Translate this page into:
Magnetoplasmonic core–shell structured Ag@Fe3O4 particles synthesized via polyol reduction process rendering dual-functionality for bacteria ablation and dyes degradation
⁎Corresponding authors. liangtian@hbust.edu.cn (Tian Liang), taochen518@163.com (Tao Chen)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Abstract
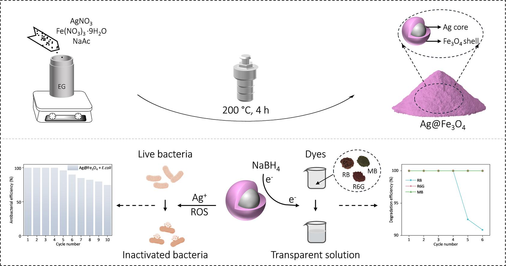
The Ag nanoparticles demonstrate potent bacteria eradication capabilities; however, their tendency to aggregate in aqueous solutions compromises the antibacterial efficacy. Furthermore, the Ag nanoparticles employed in sewage treatment are challenging to recycle, resulting in environmental pollution and resource wastage. Herein, the Ag-core Fe3O4-shell structured particles (Ag@Fe3O4) are synthesized by leveraging the reduction potential difference between Ag+/Ag0 and Fe3+/Fe2+ through a one-step polyol reduction process. The Fe3O4 shell in the Ag@Fe3O4 composite not only effectively inhibits the agglomeration of Ag, but also enhances the penetration capability of the composite into biofilms, thereby enabling Ag@Fe3O4 to possess remarkable antibacterial efficacy against Escherichia coli (E. coli). The Ag@Fe3O4 demonstrates nearly 100 % inhibition of E. coli at a concentration of 0.24 mg mL−1 (with an Ag content of 0.042 mg mL−1) while still maintaining antibacterial effectiveness of 74.6 % even after undergoing reutilization for 10 cycles. Meanwhile, due to the excellent electron conductivity of Ag and the effective adsorption capability of Fe3O4 shell towards organic dyes, Ag@Fe3O4 facilitates rapid electron transfer to organic dyes and further lead to their reduction and degradation in the presence of NaBH4. The Ag@Fe3O4 can catalytically degrade various organic dyes (including Rhodamine B, Rhodamine 6G, and Methylene blue) within only 15 min, while achieving an impressive degradation efficiency exceeding 90.9 % after 6 cycles of reutilization. The cost-effectiveness (approximately $0.17 per gram), facile magnetic recovery, along with the superior antibacterial and dye-degradation performance showcase the significant potential of Ag@Fe3O4 for medical applications and sewage treatment.
Keywords
Magnetoplasmonic
Core shell structure
Ag
Fe3O4
Antibacterial
Catalytic degradation
Organic dyes
1 Introduction
The overuse of antibiotics in recent years has accelerated the evolution of bacteria, resulting in the emergence of antibiotic-resistant “superbugs” that pose a significant threat to human health (Jia et al., 2017; Salam et al., 2023). The development of highly efficient and environmentally friendly new antibacterial materials is therefore urgently needed. The remarkable antibacterial properties of metal ions have attracted significant attention, with silver (Ag) standing out for its exceptional antibacterial activity compared to other metals (Ag > Hg > Cu > Cd > Cr > Ni > Pb > Co > Zn > Fe > Ca) (Berger et al., 1976; Fouladi-Fard et al., 2022; Razavi et al., 2021). However, conventional methods of synthesizing silver nanoparticles often result in agglomeration, leading to a significant decrease in their antibacterial efficacy (Ye et al., 2019; Zeng et al., 2018; Zhao et al., 2017).
To address the issue of agglomeration of silver nanoparticles, numerous investigations have been undertaken. Gankhuyag et al. synthesized silica-supported silver nanoparticles (SiO2@Ag) by impregnating silica particles (100–130 nm) in an AgNO3 solution using trisodium citrate as a reducing agent, which exhibited remarkable antibacterial activity against E. coli (Gankhuyag et al., 2021). Ghosh et al. developed the TiO2-Ag nanocomposite through mechanical alloying of TiO2 and Ag powder under an Ar atmosphere, resulting in significantly enhanced photocatalytic and antibacterial activities (Ghosh et al., 2020). Cinteza et al. employed an electrochemical method with a sacrificial anode to synthesize the Ag nanoparticles, further utilizing chitosan as a capping agent to enhance stabilization and biocompatibility while preserving antibacterial activity (Cinteza et al., 2018). Yang et al. fabricated the C-Zn/Ag nanocomposite by introducing Ag nanoparticles into a MOF-derived carbonized framework containing metallic Zn, which can rapidly release Ag+ ions to inactivate bacterial intracellular proteins (Yang et al., 2020).
Although much progress has been made, one challenge lies in the difficulty of recycling the nanoparticles during utilization, which could potentially result in environmental pollution and resource wastage (Mohajerani et al., 2019; Chaukura et al., 2016; Islam et al., 2021). To achieve the objectives of environmental conservation and sustainable development, we synthesized the Ag-core Fe3O4-shell structured particles (Ag@Fe3O4) by leveraging the reduction potential difference between Ag+/Ag0 and Fe3+/Fe2+ through a one-step polyol reduction process. The magnetoplasmonic core–shell Ag@Fe3O4 nanoparticles, which can be easily synthesized, not only prevent the agglomeration of Ag but also facilitate their effortless recovery. The Ag@Fe3O4 nanoparticles demonstrate exceptional antibacterial performance due to the well-dispersed Ag and sustained release of Ag+ through the porous Fe3O4 shell. At a concentration of 0.24 mg mL−1 (with an Ag content of 0.042 mg mL−1), the Ag@Fe3O4 exhibit nearly 100 % inhibition against E. coli, while maintaining a significant antibacterial efficacy of 74.6 % even after undergoing reutilization for 10 cycles. In addition to the antibacterial properties, the Ag@Fe3O4 nanoparticles also demonstrate exceptional catalytic degradation capabilities in the presence of NaBH4 towards organic dyes owing to their intricate characteristics and delicate nanostructure. The organic dye molecules are adsorbed onto the active sites on the Fe3O4 shell (possibly through electrostatic attraction and van der Waals force) while the electron transfer mediator of Ag facilitates efficient electron transfer from the nucleophilic NaBH4 to the electrophilic dye molecules, ultimately inducing reduction and degradation of the dye molecules (Amir et al., 2015; Zhang et al., 2017; Shi et al., 2021). The Ag@Fe3O4 can catalytically degrade various organic dyes (including Rhodamine B, Rhodamine 6G, and Methylene blue) within just 15 min, and also achieve a degradation efficiency of over 90.9 % after undergoing 6 cycles of reutilization.
Overall, the cost-effectiveness (approximately $0.17 per gram), facile magnetic recovery, antibacterial property, and catalytic degradation towards organic dyes render Ag@Fe3O4 particles a promising candidate for large-scale applications in antibacterial treatments and waste water treatment.
2 Materials and methods
2.1 Chemical reagents
Fe(NO3)3·9H2O was purchased from Shanghai Yien Chemical Technology Co. Ltd. AgNO3, ethylene glycol, sodium acetate (NaAc), oxalic acid, NaBH4, and Methylene blue (MB) were obtained from Sinopharm Chemical Reagent Co. Ltd., China. Rhodamine B (RB) and Rhodamine 6G (R6G) were purchased from Shanghai McLin Biochemical Technology Co., Ltd. Beef infusion powder, tryptone, yeast powder, and AGAR powder were sourced from Beijing Lan Ji Ke Technology Co., LTD. Escherichia coli (E. coli) strain was acquired from Hai Bo Biotechnology Limited.
2.2 Synthesis of materials
2.2.1 Synthesis of Ag@Fe3O4
The core–shell structured Ag@Fe3O4 particles were synthesized via a one-step polyol reduction process (Dong et al., 2019; X.h. Meng, X. Shao, H.y. Li, F.z. Liu, X.p. Pu, W.z. Li, C.h. Su, One-step hydrothermal synthesis, characterization and visible-light catalytic property of Ag-reduced graphene oxide composite, Materials Research Bulletin 48(4), 2013; Hou et al., 2017; Jiao et al., 2018; Liang et al., 2023). Specifically, Fe(NO3)3·9H2O (1.212 g), sodium acetate (2.153 g), and AgNO3 (0.076 g) were sequentially added to ethylene glycol (30 ml) and stirred for 30 min. The resulting mixture was then transferred into a Teflon-lined stainless-steel autoclave and heated at 200 °C for 4 h. Finally, the product was washed three times with deionized water, subjected to centrifugation, and dried at 60 °C overnight to obtain the Ag@Fe3O4 particles.
2.2.2 Synthesis of solid Ag cores (c-Ag)
The Ag@Fe3O4 was immersed in a 1 mol L−1 oxalic acid solution overnight, resulting in the formation of ferric oxalate and a yellow-green solution due to the reaction between Fe3O4 and oxalic acid, thus obtaining the c-Ag (Shrivastava et al., 2022; Yu et al., 2018; Zhang et al., 2014).
2.2.3 Synthesis of hollow Fe3O4 shells (s-Fe3O4)
The Ag@Fe3O4 was immersed in a 0.1 mol L−1 Fe(NO3)3 solution and subjected to ultrasonication for 30 min. Afterwards, it was rinsed with deionized water three times and finally dried, resulting in the formation of s-Fe3O4 (Chen et al., 2014; Lu et al., 2007; Zhang et al., 2018).
