Translate this page into:
Phytochemical and biological activity profiles of Thymbra linearifolia: An exclusively native species of Libyan Green mountains
⁎Corresponding authors at: Department of Medicinal Chemistry and Pharmacognosy, College of Pharmacy, Qassim University. ham.mohammed@qu.edu.sa (Hamdoon A. Mohammed), ri.khan@qu.edu.sa (Riaz A. Khan),
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
Thymbra linearifolia, a Green Mountain endemic Libyan plant species belonging to the family Lamiaceae, was investigated as the plant's first documented phytochemical and biological evaluation report. The plant is used for bronchitis, cough, asthma, and gastrointestinal ailments, including dyspepsia, colic pain, and diarrhea. The phenolic and flavonoid contents of T. linearifolia were analyzed qualitatively and quantitatively by LC-MS analysis and spectrophotometric assays. These analyses revealed the presence of 60.67 mg/g of GAE (Gallic Acid Equivalents) and 26.79 mg/g of RE (Rutin Equivalents) of the phenolics and flavonoids, respectively, in the aerial parts of the plants’ ethanolic extract. Cyanidin-3-O-rutinoside (2.26 %), pelargonidin-3-O-rutinoside (2.36 %), gallocatechin (2.94 %), taxifolin (1.22 %), kaempferol-3-O-rutinoside (0.81 %), quercetin-3-O-retinosie (1.20 %), luteolin (0.99 %), apigenin (2.0 %), dimethoxy luteolin (2.97 %), and pinobanksin-3-O-acetate (2.56 %) were detected to be the major flavonoids, whereas the syringic acid hexoside (1.38 %), and rosmarinic acid (2.54 %) were among the most abundant phenolic acids in the plant’s extract. The DPPH scavenging activity was measured at an IC50 value of 34.48 μg/mL. In addition, potential reducing, metal chelating, and free radical scavenging potentials were exhibited in the in vitro assays, i.e., ABST, ORAC, FRAP, and MC. The remarkable antioxidant activity of the plant was attributed to its phenolic and flavonoid contents. The plant extract had strong cytotoxic effects on three types of cancer cell lines, i.e., MCF-7, HepG2, and Panc-1, with IC50 values of 24.023, 22.94, and 33.30 μg/mL, respectively. Furthermore, the plant extract also exhibited cytotoxic effects greater than the standard anticancer drug, doxorubicin, against all the tested cell lines. The in-silico binding studies also demonstrated that the phytochemical constituents of T. linearifolia have a high binding affinity against the epidermal growth factor receptor (EGFR) protein, which supported the anticancer potentials of the plant. The antimicrobial assay conducted with the well-diffusion method revealed the moderate effects of the plant’s extract against Bacillus subtilis, Staphylococcus aureus, Saccharomyces cerevisiae, and Candida albicans. However, weak antimicrobial effects were recorded against the Gram-negative bacteria, Escherichia coli, and Salmonella typhimurium. These findings corroborated the immense health benefits of the plant, and substantiated the medicinal value claims of the plant, which is currently under use in different forms as part of the locals’ folk-medicinal chest.
Keywords
Thymbra linearifolia
Anthocyanins
Cytotoxicity
Antioxidant
Antimicrobial
Molecular structure
1 Introduction
Significant numbers of medicinal plants are concurrently used all over the world for their antibacterial, anticancer, and antioxidant properties. Plants are also in use as part of traditional and complementary medicine, including for finding new structural leads, developing new molecular templates, and subsequently discovering new drug candidates (Mohammed and Khan, 2022; Shanaida et al., 2020; Upton et al., 2016). A detailed phytochemical screening is an essential prerequisite to exploring, establishing, and comprehending the therapeutic significance of the natural extracts and their active constituents. The analysis is also a must for standardizing the crude extracts from the plant that hold a plethora of active ingredient(s), synergistic chemical entities, and perhaps the much-sought-after pharmaceutical agent(s) to be obtained from different plant species (Falzon and Balabanova, 2017). The biological activities, therapeutic potential, medicinal properties, and probable toxicity, as well as, the perceived and observed side effects of medicinal plants, are all directly connected to their phytoconstituents, particularly the secondary metabolites, e.g., essential oils, alkaloids, saponins, flavonoids, and phenolic acids, and their doses and duration of usage (Hussein and El-Anssary, 2019). The structural variations in the natural products, their abundance, concentration, and enrichments in the plant’s extract are affected by the plant’s environmental conditions, and abiotic stress factors, which are among the key components affecting the biological activities of medicinal plants (Mohammed, 2009; Mohammed et al., 2019; Ncube et al., 2012). For instance, the presence, frequency, and positional attachments of the hydroxyl group(s) in phenolic compounds, an essential structural feature for antioxidant and antibacterial properties, interact differently with the bacterial cell-wall, and in multiple ways towards different mechanistic pathways to produce anti-bacterial activity at different response levels as the bio-effects of the structure-led outcome (Lai and Roy, 2004).
Thymbra linearifolia belongs to the family Lamiaceae, which is abundant in herbs, shrubs, and trees, and comprises several economically valuable plant species producing essential oils, spices, and crude medicines, which comprise using leaves, flowers, seeds, roots, and other aerial parts as cold-water extracts, decoctions, oils, and plant parts’ powders to be taken orally and used topically. Several plant species of the Lamiaceae family are planted for edible, nutritional, ornamental, and lumber purposes (Figueiredo et al., 2010). Phytochemically, the volatile oils, and phenolics and flavonoids-based structures are common, and at times, major constituents. The bioactive oils are mainly comprised of several ingredients, including cineol, carvacrol, and thymol with potent antioxidant capabilities (Mohammed et al., 2020). T. linearifolia, abundant and exclusively found in the Green Mountain regions, is locally called “Zahter” in the vernacular language. The plant's occurrence, and geographic distributions have been described in 2018 by Christian BräuChler as endemic to Libya. The three growing locations of the plant are between Jebel Al-Akhdar, Benghazi and Taucheira (BräuChler, 2018). Furthermore, Christian BräuChler has recently reclassified the plant from genus Satureja to Thymbra, as the phylogenetic analyses have made it to be placed in Thymbra, a lineage well distant from the genus Satureja. The shape of the calyx and the orientation of the lobes are two other important morphological features differences between the Satureja and Thymbra (BräuChler, 2018). The plants from genus Thymbra have been reported for their abundance of phenolic compounds, e.g., benzoic acid and its derivatives, and carvacrol (Çelik et al., 2021; Delgado-Adámez et al., 2017; Sengun et al., 2021). Earlier investigations have found significant antibacterial and antioxidant activities in Thymbra species plants, other than T. linearifolia (Çelik et al., 2021; Erci et al., 2018; Sengun et al., 2021). To the best of our knowledge, currently there is no reported work on the phytochemical investigations and bioactivity property evaluations of the plant, T. linearifolia.
The current work is an attempt to explore the phytochemical constituents and evaluate the bioactivity profile of T. linearifolia, the first report for the exclusively distributed plant. Given the plant’s prevalent, but localized, use for managing physiological symptoms of many hormonal and non-hormonal disorders, diseases of the respiratory system, gastrointestinal tract, and wound healing, together with the plant’s use for enhancing the overall activity of the body as a vigor enhancer, blood purifier, and tonic, the plant's chemo-pharmacological tests were necessary steps. In this context, the plant’s qualitative and quantitative phytochemical profiling through colorimetric assays, and LC-MS analysis, in addition to its antioxidant, antimicrobial, and cytotoxic activity evaluations on different cell lines, were carried out.
2 Materials and methods
2.1 Chemicals and reagents
All chemicals and reagents were of analytical grade; aq. ethanol 95 %, Folin-Ciocalteu's reagent, gallic acid, rutin, Trolox, ethylene diamine tetra acetic acid (EDTA), aluminum chloride (AlCl3), potassium persulfate, Ferric chloride (FeCl3), sodium carbonate (Na2CO3), dimethyl sulfoxide (DMSO), trichloro acetic acid (TCA), 2,2-diphenyl-1-picryl-hydrazine-hydrate (DPPH), 2,2′-Azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS), fluorescein, 2,2′-Azobis(2-amidinopropane) dihydrochloride (AAPH), 2,4,6-Tris(2-pyridyl)-s-triazine (TPTZ) were purchased from Sigma-Aldrich, USA. Sulforhodamine B (SRB) was obtained from Invitrogen, Thermo Fisher Company, USA.
