Translate this page into:
Discovery of novel 3-(piperazin-1-yl)propan-2-ol decorated carbazole derivatives as new membrane-targeting antibacterial agents
⁎Corresponding authors. xiangzhou@gzu.edu.cn (Xiang Zhou), syang@gzu.edu.cn (Song Yang)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
Abstract
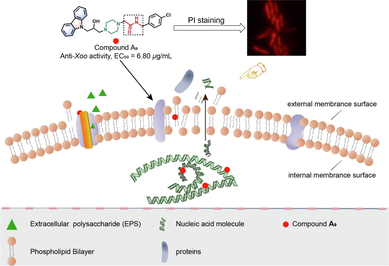
The increasing resistance problems of pathogens have drastically reduced the efficiency of existing drugs and agricultural chemicals, increasing the difficulty of preventing and controlling bacterial diseases. To create effective bactericide alternatives, a series of novel 3-(piperazin-1-yl)propan-2-ol decorated carbazole derivatives was fabricated, and their bioactivities and drug-likeness properties were evaluated. The in vitro bioassays showed that target molecules had outstanding antibacterial activity against Xanthomonas oryzae pv. oryzae (Xoo), Xanthomonas axonopodis pv. citri (Xac), and Pseudomonas syringae pv. actinidiae (Psa), with optimal EC50 values of 6.80 μg/mL (A9), 6.37 μg/mL (A9), and 10.75 μg/mL (A10), respectively. Furthermore, a series of biochemical assays indicated that title molecules could negatively impact the function of bacteria by irreversibly damaging their cell membranes, resulting in cytoplasmic components leakage (nucleic acids and proteins). In addition, phytotoxicity test and in silico toxicity predictions suggested that compound A9 had low phytotoxicity, and possessed acceptable drug-like properties. Our results showed that carbazole derivatives containing an 3-(piperazin-1-yl)propan-2-ol moiety were promising skeletons to develop novel bactericides with low toxicity for controlling refractory microbial diseases by targeting cell membranes.
Keywords
Carbazole derivatives
Cell membrane
Toxicity
Microbial diseases
Drug discovery
- BT
-
bismerthiazol
- TC
-
thiodiazole copper
- Xoo
-
Xanthomonas oryzaepv.oryzae
- Xac
-
Xanthomonas axonopodispv.citri
- Psa
-
Pseudomonas syringaepv. actinidiae
- SEM
-
Scanning electron microscopy
- HRMS
-
High-resolution mass spectrometry
- PI
-
Propidium iodide
- SARs
-
Structure–activity relationships analysis
- FTIR
-
Fourier-transform infrared spectroscopy
Abbreviations
1 Introduction
Plant bacterial diseases are posed by numerous phytopathogenic bacteria, which are difficult to control, and cause an irretrievable loss of agricultural production globally (Ali et al., 2022; Zeng et al., 2022). Some intractable phytopathogenic bacteria, such as Xanthomonas oryzae pv. oryzae (Xoo) (Sciallano et al., 2022; Zheng et al., 2021; Yang et al., 2021), Xanthomonas axonopodis pv. citri (Xac) (Zhu et al., 2022; Gao et al., 2017), and Pseudomonas syringae pv. actinidiae (Psa) (Tahir et al., 2020; Liu et al., 2022), are quite problematic for growers and can invade a variety of crops such as rice, citrus, and kiwi fruit (Moon et al., 2022; Nayem et al., 2020; Wicaksono et al., 2018). The use of chemical bactericides is currently still the most effective way to manage these diseases. Unfortunately, the existing bactericides [bismerthiazol (BT), and thiodiazole copper (TC)] possess a reduced control efficiency duo to emerging an increased bacterial resistance caused by long-term use of pesticides (Li et al., 2018; Song et al., 2022; Liu et al., 2022a, 2022b). Even worse, the use of large doses of traditional pesticides leads to environmental pollution and adversely affects human health (Zhou et al., 2022a, 2022b). Therefore, it is necessary to excavate new green pesticides with high efficiency and low toxicity. Notably, targeting membrane agents have become an attractive strategy for treating pathogenic microorganisms due to breaking the bacterial cell membrane, and killing the bacteria, thereby restricting the resistance risk (Côté et al., 2019; Cai et al., 2022).
Natural products, as a source of inspiration for pesticides, have attracted much attention due to their diverse structure, abundance of resources, and low negative effect on the environment (Zhou et al., 2022a, 2022b; Pereira, et al., 2022; Al-Yousef et al., 2021; Zang et al., 2020; Thawabteh et al., 2019). In particular, as displayed in Fig. 1, carbazole alkaloids are a crucial structural motif that is prevalent in the natural world, and many naturally and structurally modified carbazole derivatives possess extensive biological activity (Cheng et al., 2021), including antibacterial (Salih et al., 2016), antifungal (Nalli et al., 2016), antitumor (Fu et al., 2021), antioxidative (Bandgar et al., 2012), anti-inflammatory activity (Pattanashetty et al., 2018). Some carbazole alkaloids, such as ellipticine (Grau et al., 2020), carvedilol (Zhu et al., 2013), and carprofen (Parra et al., 2016), have been used in disease prevention in humans. In our previous work, carbazole derivatives containing isopropanolamine or 1, 2, 3-triazole fragments proved to be potential antimicrobial agents with obviously negative effects on the cell membrane (Zhao et al., 2019; Huang et al., 2021). Therefore, carbazoles represent a promising skeleton for use in new drugs and pesticides.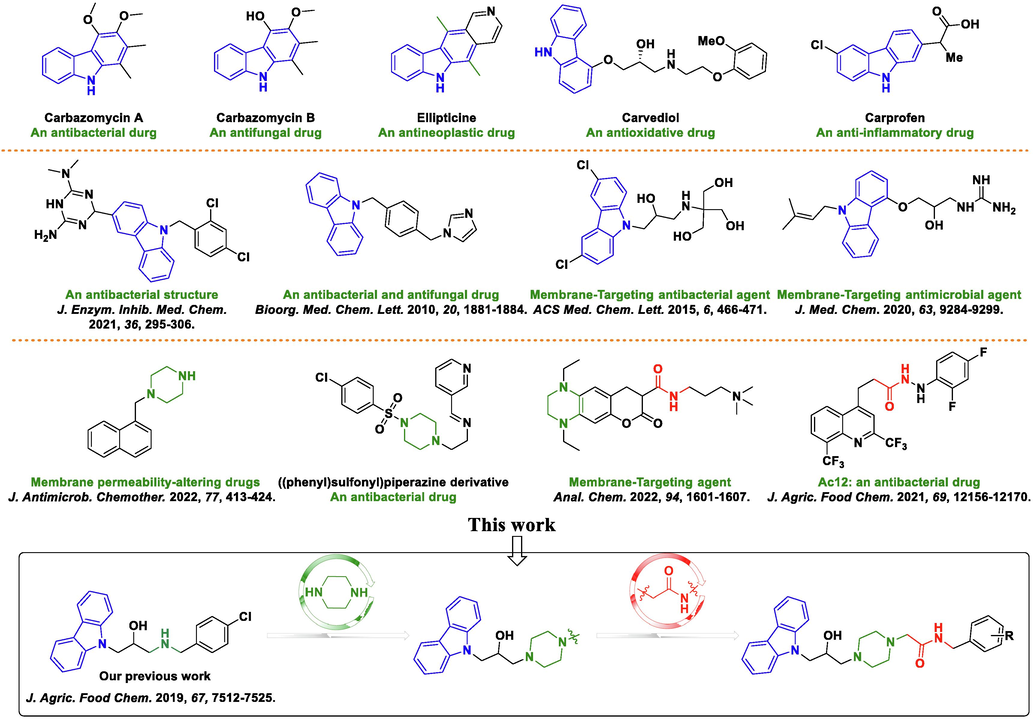
Some bioactive structures containing carbazole, piperazine, and amide moieties and the design strategy for target molecules.