2.3 Materials characterization
The morphology and microstructure of the samples were observed using a Field Emission Scanning Electron Microscope (FE-SEM, SU8010, Hitachi, Japan) and a Transmission Electron Microscope (TEM, FEI Tecnai G2 F20 S-TWIN, FEI, USA). The Atomic Absorption Spectrometer (AAS, PinAAcle 900F, Perkin Elmer, USA) is utilized for the determination of the elemental composition of materials. The Dynamic Light Scattering (DLS, Zetasizer Nano ZS90, Malvern, UK) is employed for the analysis of particle size distribution and polydispersity index (PDI) of the Ag@Fe3O4 particles. The crystal structure of the samples was analyzed using D8-FOCUS powder X-ray diffraction (XRD, D8-FOCUS, Bruker, Germany) with Cu Kα target (λ = 0.154 nm) at a voltage of 40 KV, a current of 35 mA, and a scanning speed of 10° min−1. The absorption peaks of the samples were determined using an Ultraviolet–Visible Spectrophotometer (UV, 1901 PC, Shanghai AUCY, China) with a scanning range from 200 to 800 nm. The Brunauer-Emmett-Teller (BET, ASAP 2460, Micromeritics, USA) measurements were utilized to measure the specific surface area of materials, while the pore size distribution of the materials was determined utilizing the Barrett-Joyner-Halenda (BJH) model. The magnetization curves of the samples were measured using a Vibrating Sample Magnetometer (VSM, 7404, Lake Shore, USA). Fourier Transform Infrared Spectroscopy (FT-IR, Spectrum two, Perkin Elmer, USA) was employed to analyze the composition and structural information of the materials. The X-ray Photoelectron Spectrometer (XPS, K-Alpha, thermo scientific, USA) is utilized for the validation of the electronic state and chemical composition of the materials. The Electron Spin Resonance (ESR, EMXplus-6/1, Bruker, Germany) is used to qualitatively and quantitatively detect the unpaired electrons present in atoms or molecules of materials.
2.4 Antibacterial experiments
This study assesses the antibacterial efficacy of the samples using E. coli as the test bacteria. Liquid medium preparation: a solution containing peptone (2 g), beef extract (0.6 g), and NaCl (1 g) dissolved in distilled water (200 ml) was sterilized at 121 °C for 30 min. Solid medium preparation: a mixture containing peptone (2 g), yeast extract (1 g), NaCl (2 g), and agar (3 g) dissolved in distilled water (200 ml) was sterilized at 121 °C for 30 min. E. coli activation: E. coli liquid (1 mL) was inoculated into liquid medium (200 mL) and incubated overnight at 37 °C in a constant temperature oscillator (Shakoori and Muneer, 2002).
2.4.1 Measurement for the diameter of inhibition zone (DIZ)
The activated E. coli was first diluted to a concentration of 5 × 106 Colony-Forming Units (CFU) per milliliter using physiological saline. Subsequently, 100 μL of the diluted E. coli suspension was evenly spread onto the solid culture medium and placed within an Oxford cup. The c-Ag, s-Fe3O4, and Ag@Fe3O4 suspensions at various concentrations were placed in separate Oxford cups and incubated at 37 °C for 12 h prior to measuring the diameter of the inhibition zone (Aldosari et al., 2023; Xiong et al., 2013).
2.4.2 Measurement of the minimum inhibitory concentration (MIC)
The MIC refers to the minimum antibacterial concentration that can inhibit bacterial growth (Tang and Zheng, 2018; Mei et al., 2014). A negative control group (only broth) and a positive control group (mixture of bacteria and broth) were set up for comparison. The synthesized particles of various weight (1–10 mg), bacterial solution (100 μL with a concentration of 106 colony forming units (CFU) per mL), and liquid broth (25 mL) were mixed thoroughly and cultured in a constant temperature incubator at 37 °C for 10 h. When the concentration of the composite material in the test tube surpasses the MIC, it effectively impedes bacterial growth and reproduction, resulting in a visibly clear solution within the test tube. Conversely, if the concentration of the composite material is below the MIC, it fails to hinder bacterial reproduction and growth, leading to a persistently turbid solution. Therefore, the MIC value of the composite material can be determined by visually observing the turbidity change in the test tube solution before and after incubation (Parvekar et al., 2020).
2.4.3 Cyclic antibacterial ability
The synthesized particles (10 mg) and a fresh bacterial solution (5 mL at a concentration of 106 CFU per mL) were introduced into a sterile reagent bottle and thoroughly mixed, followed by incubation at room temperature for 150 min. The above suspension was subjected to magnetic separation, and the supernatant containing bacteria (100 μL) was extracted. Subsequently, it was diluted 1000-fold with normal saline before being spread onto solid medium and incubated at 37 °C for 24 h. The colony count was subsequently conducted, followed by the calculation of the bacterial inhibition rate. This was denoted as the first cycle of the antibacterial experiment. After the aforementioned magnetic separation process, the residual bacterial solution was discarded, and the unwashed separated particles were mixed with another fresh bacterial solution (5 mL at a concentration of 106 CFU per mL) (Yong et al., 2018). The subsequent steps were then repeated for the next cycle.
The bacterial inhibition rate is calculated as follows (Wang et al., 2020; Shuai et al., 2020): where R (%) is the bacterial inhibition rate, B denotes the colony count in the control experiment without any treatment, and C represents the colony count after adding the Ag@Fe3O4.
2.5 Catalytic degradation experiments
The organic dyes of Rhodamine B (RB), Rhodamine 6G (R6G), and Methylene blue (MB) exhibit responsiveness to UV–visible (UV–vis) light, and the intensity of their absorption peaks demonstrates a positive correlation with concentration according to the Beer-Lambert Law. Therefore, the concentration of the organic dyes can be effectively monitored by employing UV–vis spectrophotometry. The synthesized particles (20 mg) were dispersed in an aqueous solution of organic dyes (RB, R6G, MB; 20 ml, 5 mg L−1) under continuous agitation, followed by the addition of sodium borohydride (1.5 mg). The catalytic degradation activity of the particles was assessed by monitoring changes in dye concentration through measuring variations in the main absorbance peak within the UV–vis spectrum.
The degradation rate (R) is calculated as follows (Sakir and Onses, 2019; Hao et al., 2013): where Co (mg L−1) and Ct (mg L−1) represent the initial and residual concentration of the dyes, respectively. The recyclability of the samples was evaluated by conducting repeated experiments as described above. After each cycle, the samples were separated using a magnet and then subjected to another round of testing. The recyclable catalytic efficiency corresponds to the degradation rate calculated in each cycle.
3 Results and discussion
The synthesis procedure of Ag@Fe3O4 core–shell nanoparticle involves a polyol reduction process by a solvothermal approach (Fig. 1). Specifically, AgNO3 and Fe(NO3)3·9H2O are dissolved in ethylene glycol along with sodium acetate, forming a clear solution. The mixture is then transferred to an autoclave and heated at 200 °C for 4 h to obtain the Ag@Fe3O4 composite. Due to the higher standard hydrogen electrode (SHE) potential of Ag+/Ag0 (
) compared to that of Fe3+/Fe2+ (
), the reduction rate of Ag+ to Ag0 surpasses that of Fe3+ to Fe2+, leading to the initial formation of Ag cores which serve as seeds for the subsequent deposition of Fe3O4 shells (Zhang et al., 2012). The formation of Fe3O4 (magnetite) shells involves a specific redox process where Fe3+ is partially reduced to Fe2+ in the presence of polyol. After the partial reduction of Fe3+ to Fe2+, the Fe2+ and the remaining Fe3+ ions in the solution combine together and result in the precipitation of Fe3O4 as a solid under alkaline conditions. During the reaction process, AgNO3 and Fe(NO3)3·9H2O provide the necessary metal ions for the formation of Ag cores and Fe3O4 shells, respectively. Ethylene glycol plays a pivotal role as a reducing agent, facilitating the reduction of metal ions. Sodium acetate may function as a stabilizing agent that regulates the reaction kinetics. The combination of the aforementioned chemical reagents in the solvothermal reaction ultimately results in the formation of magnetoplasmonic core–shell structured Ag@Fe3O4 particles. Due to their intricate characteristics and delicate nanostructure, the Ag@Fe3O4 nanoparticles possess dual functionality for bacteria ablation and dye degradation.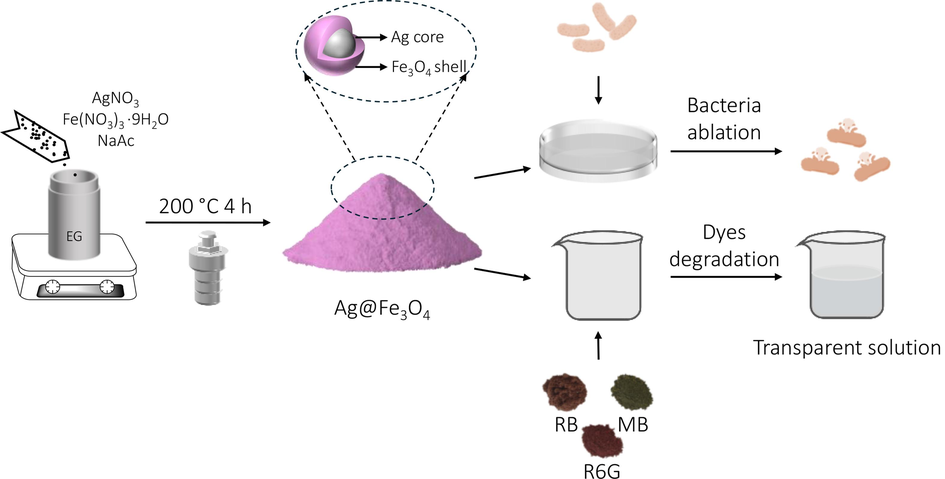
The scheme of synthesizing magnetoplasmonic core–shell structured Ag@Fe3O4 particles and their dual-functionality.
