
2019
490
lamellarin B1-20-sulfate
2019
491
lamellarin D-8-sulfate
2019
492
lamellarin B2-20-sulfate
2019
493
ireneamide A
2020
Cnemidocarpa irene
Oshima-Kojima Islet off the Oshima Peninsula, Hokkaido, Japan
(Miyako et al., 2020)
494
ireneamide B
2020
495
ireneamide C
2020
496
6-biopterin-2′-sulfate
2020
497
6-biopterin-1′-2′-disulfate
2020
498
3-methyl-6-biopterin-2′-sulfate
2020
499
siladenoserinol A
2013
a tunicate of the family Didemnidae
NorthSulawesi, Indonesia
(Nakamura et al., 2013)
500
siladenoserinol B
2013
501
siladenoserinol C
2013
502
siladenoserinol D
2013
503
siladenoserinol E
2013
504
siladenoserinol F
2013
505
siladenoserinol G
2013
506
siladenoserinol H
2013
507
siladenoserinol I
2013
508
siladenoserinol J
2013
509
siladenoserinol K
2013
510
siladenoserinol L
2013
1.2.2.4 Marine echinoderms
Echinoderms are a kind of deuterostomes, which account for up to 90% of benthic biomass in the abyssal seafloor. The common sea stars, sea urchins, sea cucumbers, and sea snake tails all are echinoderms. At present, about 6000 species of echinoderms were widely distributed from shallow sea to thousands of meters deep sea, which can be divided into five classes including Crinoidea, Holothurioidea, Asteroidea, Echinoidea and Ophiuroidea.
22 (511–532) sulphur-containing alkaloids were reported from the marine cnidarians (Fig. 8 and Table 5). Ovothiols are histidine-derived thiols that are receiving great interest for their biological activities in human model systems. Among them, ovothiol A (514) is one of the strongest natural antioxidants (Osik et al., 2021). It’s worth noting that hypalocrinins are the first naturally occurring anthraquinones and anthraquinone biaryls conjugated with taurine. Hypalocrinins A-E (520–524) are five new water-soluble amido- and aminoanthraquinone pigments and hypalocrinin F-G (525–526) are two new amidoanthraquinone biaryls, which all are quite unusual among natural products (Wolkenstein et al., 2019). Likewise, microdiscusols A-F (527–532), six new polyhydroxylated steroids conjugated with taurine, are rare new polyhydroxylated steroids conjugated with taurine (Kicha et al., 2019).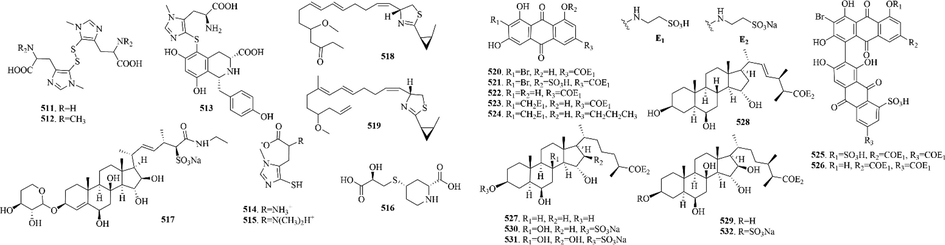
Sulphur-containing alkaloids from marine echinoderms.
No.
Compounds
Time
From
Location
Ref.
Marine echinoderms
511
bis(l-methyl-L-histidin-5-yl)disulphide
1986
unfertilized echinoderm eggs
–
(Faulkner, 1986)
512
bis(Nα.Nα,l-trimethyI-L-histidin-5-yl)disulphide
1986
unfertilized echinoderm eggs
513
imbricatine
1986
Dermasterias imbricata
–
(Pathirana and Andersen, 1986)
514
ovothiol A
1986
Evasterias troschelii
–
(Turner et al., 1987)
515
ovothiol C
1986
Strongylocentrotus purpuratus
–
516
pucherrimine
2000
Hemicentrotus pulcherrimus
Japanese sea
(Murata and Sata, 2000)
517
fisherioside A
2012
Leptasterias fisheri
Sakhalin island, Sea of Okhotsk
(Kicha et al., 2012)
518
curacin E
2016
Ophiocoma scolopendrina
Kabira Reef, Ishigaki island, Okinawa, Japan
(Ueoka et al., 2016)
519
curacin A
2016
520
hypalocrinin A
2019
Hypalocrinus naresianus
Shima Spur, Kumano-nada Sea, Japan
(Wolkenstein et al., 2019)
521
hypalocrinin B
2019
522
hypalocrinin C
2019
523
hypalocrinin D
2019
524
hypalocrinin E
2019
525
hypalocrinin F
2019
526
hypalocrinin G
2019
527
microdiscusol A
2019
Asterias microdiscus
Eastern part of the Chukchi Sea,Arctic Ocean
(Kicha et al., 2019)
528
microdiscusol B
2019
529
microdiscusol C
2019
530
microdiscusol D
2019
531
microdiscusol E
2019
532
microdiscusol F
2019
1.2.2.5 Marine molluscs
Molluscs, a kind of soft marine animal usually with a calcareous shell, are the largest group of animals in the ocean, with more than 100,000 species, more than half of which live in the ocean. Mollusks have 7 classes, including Aplacophora, Bivalvia, Monoplacophora, Polyplacophora, Scaphopoda, Gastropoda and Cephalopoda. These mollusks are widely distributed, from the cold, temperate to tropical, from the highest point of the intertidal zone to 10,000 m deep at the bottom of the ocean.
16 (533–543, 82–83, 369, 381–382) sulphur-containing alkaloids were reported from the marine molluscs (Fig. 9 and Table 6). Notably, pteriatoxins A-C (535–537) are a group of cyclic imine toxins only isolated from Japanese shellfish, which can cause rapid death in mouse (Selwood et al., 2010). Now pteriatoxins have been considered emerging toxins in the European Union and a scientific opinion has been published by the European Food Safety Authority in which an assessment of the risks to human health related to their consumption has been carried out (Moreiras et al., 2020).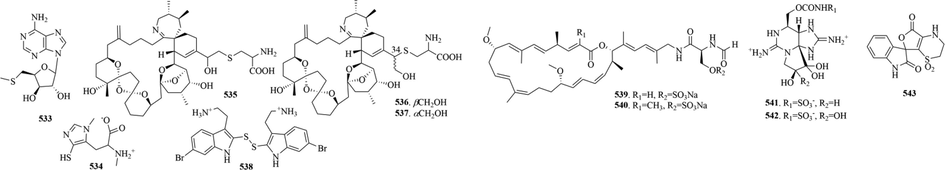
Sulphur-containing alkaloids from marine molluscs.
No.
Compounds
Time
From
Location
Ref.
Marine Molluscs
533
9-(5-deoxy-5-methylthio-β-D-xylofuranosyl)adenine
1986
Extracts of the digestive gland of the dorid nudibranchDoris verrucosa
–
(Faulkner, 1988)
534
ovothiol B
1986
Chlamys hastata
–
(Turner et al., 1987)
369
shermilamine B
1990
Chelynotus semperi
Mante Channel, Pohnpei,Federated States of Micronesia
(Carroll and Scheuer, 1990)
381
kuanoniamine A
1990
382
kuanoniamine B
1990
82
kuanoniamine C
1990
83
kuanoniamine D
1990
535
pteriatoxin A
2001
Pteria penguin
Okinawa, Japan
(Takada et al., 2001)
536
pteriatoxin B
2001
537
pteriatoxin C
2001
538
the disulfide-linked dimer of 6-bromo-2-mercaptotryptamine
2003
Calliostoma canaliculatum
Monterey Bay, California
(Kelley et al., 2003)
539
iejimalide C
2006
Eudistoma cf. rigida
Okinawa, Japan
(Kikuchi et al., 1991)
540
Iejimalide D
2006
541
11β-hydroxy-N-sulfocarbamoylsaxitoxin
2008
Wild mussels (Mytilus edulis andMytilus trossulus)
Eastern Canada coasts
(Dell’Aversano et al., 2008)
542
11,11-dihydroxy-N-sulfocarbamoylsaxitoxin
2008
543
orbicularisine
2017
Codakia orbicularis
Guadeloupe
(Goudou et al., 2017)
1.2.2.6 Marine bryozoans
Bryozoans are bryophyte-like animals, which had complete digestive apparatus, including the mouth, esophagus, stomach, intestines and anus. The individual bryozoans are small and undivided, with a body cavity. Their bones are formed by a layer of colloid which was secreted in vitro. They can devour microorganisms and organic impurities in water and have a positive effect on water purification.
10 (544–553) sulphur-containing alkaloids were reported from the marine bryozoans (Fig. 10 and Table 7). Perfragilin A (547) and B (545) were isolated from Membranipora perfragilis. As cytotoxic isoquinolines quinone, they contain a relatively uncommon thiomethyl ether functionality. And Both perfragilin A and B were toxic to murine leukemia cells (P388), with perfragilin B being considerably more potent: ED50= 0.8 and 0.07 μg/ml, respectively (Choi et al., 1993).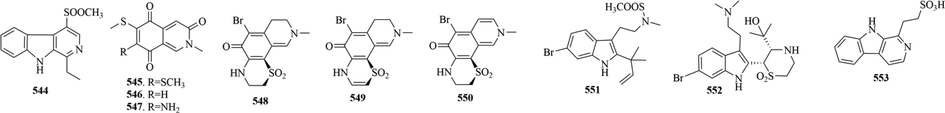
Sulphur-containing alkaloids from marine bryozoans.
No.
Compounds
Time
From
Location
Ref.
Marine Bryozoans
544
1-ethyl-4-methylsulfone-β-carboline
1991
Cribricellina cribraria
New Zealand
(Prinsep et al., 1991)
545
perfragilin B
1993
Membranipora perfragilis
Rapid Bay, South Australia
(Choi et al., 1993)
546
2-methyl-6-methylthioisoquinoline-3,5,8(2H)trione
1993
Blflustra perfragilis
Bass Strait
(Blackman et al., 1993)
547
perfragilin A
1993
Membranipora perfragilis
Rapid Bay, South Australia
(Choi et al., 1993)
548
euthyroideone A
1998
Euthyroides episcopalis
Fiordland, New Zealand
(Morris and Prinsep, 1998)
549
euthyroideone B
1998
550
euthyroideone C
1998
551
N-(2-[6-bromo-2-(1,1-dimethyl-2-propenyl)-1H-indol-3-yl]ethyl)-N-methy-lmethanesulfonamide
2002
Flustra foliacea
“Steingrund”, North Sea, Helgoland, Germany
(Peters et al., 2002)
552
flustramine R
2020
Flustra foliacea
Iceland
(Di et al., 2020)
553
orthoscuticelline E
2020
Orthoscuticella ventricosa
Korora beach, Coffs Harbour, NSW, Australia
(Kleks et al., 2020)
1.2.2.7 Other marine animals
22 (554–575) sulphur-containing alkaloids were reported from other marine animals (Fig. 11 and Table 8). Of them, nebulosins A-P (560–575) were reported from the northeastern Atlantic marine terebellid Eupolymnia nebulosa. It’s worth noting that nebulosins feature an unprecedented highly substituted thiolane ring leading to up to four contiguous chiral centers (Calabro et al., 2020).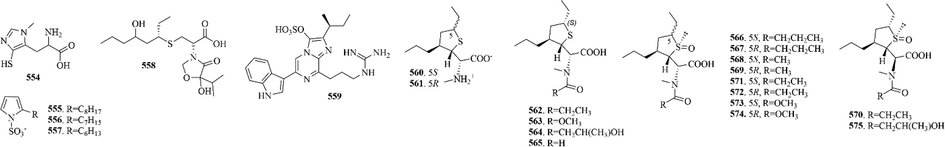
Sulphur-containing alkaloids from other marine animals.
No.
Compounds
Time
From
Location
Ref.
Other marine animals
554
L-ovithiol A
1999
Platynereis dumerilii
–
(Röhl et al., 1999)
555
2-n-octylpyrrole sulfamate
2003
Cirriformia tentaculata
Florida
(Barsby et al., 2003)
556
2-n-heptylpyrrole sulfamate
2003
557
2-n-hexylpyrrole sulfamate
2003
558
thelepamide
2014
Thelepus crispus
Friday Harbor, WA, U.S.A.
(Rodríguez et al., 2014)
559
cypridina luciferyl sulfate
2014
Vargula hilgendorfii
Chita, Aichi, Japan
(Nakamura et al., 2014)
560
nebulosin A
2020
Eupolymnia nebulosa
Intertidal area of Corranroo, West coast of Ireland
(Calabro et al., 2020)
561
nebulosin B
2020
562
nebulosin C
2020
563
nebulosin D
2020
564
nebulosin E
2020
565
nebulosin F
2020
566
nebulosin G
2020
567
nebulosin H
2020
568
nebulosin I
2020
569
nebulosin J
2020
570
nebulosin K
2020
571
nebulosin L
2020
572
nebulosin M
2020
573
nebulosin N
2020
574
nebulosin O
2020
575
nebulosin P
2020
1.3 Marine microorganism
1.3.1 Dinoflagellates
Dinoflagellates are a group of single cells with double flagella, whose shape is variable. They have both plant and animal characteristics, which could perform photosynthesis and move by the rotation of two flagella. Dinoflagellates are widely distributed, especially in tropical oceans. When the light and water temperature are appropriate, dinoflagellates can multiply in a short period of time to become the main feed of marine animals.
13 (576–588) sulphur-containing alkaloids were reported from dinoflagellates (Fig. 12 and Table 9). Among them, compounds 576–585 are thought as the carbamoyl-N-sulfo derivatives of saxitoxin and neosaxitoxin (Hall et al., 1984). In addition, it’s worth noting that symbioimine (586) and neosymbioimine (587) both have a characteristic 6,6,6-tricyclic iminium ring structure and an aryl sulfate moiety. And the plausible biogenetic pathway of them can be explained by an intramolecular Diels-Alder reaction followed by imine cyclization (Kita et al., 2005).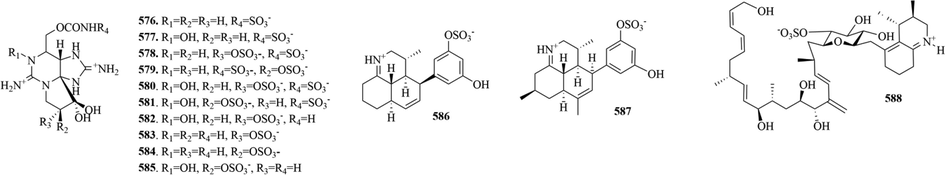
Sulphur-containing alkaloids from dinoflagellates.
No.
Compounds
Time
From
Location
Ref.
Dinoflagellates
576
toxin B1
1984
Protugonyaulax catenella
Northeast Pacific
(Hall et al., 1984)
577
toxin B2
1984
578
toxin C1
1984
579
toxin C2
1984
580
toxin C3
1984
581
toxin C4
1984
582
GTX-1
1984
583
GTX-2
1984
584
GTX-3
1984
585
GTX-4
1984
586
symbioimine
2004
Symbiodinium sp.
Sesoko Island, Okinawa, Japan.
(Kita et al., 2004)
587
neosymbioimine
2005
(Kita et al., 2005)
588
ovataline
2022
Ostreopsis cf. ovata
Kimyong, Jeju island
(Lee et al., 2022)
1.3.2 Cyanobacteria
Cyanobacteria, also known as blue-green algae, are large, single-celled prokaryotes with a long evolutionary history. They have chlorophyll which enabled them to perform oxygen-producing photosynthesis. The photosynthesis of cyanobacteria is also thought as the reason why the earth's atmosphere develops from an anaerobic state to an aerobic state. At present, there are about 2000 species of cyanobacteria, which are mainly divided into two classes: Chroococcus and Phytoplankton. As highly adaptable organisms, they are widely distributed in all kinds of natural water bodies, soil and some organisms, even in the rock surface and other harsh environments.
41 (589–627, 61, 519) sulphur-containing alkaloids were reported from cyanobacteria (Fig. 13 and Table 10). Curacins A-D (519, 591–592 and 594) are toxic metabolites isolated from the cyanobacteria, which are thought of as antimitotic agents. In addition, lyngbyabellins are a kind of depsipeptide derivatives, whose typical structural features are two thiazole rings and a chlorinated 2-methyloctanoate residue (Choi et al., 2012). They generally display various activities such as cytotoxicity, antimalarial, and antifouling activities (Fathoni et al., 2020). Lyngbyabellins O (613) and P (614) both exhibit strong antifouling activity, which may be related to the fact that compounds don’t have a side chain (Petitbois et al., 2017). Notably, aulosirazoles A-C (623–625) are the structurally unique isothiazolonaphthoquinone aulosirazole, which possess selective antitumor cytotoxicity. Although its mechanism of action is unknown, biological evaluation of them identified one potential target as the immunoregulatory enzyme indoleamine-2,3-dioxygenase (IDO) (Blunt et al., 2015).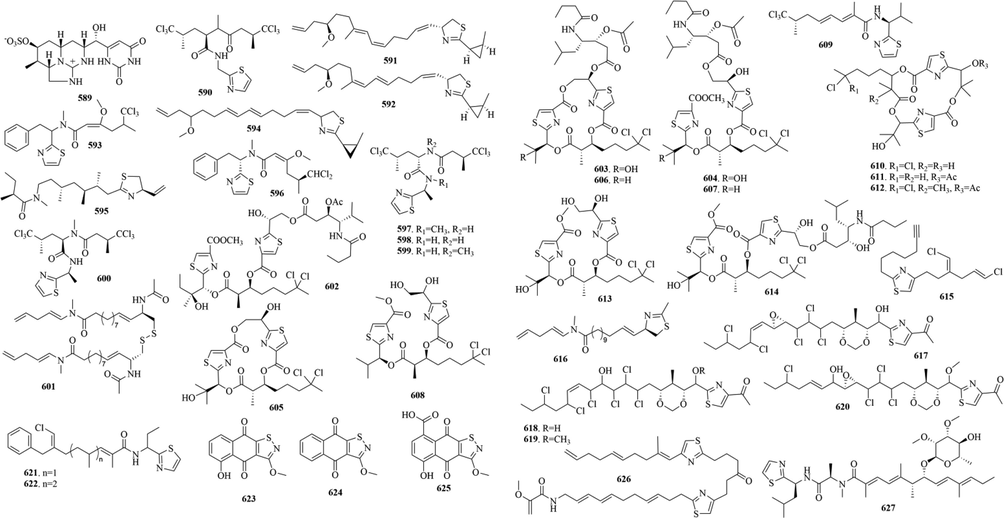
Sulphur-containing alkaloids from cyanobacteria.
No.
Compounds
Time
From
Location
Ref.
Cyanobacteria
589
cylindrospermopsin
1992
Cylindrospermopsis raciborskii
Palm Island, Queensland, Australia
(Ohtani et al., 1992)
590
13-demethylisodysidenin
1993
Oscillatoria spongeliae
–
(Faulkner, 1995)
591
curacin B
1995
Lyngbya majuscula
Curaçao, Caribbean sea
(Yoo and Gerwick, 1995)
592
curacin C
1995
593
barbamide
1996
(Orjala and Gerwick, 1996)
519.
curacin A 519
1998
Virgin Islands, British
(Márquez et al., 1998)
594
curacin D
1998
595
kalkitoxin
2000
Curaçao, Caribbean sea
(Yokokawa et al., 2004)
596
dechlorobarbamide
2000
(Sitachitta et al., 2000)
597
pseudodysidenin
2001
Lyngbya majuscula
Boca del Drago Beach, Bocas del Toro, Panama
(Jiménez and Scheuer, 2001)
598
nordysidenin
2001
599
dysidenin
2001
600
isodysidenin
2001
61.
dysideathiazole 61
2001
601
somocystinamide A
2002
Lyngbya majuscula andSchizothrix sp.
Fijian
(Nogle and Gerwick, 2002)
602
lyngbyabellin D
2003
Lyngbya sp.
Guam, U.S.A.
(Williams et al., 2003)
603
lyngbyabellin E
2005
Lyngbya majuscula
Alotau Bay, Papua New Guinea
(Han et al., 2005)
604
lyngbyabellin F
2005
605
lyngbyabellin G
2005
606
lyngbyabellin H
2005
607
lyngbyabellin I
2005
608
dolabellin
2005
609
herbamide B
2010
Bocas del Toro, Panama
(Balunas et al., 2010)
610
hectochlorin B
2015
Moorea producens
–
(Paul and Boudreau, 2015)
611
hectochlorin C
2015
612
hectochlorin D
2015
613
lyngbyabellin O
2017
Okeania sp.
Algetah Alkabira reef, Jeddah, Saudi Arabia
(Petitbois et al., 2017)
614
lyngbyabellin P
2017
615
trichothiazole A
2017
Trichodesmium sp.
Gulf of Mexico
(Belisle et al., 2017)
616
laucysteinamide A
2017
Caldora penicillata
Lau Lau Bay, Saipan
(Zhang et al., 2017a)
617
aranazole A
2018
Fischerella sp. PCC 9339
–
(Moosmann et al., 2018)
618
aranazole B
2018
619
aranazole C
2018
620
aranazole D
2018
621
isoconulothiazole B
2019
Trichodesmium sp.
Mayaguana Island, Bahamas
(Teta et al., 2019)
622
conulothiazole C
2019
623
aulosirazole A
2022
Nostoc sp. UIC 10771
Reykjavık, Iceland
(Davis et al., 2022)
624
aulosirazole B
2022
625
aulosirazole C
2022
626
caldorazole
2022
Caldora sp.
Ishigaki Island, Okinawa, Japan
(Ohno et al., 2022)
627
iezoside
2022
Leptochromothrix valpauliae
Ie Island, Okinawa, Japan,
(Kurisawa et al., 2022)
1.3.3 Marine bacteria
Marine bacteria are the most important members of marine microorganisms, which are widely distributed and abundant in the ocean. The common bacteria include Pseudomonas, Vibrio, Achromobacter, Nocardia and Streptomyces. Almost all known bacteria can be found in the marine environment. Meanwhile, most marine bacteria are decomposers, which play an important role in the whole process of marine material decomposition and transformation. Moreover, because the deep-sea environment has the characteristics of high salt, high pressure, low temperature and low nutrition, the physiological and ecological characteristics of deep-sea bacteria are very different from those of terrestrial bacteria. This is also the reason why scientists are paying more attention to deep-sea bacteria.
