Translate this page into:
Studies of phytochemical constituents by UPLC-QTOF-MS/MS of black hulless barley bran and its antioxidation and α-glucosidase inhibition effect
⁎Corresponding authors. yanwan@cdu.edu.cn (Yan Wan), xiachen3722496@163.com (Chen Xia)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Anthocyanidins and flavonoids are important components in plant. In present study, the anthocyanidins and flavonoids in black hulless barley bran were identified by operating UPLC-QTOF-MS/MS. Among them, chrysoeriol 7-O-glucouronid, luteolin 7-O-glucouronid, chrysoeriol, and luteolin were quantified by using UPLC. Additionally, the total phenolic content (TPC), total flavonoids content (TFC) and total anthocyanin content (TAC) were determined. Furthermore, the antioxidant activity (via ferric reducing, DPPH and ABTS method) and α-glucosidase inhibition effect were estimated. From the results, fifty-one constituents comprised 19 anthocyanidins and 32 flavonoids were detected in hulless barley bran fractions. Wherein compounds 1 and 9–19 (belong to anthocyanidins), and 21, 25–32, 35–42, 44–50 (belong to flavonoids) were first identified in hulless barley. The contents of chrysoeriol 7-O-glucouronid luteolin 7-O-glucouronid, chrysoeriol and luteolin were higher in E80% than that of in E40%, but were not detected in E20%. Another, the TAC exhibited the trend of E40% > E80% > E20%, and the TPC and TFC showed same trend of E40% > E20% > E80%. The higher TPC and TFC showed stronger antioxidant activity. However, no significant correlation between TAC, TPC, TFC and α-glucosidase inhibitory activity was observed. These results provided the anthocyanidins and flavonoids information, antioxidant value and α-glucosidase inhibition potential, which could provide reference in the development of black hulless barley bran.
Keywords
Black hulless barley bran
Phytochemical profiling
Antioxidant activity
UPLC-QTOF-MS/MS
1 Introduction
Hulless barley, Hordeum vulgare L. var. Nudum Hook.f., is a widely cultivated cereal crop mainly growing in highland areas around the world, including Qinghai-Tibet Plateau in China, central Siberian Highlands in Russia, the north-western Hinalayas, and highland areas of Canada and Germany (Zhang et al., 2019). Hulless barley is a special cereal crop which possess high content of protein, vitamins and dietary fiber,but low content of sugar and fat, meeting the needs of a modern and healthy lifestyle (Guo et al., 2020). In addition, β-glucan content in huless barley is rich, which have the potential to lower the synthesis of cholesterol and reduce the concentration of low-density blood lipid (Zhang et al., 2019). Moreover, abundant phenolic compounds in hulless barley, including flavonoids, phenolic acids, and anthocyanins (Ge et al., 2020;Yang et al., 2023), endowed it with the ability of anticancer, antimicrobial, antioxidative properties, and reduction of the risk of chronic diseases (Idehen et al., 2017; Hu et al., 2023; Olatunji et al., 2022; Jayeoye et al., 2021). So hulless barley has attracted increased focus because of its beneficial composition and health value in recent years.
Whole grains consist of starchy endosperm, germ, and bran (Liu, 2007). The hull and bran fractions of cereals was usually discarded in order to produce various types of products for consumption. However, the discarded fractions of hulless barley, composed of the bran and hull, accounting for 30 % of the total grain weight, are waste and poorly processed (Jadhav et al., 1998). The hulless barley bran contain amounts of phytochemical components, such as β-glucans, dietary fibre, flavonoids, phenolic acids, and anthocyanin, which act as functional phytochemicals in hull-less barley grain (Šimić et al., 2019; Xiang et al., 2021). In two hull-less and two waxy barleys, the β-glucan content in bran fractions was of the highest (Wiege et al., 2016). While the concentration of β-glucan in shorts fraction was highest (8.12–13.01 %), compared with bran and flour in six hull-less barley cultivars (Zheng et al., (2011). Besides, the bran of hulless barley expressed 68 % and 56 % of the total ABTS antioxidant capacity and the total DPPH antioxidant capacity,respectively, despite it accounted for 7 % of the total grain weight (Xiang et al., 2021). Furthermore, ferulic acid in bran was also highest in hull-less barley (Šimić et al., 2019), similar result showing ferulic acid content was highest in the outer layers in cereals (López-Pereaa et al., 2019; Ndolo et al., 2013; Vaher et al., 2010).
Hulless barley are richer in anthocyanins and flavonoids, but little report exists on the bran of black grains. Thus, in present study, the anthocyanins and flavonoids in black hulless barley bran were identified by using ultra-high-performance liquid chromatography-diode array with quadrupole time-of-flight and tandem mass spectrometry (UPLC-QTOF-MS/MS). Besides, the four flavonoids was quantified by operating ultra high performance liquid chromatography (UPLC). Furthermore, the total phenolic content (TPC), total flavonoid content (TFC) and the total anthocyanin content (TAC) in black hulless barley bran were determined. The antioxidant activities as well as α-glucosidase inhibition effect were also evaluated. This study can provide valuable reference for the utilization of black hulless barley, particularly for the consumption of anthocyanins and flavonoids of black barley bran.
2 Materials and methods
2.1 Materials
The ‘Longzi’, purchased from Tibet Chunguang Food Co. Ltd. (Xizang, China), is a hulless barley variety with black grain color, 10.1 kg bran was obtained from 100 kg hulless barley grain which was miled using a NZJ15/15 grain mill (Changzhou Weihai Machinery Technology Co., Ltd. Changzhou, China). Then these bran were ground into powder and screened through a 60-mesh sieve. These bran powder were kept at −20 °C for further tests.
2.2 Chemicals and reagents
Analytical-grade methanol were obtained from Chengdu Kelong Chemical Reagent Works (Chengdu, China). Chromatographic-grade methanol and acetonitrile were obtained from Sigma-Aldrich (St. Louis, MO, USA).
AB-8 Macroporous Resin was purchased from Shanghai Macklin Biochemical Technology Co., Ltd. (Shanghai, China). Briefly, the resins were washed 3 times with distilled water, then soaked for 24 h with ethanol. Later the resins were washed thoroughly until no alcohol smell with distilled water, then soaked with 4 % HCl for 2 h. Again, the resins were washed to neutral with distilled water, then were soaked with 4 % NaOH for 2 h. Finally, the resins were kept washing until the washing fluid became neutral with distilled water. The water in the resins were removed by filtering before use.
2.3 Sample preparation
100 g sample bran was ultrasound extracted by 800 mL of petroleum ether at 40 °C for 1 h in the KH2200DE ultrasonic instrument (Kunshan Ultrasound Instrument Co., Jiangsu, China). The mixture was filtered. Then the filter residue was collected and dried in the shade. 600 mL of 80 % ethanol together with 0.1 % hydrochloric acid was added into the filter, and was extracted 3 times at 40 °C, each time for 40 min. The three filtrates were mixed and evaporated to 100 mL at 50 °C under vacuum with a rotary evaporator (Hei-VAP, Heidolph Instruments, Schwabach, Germany). Then 100 mL of sample solution was allowed to flow at a rate of 1.0 BV/h through the glass column (diameter 3 cm × 35 cm high), then was adsorbed for 1 h. Then the column was washed with 1000 mL of 0.5 % hydrochloric acid water at a flow rate 2 BV/h, and later washed with 1000 mL eluted solution (20 %, 40 %, 80 % ethanol solution containing 0.5 % hydrochloric acid, respectively), collected 20 %, 40 %, 80 % eluted part. The eluted solution was concentrated under reduced pressure at 50 °C and freeze-dried at −80 °C by FDU-2110 Freeze dryer (Tokyo Rikakikai CO., LTD., Tokyo, Japan). The eluted sample was obtained as follows: 20 % ethanol solution (E20%, 0.5557 g), 40 % ethanol solution (E40%, 0.8994 g), 80 % ethanol solution (E80%, 0.3341 g).
2.4 Anthocyanin and flavonoid compounds identification using UPLC-QTOF-MS/MS
To analyze anthocyanin and flavonoid compounds in the hulless barley bran, an ultra-high-performance liquid chromatography (UPLC) system (Waters Corporation; Milford, MA, USA) with a diode array detector (DAD) was performed. The detector was coupled to a Waters Xevo G2-XS quadrupole time-of-flight (Q-TOF) micro-mass spectrometer (Waters, Manchester, UK), equipped with an electrospray ionization (ESI) source acting on positive mode. Briefly, the chromatographic separation was operated by using the Waters BEH C18 column (2.1 mm × 100 mm, 1.7 μm particle size; Waters Corporation; Milford, MA, USA) as follows: the injection volume of 1 µL, as well as the elution was fulfilled in 18 min with a flow rate of 0.3 mL/min. The compositions of mobile phase were 0.1 % formic acid in water (solvents A) and acetonitrile (solvents B). The gradient conditions were settled as follows: 5 %–10 % B for 0–2 min, 10 %–20 % B for 2–10 min, 20 %–40 % B for 10–15 min, 40 %-70 % B for 15–17 min, 70 %–100 % B for 17–18 min. The DAD spectra for phenolic compounds were all examined at 320 nm. The mass spectrometer (MS) parameters were settled: capillary voltage of 2.5 kV, source default voltage of 80 V, source temperature at 250 °C, desolvation temperature at 120 °C, cone voltage of 40 V, cone gas flow of 50 L/h, as well as desolvation gas flow of 600 L/h. Finally, the MS analysis was accomplished by operating mass scanning from m/z 50 to 1500.