3.1 Materials characterization
3.1.1 SEM and TEM analysis
The scanning electron microscope (SEM) images reveal that the Ag@Fe3O4 particles exhibit a high degree of monodispersity, showcasing nearly spherical morphology with an average diameter of approximately 220 nm (Fig. 2a). Upon closer examination of the magnified SEM image, it becomes evident that these spheres possess a non-smooth surface with the presence of numerous small nanoparticles (Fig. 2b). The observed disparity between the inner core and outer shell in the transmission electron microscope (TEM) images of Ag@Fe3O4 implies the presence of core/shell structures (Fig. 2c, d). In these images, the Ag cores exhibit a dark appearance while the Fe3O4 shells appear lighter due to Ag's higher electron density, resulting in reduced electron transmission (M.E.n.F. Brollo, RománLópez-Ruiz, D. Muraca, S.J.A. Figueroa, K.R. Pirota, M. Knobel, , 2014). The silver cores demonstrate a nearly uniform size of approximately 70 nm, with multiple tiny Fe3O4 NPs comprising the shells that possess an average thickness of around 75 nm. The high-angle annular dark-field (HAADF) image (Fig. 2e) and energy dispersive X-ray spectrometer (EDX) mapping images (Fig. 2f) of Ag@Fe3O4 clearly demonstrate the distribution of Fe, O, and Ag within the Ag@Fe3O4 composite, confirming the successful integration of these elements in the Ag@Fe3O4 as well as the Ag-core Fe3O4-shell structure.
(a, b) SEM, (c, d) TEM, (e) HAADF image, and (f) EDX mapping images of the core–shell structured Ag@Fe3O4 nanoparticles; (g) SEM and (h) TEM images of the hollow s-Fe3O4 particles.
The morphology and microstructure of the control samples, including Fe3O4 shells (s-Fe3O4) and Ag cores (c-Ag), are also examined. Results indicate that s-Fe3O4 exhibit a rough texture and discernible pores with a hollow structure (Fig. 2g, h), while c-Ag possess a spherical morphology (Fig. S1, Supporting Information), and their dimensions are consistent with the shell and core of Ag@Fe3O4, respectively. The weight percentages of Fe3O4 and Ag in the Ag@Fe3O4 are determined through the Atomic Absorption Spectroscopy analysis (Table S1, Supporting Information), which are 79.2 wt% and 20.8 wt%, respectively.
3.1.2 DLS analysis
The DLS technique is employed for the analysis of particle size distribution and polydispersity index (PDI) of the Ag@Fe3O4 particles. The DLS pattern (Fig. 3a) reveals that the average hydrodynamic diameter of Ag@Fe3O4 is 211.3 nm, which aligns with the TEM results. The detected polydispersity index is 0.213, confirming a relatively narrow size distribution of the Ag@Fe3O4 nanoparticles (Gong et al., 2007; Tayyaba et al., 2020).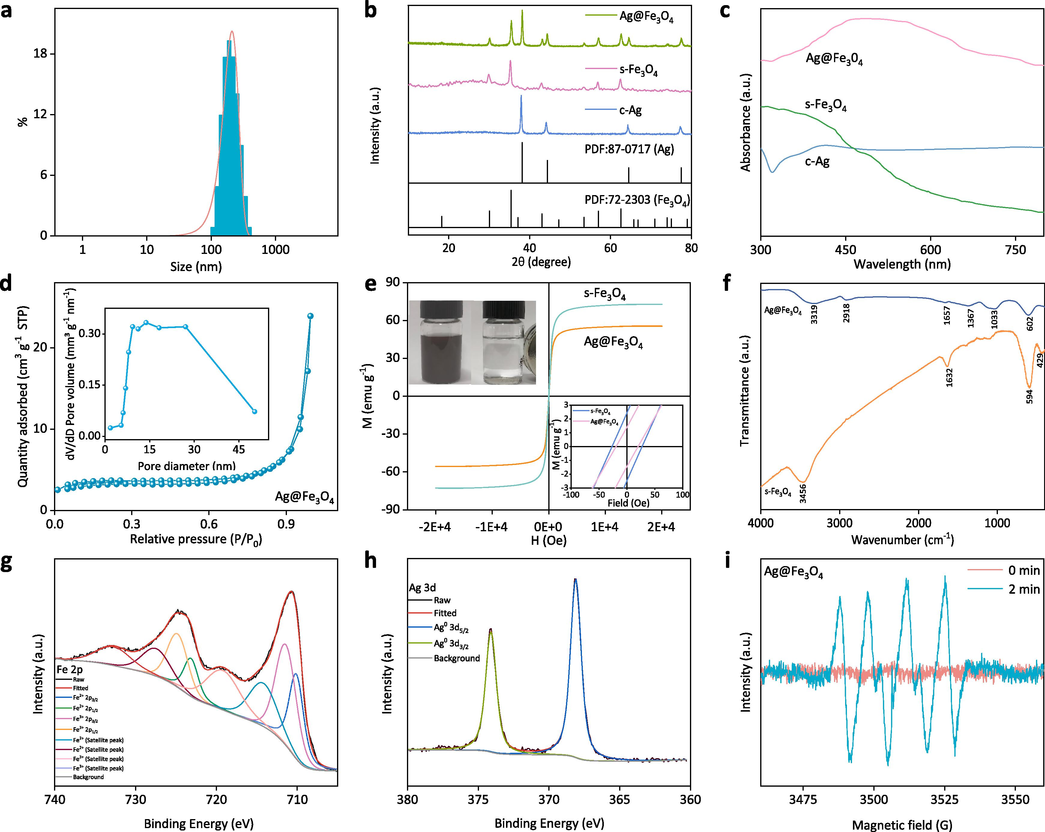
(a) The size distribution of the Ag@Fe3O4 nanoparticles; (b) XRD patterns and (c) UV–vis spectra of the c-Ag, s-Fe3O4, and Ag@Fe3O4; (d) N2 adsorption/desorption isotherm and pore size distribution of the Ag@Fe3O4; (e) magnetic hysteresis loops of s-Fe3O4 and Ag@Fe3O4, the insets are magnetic recovery images of Ag@Fe3O4 and magnified magnetic hysteresis loops of s-Fe3O4 and Ag@Fe3O4; (f) FTIR spectra of the s-Fe3O4 and Ag@Fe3O4; high-resolution XPS spectra of (g) Fe 2p and (h) Ag 3d in Ag@Fe3O4; (i) ESR spectra of Ag@Fe3O4 in aqueous solution at the time of 0 min and 2 min.
3.1.3 XRD analysis
X-ray Diffraction (XRD) patterns provide valuable information on the crystalline structure and composition of materials. The prepared samples all exhibit distinct diffraction peaks, indicating their excellent crystallinity (Fig. 3b). The peaks of c-Ag at 2θ = 38°, 44.2°, 64.3°, and 77.4° correspond to the (111), (200), (220), and (311) crystal planes of Ag, respectively, as identified by the Joint Committee of Powder Diffraction Society PDF card No. 87–0717 (Akhavan, 2009; Jeon et al., 2003; Rajesh, 2012). The peaks of s-Fe3O4 at 2θ = 30.2°, 35.5°, 43.1°, 53.4°, 56.9°, 62.6° and 74.1° can be assigned to the (220), (311), (400), (422), (511), (440) and (533) crystal planes of Fe3O4, respectively, according to the PDF card No. 72–2303 (Wei et al., 2012; Vijayakumar et al., n.d.). The successful synthesis of Ag@Fe3O4 is confirmed by the presence of both Ag and Fe3O4 diffraction peaks in its pattern.
3.1.4 UV–vis analysis
The UV–vis spectroscopy offers valuable insights into the electronic structure of diverse materials, as the absorption of light at distinct wavelengths by different materials is attributed to their specific electronic transitions. Fe3O4 primarily absorbs light in the ultraviolet (UV) region due to electronic transitions involving its d orbitals. In the visible region, Fe3O4 does not have electronic transitions that coincide with the energy levels of visible light, resulting in no significant absorption peaks in this region, which is consistent with the spectrum of s-Fe3O4 (Fig. 3c) (Joshi et al., 2014; Sheng and Xue, 2012). The absorption peak of c-Ag is observed at 417 nm (Chen et al., 2010; Height et al., 2006), which is attributed to the phenomenon of surface plasmon resonance (SPR), where light energy is absorbed and leads to the collective oscillation of free electrons on the surface of silver nanoparticles. The peak for Ag@Fe3O4 is located at 486 nm, demonstrating a red shift in comparison to Ag. The observed outcome can be attributed to the fact that the refractive index of Fe3O4 (2.420) is higher than that of silver (0.399), resulting in a red shift of the SPR peak when silver core is coated with Fe3O4 shell (Anandan et al., 2008).