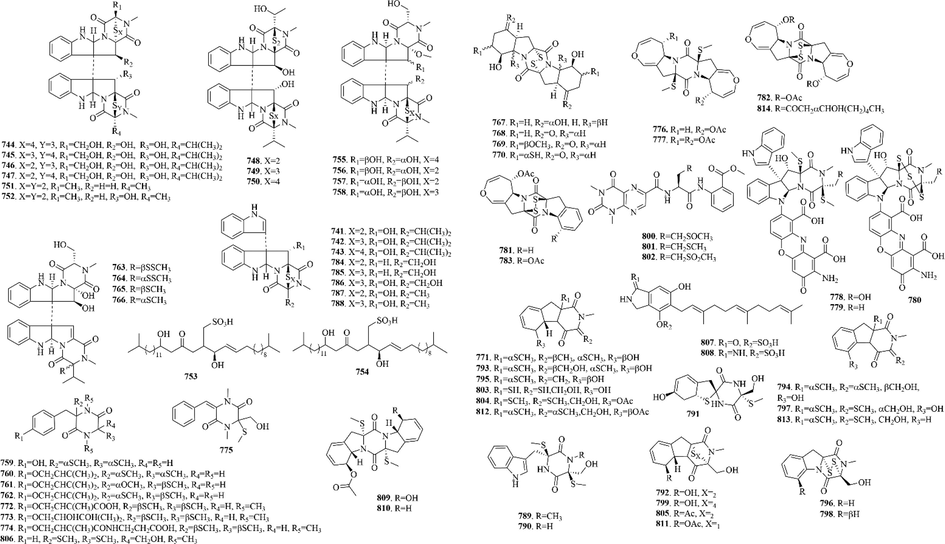
110 (628–737) sulphur-containing alkaloids were reported from marine bacteria (Fig. 14 and Table 11). Notably, thiomarinols are a kind of naturally occurring double‐headed antibiotic, whose structure comprises two antimicrobial subcomponents, pseudomonic acid analogue and holothin, linked by an amide bond (Dunn et al., 2015). Such ingredients usually have excellent antibacterial activity and can even be effective against MRSA (Shiozawa et al., 1995). And sulfadixiamycins A-C (685–687), sulfonyl‐bridged alkaloid dimers, are isolated from recombinant Streptomyces species. They have both aromatic sulfonamide and diarylsulfone substructures. In addition, sungeidines A-H (724–731), a class of microbial secondary metabolites with unique structural features, are likely to be assembled from two octaketide chains following processing by oxygenases/oxidases and cyclases.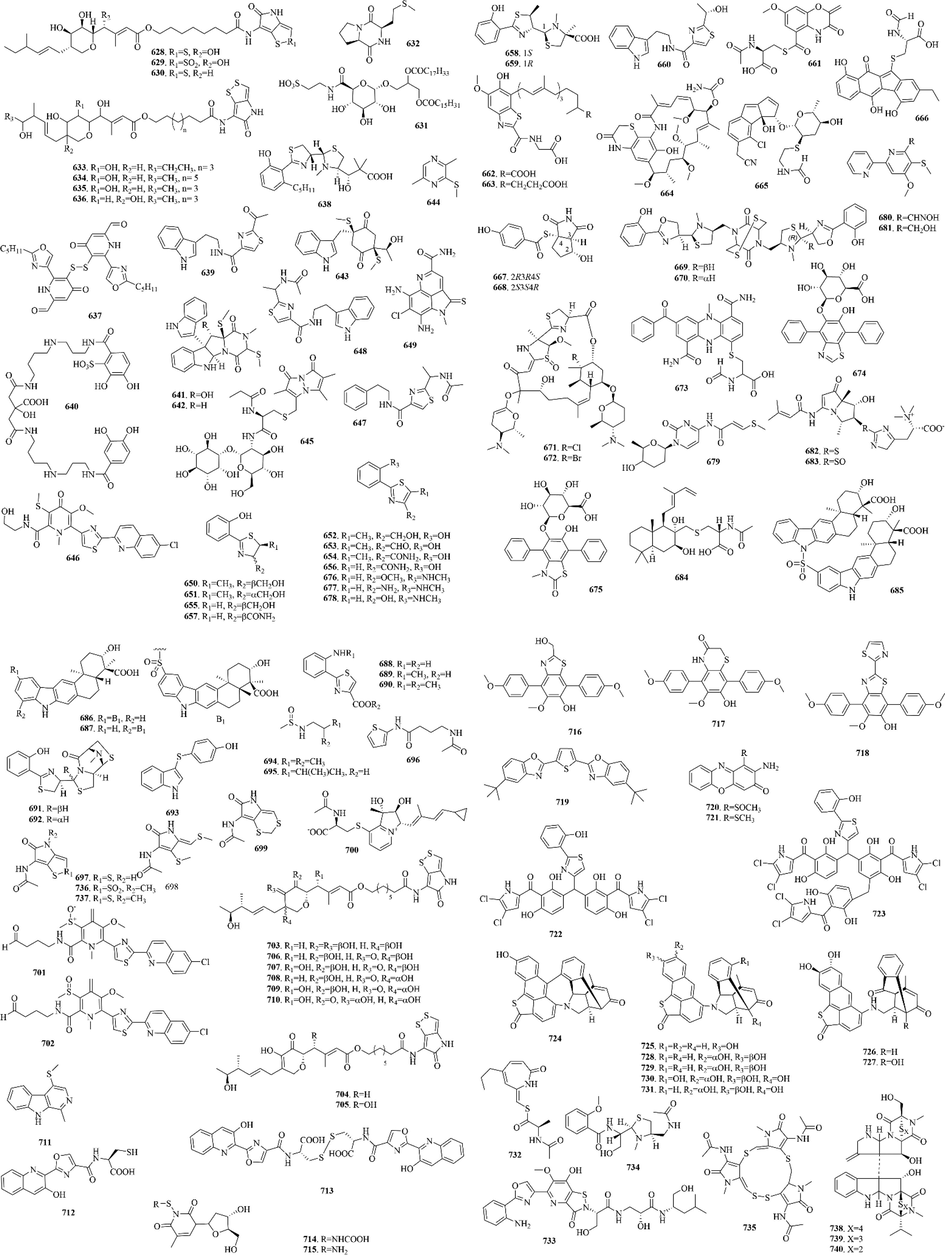
Sulphur-containing alkaloids from marine bacteria.
Sulphur-containing alkaloids from marine bacteria.
No.
Compounds
Time
From
Location
Ref.
Marine bacteria
628
thiomarinol A(Thiomarinol)
1993
Alteromonas raw sp. nov. SANK 73390
–
(Shiozawa et al., 1993)
629
thiomarinol B
1995
(Shiozawa et al., 1995)
630
thiomarinol C
1995
631
1,2-diacyl-3-α-d-glucuronopyranosyl-sn-glycerol taurineamide
1996
Hyphomonas jannaschiana
–
(Batrakov et al., 1996)
632
cyclo(L-Pro-L-Met)
1996
Pseudomonas aeruginosa
Ross Island, Antarctica
(Jayatilake et al., 1996)
633
thiomarinol D
1997
Alteromonas raw sp. nov. SANK 73390
–
(Shiozawa et al., 1997)
634
thiomarinol E
1997
635
thiomarinol F
1997
636
thiomarinol G
1997
637
B-90063
1998
Blastobacter sp. SANK 71894
Japan
(Sachiko Takaishi et al., 1998)
638
agrochelin
1999
Agrobacterium sp.
–
(Cañedo et al., 1999)
639
bacillamide A
2011
Bacillus sp.
Masan Bay, Korea.
(Zou et al., 2011)
640
petrobactin sulfonate
2004
Marinobacter hydrocarbonoclasticus
–
(Hickford et al., 2004)
641
gliocladin A
2004
Gliocladium sp.
Coast of Kata, Wakayama Prefecture, Japan
(Yoshihide Usami, 2004)
642
gliocladin B
2004
643
glioperazine
2004
644
2,5-dimethyl-3-(methylsulfanyl) pyrazine
2005
Alphaproteobacteria Loktanella sp.
North Sea
(Dickschat et al., 2005)
645
N-propionyl-desacetyl-mycothiol
2008
MAR2 strain CNQ703
Guam
(Newton et al., 2008)
646
lodopyridone
2009
Saccharomonospora sp.
La Jolla, California
(Maloney et al., 2009)
647
neobacillamide A
2009
Bacillus vallismortis
Sanya island, South China Sea
(Yu et al., 2009)
648
bacillamide C
2009
649
ammosamide A
2009
Streptomyces sp.
Bahamas
(Hughes et al., 2009)
650
pulicatin A
2010
Streptomyces sp.
Mactan island, Cebu, Philippines
(Lin et al., 2010)
651
pulicatin B
2010
652
pulicatin C
2010
653
pulicatin D
2010
654
pulicatin E
2010
655
aerugine
2010
656
pulicatin F
2010
657
pulicatin G
2010
658
watasemycin A
2010
659
watasemycin B
2010
660
bacillamide B
2009
Bacillus endophyticus
Sanya island, South China Sea
(Yu et al., 2009),(Sun et al., 2015)
661
benzoxacystol
2011
Streptomyces griseus
deep sea sediment, Canary Basin
(Nachtigall et al., 2011)
662
erythrazole A
2011
Erythrobacter sp.
Trinity Bay, Galveston, Texas, U.S.A.
(Hu and MacMillan, 2011)
663
erythrazole B
2011
664
heronamycin A
2012
Streptomyces sp.
Heron island, Queensland, Australia
(Raju et al., 2012)
665
cyanosporaside F
2013
Streptomyces sp.
Bahamas
(Lane et al., 2013)
666
(−)-homoseongomycin
2013
Salinispora pacifica DPJ-0019
–
(Woo et al., 2013)
667
nitrosporeusine A
2013
Streptomyces nitrosporeus
Arctic Chukchi Sea
(Yang et al., 2013)
668
nitrosporeusine B
2013
669
tetroazolemycin A
2013
Streptomyces olivaceus
southwest Indian Ocean
(Liu et al., 2013)
670
tetroazolemycin B
2013
671
forazoline A
2014
Actinomadura sp. WMMB-499
Florida, U.S.A.
(Wyche et al., 2014)
672
forazoline B
2014
673
dermacozine J
2014
Dermacoccus abyssi
Challenger Deep, Mariana Trench
(Wagner et al., 2014)
674
echoside D
2014
Streptomyces sp.
Jimei, China
(Deng et al., 2014)
675
echoside E
2014
676
anithiactin A
2014
Streptomyces sp.
Jaebu island, South Korea
(Kim et al., 2014b)
677
anithiactin B
2014
678
anithiactin C
2014
679
streptcytosine B
2014
Streptomyces sp.
Iriomote island, Japan
(Bu et al., 2014)
680
collismycin B
2014
681
SF2738 C
2014
682
spithioneine A
2015
Streptomyces spinoverrucosus
Bahamas
(Fu and MacMillan, 2015a)
683
spithioneine B
2015
684
N-acetyl-S-(((1R,2S,3S,4aS,8aS)-2,3-dihydroxy-5,5,8a-trimethyl-1-((E)-3-methylpenta-2,4-dien-1-yl)decahydronaphthalen-2-yl)methyl)-L-cysteine
2015
Streptomyces sp.
Parangipettai, India
(Shanthi et al., 2015)
685
sulfadixiamycin A
2015
Streptomyces sp.
–
(Baunach et al., 2015)
686
sulfadixiamycin B
2015
687
sulfadixiamycin C
2015
688
thiasporine A
2015
Actinomycetospora chlora
Vava’u, Tonga
(Fu and MacMillan, 2015b),(Seitz et al., 2016)
689
thiasporine B
2015
690
thiasporine C
2015
691
ulbactin F
2016
Brevibacillus sp.
Ohtsuchi, Iwate, Japan
(Igarashi et al., 2016)
692
ulbactin G
2016
693
4-(1H-indol-3-yl-sulfanyl)phenol
2016
Vibrio splendidus
S. Orkney island
(Nair et al., 2016)
694
N-isobutylmethanesulfinamide
2017
Salinispora pacifica
Fiji
(Harig et al., 2017)
695
N-isopentylmethanesulfinamide
2017
696
3-acetylamino-N-2-thienyl-propanamide
2017
Streptomyces sp. Q24
Zhuhai, Guangdong, China
(Ye et al., 2017)
697
holomycin
2017
Streptomyces sp. DT-A37
Dongtou, Wenzhou, Zhejiang Province, P. R. China
(Ding et al., 2017)
698
(1Z)-S,S'-dimethyldihydroholomycin
2017
699
holomycin A
2017
700
streptopertusacin A
2017
Streptomyces sp. HZP-2216E
Turtle Islet located,South China Sea
(Zhang et al., 2017b)
701
lodopyridone B
2017
Saccharomonospora sp.
La Jolla Submarine Canyon, California
(Le et al., 2017)
702
lodopyridone C
2017
703
8-hydroxythiomarinol C
2017
Pseudoalteromonas sp.
–
(Gao et al., 2017)
704
6,7-diketothiomarinol C
2017
705
6,7-diketothiomarinol A
2017
706
7-ketothiomarinol C
2017
707
7-ketothiomarinol A
2017
708
8-epi-7-ketothiomarinol C
2017
709
8-epi-7-ketothiomarinol A
2017
710
8-epi-7-epi-6-ketothiomarinol A
2017
711
1-methyl-4-methylthio-β-carboline
2017
Pseudomonas benzenivorans
California State Beaches
(Lorig-Roach et al., 2017)
712
(2-(3-hydroxyquinolin-2-yl)oxazole-4-carbonyl)-L-cysteine
2018
Streptomyces cyaneofuscatus M−157
Avilés Canyon, Cantabrian Sea
(Ortiz-López et al., 2018)
713
(2R,2′R)-3,3′-disulfanediylbis(2-(2-(3-hydroxyquinolin-2-yl)oxazole-4-carboxamido)propanoic acid)
2018
714
thymidine-3-mercaptocarbamic acid
2019
Streptomyces sp.
Red Sea
(Shaala et al., 2019)
715
thymidine-3-thioamine
2019
716
nocarterphenyl A
2019
Nocardiopsis sp. OUCMDZ-4936
Dongzhaigang Mangrove Reserve, China
(Wang et al., 2019)
717
nocarterphenyl B
2019
718
nocarterphenyl D
2021
Nocardiopsis sp. HDN154086
South China Sea
(Chang et al., 2021)
719
2,5-bis(5-tert-butyl-2-benzoxazolyl)thiophene
2019
Streptomyces sp. G278
Cu Lao Cham - Quang Nam, Vietnam
(Cao et al., 2019a)
720
questiomycin C
2019
Alteromonas sp. D
Hiroshima-bay, Hiroshima, Japan
(Umetsu et al., 2019)
721
questiomycin D
2019
722
mindapyrrole B
2019
Pseudomonas aeruginosa
Sultan Kudarat, Mindanao, Philippines
(Lacerna et al., 2019)
723
mindapyrrole C
2019
671
forazoline A
2020
Actinomadura sp. WMMB-499
–
(Zhang et al., 2020a)
724
sungeidine A
2020
Micromonospora sp.
Sungei Buloh Wetland Reserve, Singapore
(Low et al., 2020)
725
sungeidine B
2020
726
sungeidine C
2020
727
sungeidine D
2020
728
sungeidine E
2020
729
sungeidine F
2020
730
sungeidine G
2020
731
sungeidine H
2020
732
monathioamide A
2020
Pseudomonas sp. ZZ820R
Zhoushan Archipelago, Zhejiang, China
(Yi et al., 2020a)
733
levesquamide
2020
Streptomyces sp. RKND-216
Burnt Point, PE, Canada
(Liang et al., 2020)
734
streptothiazomycin A
2020
Streptomyces sp. SY1965
Mariana Trench
(Yi et al., 2020b)
735
thiolopyrrolone A
2022
Streptomyces sp. BTBU20198885
Xiamen, China
(Song et al., 2022)
736
2,2-dioxidothiolutin
2022
737
thiolutin
2022
1.3.4 Marine fungi
The distribution of fungi in the ocean mainly depended on the distribution of hosts. According to their habitat habits, marine fungi could be divided into four basic ecological types: (1) woody fungi. The largest number and most widely distributed higher fungi in marine waters is saprophytic life. (2) Parasitic algae fungi. It accounted for about 1/3 of the number of marine fungal species, most of which were ascomycetes. (3) Seaweed fungi. The number of seaweed fungi was small and mostly inhabited the leaves. (4) Parasitic animal fungi. Only parasitic in the exoskeleton and shell. Marine fungi participate in the decomposition of marine organic matter and the regeneration of inorganic nutrients and continuously provide effective nutrition for marine plants.
197 (738–934) sulphur-containing alkaloids were reported from marine fungi (Fig. 15 and Table 12). It’s worth noting that leptosins, amphiepicoccins (912–921), penicisulfuranols (869–874), gliotoxins and chetracins E, F, C (875, 876, 877) all are epipolythiodioxopiperazines (ETPs) which are a class of biologically active fungi secondary metabolites characterized by a unique bridged disulfide or polysulfide dioxopiperazine six-membered ring (Gardiner et al., 2005). These compounds occur in many fungi. And due to their broad spectra of bioactivities, ETPs have drawn wide attention in recent years (Jiang and Guo, 2011). Moreover, graphiumins A-J (814–823), rostratins A-D (767–770), aranotins (777, 781–783, 904) and eutypellazines A-S (849–866) all are thiodiketopiperazine alkaloids. Among them, eutypellazines N-P (861–863) are characteristic of unique spirocyclic skeletons. Meanwhile, eutypellazines N-O bearing a spirocyclic tetrahydro- benzothiophene motif is found in wide-type fungus for the first time (Niu et al., 2017b). In summary, thiodioxopiperazines are the main type of sulphur-containing alkaloids in fungi.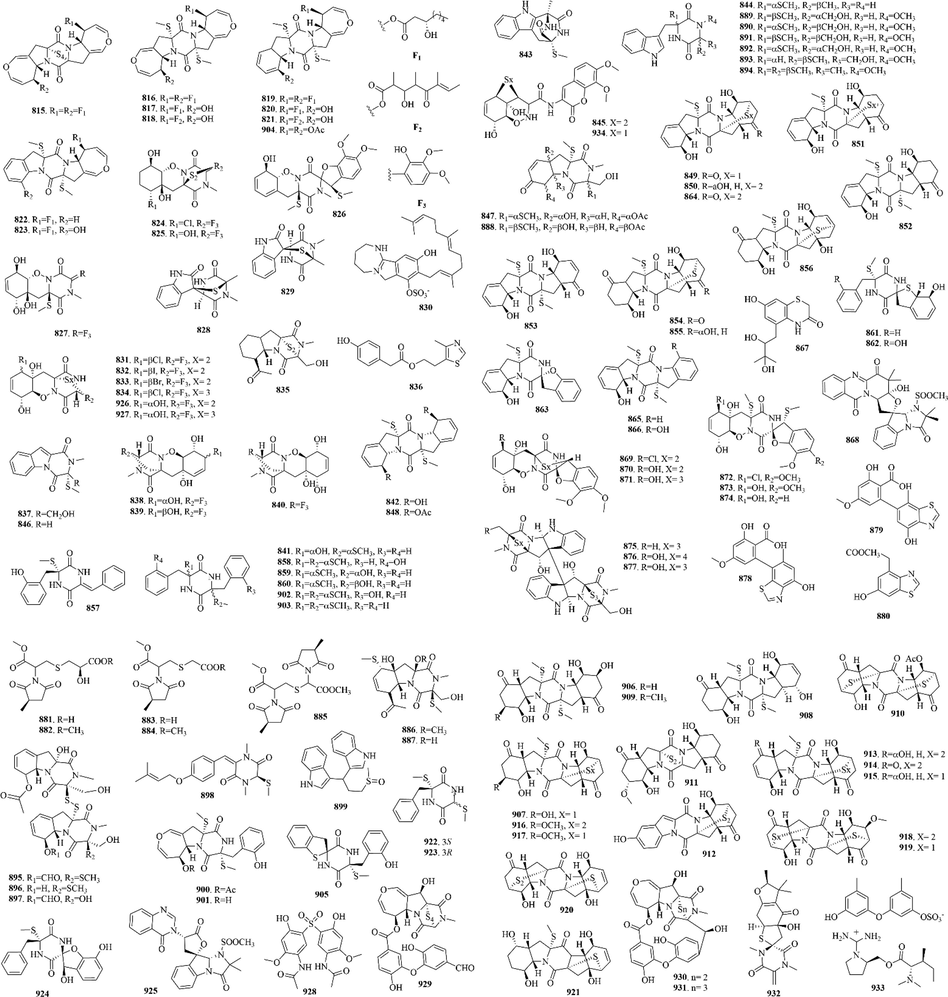
Sulphur-containing alkaloids from marine fungi.
No.
Compounds
Time
From
Location
Ref.
Marine fungi
738
leptosin A
1994
Leptosphueriu sp.
Tanabe Bay, Japan
(Takahashi et al., 1994)
739
leptosin B
1994
740
leptosin C
1994
741
leptosin D
1994
742
leptosin E
1994
743
leptosin F
1994
744
leptosin G
1995
(Takahashi et al., 1995b)
745
leptosin G1
1995
746
leptosin G2
1995
747
leptosin H
1995
748
leptosin K
1995
(Takahashi et al., 1995a)
749
leptosin K1
1995
750
leptosin K2
1995
751
11,11′-dideoxyverticillin A
1999
Penicillium sp. CNC-350
–
(John, 1999)
752
11′-deoxyverticillin A
1999
753
flavochristamide A
2000
Flavobacterium sp.
lshikari Bay, Hokkaido
(Kohayashi et al., 1995)
754
flavochristamide B
2000
755
leptosin M
2002
Leptosphaeria sp.
Tanabe Bay, Japan
(Yamada et al., 2002)
756
leptosin M1
2002
757
leptosin N
2002
758
leptosin N1
2002
759
fusaperazine A
2002
Fusarium chlamydosporum
–
(Blunt et al., 2004)
760
fusaperazine B
2002
761
Sch 54,794
1993
(Chu et al., 1993)
762
Sch 54,796
1993
763
leptosin O
2004
Leptosphaeria sp.
Tanabe Bay, Japan
(Takeshi Yamada, 2004)
764
leptosin P
2004
765
leptosin Q
2004
766
leptosin R
2004
767
rostratin A
2004
Exserohilum rostratum
Lanai Island, Hawaii
(Tan et al., 2004)
768
rostratin B
2004
769
rostratin C
2004
770
rostratin D
2004
771
dehydroxybisdethiobis(methylthio)gliotoxin
2006
Pseudallescheria sp.
