Translate this page into:
Phase behavior-microstructure-crystallization kinetics correlations in semi-crystalline/amorphous PCL/SAN mixtures filled with nanosilica
⁎Corresponding authors. f.hemmati@ut.ac.ir (Farkhondeh Hemmati), H.Khonakdar@ippi.ac.ir (Hossein Ali Khonakdar)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Abstract
In this study, the effects of poly(ε-caprolactone) (PCL) molecular weight and the type and presence of nanosilica on the non-isothermal crystallization behavior of PCL in the PCL/poly(styrene-co-acrylonitrile) (SAN)/nanosilica systems have been quantitatively investigated. The PCL/SAN system have been chosen as an ideal model system because of the special phase behavior of the blends, which include a lower-critical solution temperature (LCST) phase diagram over a virtual upper-critical solution temperature. By this choose, the great importance of phase separation, mutual phase dissolution and preferential nanoparticle migration to one of the polymeric phases on the crystallization behavior during a commercial production process has been highlighted. The addition of both hydrophilic (Si) and hydrophobic (SiR) silica nanoparticles to virgin PCLs with different molecular weights retards the crystallization process. However, the presence of these nanoparticles in PCL/SAN blends impacts the PCL crystallization kinetics in opposite ways, depending on the nanosilica type, PCL molecular weight and melt cooling rate. The reasons for the observed opposite trends are the changes in the LCST-type phase diagram position by altering the PCL molecular weight and nanosilica type, the selective migration of nanoparticles as well as the dispersion state of nanofillers. The presence of both Si and SiR nanosilicas increases the crystallization activation energy, respectively, by 4.2% and 2.4% for the PCL/SAN blend conatining the PCL with lower molecular weight. While the addition of both Si (11%) and SiR (6.7%) reduces this energy for the blend conatining the PCL with higher molecular weight.
Keywords
Phase behavior
Poly(ε-caprolactone)
Nanocomposite
Blends
Crystallization
1 Introduction
During the past decades, blending of polymers as an available and popular economical method has been attracted a lot of attention to modify the properties of polymeric products (Higgins et al., 2005; Ghafouri et al., 2022). For partial miscible polymer blends containing a semi-crystalline phase and an amorphous phase, it is possible to study the phase behavior and performance by following the crystallization kinetics of the semi-crystalline phase (Moeinifar et al., 2020). The final properties of polymer blends depend on the properties of their pure components, miscibility and multiphase morphology (Salkhi Khasraghi et al., 2022; Svoboda et al., 1999). Polymer blends including a semi-crystalline poly(ε-caprolactone) (PCL) component and an amorphous phase have been widely studied in recent years (Madbouly and Ougizawa, 2004; Hemmati et al., 2014; Mohtaramzadeh et al., 2020). PCL is an aliphatic and hydrophobic semi-crystalline polyester that is commonly used in biomedical applications such as tissue engineering and drug delivery (Maghoul et al., 2023; Khadivi et al., 2023).
PCL in the molten state is miscible with various polymers, such as poly(styrene-co-acrylonitrile) (SAN) and poly(vinyl methyl ether), at the molecular level (Mohamed and Yusoh, 2016; Woodruff and Hutmacher, 2010). Based on the researches, PCL with SAN shows miscible behavior when the copolymer contains 8 to 28 % by weight of acrylonitrile comonomer (Moeinifar et al., 2020; Schulze et al., 1993). In polymer blends containing semi-crystalline/amorphous regions, such as PCL/SAN blends, the reduction in the kinetics of spherulites growth is attributed to chain movements, the free energy of nucleation and the competition between the progressive growth rate of the crystal surface and the penetration of amorphous phase chains into the interlayer regions (Luyten et al., 1997).
PCL/SAN blends have lower critical solution temperature (LCST) phase behavior at high temperatures and at lower temperatures, these mixtures show an apparent upper critical solution temperature (UCST) phase diagram below the melting point of PCL. The virtual UCST phase diagram is due to the crystallization process of PCL chains in the presence of amorphous SAN macromolecules (Schulze et al., 1993; Svoboda et al., 1994). LCST phase diagram, spinodal decomposition kinetics, dissolution of phases in each other and PCL phase crystallization in PCL/SAN blends have been studied by many scientists (Salkhi Khasraghi et al., 2022; Su and Lin, 2004; Naziri et al., 2019). Since the isothermal crystallization kinetics of PCL melt in PCL/SAN blends is greatly affected by the presence of amorphous SAN chains, Madbouly et al. investigated the isothermal crystallization process of PCL/SAN (80/20) blend containing 27.5 % by weight of acrylonitrile. The authors found that the half-crystallization time of PCL in the blends changed by annealing at temperatures above the LCST phase diagram. It was found that the crystallization kinetics of PCL in the blend was greatly enhanced by liquid–liquid phase separation (Salkhi Khasraghi et al., 2022).
Polymer blends that exhibit phase separation may not always perform optimally in service, therefore, the development of suitable interfacial additives or compatibilizers has always been one of the major topics in the research of polymer blends (Svoboda et al., 2008). In addition to the positive effects of nanoparticles on the final properties of polymer blends, the introduction of nanoparticles into blends as compatibilizers has been widely reported as well (Madbouly et al., 2006; Azizli et al., 2020; Mdletshe et al., 2015). The final properties and performance of a composite system containing nanofiller depend on the localization and dispersion state of nanofiller (Mdletshe et al., 2015; Crosby and Lee, 2007; Qian et al., 2016; Naziri et al., 2020).
Numerous studies have been dedicated to PCL and its mixtures aimed at increasing the rate of biodegradation and crystallization (Hosseiny et al., 2020; Arshian et al., 2023). Composite membranes of the PCL/chitosan (CS) blends were prepared by applying the lyophilization method by Zhang et al. at different blend ratios (Fakhri et al., 2021). Their findings demonstrated the importance of the blend composition on the morphology and mechanical performance of the membranes. The blend with 10/90 w/w CS/PCL composition had the optimum properties suggested for tissue engineering applications. At this composition, the crystallinity degree and membrane porosity increased by 29.97 % and 85.61 %, respectively (Fakhri et al., 2021). The influences of poly(propylene carbonate) (PPC) on the thermal, mechanical and rheological properties of the PCL/PPC blends were studied by Li et al. in 2022 (Albertsson and Varma, 2002). It was reported, in these immiscible blends, that the presence of PPC phase accelerated the PCL crystallization process and reduced the PCL crystallinity degree. As a result, considerable enhancements in the PCL stiffness, strength and melt elasticity were obtained by the melt-blending of PCL with PPC (Albertsson and Varma, 2002).
In a recently published work, the effects of the PCL and pre-stretch rate (of the extrusion-casting-thermal stretching process) on the mechanical performance and crystallinity of the poly(glycolic acid)/PCL blends have been investigated. Their findings show that high tensile strength and elongation-at-break were achieved at higher molecular orientation and lower crystallinity by applying higher pre-stretching temperature (Zhang et al., 2021). In another research, the effects of PCL presence and content on the properties of the air-jet spun poly(3-hydroxybutyrate-co-3-hydroxyvalerate) (PHBV)/PCL fibrous scaffolds were evaluated. The results confirm the beneficial role of PCL on the crystallinity, thermal stability and mechanical properties of the PCL/PHBV blend scaffolds (Li et al., 2022). Mathematical modeling was applied, in another recently published study, for optimizing the mechanical properties of the melt-blown films of the ternary blends based on poly(lactic acid) (PLA)/PCL/cellulose acetate butyrate (CAB). The improved miscibility of the components caused the attainment of the films with the largest strain at break at the composition of 55–85 wt% PLA, 5–20 wt% PCL and 10–25 wt% CAB (Xu et al., 2023). All of these works clarify the importance of processing, phase miscibility and crystallinity in controlling the blend properties.
Isothermal and non-isothermal crystallization kinetics of pure PCL and its blends have been also reported in the presence of nanoparticles (Madbouly and Ougizawa, 2004; Kalva et al., 2023; Tuancharoensri et al., 2023). Jing Qian et al. investigated the morphology and crystallization behavior of PCL/SAN in a (20/80) blend containing 30 % by weight of AN comonomer in SAN in the presence of hydrophobic and hydrophilic nanosilica. It was found that hydrophilic nanosilica had a significant effect on the morphology of PCL/SAN blend compared to hydrophobic nanosilica. The nucleation ability of hydrophobic nanosilica in PCL showed its limitations because the effect of nanosilica on crystal growth is always greater than its heterogeneous nucleation effect (Mdletshe et al., 2015).
In our previous works, the effects of spherical nanosilica particles on the LCST phase diagram of PCL/SAN blends and the non-isothermal crystallization kinetics of PCL in the blends were studied. In this research, it was observed that the thermodynamic equilibrium LCST phase diagram of PCL/SAN blends unexpectedly shift to lower temperatures by adding hydrophilic nanosilica. The migration of nanosilica particles to SAN-rich regions, especially at lower cooling rates of rheometric tests, has led to unfavorable PCL/SAN enthalpy interactions and much larger dynamic asymmetry of polymer components (Maghoul et al., 2023). In another work, the non-isothermal crystallization of PCL chains in PCL/SAN blends was investigated in the presence of two types of hydrophilic and hydrophobic nanosilica. Hydrophobic silica nanoparticles significantly accelerated the crystallization process of PCL in PCL/SAN (80/20) blend, while the hydrophilic silica nanoparticles contributed only slightly to the PCL crystallization process. The reason for this is the selective migration of hydrophilic silica nanoparticles to the phase-separated regions of SAN during the non-isothermal DSC test (Mohtaramzadeh et al., 2020).
Studying the kinetic phenomenon of the non-isothermal crystallization of PCL in PCL/SAN blends in the presence of nanoparticles is very complicated and challenging. The complexity originates from the presence of the LCST phase diagram of PCL/SAN, selective migration of nanoparticles to one of two phases after phase separation, effects of nanoparticles on phase separation boundary and kinetics of mutual dissolution of two polymers during the test. Although several studies have been conducted on the crystallization of PCL in the PCL/SAN blends in the absence and presence of nanoparticles, the effects of nanoparticles on the LCST phase behavior of PCL/SAN and the migration of nanoparticles during the non-isothermal DSC test have rarely been considered. Therefore, in this research, in continuation of the group's previous works, the crystallization kinetics and crystallization activation energy of PCL in PCL/SAN blends in the presence of hydrophilic and hydrophobic silica nanoparticles are quantitatively studied. In addition to the type of nanoparticles, in this research, the effects of PCL molecular weight on the non-isothermal kinetics of the PCL crystallization and the phenomena that occur during the non-isothermal DSC test are fully investigated. The main objective of this study is getting an insight into the effects of phenomena involved in the cooling process of a filled multi-component system (with LCST phase behavior) as a model system on the crystallization kinetics. The reason for this selection is the similarity of a non-isothermal crystallization of a polymer in the presence of amorphous polymer and nanoparticles with what is done during the cooling process of the industrial production of the multiphase polymeric products.
2 Experimental
2.1 Materials
Two grades of PCL, the first one 704,105 grade with Mn = 45 kg.mol−1 and melting point ( ) of 56 °C and the second one 440,744 grade with Mn = 80 kg.mol−1 and of 60 °C, were purchased from Sigma-Aldrich Co. (Darmstadt, Germany). These two grades were assigned as PCL-1 and PCL-2, respectively. Poly(styrene-co-acrylonitrile) copolymer, SAN, with the grade of 182,850 (Mw = 165 kg mol−1 and 25 wt% content of acrylonitrile (AN)) was also supplied from Sigma-Aldrich Co (Darmstadt, Germany). Two grades of fumed silica with a 12 nm primary particle size, hydrophilic fumed silica (Aerosil® 200 with a 200 m2/g specific surface area) and hydrophobic fumed silica (Aerosil® R104 with a 150 m2/g specific surface area), were purchased from Evonik Co. (Essen, Germany). These nanosilica grades were represented as Si (hydrophilic) and SiR (hydrophobic) nanoparticles, respectively.
