Translate this page into:
Mxene Ti3C2Tx derived lamellar Ti3C2Tx-TiO2-CuO heterojunction: Significantly improved ammonia sensor performance
⁎Corresponding authors. xiaolei.ye@utbm.fr (Xiaolei Ye), yanglikmust@163.com (Li Yang)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Ammonia (NH3), as main industrial gas emissions, are extremely harmful to the human beings. So the detection of NH3 concentrations in special environments at room temperature (25 °C) is essential. Herein, the composite MXene Ti3C2Tx-TiO2-CuO was successfully prepared by one-step in-situ oxidation method using Cu(NO3)2·6H2O as a precursor. The morphology of Ti3C2Tx-TiO2-CuO shows layered accordion-like structure. Ti3C2Tx-TiO2-CuO sensor exhibits improved NH3 gas sensing response of 56.9 toward 100 ppm NH3 at room temperature under ultraviolet (UV) light exposure, which is 89.8 % higher than that of pristine Ti3C2Tx material. The improved gas sensing performance is attributed to the irradiation of UV light, the retention of Ti3C2Tx layered structure and the construction of TiO2-CuO heterojunction. Further, the possible gas sensing mechanism of the material was proposed and discussed.
Keywords
MXene Ti3C2Tx
UV irradiation
MXene-TiO2-CuO heterojunction
Gas sensor
NH3
1 Introduction
In the past decades, atmospheric environmental pollution has been a major problem plaguing most countries in the world (Chan and Yao, 2008). NH3, as one of the atmospheric pollutants, is the main cause of acid rain. Long-term inhalation of NH3 will cause pulmonary edema, respiratory distress syndrome and other diseases, which will cause serious harm to the human body (Shkir et al., 2022; Qin et al., 2022; Khudher and Abd, 2021; Alothman and Wabaidur, 2019; Late et al., 2014; Wu et al., 2018). Therefore, the development and preparation of gas sensing materials with excellent NH3 gas sensing properties have always been an important topic of concern. Addressing the above issues, researchers have carried out extensive and in-depth research on the exploration of gas sensing materials and the preparation of gas sensors. Among many gas sensing materials, metal oxide semiconductor (MOS) has received extensive researches and attentions, such as SnO2, TiO2, ZnO, CuO, WO3 and so on (Lei et al., 2020; Meng et al., 2015; Xu et al., 2022; Aliha et al., 2013; Kou et al., 2016; Li et al., 2022; Zhou et al., 2021; Wang et al., 2021). Shahabuddin et al prepared NH3 gas sensor based on SnO2 films modified with metallic clusters (MCS). And they investigated the effect of ion species on the gas sensing properties of SnO2. The results show that the integration of Pt is beneficial for enhancing the gas sensing properties of SnO2, it shows a high response value of 25.7 toward 450 ppm NH3 at 230 °C (Shahabuddin et al., 2014). Srinivasulu et al fabricated a 2D ZnO gas sensor, which displays ultra-fast gas sensing response to NH3. The sensor exhibits a maximum response of 80% toward NH3 at 250 °C (Kanaparthi and Singh, 2020). Obviously, MOS-based gas sensors have shown excellent gas sensitivity. However, they usually require high operating temperatures to ensure electrons transfer from Valence Band (VB) to Conduction Band (CB). The high working temperature makes the gas sensor unstable, and confines the utilization in complex atmosphere environment and flexible devices. Thus, it is important to search a low/room-temperature gas sensing material to replace MOS or devise a novel method to reduce operating temperature.
MXene as a promising two-dimensional (2D) material introduces the special characteristics and advantages, such as high specific surface area, abundant functional groups. Typically, Ti3C2Tx (Tx represents, -F, –OH and -O) is the most widely studied MXene because of its superior electrical conductivity, organ-like structure and mature fabrication process (Akhtar et al., 2021; Xu et al., 2017; Deng et al., 2022; Yuan et al., 2018; Chen et al., 2019; Xie et al., 2018), which make it the preferred material for gas sensing field. Although several works have been done to investigate the gas sensing properties of MXene and they have shown good selectivity for NH3 and VOCs (Sun et al., 2020; Kim et al., 2018; Gasso et al., 2022; Li et al., 2021; Zhao et al., 2021; Zhao et al., 2020; Zhang et al., 2022). There are still large challenges for improvement in the gas sensitivity of the material. On the basis of the working mechanism of resistive gas sensor, the gas sensitivity can be enhanced by constructing the heterostructure to build energy level interleaving effect, which can effectively promote the separation of free carriers, so that more free charges can take part in the gas–solid reaction and improve the gas sensitivity (Miller et al., 2014; Hang et al., 2017).
As is well known, titanium dioxide (TiO2) has been widely studied in photocatalytic and gas sensing fields because of high stability, high electrochemical performance, and excellent catalytic performance (Lai et al., 2010; Chen et al., 2012; Wang et al., 2011; Zhang et al., 2014). At the same time, in order to improve the gas sensing properties of TiO2 and broaden its application in gas sensing materials, a lot of works have been done to combine TiO2 with CuO to form p-n heterojunction. The construction of p-n heterojunction is conducive to electron-hole separation, so that more carriers participate in interfacial chemical reactions. Alev et al. (Alev and Sennik, 2018) prepared CuO thin film-TiO2 nanotube heterojunction composites by thermal oxidation evaporation method. The results show that the heterojunction materials have better gas sensing properties and lower working temperature than pure CuO and TiO2. At the same time, the formation of heterojunction materials can enhance gas sensing properties toward H2 and reduce the gas sensing properties of VOCs and NO2, so as to improve the gas sensing selectivity of materials. In summary, the construction of TiO2-CuO heterojunction can effectively enhance the gas sensitivity of materials. What’s more, CuO is a narrow band gap semiconductor, and the combination of CuO and TiO2 can improve the photocatalytic performance. With the help of the photosensitive properties of CuO-TiO2, the photothermal combined excitation can be realized by means of light excitation, which can improve the carrier migration speed and improve its gas sensitivity. It is worth noting that MXene Ti3C2Tx may be in-situ transformed partly to an n-type TiO2 semiconductor via oxidation to form MXene-TiO2 heterojunction and it can still retain the original organ-like layered structure. This special structure can provide high-speed channels for electron transmission, so as to improve the gas sensitivity of the material (Wang et al., 2009). Thus, one-step in-situ oxidation could be applied to fabricate Ti3C2Tx-TiO2-CuO heterojunction to enhance the gas sensitivity of MXene. What’s more, changing the free carrier excitation source from traditional thermal excitation to light excitation is also an effect method to reduce operating temperature. The sensing materials could be irradiated by sufficient light energy, which make the valence band (VB) electron transition to obtain free electrons to achieve gas sensing response (Karaduman et al., 2014; Cui et al., 2016). In addition, light irradiation is conducive to boosting the response and improving the response/recovery features. Therefore, combining heterojunction construction with photo excitation will be a new breakthrough in the field of MXene gas sensing materials.