3.1.5 BET analysis
The specific surface area and pore characteristic (Fig. 3d) of Ag@Fe3O4 are determined by the BET analysis. According to the classification by the International Union of Pure and Applied Chemistry (IUPAC), the N2 adsorption/desorption isotherm of Ag@Fe3O4 exhibits a Type IV curve with a hysteresis loop, indicating its mesoporous structure (Zhao et al., 2011). The data analysis confirms that the Ag@Fe3O4 nanoparticles exhibit a relatively high specific surface area of 13.8 m2 g−1 and possess abundant mesopores with an average size of 29.9 nm. The high specific surface area ensures thorough contact between the active sites on Ag@Fe3O4 and the target (bacteria or organic dyes), while the abundant mesopores facilitate efficient mass transfer of Ag+ or e− from Ag@Fe3O4 to the target, both of which contribute to achieving exceptional antibacterial and dye degradation performance.
3.1.6 Magnetic hysteresis loops analysis
The Vibrating Sample Magnetometer (VSM) pattern typically exhibits a hysteresis loop, illustrating the correlation between magnetic field strength and material magnetization, thereby offering insights into the magnetic behavior of the material. The magnetization curve of the s-Fe3O4 and Ag@Fe3O4 particles at room temperature does not exhibit any noticeable remanence or coercivity, indicating their superparamagnetic behavior (Fig. 3e). The magnetic hysteresis loops indicate that the saturation magnetization of Ag@Fe3O4 is approximately 55.5 emu g−1, which is lower than that of s-Fe3O4 (72.8 emu g−1). The decrease can be attributed to the diamagnetic nature of Ag, which, when combined with the Fe3O4 shell, attenuates the magnetic properties of the s-Fe3O4. Nonetheless, the Ag@Fe3O4 still exhibits strong magnetic responsivity, as it has been reported that the application of a magnetic field can effectively facilitate the separation of materials from aqueous solutions when their saturation magnetization exceeds 16.3 emu·g−1 (Liu et al., 2015). The strong magnetic responsivity of Ag@Fe3O4 is directly demonstrated in the inset in Fig. 3e, where the particles easily attract towards an external magnetic field when a cylindrical NdFeB permanent magnet is positioned adjacent to the vial containing the Ag@Fe3O4 suspension. During the process, the Ag@Fe3O4 particles rapidly adhere to the inner walls of the vials within 20 s, resulting in a transparent remaining liquid.
3.1.7 FTIR analysis
The distinct peaks in the FTIR spectra can provide valuable information regarding the chemical composition and structure characteristic of the material. The peaks observed at approximately 594 cm−1 and 429 cm−1 in the Ag@Fe3O4 spectrum (Fig. 3f) correspond to the stretching vibrations of the tetrahedral (Fe3+–O2−) and octahedral (Fe2+–O2−) groups, respectively, confirming the presence of Fe3O4 (Husain et al., 2019). The absorption bands at around 3456 cm−1 and 1632 cm−1 can be attributed to vibrational peaks originating from the hydroxyl group on the surface of Ag@Fe3O4. Apart from the difference in peak intensity, the peak position of s-Fe3O4 is approximately similar to that of Ag@Fe3O4 due to silver's lack of absorption in the infrared region.
3.1.8 XPS analysis
The XPS is utilized for the validation of the electronic state and chemical composition of the particles. The XPS full spectrum of Ag@Fe3O4 exhibits characteristic peaks corresponding to Fe 2p, O 1 s, and Ag 3d (Fig. S2a, Supporting Information). The peaks observed at 710.2 eV, 711.6 eV, 723.3 eV, and 725.0 eV in the Fe 2p spectrum correspond to Fe2+ 2p3/2, Fe3+ 2p3/2, Fe2+ 2p1/2 and Fe3+ 2p1/2, respectively, which is in consistent with the typical peaks in Fe3O4 (Fig. 3g) (Ma et al., 2015; Liang et al., 2018; Wilson and Langell, 2014; Yamashita and Hayes, 2008). The other four peaks are satellite peaks. The O 1 s spectrum can be deconvoluted into two distinct peaks, specifically at 529.8 eV (Fe-O) and 530.8 eV (H-O) (Fig. S2b, Supporting Information), which are associated with the Fe3O4 and surface hydroxyl, respectively (Mu et al., 2011). The Ag 3d spectrum is deconvoluted into two distinct peaks representing Ag 3d5/2 (368.0 eV) and Ag 3d3/2 (374.0 eV) (Fig. 3h), which align with the established binding energy of metallic silver (Lou et al., 2013; Mottaghi et al., 2014). The XPS results prove the successful synthesis of the Ag@Fe3O4 composite.
3.1.9 ESR analysis
The ESR technique is used to qualitatively and quantitatively detect the unpaired electrons present in atoms or molecules of materials, as well as explore the structural properties of their surroundings. The ESR patterns (Fig. 3i) demonstrate the absence of any signal peaks without light (0 min), while six characteristic peaks corresponding to superoxide radical (•O2−) are observed under a mercury lamp for 2 min. The findings indicate that under light conditions, the Ag@Fe3O4 exhibits oxidative properties (Lee et al., 2022; Zhao et al., 2019).
3.2 Antibacterial performance and its mechanism
3.2.1 Measurement for the diameter of inhibition zone (DIZ)
The antibacterial effectiveness of Ag@Fe3O4 against E. coli is evaluated through the assessment for diameter of inhibition zone (DIZ), minimum inhibitory concentration (MIC), and cycling antibacterial assay. The absence of an inhibition zone around s-Fe3O4 at both concentrations suggests that s-Fe3O4 does not possess antibacterial properties and is incapable of forming an inhibition zone (Fig. 4a). In contrast to s-Fe3O4, both c-Ag and Ag@Fe3O4 exhibit distinct inhibition zones, indicating their antibacterial properties. The DIZ of c-Ag and Ag@Fe3O4 is observed to increase with their concentration, indicating a positive correlation between antibacterial activity and particle concentration. The DIZ of Ag@Fe3O4 is evidently larger than that of c-Ag at equivalent concentrations, thereby indicating the superior antibacterial activity of Ag@Fe3O4. This is attributed to the tendency of c-Ag to aggregate, which results in a reduced surface area available for bacterial interaction and consequently hinders the release of silver ions responsible for antibacterial activity. In the case of Ag@Fe3O4, the porous Fe3O4 shells prevent the aggregation of Ag cores and facilitate the sustained release of silver ions, thereby contributing to its superior antibacterial performance.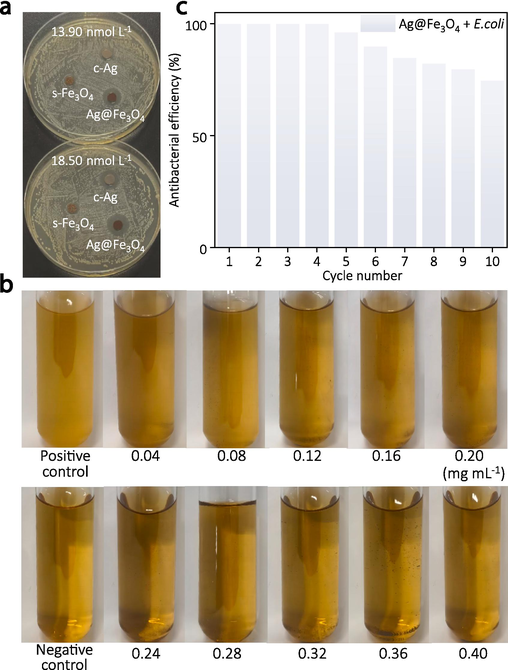
(a) Inhibition zones of the as-synthesized c-Ag, s-Fe3O4, and Ag@Fe3O4 with different concentrations against E. coli, (b) measurement of the minimum inhibitory concentration of the Ag@Fe3O4, (c) cycling antibacterial assessment of the Ag@Fe3O4.
3.2.2 Measurement of the minimum inhibitory concentration (MIC)
The broth microdilution method is employed to investigate the bactericidal effect of Ag@Fe3O4 at various concentrations, determining its MIC and quantitatively evaluating its efficacy against bacteria. A negative control group (only broth) and a positive control group (mixture of bacteria and broth) are set up for comparison. The bacteria continue to proliferate in the positive control group, resulting in the turbidity of the solution. The negative control group, however, exhibits a transparent solution due to the absence of bacteria (Fig. 4b). The solutions in the upper row tubes (0.04–0.20 mg mL−1) demonstrate a turbid state similar to that of the positive control group, indicating the sustained proliferation and growth of bacteria at these concentrations of the composite material. Conversely, the solutions in the lower row tubes (0.24–0.40 mg mL−1) exhibit clarity resembling that of the negative control group, thereby suggesting the inhibitory effect of the composite material on bacterial growth and reproduction at these concentrations. Consequently, the MIC for Ag@Fe3O4 is determined as 0.24 mg mL−1 (equivalent to 0.042 mg mL−1 in terms of Ag), representing the lowest concentration capable of impeding bacterial growth. The low MIC indicates the superior antibacterial performance of Ag@Fe3O4, which can be attributed to its intricate nanostructure. The porous Fe3O4 shell not only hinders the aggregation of Ag cores but also extends the release of silver ions, thereby contributing to an efficient and enduring antibacterial effect.