2.2 Sample preparation
The binary blends of PCL/SAN and the nanosilica-filled systems, with various SAN contents (0, 10, 20, 30, 40, 50, and 100 wt%) and two different molecular weights ( ) of PCL, were melt-compounded using a micro-compounder, DACA Instruments (Santa Barbara, California) at a rotor speed of 100 rpm and a temperature of 160 °C for 10 min. The nanosilica loadings (Si or SiR) were 3 phr in the PCL/SAN/silica nanocomposites. The components were fed to the compounder simultaneously. All samples were then compression molded using a lab-scale hot press (Davenport 25 tons) to sheets with an average thickness of 1 mm. The fabricated samples are referred to PxSy-z blends as well as PxSy-z-3Si and PxSy-z-3SiR nanocomposites, where x, y, and z represent the PCL (wt%) and SAN (wt%) weight percentages, and the applied PCL grade in the sample, respectively. In the same way, the virgin PCL grades and PCL/silica nanocomposites are represented as PCL-z and PCL-z-3Si or PCL-z-3SiR nanocomposites.
2.3 Characterizations
The effect of silica nanoparticles and the of PCL on the PCL’s crystallization behavior in semicrystalline PCL/SAN blends were assessed using differential scanning calorimeter (DSC, TA Instruments (Trios)) through a non-isothermal study. The specimens were first heated from −100 to 130 °C under the nitrogen environment (with a flux of 50 ml/min) at a heating rate of 10 °C/min to erase the thermal history. Then, the samples were cooled at various cooling rates of 2, 5, 10, and 20 °C/min from 130 to −100 °C. The indium and tin standard samples were applied to calibrate the temperature and heat flow. The time of attaining 50 % of final crystallinity (the crystallization half-time ( )) was calculated to assess the crystallization kinetics.
The dispersion state of nanoparticles and the bulk of some samples were studied using transmission electron microscope (TEM) applying a Philips CM-30 and field emission scanning electron microscope (FESEM) using Carl Zeiss Group Ultra Plus (Jena, Germany). The ultramicrotome (Leica Biosystems©, Germany) was applied to cut samples at −180 °C. The samples were then sputtered with platinum at a 3 nm rough thickness and examined.
3 Results and discussion
3.1 Results
In this study, non-isothermal DSC studies were conducted on virgin PCL, PCL/SAN blends, PCL/Silica and PCL/SAN/Silica nanocomposites at cooling rates of 2, 5, 10, and 20 °C/min. The thermograms obtained are depicted in Fig. 1. For all samples, the exothermic crystallization peak shifts to lower temperatures, intensifies and broadens with increasing cooling rate. Furthermore, at a constant cooling rate, by adding the SAN component to PCL as well as the increase of
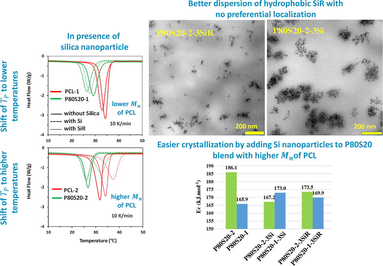
of PCL, the crystallization peak is shifted to a lower temperature, flattened, and the area under the peak decreases. The addition of silica nanoparticles affects the crystallization peak of the samples in different ways depending on the nature of the nanoparticles (hydrophilic or hydrophobic) as well as Mn of PCL. In PCL-1-based nanocomposites and P80S20-1-based nanocomposites, the incorporation of nanoparticles broadens the crystallization peak and shifts it to lower temperatures. The shift of the crystallization peak is more dominant with the incorporation of Si nanoparticle. On the contrary, in PCL-2-based nanocomposites, the incorporation of nanoparticles broadens the crystallization peak and shifts it to higher temperatures. The width of the crystallization peak increases with the addition of SiR nanoparticles and is transferred to higher temperatures. In nanocomposites based on P80S20-2, a trend similar to that of PCL-2 nanocomposites can be seen. However, with the addition of Si to P80S20-2, a slight shift to a lower temperature occurs. Similar to P80S20-1 and P80S20-2 samples, hydrophilic and hydrophobic nanosilica fillers affect the PCL crystallization peak of PCL/SAN blends in other compositions. The DSC thermograms of PCL-1-based PCL/SAN blends and nanocomposites are represented in supporting information in Figure S1.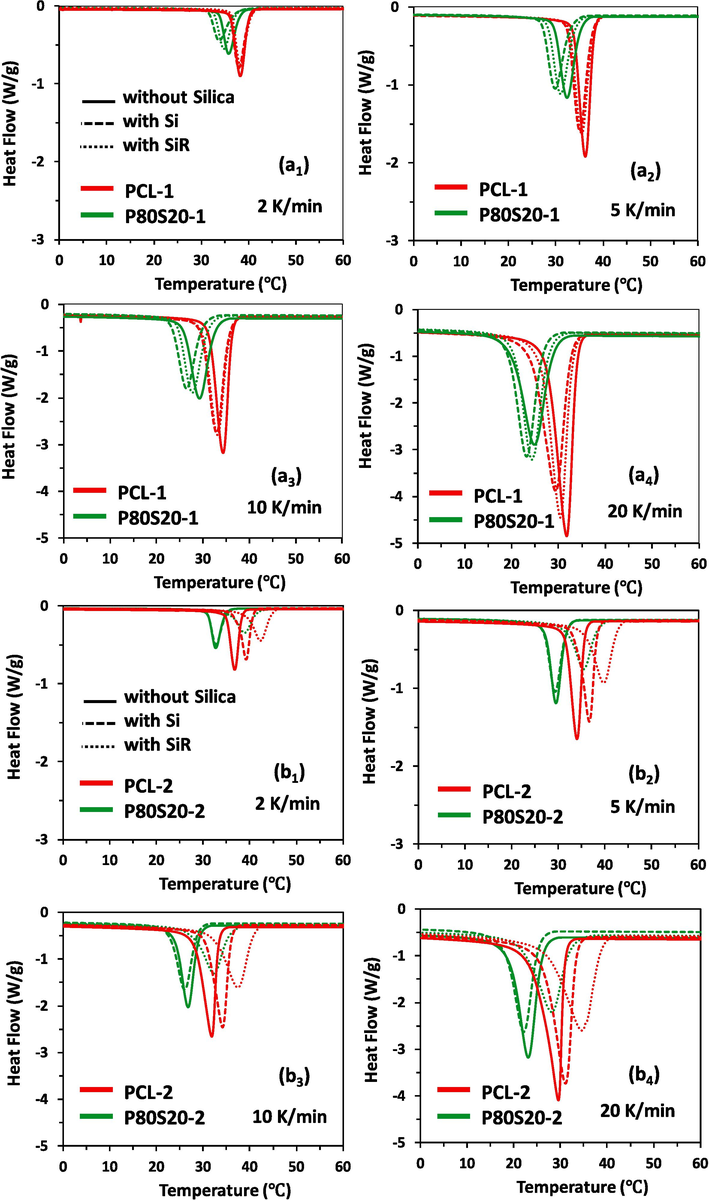
Heat flow curves for the non-isothermal crystallization of virgin PCL-1, P80S20-1, and their nanocomposites filled with Si and SiR at different cooling rates: (a1) 2 K/min, (a2) 5 K/min, (a3) 10 K/min, and (a4) 20 K/min, as well as PCL-2 and P80S20-2 and their nanocomposites filled with Si and SiR at different cooling rates: (b1) 2 K/min, (b2) 5 K/min, (b3) 10 K/min, and (b4) 20 K/min.
For a better understanding of non-isothermal crystallization kinetics, various models can be applied, such as the Avrami, Ozawa, Jeziorny, and Mo theories (Dhanvijay et al., 2012; Achla, 2017; Avrami, 1940). These models have been frequently applied for studying the non-isothermal crystallization kinetics of PCL-based blends (Kalva et al., 2023; Ozawa, 1971; Jeziorny, 1978; Choi and Kwak, 2004).
3.1.1 Avrami and Jeziorny models
The Avrami equation is the most frequent model utilized for the isothermal crystallization analysis. Modified Avrami is used to examine the crystallization process in this research, according to the following equations:
here n,
, and
are respectively the Avrami exponent, the crystallization rate constant, and crystallization half-time. The n value is attributed to the crystal growth dimensions and the nucleation mechanism. Fig. 3 shows
vs.
curves for samples. The slope and intercept of curves were used to calculate the values of n and
, respectively. The curves appeared at three stages with different slopes, which were considered as regime I (
,
, regime II (
,
, and regime III (
,
). The Avrami parameters including n,
, and
for regime I-III are gathered in Table 1. Regime I and II belong to the primary crystallization of PCL phase, while regime III shows the secondary crystallization stage. As can be seen in Table 1, the n value is nearly constant throughout each regime. A change in n value will indicate a change in the crystallization mechanism. The transition between the primary and secondary crystallization processes is therefore detectable by the n value. It should be mentioned that the values of calculated n,
, and
by considering the whole of
vs.
curves as a single stage are also assessed and reported in Table 2. All of the theoretically calculated values for the crystallization half-time (Eq. (3)) were quite similar to the corresponding experimental values that were directly calculated from the
vs. time curves.
