Translate this page into:
Exploring the molecular mechanism of Parkinson's disease triggered by combined atrazine and simazine exposure
⁎Corresponding author. bihaoran1989@sina.com (Haoran Bi)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Occupational exposure to herbicides is an important factor for the development of Parkinson's disease (PD). Atrazine (ATR) and simazine (SIM) are dopamine neurotoxins that can cause PD-like symptoms, but the mechanism is unknown. We analyzed the differentially expressed genes (DEGs) in the substantia nigra of PD patients and controls by using the PD dataset GSE20333 from the Gene Expression Omnibus (GEO) database. The potential intersectional targets of ATR and SIM combined exposure induced PD were screened by network pharmacology. The intersection targets were used for enrichment analysis and to construct protein–protein interaction networks. The binding potentials between hub targets and triazine herbicides were validated using molecular docking, and the cellular exposure models were also constructed to validate the screened results. We identified 934 genes that showed differential expression between individuals with PD and healthy controls. Using network pharmacology, we further refined our search to 151 targets of herbicides known to contribute to PD, and identified eight potential core targets: CDK2, CYCS, FOS, LCK, NTRK2, PDGFRB, PGR, and VEGFA. Finally, molecular docking and molecular biological detection results showed that ATR and SIM each exhibit strong binding affinity to CDK2, CYCS and PGR, both ATR and SIM exposure alone or in combination can affect the expression of core targets. Our study indicates that combined exposure to ATR and SIM may play a critical role in the development of PD and offers a new perspective on occupational prevention and treatment strategies.
Keywords
Triazine herbicides
Parkinson's disease
Atrazine
Simazine
Environmental factors
1 Background
Parkinson's disease (PD) ranks as the second most prevalent neurodegenerative disease characterized by a progressive decline in neurological function, trailing only Alzheimer's disease (Jiang et al., 2020). The main clinical manifestations of PD consist of asymmetrical bradykinesia, resting tremor, rigidity, and postural instability (Cabezas et al., 2014; Daviaud et al., 2014). The gradual loss of dopamine-producing neurons in the midbrain is the main contributor to motor dysfunction in PD (Grealish et al., 2010). PD is influenced by both genetic and environmental factors (Kim et al., 2022) and, in particular, pesticide exposure is a well-established environmental risk factor (Zhang et al., 2018).
Triazine herbicides are commonly used in the cultivation of crops like maize, sugarcane, and sorghum, and may accumulate in the soil, groundwater, and food products, posing potential health risks to agricultural practitioners (Buglak et al., 2019; Xu et al., 2019). Atrazine (ATR) and simazine (SIM) are typical triazine herbicides, which are used widely worldwide. At present, in vivo and in vitro experiments have shown that ATR or SIM exposure can damage dopaminergic neurons and cause PD-like symptoms (Hossain and Filipov, 2008; Walters et al., 2015; Li et al., 2017). Considering the chemical structural similarity of ATR and SIM, the combined exposure to these herbicides might be an important environmental factor contributing to the development of PD. However, there are few studies on the combination of ATR and SIM exposure, and the mechanisms that may cause PD are still unknown.
In this study, we obtained differentially expressed genes (DEGs) for PD by analyzing brain nigrostriatal microarray data from PD patients and controls. The differential targets of combined ATR and SIM exposure leading to PD were further screened by network pharmacology and validated by molecular docking, as well as molecular biological detection. Our findings suggest that the combined exposure of ATR and SIM could be associated with the onset of PD. This implies the necessity for additional investigation into the molecular mechanisms that underlie this connection.
2 Materials and methods
2.1 Genetic microarray data sources
The dataset GSE20333 was obtained from the Gene Expression Omnibus (GEO) database (https://www.ncbi.nlm.nih.gov/). The experiment was based on the GPL201 platform and conducted using the [HG-Focus] Affymetrix Human HG-Focus Target Array, which analyzed 12 substantia nigra of the midbrain. The samples included six healthy control patients and six PD patients, who were diagnosed clinically and neuropathologically.
2.2 DEG analysis
We obtained the raw data in MINiML file format. Subsequently, we used the “normalize quantiles” function from the preprocess Core package in R software (version 3.4.1) to normalize the microarray data. To convert probes into gene symbols, we used the annotation information provided in the normalized data platform. Any probes matching multiple genes were removed from the datasets. Finally, we calculated the average expression value of genes that were measured by multiple probes to obtain the final expression value. The DEGs with a P value < 0.05 and |log2FC |≥ 0.585 were identified as PD-related genes.
2.3 Network pharmacological analysis
To predict potential targets of ATR and SIM, we consulted several databases, including SwissTargetPrediction (https://www.swisstargetprediction.ch), the Comparative Toxicogenomics Database (CTD, https://ctdbase.org/), STITCH (https://stitch.embl.de/), and GeneCards (https://www.genecards.org/). We obtained the 2D structures of ATR and SIM from the PubChem database (https://pubchem.ncbi.nlm.nih.gov/) and uploaded them to these four databases for analysis. The SwissTargetPrediction and CTD screening conditions were set to default with the exception of specifying the species as Homo sapiens. The STITCH database settings were also set to default, except for specifying the species as Homo sapiens and setting the minimum required interaction score to 0.4.
To identify the intersection targets of triazine herbicides that may contribute to PD, we used the Draw Venn Diagram tool. Once we found the intersecting targets, we built a protein–protein interaction (PPI) network using the STRING database for the species Homo sapiens with a minimum required interaction score of 0.4. We then visualized the PPI network using Cytoscape 3.6.0.
