Translate this page into:
Production of higher toxic intermediates of organic pollutants during chemical oxidation processes: A review
⁎Corresponding author. longtao@nies.org (Tao Long)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
Abstract
The application of toxicity assessment methods in oxidation process was evaluated. A list of chemicals which easily generate more toxic intermediates were proposed. Products with oxygen, halogens, and coupling have a tendency to increase toxicity. The halogen ion and DOM have the potential to enhance the toxicity. Strategies to reduce residual toxicity and further research focus are suggested.
Abstract
Incomplete degradation of organic pollutants might lead to the increased environmental risk because of the potentially toxic intermediates, especially during chemical treatment processes. However, toxicity changes of common pollutants during chemical oxidation are still unclear because the toxicity results were easily influenced by oxidation means, reaction condition, toxicity assessment method, etc. This review summarized and evaluated the toxicity test and prediction methods commonly used in oxidation process, and then the potential toxic intermediates and their possible producing pathways was proposed as for polycyclic aromatic hydrocarbon, phenols, azo dyes, organophosphorus, organonitrogens, organochlorine pesticides, pharmaceuticals of different classes. Various intermediates, especially the oxygen-containing ones, such as hydroquinone-, benzoquinone-, catechol-like products and halogenated ones are prone to have higher toxicity while some of the dimers and polymers are highly toxic and refractory. In addition, environmental elements such as DOM and halogen ion have the potential to enhance the toxicity. The strategies to reduce the residual toxicity were usually to strengthen the reaction, for example, to increase the oxidant concentration or prolong reaction time. Coupling of multiple technologies is also an effective strategy.
Keywords
Oxidation intermediate
Toxicity assessment
Pesticide
Pharmaceutical
Toxicity elimination
1 Introduction
Chemical oxidation is a popular technology applied in the water treatment and soil remediation processes due to the benefits of its low energy consumption, low cost, and applicability for a variety of organic pollutants. The widely used chemical oxidation methods include Fenton and Fenton-like, permanganate, persulfate, peroxymonosulfate, chlorination, sonolysis, photolytic and photocatalytic degradation, ozonation, ferrate oxidation, etc. The oxidants either utilize their own oxidizing properties or produce highly reactive species, such as hydroxyl radicals (•OH), sulfate radicals, superoxide radicals (•O2–), singlet oxygen, to degrade organic pollutants (Kanakaraju et al., 2018; Wang and Wang, 2020). When chemical oxidation technology is used to treat water or soil, a continuously decreased toxicity is a desirable benefit. However, in some cases the intermediates are more toxic or even less biodegradable than the parent compound, and if they were discharged to the environment, they might pose threats to the ecosystem or human health and have the potential to cause secondary environmental problems (Usman et al., 2016; Wang and Wang, 2021). For example, 2-anilino-5-[(4-methylpentan-2-yl)amino]cyclohexa-2,5-diene-1,4-dione (6PPD-quinone), an ozonation product of the globally ubiquitous tire rubber antioxidant N-(1,3-dimethylbutyl)-N'-phenyl-p-phenylenediamine (6PPD), has the potential to kill adult salmon (Seiwert et al., 2022; Tian et al., 2021). Therefore, when assessing the overall performance of a chemical oxidation treatment technique, the toxicity and persistency of reaction intermediates should also be taken into account, but there is still no general agreement or standard on the assessment method. Although that some studies have measured or calculated the toxicity of oxidative intermediates (Cao et al., 2013; Li et al., 2022a; Magdeburg et al., 2012; Trovó et al., 2013), they were fragmented and most of the intermediates are less harmful than the parent compound. Seldom studies have systematically examined the potential highly toxic intermediates and their formation pathway. Therefore, it is still difficult to identify which pollutants should be given special consideration when considering chemical oxidation treatment.
In this work, the toxicity test methods suitable for evaluating toxicity change during chemical oxidation process were summarized. Then almost a thousand of articles were screened and five categories of pollutants, namely polycyclic aromatic hydrocarbons (PAHs), phenol, azo dyes, pesticide, and pharmaceuticals, obtained relatively focused attention as having the potential to increase toxicity. An inventory of potential highly toxic intermediates of these pollutants was thus established, and their generation mechanisms were summarized. Furthermore, the environmental elements that might make chemical oxidation treatments more harmful are summarized, and potential solutions to lessen residual toxicity are proposed. This review will provide insights to establish a more comprehensive approach to assess the overall effectiveness and environmental benefits of chemical oxidation treatment techniques, help identify which pollutants should be given special consideration when considering chemical oxidation treatment, and provide some reference on how to control this potential secondary risk.
2 Toxicity assessment methods
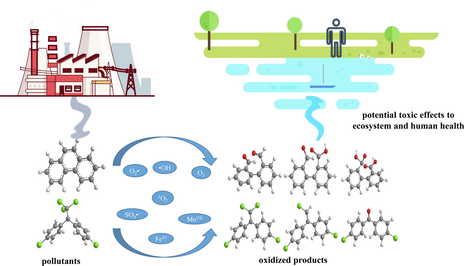
The common toxicity assessment methods reflecting oxidation effectiveness are summarized in this section. A total of 189 papers reporting changes in toxicity caused by oxidation were collected, and the application ratio of different toxicity assessment methods is summarized in Fig. 1. As some studies used more than one method, a total of 241 tests were recorded. Results indicated that the most widely used method was luminescence inhibition test, which was adopted in 34.4% of the reviewed tests, and Aliivibrio fischeri (including Vibrio fischeri (V. fischeri)) were the most commonly used test species. The frequency of other test techniques' use is as follows: aquatic toxicity test (using indicator species like brine shrimp, fish, protozoa, and fleas) > bacteriostatic impact test > growth inhibition observation of plants. Other test methods such as genotoxicity, estrogenic activity, endocrine disrupting effects were also applied, but in only a few studies. It is noteworthy that remaining oxidants and radicals must be completely eliminated before conducting any toxicity assessments due to their obvious impact on the toxicity (Gernjak et al., 2006; Mantzavinos and Kalogerakis, 2005; Ribeiro et al., 2015).
Application ratio of different toxicity assessment methods in oxidation process.
2.1 Luminescence inhibition test
The luminescent bacteria assay is the most frequently employed acute toxicity test method due to its convenient operation, high sensitivity, and low cost (Parvez et al., 2006; Zhang et al., 2017). This method was used with various pollutants in either water or soil medium (Lu et al., 2010; Usman et al., 2016). Studies have revealed a strong association between the toxicity of organic pollutants and the luminance within a certain concentration range of pollutants (Ma et al., 2014). The marine luminescent bacterium V. fischeri, Photobacterium phosphoreum (P. phosphoreum) and freshwater bacterium Vibrio qinghaiensis Q67 are the three most commonly used luminescent bacteria (Wang and Wang, 2019; Xiong et al., 2020), in which V. fischeri is used in the international standard method (International Organization for Standardization, 2007), and P. phosphoreum is used in the national standard method of China (Ministry of Ecology and Environment of The People's Republic of China, 1995). Marine luminescent bacteria need to add a certain amount of NaCl (2%∼5%) under the determination conditions, which is different from the freshwater test system. Therefore, in 1985, Chinese scholars introduced Vibrio qinghaiensis Q67 as the test object of chemical toxicity test and it was gradually widely used (Zhu et al., 1994). In the supporting information, Table S1 provides an overview of the most widely used international published toxicity standard test methods.
2.2 Aquatic toxicity test
Common aquatic toxicity test methods have been summarized in Whole Effluent Toxicity methods by U.S. Environmental Protection Agency (U.S. EPA) (EPA, 2016). The test species used to assess the oxidation-induced toxicity include water flea, algae, fish, brine shrimp, protozoa, and so on.
Water fleas are widely distributed in freshwater, among which Daphnia magna (D. magna) is the most commonly used test organism to evaluate the toxicity, and its median lethal concentration (LC50) and median effect concentration (EC50) by activity inhibitory are usually used as acute toxicity evaluation indexes. For example, Garcia-Muñoz et al. (2020) used the 48 h LC50 and EC50 values of D. magna to determine the toxicity changes of glyphosate and myclobutanil pesticides in photocatalytic degradation. Dehghan et al. (2019) measured the LC50 of D. magna at 24, 48, 72 and 96 h, respectively, so as to assess the toxicity change of metalaxyl pesticides under the graphene/Fe3O4/ZnO ternary nanomaterials catalyzed photodegradation.
Algae, which are crucial to the balance and stability of aquatic ecosystems, is another widely used organism because of their small size, rapid reproduction, ease of isolation and culture. The toxicity symptoms could be observed directly at the cellular level as well. Common evaluation indexes include the inhibition effect on algae growth and the effect on photosynthesis. Zhang et al. and Rao et al. (Rao et al., 2019; Zhang et al., 2020) observed the growth inhibition to Chlorella vulgaris (C. vulgaris) to track the toxicity change during sulfamethoxazole (SMX) and diclofenac oxidation in UV-persulfate and LaFeO3-catalyzed peroxymonosulfate systems, respectively. Other algas such as Selenastrum capricornutum, Chlorella pyrenoidos, and Desmodesmus subspicatus were also used as the test organisms in such studies (Fang et al., 2013; Kim et al., 2012; Zhang et al., 2016; Zhu et al., 2016).
Fish bioassay could be used to predict the possible effects on vertebrates, and even humans (Kime, 1999; Wester et al., 2004). Zebrafish is the most widely used fish species as the standard test organism for acute toxicity test. In addition to acute mortality, the main toxicity indicators also include embryo hatching rate, teratogenic effect, endocrine disrupting effect, etc. Willis and Oris (Willis and Oris, 2014) comprehensively evaluated the photocatalytic degradation toxicity of 4-nitrophenol with zebrafish mortality, hatching rate and phenotypic abnormalities. de Sousa Filho et al. (2019) evaluated the toxicity of etoposide solution (an antineoplastic agent) after photodegradation by using zebrafish embryo acute lethal effect, comet assay and micronucleus test.
Immobilization effects of other protozoa such as Tetrahymena pyriformis, Caenorhabditis elegans, Chironomus riparius and brine shrimp (Artemia salina) were also used at a low frequency according to our statistics.
2.3 Other toxicity tests
Microorganism test were mainly used to assess the bacteriostatic effect of antibiotics or phenol toxicity of in oxidation process, and Escherichia coli (E. coli) was the most commonly used (Carneiro et al., 2020; Huang et al., 2020; Liu et al., 2020; Olak-Kucharczyk and Ledakowicz, 2017; Zhang et al., 2018).
Seed germination rate, root elongation inhibitory effect, and growth effect (biomass, height, length, etc.) of plants such as Lemna minor, broad beans, onion, lettuce are also important indictors to measure the toxicity during oxidation process, which could be used in both wastewater and soil (Castro et al., 2014; Hou et al., 2014; Magdeburg et al., 2012; Zhang et al., 2004).
In addition to the above biological experiments, a few studies have looked into how other biological cells' genotoxicity, estrogen activity, and endocrine disrupting effects change as a result of chemical oxidation. Silva et al. (2021) tested the genetic toxicity of onion bulb root cells using granular activated carbon combined with chemical oxidant to treat wastewater. Cédat et al. (2016) tested the change of estrogenic activity in wastewater in UV photocatalysis and UV/H2O2 system. Because these test methods are more complex and time-consuming than acute toxicity, their application rate is relatively low.