2.2 Collection, identification, and extraction of the plant material
Thymbra linearifolia L aerial parts were collected from the Green Mountain (Jabel Al-Akhḍar) in 2021, and identified by Dr. Christian BräuChler, Head of the Botany Department, Natural History Museum, Vienna, Austria. The voucher specimen was kept at the herbarium of the Benghazi University, Faculty of Science, Botany Department, with the herbarium record number '1–20581′. The collected plant materials were subjected to shade-drying at room temperature (RT), reduced in size through a mill-cutter, and stored in tight plastic containers for further investigations. Dried, homogenized aerial parts of the plant material (250 g) was extracted with aq. ethanol 95 % (analytical grade, Solveco company, 3x500 mL, three successive extractions) for 24 h in a water bath fixed at 40 °C temperature. The extract was filtrated, and dried under vacuum at 40 °C using rotatory evaporator.
2.3 Total phenolic contents (TPC)
Accurately, 0.5 mL of the herbal extract (1 mg/mL) was treated with 2 mL of Folin-Ciocalteu's reagent to estimate the total polyphenols content (TPC). The extract and reagent were thoroughly mixed with 2.5 mL of 7.5 % Na2CO3, and the final mixture was set aside for 30 min in dark. The absorbance of the mixture was recorded at 760 nm wavelength using a spectrophotometer (Jasco V-630, Japan). Another mixture consisting of the Folin-Ciocalteu's reagent, distilled water (instead of the plant’s extract), and Na2CO3 were used as a negative blank. Gallic acid was employed as the standard, and a 6-point standard curve (0–50 mg/L) was created (supplementary file). The TPC of T. linearifolia extract was quantified as gallic acid equivalents (mg GA/g) for the dried extract weight. The sample was examined twice (Attard, 2013; Singleton and Rossi, 1965).
2.4 Total flavonoid contents (TFC)
To evaluate the TFC, 1 mL of diluted plant extract (1 mg/mL) was mixed with 1 mL of 2 % AlCl3. After 15 min at RT incubation, the absorbance of the mixture was recorded at 430 nm wavelength using Jasco V-630 spectrophotometer. Standard calibration curve of the rutin was plotted (0–50 mg/L) (supplementary file). The TFC of the plant was expressed as rutin equivalents/g (RE) of the dried extract. All samples were analyzed twice under identical experimental conditions (Djeridane et al., 2006; Elshibani et al., 2020).
2.5 LC-MS analysis
All solvents used in the LC-MS analysis were of analytical grade. Shimadzu ExionLC (Shimadzu, Kyoto, Japan) equipped with a TurboIonSpray, SCIEX X500R QTOF (SCIEX, Framingham, MA, USA) was used for the extract’s scanning. Accurately, 1 mg of the extract was dissolved in 2 mL of DMSO and centrifuged at 5000 rpm for 2.0 min. Accurately, 1.0 mL of the extract solution was transferred to auto-sampler, and the injection volume was adjusted to 3.0 µL. The instrument was operated using ion-source-gas-1, 50 psi, ion-source-gas-2, 50 psi, and ion-funnel-electrospray-source. The instrument parameters were adjusted as capillary voltage (Negative, −4000 V), nebulizer gas (2.0 bar), nitrogen flow (8 L/min), and dry temperature (200 °C). The mass accuracy was less than 1 ppm, the mass resolution was 50,000 FSR (Full Sensitivity Resolution), and the TOF repetition rate was up to 20 kHz. The chromatographic separation was performed on InertSustain® C18 reverse-phase (RP) column, 100 × 2.1 mm, 3.0 µm (GL-Science, Japan) at 50 °C, auto-sampler temperature 8.0 °C, with a flow rate of 0.35 mL/min and a total run time of 40 min using the gradient elution. The eluents consisted of mobile phase A as 0.1% formic acid in water, and mobile phase B as 100 % acetonitrile.
2.6 Antioxidant assays
2.6.1 DPPH assay
A free radical, DPPH assay, was conducted according to the method described earlier (Faso, 2016), with certain modifications to suit the 96-well plate assay. Briefly, 150 μL of DPPH reagent solution (2 mg DPPH in 50 mL methanol) was thoroughly mixed with 5 μL of the plant’s extract solution in a 96-well plate (n = 3), and incubated for 30 min at RT. The color fading of the DPPH was measured at 517 nm wavelength using a microplate reader (FluoStar Omega, Ortenberg, Germany), and the IC50 values were calculated using GraphPad Prism-5® software (GraphPad Prism®, USA ). Data are represented as mean ± SD, as according. Percentage (%) inhibition equals to [(Average absorbance of blank) - (average absorbance of the test)] (divided by) [(Average absorbance of the blank)] × 100.
2.6.2 ABTS assay
The ABTS radical cation decolorization analysis was used to investigate the free radicals scavenging capacity of the extract. The analysis was performed in microplates following the technique outlined by Arnao et al (Arnao et al., 2001), with few modifications. Briefly, 192 mg of ABTS was dissolved in deionized water, and transferred to a 50 mL volumetric flask. The final volume was made with the addition of distilled water. Accurately, 1 mL of the previously prepared ABTS solution was mixed with 17 µL of 140 mM potassium persulfate, and stored for 24 hr in dark. The mixture was then diluted to 50 mL using methanol to attain the final ABTS dilution. In a 96-well plate (n = 6), 190 µL of the freshly produced ABTS reagent was combined with 10 µL of the sample, and standard separately, and the preparation was placed in an incubator for 30 min in dark at RT. At the end of incubation, the drop in the intensity of ABTS color was observed at 734 nm wavelength using a microplate reader (FluoStar Omega, Ortenberg, Germany). The scavenging activity of the plant extract was calculated as Trolox equivalent/g (TE) of the plant’s extract using the generated calibration curve of the standard antioxidant compound, Trolox (supplementary file).
2.6.3 ORAC assay
The Oxygen Radical Absorbance Capacity (ORAC) assay was performed using the technique of Liang et al (Liang et al., 2014), with some modifications. Briefly, 10 µL of the prepared plant’s extract sample was incubated with 30 µL of fluorescein (100 nM) for 10 min at 37 °C. For background detection, the measurements of fluorescence (485 EX, 520 EM, nm) were conducted for three rounds (90 sec each). Following that, 70 µL of newly prepared 2,2′-Azobis(2-amidinopropane) dihydrochloride (AAPH) (300 mM) reagent was immediately added to each well. The measurements of florescence (485 EX, 520 EM, nm) were carried out for 60 min (40 cycles, each 90 sec). The anti-ORAC activity of the plant extract was calculated as Trolox equivalent/g (TE) of the plant’s extract using the linear regression curve of Trolox standard as the antioxidant compound, Trolox (supplementary file).
2.6.4 FRAP assay
The FRAP test was carried out in microplates according to the technique described by Benzie et al with certain modifications (Benzie and Strain, 1996). A freshly prepared TPTZ reagent (300 mM, acetate buffer pH 3.6), 10 mM TPTZ in 40 mM HCl, and 20 mM FeCl3, in a ratio of 10:1:1 v/v/v, respectively). In a 96-well plate, 190 µL of freshly prepared TPTZ reagent was combined with 10 µL of the test sample, and the mixture was kept at RT for 30 min in dark. At the end of the incubation, the resultant blue color was detected at 593 nm wavelength using a microplate reader (FluoStar Omega, Ortenberg, Germany). The activity of the extract was calculated as mg Trolox equivalent per gram (TE) of the plant's dried extract using the FRAP–Trolox calibration curve (supplementary file).
