Translate this page into:
Study on establishment of residual analysis methods and residue degradation regulation of natural pyrethrin in barley plants and soil on Qinghai Plateau
⁎Corresponding authors. hongyuchenred@163.com (Hongyu Chen), lwbabylw@163.com (Wei Li)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Abstract
The purpose of this study is to develop an accurate and efficient assay for the detection of pyrethroid residues and degradation in barley and soil in the Qinghai Plateau region. A sensitive and selective method was developed for the determination of pyrethrin residues in highland barley (kernel, stem, and root) and soil in the Qinghai Plateau by liquid chromatography (LC-MS/MS). Multi-residue analysis of pyrethrin in samples involves simple extraction with acetonitrile, salt-out with NaCl, MgSO4 absorption of water, cleaning with GCB and PSA, respectively, followed by separation with XR-ODS II column, and multi-reaction detection mode detection using a positive mode electrospray ionization source (ESI + ). The results showed that the 6 different components of pyrethrin had a good detection effect in the concentration range of 0.01 ∼ 1.0 mg/L, and the correlation coefficient was greater than 0.9990. The limits of quantitation of each component on each substrate were 0.01 ∼ 0.05 mg/kg, the average daily recoveries were 87.58 ∼ 95.44 %, and the relative standard deviations were 0.4 ∼ 3.8 %. The half-life of pyrethrin in barley grains, stems and soil was 0.77 ∼ 2.57 d. In soil and highland barley, the maximum dose of pyrethrin and twice the maximum dose of pyrethrin were applied, and the final residue 7 days after treatment was less than 0.2 mg/kg. The degradation behavior of pyrethrin in soil at three test sites showed that organic carbon and pH were important factors affecting the degradation of pyrethrin, and the effect of pH on the degradation of six pyrethrin components was greater than that of organic carbon. These studies provide theoretical basis for environmental safety evaluation caused by pyrethrin use in the Qinghai Plateau.
Keywords
Highland barley
Pyrethrins
The degradation dynamics rhythm
Terminal residue
Soil degradation
1 Introduction
Highland barley(Hordeum vulgare L. var. nudum Hook.f.), the smallest staple grain and the largest coarse grain in China, mainly grows on the Qinghai-Tibet Plateau and attracts much attention for its unique nutritional value (Guo et al., 2020; Mengqian et al., 2020). In recent years, with the continuous improvement of living standards and the continuous evolution of the disease spectrum, people's demand for nutritious and healthy food has become more and more urgent. Highland barley has become a cash crop with broad application prospects (Yang et al., 2010; Dai et al., 2014; Zeng et al., 2018). It was found that highland barley has the potential to lower blood sugar, reduce blood lipids, and improve metabolic syndrome due to functional active substances such as β-glucan and polyphenols (Mengqian et al., 2020). Highland barley planting consumption demand increases, and planting highland barley can harvest higher economic benefits (Lyu et al., 2022). In the growth, grouting and ripening process of highland barley, there will be diseases and pests, which are caused by many aspects, such as planting techniques, cultivation conditions and the climate of the living environment (Liu et al., 2012). In recent years, an increasing number of pesticides have been used in Highland barley production to prevent and control army worm, aphids, plasma suckers, etc. (Deng et al., 2018). Therefore, the presence of pesticide residues in Highland barley poses a potential threat to the health of those who consume it, especially if pesticides are used improperly or unavoidably to protect against pests and decay during cultivation (Zeng et al., 2015; Obadi et al., 2021). Pyrethrins, because they are fast-acting and toxic to insects at low doses and have a lethal effect, have been used more and more widely in Highland barley production to prevent and control numerous insect pests (Zhu et al., 2017).