Since the analysis on intermediates in soil is much more difficult than that in aqueous media, studies on the toxicity changes of organic contaminants following oxidation in soil are far less common, and such studies are mainly focused on PAH and pesticides. It is noteworthy that the results of soil toxicity tests may be related to the impact on soil properties, in addition to the transformation of the contaminants themselves (O Mahony et al., 2006; Olmez-Hanci et al., 2014; Sirguey et al., 2008; Tsitonaki et al., 2008).
2.4 Toxicity prediction method
In addition to conducting overall toxicity tests on the reaction system, toxicity prediction based on intermediate identification is extensively adopted. The intermediates could be analyzed with liquid chromatography/mass spectrometry (LC/MS), gas chromatograph/mass spectrometer (GC/MS), ion chromatography, etc. Then, the toxicity of the intermediates could be predicted with quantitative structure–activity relationship (QSAR) method (Mansouri et al., 2018), or by consulting literatures. The QSAR method is used more commonly because it can eliminate the problem resulted from inconsistent toxicity data sources and is easier to operate. The free software Ecological Structure-Activity Relationship Model (ECOSAR) and Toxicity Estimation Software Tool (T.E.S.T.) developed by the U.S. EPA are the most commonly used toxicity prediction software.
2.4.1 Ecosar
ECOSAR has a QSAR database of aquatic toxicity overlaid with an expert decision tree for choosing the proper chemical categories based on chemical structures. It classifies chemicals into 111 categories of neutral organic compounds, highly toxic organic compounds, surfactants, polymers, etc., and has 704 QSAR models. It can assess the toxicity of chemicals by entering the chemical name, CAS number and other information, or drawing the compound structure diagram. ECOSAR can be used to predict chemicals’ acute and chronic toxicity to aquatic organisms such as fish, aquatic invertebrates and plants. It also compares the toxicity prediction endpoint to the compound’s water solubility. ECOSAR is only applicable to the prediction of acute toxicity for compounds with logKow ≤ 5.0 and chronic toxicity for compounds with logKow ≤ 8.0.
2.4.2 T.E.S.T.
Similar to ECOSAR, the user can predict the toxicity of a compound by entering its information in T.E.S.T. software. T.E.S.T. had seven toxicity endpoints, including 96-hour fathead minnow LC50, 48-hour D. magna LC50, tetrahymena pyriformis 50 percent growth inhibition concentration (IGC50), oral rat 50 percent lethal dose (LD50), bioconcentration factors and developmental toxicity of fish, and Ames mutagenicity. In contrast to ECOSAR, T.E.S.T. is not only suitable for aquatic toxicity prediction (Ji et al., 2020; Li et al., 2022a; Liu et al., 2020). As a result, the toxicity of intermediate oxidation products in soil was usually predicted with T.E.S.T. (Shi et al., 2017; Wu et al., 2022; Yuan et al., 2020).
As shown in Fig. 1, ECOSAR was used in 35 studies and T.E.S.T. was used 17 times. The use ratio of toxicity prediction method was about 23.2% in the collected studies.
2.5 Comparison of different toxicity assessment methods
Acute toxicity test is the most often utilized test index in the toxicity assessment of the chemical oxidation process of pollutants. Other toxicity assessment methods are relatively rarely used due to complicated procedures, long time, and lack of standard methods. However, existing studies have shown that different toxicity test methods may lead to differences in conclusions. For example, for phenol, although the oxidation intermediate is more toxic according to the toxicity database, the direct toxicity test of the reaction solution has reached the opposite conclusion (Zazo et al., 2007); for SMX, when C. vulgaris is used as the test substance, the solution toxicity will slightly increase after photocatalytic treatment (Zhang et al., 2020). This is consistent with the results from the V. fischeri assay (Zhang et al., 2016), but not the results from D. magna assay (Yazdanbakhsh et al., 2020). In addition, some pollutants have specific toxicity. For example, organochlorine pesticides often act by poisoning the nervous system of the target organism (Costa, 2015). For another, chemicals such as nalidixic acid, chloramphenicol and streptomycinsulfate have delayed toxicity. The standard rapid toxicity determination method will make the measured value smaller, resulting in the easily overlooked harm to the environment (Froehner et al., 2000). Therefore, diversified toxicity endpoints should be covered when selecting toxicity test indicators according to the risk assessment needs and the toxicity mechanisms of the parent compound. Toxicity test methods could reflect the real toxicity of the oxidation system objectively, but toxicity experiments are costly and the results are more sensitive to reaction conditions.
Using the prediction method to assess product toxicity can effectively reduce the number of toxicity experiments, but it has the following shortcomings: (1) Not all toxicity endpoints can be obtained in a predictive manner, especially the ecotoxicity and mammalian chronic toxicity that can be used for soil ecological and human health risk assessment; (2) Degradation products need to be identified before prediction, and inaccurate identification of degradation products may lead to misjudgment of toxicity; (3) The prediction method is only applicable to a single organic compound, while the toxicity of the pollutant oxidation process is often formed by the combination of pollutant itself and a variety of intermediate products. Using the prediction method may cause a big difference compared with the actual toxicity. The combined use of toxicity test and prediction method would deepen the understanding on mechanisms of increased toxicity.
3 Highly toxic products of typical pollutants
3.1 PAHs
PAHs are widely distributed in soil and sediment, with carcinogenic, teratogenic and mutagenic effects. Some PAHs are difficult to biodegrade (Musa Bandowe et al., 2010; Wilson and Jones, 1993; Zhao et al., 2021). Sixteen kinds of PAHs have been listed as priority pollutants by U.S. EPA based on their wide distribution and strong toxicity.
The oxidation intermediates of PAH mainly consist of oxygen-containing polycyclic aromatic hydrocarbons (oxy-PAHs) such as quinone, ketone, aldehyde, carboxylic acid, etc. (Forsey et al., 2010; Lundstedt et al., 2006). Some oxy-PAHs have high toxicity and mutagenicity (Chesis et al., 1984; Reisen and Arey, 2002; Xue and Warshawsky, 2005), such as benzo[a]pyrene quinone and phenanthrenequinone, which show mutagenic activity and can directly react with DNA (Chesis et al., 1984). The epoxy products generated by oxidation of acenaphthene and acenaphthylene can also act as electrophilic reagents to cause DNA damage (Reisen and Arey, 2002). The main photooxidation product of phenanthrene, i.e., 9,10-phenanthraquinone, has been proved to have a stronger potency to inhibit duckweed growth and luminous intensity of luminescent bacteria than the parent compound (McConkey et al., 1997). According to the test of Lemna gibba L. G3 growth inhibition effects conducted to 11 main photo-oxidation products (anthraquinones and hydroxyanthraquinones) of anthracene, about half of the further photo-degradation products of anthraquinones were more hazardous than their parent compounds. Among them, 2-hydroxyanthraquinone is the most toxic compound in anthracene oxidation products, and is not subject to further photooxidation, showing the risk of persistent environmental damage (Mallakin et al., 1999). Liao et al. (2014) studied the formation and transformation of PAH reaction by-products in the persulfate oxidation process of PAHs from the soil of a coking plant. The results showed that there were oxy-PAH in the soil, such as 9H-fluoren-9-one, 1H-phenalen-1-one, 1(2H)-acenaphthylenone and 1,8-naphthalic anhydride, and more oxy-PAHs were generated after oxidation, including naphthalene 1-(2-hydroxypropyl), 1H-phenalen-1-one, dibenzofuran, 9H-fluoren-9-one, 1,8-naphthalic anhydride. Among them, the naphthalene 1-(2-hydroxypropyl) and 1,8-naphthalic anhydride of the highest content were presumed to be the oxidation intermediates of 1-methyl anthraquinone and acenaphthylene, respectively, and the rate of further oxidative removal was slow. But fortunately, PAHs and most of its oxidation products were removed after sufficient oxidation, reducing the integrated environmental risk. Shemer and Linden (2007) used photocatalytic oxidation to degrade the mixed pollution solutions of fluorene (FLU), dibenzofuran (DBF), dibenzothiophene (DBT) in water, and found that the toxicity of the mixed solution was enhanced during the oxidation process, which may be related to the oxidation intermediate product oxy-PAHs. In addition to oxy-PAHs, the ozonation of phenanthrene will also result to the formation of biphenyl compounds, which are difficult to biodegrade (O Mahony et al., 2006; Stehr et al., 2001). Toxicity tests by Stehr et al. (2001) using Bacillus subtilis and garden cress Lepidium sativum showed that compared with phenanthrene itself, the soil after ozonation showed higher dehydrogenase inhibition toxicity or cress growth inhibition, which may be attributed to intermediate products. The main intermediate products were identified as biphenyl compounds including 2,2′-biphenyldialdehyde, 2′-formyl-2-biphenylic acid and diphenic acid. What’s more, the oxidizable and biodegradable properties of pollutants were not significantly improved after ozonation. A list of potentially produced highly hazardous intermediates from the oxidation of various pollutants is presented in Table 1.
No.
Category
pollutants
oxidizing agent
reaction medium
toxicity test method
proposed highly toxic intermediates
refractory or not
references
1
PAH
phenanthrene
simulated solar radiation (SSR)
water
luminescence of Photobacterium phosphoreum, growth inhibition of Lemna gibba (duckweed)
9,10-phenanthrenequinone (oxy-PAHs)
Y
(McConkey et al., 1997)
2
PAH
phenanthrene
ozone
soil
contact assay with Bacillus subtilis and Root elongation test with Lepidium sativum
2,2′-biphenyldialdehyde,2′-formyl-2-biphenylic acid, diphenic acid
(biphenyls)Y
(Stehr et al., 2001)
3
PAH
anthracene
photosynthetically active radiation (PAR) and SSR
water
growth inhibition of the duckweed Lemna gibba L. G-3
2-hydroxyanthraquinone (oxy-PAHs)
Y
(Mallakin et al., 1999)
4
phenol
phenols
Fenton, wet catalytic oxidation, catalytic oxidation
water
Microtox toxicity test
catechol, hydroquinone, p-benzoquinone and their derivative compounds
N
(Santos et al., 2004; Santos et al., 2006; Svenson and Hynning, 1997; Zazo et al., 2007)
5
phenol
2-chlorophenol
Fenton
water
retrieve toxic equivalency factor data
PCDD/PCDF
Y
(Altarawneh et al., 2007; Vallejo et al., 2015)
6
phenol
bisphenol A
thermally activated persulfate
water
luminescence inhibition with V. fischeri
2,3-dimethyl benzoic acid (DMBA)
N
(Olmez-Hanci et al., 2013)
7
phenol
bisphenol A
ferrate(VI)
water
luminescence inhibition with V. fischeri
benzoquinone, hydroquinone, styrene
N
(Han et al., 2015)
8
phenol
bisphenol A
cyclodextrin enhanced photocatalytic system
water
T.E.S.T.