2.4 Gene ontology (GO) and kyoto encyclopedia of genes and genomes (KEGG) enrichment analysis
We used the Metascape database to conduct an enrichment analysis of GO terms and KEGG pathways (species Homo sapiens), with statistical significance determined at P < 0.05. The top 10 GO terms for biological process (BP), cellular component (CC), and molecular function (MF), as well as the top 10 KEGG pathways, were selected for further investigation.
2.5 Hub target screening
To calculate the top 10 hub targets, we used six cytoHubba methods: closeness, degree, edge percolated component (EPC), maximal clique centrality (MCC), maximum neighborhood component (MNC), and radiality. We then used the Draw Venn Diagram tool to intersect the top 10 targets obtained by each of the six methods, resulting in the identification of hub targets.
2.6 Molecular docking
We obtained the 3D structures of ATR and SIM from the PubChem database, which were then converted into the Mol2 format via UCSF Chimera software version 1.16. To prepare the core targets for molecular docking, we retrieved their 3D structures from the Protein Data Bank and imported them into UCSF Chimera software for hydrogenation, dehydration, ligand removal, and other necessary processes. The SwissDock online platform was used to perform molecular docking of the triazine herbicides and hub targets, which was done to evaluate their binding activity. The results of the docking process were visualized through UCSF Chimera software.
2.7 Cell culture and treatment
SH-SY5Y neuroblastoma cells were purchased from Shanghai Guandao Biological Engineering Co., Ltd (Shanghai, China). The cells were cultured in MEM/F12 (Hyclone, Logan, UT, USA) containing 15 % fetal bovine serum (ScienCell, Carlsbad, CA, USA) and 1 % double antibody (Hyclone). ATR (97 % pure) and SIM (96 % pure) was obtained from Aladdin Reagent Co., Ltd (Shanghai, China). Solutions of ATR and SIM (100, 200, 400, 600, and 800 μM) used for cell treatment were prepared by dissolving in DMSO (MP Biomedicals, OH, USA), with a final DMSO concentration < 0.1 %.
2.8 Cell viability assay and real-time quantitative polymerase chain reaction (qPCR)
Cell viability was assessed using a Cell Counting Kit-8 (CCK8) kit (Biosharp, Hefei, China) following the manufacturer’s instructions. The total RNA was isolated using TRIzol reagent and synthesized into cDNA for quantification according to the manufacturer’s instructions (Takara, Tokyo, Japan). The above methods were the same as in our previous study (Li and Bi, 2021). The primer sequences (Table S1) were designed and synthesized by ServiceBio Co., Ltd (Wuhan, China).
2.9 Statistical analysis
All statistical analyses were performed using SPSS 26.0 software (SPSS, Chicago, IL, USA), the results were represented as the mean ± standard error of the mean (SEM). The data were analyzed with a one-way ANOVA. Multiple group comparisons used Dunnett’s multiple tests and p-values less than 0.05 (p < 0.05) indicated significant differences.
3 Results
3.1 DEG screening and network pharmacological analysis
In total, we identified 8416 mRNAs, of which 934 were differentially expressed (802 upregulated and 132 downregulated, Fig. 1a and b). Through the SwissTargetPrediction, CTD, STITCH, and GeneCards databases, we obtained 3063 targets for ATR, 31 targets for SIM, and 3076 targets of triazine herbicides after removing duplicate targets. Through Venn analysis, we finally obtained 151 intersection targets (122 upregulated and 29 downregulated, Fig. 1c). The intersectional targets were used to construct a PPI network using the STRING database, which included 151 nodes and 181 edges with an average node degree of 2.4 (Fig. 1d, Table S2).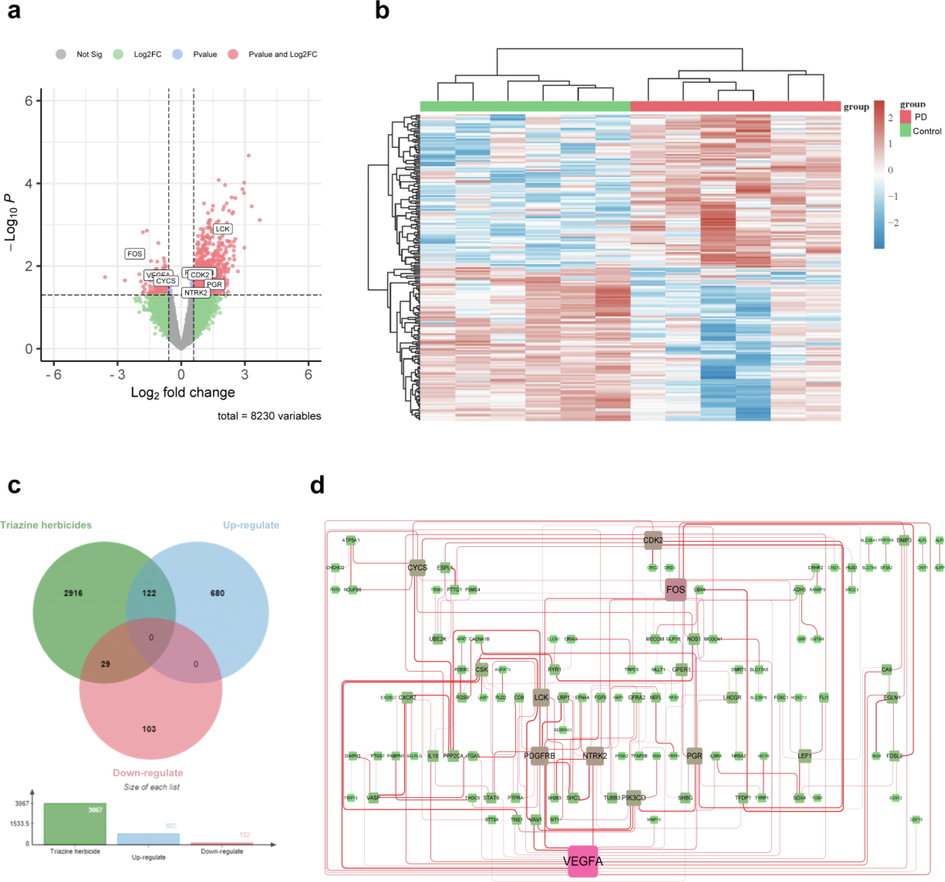
DEG screening and network pharmacological analysis. (a) Volcano plot of DEGs between PD patients and control. (b) Heatmap of DEGs between PD patients and control. (c) The Venn map of triazine herbicide-related targets and PD-related targets. (d) The PPI network. The width of each edge is proportional to the combined score, whereas the color (from green to pink) and size of each node are proportional to interaction strength.