Pyrethrins comprise primarily of insecticidal compounds extracted from the blooms of the pyrethrum plant. It is composed of 6 components, including pyrethrin I, pyrethrin II, cinerin I, cinerin II, jasmolin I and jasmolin II (Fig. 1, Table 1). Pyrethrins pose a potential health risk to humans, as they can enter the body through various means such as skin contact, inhalation, and ingestion, depending on the type of food. It is important to note that they are not entirely harmless (Chrustek et al., 2018). Most pyrethroids have different isomers, each with different biological activity and therefore different toxicity, they are highly hydrophobic, photosensitive, easily hydrolyzed, and degrade in less than 60 days (Aznar-Alemany and Eljarrat 2020). The mechanism of action of pyrethrins involves regulating the outflow of sodium ions from nerve cells, resulting in continuous nerve stimulation, repeated opening of sensory nerve fibers and motor nerve axes, and eventually leading to paralysis and death of pests, which is a typical nerve agent (Norris and Bloomquist, 2022). pyrethrins pose little danger to the human digestive system, through which they enter the body and are hydrolyzed and destroyed in the stomachs of most mammals (Bradberry et al., 2005). However, certain doses of pyrethroids can affect human health, pyrethroid exposure and oxidative stress can damage male sperm quality, and pyrethroid exposure during pregnancy and lactation can affect the reproductive system of F1 offspring (Zhang et al., 2021). Pyrethrins is genotoxic and cytotoxic to cultured human lymphocytes. This toxicity might be related to the induction of oxidative stress inside the cells (Azab et al., 2017). This natural pyrethrin mixture has been used safely worldwide for the past 160 years as an important plant insecticide, with lethal effects due to its fast action, low dose and toxic effect on insects (Sun et al., 2020). Lu et al. detected pyrethrins residues in tea by using ultra high-performance liquid chromatography-tandem mass spectrometry (Lu et al., 2010). Feng et al. tested the residue of pyrethrins in radish by using gas chromatography-mass spectrometry and concluded that the degradation of pyrethrins in greenhouse was faster than that in open air (Feng et al., 2018). Petrarca et al. determined the residue of pyrethrins in baby food by liquid chromatography-triple quadrupole mass spectrometry (Petrarca et al., 2017). Pan et al. conducted digestion and residue of pyrethrins in leaf lettuce under greenhouse and open field conditions. Using gas chromatography-mass spectrometry, they concluded that the pyrethrins disappearance half-life of field lettuce was less than that of greenhouse lettuce, which might be related to rainfall in an open environment (Pan et al., 2017). Ruiz et al. verified the quantitative analysis method of pyrethrins on lemon and apricot by using high performance liquid chromatography-mass spectrometry (Ruiz et al., 2011). Rawn et al. used quechers method to analyze pyrethrins and pyrethroids in fish tissues, and gas chromatogen-mass spectrometry was used (Rawn et al., 2010). Lorsirigool et al. researched the use and abuse of pyrethrins and pyrethroids in veterinary medicine (Lorsirigool et al., 2022). Amjad et al. carried out the residue of pyrethrins in okra and eggplant, and used high performance liquid chromatography to detect it (Amjad et al., 2020). Xu et al. extracted pyrethroid pesticide residues from tea by QuEChERS method using multi-wall carbon nanotubes (MWCNTs), analyzed and determined them by gas chromatogre-series mass spectrometry (GC–MS/MS), and established a highly sensitive and reliable method for the detection of pesticide residues in tea (Ruihan et al., 2022).
Nowadays, most of the pyrethroid residue detection methods are based on gas chromatography with electron capture detection and Liquid Chromatography–UV Detection method (Wongsa and Burakham 2012; Zhang et al., 2016). Highly efficient proportional fluorescent probes based on bis-dihydroxyboron fluorescein complexes for the detection of pyrethroid residues in fruit juices are characterised by complexity and high cost (Norris and Bloomquist, 2022). And most of the testing for pyrethroid residues is now on vegetables, tea and processed agricultural products (Hou et al., 2019; Xu et al., 2022). As far as we know, the analysis method and resolution of pyrethrins residues in barley have not been reported. The aim of this study was to provide the detection method and digestion dynamics of pyrethrin in Qinghai plateau barley. In view of this, a QuEChERS-LC-MS/MS method was developed for the rapid determination of pyrethrin residues in highland barley and soil. It will provide technical support for the safety of highland barley and provide methodological reference for the determination of other types of pesticides in the soil environment. The residual degradation rule of pyrethrins in highland barley and soil was obtained to provide data support for the safety and risk evaluation of pyrethrins in highland barley and soil.
