Translate this page into:
Polycyclic aromatic hydrocarbons (PAHs) in Egyptian red sea sediments: Seasonal distribution, source Identification, and toxicological risk assessment
⁎Corresponding author at: Department of Chemistry, College of Science, Qassim University, Buraidah 51452, Saudi Arabia. a.younis@qu.edu.sa (Alaa M. Younis)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
The RQPAHs estimated for the samples revealed that the study region was at a high-risk level. The composition profile revealed that sediments were predominantly composed of high molecular weight (4-ring) PAHs. The majority of the study area appeared to have a pyrogenic origin for the PAHs based on the diagnostic ratios. PAHs in sediments in some locations along the study area may possess the capacity to cause cancer, particularly in children.
Abstract
This study investigated the concentrations of 16 polycyclic aromatic hydrocarbons (PAHs) in sediments from the shoreline of the Red Sea. 16 PAH concentrations ranged from 1.43 to 4990.69 µg/g dry weight to 1.64 to 6397.69 µg/g dry weight during spring and autumn, respectively. Most sites with the highest PAH concentrations were (4–6 rings) compounds. The sources of PAHs were analyzed using principal component analysis (PCA) and diagnostic ratios between combinations of PAHs. The results indicated that the pyrogenic source was the primary source of PAHs. The results showed that surface sediment from all sample sites presents low to sporadic toxicity concerns to marine organisms. Total carcinogenicity and mutagenicity of eight priority PAHs ranged from 0.03 to 863.96 and 0.09 to 1197.85 µg/g dry weight, respectively (TEQBaP and MEQBaP). BaP accounted for most of the influence among the eight carcinogenic PAHs and could be employed as a particular PAH toxicity indicator. Using the toxic equivalent quotient (TEQcarc) and incremental lifetime cancer risk (ILCR) to estimate human health risk, it has been determined that sediment contaminated with PAHs in some study area stations may possess the capacity to cause cancer, particularly in children. The ecological risk posed by PAHs was evaluated using a risk quotient (RQ). The RQPAHs estimated for the samples revealed that the study region was at a high-risk level. Anthropogenic hydrocarbon inputs that were more pronounced in locations connected to maritime activity are described in this study. Further monitoring studies encompassing different environmental matrices about PAHs pollution and their toxicological relevance should be conducted.
Keywords
Risk assessment
Polycyclic aromatic hydrocarbons
Cancer risk
Ecological risk
Environmental monitoring
Red Sea coast
Egypt
1 Introduction
One of the environment's most well-known and substantial classes of persistent organic contaminants is polycyclic aromatic hydrocarbons (PAHs). PAHs have recently drawn much attention due to their short-term toxicity, mutagenicity, and carcinogenicity worldwide (Hussain et al., 2015). Due to their widespread occurrence and associated health risk, 16 PAHs were selected by the US Environmental Protection Agency as environmental contaminants. The International Agency for Research on Cancer has classified seven of these chemicals as confident (Group 1), probable (Group 2A), or possibly (Group 2B) carcinogens (Yang et al., 2014a;Yang et al., 2014b). In the ecosystem, PAHs are pervasive, hazardous, and extensively present. Water, suspended materials, and sediments are specifically contaminated by PAHs (Yang et al., 2013;Bai et al., 2014;Keshavarzifard et al., 2014;Younis et al., 2018;El-Naggar et al., 2021). Pollution of aquatic ecosystems by metals and PAHs, particularly in sediments and water, is one of the most pressing contamination issues due to their toxicity, abundance, significant non-degradability, and consequent bioaccumulation (Younis et al., 2018;Younis, 2019;Younis et al., 2019;Soliman et al., 2020;El-Naggar et al., 2021;Hanafy et al., 2021;Younis et al., 2022).
There are two primary categories of sources for PAHs: anthropogenic and natural. Pyrogenic and petrogenic sources combine to form anthropogenic sources of PAHs. Pyrogenic PAHs are often composed of 4–6 ringed high molecular weight PAHs, such as benzo(a)pyrene (BaP), fluoranthene (Flu), benzo(b) fluoranthene (BbF),benzo(k)fluoranthene(BkF), chrysene (Chr), pyrene (Pyr), benzo (a)anthracene (BaA) (Liu et al., 2009b;Riccardi et al., 2013). Pyrogenic is primarily found in incomplete burning of organic substances, including that produced by burning fossil fuels and biomass (Zakaria et al., 2002;Khairy et al., 2009;Liu et al., 2009b). As a result of their higher quantities of non-alkylated PAHs than petrogenic sources, it is anticipated that pyrogenic sources will be more stable at high temperatures and hazardous (Khairy et al., 2009;Saha et al., 2009;Beyer et al., 2010). Naphthalene (Naph), acenaphthylene (Acthy), acenaphthene (Ace), and fluorine (F) are examples of petrogenic PAHs. They are low molecular weight or alkyl-substituted PAHs and have two to three rings. It is suspected that these hydrocarbons resulted from oil spills involving crude and fuel oil, new or old crankcase oil, recurring or unintentional pipeline leaks on land and at sea, and home and industrial wastes (Zakaria et al., 2002;Liu et al., 2009b;Saha et al., 2009;Riccardi et al., 2013).
Like the hydrocarbons of interest, sediments are regarded as superior contaminant sinks. These substances easily bind to particle matter. Hydrophobic contaminants are stored in bottom sediments. As a result, long after pollution input has stopped, sediments can retain and release contaminants that affect biota (da Luz et al., 2010).
Because of PAHs' occasionally pervasive and long-lasting effects on environmental media, they eventually become accumulated and permanent in aquatic system bed sediments. This occurs due to most PAHs' strong sorption to organic materials in sediments and resistance to bacterial breakdown in anoxic environments due to their high hydrophobicity. PAHs are also discharged into the water as an uninterrupted source when favorable environmental conditions, such as dissolved oxygen, organic matter, salinity, and pH (Tepe et al., 2022). PAHs endangers the aquatic marine ecosystem by bioaccumulating in food chains and persisting in their course despite difficulty or resistance (Chen and Chen, 2011). Depending on how close a location is to areas of anthropogenic activity, PAH levels in sediment change (Bihari et al., 2006).
The Red Sea is strategically vital since it separates the Middle East, the Far East, Europe, and Asia from the African continent. Because it serves as a natural boundary between the western and eastern coasts of the Arabian Peninsula and a crucial route for unarmed oil traffic from the Bab El-Mandeb in the south to the Suez Canal in the north, the Red Sea's geopolitical location is significant.
According to Rasul et al. (2015), the Red Sea is an oligotrophic ecosystem with enormous coral reef systems that make up 3.8% of the world's coral reefs. These have been prominent tourist attractions in Egypt's coastal areas for decades. The Red Sea's great biodiversity is partly due to a wide variety of tropical ecosystems, including mangrove forests, islands, shoals, and coastal lagoons. Along the Red Sea coast, industrial and human activities have increased dramatically, necessitating the need for up-to-date information to serve that can help the decision-maker establish effective management plans for the study area, which is of global importance. These activities have frequently encountered difficulties in disposing of the hazardous waste they generate.
Few research has been published on the health effects of concurrent exposure to PAHs in multiple environmental media through major exposure routes (Ma et al., 2021). Research on health risk assessments for simultaneous exposure to PAHs from multiple media in the same environment is lacking. It has yet to be published, making it difficult to conclude the potential dangers of PAHs for organisms. Considering the above, this study seeks to answer related questions about PAH pollution and its potential cancer risk in marine sediments on the Red Sea coast. Therefore, the current study aims to investigate the spatial and temporal distribution of 16 PAHs and their composition in surface sediment from the Red Sea coast. After identifying the significant pollution sources, the ecological risks related to PAHs in surface sediments have also been examined. The health risks of PAHs were identified for the first time in the study area.
2 Materials and methods
2.1 Sample preparation
During spring and autumn, sediments were collected from 14 different positions along the Red Sea coast (Fig. 1). Considering the anticipated contaminated area brought on by anthropogenic activity; the locations were chosen to span around 450 km. The precise location of each site was defined using the global positioning system (GPS). The locations of sampling are depicted in (Fig. 1). A stainless-steel grab was utilized to gather the sediments, which were then scraped off with a stainless-steel spatula, scooped into wide-mouth glass bottles that had already been cleaned, frozen, and brought to the lab, where they were kept at −20 °C until analysis. Following established procedures, the samples were examined for aromatic hydrocarbons (UNEP/FAO/IAEA/IOC, 1991; UNEP/IOC/IAEA, 1992).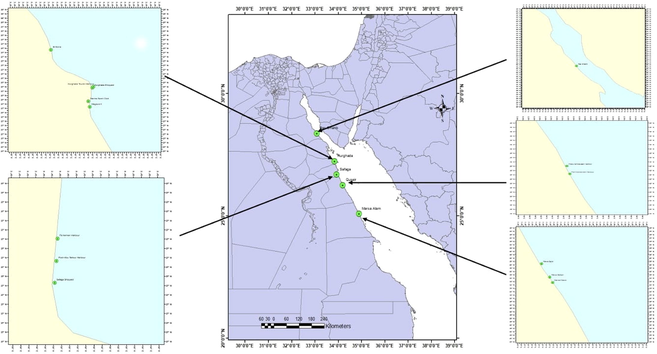
The location of the samplings along the study area.
Several parameters, such as temperature, pH, levels of nutrients, bacterial population size, etc., can influence the efficiency of PAH degradation in marine sediments (Shahriari Moghadam et al., 2014). Therefore, individual samples were taken out of the deep freezer and given roughly 5 h to thaw at room temperature. We used the method of (Gaudette et al., 1974) to determine the total organic carbon (TOC). Each sample was properly blended before 30 g of sediment and 90 g of anhydrous sodium sulfate were added. The sediment sample was then sonicated for roughly 3 min at settings that would produce effective extraction in an ultrasonic bath with 100 ml of 1:1 v/v dichloromethane/acetone. The extract was then filtered and dried using glass wool and anhydrous sodium sulfate. Utilizing a rotary evaporator and nitrogen gas, dilute the extract to 1 ml (UNEP/IOC/IAEA, 1992).
The extract was cleaned up and fractionated through a silica/alumina column. Before being partially deactivated with 5% water, the silica and alumina were activated at 200 °C for four hours to remove moisture. The chromatographic column was created by slurry packing 10 ml of silica, 10 ml of alumina, and 1 g of anhydrous sodium sulfate. To elute the first fraction (aliphatic hydrocarbon), 40 ml of hexane was used, followed by 40 ml of hexane/dichloromethane (9:1) and 20 ml of hexane/dichloromethane (1:1). (Which combined contain PAHs). The resulting fractions were mixed with the copper powder. The elimination of sulfur that hampered GC–MS analysis was assumed to cause the copper's color to change from brassy red to black (Sundberg et al., 2005). Finally, eluted samples were reduced to a volume of 0.2 ml by concentration using an N2 gas stream.