3.2.3 Cycling antibacterial assay
The reusability of Ag@Fe3O4 nanocomposite is evaluated by conducting a 10-cycle antibacterial experiment against E. coli (Fig. 4c). The results demonstrate that the antibacterial efficacy of Ag@Fe3O4 against E. coli remained stable during the first four cycles; however, it started to decline from the fifth cycle onwards, with an antibacterial efficiency of 74.6 % by the tenth cycle. This phenomenon can be attributed to the inevitable loss of Ag@Fe3O4 over multiple cycles, which leads to a diminished inhibitory effect on bacteria. Additionally, bacterial adhesion onto the surface of the material during cycling could hinder direct contact between Ag@Fe3O4 and bacteria, thereby reducing its effectiveness. In general, the Ag@Fe3O4 composite exhibits exceptional cyclic antibacterial performance due to its delicate structure, in which the porous Fe3O4 shell not only ensures sustained release of silver ions but also demonstrates excellent magnetic properties for efficient and high recovery rates under an external magnetic field.
The aforementioned advantages of Ag@Fe3O4 function synergistically and contribute to its exceptional antibacterial performance and reutilization, surpassing that of numerous previously reported Ag-based composites (Table S2, Supporting Information), thereby demonstrating its significant potential for medical applications and sewage treatment.
3.2.4 Possible antibacterial mechanism
The Ag@Fe3O4 composite demonstrates remarkable antibacterial efficacy, and multiple potential antibacterial mechanisms are implicated (Fig. 5).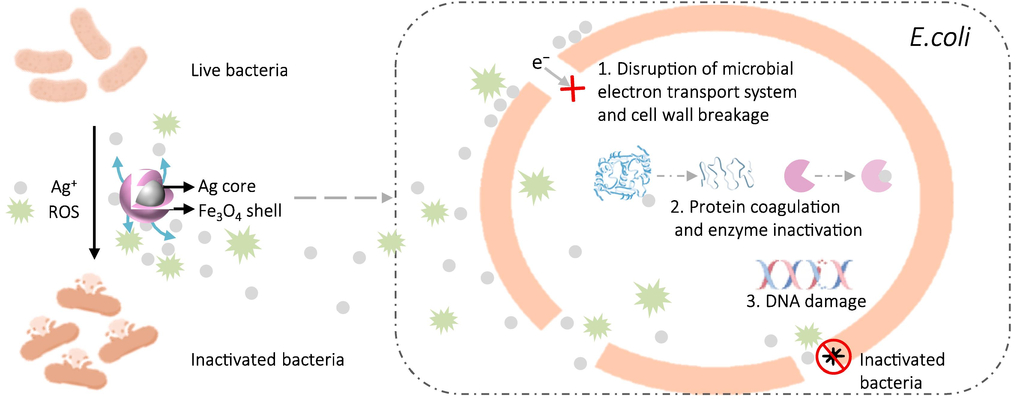
The diagram illustrating the possible antibacterial mechanism of the Ag@Fe3O4.
Firstly, the negatively charged bacterial cell wall strongly binds to Ag+ through Coulombic attraction. On one hand, these Ag+ impact the permeability of the bacterial membrane and disrupt both the microbial electron transport system and material transport system. On the other hand, the interaction between Ag+ and the cell wall triggers a peptidoglycan reaction, resulting in the degradation of crucial components and functional impairments within bacteria. Furthermore, penetration of Ag+ into the cell wall causes rupture and leakage of cellular fluid, ultimately resulting in bacterial death (Akhavan, 2009; Sotiriou and Pratsinis, 2010).
Secondly, upon penetration of the cell wall and entry into the bacterial cell, Ag+ selectively binds to specific groups (such as sulfhydryl group) within the bacterial cells, inducing protein coagulation. This process disrupts the enzymatic activity responsible for cellular synthesis, thereby impairing the division and reproduction ability of bacteria, ultimately leading to their demise (Yuranova et al., 2003; Ding et al., 2017).
Thirdly, the silver ions can induce DNA damage in bacteria. On one hand, silver ions can disrupt the structure and function of specific proteins, thereby reducing the efficiency of oxidative phosphorylation and impeding ATP synthesis, which results in impairment to DNA replication. On the other hand, the presence of silver ions induces the generation of reactive oxygen species (ROS, including the superoxide radical of •O2− confirmed by the ESR analysis), which can displace hydrogen atoms within DNA molecules. This displacement results in structural deformations in bacterial DNA, thereby impeding the synthesis of DNA and proteins and ultimately rendering the bacteria inactive (Li et al., 2005; Sondi and Salopek-Sondi, 2004; Li et al., 2021; Zhang et al., 2023).
The porous Fe3O4 shell in the Ag@Fe3O4 effectively impedes the aggregation of Ag, ensuring a sustained release of silver ions and exhibiting exceptional magnetic properties. All these factors of Ag@Fe3O4 function synergistically to efficiently eradicate bacteria and achieve a high recovery rate, offering great potential for the development of medical devices, antibacterial coatings, and sewage treatment applications.
3.3 Catalytic degradation of dyes and its mechanism
3.3.1 Catalytic degradation of dyes
The as-synthesized Ag@Fe3O4 and its control samples of c-Ag and s-Fe3O4 are subjected to the degradation experiment of organic dyes (RB, R6G, and MB) in the presence of NaBH4. The detailed experimental parameters are as follows: the catalytic temperature is room temperature (∼25 °C), the dye concentration is 5 mg L−1, the solution pH is 7, and the dosage of Ag@Fe3O4 is 20 mg per each catalytic experiment. The results indicate very little degradation of RB after 15 min by either pure Ag@Fe3O4 (Fig. 6a) or pure NaBH4 (Fig. 6b), while the presence of both Ag@Fe3O4 and NaBH4 achieves near complete degradation of RB within only 5 min (Fig. 6c). The corresponding digital images visually demonstrate the degradation effect of both Ag@Fe3O4 and NaBH4 towards RB since the solution color gradually faded with the reaction time (Fig. 6d), confirming their highly efficient degradation effect. The degradation effect is further quantitatively assessed by calculating their degradation efficiency (Fig. 6e). The degradation efficiency of both Ag@Fe3O4 and NaBH4 towards RB can reach nearly 100 % within 5 min, whereas after 15 min, the pure Ag@Fe3O4 and pure NaBH4 only achieve approximately 18.7 % and 36.8 %, respectively. The degradation efficiency for the control samples of c-Ag and s-Fe3O4 in the presence of NaBH4 after 5 min are 48.2 % and 83.8 %, respectively, also exhibiting a limited degradation effect towards RB when compared to Ag@Fe3O4 (Figs. S3, S4, S5, Supporting Information).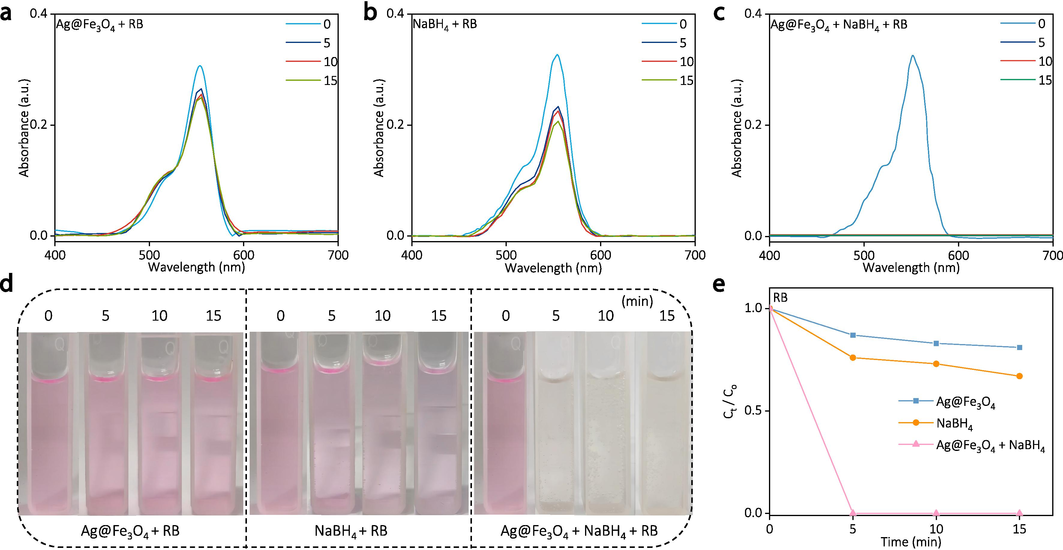
The degradation of RB by pure Ag@Fe3O4, pure NaBH4, and “Ag@Fe3O4 + NaBH4”: (a-c) the UV–vis spectra at different time, (d) corresponding digital images, and (e) degradation efficiency over time.