3.2 GO and KEGG enrichment analysis
To investigate the potential mechanisms underlying triazine herbicide-induced PD, we conducted GO and KEGG pathway enrichment analyses. The overlapping targets were enriched in the BP terms “transmembrane receptor protein tyrosine kinase signaling pathway”, “response to hormone”, and “regulation of sequestering of calcium ion”; the CC terms “transcription regulator complex”, “asymmetric synapse”, and “neuron to neuron synapse”; and the MF terms “DNA-binding transcription activator activity”, “hormone binding”, and “phosphatase activity” (Fig. 2a–c). The KEGG pathway analysis revealed that the common targets were enriched in the terms “Calcium signaling pathway”, “Ras signaling pathway”, and “Estrogen signaling pathway” (Fig. 2d).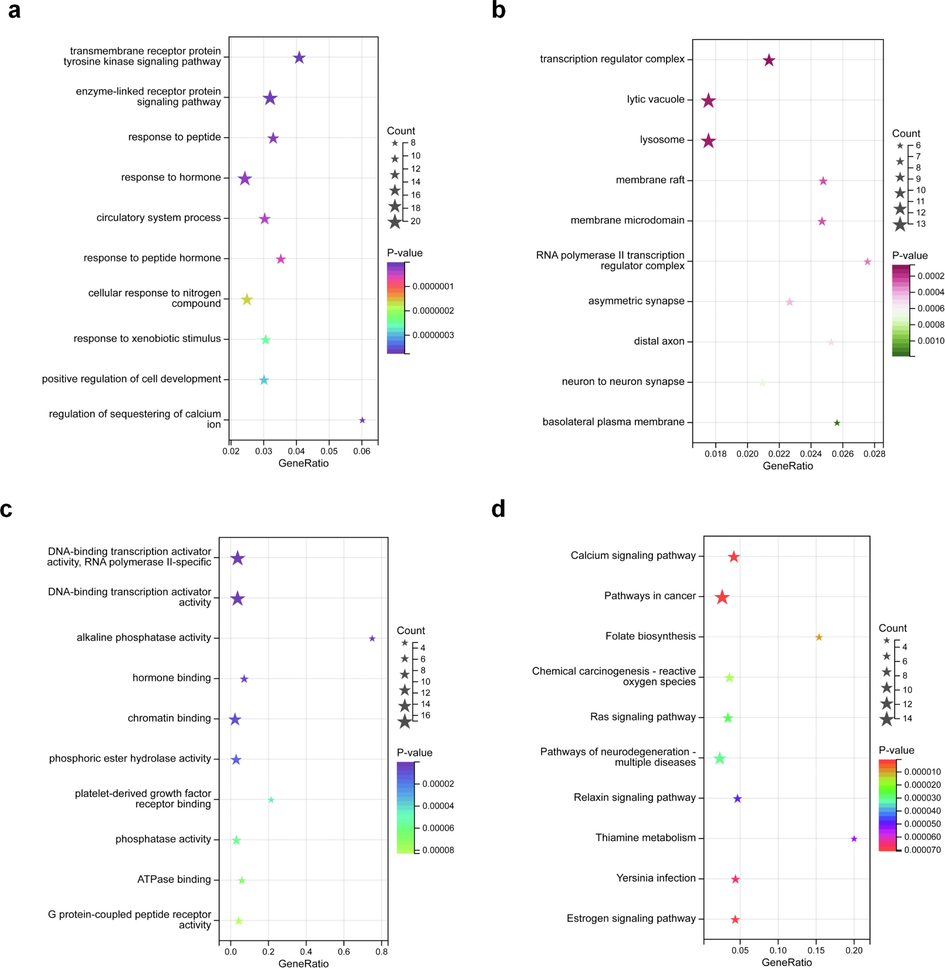
GO and KEGG enrichment analysis. (a) BP enrichment analysis, (b) CC enrichment analysis, (c) MF enrichment analysis, and (d) KEGG pathway enrichment analysis.