In an effort to extend the potential of carbazoles, we found that some molecules possessed outstanding antimicrobial activity via targeting the cell membrane (Lin et al., 2020; Xue et al., 2021; Zhang et al., 2010). Thus, carbazoles could be regarded as a template for the fabrication and development of membrane-targeting agents (Hurley et al., 2015). More interestingly, some compounds containing piperazine (Casalone et al., 2022; Prasad et al., 2022) and amide (Feng et al., 2022a, 2022b) scaffolds also possess remarkable antimicrobial activity via targeting the cell membrane, such as an ((phenyl)sulfonyl)piperazine derivative (Prasad et al., 2022) and compound Ac12 (Chen et al., 2021). Therefore, rational molecular design might lead to the development of new membrane-targeting agents.
Herein, to explore more effective membrane-targeting agents, a series of novel 3-(piperazin-1-yl)propan-2-ol decorated carbazole derivatives were designed and synthesized via a molecular hybridization strategy. Furthermore, the antimicrobial behavior of the title molecule was investigated by various biochemical assays. To verify the toxic active of title molecules, we also investigated the phytotoxicity and in silico ‘drug-likeness’ of the synthetic compounds.
2 Materials and methods
2.1 Instruments and chemicals
NMR spectra were recorded on a JEOL–ECX 500 (500 MHz, JEOL Ltd., Tokyo, Japan) or Bruker Biospin AG-400 instrument (400 MHz, Bruker Optics, Switzerland). High-resolution mass spectrometry (HRMS) data was performed with a Q-Exactive Orbitrap MS apparatus (Thermo Fisher Scientific, USA). Scanning electron microscopy (SEM) images were obtained by a Nova NanoSEM 450 (FEI, USA). Fluorescent images were obtained by an Olympus BX53 microscope (Tokyo, Japan). Optical density (OD) was recorded on a Cytation™ 5 multi-mode reader (BioTek Instruments, Inc., USA). Cell membrane permeability assay was measured using a DDS-307 electrical conductivity analyzer. All chemicals and solvents were purchased from commercial sources.
2.2 Experimental section
The antibacterial activity in vitro and in vivo, morphological study, growth curve, and the cell membrane permeability assay were conducted via previously reported methods (Feng et al., 2022a, 2022b; Wang et al., 2022).
2.3 Propidium iodide (PI) staining experiment for Xoo cells
To further observe the damage to the bacteria cell membrane (Gan et al. 2020), the active compound A9 (anti-Xoo, EC50 = 6.80 μg/mL) was chosen to carry out a PI staining assay. Briefly, Xoo cells at logarithmic phase (OD595 = 0.6–0.8) were collected via centrifugation (8000 rpm, 2 min, 4 °C), and resuspended with phosphate buffer saline (PBS, 10 mM, pH = 7.2) to an OD595 value of 0.1. Then, 1 mL of the resuspended Xoo cells was incubated with compound A9 at various concentrations (6.8, 13.6, and 27.2 μg/mL) in a shaker (28 °C, 180 rpm) for 14 h. The same volume of DMSO was used as a negative control. After that, 100 μL of the treated Xoo cultures was incubated with 10 μL of PI dye solution (20 μg/mL) for 20 min. Finally, fluorescence images of the treated Xoo cells were collected.
2.4 Bioassay of intracellular substance leakage
The leakage of nucleic acid and protein in Xoo cells was determined according to a previously reported method (Xiang et al., 2018; Cockrell et al., 2015), and the active compound A9 (anti-Xoo, EC50 = 6.80 μg/mL) was chosen as the treatment agent. Briefly, Xoo cells at logarithmic phase (OD595 = 0.6–0.8) were collected via centrifugation (8000 rpm, 2 min, 4 °C), and resuspended in PBS buffer to an OD595 value of 0.1. Then, 1 mL of the resuspended Xoo cells (OD595 = 0.1) was incubated with different dosages of compound A9 in a shaker for 14 h. After that, the supernatants of the samples were collected via centrifugation (8000 rpm, 2 min, 4 °C). Finally, the content of nucleic acid and protein in the supernatants was immediately detected using an ultra-micro spectrophotometer instrument (NanoPhotometer N50).
2.5 Fourier-transform infrared spectroscopy (FTIR)
FTIR was applied to intensively investigate the interaction between bacterial cell membrane and title compounds (Cockrell et al., 2015; Poonprasartporn and Chan, 2021). Briefly, after undergoing the same treatment as the experiment in Section 2.3, 13.6 μg/mL of compound A9 and equivoluminal DMSO were incubated with Xoo bacterial suspension (OD595 = 0.1) in a shaker (28 °C, 180 rpm) for 14 h. The bacterial suspension was then harvested by centrifugation (8000 rpm, 2 min, 4 °C), and then re-suspended by PBS. Samples were dried and then stored in the freeze dryer for backup.
All FTIR spectra were recorded by the Nicolet iS50 FT-IR spectrophotometer in transmission mode, and the FTIR data were obtained in the wavelength range of 4000–400 cm−1.
2.6 Effect of compound A9 on extracellular polymeric substances (EPS)
The effect of molecule A9 on the EPS production of Xoo cells was assayed via our previously reported method (DuBois et al., 2002; Qi et al., 2022). Briefly, 0.1 (OD595) of Xoo cells was incubated with compound A9 at final dosages of 1.7, 3.4, 6.8, and 13.6 μg/mL in a shaker for 3 d. Finally, the relative production of EPS in different samples was measured according to the xanthan gum standard curve. Each assay was repeated at three times independently.
2.7 Scanning electron microscopy analysis
A morphological study of the Xoo cells was assayed referring to previous works (Feng et al., 2022a, 2022b; Wang et al., 2023). Briefly, 15 mL of Xoo cells (OD595 = 0.2) was transferred to sterile 50 mL centrifuge tubes. After incubating with different dosages (0, 6.8, 13.6, 27.2, and 54.4 μg/mL) of compound A9 in a shaker for 14 h, the Xoo cells were gently washed with PBS, fixed with 2.5 % glutaraldehyde, and gradually dehydrated with different concentration of ethanol. Finally, the samples were freeze-dried, gold-sprayed, and captured by SEM.
2.8 The phytotoxicity and in silico pharmacokinetics prediction
Firstly, in ChemDraw (version Ultra 14.0, PerkinElmer Informatics, Waltham, MA, USA), the structure of molecule A9 was transformed into their SMILES format. Drug-likeness predictions were then conducted using ADMETlab 2.0 (https://admetmesh.scbdd.com) accessed on 26 Oct. 2022.
2.9 Statistical analysis
All experiments were carried out at least in triplicate. Growth curves and extracellular substance leakage results were processed using Origin 2021 software. Statistical analyses were executed using one-way ANOVA in the SPSS 20.0 software. The bioassay results were reported as the mean ± SD.