2 Materials and methods
2.1 Materials and reagents
Methanol, acetonitrile, acetic acid, formic acid, ammonium formate, ammonium acetate (chromatographic grade, Merck, Darmstadt, Germany); anhydrous magnesium sulfate, sodium chloride and ammonia (analytical grade, Sinopharm Chemical Reagent Co., Ltd., Shanghai, China); ethylenediamine-N-propyl silane (PSA), graphitized carbon black (GCB), Florisil and octadecyl bonded silica gel (C18) (Angela Technologies, Tianjin, China). Pyrethrins standard (purity, 77.1 %, Beijing Qinchengyixin Technology, Beijing, China); 1.5 % pyrethrins aqueous emulsion (Inner Mongolia Qingyuanbao Biotechnology Co., Ltd., Bayannur, China).
2.2 Field experiments
Field experiments were designed according to the requirements stated in the Guidelines for Pesticide Residue Tests and the Standard Operating Procedures for Pesticide Registration Residue Field Tests (inspection 2016, China 2018).The study including a dissipation dynamic experiment and a final residue experiment was conducted at the experimental field, Institute of Plant Protection, Qinghai Academy of Agriculture and Forestry Sciences in the year 2021.
Dissipation experiments: The design of the field experiment for dissipation is shown in Table 2. There were seven treatments including six pyrethrin treatments and one control treatment. Each treatment consisted of three replicate plots and each plot was 25 m2. In the control treatment, no pesticide was used but the same amount of water was sprayed throughout the period. The plots of different treatments were separated by a buffer area of 25 m2. For the purpose of evaluating the dissipation of pyrethrins in whole plants, 1.5 % pyrethrin EW was dissolved in water and applied at active constituent 27 g a.i./ha and 54 g a.i./ha on the surface of plants. Dissipation experiments in soil: Since the plants of highland barley are higher and denser in the middle growth stage, stalk and leaf spray is adopted, and the original deposition amount in the soil is very low. Therefore, this experiment adopts the blank land without plant cover near the highland barley field to conduct the dynamic test of pyrethrins soil residue digestion. Each plot was 8 m2 and 1.5 % pyrethrin EW was dissolved in water and applied at active constituent 54 g a.i./ha on the surface of soil. Whole plants(about 1 kg) and soil (0–10 cm depth, 1–2 kg) were randomly collected at 2 h, 1, 2, 3, 5, 7, 10, 14, 21, and 28 days after application.
Residue experiments: two application rates of 27 g a.i./hm2 (recommended rate) and 54 g a.i./hm2 (twice the recommended rate) were set; experiments were conducted in accordance with the Guidelines for Testing Pesticide Residues in Agricultural Crops (NY/T788-2004)(China, 2004). Each treatment was applied 1, 2 and 3 times, with 3 replicates for each treatment. Each plot was 25 m2 in highland barley plant and the sampling intervals were 1 d, 3 d and 5 d from the last application. Each plot was 8 m2 in soil and the sampling intervals were 3 d, 5 d and 7 d from the last application. There was a blank control and a protection zone between treatments.
2.3 LC-MS/MS conditions
LC-MS/MS analysis was conducted with an 8040 Shimadzu, Kyoto, Japan LC system.Mass spectrometry conditions: Ion source: EIS+; Scanning mode: positive ion mode; the atomization gas flow rate was 3.0 L/min; the drying gas flow rate was 15 L/min; the DL temperature was 250 °C; the heating block temperature was 450 °C. Precursor ion scanning and product ion scanning were performed to obtain the mass charge ratio of precursor ion and product ion, and then quantitative ion and qualitative ion were obtained for qualitative and quantitative analysis of pyrethrins. By optimizing the mass spectrum parameters such as spray voltage and collision energy, the peak pattern, sensitivity and separation degree of the six analytes were optimized.
Chromatographic conditions: the analytical column was a Shim-pack XR-ODSⅡ(75 mm × 2.0 mm, i. d., 2.2 µm); the flow rate was 0.3 mL/min; the injection volume was 3 μL; the column temperature was 40 °C; the mobile phase included A as acetonitrile and B as 0.1 % formic acid aqueous solution; the mobile phase gradient elution was: 0 ∼ 10 min, 40 ∼ 20 % B; 10 ∼ 10.1 min, 20 ∼ 0 % B; 10.1 ∼ 12 min, 0 % B; 12 ∼ 12.1 min, 0 ∼ 40 % B; 12.1 ∼ 16 min, 40 % B.