2.5 Quantification of flavonoid compounds using UPLC
The characteristic flavonoid compounds in extracts were determined according to the method of Xiang et al. (2021). An UPLC instrument, equipped with a DAD detector (Agilent LC1290 series, Agilent Technologies, USA), and a 100 mm × 2.1 mm, 1.7 μm particle size Waters BEH C18 (Waters Technologies, USA) were employed in analysis process. The gradient program of mobile phase was assigned as follows: 0–2 min for 5 %-10 % B, 2–10 min for 10 %-20 % B, 10–15 min for 20–40 % B, 15–17 min for 40 %-70 % B, 17–18 min for 70 %-95 % B, 18–20 min for 95 % B at the 0.3 mL/min flow rate. Then detection was carried out at 30 ˚C for column temperature with 1 μL injection volume and 350 nm detection wavelength. Compounds of four flavonoid were quantified with the corresponding standard compounds, and the contents were expressed as µg flavonoid/g dry weight (DW).
2.6 Total anthocyanin content determination
The total anthocyanin content (TAC) was evaluated by the method of Xia et al. (2022) with some modifications, by the Agilent LC-1260 high-performance liquid chromatography (HPLC) system (Agilent, Santa Clara, CA, USA) fitted with a diode array detector (DAD). Then chromatographic separation was performed with an Infinity Lab Poroshell 120 PFP column (4.6 × 100 mm, 2.7 µm particle size, Agilent, Santa Clara, CA, USA). Based on a previous study (Xia et al., 2021), the gradient program of mobile phase was set at 5 %–10 % B (0–10 min), 10 %–20 % B (10–20 min), 20 %–40 % B (20–35 min), 40 %–70 % B (35–40 min), 70 %–95 % B (40–45 min), with a flow rate of 0.8 mL/min. The column temperature was the same as that in section 2.5 with injection volume of 5 µL and the detection wavelength of 525 nm. To evaluate TAC, the.Cyanidin-3-glucoside was utilized as an external standard. The regression equation was y = 3.4613x − 13.562, R2 = 0.9999, with a linear range of 7.81 to 1000 μg/mL. The content of TAC were expressed as mg cyanidin-3-glucoside/g dry weight (DW).
2.7 Total phenolic content analysis
The total phenolic content (TPC) was tested according to the Folin-Ciocalteu colorimetric method. 20 μL Folin-Ciocateu reagent was mixed with 20 μL sample solution, shaken and kept at room temperature for 5 min. Then the mixture was added with 160 μL Na2CO3 solution (5 %, w/v, in water) and reacted at room temperature for 1 h, avoiding light. The absorbance of mixture was monitored at 765 nm. The regression equation of gallic acid was y = 0.0069x + 0.0273 (R2 = 0.9981), with a linear range of 3.31 to 212 μg/mL. The content of TPC was expressed as milligram gallic acid equivalent (GAE) per gram.
2.8 Measurement of total flavonoid content
The total flavonoid content (TFC) was quantified using the method of Xiang et al. (2022), with some modifications. 20 μL sample solution was added with 10 μL 25 % (m/v) NaNO2, mixed and maintained at room temperature for 6 min. Then 10 μL 10 % AlCl3 was mixed and reacted for 5 min. Subsequently, 30 μL 1 mol/L NaOH together with 100 μL distilled water were added to the mixture. The the absorbance was determined at 510 nm. The concentration of TFC was calculated using a standard curve (y = 0.0013x + 0.0037, R2 = 0.9985).
2.9 Analysis of antioxidant activity
The DPPH radical scavenging activity was measured by the method of Yu et al. (2021), with some modification. Briefly, 100 μL sample solution,diluted to the appropriate ratio, was mixted with 100 μL 128.5 μg/mL DPPH solution (dissolved in 80 % methanol), then was incubated at room temperature for 30 min in a darkness before determination at 517 nm. takeing Trolox as a positive control. Then the scavenging activity of DPPH radical was calculated as following.
Where Asample was the absorbance of tested sample solutions, while Acontrol was the absorbance of the control reaction where the sample was replaced by 80 % methanol.
The ABTS radical scavenging activity was assayed by the method of Yu et al. (2021), with modifications. The working ABTS reagent containing 2.45 mM K2S2O8 and 7 mM ABTS, was incubated for 16 h without light. When the absorbance of ABTS reagent was 7.0, it was diluted by pH = 7.4 PBS solution.Later, the volume of 40 μL sample solution (diluted to the appropriate ratio) was incubated with ABTS reagent (160 μL) for 6 min at room temperature in a darkness. The absorbance was subsequently recorded at 734 nm. Trolox was also used as a positive control. The scavenging activity of ABTS radical was measured by the Eq.1.
The reducing power was determined as method described previously with some modifications (Dudonné et al., (2009). To prepare the working FRAP reagent, 0.3 M CH3COONa (pH = 3.6), 10 mM 2,4,6-tri(2-pyridyl)-s-triazine (TPTZ, in HCl), as well as 20 mM FeCl3 was mixed in a ratio of 10:1:1. Then 30 μL sample solutions, which was diluted to the appropriate ratio, was incubated with 265 μL FRAP reagent at 37 °C in a water bath for 30 min. Similarly using Trolox as a positive control, the absorbance of 593 nm was measured. A sample blank test was also performed using sodium acetate buffer. The reduction activity of the extract was reflected by the solution absorbance. The sample owned stronger reducing power as with the bigger solution absorbance.
2.10 The inhibition effect of α-glucosidase
The α-glucosidase inhibition effect was measured according to the method described by Yang et al. (2021) with slight modifications. PBS (0.1 M, pH = 6.9) was used to prepare the reagent of α-glucosidase and pNPG. Sample solution (50 μL), which was diluted to the appropriate ratio, was incubated with α-glucosidase reagent (1U/mL, 500 μL) in a 37 ˚C water bath for 10 min, and then added with 50 μL of 5 mM pNPG. After kept in a 37 ˚C water bath for 5 min, 50 μL of 1 M Na2CO3 was added into the mixture system. Then the absorbance of 405 nm was recorded, by setting the absorbance as a positive control. To calculate the enzyme inhibition activity, the following equations were used:
A1:sample + PBS + pNPG + Na2CO3.
A2:PBS + α-glucosidase + pNPG + Na2CO3.
A3:PBS + PBS + pNPG + Na2CO3.
2.11 Statistical analysis
All determined experiments were performed in triplicate otherwise specified. The results were expressed as mean ± standard deviation (SD). The stastical analysis was determined by one-way ANOVA, followed by Student-Newman-Keuls (SNK) test (version 20.0, IBM SPSS Statistics, Armonk, NY USA). Pearson's correlation coefficient was used to determine the correlation analysis among TAC, TPC, TFC, four quantified flavonoids, antioxidant activity and inhibition of a-glucosidase. Value with p < 0.05 were consideried statistically significant.
3 Results and discussion
3.1 Identification of anthocyanin and flavonoid compounds
51 compounds including 19 anthocyanidins and 32 flavonoids in black hulless barley bran fractions were characterized and identified using authentic standards, published references, as well as MS/MS fragmentation patterns in total (Table 1). And the chemical structure, characteristics of secondary ion mass spectrum in positive, and fragmentation patterns of Compounds 26 and 38 were exhibited in Fig. 1. The MS spectra of the identified compounds in E80% fraction was supported in Supplementary material 1. The anthocyanidins contained 18 cyanidin derivatives and one pelargonidin 3-O-glucoside. The dominant flavonoids in extracts were chrysoeriol and luteolin, and the sugars were glucose, galactoside, glucuronide. From the Table 1, the extracts contain 0, 18 and 9 cyanidin derivatives for E20%, E40%, E80%, respectively. These results were consistent with TAC content determined in present study. Another, the extracts contain 0, 24 and 27 flavonoids for E20%, E40%, E80%, respectively. -, No detected. +, Detected. *, identified by reference compound. #, first identified in hulless barley.
Peak no.