3 Results and discussion
3.1 Chemistry
A facile synthetic route for 3-(piperazin-1-yl)propan-2-ol decorated carbazole derivatives was proposed. First, 9H-carbazole was firstly reacted with epibromohydrin to generate intermediate 1. Second, intermediate 1 was ring-opened with 1-boc piperazine, and then intermediate 2 bearing a piperazine group was obtained through removal of the boc group. (The more detail synthesis route could be found in Figure S1-S2). The target compounds A1–A13 were finally obtained by nucleophilic substitution reaction of intermediate 2 with intermediate 3 (Fig. 2&Figure S3). Similarly, the target compounds A14–A18 were synthesized via a ring-opening reaction between intermediate 1 and different 1-substituted piperazines (Fig. 3), the general procedure for the preparation of the compounds A19 and A20 could be found in Figure S4).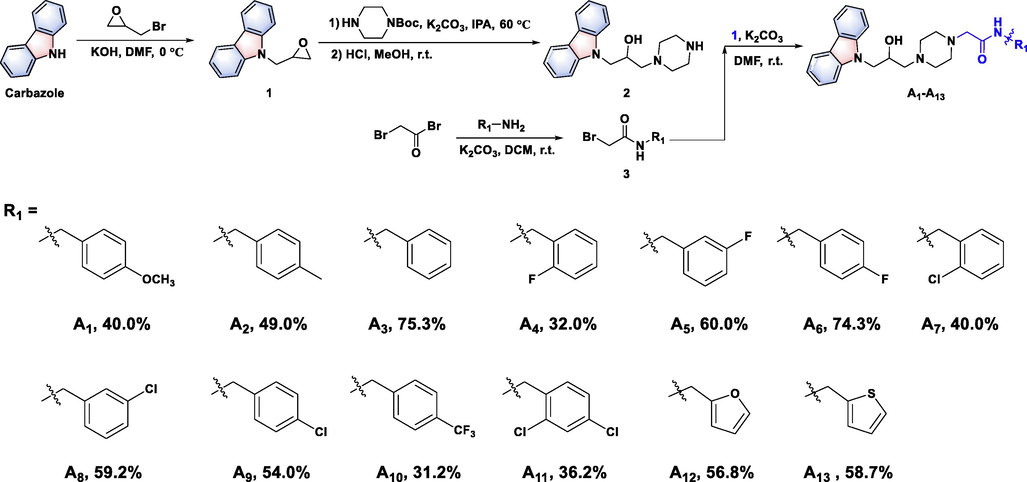
Synthesis route for target molecules A1–A13.
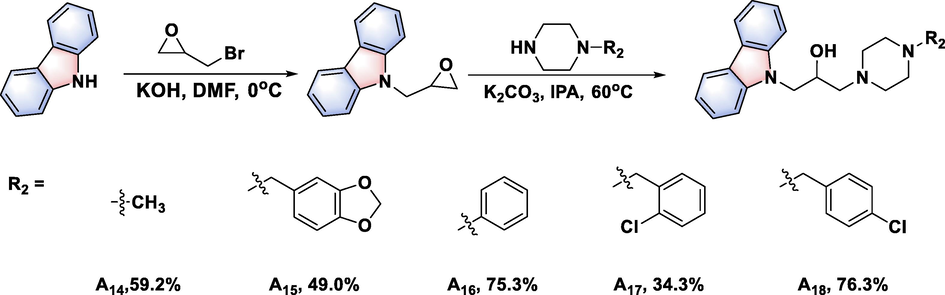
Synthetic route for compounds A14–A18.
All of the title molecules were verified by 1H NMR, 13C NMR, 19F NMR and HRMS. For instance, in 1H NMR spectra, three double peaks (d) occurred in the range of δ 8.08–8.13, 7.62–7.65 and 7.47–7.39 ppm, and the triplet peak (t) displayed at δ 7.16–7.20 ppm, which belonged to protons of the carbazole ring. The double peak (d) located in δ 8.09–8.36 ppm was signed as the characteristic peak of N–H (A1–A13). Other peaks located in δ 7.04–7.60 ppm were attributed to the phenyl group. The doublet of doublets peaks (dd) at δ 4.46–4.54 ppm and 4.26–4.33 ppm was generated by the methylene group which connected between isopropanol and carbazole moiety. The single peak (s) between 4.01 ppm and 4.28 ppm was the CH2-CH-CH2 moiety. The peaks in the range of δ 2.93–3.04 ppm and δ 3.36–3.55 ppm were the characteristic of the piperazine-CH2 of compounds A1–A13 and A15–A18. The protons of alkyl linker (isopropanol-CH2-piperazine) could be identified at δ 2.50–2.60 ppm. Multiple peaks at δ 2.30–2.55 ppm were the signals of the piperazine moiety. For compounds A1–A13, the single peak (s) at δ 4.22–4.40 ppm was singed as the benzyl-CH2. Moreover, in the 13C NMR spectra, the characteristic peaks of the carbazole moiety were signaled at δ 141.02–141.21, 125.96–125.77, 122.53–122.45, 120.30–120.55 and 119.06–119.27 ppm, respectively. Notably, a strong peak at δ 169.43–170.34 ppm was the characteristic peak of the carbonyl group of compounds A1–A13 and A19. Other signals at δ 159.25–113.78 represented the characteristic peaks of phenyl group. The signals of the piperazine moiety were observed at δ 52.74–53.90 and 47.90–48.14 ppm, and the characteristic peaks of isopropanol moiety were validated at δ 67.80–67.41, 62.43–62.08 and 59.78–53.84 ppm. Specially, split peaks at δ 160.46–162.66, 129.24–130.63, 123.61–124.75 and 114.28–115.50 ppm belonged to the carbon of phenyl-F for compounds A4–A6, respectively. In addition, in 19F NMR spectra, the phenyl-F for compounds A4–A6 and the CF3 signals of compounds A10 mainly occurred at δ −119.17, −113.59, −116.28 and −60.76 ppm, respectively. Finally, for the all the synthesized target compounds, their exact molecular weight could be found in HR(ESI)MS spectra by comparing with the theoretical values. The above analysis indicated that the title compounds were successfully prepared, and detailed date could be found in the Supplementary Materials. (Figure S5-68).
3.2 Antibacterial activities and SAR analysis
A preliminary in vitro bioassay of the title compounds was performed with the turbidimeter test (Wang et al., 2023). The commercial bactericides TC and BT were used as positive controls. The bioassay outcomes (Table 1) revealed that most of the target compounds showed remarkable bactericidal activity against all the tested bacterial strains, affording EC50 values that were much lower than those of BT and TC. Especially, compound A9 proved to have the most potent antibacterial activity against Xoo and Xac, with EC50 values of 6.80 and 6.37 μg/mL, respectively. In addition, compounds A1, A2–A8, A10 and A11 displayed excellent anti-Xoo potency, with EC50 values ranging from 7.01 to 20.17 μg/mL. Moreover, compounds A3, A7, A8, A10, and A11 revealed excellent anti-Xac activity, with EC50 values of 9.44, 7.98, 8.00, 7.67 and 9.08 μg/mL, respectively, which were quite better than TC (62.71 μg/mL) and BT (79.36 μg/mL). Interestingly, compounds A12 and A13 had lower biological activity against Xoo than other compounds, with EC50 values of 43.42 μg/mL and 43.03 μg/mL. For anti-Psa activity, compared with the control agents BT (>100 μg/mL) and TC (>100 μg/mL), compounds A6 and A10 showed significant antibacterial activity, with EC50 values of 16.67 μg/mL and 10.75 μg/mL, respectively.
Compd.