2.7 Antiproliferative assay
The SRB assay was used to investigate cancer cells viability in presence of the plant’s extract. An aliquot of 100 μL suspension of the cells (5x103 cells) were seeded into 96-well plates, and were kept incubated for 24 hr. Another aliquot of 100 μL media comprising T. linearifolia extract at various concentrations were delivered to the cells. Following 72 hr of exposure to the plant’s extract, the cells were maintained by substituting the media with 150 μL of 10 % TCA, and kept in an incubator at 4 °C for an hr. After discarding the TCA, the cells were rinsed five times with deionized water. Aliquots of 70 μL SRB solution (0.4 %, w/v) were added, and allowed to stand at RT for 10 min in dark, and plates washed with 1 % acetic acid. Afterwards, 150 μL of TRIS (10 mM) was added to dissolve the protein-bound SRB stain. A BMG LABTECH®- FLUOstar Omega microplate reader (Ortenberg, Germany) was used to measure the absorbance at 540 nm wavelength (Skehan et al., 1990).
2.8 Antimicrobial assay
2.8.1 Disk diffusion assay
Antibacterial potentials were measured using a modified Kirby-Bauer disk diffusion susceptibility test (Hudzicki, 2009). Disks containing the plant’s extract (50 µL) were prepared, and attached to the surface of the agar plates, inoculated with the test bacterium (Escherichia coli ATCC 8739, Bacillus subtilis ATCC 6633, Staphylococcus aureus ATCC 29213,
Salmonella typhimurium ATCC 14028, Candida albicans ATCC 10231, and Saccharomyces cerevisiae ATCC 9763). The negative control was made of the same volume of DMSO, while the positive control was standard disks of the standard antibacterial agent(s). The plates were kept inverted in the incubator for one day at 37 °C. After incubation, the plates were inspected to measure the zones of inhibition. Diameters less than 5 mm were considered as having no effects. All the experiment was triplicated.
2.8.2 Broth microdilution method
The broth microdilution assay was used to determine the plant’s extract minimal inhibitory concentration (MIC) required for the growth inhibition of the microorganisms under test. Serial dilutions of the plant’s extract (10 mg/mL) were prepared in 96-well plates using 100 µL of the extract, and 100 µL of the Muller Hinton Broth (MHB). Afterwards, 100 µL of the microbial inoculums were added to each well with final concentration of 5.0 X 105 CFU/mL. In addition, another 100 µL from each microbial organism suspensions were diluted, and cultured externally to confirm inoculum density. A growth control well containing inoculated broth, without extract, and a negative control well containing only the broth without extract and microbial organism were added to each sample plate. The plates were incubated at 37.0 ± 1.0 °C for 24.0 ± 2.0 hr, except for Bacillus subtilis, which was incubated at 30.0 ± 1.0 °C for 24 hr. After incubation, the plates were removed from the incubator, and placed in dark to check the microbial growths, wherein the turbidity of the solution was an indication of the microbial growth, thereby confirming the validity of the test.
2.9 Docking method
Molecular dockings of the secondary metabolite compounds identified from the plant’s extract were run by the molecular operating environment (MOE) 2019.012 suite against epidermal growth factor receptor (EGFR). X-rays diffraction (XRD) data-domains of the EGFR complexed with BDBM50432373 ligand was obtained from the Protein Data Bank (https://www.rcsb.org/structure/3W33) (Kawakita et al., 2013), and protein and ligands preparations were performed by MOE, including gas tier charges through the MMFF 94x force-field, by adding hydrogen atoms, removing the water molecules, 3D-protonation of the structure, and minimizing of the protein structure. The screened metabolites were subjected to partial charges addition, and energy minimizations (El-Shershaby et al., 2021a; Ma et al., 2021). The docking site was chosen to be the co-crystallized ligand site. Triangle matcher, and London dG were selected as the placement, and the scoring methodology, respectively, which was able to create 100 poses. Moreover, the rigid receptor, and GBVI/WSA dG were selected as the placement, and the scoring methodology to extract the best 10 poses were produced from 100 poses for each docked molecule (El-Shershaby et al., 2021b). The output from MOE software was further visualized by Discovery Studio® 4.0 software for ligand–protein interactions. ligand (W19).
2.10 Statistical analysis
Results were presented as mean ± SD (standard deviation). Statistical analysis was conducted by applying Student’s t-test and P-values less than 0.05 were recorded as significant. Correlations between acquired data were analyzed using the correlation coefficient statistical tool in the MS Excel software.
3 Results and discussion
3.1 Total phenolics and flavonoids contents in T. Linearifolia
The quantitative analysis results of the phenolics and flavonoid contents in the plant extract are presented in Table 1, which revealed the presence of 60.67 mg/g GAE, and 26.79 mg/g RE, of the phenolics, and the flavonoid contents, respectively. The results were consistent with the LC-MS analysis findings (Table 2), showed 5.19 %, and 23.5 % of the phenolic acids, and flavonoids, respectively, which were calculated relative to the total peaks area in the LC chromatogram. The results presented in Table 2 exhibited considerable numbers of phenolic acids and flavonoid contents, which may have contributed to the tested and confirmed biological activities of the plant’s extract as antimicrobial, cytotoxic, and antioxidant nature. There are no published information on the presence of phenolics and flavonoid contents in T. linearifolia. Nonetheless, compared to other Thymbra species, the current findings showed lesser quantities of the TFC. Another Thymbra species, Thymbra spicata, is reported to contain 201.4–250 mg/g GAE as the phenolics content equivalent, which is much higher than the current findings at 60 mg/g GAE for the phenolic contents from T. linearifolia (Diab et al., 2022; Eruygur et al., 2017; Khalil et al., 2019). The current findings on the flavonoid contents also revealed that T. linearifolia has lower quantities of flavonoid compounds in comparison to other well-known and well investigated Thymbra species, T. spicata. The T. spicata was shown to possess 78.17 mg QE as the flavonoid contents’ equivalent quantity in the plant species growing in Turkey. However, the contents of the flavonoids were higher in the current study compared to the contents in T. spicata growing in Lebanon at 9.04 mg QE (Khalil et al., 2019) as against 26.76 mg RE in the current study for T. linearifolia, which reflected the inter-species variations, owing to the genetic makeup, and probably also due to the environmental factors of the geographical significance as well as other biotic and abiotic factors. However, the compared standards of the QE (Quercetin Equivalents) and the RE (Rutin Equivalents) can have qualitative measurements effects owing to the structural resemblance of the nearest types (both with 3′,4′,5,7-tetra hydroxy substitutions), and presence of glycosidal (rutin, 3-O-di-glycoside), and non-glycosidal (quercetin) entities between the two flavonoids, and for which no conclusive data on quantitation difference is available. *Compounds identification is based on their mass fragmentation patterns and the literature mass spectral data.
Test
TPC
TFC
DPPH
ABTS
ORAC
FRAP
MC
Units
mg/g, GAE
mg/g, RE
IC50, μg/mL
µM, TE
μM, EE
Results
60.67 ± 2.4
26.79 ± 1.53
34.48 ± 1.23
2328.35 ± 103.20
3824.13 ± 210.48
479.69
± 43.8519.47 ±
1.79
Serial
Rt (minutes)
Constituent Identity
MS: (m/z):
[M−H]-/ [M]-
Abundance (%)
MS/MS (m/z)
1.
0.99
Quinic acids
191.0246
0.07
162, 111, 85, 34 (El-Shazly et al., 2022)
2.
2.36
Protocatechuic acid glucoside
315.0798
0.13
153, 152, 109, 108 (El-Shazly et al., 2022)
3.
3.75
4-Hydroxy benzoic acid
137.0277
0.06
108, 94, 93 (Hossain et al., 2010)
4.
9.06
Gallocatechin isomer
305.0764
2.94
225 (Taamalli et al., 2015)
5.
9.65
Vicenin-2 (apigenin-6,8-di-C-β-D-gluco-pyranoside)
593.1633
0.34
503, 473, 383, 353 (Pontes et al., 2022)
6.
10.40
Cyanidin-3-O-rutinoside
595.1782
2.62
287 (Ross et al., 2007)
7.
10.49
Unknown, Cyanidin derivative
658.1761
0.71
595, 287
8.