Uljin, Gyeongbuk, Korea
(Li et al., 2006)
772
bilain A
2007
Penicillium bilaii
Huon estuary, Tasmania
(Capon et al., 2007)
773
bilain B
2007
774
bilain C
2007
775
(Z)-6-benzylidene-3-hydroxymethyl-1,4-dimethyl-3-methylsulfanylpiperazine-2,5-dione
2008
Order Pleosporales CRIF2
Surin Island
(Prachyawarakorn et al., 2008)
776
alternarosin A
2009
Alternaria raphani
Qingdao, China
(Wang et al., 2009)
777
bisdethiobis(methylthio)acetylaranotin
2009
778
plectosphaeroic acid A
2009
Plectosphaerella cucumerina
Barkley Sound, British Columbia
(Carr et al., 2009)
779
plectosphaeroic acid B
2009
780
plectosphaeroic acid C
2009
781
deoxyapoaranotin
2011
Arthrinium versicolor
East Sea, Korea
(Choi et al., 2011)
782
acetylaranotin
2011
783
acetylapoaranotin
2011
784
luteoalbusin A
2012
Acrostalagmus luteoalbus
South China Sea
(Wang et al., 2012a)
785
luteoalbusin B
2012
786
T988A
2012
787
gliocladines C
2012
788
gliocladines D
2012
789
chetoseminudin B
2012
790
chetoseminudin C
2012
791
spirogliotoxin
2012
Aspergillus fumigatus YK-7
Yingkou, China
(Wang et al., 2012b)
792
gliotoxin
793
bisdethiobis(methylthio)gliotoxin
794
didehydrobisdethiobis(methylthio)gliotoxin
795
bis(dethio)-10a-methylthio-3a-deoxy-3,3a-didehydrogliotoxin
2012
Penicillium sp.
Suruga Bay, Japan
(Sun et al., 2012)
796
6-deoxy-5a,6-didehydrogliotoxin
2012
797
bis(dethio)bis(methylthio)-5a,6-didehydrogliotoxin
2012
798
5a,6-didehydrogliotoxin
2012
799
gliotoxin G
2012
800
penilumamide
2014
Aspergillus sp.
Xisha islands,South China Sea
(Reddy et al., 2017)
801
penilumamide B
2014
802
penilumamide C
2014
803
reduced gliotoxin
2014
Neosartorya pseudofischeri
Hainan Sanya National Coral Reef Reserve, China
(Liang et al., 2014)
804
acetylgliotoxin
805
bis-N-norgliovictin
806
6-acetylbis(methylthio)gliotoxin
807
chartarutine C
2014
Stachybotrys chartarum
Beibuwan Bay, China
(Li et al., 2014)
808
chartarutine D
2014
809
cladosporin A
2015
Cladosporium sp.
Yangshashan Bay, Ningbo, Zhejiang, China
(Gu et al., 2015)
810
cladosporin B
2015
811
6-acetylmonodethiogliotoxin
2015
Dichotomomyces cejpii
Bare island, Sydney, Australia
(Harms et al., 2015)
812
6-acetylbisdethiobis(methylthio)gliotoxin
2015
813
5a,6-anhydrobisdethiobis(methylthio)-gliotoxin
2015
814
graphiumin A
2015
Graphium sp.
Ishigaki island, Okinawa, Japan
(Fukuda et al., 2015)
815
graphiumin B
2015
816
graphiumin C
2015
817
graphiumin D
2015
818
graphiumin E
2015
819
graphiumin F
2015
820
graphiumin G
2015
821
graphiumin H
2015
822
graphiumin I
2015
823
graphiumin J
2015
824
adametizine A
2015
Penicillium adametzioides
Hainan island, China
(Liu et al., 2015b)
825
adametizine B
2015
826
peniciadametizine A
2015
Penicillium adametzioides
Wenchang, Hainan, China
(Liu et al., 2015c)
827
peniciadametizine B
2015
828
pseudellone A
2015
Pseudallescheria ellipsoidea F42 − 3
National Coral Reef Reserve, Hainan, China
(Liu et al., 2015a)
829
pseudellone B
2015
830
stachybotrin G
2015
Stachybotrys chartarum MXH-X73
Xisha island, China
(Ma et al., 2015)
831
DC1149B
2015
Trichoderma cf. brevicompactum
Palau
(Yamazaki et al., 2015a)
832
iododithiobrevamide
2015
833
DC1149R
2015
834
chlorotrithiobrevamide
2015
(Yamazaki et al., 2015b)
835
acetylgliotoxin G
2015
Dichotomomyces cejpii
Pecém’s offshore port, Ceará, Brazil
(Rodrigues et al., 2015)
836
acaromyester A
2016
Acaromyces ingoldii
South China Sea
(Gao et al., 2016)
837
dichotocejpin A
2016
Dichotomomyces cejpii FS110
South China Sea
(Fan et al., 2016)
838
pretrichodermamide D
2016
Penicillium sp. KMM 4672
Vietnam, South China Sea
(Yurchenko et al., 2016)
839
pretrichodermamide E
2016
840
pretrichodermamide F
2016
841
pseuboydone C
2016
Pseudallescheria boydii
Hainan Sanya National Coral Reef Reserve, China
(Lan et al., 2016)
842
pseuboydone D
2016
843
lasiodipline F
2016
Pseudallescheria ellipsoidea F42-3
(Wang et al., 2016)
844
pseudellone D
2016
845
dithioaspergillazine A
2016
Trichoderma cf. brevicompactum
Palau
(Yamazaki et al., 2016)
846
dichocerazine A
2017
Dichotomomyces cejpii F31-1
Hainan Sanya National Coral Reef Reserve, China
(Chen et al., 2017b)
847
dichocerazine B
2017
848
haematocin
2017
849
eutypellazine A
2017
Eutypella sp. MCCC 3A00281
South Atlantic Ocean
(Niu et al., 2017a)
850
eutypellazine B
2017
851
eutypellazine C
2017
852
eutypellazine D
2017
853
eutypellazine E
2017
854
eutypellazine F
2017
855
eutypellazine G
2017
856
eutypellazine H
2017
857
eutypellazine I
2017
858
eutypellazine J
2017
859
eutypellazine K
2017
860
eutypellazine L
2017
861
eutypellazine N
2017
(Niu et al., 2017b)
862
eutypellazine O
2017
863
eutypellazine P
2017
864
eutypellazine Q
2017
865
eutypellazine R
2017
866
eutypellazine S
2017
867
gliomastin B
2017
Gliomastix sp.
Ain El-Sokhna, Eygpt
(Elnaggar et al., 2017)
868
scedapin C
2017
Scedosporium apiospermum
Hainan Sanya National Coral Reef Reserve, China
(Huang et al., 2017)
869
penicisulfuranol A
2017
Penicillium janthinellum HDN13-309
Hainan, China
(Zhu et al., 2017)
870
penicisulfuranol B
2017
871
penicisulfuranol C
2017
872
penicisulfuranol D
2017
873
penicisulfuranol E
2017
874
penicisulfuranol F
2017
875
chetracin E
2018
Acrostalagmus luteoalbus HDN13-530
Liaodong Bay, China
(Yu et al., 2018)
876
chetracin F
2018
877
chetracin C
2018
878
altenusinoide A
2018
Alternaria sp. SCSIOS02F49
Xuwen County, Guangdong, China
879
altenusinoide B
2018
880
methyl 2-(6-hydroxybenzothiazol-4-yl) acetate
2018
881
violaceimide A
2018
Aspergillus violaceus
South China Sea
(Yin et al., 2018)
882
violaceimide B
2018
883
violaceimide C
2018
884
violaceimide D
2018
885
violaceimide E
2018
886
geospallin A
2018
Geosmithia pallida FS140
(Sun et al., 2018)
887
geospallin B
2018
888
geospallin C
2018
889
(+)-acrozine A
2019
Acrostalagmus luteoalbus TK-43
Sinop, Turkey
(Cao et al., 2019b)
890
(–)-acrozine A
2019
891
(+)-acrozine B
2019
892
(–)-acrozine B
2019
893
acrozine F
2021
(Cao et al., 2021)
894
acrozine G
2021
895
dechdigliotoxin A
2019
Dichotomomyces cejpii
South China Sea
(Liu et al., 2019b)
896
dechdigliotoxin B
2019
897
dechdigliotoxin C
2019
898
fusaperazine F
2019
Penicillium crustosum HDN153086
Prydz Bay, Antarctica
(Liu et al., 2019a)
899
pseudboindole B
2019
Pseudallescheria boydii F44-1
Hainan Sanya National Coral Reef Reserve, China
(Yuan et al., 2019)
900
emestrin L
2020
Aspergillus terreus
Weizhou coral reefs, South China Sea
(Wu et al., 2020)
901
emestrin M
2020
902
emethacin C
2020
903
emethacin B
2020
904
bisdethiobis(methylsulfanyl)acetylapoaranotin
2020
905
spiroepicoccin A
2020
Epicoccum nigrum
–
(Li et al., 2020d)
906
7-dehydroxyepicoccin H
2020
Epicoccum nigrum SD-388
Western Pacific
(Chi et al., 2020b)
907
7-hydroxyeutypellazine F
2020
908
5′-hydroxy-6′-ene-epicoccin G
2020
(Chi et al., 2020a)
909
7-methoxy-7′-hydroxyepicoccin G
2020
910
8′-acetoxyepicoccin D
2020
911
7′-demethoxyrostratin C
2020
912
amphiepicoccin A
2020
Epicoccum nigrum HDN17-88
Western Pacific
(Wang et al., 2020)
913
amphiepicoccin B
2020
914
amphiepicoccin C
2020
915
amphiepicoccin D
2020
916
amphiepicoccin E
2020
917
amphiepicoccin F
2020
918
amphiepicoccin G
2020
919
amphiepicoccin H
2020
920
amphiepicoccin I
2020
921
amphiepicoccin J
2020
922
citriperazine A
2020
Penicillium sp. KMM 4672
South China Sea
(Yurchenko et al., 2020)
923
citriperazine B
2020
924
citriperazine C
2020
925
scetryptoquivaline A
2020
Scedosporium apiospermum F41-1
Hainan Sanya National Coral Reef Reserve, China
(Li et al., 2020a)
926
5-epi-pretrichodermamide A
2020
Trichoderma cf. brevicompactum
Palau
(Yamazaki et al., 2020)
927
5-epi-trithiopretrichodermamide A
2020
928
pensulfonamide
2021
Penicillium aculeatum
Red Sea (Egypt)
(Hawas et al., 2022)
929
secoemestrin C
2021
Aspergillus quadrilineatus FJJ093
Jeju Island, Republic of Korea
(Hwang et al., 2021)
930
emestrin
2021
931
emestrin B
2021
932
talaromanloid A
2022
Talaromyces mangshanicus BTBU20191089
–
(Zhang et al., 2022)
933
ochraceopetalin
2021
Aspergillus ochraceopetaliformis
–
(Park et al., 2021b)
934
aspergillazine A
2005
Spicaria elegans
Jiaozhou Bay, China
(Liu et al., 2005)
1.3.5 Mangroves bacteria, fungi and other marine microorganism
Mangrove is a special ecosystem for the transition from land to sea. In recent years, mangrove bacteria and fungi have gradually become the focus of research. 2 (935–936) and 34 (937–968, 767, 909) sulphur-containing alkaloids were reported from mangroves bacteria and fungi, respectively. Brocazines A-F (937–942), phomazines A-C (943–945), epicorazines A-C (946–948), penicibrocazines A-E (955–959) and penispirozines A-D (964–967) all are thiodiketopiperazines alkaloids. Moreover, epicoccins A-E (949–953) are epipolythiodioxopiperazines. This shows that thiodioxopiperazines are the main type of sulphur-containing alkaloids in fungi again. Notably, spirobrocazines A-B (961–962) are characteristic of a unique spirocyclic skeleton (Meng et al., 2016) and penispirozine B (965) possesses a 6/5/6/5/6 pentacyclic ring system with two rare spirocyclic centers (Zhu et al., 2020). In addition, penispirozine A (964) has an unusual pyrazino[1,2]oxazadecaline coupled with a thiophane ring system and trichodermamide G (968) has a similar cyclic system. In addition, 4 (969–972) sulphur-containing alkaloids, new pigments with an unprecedented skeleton, were reported from marine ciliates Pseudokeronopsis riccii (Fig. 16 and Table 13).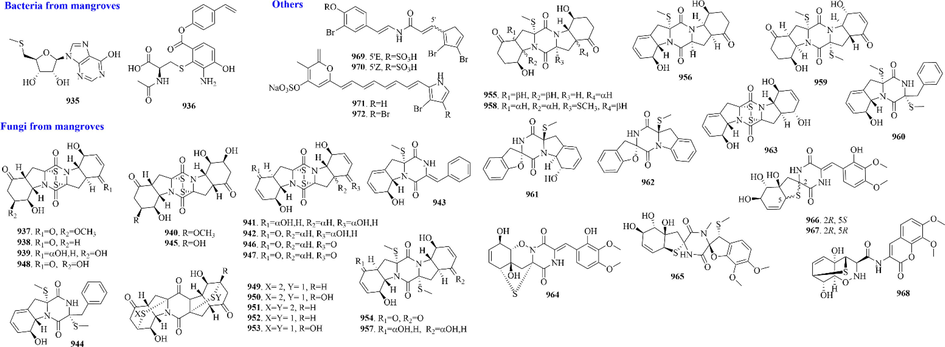
Sulphur-containing alkaloids from mangroves bacteria, fungi, other marine microorganism.
No.
Compounds
Time
From
Location
Ref.
Mangroves bacteria
935
9-((2R,3R,4S,5S)-3,4-dihydroxy-5-((methylthio)methyl)tetrahydrofuran-2-yl)-6-hydroxy-9H-purin-3-ium
2014
Micromonospora sp. K310
Butre river, Ghana
(Kyeremeh et al., 2014)
936
bagremycin C
2017
Streptomyces sp. Q22
Qiao Mangrove Forest, Zhuhai City, Guangdong, China
(Chen et al., 2017a)
Mangroves fungi
937
brocazine A
2014
Penicillium brocae MA-231
Hainan island, China
(Meng et al., 2014)
938
brocazine B
2014
939
brocazine C
2014
940
brocazine D
2014
941
brocazine E
2014
942
brocazine F
2014
943
phomazine A
2014
Phoma sp. OUCMDZ-1847
Wenchang, China
(Kong et al., 2014)
944
phomazine B
2014
945
phomazine C
2014
946
epicorazine A
2014
947
epicorazine B
2014
948
epicorazine C
2014
949
epicoccin A
2014
950
epicoccin B
2014
951
epicoccin C
2014
952
epicoccin D
2014
953
epicoccin E
2014
954
exserohilone A
2014
767.
rostratin A
2014
955
penicibrocazine A
2015
Penicillium brocae
Hainan island, China
(Meng et al., 2015)
956
penicibrocazine B
2015
957
penicibrocazine C
2015
958
penicibrocazine D
2015
959
penicibrocazine E
2015
960
analog
2015
961
spirobrocazine A
2016
Penicillium brocae MA-231
(Meng et al., 2016)
962
spirobrocazine B
2016
963
brocazine G
2016
964
penispirozine A
2020
Penicillium janthinellum HDN13-309
(Zhu et al., 2020)
965
penispirozine B
2020
966
penispirozine C
2020
967
penispirozine D
2020
968
trichodermamide G
2020
Trichoderma harzianum D13
(Zhao et al., 2020)
909.
aspergillazine A
2020
Other marine microorganism
969
keronopsamide B
2010
Pseudokeronopsis riccii
Tyrrhenian Coast, Sardinia, Italy
(Guella et al., 2010)
970
keronopsamide C
2010
971
keronopsin A1
2010
972
keronopsin A2
2010
1.4 Bioactivities of Marine-Derived Sulphur-containing alkaloids
The biological activities of marine-derived sulphur-containing alkaloids have been studied extensively. As listed in Table 14, marine-derived sulphur-containing alkaloids had a broad range of bioactive properties including cytotoxicity, antibacteria, antifungi, antimitotic, antiviral, and other activities.
In summary, while research on the biological activity of marine sulphur-containing alkaloids has explored a wide range of directions, the primary focus remains on their cytotoxicity against tumour cells. Over the past four decades, numerous compounds with potent cytotoxic properties have been discovered, displaying strong efficacy against various types of tumour cells. Here, we have summarized the compounds with superior activity according to the type of tumour they target.
1.5 Cytotoxicity
1.5.1 Leukemia
Leukemia is a collection of malignant tumours that affect the blood system. Clonal leukemia cells undergo uncontrolled proliferation and accumulate in the bone marrow and other haematopoietic tissues due to impaired differentiation, apoptosis, and other mechanisms, ultimately inhibiting normal haematopoietic function. (Whiteley et al., 2021). Several marine sulphur-containing alkaloids have demonstrated cytotoxicity against different types of leukemia cells. For instance, prianosin A (22), C (29), D (30), varamine A (370), B (371), diplamine (372), and eudistomidin J (455) exhibited IC50 values of 0.037, 0.15, 0.18, 0.03, 0.05, 0.02, and 0.047 μg/ml, respectively, against L1210 cells. (Kobayashi et al., 1987; Cheng et al., 1988; Molinski and Ireland, 1989; Charyulu et al., 1989; Suzuki et al., 2011). Similarly, compounds such as discorhabdin B (32), discorhabdin W (145), (6R,8S)-1-thiomethyldiscorhabdin G*/I (184), 16a,17a-dehydrodiscorhabdin W (185), discorhabdin G*/I (146), discorhabdin A (31), dercitin (27), curacin E (518), agrochelin (638), eudistomidin J (455), pateamine (93) have been reported to inhibit P388 cells with IC50 values of 0.084, 0.087, 0.28, 0.45, 0.6, 0.13, 0.11, 0.081, 0.02, 0.053 µM and 43, 0.15 ng/ml, respectively. (Lang et al., 2005; Grkovic and Copp, 2009; Burres et al., 1989; Suzuki et al., 2011; Northcote et al., 1991; Ueoka et al., 2016; Cañedo et al., 1999). Meanwhile, compounds such as perfragilins B (545), leptosin A (738), B (739), C (740), D (741), E (742), F (743), G (744), G1 (745), G2 (746), H (747), K (748), K1 (749) and K2 (750), N (757), N1 (758) and P (764) exhibited EC50 values of 70, 1.85, 2.40, 1.75, 86, 46, 56, 4.6, 4.3, 4.4, 3.0, 3.8, 2.2, 2.1 180, 190 and 100 ng/ml, respectively, against P388 cells. (Choi et al., 1993; Takahashi et al., 1994; Takahashi et al., 1995b; Takahashi et al., 1995a; Yamada et al., 2002; Takeshi Yamada, 2004). Among them, shishijimicin A (431), B (432), C (433) and namenamicin (434) have shown excellent cytotoxicity against P388 cells with IC50 values of 0.47, 2.0, 1.7 and 3.3 pg/ml, respectively. (Oku et al., 2003).
Moreover, chetracin E (875) exhibited IC50 values of 0.4 μM against K562 cells. (Yu et al., 2018). Moreover, somocystinamide A (601) and 14-methyleudistomidin C (418) exhibited IC50 values of 60 nM and 0.57 μg/ml against Molt4 cells, and compound 601 also inhibited CEM cells with an IC50 of 14 nM. (Wrasidlo et al., 2008; Rashid et al., 2001). It is worth noting that dercitin (27) demonstrated cytotoxicity against HL-60 and HL-60/AR cells by reducing DNA replication, with IC50 values of 0.15 and 0.24 μM, respectively. (Burres et al., 1989). Meanwhile, somocystinamide A (601) exhibited cytotoxicity against Jurkat cells with an IC50 value of only 3 nM. (Wrasidlo et al., 2008).
1.5.2 Lymphomas
Lymphomas are a heterogeneous group of malignant tumors that originate from the lymphatic hematopoietic system. Although these tumors typically arise in the lymph nodes, the distribution of the lymphatic system allows them to spread throughout the body and invade nearly any tissue or organ. (Jiang et al., 2017). Prianosin A (22), C (29), and D (30) exhibited strong cytotoxicity against L5178Y cells with IC50 values of 0.014, 0.024, and 0.048 μg/mL, respectively. (Kobayashi et al., 1987; Cheng et al., 1988).
1.5.3 Colorectal cancer
Colorectal cancer is a prevalent malignant tumor that includes colon and rectal cancers. Tumor cells can metastasize to lymph nodes through lymphatic vessels or to the liver, lungs, and bones through the bloodstream. The primary treatment regimen is currently a combination of chemotherapy, with chemotherapeutic agents such as 5-fluorouracil, oxaliplatin, irinotecan, and other drugs. (Biller et al., 2021). HCT116 cells are a commonly used in vitro model of colorectal cancer. Discorhabdin A (31), (-)-(1R,2R,6R,8S,6′S)-discorhabdin B dimer (316), latrunculone A (168), patellazole A (360), B (361), C (362), acetylgliotoxin (804), reduced gliotoxin (803), chetracin E (875), C (877), epicorazine A (946) and rostratin C (769) have been reported to inhibit HCT116 cells with IC50 values of 7, 160, 480, 0.62, 0.39, 4.7, 0.66, 0.62, 5.6, 890, 430, 400, 300, 330 nM and 0.76 μg/ml, respectively. (Antunes et al., 2004; Li et al., 2020b; Amagata et al., 2008; Richardson et al., 2005; Liang et al., 2014; Liang et al., 2014; Yu et al., 2018; Tan et al., 2004). In addition, compound 360, 361 and 362 also exhibited IC50 values of 0.66, 0.62 and 5.6 nM against HCT 116 cells p53–/–. Meanwhile, discorhabdin I (131), L (132), tanjungide A (473), agrochelin (638), dercitin (27), latrunculin A (16), ecteinascidin 743 (376), 729 (375), 597 (393), 583 (394) and 594 (395) exhibited cytotoxicity against HT-29 cells with GI50 values of 0.35, 0.12 and 0.19 µM and IC50 values of 0.268, 0.063 µM, 60, 0.5, 0.5, 2.0, 10 and 25 ng/ml, respectively. (Reyes et al., 2004; Murcia et al., 2014; Cañedo et al., 1999; Burres et al., 1989; Longley et al., 1993; Sakai et al., 1996).
RKO cells and COLO-205 cells are two other in vitro cellular models of colon cancer. A study showed that gliotoxin (792) and reduced gliotoxin (803) were cytotoxic to RKO cells (IC50 values of 0.8 and 0.41 µM, respectively), while 14-methyleudistomidin C (418) was cytotoxic to COLO-205 cells (IC50= 0.42μg/ml). (Liang et al., 2014; Rashid et al., 2001).
1.5.4 Pancreatic cancer
Pancreatic cancer is a highly prevalent malignant disease of the gastrointestinal tract with a very low survival rate. Patients with untreated pancreatic cancer typically have a survival time of approximately four months. (Park et al., 2021a). In vitro studies have shown that discorhabdin T (156), U (157), and DC1149B (831) are effective inhibitors of PANC-1 cells, with IC50 values of 0.7, 0.069 and 0.02 µM, respectively. (Gunasekera et al., 2003; Tang et al., 2020).