Rt
(min)[M + H]+
(m/z)Formula
Fragment ions
Identified compounds
20 %
40 %
80 %
#1
4.688
773.2142
C33H41O21
287
Cyanidin 3,3′,5-tri-O-glucoside
–
+
+
2
4.748
611.1701
C27H31O16
287
Cyanidin 3,5-O-diglucoside
–
+
+
3
5.231
449.1171
C21H21O11
287
Cyanidin 3-O-glucoside
–
+
+
4
5.261
611.1701
C27H31O16
287
Cyanidin-O-diglucoside
–
+
+
5
5.575
611.1802
C27H31O16
287
Cyanidin-O-diglucoside
–
+
–
6
5.942
433.1229
C21H21O10
271
Pelargonidin 3-O-glucoside
–
+
+
7
6.624
535.116
C24H23O14
287
Cyanidin 3-O-(6″-O-malonyl)-galactoside
–
+
+
8
7.393
535.116
C24H23O14
287
Cyanidin 3-O-(6″-O-malonyl)-galactoside
–
+
+
#9
8.219
783.1716
C35H43O20
287
Cyanidin 3,5-di-O-[6″-O-(malonyl)-galactoside]
–
+
–
#10
8.503
783.1716
C35H43O20
287
Cyanidin 3,5-di-O-[6″-O-(malonyl)-glucoside]
–
+
–
#11
8.785
621.1219
C27H25O17
287
Cyanidin 3-O-(3″,6″-O-dimalonyl)-glucoside
–
+
–
#12
9.554
621.1219
C27H25O17
287
Cyanidin 3-O-(3″,6″-O-dimalonyl)-glucoside
–
+
+
#13
9.898
549.1324
C25H25O14
287
Cyanidin 3-O-(6″-O-succinyl)-glucoside
–
+
–
#14
10.436
563.1505
C25H23O15
287
Cyanidin 3-O-(6″-O-succinyl)-glucuronide
–
+
+
#15
11.09
635.1276
C28H27O17
287
Cyanidin 3-O-(3″,6″-O-dimalonyl)-glucuronide
–
+
–
#16
11.518
635.1379
C28H27O17
287
Cyanidin 3-O-(3″,6″-O-dimalonyl)-glucuronide
–
+
–
#17
11.577
563.1505
C25H23O15
287
Cyanidin 3-O-(6″-O-succinyl)-glucuronide
–
+
–
#18
12.468
649.1435
C29H29O17
287
Cyanidin 3-O-(3″,6″-O-disuccinyl)-galactoside
–
+
+
#19
12.753
649.1435
C29H29O17
287
Cyanidin 3-O-(3″,6″-O-disuccinyl)-galactoside
–
+
+
20
6.434
463.1313
C22H22O11
301,286,258,229
Chrysoeriol-O-glucoside
–
+
–
#21
9.328
549.1324
C25H24O14
301,286,258
Chrysoeriol 7-O-(6″-O-malonyl)-galactoside
–
+
–
22
9.847
433.1187
C21H20O10
313,283
Isovitexin or vitexin
–
+
+
*23
10.476
449.1127
C21H20O11
287,153
Luteolin 7-O-glucoside
–
–
+
*24
10.568
463.0961
C21H18O12
287,153
Luteolin 7-O-glucuronide
–
+
+
#25
10.622
639.1653
C25H24O14
301,286,258
Chrysoeriol-O-glucoside-O-glucuronide
–
+
–
#26
11.136
639.1653
C27H26O18
477,301,286,258,229,153
Chrysoeriol 7-O-glucuronide 4′-O-glucoside
–
+
+
#27
11.246
653.1418
C27H24O19
301,286,258,229,153
Chrysoeriol 7-O-diglucuronide
–
–
+
#28
12.216
653.1313
C27H24O19
301,286,258,229,153
Chrysoeriol 7-O-diglucuronide
–
+
#29
12.274
609.1743
C26H24O17
463,301,286,258
Chrysoeriol-O-rhamnoside-O-glucoside
–
+
+
#30
12.476
639.1653
C27H26O18
477,301,286,258
Chrysoeriol-O-glucoside-O-glucuronide
–
+
+
#31
12.675
639.1705
C27H26O18
477,301,286,258
Chrysoeriol-O-glucoside-O-glucuronide
–
+
+
#32
12.723
577.1584
C26H24O15
301,286,258
Chrysoeriol 7-O-(6″-O-succinyl)-glucuronide
–
+
+
*33
12.734
463.1313
C21H18O12
301,286,258,229,183
Chrysoeriol 7-O-glucoside
–
+
+
*34
12.807
477.1062
C22H20O12
301,286,258,229,153
Chrysoeriol 7-O-glucuronide
–
+
+
#35
12.93
609.149
C27H28O16
477,301,286,258,229
Chrysoeriol-O-pentoside-O-glucuronide
–
–
+
#36
13.519
477.1062
C22H20O12
287,153
Luteolin 7-O-methylglucuronide
–
+
#37
13.6
563.1116
C26H27O14
301,286,258
Chrysoeriol 7-O-(6″-O-succinyl)-glucoside
–
–
+
#38
13.918
549.1324
C25H24O14
463,301,286,258,229,153
Chrysoeriol 7-O-(6″-O-malonyl)-glucoside
–
+
+
#39
13.954
653.1679
C27H24O19
491,301,286,258
Chrysoeriol-O-methylglucuronide-O-glucoside
–
+
–
#40
14
563.1116
C26H27O14
301,286,258,229
Chrysoeriol 7-O-(6″-O-succinyl)-glucoside
–
+
+
#41
14.296
653.1783
C27H24O19
491,301,286,258
Chrysoeriol-O-methylglucuronide-O-glucoside
–
+
#42
14.37
491.121
C24H26O11
287,153
Luteolin 7-O-(6″-O-acetyl)-glucoside
–
+
+
*43
14.454
287.0577
C15H10O6
153
Luteolin
–
+
+
#44
14.656
491.121
C24H26O11
301,286,258,229,153
Chrysoeriol 7-O-methylglucuronide
–
+
+
#45
14.709
667.1954
C30H34O17
505,301,286,258
Chrysoeriol-O-acetylglucoside-O-glucoside
–
+
+
#46
14.851
667.206
C30H34O17
505,301,286,258
Chrysoeriol-O-acetylglucoside-O-glucoside
–
+
+
#47
15.306
753.1874
C33H36O20
447,301,286,258
Chrysoeriol-O-glucuronide-O-(6″-O-succinyl)-glucuronide
–
–
+
#48
15.307
667.1954
C30H34O17
505,301,286,258
Chrysoeriol-O-acetylglucoside-O-glucoside
–
+
+
#49
15.446
505.1364
C24H24O12
301,286,258,229
Chrysoeriol 7-O-(6″-O-acetyl)-glucoside
–
+
+
#50
15.858
577.1584
C26H24O15
301,286,258,229
Chrysoeriol 7-O-(6″-O-succinyl)-glucuronide
–
–
+
*51
15.899
301.0773
C16H12O6
286,258,229,153
Chrysoeriol
–
+
+
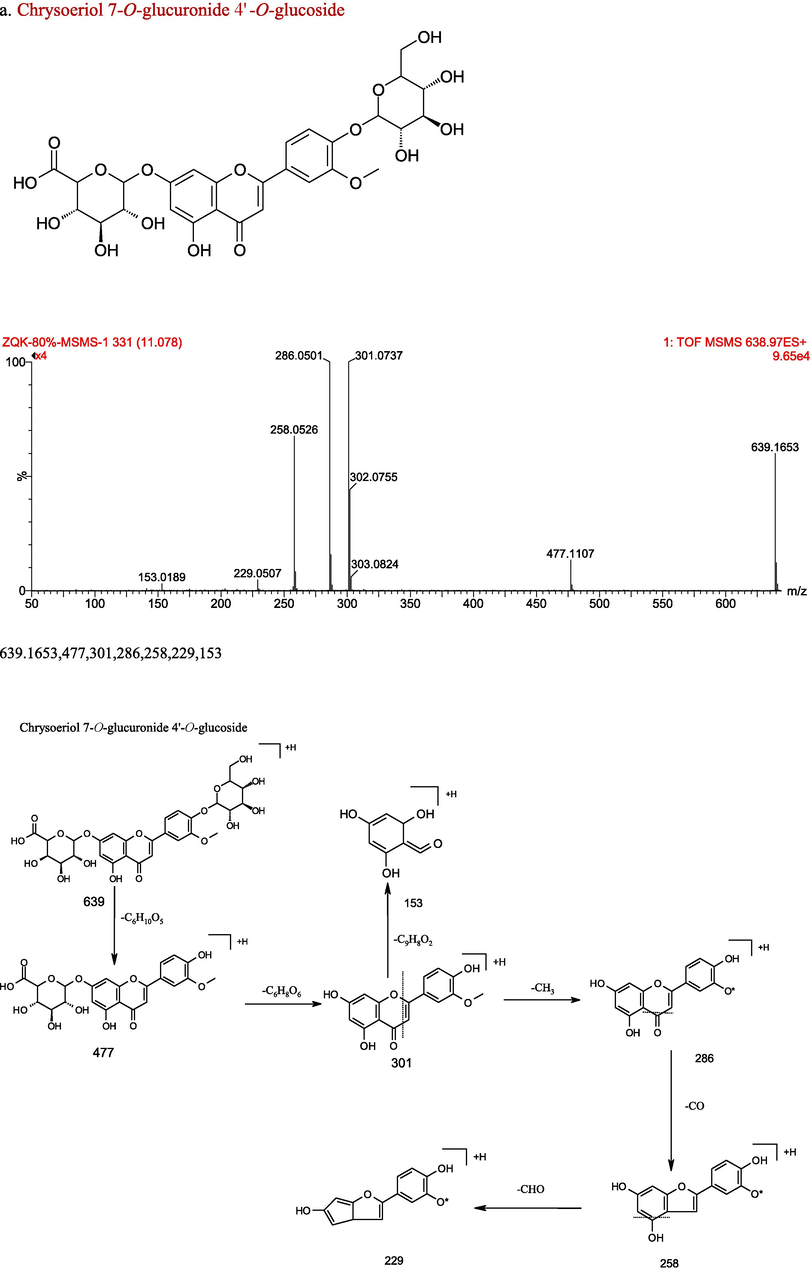
The chemical structure, characteristics of secondary ion mass spectrum in positive, and fragmentation patterns of Chrysoeriol 7-O-glucuronide 4′-O-glucoside (a) and Chrysoeriol 7-O-(6″-O-malonyl)-glucoside (b).