2.2 Instrumentation (GC–MS)
Using a gas chromatography-mass spectrometer (GC–MS), PAHs were measured (QP2010 Ultra, Shimadzu, Japan). The following GC procedures were used: an autosampler, an injection volume of 1 μl, an injector temperature of 280 °C, a pressure of 150 kPa, splitless injection mode with a purge flow of 3 ml/min, helium was used as the carrier gas (99.999%) with a flow rate of 1.0 ml/min and an average velocity of 54.6 cm/sec, and compounds were separated on an RTX-5MS capillary Initial temperature set at 60 °C; 20 °C/min to 160 °C; 3 °C/min to 280 °C, kept for 6 min; 20 °C/min to 300 °C, held for 5 min; were the settings for the oven. The electron impact (EI) ionization, ion source temperature of 230 °C, transfer line temperature of 260 °C, and full scan mode for the masses between 35 and 500 m/z were all used in the mass spectrometer processes. GC–MS software from Shimadzu was used throughout the project to process the data automatically.
2.3 Analytical quality assurance
The samples were stored in clean containers after being covered with aluminum foil. The solvents, reagents, and standards used were high purity (above 98%) and analytical grade. After cleaning the equipment and glassware with water and liquid soap, deionized water, acetone, and finally, oven drying at 150 °C, the glassware was rinsed. There were established blanks, recovery tests, and detection limits. In order to determine the detection limits, a signal-to-noise ratio of 3:1 was used. For recovery experiments, sediment samples from the Red Sea coast were used as blanks. Validation studies were carried out by mixed standards with concentrations ranging from 1 to 8.33 ppb introduced into the samples. Acceptable recovery values for PAHs from sediments (n = 6) were found to vary between 76 and 109.7%. The detection limits for sediments were from 0.003 to 0.009 ng/g for acenaphthene, Fluoranthene, Anthracene, phenanthrene, benzo[a]anthracene, and benzo[k] fluoranthene, and from 0.015 to 0.043 ng/g for benzo[a]pyrene, benzo[b]fluoranthene, pyrene. Analyses that produced findings below these thresholds were labeled “Not Detected.”.
2.4 Statistical analysis
An IBMSPSS 25.0 (SPSS Inc., USA) was used for the data treatment and multivariate statistical analysis. Data normality was assessed using Kolmogorov-Smirnov (K-S) tests. One-way analysis of variance (ANOVA) for variables with normal distribution and the Kruskal-Wallis test for variables without normal distribution were used to examine the statistical significance of the differences. To locate likely sources of the PAHs, PCA was carried out using a varimax rotation. The Bartlett and Kaiser-Meyer-Olkin (KMO) tests were employed to gauge the PCA's validity. The PAH concentration ratios were used to calculate the source diagnostic indices.
3 Results and discussion
3.1 Distribution of PAHs in sediments
In the current study, the total PAH levels in surficial sediments varied from 1.64 to 6397.69 µg/g with a mean concentration of 1192.53 µg/g in the autumn season and from 1.43 to 4990.69 µg/g with a mean concentration of 901.29 µg/g in the spring season (Tables 1 & 2). Station 5 at Hurghada Shipyard, which is affected by dockyards and ship repair activities, has the highest PAH concentration during the autumn and spring seasons. The sediments were found to be contaminated with PAHs throughout the study, with higher concentrations recorded in stations, particularly those that were close to the Marine Sports Club Hurghada (station 4: 2196.43 and 1902.73 µg/g in the autumn and spring, respectively), Pre-Hamraween harbor (station 10: 2684.92 and 1561.12 µg/g in the autumn and spring, respectively), Safaga shipyard (station 7: 1866.40 and 1438 µg/g dw in the autumn and spring, respectively), Hurghada Tourist Harbor (station 6: 872.81 and 512.37 µg/g dw in the autumn and spring respectively), fishermen harbor in Safaga (station 9: 765.33 and 596.94 µg/g dw in the autumn and spring, respectively), Post-Hamraween harbor (station 11: 720.14 and 618.02 µg/g dw in the autumn and spring, respectively) and Marina Marsa in Marsa Alam (station 13: 529.96 and 441.88 µg/g dw in the autumn and spring, respectively). Naph = naphthalene, Acthy = acenaphthylene, Ace = acenaphthene, F = fluorene, Phe = phenanthrene, Ant = anthracene, Flu = fluoranthene, Pyr = pyrene, BaA = benzo[a]anthracene, Chr = chrysene, BbF = benzo[b]fluoranthene, BkF = benzo[k]fluoranthene, BaP = benzo[a]pyrene, DahA = dibenzo[a,h]anthracene, BP= benzo [ghi] perylene, IP = indeno [1,2,3-cd] pyrene, ∑PAHs = total PAHs in sediment.
Station
Naph
Acthy
Ace
F
Phe
Ant
Flu
Pyr
BaA
Chr
BbF
BkF
BaP
BP
DahA
IP
∑PAHs
1
0.82
ND
0.36
2.16
10.86
15.58
18.91
32.62
11.02
149.28
5.37
1.93
4.22
ND
ND
35.10
288.22
2
0.23
ND
0.13
ND
0.17
0.15
0.29
0.96
5.59
7.94
2.51
ND
1.19
ND
ND
4.72
23.88
3
0.68
0.44
0.41
0.55
0.99
1.09
2.66
1.39
2.35
1.86
1.28
0.75
1.06
ND
ND
1.74
17.24
4
5.70
1.17
7.79
34.39
206.13
105.28
454.32
513.83
321.19
102.95
91.07
10.60
76.67
ND
ND
265.34
2196.43
5
17.86
2.17
18.16
38.66
220.87
58.41
487.13
739.92
474.81
989.49
769.67
583.18
142.92
ND
ND
1854.40
6397.67
6
2.36
0.08
0.23
11.38
0.47
13.60
1.30
1.39
2.19
19.31
2.76
710.46
86.50
ND
ND
20.79
872.81
7
2.91
2.01
1.45
31.64
35.38
46.12
116.93
136.99
185.74
216.95
35.77
688.76
165.41
ND
ND
200.35
1866.40
8
2.77
ND
0.00
3.02
0.96
1.27
18.13
2.81
29.71
5.30
7.57
18.69
6.56
ND
ND
3.77
100.56
9
0.79
0.84
3.01
6.89
12.01
5.60
28.07
27.42
22.81
36.05
105.24
225.29
60.03
ND
ND
231.29
765.33
10
0.19
2.07
1.09
2.68
72.35
5.76
333.81
247.37
404.87
518.12
552.08
158.62
268.75
ND
ND
117.16
2684.92
11
0.71
0.00
0.63
0.82
3.04
5.26
151.19
5.85
20.03
15.84
15.82
424.63
6.36
ND
ND
69.95
720.14
12
ND
0.17
0.99
6.89
76.33
2.08
52.82
25.74
21.94
12.89
16.10
2.02
9.97
ND
ND
2.32
230.26
13
2.48
1.14
1.88
11.60
123.81
72.98
59.58
28.97
43.08
19.02
22.81
69.58
68.85
ND
ND
4.16
529.96
14
0.50
ND
ND
ND
0.26
0.23
0.27
ND
ND
ND
0.37
ND
ND
ND
ND
ND
1.64
Min.
ND
ND
ND
ND
0.17
0.15
0.27
ND
ND
ND
0.37
ND
ND
0.00
0.00
ND
1.64
Max.
17.86
2.17
18.16
38.66
220.87
105.28
487.13
739.92
474.81
989.49
769.67
710.46
268.75
0.00
0.00
1854.40
6397.67
Average
2.92
1.01
2.78
12.56
54.55
23.82
123.24
135.79
118.87
161.15
116.32
241.21
69.11
ND
ND
216.24
1192.53
Station
Naph
Acthy
Ace
F
Phe
Ant
Flu
Pyr
BaA
Chr
BbF
BkF
BaP
BP
DahA
IP
∑PAHs
1
0.80
ND
0.33
1.89
11.33
14.88
17.88
33.04
7.95
144.12
5.34
1.42
4.25
ND
ND
34.30
277.53
2
0.24
ND
0.15
ND
0.16
ND
0.22
0.68
4.98
7.36
1.97
ND
0.91
ND
ND
3.68
20.34
3
0.98
0.43
0.64
0.53
0.77
0.84
0.97
1.27
1.15
1.11
0.77
0.48
0.66
ND
ND
0.85
11.46
4
4.59
0.89
6.04
30.92
40.77
30.54
351.86
435.69
264.45
37.31
52.38
321.32
68.76
ND
ND
257.23
1902.73
5
13.99
1.78
17.14
36.47
198.16
55.10
432.20
698.06
367.40
688.33
838.65
8.83
635.69
ND
ND
999.03
4990.84
6
2.07
0.08
0.16
8.91
0.37
7.54
0.71
1.13
1.56
15.58
2.12
292.83
56.55
ND
ND
122.76
512.37
7
2.69
1.84
1.28
28.75
33.55
39.12
75.98
121.02
147.26
181.52
33.47
514.30
113.67
ND
ND
143.54
1438.00
8
2.12
ND
ND
1.86
0.60
0.84
13.72
2.04
24.21
4.21
6.79
16.93
4.51
ND
ND
3.27
81.11
9
0.73
0.76
2.88
6.36
11.52
4.96
17.54
24.66
16.19
31.93
81.21
137.94
39.89
ND
ND
220.38
596.94
10
0.24
1.33
1.05
2.35
61.77
4.74
303.18
176.95
329.78
42.53
359.90
93.00
144.48
ND
ND
39.84
1561.12
11
0.62
ND
0.68
0.80
3.30
4.53
142.21
4.32
11.17
12.28
13.87
366.58
6.61
ND
ND
51.06
618.02
12
ND
0.21
0.88
5.79
37.55
2.07
44.99
20.70
17.17
11.56
13.32
1.43
6.74
ND
ND
1.85
164.27
13
2.23
0.96
1.16
9.48
90.60
64.80
50.21
26.15
37.01
17.27
16.97
56.76
64.15
ND
ND
4.12
441.88
14
0.33
ND
ND
ND
0.26
0.23
0.27
ND
ND
ND
0.34
ND
ND
ND
ND
ND
1.43
Min
ND
ND
ND
ND
0.16
ND
0.22
ND
ND
ND
0.34
ND
ND
0.00
0.00
ND
1.43
Max
13.99
1.84
17.14
36.47
198.16
64.80
432.20
698.06
367.40
688.33
838.65
514.30
635.69
0.00
0.00
999.03
4990.84
Average
2.43
0.92
2.70
11.18
35.05
17.71
103.71
118.90
94.64
91.93
101.94
150.98
88.22
ND
ND
144.76
901.29
Water emission sources and the physicochemical characteristics of sediments may be the main variables affecting the variations in PAH levels. In both seasons, stations 4, 5, 6, 7, 9, 10, 11, and 13 were adjacent to dockyards and areas with a lot of shipping activity. These sites were affected by the discharge of PAH from raw sewage, wastewater from industry, household activities, and other types of urban effluents.