1.5.5 Breast cancer
Breast cancer is a common malignant tumor that affects women. It occurs due to the uncontrolled proliferation of epithelial cells in the breast, influenced by various carcinogenic factors. Common early symptoms include breast lumps, nipple discharge, and swollen lymph nodes in the armpits, while in advanced stages, cancer cells can metastasize to distant organs, leading to life-threatening multi-organ lesions. (Harbeck et al., 2019). Kuanoniamine C (82) and A (381) have demonstrated cytotoxicity against MCF-7 cells with GI50 values of 0.81 and 0.12 nM, respectively, as well as against MDA-MB-231 cells with GI50 values of 10.23 and 0.73 nM, respectively. (Kijjoa et al., 2007). Curacin A (519), B (591), D (594), Luteoalbusin A (784), B (785), T988A (786), Gliocladine C (787) and D (788) were reported to inhibit MCF-7 cells with IC50 values of 0.038, 0.32, 0.34, 0.23, 0.25, 0.91, 0.23, and 0.65 µM, respectively. (Verdier-Pinard et al., 1998; Márquez et al., 1998; Wang et al., 2012a).
1.5.6 Lung cancer
Lung cancer is a malignant tumor that originates in the lining or glands of the bronchi in the lungs. It is one of the fastest-growing malignancies in terms of morbidity and mortality and poses a serious threat to public health. Currently, there are two main classifications of lung cancer: small cell lung cancer and non-small cell lung cancer, which can be further divided into adenocarcinoma, squamous cell carcinoma, large cell carcinoma, bronchoalveolar carcinoma, and others depending on the pathology. (Hirsch et al., 2017). Several compounds have been reported to inhibit lung cancer cell growth. Dercitin (27), somocystinamide A (601), ecteinascidin 743 (376), latrunculin A (16), ecteinascidin 729 (375), 597 (393), 583 (394), 594 (395), agrochelin (638), chetracin E (875), and C (877) have been reported to inhibit A549 cells with IC50 of 7, 160, 480, 0.62, 0.39, 4.7, 0.66, 0.62, 5.6, 890, 430, 400, 300, 330 nM and 0.76 μg/ml, respectively. (Burres et al., 1989; Wrasidlo et al., 2008; Simoens et al., 2003; Longley et al., 1993; Sakai et al., 1996; Cañedo et al., 1999; Yu et al., 2018). Lyngbyabellin E-I (603–607) have also been shown to inhibit NCI-H460 cells, with IC50 values ranging from 0.2 to 2.2 µM. (Williams et al., 2003). Meanwhile, kuanoniamine C (82) and A (381) demonstrated cytotoxicity against MCF-7 cells, with GI50 values of 0.81 and 0.12 nM, respectively. (Kijjoa et al., 2007). Additionally, Chetracin E (875) and C (877) exhibited IC50 values of 0.2 and 0.8 μM, respectively, against NCI-H1975 cells. (Yu et al., 2018). Notably, compound 376 showed significant inhibition of NCI-H292 cells with an IC50 value of 1.1 nM. (Simoens et al., 2003).
1.5.7 Cervical cancer
Cervical cancer is a prevalent malignancy in women, and persistent high-risk HPV infection is a well-established major risk factor for the disease. More than 90% of cervical cancers are associated with high-risk HPV infection. (Cohen et al., 2019). In vitro, Hela cells are widely used as a model for cervical cancer. Among tested compounds, iejimalide C (539), caldorazole (626), and iezoside (627) exhibited IC50 values of 2.7, 23, and 6.8 nM, respectively, against HeLa cells. Compound 626 also inhibited HeLa S3 and HeLa S3Mer- cells, with IC50 values of 44 and 48 nM, respectively. (Kazami et al., 2014; Ohno et al., 2022; Kurisawa et al., 2022). Notably, shishijimicin A (431), B (432), C (433), and namenamicin (434) showed excellent cytotoxicity against HeLa cells, with IC50 values of 1.8, 3.3, 6.3, and 43 pg/ml, respectively. (Oku et al., 2003).
1.5.8 Melanoma
Melanoma is a highly malignant tumor that develops from melanocytes commonly found in the skin. Due to its aggressive nature, melanoma is prone to infiltrative growth and metastasis, making it one of the deadliest types of skin cancer. (Schadendorf et al., 2018). Agrochelin (638), ecteinascidin 743 (376), 729 (375), 597 (393), 583 (394), and 594 (395) have been reported to inhibit MEL-28 cells with IC50 values of 0.268 µM, 5.0, 5.0, 2.0, 5.0, and 25 ng/ml, respectively (Cañedo et al., 1999; Sakai et al., 1996).
1.5.9 Ovarian cancer
Ovarian cancer is a malignant tumor that grows on the ovary, with 90% to 95% of cases being primary ovarian cancers. Despite having a lower incidence rate than cervical and endometrial cancers, ovarian cancer has the highest mortality rate among gynecological cancers, ranking first. (Matulonis et al., 2016). 14-methyleudistomidin C (418) and aulosirazole A-B (623–624) demonstrated IC50 values of 0.98 μg/ml, 0.301, and 0.6 µM, respectively, against OVCAR-3 cells (Davis et al., 2022; Rashid et al., 2001). Additionally, brocazine G (963) exhibited inhibition of A2780 and A2780 CisR cells with IC50 values of 664 and 661 nM, respectively (Meng et al., 2016).
1.5.10 Others
In addition to their activity against the aforementioned tumour cells, sulphur-containing marine alkaloids have demonstrated promising cytotoxic properties against other types of cancer cells. For instance, luteoalbusin A (784), B (785), T988A (786), gliocladine C (787) and D (788) were reported to inhibit the growth of SF-268 and HepG-2 cells, with IC50 values ranging from 0.46 to 2.49 µM. Moreover, kuanoniamine C (82) and A (381) exhibited excellent cytotoxicity against SF-268 cells, with IC50 values of 33.16 and 4.67 nM, respectively. (Kijjoa et al., 2007). Of particular note, shishijimicin A (431), B (432), C (433) and namenamicin (434) displayed potent cytotoxicity against 3Y1 cells, with IC50 values of 2.0, 3.1, 4.8 and 13 pg/ml. (Oku et al., 2003).
1.5.11 Structure-activity relationships of the cytotoxicity
It is noteworthy that discorhabdins, curacins, tanjungides, leptosins, and latrunculins exhibit better cytotoxicity than any other reported compounds. Thus, the structure–activity relationships of the cytotoxicity of these compounds have been summarized for further investigation.
As depicted in Fig. 17a, the cytotoxicity of discorhabdins is decreased when there is a double bond between C-4 and C-5 of discorhabdins, which is also confirmed in Fig. 17b. When there is a hydroxyl substitution at C-3′, the cytotoxicity of the compound is increased. Fig. 17c reveals that the presence of substituents at C-2′ affects the cytotoxicity of the compound, and the degree of influence is related to the nature of the substituents, as confirmed in Fig. 17d. Simultaneously, Fig. 17c also shows that replacing the carbonyl group at C4′ with more hydroxyl groups does not affect the cytotoxicity, indicating that the presence of a carbonyl group does not affect cytotoxicity. Fig. 17d shows that the cytotoxicity of the compound is reduced when there is a double bond between C-5′ and C-6′, and converting the N atom at the C-6 position to NH+ also reduces the cytotoxicity. The cytotoxicity of latrunculins is strongly related to the configuration at C-18. When C-18 has an R-configuration, the cytotoxicity of the compounds increases, and when it has an S-configuration, the cytotoxicity decreases (Fig. 17e). When the hydroxyl group at C-17 is replaced by methoxy, the toxicity of the compound is also weakened. In addition, we found that as the lactone ring of these compounds becomes larger, their cytotoxicity increases, which may be related to the increase in the number of hydroxyl groups on the lactone ring.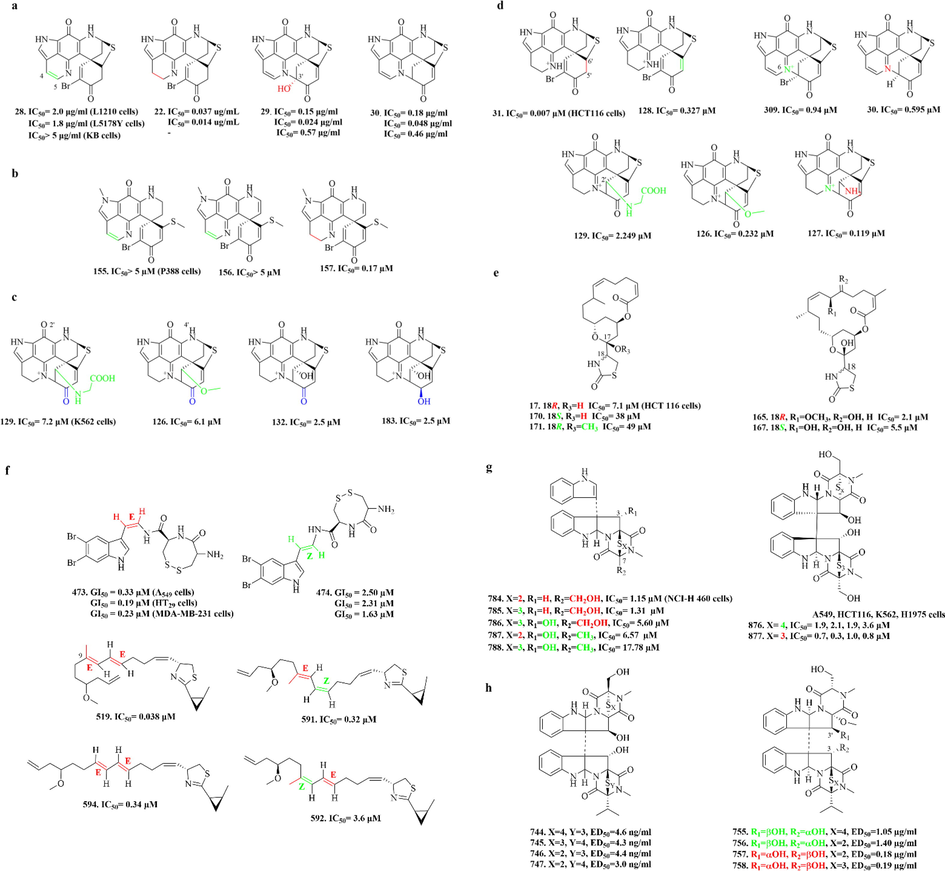
Structure-activity relationships of the cytotoxicity.
The cytotoxicity of curacins and tanjungides is related to the configuration of the double bond in their structures. When the double bond is of the E-type, the cytotoxicity of the compounds increases, and when it is of the Z-type, it decreases. If there is a methyl group at C-9 of curacins, the cytotoxicity of the compound also increases (Fig. 17f). The cytotoxicity of leptosins is related to the number of sulfur atoms in the sulfur bridge. As the number of sulfur atoms increases, the cytotoxicity is attenuated, but if the sulfur bridge is missing, the cytotoxicity of leptosins is greatly reduced. It can be seen that the sulfur bridge is an important cytotoxic basis of leptosins. Cytotoxicity is reduced in the presence of methyl group at C-7, and increased in the presence of hydroxymethyl groups. Cytotoxicity is similarly reduced in the presence of hydroxyl groups in C-3. (Fig. 17g). In addition, the configuration of the hydroxyl groups at the C-3 and C-3′ positions will affect the cytotoxicity. The cytotoxicity will decrease when both are S-configurations, and it will increase when both are R-configurations. (Fig. 17h).
2 Conclusions and outlook
This review summarized current research regarding the chemical and bioactivity diversity of marine-derived sulphur-containing alkaloids from 1992 to 2022. More than 972 sulphur-containing alkaloids have been isolated and identified from the marine. Meanwhile, modern pharmacological research revealed that the sulphur-containing alkaloids have significant pharmacological properties including cytotoxicity, anti-proliferation, anti-virus, anti-inflammatory, antioxidant, antibacteria, antifungal, anti-malarial, antiparasitic and enzyme inhibitory activity. Regardless, there are still several aspects that need to be concerned in the further development of marine-derived sulphur-containing alkaloids.
Firstly, as shown in Fig. 1, sponges are the dominant producer of marine-derived sulphur-containing alkaloids, yielding 316 of these 972 compounds (32.51%). Marine animal tunicates also produce massive sulphur-containing alkaloids with a combined percentage of 16.26%. In addition, marine fungi and bacteria are also important sources, producing 20.27% and 11.32%, respectively, of the alkaloids reviewed. It can be seen that almost 80.36% of marine-derived sulphur-containing alkaloids are from marine sponges, fungi, tunicates and bacteria. This suggests that we can focus more on marine sponges, fungi, tunicates and bacteria in the search for more marine-derived sulphur-containing alkaloids.
Secondly, sulphur-containing alkaloids obtained from sponges have fallen since 2010, while microbes, especially fungi, have grown to be important producers (Fig. 2). There may be two reasons for this. (1) with the increasing demand for new bioactive substances, the attraction of terrestrial fungi in drug screening has gradually decreased. Scientists began to pay attention to marine fungi living in complex environments such as high pressure, high salt and low temperature. Meanwhile, the complex environment makes the secondary metabolites of marine fungi have diverse structures and unique biological activities, which attracts scientists to increase the research and development of marine fungi. (2) Biochemists have begun to generally acknowledge that sampling slow-growing sessile organisms to identify natural products is not an economical and environmentally friendly approach. Fungi can reproduce indefinitely under suitable conditions, and their genomes can be easily mined for targeted metabolites. This also makes fungi get more attention.
Thirdly, sulphur-containing alkaloids have been shown to have a variety of biological activities such as cytotoxicity, anti-proliferation, anti-virus, anti-inflammatory, antioxidant, antibacteria, antifungal, anti-malarial, antiparasitic and enzyme inhibitory activity. Of them, ecteinascidin 743 (yondelis) has been approved by the European Union in October 2007 for the treatment of advanced soft tissue tumors, which became the first modern marine drug (Menchaca et al., 2003). Thiomarinols, a kind of naturally occurring double-headed antibiotic, usually have excellent antibacterial activity and can even be effective against MRSA (Shiozawa et al., 1995). But more sulphur-containing alkaloids with various activities also need to be found in the marine. Of course, it can not be ignored that monomeric compounds with outstanding pharmacological activities can be considered the source of new drugs with excellent therapeutic effects. For example, shishijimicin A (431), B (432), C (433) and namenamicin (434) exhibited extremely cytotoxic to 3Y1, HeLa and P388 cells with IC50 values from 0.47 to 43 pg/ml. Gliotoxin (792), acetylgliotoxin (804) and reduced gliotoxin (803) have been reported to inhibit HCT-116 and RKO cells with IC50 values from 0.41 to 4.49 µM. Chetracin E (875), F (876) and C (877) also have cytotoxicity against A549, HCT116, K562, H1975 cells with GI50 from 0.2 to 3.6 μM.
Fourth, although many compounds with excellent activity have been found in sulphur-containing alkaloids, only a few components have been fully and deeply studied, and a large number of active components have only been tested for simple biological activity. This may be related to active compounds being too few to support a more in-depth study of the mechanism. Such problems usually need to be solved by chemical synthesis, but it is not realistic to synthesize each compound without purpose. At present, it is proposed to simulate the combination of active components and target receptors by molecular docking technology to achieve preliminary screening of active compounds, which may be a method to reduce the workload (Chen et al., 2020). All in all, it is still necessary to further study the mechanism of active sulphur-containing alkaloids to provide a scientific basis for the development of new drugs.
Fifth, the remarkable chemical diversity and biological activities of marine-derived sulphur-containing alkaloids make them attractive candidates for drug discovery and development. With the continued development of advanced techniques for marine natural product isolation, structure elucidation, and biosynthesis studies, we can expect to discover even more diverse and potent sulphur-containing alkaloids.The future impact of marine-derived sulphur-containing alkaloids in the drug discovery avenue will be more significant too.
Finally, the ocean is a huge treasure house of medicine awaiting human exploration. Among the natural marine products, sulphur-containing alkaloids are important potential drugs that deserve further research and development. This review could be a useful tool in assisting researchers in the selection of interesting species or isolated compounds for further studies, as well as expand the research of marine-derived sulphur-containing alkaloids.
Author contributions
Z.L. Zhang, Y.Z. Li, W. Wang and Y. Sun searched and collected literature; Z.L. Zhang and Y.Z. Li carried out the writing work; X.M. Song and D.D. Zhang designed this review. All authors have read and agreed to the published version of the manuscript.
Acknowledgements
We thank the foundations of the National Natural Science Foundation of China (82104368) and Shaanxi Provincial Science and Technology Department Project (2021JQ-744) for financial support of this study.
References
- Alkaloids from the Tunicate Polycarpa aurata from Chuuk Atoll. J. Org. Chem.. 1996;61:2709-2712.
- [CrossRef] [Google Scholar]
- Nordehydrocyclodercitin, a hexacyclic pyridoacridine alkaloid from the marine ascidian, Aplidium sp. Natural Product Res.. 2007;21:782-786.
- [CrossRef] [Google Scholar]
- Latrunculin with a highly oxidized thiazolidinone ring: structure assignment and actin docking. Org. Lett.. 2007;9:4773-4776.
- [CrossRef] [Google Scholar]
- Conicaquinones A and B, two novel cytotoxic terpene quinones from the mediterranean ascidian Aplidium conicum. Eur. J. Org. Chem.. 2003;2003:898-900.
- [CrossRef] [Google Scholar]
- Novel bioactive bromopyrrole alkaloids from the Mediterranean sponge Axinella verrucosa. Bioorg. Med. Chem.. 2006;14:17-24.
- [CrossRef] [Google Scholar]
- Palyosulfonoceramides A and B: Unique Sulfonylated Ceramides from the Brazilian Zoanthids Palythoa caribaeorum and Protopalyhtoa variabilis. Mar. Drugs 2012:2846-2860.
- [CrossRef] [Google Scholar]
- Evaluation of new sesquiterpene quinones from two Dysidea sponge species as inhibitors of protein tyrosine kinase. J. Org. Chem.. 1992;57:6604-6607.
- [CrossRef] [Google Scholar]
- Interrogating the bioactive pharmacophore of the Latrunculin Chemotype by investigating the metabolites of two taxonomically unrelated sponges. J. Med. Chem.. 2008;51:7234-7242.
- [CrossRef] [Google Scholar]
- Cytotoxic Pyrroloiminoquinones from Four New Species of South African Latrunculid Sponges. J. Nat. Prod.. 2004;67:1268-1276.
- [CrossRef] [Google Scholar]
- Kottamide E, the first example of a natural product bearing the amino acid 4-amino-1,2-dithiolane-4-carboxylic acid (Adt) Tetrahedron Lett.. 2003;44:8963-8965.
- [CrossRef] [Google Scholar]
- Isodiplamine, cystodytin K and lissoclinidine: novel bioactive alkaloids from the New Zealand ascidian Lissoclinum notti. Tetrahedron. 2002;58:9779-9783.
- [CrossRef] [Google Scholar]
- Brominated tyrosine metabolites from an unidentified sponge. J. Org. Chem.. 1987;52:3584-3586.
- [CrossRef] [Google Scholar]
- Thiazole and imidazole metabolites from the ascidian Aplydium pliciferum. Tetrahedron Lett.. 1988;29:1099-1102.
- [CrossRef] [Google Scholar]
- Nagelamides K and L, Dimeric Bromopyrrole Alkaloids from Sponge Agelas species. Org. Lett.. 2008;10:2099-2102.
- [CrossRef] [Google Scholar]
- Nagelamides Q and R, Novel Dimeric Bromopyrrole Alkaloids from Sponges Agelas sp. Org. Lett.. 2009;11:1785-1788.
- [CrossRef] [Google Scholar]
- Dragonamide E, a Modified Linear Lipopeptide from Lyngbya majuscula with Antileishmanial Activity. J. Nat. Prod.. 2010;73:60-66.
- [CrossRef] [Google Scholar]
- Defensive 2-Alkylpyrrole Sulfamates from the Marine Annelid Cirriformia tentaculata. J. Nat. Prod.. 2003;66:1110-1112.
- [CrossRef] [Google Scholar]
- Lipid composition of the gram-negative, budding, seawater bacterium Hyphomonas jannaschiana lacking in phospholipids. Biochimica et Biophysica Acta (BBA) - Lipids and Lipid Metabolism. 1996;1303:39-46.
- [CrossRef] [Google Scholar]
- Bacterial synthesis of unusual sulfonamide and sulfone antibiotics by flavoenzyme-mediated sulfur dioxide capture. Angew. Chem. Int. Ed.. 2015;54:13279-13283.
- [CrossRef] [Google Scholar]
- Trichothiazole A, a dichlorinated polyketide containing an embedded thiazole isolated from Trichodesmium blooms. Tetrahedron Lett.. 2017;58:4066-4068.
- [CrossRef] [Google Scholar]
- Antillatoxin and kalkitoxin, ichthyotoxins from the tropical cyanobacterium Lyngbya majuscula, induce distinct temporal patterns of NMDA receptor-mediated neurotoxicity. Toxicon. 1999;37:1645-1648.
- [CrossRef] [Google Scholar]
- Diagnosis and treatment of metastatic colorectal cancer: a review. JAMA. 2021;325:669-685.
- [CrossRef] [Google Scholar]
- Two sulfur-containing isoquinoline alkaloids from the bryozoan Biflustra perfragilis. J. Aust. J. Chem.. 1993;46:213-220.
- [CrossRef] [Google Scholar]
- On the chemistry of Latrunculins A and B. Liebigs Ann. Chem.. 1989;1989:1171-1188.
- [CrossRef] [Google Scholar]
- The stereochemistry of Eudistomins C, K, E, F AND L. Tetrahedron Lett.. 1987;28:1825-1826.
- [CrossRef] [Google Scholar]
- Synthesis and intracellular redox cycling of natural quinones and their analogues and identification of Indoleamine-2,3-dioxygenase (IDO) as potential target for anticancer activity. Angew. Chem. Int. Ed.. 2015;54:8740-8745.
- [CrossRef] [Google Scholar]
- Structures and antimicrobial activities of pyridoacridine alkaloids isolated from different chromotypes of the Ascidian Cystodytes dellechiajei. J. Nat. Prod.. 2010;73:1044-1048.
- [CrossRef] [Google Scholar]
- Additional cytotoxic pyridoacridine alkaloids from the ascidian cystodytes violatinctus and biogenetic considerations. J. Nat. Prod.. 2013;76:1801-1805.
- [CrossRef] [Google Scholar]
- Expanding the described metabolome of the marine cyanobacterium moorea producens JHB through orthogonal natural products workflows. PLoS ONE. 2015;10:e0133297.
- [CrossRef] [Google Scholar]
- A new β-carboline alkaloid isolated from the marine sponge Hyrtios erecta. Tetrahedron Lett.. 1996;37:3457-3460.
- [CrossRef] [Google Scholar]
- Lamellarin Sulfates from the Pacific Tunicate Didemnum ternerratum. J. Nat. Prod.. 2019;82:2000-2008.
- [CrossRef] [Google Scholar]
- New pyridoacridine alkaloids from the purple morph of the ascidian Cystodytes dellechiajei. Tetrahedron Lett.. 2011;52:3041-3044.