Xoo
Xac
Psa
regression equation
EC50 (μg/mL)a
regression equation
EC50 (μg/mL)a
regression equation
EC50 (μg/mL)a
A1
y = 5.5994x − 2.4072
20.03 ± 1.31
y = 5.3636x − 1.9743
19.97 ± 0.54
y = 2.6269x ± 1.4257
22.94 ± 0.48
A2
y = 7.4634x − 1.5724
7.60 ± 0.12
y = 4.0136x + 0.8309
10.93 ± 1.00
y = 2.6722x + 0.6665
41.85 ± 2.23
A3
y = 3.4381x + 1.7449
8.85 ± 0.30
y = 2.4192x + 2.6410
9.44 ± 0.24
y = 1.5671x + 2.9197
21.27 ± 1.55
A4
y = 5.3659x − 1.9700
19.90 ± 0.78
y = 3.8595x + 0.1020
18.57 ± 0.99
> 100
A5
y = 5.6853x − 2.4181
20.17 ± 0.75
y = 6.8270x + 2.5643
12.82 ± 0.55
> 100
A6
y = 5.7575x − 1.0896
11.42 ± 0.07
y = 6.1388x − 1.4993
11.45 ± 0.20
y = 1.6058x + 3.0377
16.67 ± 1.52
A7
y = 4.9967x + 0.2835
8.78 ± 0.10
y = 2.8562x + 2.4239
7.98 ± 0.35
y = 2.5163x + 0.5876
56.69 ± 8.64
A8
y = 8.2138x − 3.2233
10.03 ± 0.17
y = 4.3883x + 1.0392
8.00 ± 0.68
>100
A9
y = 11.207x − 4.3286
6.80 ± 0.16
y = 3.2233x + 2.4078
6.37 ± 0.52
y = 3.191x + 0.171
32.61 ± 5.81
A10
y = 4.1642x − 1.4784
7.01 ± 0.44
y = 2.9768x + 2.3660
7.67 ± 0.40
y = 2.7091x + 2.2057
10.75 ± 0.68
A11
y = 12.358x − 5.6733
7.31 ± 0.97
y = 4.8225x + 0.3799
9.08 ± 0.56
>100
A12
y = 5.8677x + 4.6092
43.42 ± 2.18
y = 3.1705x + 0.3144
25.49 ± 2.79
y = 2.6061x + 0.0748
77.60 ± 2.02
A13
y = 43.945x − 66.794
43.03 ± 1.61
y = 6.7760x − 4.0359
21.55 ± 2.27
>100
BTb
y = 9.8313x − 9.5823
30.43 ± 0.33
y = 3.5826x − 1.8056
79.36 ± 4.97
>100
TCb
y = 7.6119x − 9.5933
82.64 ± 2.57
y = 9.4833x − 12.045
62.71 ± 2.32
>100
The structure–activity relationships analysis (SARs) of target compounds A1–A13 against Xoo were summarized as follows: 1) compared with compound A3, the introduction of electron-withdrawing groups on the benzene ring was commonly more beneficial to enhance antibacterial potency compared to electron-donating groups, such as compounds A9 (4-Cl, 6.80 μg/mL), A10 (4-CF3, 7.01 μg/mL), A6 (4-F, 11.42 μg/mL), A3 (4-H-Ph, 8.85 μg/mL), and A1 (4-OCH3-Ph, 20.03 μg/mL); 2) the positions of substituents on the benzene ring were generally affected the anti-Xoo ability, as the following order: A9 (4-Cl, 6.80 μg/mL) > A7 (2-Cl, 8.78 μg/mL) > A8 (3-Cl, 10.03 μg/mL), and A6 (4-F, 11.42 μg/mL) > A4 (2-F, 19.90 μg/mL) > A5 (3-F, 20.17 μg/mL); 3) the compounds that introduced a benzyl amine moiety were much better than those with a five-membered heterocyclic methylamine, namely, the activity of compounds A1–A11 was superior to that of compounds A12–A13. As for anti-Xac and anti-Psa activity, the similar trends were observed. All the analyses suggested that introducing a 4-electron-withdrawing substituted benzyl amine moiety to the 3-(piperazin-1-yl)propan-2-ol decorated carbazole derivatives could significantly enhance antibacterial potency. Especially, the 4-Cl-benzyl moiety-containing compound A9 afforded the most active EC50 values of 6.80 μg/mL and 6.37 μg/mL toward Xoo and Xac, respectively, and compound A10 (4-CF3-benzyl) was the most active molecule of all the title compounds in terms of anti-Psa activity.
To study the effect of amide groups on antibacterial potency, a new series of 3-(piperazin-1-yl)propan-2-ol decorated carbazole derivatives (A14–A18) was designed and synthesized (Fig. 3), the compounds A19 and A20 could be found in Figure S4). The in vitro bioassays (Table 2) showed that the antibacterial potency of title molecules A14–A20 was dramatically decreased after the amide group was removed, affording EC50 values of over 100 μg/mL toward all the tested phytopathogenic bacteria (Xoo, Xac, and Psa); except for compound A15, which afforded a moderate EC50 value of 40.63 μg/mL against Xoo, and compound A17, which afforded outstanding biological activity with EC50 values of 11.42 μg/mL against Xoo and 16.35 μg/mL against Xac. Furthermore, compared with compound A3, compound A16 afforded EC50 values of over 100 μg/mL toward all the tested phytopathogenic bacteria (Xoo, Xac, and Psa). Similar results were found in other compounds, such as compounds A7, A8 and A9, affording the remarkable antibacterial activities than compounds A17 and A18. However, when introducing benzoyl unit and (phenyl)sulfonyl unit to parent skeleton, the compounds A19 and A20 were inactive for fighting against three pathogenic bacteria. These results suggested that the amide groups of compounds A1–A14 were key active cores for their excellent bactericidal competence.
Compd.
Xoo
Xac
Psa
regression equation
EC50 (μg/mL)a
regression equation
EC50 (μg/mL)a
regression equation
EC50 (μg/mL)a
A14
y = 5.6818x − 4.31718 4.3171
40.63 ± 1.65
>100
>100
A15
>100
>100
>100
A16
>100
>100
>100
A17
y = 3.1182x + 1.7020
11.42 ± 1.59
y = 2.1058x + 2.4445
16.35 ± 1.35
>100
A18
> 100
>100
>100
A19
> 100
>100
>100
A20
> 100
>100
>100
BTb
y = 9.8313x − 9.5823
30.43 ± 0.33
y = 3.5826x − 1.8056
79.36 ± 4.97
>100
TCb
y = 7.6119x − 9.5933
82.64 ± 2.57
y = 9.4833x − 12.045
62.71 ± 2.32
>100
Finally, a detailed SAR analysis of the title molecules was depicted in Fig. 4.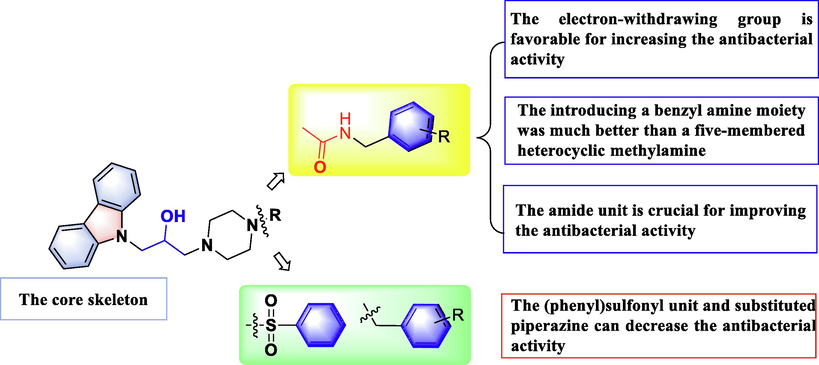
Diagram of structure–activity relationship (SAR) analyses.
3.3 Growth curves of Xoo cells triggered by compound A9
Based on in vitro bactericidal bioassays, the most active compound A9, was selected to investigate the antibacterial mechanism, and the growth curve test was used to evaluate the effect of compound A9 on Xoo (Qi et al., 2022; Zhou et al., 2021). As shown in Fig. 5, compared to the control group (0 μg/mL), cell growth was not obviously affected after treating with compound A9 at concentrations of 3.4, 6.8, and 13.6 μg/mL, respectively. However, significant change occurred when Xoo cells were incubated with compound A9 at 27.2 μg/mL. The results showed that compound A9 could significantly affect the growth of phytopathogenic bacteria at concentrations being over 27.2 μg/mL.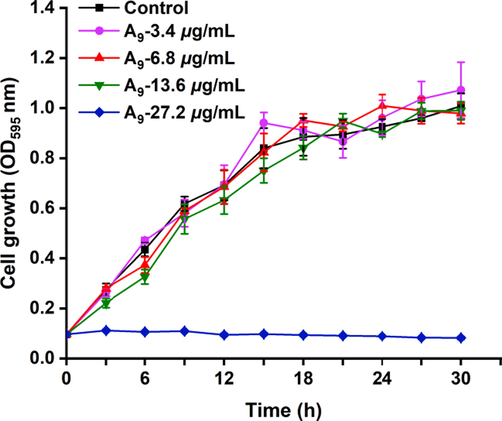
Growth curve of Xoo treated with compound A9 at 0, 3.4, 6.8, 13.6, and 27.2 μg/mL.