The present investigation also revealed that autumn had higher overall PAH concentrations than spring. Analyzing the spatial and temporal variability of PAH levels in sediments necessitates taking into account two types of parameters: (1) Intrinsic parameters, which are mainly connected to the contaminant's physicochemical properties, and (2) different abiotic and biotic variables are both important (Gdara et al., 2017).
The distribution and separation of average individual PAHs (Tables 1 & 2) were noted in the examined area. DahA and BP levels were not found at autumn and spring sampling stations. Following in order of decline in autumn were the remaining PAHs: BkF > IP > Chr > Pyr > Flu > BaA > BbF > BaP > Phe > Ant > F > Naph > Ace > Acthy. Whereas the following is the order for spring: BkF > IP > Pyr >Flu > BbF > BaA > Chr > BaP > Phe > Ant > F > Ace > Naph > Acthy.
This study used regression analysis to examine the connection between PAH concentration and organic carbon content (TOC%). The linear regressions in the autumn and spring seasons demonstrate a strong association between PAHs and %TOC (P < 0.01) (Fig. 2). It is widely known how important sedimentary organic matter is to partition PAHs in sediments. (Chiou et al., 1998) discovered that the numerous aromatic portions of the organic matter were primarily responsible for the high partitioning of PAHs to sedimentary organic matter. For them, the organic soil was like a “heterogeneous polymer” in nature, where PAHs interacted most strongly with aromatic regions.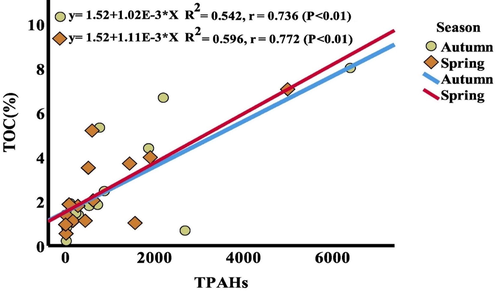
Regressions of total PAHs with %TOC at the location of the study area.
The varied geological features of the sites, the volume of samples analyzed, and the set of PAH compounds evaluated make it challenging to compare the quantities of PAHs in sediments along the study area with those found in other coastal regions. A broad understanding of pollution levels seen elsewhere is frequently beneficial to determine the pollution level in our study area. Baumard et al., (1998) categorized sediment pollution into four categories: low, which is equal to 0.0–0.1 µg/g of dry weight; moderate; 0.1–1.0 µg/g; high, 1.0–5.0 µg/g; and very high, which is above 5.0 µg/g. Therefore, the PAH pollution levels in the current investigation were high to very high (1.64–6397.67 µg/g in autumn and 1.43–4990.84 µg/g in spring). The sediments taken from all stations in both seasons, except station 14, can be classified as severely polluted based on the PAH levels found in the examined areas Table 3. N* Number of PAH compounds.
Location
Min–Max (µg/g)
Pollution level
N*
References
present study
1.64– 6397.67 in autumn
high to very high
16
─
1.43– 4990.84 in spring
high to very high
─
Baltic Sea
0.80–1.90
Moderate to high
15
(Witt, 1995)
Baltic Sea
0.00396–30.10
low to very high
24
(Baumard et al., 1999)
Gulf of Naples, Southern Italy
0.092–12.561
low to very high
15
(Romano et al., 2004)
The Sea of Marmara (Turkey)
0.120–11.40
Moderate to very high
14
(Tolun et al., 2006)
The Sea of Marmara (Turkey)
0.05–13.482
low to very high
14
(Ünlü and Alpar, 2006)
Mediterranean coast (Egypt)
0.088–6.338
low to very high
16
(El Nemr et al., 2007)
Mediterranean coast (Egypt)
0.21–1.02
Moderate to high
16
(El Nemr et al., 2013)
Mediterranean coast (Tunisia)
0.0256– 0.5768 in winter
0.0449– 0.3958 in summerlow to moderate
low to moderate17
(Nouira et al., 2013)
Red Sea coast (Egypt)
0.0074–0.457
low to moderate
16
(Salem et al., 2014)
Red Sea coast (Egypt)
0.132–5.182
Moderate to very high
16
(El Nemr et al., 2014)
Red Sea coast (Egypt)
1.01–1513.27
high to very high
15
(Abdallah et al., 2016)
Suez Gulf, Red sea (Egypt)
0.0066–1.77
Low to High
16
(Ahmed et al., 2017)
Red Sea coast (Saudi Arabia)
1.17–3.0
high
16
(Al-Mur, 2019)
North Seas
0.00046–0.227
Low to moderate
18
(Wang et al., 2020)
Kerala, India
0.001–55.83
Low to very high
16
(Yahiya and Miranda, 2021)
Tourist Beaches of Bushehr, Persian Gulf
0.193–0.726
Moderate
16
(Ghasemi and Keshavarzifard, 2022)
Giresun coastline of southeastern Black Sea (Turkey)
0.04914–0.44426
Low to moderate
16
(Tepe et al., 2022)
Egyptian Mediterranean coast
13.156–34.852
very high
16
(El-Maeadny et al., 2023)
The TOC, 16PAHs, and PAH contents did not follow a normal distribution ((P < 0.05), as determined by the K-S test for a single sample. There were no statistically significant differences (P > 0.05) across the stations or seasons when employing the non-parametric Kruskal-Wallis test (K-W).
3.2 Percent composition of PAHs
Although Dibenzo[ah]anthracene and Benzo(ghi)perylene in both seasons were under the detection threshold, sixteen different PAHs were found in the sediments. These 16 PAHs were divided into five classes based on the number of aromatic rings they contained (2–6). (Fig. 3) shows the PAHs' % content based on ring groups. In both seasons, typically, the 4-ring group was the predominant, followed by the 5-ring group. However, significant variances were seen depending on the seasons and locations. During the autumn and spring seasons at station 14, the 2- and 3-ring congeners known as low molecular weight (LMW) PAHs predominated, making up 61.23% and 57.10% of the total PAHs, respectively.
Composition of PAH of the ring according to the percentage of aromatic rings in the study area's sediments during the autumn and spring.
For example, the existence of fused aromatic rings is correlated with higher carcinogenicity of PAHs. chrysene, benzo(a)anthracene, benzo(a)pyrene, benzo(k)fluoranthene, benzo(b)fluoranthene, benzo(g,h,i)perylene, dibenzo(a,h)anthracene, and indeno(1,2,3-cd)pyrene are eight polycyclic aromatic hydrocarbons (PAHs) (Car-PAHs) commonly (IARC, 1983; Menzie et al., 1992). Total quantities of these compounds in sediments from the study region ranged from 0.37 to 4814.48 µg/g during the autumntime, accounting for 22.40–96.47% of all PAHs. The overall amount of (Car-PAHs) varied from 0.34 to 3537.93 µg/g during the springtime, making up between 23.39 and 95.93% of the total PAH concentrations.
Naphthalene, Anthracene, and phenanthrene are low-molecular-weight PAHs that are more easily broken down and are of particular concern (Fernandes et al., 1997;Macıas-Zamora et al., 2002). As a result, sediments from the river and marine habitats frequently show that HMW PAHs are present at higher concentrations than LMW PAHs (Mandalakis et al., 2014;Liu et al., 2015). The type of emission was the primary variable affecting the seasonal composition of PAHs. According to numerous studies, the prevalence of alkylated PAHs, besides 2- and 3-ring PAHs, petrogenic sources of contamination, including crude oil and unburned petroleum and its byproducts, are indicated. However, 4- to 6-ring PAHs are associated with pyrogenic sources, including the incomplete combustion of organic molecules (Cerniglia, 1993;Xu et al., 2007). Additionally, seasonal variations in temperature, sunlight intensity, and the efficiency of photochemical breakdown (Fu et al., 2009) altered the buildup of PAHs in the atmosphere, impacting the sediment loads. Additionally, in fluids and sediments, 2-, 3-, and 4-ring PAHs were easier to biodegrade and volatilize than 5- and 6-ring PAHs (Wang et al., 2010).
3.3 Identifying the source of PAHs in sediment
Most polycyclic aromatic chemicals in the environment are either petrogenic or pyrogenic, both of which are mostly attributed to human activity. When fossil fuels are burned, pyrogenic chemicals are released into the environment. In comparison, pyrogenic compounds are specific types of substances that can be extracted from petroleum. Compounds having two or three benzene rings and a low molecular weight (LPAH) are common in petrogenic sources. In contrast, pyrogenic sources typically produce higher molecular weight (HPAH) compounds with four to six benzene rings (Neff, 1979;Zakaria et al., 2002). Its diagnostic ratio is the most effective and frequent method for detecting the PAH compound's origin (Douben, 2003). By contrasting the estimated using diagnostic ratios, the provenance of PAH molecules derived from various sources can be ascertained. LPAHs/HPAHs, Phe/Ant, Flu/Pyr, Ant/(Ant + Phe), Flu/(Flu + Pyr), BaA/(BaA + Chr), and COMB/PAHs are the ratios that are most frequently employed.
The current investigation uses a ratio of roughly two and three rings to four-six rings to distinguish between the PAH source (Soclo et al., 2000;De Luca et al., 2005;Wang et al., 2006). Naph, Acthy, Ace, F, Phe, and Ant are among the LPAHs; Flu, Pyr, BaA, Chr, BbF, BkF, BaP, IP, DahA, and BP are among the HPAHs. Except for stations (13 and 14) in the autumn and spring seasons, respectively, which were produced from petrogenic sources since LPAHs/HPAHs ratios were more than 1, all stations under study had LPAHs/HPAHs ratios below 1 signify pyrogenic sources for these chemicals (Table 4).