Herein, we demonstrate the achievement of room temperature gas sensing materials is in accordance with the formation of MXene Ti3C2Tx-TiO2-CuO heterojunction and the illumination of UV light. TiO2 were formed in-situ on the in-layer and surface of MXene to construct MXene-TiO2 junction, which was achieved through one-step in-situ oxidation. The existence of Ti3C2Tx-TiO2-CuO junctions was confirmed using field emission scanning electron microscopy (FESEM). The optimized MXene Ti3C2Tx-TiO2-CuO material demonstrated gas sensitivity superior to that of pristine Ti3C2Tx, TiO2-CuO and Ti3C2Tx-TiO2 toward NH3. The existence of UV light is beneficial to the gas sensitivity of Ti3C2Tx-TiO2-CuO materials.
2 Materials and methods
2.1 Methods of sample preparation
Synthesis of Ti3C2Tx MXene: 1 g lithium fluoride (LiF ≥ 99 %) powder was dissolved in 10 mL 9 mol/L hydrochloric acid (HCl) and stirred at room temperature for 30 min. 1 g of Ti3AlC2 MAX powder was slowly added into the above solution in the ice bath, and then the solution was sealed and transferred to oil bath at 35 °C for continuous stirring for 24 h. The obtained precipitate was repeatedly centrifuged and washed with deionized (DI) water at 3500 r/min for 5 min each time until the pH value of the solution was neutral. At last, the as-prepared samples were put into an oven for drying at 80 °C.
Synthesis of Ti3C2Tx-TiO2-CuO: Different mass (0.01 g, 0.05 g and 0.1 g) copper nitrate (Cu(NO3)2·6H2O) was dispersed uniformly in 1 mL DI water, respectively. 1 g Ti3C2Tx powder was put into the above dark blue solution, and the black uniform Ti3C2Tx /Cu(NO3)2·6H2O solution was obtained by magnetic stirring for 15 min at room temperature. Stand in dark at room temperature for 24 h to fully volatilize the liquid phase and obtain black precipitate. The obtained samples were then placed in an oven and dried at 70 °C for 8 h. Then put into a tube furnace and sintered at 500 °C for 30 min under the protection of argon (Ar, 99.99%) to obtain Ti3C2Tx-TiO2-CuO composites with different CuO contents.
Ti3C2Tx-TiO2 and TiO2-CuO composites were also prepared to investigate the effects of Ti3C2Tx and CuO additions on the material performances. The preparation details are as follows: 1 g of Ti3C2Tx powder was put into the Al2O3 ceramic crucible, and sintered at 200 °C for 60 min in air atmosphere to obtain black powdered Ti3C2Tx-TiO2 composites. 0.1 g Cu(NO3)2·6H2O and 1 g Ti3C2Tx powder were dispersed uniformly in 1 mL DI water by using magnetic stirring. Then stand in dark at room temperature for 24 h to fully volatilize the liquid phase and obtain black precipitate. After drying at 70 °C for 8 h in an oven, the obtained samples were put into a tuber furnace and sintered at 500 °C in air atmosphere for 30 min to obtain TiO2-CuO composite (Fig. 1). To better distinguish samples, Ti3C2Tx-TiO2-CuO composites with 0.01 g, 0.05 g and 0.1 g Cu(NO3)2·6H2O were labeled as TTC-1, TTC-2 and TTC-3, respectively. Ti3C2Tx-TiO2 and TiO2-CuO complex were labeled as TT and TC, respectively.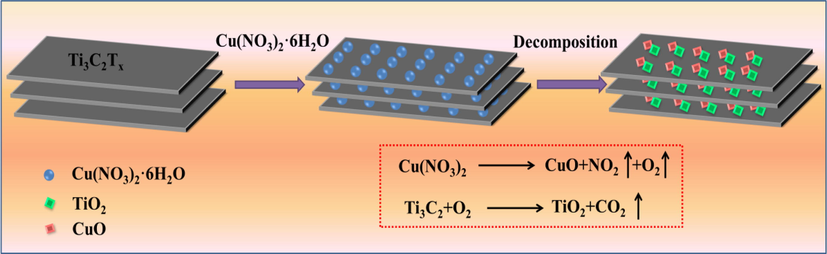
The schematic diagram of the manufacture of Ti3C2Tx-TiO2-CuO.
2.2 Samples characterization
The morphologies of samples were observed by a field emission scanning electron microscope (FESEM, JSM-5610L). The distribution of Ti and Cu and the location of TiO2 and CuO were confirmed by using Energy Disperse Spectroscopy (EDS). N2 adsorption/desorption (Autosorb-1-C) method was used to measure specific surface area at 77.4 K. X-ray diffraction (XRD, Rigaku D/max-2500) was applied to study the crystal phase in the 2θ range from 10° to 90°.
2.3 Measurement of gas sensing performance
In order to get the dynamic resistance of sensors, the prepared samples were printed on the chips composed with heating electrode and measuring electrode by using screen printing process. It is worth noting that the detail information about the chips is described in supply materials (Hou et al., 2021; Hou et al., 2021). Then the chips with different samples were put into an oven to evaporate and remove moisture at 70 °C for 40 min and annealed at 400 °C for 2 h under Ar atmosphere.
The prepared chips were mounted in a four-channel gas sensitivity detection equipment to measure gas sensing sensitivities. Target gases were diluted with air, and desired gas concentrations were obtained by controlling the flow rates in a home-made gas delivery system. The operating temperature were maintained range from 25 to 300 °C to investigate the gas sensitivity of samples at different temperatures. The resistance signal of samples was collected by data collection module (Agilent 34970A). The gas responses of samples were calculated based on the change of resistance, and the detail formula is shown as following:
where is resistance difference under air and target gas. equals to the resistance under air.