catechol derivatives formed by bond breaking such as 4-prop-1-en-2-ylbenzene-1,2-diol; hydroxylated products such as 5-hydroxybisphenol A, 4,5-bisphenol-o-quinone; isopropyl removal product such as 4,4′-dihydroxybiphenyl; coupling products
The biphenyl and coupling products are refractory
(Zhou et al., 2020)
9
phenol
bisphenol S (BPS)
peroxydisulfate (PDS) ordissolved oxygen (DO) in ultrasound (US)
systemwater
luminescence inhibition with V. fischeri
bond breaking product (p-hydroxybenzenesulfonic acid), oxygen-connected coupling product and hydroxylated product (BPS catechol)
The coupling product is refractory
(Lu et al., 2019)
10
phenol
bisphenol F
ferrate [Fe(VI)]
water
luminescence inhibition with Photobacterium phosphoreum and ECOSAR
coupling products and demethylation product
Y
(Yang et al., 2020)
11
azo dye
AO7
Electro-Fenton process using carbon felt cathode
water
luminescence inhibition with Vibrio fischeri
1,2-naphthaquinone, 1,4-benzoquinone
N
(Le et al., 2016)
12
azo dye
nitrotoluenesulfonic acid
wet air oxidation
water
toxicity test on the enriched nitrosomonas culture
cresol, nitrobenzene, phenol, quinolinol, 2-chlorophenol (oxy-products)
N
(Hao and Phull, 1993)
13
pesticide
chlorpyrifos
ultrasonic irradiation
water
luminescence inhibition with Vibrio qinghaiensis Q67
chlorpyrifos oxon
Y
(Zhang et al., 2011)
14
pesticide
alachlor
Fenton
water
luminescence inhibition with Vibrio fischeri
2,6-diethylaniline
Y
(Sanchis et al., 2014)
15
pesticide
diuron
Fenton
water
luminescence inhibition with Vibrio fischeri
3,4-dichloroaniline
Y
(Sanchis et al., 2014)
16
pesticide
atrazine
Fenton
water
luminescence inhibition with Vibrio fischeri
desethyldesiso-propyl-2-hydroxyatrazine
Y
(Sanchis et al., 2014)
17
pesticide
fluopyram
UV light (λ ≥ 200 nm) and SSR (λ ≥ 290 nm).
water
luminescence inhibition with Vibrio fischeri
hydroxylated products
Y
(Dong and Hu, 2016)
18
pesticide
DDT
Fenton-like system with zero-valent iron, EDTA and air (ZVI/EDTA/Air), photodegradation, peroxymonosulfate activation by MnO2
soil and water
influence on algae growth
2,2-bis(4-chlorophenyl)-1-chloroethylene (4,4′-DDMU), DDE, dichlorobenzophenone
Y
(Cao et al., 2013; Hong et al., 1997; Liu et al., 2021; Megharaj et al., 1999)
19
pharmaceuticals
florfenicol
electrochemical advanced oxidation
water
luminescence inhibition with Vibrio fischeri
chlorine-containing hydroxylated products and inorganic chlorines
N
(Periyasamy et al., 2022)
20
pharmaceuticals
lomefloxacin
direct, indirect, and self-sensitized photolysis
water
luminescence inhibition with Vibrio fischeri
product derived by defluorination and piperazing ring oxidation
N
(Zhang et al., 2019)
21
pharmaceuticals
diclofenac
•OH oxidation by pulse radiolysis experiment
water
luminescence inhibition with Vibrio fischeri
chloroderivatives
N
(Yu et al., 2013)
21
pharmaceuticals
carbamazepine
UV-radiation, chlorine dioxide
water
luminescence inhibition with Vibrio fischeri, biomass growth of Pseudokirchneriella subcapitata, daphnia immobilization
Acridine, 9(10H)-acridone
UV irradiation could enhance the biodegradability of carbamazepine residues
(Donner et al., 2013; Kosjek et al., 2009)
3.2 Phenols
Phenolic compounds have gained extensive attention because of their high toxicity and the frequent presence in industrial wastewaters and natural environment (Ding et al., 1995; Ekpe et al., 2022). There have been several literatures focusing on the ecotoxicity of phenols after oxidation treatment, and most of them reported an enhanced toxicity in the early stage of phenol oxidation (Santos et al., 2004; Santos et al., 2006; Xia et al., 2017; Zazo et al., 2007). The reason was the production of intermediates such as hydroquinone, 1,4-benzoquinone and catechol, but the intermediates could further transform into low-toxicity organic acids, such as acetic, oxalic, and maleic acids. The typical evolution process of phenol was shown in Fig. 2. However, several studies found that the ecotoxicity was reduced in neutral or basic medium (Santos et al., 2006; Zazo et al., 2007).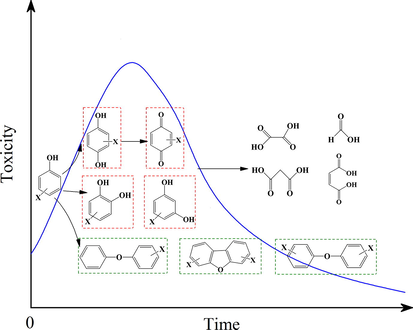
The representative degradation pathway and toxicity evolution of phenol. The chemicals in red box are more toxic than the parent, and those in green are refractory in addition to highly toxic.
Noticeable toxicity increases have been also reported during the oxidation process of cresol, nitrophenol and chlorophenol, and it could be attributed to the production of substituted hydroquinone, 1,4-benzoquinone and catechol (Santos et al., 2006; Svenson and Hynning, 1997). It is noteworthy that toxic polychlorinated dibenzo-p-dioxins and dibenzofurans (PCDD/Fs) formation was observed in the oxidation of chlorophenols, which might cause further toxicity increase in oxidation process, and changing the operating conditions is conducive to mitigate toxic byproducts (Altarawneh et al., 2007; Fukushima and Tatsumi, 2001; Vallejo et al., 2013; Vallejo et al., 2015; Weber, 2007).
There was also an increase in ecotoxicity during the initial stages of bisphenol A (BPA) oxidation. A significant correlation existed between the toxicity patterns and its major transformation products, such as benzoquinone, hydroquinone, styrene, p-isopropyl phenol, etc. (Han et al., 2015; Olmez-Hanci et al., 2013; Zhou et al., 2020) Richard et al. (2014) found that the UV/H2O2 treatment of BPA revealed cytotoxic by-products but did not identify the specific compounds. In addition, the acute toxicity assay suggested that the degradation products of bisphenol S (BPS) and bisphenol F (BPF) were more toxic than their parent compounds, and the highly toxic products were identified as self-coupling dimers or polymers, such as 4,4′-dihydroxybibenzyl and 4,4′-dihydroxybiphenyl. They could undergo further oxidation into hydroxylation products during the reaction (Lu et al., 2019; Yang et al., 2020). Conversely, dimers generated during BPA oxidation seems not to increase the acute toxicity during photo-Fenton-like treatment, which might be due to the limited amount of products (Molkenthin et al., 2013).
3.3 Azo dyes
Some studies have reported the highly toxic oxidation intermediates of azo dyes (de Luna et al., 2014; Glaze, 1986; Neamtu et al., 2003; Wang et al., 2003). For example, Wang et al. (2003) confirmed the formation of high toxic by-products of C.I. Remazol Black 5 by the luminescence test (V. fischeri) and the neutral red cytotoxicity assay (rat hepatoma cells). Neamtu et al. (2003) observed the temporary increase in luminescent bacteria inhibition during the UV/H2O2 treatment of C.I. Reactive Yellow 84. Vat Green 3, Reactive Black 5, Acid Orange 7 (AO7), and Food Red 17 also produced more hazardous degradation products than the parent dyes, according to acute test with Daphnia similis and chronic tests with Ceriodaphnia dubia and Pseudokirchneriella subcapitata (de Luna et al., 2014). It has been pointed out that the reagent dosages and treatment period played a crucial role in the detoxification of dyes, meaning that the toxic intermediates could be degraded readily (Castro et al., 2014; de Luna et al., 2014; Khadhraoui et al., 2009; Zhao et al., 2010). However, seldom studies determined the structures of the highly toxic intermediate (Glaze, 1986). The toxicity of AO7 during electro-Fenton treatment increased rapidly at the early stage, in line with the emergence of intermediate hazardous aromatic compounds including 1,2-naphthaquinone (NAPQ) and 1,4-benzoquinone (BZQ), which were formed through the breakage of the azo linkage (Alderete et al., 2021; Le et al., 2016; Velegraki et al., 2006; Zhao et al., 2010). Fig. 3 is the schematic diagram on the toxicity transformation mechanism of AO7.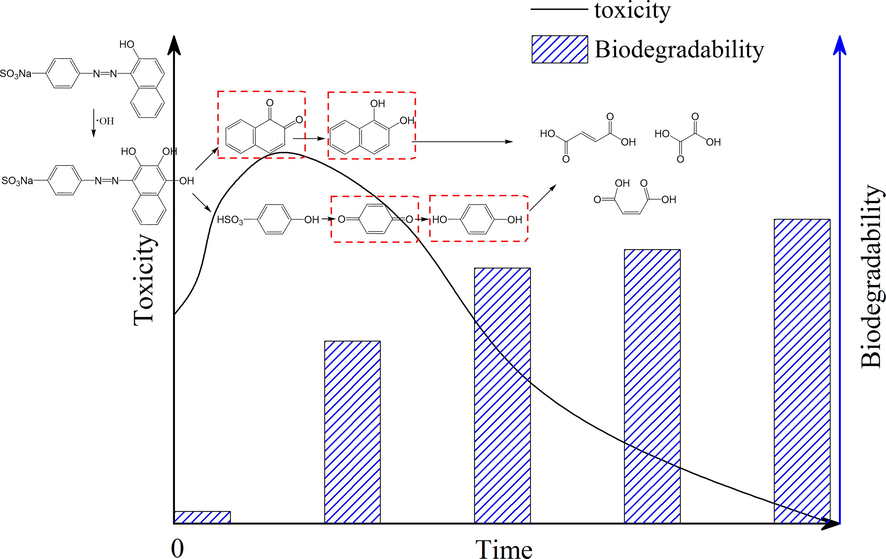
The toxicity and biodegradability evolution related to the degradation intermediates of AO7. The chemicals in red box are more toxic than the parent.
In addition, as the commercial azo dyes usually contain some impurities such as synthetic precursors, dispersing agents, and by-products, the toxicity of the impurities during oxidation should been paid attention (Zhang et al., 2004). As the important raw material of dyestuff, nitrobenzene and diphenylamine experienced a trend of toxicity that was initially increasing and then reducing during photocatalytic treatment. The toxic may be related with the oxygenated intermediates (Bolduc and Anderson, 1997; Li et al., 2009). Wet air oxidation of nitrotoluenesulfonic acid (NTSA) also produced some more toxic intermediates than NTSA itself such as cresol, nitrobenzene, phenol, quinolinol, 2-chlorophenol, etc (Hao and Phull, 1993).