The superior degradation efficiency of Ag@Fe3O4 towards RB can be attributed to multiple factors. Firstly, the Fe3O4 shell effectively prevents the agglomeration of the Ag core, thereby preserving a high specific surface area and activity for the catalyst (M.a. Ghasemzadeh, J.s. Ghomi, S. Zahedi, Fe3O4 nanoparticles: A highly efficient and easily reusable catalyst for the one-pot synthesis of xanthene derivatives under solvent-free conditions, Journal of the Serbian Chemical Society 78(6), 2013; Wang et al., 2021). Secondly, the presence of mesopores and abundant active sites in the Fe3O4 shell enhances dye adsorption efficiency and facilitates increased contact between dye molecules and Ag core. Thirdly, the Ag with high electrical conductivity serves as an electron mediator, capturing electrons from the nucleophilic NaBH4 and transferring them to the electrophilic RB during the reaction, ultimately resulting in efficient degradation of the RB (ZhongJie Jiang, 2005; Zheng and Wang, 2012). The degradation performance of RB by Ag@Fe3O4 surpasses that of pure c-Ag and s-Fe3O4, owing to the synergistic effect resulting from the aforementioned advantages of Fe3O4 shell and Ag core.
Similarly, the Ag@Fe3O4 demonstrates remarkable degradation efficiency towards various organic dyes, including R6G (Fig. 7) and MB (Fig. 8). The Ag@Fe3O4 can completely degrade R6G within 5 min in the presence of NaBH4, outperforming the control samples of pure Ag@Fe3O4 and pure NaBH4, as evidenced by the UV–vis spectra and corresponding digital images (Fig. 7).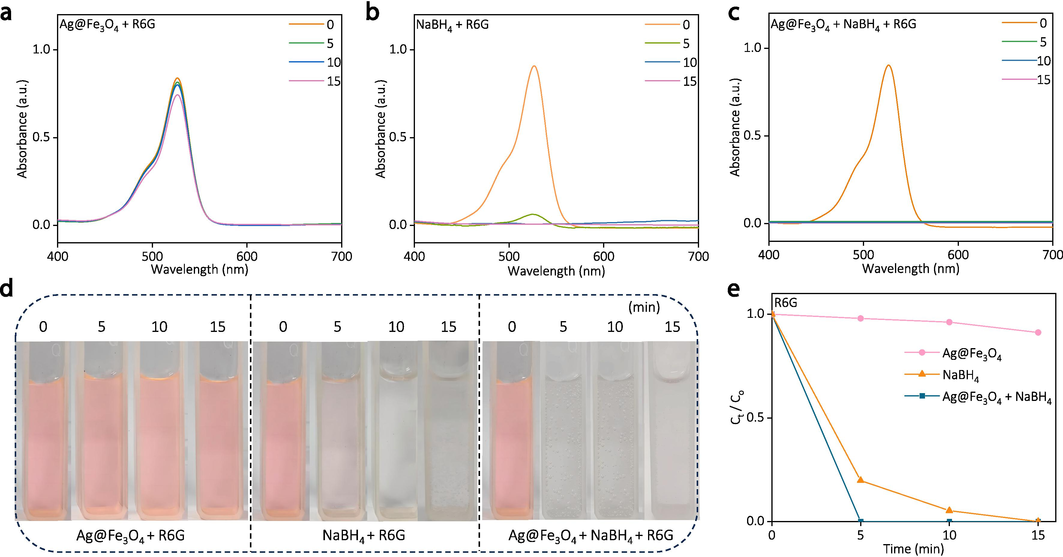
The degradation of R6G by pure Ag@Fe3O4, pure NaBH4, and “Ag@Fe3O4 + NaBH4”: (a-c) the UV–vis spectra at different time, (d) corresponding digital images, and (e) degradation efficiency over time.

The degradation of MB by pure Ag@Fe3O4, pure NaBH4, and “Ag@Fe3O4 + NaBH4”: (a-c) the UV–vis spectra at different time, (d) corresponding digital images, and (e) degradation efficiency over time.
The Ag@Fe3O4 also exhibit the superior degradation performance towards MB (Fig. 8). The remarkable degradation capability of Ag@Fe3O4 towards RB, R6G, and MB proves its relatively universal effect on degrading organic dyes, thereby indicating its potential for practical application in dye-containing wastewater treatment.
3.3.2 Cycling degradation property
To assess the practical applicability of Ag@Fe3O4 in wastewater treatment, the investigation into its cycling degradation property is conducted. During the experiment, Ag@Fe3O4 is easily reused by being collected with a magnet. The results demonstrate that even after 6 consecutive cycles of reutilization, the Ag@Fe3O4 still exhibits a remarkable degradation efficiency of 90.9 % towards RB, highlighting its exceptional potential for practical wastewater treatment (Fig. 9a). Similarly, the Ag@Fe3O4 exhibit excellent catalytic degradation efficiency towards Rhodamine 6G (Fig. 9b) and Methylene blue (Fig. 9c) after 6 cycles, both of which achieving 100 %.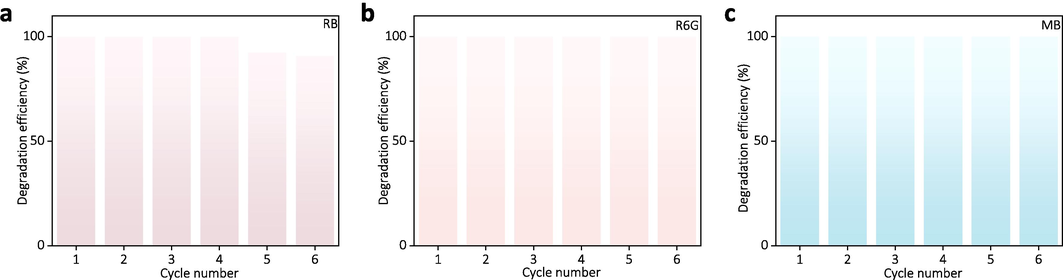
Cycling degradation performance of the Ag@Fe3O4 towards (a) RB, (b) R6G, and (c) MB.
The decrease in the degradation efficiency of Ag@Fe3O4 towards organic dyes with increasing cycle number may be attributed to several factors. Firstly, during the process of magnetic recovery, some loss of Ag@Fe3O4 nanoparticles is inevitable (such as incomplete separation of nanoparticles from the solution and adherence of particles to container surfaces), leading to a reduction in degradation efficiency during subsequent cycles due to the decrease in available catalyst. Secondly, during cycling, the surface of Ag@Fe3O4 nanoparticles can become fouled with reaction intermediates, by-products, or residual organic dyes, thereby obstructing active sites on the nanoparticles and diminishing their catalytic efficiency. Thirdly, the leaching and loss of the silver component in Ag@Fe3O4 during the degradation process can result in a reduction of catalytic activity, as silver plays a crucial role in the degradation mechanism (Wang et al., 2021).
Despite a decrease in degradation efficiency with an increasing cycle number, the overall degradation efficiency of RB, R6G, and MB can still exceed 90.9 % after 6 cycles, highlighting the superior degradation efficiency of Ag@Fe3O4 towards organic dyes. The catalytic degradation efficiency of the obtained Ag@Fe3O4 towards various organic dyes surpasses that of many previously reported Ag-based composites (Table S3, Supporting Information). Additionally, the Ag@Fe3O4 is cost-effective (around $0.17 per gram in terms of chemical reagents, the detailed calculation can be seen in Table S4, Supporting Information), underscoring its potential for practical applications in dye-containing wastewater treatment.
3.3.3 Possible degradation mechanism
The Ag@Fe3O4 exhibit exceptional catalytic degradation properties towards organic dyes, involving multiple potential mechanisms. The degradation of RB can be taken as an example (Figure S6, Supporting Information). The cationic dye RB is electrostatically attracted to the hydroxyl groups on the surface of the Fe3O4 shell or adsorbed onto the active sites on the Fe3O4 shell through van der Waals force. The Ag serves as an electron transfer mediator, facilitating the efficient electron transfer from the nucleophilic NaBH4 to the electrophilic RB molecules. The electrons induce reduction of the chromophoric structure and disruption of the conjugated system in RB, leading to the decolorization of the dye. The final products may consist of Leuco-Rhodamine B, aminated and de-ethylated derivatives, as well as inorganic byproducts (potentially including borates) (Liu et al., 2023; Wang et al., 2016). The degradation process of R6G (Fig. S7, Supporting Information) and MB (Fig. S8, Supporting Information) is similar to that of RB, except for the potential final products differing from each other.
4 Conclusions
The magnetoplasmonic core–shell structured Ag@Fe3O4 particles have been synthesized via a one-step polyol reduction process, exhibiting dual functionality for bacteria eradication and organic dyes degradation. The delicate nanostructure of the Ag@Fe3O4 not only enhances the antibacterial efficacy and catalytic degradation efficiency of the particles but also exploits the magnetic properties of Fe3O4 for facile recovery and reutilization. The Ag@Fe3O4 exhibits nearly 100 % inhibition of E. coli at a concentration of 0.24 mg mL−1 (with an Ag content of 0.042 mg mL−1), while maintaining antibacterial efficacy at 74.6 % even after undergoing reutilization for 10 cycles. Meanwhile, the Ag@Fe3O4 exhibits remarkable efficacy in catalytically degrading various organic dyes (including Rhodamine B, Rhodamine 6G, and Methylene blue) within only 15 min in the presence of NaBH4, while achieving an impressive degradation efficiency exceeding 90.9 % after undergoing 6 cycles. The cost-effectiveness (approximately $0.17 per gram) and facile magnetic recovery of Ag@Fe3O4 particles further augment their practicality for large-scale applications in antibacterial treatments and environmental remediation, thereby promoting both ecological conservation and efficient resource utilization. The future research could focus on optimizing the synthesis parameters and scalability of the production process, functionalizing the surface with specific ligands or coatings to enhance interaction with target molecules or bacteria, combining Ag@Fe3O4 with other nanomaterials to create hybrid systems that offer synergistic effects, and integrating the nanoparticles into practical devices or systems for antibacterial applications and water purification (such as filters, coatings, and sensors).