- [CrossRef] [Google Scholar]
- Anti-Mycobacterial Nucleoside Antibiotics from a Marine-Derived Streptomyces sp. TPU1236A. Marine Drugs 2014:6102-6112.
- [Google Scholar]
- Antitumor activity and nucleic acid binding properties of dercitin, a new acridine alkaloid isolated from a marine Dercitus species sponge. Cancer Res. 1989;49:5267-5274.
- [Google Scholar]
- Nebulosins: Trisubstituted Thiolane Natural Products from the Northeastern Atlantic Annelid Eupolymnia nebulosa. J. Org. Chem.. 2020;85:14026-14041.
- [CrossRef] [Google Scholar]
- Agrochelin, a new cytotoxic alkaloid from the marine bacteria Agrobacterium sp. Tetrahedron Lett.. 1999;40:6841-6844.
- [CrossRef] [Google Scholar]
- Halenaquinone and xestoquinone derivatives, inhibitors of Cdc25B phosphatase from a Xestospongia sp. Bioorg. Med. Chem.. 2005;13:999-1003.
- [CrossRef] [Google Scholar]
- Isolation and characterization of three pairs of indolediketopiperazine enantiomers containing infrequent N-methoxy substitution from the marine algal-derived endophytic fungus Acrostalagmus luteoalbus TK-43. Bioorg. Chem.. 2019;90:103030
- [CrossRef] [Google Scholar]
- Uncommon N-Methoxyindolediketopiperazines from Acrostalagmus luteoalbus, a Marine Algal Isolate of Endophytic Fungus. Chin. J. Chem .. 2021;39:2808-2814.
- [CrossRef] [Google Scholar]
- Antimicrobial metabolites from a marine-derived Actinomycete Streptomyces sp. G278. Nat. Prod. Res.. 2019;33:3223-3230.
- [CrossRef] [Google Scholar]
- Citromycetins and Bilains A-C: new Aromatic Polyketides and Diketopiperazines from Australian Marine-Derived and Terrestrial Penicillium spp. J. Nat. Prod.. 2007;70:1746-1752.
- [CrossRef] [Google Scholar]
- Occurrence of a taurine derivative in an antarctic glass sponge. Nat. Product Commun.. 2014;9 1934578X1400900408
- [CrossRef] [Google Scholar]
- Plectosphaeroic Acids A, B, and C, Indoleamine 2,3-Dioxygenase Inhibitors Produced in Culture by a Marine Isolate of the Fungus Plectosphaerella cucumerina. Org. Lett.. 2009;11:2996-2999.
- [CrossRef] [Google Scholar]
- Leptoclinidamines A−C, Indole Alkaloids from the Australian Ascidian Leptoclinides durus. J. Nat. Prod.. 2009;72:696-699.
- [CrossRef] [Google Scholar]
- Dysinosin A: a novel inhibitor of Factor VIIa and thrombin from a new genus and species of australian sponge of the family Dysideidae. J. Am. Chem. Soc.. 2002;124:13340-13341.
- [CrossRef] [Google Scholar]
- Kuanoniamines A, B, C, and D: pentacyclic alkaloids from a tunicate and its prosobranch mollusk predator Chelynotus semperi. J. Org. Chem.. 1990;55:4426-4431.
- [CrossRef] [Google Scholar]
- A second shermilamine alkaloid from a tunicate Trididemnum sp. J. Org. Chem.. 1989;54:4231-4232.
- [CrossRef] [Google Scholar]
- The unique 6-(p-hydroxyphenyl)-2H-3,4-dihydro-1,1-dioxo-1,4-thiazine and the new tripeptide l-glu-gly-4-hydroxystirylamine from the marine sponge anchinoe tenacior. Tetrahedron Lett.. 1994;35:2421-2422.
- [CrossRef] [Google Scholar]
- Chemical investigation of the Indonesian Tunicate Polycarpa aurata and evaluation of the effects against schistosoma mansoni of the novel alkaloids Polyaurines A and B. Mar. Drugs 2019
- [CrossRef] [Google Scholar]
- Indoleamine 2,3-Dioxygenase inhibitors isolated from the sponge xestospongia vansoesti: structure elucidation, analogue synthesis, and biological activity. Org. Lett.. 2014;16:6480-6483.
- [CrossRef] [Google Scholar]
- Antibacterial p-Terphenyl with a Rare 2,2′-Bithiazole Substructure and Related Compounds Isolated from the Marine-Derived Actinomycete Nocardiopsis sp. HDN154086. J. Nat. Prod.. 2021;84:1226-1231.
- [CrossRef] [Google Scholar]
- Dihydroxystyrene Metabolites from an Association of the Sponges Poecillastra wondoensis and Jaspis sp. J. Nat. Prod.. 2008;71:779-783.
- [CrossRef] [Google Scholar]
- Diplamine, a cytotoxic polyaromatic alkaloid from the tunicate diplosoma sp. Tetrahedron Lett.. 1989;30:4201-4202.
- [CrossRef] [Google Scholar]
- Cytotoxic Bagremycins from Mangrove-Derived Streptomyces sp. Q22. J. Nat. Prod.. 2017;80:1450-1456.
- [CrossRef] [Google Scholar]
- Recent advances in molecular docking for the research and discovery of potential marine drugs. Mar. Drugs 2020
- [CrossRef] [Google Scholar]
- Diverse secondary metabolites from the marine-derived fungus Dichotomomyces cejpii F31–1. Mar. Drugs 2017
- [CrossRef] [Google Scholar]
- Prianosins B, C, and D, novel sulfur-containing alkaloids with potent antineoplastic activity from the Okinawan marine sponge Prianos melanos. J. Org. Chem.. 1988;53:4621-4624.
- [CrossRef] [Google Scholar]
- Cytotoxic thiodiketopiperazine derivatives from the deep sea-derived fungus Epicoccum nigrum SD-388. Mar. Drugs 2020
- [CrossRef] [Google Scholar]
- New antibacterial thiodiketopiperazines from the deep sea sediment-derived fungus Epicoccum nigrum SD-388. Chem. Biodivers.. 2020;17:e2000320.
- [Google Scholar]
- Violatinctamine, a new heterocyclic compound from the marine tunicate Cystodytes cf. violatinctus. Tetrahedron Lett.. 2004;45:7925-7928.
- [CrossRef] [Google Scholar]
- Lyngbyabellins K-N from two palmyra atoll collections of the marine cyanobacterium Moorea bouillonii. Eur. J. Org. Chem.. 2012;2012:5141-5150.
- [CrossRef] [Google Scholar]
- Perfragilins A and B, Cytotoxic Isoquinolinequinones from the Bryozoan Membranipora perfragilis. J. Nat. Prod.. 1993;56:1431-1433.
- [CrossRef] [Google Scholar]
- Apoptosis-inducing effect of diketopiperazine disulfides produced by Aspergillus sp. KMD 901 isolated from marine sediment on HCT116 colon cancer cell lines. J. Appl. Microbiol.. 2011;110:304-313.
- [CrossRef] [Google Scholar]
- Two novel diketopiperazines isolated from the fungus Tolypocladium sp. Tetrahedron Lett.. 1993;34:7537-7540.
- [CrossRef] [Google Scholar]
- A novel chlorinated ketide amino acid, herbamide A, from the marine sponge Dysidea herbacea. Tetrahedron Lett.. 1995;36:1185-1188.
- [CrossRef] [Google Scholar]
- Discovery of new pyridoacridine alkaloids from Lissoclinum cf. badium that inhibit the ubiquitin ligase activity of Hdm2 and stabilize p53. Bioorganic Medicinal Chem.. 2008;16:10022-10028.
- [CrossRef] [Google Scholar]
- Shermilamine A: A pentacyclic alkaloid from a tunicate. J. Org. Chem.. 1988;53:4619-4620.
- [CrossRef] [Google Scholar]
- A biologically active 1,2,3-trithiane derivative from the New Zealand ascidain Aplidium sp D. Tetrahedron Lett.. 1989;30:3703-3706.
- [CrossRef] [Google Scholar]
- Mycothiazole, a polyketide heterocycle from a marine sponge. J. Am. Chem. Soc.. 1988;110:4365-4368.
- [CrossRef] [Google Scholar]
- The marine sponge Agelas citrina as a source of the new pyrrole–imidazole alkaloids citrinamines A-D and N-methylagelongine. Beilstein J. Org. Chem.. 2015;11:2029-2037.
- [CrossRef] [Google Scholar]
- Cytotoxic and Protein Kinase Inhibiting Nakijiquinones and Nakijiquinols from the Sponge Dactylospongia metachromia. J. Nat. Prod.. 2014;77:218-226.
- [CrossRef] [Google Scholar]
- New Lamellarin Alkaloids from the Australian Ascidian, Didemnum chartaceum. J. Nat. Prod.. 1999;62:419-424.
- [CrossRef] [Google Scholar]
- Thiaplakortones A-D: antimalarial thiazine alkaloids from the australian marine sponge Plakortis lita. J. Org. Chem.. 2013;78:9608-9613.
- [CrossRef] [Google Scholar]
- Aulosirazoles B and C from the Cyanobacterium Nostoc sp. UIC 10771: analogues of an Isothiazolonaphthoquinone Scaffold that Activate Nuclear Transcription Factor FOXO3a in Ovarian Cancer Cells. J. Nat. Prod.. 2022;85:540-546.
- [CrossRef] [Google Scholar]
- Exiguaquinol: a novel pentacyclic hydroquinone from Neopetrosia exigua that inhibits helicobacter pylori MurI. Org. Lett.. 2008;10:2585-2588.
- [CrossRef] [Google Scholar]
- Coscinolactams A and B: new nitrogen-containing sesterterpenoids from the marine sponge Coscinoderma mathewsi exerting anti-inflammatory properties. Tetrahedron. 2009;65:2905-2909.
- [CrossRef] [Google Scholar]
- Neamphine, a sulfur containing aromatic heterocycle isolated from the marine sponge neamphius huxleyi. Tetrahedron Lett.. 1991;32:2707-2710.
- [CrossRef] [Google Scholar]
- Corallistine, a new polynitrogen compound from the sponge Corallistes fulvodesmus L. & L. Tetrahedron Lett.. 1989;30:1534-1538.
- [CrossRef] [Google Scholar]
- Isolation and Structure Elucidation of New and Unusual Saxitoxin Analogues from Mussels. J. Natural Products. 2008;71:1518-1523.
- [CrossRef] [Google Scholar]
- p-Terphenyl O-β-glucuronides, DNA topoisomerase inhibitors from Streptomyces sp. LZ35ΔgdmAI. Bioorg. Med. Chem. Lett.. 2014;24:1362-1365.
- [CrossRef] [Google Scholar]
- Alisiaquinones and Alisiaquinol, dual inhibitors of plasmodium falciparum enzyme targets from a new caledonian deep water sponge. J. Nat. Prod.. 2008;71:1189-1192.
- [CrossRef] [Google Scholar]
- Bromotryptamine and imidazole alkaloids with anti-inflammatory activity from the bryozoan Flustra foliacea. J. Nat. Prod.. 2020;83:2854-2866.
- [CrossRef] [Google Scholar]
- Novel Pyrazines from the Myxobacterium Chondromyces crocatus and Marine Bacteria. Eur. J. Org. Chem.. 2005;2005:4141-4153.
- [CrossRef] [Google Scholar]
- A new discorhabdin from two sponge genera. J. Nat. Prod.. 1999;62:636-637.
- [CrossRef] [Google Scholar]
- Derivatives of holomycin and cyclopropaneacetic acid from streptomyces sp. DT-A37. Chem. Biodivers.. 2017;14:e1700140.
- [Google Scholar]
- Structures and mechanisms of antitumor agents: Xestoquinones uncouple cellular respiration and disrupt HIF signaling in human breast tumor cells. J. Nat. Prod.. 2012;75:1553-1559.
- [CrossRef] [Google Scholar]
- New chlorinated metabolites from the tropical marine sponge Dysidea herbacea. J. Aust. J. Chem.. 1997;50:139-144.
- [CrossRef] [Google Scholar]
- Enzymatic Basis of “Hybridity” in Thiomarinol Biosynthesis. Angew. Chem. Int. Ed.. 2015;54:5137-5141.
- [CrossRef] [Google Scholar]
- Bioactive Pyridoacridine Alkaloids from the Micronesian Sponge Oceanapia sp. J. Nat. Prod.. 1998;61:301-305.
- [CrossRef] [Google Scholar]
- Bioactive Natural and Semisynthetic Latrunculins. J. Nat. Prod.. 2006;69:219-223.
- [CrossRef] [Google Scholar]
- Sulfur-Containing Polybromoindoles from the Formosan Red Alga Laurencia brongniartii. J. Nat. Prod.. 2005;68:815-817.
- [CrossRef] [Google Scholar]
- Discorhabdins revisited: cytotoxic alkaloids from southern australian marine sponges of the genera higginsia and spongosorites. J. Nat. Prod.. 2009;72:460-464.
- [CrossRef] [Google Scholar]
- Hydroquinone derivatives from the marine-derived fungus Gliomastix sp. RSC Adv.. 2017;7:30640-30649.
- [CrossRef] [Google Scholar]
- Nagelamides A−H, New Dimeric Bromopyrrole Alkaloids from Marine Sponge Agelas Species. J. Nat. Prod.. 2004;67:1262-1267.
- [CrossRef] [Google Scholar]
- Chlorinated Thiazole-Containing Polyketide-Peptides from the Caribbean Sponge Smenospongia conulosa: Structure Elucidation on Microgram Scale. Eur. J. Org. Chem.. 2016;2016:2871-2875.
- [CrossRef] [Google Scholar]
- Baculiferins A-O, O-sulfated pyrrole alkaloids with anti-HIV-1 activity, from the Chinese marine sponge Iotrochota baculifera. Bioorg. Med. Chem.. 2010;18:5466-5474.
- [CrossRef] [Google Scholar]
- Dichotocejpins A-C: New Diketopiperazines from a Deep-Sea-Derived Fungus Dichotomomyces cejpii FS110. Mar. Drugs 2016
- [CrossRef] [Google Scholar]
- Bioactivities of Lyngbyabellins from Cyanobacteria of Moorea and Okeania Genera. Molecules 2020
- [CrossRef] [Google Scholar]
- Two novel pyrrole-imidazole alkaloids from the Mediterranean sponge Agelas oroides. Tetrahedron Lett.. 2000;41:9917-9922.
- [CrossRef] [Google Scholar]
- Discorhabdin R: A New Antibacterial Pyrroloiminoquinone from Two Latrunculiid Marine Sponges, Latrunculia sp. and Negombata sp. J. Nat. Prod.. 2000;63:1527-1528.
- [CrossRef] [Google Scholar]
- 2-Guanidinoethanesulfonyl sesquiterpenes from the marine sponge Agelas nakamurai. Tetrahedron Lett.. 2022;103:153964
- [CrossRef] [Google Scholar]
- Spithioneines A and B, Two New Bohemamine Derivatives Possessing Ergothioneine Moiety from a Marine-Derived Streptomyces spinoverrucosus. Org. Lett.. 2015;17:3046-3049.
- [CrossRef] [Google Scholar]
- Thiasporines A-C, Thiazine and Thiazole Derivatives from a Marine-Derived Actinomycetospora chlora. J. Nat. Prod.. 2015;78:548-551.
- [CrossRef] [Google Scholar]
- Graphiumins, new thiodiketopiperazines from the marine-derived fungus Graphium sp. OPMF00224. J. Antibiot.. 2015;68:620-627.
- [CrossRef] [Google Scholar]
- Secondary Metabolites from the Deep-Sea Derived Fungus Acaromyces ingoldii FS121. Molecules 2016
- [CrossRef] [Google Scholar]
- Selected Mutations Reveal New Intermediates in the Biosynthesis of Mupirocin and the Thiomarinol Antibiotics. Angew. Chem. Int. Ed.. 2017;56:3930-3934.
- [CrossRef] [Google Scholar]
- The epipolythiodioxopiperazine (ETP) class of fungal toxins: distribution, mode of action. functions and biosynthesis.. 2005;151(1021–1032)
- [CrossRef] [Google Scholar]
- Screening and Biological Effects of Marine Pyrroloiminoquinone Alkaloids: Potential Inhibitors of the HIF-1α/p300 Interaction. J. Nat. Prod.. 2016;79:1267-1275.
- [CrossRef] [Google Scholar]
- Orbicularisine: A Spiro-Indolothiazine Isolated from Gills of the Tropical Bivalve Codakia orbicularis. J. Nat. Prod.. 2017;80:1693-1696.
- [CrossRef] [Google Scholar]
- Psammaplin Metabolites New and Old: An NMR Study Involving Chiral Sulfur Chemistry %J. Aust. J. Chem.. 2010;63(867–872)
- [CrossRef] [Google Scholar]
- New natural products in the discorhabdin A- and B-series from New Zealand-sourced Latrunculia spp. sponges. Tetrahedron. 2009;65:6335-6340.
- [CrossRef] [Google Scholar]
- Isolation and Characterization of Diastereomers of Discorhabdins H and K and Assignment of Absolute Configuration to Discorhabdins D, N, Q, S, T, and U. J. Nat. Prod.. 2010;73:1686-1693.
- [CrossRef] [Google Scholar]
- Bioactive Metabolites from the Caribbean Sponge Aka coralliphagum. J. Nat. Prod.. 2007;70:504-509.
- [CrossRef] [Google Scholar]
- Preparative Separation of Sulfur-Containing Diketopiperazines from Marine Fungus Cladosporium sp. Using High-Speed Counter-Current Chromatography in Stepwise Elution Mode. Mar. Drugs 2015:354-365.
- [CrossRef] [Google Scholar]
- Keronopsamides, a New Class of Pigments from Marine Ciliates. Eur. J. Org. Chem.. 2010;2010:427-434.
- [CrossRef] [Google Scholar]
- Isolation and Structure Elucidation of 34-Sulfatobastadin 13, an Inhibitor of the Endothelin A Receptor, from a Marine Sponge of the Genus Ianthella. J. Nat. Prod.. 1993;56:1613-1617.
- [CrossRef] [Google Scholar]
- Discorhabdins S, T, and U, New Cytotoxic Pyrroloiminoquinones from a Deep-Water Caribbean Sponge of the Genus Batzella. J. Nat. Prod.. 2003;66:1615-1617.
- [CrossRef] [Google Scholar]
- New cytotoxic acridine alkaloids from two deep water marine sponges of the family Pachastrellidae. Tetrahedron Lett.. 1989;30:4359-4362.
- [CrossRef] [Google Scholar]
- Pyridoacridine alkaloids from deep-water marine sponges of the family Pachastrellidae: structure revision of dercitin and related compounds and correlation with the kuanoniamines. J. Org. Chem.. 1992;57:1523-1526.
- [CrossRef] [Google Scholar]
- Selective cytotoxic activity of the marine-derived batzelline compounds against pancreatic cancer cell lines. Anticancer Drugs. 2009;20
- [CrossRef] [Google Scholar]
- Dinoflagellate neurotoxins related to saxitoxin: Structures of toxins C3 and C4, and confirmation of the structure of neosaxitoxin. Tetrahedron Lett.. 1984;25:3537-3538.
- [CrossRef] [Google Scholar]
- A new bioactive sesquiterpenoid quinone from the mediterranean sea marine sponge Dysidea avara. Nat. Prod. Commun.. 2013;8
- [CrossRef] [Google Scholar]
- Isolation and structure of five lyngbyabellin derivatives from a Papua New Guinea collection of the marine cyanobacterium Lyngbya majuscula. Tetrahedron. 2005;61:11723-11729.
- [CrossRef] [Google Scholar]
- Nitrogen-Containing Volatiles from Marine Salinispora pacifica and Roseobacter-Group Bacteria. J. Nat. Prod.. 2017;80:3289-3295.
- [CrossRef] [Google Scholar]
- Epipolythiodiketopiperazines from the Marine Derived Fungus Dichotomomyces cejpii with NF-κB Inhibitory Potential. Mar. Drugs 2015:4949-4966.
- [CrossRef] [Google Scholar]
- Preclinical Evaluation of Discorhabdins in Antiangiogenic and Antitumor Models. Mar. Drugs 2018
- [CrossRef] [Google Scholar]
- New Alkaloids from the Mediterranean Sponge Hamigera hamigera. J. Organic Chem.. 2004;2:88-100.
- [CrossRef] [Google Scholar]
- Bioactive sulfonyl metabolites from the Red Sea endophytic fungus Penicillium aculeatum. Nat. Product Res.. 2022;36:2713-2721.
- [CrossRef] [Google Scholar]
- A new imidazole alkaloid from the marine sponge Leucetta microrhaphis. J. Org. Chem.. 1992;57:2176-2178.
- [CrossRef] [Google Scholar]
- Novel Adociaquinone Derivatives from the Indonesian Sponge Xestospongia sp. Mar. Drugs 2015:2617-2628.
- [CrossRef] [Google Scholar]
- Hainanerectamines A-C. Alkaloids from the Hainan Sponge Hyrtios erecta.. 2014;12:3982-3993.
- [Google Scholar]
- Nouveau derive indolique du thiadiazole-1,2,4, isole d'un tunicier (Dendrodoa grossularia) Tetrahedron Lett.. 1980;21:1457-1458.
- [CrossRef] [Google Scholar]
- Alkaloids from marine organisms. Part 8: Isolation of bisdemethylaaptamine and bisdemethylaaptamine-9-O-sulfate from an Indonesian Aaptos sp. marine sponge. Tetrahedron. 2004;60:6101-6104.
- [CrossRef] [Google Scholar]
- From anti-fouling to biofilm inhibition: New cytotoxic secondary metabolites from two Indonesian Agelas sponges. Bioorg. Med. Chem.. 2010;18:1297-1311.
- [CrossRef] [Google Scholar]
- Petrobactin Sulfonate, a New Siderophore Produced by the Marine Bacterium Marinobacter hydrocarbonoclasticus. J. Nat. Prod.. 2004;67:1897-1899.
- [CrossRef] [Google Scholar]
- Thiomarinol, a new hybrid antimicrobial antibiotic produced by a marine bacterium. J. Antibiot.. 1993;46:1834-1842.
- [CrossRef] [Google Scholar]
- Thiomarinols D, E, F and G, New Hybrid Antimicrobial Antibiotics Produced by a Marine Bacterium; Isolation, Structure, and Antimicrobial Activity. J. Antibiot.. 1997;50:449-452.
- [CrossRef] [Google Scholar]
- Thiomarinols B and C, New Antimicrobial Antibiotics Produced by a Marine Bacterium. J. Antibiot.. 1995;48:907-909.
- [CrossRef] [Google Scholar]
- Tedaniophorbasins A and B—Novel Fluorescent Pteridine Alkaloids Incorporating a Thiomorpholine from the Sponge Tedaniophorbas ceratosis. Mar. Drugs 2021
- [CrossRef] [Google Scholar]
- Lung cancer: current therapies and new targeted treatments. Lancet. 2017;389:299-311.