Sample
n1
Zt1
Zc1
1(1/2)
n2
Zt2
Zc2
2(1/2)
n3
Zt3
Zc3
3(1/2)
PCL-1
2
1.093
1.87E-03
0.043
2.25E + 02
5.152
0.008
0.092
2.354
1.291
0.557
0.746
1.185
5
0.957
1.47E-03
0.271
6.21E + 02
4.606
0.686
0.927
1.002
1.134
1.834
1.129
0.424
10
0.901
1.46E-03
0.520
9.35E + 02
4.237
6.266
1.201
0.595
1.202
3.488
1.133
0.261
20
1.167
4.33E-03
0.762
7.74E + 01
4.221
42.609
1.206
0.377
1.496
6.437
1.098
0.225
PCL-1-3Si
2
1.313
2.90E-03
0.054
6.48E + 01
4.725
0.006
0.080
2.700
1.422
0.414
0.644
1.436
5
0.858
7.32E-04
0.236
2.94E + 03
4.559
0.228
0.744
1.283
1.577
1.090
1.017
0.750
10
0.988
1.50E-03
0.522
4.97E + 02
4.510
2.276
1.086
0.768
1.605
2.588
1.100
0.440
20
0.692
5.24E-04
0.685
3.26E + 04
4.164
15.076
1.145
0.477
1.864
5.374
1.088
0.333
PCL-1-3SiR
2
1.117
1.83E-03
0.043
2.03E + 02
4.938
0.004
0.063
2.838
1.497
0.360
0.600
1.550
5
0.909
1.05E-03
0.254
1.27E + 03
4.889
0.173
0.704
1.329
1.561
1.077
1.015
0.754
10
0.686
5.05E-04
0.468
3.74E + 04
4.391
2.549
1.098
0.743
1.447
2.688
1.104
0.392
20
0.817
8.10E-04
0.701
3.87E + 03
4.045
22.423
1.168
0.423
1.702
6.302
1.096
0.273
P80S20-1
2
0.995
1.48E-03
0.039
4.80E + 02
5.147
0.002
0.041
3.210
1.262
0.390
0.624
1.578
5
1.107
2.91E-03
0.311
1.40E + 02
5.123
0.058
0.566
1.624
1.339
0.932
0.986
0.802
10
1.177
4.33E-03
0.580
7.48E + 01
5.045
0.813
0.980
0.969
1.317
1.906
0.980
0.969
20
1.147
8.15E-03
0.786
4.82E + 01
4.698
8.177
1.111
0.591
1.634
3.999
1.072
0.342
P80S20-1-3Si
2
1.375
2.16E-03
0.047
6.64E + 01
4.841
0.001
0.026
4.176
1.801
0.116
0.341
2.694
5
1.235
3.38E-03
0.320
7.44E + 01
4.803
0.043
0.534
1.781
1.575
0.683
0.927
1.009
10
1.254
5.64E-03
0.596
4.64E + 01
4.532
0.814
0.980
0.965
1.489
1.851
1.063
0.517
20
1.267
1.04E-02
0.796
2.74E + 01
4.311
11.800
1.131
0.518
1.585
4.707
1.081
0.299
P80S20-1-3SiR
2
1.284
3.18E-03
0.056
6.62E + 01
5.026
0.002
0.046
3.178
1.386
0.336
0.580
1.684
5
1.228
4.35E-03
0.337
6.22E + 01
4.689
0.083
0.608
1.571
1.344
0.936
0.987
0.800
10
1.119
4.23E-03
0.579
9.54E + 01
4.485
0.968
0.997
0.928
1.216
2.005
1.072
0.417
20
1.262
8.96E-03
0.790
3.13E + 01
4.468
11.421
1.129
0.534
1.413
4.542
1.079
0.264
PCL-2
2
0.966
2.57E-03
0.051
3.29E + 02
5.409
0.006
0.081
2.372
1.344
0.449
0.670
1.381
5
0.928
2.83E-03
0.309
3.75E + 02
4.310
0.745
0.943
0.983
1.410
1.482
1.082
0.583
10
1.285
9.20E-03
0.626
2.89E + 01
4.096
6.044
1.197
0.589
1.467
2.985
1.056
0.370
20
1.590
3.86E-02
0.850
6.15E + 00
4.054
39.838
1.202
0.368
1.496
5.133
1.085
0.262
PCL-2-3Si
2
1.155
2.23E-03
0.047
1.44E + 02
4.941
0.014
0.117
2.210
1.413
0.389
0.624
1.504
5
0.968
1.40E-03
0.269
6.07E + 02
4.673
0.472
0.861
1.086
1.571
1.139
1.026
0.729
10
0.929
9.05E-04
0.496
1.27E + 03
4.360
5.439
1.185
0.623
1.488
2.668
1.050
0.404
20
1.567
2.42E-02
0.830
8.52E + 00
4.079
28.379
1.182
0.402
1.633
4.962
1.083
0.300
PCL-2-3SiR
2
1.323
2.99E-03
0.055
6.14E + 01
4.544
0.003
0.059
3.204
1.564
0.166
0.408
2.493
5
0.940
1.58E-03
0.275
6.49E + 02
4.168
0.146
0.680
1.453
1.799
0.517
0.876
1.177
10
0.770
1.09E-03
0.505
4.38E + 03
4.059
1.406
1.035
0.840
1.852
1.407
1.017
0.682
20
0.630
5.15E-04
0.685
9.25E + 04
3.919
8.810
1.115
0.523
1.953
3.414
1.063
0.442
P80S20-2
2
1.177
5.26E-03
0.073
6.32E + 01
5.839
0.001
0.027
3.251
1.373
0.312
0.558
1.791
5
1.075
2.87E-03
0.310
1.65E + 02
5.449
0.124
0.658
1.372
1.465
0.898
0.979
0.838
10
1.024
2.64E-03
0.552
2.30E + 02
4.587
3.128
1.121
0.720
1.101
2.384
1.091
0.326
20
1.432
1.59E-02
0.813
1.40E + 01
4.405
20.123
1.162
0.465
1.405
4.193
1.074
0.278
P80S20-2-3Si
2
1.277
3.26E-03
0.057
6.65E + 01
5.370
0.001
0.036
3.223
1.412
0.286
0.535
1.870
5
1.063
2.14E-03
0.292
2.31E + 02
4.881
0.153
0.687
1.362
1.436
0.935
0.987
0.812
10
1.180
4.68E-03
0.585
6.92E + 01
4.720
2.268
1.085
0.778
1.362
2.093
1.077
0.444
20
1.505
1.83E-02
0.819
1.12E + 01
4.245
18.043
1.156
0.464
1.416
4.216
1.075
0.279
P80S20-2-3SiR
2
1.219
4.70E-03
0.069
6.00E + 01
4.596
0.001
0.036
3.911
1.806
0.093
0.305
3.038
5
1.125
8.24E-03
0.383
5.13E + 01
4.164
0.076
0.597
1.701
1.698
0.436
0.847
1.315
10
1.657
2.60E-02
0.694
7.25E + 00
4.080
0.732
0.969
0.987
1.715
1.174
1.016
0.736
20
1.003
5.13E-03
0.768
1.33E + 02
3.911
4.037
1.072
0.637
1.894
2.892
1.055
0.470
Sample
n
Zt
Zc
(1/2)
R2
PCL-1
2
3.069
0.029
0.172
2.797
0.932
5
3.189
0.399
0.832
1.189
0.950
10
3.222
2.351
1.089
0.685
0.962
20
3.413
11.798
1.131
0.436
0.973
PCL-1-3Si
2
3.011
0.024
0.157
3.035
0.943
5
3.244
0.206
0.729
1.454
0.952
10
3.389
1.178
1.017
0.855
0.956
20
3.404
6.328
1.097
0.522
0.971
PCL-1-3SiR
2
3.084
0.020
0.141
3.170
0.931
5
3.314
0.180
0.710
1.502
0.939
10
3.353
1.267
1.024
0.835
0.958
20
3.366
9.108
1.117
0.465
0.976
P80S20-1
2
3.023
0.012
0.110
3.810
0.928
5
3.166
0.099
0.630
1.847
0.929
10
3.304
0.476
0.928
1.120
0.948
20
3.328
2.852
1.054
0.654
0.959
P80S20-1-3Si
2
2.973
0.008
0.088
4.529
0.935
5
3.156
0.083
0.608
1.961
0.939
10
3.200
0.566
0.945
1.066
0.952
20
3.322
4.159
1.074
0.583
0.963
P80S20-1-3SiR
2
2.899
0.017
0.129
3.624
0.936
5
3.029
0.120
0.654
1.785
0.943
10
3.168
0.567
0.945
1.065
0.955
20
3.244
3.632
1.067
0.600
0.959
PCL-2
2
2.936
0.029
0.169
2.963
0.925
5
2.998
0.464
0.858
1.143
0.951
10
3.106
2.395
1.091
0.671
0.965
20
3.058
8.470
1.113
0.441
0.971
PCL-2-3Si
2
2.785
0.035
0.187
2.923
0.924
5
3.215
0.306
0.789
1.290
0.951
10
3.318
2.002
1.072
0.726
0.959
20
3.167
7.045
1.103
0.481
0.972
PCL-2-3SiR
2
2.775
0.016
0.127
3.873
0.945
5
2.967
0.144
0.678
1.700
0.957
10
3.092
0.798
0.978
0.956
0.960
20
3.145
3.525
1.065
0.596
0.972
P80S20-2
2
2.889
0.016
0.126
3.697
0.920
5
3.290
0.157
0.690
1.572
0.910
10
3.382
1.469
1.039
0.801
0.947
20
3.145
4.255
1.075
0.562
0.962
P80S20-2-3Si
2
2.904
0.015
0.122
3.755
0.930
5
3.159
0.152
0.686
1.616
0.942
10
3.155
0.891
0.989
0.923
0.949
20
3.088
4.090
1.073
0.563
0.968
P80S20-2-3SiR
2
2.663
0.014
0.117
4.368
0.939
5
2.716
0.111
0.645
1.961
0.955
10
2.910
0.499
0.933
1.120
0.967
20
3.068
2.146
1.039
0.692
0.963
For correction of the
parameter in the Avrami equation to include the non-isothermal conditions, the Jeziorny equation is also used which is illustrated in Eq. (4):
The values of samples are reported in Table 1 and Table 2 respectively for a three-stage and one-stage crystallization.
According to Table 1, the n value rapidly increased throughout the early phases of the main process over stage 1 to stage 2. The Avrami exponent of samples, in the first stage, , and third stage, , respectively is mostly close to and in the range between 1 and 2. However, the second stage’s Avrami exponent, , is between 3.92 and 5.53 range, indicating three-dimensional (3D) crystallite growth, as well as heterogeneous and athermal nucleation, for the primary crystallization stage. There was a reduction in values by increasing the cooling rate, while the values of and increased. As well from Table 1, the incorporation of Si and SiR nanoparticles roughly leads to a reduction of as well as n (Avrami exponent of single-stage crystallization) compared to PCL and PCL80/SAN20 blend samples. P80S20 blends show a higher value of n2 compared to PCL. However, increasing the molecular weight of PCL mostly leads to a decrease in n values.
Additionally, and (crystallization rate constant of single-stage crystallization) and consequently change differently with the incorporation of Si and SiR nanoparticles in pure PCL and P80S20 blends. With the incorporation of Si and SiR nanoparticles into PCL-1 and PCL-2, there was a reduction in and values and an increment of at whole cooling rates, only there was an increase of and a decrease of in PCL-2-3Si compared to PCL-2 at just one cooling rate of 2 K/min. A reduction in and values means a decrease in the overall rate of bulk crystallization, in other words overall a slower crystallization rate, by the addition of nanoparticles to pure PCL samples. Indeed, the heat flow curves for non-isothermal crystallization of PCL samples become broader and less sharp through the incorporation of nanoparticles.
In the P80S20-1 blends, the addition of Si leads to a reduction in at two lower cooling rates (2 and 5 K/min) and an increment of at two higher cooling rates (10 and 20 K/min). While in all cooling rates with the addition of SiR, an increase in was obtained. Interestingly, P80S20-2 blends render an adverse trend. The incorporation of Si increases at two lower cooling rates (2 and 5 K/min), while decreasing it at higher cooling rates. Besides, the incorporation of SiR decreases the of P80S20-2 roughly in all studied cooling rates. The reduction of values observed in pure PCL samples due to the addition of nanoparticles indicates the negative effect of nanoparticles on the crystallization kinetics of PCL. However, the reducing effect of SiR incorporation on in PCL is more pronounced compared to Si, especialy at higher cooling rates.
Crystallization of the samples starts at lower temperatures through the increment of
of PCL, as well as the addition of SAN in the P8S20 blends (see third column of Fig. 2). Interestingly, the incorporation of Si and SiR nanoparticles affects the start temperature of crystallization of samples in a different trend. In PCL-1 and P80S20-1-based nanocomposite, the start temperature shifts to the lower temperature in the presence of both hydrophilic and hydrophobic silicas. Although, in the higher
PCL-2-based nanocomposites, the start temperature moves to higher temperatures.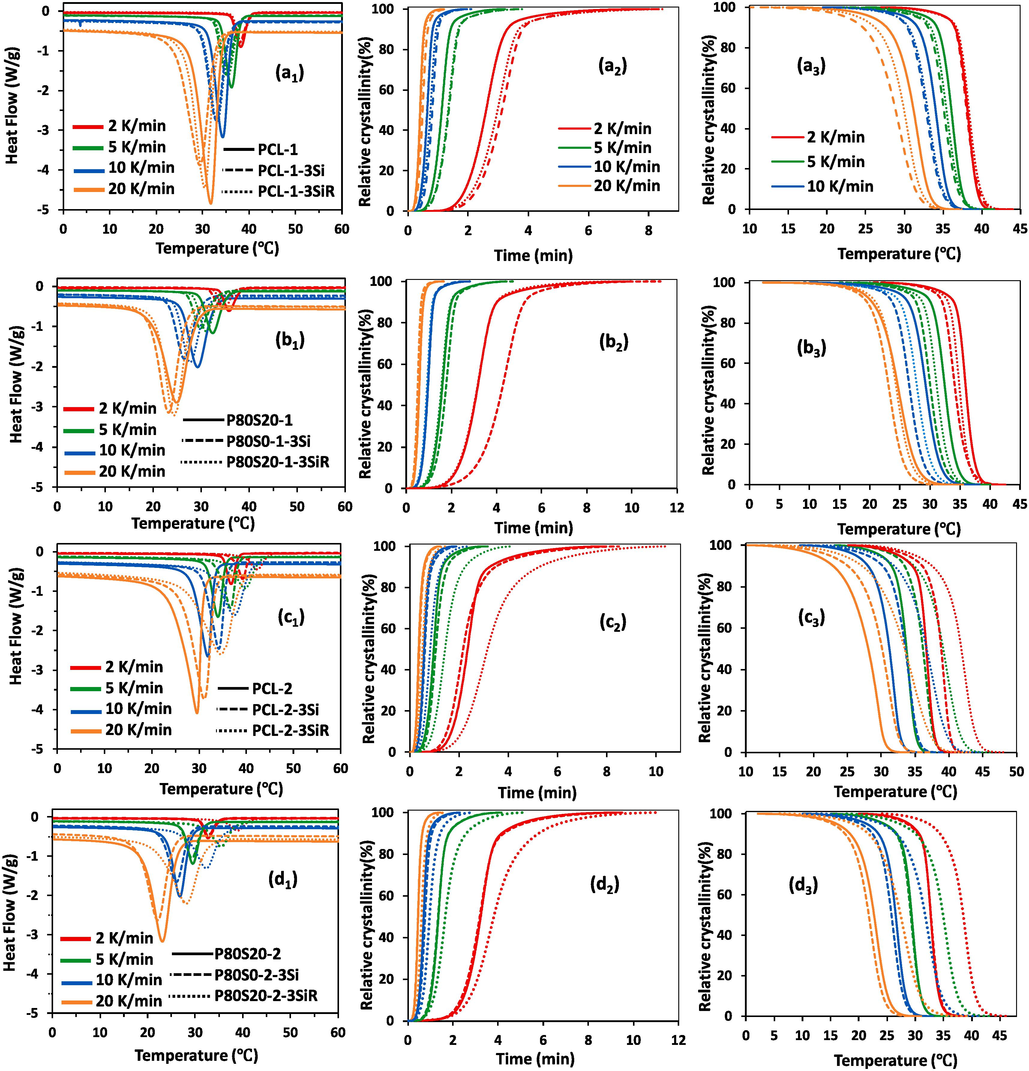
Heat flow curves and relative degree of crystallinity, X(T), versus time and temperature at different cooling rates for the non-isothermal crystallization of (a1, a2, a3) virgin PCL-1, PCL-1-3Si, and PCL-1-3SiR, (b1, b2, b3) P80S20-1, P80S20-1-3Si, and P80S20-1-3SiR, (c1, c2, c3) virgin PCL-2, PCL-2-3Si, and PCL-2-3SiR, (d1, d2, d3) P80S20-2, P80S20-2-3Si, and P80S20-2-3SiR.