3.4 Pesticide
3.4.1 Organophosphorus
Pesticides are produced in vast quantities and employed in a variety of agricultural processes. They were distributed in wastewaters, soil and groundwater from pesticide manufacturing plants, and chemical oxidation have been proven useful for their removal (Bhandari et al., 2020; Paul et al., 2006). The most widely utilized pesticides is organophosphorus (OP), which accounts for 40% of global use and roughly 70% of consumption in the United States (Vasseghian et al., 2022). OPs have an irreversible inhibition of acetylcholinesterase (AChE) enzyme, which would cause acetylcholine to accumulate and cholinergic receptors to become overactive in various regions of the body (Chowdhary et al., 2014; Pope, 1999). Toxicity increase of OPs during chemical oxidation have been noticed in some studies. For example, as for malathion (S-1,2-bis(ethoxycarbonyl)ethyl-O,O-dimethyl phosphorodithioate), which has the highest rate of the total OP pesticide consumption (Vasseghian et al., 2020; Vasseghian et al., 2022), the treated effluent’s luminescence and AChE toxicity all dramatically increased during the Fenton, photo-Fenton, and sono-photo Fenton processes. Malaoxon, diethyl maleate, diethyl malate, ethyl 2-hydroxysuccinate, and d-malate were recognized as the intermediates by oxidative desulfuration, the replacement of oxygen for sulfur on the P = S bond through oxidation, and hydrolysis processes mechanisms (Vasseghian et al., 2022). We calculated the acute toxicities of the intermediates with T.E.S.T. (version 5.1), and the results showed the malathion oxon had the higher acute oral toxicity to rat than the parent compound (see Table S2 in Supporting Information), which was consistent with the results from previous study with the LC50 to larval Rana boylii as the indicator (Sparling and Fellers, 2007). Ohashi et al. (1994) proposed the ozonation products of 17 kinds of organophosphorous pesticides, but the toxicity change was not clear. So the toxicities of the intermediates were also calculated with T.E.S.T. software in this work and shown in Table S2. The results showed that the oxon products usually had lower acute toxicity to D. magna but higher oral cute toxicity to rat. It may be because that the oxon products have lower solubilities, but higher lipophilicity. Regarding chlorpyrifos, Zhang et al. (2011) found that it became more harmful as evidenced by the luminous bacterium's increasing light emission after ultrasonic irradiation for 30 min and 60 min, and this may contribute to the formation of chlorpyrifos oxon, which has been proven to be 100 times more toxic than its parent compound (Sparling and Fellers, 2007). Sheikhi et al. (2021) revealed that the main products of chlorpyrifos had lower acute and chronic toxicity than their parent, but some of the products were classified as very toxic according to the Globally Harmonized System of Classification and Labeling of Chemicals (GHS). Generally, despite the toxicity results differ with the oxidization technology, toxicity assessment method, reaction condition, and so on, much attention should be paid to the degradation byproducts’ toxicity of chlorpyrifos (Liu et al., 2019; Sheikhi et al., 2021; Zhang et al., 2011).
3.4.2 Organonitrogens
Sanchis et al. (2014) investigated the Fenton oxidation intermediates, ecotoxicity and biodegradability of three organonitrogenous herbicides, i.e. alachlor, diuron and atrazine. The ecotoxicity was somewhat negatively impacted by the oxidation process, but fortunately, significant improvement of biodegradability was observed. The 2,6-diethylaniline, 3,4-dichloroaniline and desethyldesiso-propyl-2-hydroxyatrazine (DEIHA) partly contribute to the increase of ecotoxicity of alachlor, diuron and atrazine, respectively. Similar results were also revealed in other oxidation processes (Farré et al., 2005; Malato et al., 2003a; Osano et al., 2002). In addition to the above herbicides, the increase of ecotoxicity during oxidation processes were also observed as for isoproturon, chlorfenvinfos, methomyl, metoxuron, fluopyram, etc. (Dong and Hu, 2016; Farré et al., 2005; Lapertot et al., 2006; Malato et al., 2003b; Parra et al., 2002). The toxic formylated and demethylated compounds forming by the –N(R)CHO functions (where R might be H or CH3) addition or –N(CH3)2 removal could account for the enhanced toxicity. The N-formylated compounds were usually formed from phenylurea herbicides in the presence of •OH, but they seemed not to be generated when reacting with 3CDOM* (the excited triplet states of chromophoric dissolved organic matter which cloud be produced during direct photolysis) (Fabbri et al., 2015; Tixier et al., 2001). The hydroxylated products might also contribute to the toxicity increase (Dong and Hu, 2016; Hu et al., 2019; Li et al., 2020).
3.4.3 Organochlorine
Organochlorine pesticides (OCPs) are a major concern due to their high toxicity, stability, and resistance to degradation in the environment. Most organochlorine pesticides have been forbidden in most nations during the past 40 years, with the exception of lindane and dichlorodiphenyltrichloroethane (DDT), which still have some applications (Costa, 2015). But OCPs such as DDT, endosulfans, pentachlorophenol (PCP), toxaphenes, lindane still have widespread environmental residue (Li et al., 2016; Olisah et al., 2020; Wong et al., 2010; Yadav et al., 2016).
A trace quantity of the more hazardous 4,4′-DDMU could be produced when DDT was subjected to the Fenton or activated persulfate treatment (Cao et al., 2013; Hong et al., 1997; Liu et al., 2021). During oxidation processes of PCP, high toxicity products such as octachlorodibenzo-p-dioxin/furan (OCDD/OCDF), tetrachloro-1,4-benzoquinone (TCBQ) and trichlorohydroxy-1,4-benzoquinone (OH-TrCBQ) were observed and it is noteworthy that a continuous flow chemiluminescence method to monitor TCBQ and OH-TrCBQ generation during photocatalytic oxidation process of PCP was developed by Ma et al, which provide some inspiration for dynamic monitoring of highly toxic intermediate oxidation products (Ma et al., 2018; Chen et al. 2020). Other organochlorines usually experienced a toxicity decrease process (Begum et al., 2017; Ikehata and El-Din, 2006; Ribeiro et al., 2015).
3.5 Pharmaceuticals
3.5.1 Fluoroquinolones (FQs)
FQs, the powerful and broad-spectrum antimicrobial agents, have been employed more often in both human and veterinary medicine since the late 1980 s (Feng et al., 2018; Redgrave et al., 2014). Although in most cases the oxidation was a detoxification process as for FQs, some studies have revealed its toxicity increase during oxidation processes (Bobu et al., 2013; Carneiro et al., 2020; Feng et al., 2018; Yang et al., 2016). For example, Qiu et al. (2019) found that the acute toxicity indicated by death and incubation number of zebrafish embryos of pefloxacin reaction solutions was firstly shown to increase and then be followed by a drop with increasing irradiation time of photolysis, but they did not identify the effective intermediates. The basic structure of FQ is shown in Fig. 4, and according to previous studies, FQs were often oxidized by cleavage or oxidation of piperazine ring, and decarboxylation of quinolone substituents. When FQs contain more than one fluorine, defluorination was also a degradation route (Li et al., 2022a; Zhang et al., 2019; Zhu et al., 2016). The toxicity of ofloxacin during the electrochemical oxidation on a BDD electrode process was evaluated with the EC50 values by the algal (desmodesmus subspicatus) growth inhibition tests. The toxicity significantly increased, and it is revealed that the oxidation of the piperizinyl ring is not sufficient to eliminate the toxicological effects of FQs and would even increase it (Zhu et al., 2016). Similar conclusion has also been driven by Zhang et al. and Ge et al. (Ge et al., 2018; Zhang et al., 2019). Zhang et al. (2019) found that the degradation products of lomefloxacin exhibited greater toxicity, and the possible intermediate was formed by defluorination and breakage of piperizinyl ring. Ge et al. (2018) examined the photomodified toxicity of gatifloxacin using an E. coli activity assay. The light-irradiated solutions' antibacterial activity did not considerably decline during the first photodegradation phase (0 − t1/2) as the photoreactions had not alter the core aromatic backbone of quinolone substitutes. Li et al. (2022a) investigated the intermediates of ofloxacin treated with heat-activated persulfate and calculated their oral rat LD50 values, mutagenicity and developmental toxicity with T.E.S.T. and ECOSAR respectively. The products formed by β-elimination on the piperazine substituent appeared to have a higher mutagenicity and developmental toxicity, while products formed by the cleavage of quinolone seemed to be more mutagenic. A greater acute toxicity to rats was seen when the quinolone and piperazine rings were both removed. In conclusion, the destruction of fluorinated aromatic ring might be the key of detoxification, which required futher investigation.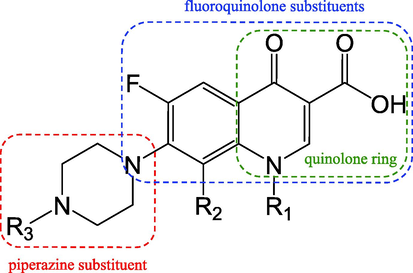
The characteristic structure of fluoroquinolone.
3.5.2 Sulfonamides (SAs)
The toxicity increase of SAs during oxidation or photolysis was also found in many previous studies (Qiu et al., 2019; Trovó et al., 2009; Xu et al., 2014; Zhang et al., 2016). Li et al. (2021) reviewed the abiotic transformation and ecotoxicity alteration of SAs during water treatment and environmental processes. The typical structural transformation of SAs include oxidation of aniline moiety, SO2 extrusion, cleavage of sulfonamide group, transformation on the heterocyclic ring, and so on (Zhang et al., 2020). The typical transformation processes are summarized in Fig. 5, and the highly toxic intermediates are identified according to the previous studies. The formed dimers connecting through –NH–NH– have been previously demonstrated to be more detrimental to aquatic species, and exhibit carcinogenicity (Zhuang et al., 2019). Additionally, it has been established that the nitroso-, nitro-, and hydroxylamine-SAs were more hazardous than the parent SAs due to their stronger propensities to bind to dihydropteroate synthase (Majewsky et al., 2014; Shad et al., 2018), and the hydroxylamine-SAs usually have a higher cytotoxicity. Both acute and chronic toxicity of SO2 extrusion products to typical aquatic species were also revealed to be much higher than the parent compound (Kråkström et al., 2020; Yang et al., 2018). One important product, 2-aminopyrimidine, which is formed through the cleavage of sulfonamide bond (S-N) during several advanced oxidation processes (AOPs) (Fabiańska et al., 2014; Neafsey et al., 2010; Wang et al., 2010; Zou et al., 2014), showed substantially higher acute and chronic toxicity for aquatic species than sulfadiazine based on ECOSAR computation [1 4 3]. In addition, acute toxicity and genotoxic potentials were increased during chlorination processes with the emergency of DBPs of SAs (Fu et al., 2018; Zhang et al., 2020).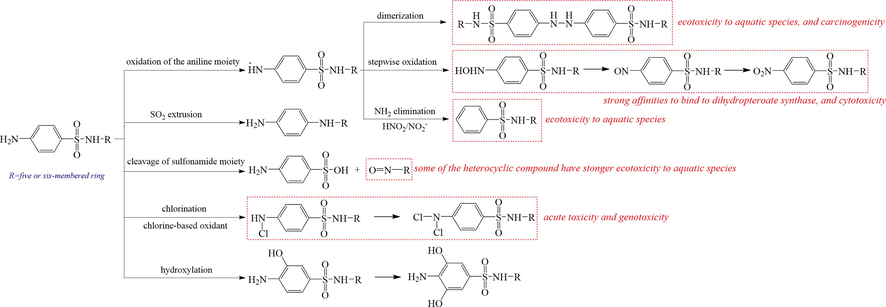
Typical transformation pathways and product toxicity of SAs. The chemicals in red box are more toxic than the parent.