3.4 Morphological study of Xoo cells
To study the morphological impact of the title compound on the tested bacteria, Xoo cells were treated with the active compound A9 and then observed via SEM (Wang et al., 2022). As displayed in Fig. 6, the surface of the control Xoo was smooth and full. However, many damaged cells appeared after treating with compound A9 at 13.6 μg/mL, and more cells were corrugated or even seriously malformed and cracked with increased concentration (Fig. 6c and 6d). These findings suggested that compound A9 could negatively impact the physiological function and morphology of bacteria, which might be key factors for bacterial death.
Scanning electron microscopic results of Xoo cells after treating with compound A9 at (a) 0 μg/mL, (b) 13.6 μg/mL, (c) 27.2 μg/mL, and (d) 54.4 μg/mL. The scale bars represent 1 μm.
3.5 PI staining experiment for Xoo cells
PI, an effective indicator dye, emits red fluorescence upon intercalating with DNA (Zhang et al., 2018), and the intensity can indirectly reflect the vitality and membrane permeability of cells. As shown in Fig. 7, the fluorescence intensity increased with an increase in the dosage of compound A9. Specifically, the sample treated by compound A9 at 6.8 μg/mL showed weak red fluorescence intensity, which was comparative to that of blank controls (0 μg/mL, healthy cells). However, slight red fluorescence was observed at 13.6 μg/mL (Fig. 7g), and strong red fluorescence (Fig. 7h) was observed at 27.2 μg/mL, which suggested that the damage to the Xoo cell membrane increased with increasing dosage of compound A9. The results showed that cell membrane permeability could be negatively affected by compound A9, which was consistent with the SEM results and may be a key factor for causing cell death.
Morphology of Xoo cells stained with PI after incubation with compound A9 at (a,e) 0 μg/mL, (b,f) 6.8 μg/mL, (c,g) 13.6 μg/mL, and (d,h) 27.2 μg/mL. The image a-d was obtained from regular observation and the image e-h was obtained from fluorescent observation for Xoo cells. The scale bars represent 10 μm.
3.6 Compound A9 induced leakage of cellular nucleic acids and proteins
As an important indicator of cell membrane damage, the leakage of nucleic acids and proteins from Xoo cells was verified (Xiang et al., 2018; Ning et al., 2019). As shown in Fig. 8, the lowest extracellular protein content and nucleic acid leakage was seen in the control treatment at 4852.0 µg/mL and 280.1 µg/mL, respectively, which indicated that the cell wall and membrane was unbroken. However, a distinct increase in extracellular protein and nucleic acid content was observed after treating with compound A9, with the leakage amount being positively correlated with concentration, indicating that compound A9 could cause evident damage to the integrity of Xoo and resulted in the leakage of intracellular nucleic acids, proteins, and other functional molecules.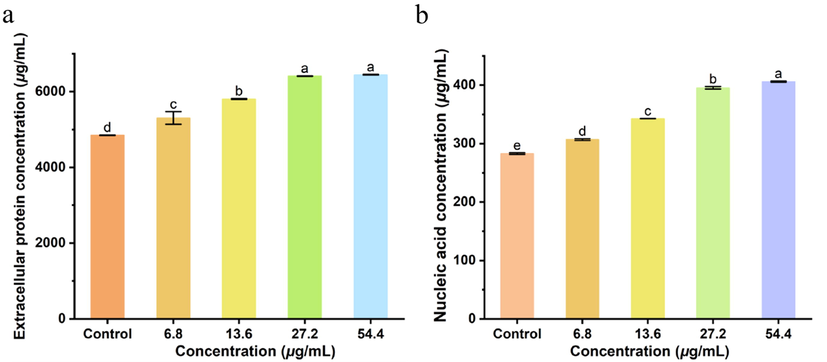
The concentration of extracellular protein and nucleic of Xoo cells after incubation with various concentrations of A9: 0 μg/mL (CK), 1.7 μg/mL, 3.4 μg/mL, 6.8 μg/mL and 13.6 μg/mL.
3.7 Effects of compound A9 on extracellular polysaccharide production
Many studies had shown that EPS could not only enhance the resistance of bacteria to harsh environments, but it could also be considered a virulence factor in evading host defenses (Qi et al., 2022). In one study, xanthan gum, as one of the main components of EPS, was produced on cell membranes (Sutherland, 1982), which served as a key indicator of the viability of phytopathogenic Xanthomonas (Picchi et al., 2021). As shown in Fig. 9, the EPS yield of Xoo cells (199.16 μg/mL) treated with compound A9 at 1.7 μg/mL was slightly lower than that of the control group (221.76 μg/mL) and displayed more significant decreases at elevated dosages. For example, the EPS yield decreased to 100.58 and 25.89 μg/mL when incubated with compound A9 at 3.4 and 6.8 μg/mL, respectively. Our findings indicated that molecule A9 could significantly suppress the production of EPS in phytopathogenic bacteria.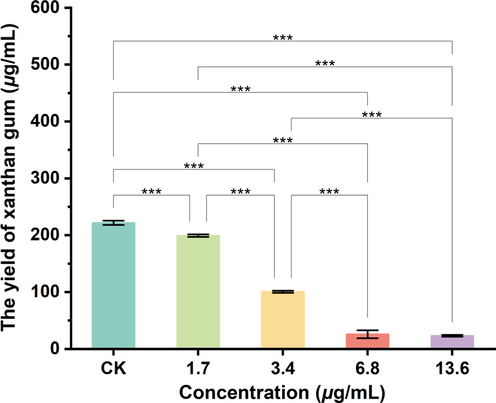
Inhibition of the reduction of extracellular polysaccharide of Xoo cells in vivo protein content after 72 h incubation in various concentrations of A9, respectively. 0 μg/mL(CK), 1.7 μg/mL, 3.4 μg/mL, 6.8 μg/mL and 13.6 μg/mL. (*p < 0.05, **p < 0.01, ***p < 0.001).
3.8 Fourier transform infrared spectroscopy
The IR bands of different biomolecular characteristics were investigated: lipids (3000–2800 cm−1), protein/amides I and II (1700–1500 cm−1), phospholipids/DNA/RNA (1500–1185 cm−1), polysaccharides (1185–900 cm−1), and the fingerprint region (900–600 cm−1) (He et al., 2021). As shown in Fig. 10, the effects of compound A9 and Xoo, individually and combined, on the bacterial membrane were analyzed via FT-IR. The outcomes reflected the changes of the molecular composition of Xoo bacteria before and after treating with compound A9. Upon treatment with 54.4 µg/mL of compound A9 + Xoo cells, the peak intensity at 3000 cm−1 and 2800 cm−1 was slightly increased compared to the control group. And the evident change was found at 1185–900 cm−1, which belonged to IR bands of polysaccharides. Meanwhile, the results were agreed with similar literature (Ojeda et al., 2008; Stenclova et al., 2019). Interestingly, compared with the control, an enhanced vibration peaks at 1500–1700 cm−1 was observed, which belonged to C—N stretching, C⚌O stretching, N—H bending and stretching, and protein/amides I and II. These outcomes were consistent with previous works (Zhou et al., 2021; Biswas et al., 2019). When the cell membrane was disturbed, the corresponding absorption peak intensity increased significantly, reflecting cell damage and death. Indicating that compound A9 could potentially change the Xoo cell membrane.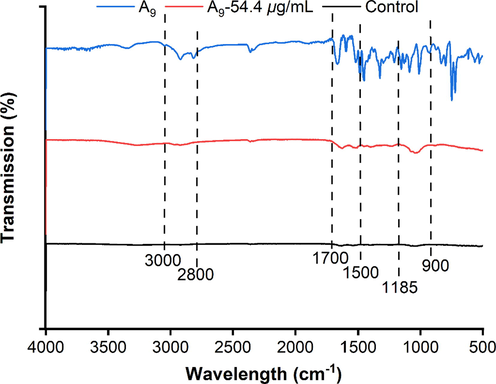
FTIR spectra (500–4000 cm−1) of Xoo cells after incubation with different concentrations (0 μg/mL, 6.8 μg/mL, and 13.6 μg/mL) of compound A9.