3 Results and discussion
In order to study the information of the prepared samples, XRD was used to analysis phase structure of all samples and the results are shown in Fig. 2. Fig. 2(a) shows the XRD patterns of Ti3AlC2 before and after hydrofluoric acid etching. It can be seen that there exist Ti3AlC2 peak in the spectrum before etching, and the characteristic peaks of Ti3C2Tx appeared at 18.2°, 27.5°, 35.9°, 41.7° and 60.4° in the spectrum after etching. At the same time, there was no residual peak of Ti3AlC2 in the spectrum, indicating that the Al layer in Ti3AlC2 was etched completely by hydrofluoric acid and the pristine Ti3C2Tx material was successfully prepared. Fig. 2(b) displays X-ray diffraction (XRD) patterns of each sample fabricated with one-step in-situ oxidation method. Ti3C2Tx, TiO2 (25.34°, 37.9°, 48.17°, 54.02° and 55.16°) can be observed in TTC-1, TTC-2 and TTC-3 spectra, indicating that TiO2 has been loaded on Ti3C2Tx, while Ti3C2Tx phase still exists in the three samples, demonstrating that Ti3C2Tx is not completely oxidized. CuO phase was not found in the diffraction pattern of all samples, which is related to the low content of CuO. XRD cannot identify the phase when the content is less than 5% (Amparo et al., 2008; Ciżmana, 2020). There is no diffraction peak of MXene, which demonstrates MXene has been transferred to TiO2 completely. In the diffraction pattern of TT sample, Ti3C2Tx and TiO2 diffraction peaks can also be observed, proving that Ti3C2Tx and TiO2 were successfully combined.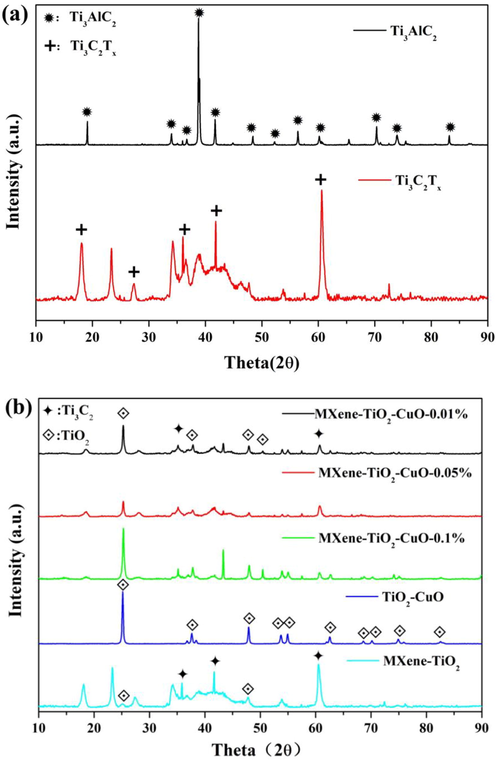
XRD spectra of Ti3AlC2, Ti3C2Tx, TTC-1、TTC-2, TTC-3, TT and TC composites.
In order to investigate Ti3C2Tx and TiO2-CuO compound state, FESEM was applied to check the microstructure of all samples. The results are shown in Fig. 3. It can be seen from Fig. 3(a)-3(b), 2D Ti3C2Tx prepared by etching Ti3AlC2 with fluoride salt (LiF, HCl) system has typical accordion-like morphology, which is composed of a large number of layers, and the layers are arranged in neat rows in the absence of impurities. Obvious white particles can also be observed on the surface of Ti3C2Tx composited with different contents of CuO (Fig. 3(c-e)). In order to trace the distribution of TiO2 and CuO, EDS was used to characterize and analyze the distribution of different elements. Taking TTC-1 for example, TTC-1 was characterized and analyzed by EDS, the results are shown in Fig. 4(a–d). C, O, F, Al, Ti and Cu elements appear at points 1 and 2. Cu account for a large proportion in point 1, and the Ti mainly concentrates at point 2 and 3, indicating that the scab attached to the surface of Ti3C2Tx is TiO2, and the white particle is CuO, which proves that the TiO2-CuO heterojunction is successfully combined with Ti3C2Tx. The adhesion of TiO2-CuO heterojunction on the surface of Ti3C2Tx is conducive to increasing the specific surface area of the material and improving the gas adsorption surface active sites, thereby improving the gas sensitivity of the material.
FESEM images of Ti3AlC2、Ti3C2Tx、TTC-1、TTC-2、TTC-3、TT and TC.
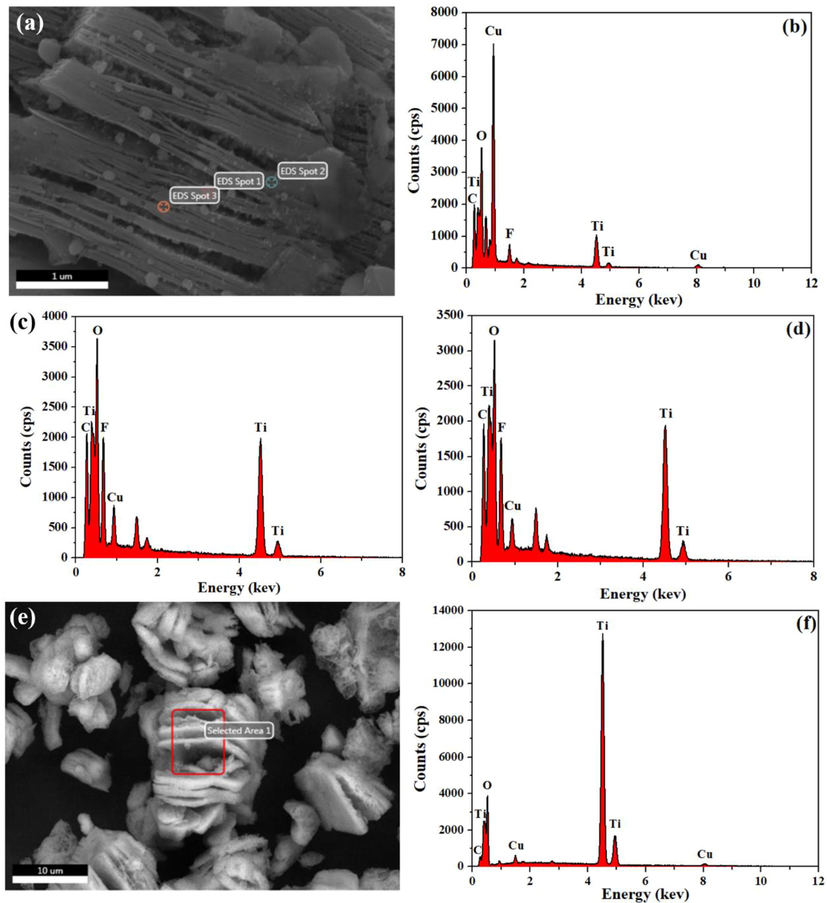
EDS spectra of TTC-1 (a, b, c, d) and EDS mapping of the marked domain of TC (e, f).