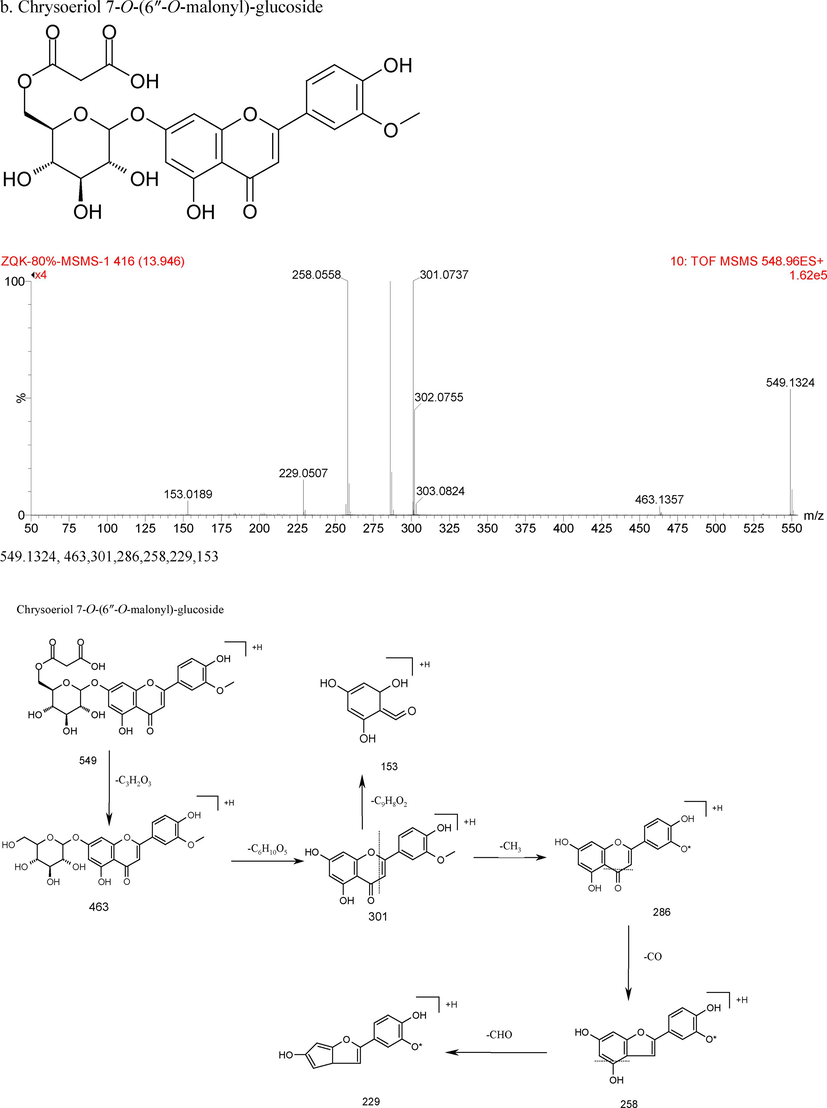
The chemical structure, characteristics of secondary ion mass spectrum in positive, and fragmentation patterns of Chrysoeriol 7-O-glucuronide 4′-O-glucoside (a) and Chrysoeriol 7-O-(6″-O-malonyl)-glucoside (b).
Compound 1 displayed a molecular ion at m/z 733, together with a fragment ion at m/z 287 ([M + H-162–162–162]+ loss of three glucose moieties, established as cyanidin 3,3′,5-tri-O-glucoside. Compound 2, 4 and 5 exhibited a molecular ion [M + H]+ at m/z 611, along with a fragment ion at m/z 287 ([M + H-162–162]+ loss of two glucose moieties), confirmed as cyanidin 3,5-O-diglucoside isomers published previously (Fischer et al., 2011; Hou et al., 2013; Hao et al., 2015). Compound 3 held a molecular ion [M + H]+ at m/z 449 as well as a fragment ion at m/z 287 ([M + H-162]+ loss of a glucose moiety), identified as cyanidin 3-O-glucoside by reference to previous report (Fischer et al., 2011; Hou et al., 2013; Hao et al., 2015). Compound 6 presented a molecular ion at m/z 433, together with a fragment ion at m/z 271 ([M + H-162]+ loss of one glucose moiety, confirmed as pelargonidin 3-O-glucoside published previously (Mullen et al., 2008). Compounds 7 and 8 showed a similar parent ion at m/z 535, as well as a fragment at m/z 287 ([M + H-162–86]+ loss of one glucose and one malonic acid), certified as cyanidin 3-O-(6″-O-malonyl)-glucoside isomers (Viacava et al., 2017; Chen et al., 2012). During the grain color-changing and maturation stages, cyanidin 3-O-(6″-O-malonyl)-glucoside together with cyanidin 3-O-glucoside were significantly increased in the purple huless barley grains (Xu et al., 2022). Compounds 9 and 10 showed a similar parent ion at m/z 783, and a fragment at m/z 287 ([M + H-162–162–86–86]+ loss of two glucose moieties and two malonyl moieties), affirmed as cyanidin 3,5-di-O-[6″-O-(malonyl)]-glucoside isomers (Tatsuzawa et al., 2011). Compounds 11 and 12 showed a similar parent ion at m/z 621, along with a fragment at m/z 287 ([M + H-162–86–86]+ loss of one glucose moiety and two malonyl moieties), identified as cyanidin 3-O-(3″,6″-O-dimalonyl)-glucoside isomers, which structure was confirmed by Chen et al. (2012) and Nakayama et al. (1997). Compounds 13 showed a parent ion at m/z 549, as well as a fragment at m/z 287 ([M + H-162-100]+ loss of one glucose moiety and one succinyl moiety), confirmed as cyanidin 3-O-(6″-O-succinyl)-glucoside which was also identified in Phragmites australis (Fossen and Øyvind, 1998). Compounds 14 and 17 showed a same parent ion at m/z 563, together with a fragment at m/z 287 ([M + H-176-100]+ loss of one glucuronyl moiety and one succinyl moiety), established as cyanidin 3-O-(6″-O-succinyl)-glucuronide isomers. Compounds 15 and 16 showed a same parent ion at m/z 635, and a fragment at m/z 287 ([M + H-176-86-86]+ loss of one glucuronyl moiety and two malonyl moieties), identified as cyanidin 3-O-(3″,6″-O-dimalonyl)-glucuronide isomers. Compounds 18 and 19 showed a same parent ion at m/z 649, as well as a fragment at m/z 287 ([M + H-162-100-100]+ loss of one glucose moiety and two succinyl moieties), confirmed as cyanidin 3-O-(3″,6″-O-disuccinyl)-glucoside isomer. Up to present, there is no identification details about Compounds 1, 14-19 in published papers.
Compounds number 23, 24, 33, 34, 43 and 51 were unambiguously established as chrysoeriol-7-O-glucoside, luteolin-7-O-glucoside, luteolin 7-O-glucuronide, chrysoeriol 7-O-glucuronide, luteolin as well as chrysoeriol, respectively, based on the comparisons of MS fragment characteristics with authentic standards, and combination with previous studies (Petar et al., 2007; Kumar et al., 2017). Compounds number 20–22, 25–32, 35–42, and 44–50 were preliminary identified by operating a combination of MS spectra in the published references.
Compound 22 exhibited the parent ion [M + H]+ at m/z 433, and fragment ion at m/z 313 ([M + H-C3H6O3]+), 283 ([M + H-C4H8O4]+). Through comparison with corresponding MS data, it was preliminary identified as isovitexin or vitexin (Peng et al., 2005). Compound 51 showed positive molecular ion [M + H]+ at m/z 301, together with fragment into ions at m/z 286 ([M + H-CH3]+), 258 ([M + H-CH3-CO]+), 229 ([M + H-CH3-CO-HCO]+), 153(RDA 1/3). Furthermore, it was identified as chrysoeriol by using authentic standards, which was also reported by Petar et al. (2007). These fragments allow us to rapidly distinguish the derivatives of chrysoeriol in chemical profiling. Compounds 20–21,25–32,35–41, 44–50 showed the same fragment ion at 301, 286, 258, which indicated that they were chrysoeriol derivatives. Compound 20 showed the parent ion [M + H]+ at m/z 463, and the MS2 ions at m/z 301 ([M + H-162]+ loss of one glucose moiety), 286, 258, 229, which indicated that it was chrysoeriol -O-glucoside isomer. Compounds 21 and 38 showed the deprotonated [M + H]+ ion with m/z 549, further fragmented into ions at m/z 301 ([M + H-162-86]+ loss of one glucose moiety and one malonyl moiety), thus they were identified as chrysoeriol 7-O-(6″-O-malonyl)-glucoside isomers (Conrad et al., 2009). Compounds 25, 26, 30 and 31 presented the same parent ion [M + H]+ at m/z 639, demonstrating their possibility as isomers. They produced MS2 ions at m/z 477 ([M + H-162]+ loss of one glucose molecule), 301 ([M + H-162-176]+ loss of one glucose molecule and one glucuronyl group), so they were comfirmed as chrysoeriol-O-glucoside-O-glucuronide isomer. Compounds 27 and 28 showed the same parent ion at m/z 653, along with fragment ions at m/z 477 ([M + H-176-176]+ loss of two glucuronyl groups), and 286, 258, 229, 153. so they were recognized as chrysoeriol-7-O-diglucuronide isomers. Compound 29 presented the parent ion at m/z 609, fragment ion at m/z 463 ([M + H-146]+ loss of one rhamnoside group) and 301 ([M + H-146-162]+ loss of one rhamnoside group and one glucoside group). Based on the fragments and relevant literature (Petar at al., 2007), compound 29 was identified as chrysoeriol-O-rhamnoside-O-glucoside. Compounds 35 showed the parent ion at m/z 609, together with fragment ion at m/z 477 ([M + H-132]+ loss of one pentoside group), and 301 ([M + H-132-176]+ loss of one pentoside group and one glucuronyl group), which indicated it was chrysoeriol-O-pentoside-O-glucuronide. Compounds 37 and 40 showed same parent ion at m/z 563, as well as fragment ion at m/z 301 ([M + H-162-100]+ loss of one glucose molecule and one succinyl group), which indicated they were chrysoeriol 7-O-(6″-O-succinyl)-glucoside isomers. Compounds 39 and 41 showed same parent ion at m/z 653, along with fragment ion at m/z 491 ([M + H-162]+ loss of one glucose molecule), 301 ([M + H-162-176-14]+ loss of one glucuronyl group as well as a methyl group from fragment ion at m/z 491), indicating they were chrysoeriol-O-methylglucuronide-O-glucoside isomers. Compound 44 showed fragmented into ion at m/z 301 ([M + H-176-14]+ loss one glucuronyl group and a methyl group), indicated it was chrysoeriol-O-methylglucuronide. Compounds 45,46 and 48 showed same parent ion at 667, and fragment ions at m/z 505 ([M + H-162]+ loss of one glucose molecule), and 301 ([M + H-162-162-42]+ loss of two glucose moieties and one acetyl moiety), which indicated they were chrysoeriol-O-acetylglucoside-O-glucoside isomers. Compound number 47 showed parent ion at 753, as well as fragment ions at m/z 447 ([M + H-132-132-42]+ (loss of two pentose groups along with one acetyl group), 301 ([M + H-132-132-42-146]+ loss of two pentose groups, one acetyl grouploss and one rhamnose moiety), which indicated it was chrysoeriol-O-digpentose-(6″-O-acetyl)-rhamnose. Compound 49 showed the ions of fragment was at m/z 301 ([M + H-162-42]+, lossing of one glucose molecule as well as one acetyl group), which indicated it was chrysoeriol 7-O-(6″-O-acetyl)-glucoside. Compound 50 showed ions of fragment was at m/z 301 ([M + H-176-100]+, lossing of one glucuronyl group and one succinyl group), which indicated it was chrysoeriol 7-O-(6″-O-succinyl)-glucuronide.