3.3 Hub target identification
The top 10 targets were calculated by the closeness, degree, EPC, MCC, MNC, and radiality methods (Fig. 3a–f, Table S3). Through Venn analysis, cyclin-dependent kinase 2 (CDK2), cytochrome c (CYCS), Fos proto-oncogene (FOS), Src family tyrosine kinase (LCK), BDNF/NT-3 growth factors receptor (NTRK2), platelet-derived growth factor receptor beta (PDGFRB), progesterone receptor (PGR), and vascular endothelial growth factor A (VEGFA) were identified as the top eight hub targets (Fig. 3g), and PPI networks of CDK2, CYCS, FOS, LCK, NTRK2, PDGFRB, PGR, and VEGFA were constructed through the STRING database (Fig. 3h).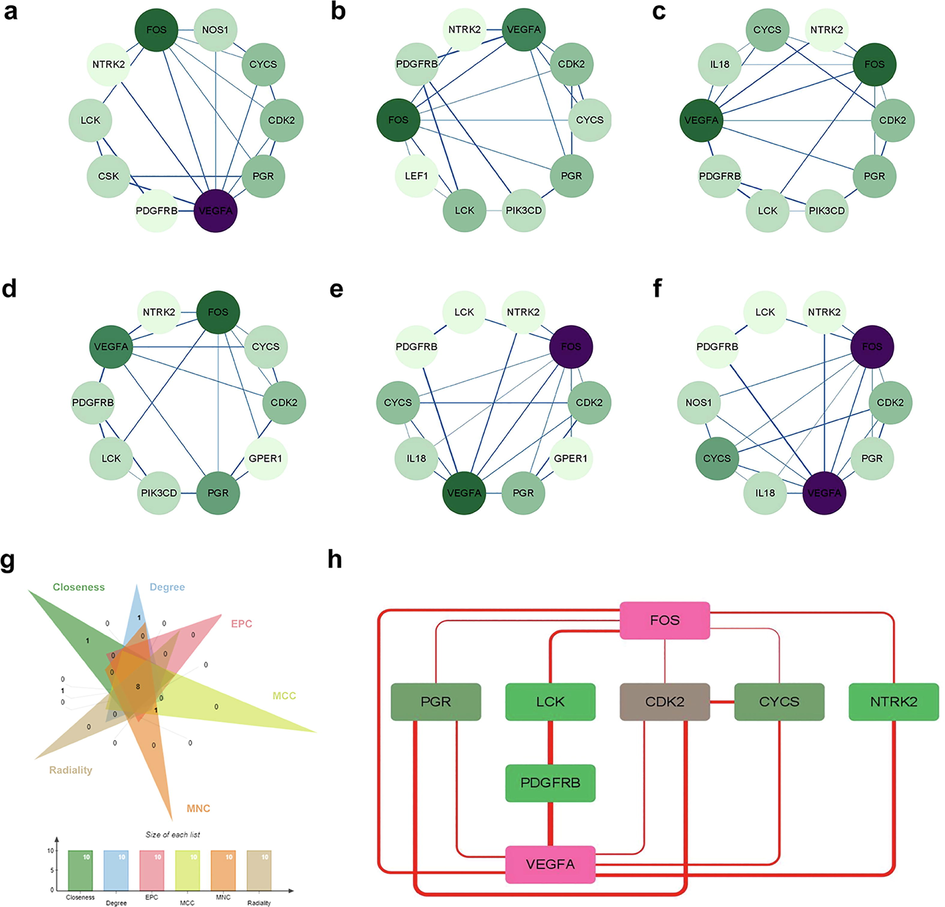
Hub target identification. Top 10 targets calculated through (a) the closeness method, (b) the degree method, (c) the EPC method, (d) the MCC method, (e) the MNC method, and (f) the radiality method. The width of each edge is proportional to the combined score, whereas the color (light green to purple). (g) A Venn map of the top eight core targets. (h) The PPI network of the eight targets.
3.4 Molecular docking
To further investigate the ability of ATR and SIM to bind to the top eight core targets, we performed a molecular docking analysis. The binding energy between ATR and CDK2 was −7.38 kcal/mol (Fig. 4a), between ATR and CYCS it was −7.68 kcal/mol (Fig. 4b), between ATR and PGR it was −7.42 kcal/mol (Fig. 4c), between SIM and CDK2 it was −7.13 kcal/mol (Fig. 4d), between SIM and CYCS it was −7.56 kcal/mol (Fig. 4e), and between SIM and PGR it was −7.26 kcal/mol (Fig. 4f). The binding energies of the remaining targets to both ATR and SIM were greater than −7 (Table S4).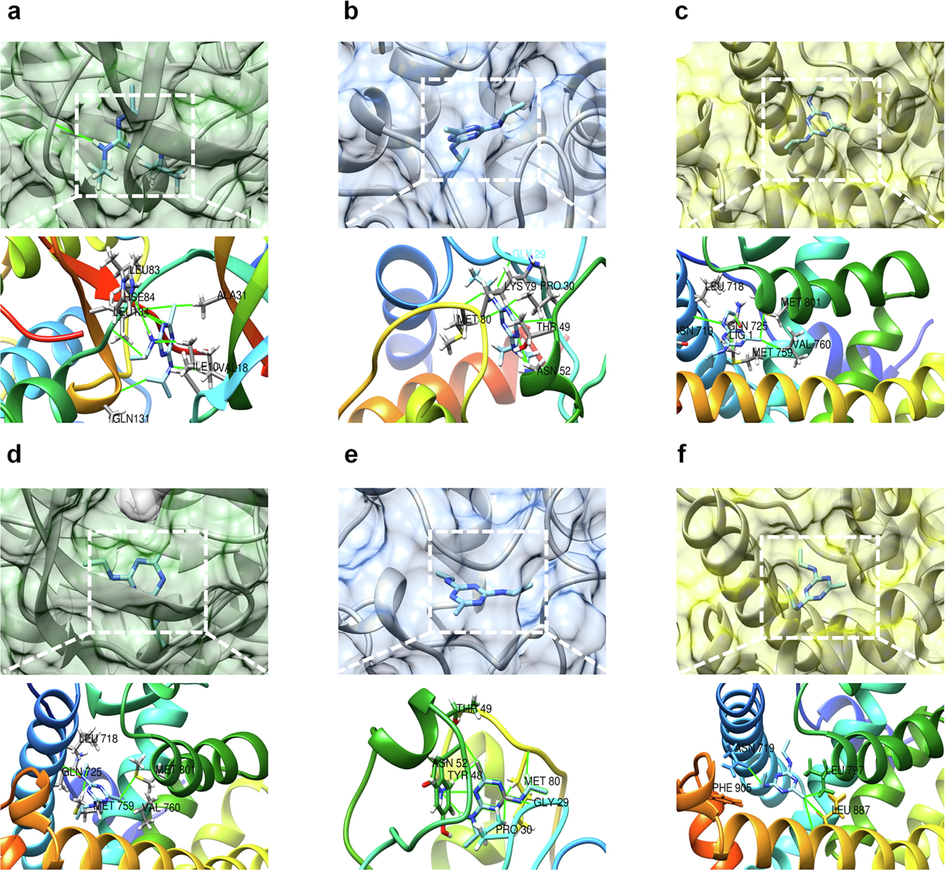
Molecular docking. (a) ATR-CDK2, (b) ATR-CYCS, (c) ATR-PGR, (d) SIM-CDK2, (e) SIM-CYCS, and (f) SIM-PGR.
3.5 Cell viability assay and qPCR
To investigate the dopaminergic neurotoxicity effect of ATR and SIM alone or in combination, the SH-SY5Y cells were treated by various concentrations of ATR and SIM for 72 h, followed by a CCK-8 assay. We found that both ATR (Fig. 5 a-b, ANOVA: F (5,30) = 1.29, P < 0.01) and SIM (Fig. 5 c-d, ANOVA: F (5,30) = 0.36, P < 0.01) decreased the cell viability in a dose-dependent manner. The IC50s for ATR and SIM in SH-SY5Y cells were 396.2 μM and 69.1 μM respectively. The qPCR analysis