![Plots of Log [-ln(1 - X(T))] versus Log (t) used in the Avrami method for (a1, a2, a3) virgin PCL-1, PCL-1-3Si, and PCL-1-3SiR, (b1, b2, b3) P80S20-1, P80S20-1-3Si, and P80S20-1-3SiR, (c1, c2, c3) virgin PCL-2, PCL-2-3Si, and PCL-2-3SiR, (d1, d2, d3) P80S20-2, P80S20-2-3Si, and P80S20-2-3SiR at different cooling rates.](/content/184/2024/17/2/img/10.1016_j.arabjc.2023.105522-fig4.png)
Plots of Log [-ln(1 - X(T))] versus Log (t) used in the Avrami method for (a1, a2, a3) virgin PCL-1, PCL-1-3Si, and PCL-1-3SiR, (b1, b2, b3) P80S20-1, P80S20-1-3Si, and P80S20-1-3SiR, (c1, c2, c3) virgin PCL-2, PCL-2-3Si, and PCL-2-3SiR, (d1, d2, d3) P80S20-2, P80S20-2-3Si, and P80S20-2-3SiR at different cooling rates.
Our recent research shows that the increase of SAN concentration in the PCL/SAN blends slows the chain crystallization of PCL and results in significantly lower and higher (Mohtaramzadeh et al., 2020). This observation is related to a diluting effect of the SAN component on the crystallization of PCL. In agreement with our previous finding, according to Tables 1 and 2, the P80S20 blends present lower compared to PCL samples. In addition, the decreases by increasing the of PCL, as well as incorporating SAN into the P8S20 blends.
A notable feature of Table 1 is that the hydrophobic SiR in virgin PCL and P80S20 samples affects and more considerably in comparison with hydrophilic Si nanoparticle. In other words, SiR decreases more profoundly compared to Si and a slower crystallization rate is attained in SiR-filled nanocomposite. Although, in some of the samples such as P80S20-1, the addition of SiR accelerates the crystallization of PCL. Also, the presence of SiR has comparatively more profound impact on the higher -based PCL-2 and P80S20-2 crystallization kinetics. The adverse changes in the PCL crystallization kinetics of P80S20-1 compared to P80S20-2 with the incorporation of SiR could be concluded.
3.1.2 Ozawa model
The Ozawa model derived from the Avrami theory is commonly used to explain the dynamics of the non-isothermal crystallization of polymers. The substitution of the cooling rate,
, for the time variable, t, is the fundamental modification applied to the Avrami model in the Ozawa extension. In the Ozawa model, by the assumption of a constant cooling rate for the molten polymer, the Avrami equation is modified to deal with non-isothermal behavior (Achla, 2017). The Eqs. (5) and (6) are applied to calculate the crystallinity degree,
, at a constant temperature of T according to Ozawa model, as follows: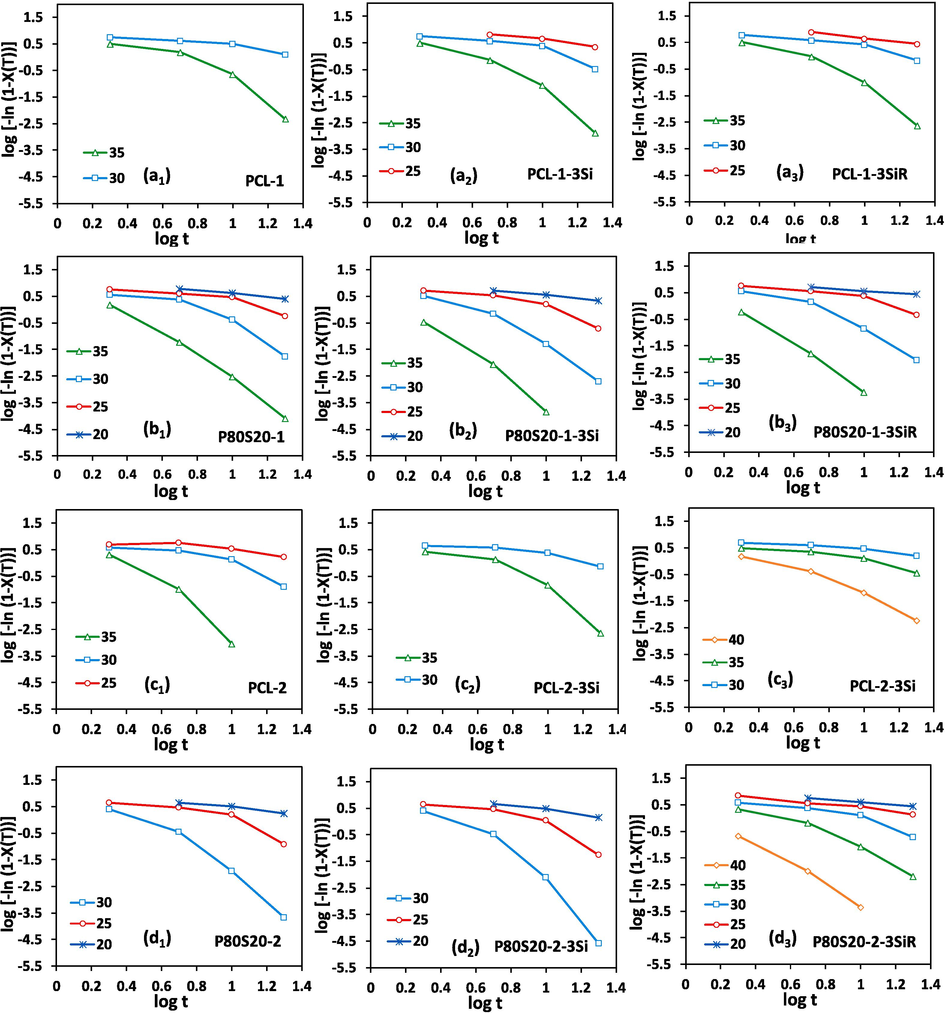
Ozawa analysis of the non-isothermal crystallization data for Plots of log(ϕ) versus log(t) for the non-isothermal crystallization of (a1, a2, a3) virgin PCL-1, PCL-1-3Si, and PCL-1-3SiR, (b1, b2, b3) P80S20-1, P80S20-1-3Si, and P80S20-1-3SiR, (c1, c2, c3) virgin PCL-2, PCL-2-3Si, and PCL-2-3SiR, (d1, d2, d3) P80S20-2, P80S20-2-3Si, and P80S20-2-3SiR.
Sample
T(°C)
m
R2
Sample
T(°C)
M
R2
PCL-1
40
PCL-2
40
35
2.729
49.24
0.862
35
4.721
82.41
0.957
30
0.633
10.37
0.876
30
1.399
16.77
0.792
25
25
0.472
8.77
0.717
20
20
PCL-1-3Si
40
PCL-2-3Si
40
35
3.3226
68.55
0.916
35
2.998
55.82
0.857
30
1.1442
18.37
0.806
30
0.745
9.55
0.820
25
0.8098
26.58
0.973
25
20
20
PCL-1-3Si
40
PCL-2-3SiR
40
2.406
11.75
0.960
35
3.070
55.32
0.914
35
0.894
7.25
0.871
30
0.830
11.50
0.827
30
0.485
7.69
0.921
25
0.546
14.52
0.967
25
20
20
P80S20-1
40
P80S20-2
40
35
4.233
18.09
0.992
35
30
2.289
38.62
0.849
30
4.087
92.00
0.953
25
0.920
14.17
0.800
25
1.461
19.77
0.789
20
0.639
17.20
0.994
20
0.686
14.27
0.953
P80S20-1-3Si
40
P80S20-2-3Si
40
35
4.797
11.80
0.987
35
30
3.224
56.30
0.953
30
4.944
237.85
0.924
25
1.386
20.99
0.849
25
1.794
27.73
0.802
20
0.626
14.42
0.982
20
0.869
19.80
0.962
P80S20-1-3SiR
40
P80S20-2-3SiR
40
3.806
3.34
0.991
35
4.317
16.90
0.996
35
2.509
19.59
0.946
30
2.604
39.83
0.930
30
1.240
12.72
0.852
25
1.033
15.26
0.851
25
0.682
11.40
0.976
20
0.466
10.97
0.996
20
0.523
13.22
1.000
3.1.3 Mo model
The other theory for studying the non-isothermal crystallization of polymers is the modified approach proposed by Mo et al. This approach results from a combination of the Avrami and Ozawa equations. Accordingly, the crystallinity degree in this theory is related to the cooling rate (
) and the crystallization time (t) or temperature (
) (Choolaei et al., 2017). The
is related to the t through Eq. (7) at a specific degree of crystallinity, as follows: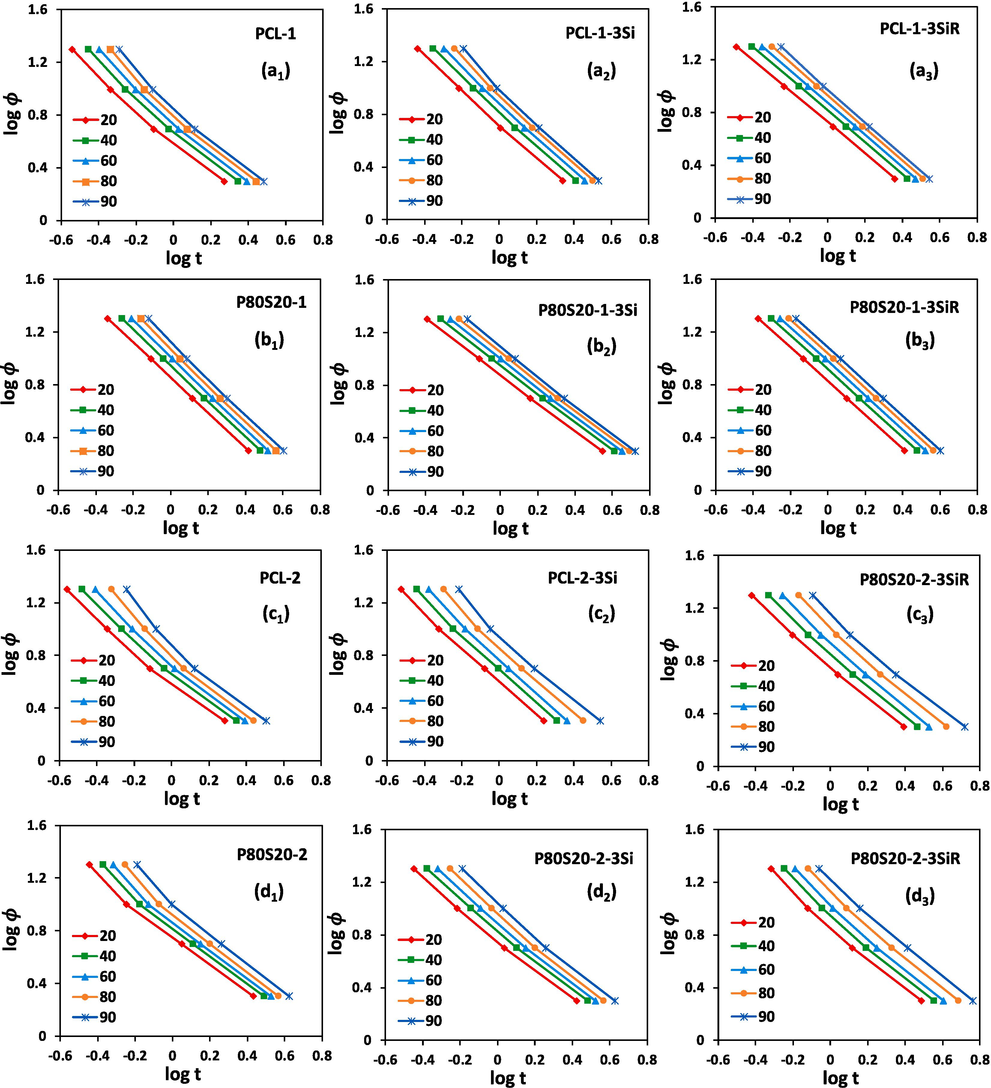
Plots of log(ϕ) versus log(t) for the non-isothermal crystallization of (a1, a2, a3) virgin PCL-1, PCL-1-3Si, and PCL-1-3SiR, (b1, b2, b3) P80S20-1, P80S20-1-3Si, and P80S20-1-3SiR, (c1, c2, c3) virgin PCL-2, PCL-2-3Si, and PCL-2-3SiR, (d1, d2, d3) P80S20-2, P80S20-2-3Si, and P80S20-2-3SiR.