Diagnostic ratio
Value
Source
Reference
∑LMW/∑HMW
<1
Pyrogenic
(Soclo et al., 2000;De Luca et al., 2005;Wang et al., 2006)
>1
Petrogenic
∑COMB/∑PAHs
<0.3
Petrogenic
(Ravindra et al., 2008;Liu et al., 2017a)
0.3 – 0.7
mixed sources
>0.7
high-temperature combustion
Anth/(Anth + Phe)
<0.1
Petrogenic
(Yunker et al., 2002)
>0.1
Pyrogenic
Phe/Anth
>10
Petrogenic
(Sicre et al., 1987;Budzinski et al., 1997)
<10
Pyrogenic
Flu/Pyr
<1.0
Petrogenic
(Sicre et al., 1987;Budzinski et al., 1997)
>1.0
Pyrogenic
Flu/(Flu + Pyr)
<0.4
Petrogenic
(Yunker et al., 2002)
0.4 – 0.5
Fossil fuel combustion
>0.5
Grass, wood, coal combustion
BaA/(BaA + Chr)
<0.2
0.2 – 0.35Petrogenic
mixed petrogenic and pyrogenic(Yunker et al., 2002)
>0.35
Combustion
TIPAH
< 4
Petrogenic
(Orecchio, 2010)
> 4
Pyrogenic
Both the Phe/Ant and Flu/Pyr systems were utilized in the current study to look into potential PAH sources along the Red Sea shoreline. Flu/Pyr > 1.0 was thought to imply pyrolytic origin (combustion sources), while Flu/Pyr < 1.0 was typically associated with a petrogenic source (petroleum). Phe/Ant ˃10 was primarily from a petrogenic source, although Phe/Ant < 10 was typically viewed as a sign that the source was pyrolytic (Sicre et al., 1987;Budzinski et al., 1997). This study's Phe/Ant ratios, greater than ten at stations (10 and 12) during both seasons, point to petrogenic origins. Additionally, the Phe/Ant ratio of the remaining sites in both seasons is under 10, indicating that the sources of the PAHs are combustion-related. One possible cause in the area under investigation is the incomplete combustion of fuel used by the boats' and vehicles' engines.
Seasons of autumn and spring, respectively, the ratio of Flu/Pyr < 1.0 at locations (1, 2, 4, 5, 6, and 7) and stations (1, 2, 3, 4, 5, 6, 7, and 9) indicated that they were from petroleum sources. The other locations in both seasons had Flu/ Pyr values greater than 1, showing that combustion was the main source of PAHs (Table 5). The genesis of PAHs is petrogenic when the ratio of Ant/(Ant + Phe) is below or equal to 0.1 and pyrolytic when it is>0.1 (Yunker et al., 2002).
Season
1
2
3
4
5
6
7
8
9
10
11
12
13
14
Phe/Ant
Autumn
0.70
1.14
0.91
1.96
3.78
0.03
0.77
0.76
2.14
12.56
0.58
36.76
1.70
1.14
Spring
0.76
ND
0.92
1.34
3.60
0.05
0.86
0.71
2.32
13.03
0.73
18.15
1.40
1.11
Flu/Pyr
Autumn
0.58
0.31
1.92
0.88
0.66
0.94
0.85
6.45
1.02
1.35
25.84
2.05
2.06
ND
Spring
0.54
0.33
0.76
0.81
0.62
0.63
0.63
6.74
0.71
1.71
32.94
2.17
1.92
ND
Flu/(Flu + Pyr)
Autumn
0.37
0.23
0.66
0.47
0.40
0.48
0.46
0.87
0.51
0.57
0.96
0.67
0.67
ND
Spring
0.35
0.25
0.43
0.45
0.38
0.39
0.39
0.87
0.42
0.63
0.97
0.68
0.66
ND
Ant/(Ant + Phe)
Autumn
0.59
0.47
0.52
0.34
0.21
0.97
0.57
0.57
0.32
0.07
0.63
0.03
0.37
0.47
Spring
0.57
ND
0.52
0.43
0.22
0.95
0.54
0.58
0.30
0.07
0.58
0.05
0.42
0.47
BaA/(BaA + Chr)
Autumn
0.07
0.41
0.56
0.76
0.32
0.10
0.46
0.85
0.39
0.44
0.56
0.63
0.69
ND
Spring
0.05
0.40
0.51
0.88
0.35
0.09
0.45
0.85
0.34
0.89
0.48
0.60
0.68
ND
COMP/∑PAHs
Autumn
0.90
0.97
0.76
0.84
0.94
0.97
0.94
0.92
0.96
0.97
0.99
0.62
0.60
0.39
Spring
0.89
0.97
0.63
0.94
0.94
0.96
0.93
0.93
0.95
0.95
0.98
0.72
0.62
0.43
LMW/HMW
Autumn
0.12
0.03
0.32
0.20
0.06
0.03
0.07
0.09
0.04
0.03
0.01
0.60
0.68
1.58
Spring
0.12
0.03
0.58
0.06
0.07
0.04
0.08
0.07
0.05
0.05
0.02
0.39
0.62
1.33
TI
Autumn
7.15
7.33
9.68
8.34
4.71
11.38
9.12
12.09
6.38
4.37
11.53
5.10
8.86
4.68
Spring
6.82
2.02
8.85
9.78
4.87
10.95
8.59
12.29
5.73
6.72
10.60
5.22
9.22
4.74
These combined ratios were used in the current study to accurately estimate the origin of PAH compounds. The PAHs at station 5 during the autumn and stations 5 and 9 during the spring were of mixed origin, according to the ratio of BaA/(BaA + Chr) (Yunker et al., 2002). Stations 1 and 6′s BaA/(BaA + Chr) ratios of 0.2 over the course of both seasons suggest that the PAH compounds were sourced from petroleum. BaA/(Ba + Chr) ratios>0.35 were found in the remaining sites, proving that pyrolysis was the predominant PAH source. Based on the ratio of Ant to (Ant + Phe), PAH levels at stations (10 & 12) in the autumn and spring originated from petroleum, whereas in contrast, the remaining sites in both seasons originated from a combustion source. During the autumn and spring, the ratio of Flu/ (Flu + Pyr) at locations (4, 5, 6, and 7) and stations (3, 4, and 9) was between 0.4 and 0.5. It showed that the PAHs in these stations originated from sources of petroleum burning. Nonetheless, during the autumn, the Flu/ (Flu + Pyr) ratio for locations (1 and 2) and locations (1, 2, 5, 6, and 7) during the fall and spring seasons were<0.4. Consequently, it can be assumed that PAH chemicals at these sites originated from contamination by petroleum sources. Other stations observed Flu/ (Flu + Pyr) > 0.5, indicating that fossil fuel burning constituted the main source of PAHs (Yunker et al., 2002).
COMB concentrations ranged from 0.63 to 6041.53 µg/g in the autumn and from 0.62 to 4668.20 µg/g in the spring. The highest COMB/PAHs ratio (0.99 and 0.98) was observed at station 11 during both seasons, indicating that the PAHs at this location was primarily derived from combustion. In addition, the high COMB/PAHs ratio values suggested that widespread combustion activities influenced the PAHs in sediment along the investigation area (Ravindra et al., 2008; Liu et al., 2017a).
The overall TIPAH index was also computed using the following formula: (Orecchio, 2010).
In general, LPAH/HPAH, and TIPAH values indicated that pyrogenic sources are prevalent; hence, it is reasonable to assume that the primary source of the PAHs in this study was pyro genesis.
3.4 Assessment of sediment quality and toxic potential
3.4.1 PAHs Evaluation using biological threshold
Sediment quality guidelines (SQGs) are valuable techniques for assessing PAH contamination in marine sediments. The ecological effects range low (ERL), and biological effects vary median (ERM) are common ecological risk assessment criteria (Long et al., 1995). The probable effects level (PEL) and the threshold effects level (TEL) are additional (Macdonald et al., 1996). This study used two sets of SQGs to assess the ecological toxicity of specific PAHs to aquatic species, including ERL/ERM and TEL/PEL values. These two sets of SQGs facilitated the construction of massive toxicity databases for marine creatures and their application to evaluating major chemistry/toxicity connections for individual PAHs (McCready et al., 2006;Liu et al., 2009a). The sediment is categorized as having the lowest potential for adverse effects on organisms that live in the sediment. As a result, as being the least contaminated if the concentration of PAHs in the sediment is lower than ERL/TEL. The ≥ ERL/TEL concentration ranges and < ERM/PEL reflected transitional zones from predominately non-toxic to predominately toxic environments. The sensitivity of the toxicity tests and several geological conditions may significantly impact actual toxicity in these concentration ranges. PEL and ERM values represent the chemical concentrations above which there is a relatively high risk of harm to sediment-dwelling species.
Table 6 presents the ERL/ERM and TEL/PEL values for individual PAHs. Within the three PAH concentration ranges a value of (<ERL/TEL) indicates that the pollutants cause ecological problems infrequently. However, a value of (≥ERL/TEL and < ERM/PEL) suggests that the contaminants produce ecological risks occasionally. A value of (≥ERM / PEL) shows that pollutants frequently pose ecological concerns (Long and MacDonald, 1998;Liu et al., 2009a). With only a few exceptions, as shown in (Table 6) for all sampling sites, the majority of individual PAH concentrations reached < ERLs. These exceptions include Acenaphthene and Pyrene in station 5 during the autumn and spring seasons, Fluorene in stations 4, 5, and 7 during the autumn and spring seasons, Anthracene in station 4 during the autumn season, Benzo[a]anthracene in station 4, 5, and 10 during the autumn and spring seasons, Chrysene in station 5 and 10 during the autumn; station 5 during the spring season, Benzo[b]Fluoranthene in station 5 and 10 during the autumn and spring season, Benzo[k] Fluoranthene in stations(4 and 5) in spring and autumn seasons, and stations 6, 7 and 11 in both seasons and Benzo[a]Pyrene in station 5 during the autumn season. As a result, site sediments may be linked to decreased toxicity. Comparable results were obtained when PAH levels in sediments were examined in light of TEL and PEL SQG values (Table 6). The only TELs that were much lower than their own were those of Naph, Acthy, and DahA. Most PAH concentrations, including Ace, F, Phe, Ant, Flu, Pyr, BaA, Chr, and BaP, exceeded the corresponding TELs but not the PELs, indicating that the pollutants can occasionally be hazardous to the environment. ND = Not detected, St. = station.