demonstrated that exposure to SIM (35 μM, 1/2 IC50) alone or in combination with ATR (200 μM, 1/2 IC50) for 72 h significantly increased CDK2 mRNA expression (Fig. 5 e, ANOVA: F (3,20) = 4.517, P < 0.05, SIM vs. control: P < 0.05, mixture vs. control: P < 0.01). ATR alone or combined with SIM for 72 h significantly elevated PGR mRNA levels (Fig. 5 f, ANOVA: F (3,20) = 6.293, P < 0.01, ATR vs. control: P < 0.05, mixture vs. control: P < 0.01). Conversely, SIM exposure alone or in combination with ATR for 72 h significantly reduced CYCS mRNA expression (Fig. 5 g, ANOVA: F (3,20) = 5.342, P < 0.01, SIM vs. control: P < 0.05, mixture vs. control: P < 0.01).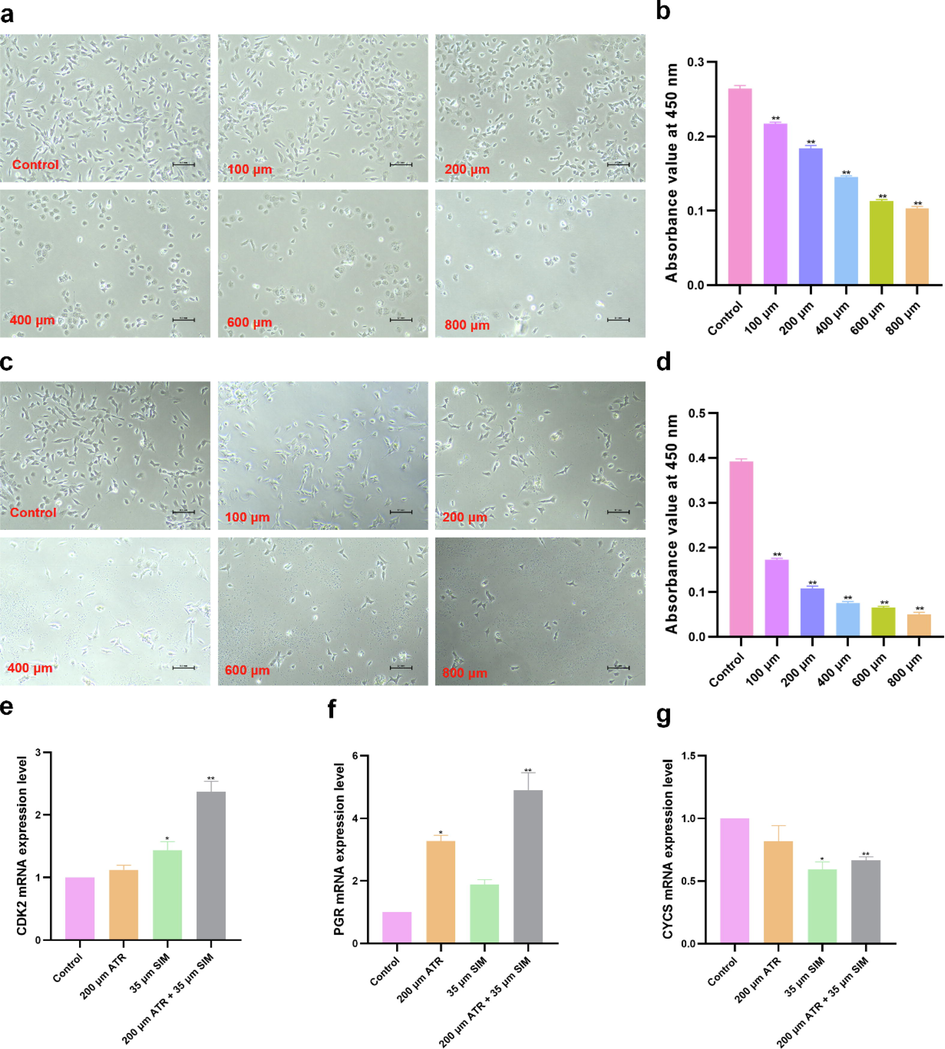
Cell viability assay and qPCR. (a) SH-SY5Y cellular model exposure to ATR, (b) Effects of ATR on the cytotoxicity of SH-SY5Y cells, (c) SH-SY5Y cellular model exposure to SIM, (d) Effects of SIM on the cytotoxicity of SH-SY5Y cells, (e) CDK2 mRNA expression level, (f) PGR mRNA expression level, and (g) CYCS mRNA expression level. Expression levels were standardized to GAPDH. Each column represents the mean ± S.E.M, *P < 0.05 vs. control, **P < 0.01 vs. control.
4 Discussion
Although the majority of PD cases are believed to occur randomly, environmental factors may have a significant impact on the onset of the disease (Xia et al., 2017). According to epidemiological studies (Fernandez-Abascal et al., 2018), there may be a relationship between pesticide exposure and PD. Some pesticides, such as rotenone, have even been used to induce PD in animal models (Zhang et al., 2019). In the present study, we preliminarily explored the molecular biological mechanism of PD induced by combining ATR and SIM exposure through bioinformatics algorithms. We first screened aberrantly expressed mRNAs between PD patients and control by mining GSE20333 from GEO datasets, and acquired 934 DEGs (802 up-regulated and 132 down-regulated). Subsequently, a potential 151 ATR and SIM targets (122 up-regulated and 29 down-regulated) for the inducement of PD were predicted using network pharmacology. Additionally, GO and KEGG analyses were performed to investigate the biological characteristics of the potential targets, and eight hub targets were further screened for validation using molecular docking. Finally, we constructed ATR and SIM alone or in combination exposure cellular models to validate our screened results.