Compound 43 was identified as luteolin, using authentic standards. It showed positive molecular ion [M + H]- at m/z 287 and further fragmented into ion at m/z 153, similar to the result reported by Kumar et al. (2017). Compounds 36 and 42 showed the ions of fragments at m/z 287 and 153, suggesting they were luteolin derivatives. As the fragment of compound 36 was at m/z 287 ([M + H-176-14]+ loss of one glucuronyl group as well as one methyl group, it was further established as luteolin 7-O-methylglucuronide. Compound 42 exhibited the pseudomlecular ion at m/z 491, together with the fragment ion at m/z 287 ([M + H-162-42]+ losing of one glucoside group and one acetyl group), so it was vertified as luteolin 7-O-(6″-O-acetyl)-glucoside. However, identification details about compounds 25–28, 30–32, 35–37, 39–42, 44–50 were not found in published papers. And the compounds 1, 9–19, 21, 25–32, 35–42, 44–50 were first identified in hulless barley.
3.2 Quantification
Chrysoeriol, chrysoeriol 7-O-glucouronid, luteolin as well as luteolin 7-O-glucouronid were further quantified using UPLC, which exhibited as the higher content than other flavonoids in hulless barley in our research and also proved in other literature (Xiang et al., 2021). As shown in Table 2, the four flavonoids were all not detected in E20%, and higher content in E80% than that of in E40%. This was likely that the polarity of these four flavonoids were closer to that of E80%. As widely distributed in the plant kingdom, Luteolin and its glycosides were proved to possess the potential activities to antioxidant, anti-inflammatory, antimicrobial as well as anticancer (Bangar et al., 2023; Wang et al., 2021). Compared with other cereal (Tiozon et al., 2022; Suchowilska et al., 2020), black hulless barley exhibited higher content of luteolin (1.79 mg/g in E40%, and 6.82 mg/g in E80%) than that of rice, sorghum and wheat (0.5–1 mg/100 g, 0–18.2 mg/100 g, 3.1–4.14 mg/100 g, respectively). The higher content of flavonoid in hulless barley might be correlated with its cultivated place-Tibetan Plateau (>4,000 m above sea level), which is characterized by the high UV-B radiation (about 65 kJ m−2 in the summer), low temperatures (average yearly temperature 7.6 °C), and low barometric pressure (about 650 mbar) (Norsang et al., 2009). On the evolution of the downstream phenylpropanoid pathway, most plants have adopted flavonoids as the major line of defense in resisting UV-B radiation (Zeng et al., 2019). Chrysoeriol, a 30-O-methoxy flavone, is a methoxy derivative of luteolin observed in numerous plants and observed as several derivatives. (Boro et al., 2023; Bangar et al., 2023). It has the anticancer, anti-inflammatory, antibacterial, antifungal, anti-osteoporosis, anti-insecticide, and neuroprotective actions (Boro et al., 2023). However, little information about the quantitative analysis of chrysoeriol in other cereal in published papers. Different lowercase superscripts within the same column indicate significant difference (p < 0.05).
Flavonoid compounds
Equation
R2
Linear range (µg/mL)
Content (mean ± SD, mg/g)
E20%
E40%
E80%
Luteolin 7-O-glucouronid
y = 11.245 x + 2.217
0.9999
2–200
0
8.56 ± 0.04b
9.45 ± 0.04a
Chrysoeriol 7-O-glucouronid
y = 9.3088 x - 64.583
0.9994
2.5–225
0
29.97 ± 0.13b
132.78 ± 0.61a
Luteolin
y = 10.11 x -7.34
0.9999
2–200
0
1.79 ± 0.01b
6.82 ± 0.03a
Chrysoeriol
y = 12.366 x + 1.0054
0.9999
2.46–246
0
3.20 ± 0.02b
28.67 ± 0.04a
TAC
y = 3.4613 x - 13.562
0.9999
7.81–1000
8.53 ± 0.78c
176.26 ± 7.56a
31.32 ± 0.45b
TPC
y = 0.0069 x + 0.0273
0.9981
3.31–212
190.80 ± 3.79b
237.83 ± 1.28a
133.18 ± 5.22c
TFC
y = 0.0013 x + 0.0037
0.9985
1.56–200
107.55 ± 4.73b
125.16 ± 5.13a
63.97 ± 5.45c
As an important group of water-soluble pigments in plants, anthocyanins would produce red, purple, blue as well as intermediate hues in plant tissues (Clifford, 2000). Another, anthocyanins is significant because of their antioxidant, anticancer and antidiabetes activities and human nutrition (Konczak and Zhang, 2004; Stintzing and Carle, 2004). From Table 2, TAC showed the trend of E40% > E80% > E20%, which indicated that large amount of anthocyanins in black hulless barley bran were resolved in 40 % ethanol. This result was accordance with the UPLC-QTOF-MS/MS result (Table 1), no anthocyanidins and flavonoids was detected in E20%, 19 anthocyanidins and 24 flavonoids were found in E40%, 10 anthocyanidins and 28 flavonoids were found in E80%.
Being recognized as a major group of phytochemicals, phenolic compounds contribute largely to the antioxidant capacity in cereals (Van Hung, 2016; Šimić et al., 2019). The hulless barley was also a rich cereal of phenolic compounds (Zeng et al., 2019; Ge et al., 2020). In present study, the TPC and TFC in E40% were 237.83 and 125.16 mg/g, respectively. The total phenolic content was significantly affected by cultivars, milling conditions and milling fractions (Šimić et al., 2019; Moza and Gujral, 2017). From Table 2, the TPC and TFC showed same trend of E40% > E20% > E80%. Ethanol is a polar-protic solvent which is preferential to extract low molecular weight compounds, such as glycosylated and non-glycosylated phenolic compounds (Yusoff et al., 2022). However, different ethanol concentration exhibited different capacity on extraction content and type of polyphenols. Bai (2014) also reported that 40 % ethanol as the extraction solvent could obtained higher content polyphenols in apple pomace than using 20 % and 80 % ethanol. Furthermore, as shown in Table 2, not a single flavonoid or phenolic acid compound was identified with UPLC-MS/MS, E20% exhibited higher TPC and TFC than those of E80%. In our previous study (Deng et al., 2021), ferulic acid was the predominant phenoilc acid in hulless barley, ranging from 266.6 to 760.64 μg/g. Furthermore, Ge et al. (2020) identified 156 phenolic substances, including monophenol, phenolic acids, flavonoids and other polyphenols. And the contents of chlorogenic acid (16.898 μg/g), quercitrin (184.093 μg/g), vitexin (253.012 μg/g) were more higher than other quatified polyphenols in black hulless barley. These implied that there were abundance of flavonoid or polyphenols compound in E20%.
3.3 Antioxidant properties
Antioxidants are believed as the essential for the body defense system against oxidative stress (Kancheva and Kasaikina, 2013). Thus antioxidant capacity has become a desirable and essential quality characteristic of plant. In present study, the antioxidant activity of three extracts were examined through the ferric reducing antioxidant potential (FRAP) tests, and DPPH, ABTS radical scavenging assays.