- [CrossRef] [Google Scholar]
- Erythrazoles A-B, Cytotoxic Benzothiazoles from a Marine-Derived Erythrobacter sp. Org. Lett.. 2011;13:6580-6583.
- [CrossRef] [Google Scholar]
- A New N-Acyl Taurine from the South China Sea Marine Sponge Callyspongia sp. Chem. Nat. Compd.. 2015;51:540-541.
- [CrossRef] [Google Scholar]
- Carbamate derivatives and sesquiterpenoids from the South China Sea gorgonian Melitodes squamata. Beilstein J. Org. Chem.. 2012;8:170-176.
- [CrossRef] [Google Scholar]
- A New 1,4-Diazepine from. South China Sea Marine Sponge Callyspongia Species.. 2010;15(871–877)
- [Google Scholar]
- A new taurine derivative from South China Sea marine sponge Axinella sp. Nat. Prod. Res.. 2013;27:1537-1541.
- [CrossRef] [Google Scholar]
- Amino Acid-Directed Strategy for Inducing the Marine-Derived Fungus Scedosporium apiospermum F41–1 to Maximize Alkaloid Diversity. Org. Lett.. 2017;19:4888-4891.
- [CrossRef] [Google Scholar]
- The Ammosamides: Structures of Cell Cycle Modulators from a Marine-Derived Streptomyces Species. Angew. Chem. Int. Ed.. 2009;48:725-727.
- [CrossRef] [Google Scholar]
- Inhibitory Effects of Epipolythiodioxopiperazine Fungal Metabolites on Isocitrate Lyase in the Glyoxylate Cycle of Candida albicans. Mar. Drugs 2021
- [CrossRef] [Google Scholar]
- Ulbactins F and G, Polycyclic Thiazoline Derivatives with Tumor Cell Migration Inhibitory Activity from Brevibacillus sp. Org. Lett.. 2016;18:1658-1661.
- [CrossRef] [Google Scholar]
- Gesashidine A, a β-Carboline Alkaloid with an Imidazole Ring from a Thorectidae Sponge. J. Nat. Prod.. 2005;68:1109-1110.
- [CrossRef] [Google Scholar]
- Reticulatins A and B and hyrtioreticulin F from the marine sponge Hyrtios reticulatus. Tetrahedron. 2013;69:7051-7055.
- [CrossRef] [Google Scholar]
- Isolation of Ciliatamide D from a Marine Sponge Stelletta sp. and a Reinvestigation of the Configuration of Ciliatamide A. J. Nat. Prod.. 2013;76:755-758.
- [CrossRef] [Google Scholar]
- Three new sesquiterpene aminoquinones from a Vietnamese Spongia sp. and their biological activities. J. Nat. Med.. 2018;72:298-303.
- [CrossRef] [Google Scholar]
- Salvadenosine, a 5′-Deoxy-5′-(methylthio) Nucleoside from the Bahamian Tunicate Didemnum sp. J. Org. Chem.. 2014;79:9992-9997.
- [CrossRef] [Google Scholar]
- Metabolites from an Antarctic Sponge-Associated Bacterium, Pseudomonas aeruginosa. J. Nat. Prod.. 1996;59:293-296.
- [CrossRef] [Google Scholar]
- Discorhabdins from the Korean Marine Sponge Sceptrella sp. J. Nat. Prod.. 2010;73:258-262.
- [CrossRef] [Google Scholar]
- Bacillamide, a novel algicide from the marine bacterium, Bacillus sp. SY-1, against the harmful dinoflagellate. Cochlodinium polykrikoides. Tetrahedron Letters. 2003;44:8005-8007.
- [CrossRef] [Google Scholar]
- Lymphoma classification update: T-cell lymphomas, Hodgkin lymphomas, and histiocytic/dendritic cell neoplasms. Expert Rev. Hematol.. 2017;10:239-249.
- [CrossRef] [Google Scholar]
- Epipolythiodioxopiperazines from Fungi: Chemistry and Bioactivities. Mini-Rev. Med. Chem.. 2011;11:728-745.
- [CrossRef] [Google Scholar]
- Marine Natural Products in Medicinal Chemistry. ACS Med. Chem. Lett.. 2018;9:959-961.
- [CrossRef] [Google Scholar]
- Novel marine sponge derived amino acids 13. Additional psammaplin derivatives from Psammaplysilla purpurea. Tetrahedron. 1991;47:2097-2102.
- [CrossRef] [Google Scholar]
- Mauritamide A and accompanying oroidin alkaloids from the sponge agelas mauritiana. Tetrahedron Lett.. 1994;35:1375-1378.
- [CrossRef] [Google Scholar]
- New Lipopeptides from the Caribbean Cyanobacterium Lyngbya majuscula. J. Nat. Prod.. 2001;64:200-203.
- [CrossRef] [Google Scholar]
- Halichondria sulfonic acid, a new HIV-1 inhibitory guanidino-sulfonic acid, and halistanol sulfate isolated from the marine sponge Halichondria rugosa Ridley & Dendy. Nat. Prod. Res.. 2006;20:1129-1135.
- [CrossRef] [Google Scholar]
- Potent antioxidant activity of a dithiocarbamate-related compound from a marine hydroid. Biochem. Pharmacol.. 1999;58:1313-1319.
- [CrossRef] [Google Scholar]
- The Unexpected Isolation of CTP-431, a Novel Thiopyrone from the Sponge Cacospongia mycofijiensis. J. Org. Chem.. 2008;73:7255-7259.
- [CrossRef] [Google Scholar]
- Wondonin, a novel compound, inhibits hypoxia-induced angiogenesis through hypoxia-inducible factor 1 alpha. FEBS Lett.. 2007;581:4977-4982.
- [CrossRef] [Google Scholar]
- Ianthesine E, a new bromotyrosine-derived metabolite from the Great Barrier Reef sponge Pseudoceratina sp. Nat. Prod. Res.. 2008;22:1257-1263.
- [CrossRef] [Google Scholar]
- Polycarpine dihydrochloride: A cytotoxic dimeric disulfide alkaloid from the indian ocean ascidian Polycarpa clavata. Tetrahedron Lett.. 1996;37:2369-2372.
- [CrossRef] [Google Scholar]
- Latrunculins: NMR study, two new toxins and a synthetic approach. Tetrahedron. 1985;41:1905-1914.
- [CrossRef] [Google Scholar]
- Iejimalide C Is a Potent V-ATPase Inhibitor, and Induces Actin Disorganization. Biol. Pharm. Bull.. 2014;37:1944-1947.
- [CrossRef] [Google Scholar]
- Novel Amino Acid Derived Natural Products from the Ascidian Atriolum robustum: Identification and Pharmacological Characterization of a Unique Adenosine Derivative. J. Med. Chem.. 2004;47:2243-2255.
- [CrossRef] [Google Scholar]
- Characterization of a Novel Gastropod Toxin (6-Bromo-2-mercaptotryptamine) That Inhibits Shaker K Channel Activity. J. Biol. Chem.. 2003;278:34934-34942.
- [CrossRef] [Google Scholar]
- 2-Debromonagelamide U, 2-Debromomukanadin G, and 2-Debromonagelamide P from Marine Sponge Agelas sp. Heterocycles. 2015;90:425-431.
- [CrossRef] [Google Scholar]
- Fisherioside a, a new steroidal glycoside from the starfish Leptasterias fisheri. Chem. Nat. Compd.. 2012;48:806-809.
- [CrossRef] [Google Scholar]
- Six new polyhydroxylated steroids conjugated with taurine, microdiscusols A-F, from the Arctic starfish Asterias microdiscus. Steroids. 2019;150:108458
- [CrossRef] [Google Scholar]
- Anticancer Activity Evaluation of Kuanoniamines A and C Isolated from the Marine Sponge Oceanapia sagittaria, Collected from the Gulf of Thailand. Mar. Drugs 2007:6-22.
- [CrossRef] [Google Scholar]
- Iejimalides C and D, new antineoplastic 24-membered macrolide sulfates from the okinawan marine tunicate Eudistoma cf. rigida. Tetrahedron Lett.. 1991;32:797-798.
- [CrossRef] [Google Scholar]
- Microxine, a New cdc2 Kinase Inhibitor from the Australian Marine Sponge Microxina Species. J. Nat. Prod.. 2001;64:525-526.
- [CrossRef] [Google Scholar]
- Suvanine Sesterterpenes and Deacyl Irciniasulfonic Acids from a Tropical Coscinoderma sp. Sponge. Journal of Natural Products. 2014;77:1396-1403.
- [CrossRef] [Google Scholar]
- Meroterpenoids from a Tropical Dysidea sp. Sponge. Journal of Natural Products. 2015;78:2814-2821.
- [CrossRef] [Google Scholar]
- Anithiactins A-C, Modified 2-Phenylthiazoles from a Mudflat-Derived Streptomyces sp. J. Nat. Prod.. 2014;77:2716-2719.
- [CrossRef] [Google Scholar]
- Symbioimine Exhibiting Inhibitory Effect of Osteoclast Differentiation, from the Symbiotic Marine Dinoflagellate Symbiodinium sp. J. Am. Chem. Soc.. 2004;126:4794-4795.
- [CrossRef] [Google Scholar]
- Symbioimine and neosymbioimine, amphoteric iminium metabolites from the symbiotic marine dinoflagellate Symbiodinium sp. Bioorg. Med. Chem.. 2005;13:5253-5258.
- [CrossRef] [Google Scholar]
- Orthoscuticellines A-E, β-Carboline Alkaloids from the Bryozoan Orthoscuticella ventricosa Collected in Australia. J. Nat. Prod.. 2020;83:422-428.
- [CrossRef] [Google Scholar]
- Prianosin A, a novel antileukemic alkaloid from the okinawan marine sponge Prianos melanos. Tetrahedron Lett.. 1987;28:4939-4942.
- [CrossRef] [Google Scholar]
- Eudistomidins B, C, and D: novel antileukemic alkaloids from the Okinawan marine tunicate Eudistoma glaucus. J. Org. Chem.. 1990;55:3666-3670.
- [CrossRef] [Google Scholar]
- Tauroacidins A and B, new bromopyrrole alkaloids possessing a taurine residue from Hymeniacidon sponge. Tetrahedron. 1997;53:16679-16682.
- [CrossRef] [Google Scholar]
- Spongiacysteine, a Novel Cysteine Derivative from Marine Sponge Spongia sp. Chem. Lett.. 2004;33:1262-1263.
- [CrossRef] [Google Scholar]
- Flavocristamides A and B, new DNA polymerase α inhibitors from a marine bacterium Flavobacterium sp. Tetrahedron. 1995;51:10487-10490.
- [CrossRef] [Google Scholar]
- Thiodiketopiperazines from the Marine-Derived Fungus Phoma sp. OUCMDZ-1847. J. Nat. Prod.. 2014;77:132-137.
- [CrossRef] [Google Scholar]
- Three New Alkaloids from the Marine Tunicate Cystodytes violatinctus. J. Org. Chem.. 1998;63:4601-4603.
- [CrossRef] [Google Scholar]
- Cycloshermilamine D, a New Pyridoacridine from the Marine Tunicate Cystodytes violatinctus. J. Nat. Prod.. 2000;63:830-831.
- [CrossRef] [Google Scholar]
- Phloeodictines A and B: new antibacterial and cytotoxic bicyclic amidinium salts from the new caledonian sponge, Phloeodictyon sp. J. Org. Chem.. 1992;57:3832-3835.
- [CrossRef] [Google Scholar]
- Nagelamides M and N, new bromopyrrole alkaloids from sponge Agelas species. Tetrahedron. 2008;64:10810-10813.
- [CrossRef] [Google Scholar]
- Ircinamine, a Novel Cytotoxic Alkaloid from Ircinia sp. Chem. Lett.. 2002;31:464-465.
- [CrossRef] [Google Scholar]
- Structural Determination, Total Synthesis, and Biological Activity of Iezoside, a Highly Potent Ca2+-ATPase Inhibitor from the Marine Cyanobacterium Leptochromothrix valpauliae. J. Am. Chem. Soc.. 2022;144:11019-11032.
- [CrossRef] [Google Scholar]
- Bromopyrrole Alkaloids from a Marine Sponge Agelas sp. Chem. Pharm. Bull.. 2014;62:499-503.
- [CrossRef] [Google Scholar]
- Agelamadin F and tauroacidin E, bromopyrrole alkaloids from an Okinawan marine sponge Agelas sp. Tetrahedron Lett.. 2015;56:4502-4504.
- [CrossRef] [Google Scholar]
- Butremycin, the 3-Hydroxyl Derivative of Ikarugamycin and a Protonated Aromatic Tautomer of 5′-Methylthioinosine from a Ghanaian Micromonospora sp. K310. Mar. Drugs 2014:999-1012.
- [CrossRef] [Google Scholar]
- Mindapyrroles A-C, Pyoluteorin analogues from a shipworm-associated bacterium. J. Nat. Prod.. 2019;82:1024-1028.
- [CrossRef] [Google Scholar]
- Eudistomins from the New Zealand Ascidian Ritterella sigillinoides. J Australian Journal of Chemistry.. 1989;42:1201-1206.
- [CrossRef] [Google Scholar]
- Eudistomin K sulfoxide - an antiviral sulfoxide from the New Zealand ascidian Ritterella sigillinoides. Tetrahedron Lett.. 1988;29:2255-2256.
- [CrossRef] [Google Scholar]
- Exploration of the Electrophilic Reactivity of the Cytotoxic Marine Alkaloid Discorhabdin C and Subsequent Discovery of a New Dimeric C-1/N-13-Linked Discorhabdin Natural Product. Mar. Drugs 2020
- [CrossRef] [Google Scholar]
- Secondary metabolites with chemical diversity from the marine-derived fungus Pseudallescheria boydii F19–1 and their cytotoxic activity. RSC Adv.. 2016;6:76206-76213.
- [CrossRef] [Google Scholar]
- Structures and Comparative Characterization of Biosynthetic Gene Clusters for Cyanosporasides, Enediyne-Derived Natural Products from Marine Actinomycetes. J. Am. Chem. Soc.. 2013;135:4171-4174.
- [CrossRef] [Google Scholar]
- Discorhabdin W, the First Dimeric Discorhabdin. J. Nat. Prod.. 2005;68:1796-1798.
- [CrossRef] [Google Scholar]
- Lodopyridones B and C from a marine sediment-derived bacterium Saccharomonospora sp. Bioorg. Med. Chem. Lett.. 2017;27:3123-3126.
- [CrossRef] [Google Scholar]
- Halenaquinone Derivatives from Tropical Marine Sponge Xestospongia sp. Heterocycles. 2012;85:895-910.
- [CrossRef] [Google Scholar]
- In vitro and in vivo anti-Vibrio vulnificus activity of psammaplin A, a natural marine compound. Mol Med Rep. 2016;14:2691-2696.
- [CrossRef] [Google Scholar]
- Ovataline: A Polyketide Isolated from the Benthic Dinoflagellate Ostreopsis cf. ovata with 5α-Reductase Inhibitory Activity in RWPE-1 Prostatic Cells. Org. Lett.. 2022;24:4182-4186.
- [CrossRef] [Google Scholar]
- Potential Antidiabetic Fumiquinazoline Alkaloids from the Marine-Derived Fungus Scedosporium apiospermum F41–1. J. Nat. Prod.. 2020;83:1082-1091.
- [CrossRef] [Google Scholar]
- Stereochemical Elucidation of Natural Products from Residual Chemical Shift Anisotropies in a Liquid Crystalline Phase. J. Am. Chem. Soc.. 2020;142:2301-2309.
- [CrossRef] [Google Scholar]
- New Discorhabdin B Dimers with Anticancer Activity from the Antarctic Deep-Sea Sponge Latrunculia biformis. Mar. Drugs 2020
- [CrossRef] [Google Scholar]
- A New Antibacterial Dioxopiperazine Alkaloid Related to Gliotoxin from a Marine Isolate of the Fungus Pseudallescheria. J. Antibiot.. 2006;59:248-250.
- [CrossRef] [Google Scholar]
- Epimeric methylsulfinyladenosine derivatives from the marine ascidian Herdmania momus. Bioorg. Med. Chem. Lett.. 2013;23:4701-4704.
- [CrossRef] [Google Scholar]
- Isoindolinone-type alkaloids from the sponge-derived fungus Stachybotrys chartarum. Tetrahedron. 2014;70:7010-7015.
- [CrossRef] [Google Scholar]
- New Discorhabdin Alkaloids from the Antarctic Deep-Sea Sponge Latrunculia biformis. Mar. Drugs 2019
- [CrossRef] [Google Scholar]
- Tridiscorhabdin and Didiscorhabdin, the First Discorhabdin Oligomers Linked with a Direct C-N Bridge from the Sponge Latrunculia biformis Collected from the Deep Sea in Antarctica. J. Nat. Prod.. 2020;83:706-713.
- [CrossRef] [Google Scholar]
- Unusual anti-inflammatory meroterpenoids from the marine sponge Dactylospongia sp. Org. Biomol. Chem.. 2018;16:6773-6782.
- [CrossRef] [Google Scholar]
- Kalkitoxin Reduces Osteoclast Formation and Resorption and Protects against Inflammatory Bone Loss. Int. J. Mol. Sci. 2021
- [CrossRef] [Google Scholar]
- Discovery of an Isothiazolinone-Containing Antitubercular Natural Product Levesquamide. J. Org. Chem.. 2020;85:6450-6462.
- [CrossRef] [Google Scholar]
- Exploring the Chemodiversity and Biological Activities of the Secondary Metabolites from the Marine Fungus Neosartorya pseudofischeri. Mar. Drugs 2014:5657-5676.
- [CrossRef] [Google Scholar]
- Advances in exploring the therapeutic potential of marine natural products. Pharmacol. Res.. 2019;147:104373
- [CrossRef] [Google Scholar]
- Pulicatins A−E, Neuroactive Thiazoline Metabolites from Cone Snail-Associated Bacteria. J. Nat. Prod.. 2010;73:1922-1926.
- [CrossRef] [Google Scholar]
- Computationally assisted structure elucidation of new 2-guanidinoethanesulfonyl sesquiterpenoid alkaloids: Agelasidines G-I from the marine sponge Agelas nakamurai. Tetrahedron. 2022;133077
- [CrossRef] [Google Scholar]
- Tridentatols D−H, Nematocyst Metabolites and Precursors of the Activated Chemical Defense in the Marine Hydroid Tridentata marginata (Kirchenpauer 1864) J. Nat. Prod.. 2002;65:681-684.
- [CrossRef] [Google Scholar]
- Tridentatols A-C, novel natural products of the marine hydroid Tridentata marginata. Tetrahedron Lett.. 1996;37:9131-9134.
- [CrossRef] [Google Scholar]
- Lissoclinotoxins: Antibiotic polysulfur derivatives from the tunicate Lissoclinum perforatum. Revised structure of lissoclinotoxin A. Tetrahedron. 1994;50:5323-5334.
- [CrossRef] [Google Scholar]
- Dechdigliotoxins A-C, Three Novel Disulfide-Bridged Gliotoxin Dimers from Deep-Sea Sediment Derived Fungus Dichotomomyces cejpii. Mar. Drugs 2019
- [CrossRef] [Google Scholar]
- Trichodermamide A and aspergillazine A, two cytotoxic modified dipeptides from a marine-derived fungus Spicaria elegans. Arch Pharm Res. 2005;28:1042-1046.
- [CrossRef] [Google Scholar]
- Pseudellones A-C, Three Alkaloids from the Marine-Derived Fungus Pseudallescheria ellipsoidea F42–3. Org. Lett.. 2015;17:5156-5159.
- [CrossRef] [Google Scholar]
- Bisthiodiketopiperazines and Acorane Sesquiterpenes Produced by the Marine-Derived Fungus Penicillium adametzioides AS-53 on Different Culture Media. J. Nat. Prod.. 2015;78:1294-1299.
- [CrossRef] [Google Scholar]
- Peniciadametizine A, a Dithiodiketopiperazine with a Unique Spiro[furan-2,7'-pyrazino[1,2-b][1,2]oxazine] Skeleton, and a Related Analogue, Peniciadametizine B, from the Marine Sponge-Derived Fungus Penicillium adametzioides. Mar. Drugs 2015:3640-3652.
- [CrossRef] [Google Scholar]
- Tetroazolemycins A and B, Two New Oxazole-Thiazole Siderophores from Deep-Sea Streptomyces olivaceus FXJ8.012. Mar. Drugs 2013:1524-1533.
- [CrossRef] [Google Scholar]
- Secondary metabolites from Antarctic marine-derived fungus Penicillium crustosum HDN153086. Nat. Prod. Res.. 2019;33:414-419.
- [CrossRef] [Google Scholar]
- Evaluation of Marine Sponge Metabolites for Cytotoxicity and Signal Transduction Activity. J. Nat. Prod.. 1993;56:915-920.
- [CrossRef] [Google Scholar]
- Evaluating Nitrogen-Containing Biosynthetic Products Produced by Saltwater Culturing of Several California Littoral Zone Gram-Negative Bacteria. J. Nat. Prod.. 2017;80:2304-2310.
- [CrossRef] [Google Scholar]
- Sungeidines from a Non-canonical Enediyne Biosynthetic Pathway. J. Am. Chem. Soc.. 2020;142:1673-1679.
- [CrossRef] [Google Scholar]
- Application of marine natural products in drug research. Bioorg. Med. Chem.. 2021;35:116058
- [CrossRef] [Google Scholar]
- Stachybotrin G, a sulfate meroterpenoid from a sponge derived fungus Stachybotrys chartarum MXH-X73. Tetrahedron Lett.. 2015;56:7053-7055.
- [CrossRef] [Google Scholar]
- Kuanoniamine C stimulates bortezomib-induced cell death via suppression of glucose-regulated protein 78 in osteosarcoma. Biochem. Biophys. Res. Commun.. 2020;527:289-296.
- [CrossRef] [Google Scholar]
- Structure of (−)-Neodysidenin from Dysidea herbacea. Implications for Biosynthesis of 5,5,5-Trichloroleucine Peptides. Org. Lett.. 2000;2:2721-2723.
- [CrossRef] [Google Scholar]
- Lodopyridone, a Structurally Unprecedented Alkaloid from a Marine Actinomycete. Org. Lett.. 2009;11:5422-5424.
- [CrossRef] [Google Scholar]
- Curacin D, aN antimitotic agent from the marine Cyanobacterium Lyngbya majuscula. Phytochemistry. 1998;49:2387-2389.
- [CrossRef] [Google Scholar]
- 1-O-Sulfatobastadins-1 and -2 from Ianthella basta (Pallas). Antagonists of the yR1-FKBP12 Ca2+. Channel.. 2004;2(176–184)
- [CrossRef] [Google Scholar]
- Thiomycalolides: New Cytotoxic Trisoxazole-Containing Macrolides Isolated from a Marine Sponge Mycale sp. J. Nat. Prod.. 1998;61:663-666.