10.59
Cyanidin-3-O-glucoside
449.1196
0.09
287 (Ross et al., 2007)
9.
10.72
Taxifolin
303.0572
1.22
285, 217, 151, 125 (El-Shazly et al., 2022)
10.
10.92
Keampferol-3-O-rutinoside
593.1630
0.81
284, 285 (Wojdyło and Nowicka, 2019)
11.
11.04
Keampferol-3-O-glucoside
447.1038
0.14
284, 285 (Wojdyło and Nowicka, 2019)
12.
11.09
luteolin-3-O-glucuronide
461.0840
0.11
285 (Hossain et al., 2010)
13.
11.11
Pelargonidin-3-O- rutinoside
579.1828
2.36
272, 271 (Kajdžanoska et al., 2010)
14.
11.34
Pelargonidin-3-O-glucoside
433.1247
0.04
271 (Kajdžanoska et al., 2010)
15.
11.44
Apigenin-7-O-rutinoside
577.1688
0.28
269 (Hossain et al., 2010)
16.
11.45
Isomeric to pelargonidin-3-O- rutinoside
580.1891
1.47
579, 271
17.
11.49
Quercetin-3-O-rutinoside
609.1939
1.20
301, 302, 303 (Wojdyło and Nowicka, 2019)
18.
11.56
Syringic acid hexoside
360.0877
1.38
198, 197, 161, 135, 133, 123 (Fang et al., 2002)
19.
11.64
Rosmarinic acid
359.0841
2.54
198, 197, 179, 161, 135 (Hossain et al., 2010)
20.
11.66
Syringic acid
197.0507
0.20
179, 135 (Hossain et al., 2010)
21.
11.69
Caffeic acid
179.0397
0.08
161, 135 (Hossain et al., 2010)
22.
12.45
Salvianolic acid
718.1662
0.36
519 (Irakli et al., 2021)
23.
12.50
Luteolin di hexoside
609.1386
0.11
447, 285 (Choi et al., 2018)
24.
12.63
Cyanidin-3-O-sophoroside
611.1541
0.07
287 [M−H], 449 [M−glu] (Ross et al., 2007b)
25.
13.27
Luteolin
285.0466
0.99
133, 175, 217 (Mohammed et al., 2022)
26.
13.64
Hydroxy benzoic acid-O-hexoside
299.0629
0.37
137, 119 (Hossain et al., 2010)
27.
13.94
Kaempferol
285.0480
0.23
271, 119 (Mohammed et al., 2022)
28.
14.22
Apigenin
269.0510
2.00
225, 151, 171 (Taamalli et al., 2015)
29.
15.28
Thymol
149.0645
0.06
150, 135 (Hossain et al., 2010)
30.
16.52
Methyl apigenin
283.0681
0.24
269, 268 (Hossain et al., 2010)
31.
18.26
Dimethoxy luteolin
313.1867
2.97
299, 284, 283, 254 (Fathoni et al., 2017)
32.
21.31
Pinobanksin-3-O-acetate
313.1865
2.56
181, 180, 165, 151 (Pellati et al., 2011)
33.
23.00
Ursolic acid
455.3614
2.63
Total relative percentage
31.38
3.2 LC-MS profiling of the plant’s ethanolic extract
The LC-MS analysis was performed to identify the phenolic acids and flavonoids present in the plant’s extract. Out of hundreds of the peaks in the negative and positive modes chromatograms of the plant’s extract, only thirty-three compounds were identified based on the known MS (mass) spectral data and the mass fragmentation patterns. The molecular ion peak of the each identified constituent, its mass fragmentation pattern, and the MS/MS fragments were compared to the literature data. The analysis showed the presence of both the phenolic acids, and flavonoids in considerable quantities with a relative percentages (RP) of 28.75 %, as calculated for a total of 32 phenolics and flavonoid compounds. The analysis revealed the presence of nine phenolic acids, i.e., quinic acid, protocatechuic acid glucoside, 4-hydroxybenzoic acid, syringic acid hexoside, rosmarinic acid, syringic acid, caffeic acid, salvianolic acid, and hydroxy benzoic acid-O-hexoside, with a total relative percentage of these acids calculated at 5.19 %, when calculated based on the total peaks’ areas in the negative ion mode chromatogram of the plant’s extract. These phenolic acids were identified based on their characteristic mass fragments in the mass spectrum. For example, the quinic acid was identified based on the presence of the molecular ion peak at m/z 191.0246 [M−H], and fragment ions mass peaks at m/z 162, 111, 85, and 34, which were reported as characteristic fragments for the compound (El-Shazly et al., 2022). Similarly, the presence of fragment ions masses at m/z 108, 94, and 93, with peak at m/z 137.0277 [M−H] for the molecular ion peak led to the identification of 4-hydroxy benzoic acid (Hossain et al., 2010). In addition, the most abundant phenolic acid in the extract, rosmarinic acid, being one of the most abundant phenolic compounds in the Lamiaceae family, was identified through the presence of the molecular ion peak at m/z 359.0841, and fragment masses at m/z 198, 197, 179, 161, and 135 (Hossain et al., 2010). The phenolic acid glycosides, syringic acid hexoside (RP, 1.38 %), and hydroxy benzoic acid-O-hexoside (RP, 0.37 %) were identified through the presence of the de-hexoside fragments mass at m/z 197 [syringic acid-(H + hexose)], and mass at m/z 137 [hydroxy benzoic acid-(H + hexose)], respectively (Fang et al., 2002; Hossain et al., 2010). These phenolic glycosides were also identified based on their mass fragmentation patterns of their respective glycoses (Fang et al., 2002; Hossain et al., 2010). The mass-based identification was well supported from the known mass spectral data of these compounds and ruled out the possibilities of other similar mass compounds based on their decimal four place HR-MS (high resolution mass) molecular ion spectra. Besides the phenolic acids, twenty-two flavonoids of different classes, i.e., flavonol, flavone, dihydroflavonol, catechins, and anthocyanins were identified. Among all the identified flavonoids, the gallocatechin isomer (2.94 %), cyanidin-3-O-rutinoside (2.62 %), taxifolin (1.22 %), keampferol-3-O-rutinoside (0.81 %), pelargonidin-3-O-rutinoside (2.36 %), pelargonidin-3-O-rutinoside (isomeric) (1.47 %), quercetin-3-O-rutinoside (1.20 %), luteolin (0.99 %), apigenin (2.00 %), dimethoxy luteolin (2.97 %), and pinobanksin-3-O-acetate (2.56 %) were found as the major flavonoids in the plant extract (Table 2). A total percentage of the identified flavonoids of the identified compounds from the plant extract was calculated at 23.5 %, wherein the anthocyanins were the most abundant flavonoids, with a relative percentage of 7.36 %. All the identifications of the T. linearifolia flavonoids were based on the observations of the molecular ions and fragmentation ions peaks for each constituent compounds, and which were consistent with the reported mass values of the