3.5.3 Others
Apart from fluoroquinolones and sulfonamides, high toxic intermediates have also been found in other pharmaceuticals. Typically, the halogen-containing chemicals have potential high toxicity via transformation. For example, propranolol (PRO), one of the most commonly used beta blockers, enhanced the luminescence inhibition during UV-LED/chlorine treatment, which might be correlated with the chlorine-containing by-products (Xiong et al., 2020). Diclofenac (DCF), a synthetic non-steroidal anti-inflammatory medication, is one of the most widely prescribed painkillers. Hydroxylation, ring-opening, decarboxylation and cyclation reaction might occur during the processes, and the toxicity of DCF solution experienced an increase stage during some processes (Calza et al., 2006; Lu et al., 2017; Wang et al., 2015; Yu et al., 2013; Zhao et al., 2009). The toxicity might be related with the production of the chloroderivatives (Lu et al., 2017). Most of the chlorine-containing products, such as hydroxylated DCF, chlorinated DCF, 2,6-dichloro-N-(2-methylphenyl)aniline, might be responsible for the toxicity (Gao et al., 2022; Li et al., 2017; Qiu et al., 2020). Similarly, the increase of toxicity occurred during the florfenicol (FLO) degradation due to chlorinated byproducts and inorganic ones (ClO2-, ClO3- and ClO4-) formed, which are more toxic than the parent FLO (Periyasamy et al., 2022). Similar phenomenon was also observed on nitrogenous heterocyclic compounds. Carbamazepine produced acridine and acridone during UV-radiation and chlorine dioxide oxidation, which have been found to have higher ecotoxicity (Donner et al., 2013; Kosjek et al., 2009).
4 The enhanced toxicity induced by environmental elements
Environmental elements such as dissolved organic matter (DOM) and halides have been reported to have the potential to enhance the toxicity after oxidation. The enhanced toxicity on V. fischeri and D. magna have been reported for DOM-spiked ofloxacin solutions after photo-Fenton processes. However, the increased toxic effects might be attributed to the byproducts generated from DOM but not from the transformation intermediates of ofloxacin (Michael et al., 2010; Michael et al., 2013). Li et al. (2022b) found that a ClO2 pre-oxidation process might be useful in reducing the genotoxicity and cytotoxicity of UV/chlorine-treated DOM-containing water. On the contrary, sometimes DOM helps reduce the toxicity. For example, the cross-coupling products combining SAs and natural organic matter (NOM) constituent via the amide nitrogen might lessen ecotoxicity due to their lower mobility (Li et al., 2020; Wang et al., 2020). Franklin pointed out that the DOM source affects DBP formation, and the organic nitrogen and protein-like content drove differences (Franklin et al., 2021), and Xie et al. discovered that organic matters with a molecular weight (MW) of between 1 k and 100 k Da are more likely to generate organic halogens than those with a MW of lower than 1 k Da and higher than 100 k Da. (Xie et al., 2020).
Halides are also noteworthy and usually have a combined effect with DOM. The growth inhibition ratios of SMX to C. vulgaris increased with increasing halogen ion (Cl-, Br-) content under the UV/PS and UV/NaClO oxidation system (Zhang et al., 2020). Similar results were found in the Co(II)/peroxymonosulfate, ozone system, electrochemical oxidation, and so on (Fang et al., 2016; He et al., 2022; Xiang et al., 2019; Xu et al., 2022). It has been reported that the cytotoxic index value of brominated byproducts was much higher than chlorinated ones, and iodo-products which have mammalian cell toxicity should also been addressed (Richardson et al., 2008). The halogenated organic products, which were formed from reactions of pollutants with halogen radicals or hypohalous acid, can interpret the phenomenon of toxicity enhancement. The description on increased toxicity induced by interaction between DOM and halogen elements is available in an excellent review compiled by Yang et al. and Ike et al. (Ike et al., 2019; Yang et al., 2022). An interaction between NO2– and DOM was also found to be have the potential to enhance the toxicity (Yang et al., 2021; Yang et al., 2022).
5 Strategies to reduce the residual toxicity
As in most cases the high toxicity resulted from the inadequate degradation of pollutants, it is necessary to take some measures to strengthen the reaction, for example, to increase the oxidant concentration or prolong reaction time. Sanchis et al. (2014) has proved that the detected toxicity products decreased significantly as the amount of oxidant increased. According to Liu et al. (2017), prolonging the photo-Fenton oxidation time could lessen the toxicity of methylene blue solution. Furthermore, coupling of multiple technologies is an effective strategy. For example, the coupling of ozone and electrochemical methods can reduce the toxicity of the phenol degradation process (Gao et al., 2022), and combining solar photo-Fenton and biological treatment can effectively remove the pharmaceuticals in wastewater and alleviate the residual toxicity (Sirtori et al., 2009). Likewise, biodegradation after Fenton-like oxidation could effectively reduce the toxicity of petroleum-contaminated soil (Lu et al., 2010), and the products of carbamazepine were also proved to be more susceptible to biological treatment than the parent (Kosjek et al., 2009). Membrane technology can be also used following AOPs to further remove toxicity. Żyłła et al. (2019) found that the combined use of UV/H2O2 and nanofiltration process facilitated the chemical conversion of the tested chemicals as well as the elimination of their poisonous oxidation byproducts from the solution, and Zhi et al. (2020) draw the similar conclusion.
Optimization of oxidation catalytic materials to regulate the reactive oxygen species (ROS) generation is another way to change the pollutant conversion pathways and reduce product toxicity. For example. Chen et al. (Chen et al., 2020; 2022) synthesized the polydopamine modified TiO2 nanoparticles (P25/PDA) and TiO2/polydopamine/ bismuth oxybromide (P25/PDA/BiOBr) materials respectively to improve the PCP photodegradation process and depress generation of TCBQ and OH-TrCBQ through regulating the dominant ROS to be H2O2, •O2–, electrons, et al., but not •OH.
In addition, controlling environmental conditions was also effective in some cases. Chen et al. and Sun et al. (Chen et al., 2021; Sun et al., 2019) proved that pH might affect the generation of nitro(so) and chlorinated products as well as toxicity alterations when phenol was treated with UV light and monochloramine. The pH was also an influencing factor on the formation of DBPs and toxicity variation during the UV/chlorine AOPs. Moreover, the contribution of various ROS and the degradation pathway during SMX oxidation varied intensively under different pH. The residual toxicity was better eliminated along with the increase of pH (pH-dependent transformation products and residual toxicity evaluation of sulfamethoxazole degradation through non-radical oxygen species involved process). The presence of humic substances would change oxidative conversion pathways of PCP and avoid OCDD formation in some cases (Fukushima et al., 2003; Fukushima et al., 2010; Fukushima and Tatsumi, 2001).
6 Conclusion and perspectives
Incomplete degradation of organic pollutants in wastewater or soil would lead to unintended formation of potentially toxic intermediates, and sometimes the complete conversion of the parent compound or even a decrease in the amount of organic carbon in the reaction solution is insufficient to detoxify it. Therefore, only monitoring the pollutant attenuation is not enough to reflect the treatment effect, and analysis on intermediates or toxicity is necessary. Acute toxicity test is commonly used in the oxidation process to assess the negative environmental impacts of the intermediates, and the luminescence inhibition is the predominant indicator. The prediction based on QSAR following product identification is also frequently applied. As each method has pros and cons of its own, combination of both is recommended. Although test species, reaction conditions, oxidation methods, and evaluation endpoints all had an impact on toxicity, particular attention should be paid to all substances that have the potential to produce more hazardous intermediates. Higher toxic intermediates have been found as for PAH, phenols, azo dyes, organophosphorus, organonitrogens, organochlorine pesticide, pharmaceutical chemicals, and so on. The hydroquinone-, benzoquinone-, catechol-like and halogenated products usually have higher ecotoxicity while the dimers and polymers are refractory and prone to accumulate in the soil. In addition, DOM and halogen ion have the potential to enhance the oxidative toxicity. The strategies to reduce the residual toxicity were also summarized in this work. For the deeper understanding of the oxidation product toxicity of pollutants, more efforts can be made from the following aspects: (1) to investigate the occurrence and distribution of persistent oxidation products; (2) to study the toxicity effects of the products at multiple trophic levels; (3) to strengthen the residual toxicity studies in soil; (4) to consider the toxicity change as a standardized measure of remediation effectiveness; (5) to further clarify the relationship between the transformation pathway of the pollutants and key action species.
Acknowledgements
This work was supported by National Key Research and Development Project of China (Grant number 2018YFC1801101), National Natural Science Foundation of China (Grant number 21707041), and Central Public-interest Scientific Institution Basal Research Fund (GYZX220202).
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Evaluation of toxicity and mutagenicity of a synthetic effluent containing azo dye after Advanced Oxidation Process treatment. Chemosphere. 2021;263:128291
- [Google Scholar]
- Quantum chemical investigation of formation of polychlorodibenzo-p-dioxins and dibenzofurans from oxidation and pyrolysis of 2-chlorophenol. J. Phys. Chem. A. 2007;111:2563-2573.
- [Google Scholar]
- Degradation of endosulfan and lindane using Fenton’s reagent. Appl. Water Sci.. 2017;7:207-215.
- [Google Scholar]
- Concentration and distribution of pesticide residues in soil: Non-dietary human health risk assessment. Chemosphere. 2020;253:126594
- [Google Scholar]
- Comparison of different advanced oxidation processes for the degradation of two fluoroquinolone antibiotics in aqueous solutions. J. Environ. Sci. Health. A. 2013;48:251-262.
- [Google Scholar]
- Enhancement of the biodegradability of model wastewater containing recalcitrant or inhibitory chemical compounds by photocatalytic pre-oxidation. Biodegradation. 1997;8:237-249.
- [Google Scholar]
- Photocatalytic degradation study of diclofenac over aqueous TiO2 suspensions. Appl. Catal. B- Environ.. 2006;67:197-205.
- [Google Scholar]
- Remediation of DDTs contaminated soil in a novel Fenton-like system with zero-valent iron. Chemosphere. 2013;90:2303-2308.
- [Google Scholar]
- Comparing the electrochemical degradation of the fluoroquinolone antibiotics norfloxacin and ciprofloxacin using distinct electrolytes and a BDD anode: Evolution of main oxidation byproducts and toxicity. J. Environ. Chem. Eng.. 2020;8:104433
- [Google Scholar]
- Combination of advanced oxidation processes and biological treatment for the removal of benzidine-derived dyes. Environ. Prog. Sustain.. 2014;33:873-885.
- [Google Scholar]
- Are UV photolysis and UV/H2O2 process efficient to treat estrogens in waters? Chemical and biological assessment at pilot scale. Water Res.. 2016;100:357-366.
- [Google Scholar]
- Formation of nitro(so) and chlorinated products and toxicity alteration during the UV/monochloramine treatment of phenol. Water Res.. 2021;194:116914
- [Google Scholar]
- Dynamic monitoring and regulation of pentachlorophenol photodegradation process by chemiluminescence and TiO2/PDA. J. Hazard. Mater.. 2020;399:123073
- [Google Scholar]
- Dynamic control of pentachlorophenol photodegradation process using P25/PDA/BiOBr through regulation of photo-induced active substances and chemiluminescence. Chemosphere. 2022;307:135914
- [Google Scholar]
- Mutagenicity of quinones: Pathways of metabolic activation and detoxification. Proc. Natl. Acad. Sci. U S A. 1984;81:1696-1700.
- [Google Scholar]
- Chapter 9 - The neurotoxicity of organochlorine and pyrethroid pesticides. In: Lotti M., Bleecker M.L., eds. Handbook of Clinical Neurology. Elsevier; 2015. p. :135-148.