PAHs
Samples concentrations (µg/g X10 -3)
TEL
PEL
US NOAA
Sample locations > ERL ─ < ERM
Sample locations > TEL ─ < PEL
Autumn
Spring
ERL
ERM
Autumn
Spring
Autumn
Spring
Naph
ND − 17.72
ND − 13.99
34.6
391
160
2100
─
─
─
─
Acthy
ND − 2.17
ND − 1.84
5.87
128
44
640
─
─
─
─
Ace
ND − 18.16
ND − 17.14
6.71
88.9
16
500
St. 5
St. 5
St. 4, 5
St. 5
F
ND − 38.55
ND − 35.47
21.2
144
19
540
St. 4, 5, 7
St. 4, 5, 7
St. 4, 5, 7
St. 4,5,7
Phe
ND − 220.87
ND − 198.16
86.7
544
240
1500
─
─
St. 4, 5, 13
St. 5, 13
Ant
0.15–105.28
ND − 64.80
46.9
245
85.3
1100
St. 4
─
St. 4, 5, 13
St. 5, 13
Flu
0.27–487.13
0.22–432.20
113
1494
600
5100
─
─
St. 4,5,7,10,11
St. 4,5,10,11
Pyr
ND − 739.92
ND − 698.06
153
1398
665
2600
St. 5
St. 5
St. 4,5,10
St. 4,5,10
BaA
ND − 474.81
ND − 367.40
74.8
693
261
1600
St. 4, 5, 10
St. 4,5, 10
St. 4,5,7,10
St. 4,5,7,10
Chr
ND − 989.49
ND − 688.33
108
846
384
2800
St. 5, 10
St. 5
St. 1,5,7,10
St. 1,5,7
BbF
0.37–769.69
0.34–838.65
─
─
320
1800
St. 5,10
St. 5, 10
─
─
BkF
ND − 710.46
ND − 514.30
─
─
280
1620
St. 5,6,7,11
St. 4, 6,7,11
─
─
BaP
ND − 268.75
ND − 635.69
88.8
763
430
1600
─
St. 5
St. 5,7,10
St. 5,7,10
DahA
ND
ND
6.22
135
63.4
260
─
─
─
─
BP
ND
ND
─
─
430
1600
─
─
─
─
IP
ND − 1854.40
ND − 999.03
─
─
─
─
─
─
─
─
3.4.2 Mutagenic and carcinogenic risks of PAHs in surface sediment
The TEQBaP and MEQBaP were computed using the following equations (Safe, 1998;Jung et al., 2010;Benson et al., 2017):
Where Ci is the concentration of a single PAH, TEFi is the toxic equivalence factor, and MEFi is the mutagenic equivalence factor, respectively (Nisbet and LaGoy, 1992;Durant et al., 1996;Durant et al., 1999).
While the total MEQ ranged from 0.09 to 1030.11 µg/g and ranged from 0.09 to 1197.85 µg/g in the autumn and spring seasons, respectively, the total TEQ values for red sea coast samples varied from 0.04 to 521.02 µg/g and ranged from 0.03 to 863.96 µg/g. During the autumn and spring seasons, station 5 (Hurghada shipyard) recorded the highest levels for both BaP TEQ and BaP MEQ, indicating a highly contaminated local source that may depend on dockyards and ship maintenance Table 7.
1
2
3
4
5
6
7
8
9
10
11
12
13
14
Total TEQ
Autumn
11.06
2.55
1.69
146.52
521.02
160.31
278.64
12.59
118.85
397.20
59.56
14.34
83.00
0.04
Spring
10.59
2.04
1.00
158.67
863.96
98.63
199.34
9.67
85.78
227.15
51.00
10.23
75.81
0.03
Total MEQ
Autumn
20.10
3.87
2.22
210.95
1030.11
172.29
331.14
14.20
185.30
502.55
80.62
16.96
87.35
0.09
Spring
19.47
3.07
1.29
219.25
1197.85
127.74
238.27
11.14
145.55
284.79
67.36
12.41
79.25
0.09
The average contribution of the following eight cancer-causing PAHs to the total TEQcarc of surface sediments: In the autumn season, the average TEQcarc contributions from eight carcinogenic PAHs were ranked as follows: BaP (51.84%) > BkF(17.17%) > BbF (13.58%) > IP (12.46%) > BaA (11.02%) > Chr(1.90%). In the spring season, the mean contribution of these eight PAHs was as follows: BkF (16.71%), BbF (13.40%), IP (11.50%), BaA (10.55%), and Chr (1.77%) are all higher than BaP (54.02%). Thus, most of the toxicological risks that the examined area experienced appeared to be caused by BaP and BkF (Fig. 4). Similar outcomes for the contribution of BaP to overall TEQ were discovered elsewhere. Both (Savinov et al., 2003) and (Gdara et al., 2017) showed BaP contribution to be between 49 and 76 percent, demonstrating the value of employing BaP as a marker for carcinogenic PAHs that are impacting sediments.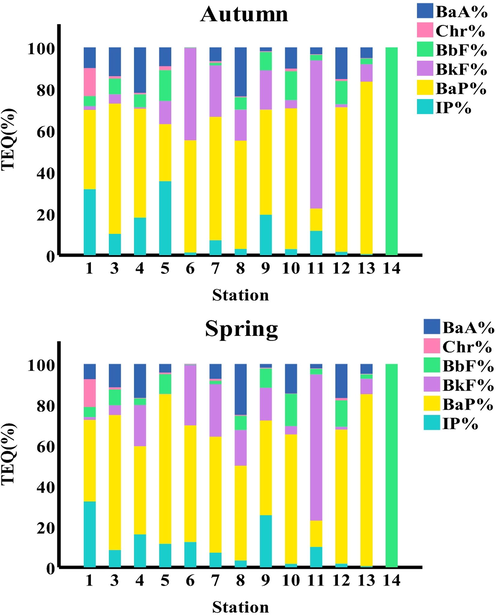
BaA, Chr, BbF, BkF, BaP, and IP contributions to the overall TEQ values along the study area.
3.4.3 Ecological risk assessment
Based on risk quotients, the ecological danger of PAHs in water and sediments was determined during this experiment (RQ). The quality values of negligible concentrations (NCs) and maximum permissible concentrations (MPCs) of PAHs in the medium were reported by (Kalf et al., 1997). NCs are the environmental concentrations below which it is anticipated that there will be little risk of adverse consequences. MPCs are the environmental concentrations over which the danger of unfavorable effects is considered unbearable for ecosystems (Crommentuijn et al., 2000). Thus, the following formulas were used to derive RQ, RQ(NCs), and RQ(MPCs):
Where CPAHs represented the concentration of a single PAH in the medium, CQV represented the associated quality values of a specific PAH in the medium, and RQ risk quotient values. Only ten distinct PAHs, nevertheless, could be detected using this technique. The ecological danger of the remaining six individual PAHs could not be assessed using this method (Acthy, Ace, Flu, Pyr, BbF, DaA). To deal with this issue, a novel approach proposed by (Cao et al., 2010) was used in this study, and RQ-PAHs, RQ-PAHs (NCs), and RQ-PAHs (MPCs) were exposed to the equations below:
RQ (NCs) < 1.0 demonstrated that PAHs' potential limited contribution to ecological risk, whereas RQ (MPCs) > 1.0 would indicate that the pollution of the individual PAHs posed a serious concern, according to other studies (Sun et al., 2009;Cao et al., 2010). If RQ (NCs) > 1.0 and RQ (MPCs) < 1.0, it is implied that PAHs pose a moderate ecological concern and that the necessary remedial or control measures must be implemented.
All of the RQ (NCs) for different PAHs in sediment were greater than one during the autumn and spring seasons, as shown in (Table 8), indicating that the sediment of the research area posed a significant level of ecological risk. In general, RQ (PAHs(NCs)) and RQ (PAHs(MPCs)) in sediments at all sites in the autumn and spring were high ecological risk of PAHs (RQ (PAHs(NCs)) > 800, RQ (PAHs(MPCs)) > 1) except station 14, which was regarded a moderate risk.
Station
RQ(NCs) of 2 rings%
RQ(NCs) of 3 rings%
RQ(NCs) of 4 rings%
RQ(NCs) of 5 rings%
RQ(NCs) of 6 rings%
Autumn
Spring
Autumn
Spring
Autumn
Spring
Autumn
Spring
Autumn
Spring
1
1.12
1.12
32.79
32.24
61.67
62.18
3.29
3.33
1.13
1.14
2
4.41
5.68
7.32
5.15
66.03
67.67
20.07
19.42
2.17
2.08
3
9.40
15.28
44.26
47.72
37.47
31.07
8.30
5.62
0.57
0.31
4
0.55
0.59
22.28
11.74
72.68
81.37
3.88
5.51
0.61
0.79
5
1.06
0.89
11.71
11.67
64.43
63.02
20.19
22.90
2.61
1.51
6
2.86
4.35
36.02
41.14
3.40
4.56
57.12
43.82
0.60
6.13
7
0.70
0.76
25.11
26.15
57.98
58.22
15.06
13.90
1.14
0.97
8
9.77
9.66
18.55
15.13
55.95
57.28
15.42
17.58
0.32
0.35
9
0.62
0.70
17.38
19.69
33.30
34.79
44.44
39.82
4.27
4.99
10
0.03
0.04
4.48
5.25
63.19
65.88
31.93
28.65
0.37
0.18
11
1.09
1.15
13.29
14.65
35.19
31.81
47.89
50.16
2.54
2.24
12
0.00
0.00
40.31
34.69
51.14
55.85
8.48
9.38
0.07
0.07
13
1.19
1.25
65.07
64.27
25.81
26.96
7.88
7.46
0.05
0.06
14
50.14
40.47
34.25
41.45
1.44
1.79
14.17
16.29
0.00
0.00
Individual PAH contributions to RQ (NCs) and their contributions to RQ (MPCs) were extremely similar. It was discovered that the ecological load in sediment varied seasonally and was mostly influenced by the 4-ring PAH RQ (NCs) ratio.
While 5-ring PAHs made up the majority of the ecological risk load in the sediment at the stations (6, 9, and 11) in both seasons, 3-ring PAH contributions to RQ (NCs) in sediment at the stations (3 and 13) were>4-ring in the autumn and spring seasons. The main ecological risk load in the sediment at station 14 in the autumn and spring was the RQ (NCs) of 2 + 3-ring PAHs.
3.4.4 PAHs' health risks
The hazardous level of all PAHs, equal to the toxic concentration of BaP, determines the carcinogenic risk value. This study evaluated the potential health concerns posed by PAHs in sediments through ingestion and skin contact. The following formulae are suggested for these two pathways to calculate exposure (USEPA, 2004).
Where, Cancer Risk ingest is cancer risk via ingestion from sediment; Csed is the concentration equivalent to B[a]P toxicity in sediment; IngR is the ingestion rate: 100 mg day−1 for adults and 200 mg day−1 for children; EF is the exposure frequency: 350 days year−1; ED is the exposure duration: 30 years for adults and 6 years for children; BW is the body weight: 70 kg for adults and 16 kg for children; AT is the average day: 10,950 days for adults and 2190 days for children; CF is the unit conversion factor: 10−6 kg mg−1; SFO is the oral slope factor: 7.3 mg (kg day)−1; Cancer Risk dermal is the cancer risk via dermal contact of sediment; SA is the exposed skin surface area: 5700 cm2; AF is the adherence factor from the sediment to skin: 0.07 mg cm−2 for adults and 0.2 mg cm−2 for children; ABS is the dermal absorption from the sediment: 0.13.
Where cancer-causing sediment may be ingested; The ingestion rate (IngR) is 100 mg for adults and 200 mg for children; the exposure frequency (EF) is 350 days per year; the exposure duration (ED) is 30 years for adults and six years for children; and the concentration (Csed) of B[a]P in sediment is equal to its toxicity. Body weight abbreviated as “BW,” is 70 kg for adults and 16 kg for children daily, or 10,950 days and 2190 days, respectively.