fragments of the corresponding compounds in their mass spectra. The anthocyanins, for example, were identified based on the mass values of the aglycones fragments presence for each compound. The presence of a molecular ion peak at m/z 595.1782 [M−H]-, and the fragment’s mass peak at m/z 287 [M-(H + rutinoside)]- revealed the detachment of the rutinoside moiety [308 atomic mass units (amu), and led to the identification of the compound as cyanidin-3-O-rutinoside (Fig. 1, LC, Rt 10.40 min). However, cyanidin-3-O-glucoside (Rt 10.59 min) showed the molecular ion peak at m/z 449.1196 [M−H]- and a fragment peak at m/z 287 [M-(H + glu)]- after the removal of 162 amu, which revealed the detaching of the glucoside moiety, and supported the identification of the compound (Ross et al., 2007). The keampferol-3-O-rutinoside (Fig. 1, Rt 10.92) showed a molecular ion peak at m/z 593.1630 [M−H]- and mass fragments at m/z 285, after removal of 308 amu, which revealed the detachment of the rutinoside moiety, and the identity of the compound (Wojdyło and Nowicka, 2019). The pelargonidin-3-O- rutinoside (Fig. 1, Rt 11.11) was identified by the presence of m/z 579.1828 [M−H]- and mass fragments at m/z 271, thereby indicating the aglycone part of the compound, and detachment of the rutinoside glycosidal part (Kajdžanoska et al., 2010). The quercetin-3-O-rutinoside was identified by the presence of molecular ion peak at m/z 609.1939 [M−H]-, and mass fragments at m/z 301 (Fig. 1), also indicating the presence of the aglycone part of the glycoside compound, together with the detachment of the rutinoside, the glycone moiety (Wojdyło and Nowicka, 2019). The identified flavonoid aglycones, i.e., taxifolin, luteolin, kaempferol, apigenin, methyl apigenin, and dimethoxy luteolin, were identified based on the presence of their corresponding molecular ion peaks, and MS/MS fragmentations of each compound as compared with the literature (El-Shazly et al., 2022; Fathoni et al., 2017; Hossain et al., 2010; Mohammed et al., 2022; Taamalli et al., 2015).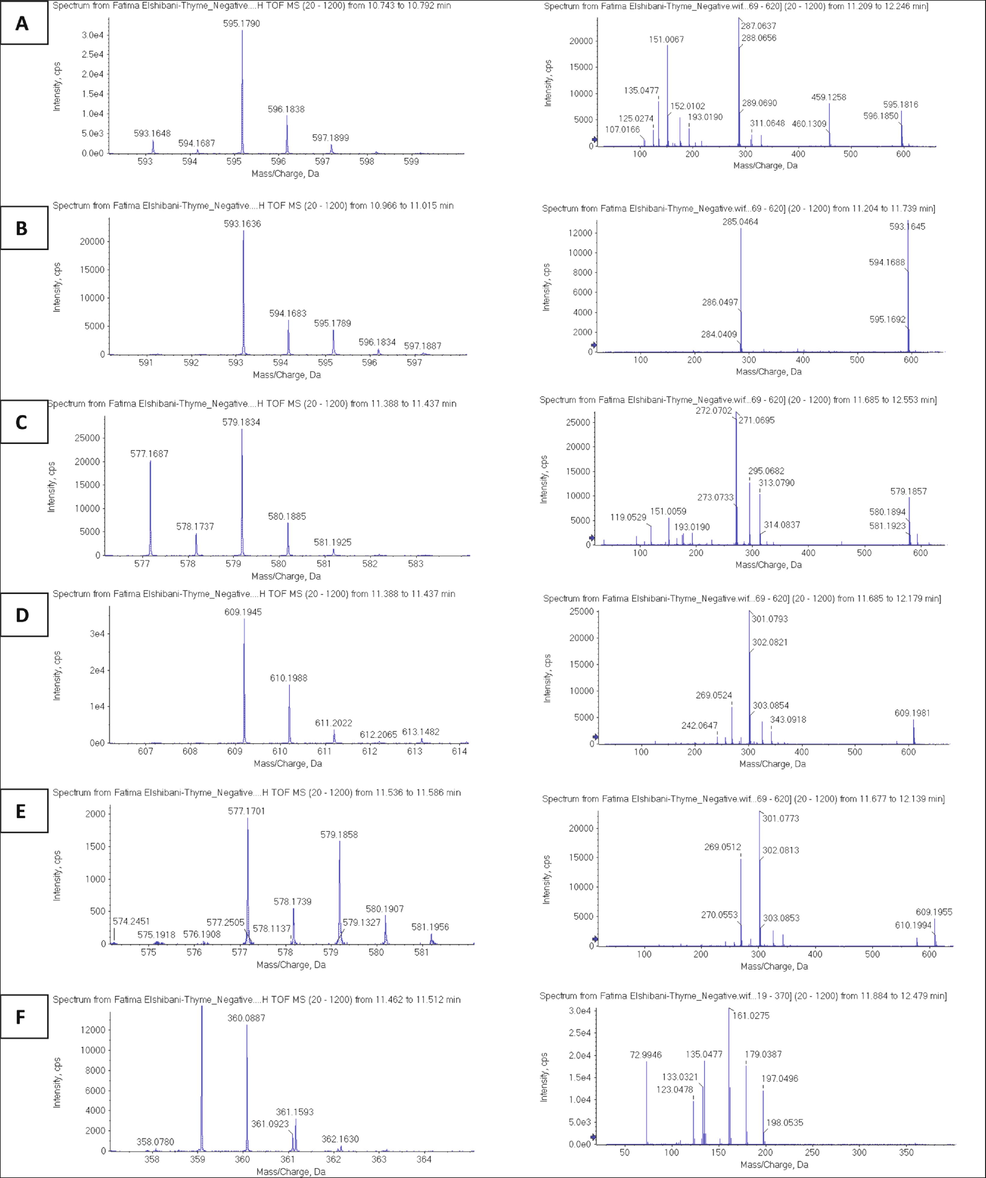
MS-MS based parent and daughter mass ions for the major identified glycosides, cyanidin-3-O-rutinoside (A), keampferol-3-O-rutinoside (B), pelargonidin-3-O-rutinoside (C), quercetin-3-O-rutinosid (D), apigenin-7-O-rutinoside (E), syringic acid hexoside (F).
Some of these major identified compounds have also been detected in other Thymbra species plants’ extracts as well. For example, rosmarinic acid, syringic acid, rutin, quercetin, luteolin, gallocatechin, and apigenin were identified in Thymbra spicata (Bener, 2019; Diab et al., 2022). In addition, glycosidal flavonoids, which were identified as the major class of compounds present in T. linearifolia, have also been found in several other Thymbra species (Mohammadi, et al., 2019). The quantitative analysis by the colorometric assays, and LC-MS based identification has fewer points in common with respect to the identification of the types of the products, i.e., phenolics and flavonoids, and the relative percentages and structural identity of the constituents’ in the plant’s extract. However, it substantiated the presence of these classes of compounds in both the analyses, and confirmed the presence of these compounds in the plant’s extract for the purpose of phytochemical profiling, and comparative studies with the plants in the Thymbra genus.
3.3 Antioxidant activity of T. linearifolia
The antioxidant activity of T. linearifolia was assessed by different assay methods. The in vitro assay methods were selected to evaluate the free radicals capturing activity of the plant extract, which included DPPH, ABTS, and ORAC. The additional methods were the ferric-reducing (FRAP), and metal chelating (MC) activities of the plant’s ethanolic extract. The results shown in Table 1 exhibited marked antioxidant activity of the plant, which is consistent with the findings of the LC-MS and TPC and TFC phytochemical analysis. It revealed the presence of considerable quantities and different constituents from the phenolics and flavonoids class of products in the plant extract. A higher proportion of the known antioxidant compounds were detected by the LC-MS analysis. The encountered anthocyanins have been reported to exert antioxidant activity with several mechanisms, including free radical capturing (Abdellatif et al., 2021; Miguel, 2011), reducing (García-Alonso et al., 2004), and metal chelating potentials (Zhao and Yuan, 2021). Certain other Thymbra species have also been reported for their potential antioxidant activity, and the presence of different types of phenolics and flavonoids (Bower et al., 2014; Gedikoğlu et al., 2019). The DPPH scavenging activity of T. spicata has been reported (Gumus et al., 2011), and it revealed higher DPPH scavenging activity (IC50 value at10.52 μg/mL), as compared to the currently studied, T. linearifolia (IC50 at 34.48 μg/mL).
3.4 Cytotoxic activity of T. Linearifolia extract
The ethanolic extract of T. linearifolia showed substantial anti-proliferative activity against all tested cancerous cell lines, MCF-7, HepG2, and Panc-1 with IC50 values at 24.023, 22.94, and 33.3 µg/mL, respectively. The antiproliferative effects of the plant extract was compared with the standard, DOX (doxorubicin), at the highest dose-level concentration of 100 µg/mL (Table 3). The results revealed a substantial reduction in cell proliferations when treated with the plant’s extract, i.e., the breast cancer (MCF-7), and hepatic cellular carcinomas (HepG2) cell lines' viabilities were reduced by 98.29 % by the plant extract, as compared to 73.05 %, and 76.98 % reductions of cell viability, respectively, when treated with the same dose of the DOX. These marked findings of T. linearifolia cytotoxicity is of immense potential for further studies, and holds promise for development of the anticancer activity constituent(s) from the plant.