- [Google Scholar]
- Aquatic toxicity of dyes before and after photo-Fenton treatment. J. Hazard. Mater.. 2014;276:332-338.
- [Google Scholar]
- Toxicological study of the degradation products of antineoplastic agent etoposide in commercial formulation treated by heterogeneous photocatalysis using SrSnO3. Environ. Sci. Pollut. R.. 2019;26:4224-4233.
- [Google Scholar]
- Visible-light-driven photocatalytic degradation of Metalaxyl by reduced graphene oxide/Fe3O4/ZnO ternary nanohybrid: Influential factors, mechanism and toxicity bioassay. J. Photoch. Photobio. A: Chem. 2019;375:280-292.
- [Google Scholar]
- Catalytic supercritical water oxidation: Phenol conversion and product selectivity. Environ. Sci. Technol.. 1995;29:2748-2753.
- [Google Scholar]
- Photodegradation of the novel fungicide fluopyram in aqueous solution: Kinetics, transformation products, and toxicity evolvement. Environ. Sci. Pollut. R.. 2016;23:19096-19106.
- [Google Scholar]
- Ecotoxicity of carbamazepine and its UV photolysis transformation products. Sci. Total Environ.. 2013;443:870-876.
- [Google Scholar]
- Long-term degradation of toluene and phenol in soil: Identification of transformation products and pathways via HRMS-based suspect and non-target screening. J. Hazard. Mater.. 2022;430:128429
- [Google Scholar]
- Photochemical transformation of phenylurea herbicides in surface waters: A model assessment of persistence, and implications for the possible generation of hazardous intermediates. Chemosphere. 2015;119:601-607.
- [Google Scholar]
- Electrochemical degradation of sulfonamides at BDD electrode: Kinetics, reaction pathway and eco-toxicity evaluation. J. Hazard. Mater.. 2014;280:579-587.
- [Google Scholar]
- Advanced oxidation kinetics and mechanism of preservative propylparaben degradation in aqueous suspension of TiO2 and risk assessment of its degradation products. Environ. Sci. Technol.. 2013;47:2704-2712.
- [Google Scholar]
- Enhanced AOX accumulation and aquatic toxicity during 2,4,6-trichlorophenol degradation in a Co(II)/peroxymonosulfate/Cl− system. Chemosphere. 2016;144:2415-2420.
- [Google Scholar]
- Degradation of some biorecalcitrant pesticides by homogeneous and heterogeneous photocatalytic ozonation. Chemosphere. 2005;58:1127-1133.
- [Google Scholar]
- Metal-mediated oxidation of fluoroquinolone antibiotics in water: A review on kinetics, transformation products, and toxicity assessment. J. Hazard. Mater.. 2018;344:1136-1154.
- [Google Scholar]
- Oxidation kinetics of polycyclic aromatic hydrocarbons by permanganate. Chemosphere. 2010;79:628-636.
- [Google Scholar]
- Terrestrial dissolved organic matter source affects disinfection by-product formation during water treatment and subsequent toxicity. Environ. Pollut.. 2021;283:117232
- [Google Scholar]
- Bioassays with Vibrio fischeri for the assessment of delayed toxicity. Chemosphere. 2000;40:821-828.
- [Google Scholar]
- New insights into the chlorination of sulfonamide: Smiles-type rearrangement, desulfation, and product toxicity. Chem. Eng. J.. 2018;331:785-793.
- [Google Scholar]
- Effects of humic substances on the pattern of oxidation products of pentachlorophenol induced by a biomimetic catalytic system using tetra(p-sulfophenyl)porphineiron(III) and KHSO5. Environ. Sci. Technol.. 2003;37:386-394.
- [Google Scholar]
- Degradation pathways of pentachlorophenol by photo-Fenton systems in the presence of iron(III), humic acid, and hydrogen peroxide. Environ. Sci. Technol.. 2001;35:1771-1778.
- [Google Scholar]
- Influence of humic acid type on the oxidation products of pentachlorophenol using hybrid catalysts prepared by introducing iron(III)-5,10,15,20-tetrakis(p-hydroxyphenyl) porphyrin into hydroquinone-derived humic acids. Chemosphere. 2010;78:1155-1159.
- [Google Scholar]
- Comparative investigation of diclofenac degradation by Fe2+/chlorine and Fe2+/PMS processes. Sep. Purif. Technol.. 2022;297:121555
- [Google Scholar]
- Reaction pathways, kinetics and toxicity assessment during the photocatalytic degradation of glyphosate and myclobutanil pesticides: Influence of the aqueous matrix. Chem. Eng. J.. 2020;384:123315
- [Google Scholar]
- Exploring the aquatic photodegradation of two ionisable fluoroquinolone antibiotics – Gatifloxacin and balofloxacin: Degradation kinetics, photobyproducts and risk to the aquatic environment. Sci. Total Environ.. 2018;633:1192-1197.
- [Google Scholar]
- Solar photo-Fenton treatment – Process parameters and process control. Appl. Catal. B-Environ.. 2006;64:121-130.
- [Google Scholar]
- Chemistry of ozone, by-products, and their health effects, in ozonation: Recent advances and research needs. Denver: AWWARF; 1986.
- Degradation of bisphenol A by ferrate(VI) oxidation: Kinetics, products and toxicity assessment. Chem. Eng. J.. 2015;262:34-40.
- [Google Scholar]
- Wet oxidation of nitrotoluenesulfonic acid: Some intermediates, reaction pathways, and byproduct toxicity. Environ. Sci. Technol.. 1993;27:1650-1658.
- [Google Scholar]
- Effect of pH and Cl- concentration on the electrochemical oxidation of pyridine in low-salinity reverse osmosis concentrate: Kinetics, mechanism, and toxicity assessment. Chem. Eng. J.. 2022;449:137669
- [Google Scholar]
- Identification of Photodegradation Products of DDT in Water. Anal. Sci.. 1997;13:75-82.
- [Google Scholar]
- Seed germination, root elongation, root-tip mitosis, and micronucleus induction of five crop plants exposed to chromium in fluvo-aquic soil. Environ. Toxicol. Chem.. 2014;33:671-676.
- [Google Scholar]
- Mechanism and identify photolysis products of fluopyram under TiO2: Experiments, DFT and ab initio Molecular dynamics study. SDRP J. Earth Sci. Environ. Stud.. 2019;4:681-690.
- [Google Scholar]
- Transformation of m-aminophenol by birnessite (δ-MnO2) mediated oxidative processes: Reaction kinetics, pathways and toxicity assessment. Environ. Pollut.. 2020;256:113408
- [Google Scholar]
- Oxidation byproducts from the degradation of dissolved organic matter by advanced oxidation processes – A critical review. Water Res.. 2019;164:114929
- [Google Scholar]
- Aqueous pesticide degradation by hydrogen peroxide/ultraviolet irradiation and Fenton-type advanced oxidation processes: A review. J. Environ. Eng. Sci.. 2006;5:81-135.
- [Google Scholar]
- Water quality – Determination of the inhibitory effect of water samples on the light emission of Vibrio fischeri (Luminescent bacteria test) ISO 2007:11348-111341.
- [Google Scholar]
- Oxidation degradation of tri(dichloropropyl) phosphate by UV/H2O2 system: Degradation pathways and risk assessment of intermediates. J. Environ. Chem. Eng.. 2020;8:104513
- [Google Scholar]
- Advanced oxidation process-mediated removal of pharmaceuticals from water: A review. J. Environ. Manage.. 2018;219:189-207.
- [Google Scholar]
- Discoloration and detoxicification of a Congo red dye solution by means of ozone treatment for a possible water reuse. J. Hazard. Mater.. 2009;161:974-981.
- [Google Scholar]
- Degradation and toxicity assessment of sulfamethoxazole and chlortetracycline using electron beam, ozone and UV. J. Hazard. Mater.. 2012;227–228:237-242.
- [Google Scholar]
- A strategy for assessing the effects of xenobiotics on fish reproduction. Sci. Total Environ.. 1999;225:3-11.
- [Google Scholar]
- Fate of Carbamazepine during Water Treatment. Environ. Sci. Technol.. 2009;43:6256-6261.
- [Google Scholar]
- Catalytic ozonation of the antibiotic sulfadiazine: Reaction kinetics and transformation mechanisms. Chemosphere. 2020;247:125853
- [Google Scholar]
- Enhancing biodegradability of priority substances (pesticides) by solar photo-Fenton. Water Res.. 2006;40:1086-1094.
- [Google Scholar]
- Toxicity removal assessments related to degradation pathways of azo dyes: Toward an optimization of electro-Fenton treatment. Chemosphere. 2016;161:308-318.
- [Google Scholar]
- Heat-activated persulfate oxidative degradation of ofloxacin: Kinetics, mechanisms, and toxicity assessment. Chem. Eng. J.. 2022;433:133801
- [Google Scholar]
- Photodegradation kinetics, transformation, and toxicity prediction of ketoprofen, carprofen, and diclofenac acid in aqueous solutions. Environ. Toxicol. Chem.. 2017;36:3232-3239.
- [Google Scholar]
- ClO2 pre-oxidation changes dissolved organic matter at the molecular level and reduces chloro-organic byproducts and toxicity of water treated by the UV/chlorine process. Water Res.. 2022;216:118341
- [Google Scholar]
- Degradation of diphenylamine by persulfate: Performance optimization, kinetics and mechanism. J. Hazard. Mater.. 2009;164:26-31.
- [Google Scholar]
- Degradation of fluopyram in water under ozone enhanced microbubbles: Kinetics, degradation products, reaction mechanism, and toxicity evaluation. Chemosphere. 2020;258:127216
- [Google Scholar]
- Characteristics of residual organochlorine pesticides in soils under different land-use types on a coastal plain of the Yellow River Delta. Environ. Geochem. Hlth.. 2016;38:535-547.
- [Google Scholar]
- Significant effect of evaporation process on the reaction of sulfamethoxazole with manganese oxide. Environ. Sci. Technol.. 2020;54:4856-4864.
- [Google Scholar]
- Abiotic transformation and ecotoxicity change of sulfonamide antibiotics in environmental and water treatment processes: A critical review. Water Res.. 2021;202:117463
- [Google Scholar]
- Identification of persulfate oxidation products of polycyclic aromatic hydrocarbon during remediation of contaminated soil. J. Hazard. Mater.. 2014;276:26-34.
- [Google Scholar]
- Efficient transformation of DDT with peroxymonosulfate activation by different crystallographic MnO2. Sci. Total Environ.. 2021;759:142864
- [Google Scholar]
- Oxidative degradation of chlorpyrifos using ferrate(VI): Kinetics and reaction mechanism. Ecotoxicol. Environ. Saf.. 2019;170:259-266.
- [Google Scholar]
- Enhanced catalytic degradation of methylene blue by α-Fe2O3/graphene oxide via heterogeneous photo-Fenton reactions. Appl. Catal. B-Environ.. 2017;206:642-652.
- [Google Scholar]
- Insights into degradation pathways and toxicity changes during electro-catalytic degradation of tetracycline hydrochloride. Environ. Pollut.. 2020;258:113702
- [Google Scholar]
- Degradation of diclofenac by UV-activated persulfate process: Kinetic studies, degradation pathways and toxicity assessments. Ecotox. Environ. Safe.. 2017;141:139-147.