Fig. 4(e) exhibits the microstructure of the TT sample. After sintering at 200 °C, fine particles appear on the surface of Ti3C2Tx, and Ti3C2Tx still maintains a layered structure, indicating that low temperature sintering cannot cause damage to the structure of Ti3C2Tx material. Combined with the analysis results of XRD in Fig. 3, the Ti3C2Tx-TiO2 composite was successfully prepared. The TiO2-CuO sample prepared by high temperature (500° C) sintering in air atmosphere shows a rough stacked morphology (Fig. 3(g-h)), and the original smooth and angular morphology disappeared, combined the results of XRD in the diffraction peak of TC, indicating that Ti3C2Tx was completely oxidized to TiO2. EDS surface scanning characterization was carried out to verify the existence of CuO and the results are shown in Fig. 4(d-e). The scanned area contains C, O, Ti, Al and Cu elements, indicating the existence of CuO phase, but the content of Cu element is less than 5%, which is the reason why CuO phase cannot appear in the XRD TC sample pattern.
Table 1 shows the specific surface area of raw Ti3C2Tx and Ti3C2Tx based composites. It can be seen from the result that TTC composites has a higher specific surface area. And the value of TTC-1 sample is 80.9 m2 /g, which is 70.6 % higher than that of raw Ti3C2Tx sample. There is no obvious or even decreasing trend in specific surface area of TT sample, because Ti3C2Tx was in-situ oxidized into TiO2, which destroyed the original layered structure and reduced the specific surface area of the sample.
Sample
Ti3C2Tx
TTC-1
TTC-2
TTC-3
TT
TC
Specific surface areas (m2/g)
23.71
80.90
65
54.70
22.50
45.10
In order to study the effect of TiO2-CuO and CuO addition on the gas sensitivity of Ti3C2Tx materials, the gas sensing response values of different samples were tested. Firstly, the gas sensitivity values of all samples to 100 ppm NH3 under UV irradiation at different working temperatures were studied. The results are shown in Fig. 5(a). The gas sensitivity of different samples shows high dependence on working temperature. After the composite of Ti3C2Tx and Cu(NO3)2 with different contents (0.01 %, 0.05 % and 0.1 %), and the gas sensing properties of TTC-1, TTC-2 and TTC-3 are improved at all temperatures. Among them, TTC-1 shows the highest gas sensing response in the measurement at 100 °C when the temperature limited within 100 °C. This is because Ti3C2Tx was compounded with 0.01 % Cu(NO3)2, and the generated TiO2-CuO was attached to the surface of Ti3C2Tx material, which increased the specific surface area of the material and provided more active sites for gas adsorption, thereby significantly improving the gas sensing properties. With increasing the content of Cu(NO3)2, the decomposed O2 increases, and the TiO2-CuO particles grow up. The large particles could partially block the Ti3C2Tx layer, which is not conducive to electronic transmission, so that the gas sensitivity of the material is not significantly improved or even unfavorable. The gas response value of TTC-1 is 56.9% toward 100 ppm NH3 at room temperature, which is 89.8 % higher than that of raw Ti3C2Tx sample. At the same time, the optimal operating temperature of TTC-1 sample is 100 °C, and gas sensitivity response value is 91.1 %, indicating the addition of TiO2-CuO is beneficial for the improvement of gas performance of Ti3C2Tx. For TC sample, the gas sensing performance is lower than that of raw Ti3C2Tx at 25–100 °C. The target gas does not have enough energy to react with the adsorbed oxygen on the material surface at low temperature, and the carrier transition ability is insufficient, which makes the response value low. As the temperature increases, the gas response value of the TC sample shows higher than that of pristine Ti3C2Tx sample at 150–300 °C. At the moment, the target gas molecules become sufficiently active, and the carriers obtain sufficient energy to transition to the surface of the material for adsorption chemical reaction, resulting in a significant increase in the response value. The optimal gas sensing temperature of TC sample is 200 °C. It is worth mentioning that STTC-1 (100 °C) > STC (200 °C). This is because Ti3C2Tx material in TTC-1 is not completely oxidized and still retains the layered structure, it can provide high-speed migration channels for carriers in the process of gas sensing reaction, effectively avoids the previous carrier needs to cross the grain boundary barrier to complete the transmission, improving the gas sensitivity of the material. In order to investigate the influence of CuO addition on the gas sensitivity of materials, the gas sensing properties of TT samples were also tested. The results exhibits that the existence of CuO can increase the gas sensitivity of samples, which is because CuO could form TiO2-CuO heterojunction with TiO2. The construction of heterojunction is accompanied by the generation of energy level interleaving, which is beneficial to promoting the separation of holes and electrons, providing more carriers for the gas sensing reaction, thus increasing the gas sensing properties. Fig. 6 shows the gas sensing response of different samples to different concentrations of NH3 (10–1000 ppm) under UV irradiation at room temperature. It can be concluded from the result that the gas sensing values of all samples are positively correlated with the gas concentration, and the gas sensing response values of TTC-1 samples to NH3 at any gas concentration are higher than those of other samples. The gas sensing response to 10 ppm NH3 is 10.4, thus TTC-1 shows a good low detection limit.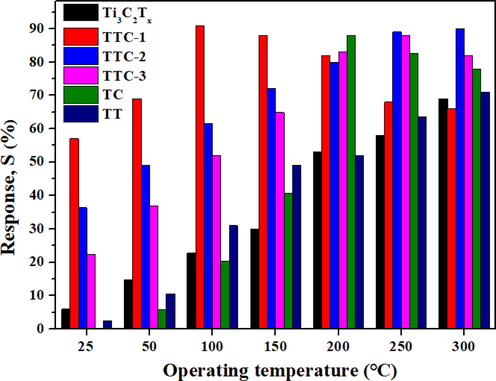
Gas response of different samples toward 100 ppm NH3 at different operating temperature under UV irradiation.