KEGG analysis showed the significant enrichment of DEGs in the calcium signaling and Ras signaling pathways. A previous study used spatial transcriptome profiling of the hippocampal single cell microzone and found that the calcium signaling pathway plays a key role in the PD mouse model (Jia et al., 2023). It also has been reported that neuromelanin loss in PD patients can result in increased calcium signaling due to the loss of a crucial calcium storage site, and ultimately leads to the production of reactive oxygen species (ROS) and cell death within the substantia nigra (Knörle, 2018). ATR is reported to have the ability to activate the calcium signaling pathway and enhance the rate of androgenesis caused by hCG in the Leydig cells of rats (Pogrmic-Majkic et al., 2016). Meanwhile, SIM exposure could affect cytoplasmic Ca2+ homeostasis, thereby increasing ROS levels and interfering with the ability of mouse oocytes to mature (Shang et al., 2021). Therefore, we proposed that the development of PD may be attributed largely to the impairment of the calcium signaling pathway resulting from exposure to triazine herbicides.
The Ras signaling pathway is the first well-established MAPK pathway that plays critical roles in cellular differentiation, development, survival, and proliferation (Zhao and Luo, 2022). Abnormalities in the Ras-MAPK signaling pathway are critical in the process of dopaminergic neuron loss in PD (Nataraj et al., 2017). Perlecan-conjugated laminin 511/521-E8 fragment treatment enhanced the maturation and neurite extension of the grafted dopamine progenitors by activating Ras signaling, which could be an efficient cell replacement therapy for PD (Adachi et al., 2022). It has been reported that ATR could promote the development of mammary tumors in human c-Ha-Ras proto-oncogene transgenic rats (Fukamachi et al., 2004). Moreover, our previous study suggested that ATR induced hippocampal neurotoxicity by affecting the Ras-MAPK signaling pathway in rats (Li et al., 2019). To our knowledge, no studies have shown that SIM exposure can affect the Ras signaling pathway, but given the similarity in chemical structure, we hypothesized that combined ATR and SIM exposure may contribute to PD pathogenesis through the Ras signaling pathway.