- [Google Scholar]
- Remediation of petroleum-contaminated soil after composting by sequential treatment with Fenton-like oxidation and biodegradation. Bioresource Technol.. 2010;101:2106-2113.
- [Google Scholar]
- Sonolytic degradation of bisphenol S: Effect of dissolved oxygen and peroxydisulfate, oxidation products and acute toxicity. Water Res.. 2019;165:114969
- [Google Scholar]
- Transformation of PAHs during ethanol-Fenton treatment of an aged gasworks' soil. Chemosphere. 2006;65:1288-1294.
- [Google Scholar]
- Bioassay based luminescent bacteria: Interferences, improvements, and applications. Sci. Total Environ.. 2014;468–469:1-11.
- [Google Scholar]
- Dynamic tracking of highly toxic intermediates in photocatalytic degradation of pentachlorophenol by continuous flow chemiluminescence. Environ. Sci. Technol.. 2018;52:2870-2877.
- [Google Scholar]
- Whole effluent toxicity assessment at a wastewater treatment plant upgraded with a full-scale post-ozonation using aquatic key species. Chemosphere. 2012;88:1008-1014.
- [Google Scholar]
- Antibacterial activity of sulfamethoxazole transformation products (TPs): General relevance for sulfonamide TPs modified at the para position. Chem. Res. Toxicol.. 2014;27:1821-1828.
- [Google Scholar]
- Applied studies in solar photocatalytic detoxification: An overview. Sol. Energy. 2003;75:329-336.
- [Google Scholar]
- Photocatalytic treatment of diuron by solar photocatalysis: Evaluation of main intermediates and toxicity. Environ. Sci. Technol.. 2003;37:2516-2524.
- [Google Scholar]
- Impacts of structural photomodification on the toxicity of environmental contaminants: anthracene photooxidation products. Ecotoxicol. Environ Saf.. 1999;43:204-212.
- [Google Scholar]
- OPERA models for predicting physicochemical properties and environmental fate endpoints. J. Cheminform.. 2018;10:10.
- [Google Scholar]
- Treatment of olive mill effluents: Part I. Organic matter degradation by chemical and biological processes-an overview. Environ. Int.. 2005;31:289-295.
- [Google Scholar]
- Toxicity of a PAh photooxidation product to the bacteriaPhotobacterium phosphoreum and the duckweedLemna gibba: Effects of phenanthrene and its primary photoproduct, phenanthrenequinone. Environ. Toxicol. Chem.. 1997;16:892-899.
- [Google Scholar]
- Effects of DDT and its metabolites on soil algae and enzymatic activity. Biol. Fert. Soils. 1999;29:130-134.
- [Google Scholar]
- Solar Fenton and solar TiO2 catalytic treatment of ofloxacin in secondary treated effluents: Evaluation of operational and kinetic parameters. Water Res.. 2010;44:5450-5462.
- [Google Scholar]
- Light-induced catalytic transformation of ofloxacin by solar Fenton in various water matrices at a pilot plant: Mineralization and characterization of major intermediate products. Sci. Total Environ.. 2013;461–462:39-48.
- [Google Scholar]
- Ministry of Ecology and Environment of The People's Republic of China, 1995. Water quality—Determination of the acute toxicity—Luminescent bacterial test. GB/T 15441-1995.
- Photo-Fenton-like treatment of BPA: Effect of UV light source and water matrix on toxicity and transformation products. Water Res.. 2013;47:5052-5064.
- [Google Scholar]
- Polycyclic aromatic hydrocarbons (PAHs) and their oxygen-containing derivatives (OPAHs) in soils from the Angren industrial area. Uzbekistan. Environ. Pollut.. 2010;158:2888-2899.
- [Google Scholar]
- Degradation of sulfonamides in aqueous solution by membrane anodic fenton treatment. J. Agric. Food Chem.. 2010;58:1068-1076.
- [Google Scholar]
- Oxidation of commercial reactive azo dye aqueous solutions by the photo-Fenton and Fenton-like processes. J. Photochem. Photobiol. A: Chem.. 2003;161:87-93.
- [Google Scholar]
- O Mahony, M.M., Dobson, A.D.W., Barnes, J.D., Singleton, I., 2006. The use of ozone in the remediation of polycyclic aromatic hydrocarbon contaminated soil. Chemosphere 63, 307-314.
- Ozonation products of organophosphorous pesticides in water. Eisei Kagaku. 1994;40:185-192.
- [Google Scholar]
- Advanced oxidation of preservative agents in H2O2/UVC system – Kinetics study, transformation products and toxicity assessment. J. Hazard. Mater.. 2017;333:348-357.
- [Google Scholar]
- Occurrence of organochlorine pesticide residues in biological and environmental matrices in Africa: A two-decade review. Heliyon. 2020;6:e3518.
- [Google Scholar]
- Bisphenol A treatment by the hot persulfate process: Oxidation products and acute toxicity. J. Hazard. Mater.. 2013;263:283-290.
- [Google Scholar]
- Investigation of the toxicity of common oxidants used in advanced oxidation processes and their quenching agents. J. Hazard. Mater.. 2014;278:330-335.
- [Google Scholar]
- Comparative toxic and genotoxic effects of chloroacetanilides, formamidines and their degradation products on Vibrio fischeri and Chironomus riparius. Environ. Pollut.. 2002;119:195-202.
- [Google Scholar]
- New integrated photocatalytic-biological flow system using supported TiO2 and fixed bacteria for the mineralization of isoproturon. Appl. Catal. B-Environ.. 2002;36:131-144.
- [Google Scholar]
- A review on advantages of implementing luminescence inhibition test (Vibrio fischeri) for acute toxicity prediction of chemicals. Environ. Int.. 2006;32:265-268.
- [Google Scholar]
- Bacterial community structure of a pesticide-contaminated site and assessment of changes induced in community structure during bioremediation. FEMS Microbiol. Ecol.. 2006;57:116-127.
- [Google Scholar]
- Insight into BDD electrochemical oxidation of florfenicol in water: Kinetics, reaction mechanism, and toxicity. Chemosphere. 2022;288:132433
- [Google Scholar]
- Organophosphorus pesticides: Do they all have the same mechanism of toxicity? J. Toxicol. Environ. Health B. 1999;2:161-181.
- [Google Scholar]
- Ozonation of diclofenac in the aqueous solution: Mechanism, kinetics and ecotoxicity assessment. Environ. Res.. 2020;188:109713
- [Google Scholar]
- Photolysis of enrofloxacin, pefloxacin and sulfaquinoxaline in aqueous solution by UV/H2O2, UV/Fe(II), and UV/H2O2/Fe(II) and the toxicity of the final reaction solutions on zebrafish embryos. Sci. Total Environ.. 2019;651:1457-1468.
- [Google Scholar]
- Efficient degradation of diclofenac by LaFeO3-Catalyzed peroxymonosulfate oxidation–-kinetics and toxicity assessment. Chemosphere. 2019;218:299-307.
- [Google Scholar]
- Fluoroquinolone resistance: Mechanisms, impact on bacteria, and role in evolutionary success. Trends Microbiol.. 2014;22:438-445.
- [Google Scholar]
- Reactions of Hydroxyl Radicals and Ozone with Acenaphthene and Acenaphthylene. Environ. Sci. Technol.. 2002;36:4302-4311.
- [Google Scholar]
- An overview on the advanced oxidation processes applied for the treatment of water pollutants defined in the recently launched Directive 2013/39/EU. Environ. Int.. 2015;75:33-51.
- [Google Scholar]
- Toxicity of the micropollutants Bisphenol A, Ciprofloxacin, Metoprolol and Sulfamethoxazole in water samples before and after the oxidative treatment. Int. J. Hyg. Envir. Heal.. 2014;217:506-514.
- [Google Scholar]
- Occurrence and mammalian cell toxicity of iodinated disinfection byproducts in drinking water. Environ. Sci. Technol.. 2008;42:8330-8338.
- [Google Scholar]
- Coupling Fenton and biological oxidation for the removal of nitrochlorinated herbicides from water. Water Res.. 2014;49:197-206.
- [Google Scholar]
- Evolution of toxicity upon wet catalytic oxidation of phenol. Environ. Sci. Technol.. 2004;38:133-138.
- [Google Scholar]
- Wet oxidation of phenol, cresols and nitrophenols catalyzed by activated carbon in acid and basic media. Appl. Catal. B-Environ.. 2006;65:269-281.
- [Google Scholar]
- Abiotic oxidative transformation of 6-PPD and 6-PPD quinone from tires and occurrence of their products in snow from urban roads and in municipal wastewater. Water Res.. 2022;212:118122
- [Google Scholar]
- Understanding the ozonated degradation of sulfadimethoxine, exploration of reaction site, and classification of degradation products. Chemosphere. 2018;212:228-236.
- [Google Scholar]
- Chlorpyrifos removal from aqueous solution through sequential use of coagulation and advanced oxidation processes: By-products, degradation pathways, and toxicity assessment. Environ. Technol. Inno.. 2021;23:101564
- [Google Scholar]
- Photolysis, oxidation and subsequent toxicity of a mixture of polycyclic aromatic hydrocarbons in natural waters. J. Photoch. Photobio. A. 2007;187:186-195.
- [Google Scholar]
- Chemical oxidation of bis(2-chloroethyl) ether in the Fenton process: Kinetics, pathways and toxicity assessment. Chemosphere. 2017;180:117-124.
- [Google Scholar]
- Combined AOP/GAC/AOP systems for secondary effluent polishing: Optimization, toxicity and disinfection. Sep. Purif. Technol.. 2021;263:118415
- [Google Scholar]
- Decontamination industrial pharmaceutical wastewater by combining solar photo-Fenton and biological treatment. Water Res.. 2009;43:661-668.
- [Google Scholar]
- Comparative toxicity of chlorpyrifos, diazinon, malathion and their oxon derivatives to larval Rana boylii. Environ. Pollut.. 2007;147:535-539.
- [Google Scholar]
- Stehr, J., M Ller, T., Kamnerdpetch, C., Scheper, T., Svensson, K., 2001. Basic examinations on chemical pre-oxidation by ozone for enhancing bioremediation of phenanthrene contaminated soils. Appl. Microbiol. Biot. 57, 803-809.
- The influence of the UV/chlorine advanced oxidation of natural organic matter for micropollutant degradation on the formation of DBPs and toxicity during post-chlorination. Chem. Eng. J.. 2019;373:870-879.
- [Google Scholar]
- Increased aquatic toxicity following photolytic conversion of an organochlorine pollutant. Chemosphere. 1997;34:1685-1692.
- [Google Scholar]
- A ubiquitous tire rubber-derived chemical induces acute mortality in coho salmon. Science. 2021;371:185-189.
- [Google Scholar]
- Degradation products of a phenylurea herbicide, diuron: Synthesis, ecotoxicity, and biotransformation. Environ. Toxicol. Chem.. 2001;20:1381-1389.
- [Google Scholar]
- Photodegradation of sulfamethoxazole in various aqueous media: Persistence, toxicity and photoproducts assessment. Chemosphere. 2009;77:1292-1298.
- [Google Scholar]
- Degradation of the antibiotic chloramphenicol by photo-Fenton process at lab-scale and solar pilot plant: Kinetic, toxicity and inactivation assessment. Sol. Energy. 2013;97:596-604.