Cell lines
MCF-7
HepG2
PANC-1
T. linearifolia extract
1.71 ± 0.57
1.71 ± 0.93
6.92 ± 2.57
DOX (Doxorubicin)
26.95 ± 0.72
23.02 ± 0.31
18.93 ± 0.36
Assumptively, the plant's potential cytotoxicity can be attributed to the presence of phenolic acids and flavonoids, which are well-known for their anticancer activity (Ali and Neda, 2011; Bonta, 2020; Mohammed, 2020). Some of the major identified phenolics, such as rosmarinic acid, and syringic acid, have previously been reported to have anticancer activity (Gheena and Ezhilarasan, 2019; Hossan et al., 2014). The latter has been linked to specific mechanisms of apoptosis in HepG2 cells, including downregulation of Bcl-2 gene expressions, and upregulation of caspases-3 and caspase-9, cytochrome c, Apaf-1, Bax, and p53 related pathways and components (Gheena and Ezhilarasan, 2019). Anthocyanins, as one of the major classes of compounds in the extract, have been reported for their potential anticancer activity through different mechanisms, including controlling the viability and proliferation of cancerous cells, thereupon arresting the cell cycle, and inducing the apoptosis (de Arruda Nascimento et al., 2022). Anthocyanins have also been reported for their specific, and critical roles in supporting liver cells, and suppressing the proliferation of hepatocellular carcinoma, and HepG2 cells (Mohammed and Khan, 2022). Compared to the other Thymbra species, the T. spicata, the current findings amply indicated that the T. linearifolia possessed better cytotoxic effects against the breast cancer cell line, MCF-7, with an IC50 value of 24.02 µg/mL, as compared with the 340 µg/mL for the T. spicata (Eruygur et al., 2017). These activities may have resulted from individual compounds, or may have been an end outcome of the synergistic actions of these known, together with unknown/unidentified compounds.
3.5 Antimicrobial activity of T. linearifolia
The results in Table 4 indicated moderate to weak antibacterial activity of T. linearifolia against all tested microbial strains. The extract showed very weak activity against Gram-negative bacteria, E. coli ATCC 8739, and S. typhimurium ATCC 14028, with IZD values equaling to 5 and 6 mm, and MIC values at 1.25 and 2.74 µg/mL, respectively. However, a moderate antimicrobial effect was exhibited by the plant’s extract against Gram-positive, B. subtilis ATCC 6633, and Staphylococcus aureus ATCC 29213, as well as against the Gram-negative S. cerevisiae ATCC 9763, which showed IZD values near to 15 mm, and MIC values at 0.59, 0.50, and 0.86 µg/mL, respectively. The antimicrobial results also indicated substantial antifungal activity for the T. linearifolia ethanolic extract, that showed 17 mm IZD, and MIC value at 0.63 µg/mL against C. albicans ATCC 10231. IZD, inhibition zone diameter in millimeters; MIC, minimal inhibitory concentration.
Microbial Organism
IZD (mm)
MIC
(µg/mL)
Escherichia coli ATCC 8739
5 mm
1.25
Bacillus subtilis ATCC 6633
15 mm
0.59
Staphylococcus aureus ATCC 29213
15 mm
0.50
Salmonella typhimurium ATCC 14028
6 mm
2.74
Saccharomyces cerevisiae ATCC 9763
15 mm
0.86
Candida albicans ATCC 10231
17 mm
0.63
As compared with reported antimicrobial activity of T. spicata, which showed relatively higher activity against E. coli (IZD 13 mm, as compared to 5 mm of the current findings), Staphylococcus aureus (IZD 35 mm, as compared to 15 mm of current findings), S. typhimurium (IZD 11 mm, as compared to 6 mm of the current investigation). The significant variations in the antimicrobial activity can also be attributed to the extraction methods used for the extraction of T. spicata by microwave-assisted extraction (Gedikoğlu et al., 2019). This notion of method of extraction based differences could be supported by reported antimicrobial data obtained by Sengun, et al. for T. spicata that showed lower IZD value for the plant extract against Staphylococcus aureus at 15 ± 7 mm (Sengun et al., 2021).
3.6 Docking study for cytotoxicity
To predict the cytotoxicity levels of the constituent compounds, an in-silico modeling study was run against EGFR target ligand, wherein the inhibition of EGFR ultimately led to the blockade of the growth pathways, indicating the promising anti-cancer potentials of the compounds (Kawakita et al., 2013). The lower binding energy was an indication of a higher binding efficiency. The docking scores of the screened products are listed in Table 5. The BDBM50432373 was used as a reference compound for EGFR inhibitions comparisons. It was found that the flavonoid glycosides, i.e., cyanidin-3-O-rutinoside, keampferol-3-O-rutinoside, pelargonidin-3-O- rutinoside, and quercetin-3-O-rutinoside showed higher binding affinities with docking scores ranging between −9.9 kcal/mol to −10.5 kcal/mol, which were very close to the referral ligand, BDBM50432373. In addition, the flavonoids. i.e., gallocatechin, taxifolin, luteolin, apigenin, dimethoxy luteolin, and pinobanksin-3-O-acetate showed favorable binding affinity against the EGFR, and the docking scores were between −8.2 kcal/mol to −8.9 kcal/mol. The in-silico ligand - protein interactions also showed that the screened compounds which all are of secondary metabolic origins in the plant, participated in the formation of H (hydrogen)-bonds with the amino acids of the EGFR protein, which included Lys745, Met793, Asn842, and Asp855. The compounds that were involved in the hydrophobic interactions with the amino acids Leu718, Phe723, Val726, Ala743, Thr790, Gly796, Leu844, Thr854) through pi- alkyl interactions, and/or pi-sigma interactions, were also found out (Fig. 2). These predictive findings suggested the strong anticancer activities of the individual compounds, as well as combined effects of different other compounds of phenolics and flavonoids nature from the extract, which has been currently in silico modeled, to be of synergistic nature, or otherwise in their activity elicitations. However, possibility of unidentified constituent(s) participating in the experimentally observed anticancer activities cannot be ruled out.