Sample
X(T)
α
F(
Min R2
Sample
X(T)
α
F(
Min R2
PCL-1
20
1.219
4.111
0.990
PCL-2
20
1.180
4.056
0.981
40
1.240
5.036
40
1.203
4.929
60
1.254
5.859
60
1.240
5.731
80
1.265
6.783
80
1.302
6.883
90
1.274
7.702
90
1.316
8.433
PCL-1-3Si
20
1.287
5.327
0.996
PCL-2-3Si
20
1.298
4.023
0.990
40
1.307
6.702
40
1.323
5.004
60
1.320
7.800
60
1.339
5.99
80
1.344
9.049
80
1.321
7.57
90
1.366
10.198
90
1.299
9.486
PCL-1-3SiR
20
1.178
5.327
0.999
PCL-2-3SiR
20
1.224
5.849
0.995
40
1.205
6.548
40
1.25
7.439
60
1.217
7.489
60
1.267
9.055
80
1.235
8.543
80
1.256
11.585
90
1.259
9.543
90
1.216
14.282
P80S20-1
20
1.332
7.19
0.999
P80S20-2
20
1.115
5.861
0.992
40
1.355
8.88
40
1.14
6.941
60
1.372
10.309
60
1.161
7.827
80
1.384
11.899
80
1.194
9.087
90
1.386
13.452
90
1.207
10.892
P80S20-1-3Si
20
1.069
7.586
0.999
P80S20-2-3Si
20
1.146
5.851
0.996
40
1.079
8.98
40
1.163
6.987
60
1.089
10.065
60
1.186
7.998
80
1.101
11.272
80
1.219
9.391
90
1.115
12.405
90
1.22
11.161
P80S20-1-3SiR
20
1.277
6.733
0.982
P80S20-2-3SiR
20
1.231
7.554
0.994
40
1.288
8.264
40
1.243
9.29
60
1.293
9.484
60
1.249
10.929
80
1.296
10.842
80
1.234
13.477
90
1.295
12.109
90
1.209
16.252
According to plots in Fig. 5, a good agreement between the Mo analysis and experimental data can be concluded. The R2 values higher than 0.98 result from fitting the Mo model to the data for all samples. Furthermore, there is an increase in the value of by the increasing relative crystallinity, which refers to the attainment of higher crystallinity degrees at higher at a constant time, t. Almost constant values for , independent of , are reported (Choolaei et al., 2017). In addition, the values presented in Table 4 alter very little with the increase of . A reduced value was expected through the acceleration of the crystallization process (Jana and Cho, 2010).
A higher value is resulted for PCL-1 compared to those of PCL-2 samples in all measured , demonstrating that the higher molecular weight PCL-2 can crystallize at a faster rate, which is in line with the result of modified Avrami. In addition, F(TC) increased with the incorporation of amorphous SAN in the P80S20 blends, which is consistent with modified Avrami and points to slower non-isothermal crystallization kinetics of PCL in P80S20 blends with SAN incorporation.
According to Table 4, Si and SiR incorporation occurred in different trends for different samples.
For virgin PCL-1 and PCL-2 samples, the incorporation of Si and SiR resulted in an increment in at whole , only there was a decrease in for PCL-2-3Si compared to PCL-2 at 20 % . Interestingly, in PCL-1, the incorporation of Si led to a higher increment of in all measured , while the addition of SiR increased more dominantly in PCL-2. This means that Si reduces the crystallization rate more than SiR in PCL-1, while the trend is opposite in higher molecular weight PCL-2 and the incorporation of SiR further decreases the crystallization rate.
In P80S20-1, the addition of Si increases at of 20 % and 40 %, while decreasing at higher . However, the addition of SiR to P80S20-1 decreases the at all s. In P80S20-2, the incorporation of both Si and SiR results in an increment in at whole s, only there was a decrease in for P80S20-2-3Si compared to PCL-2 at of 20 %. The data resulting from the Mo model, which is in agreement with the Avrami model, indicates that the incorporation of Si and SiR to P80S20-1 increases the rate of crystallization, especially at higher s. In contrast, it demonstrates that the incorporation of Si and SiR into P80S20-2 decreases the rate of crystallization. of P80S20-1 and P80s20-2 is considerably influenced by the incorporation of SiR compared to Si. Indeed, hydrophobic SiR affects the PCL crystallization process more effectively in the P80S20 blend.
3.1.4 Activation energy of non-isothermal crystallization kinetics
3.1.4.1 Kissinger method
Kissinger method is applied to assess the activation energy of crystallization (
) (Ebadi-Dehaghani et al., 2015; Kissinger, 1956) considering the crystallization peak temperature (
) at a constant cooling rate of
as below according to Eq. (8), where
is the universal gas constant.
The plots of
vs.
for all samples were depicted in Fig. 6, which shows a good linear relation. The slopes of Kissinger plots is used to calculate the
, which are given in Table 5.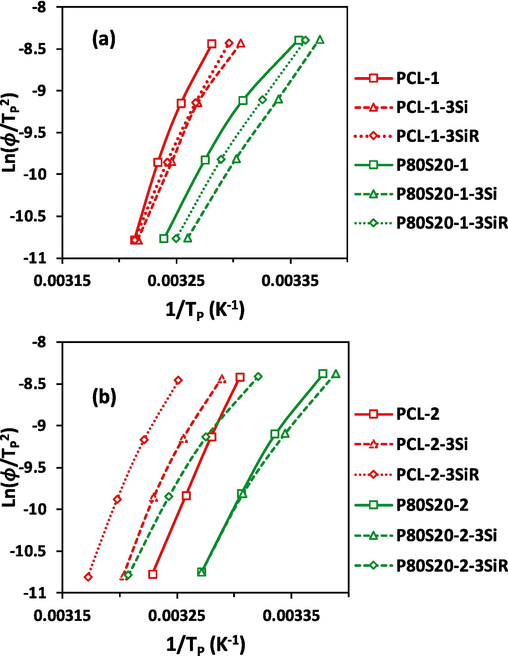
Kissinger plots for (a) virgin PCL-1, PCL-1-3Si, PCL-1-3SiR, P80S20-1, P80S20-1-3Si, and P80S20-1-3SiR, (b) virgin PCL-2, PCL-2-3Si, PCL-2-3SiR, P80S20-2, P80S20-2-3Si, and P80S20-2-3SiR.
Sample
Sample
PCL-1
285.21
PCL-2
256.97
PCL-1-3Si
235.91
PCL-2-3Si
226.66
PCL-1-3SiR
218.00
PCL-2-3SiR
247.39
P80S20-1
165.91
P80S20-2
186.05
P80S20-1-3Si
172.96
P80S20-2-3Si
167.18
P80S20-1-3SiR
169.89
P80S20-2-3SiR
173.53
is considered an activation energy needed to make motion of the macromolecular segments toward the growing surface of crystalline lamellae. The smaller values for lead to easier crystallization of polymer chains (Su and Lin, 2004). As expected, the higher the molecular weight PCL, the lower the . The activation energy of crystallization is gradually reduced by the growth of the of polymers. The incorporation of the SAN components in P80S20 blends reduces the activation energy of PCL crystallization, relating to the hindrance of amorphous SAN on the PCL crystallization process, which is in agreement with the Avrami and Jeziorny results. An interesting point of Table 5 is that the P80S20-1 has a lower compared to P80S20-2. In other words, the presence of SAN facilities the crystallization of PCL in P80S20-1 more compared to P80S20-2.
is decreased by adding 3 wt% of both Si and SiR to PCL-1 and PCL-2. The reduction of by the incorporation of nanofiller is more obvious in lower molecular weight PCL-1 (17 % reduction for the addition of Si and 24 % reduction for SiR). Indeed, a very small change is observed by the addition of nanosilica to PCL-2 (12 % reduction for the addition of Si and 4 % reduction for SiR). The presence of SiR reduces the of PCL-1 more considerably than Si. While the reverse effect has been observed by the incorporation of nanofiller into PCL-2, Si leads to a higher reduction of in PCL-2. The lower of silica-filled PCL-1 and PCL-2 compared to neat PCL-1 and PCL-2, considering the improved degree of crystallinity of both neat PCL in the presense of Si and SiR, indicates a positive effect of nanosilica on the PCL crystallization process. However, the presence of hyrophobic SiR in PCL-1 facilitates the crystallization more effectively.
Intriguingly, the increment of is resulted by the incorporation of both Si (4.2 %) and SiR (2.4 %) to P80S20-1. While the addition of both Si (11 %) and SiR (6.7 %) reduces the of nanocomposites compared to P80S20-2. The data of the Kissinger model are in agreement with the results of the Avrami and Jeziorny equations.
Considering the variation of
at different cooling rates (see Table 6) will also shed light on the effect of nanosilica on the crystallization of PCL. According to Table 6, by increasing the
of PCL, as well as incorporating SAN into the P8S20 blends,
shifts to lower temperatures. Interestingly,
is affected differently by the addition of Si and SiR nanoparticles to the pure PCL and P80S20 blends depending on the
of PCL in the sample formulations. Incorporation of Si and SiR nanoparticles into a lower
PCL-1 and P80S20-1 leads to a shift in
to lower temperatures, while in higher
PCL-2 and P80S20-2,
shifts to higher temperatures. Only in the P80S20-2 sample, adding 3 wt% hydrophilic silica did not cause any noticeable changes in
.