- [CrossRef] [Google Scholar]
- Ovarian cancer. Nature Reviews Disease primers. 2016;2:16061.
- [CrossRef]
- Inhibition of Topoisomerase II Catalytic Activity by Pyridoacridine Alkaloids from a Cystodytes sp. Ascidian: A Mechanism for the Apparent Intercalator-Induced Inhibition of Topoisomerase II. J. Med. Chem.. 1994;37:3819-3827.
- [CrossRef] [Google Scholar]
- (–)-Agelasidine A from Agelas clathrodes. Zeitschrift für Naturforschung C. 2006;61:472-476.
- [CrossRef] [Google Scholar]
- Synthesis of Natural Ecteinascidins (ET-729, ET-745, ET-759B, ET-736, ET-637, ET-594) from Cyanosafracin B. J. Org. Chem.. 2003;68:8859-8866.
- [CrossRef] [Google Scholar]
- Brocazines A-F, Cytotoxic Bisthiodiketopiperazine Derivatives from Penicillium brocae MA-231, an Endophytic Fungus Derived from the Marine Mangrove Plant Avicennia marina. J. Nat. Prod.. 2014;77:1921-1927.
- [CrossRef] [Google Scholar]
- Three Diketopiperazine Alkaloids with Spirocyclic Skeletons and One Bisthiodiketopiperazine Derivative from the Mangrove-Derived Endophytic Fungus Penicillium brocae MA-231. Org. Lett.. 2016;18:5304-5307.
- [CrossRef] [Google Scholar]
- Penicibrocazines, A-E, Five new sulfide diketopiperazines from the marine-derived endophytic fungus penicillium brocae. Mar. Drugs 2015:276-287.
- [CrossRef] [Google Scholar]
- Conithiaquinones A and B, Tetracyclic Cytotoxic Meroterpenes from the Mediterranean Ascidian Aplidium conicum. Eur. J. Org. Chem.. 2013;2013:3241-3246.
- [CrossRef] [Google Scholar]
- Unusual Sulfamate Indoles and a Novel Indolo[3,2-a]carbazole from Ancorina sp.1. J. Org. Chem.. 2002;67:6671-6677.
- [CrossRef] [Google Scholar]
- Saccharothrix algeriensis NRRL B-24137, the first non-Streptomyces actinobacterium, produces holomycin after cystine feeding. Arch. Microbiol.. 2020;202:2509-2516.
- [CrossRef] [Google Scholar]
- Diverse Aromatic Metabolites in the Solitary Tunicate Cnemidocarpa irene. J. Nat. Prod.. 2020;83:3156-3165.
- [CrossRef] [Google Scholar]
- Varamines A and B, new cytotoxic thioalkaloids from Lissoclinum vareau. J. Org. Chem.. 1989;54:4256-4259.
- [CrossRef] [Google Scholar]
- Aranazoles: Extensively Chlorinated Nonribosomal Peptide-Polyketide Hybrids from the Cyanobacterium Fischerella sp. PCC 9339. Org. Lett.. 2018;20:5238-5241.
- [CrossRef] [Google Scholar]
- (-)-Agelasidine C and (-)-Agelasidine D, Two New Hypotaurocyamine Diterpenoids from the Caribbean Sea Songe Agelas clathrodes. J. Nat. Prod.. 1992;55:389-394.
- [CrossRef] [Google Scholar]
- Analysis of Cyclic Imines in Mussels (Mytilus galloprovincialis) from Galicia (NW Spain) by LC-MS/MS. Int. J. Environ. Res. Public Health 2020
- [CrossRef] [Google Scholar]
- The marine sponge metabolite mycothiazole: A novel prototype mitochondrial complex I inhibitor. Bioorg. Med. Chem.. 2010;18:5988-5994.
- [CrossRef] [Google Scholar]
- Kalkitoxin Inhibits Angiogenesis, Disrupts Cellular Hypoxic Signaling, and Blocks Mitochondrial Electron Transport in Tumor Cells. Mar. Drugs 2015:1552-1568.
- [CrossRef] [Google Scholar]
- A revised structure for the marine bromoindole derivative citorellamine. Tetrahedron Lett.. 1987;28:749-752.
- [CrossRef] [Google Scholar]
- Amaranzoles B−F, Imidazole-2-carboxy Steroids from the Marine Sponge Phorbas amaranthus. C24-N- and C24-O-Analogues from a Divergent Oxidative Biosynthesis. J. Org. Chem.. 2010;75:2453-2460.
- [CrossRef] [Google Scholar]
- Euthyroideones, Novel Brominated Quinone Methides from the Bryozoan Euthyroides episcopalis. J. Org. Chem.. 1998;63:9545-9547.
- [CrossRef] [Google Scholar]
- Isolation and Structure of Pulcherrimine, a Novel Bitter-Tasting Amino Acid, from the Sea Urchin (Hemicentrotus pulcherrimus) Ovaries. J. Agric. Food Chem.. 2000;48:5557-5560.
- [CrossRef] [Google Scholar]
- Eudistomidins E and F, new β-carboline alkaloids from the okinawan marine tunicate Eudistoma glaucus. Tetrahedron Lett.. 1991;32:3539-3542.
- [CrossRef] [Google Scholar]
- Tanjungides A and B: New Antitumoral Bromoindole Derived Compounds from Diazona cf formosa. Isolation and Total Synthesis. Mar. Drugs 2014:1116-1130.
- [CrossRef] [Google Scholar]
- Anti-infective Discorhabdins from a Deep-Water Alaskan Sponge of the Genus Latrunculia. J. Nat. Prod.. 2010;73:383-387.
- [CrossRef] [Google Scholar]
- Benzoxacystol, a benzoxazine-type enzyme inhibitor from the deep-sea strain Streptomyces sp. NTK 935. J. Antibiot.. 2011;64:453-457.
- [CrossRef] [Google Scholar]
- Marine Bacteria, XLVII – Psychrotolerant Bacteria from Extreme Antarctic Habitats as Producers of Rare Bis- and Trisindole Alkaloids. Planta Med. 2016;82:910-918.
- [Google Scholar]
- Siladenoserinols A-L: New Sulfonated Serinol Derivatives from a Tunicate as Inhibitors of p53–Hdm2 Interaction. Org. Lett.. 2013;15:322-325.
- [CrossRef] [Google Scholar]
- Identification of 3-enol sulfate of Cypridina luciferin, Cypridina luciferyl sulfate, in the sea-firefly Cypridina (Vargula) hilgendorfii. Tetrahedron. 2014;70:2161-2168.
- [CrossRef] [Google Scholar]
- Agelasidine-A, a novel sesquiterpene possessing antispasmodic activity from the okinawa sea sponge Agelas sp. Tetrahedron Lett.. 1983;24:4105-4108.
- [CrossRef] [Google Scholar]
- Physiologically active marine natural products from Porifera. VIII. Agelasidines. Novel hypotaurocyamine derivatives from the Okinawan sea sponge Agelas nakamurai Hoshino. J. Org. Chem.. 1985;50:2494-2497.
- [CrossRef] [Google Scholar]
- Penarolide Sulfates A1 and A2, New α-Glucosidase Inhibitors from a Marine Sponge Penares sp. Tetrahedron. 2000;56:8977-8987.
- [CrossRef] [Google Scholar]
- Penasulfate A, a New α-Glucosidase Inhibitor from a Marine Sponge Penares sp. J. Nat. Prod.. 2004;67:1346-1350.
- [CrossRef] [Google Scholar]
- Five New Sulfur-containing Polybrominated Bisindoles from the Red Alga Laurencia brongniartii. Heterocycles. 2005;65:2675-2682.
- [CrossRef] [Google Scholar]
- Structure revision of the rare sponge metabolite echinosulfone A, and biosynthetically related echinosulfonic acids A-D. Tetrahedron Lett.. 2020;61:151651
- [CrossRef] [Google Scholar]
- An N-acyl homolog of mycothiol is produced in marine actinomycetes. Arch. Microbiol.. 2008;190:547-557.
- [CrossRef] [Google Scholar]
- Three New Pyridoacridine Type Alkaloids from a Singaporean Ascidian. J. Nat. Prod.. 2002;65:1198-1200.
- [CrossRef] [Google Scholar]
- Eutypellazines A-M, thiodiketopiperazine-type alkaloids from deep sea derived fungus Eutypella sp. MCCC 3A00281. RSC Adv.. 2017;7:33580-33590.
- [CrossRef] [Google Scholar]
- Eutypellazines N−S, new thiodiketopiperazines from a deep sea sediment derived fungus Eutypella sp. with anti-VRE activities. Tetrahedron Lett.. 2017;58:3695-3699.
- [CrossRef] [Google Scholar]
- Somocystinamide A, a Novel Cytotoxic Disulfide Dimer from a Fijian Marine Cyanobacterial Mixed Assemblage. Org. Lett.. 2002;4:1095-1098.
- [CrossRef] [Google Scholar]
- Pateamine: a potent cytotoxin from the New Zealand Marine sponge, mycale sp. Tetrahedron Lett.. 1991;32:6411-6414.
- [CrossRef] [Google Scholar]
- Stereochemistry of theonezolides A-C. Tetrahedron Lett.. 2013;54:783-787.
- [CrossRef] [Google Scholar]
- Isolation of Caldorazole, a Thiazole-Containing Polyketide with Selective Cytotoxicity under Glucose-Restricted Conditions. Org. Lett.. 2022;24:4547-4551.
- [CrossRef] [Google Scholar]
- Cylindrospermopsin: a potent hepatotoxin from the blue-green alga Cylindrospermopsis raciborskii. J. Am. Chem. Soc.. 1992;114:7941-7942.
- [CrossRef] [Google Scholar]
- Ianthesines A-D, Four Novel Dibromotyrosine-Derived Metabolites from a Marine Sponge, Ianthella sp. Tetrahedron. 2000;56:5813-5818.
- [CrossRef] [Google Scholar]
- Shishijimicins A−C, Novel Enediyne Antitumor Antibiotics from the Ascidian Didemnum proliferum. J. Am. Chem. Soc.. 2003;125:2044-2045.
- [CrossRef] [Google Scholar]
- Antiparasitic Activity of Bromotyrosine Alkaloids and New Analogues Isolated from the Fijian Marine Sponge Aplysinella rhax. Chem. Biodivers.. 2020;17:e2000335.
- [Google Scholar]
- Inhibition of the Quorum Sensing System, Elastase Production and Biofilm Formation in Pseudomonas aeruginosa by Psammaplin A and Bisaprasin. Molecules 2022
- [CrossRef] [Google Scholar]
- Lepadins I-K, 3-O-(3′-Methylthio)acryloyloxy-decahydroquinoline Esters from a Bahamian Ascidian Didemnum sp. Assignment of Absolute Stereostructures. J. Org. Chem.. 2018;83:13670-13677.
- [CrossRef] [Google Scholar]
- Barbamide, a Chlorinated Metabolite with Molluscicidal Activity from the Caribbean Cyanobacterium Lyngbya majuscula. J. Nat. Prod.. 1996;59:427-430.
- [CrossRef] [Google Scholar]
- New 3-Hydroxyquinaldic Acid Derivatives from Cultures of the Marine Derived Actinomycete Streptomyces cyaneofuscatus M-157. Mar. Drugs 2018
- [CrossRef] [Google Scholar]
- Kinetic Studies of Antioxidant Properties of Ovothiol A. Antioxidants 2021
- [CrossRef] [Google Scholar]
- Echinosulfonic Acids A−C and Echinosulfone A: Novel Bromoindole Sulfonic Acids and a Sulfone from a Southern Australian Marine Sponge, Echinodictyum. J. Nat. Prod.. 1999;62:1246-1249.
- [CrossRef] [Google Scholar]
- Ochraceopetalin, a Mixed-Biogenetic Salt of Polyketide and Amino Acid Origins from a Marine-Derived Aspergillus ochraceopetaliformis Fungus. Mar. Drugs 2021
- [CrossRef] [Google Scholar]
- Imbricatine, an unusual benzyltetrahydroisoquinoline alkaloid isolated from the starfish Dermasterias imbricata. J. Am. Chem. Soc.. 1986;108:8288-8289.
- [CrossRef] [Google Scholar]
- Enantiomeric 1,2,3-Trithiane-Containing Alkaloids and Two New 1,3-Dithiane Alkaloids from New Zealand Ascidians. J. Org. Chem.. 2001;66:8257-8259.
- [CrossRef] [Google Scholar]
- Anti-inflammatory Thiazine Alkaloids Isolated from the New Zealand Ascidian Aplidium sp.: Inhibitors of the Neutrophil Respiratory Burst in a Model of Gouty Arthritis. J. Nat. Prod.. 2007;70:936-940.
- [CrossRef] [Google Scholar]
- Antimicrobial Diterpene Alkaloids from an Agelas citrina Sponge Collected in the Yucatán Peninsula. Mar. Drugs 2022
- [CrossRef] [Google Scholar]
- New β-Carboline Alkaloids from the Andaman Sea Sponge Dragmacidon sp. J. Nat. Prod.. 2004;67:2113-2116.
- [CrossRef] [Google Scholar]
- Sargassulfamide A, an Unprecedented Amide Derivative from the Seaweed Sargassum naozhouense. Chem. Nat. Compd.. 2020;56:98-100.
- [CrossRef] [Google Scholar]
- Peng C., Gunaherath G.M.K.B., Piggott A.M., Khalil Z., Conte M., and Capon R.J. (2010) 9-(5-Deoxy-5-thio--<sc>d</sc>-xylofuranosyl)adenine Disulfide from the Southern Australian Marine Sponge Trachycladus laevispirulifer: the First Natural Occurrence of a Nucleoside Disulfide %J Australian Journal of Chemistry. 63:873-876. https://doi.org/10.1071/CH09645.
- Cytotoxic pigments from new zealand sponges of the genus latrunculia : discorhabdins a, b and c. Tetrahedron. 1988;44:1727-1734.
- [CrossRef] [Google Scholar]
- Four New Bromotryptamine Derivatives from the Marine Bryozoan Flustra foliacea. J. Nat. Prod.. 2002;65:1633-1637.
- [CrossRef] [Google Scholar]
- Serinolamides and Lyngbyabellins from an Okeania sp. Cyanobacterium Collected from the Red Sea. J. Natural Products. 2017;80:2708-2715.
- [CrossRef] [Google Scholar]
- Isolation of Psammaplin A 11‘-Sulfate and Bisaprasin 11‘-Sulfate from the Marine Sponge Aplysinella rhax. J. Nat. Prod.. 2000;63:393-395.
- [CrossRef] [Google Scholar]
- New Cytotoxic 1,2,4-Thiadiazole Alkaloids from the Ascidian Polycarpa aurata. Org. Lett.. 2013;15:2230-2233.
- [CrossRef] [Google Scholar]
- Psammaplins from the Sponge Pseudoceratina purpurea: Inhibition of Both Histone Deacetylase and DNA Methyltransferase. J. Org. Chem.. 2003;68:3866-3873.
- [CrossRef] [Google Scholar]
- Callyspongisines A-D: bromopyrrole alkaloids from an Australian marine sponge, Callyspongia sp. Org. Biomol. Chem.. 2014;12:1579-1584.
- [CrossRef] [Google Scholar]
- Diketopiperazines and Phthalides from a Marine Derived Fungus of the Order Pleosporales. Planta Med. 2008;74:69-72.
- [Google Scholar]
- New Cytotoxic β-Carboline Alkaloids from the Marine Bryozoan, Cribricellina cribraria. J. Nat. Prod.. 1991;54:1068-1076.
- [CrossRef] [Google Scholar]
- Sinulasulfoxide and sinulasulfone, sulfur-containing alkaloids from the Indonesian soft coral Sinularia sp. Tetrahedron Lett.. 2012;53:3937-3939.
- [CrossRef] [Google Scholar]
- Phenolic constituents of Psammaplysilla. Tetrahedron Lett.. 1987;28:3229-3232.
- [CrossRef] [Google Scholar]
- Novel cytotoxic topoisomerase II inhibiting pyrroloiminoquinones from Fijian sponges of the genus Zyzzya. J. Am. Chem. Soc.. 1993;115:1632-1638.
- [CrossRef] [Google Scholar]
- Heronamycin A: a new benzothiazine ansamycin from an Australian marine-derived Streptomyces sp. Tetrahedron Lett.. 2012;53:1063-1065.
- [CrossRef] [Google Scholar]
- Marine Natural Products from Tunicates and Their Associated Microbes. Mar. Drugs 2021
- [CrossRef] [Google Scholar]
- New Cytotoxic N-Methylated β-Carboline Alkaloids from the Marine Ascidian Eudistoma gilboverde. J. Nat. Prod.. 2001;64:1454-1456.
- [CrossRef] [Google Scholar]
- New lamellarin alkaloids from an unidentified ascidian from the Arabian Sea. Tetrahedron. 1997;53:3457-3466.
- [CrossRef] [Google Scholar]
- A straight forward and first total synthesis of Penilumamides B-D. Tetrahedron Lett.. 2017;58:3347-3349.
- [CrossRef] [Google Scholar]
- Lamellarin α 20-Sulfate, an Inhibitor of HIV-1 Integrase Active against HIV-1 Virus in Cell Culture. J. Med. Chem.. 1999;42:1901-1907.
- [CrossRef] [Google Scholar]
- Preclinical pharmacology of ecteinascidin 729, a marine natural product with potent antitumor activity. Cancer Chemother. Pharmacol.. 1996;38:329-334.
- [CrossRef] [Google Scholar]
- Two novel alkaloids from the South China Sea marine sponge Dysidea sp. J. Antibiot.. 2010;63:699-701.
- [CrossRef] [Google Scholar]
- Discorhabdins I and L, Cytotoxic Alkaloids from the Sponge Latrunculia brevis. J. Nat. Prod.. 2004;67:463-465.
- [CrossRef] [Google Scholar]
- Eight-Membered Cyclic 1,2,3-Trithiocane Derivatives from Perophora viridis, an Atlantic Tunicate. Eur. J. Organic Chem. 2002:2400-2404.
- [CrossRef] [Google Scholar]
- The patellazoles inhibit protein synthesis at nanomolar concentrations in human colon tumor cells. Anticancer Drugs. 2005;16
- [CrossRef] [Google Scholar]
- Eudistomins A-Q,beta.-carbolines from the antiviral Caribbean tunicate Eudistoma olivaceum. J. Am. Chem. Soc.. 1987;109:3378-3387.
- [CrossRef] [Google Scholar]
- Cnidarians as a Source of New Marine Bioactive Compounds—An Overview of the Last Decade and Future Steps for Bioprospecting. Mar. Drugs 2011:1860-1886.
- [CrossRef] [Google Scholar]
- Bioprospection of Cytotoxic Compounds in Fungal Strains Recovered from Sediments of the Brazilian Coast. Chem. Biodivers.. 2015;12:432-442.
- [CrossRef] [Google Scholar]
- Two bromotyrosine-cysteine derived metabolites from a sponge. Tetrahedron Lett.. 1987;28:4989-4992.
- [CrossRef] [Google Scholar]
- Thelepamide: an unprecedented ketide-amino acid from Thelepus crispus, a Marine Annelid Worm. Org. Lett.. 2014;16:464-467.
- [CrossRef] [Google Scholar]
- Highly Polar Spiroisoxazolines from the Sponge Aplysina fulva. J. Nat. Prod.. 2007;70:1191-1194.
- [CrossRef] [Google Scholar]
- ʟ-Ovothiol A: The Egg Release Pheromone of the Marine Polychaete Platynereis Dumerilii: Annelida. Polychaeta. 1999;54:1145-1174.
- [CrossRef] [Google Scholar]
- Citorellamine, a new bromoindole derivative from polycitorella mariae. Tetrahedron Lett.. 1985;26:4303-4306.
- [CrossRef] [Google Scholar]
- Echinosulfonic acid D: an ESI MS n evaluation of a new cytotoxic alkaloid from the New-Caledonian sponge Psammoclemma sp. Nat. Prod. Res.. 2005;19:75-79.
- [CrossRef] [Google Scholar]
- Pteridine-, thymidine-, choline- and imidazole-derived alkaloids from the Australian ascidian, Leptoclinides durus. Org. Biomol. Chem.. 2013;11:261-270.
- [CrossRef] [Google Scholar]
- Indolo[3,2-a]carbazoles from a Deep-Water Sponge of the Genus Asteropus. J. Nat. Prod.. 2013;76:1989-1992.
- [CrossRef] [Google Scholar]
- B-90063, a Novel Endothelin Converting Enzyme Inhibitor Isolated from a New Marine Bacterium, Blastobacter sp. SANK 71894. J. Antibiot.. 1998;51:805-815.
- [CrossRef] [Google Scholar]
- Ecteinascidins: Putative Biosynthetic Precursors and Absolute Stereochemistry. J. Am. Chem. Soc.. 1996;118:9017-9023.
- [CrossRef] [Google Scholar]
- Isolations of N-Methyl-d-aspartic Acid-Type Glutamate Receptor Ligands from Micronesian Sponges. J. Nat. Prod.. 2003;66:784-787.
- [CrossRef] [Google Scholar]
- Batzellines A, B, and C. Novel pyrroloquinoline alkaloids from the sponge Batzella Sp. Tetrahedron Lett.. 1989;30:2517-2520.
- [CrossRef] [Google Scholar]
- Psammaplin A inhibits hepatitis C virus NS3 helicase. J. Nat. Med.. 2013;67:765-772.
- [CrossRef] [Google Scholar]
- Sagitol, a pyridoacridine alkaloid from the sponge Oceanapia sagittaria. Tetrahedron Lett.. 1996;37:9147-9148.
- [CrossRef] [Google Scholar]
- Cytotoxic isoquinoline quinones from sponges of the genus Petrosia. Nat. Prod. Res.. 2004;18:89-93.
- [CrossRef] [Google Scholar]
- Ircinamine B, bioactive alkaloid from marine sponge Dactylia sp. Tetrahedron Lett.. 2006;47:7871-7873.
- [CrossRef] [Google Scholar]
- Aldose reductase inhibitors from a marine sponge, Dictyodendrilla sp. J. Org. Chem.. 1993;58:7632-7634.
- [CrossRef] [Google Scholar]
- Melanoma. Lancet. 2018;392:971-984.
- [CrossRef]
- Xesto- and halenaquinone derivatives from a sponge, Adocia sp., from Truk lagoon. J. Org. Chem.. 1988;53:3922-3925.
- [CrossRef] [Google Scholar]
- Didemnolines A-D, new N9-substituted β-carbolines from the marine ascidian Didemnum sp. Tetrahedron. 1995;51:10125-10130.
- [CrossRef] [Google Scholar]
- Five new alkaloids from the tropical ascidian, Lissoclinum sp. lissoclinotoxin A is chiral. J. Org. Chem.. 1994;59:6600-6605.