Compound
Binding Scores (kcal/mol)
Interacting Amino Acid Residues
Gallocatechin
−8.2
Leu718, Val726, Ala743, Lys745, Leu844, Asp855,
Cyanidin 3-O-rutinoside
−10.5
Val726, Ala743, Lys745, Thr790, Met793, Arg841, Asn842, Thr854
Taxifolin
−8.6
Ala743, Lys745, Leu788, Met793
Keampferol-3-O-rutinoside
−10.1
Leu718, Val726, Lys745, Met793, Gly796, Asn842, Leu844, Thr854, Asp855
Pelargonidin-3-O- rutinoside
−10.0
Leu718, Phe723, Val726, Ala743, Thr790, Asp837, Arg841, leu844, Thr854
Quercetin-3-O-rutinoside
−9.9
Leu718, Ala743, Lys745, Thr790, Met793, Arg841, Leu844, Thr854, Lys875
Luteolin
−8.9
Leu718, Val726, Ala743, Lys745, Met793
Apigenin
−8.8
Leu718, Val726, Ala743, Lys745, Met793, Gly796
Dimethoxy luteolin
−9.0
Lys745, Met766, Cys775, Thr854, Leu858
Pinobanksin-3-O-acetate
−8.4
Leu718, Val726, Ala743, Lys745, Met793, Leu844
BDBM50432373
−10.5
Ser720, Val726, Ala743, Lys745, Leu788, Met793, Leu844, Phe856
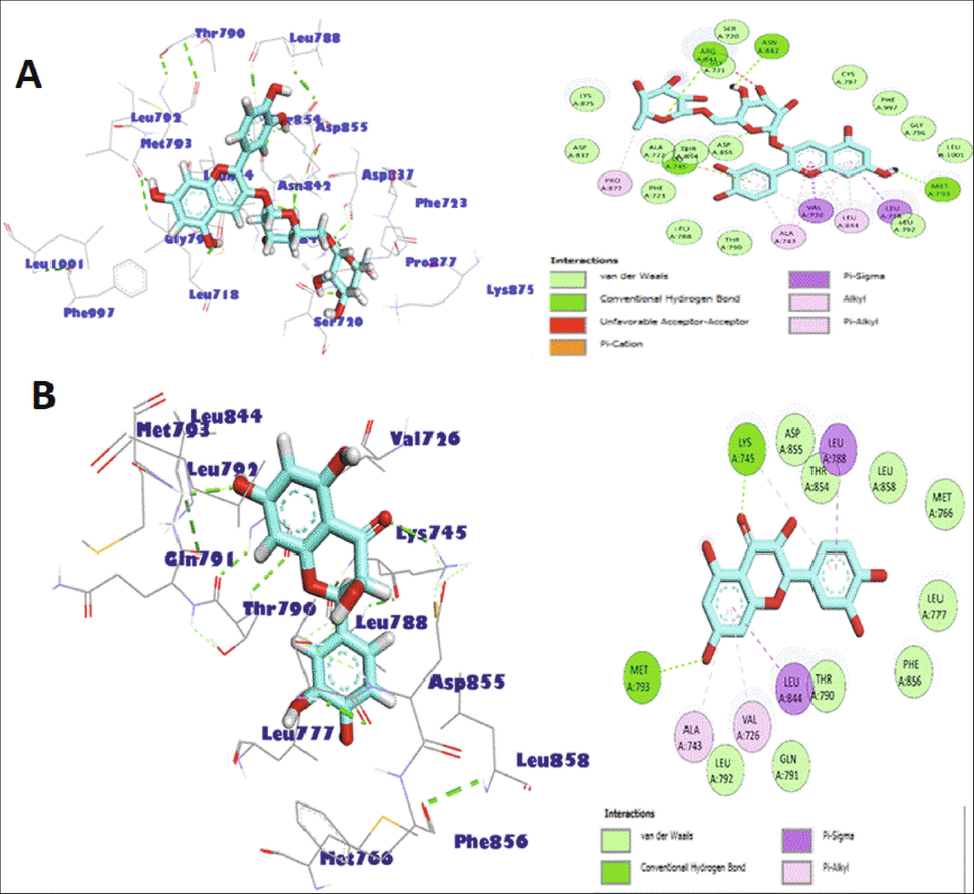
Molecular interactions of cyanidin-3-O-rutinoside (A), and taxifolin (B) with EGFR. The H-(hydrogen) bonds are represented as green dashed lines.
4 Conclusion
The current findings are the first report for the Lamiaceae species plant, Thymbra linearifolia, especially native to the Mediterranean region of North African nation, Libya, and confined to its specific location in the Jabel Al-Akhdar area. The current investigations, up to larger extent, revealed the importance of the plant, and also compared the plant’s profile with the other abundant species, T. spicata, in other geographic locations of the world, in both the phytochemical constituents’ presence, and biological activity aspects of the antimicrobial and antioxidant activities, and the strong cytotoxic properties. The phytochemical investigations of the plant’s ethanolic extract constituents, achieved through the LC-MS and color-based assays, revealed the presence of considerable quantities of the phenolic acids and flavonoids, and thereby indicated the abundance of flavonoids aglycones and their glycosides, as the major identified constituents. The abundant presence of phenolic acids, of which syringic and rosmarinic acids were major products, were also confirmed. The docking scores of T. linearifolia constituents were at a minimum against the EGFR ligand, which indicated the strong binding affinities and least energy requirements. In the light of the strong cytotoxic properties observed, detailed chemical analysis, and further biological activity of the crude extract at pre-clinical stages, and cytotoxic bioactivity evaluations of the individual compounds isolated from the plant’s extract are recommended and planned.
Funding
This research received no external or internal funding.
Acknowledgments
Researchers would like to thank the Deanship of Scientific Research, Qassim University for funding publication of this project.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Anthocyanins-rich pomegranate cream as a topical formulation with anti-aging activity. J. Dermatolog. Treat.. 2021;32:983-990.
- [Google Scholar]
- Flavonoids and phenolic acids: Role and biochemical activity in plants and human. J. Med. plants Res.. 2011;5:6697-6703.
- [Google Scholar]
- The hydrophilic and lipophilic contribution to total antioxidant activity. Food Chem.. 2001;73:239-244.
- [Google Scholar]
- A rapid microtitre plate Folin-Ciocalteu method for the assessment of polyphenols. Open Life Sci.. 2013;8:48-53.
- [Google Scholar]
- Modeling and optimizing microwave-assisted extraction of antioxidants from Thymbra Spicata L. and characterization of their phenolic constituents. Food Sci. Biotechnol.. 2019;28:1733-1745.
- [Google Scholar]
- The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: the FRAP assay. Anal. Biochem.. 1996;239:70-76.
- [Google Scholar]
- Dietary phenolic acids and flavonoids as potential anti-cancer agents: current state of the art and future perspectives. Anti-Cancer Agents Med. Chem. (Formerly Curr. Med. Chem. Agents). 2020;20:29-48.
- [Google Scholar]
- Bioactive compounds from culinary herbs inhibit a molecular target for type 2 diabetes management, dipeptidyl peptidase IV. J. Agric. Food Chem.. 2014;62:6147-6158.
- [Google Scholar]
- Delimitation and revision of the genus Thymbra (Lamiaceae) Phytotaxa. 2018;369:15-27.
- [Google Scholar]
- Biological activity, and volatile and phenolic compounds from five Lamiaceae species. Flavour Fragr. J.. 2021;36:223-232.
- [Google Scholar]
- LC-MS/MS profiling of polyphenol-enriched leaf, stem and root extracts of Korean Humulus japonicus Siebold & Zucc and determination of their antioxidant effects. Biomed. Chromatogr.. 2018;32(e4171):1-8.
- [Google Scholar]
- In vitro anticancer properties of anthocyanins: a systematic review. Biochim. Biophys Acta (BBA)-Reviews Cancer 2022:188748.
- [Google Scholar]
- Chemical composition and bioactivity of essential oils from flower and fruit of Thymbra capitata and Thymus species. J. Food Sci. Technol.. 2017;54:1857-1865.
- [Google Scholar]
- Influence of simulated in vitro gastrointestinal digestion on the phenolic profile, antioxidant, and biological activity of Thymbra spicata L. Extracts. Antioxidants. 2022;11(1778):1-18.
- [Google Scholar]
- Antioxidant activity of some Algerian medicinal plants extracts containing phenolic compounds. Food Chem.. 2006;97:654-660.
- [Google Scholar]
- LC-MS/MS profiling, antibiofilm, antimicrobial and bacterial growth kinetic studies of Pluchea dioscoridis extracts. Acta Chromatogr.. 2022;34:338-350.
- [Google Scholar]
- The antimicrobial potential and pharmacokinetic profiles of novel quinoline-based scaffolds: synthesis and in silico mechanistic studies as dual DNA gyrase and DHFR inhibitors. New J. Chem.. 2021;45:13986-14004.
- [Google Scholar]
- From triazolophthalazines to triazoloquinazolines: A bioisosterism-guided approach toward the identification of novel PCAF inhibitors with potential anticancer activity. Bioorg. Med. Chem.. 2021;42(116266):1-16.
- [Google Scholar]
- Estimation of phenolic content, flavonoid content, antioxidant properties and alpha-amylase inhibitory activities of Capparis spinosa L. J. Pharmacognocy Photochem.. 2020;9:24-28.
- [Google Scholar]
- Green synthesis of silver nanoparticles using Thymbra spicata L. var. spicata (zahter) aqueous leaf extract and evaluation of their morphology-dependent antibacterial and cytotoxic activity. Artif. Cells Nanomed. Biotechnol.. 2018;46:150-158.
- [Google Scholar]
- A study on the antioxidant, antimicrobial and cytotoxic activity of Thymbra spicata L. var. spicata ethanol extract. Cumhur. Med. J.. 2017;39:531-538.