Sample
(°C)
2
5
10
20
2
5
10
20
PCL-1
46.88
43.73
41.94
39.93
38.24
36.25
34.35
31.8
PCL-1-3Si
46.42
44.37
43.20
41.70
37.94
35.08
32.92
29.45
PCL-1-3SiR
44.49
44.63
43.29
40.09
38.24
35.48
33.12
30.41
P80S20-1
45.62
42.82
41.77
39.25
35.75
32.37
29.29
24.87
P80S20-1-3Si
43.42
42.39
39.70
38.10
33.76
29.84
26.47
23.26
P80S20-1-3SiR
43.97
43.13
41.90
38.55
34.72
31.05
27.74
24.34
PCL-2
38.26
36.12
34.81
34.75
36.75
33.98
31.9
29.6
PCL-2-3Si
38.16
36.18
35.88
35.49
39.21
36.64
34.18
31.06
PCL-2-3SiR
38.43
36.22
35.35
35.33
42.26
39.73
37.48
34.61
P80S20-2
36.14
36.13
34.82
34.68
32.69
29.48
26.81
23.11
P80S20-2-3Si
36.90
35.02
33.35
31.09
32.7
29.34
26.02
22.14
P80S20-2-3SiR
37.09
36.08
34.24
33.37
38.79
35.39
32.32
28.16
Table 6, also lists the values of the calculated degree of crystallinity (
) of the samples according to Eq. (9), as follows:
The addition of both Si and SiR led to an increment in the crystallinity of PCL in both PCL-1 and PCL-2-based nanocomposites, especially at higher cooling rates, which can be related to the nucleation effect of nanoparticles. The nucleation effect of nanoparticles leads to a reduction in . In the case of P80S20-based nanocomposites, mainly a slight decrease in crystallinity is estimated compared to P80S20 blends. This reduction is more considerable in Si-filled samples compared to SiR-filled samples.
According to Table 6, there is a decrease in by increasing the of PCL, as well as incorporating SAN in the P80S20 blends. Although the incorporation of Si and SiR nanoparticles into pure PCL samples slows down the crystallization rate ( ) of PCL, it leads to an increment in , especially at higher cooling rates. This means the nucleation effect of nanoparticles in PCL/Silica nanocomposites.
3.1.4.2 Friedman method
The Friedman approach (Jafari et al., 2019; Friedman, 1964) is another model used for a non-isothermal study based on the assessment of the effective energy barrier for crystallization according to the Eq. (10),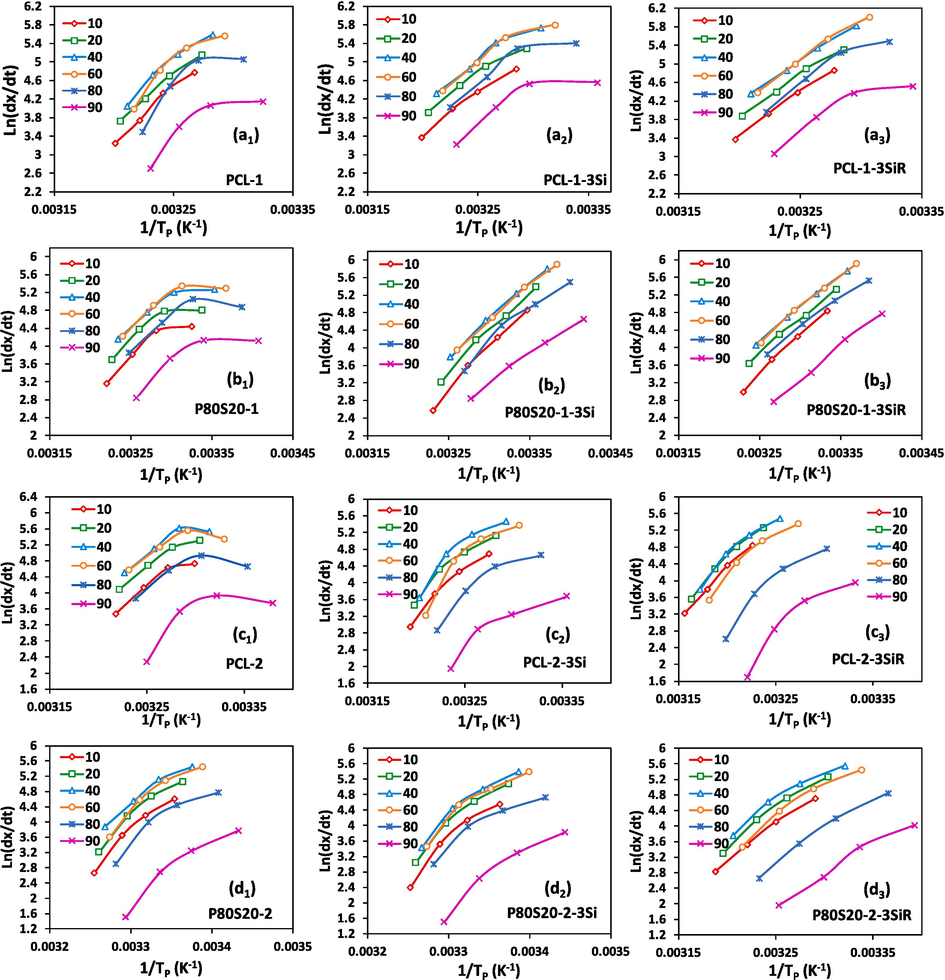
Plots of ln (dx/dt)x,i vs 1/Tx in different crystallinity degrees for (a1, a2, a3) virgin PCL-1, PCL-1-3Si, and PCL-1-3SiR, (b1, b2, b3) P80S20-1, P80S20-1-3Si, and P80S20-1-3SiR, (c1, c2, c3) virgin PCL-2, PCL-2-3Si, and PCL-2-3SiR, (d1, d2, d3) P80S20-2, P80S20-2-3Si, and P80S20-2-3SiR.
Sample
XC(T)
ΔEX
R2
Sample
XC(T)
ΔEX
R2
PCL-1
10
195.00
0.986
PCL-2
10
131.27
0.923
20
174.18
0.992
20
125.03
0.952
40
175.62
0.966
40
104.07
0.823
60
167.55
0.890
60
67.65
0.645
80
146.70
0.757
80
55.86
0.520
90
118.92
0.762
90
83.84
0.565
PCL-1-3Si
10
158.13
0.977
PCL-2-3Si
10
176.79
0.965
20
118.09
0.979
20
160.37
0.940
40
121.99
0.974
40
164.31
0.894
60
119.41
0.937
60
177.58
0.865
80
90.09
0.843
80
135.14
0.873
90
86.59
0.801
90
110.53
0.866
PCL-1-3SiR
10
152.28
0.977
PCL-2-3SiR
10
167.05
0.918
20
143.35
0.979
20
162.51
0.941
40
141.36
0.974
40
168.83
0.949
60
148.42
0.937
60
191.94
0.981
80
125.65
0.843
80
196.07
0.993
90
105.39
0.801
90
161.68
0.878
P80S20-1
10
101.79
0.992
P80S20-2
10
161.77
0.959
20
81.87
0.994
20
148.05
0.952
40
77.65
0.996
40
122.67
0.966
60
68.78
0.997
60
132.45
0.928
80
60.23
0.966
80
131.53
0.928
90
66.95
0.993
90
119.38
0.889
P80S20-1-3Si
10
162.61
0.992
P80S20-2-3Si
10
160.19
0.940
20
151.73
0.994
20
146.10
0.951
40
139.55
0.996
40
133.14
0.944
60
132.72
0.997
60
122.49
0.928
80
126.60
0.966
80
100.65
0.921
90
107.64
0.993
90
126.50
0.940
P80S20-1-3SiR
10
147.79
0.996
P80S20-2-3SiR
10
152.18
0.992
20
129.46
0.997
20
150.08
0.974
40
124.04
0.998
40
127.72
0.956
60
125.87
0.996
60
132.44
0.957
80
111.62
0.993
80
136.12
0.981
90
126.86
0.996
90
125.43
0.980
The results from Table 7 show that, in general, there is a decreasing trend for with an increase of for most of the samples. The addition of SAN to PCL-1 in P80S20-1 reduces , which is in agreement with the Kissinger technique. As well, it is consistent with the findings of Avrami, Jeziorny, and Mo equations. While the increment of is resulted by the addition of SAN to PCL-2 in P80S20-2. The observed trend for P80S20 blends indicates the ease of PCL crystallization process for PCL-1, while its hardening for PCL-2 in the presence of amorphous SAN. This was also confirmed in our latest research, whereas the higher effective activation energy resulted in P90S10-2 and P80S20-2 compared to PCL-2. Although it should be considered that the crystallinity of PCL in the P80S20 blend compared to pure PCL is reduced in P80S20-1, but in P80S20-2 it is almost the same.
reduces by adding both Si and SiR to pure PCL-1, while it increases by the addition of both nanosilica in the case of P80S20-1. This data is similar to the trend observed in the Kissinger technique and in agreement with the results of the Avrami, Jeziorny, and Mo equations. Indeed, the incorporation of Si and SiR accelerates the crystallization kinetics of PCL, while it slows it down for P80S20-1. SiR affects the trend more considerably at lower crystallinity degrees, while the effect of Si is more dominant at higher crystallinity degrees.
The contradictory data from those of the Kissinger model is the increment of in PCL-2 by incorporation of Si and SiR, although the reduction of of PCL-2 in the Kissinger model in the presence of nanoparticles is small. For P80S20-2-based nanocomposites, decreased at lower , while it increased at higher due to the incorporation of Si and SiR.
3.2 Discussion
By adding Si and SiR nanoparticles to PCL-1 and PCL-2 samples, in general, at all cooling rates (unless 2 K/min for PCL-2-3Si), the and values reduce and the value increases (see Tables 1 and 2). It means that adding 3 phr of hydrophilic and hydrophobic nanosilica to PCL-1 and PCL-2 samples leads to a lower crystallization rate of PCL.
However, the incorporation of Si and SiR into P80S20 blends has yielded interesting data. The addition of Si to P80S20-1 leads to the deceleration of crystallization kinetics of PCL at two lower cooling rates (2 and 5 K/min) compared to P80S20-1, while it accelerates it at the two higher cooling rates (10 and 20 K/min). However, the effect of Si on P80S20-2 is vice versa. Indeed, the incorporation of Si to P80S20-2 accelerates the crystallization rate at two lower cooling rates, while slowing down it at two higher rates. With the addition of SiR, the of P80S20-1-3SiR increased at all cooling rates compared to P80S20-1, which means speeding up the crystallization rate of P80S20-1 in the presence of SiR. Interestingly, the addition of SiR to P80S20-2 considerably decreases the at all cooling rates. Indeed, based on the kinetics analysis’s result, the crystallization process of PCL in P80S20-1 is considerably accelerated in the presence of the hydrophobic SiR, whereas the hydrophilic Si contributes only slightly to the PCL crystallization process in P80S20-1 (only at higher cooling rates). These reverse effects of Si and SiR on the crystallization kinetics of PCL in PCL/SAN blends are related to the preferential localization of Si/SiR in the biphasic PCL/SAN blends, different influences of silica nanoparticles on the LCST-type phase behavior of PCL/SAN blends and changes in the LCST phase diagram position by altering the PCL molecular weight.