3.9 The phytotoxicity and in silico ‘drug-likeness’ evaluation of the title compound
Finally, the most important step for pharmaceutical development is assessing the potential of lead compounds in the early time (Zhou et al., 2022a, 2022b). Therefore, to evaluate whether molecule A9 has potential as a lead compound, the toxic activity and pharmacokinetic information was obtained. For instance, the phytotoxicity was assayed by rice plant and displayed in Fig. 11, after 7 day incubation, compound A9 exhibited non-toxicity toward rice (Zhou et al., 2021; Mukhtar et al., 2020). Furthermore, the ADMET and drug-like properties of compound A9 were achieved by ADMETlab 2.0 software (Xiong et al., 2021). The physicochemical, ADMET, and drug-like properties of the selected molecule A9 are shown in Fig. 12 and Table 3. Remarkably, compound A9 exhibited reasonable physicochemical, ADMET, and pharmaceutical properties, with molecular weight = 490.21, logS = − 3.946, logP = 4.234, and logD = 3.883. Furthermore, the ADMET and drug-like properties of compound A9 were determined: 1) it possessed definite absorption capacity with excellent activity in human intestinal absorption (HIA), and its permeability in Madin–Darby canine kidney cells (MDCK) (Pgp) was 1.5 × 10−5 cm/s; 2) it had a low VD value; 3) it was conventionally safe (e.g., low skin sensitization, eye irritation, and eye corrosion); and 4) it had great potential drug-likeness properties, including the Lipinski rule and the golden triangle rule. In addition, the metabolism property of compound A9 was provided in Table S1. In short, the above results indicated that compound A9 had acceptable pharmacokinetic characteristics and low phytotoxicity, suggesting a promising antimicrobial selectivity.
The phytotoxicity of compound A9.
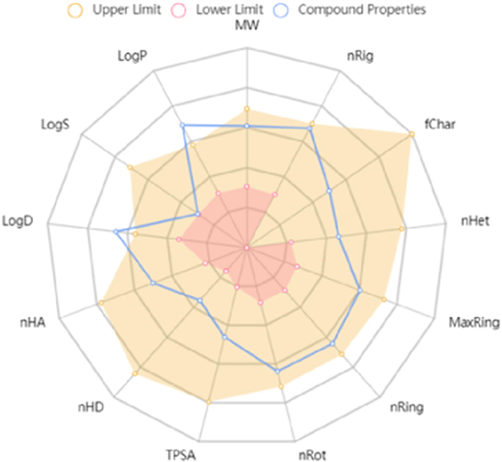
Physicochemical properties of compound A9.
Property
Value
Decision
Absorption
Caco-2 Permeability
−5.433
Medium
Madin–Darby canine kidney cells (MDCK) (Pgp) permeability (cm/s)
0.000015
Excellent
Human intestinal absorption (HIA) (%)
0.103
Excellent
Distribution
Plasma protein binding (PPB)(%)
96.88
Bad
Volume distribution (VD) (L/kg)
1.98
Excellent
Excretion
Clearance (mL/min/kg)
10.526
Excellent
The half-life (T1/2) (h)
0.054
Toxicity
Rat oral acute toxicity
0.723
Bad
Skin sensitization
0.131
Excellent
Eye corrosion
0.003
Excellent
Eye irritation
0.007
Excellent
Respiratory toxicity
0.845
Bad
Drug-likeness
Lipinski rule
Accepted
Excellent
Golden triangle
Accepted
Excellent
4 Conclusion
In summary, a series of 3-(piperazin-1-yl)propan-2-ol decorated carbazole analogues was fabricated, and assessed for antibacterial potency. The in vitro bioassay indicated that most of the target molecules had remarkable bactericidal potency against malignant phytopathogenic bacteria, including Xoo, Xac, and Psa, with EC50 values of 6.80, 6.37, and 10.75 μg/mL, respectively, which were much better than those of commercial bactericides BT (30.43, 79.36 and > 100 μg/mL) and TC (82.64, 62.71, and > 100 μg/mL). Furthermore, the SAR analysis suggested that the electron-withdrawing substituted benzyl amine moiety and the amide groups of compounds A1–A14 served as the key active core for their excellent bactericidal competence. Moreover, the SEM, PI staining, and cellular nucleic acids and proteins leakage experiments indicated that compound A9 could negatively impact the physiological function and morphology of the bacteria cell membrane, change its permeability, and lead to the leakage of cellular nucleic acids and proteins, which may be the key factor for bacterial death. The corresponding action mechanism was displayed in Fig. 13. In addition, compound A9 also could significantly inhibit the production of EPS in phytopathogenic bacteria, which indirectly explained the effect of compound A9 on the bacteria cell membrane. Finally, phytotoxicity test exhibited that compound A9 had low phytotoxicity, and in silico predictions suggested that compound A9 possessed acceptable drug-like properties. Our results showed that compound A9 could be regarded as a potential lead compound to develop novel membrane-targeting agents for treating pathogenic microorganisms’ infection.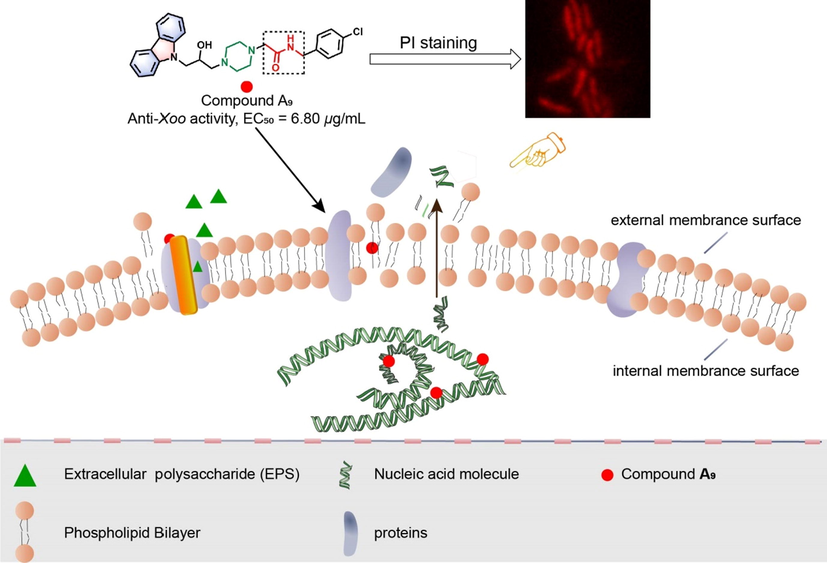
The hypothesized mechanism of action of compound A9.
CRediT authorship contribution statement
Ying-Guo Ding: Investigation, Formal analysis, Methodology, Writing – original draft. Ai-Qun Chen: Investigation. Na Wang: Investigation, Formal analysis. Zhou-Qing Long: Investigation, Formal analysis. Hong-Wu Liu: Investigation, Formal analysis. Jiao Xie: Investigation. Shi-Tao Liu: Visualization. Pu-Ying Qi: Visualization. Xiang Zhou: Investigation, Funding acquisition, Writing – review & editing. Li-Wei Liu: Investigation, Formal analysis. Song Yang: Supervision, Funding acquisition, Project administration, Methodology, Writing – review & editing.