CRediT authorship contribution statement
Qing Shen: Writing – original draft, Formal analysis, Data curation. Luping Zhang: Writing – review & editing. Yuan Zhao: Writing – review & editing. Xiaobing Han: Writing – review & editing. Jie Gao: Writing – review & editing. Yuesheng Li: Writing – review & editing. Xiaoming Zhu: Writing – review & editing. Tian Liang: Writing – review & editing, Formal analysis, Conceptualization. Tao Chen: Writing – review & editing.
Acknowledgments
This work was financially supported by the Scientific Research Foundation of Hubei University of Science and Technology, China (2022ZX10, BK202314, 2022T03).
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Lasting antibacterial activities of Ag-TiO2/Ag/a-TiO2 nanocomposite thin film photocatalysts under solar light irradiation. J. Colloid Interface Sci.. 2009;336(1):117-124.
- [CrossRef] [Google Scholar]
- Synthesis and characterization of magnetic Ag-Fe3O4@polymer hybrid nanocomposite systems with promising antibacterial application. Drug Dev. Ind. Pharm.. 2023;49(12):723-733.
- [CrossRef] [Google Scholar]
- Rapid color degradation of organic dyes by Fe3O4@His@Ag recyclable magnetic nanocatalyst. J. Ind. Eng. Chem.. 2015;27:347-353.
- [CrossRef] [Google Scholar]
- Sonochemical synthesis of Au-Ag core-shell bimetallic nanoparticles. J. Phys. Chem. C. 2008;112(39):15102-15105.
- [CrossRef] [Google Scholar]
- Antifungal properties of electrically generated metallic ions. American Society for Microbiology. 1976;10(5):856-860.
- [CrossRef] [Google Scholar]
- Compact Ag@Fe3O4 Core-shell nanoparticles by means of single-step thermal decomposition reaction. Sci. Rep.. 2014;4(1):6839.
- [CrossRef] [Google Scholar]
- Potential uses and value-added products derived from waste polystyrene in developing countries: A review. Resour. Conserv. Recycl.. 2016;107:157-165.
- [CrossRef] [Google Scholar]
- Size-Controlled and Size-Designed Synthesis of Nano Submicrometer Ag Particles. Cryst. Growth Des.. 2010;10(8):3378-3386.
- [CrossRef] [Google Scholar]
- Amorphous InGaZnO Thin Film Transistors with Wet-Etched Ag Electrodes. ECS Solid State Lett.. 2014;3(6):Q29-Q32.
- [CrossRef] [Google Scholar]
- Chitosan-stabilized Ag nanoparticles with superior biocompatibility and their synergistic antibacterial effect in mixtures with essential oils. Nanomaterials. 2018;8(10):826.
- [CrossRef] [Google Scholar]
- Au-Ag core-shell nanoparticles for simultaneous bacterial imaging and synergistic antibacterial activity, Nanomedicine: Nanotechnology. Biology and Medicine. 2017;13(1):297-305.
- [CrossRef] [Google Scholar]
- One-step hydrothermal synthesis of Ag decorated TiO2 nanoparticles for dye-sensitized solar cell application. Renew. Energy. 2019;135:1207-1212.
- [CrossRef] [Google Scholar]
- The surface modification of spherical ZnO with Ag nanoparticles: A novel agent, biogenic synthesis, catalytic and antibacterial activities. Arab. J. Chem.. 2022;15(3):103658
- [CrossRef] [Google Scholar]
- One-Pot Synthesis of SiO2@Ag Mesoporous Nanoparticle Coating for Inhibition of Escherichia coli Bacteria on Various Surfaces. Nanomaterials. 2021;11(2):549.
- [CrossRef] [Google Scholar]
- Fe3O4 nanoparticles: A highly efficient and easily reusable catalyst for the one-pot synthesis of xanthene derivatives under solvent-free conditions. J. Serbian Chem. Soc.. 2013;78(6):769-779.
- [CrossRef] [Google Scholar]
- Enhanced photocatalytic and antibacterial activities of mechanosynthesized TiO2-Ag nanocomposite in wastewater treatment. J. Mol. Struct.. 2020;1211:128076
- [CrossRef] [Google Scholar]
- Preparation and antibacterial activity of Fe3O4@Ag nanoparticles. Nanotechnology. 2007;18(28):285604
- [CrossRef] [Google Scholar]
- Mild aqueous synthesis of urchin-like MnOx hollow nanostructures and their properties for RhB degradation. Chem. Eng. J.. 2013;229:134-143.
- [CrossRef] [Google Scholar]
- Ag-ZnO catalysts for UV-photodegradation of methylene blue. Appl Catal B. 2006;63(3–4):305-312.
- [CrossRef] [Google Scholar]
- The preparation and lithium battery performance of core-shell SiO2@Fe3O4@C composite. Ceram. Int.. 2017;43(14):11505-11510.
- [CrossRef] [Google Scholar]
- Synthesis and characterization of Fe3O4 magnetic nanoparticles from iron ore. J. Phys. Conf. Ser.. 2019;1242(1):012021
- [CrossRef] [Google Scholar]
- A critical review on silver nanoparticles: From synthesis and applications to its mitigation through low-cost adsorption by biochar. J. Environ. Manage.. 2021;281:111918
- [CrossRef] [Google Scholar]
- Preparation and antibacterial effects of Ag-SiO2 thin films by sol-gel method. Biomaterials. 2003;24(27):4921-4928.
- [CrossRef] [Google Scholar]
- Stable nanocomposite based on PEGylated and silver nanoparticles loaded graphene oxide for long-term antibacterial activity. ACS Appl. Mater. Interfaces. 2017;9(18):15328-15341.
- [CrossRef] [Google Scholar]
- Facile hydrothermal synthesis of Fe3O4@cellulose aerogel nanocomposite and its application in Fenton-like degradation of Rhodamine B. Carbohydr. Polym.. 2018;189:371-378.
- [CrossRef] [Google Scholar]
- Catalytic properties of silver nanoparticles supported on silica spheres. J. Phys. Chem. B. 2005;109(5):1730-1735.
- [CrossRef] [Google Scholar]
- Quantitative SERS-based detection using Ag-Fe3O4 nanocomposites with an internal reference. J. Mater. Chem. C. 2014;2(46):9964-9968.
- [CrossRef] [Google Scholar]
- Magnetically sensitive TiO2 hollow sphere/Fe3O4 core-shell hybrid catalyst for high-performance sunlight-assisted photocatalytic degradation of aqueous antibiotic pollutants. J. Alloy. Compd.. 2022;902:163612
- [CrossRef] [Google Scholar]
- Synergistic antibacterial effects of β-lactam antibiotic combined with silver nanoparticles. Nanotechnology. 2005;16(9):1912-1917.
- [CrossRef] [Google Scholar]
- Ag/AgX nanostructures serving as antibacterial agents: achievements and challenges. Rare Met.. 2021;41(2):519-539.
- [CrossRef] [Google Scholar]
- High-energy flexible quasi-solid-state lithium-ion capacitors enabled by a freestanding rGO-encapsulated Fe3O4 nanocube anode and a holey rGO film cathode. Nanoscale. 2018;10(37):17814-17823.
- [CrossRef] [Google Scholar]
- Dichroic switching of core-shell plasmonic nanoparticles on reflective surfaces. Exploration 2023:20210234.
- [CrossRef] [Google Scholar]
- Microbial preparation of magnetite reduced graphene oxide nanocomposites for the removal of organic dyes from aqueous solutions. RSC Adv.. 2015;5(116):95857-95865.
- [CrossRef] [Google Scholar]
- Recent advances of magnetite (Fe3O4)-based magnetic materials in catalytic applications. Magnetochemistry. 2023;9(4):110.
- [CrossRef] [Google Scholar]
- Fast-generation of Ag3PO4 concave microcrystals from electrochemical oxidation of bulk silver sheet. CrstEngComm. 2013;15(25):5070-5075.
- [CrossRef] [Google Scholar]
- Fabrication of cubic nanocages and nanoframes by dealloying Au/Ag alloy nanoboxes with an aqueous etchant based on Fe(NO3)3 or NH4OH. Nano Lett.. 2007;7(6):1764-1769.
- [CrossRef] [Google Scholar]
- Growth Mechanism and Electrical and Magnetic Properties of Ag-Fe3O4 Core-Shell Nanowires. ACS Appl. Mater. Interfaces. 2015;7(29):16027-16039.
- [CrossRef] [Google Scholar]
- Polymer-Ag Nanocomposites with Enhanced Antimicrobial Activity against Bacterial Infection. ACS Appl. Mater. Interfaces. 2014;6(18):15813-15821.
- [CrossRef] [Google Scholar]
- Nanoparticles in construction materials and other applications and implications of nanoparticle use. Materials. 2019;12(19):3052.