The high correlation coefficients between absorbance and tested concentrations of E80%, E40%, E20% and trolox (0.9956, 0.9992, 0.9985 and 0.9999), demonstrated that the reducing power of the three samples together with trolox was dose-dependent. As shown in Fig. 2a, the reducing power followed the trend as trolox > E40% > E20% > E80%. The scavenging effect on DPPH radical was also in a concentration dependent manner (Fig. 2b). The DPPH IC50 values, the concentration of sample to inhibition 50 % of the radical, were 15.02 ± 0.22, 99.52 ± 2.36, 45.49 ± 0.62, 53.93 ± 0.47 μg/mL for trolox, E80%, E40%, E20%, respectively (P < 0.01). The sequence of scavenging effect was trolox > E40% > E20% > E80%. This result was consistent with reducing power analysis. And the three extracts were all exceed 80 % scavenging effect on DPPH radical within a certain concentration range. As shown in Fig. 2c, the scavenging effect all samples on ABTS radical was also concentration dependent. The IC50 values of trolox, E80%, E40% and E20% were 44.40 ± 1.06, 47.80 ± 1.60, 25.60 ± 1.33 and 28.02 ± 0.91 μg/mL, respectively (P < 0.01). The sequence of scavenging effect was trolox > E40% > E20% > E80%. The similar trend were exhibited in reducing power and DPPH assays (Fig.a-b). And the three extracts were all exceed 90 % scavenging effect on ABTS radical within a certain concentration range.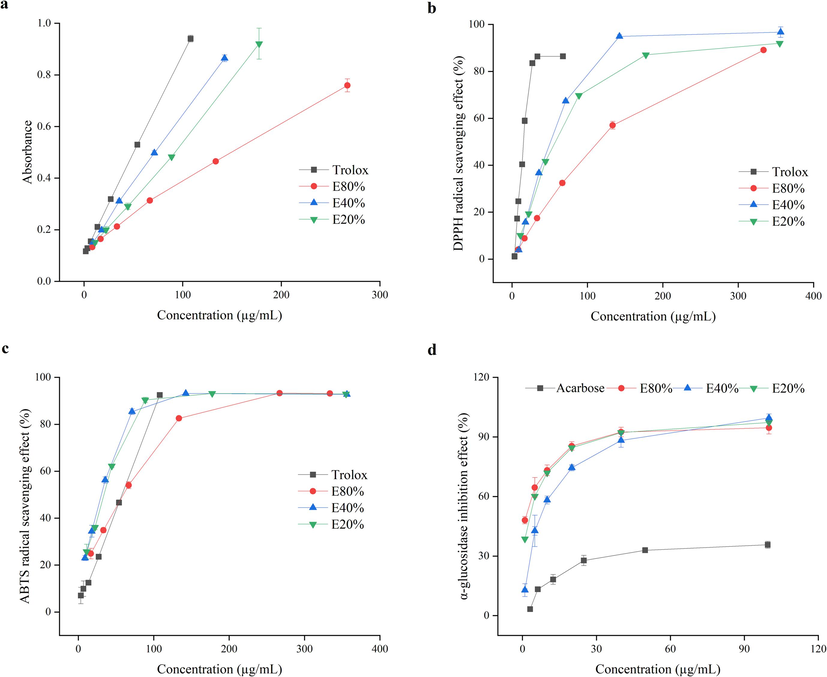
The antioxidant activity (a, reducing power. b, DPPH. c, ABTS.) and α-glucosidase inhibition effect of extracts in black hulless barley bran (d).
The reaction mechanism of 2,2-azinobis (3-ethyl-benzothiazoline-6-sulphonic acid) (ABST), 2,2-diphenyl-1-picrylhydrazyl (DPPH), and ferric reducing antioxidant potential (FRAP) tests all belong to the single electron transfer mode (Kancheva and Kasaikina, 2013), which maybe the main reason for the phenomenon that three extracts have similar change trends in antioxidant activity: E40% > E20% > E80%. Another, the same trends were discovered in TPC and TFC (Table 2). Phenolic compounds are considered as the major antioxidants in cereals (Van Hung, 2016; Ge et al., 2020). So phenolics and flavonoids might be the major antioxidant compounds in hulless barley samples.
3.4 α-glucosidase inhibition effect assay
Due to gastro-intestinal intolerability, side effects as well as high cost, the use of commercial α-glucosidase inhibitors (acarbose, miglitol and voglibose) is restricted (Andrea and Davide, 2014). Plant extracts, such as cranberry (Apostolidis et al., 2006), tea polyphenols (Kan et al., 2021), brown rice bound phenolics (Ye et al., 2022) were investigated to have strong α-glucosidase inhibitory effect, potentially demonstrating their efficacy in effective management of obesity and diabetes mellitus. Our result also showed obvious dose-dependent manner when referred to inhibition effect on α-glucosidase. The IC50 values were 188.42 ± 36.63, 1.47 ± 0.19, 5.99 ± 1.81, 2.33 ± 0.34 μg/mL for acarbose, E80%, E40%, E20%, respectively. The IC50 values of three sample were significantly lower than those of acarbose (p < 0.01). The inhibition effect followed the pattern as: E80% > E20% > E40% > acarbose. The results manifested that compared with acarbose, these three samples were preferable α-glucosidase inhibitors in vitro, which containing exceed 90 % α-glucosidase inhibition effect within a certain concentration range. Another, from the Table 1, 0, 24 and 27 flavonoids were identified in E20%, E40%, E80%, respectively. But E80% contained lower content of TFC. Yang et al. (2021) reported that the number of hydroxyl groups on the A-ring might of flavonoids served as the most effective α-glucosidase inhibitor. So it was hypothesized that there were more specific flavonoid substances, which had the good effect on inhibition of α-glucosidase, in E80% portion.
3.5 Correlation analysis
The high negative correlations were exhibited between the TPC and the antioxidant activities (presented by IC50 value, which smaller means stronger antioxidant capacity) measured by reducing power, DPPH, ABTS (r = -949, r = -0.948, r = -0.820, p < 0.01,Table 3), representing that the higher TPC, the stronger antioxidant activities, in agreement with our previous studies on hulless barley (Deng et al., 2021). Similarly, TPC in colored barley were found to be the main contributors of DPPH and ABTS free radical scavenging activities (Suriano et al., 2018). Plant polyphenols are multifunctional, which could be served as hydrogen donating antioxidants, reducing agents, as well as singlet oxygen quenchers (Rice-Evans et al., 1996). **p < 0.01 (2-tailed). *p < 0.05 (2-tailed).
TAC
TPC
TFC
Reducing power
DPPH (used IC50 value, μg/mL)
ABTS (used IC50 value, μg/mL)
Inhibition of a-glucosidase
(used IC50 value, μg/mL)Luteolin 7-O-glucouronid
Chrysoeriol 7-O-glucouronid
Luteolin
Chrysoeriol
TAC
1
0.758*
0.624
−0.542
−0.518
−0.472
0.886**
0.53
−0.188
−0.142
−0.291
TPC
1
0.978**
−0.949**
−0.948**
−0.920**
0.842**
−0.145
−0.779*
−0.750*
−0.841**
TFC
1
−0.971**
−0.981**
−0.955**
0.776*
−0.318
−0.870**
−0.849**
−0.916**
Reducing power
1
0.994**
0.991**
−0.705*
0.415
0.923**
0.895**
0.949**
DPPH
(used IC50 value, μg/mL)
1
0.988**
−0.688*
0.446
0.937**
0.915**
0.964**
ABTS
(used IC50 value, μg/mL)
1
−0.644
0.48
0.947**
0.923**
0.965**
Inhibition of a-glucosidase
(used IC50 value, μg/mL)
1
0.219
−0.432
−0.399
−0.522
Luteolin 7-O-glucouronid
1
0.728*
0.760*
0.653
Chrysoeriol 7-O-glucouronid
1
0.993**
0.989**
Luteolin
1
0.988**
Chrysoeriol
1
Additional, the high negative correlations were found between TFC and the antioxidant activities (presented by IC50 value) inluding reducing power, DPPH, ABTS (r = -971, r = -0.981, r = -0.955, p < 0.01), suggesting that flavonoids were also the important ingredients of antioxidant activity in huless barley. Antioxidant activities of plants were closely associate with their polyphenols and flavonoids (Scalbert et al., 2005). The antioxidant activity of the flavonoids relies on their capability, which contribute protons and electrons to resist the impact of energetic oxidants, for instance free radicals (Martins et al., 2004).
The positive correlations between α-glucosidase inhibition effect and TAC, TPC and TFC were displayed in Table 3, indicating that total anthocyanin, total phenolic compounds and total flavonoids were not significant effective on a-glucosidase inhibitory activity. It was reported that phenolic phytochemicals from plant sources might be efficiently a-glucosidase inhibitors (Apostolidis and Lee, 2010; Wu and Xu, 2014; Nguyen et al., 2022). The catalytic activity of α-glucosidase could be reduced because of the reversible combination of polyphenols and glucosidase (Wang, 2017). However, the significant inhibition effect of TAC, TPC and TFC on α-glucosidase were not found in present study. β-glucan have the potential to lower the synthesis of cholesterol and reduce the concentration of low-density blood lipid, and hulless barley is the richest cereal in terms of β-glucan content (Zhang et al., 2019). But Tian et al. (2013) reported that the highland barley extract had a better effect than single β-glucans on hypoglycemic activity. Bellesia and Tagliazucchi (2014) pointed that the binding force and stability of polyphenols and a-glucosidase are different, largely depending on the structure of polyphenols, including structure of parent nucleus, type and number of substituent, and binding site. Yao et al. (2022) reported that hyperoside and scoparone might be the main active substance in hulless barley on lipid-lowering effects. So β-glucans, hyperoside, scoparone and other flavonoids might be the main activity compounds on α-glucosidase inhibitory activity.