The bulk morphology of cryo-microtomed P80S20-1-3SiR, P80S20-2-3Si, P80S20-2-3SiR, P50S50-1-3Si and P50S50-1-3SiR was studied by TEM and reported on Fig. 8 and Fig. 9 to illustrate the effects of the nanofiller type and PCL molecular weight on the localization and dispersion state of silica nanoparticles, respectively. As can be found from the images in Fig. 8, the dispersion state of hydrophobic nanosilica (SiR) is better than the one for hydrophilic nanosilica (Si) for both blends containing PCL-1 and PCL-2. Additionally, from Fig. 9, it can be concluded that the SiR dispersion state improves by increasing PCL molecular weight. The presence of smaller silica nanoparticle agglomerates and aggregates can prove these facts. The hydrophilic nature and lower specific surface area of SiR nanoparticles as well as the higher viscosity of the PCL-2-based blends during the melt-compounding process cause better dispersion of hydrophobic nanosilica, especially in the PCL-2-based blends. The dispersion state of nanoparticles has a considerable effect on the PCL crystallization kinetics.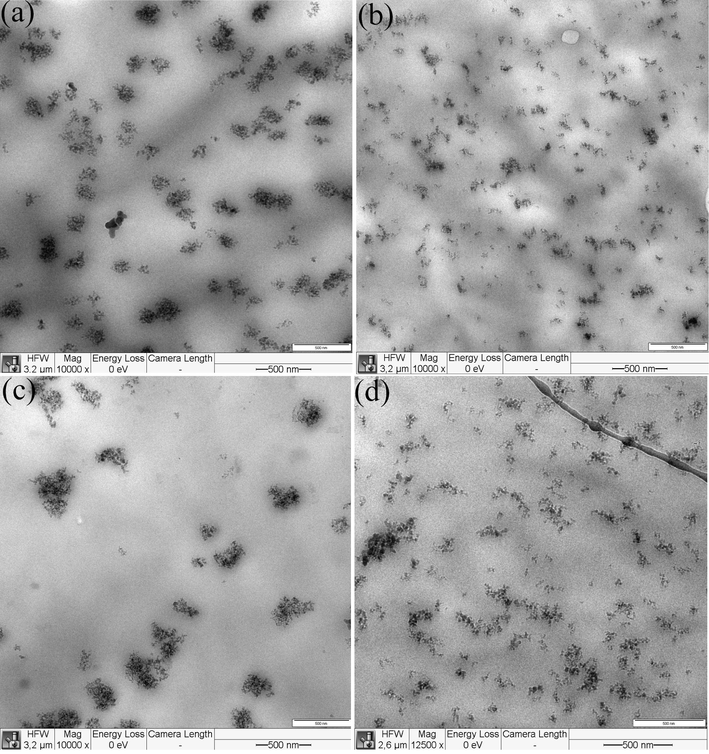
TEM image of: (a) P80S20-2-3Si, (b) P80S20-2-3SiR, (c) P50S50-1-3Si and (d) P50S50-1-3SiR.

TEM image of: (a)-(b) P80S20-1-3SiR and (c)-(d) P80S20-2-3SiR.
Besides these data, from the TEM images of Figs. 8 and 9, clearly no selective localization for Si and SiR nanoparticles can be perceived. The staining of the cryo-microtomed specimens did not increase the contrast of PCL-rich and SAN-rich domains. However, a closer look at the TEM images shows that the surface has some darker and brighter areas. These areas can be considered as PCL-rich and SAN-rich domains, since these polymers have different mechanical properties. By considering that, no preferential localization for Si and SiR nanoparticles can be perceived in both PCL-1 and PCL-2-based nanocomposites. It is noteworthy that for the TEM image preparation, the cryo-microtomy was done on the samples without any annealing after the melt-compounding process. Indeed, in these samples, Si and SiR nanoparticles are localized in both PCL-rich and SAN-rich regions, as well as the PCL/SAN interface for both PCL molecular weights.
The preferential localization of Si and SiR nanoparticles at the interface of PCL and SAN-rich domains was reported in our previous work according to the theoretically calculated wettability parameters of hydrophilic Si and hydrophobic SiR in PCL/SAN blends (Mohtaramzadeh et al., 2020). However, the microscopic observations on the samples without annealing after melt-compounding mostly show non-selective localization of nanoparticles. In our previous paper, it was demonstrated that the hydrophilic nanosilica particles migrate to the SAN-rich domains by annealing the samples, owing to the higher interfacial tensions of PCL/Si and SAN/Si pairs compared to the interfacial tension of PCL/SAN pair. Both theoretical calculation of interfacial tensions and TEM images on the annealed samples supported the Si nanoparticles migration to the SAN-rich domains during the DSC tests (Mohtaramzadeh et al., 2020). In contrast, the hydrophobic nanosilica is preferentially localized at PCL-rich and SAN-rich domains as well as PCL/SAN interface (supported by both theoretical calculations and experimental evidence) (Mohtaramzadeh et al., 2020). During the non-isothermal DSC experiments, the samples were heated to 130 °C to erase the thermal history. At this segment of the DSC test, the entrance to the phase-separated region of LCST phase diagram and the Si nanoparticle migration to the SAN-rich domains would probably happen. Considering different cooling rates of the DSC tests, the occurrence of these kinetic-controlled phenomena will be more noticeably observed at lower cooling rates, i.e., 2 and 5 °C/min.
In addition to the nanosilica particle migration during the non-isothermal DSC test, the position of the LCST-type phase diagram of PCL/SAN blends has a profound impact on the crystallization kinetics of the PCL phase. Indeed, the amount of dissolved amorphous SAN chains that can retard the PCL-phase crystallization is determined by the PCL/SAN phase separation and mutual dissolution. It is affected by the LCST phase diagram position and the non-isothermal DSC ramp rate. According to our previous works, the position of the phase boundary of the PCL/SAN LCST-type phase diagram changes by altering the PCL molecular weight and adding hydrophilic and hydrophobic nanosilica (Maghoul et al., 2023; Khadivi et al., 2023; Mohtaramzadeh et al., 2020). In one of these works, it has shown that the addition of SiR nanoparticles to PCL/SAN blend causes the shift of the equilibrium LCST phase diagram to higher temperatures. Contrary to the compatibilization effect of SiR nanofiller on the PCL/SAN phase behavior, the presence of hydrophilic nanosilica, Si, shifted the phase diagram to lower temperatures and decreased the miscibility window area (Maghoul et al., 2023).
In another work of our group on the non-isothermal crystallization kinetics of PCL in PCL/SAN blends containing PCL grades with different molecular weights, it was shown that the PCL crystallization in the blend with lower molecular weight of PCL was comparatively easier (Khadivi et al., 2023). In that work, it was found that the plausible reason for the easier crystallization in P80S20-1 blend was the presence of the larger amount of dissolved SAN chains in the PCL-rich domains, which was induced by phase separation during DSC experiment at higher temperatures. In contrast, the LCST-type phase diagram for P80S20-2 was probably shifted to higher temperatures and during the heating segment of the DSC test, the sample did not succeed in entering the phase-separated region of the LCST phase diagram (Khadivi et al., 2023). More dissolved amorphous SAN chains in the P80S20-2 blend brought about higher activation energy of PCL crystallization. Better compatibility of PCL and SAN phases in PCL-1 and PCL-2-based systems can be clarified by comparing the SEM images of these mixtures, which were prepared by examining the cryo-microtomed surfaces. Fig. 10 and Figure S2 (in the supporting information) demonstrate the lower size of rough areas that are related to the SAN-rich domains. Contrary to the PCL-1-based systems in Fig. 10, no visible rough areas can be discerned on the SEM images of PCL-2-based mixtures, confirming the higher miscibility of PCL and SAN in the blend containing the PCL polymer with higher molecular weight.
SEM image of the cryo-microtomed surface of: (a) P80S20-1, (b) P80S20-2, (c) P80S20-1-Si3, (d) P80S20-2-3Si, (e) P80S20-1-3SiR and (f) P80S20-2-3SiR. The white arrows mark the SAN-rich domains.
Considering the above evidence can help someone interpret the obtained data showing opposite effects of Si and SiR on the PCL crystallization kinetics in PCL-1 and PCL-2-based blends. For the SiR nanosilica influence on the crystallization of P80S20-1 and P80S20-2 blends, it should be kept in mind that SiR nanoparticles have no selective localization state in PCL/SAN blends and LCST-type phase diagram is most likely shifted to higher temperatures in PCL-2-based blends. As a direct result, P80S20-2-3SiR does not enter the phase-separated region during the heating segment of the DSC experiment. In contrast, P80S20-1-3SiR nanocomposite most probably enters the phase-separated region during the examination. Therefore, less amount of dissolved SAN chains alongside PCL chains during crystallization makes the poisoning SAN effect less strong. By that, SiR nanoparticles with good dispersion state attain the opportunity to accelerate the PCL crystallization process by providing larger heterogeneous nucleation sites. Thus, SiR nanofiller accelerates PCL crystallization in P80S20-1 and oppositely retards PCL crystallization in P80S20-2 blend.
For the adverse effects of the hydrophilic nanosilica on the PCL crystallization kinetics of PCL-1 and PCL-2-based blends, which also depend on the DSC cooling rate, four notable points should be considered. First of all, Si nanoparticles do not disperse as well as SiR nanofiller in PCL/SAN blends. Second, hydrophilic nanosilica thermodynamically prefers to migrate to the SAN-rich domains. Third, Si nanofiller does not improve better PCL/SAN miscibility like hydrophobic nanosilica. Fourth, the PCL-2-based blend has higher compatibility than the PCL-1-based mixture. As a result, the LCST-phase diagram of PCL/SAN will be positioned at a lower temperature for P80S20-1-3Si. During the first segments of the DSC experiment, the nanocomposite enters the phase-separated region. By the occurrence of PCL/SAN phase separation, less amount of dissolved SAN in the PCL-rich phase causes a weaker retardation effect on the crystallization. Therefore, Si nanoparticles will be successful to accelerate the PCL crystallization at cooling rates of 10 and 20 K/min. While, at lower cooling rates (2 and 5 K/min), the preferential migration of hydrophilic nanosilica to the SAN-rich phase makes the acceleration effect of nanoparticles less intensified. Thus, a retardation effect for Si in P80S20-1-3Si is observed at lower cooling rates of 2 and 5 K/min.
For P80S20-2-3Si nanocomposite, the PCL polymer has a higher molecular weight. In this blend, PCL and SAN have higher compatibility. However, Si nanoparticles do not shift the LCST-type phase diagram to higher temperatures like SiR ones. As a direct result, PCL/SAN phase separation cannot happen at higher cooling rates of 10 and 20 K/min. Indeed, a higher amount of dissolved SAN chains diminishes the PCL crystallization rate in the P80S20-2-3Si sample at higher cooling rates. Contrary to that, the P80S20-2-3Si nanocomposite most likely encounters the phase separation to some extent at lower cooling rates of 2 and 5 K/min. Therefore, the phase separation of amorphous SAN chains provides the opportunity for silica nanoparticles with a better dispersion state in P80S20-2-3Si than in P80S20-1-3Si to assist the PCL crystallization process.
Following the aim of this work, it has been shown that the non-isothermal crystallization behavior is a complex phenomenon in the semi-crystalline/amorphous polymeric systems containing nanoparticles. The crystallization kinetics in these systems has a significant effect on the product’s final properties. The findings clarify that different factors like the system phase behavior, the nanoparticle preferential migration to one of the phases, the molecular weight of the polymeric components and the position of the miscibility window noticeably influence the crystallization kinetics.
4 Conclusions
In this work, the semicrystalline/amorphous PCL/SAN mixtures, with LCST-type phase diagram up above the crystallization temperature of PCL, were chosen to study the effects of PCL molecular weight and nanosilica type on the non-isothermal crystallization kinetics. The crystallization kinetics variables and activation energy of PCL were determined by fitting theoretical models on the experimental data including Avrami, Jeziorny, Ozawa, Mo, Kissinger and Friedman models. Among different applied theoretical models, the best fitting of the experimental data is obtained for the Avrami, Jeziorny, Mo and Kissinger models. The largest R2s are attained for the determination of and (considering a three-stage crystallization), and . These kinetic variables are the most useful parameters for showing the changes in the PCL crystallization rate against the PCL molecular weight, nanosilica type and DSC cooling rate. According to these data, the following results are concluded:
While the presence of both hydrophilic and hydrophobic nanosilica fillers has a retardation effect on the PCL crystallization kinetics, the presence of these nanoparticles in the PCL/SAN blends affects the PCL crystallization kinetics through opposite trends depending on the PCL molecular weight, nanosilica type and DSC cooling rate. The hydrophobic nanosilica accelerates the PCL crystallization in the PCL/SAN blend containing PCL with lower molecular weight, regardless of the DSC cooling rate. In contrast, this nanofiller retards the PCL crystallization for the blend based on the higher molecular weight PCL. The plausible reasons for these opposite trends are the probable shift of the LCST phase diagram by increasing the PCL molecular weight, relatively better dispersion state and no preferential localization state of this type of nanosilica. Nonetheless, the influences of hydrophilic nanosilica on the PCL crystallization in the PCL/SAN mixture also depend on the cooling rate of the DSC test. The Si nanoparticles decrease the PCL crystallization rate at a lower DSC ramp rate, while these increase crystallizations at higher cooling rates, albeit for the PCL/SAN blend including lower molecular weight PCL. For the blend based on the higher molecular weight PCL, the Si effects on the PCL crystallization are totally opposite. As mentioned, the changes in the LCST phase diagram induced by the nanosilica presence and PCL molecular weight, selective migration of nanosilica during the test and nanosilica dispersion state are the important factors to cause the opposite trends in the crystallization behavior of PCL in the PCL/SAN blends. Overall, according to the findings, it can be concluded that the non-isothermal crystallization of polymers in hybrid systems with a special phase behavior like the one studied here is a complex phenomenon, that is impacted by several factors. Each of these factors can alter the crystallization rate in different ways. In industrial-scale production of similar systems, these factors should be considered to optimize the crystallization and final properties of the products.