Cancer Risk Dermal is the risk of developing cancer from dermal contact with contaminated sediment. The surface area (SA) of visible skin is 5700 cm2. The AF, or adherence factor, is 0.07 mg cm−2 for adults and 0.2 mg cm−2 for children when the sediment adheres to the skin. The dermal absorption of the sediment (ABS) is 0.13. Since there are many degrees of cancer risk, the characteristics of each can be stated qualitatively as follows: Minimal risk exists when the predicted value is ≦10−6, a small risk exists between 10−6 < to < 10−4, a moderate risk exists between 10−4≦ to < 10−3, a high risk exists between 10−3≦ to < 10−1, and a very high risk exists when the value is 10−1≦ (Man et al., 2013).
In this study, the Cancer Risk ingest for adults in autumn and spring varied from 4.00 × 10-7 to 5.21 × 10-3 and from 3.00 × 10-7 to 8.64 × 10-3, respectively. In contrast, the ranges for children in autumn and spring varied from 3.50 × 10-6 to 4.56 × 10-2 and from 2.63 × 10-6 to 7.56 × 10-2, representing a range from very low to high risk for adults and from low to high risk for children (Table 9). Dermal cancer incidence rates in adults in autumn and spring were 2.07 × 10-7 to 2.70 × 10-3 and 1.56 × 10-7 to 4.48 × 10-3, respectively, and it ranged from 2.59 × 10-6 to 3.38 × 10-2 and 1.95 × 10-6 to 5.60 × 10-2 for children, indicating adults were at very low to high risk, while kids were also at low to high risk. According to the findings of this study, practically all samples that were polluted with PAHs had CRs that surpassed, in comparison, the USEPA-recommended level (10−6), indicating possible health hazards. Sediments in the study area pose a greater threat to children's health than adults, according to the overall CRs value. According to the analysis results, station 5 recorded the highest CRsing and CRsdermal values in the spring and autumn. Consequently, PAH-contaminated sediments in a few study sites could have a severe cancer-causing impact, especially on young people. Compared to adults, children may absorb more harmful substances through dermal and gastrointestinal contact due to their shorter stature and a higher surface area-to-volume ratio (Verbrugge, 2008;Ghasemi and Keshavarzifard, 2022) This is because children tend to engage in more hand-to-mouth activities, weigh less, and consume more per unit of body mass than adults.
1
2
3
4
5
6
7
8
9
10
11
12
13
14
CRSing for Adult
Autumn
1.11E-04
2.55E-05
1.69E-05
1.47E-03
5.21E-03
1.60E-03
2.79E-03
1.26E-04
1.19E-03
3.97E-03
5.96E-04
1.43E-04
8.30E-04
4E-07
Spring
1.06E-04
2.04E-05
1.00E-05
1.59E-03
8.64E-03
9.86E-04
1.99E-03
9.67E-05
8.58E-04
2.27E-03
5.10E-04
1.02E-04
7.58E-04
3E-07
Risk level for Adult
Autumn
Moderate
Low
Low
High
High
High
High
Moderate
High
High
Moderate
Moderate
Moderate
Very Low
Spring
Moderate
Low
Low
High
High
Moderate
High
Low
Moderate
High
Moderate
Moderate
Moderate
Very Low
CRSing for Chlidern
Autumn
9.68E-04
2.23E-04
1.48E-04
1.28E-02
4.56E-02
1.40E-02
2.44E-02
1.10E-03
1.04E-02
3.48E-02
5.21E-03
1.26E-03
7.26E-03
3.5E-06
Spring
9.27E-04
1.79E-04
8.75E-05
1.39E-02
7.56E-02
8.63E-03
1.74E-02
8.46E-04
7.51E-03
1.99E-02
4.46E-03
8.95E-04
6.63E-03
2.63E-06
Risk level for Childern
Autumn
Moderate
Moderate
Moderate
High
High
High
High
High
High
High
High
High
High
Low
Spring
Moderate
Moderate
low
High
High
High
High
Moderate
High
High
High
Moderate
High
Low
CRSdermal for Adult
Autumn
5.74E-05
1.32E-05
8.77E-06
7.60E-04
2.70E-03
8.32E-04
1.45E-03
6.53E-05
6.16E-04
2.06E-03
3.09E-04
7.44E-05
4.31E-04
2.07E-07
Spring
5.49E-05
1.06E-05
5.19E-06
8.23E-04
4.48E-03
5.12E-04
1.03E-03
5.02E-05
4.45E-04
1.18E-03
2.65E-04
5.31E-05
3.93E-04
1.56E-07
Risk level for Adult
Autumn
Low
Low
Low
Moderate
High
Moderate
High
Low
Moderate
High
Moderate
Low
Moderate
Very Low
Spring
Low
Low
Low
Moderate
High
Moderate
High
Low
Moderate
High
Moderate
Low
Moderate
VeryLow
CRSdermal for Childern
Autumn
7.17E-04
1.65E-04
1.10E-04
9.50E-03
3.38E-02
1.04E-02
1.81E-02
8.16E-04
7.71E-03
2.58E-02
3.86E-03
9.30E-04
5.38E-03
2.59E-06
Spring
6.87E-04
1.32E-04
6.48E-05
1.03E-02
5.60E-02
6.40E-03
1.29E-02
6.27E-04
5.56E-03
1.47E-02
3.31E-03
6.63E-04
4.92E-03
1.95E-06
Risk level for Childern
Autumn
Moderate
Moderate
Moderate
High
High
High
High
Moderate
High
High
High
Moderate
High
Low
Spring
Moderate
Moderate
Low
High
High
High
High
Moderate
High
High
High
Moderate
High
low
3.5 Principal component analysis
For the first main component, the variance percentage was 46.96%. The component matrix showed that the four-ring PAHs Flu, Pyr, BaA, and Chr, the five- and six-ring PAHs BbF, BaP and IP, and the two- and three-ring PAHs Naph, Acthy, Ace and Phe had the highest contribution rates. Therefore, the majority of PAHs that were either petrogenic or pyrogenic showed a high loading in PC1. As a result, PAH genesis in Red Sea coast sediments is influenced by both petrogenic and pyrogenic sources. Additionally, these substances are mostly composed of high molecular weight PAHs with 4–6 rings and primarily result from incomplete combustion and pyrolysis of fuel (Chen et al., 2012; Zhang et al., 2012). High Naph loading is another thing to note. Acthy and Ace oil spills are where the majority of naphtha is found (Ekpo et al., 2012). Phe and Flu are thermodynamically stable 3- and 4-ring PAHs, and their abundance is assumed to indicate petrogenic origins (Saha et al., 2009; Wang et al., 2011).
The component matrix revealed that several three-ring PAHs had substantial amounts of Ace, F, Phe, and Ant. The variance percentage of the second main component was 26.84%. The primary sources of three-ring PAHs with low molecular weight were petrogenic or low-temperature combustion processes (Harrison et al., 1996;Jenkins et al., 1996). In addition, Ace, Phe, Ant, and Flu are common coal compounds that are not entirely burned (Larsen and Baker, 2003). According to a previous study (Yunker et al., 2002;Zakaria et al., 2002;Boonyatumanond et al., 2006) Phe and F were identified in larger proportions in crude oil, crankcase oil, and unburned fuel, and it was believed that they originated from diesel engine soot. The Ace, F, Phe, Ant, Flu, and Pyr contexts were substantially laden with TOC due to the presence of these components (PC2). These compounds played an important part in the silt's organic matter.
As indicated by the third principal component variance percentage of 11.42 percent, there was a great deal of BkF in the component matrix. BkF is one of the markers of gasoline and diesel engine emissions (Zakaria et al., 2002;Larsen and Baker, 2003;Boonyatumanond et al., 2006;Liu et al., 2017b).
The results of our PCA indicate that incomplete combustion and pyrolysis of fuel are the principal pyrogenic sources of PAHs in surface sediments. In comparison, the inputs from the petrogenic source, such as oil spills, are insignificant (Fig. 5).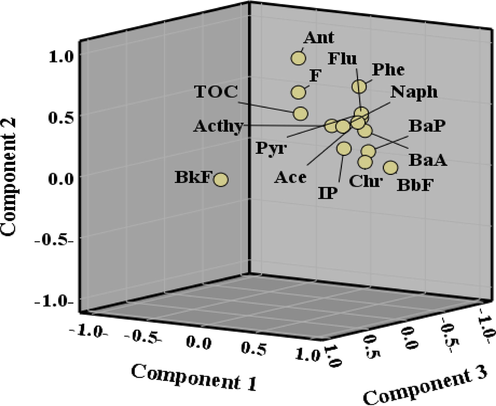
The component plot of PAHs in the investigated sediment samples.
4 Conclusions
The study area's fourteen surface sediment locations tested sixteen polycyclic aromatic hydrocarbons (PAHs). The composition profile revealed that sediments were predominantly composed of high molecular weight (4-ring) PAHs. The majority of the study area appeared to have a pyrogenic origin for PAHs based on the diagnostic ratios. According to the RQNCs and RQMPCs values, the bulk of the PAHs examined in this area constitute a significant ecological risk level. According to the ecotoxicological risk, PAH levels were found in several stations (ERL/TEL and ERM/PEL), suggesting that the toxicity risk posed by PAHs was minimal and intermittent. The numerical effect-based sediment quality standards are met by this (SQGs). The TEQs ranged from 0.03 to 863.96 µg/g dry weight for benzo(a)pyrene and 0.09 to 1197.85µg/g dry weight for benzo[k] Fluoranthene, respectively (the two most toxic). The estimated cancer risk from PAH exposure in children and adults leads to a very low to high risk for adults and a low to high risk for children, which may be quite alarming. The levels of 16PAHs in the current investigation were high when compared to those discovered elsewhere. All activities creating more severe pollution in the area should be restricted to preserve the ecosystem's health for aquatic species and the human lives that benefit from them along the food chain. From the previous conclusion, we recommend the following:
-
This study's findings may help inform an additional discussion on the standard requirements for long-term monitoring of PAH and the existing legislation that should be used to address PAH pollution in Egypt's coastal areas and raise awareness among scientists and government agencies about the risks of cancer caused by pollution levels in the marine environment.
-
Due to many economic activities along the Red Sea coast, further monitoring studies encompassing different environmental matrices, the biological impact, and possible factors and diseases related to environmental pollution should be conducted.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Monitoring of pollution in sediments of the coasts in Egyptian Red Sea. Egypt. J. Pet.. 2016;25:133-151.
- [CrossRef] [Google Scholar]
- Levels, compositions, and quality of some Egyptian surface sediments from Suez Gulf, as integrated from polycyclic hydrocarbons. Energy Sources Part A.. 2017;39:664-672.
- [Google Scholar]
- Assessing the ecological risks from hydrocarbons in the marine coastal sediments of Jeddah. Red Sea. Environ. Monit. Assess.. 2019;191:180.