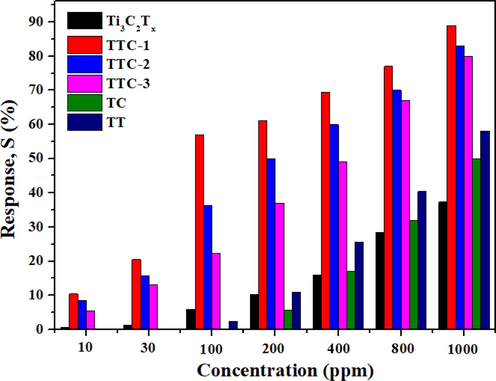
Gas response of different samples toward different concentration NH3 at room temperature under UV irradiation.
According to the comparative study on the gas sensing properties of all samples, it can be seen that TTC-1 is the optimal gas sensing material. As an important index for evaluating the gas sensing material, the gas sensing selectivity of TTC-1 was tested, and the gas sensing responses toward 100 ppm NH3, methanol, ethanol, toluene, NO2, CO and formaldehyde were detected, respectively, and the results are displayed in Fig. 7(a). The optimal response value S (%) of the sample towards different gases is as follows: Sammonia (56.9) > Sethanol (36) > SCO (26.9) > Snitrogen dioxide (25) > Smethanol (19.9) > Sformaldehyde (9.96) > Stoluene (9.1). The minimum response ratio (αmin = Sammonia/Sethanol) of TTC-1 sample to NH3 and other gases with the same concentration reached 1.9, and the maximum response ratio (αmax = Sammonia/Stoluene) is 6.3, indicating that TTC-1 sample has excellent gas sensitivity selectivity. Fig. 7(b-c) evaluates the gas sensing stability of TTC-1 sample. The gas sensing response of 100 ppm NH3 was detected for 10 times within 10 days, and 10 cycles were tested each time. It is found that the gas sensing response of the material fluctuated within the range of ± 2.5 %, and the device shows good cycling stability. It is worth mentioning that resistance of TTC-1 show reducint tendency, putting air back, the resistance increase, indicating that TTC-1 is an n-type semiconductor.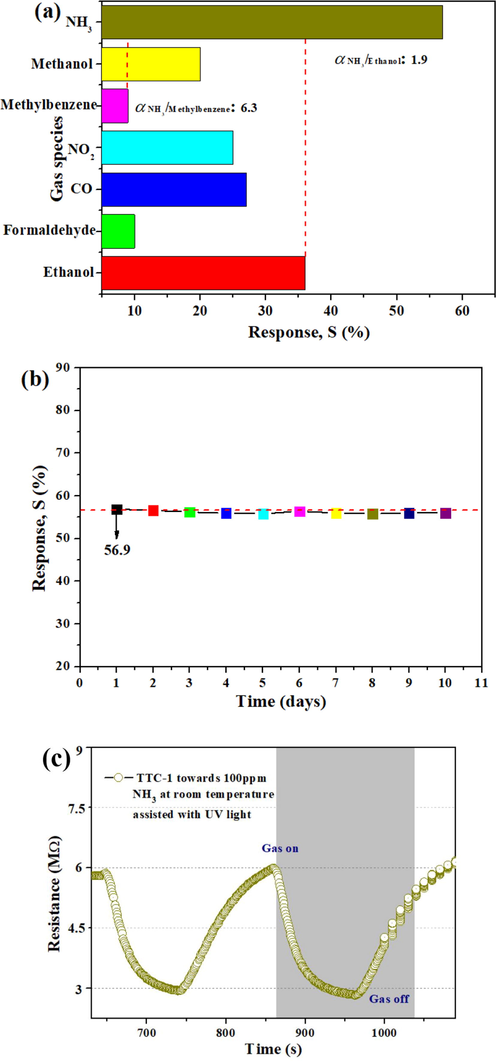
(a) Gas response of TTC-1 toward 100 ppm different species of gas at room temperature under UV irradiation. (b) The stability of TTC-1 at room temperature to 100 ppm NH3 under UV irradiation. (c) Dynamic response-recovery curve of TTC-1 towards NH3 at room temperature assisted with UV light.
Response time is another important parameter to evaluate the gas sensing materials. The response-recovery time of different samples toward 100 ppm NH3 with UV irradiation at room temperature were measured, the results are shown in Table S1. The response time of TTC-1 toward 100 ppm NH3 is 75 s and recovery time is 80 s, which exhibites excelltent response-recovery properties. TC also shows good response-recovery properties, proving that Ti3C2Tx layer structure plays an important role in carriers transportation.
In order to measure the influence of this work on enhancing the gas sensing performance of MXene materials, the TTC-1 samples obtained in this work were compared with the gas sensing response of 100 ppm NH3 reported in other literatures. The results are shown in Table 2. By comparison, the gas sensing response values of TTC-1 samples in this work shows more excellent performacne than those reported in other literatures. It can be concluded that the preparation of MXene composites with TiO2-CuO heterostructure is an outstanding method to enhance the gas sensitivity of Ti3C2Tx, and the Ti3C2Tx-TiO2-CuO has a good application prospect for the low temperature detection of NH3.