No significantly negative correlations were obtained among antioxidant activities and the four compounds, including luteolin, luteolin 7-O-glucouronid, chrysoeriol and chrysoeriol 7-O-glucuronide, suggesting those four flavonoids might make little contributions to the antioxidant activities in hulless barley. The similar results were perceived in our previous study (Deng et al., 2021). In scavenging the DPPH radicals, earlier reports found that chrysoeriol was inactive, and chrysoeriol glycoside also exhibited not very efficient (Mishra et al., 2003). And luteolin 7-O-β-glucuronide was reported not relatively effective radical scavenger (Özgen et.al., 2011). Although luteolin and its derivatives feature a conjugated system, their antioxidant activity is generally lower compared to other flavonoids, particularly flavonols. This reduced activity is attributed to the absence of the 3-OH group, which eliminates a potential active site (Charlton et al., 2023).
Significantly positive correlations were found between luteolin, luteolin 7-O-glucouronid, chrysoriol, chrysoeriol 7-O-glucouronid. Luteolin, commonly named 3′,4′,5,7-tetrahydroxyflavone, is a flavone-based polyphenol naturally found as glycosides, with a phenylpropanoid biosynthesis route (Bangar et al., 2023). As a 30-O-methoxy flavone, chrysoeriol is a chemically derived product of luteolin (Aboulaghras et al., 2022).
4 Conclusion
By employing UPLC-QTOF-MS/MS, fifty-two constituents comprised 19 anthocyanidins and 32 flavonoids in black hulless barley bran fractions were characterized and identified. Among them, luteolin, luteolin 7-O-glucouronid, chrysoeriol and chrysoeriol 7-O-glucouronid were quantified by using UPLC-DAD, and they were all not detected in E20%, and higher content in E80% than that of in E40%. Another, the stronger antioxidant activity was obtained under higher TPC and TFC content, indicating that phenolics and flavonoids might be the key antioxidant compounds in black hulless barley bran. However, there were no significant correlation between TAC, TPC, TFC and α-glucosidase inhibitory activity. The current study could give improtant references for the utilization of black hulless barley bran, particularly for their consumption of anthocyanins and flavonoids of black barley bran.
Funding
This study was supported by the 1 + 9 unveiling of for tackling key scientific and technological problems - Functional food core technology, Sichuan Academy of Agricultural Sciences (1 + 9KJGG007); Cooperation project between people’s Government of Ganzi Tibetan Autonomous Prefecture and Sichuan Academy of Agricultural Sciences (2021), China; Innovation Team Project of Sichuan Province Authentic Chinese Medicine (Grant number SCCXTD-2020-19).
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Health benefits and pharmacological aspects of chrysoeriol. Pharmaceuticals. 2022;15(8):973.
- [CrossRef] [Google Scholar]
- In vitro potential of ascophyllum nodosum phenolic antioxidant-mediated α-glucosidase and α-amylase inhibition. J. Food Sci.. 2010;75(3):H97-H102.
- [CrossRef] [Google Scholar]
- Potential of cranberry-based herbal synergies for diabetes and hypertension management. Asia Pac. J. Clin. Nutr.. 2006;15:433-441.
- [CrossRef] [Google Scholar]
- Isolation and identification of polyphenols from apple pomace and their antioxidant and anti-inflammatory characters in vitro [D]. Northwest A&F University; 2014. p. :32.
- Luteolin: A flavone with myriads of bioactivities and food applications. Food Biosci.. 2023;52:102366
- [CrossRef] [Google Scholar]
- Cocoa brew inhibits in vitro α-glucosidase activity: The role of polyphenols and high molecular weight compounds. Food Res. Int.. 2014;64:439-445.
- [CrossRef] [Google Scholar]
- Boro, A., Shanmugam, R., Latha, A.S., Rajan, A.P., Al-Dhabi, N.A., Mariadhas, V.A., Arumugam, V.A., Balasubramanian, B., 2023. Chrysoeriol: Derivatives, Resources, Biosynthetic Pathway, Bioavailability, and Bioactivities. In: Xiao, J. (eds) Handbook of Dietary Flavonoids. Springer, Cham. https://doi.org/10.1007/978-3-030-94753-8_16-1.
- Structural features of small molecule antioxidants and strategic modifications to improve potential bioactivity. Molecules. 2023;28(3):1057.
- [CrossRef] [Google Scholar]
- The identification of flavonoids and the expression of genes of anthocyanin biosynthesis in the chrysanthemum flowers. Biol. Plantarum. 2012;56(3):458-464.
- [CrossRef] [Google Scholar]
- Anthocyanins – nature, occurrence and dietary burden. J. Sci. Food Agri.. 2000;80:1063-1072.
- [Google Scholar]
- Deng, J.L., Xiang, Z.Y., Xia, C., Lin, C.B., Zhu, Y.q., Yang, K.J., Liu, T.H., Xia, C., Chen, J., Zhang, W.H., Zhang, Y.H., & Zhu, B.Y. (2021). Identification and quantification of free, esterified, and insoluble-bound phenolics in grains of hulless barley varieties and their antioxidant activities. LWT-Food Sci. Technol. 151, 112001. https://doi.org/10.1016/j.lwt.2021.112001.
- Flavonoid glucuronides and a chromone from the aquatic macrophyte Stratiotes aloides[J] J. Nat. Prod.. 2009;72(5):835-840.
- [Google Scholar]
- Comparative study of antioxidant properties and total phenolic content of 30 plant extracts of industrial interest using DPPH, ABTS, FRAP, SOD, and ORAC assays. J. Agric. Food Chem.. 2009;57:1768-1774.
- [CrossRef] [Google Scholar]
- Identification and quantification of phenolic compounds from pomegranate (Punica granatum L.) peel, mesocarp, aril and differently produced juices by HPLC-DAD-ESI/MS n. Food Chem.. 2011;127(2):807-821.
- [CrossRef] [Google Scholar]
- Cyanidin 3-O-(6″-Succinyl- β -glucopyranoside) and other anthocyanins from phragmites australis. Phytochemistry. 1998;49(4):1065-1068.
- [CrossRef] [Google Scholar]
- The phenolic compounds profile, quantitative analysis and antioxidant activity of four naked barley grains with different color. Food Chem.. 2020;335:127655
- [CrossRef] [Google Scholar]
- Understanding the nutrient composition and nutritional functions of highland barley (Qingke): A review. Trends Food Sci. Tech.. 2020;103:109-117.
- [CrossRef] [Google Scholar]
- Identification of anthocyanins in black rice (Oryza sativa L.) by UPLC/Q-TOF-MS and their in vitro and in vivo antioxidant activities. J. Cereal Sci.. 2015;64:92-99.
- [CrossRef] [Google Scholar]
- Identification of anthocyanins isolated from black rice (Oryza sativa L.) and their degradation kinetics. Food Res. Int.. 2013;50(2):691-697.
- [CrossRef] [Google Scholar]
- Unveiling the anticancer, antimicrobial, antioxidative properties, and UPLC-ESI-QTOF-MS/GC-MS metabolite profile of the lipophilic extract of siam weed (Chromolaena odorata) Arab. J. Chem.. 2023;16(7):104834
- [CrossRef] [Google Scholar]
- Bioactive phytochemicals in barley. J. Food Drug Anal.. 2017;25:148-161.
- [CrossRef] [Google Scholar]
- Barley: chemistry and value-added processing. Crit. Rev. Food Sci. Nutr.. 1998;38:123-171.
- [CrossRef] [Google Scholar]
- Multifarious biological applications and toxic Hg2+ sensing potentiality of biogenic silver nanoparticles based on securidaca inappendiculata hassk stem extract. Int. J. Nanomed.. 2021;16:7557-7574.
- [CrossRef] [Google Scholar]
- Kan, L., Capuano, E., Fogliano, V., Verkerk, R., Mes J.J., Tomassen, M.M.M., Oliviero, T., 2021. Corrigendum to 'Inhibition of α-glucosidases by tea polyphenols in rat intestinal extract and Caco-2 cells grown on Transwell'. Food Chem. 361, Artical 130047. https://doi.org/10.1016/j.foodchem.2021.130649.
- Bio-antioxidants - A chemical base of their antioxidant activity and beneficial effect on human health. Curr. Med. Chem.. 2013;20:4784-4805.
- [CrossRef] [Google Scholar]
- Anthocyanins-more than naturé s colours. J. Biomed. Biotechnol.. 2004;5:239-240.
- [CrossRef] [Google Scholar]
- Identification and characterization of phenolics and terpenoids from ethanolic extracts of Phyllanthus species by HPLC-ESI-QTOF-MS/MS. J. Pharm. Anal.. 2017;007(004):214-222.
- [CrossRef] [Google Scholar]
- Whole grain phytochemicals and health. J. Cereal Sci.. 2007;46(3):207-219.
- [CrossRef] [Google Scholar]
- Bioactive compounds and antioxidant activity of wheat Bran and barley husk in the extracts with different polarity. Int. J. Food Prop.. 2019;22:646-658.
- [CrossRef] [Google Scholar]
- Toward the prediction of the activity of antioxidants: experimental and theoretical study of the gas-phase acidities of flavonoids. J. Am. Soc. Mass Spectrom.. 2004;15(6):848-861.