- [CrossRef] [Google Scholar]
- Ag/Pd core-shell nanoparticles by a successive method: Pulsed laser ablation of Ag in water and reduction reaction of PdCl2. Appl. Surf. Sci.. 2014;292:892-897.
- [CrossRef] [Google Scholar]
- Highly dispersed Fe3O4 nanosheets on one-dimensional carbon nanofibers: Synthesis, formation mechanism, and electrochemical performance as supercapacitor electrode materials. Nanoscale. 2011;3(12):5034-5040.
- [CrossRef] [Google Scholar]
- The minimum inhibitory concentration (MIC) and minimum bactericidal concentration (MBC) of silver nanoparticles against Staphylococcus aureus. Biomater. Invest. Dent.. 2020;7(1):105-109.
- [CrossRef] [Google Scholar]
- Briefly brominated Ag thin films: XRD, FESEM, and optical studies of surface modification. Appl. Surf. Sci.. 2012;259:276-282.
- [CrossRef] [Google Scholar]
- Green synthesis of Ag nanoparticles in oil-in-water nano-emulsion and evaluation of their antibacterial and cytotoxic properties as well as molecular docking. Arab. J. Chem.. 2021;14(9):103323
- [CrossRef] [Google Scholar]
- Solid substrates decorated with Ag nanostructures for the catalytic degradation of methyl orange. Results Phys.. 2019;12:1133-1141.
- [CrossRef] [Google Scholar]
- Antimicrobial Resistance: A Growing Serious Threat for Global Public Health. Healthcare. 2023;11(13):1946.
- [CrossRef] [Google Scholar]
- Copper-Resistant Bacteria from Industrial Effluents and Their Role in Remediation of Heavy Metals in Wastewater. Folia Microbiol.. 2002;47:43-50.
- [CrossRef] [Google Scholar]
- Synthesis and properties of Au-Fe3O4 heterostructured nanoparticles. J. Colloid Interface Sci.. 2012;374(1):96-101.
- [CrossRef] [Google Scholar]
- Preparation of Magnetically Recoverable MPCTP-Ag Composite Nanoparticles and Their Application as High-Performance Catalysts. Langmuir. 2021;37(34):10249-10258.
- [CrossRef] [Google Scholar]
- Study on formation of Pd nanocatalyst in self-reducing silica nanotube produced by using sacrificial Fe3O4 template and its efficacy in Cr(VI) reduction. Mater. Chem. Phys.. 2022;278:125580
- [CrossRef] [Google Scholar]
- A strawberry-like Ag-decorated barium titanate enhances piezoelectric and antibacterial activities of polymer scaffold. Nano Energy. 2020;74:104852
- [CrossRef] [Google Scholar]
- I. Sondi, B. Salopek-Sondi, Silver nanoparticles as antimicrobial agent: a case study on E. coli as a model for Gram-negative bacteria, Journal of Colloid and Interface Science 275(1) (2004) 177-182. DOI: 10.1016/j.jcis.2004.02.012.
- Antibacterial activity of nanosilver ions and particles. Environ. Sci. Tech.. 2010;44(14):5649-5654.
- [CrossRef] [Google Scholar]
- Antibacterial Activity of Silver Nanoparticles: Structural Effects. Adv. Healthc. Mater.. 2018;7(13):1701503.
- [CrossRef] [Google Scholar]
- In situ self-assembled Ag-Fe3O4 nanoclusters in exosomes for cancer diagnosis. J. Mater. Chem. B. 2020;8(14):2845-2855.
- [CrossRef] [Google Scholar]
- Vijayakumar, R., Koltypin, Y., Felner, I., Gedanken, A., n.d. Sonochemical synthesis and characterization of pure nanometer-sized Fe3O4 particles. Mat. Sci. Eng. A 286 (1), 101–105. https://doi.org/10.1016/S0921-5093(00)00647-X.
- Rhodium nanoparticles supported on 2-(aminomethyl) phenols-modified Fe3O4 spheres as a magnetically recoverable catalyst for reduction of nitroarenes and the degradation of dyes in water. Catal. Lett.. 2021;152(4):1076-1085.
- [CrossRef] [Google Scholar]
- Silica coated Fe3O4 magnetic nanospheres for high removal of organic pollutants from wastewater. Chem. Eng. J.. 2016;306:280-288.
- [CrossRef] [Google Scholar]
- Hollow porous core-shell ZnFe2O4/AgCl nanocubes coated with EDTA and Ag nanoparticles for enhanced photocatalytic performances of visible-light-driven. Chem. Eng. J.. 2020;400:125908
- [CrossRef] [Google Scholar]
- Synthesis of Fe3O4 nanoparticles and their magnetic properties. Procedia Eng.. 2012;27:632-637.
- [CrossRef] [Google Scholar]
- XPS analysis of oleylamine/oleic acid capped Fe3O4 nanoparticles as a function of temperature. Appl. Surf. Sci.. 2014;303:6-13.
- [CrossRef] [Google Scholar]
- X.h. Meng, X. Shao, H.y. Li, F.z. Liu, X.p. Pu, W.z. Li, C.h. Su, One-step hydrothermal synthesis, characterization and visible-light catalytic property of Ag-reduced graphene oxide composite, Materials Research Bulletin 48(4) (2013) 1453-1457. DOI: DOI: 10.1016/j.materresbull.2012.12.037.
- Nanofibrillated cellulose as the support and reductant for the facile synthesis of Fe3O4/Ag nanocomposites with catalytic and antibacterial activity. J. Mater. Chem. A. 2013;1(47)
- [CrossRef] [Google Scholar]
- Analysis of XPS spectra of Fe2+ and Fe3+ ions in oxide materials. Appl. Surf. Sci.. 2008;254(8):2441-2449.
- [CrossRef] [Google Scholar]
- Metal-organic framework Ag-based hybrid nanoagents for rapid and synergistic bacterial eradication. ACS Appl. Mater. Interfaces. 2020;12(12):13698-13708.
- [CrossRef] [Google Scholar]
- In situ reduction of silver nanoparticles by gelatin to obtain porous silver nanoparticle chitosan composites with enhanced antimicrobial and wound-healing activity. Int. J. Biol. Macromol.. 2019;121:633-642.
- [CrossRef] [Google Scholar]
- Recyclable magnetite-silver heterodimer nanocomposites with durable antibacterial performance. Bioact. Mater.. 2018;3(1):80-86.
- [CrossRef] [Google Scholar]
- Self-template etching synthesis of urchin-like Fe3O4 microspheres for enhanced heavy metal ions removal. Langmuir. 2018;34(32):9359-9365.
- [CrossRef] [Google Scholar]
- Antibacterial textiles prepared by RF-plasma and vacuum-UV mediated deposition of silver. J. Photochem. Photobiol. A Chem.. 2003;161(1):27-34.
- [CrossRef] [Google Scholar]
- Rapid synthesis of MoS2-PDA-Ag nanocomposites as heterogeneous catalysts and antimicrobial agents via microwave irradiation. Appl. Surf. Sci.. 2018;459:588-595.
- [CrossRef] [Google Scholar]
- Facile one-step synthesis of plasmonic magnetic core shell nanostructures and their multifunctionality. J. Mater. Chem.. 2012;22(21):10779.
- [CrossRef] [Google Scholar]
- Syntheses, Plasmonic Properties, and Catalytic Applications of Ag–Rh Core-Frame Nanocubes and Rh Nanoboxes with Highly Porous Walls. Chem. Mater.. 2018;30(6):2151-2159.
- [CrossRef] [Google Scholar]
- Montmorillonite/Agarose Three-Dimensional Network Gel Sponge for Wound Healing with Hemostatic and Durable Antibacterial Properties. ACS Appl. Nano Mater.. 2023;6(18):17263-17275.
- [CrossRef] [Google Scholar]
- Silver coated magnetic microflowers as efficient and recyclable catalysts for catalytic reduction. New J. Chem.. 2017;41(23):14199-14208.
- [CrossRef] [Google Scholar]
- Iron‐oxide‐based advanced anode materials for lithium‐ion batteries. Adv. Energy Mater.. 2014;4(4):1300958.
- [CrossRef] [Google Scholar]
- Synthesis, characterization, and photocatalytic properties of Ag modified hollow SiO2/TiO2 hybrid microspheres. Appl Catal B. 2011;103(1–2):181-189.
- [CrossRef] [Google Scholar]
- Stable nanocomposite based on PEG ylated and silver nanoparticles loaded graphene oxide for long-term antibacterial activity. ACS Appl. Mater. Interfaces. 2017;9(18):15328-15341.
- [CrossRef] [Google Scholar]
- Dual sensitization effect and conductive structure of Fe3O4@mTiO2/C photocatalyst towards superior photodegradation activity for bisphenol A under visible light. J. Photochem. Photobiol. A Chem.. 2019;382:111902
- [CrossRef] [Google Scholar]
- Ag nanoparticle-entrapped hydrogel as promising material for catalytic reduction of organic dyes. J. Mater. Chem.. 2012;22(32):16552-16559.
- [CrossRef] [Google Scholar]
Appendix A
Supplementary data
Supplementary data to this article can be found online at https://doi.org/10.1016/j.arabjc.2024.106058.
Appendix A
Supplementary data
The following are the Supplementary data to this article:Supplementary Data 1
Supplementary Data 1