2.4 Sample pre-treatment
Soil: 10.0 g soil sample was added with 10 mL acetonitrile, vortex extraction for 3 min, 1 g NaCl was added, vortex for 1 min, centrifuge for 5 min at 5000 r/min, 1.5 mL supernatant, 50 mg MgSO4, 100 mg PSA, vortex for 1 min, Centrifuge at 12,000 r/min for 2 min, filter the membrane by 0.22 μm, and load the machine to be tested.
Grain: 5.0 g grains sample was added with 10 mL acetonitrile, vortexized for 3 min, 1 g NaCl was added for 1 min, vortex for 5 min at 5000 r/min, 1.5 mL supernatant, 50 mg MgSO4, 20 mg GCB, vortex for 1 min, Centrifuge at 12,000 r/min for 2 min, filter the membrane by 0.22 μm, and load the machine to be tested.
Stem: 2.0 g stems sample was added with 10 mL acetonitrile, vortexized for 3 min, 1 g NaCl was added for 1 min, vortex for 5 min at 5000 r/min, 1.5 mL supernatant, 50 mg MgSO4, 20 mg GCB, vortex for 1 min, Centrifuge at 12,000 r/min for 2 min, filter the membrane by 0.22 μm, and load the machine to be tested.
Root: 5.0 g roots sample was added with 10 mL acetonitrile, vortex extraction for 3 min, 1 g NaCl was added, vortex for 1 min, centrifuge for 5 min at 5000 r/min, 1.5 mL supernatant, 50 mg MgSO4, 100 mg PSA, vortex for 1 min, Centrifuge at 12,000 r/min for 2 min, filter the membrane by 0.22 μm, and load the machine to be tested.
2.5 Standards
A standard stock solution of pyrethrins (100 mg/L) was prepared in acetonitrile. The standard solutions required for constructing a calibration graph (0.01,0.02,0.05, 0.1, 0.5 and 1 mg/L) were prepared from the stock solution by serial dilution with acetonitrile.
2.6 Effects of different soils on degradation
The soil in the three experimental plots (Jintan Township in Haibei; Xiangrid in Haixi; Ledu in Haidong) were treated with sterilization (121 ℃、1.1 kg/cm2)and non-sterilization respectively. Adjust the relative water content of soil to 8 ∼ 10 %0.1 kg of soil was set for each treatment, and 1.5 % pyrethrins aqueous emulsion was added. The addition concentration was set at 2 mg/kg. It was cultured in a dark incubator at a temperature of 25 ℃. Appropriate amount of water was added every 2 to 3 days to keep the soil moisture constant. Soil samples were taken for testing at 2 h, 1 d, 3 d, 5 d, 7 d, 10 d, 14 d, 21 d and 28 d after the addition of medicine.
3 Results and analysis
3.1 Optimization of chromatographic conditions
Mobile phase compositions (i.e., water–methanol and water–acetonitrile) and the concentration of formic acid, ammonium acetate, and a mixture of formic acid and ammonium acetate, were optimized to achieve maximal sensitivity. Results indicated that acetonitrile–water was used as mobile phase, pyrethrins 6 components showed better separation effect and mass spectrum response than methanol–water (Fig. 1). Therefore, acetonitrile is used as the organic phase in the mobile phase. The effects of 0.1 % formic acid, 2.0 mmol/L ammonium acetate and 2.0 mmol/L ammonium formate in aqueous phase on chromatographic separation and sensitivity were also compared. When 0.1 % formic acid is added, the ionization efficiency and sensitivity of each compound are most significant. Therefore, acetonitrile-0.1 % formic acid aqueous solution was used as mobile phase in this experiment.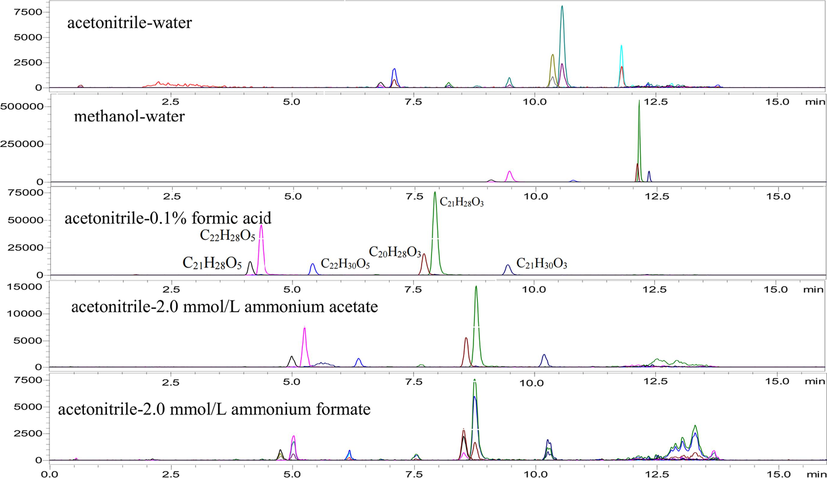
Organic phase and aqueous phase combination screening.