- [CrossRef] [Google Scholar]
- One-Pot Synthesis of 5-Hydroxy-4H-1,3-thiazin-4-ones: Structure Revision, Synthesis, and NMR Shift Dependence of Thiasporine A. Org. Lett.. 2016;18:3070-3073.
- [CrossRef] [Google Scholar]
- Isolation, Structural Determination and Acute Toxicity of Pinnatoxins E, F and G. J. Agric. Food Chem.. 2010;58:6532-6542.
- [CrossRef] [Google Scholar]
- Bioactive Diketopiperazines and Nucleoside Derivatives from a Sponge-Derived Streptomyces Species. Marine Drugs 2019
- [Google Scholar]
- Cheminformatic Insight into the Differences between Terrestrial and Marine Originated Natural Products. J. Chem. Inf. Model.. 2018;58:1182-1193.
- [CrossRef] [Google Scholar]
- Characterization of a Potential β-Lactamase Inhibitory Metabolite from a Marine Streptomyces sp. PM49 Active Against Multidrug-Resistant Pathogens. Appl. Biochem. Biotechnol.. 2015;175:3696-3708.
- [CrossRef] [Google Scholar]
- New Bromotyrosine Metabolites from the Sponge Aplysinella rhax. Tetrahedron. 2000;56:9071-9077.
- [CrossRef] [Google Scholar]
- Wondonins A and B, new bis(dihydroxystyryl)imidazoles from a two-sponge association. Tetrahedron Lett.. 2001;42:1965-1968.
- [CrossRef] [Google Scholar]
- In vitro interaction between Ecteinascidin 743 (ET-743) and radiation, in relation to its cell cycle effects. Brit. J. Cancer. 2003;89:2305-2311.
- [CrossRef] [Google Scholar]
- Biosynthetic Pathway and Origin of the Chlorinated Methyl Group in Barbamide and Dechlorobarbamide, Metabolites from the Marine Cyanobacterium Lyngbya majuscula. Tetrahedron. 2000;56:9103-9113.
- [CrossRef] [Google Scholar]
- Unique Cyclized Thiolopyrrolones from the Marine-Derived Streptomyces sp. BTBU20218885. Mar. Drugs 2022
- [CrossRef] [Google Scholar]
- A Reassignment of (−)-Mycothiazole and the Isolation of a Related Diol. J. Nat. Prod.. 2006;69:145-147.
- [CrossRef] [Google Scholar]
- Latrunculins: novel marine toxins that disrupt microfilament organization in cultured cells. Science. 1983;219:493-495.
- [CrossRef] [Google Scholar]
- Antifungal Diterpene Alkaloids from the Caribbean Sponge Agelas citrina: Unified Configurational Assignments of Agelasidines and Agelasines. Eur. J. Org. Chem.. 2012;2012:5131-5135.
- [CrossRef] [Google Scholar]
- Total synthesis of mycothiazole, a polyketide heterocycle from marine sponges. Tetrahedron. 2003;59:6579-6593.
- [CrossRef] [Google Scholar]
- Geospallins A-C: New Thiodiketopiperazines with Inhibitory Activity against Angiotensin-Converting Enzyme from a Deep-Sea-Derived Fungus Geosmithia pallida FS140. Mar. Drugs 2018
- [CrossRef] [Google Scholar]
- Synthesis and structural reconfirmation of bacillamide B. Org. Biomol. Chem.. 2015;13:4271-4277.
- [CrossRef] [Google Scholar]
- Isobatzellines A, B, C, and D. Cytotoxic and antifungal pyrroloquinoline alkaloids from the marine sponge Batzella sp. J. Org. Chem.. 1990;55:4964-4966.
- [CrossRef] [Google Scholar]
- Gliotoxin Analogues from a Marine-Derived Fungus, Penicillium sp., and Their Cytotoxic and Histone Methyltransferase Inhibitory Activities. J. Nat. Prod.. 2012;75:111-114.
- [CrossRef] [Google Scholar]
- Anti-Inflammatory Activity of Monosubstituted Xestoquinone Analogues from the Marine Sponge Neopetrosia compacta. Antioxidants 2022
- [CrossRef] [Google Scholar]
- Ecteinascidins 770 and 786 from the Thai Tunicate Ecteinascidia thurstoni. J. Nat. Prod.. 2002;65:935-937.
- [CrossRef] [Google Scholar]
- Eudistomidins H-K, new β-carboline alkaloids from the Okinawan marine tunicate Eudistoma glaucus. Bioorg. Med. Chem. Lett.. 2011;21:4220-4223.
- [CrossRef] [Google Scholar]
- S1319: A novel β2-adrenoceptor agonist from a marine sponge Dysidea sp. Bioorg. Med. Chem. Lett.. 1999;9:1361-1364.
- [CrossRef] [Google Scholar]
- Psammaplin A, a chitinase inhibitor isolated from the fijian marine sponge Aplysinella rhax. Bioorg. Med. Chem.. 2002;10:1123-1128.
- [CrossRef] [Google Scholar]
- Schulzeines A−C, New α-Glucosidase Inhibitors from the Marine Sponge Penares schulzei1. J. Am. Chem. Soc.. 2004;126:187-193.
- [CrossRef] [Google Scholar]
- Resolution of the Confusion in the Assignments of Configuration for the Ciliatamides, Acylated Dipeptides from Marine Sponges. J. Nat. Prod.. 2017;80:2845-2849.
- [CrossRef] [Google Scholar]
- Structural determination of pteriatoxins A, B and C, extremely potent toxins from the bivalve Pteria penguin. Tetrahedron Lett.. 2001;42:3495-3497.
- [CrossRef] [Google Scholar]
- Nakijinamines C-E, New Heteroaromatic Alkaloids from the Sponge Suberites Species. Org. Lett.. 2011;13:3016-3019.
- [CrossRef] [Google Scholar]
- Ishigadine A, a new canthin-6-one alkaloid from an Okinawan marine sponge Hyrtios sp. Tetrahedron Lett.. 2018;59:4500-4502.
- [CrossRef] [Google Scholar]
- Leptosins, antitumour metabolites of a fungus isolated from a marine alga. J. Chem. Soc. Perkin Trans.. 1994;1:1859-1864.
- [CrossRef] [Google Scholar]
- Potent cytotoxic metabolites from a Leptosphaeria species. Structure determination and conformational analysis. Tetrahedron. 1995;51:3483-3498.
- [CrossRef] [Google Scholar]
- Cytotoxic metabolites from a fungal adherent of a marine alga. Phytochemistry. 1995;38:155-158.
- [CrossRef] [Google Scholar]
- Leptosins O-S, Cytotoxic Metabolites of a Strain of Leptosphaeria Sp. Isolated from a Marine Alga. HETEROCYCLES. 2004;63:641-653.
- [CrossRef] [Google Scholar]
- Isolation and Structure Assignments of Rostratins A−D, Cytotoxic Disulfides Produced by the Marine-Derived Fungus Exserohilum rostratum. J. Nat. Prod.. 2004;67:1374-1382.
- [CrossRef] [Google Scholar]
- Sulfur-containing polybromoindoles from the red alga Laurencia brongniartii. Tetrahedron. 1989;45:7301-7310.
- [CrossRef] [Google Scholar]
- New Cytotoxic Macrolides from the Sponge Fasciospongia rimosa. Chem. Lett.. 1996;25:255-256.
- [CrossRef] [Google Scholar]
- Nagelamides U-W, bromopyrrole alkaloids from a marine sponge Agelas sp. Tetrahedron Lett.. 2013;54:3794-3796.
- [CrossRef] [Google Scholar]
- Nagelamides X-Z, Dimeric Bromopyrrole Alkaloids from a Marine Sponge Agelas sp. Org. Lett.. 2013;15:3262-3265.
- [CrossRef] [Google Scholar]
- Hyrtimomines D and E, bisindole alkaloids from a marine sponge Hyrtios sp. Tetrahedron Lett.. 2013;54:4038-4040.
- [CrossRef] [Google Scholar]
- Hyrtimomines, indole alkaloids from Okinawan marine sponges Hyrtios spp. Tetrahedron. 2014;70:832-837.
- [CrossRef] [Google Scholar]
- Selective cytotoxicity of epidithiodiketopiperazine DC1149B, produced by marine-derived Trichoderma lixii on the cancer cells adapted to glucose starvation. J. Nat. Med.. 2020;74:153-158.
- [CrossRef] [Google Scholar]
- Merosesquiterpenes from the marine sponge Spongia pertusa Esper and their antifungal activities. Tetrahedron Lett.. 2022;93:153690
- [CrossRef] [Google Scholar]
- Petroquinones: trimeric and dimeric xestoquinone derivatives isolated from the marine sponge Petrosia alfiani. Tetrahedron. 2016;72:5530-5540.
- [CrossRef] [Google Scholar]
- A joint molecular networking study of a Smenospongia sponge and a cyanobacterial bloom revealed new antiproliferative chlorinated polyketides. Org. Chem. Front.. 2019;6:1762-1774.
- [CrossRef] [Google Scholar]
- Siladenoserinols M-P, sulfonated serinol derivatives from a tunicate. Tetrahedron. 2018;74:7516-7521.
- [CrossRef] [Google Scholar]
- Isolation and Total Synthesis of Stolonines A-C, Unique Taurine Amides from the Australian Marine Tunicate Cnemidocarpa stolonifera. Mar. Drugs 2015:4556-4575.
- [CrossRef] [Google Scholar]
- Ovothiols, a family of redox-active mercaptohistidine compounds from marine invertebrate eggs. Biochemistry. 1987;26:4028-4036.
- [CrossRef] [Google Scholar]
- Curacin E from the Brittle Star Ophiocoma scolopendrina. J. Nat. Prod.. 2016;79:2754-2757.
- [CrossRef] [Google Scholar]
- Questiomycins, Algicidal Compounds Produced by the Marine Bacterium Alteromonas sp. D and Their Production Cue. Molecules 2019
- [CrossRef] [Google Scholar]
- New polychlorinated amino acid derivatives from the marine sponge Dysidea herbacea. J. Org. Chem.. 1993;58:6336-6343.
- [CrossRef] [Google Scholar]
- Sagitol D, a New Thiazole Containing Pyridoacridine Alkaloid from a Vietnamese Ascidian. Nat. Prod. Commun.. 2015;10:1934578X1501000915
- [CrossRef] [Google Scholar]
- Structure-Activity Analysis of the Interaction of Curacin A, the Potent Colchicine Site Antimitotic Agent, with Tubulin and Effects of Analogs on the Growth of MCF-7 Breast Cancer Cells. Mol. Pharmacol.. 1998;53:62.
- [CrossRef] [Google Scholar]
- Dermacozines H-J Isolated from a Deep-Sea Strain of Dermacoccus abyssi from Mariana Trench Sediments. J. Nat. Prod.. 2014;77:416-420.
- [CrossRef] [Google Scholar]
- Amphiepicoccins A-J: Epipolythiodioxopiperazines from the Fish-Gill-Derived Fungus Epicoccum nigrum HDN17-88. J. Nat. Prod.. 2020;83:524-531.
- [CrossRef] [Google Scholar]
- Thiaplakortone B attenuates RANKL-induced NF-κB and MAPK signaling and dampens OVX-induced bone loss in mice. Biomed. Pharmacother.. 2022;154:113622
- [CrossRef] [Google Scholar]
- Wang D., Jiang W., Kim C.K., Bokesch H.R., Woldemichael G.M., Gryder B.E., Shern J.F., Khan J., O’keefe B.R., Beutler J.A., and Gustafson K.R. (2021) Neopetrothiazide: An Intriguing Pentacyclic Thiazide Alkaloid from the Sponge Neopetrosia sp. Organic Letters 23:3278-3281. https://doi.org/10.1021/acs.orglett.1c00743.
- Cytotoxic indole diketopiperazines from the deep sea-derived fungus Acrostalagmus luteoalbus SCSIO F457. Bioorg. Med. Chem. Lett.. 2012;22:7265-7267.
- [CrossRef] [Google Scholar]
- 2,5-Diketopiperazines from the Marine-Derived Fungus Aspergillus fumigatus YK-7. Chem. Biodivers.. 2012;9:385-393.
- [CrossRef] [Google Scholar]
- Three new sulfur-containing alkaloids, polycarpaurines A, B, and C, from an Indonesian ascidian Polycarpa aurata. Tetrahedron. 2007;63:409-412.
- [CrossRef] [Google Scholar]
- Cerebrosides of the Halotolerant Fungus Alternaria raphani Isolated from a Sea Salt Field. J. Nat. Prod.. 2009;72:1695-1698.
- [CrossRef] [Google Scholar]
- Cytotoxic p-Terphenyls from a Marine-Derived Nocardiopsis Species. J. Nat. Prod.. 2019;82:3504-3508.
- [CrossRef] [Google Scholar]
- Two additional new compounds from the marine-derived fungus pseudallescheria ellipsoidea F42–3. Molecules 2016
- [CrossRef] [Google Scholar]
- Dictyodendrins A−E, the First Telomerase-Inhibitory Marine Natural Products from the Sponge Dictyodendrilla verongiformis1. J. Org. Chem.. 2003;68:2765-2770.
- [CrossRef] [Google Scholar]
- Leukaemia: a model metastatic disease. Nat. Rev. Cancer. 2021;21:461-475.
- [CrossRef] [Google Scholar]
- Continuing Studies on the Cyanobacterium Lyngbya sp.: Isolation and Structure Determination of 15-Norlyngbyapeptin A and Lyngbyabellin D. J. Nat. Prod.. 2003;66:595-598.
- [CrossRef] [Google Scholar]
- Hypalocrinins, Taurine-Conjugated Anthraquinone and Biaryl Pigments from the Deep Sea Crinoid Hypalocrinus naresianus. J. Nat. Prod.. 2019;82:163-167.
- [CrossRef] [Google Scholar]
- Insights into Lomaiviticin Biosynthesis. Isolation and Structure Elucidation of (−)-Homoseongomycin. J. Nat. Prod.. 2013;76:1238-1241.
- [CrossRef] [Google Scholar]
- Wrasidlo W., Mielgo A., Torres V.A., Barbero S., Stoletov K., Suyama T.L., Klemke R.L., Gerwick W.H., Carson D.A., Stupack D.G., 2008. The marine lipopeptide somocystinamide A triggers apoptosis via caspase 8. In: Proceedings of the National Academy of Sciences 105:2313-2318. https://doi.org/10.1073/pnas.0712198105.
- New Thiodiketopiperazine and 3,4-Dihydroisocoumarin Derivatives from the Marine-Derived Fungus Aspergillus terreus. Mar. Drugs 2020
- [CrossRef] [Google Scholar]
- Forazoline A: Marine-Derived Polyketide with Antifungal In Vivo Efficacy. Angew. Chem. Int. Ed.. 2014;53:11583-11586.
- [CrossRef] [Google Scholar]
- Leptosins M-N1, cytotoxic metabolites from a Leptosphaeria species separated from a marine alga. Structure determination and biological activities. Tetrahedron. 2002;58:479-487.
- [CrossRef] [Google Scholar]
- Induced Production of Halogenated Epidithiodiketopiperazines by a Marine-Derived Trichoderma cf. brevicompactum with Sodium Halides. J. Nat. Prod.. 2015;78:2319-2321.
- [CrossRef] [Google Scholar]
- Induced production of a new unprecedented epitrithiodiketopiperazine, chlorotrithiobrevamide, by a culture of the marine-derived Trichoderma cf. brevicompactum with dimethyl sulfoxide. Tetrahedron Lett.. 2015;56:6262-6265.
- [CrossRef] [Google Scholar]
- Induced production of a new dipeptide with a disulfide bridge by long-term fermentation of marine-derived Trichoderma cf. brevicompactum. Tetrahedron Lett.. 2016;57:5764-5767.
- [CrossRef] [Google Scholar]
- Epipolythiodiketopiperazine and trichothecene derivatives from the NaI-containing fermentation of marine-derived Trichoderma cf. brevicompactum. J. Antibiot.. 2020;73:559-567.
- [CrossRef] [Google Scholar]
- Macrophilones from the Marine Hydroid Macrorhynchia philippina Can Inhibit ERK Cascade Signaling. J. Nat. Prod.. 2018;81:1666-1672.
- [CrossRef] [Google Scholar]
- Antimicrobial Metabolites from the Paracel Islands Sponge Agelas mauritiana. J. Nat. Prod.. 2012;75:774-778.
- [CrossRef] [Google Scholar]
- Nitrosporeusines A and B, Unprecedented Thioester-Bearing Alkaloids from the Arctic Streptomyces nitrosporeus. Org. Lett.. 2013;15:5366-5369.
- [CrossRef] [Google Scholar]
- Bioactive Sulfated Sesterterpene Alkaloids and Sesterterpene Sulfates from the Marine Sponge Fasciospongia sp. J. Nat. Prod.. 2009;72:319-323.
- [CrossRef] [Google Scholar]
- Novel propanamide analogue and antiproliferative diketopiperazines from mangrove Streptomyces sp. Q24. Nat. Prod. Res.. 2017;31:1390-1396.
- [CrossRef] [Google Scholar]
- New metabolites from the marine-derived bacterium Pseudomonas sp. ZZ820R. Fitoterapia. 2020;143(104555)
- [CrossRef] [Google Scholar]
- New Antifungal Metabolites from the Mariana Trench Sediment-Associated Actinomycete Streptomyces sp. SY1965. Mar. Drugs 2020
- [CrossRef] [Google Scholar]
- Violaceimides A-E, sulfur-containing metabolites from a sponge-associated fungus Aspergillus violaceus. Tetrahedron Lett.. 2018;59:3157-3160.
- [CrossRef] [Google Scholar]
- An expeditious total synthesis of kalkitoxins: determination of the absolute stereostructure of natural kalkitoxin. Tetrahedron. 2004;60:6859-6880.
- [CrossRef] [Google Scholar]
- Curacins B and C, New Antimitotic Natural Products from the Marine Cyanobacterium Lyngbya majuscula. J. Nat. Prod.. 1995;58:1961-1965.
- [CrossRef] [Google Scholar]
- Gliocladins A - C and Glioperazine; Cytotoxic Dioxo- or Trioxopiperazine Metabolites from a Gliocladium Sp. Separated from a Sea Hare. HETEROCYCLES. 2004;63:1123-1129.
- [CrossRef] [Google Scholar]
- Neobacillamide A, a Novel Thiazole-Containing Alkaloid from the Marine Bacterium Bacillus vallismortis C89, Associated with South China Sea Sponge Dysidea avara. Helv. Chim. Acta. 2009;92:607-612.
- [CrossRef] [Google Scholar]
- Chetracins E and F, cytotoxic epipolythiodioxopiperazines from the marine-derived fungus Acrostalagmus luteoalbus HDN13-530. RSC Adv.. 2018;8:53-58.
- [CrossRef] [Google Scholar]
- Exploration of Indole Alkaloids from Marine Fungus Pseudallescheria boydii F44–1 Using an Amino Acid-Directed Strategy. Mar. Drugs 2019
- [CrossRef] [Google Scholar]
- Pretrichodermamides D–F from a Marine Algicolous Fungus Penicillium sp. KMM 4672. Marine Drugs 2016
- [CrossRef] [Google Scholar]
- Citriperazines A-D produced by a marine algae-derived fungus Penicillium sp. KMM 4672. Nat. Prod. Res.. 2020;34:1118-1123.
- [CrossRef] [Google Scholar]
- Patellazole C: a novel cytotoxic macrolide from Lissoclinum patella. J. Am. Chem. Soc.. 1988;110:7919-7920.
- [CrossRef] [Google Scholar]
- Phorbasins D−F: Diterpenyl-taurines from a Southern Australian Marine Sponge, Phorbas sp. Org. Lett.. 2008;10:1959-1962.
- [CrossRef] [Google Scholar]
- A unique indolizinium alkaloid streptopertusacin A and bioactive bafilomycins from marine-derived Streptomyces sp. HZP-2216E. Phytochemistry. 2017;144:119-126.
- [CrossRef] [Google Scholar]
- A search for kinase inhibitors and antibacterial agents: bromopyrrolo-2-aminoimidazoles from a deep-water Great Australian Bight sponge, Axinella sp. Tetrahedron Lett.. 2012;53:3784-3787.
- [CrossRef] [Google Scholar]
- A new thiodiketopiperzaine from the marine sponge Tedania sp. Nat. Prod. Res.. 2020;34:1113-1117.
- [CrossRef] [Google Scholar]
- Laucysteinamide A, a Hybrid PKS/NRPS Metabolite from a Saipan Cyanobacterium, cf. Caldora penicillata. Marine Drugs 2017
- [CrossRef] [Google Scholar]
- MS-Derived Isotopic Fine Structure Reveals Forazoline A as a Thioketone-Containing Marine-Derived Natural Product. Org. Lett.. 2020;22:1275-1279.
- [CrossRef] [Google Scholar]
- New secondary metabolites from the marine-derived fungus talaromyces mangshanicus BTBU20211089. Marine Drugs 2022
- [Google Scholar]
- Nitrosporeusine A attenuates sepsis-associated acute kidney injury through the downregulation of IL-6/sIL-6R axis activation-mediated PGC-1α suppression. Biochem. Biophys. Res. Commun.. 2019;515:474-480.
- [CrossRef] [Google Scholar]
- Antifungal nafuredin and epithiodiketopiperazine derivatives from the mangrove-derived fungus Trichoderma harzianum D13. Front. Microbiol.. 2020;11
- [CrossRef] [Google Scholar]
- Penicisulfuranols A-F, Alkaloids from the Mangrove Endophytic Fungus Penicillium janthinellum HDN13-309. J. Nat. Prod.. 2017;80:71-75.
- [CrossRef] [Google Scholar]
- Penispirozines A-H, Three Classes of Dioxopiperazine Alkaloids with Spirocyclic Skeletons Isolated from the Mangrove-Derived Penicillium janthinellum. J. Nat. Prod.. 2020;83:2647-2654.
- [CrossRef] [Google Scholar]
- Atkamine: A New Pyrroloiminoquinone Scaffold from the Cold Water Aleutian Islands Latrunculia Sponge. Org. Lett.. 2013;15:1516-1519.
- [CrossRef] [Google Scholar]
- Two New Imidazolone-Containing Alkaloids and Further Metabolites from the Ascomycete Fungus Tricladium sp. Chem. Biodivers.. 2011;8:1914-1920.
- [CrossRef] [Google Scholar]
- Computationally Assisted Discovery and Assignment of a Highly Strained and PANC-1 Selective Alkaloid from Alaska’s Deep Ocean. J. Am. Chem. Soc.. 2019;141:4338-4344.
- [CrossRef] [Google Scholar]
Appendix A
Supplementary material
Supplementary data to this article can be found online at https://doi.org/10.1016/j.arabjc.2023.105011.
Appendix A
Supplementary material
The following are the Supplementary data to this article:Supplementary data 1
Supplementary data 1