Material
Method
Temperature (°C)
Species/Concentration
Response
Ref
GO/WO3
Hydrothermal
200
NH3/100 ppm
1.17
47
2D Ti3C2Tx
Applied directly
Room temperature
NH3/100 ppm
21%
48
3-D frame Ti3C2Tx
Self-assembly
Room temperature
NH3/10 ppm
0.80%
49
Etched Ti3C2Tx
Alkaline etching
Room temperature
NH3/100 ppm
28.87%
50
Ti3C2Tx-TiO2-CuO
In-situ oxidation
Room temperature coupled with UV irradiation
NH3/100 ppm
56.90%
This work
PANI-CeO2
Gaussian process
50
NH3/10 ppm
1.5%
51
To further investigate the influence of UV light on the gas sensitivity of TTC-1 materials, the gas sensing properties of TTC-1 samples at room temperature with and without UV light were tested toward different concentrations of NH3, and the results are shown in Fig. 8. The resistance of TTC-1 toward different target gases with UV irradiation and without UV irradiation at room temperature are shown in Table S1 and S2. It can be observed that the gas sensitivities of TTC-1 samples at various gas concentrations (10–1000 ppm) under UV light are higher than those without UV light. The difference can be explained by the activation of UV to TTC-1 sample, which can promote the migration of photogenerated electrons from the VB to CB, making CB provide more free charges to react with adsorb oxygen, resulting in higher gas sensitivity of the material. Therefore, UV light is a necessary factor to enhance the gas sensitivity of TTC-1 sample.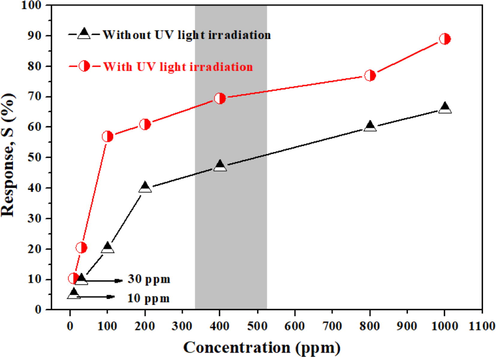
Gas response of TTC-1 toward different concentrations NH3 with and without UV light at room temperature.
According to analysizing the characterizations of all samples, it can be concluded that TTC-1 sample is an effective gas sensing material for monitoring NH3 at room temperature coupled with UV exposure. Thus, it is imperative to study the princle of gas sensitivity of TTC-1 sample. Generally, it is wildly accepted that the the gas sensing mechanism of materials can be explained by the resistance change which depends on the chemical reaction on the material surface and the capability of the adsorption and desorption of gas molecules (Wan et al., 2004). Fig. 9(a) exhibites the schematic diagram of the principle of gas sensing of TTC-1 without UV exposure. When TTC-1 is deposited in the air, the oxygen molecules are adsorbed on the surface of the material, obtaining electrons, decomposing into oxygen ions and forming an electron depletion layer, which increases the material resistance. When NH3 is introduced into the surface of TTC-1 material, NH3 reacts with oxygen anion, and the trapped electrons are released back to the CB of TTC-1 material. During the process, the electron depletion layer of the material get narrower, reducing the material resistance. Compared with the case in the absence of UV irradiation, extra photogenerated electrons will participate in each reaction under UV irradiation (Fig. 9(b)), which increases the resistance difference of the material before and after entering the NH3, thereby improves the gas sensitivity of the material.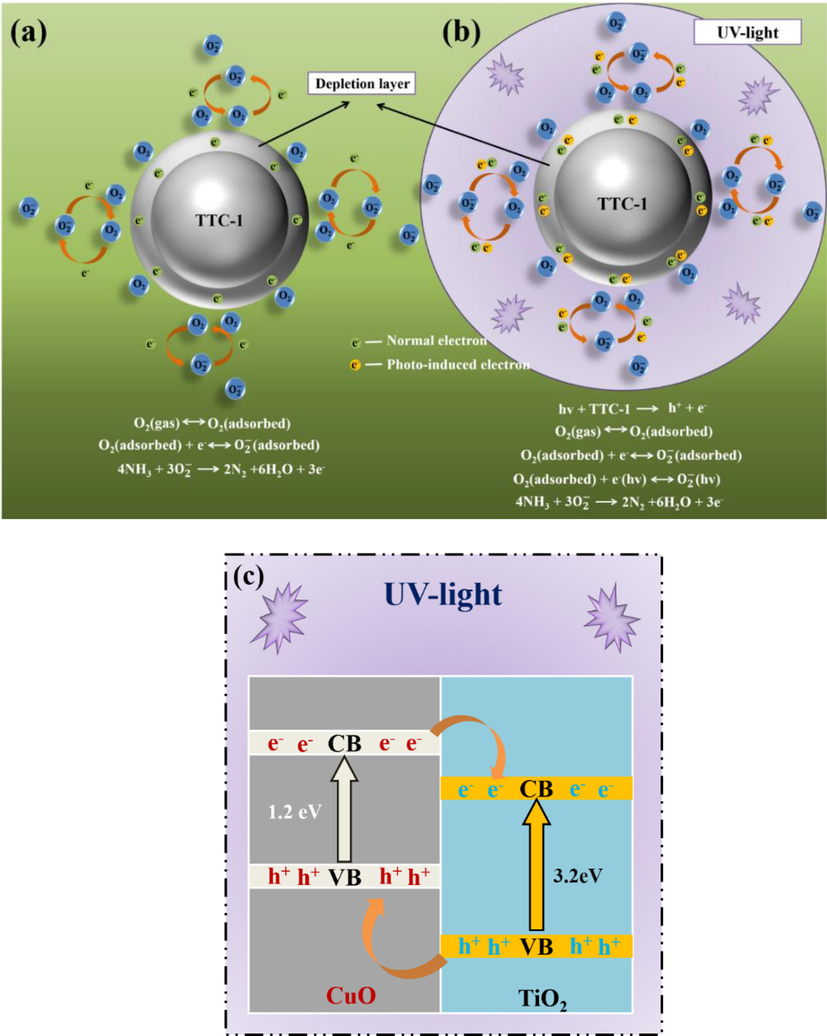
Schematic diagram of the possible gas sensing mechanism of TTC-1 under UV irradiation.
The construction of TiO2-CuO heterojunction is also the reason for enhancing the gas sensitivities of materials. The construction of heterojunction effectively reduces the rebinding rate of photogenerated electrons and holes, provides a large number of free charges for the gas sensing reaction, and ensures the effective gas sensitivity reaction. As shown in Fig. 9(c), CuO is a p-type semiconductor, and TiO2 is a n-type semiconductor. There is a carrier concentration difference at the interface between the two semiconductors. Due to the existence of carrier concentration difference, free electrons and holes migrate and diffuse to the p-zone (CuO) and the n-zone (TiO2) respectively, so that the electron-hole pairs are effectively separated, thereby enhancing the gas sensitivity of the material. It is worth noting that the TiO2-CuO heterojunction is attached to the surface of the Ti3C2Tx material, which increases the specific surface area of the material and provides a number of active sites for oxygen molecules to adsorb on the material surface. At the same time, TTC-1 sample retains the original layered structure of Ti3C2Tx material, which provides a fast channel for electron transport. What’ more, for TTC-1 sample, both for reducing gas and oxidizing gas, the increase of specific surface area, the establishment of heterojunction and the retention of MXene sheet structure are beneficial to carrier transport. Therefore, the above structural changes are also beneficial to the gas sensing response of oxidizing gas.