Acknowledgements
We acknowledge the supports form National Key Research and Development Program of China (2022YFD1700300), National Natural Science Foundation of China (21877021, 32160661, 32202359), the Guizhou Provincial S&T Project (2018[4007]), the Guizhou Province [Qianjiaohe KY number (2020)004]), Program of Introducing Talents of Discipline to Universities of China (D20023, 111 Program), and GZU (Guizhou University) Found for Newly Enrolled Talent (No. 202229).
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Advances, limitations, and prospects of biosensing technology for detecting phytopathogenic bacteria. Chemosphere. 2022;296:133773
- [CrossRef] [Google Scholar]
- Phytoconstituents and pharmacological activities of cyanobacterium Fischerella ambigua. Arab. J. Chem.. 2021;14(6):103153
- [CrossRef] [Google Scholar]
- Synthesis, biological evaluation, and docking studies of 3-(substituted)-aryl-5-(9-methyl-3-carbazole)-1H-2-pyrazolines as potent anti-inflammatory and antioxidant agents. Bioorg. Med. Chem. Lett.. 2012;22(18):5839-5844.
- [CrossRef] [Google Scholar]
- Molecular mechanism of antimicrobial activity of chlorhexidine against carbapenem-resistant Acinetobacter baumannii. PLoS One.. 2019;14(10):e0224107.
- [Google Scholar]
- Juglone Inhibits Listeria monocytogenes ATCC 19115 by Targeting Cell Membrane and Protein. Foods. 2022;11(17):2558.
- [CrossRef] [Google Scholar]
- Characterization of substituted piperazines able to reverse MDR in Escherichia coli strains overexpressing resistance-nodulation-cell division (RND) efflux pumps. J. Antimicrob. Chemother.. 2022;77(2):413-424.
- [CrossRef] [Google Scholar]
- Antifungal Exploration of Quinoline Derivatives against Phytopathogenic Fungi Inspired by Quinine Alkaloids. J. Agric. Food Chem.. 2021;69(41):12156-12170.
- [CrossRef] [Google Scholar]
- Antimicrobial Chlorinated Carbazole Alkaloids from the Sponge Associated Actinomycete Streptomyces diacarni LHW51701. Chin. J. Chem.. 2021;39(5):1188-1192.
- [CrossRef] [Google Scholar]
- Differences in Physical and Biochemical Properties of Thermus scotoductus SA-01 Cultured with Dielectric or Convection Heating. Appl. Environ. Microbiol.. 2015;81(18):6285-6293.
- [CrossRef] [Google Scholar]
- Balsacone C, a New Antibiotic Targeting Bacterial Cell Membranes, Inhibits Clinical Isolates of Methicillin-Resistant Staphylococcus aureus (MRSA) Without Inducing Resistance. Front. Microbiol.. 2019;10:2341.
- [CrossRef] [Google Scholar]
- Colorimetric Method for Determination of Sugars and Related Substances. Anal. Chem.. 2002;28(3):350-356.
- [CrossRef] [Google Scholar]
- Two Water-Soluble and Wash-Free Fluorogenic Probes for Specific Lighting Up Cancer Cell Membranes and Tumors. Anal. Chem.. 2022;94(3):1601-1607.
- [CrossRef] [Google Scholar]
- Fabrication of Isopropanolamine-Decorated Coumarin Derivatives as Novel Quorum Sensing Inhibitors to Suppress Plant Bacterial Disease. J. Agric. Food Chem.. 2022;70(20):6037-6049.
- [CrossRef] [Google Scholar]
- meso-borneol- and meso-carbazole-substituted porphyrins: multifunctional chromophores with tunable electronic structures and antitumor activities. New J. Chem.. 2021;45(4):2141-2146.
- [CrossRef] [Google Scholar]
- Synergistic Effect of Propidium Iodide and Small Molecule Antibiotics with the Antimicrobial Peptide Dendrimer G3KL against Gram-Negative Bacteria. Molecules. 2020;25(23):5643.
- [CrossRef] [Google Scholar]
- Label-free quantitative proteomic analysis of inhibition of Xanthomonas axonopodis pv. citri by the novel bactericide Fubianezuofeng. Pestic. Biochem. Phys.. 2017;138:37-42.
- [CrossRef] [Google Scholar]
- Multigram scale synthesis of polycyclic lactones and evaluation of antitumor and other biological properties. Eur. J. Med. Chem.. 2020;185:111807
- [CrossRef] [Google Scholar]
- Inactivation of Staphylococcus aureus using ultrasound in combination with thyme essential oil nanoemulsions and its synergistic mechanism. LWT-Food Sci. Technol.. 2021;147:111574
- [CrossRef] [Google Scholar]
- Rational Optimization of 1,2,3-Triazole-Tailored Carbazoles As Prospective Antibacterial Alternatives with Significant In Vivo Control Efficiency and Unique Mode of Action. J. Agric. Food Chem.. 2021;69(16):4615-4627.
- [CrossRef] [Google Scholar]
- Membrane-Targeting DCAP Analogues with Broad-Spectrum Antibiotic Activity against Pathogenic Bacteria. ACS Med. Chem. Lett.. 2015;6(4):466-471.
- [CrossRef] [Google Scholar]
- Novel bisthioether derivatives containing a 1,3,4-oxadiazole moiety: design, synthesis, antibacterial and nematocidal activities. Pest Manag. Sci.. 2018;74(4):844-852.
- [CrossRef] [Google Scholar]
- Development of Highly Potent Carbazole Amphiphiles as Membrane-Targeting Antimicrobials for Treating Gram-Positive Bacterial Infections. J. Med. Chem.. 2020;63(17):9284-9299.
- [CrossRef] [Google Scholar]
- Liu, W., Zhao, C., Liu, L., Huang, D., Ma, C., Li, R., Huang, L.L., 2022. Genome-wide identification of the TGA gene family in kiwifruit (Actinidia chinensis spp.) and revealing its roles in response to Pseudomonas syringae pv. actinidiae (Psa) infection. Int. J. Biol. Macromol. 222 (Part A), 101-113. https://doi.org/10.1016/j.ijbiomac.2022.09.154.
- Pyrido [1,2-a] Pyrimidinone Mesoionic Compounds Containing Vanillin Moiety: Design, Synthesis, Antibacterial Activity, and Mechanism. J. Agric. Food Chem.. 2022;70(34):10443-10452.
- [CrossRef] [Google Scholar]
- Oryza-Specific Orphan Protein Triggers Enhanced Resistance to Xanthomonas oryzae pv. oryzae in Rice. Front. Plant Sci.. 2022;13:859375
- [CrossRef] [Google Scholar]
- Phytotoxicity of different antibiotics to rice and stress alleviation upon application of organic amendments. Chemosphere. 2020;258:127353
- [CrossRef] [Google Scholar]
- Four new carbazole alkaloids from Murraya koenigii that display anti-inflammatory and anti-microbial activities. Org. Biomol. Chem.. 2016;14(12):3322-3332.
- [CrossRef] [Google Scholar]
- Combined effect of salt stress and Xanthomonas axonopodis pv citri on citrus (Citrus aurantifolia) Heliyon. 2020;6(2):e03403.
- [Google Scholar]
- The apoptosis of Staphylococcus aureus induced by glycinin basic peptide through ROS oxidative stress response. LWT-Food Sci. Technol.. 2019;99:62-68.
- [CrossRef] [Google Scholar]
- Characterization of the cell surface and cell wall chemistry of drinking water bacteria by combining XPS, FTIR spectroscopy, modeling, and potentiometric titrations. Langmuir.. 2008;24:4032-4040.