- [CrossRef] [Google Scholar]
- Occurrence, Distribution, Environmental Risk Assessment and Source Apportionment of Polycyclic Aromatic Hydrocarbons (PAHs) in Water and Sediments of the Liaohe River Basin. China. Bull. Environ. Contam. Toxicol.. 2014;93:744-751.
- [CrossRef] [Google Scholar]
- Polycyclic aromatic hydrocarbons in sediments and mussels of the western Mediterranean sea. Environ. Toxicol. Chem.. 1998;17:765-776.
- [CrossRef] [Google Scholar]
- Polycyclic aromatic hydrocarbons in recent sediments and mussels (Mytilus edulis) from the Western Baltic Sea: occurrence, bioavailability and seasonal variations. Mar. Environ. Res.. 1999;47:17-47.
- [CrossRef] [Google Scholar]
- Polycyclic aromatic hydrocarbons in imported Sardinops sagax: Levels and health risk assessments through dietary exposure in Nigeria. J. Food Composit. Anal.. 2017;57:109-116.
- [CrossRef] [Google Scholar]
- Analytical methods for determining metabolites of polycyclic aromatic hydrocarbon (PAH) pollutants in fish bile: a review. Environ. Toxicol. Pharmacol.. 2010;30:224-244.
- [CrossRef] [Google Scholar]
- PAH content, toxicity and genotoxicity of coastal marine sediments from the Rovinj area, Northern Adriatic. Croatia. Sci. Total Environ.. 2006;366:602-611.
- [CrossRef] [Google Scholar]
- Distribution and origins of polycyclic aromatic hydrocarbons (PAHs) in riverine, estuarine, and marine sediments in Thailand. Mar. Pollut. Bull.. 2006;52:942-956.
- [CrossRef] [Google Scholar]
- Evaluation of sediment contamination by polycyclic aromatic hydrocarbons in the Gironde estuary. Mar. Chem.. 1997;58:85-97.
- [CrossRef] [Google Scholar]
- Distribution and ecosystem risk assessment of polycyclic aromatic hydrocarbons in the Luan River. China. Ecotoxicol.. 2010;19:827-837.
- [CrossRef] [Google Scholar]
- Biodegradation of polycyclic aromatic hydrocarbons. Curr. Opin. Biotechnol.. 1993;4:331-338.
- [CrossRef] [Google Scholar]
- Distribution, origin, and potential toxicological significance of polycyclic aromatic hydrocarbons (PAHs) in sediments of Kaohsiung Harbor. Taiwan. Mar. Pollut. Bull.. 2011;63:417-423.
- [CrossRef] [Google Scholar]
- Source apportionment of polycyclic aromatic hydrocarbons (PAHs) in surface sediments of the Rizhao coastal area (China) using diagnostic ratios and factor analysis with nonnegative constraints. Sci. Total Environ.. 2012;414:293-300.
- [CrossRef] [Google Scholar]
- Partition Characteristics of Polycyclic Aromatic Hydrocarbons on Soils and Sediments. Environ. Sci. Technol.. 1998;32:264-269.
- [CrossRef] [Google Scholar]
- Maximum permissible and negligible concentrations for some organic substances and pesticides. J. Environ. Manage.. 2000;58:297-312.
- [CrossRef] [Google Scholar]
- Evaluation of surface sediment contamination by polycyclic aromatic hydrocarbons in colony Z3—(Patos Lagoon, Brazil) Microchem. J.. 2010;96:161-166.
- [CrossRef] [Google Scholar]
- Nature, distribution and origin of polycyclic aromatic hydrocarbons (PAHs) in the sediments of Olbia harbor (Northern Sardinia, Italy) Mar. Pollut. Bull.. 2005;50:1223-1232.
- [CrossRef] [Google Scholar]
- PAHs: an ecotoxicological perspective. New York, USA: John Wiley & Sons; 2003.
- Human cell mutagenicity of oxygenated, nitrated and unsubstituted polycyclic aromatic hydrocarbons associated with urban aerosols. Mutat. Res. Genet. Toxicol.. 1996;371:123-157.
- [CrossRef] [Google Scholar]
- Mutagenicity of C24H14 PAH in human cells expressing CYP1A1. Mutat. Res. Genet. Toxicol. Environ. Mutagen.. 1999;446:1-14.
- [CrossRef] [Google Scholar]
- Distributions and sources of polycyclic aromatic hydrocarbons in surface sediments from the Cross River estuary, S.E. Niger Delta. Nigeria. Environ. Monit. Assess.. 2012;184:1037-1047.
- [CrossRef] [Google Scholar]
- The distribution and sources of polycyclic aromatic hydrocarbons in surface sediments along the Egyptian Mediterranean coast. Environ. Monit. Assess.. 2007;124:343-359.
- [CrossRef] [Google Scholar]
- Aliphatic and polycyclic aromatic hydrocarbons in the surface sediments of the Mediterranean: assessment and source recognition of petroleum hydrocarbons. Environ. Monit. Assess.. 2013;185:4571-4589.
- [CrossRef] [Google Scholar]
- Distribution patterns and risks posed of polycyclic aromatic hydrocarbons contaminated in the surface sediment of the Red Sea coast (Egypt) Desalin. Water Treat.. 2014;52:7964-7982.
- [CrossRef] [Google Scholar]
- Spatial distribution, sources and risk assessment of polycyclic aromatic hydrocarbons in the surficial sediments of the Egyptian Mediterranean coast. Mar. Pollut. Bull.. 2023;188:114658
- [CrossRef] [Google Scholar]
- Seasonal and temporal influence on polycyclic aromatic hydrocarbons in the Red Sea coastal water. Egypt. Sustainability. 2021;13:11906.
- [CrossRef] [Google Scholar]
- Polyaromatic hydrocarbon (PAH) distributions in the Seine River and its estuary. Mar. Pollut. Bull.. 1997;34:857-867.
- [CrossRef] [Google Scholar]
- Composition, Distribution, and Characterization of Polycyclic Aromatic Hydrocarbons in Soil in Linfen. China. Bull. Environ. Contam. Toxicol.. 2009;82:167-171.
- [CrossRef] [Google Scholar]
- An inexpensive titration method for the determination of organic carbon in recent sediments. J. Sediment Res.. 1974;44:249-253.
- [CrossRef] [Google Scholar]
- Seasonal Distribution, Source Identification, and Toxicological Risk Assessment of Polycyclic Aromatic Hydrocarbons (PAHs) in Sediments from Wadi El Bey Watershed in Tunisia. Arch. Environ. Contam. Toxicol.. 2017;73:488-510.
- [CrossRef] [Google Scholar]
- Are the tourist beaches safe for swimming? A case study of health risks of polycyclic aromatic hydrocarbons (PAHs) in tourist beaches of Bushehr City. Environ. Monit. Assess.. 2022;194:398.
- [CrossRef] [Google Scholar]
- Spatial, seasonal distribution and ecological risk assessment of Zn, Cr, and Ni in Red Sea surface sediments. Egypt. Egypt. J. Aquat. Biol. Fish.. 2021;25:513-538.
- [CrossRef] [Google Scholar]
- Source apportionment of atmospheric polycyclic aromatic hydrocarbons collected from an urban location in Birmingham, U.K. Environ. Sci. Technol.. 1996;30:825-832.
- [CrossRef] [Google Scholar]
- Sources of polycyclic aromatic hydrocarbons in sediments of the Bharalu River, a tributary of the River Brahmaputra in Guwahati. India. Ecotoxicol. Environ. Saf.. 2015;122:61-67.
- [CrossRef] [Google Scholar]
- IARC Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans: Polynuclear Aromatic Compounds, Part 1, Chemical, Environmental and Experimental Data. Lyon, France: International Agency for Research on Cancer; 1983.
- Emission Factors for Polycyclic Aromatic Hydrocarbons from Biomass Burning. Environ. Sci. Technol.. 1996;30:2462-2469.
- [CrossRef] [Google Scholar]
- Assessment of benzo (a) pyrene-equivalent carcinogenicity and mutagenicity of residential indoor versus outdoor polycyclic aromatic hydrocarbons exposing young children in New York City. Int. J. Environ. Res. Public Health.. 2010;7:1889-1900.
- [CrossRef] [Google Scholar]
- Environmental Quality Objectives for 10 Polycyclic Aromatic Hydrocarbons (PAHs) Ecotoxicol. Environ. Saf.. 1997;36:89-97.
- [CrossRef] [Google Scholar]
- Baseline distributions and sources of Polycyclic Aromatic Hydrocarbons (PAHs) in the surface sediments from the Prai and Malacca Rivers, Peninsular Malaysia. Mar. Pollut. Bull.. 2014;88:366-372.
- [CrossRef] [Google Scholar]
- Risk assessment of polycyclic aromatic hydrocarbons in a Mediterranean semi-enclosed basin affected by human activities (Abu Qir Bay, Egypt) J. Hazard. Mater.. 2009;170:389-397.
- [CrossRef] [Google Scholar]
- Source apportionment of polycyclic aromatic hydrocarbons in the urban atmosphere: a comparison of three methods. Environ. Sci. Technol.. 2003;37:1873-1881.
- [CrossRef] [Google Scholar]
- Source apportionment of polycyclic aromatic hydrocarbons (PAHs) in surface sediments of the Huangpu River, Shanghai. China. Sci. Total Environ.. 2009;407:2931-2938.
- [CrossRef] [Google Scholar]
- Distribution, source, and ecological risk assessment of polycyclic aromatic hydrocarbons (PAHs) in surface sediments from the Hun River, northeast China. Environ. Monit. Assess.. 2015;187:290.
- [CrossRef] [Google Scholar]
- Ecological risk analysis of polycyclic aromatic hydrocarbons (PAHs) in surface sediments from Laizhou Bay. Environ. Monit. Assess.. 2009;159:429-436.
- [CrossRef] [Google Scholar]
- Distribution, source analysis, and ecological risk assessment of polycyclic aromatic hydrocarbons in the typical topsoil of the Issyk-Kul Lake Basin. Environ. Monit. Assess.. 2017;189:1-11.
- [Google Scholar]
- Aliphatic and polycyclic aromatic hydrocarbons (PAHs) in soils of the northwest Qinling Mountains: patterns, potential risk and an appraisal of the PAH ratios to infer their source. J. Environ. Sci. Health., Part A. 2017;52:320-332.
- [CrossRef] [Google Scholar]
- Recommended uses of empirically derived, sediment quality guidelines for marine and estuarine ecosystems. Hum. Ecol. Risk Assess.. 1998;4:1019-1039.
- [CrossRef] [Google Scholar]
- Incidence of adverse biological effects within ranges of chemical concentrations in marine and estuarine sediments. Environ. Manage.. 1995;19:81-97.