3.2 Optimization of mass spectrometry conditions
After optimizing the mass spectrum parameters, pyrethrin standard liquid (10.0 mg/L) was directly injected into the mass spectrum by liquid chromatography. The positive and negative ions were qualitatively ionized, and the cone-hole voltage and collision energy were optimized to obtain the optimal parent and daughter ions (the optimization results are shown in Table 1).
Compound
Retention time(min)
Ion source
Precursor (m/z)
Product (m/z)
Q1 Pre Bias (V)
CE
(eV)Q3 Pre Bias (V)
cinerin II
4.147
EIS+
361.2
107*
−10
−21
−10
77
−24
−54
−12
pyrethrin II
4.377
EIS+
373.2
161*
−26
−9
−16
jasmolin II
cinerin I
pyrethrin I
jasmolin I
5.468
7.769
7.997
9.509
EIS+
EIS+
EIS+
EIS+
375. 2
317.2
329.2
331.2133
163.1*
107
107*
77
161*
133
163.1*
107−14
−26
−14
–22
–22
–22
–22
−12
–22−20
−11
−27
−20
−53
−10
−19
−10
−24−24
−16
−16
−18
−12
−28
−12
−36
−38
3.3 Optimization of extractants
3.3.1 Selection of extractants for different matrices
Pyrethrins is easily soluble in organic solvent (methanol, acetone, acetonitrile), insoluble in water, but the use of acetone extraction, the final need for nitrogen drying, resulting in complex pre-treatment steps, so we choose methanol, acetonitrile, acetonitrile-formic acid mixed solution (99:1) as the extraction agent, soil, highland barley grains, stems and roots as the matrix to discuss. The effects of three extractants on the recovery of pyrethrins were compared. As can be seen from (Fig. 2), when a small amount of formic acid was added to acetonitrile, the recovery rate of each component was slightly lower than that of acetonitrile extraction. The average recoveries of acetonitrile formic acid and pure acetonitrile were 85.56 %∼92.11 % and 90.54 %∼94.34 %, respectively. When methanol was used as extraction agent, the recovery rate of each component was low, and when methanol was used to extract grains, the liquid phase of extraction was greener than that of acetonitrile extraction, which was easy to extract excessive pigment, resulting in difficulties in purification. Therefore, acetonitrile was selected as the solvent to extract pyrethrins from soil, barley grains, stems and roots.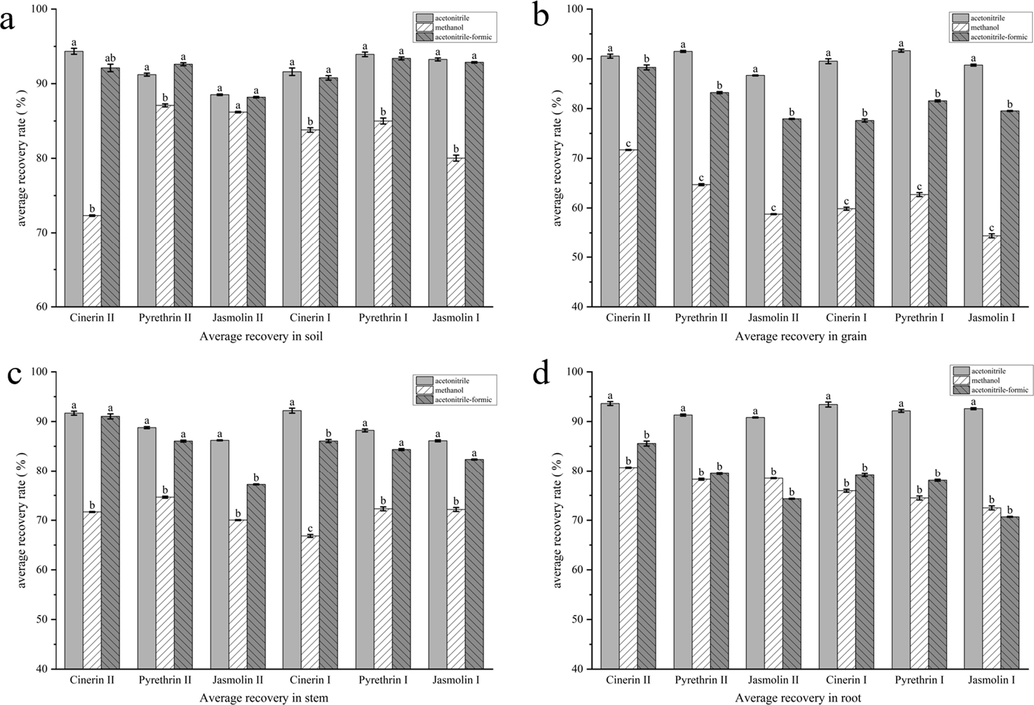
Recoveries of pyrethrins under different extractants.