In summary, the combination of UV irradiation, the construction of TiO2-CuO heterojunction and the preservation of the layered structure of the original Ti3C2Tx material led to the TTC-1 sample having higher gas sensing response and lower working temperature, making it a gas sensing material with certain advantages.
4 Conclusions
Ti3C2Tx-TiO2-CuO composites with different CuO contents were prepared by one-step in-situ oxidation method. TiO2-CuO composites were also prepared for comparison. By testing the microstructure, phase analysis and gas sensing properties of the samples, the following conclusions are obtained:
TiO2-CuO heterojunction is attached to the surface of Ti3C2Tx material, which enhances the specific surface area and provides a large number of active sites for oxygen adsorption on the material surface. Ti3C2Tx-TiO2-CuO composites still maintain the original layered structure of Ti3C2Tx, which provides a fast channel for electronic transmission.
The Ti3C2Tx-TiO2-CuO composite exhibits n-type semiconductor properties. The composite with 0.01% CuO exhibits the best gas sensing response, and the response value towards 100 ppm NH3 at room temperature can reach 56.9%, which is 89.8% higher than that of pure Ti3C2Tx. At the optimal working temperature (100° C), the response value to 100 ppm NH3 can reach 91.1 %, and the material has excellent gas sensing selectivity and stability.
The improvement of the gas sensitivity of the material is attributed to the excitation of the photogenerated electrons and holes of the material by the irradiation of UV light, which provides more free charges for the gas sensitivity reaction and makes the resistance difference of the material larger before and after the target gas is introduced. The construction of TiO2-CuO heterojunction effectively reduces the electron-hole recombination rate, provides a large number of electrons for the gas-sensitive reaction, and ensures the effective conduct of gas-sensitive reaction. The TiO2-CuO heterojunction is attached to the surface of Ti3C2Tx material, which increases the specific surface area of the material and provides more active sites for oxygen molecules to adsorb on the surface of the material. At the same time, TTC-1 sample retains the original layered structure of Ti3C2Tx material, which provides a fast channel for electron transport.
Acknowledgement
This research was funded by National Natural Science Foundation of China, No. 51864028, NSAF No. U2030207; Yunnan Fundamental Research Projects, No. 202201BE070001-018; Yunnan Province Funds for Distinguished Young Scientists, No. 2019FJ005; Science Research Foundation of Yunnan Provincial Education Department, No. 2022J0441.
References
- Synthesis and characterization of MXene/BiCr2O4 nanocomposite with excellent electrochemical properties. J Mater Res Technol.. 2021;15:2007-2015.
- [CrossRef] [Google Scholar]
- Improved gas sensing performance of p-copper oxide thin film/n-TiO2 nanotubes heterostructure. J. Alloys Comp.. 2018;749:221-228.
- [CrossRef] [Google Scholar]
- The sensing behaviour of metal oxides (ZnO, CuO and Sm2O3) doped-SnO2 for detection of low concentrations of chlorinated volatile organic compounds. Sens. Actuators, B.. 2013;B181:637-643.
- [CrossRef] [Google Scholar]
- Application of carbon nanotubes in extraction and chromatographic analysis: A review. Arab. J. Chem.. 2019;12:633-651.
- [CrossRef] [Google Scholar]
- A novel approach for calculating starch crystallinity and its correlation with double helix content: A combined XRD and NMR study. Biopolymers. 2008;89:761-768.
- [CrossRef] [Google Scholar]
- A comparative study on UV light activated porous TiO2 and ZnO film sensors for gas sensing at room temperature. Ceram. Int.. 2012;38:503-509.
- [CrossRef] [Google Scholar]
- Ti3C2Tx-Based Three-Dimensional Hydrogel by a Graphene Oxide-Assisted Self-Convergence Process for Enhanced Photoredox Catalysis. ACS Nano. 2019;13:295-304.
- [CrossRef] [Google Scholar]
- Effect of the iron content on the structure and electrical properties of sodium borosilicate glasses: XRD, TEM, Mössbauer, FTIR and DIS spectroscopy study. J Non-Cryst. Solids.. 2020;531:119847
- [CrossRef] [Google Scholar]
- UV-light illumination room temperature HCHO gas-sensing mechanism of ZnO with different nanostructures. Sens. Actuators, B.. 2016;227:220-226.
- [CrossRef] [Google Scholar]
- A novel electrode hybrid of N-Ti3C2Tx/C/CuS fabricated using ZIF-67 as an intermediate derivation for superhigh electrochemical properties of supercapacitors. J Mater Res Technol.. 2022;19:3507-3520.
- [CrossRef] [Google Scholar]
- MXene modulated SnO2 gas sensor for ultra-responsive room-temperature detection of NO2. Sens. Actuators, B.. 2022;357:131427
- [CrossRef] [Google Scholar]
- Efficient exfoliation of g-C3N4 and NO2 sensing behavior of graphene/g-C3N4 nanocomposite. Sens. Actuators, B.. 2017;248:940-948.
- [CrossRef] [Google Scholar]
- Room temperature gas sensing under UV light irradiation for Ti3C2Tx MXene derived lamellar TiO2-C/g-C3N4 composites. Appl. Surf. Sci.. 2021;535:147666
- [CrossRef] [Google Scholar]
- Improvement of Gas Sensing Property for Two-Dimensional Ti3C2Tx treated with Oxygen Plasma by Microwave Energy Excitation. Ceram. Int.. 2021;47:7728-7737.
- [CrossRef] [Google Scholar]
- Highly sensitive and ultra-fast responsive ammonia gas sensor based on 2D ZnO nanoflakes. Materials Sci. Energy Tech.. 2020;3:91-96.
- [CrossRef] [Google Scholar]
- UV light activated gas sensor for NO2 detection. Mater. Sci. Semicond. Process.. 2014;28:43-47.