- [CrossRef] [Google Scholar]
- Ex vivo permeation of carprofen from nanoparticles: A comprehensive study through human, porcine and bovine skin as anti-inflammatory agent. Int. J. Pharm.. 2016;501:10-17.
- [CrossRef] [Google Scholar]
- Design, Synthesis and Computational Studies of Novel Carbazole N-phenylacetamide Hybrids as Potent Antibacterial, Anti-inflammatory, and Antioxidant Agents. J. Heterocyclic Chem.. 2018;55(7):1765-1774.
- [CrossRef] [Google Scholar]
- Lignin nanoparticles: New insights for a sustainable agriculture. J. Clean. Prod.. 2022;345:131145
- [CrossRef] [Google Scholar]
- Modified Monosaccharides Content of Xanthan Gum Impairs Citrus Canker Disease by Affecting the Epiphytic Lifestyle of Xanthomonas citri subsp. citri. Microorganisms. 2021;9(6):1176.
- [CrossRef] [Google Scholar]
- Live-cell ATR-FTIR spectroscopy as a novel bioanalytical tool for cell glucose metabolism research. BBA Mol. Cell Res.. 2021;1868(7):119024
- [CrossRef] [Google Scholar]
- Design, synthesis, molecular docking and DFT computational insight on the structure of Piperazine sulfynol derivatives as a new antibacterial contender against superbugs MRSA. J. Mol. Struct.. 2022;1247:131333
- [CrossRef] [Google Scholar]
- Exploring an Innovative Strategy for Suppressing Bacterial Plant Disease: Excavated Novel Isopropanolamine-Tailored Pterostilbene Derivatives as Potential Antibiofilm Agents. J. Agric. Food Chem.. 2022;70(16):4899-4911.
- [CrossRef] [Google Scholar]
- Synthesis and antimicrobial activities of 9H-carbazole derivatives. Arab. J. Chem.. 2016;9:S781-S786.
- [CrossRef] [Google Scholar]
- The Complete Genome Resource of Xanthomonas oryzae pv. oryzae CIX2779 Includes the First Sequence of a Plasmid for an African Representative of This Rice Pathogen. Mol. Plant-Microbe Interact.. 2022;36(1):73-77.
- [CrossRef] [Google Scholar]
- Discovery of Epipodophyllotoxin-Derived B2 as Promising XooFtsZ Inhibitor for Controlling Bacterial Cell Division: Structure-Based Virtual Screening, Synthesis, and SAR Study. Int. J. Mol. Sci.. 2022;23(16):9119.
- [CrossRef] [Google Scholar]
- Cyclic Changes in the Amide Bands Within Escherichia coli Biofilms Monitored Using Real-Time Infrared Attenuated Total Reflection Spectroscopy (IR-ATR) Appl Spectrosc.. 2019;73:424-432.
- [CrossRef] [Google Scholar]
- Biosynthesis of microbial exopolysaccharides. Adv. Microb. Physiol.. 1982;23:79-150.
- [CrossRef] [Google Scholar]
- QTL Mapping for Resistance to Cankers Induced by Pseudomonas syringae pv. actinidiae (Psa) in a Tetraploid Actinidia chinensis Kiwifruit Population. Pathogens. 2020;9(11):967.
- [Google Scholar]
- The Biological Activity of Natural Alkaloids against Herbivores. Cancerous Cells and Pathogens. Toxins. 2019;11(11):656.
- [CrossRef] [Google Scholar]
- Discovery of novel rost-4-ene derivatives as potential plant activators for preventing phytopathogenic bacterial infection: Design, synthesis and biological studies. Pest Manag. Sci.. 2022;78(8):3404-3415.
- [CrossRef] [Google Scholar]
- Novel 1,3,4-oxadiazole thioether and sulfone derivatives bearing a flexible N-heterocyclic moiety: Synthesis, characterization, and anti-microorganism activity. Arab. J. Chem.. 2023;16(2):104479
- [CrossRef] [Google Scholar]
- Biological control of Pseudomonas syringae pv. actinidiae (Psa), the causal agent of bacterial canker of kiwifruit, using endophytic bacteria recovered from a medicinal plant. Biol. Control.. 2018;116:103-112.
- [CrossRef] [Google Scholar]
- Antibacterial activity and a membrane damage mechanism of plasma-activated water against Pseudomonas deceptionensis CM2. LWT-Food Sci. Technol.. 2018;96:395-401.
- [CrossRef] [Google Scholar]
- ADMETlab 2.0: an integrated online platform for accurate and comprehensive predictions of ADMET properties. Nucleic Acids Res.. 2021;49(W1):W5-W14.
- [CrossRef] [Google Scholar]
- Design, synthesis and evaluation of carbazole derivatives as potential antimicrobial agents. J. Enzyme. Inhib. Med. Chem.. 2021;36(1):295-306.
- [CrossRef] [Google Scholar]
- Dysregulation of ClpP by Small-Molecule Activators Used Against Xanthomonas oryzae pv. oryzae Infections. J. Agric. Food Chem.. 2021;69(27):7545-7553.
- [CrossRef] [Google Scholar]
- Overcoming acquired resistance of EGFR-mutant NSCLC cells to the third generation EGFR inhibitor, osimertinib, with the natural product honokiol. Mol. Oncol.. 2020;14(4):882-895.
- [CrossRef] [Google Scholar]
- Degradable Bactericide Constructed Using a Charge-Reversal Surfactant against Plant Pathogenic Bacteria. ACS Appl. Mater. Interfaces. 2022;14(8):10134-10141.
- [CrossRef] [Google Scholar]
- Cell permeability and nuclear DNA staining by propidium iodide in basidiomycetous yeasts. Appl. Microbiol. Biotechnol.. 2018;102(9):4183-4191.
- [CrossRef] [Google Scholar]
- Synthesis, antibacterial and antifungal activities of some carbazole derivatives. Bioorg. Med. Chem. Lett.. 2010;20(6):1881-1884.
- [CrossRef] [Google Scholar]
- Identification of Racemic and Chiral Carbazole Derivatives Containing an Isopropanolamine Linker as Prospective Surrogates against Plant Pathogenic Bacteria: In Vitro and In Vivo Assays and Quantitative Proteomics. J. Agric. Food Chem.. 2019;67(26):7512-7525.
- [CrossRef] [Google Scholar]
- Elucidation of the Pathogenicity-Associated Regulatory Network in Xanthomonas oryzae pv. oryzae. mSystems. 2021;6(2):e00789-e820.
- [CrossRef] [Google Scholar]
- The discovery of natural 4'-demethylepipodophyllotoxin from renewable Dysosma versipellis species as a novel bacterial cell division inhibitor for controlling intractable diseases in rice. Ind. Crop. Prod.. 2021;174:114182
- [CrossRef] [Google Scholar]
- Discovery of Simple Diacylhydrazine-Functionalized Cinnamic Acid Derivatives as Potential Microtubule Stabilizers. Int. J. Mol. Sci.. 2022;23(20):12365.
- [CrossRef] [Google Scholar]
- Mining and characterization of oxidative stress-related binding proteins of parthenolide in Xanthomonas oryzae pv. oryzae. Pest Manag. Sci.. 2022;78(8):3345-3355.
- [CrossRef] [Google Scholar]
- Discovery of novel N-substituted carbazoles as neuroprotective agents with potent anti-oxidative activity. Eur. J. Med. Chem.. 2013;68:81-88.
- [CrossRef] [Google Scholar]
- Antibacterial Activity of Allicin-Inspired Disulfide Derivatives against Xanthomonas axonopodis pv. citri. Int. J. Mol. Sci.. 2022;23(19):11947.
- [CrossRef] [Google Scholar]
Appendix A
Supplementary material
Supplementary data to this article can be found online at https://doi.org/10.1016/j.arabjc.2023.104991.
Appendix A
Supplementary material
The following are the Supplementary data to this article:Supplementary data 1
Supplementary data 1