The top 10 hub targets were calculated by using six methods in cytoHubba and screening eight intersecting hub targets. Then, through molecular docking, we finally predicted three targets with a high binding potential to ATR and SIM: CDK2, CYCS, and PGR. CDK2 is mainly known for its pivotal role in cell cycle progression, including DNA synthesis, G1/S phase transition, and G2 progression modulation (Zhang et al., 2022). Within the mitochondria, the hemeprotein CYCS serves as a key player in both cellular respiration and apoptosis (Kalpage et al., 2019). Studies confirm that apoptosis due to abnormal expression of CDK2 and CYCS is the main cause of dopaminergic neuron deficiency in PD (Yu et al., 2005; Wang et al., 2007). ATR has been reported to have the ability to halt the cell cycle of neural stem cells at the G1 phase and hinder the transition from the G1 to the S phase by reducing the expression levels of CDK2 genes (Shan et al., 2021). Meanwhile, transcriptome sequencing analysis demonstrated that exposure to SIM induced reproductive toxicity due to a significant decrease in the expression of genes related to the cell cycle in Xenopus laevis (Sai et al., 2015). Notably, there is no evidence that ATR or SIM exposure can lead to abnormal expression or release of CYCS. Our molecular docking results showed that ATR and SIM bind strongly to CDK2 and CYCS, and we also found that exposure to ATR and SIM alone or in combination can significantly affect the mRNA expression of CDK2 and CYCS in SH-SY5Y cells. Our results suggests that CDK2 and CYCS are the key targets of Parkinson's disease induced by combined exposure to ATR and SIM.
PGR is a transcription factor belonging to the nuclear receptor superfamily, which plays a vital role in the intracellular signaling of progesterone and is involved in a multitude of cell biological activities (Lee et al., 2022). It has been reported that PGR plays a neuroprotective and regenerative role in PD (Zhao et al., 2020). Moreover, the significance of PGR expression during the developmental stage of the mesocortical dopamine pathway is crucial for intricate cognitive behavior in adulthood (Willing and Wagner, 2016). ATR and SIM have been reported to cause estrogen-like effects, which could lead to decreased PGR binding capacity in the cytoplasm (Connor et al., 1996). In addition, evidence suggested that ATR can affect the mRNA expression level of PGR and enhance the occurrence of androgenesis induced by hCG in Leydig cells of rats (Fa et al., 2013). Integrating the molecular docking and qPCR results, we hypothesized that combined ATR and SIM exposure may induce PD pathogenesis by interfering with the expression or function of PGR.
5 Limitations
The study has some limitations currently. First, the network pharmacology analysis relies on the timeliness of the database, and the update of the data could make the study more accurate. Secondly, our study only explored the molecular mechanisms of combined ATR and SIM exposure leading to PD, while other triazine herbicides, such as propazine, remain in the environment, so more complex combined exposures need to be taken into account. Finally, the present study was based on a bioinformatics and in vitro strategies to investigate the mechanism of PD caused by combined ATR and SIM exposure, and our results need to be confirmed by further in vivo studies.
6 Conclusion
Our study used several bioinformatics methods to explore the molecular mechanisms underlying PD induced by simultaneous exposure to ATR and SIM. Our analysis identified the calcium and Ras signaling pathways as potentially significant, with CDK2, CYCS, and PGR emerging as key targets. Our study represents an initial exploration of the potential link between triazine herbicides and PD, producing results that provide valuable insights for future research. Further in vivo and in vitro investigations are warranted to confirm our findings.
CRediT authorship contribution statement
Jianan Li: Funding acquisition, Conceptualization, Investigation, Methodology, Project administration and Roles/Writing - original draft. Haoran Bi: Data curation, Formal analysis, Resources; Software, Supervision, Validation, Visualization and Writing - review & editing.
Funding
This study was supported by the China Postdoctoral Science Foundation (2022M712679) and the National Natural Science Foundation of China (82203994).
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Pretreatment with Perlecan-Conjugated Laminin-E8 Fragment Enhances Maturation of Grafted Dopaminergic Progenitors in Parkinson's Disease Model. Stem Cells Transl. Med.. 2022;11:767-777.
- [Google Scholar]
- QSAR analysis of immune recognition for triazine herbicides based on immunoassay data for polyclonal and monoclonal antibodies. PLoS One. 2019;14:e0214879.
- [Google Scholar]
- Astrocytic modulation of blood brain barrier: perspectives on Parkinson's disease. Front. Cell. Neurosci.. 2014;8:211.
- [Google Scholar]
- Failure of chloro-S-triazine-derived compounds to induce estrogen receptor-mediated responses in vivo and in vitro. Fund. Appl. Toxicol.: Off. J. Soc. Toxicol.. 1996;30:93-101.
- [Google Scholar]
- Modeling nigrostriatal degeneration in organotypic cultures, a new ex vivo model of Parkinson's disease. Neuroscience. 2014;256:10-22.
- [Google Scholar]
- Involvement of ERK1/2 signaling pathway in atrazine action on FSH-stimulated LHR and CYP19A1 expression in rat granulosa cells. Toxicol. Appl. Pharmacol.. 2013;270:1-8.
- [Google Scholar]
- β-Naphtoflavone and Ethanol Induce Cytochrome P450 and Protect towards MPP+ Toxicity in Human Neuroblastoma SH-SY5Y Cells. Int. J. Mol. Sci.. 2018;19
- [Google Scholar]
- Possible enhancing effects of atrazine and nonylphenol on 7,12-dimethylbenz[a]anthracene-induced mammary tumor development in human c-Ha-ras proto-oncogene transgenic rats. Cancer Sci.. 2004;95:404-410.
- [Google Scholar]
- The A9 dopamine neuron component in grafts of ventral mesencephalon is an important determinant for recovery of motor function in a rat model of Parkinson's disease. Brain. 2010;133:482-495.
- [Google Scholar]
- Alteration of dopamine uptake into rat striatal vesicles and synaptosomes caused by an in vitro exposure to atrazine and some of its metabolites. Toxicology. 2008;248:52-58.