- [Google Scholar]
- Phytotherapy: an introduction to herbal medicine. Prim. Care Clin. Off. Pract.. 2017;44:217-227.
- [Google Scholar]
- LC/MS/MS characterization of phenolic constituents in dried plums. J. Agric. Food Chem.. 2002;50:3579-3585.
- [Google Scholar]
- DPPH Free Radical Scavenging Activity of Two Extracts from Agelanthus dodoneifolius (Loranthaceae) Leaves. Int. J. Toxicol. Pharmacol. Res. 2016;8:29-34.
- [Google Scholar]
- Fathoni, A., Saepudin, E., Cahyana, A.H., Rahayu, D.U.C., Haib, J., 2017. Identification of nonvolatile compounds in clove (Syzygium aromaticum) from Manado, in: AIP Conference Proceedings. AIP Publishing LLC, p. 30079.
- Volatiles from Thymbra and Thymus species of the western Mediterranean basin, Portugal and Macaronesia. Nat. Prod. Commun.. 2010;5:1466-1476.
- [Google Scholar]
- Antioxidant and cellular activities of anthocyanins and their corresponding vitisins a studies in platelets, monocytes, and human endothelial cells. J. Agric. Food Chem.. 2004;52:3378-3384.
- [Google Scholar]
- Evaluation of Thymus vulgaris and Thymbra spicata essential oils and plant extracts for chemical composition, antioxidant, and antimicrobial properties. Food Sci. Nutr.. 2019;7:1704-1714.
- [Google Scholar]
- Syringic acid triggers reactive oxygen species–mediated cytotoxicity in HepG2 cells. Hum. Exp. Toxicol.. 2019;38:694-702.
- [Google Scholar]
- Effect of gamma irradiation on total phenolic contents and antioxidant activities of Satureja hortensis, Thymus vulgaris, and Thymbra spicata from Turkey. Int. J. Food Prop.. 2011;14:830-839.
- [Google Scholar]
- Characterization of phenolic composition in Lamiaceae spices by LC-ESI-MS/MS. J. Agric. Food Chem.. 2010;58:10576-10581.
- [Google Scholar]
- Rosmarinic acid: a review of its anticancer action. World J. Pharm. Pharm. Sci. 2014;3:57-70.
- [Google Scholar]
- Kirby-Bauer disk diffusion susceptibility test protocol. Am. Soc. Microbiol.. 2009;15:55-63.
- [Google Scholar]
- Plants secondary metabolites: the key drivers of the pharmacological actions of medicinal plants. Herb. Med. 2019;1
- [CrossRef] [Google Scholar]
- LC-MS identification and quantification of phenolic compounds in solid residues from the essential oil industry. Antioxidants. 2021;10(2016):1-19.
- [Google Scholar]
- HPLC-DAD-ESI-MSn identification of phenolic compounds in cultivated strawberries from Macedonia. Maced. J. Chem. Chem. Eng. 2010;29:181-194.
- [Google Scholar]
- Design and synthesis of novel pyrimido [4, 5-b] azepine derivatives as HER2/EGFR dual inhibitors. Bioorg. Med. Chem.. 2013;21:2250-2261.
- [Google Scholar]
- Antisteatotic and antioxidant activities of Thymbra spicata L. extracts in hepatic and endothelial cells as in vitro models of non-alcoholic fatty liver disease. J. Ethnopharmacol.. 2019;239:111919.
- [Google Scholar]
- Antimicrobial and chemopreventive properties of herbs and spices. Curr. Med. Chem.. 2004;11:1451-1460.
- [Google Scholar]
- Antioxidant and antiproliferative activities of twenty-four Vitis vinifera grapes. PLoS One. 2014;9(e105146):1-10.
- [Google Scholar]
- Design and synthesis of new quinoxaline derivatives as potential histone deacetylase inhibitors targeting hepatocellular carcinoma: in silico, in vitro, and SAR studies. Front. Chem.. 2021;9:1-21.
- [Google Scholar]
- Anthocyanins: Antioxidant and/or anti-inflammatory activities. J. Appl. Pharm. Sci. 2011:7-15.
- [Google Scholar]
- Effects of extract and essential oil of Thymbra spicata L. and Satureja bachtiari L. on improving quality and vase life of cut flower Gerbera jamesoni L. Iran. J. Med. Aromat. Plants Res.. 2019;35:617-634.
- [Google Scholar]
- The valuable impacts of halophytic genus suaeda; nutritional, chemical, and biological values. Med. Chem.. 2020;16(8):1044-1057.
- [Google Scholar]
- Essential oil constituents and biological activities of the halophytic plants, suaeda vermiculata forssk and salsola cyclophylla bakera growing in Saudi Arabia. J. Essent. Oil-Bearing Plants. 2019;22
- [CrossRef] [Google Scholar]
- Drying induced impact on composition and oil quality of rosemary herb, Rosmarinus Officinalis Linn. Molecules. 2020;25:2830.
- [Google Scholar]
- Phytochemical profiling, molecular docking, and in vitro anti-hepatocellular carcinoid bioactivity of Suaeda vermiculata extracts. Arab. J. Chem.. 2022;15:103950
- [CrossRef] [Google Scholar]
- Anthocyanins: traditional uses, structural and functional variations, approaches to increase yields and products’ quality, hepatoprotection, liver longevity, and commercial products. Int. J. Mol. Sci.. 2022;23:2149.
- [Google Scholar]
- Mohammed, H., 2009. Natural and synthetic flavonoid derivatives with potential antioxidant and anticancer activities. PhD thesis, Saarland University, Germany.
- Quality from the field: The impact of environmental factors as quality determinants in medicinal plants. South African J. Bot.. 2012;82:11-20.
- [Google Scholar]
- HPLC-DAD and HPLC-ESI-MS/MS methods for metabolite profiling of propolis extracts. J. Pharm. Biomed. Anal.. 2011;55:934-948.
- [Google Scholar]
- Chemical constituents and antibacterial activity of Bromelia laciniosa (Bromeliaceae): Identification and structural characterization. Phytomedicine Plus. 2022;2:100215
- [Google Scholar]
- Antiproliferative activity is predominantly associated with ellagitannins in raspberry extracts. Phytochemistry. 2007;68:218-228.
- [Google Scholar]
- Chemical compositions, total phenolic contents, antimicrobial and antioxidant activities of the extract and essential oil of Thymbra spicata L. growing wild in Turkey. J. Food Meas. Charact.. 2021;15:386-393.
- [Google Scholar]
- Development of high-performance thin layer chromatography method for identification of phenolic compounds and quantification of rosmarinic acid content in some species of the Lamiaceae family. J. Pharm. Bioallied Sci.. 2020;12:139.
- [Google Scholar]
- Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Vitic.. 1965;16:144-158.
- [Google Scholar]
- New colorimetric cytotoxicity assay for anticancer-drug screening. JNCI J. Natl. Cancer Inst.. 1990;82:1107-1112.
- [Google Scholar]
- LC-MS-based metabolite profiling of methanolic extracts from the medicinal and aromatic species Mentha pulegium and Origanum majorana. Phytochem. Anal.. 2015;26:320-330.
- [Google Scholar]
- American herbal pharmacopoeia: botanical pharmacognosy-microscopic characterization of botanical medicines. CRC Press; 2016.
- Anticholinergic effects of Actinidia arguta fruits and their polyphenol content determined by liquid chromatography-photodiode array detector-quadrupole/time of flight-mass spectrometry (LC-MS-PDA-Q/TOF) Food Chem.. 2019;271:216-223.
- [Google Scholar]
- Anthocyanins from pomegranate (Punica granatum L.) and their role in antioxidant capacities in vitro. Chem. Biodivers.. 2021;18:1-21.
- [Google Scholar]
Appendix A
Supplementary material
Supplementary data to this article can be found online at https://doi.org/10.1016/j.arabjc.2023.104775.
Appendix A
Supplementary material
The following are the Supplementary data to this article:Supplementary Data 1
Supplementary Data 1