- [CrossRef] [Google Scholar]
- Polycyclic aromatic hydrocarbons in soil-turfgrass systems in urban Shanghai: Contamination profiles, in situ bioconcentration and potential health risks. J. Clean. Prod.. 2021;289:125833
- [CrossRef] [Google Scholar]
- Development and evaluation of sediment quality guidelines for Florida coastal waters. Ecotoxicology.. 1996;5:253-278.
- [CrossRef] [Google Scholar]
- PAHs composition of surface marine sediments: a comparison to potential local sources in Todos Santos Bay, B.C. Mexico. Chemosphere. 2002;46:459-468.
- [CrossRef] [Google Scholar]
- Cancer risk assessments of Hong Kong soils contaminated by polycyclic aromatic hydrocarbons. J. Hazard. Mater.. 2013;261:770-776.
- [CrossRef] [Google Scholar]
- Distribution of aliphatic hydrocarbons, polycyclic aromatic hydrocarbons and organochlorinated pollutants in deep-sea sediments of the southern Cretan margin, eastern Mediterranean Sea: A baseline assessment. Chemosphere.. 2014;106:28-35.
- [CrossRef] [Google Scholar]
- Relationships between toxicity and concentrations of chemical contaminants in sediments from Sydney Harbour, Australia, and Vicinity. Environ. Monit. Assess.. 2006;120:187-220.
- [CrossRef] [Google Scholar]
- Exposure to carcinogenic PAHs in the environment. Environ. Sci. Technol.. 1992;26:1278-1284.
- [CrossRef] [Google Scholar]
- Polycyclic aromatic hydrocarbons in the aquatic environment. Sources, fates and biological effects. London, UK: Applied Science Publishers Ltd.; 1979.
- Toxic equivalency factors (TEFs) for polycyclic aromatic hydrocarbons (PAHs) Regul. Toxicol. Pharm.. 1992;16:290-300.
- [CrossRef] [Google Scholar]
- Polycyclic aromatic hydrocarbons (PAHs) in surface sediments of Monastir Bay (Tunisia, Central Mediterranean): distribution, origin and seasonal variations. Int. J. Environ. Anal. Chem.. 2013;93:1470-1483.
- [CrossRef] [Google Scholar]
- Contamination from polycyclic aromatic hydrocarbons (PAHs) in the soil of a botanic garden localized next to a former manufacturing gas plant in Palermo (Italy) J. Hazard. Mater.. 2010;180:590-601.
- [CrossRef] [Google Scholar]
- Introduction to the Red Sea: Its Origin, Structure, and Environment. In: Rasul N.M.A., Stewart I.C.F., eds. The Red Sea: The Formation, Morphology, Oceanography and Environment of a Young Ocean Basin. Berlin, Heidelberg: Springer; 2015. p. :1-28.
- [Google Scholar]
- Atmospheric polycyclic aromatic hydrocarbons: source attribution, emission factors and regulation. Atmos. Environ.. 2008;42:2895-2921.
- [CrossRef] [Google Scholar]
- Identification of hydrocarbon sources in contaminated soils of three industrial areas. Sci. Total Environ.. 2013;450–451:13-21.
- [CrossRef] [Google Scholar]
- Marine sediment contamination of an industrial site at Port of Bagnoli, Gulf of Naples, Southern Italy. Mar. Pollut. Bull.. 2004;49:487-495.
- [CrossRef] [Google Scholar]
- Hazard and risk assessment of chemical mixtures using the toxic equivalency factor approach. Environ. Health Perspect.. 1998;106:1051-1058.
- [CrossRef] [Google Scholar]
- Sources of sedimentary PAHs in tropical Asian waters: Differentiation between pyrogenic and petrogenic sources by alkyl homolog abundance. Mar. Pollut. Bull.. 2009;58:189-200.
- [CrossRef] [Google Scholar]
- The monitoring and risk assessment of aliphatic and aromatic hydrocarbons in sediments of the Red Sea. Egypt. Egypt. J. Aquat. Res.. 2014;40:333-348.
- [CrossRef] [Google Scholar]
- Polycyclic aromatic hydrocarbons (PAHs) and organochlorines (OCs) in bottom sediments of the Guba Pechenga, Barents Sea. Russia. Sci. Total Environ.. 2003;306:39-56.
- [CrossRef] [Google Scholar]
- Biodegradation of polycyclic aromatic hydrocarbons by a bacterial consortium enriched from mangrove sediments. J. Environ. Health Sci. Eng.. 2014;12:114.
- [CrossRef] [Google Scholar]
- Aliphatic and aromatic hydrocarbons in different sized aerosols over the Mediterranean Sea: occurrence and origin. Atmos. Environ.. 1987;21:2247-2259.
- [CrossRef] [Google Scholar]
- Origin of Polycyclic Aromatic Hydrocarbons (PAHs) in Coastal Marine Sediments: case Studies in Cotonou (Benin) and Aquitaine (France) Areas. Mar. Pollut. Bull.. 2000;40:387-396.
- [CrossRef] [Google Scholar]
- Chemical fractions and ecological risk of metals in sediments of the Bitter Lakes. Egypt. Egypt. J. Aquat. Biol. Fish.. 2020;24:167-196.
- [CrossRef] [Google Scholar]
- Distribution of polycyclic aromatic hydrocarbons (PAHs) in Henan Reach of the Yellow River, Middle China. Ecotoxicol. Environ. Saf.. 2009;72:1614-1624.
- [CrossRef] [Google Scholar]
- The distribution and relative toxic potential of organic chemicals in a PCB contaminated bay. Mar. Pollut. Bull.. 2005;50:195-207.
- [CrossRef] [Google Scholar]
- Seasonal distribution and risk assessment of polycyclic aromatic hydrocarbons (PAHs) in surface sediments from the Giresun coast of southeastern Black Sea. Mar. Pollut. Bull.. 2022;178:113585
- [CrossRef] [Google Scholar]
- Polycyclic aromatic hydrocarbon contamination in coastal sediments of the Izmit Bay (Marmara Sea): case studies before and after the Izmit Earthquake. Environ. Int.. 2006;32:758-765.
- [CrossRef] [Google Scholar]
- UNEP/FAO/IAEA/IOC, 1991. Sampling of selected marine organisms and sample preparation for the analysis of chlorinated hydrocarbons, Reference methods for marine pollution studies No. 12. Nairobi, Kenya United Nations Environment Programme.
- UNEP/IOC/IAEA, 1992. Determination of petroleum hydrocarbons in sediments. Nairobi, Kenya, UNEP.
- Distribution and sources of hydrocarbons in surface sediments of Gemlik Bay (Marmara Sea, Turkey) Chemosphere.. 2006;64:764-777.
- [CrossRef] [Google Scholar]
- USEPA, 2004. Risk Assessment Guidance for Superfund Volume 1: Human Health Evaluation Manual (part E). Washington, DC, USA, US Environmental Protection Agency.
- Verbrugge, L., 2008. Health consultation: evaluation of blue mussel samples collected in May 2006 M/V Seledang Ayu Oil Spill, Unalaska, Alaska. USA, Research report of U.S. Department of Health and Human Services, Public Health Service, Agency for Toxic Substances and Disease Registry.
- PAHs distribution in precipitation at Mount Taishan: China. Identification of sources and meteorological influences. Atmos. Res.. 2010;95:1-7.
- [CrossRef] [Google Scholar]
- Overall comparison and source identification of PAHs in the sediments of European Baltic and North Seas, Chinese Bohai and Yellow Seas. Sci. Total Environ.. 2020;737:139535
- [CrossRef] [Google Scholar]
- Sources and distribution of aliphatic and polyaromatic hydrocarbons in sediments of Jiaozhou Bay, Qingdao. China. Mar. Pollut. Bull.. 2006;52:129-138.
- [CrossRef] [Google Scholar]
- Sources and distribution of aliphatic and polycyclic aromatic hydrocarbons in Yellow River Delta Nature Reserve, China. Appl. Geochem.. 2011;26:1330-1336.
- [CrossRef] [Google Scholar]
- Polycyclic aromatic hydrocarbons in water and sediment of the Baltic Sea. Mar. Pollut. Bull.. 1995;31:237-248.
- [CrossRef] [Google Scholar]
- Polycyclic aromatic hydrocarbons in the surface sediments from Yellow River. China. Chemosphere.. 2007;67:1408-1414.
- [CrossRef] [Google Scholar]
- Distribution, sources and potential toxicological significance of polycyclic aromatic hydrocarbons (PAHs) in the south west coast of Kerala, India. Mater. Today Proc.. 2021;41:736-743.
- [CrossRef] [Google Scholar]
- Cancer risk of polycyclic aromatic hydrocarbons (PAHs) in the soils from Jiaozhou Bay wetland. Chemosphere.. 2014;112:289-295.
- [CrossRef] [Google Scholar]
- Levels, sources and potential risks of polycyclic aromatic hydrocarbons (PAHs) in multimedia environment along the Jinjiang River mainstream to Quanzhou Bay, China. Mar. Pollut. Bull.. 2013;76:298-306.
- [CrossRef] [Google Scholar]
- Concentrations, source and risk assessment of polycyclic aromatic hydrocarbons in soils from Midway Atoll, North Pacific Ocean. PLOS ONE.. 2014;9:e86441.
- [Google Scholar]
- Environmental Impacts on Egyptian Delta Lakes’ Biodiversity: A Case Study on Lake Burullus. In: Negm A.M., Bek M.A., Abdel-Fattah S., eds. Egyptian Coastal Lakes and Wetlands: Part II: Climate Change and Biodiversity. Cham, Switzerland: Springer International Publishing; 2019. p. :107-128.
- [Google Scholar]
- Assessment of polycyclic aromatic hydrocarbons in surface sediments and some fish species from the Gulf of Suez, Egypt. Egypt. J. Aquat. Biol. Fish.. 2018;22:49-59.
- [CrossRef] [Google Scholar]
- Effect of different molecular weights of chitosan on the removal efficiencies of heavy metals from contaminated water. Egypt. J. Aquat. Biol. Fish.. 2019;23:149-158.
- [CrossRef] [Google Scholar]
- Distribution and ecological risk evaluation of bioavailable phosphorus in sediments of El Temsah Lake, Suez Canal. Oceanologia.. 2022;64:287-298.
- [CrossRef] [Google Scholar]
- PAHs in the Fraser River basin: a critical appraisal of PAH ratios as indicators of PAH source and composition. Org. Geochem.. 2002;33:489-515.
- [CrossRef] [Google Scholar]
- Distribution of Polycyclic Aromatic Hydrocarbons (PAHs) in Rivers and Estuaries in Malaysia: A Widespread Input of Petrogenic PAHs. Environ. Sci. Technol.. 2002;36:1907-1918.
- [CrossRef] [Google Scholar]
- Potential source contributions and risk assessment of PAHs in sediments from Taihu Lake, China: comparison of three receptor models. Water Res.. 2012;46:3065-3073.
- [CrossRef] [Google Scholar]