- [Google Scholar]
- Effects of heat-activated persulfate oxidation on soil microorganisms. Water Res.. 2008;42:1013-1022.
- [Google Scholar]
- U.S. EPA, 2016. Clean water act analytical methods: Whole effluent toxicity methods. https://www.epa.gov/cwa-methods/whole-effluent-toxicity-methods.
- Fenton oxidation to remediate PAHs in contaminated soils: A critical review of major limitations and counter-strategies. Sci. Total Environ.. 2016;569–570:179-190.
- [Google Scholar]
- Comparative study of the destruction of polychlorinated dibenzo-p-dioxins and dibenzofurans during Fenton and electrochemical oxidation of landfill leachates. Chemosphere. 2013;90:132-138.
- [Google Scholar]
- Assessment of PCDD/Fs formation in the Fenton oxidation of 2-chlorophenol: Influence of the iron dose applied. Chemosphere. 2015;137:135-141.
- [Google Scholar]
- Pesticide decontamination using UV/ferrous-activated persulfate with the aid neuro-fuzzy modeling: A case study of Malathion. Food Res. Int.. 2020;137:109557
- [Google Scholar]
- Decontamination of toxic Malathion pesticide in aqueous solutions by Fenton-based processes: Degradation pathway, toxicity assessment and health risk assessment. J. Hazard. Mater.. 2022;423:127016
- [Google Scholar]
- Photocatalytic and sonolytic oxidation of acid orange 7 in aqueous solution. Appl. Catal. B-Environ.. 2006;62:159-168.
- [Google Scholar]
- Photodegradation of Sulfadiazine by Goethite−Oxalate Suspension under UV Light Irradiation. Ind. Eng. Chem. Res.. 2010;49:3527-3532.
- [Google Scholar]
- Oxidation of diclofenac by potassium ferrate (VI): Reaction kinetics and toxicity evaluation. Sci. Total Environ.. 2015;506–507:252-258.
- [Google Scholar]
- Thermal Activation of Peracetic Acid in Aquatic Solution: The Mechanism and Application to Degrade Sulfamethoxazole. Environ. Sci. Technol.. 2020;54:14635-14645.
- [Google Scholar]
- Oxidative removal of carbamazepine by peroxymonosulfate (PMS) combined to ionizing radiation: Degradation, mineralization and biological toxicity. Sci. Total Environ.. 2019;658:1367-1374.
- [Google Scholar]
- Reactive species in advanced oxidation processes: Formation, identification and reaction mechanism. Chem. Eng. J.. 2020;401:126158
- [Google Scholar]
- Toxicity changes of wastewater during various advanced oxidation processes treatment: An overview. J. Clean. Prod.. 2021;315:128202
- [Google Scholar]
- Ozonation of an azo dye C.I. Remazol Black 5 and toxicological assessment of its oxidation products. Chemosphere. 2003;52:1225-1232.
- [Google Scholar]
- Relevance of PCDD/PCDF formation for the evaluation of POPs destruction technologies–review on current status and assessment gaps. Chemosphere. 2007;67:S109-S117.
- [Google Scholar]
- Comparative toxicological pathology in mammals and fish: Some examples with endocrine disrupters. Toxicology. 2004;205:27-32.
- [Google Scholar]
- Acute photo-induced toxicity and toxicokinetics of single compounds and mixtures of polycyclic aromatic hydrocarbons in zebrafish. Environ. Toxicol. Chem.. 2014;33:2028-2037.
- [Google Scholar]
- Bioremediation of soil contaminated with polynuclear aromatic hydrocarbons (PAHs); a review. Environ. Pollut.. 1993;81:229-249.
- [Google Scholar]
- Organochlorine pesticides in soils of Mexico and the potential for soil–air exchange. Environ. Pollut.. 2010;158:749-755.
- [Google Scholar]
- Pyrene contaminated soil remediation using microwave/magnetite activated persulfate oxidation. Chemosphere. 2022;286:131787
- [Google Scholar]
- Natural magnetic pyrrhotite as a high-Efficient persulfate activator for micropollutants degradation: Radicals identification and toxicity evaluation. J. Hazard. Mater.. 2017;340:435-444.
- [Google Scholar]
- The influence of bromide on the degradation of sulfonamides in UV/free chlorine treatment: Degradation mechanism, DBPs formation and toxicity assessment. Chem. Eng. J.. 2019;362:692-701.
- [Google Scholar]
- Adsorbable organic halogens formed during treatment of Cl–containing wastewater by sulfate and hydroxyl radical-based advanced oxidation processes. Chem. Eng. J.. 2020;389:124457
- [Google Scholar]
- UV-LED/chlorine degradation of propranolol in water: Degradation pathway and product toxicity. Chemosphere. 2020;248:125957
- [Google Scholar]
- Photodegradation of sulfapyridine under simulated sunlight irradiation: Kinetics, mechanism and toxicity evolvement. Chemosphere. 2014;99:186-191.
- [Google Scholar]
- Ozone oxidation of 2,4,6-TCP in the presence of halide ions: Kinetics, degradation pathways and toxicity evaluation. Chemosphere. 2022;288:132343
- [Google Scholar]
- Metabolic activation of polycyclic and heterocyclic aromatic hydrocarbons and DNA damage: A review. Toxicol. Appl. Pharm.. 2005;206:73-93.
- [Google Scholar]
- Occurrence, profile and spatial distribution of organochlorines pesticides in soil of Nepal: Implication for source apportionment and health risk assessment. Sci. Total Environ.. 2016;573:1598-1606.
- [Google Scholar]
- Sulfadiazine oxidation by permanganate: Kinetics, mechanistic investigation and toxicity evaluation. Chem. Eng. J.. 2018;349:56-65.
- [Google Scholar]
- Transformation of ammonium to nitrophenolic byproducts by sulfate radical oxidation. Water Res.. 2021;202:117432
- [Google Scholar]
- Oxidation of ciprofloxacin and enrofloxacin by ferrate(VI): Products identification, and toxicity evaluation. J. Hazard. Mater.. 2016;320:296-303.
- [Google Scholar]
- Differentiation of pathways of nitrated byproduct formation from ammonium and nitrite during sulfate radical oxidation. Environ. Sci. Technol.. 2022;56:7935-7944.
- [Google Scholar]
- Multiple roles of dissolved organic matter in advanced oxidation processes. Environ. Sci. Technol.. 2022;56:11111-11131.
- [Google Scholar]
- Ferrate oxidation of bisphenol F and removal of oxidation products with ferrate resulted particles. Chem. Eng. J.. 2020;383:123167
- [Google Scholar]
- Enhanced degradation of sulfamethoxazole antibiotic from aqueous solution using Mn-WO3/LED photocatalytic process: Kinetic, mechanism, degradation pathway and toxicity reduction. Chem. Eng. J.. 2020;380:122497
- [Google Scholar]
- Degradation of diclofenac by advanced oxidation and reduction processes: Kinetic studies, degradation pathways and toxicity assessments. Water Res.. 2013;47:1909-1918.
- [Google Scholar]
- Degradation of parathion methyl in bovine milk by high-intensity ultrasound: Degradation kinetics, products and their corresponding toxicity. Food Chem.. 2020;327:127103
- [Google Scholar]
- Evolution of ecotoxicity upon Fenton's oxidation of phenol in water. Environ. Sci. Technol.. 2007;41:7164-7170.
- [Google Scholar]
- The degradation of chlorpyrifos and diazinon in aqueous solution by ultrasonic irradiation: Effect of parameters and degradation pathway. Chemosphere. 2011;82:1109-1115.
- [Google Scholar]
- Combination of enzymatic degradation by chloroperoxidase with activated sludge treatment to remove sulfamethoxazole: Performance, and eco-toxicity assessment. J. Chem. Technol. Biot.. 2016;91:2802-2809.
- [Google Scholar]
- Degradation of sulfamethoxazole by UV/persulfate in different water samples: Influential factors, transformation products and toxicity. Chem. Eng. J.. 2020;379:122354
- [Google Scholar]
- Evaluation of ecotoxicological effects of benzophenone UV filters: Luminescent bacteria toxicity, genotoxicity and hormonal activity. Ecotox. Environ. Safe.. 2017;142:338-347.
- [Google Scholar]
- Influence of chemical speciation on photochemical transformation of three fluoroquinolones (FQs) in water: Kinetics, mechanism, and toxicity of photolysis products. Water Res.. 2019;148:19-29.
- [Google Scholar]
- UV/H2O2 and UV/PDS Treatment of Trimethoprim and Sulfamethoxazole in Synthetic Human Urine: Transformation Products and Toxicity. Environ. Sci. Technol.. 2016;50:2573-2583.
- [Google Scholar]
- Effects of dye additives on the ozonation process and oxidation by-products: A comparative study using hydrolyzed C.I. Reactive Red 120. Dyes Pigments. 2004;60:1-7.
- [Google Scholar]
- Degradation of 2, 4-dichlorophenol in aqueous solution by dielectric barrier discharge: Effects of plasma-working gases, degradation pathways and toxicity assessment. Chemosphere. 2018;204:351-358.
- [Google Scholar]
- Insights into the vertical distribution of the microbiota in steel plant soils with potentially toxic elements and PAHs contamination after 60 years operation: Abundance, structure, co-occurrence network and functionality. Sci. Total Environ.. 2021;786:147338
- [Google Scholar]
- Electro-oxidation of diclofenac at boron doped diamond: Kinetics and mechanism. Electrochim. Acta. 2009;54:4172-4179.
- [Google Scholar]
- Removal of Acid Orange 7 in simulated wastewater using a three-dimensional electrode reactor: Removal mechanisms and dye degradation pathway. Chemosphere. 2010;78:46-51.
- [Google Scholar]
- Development of ozonation and reactive electrochemical membrane coupled process: Enhanced tetracycline mineralization and toxicity reduction. Chem. Eng. J.. 2020;383:123149
- [Google Scholar]
- Enhanced removal of bisphenol A by cyclodextrin in photocatalytic systems: Degradation intermediates and toxicity evaluation. Chinese Chem. Lett.. 2020;31:2623-2626.
- [Google Scholar]
- Electrochemical oxidation of fluoroquinolone antibiotics: Mechanism, residual antibacterial activity and toxicity change. Water Res.. 2016;102:52-62.
- [Google Scholar]
- A new species of luminous bacteria: Vibrio Qinghaiensis sp. Nov. Oceanologia et Limnologia Sinica. 1994;25:273-277.
- [Google Scholar]
- Degradation of sulfadimethoxine by permanganate in aquatic environment: Influence factors, intermediate products and theoretical study. Sci. Total Environ.. 2019;671:705-713.
- [Google Scholar]
- Synergistic degradation of antibiotic sulfadiazine in a heterogeneous ultrasound-enhanced Fe0/persulfate Fenton-like system. Chem. Eng. J.. 2014;257:36-44.
- [Google Scholar]
- Integration of advanced oxidation and membrane filtration for removal of micropollutants of emerging concern. Process Saf. Environ.. 2019;130:67-76.
- [Google Scholar]
Appendix A
Supplementary data
Supplementary data to this article can be found online at https://doi.org/10.1016/j.arabjc.2023.104856.
Appendix A
Supplementary data
The following are the Supplementary data to this article:Supplementary data 1
Supplementary data 1