- [Google Scholar]
- Spatial Transcriptome Profiling of Mouse Hippocampal Single Cell Microzone in Parkinson's Disease. Int. J. Mol. Sci.. 2023;24
- [Google Scholar]
- Effect of rTMS on Parkinson's cognitive function: a systematic review and meta-analysis. BMC Neurol.. 2020;20:377.
- [Google Scholar]
- Serine-47 phosphorylation of cytochrome c in the mammalian brain regulates cytochrome c oxidase and caspase-3 activity. FASEB J.. 2019;33:13503-13514.
- [Google Scholar]
- S-nitrosylated PARIS Leads to the Sequestration of PGC-1α into Insoluble Deposits in Parkinson's Disease Model. Cells.. 2022;11
- [Google Scholar]
- Neuromelanin in Parkinson's Disease: from Fenton Reaction to Calcium Signaling. Neurotox. Res.. 2018;33:515-522.
- [Google Scholar]
- Activation function 1 of progesterone receptor is required for progesterone antagonism of oestrogen action in the uterus. BMC Biol.. 2022;20:222.
- [Google Scholar]
- Integrating network pharmacology and in vitro model to investigate hippocampal neurotoxicity induced by atrazine. Toxicol. Mech. Methods 2021:1-9.
- [Google Scholar]
- The MEK/ERK/CREB signaling pathway is involved in atrazine induced hippocampal neurotoxicity in Sprague Dawley rats. Ecotoxicol. Environ. Saf.. 2019;170:673-681.
- [Google Scholar]
- Dopaminergic Dysfunction in Mammalian Dopamine Neurons Induced by Simazine Neurotoxicity. Int. J. Mol. Sci.. 2017;18
- [Google Scholar]
- Neurotrophic Effect of Asiatic acid, a Triterpene of Centella asiatica Against Chronic 1-Methyl 4-Phenyl 1, 2, 3, 6-Tetrahydropyridine Hydrochloride/Probenecid Mouse Model of Parkinson's disease: The Role of MAPK, PI3K-Akt-GSK3β and mTOR Signalling Pathways. Neurochem. Res.. 2017;42:1354-1365.
- [Google Scholar]
- Atrazine activates multiple signaling pathways enhancing the rapid hCG-induced androgenesis in rat Leydig cells. Toxicology. 2016;368–369:37-45.
- [Google Scholar]
- The Effects of Simazine, a Chlorotriazine Herbicide, on the Expression of Genes in Developing Male Xenopus laevis. Bull. Environ. Contam. Toxicol.. 2015;95:157-163.
- [Google Scholar]
- Evaluation of atrazine neurodevelopment toxicity in vitro-application of hESC-based neural differentiation model. Reprod. Toxicol.. 2021;103:149-158.
- [Google Scholar]
- Simazine perturbs the maturational competency of mouse oocyte through inducing oxidative stress and DNA damage. Ecotoxicol. Environ. Saf.. 2021;230:113105
- [Google Scholar]
- The effects of gestational and chronic atrazine exposure on motor behaviors and striatal dopamine in male Sprague-Dawley rats. Toxicol. Appl. Pharmacol.. 2015;289:185-192.
- [Google Scholar]
- PINK1 mutants associated with recessive Parkinson's disease are defective in inhibiting mitochondrial release of cytochrome c. Neurobiol. Dis.. 2007;28:216-226.
- [Google Scholar]
- Progesterone Receptor Expression in the Developing Mesocortical Dopamine Pathway: Importance for Complex Cognitive Behavior in Adulthood. Neuroendocrinology. 2016;103:207-222.
- [Google Scholar]
- Zebrafish slc30a10 deficiency revealed a novel compensatory mechanism of Atp2c1 in maintaining manganese homeostasis. PLoS Genet.. 2017;13:e1006892.
- [Google Scholar]
- Modeling microbial communities from atrazine contaminated soils promotes the development of biostimulation solutions. ISME J.. 2019;13:494-508.
- [Google Scholar]
- DNA damage induces cdk2 protein levels and histone H2B phosphorylation in SH-SY5Y neuroblastoma cells. J. Alzheimer's Dis.: JAD. 2005;8:7-21.
- [Google Scholar]
- Inhibition of the CDK2 and Cyclin A complex leads to autophagic degradation of CDK2 in cancer cells. Nat. Commun.. 2022;13:2835.
- [Google Scholar]
- Induced pluripotent stem cells in rat models of Parkinson's disease: A systematic review and meta-analysis. Biomed Rep.. 2018;8:289-296.
- [Google Scholar]
- Involvement of Akt/mTOR in the Neurotoxicity of Rotenone-Induced Parkinson's Disease Models. Int. J. Environ. Res. Public Health. 2019;16
- [Google Scholar]
- Discovery of Raf Family Is a Milestone in Deciphering the Ras-Mediated Intracellular Signaling Pathway. Int. J. Mol. Sci.. 2022;23
- [Google Scholar]
- Botanical Drug Puerarin Promotes Neuronal Survival and Neurite Outgrowth against MPTP/MPP(+)-Induced Toxicity via Progesterone Receptor Signaling. Oxid. Med. Cell. Longev.. 2020;2020:7635291.
- [Google Scholar]
Appendix A
Supplementary data
Supplementary data to this article can be found online at https://doi.org/10.1016/j.arabjc.2024.105657.
Appendix A
Supplementary data
The following are the Supplementary data to this article:Supplementary data 1
Supplementary data 1