- [CrossRef] [Google Scholar]
- Variation Resistance of different operation temperature of NO2 and NH3 gases for the Ag-doped SiC gas sensor. J. Phys: Conference Series.. 2021;1973:012140
- [CrossRef] [Google Scholar]
- Metallic Ti3C2Tx MXene Gas Sensors with Ultrahigh Signal-to-Noise Ratio. ACS Nano. 2018;12:986-993.
- [Google Scholar]
- Synthesis of Co-doped SnO2 nanofibers and their enhanced gas-sensing properties. Sens. Actuators, B.. 2016;236:425-432.
- [CrossRef] [Google Scholar]
- Nitrogen-doped TiO2 nanotube array films with enhanced photocatalytic activity under various light sources. J. Hazard. Mater.. 2010;184:855-863.
- [CrossRef] [Google Scholar]
- Single-layer MoSe2 based NH3 gas sensor. Appl. Phys. Lett.. 2014;105:233103
- [CrossRef] [Google Scholar]
- One-Step Fabrication of Nanocrystalline Nanonetwork SnO2 Gas Sensors by Integrated Multilaser Processing. Adv. Mater Technol-US.. 2020;5:2000281.
- [CrossRef] [Google Scholar]
- Preparation and Application of 2D MXene-Based Gas Sensors: A Review. Chemosensors.. 2021;9:225.
- [CrossRef] [Google Scholar]
- Mesoporous WS2-Decorated Cellulose Nanofiber-Templated CuO Heterostructures for High-Performance Chemiresistive Hydrogen Sulfide Sensors. Anal. Chem.. 2022;94(46):16160-16170.
- [Google Scholar]
- Flower-like hierarchical structures consisting of porous single-crystalline ZnO nanosheets and their gas sensing properties to volatile organic compounds (VOCs) J. Alloys Comp.. 2015;626:124-130.
- [CrossRef] [Google Scholar]
- Nanoscale metal oxide-based heterojunctions for gas sensing: a review. Sens. Actuators, B.. 2014;204:250-272.
- [CrossRef] [Google Scholar]
- Enhanced NH3 sensing performance at ppb level derived from Ti3C2Tx-supported ZnTi-LDHs nanocomposite with similar metal-semiconductor heterostructure. Sens. Actuators, B.. 2022;352:131077
- [CrossRef] [Google Scholar]
- Metal clusters activated SnO2 thin film for low level detection of NH3 gas. Sens. Actuators, B.. 2014;194:410-418.
- [CrossRef] [Google Scholar]
- High performance of the rare earth (Er, Gd & Pr) doped MoO3 thin films for advanced applications towards ammonia gas sensing. J Mater Res Technol.. 2022;20:4556-4565.
- [CrossRef] [Google Scholar]
- W18O49/Ti3C2Tx Mxene nanocomposites for highly sensitive acetone gas sensor with low detection limit. Sens. Actuators, B.. 2020;304:127274
- [CrossRef] [Google Scholar]
- Fabrication and ethanol sensing characteristics of Zn nanowire gas sensors. Appl. Phys. Lett.. 2004;84:3654-3656.
- [CrossRef] [Google Scholar]
- Metal-containing carbon nitride compounds: a new functional organic–metal hybrid material. Adv. Mater.. 2009;21:1609-1612.
- [CrossRef] [Google Scholar]
- Hydrogen-Treated TiO2 Nanowire Arrays for Photoelectrochemical Water Splitting. Nano. Lett.. 2011;11:3026-3033.
- [CrossRef] [Google Scholar]
- Conductometric room temperature ammonia sensors based on titanium dioxide nanoparticles decorated thin black phosphorus nanosheets. Sens. Actuators B. 2021;349:130770
- [CrossRef] [Google Scholar]
- A Simple Graphene NH3 Gas Sensor via Laser Direct Writing. Sensors (Basel).. 2018;18:4405.
- [CrossRef] [Google Scholar]
- Ti3C2Tx MXene as a Janus cocatalyst for concurrent promoted photoactivity and inhibited photocorrosion. Appl. Catal. B.. 2018;237:43-49.
- [CrossRef] [Google Scholar]
- Hydrothermal synthesis of one-dimensional α-MoO3 nanomaterials and its unique sensing mechanism for ethanol. Arab. J. Chem.. 2022;15:104083
- [CrossRef] [Google Scholar]
- Binder-free Ti3C2Tx MXene electrode film for supercapacitor produced by electrophoretic deposition method. Chem. Eng. J.. 2017;317:1026-1036.
- [CrossRef] [Google Scholar]
- A flexible VOCs sensor based on a 3D MXene framework with a high sensing performance. J. Mater. Chem. A.. 2018;6:18116-18124.
- [CrossRef] [Google Scholar]
- Edge-enriched MoS2 nanosheets modified porous nanosheet-assembled hierarchical In2O3 microflowers for room temperature detection of NO2 with ultrahigh sensitivity and selectivity. J. Hazrad. Mater.. 2022;15:128836
- [CrossRef] [Google Scholar]
- UV light activation of TiO2 for sensing formaldehyde: How to be sensitive, recovering fast, and humidity less sensitive. Sens. Actuators, B.. 2014;202:964-970.
- [CrossRef] [Google Scholar]
- Enhanced Blocking Effect: A New Strategy to Improve the NO2 Sensing Performance of Ti3C2Tx by γ-Poly(l-glutamic acid) Modification. ACS Sens.. 2021;6:2858-2867.
- [Google Scholar]
- A review on Ti3C2Tx-based nanomaterials: synthesis and applications in gas and humidity sensors. Rare Met.. 2020;40(6):1459-1476.
- [Google Scholar]
- Zhou, Y., Wang, Y. H., Wang, Y. J., Yu, H. C., Zhang, R. J., Li, J., Zang, Z. G., Li, X. 2021. MXene Ti3C2Tx-Derived Nitrogen-Functionalized Heterophase TiO2 Homojunctions for Room-Temperature Trace Ammonia Gas Sensing. ACS Appl. Mater. Interfaces. 13, 47:5 6485-56497. https://doi.org/10.1021/acsami.1c17429.
Appendix A
Supplementary data
Supplementary data to this article can be found online at https://doi.org/10.1016/j.arabjc.2023.104808.
Appendix A
Supplementary data
The following are the Supplementary data to this article:Supplementary data 1
Supplementary data 1