- [CrossRef] [Google Scholar]
- Effect of O-glycosilation on the antioxidant activity and free radical reactions of a plant flavonoid, chrysoeriol. Bioorg. Med. Chem.. 2003;11(13):2677-2685.
- [CrossRef] [Google Scholar]
- Influence of non-starchy polysaccharides on barley milling behavior and evaluatingbioactive composition of milled fractions. Food Chem.. 2017;218:137-143.
- [CrossRef] [Google Scholar]
- Bioavailability of pelargonidin-3-O-glucoside and its metabolites in humans following the ingestion of strawberries with and without cream. J. Agri. Food Chem.. 2008;56(3):713-719.
- [CrossRef] [Google Scholar]
- Identification of Cyanidin 3-O-(3″,6″–Dimalonyl–glucopyranoside) as a flower pigment of Chrysanthemum. Biosci. Biotech. Biochem.. 1997;61(9):1607-1608.
- [CrossRef] [Google Scholar]
- Ferulic acid fluorescence intensity profiles and concentration measured by HPLC in pigmented and non-pigmented cereals. Food Res. Int.. 2013;52:109-118.
- [CrossRef] [Google Scholar]
- α-glucosidase inhibitory activity and quantitative contribution of phenolic compounds from Vietnamese Aquilaria crassna leaves. Nat. Prod. Commun.. 2022;17(3):36-40.
- [CrossRef] [Google Scholar]
- Ground-based measurements and modeling of solar UV-B radiation in Lhasa, Tibet. Atmos. Environ.. 2009;43:1498-1502.
- [CrossRef] [Google Scholar]
- New insights on Acanthus ebracteatus Vahl: UPLC-ESI-QTOF-MS profile, antioxidant, antimicrobial and anticancer activities. Molecules. 2022;27(6):1981.
- [CrossRef] [Google Scholar]
- Relationship between chemical structure and antioxidant activity of luteolin and its glycosides isolated from Thymus sipyleus subsp. sipyleus var. sipyleus. Rec. Nat. Prod.. 2011;5(1):12-21.
- [CrossRef] [Google Scholar]
- Preparative separation of isovitexin and isoorientin from Patrinia villosa Juss by high-speed counter-current chromatography. J. Chromatog. A. 2005;1074(1–2):111-115.
- [CrossRef] [Google Scholar]
- Petar, D.M., Nigel, C.V., Renée, J.G., Geoffrey, C.K., Marina, S., Pedja, J., 2007. Flavonoids from Phlomis fruticosa (Lamiaceae) growing in Montenegro. Biochem. Syst. Ecol. 35(7), https://doi.org/462-466.10.1016/j.bse.2007.01.001.
- Structure-antioxidant activity relationships of flavonoids and phenolic acids. Free Radic. Bio. Med.. 1996;20(7):933-956.
- [CrossRef] [Google Scholar]
- Polyphenols:antioxidants and beyond. Am. J. Clin. Nutr.. 2005;81(1):215S-217S.
- [CrossRef] [Google Scholar]
- Distribution of β-glucan, phenolic acids, and proteins as functional phytonutrients of hull-less barley grain. Foods. 2019;8(12):680.
- [CrossRef] [Google Scholar]
- Effects of beta glucan in highland barley on blood glucose and serum lipid in high fat-induced C57 mouse. Chinese J. Disease Control Prevent.. 2013;47(1):55-58.
- [Google Scholar]
- Functional properties of anthocyanins and betalains in plants, food, and in human nutrition. Trends Food Sci. Tech.. 2004;15(1):19-38.
- [CrossRef] [Google Scholar]
- Concentrations of phenolic acids, flavonoids and carotenoids and the antioxidant activity of the grain, flour and bran of Triticum polonicum as compared with three cultivated wheat species. Agriculture. 2020;10(12):591.
- [CrossRef] [Google Scholar]
- Phenolic acids profile, nutritional and phytochemical compounds, antioxidant properties in colored barley grown in southern Italy. Food Res. Int.. 2018;113:221-233.
- [CrossRef] [Google Scholar]
- Malonylated anthocyanidin 3,5-diglucosides in the flowers of the genus Disa (Orchidaceae) Biochem. Syst. Ecol.. 2011;39(3):220-224.
- [CrossRef] [Google Scholar]
- Effects of beta glucan in highland barley on blood glucose and serum lipid in high fat-induced C57 mouse. Chin. J. Prev. Med.. 2013;47(1):55-58.
- [Google Scholar]
- Metabolomics based inferences to unravel phenolic compound diversity in cereals and its implications for human gut health. Trends Food Sci. Tech.. 2022;127:14-25.
- [CrossRef] [Google Scholar]
- Phenolic compounds and the antioxidant activity of the bran, flour and whole grain of different wheat varieties. Procedia Chem.. 2010;2:76-82.
- [CrossRef] [Google Scholar]
- Phenolic compounds of cereals and their antioxidant capacity. Crit. Rev. Food Sci. Nutr.. 2016;56(1):25-35.
- [CrossRef] [Google Scholar]
- Characterization of phenolic compounds in green and red oak-leaf lettuce cultivars by UHPLC-DAD-ESI-QToF/MS using MSE scan mode. J. Mass Spectrom.. 2017;52(12)
- [CrossRef] [Google Scholar]
- Inhibition effect on α-glucosidase and antioxidant activity for polyphenol extracts from Phyllanthus emblica L. Food Res. Dev.. 2017;16(16):741.
- [Google Scholar]
- Luteolin ameliorates LPS-induced acute liver injury by inhibiting TXNIP-NLRP3 inflammasome in mice. Phytomedicine. 2021;87:153586
- [CrossRef] [Google Scholar]
- Characterization of milling fractions from new sources of barley for use in food industry. Starch-Stärke. 2016;68(3–4):321-328.
- [CrossRef] [Google Scholar]
- Inhibitory effects of onion against α-glucosidase activity and its correlation with phenolic antioxidants. Int. J. Food Prop.. 2014;17(3):599-609.
- [CrossRef] [Google Scholar]
- Distribution of free and bound phenolic compounds, β-glucan, and araboxylan in fractions of milled hulless barley. LWT-Food. Sci. Technol.. 2022;169
- [CrossRef] [Google Scholar]
- Effect of processing on the release of phenolic compounds and antioxidant activity during in vitro digestion of hulless barley. Arab. J. Chem.. 2021;14:103447
- [CrossRef] [Google Scholar]
- Integrative metabolomic and transcriptomic analyses reveal the mechanisms of Tibetan hulless barley grain coloration. Front. Plant Sci.. 2022;13
- [Google Scholar]
- Yang, J.C., Wang, X.L., Zhang, C.Y., Ma, L., Wei, T., Zhao, Y.J., Peng, X., 2021. Comparative study of inhibition mechanisms of structurally different flavonoid compounds on α-glucosidase and synergistic effect with acarbose. Food Chem. 347, 129056.1-129056.7. https://doi.org/10.1016/j.foodchem.2021.129056.
- Yang Y., Fan B., Mu Y.W., Li Y., Tong L.T., Wang L.L., Liu L.Y., Li M.M., Sun P.P., Sun J., Wang F.Z., 2023. A comparative metabolomics study of polyphenols in highland barley (Hordeum vulgare L.) grains with different colors. Food Res. Int. 2023, 174: 113672. https://doi.org/10.1016/j.foodres.2023.113672.
- Evaluation of the intracellular lipid-lowering effect of polyphenols extract from highland barley in HepG2 cells. Food Sci. Hum. Well. 2022:1-14.
- [CrossRef] [Google Scholar]
- Ye, C.Y., Zhang, R.F., Dong, L.M., Chi, J.W., Huang, F., Dong, L.H., Zhang, M.W., Jia, X.C., 2022. α-Glucosidase inhibitors from brown rice bound phenolics extracts (BRBPE): Identification and mechanism. Food Chem. 372, Artical 131306.1-131306.9. https://doi.org/10.1016/j.foodchem.2021.131306.
- Variation of the Polyphenolic Composition and Antioxidant Capacity of Freshly Prepared Pomegranate Leaf Infusions over One-Day Storage. Antioxidants. 2021;10:1187.
- [CrossRef] [Google Scholar]
- A review of ultrasound-assisted extraction for plant bioactive compounds: Phenolics, flavonoids, thymols, saponins and proteins. Food Res. Int.. 2022;157:111268
- [CrossRef] [Google Scholar]
- Genome-wide dissection of co-selected UV-B responsive pathways in the UV-B adaptation of qingke. Mol. Plant. 2019
- [CrossRef] [Google Scholar]
- Assessment of β-glucans, phenols, flavor and volatile profiles of hulless barley wine originating from highland areas of China. Food Chem.. 2019;293:32-40.
- [CrossRef] [Google Scholar]
- The effect of coelitin on inflammatory factors in acute gout arthritis mice and its immune mechanism. Immunol. J.. 2021;37(10):875-881.
- [Google Scholar]
- Distribution and molecular characterization of beta-glucans from hull-less barley bran, shorts and flour. Int. J. Mol. Sci.. 2011;12:1563-1574.
- [CrossRef] [Google Scholar]
Appendix A
Supplementary material
Supplementary data to this article can be found online at https://doi.org/10.1016/j.arabjc.2024.105644.
Appendix A
Supplementary material
The following are the Supplementary data to this article:Supplementary Data 1
Supplementary Data 1