CRediT authorship contribution statement
Samaneh Salkhi Khasraghi: Conceptualization, Methodology, Investigation, Resources, Visualization, Software, Writing – original draft. Hanieh Khonakdar: Conceptualization, Methodology, Investigation, Resources, Visualization, Software, Writing – original draft. Farkhondeh Hemmati: Investigation, Resources, Writing – review & editing. Sara Zarei: Methodology, Investigation, Resources, Visualization, Software. Fatemeh Sadat Miri: Methodology, Investigation, Resources, Visualization, Software. Marcos A.L. Nobre: Investigation, Resources, Writing – review & editing. Hossein Ali Khonakdar: Investigation, Resources, Writing – review & editing.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Aliphatic polyesters: Synthesis, properties and applications. Degradable Aliphatic Polyesters 2002:1-40.
- [Google Scholar]
- Poly (lactic acid) films reinforced with hybrid zinc oxide-polyhedral oligomeric silsesquioxane nanoparticles: morphological, mechanical, and antibacterial properties. Polym. Adv. Technol.. 2023;34(3):985-997.
- [Google Scholar]
- Crystallization and melting behavior of poly (ethylene succinate) in presence of graphene nanoplatelets. Thermochim Acta. 2014;586:17-24.
- [Google Scholar]
- Kinetics of phase change. II transformation-time relations for random distribution of nuclei. J. Chem. Phys.. 1940;8(2):212-224.
- [Google Scholar]
- Compatibilizer/graphene/carboxylated acrylonitrile butadiene rubber (XNBR)/ethylenepropylenediene monomer (EPDM) nanocomposites: Morphology, compatibility, rheology and mechanical properties. J. Appl. Polym. Sci.. 2020;137(43)
- [Google Scholar]
- Architectural effects of poly (ε-caprolactone) s on the crystallization kinetics. Macromolecules. 2004;37(10):3745-3754.
- [Google Scholar]
- Influence of graphene oxide on crystallization behavior and chain folding surface free energy of poly (vinylidenefluoride-co-hexafluoropropylene) Macromol. Chem. Phys.. 2017;218(19):1700103.
- [Google Scholar]
- Polymer nanocomposites: the “nano” effect on mechanical properties. Polym. Rev.. 2007;47(2):217-229.
- [Google Scholar]
- Isothermal and nonisothermal crystallization kinetics of poly (∊-caprolactone) J. Appl. Polym. Sci.. 2012;124(2):1333-1343.
- [Google Scholar]
- Microstructure and non-isothermal crystallization behavior of PP/PLA/clay hybrid nanocomposites. J. Therm. Anal. Calorim.. 2015;121:1321-1332.
- [Google Scholar]
- Impact of poly (ε-caprolactone) on the thermal, dynamic-mechanical and crystallization behavior of polyvinylidene fluoride/poly (ε-caprolactone) blends in the presence of KIT-6 mesoporous particles. Polym. Adv. Technol.. 2021;32(11):4424-4439.
- [Google Scholar]
- Kinetics of thermal degradation of char-forming plastics from thermogravimetry. Application to a phenolic plastic. J. Polym. Sci. Part c: Polym. Symposia 1964
- [Google Scholar]
- Electrospun nanofibers of poly (lactic acid)/poly (ε-caprolactone) blend for the controlled release of levetiracetam. Polym. Eng. Sci.. 2022;62(12):4070-4081.
- [Google Scholar]
- Nonisothermal crystallization kinetics and determination of surface-folding free energy of PP/EVA/OMMT nanocomposites. J. Polym. Sci. B. 2009;47(7):674-684.
- [Google Scholar]
- Effects of organoclay on the compatibility and interfacial phenomena of PE/EVA blends with UCST phase behavior. Polym. Compos.. 2014;35(12):2329-2342.
- [Google Scholar]
- Polymer blends; stretching what we can learn through the combination of experiment and theory. Prog. Polym. Sci.. 2005;30(8–9):832-843.
- [Google Scholar]
- A correlation between morphology and mechanical performance of injected-molded PE/EVA/clay nanocomposites: Insight into phase miscibility and interfacial phenomena. J. Appl. Polym. Sci.. 2020;137(45):49401.
- [Google Scholar]
- Nonisothermal crystallization kinetic studies on melt processed poly (ethylene terephthalate)/polylactic acid blends containing graphene oxide and exfoliated graphite nanoplatelets. J. Appl. Polym. Sci.. 2019;136(23):47569.
- [Google Scholar]
- Non-isothermal crystallization of poly (ε-caprolactone)-grafted multi-walled carbon nanotubes. Compos. A Appl. Sci. Manuf.. 2010;41(10):1524-1530.
- [Google Scholar]
- Parameters characterizing the kinetics of the non-isothermal crystallization of poly (ethylene terephthalate) determined by DSC. Polymer. 1978;19(10):1142-1144.
- [Google Scholar]
- Air-jet spun PHBV/PCL blend tissue engineering scaffolds exhibit improved mechanical properties and cell proliferation. Results Mater. 2023100415
- [Google Scholar]
- Correlation between non-isothermal crystallization kinetics and morphology in poly (ε-caprolactone)/poly (styrene-co-acrylonitrile) blends considering the blend phase behavior: Effects of poly (ε-caprolactone) molecular weight. J. Appl. Polym. Sci. 2023:e54384.
- [Google Scholar]
- Variation of peak temperature with heating rate in differential thermal analysis. J. Res. Natl. Bur. Stand. 1956;57(4):217-221.
- [Google Scholar]
- Blends of biodegradable poly (ε-caprolactone) and sustainable poly (propylene carbonate) with enhanced mechanical and rheological properties. Colloid Polym. Sci. 2022:1-10.
- [Google Scholar]
- Kinetics studies of non-isothermal melt crystallization of poly (ε-caprolactone) and poly (L-lactide) Chiang Mai J. Sci. 2016;43:329-338.
- [Google Scholar]
- Morphology in binary blends of poly (vinyl methyl ether) and ∊-caprolactone-trimethylene carbonate diblock copolymer. Polymer. 1997;38(3):509-519.
- [Google Scholar]
- Nonisothermal crystallization kinetics of miscible blends of polycaprolactone and crosslinked carboxylated polyester resin. J. Macromol. Sci. Part B. 2011;50(3):427-443.
- [Google Scholar]
- Isothermal crystallization of poly (ε-caprolactone) in blend with poly (styrene-co-acrylonitrile): influence of phase separation process. Macromol. Chem. Phys.. 2004;205(14):1923-1931.
- [Google Scholar]
- Isothermal crystallization kinetics of poly (ε-caprolactone) with tetramethyl polycarbonate and poly (styrene-co-acrylonitrile) blends using broadband dielectric spectroscopy. Macromol. Chem. Phys.. 2006;207(11):978-986.
- [Google Scholar]
- Thermodynamically-equilibrium LCST phase diagram of PCL/SAN mixtures determined by thermal analysis: opposing effects of hydrophilic and hydrophobic silica nanoparticles. Thermochim Acta. 2023;724:179501
- [Google Scholar]
- Nonisothermal crystallization and microstructural behavior of poly (ε-caprolactone) and granular tapioca starch-based biocomposites. Int. J. Polym. Anal. Characterization. 2017;22(3):222-236.
- [Google Scholar]
- Studies on the effect of silicon carbide nanoparticles on the thermal, mechanical, and biodegradation properties of poly (caprolactone) J. Appl. Polym. Sci.. 2015;132(26)
- [Google Scholar]
- Study on the effects of polyhedral oligomeric silsesquioxane on compatibility, crystallization behavior and thermal stability of polylactic acid/polycaprolactone blends. Polym. Bull.. 2020;77:585-598.
- [Google Scholar]
- A review on the recent research of polycaprolactone (PCL) Adv. Mat. Res.. 2016;1134:249-255.
- [Google Scholar]
- Structure-properties correlations in poly (ε-caprolactone)/poly (styrene-co-acrylonitrile)/nanosilica mixtures: Interrelationship among phase behavior, morphology and non-isothermal crystallization kinetics. Polym. Test.. 2020;89:106593
- [Google Scholar]
- Rheologically determined phase diagram of poly (ε-caprolactone)/poly (styrene-co-acrylonitrile) blends: Role of ramp rate in dynamic measurements. J. Appl. Polym. Sci.. 2019;136(28):47750.
- [Google Scholar]
- Spherical nanoparticle effects on the lower critical solution temperature phase behavior of poly (ε-caprolactone)/poly (styrene-co-acrylonitrile) blends: separation of thermodynamic aspects from kinetics. J. Appl. Polym. Sci.. 2020;137(19):48679.
- [Google Scholar]
- Morphology and crystallization behavior of PCL/SAN blends containing nanosilica with different surface properties. J. Appl. Polym. Sci.. 2016;133(43)
- [Google Scholar]
- Tire tread performance of silica-filled SBR/BR rubber composites incorporated with nanodiamond and nanodiamond/nano-SiO2 hybrid nanoparticle. Diam. Relat. Mater.. 2022;126:109068
- [Google Scholar]
- Phase behaviour of poly (∊-caprolactone)/(polystyrene-ran-acrylonitrile) blends exhibiting both liquid-liquid unmixing and crystallization. Polymer. 1993;34(17):3704-3709.
- [Google Scholar]
- Ringed spherulites in ternary polymer blends of poly (ε-caprolactone), poly (styrene-co-acrylonitrile), and polymethacrylate. Colloid Polym. Sci.. 2004;283:182-193.
- [Google Scholar]
- Light-scattering and TEM analyses of virtual upper critical solution temperature behavior in PCL/SAN blends. Macromolecules. 1994;27(5):1154-1159.
- [Google Scholar]
- Spinodal decomposition and succeeding crystallization in PCL/SAN blends. Polymer. 1999;40(6):1459-1463.
- [Google Scholar]
- Competition of phase dissolution and crystallization in poly (ε-caprolactone)/poly (styrene-co-acrylonitrile) blend. Eur. Polym. J.. 2008;44(2):329-341.
- [Google Scholar]
- In situ compatibilized blends of PLA/PCL/CAB melt-blown films with high elongation: investigation of miscibility, morphology, crystallinity and modelling. Polymers. 2023;15(2):303.
- [Google Scholar]
- The return of a forgotten polymer—Polycaprolactone in the 21st century. Prog. Polym. Sci.. 2010;35(10):1217-1256.
- [Google Scholar]
- Effect of temperatures on stress-induced structural evolution and mechanical behaviors of polyglycolic acid/polycaprolactone blends. Polymer. 2023;283:126239
- [Google Scholar]
- Thermal stability and dynamic mechanical properties of poly (ε-caprolactone)/chitosan composite membranes. Materials. 2021;14(19):5538.
- [Google Scholar]
Appendix A
Supplementary material
Supplementary data to this article can be found online at https://doi.org/10.1016/j.arabjc.2023.105522.
Appendix A
Supplementary material
The following are the Supplementary data to this article:Supplementary data 1
Supplementary data 1
The heat flow curves for the non-isothermal crystallization of samples as well as the SEM images of PCL/SAN 50/50 samples are shown in Figure S1 and Figure S2 of Supplementary material.