3.3.2 Optimization of extractant dosage
Using highland barley grains as the substrate (Table 2), the effects of acetonitrile dosage on the recovery of pyrethrins in three volumes of extraction solvent (10 mL, 20 mL and 30 mL) were compared. We found that there was no significant difference in the recovery rate of pyrethrins components when the extraction solvent volume was 10 mL, 20 mL and 30 mL. When the volume of extraction solvent was 10 mL, the recovery of each component was 87.49 ∼ 91.81 %. When the volume of extraction solvent was 20 mL, the recoveries of each component were 85.65 ∼ 90.71 %. When the volume of extraction solvent was 30 mL, the recovery of each component was 85.35 ∼ 90.28 %. Therefore, 10 mL acetonitrile was selected as the extraction solvent in order to facilitate operation and save drugs.
Extractant dosage (mL)
Average recovery (%)
Cinerin II
Pyrethrin II
Jasmolin II
Cinerin I
Pyrethrin I
Jasmolin I
10
90.94
91.47
87.49
88.59
91.81
88.25
20
90.71
88.21
85.65
86.52
90.63
88.73
30
90.25
85.86
85.35
85.53
90.28
87.52
3.3.3 Selection of sorbent
Soil sample: The effects of anhydrous magnesium sulfate dosage of 50 mg, 100 mg and 150 mg on pyrethrins recovery were compared (Fig. 4). All the pyrethrins concentrations were 1.0 mg/kg. In the three MgSO4 levels, the average recoveries of cinerin II were 87.31 %∼89.20 %; the average recoveries of pyrethrin II were 89.78 %∼90.34 %; the average recoveries of jasmolin II were 92.45 %∼95.78 %; the average recoveries of cinerin I were 83.21 %∼84.57 %; the average recoveries of pyrethrin I were 89.01 %∼90.64 %; the average recoveries of jasmolin I were 87.67 %∼89.37 %. As the amount of anhydrous MgSO4 increased, the recovery rate of each component of pyrethrins showed no significant difference, saving the amount of anhydrous MgSO4. Therefore, 50 mg anhydrous MgSO4 was finally selected to absorb the water in the supernatant during purification.
Root sample: Effect of 20 mg, 50 mg, 100 mg, 150 mg, 200 mg PSA on pyrethrins recovery (Fig. 4). All the pyrethrins concentrations were 1.0 mg/kg. When PSA dosage increased from 20 mg to 100 mg, the recovery rate of each component of pyrethrins showed an upward trend, and the recovery rate basically stopped increasing as PSA dosage continued to increase. At 100 mg PSA level, the average recoveries were cinerin II 92.87 %, pyrethrin II 97.23 %, jasmolin II 95.33 %, cinerin I 96.13 %, pyrethrin I 96.22 %, respectively. jasmolin I 96.89 %. In conclusion, 50 mg anhydrous MgSO4 and 100 mg PSA were selected as purification agents for soil and highland barley root.
Grain sample: To compare the effects of different purifier C18, GCB, PSA and Florisil (dosage: 10, 20, 30, 50, 70 mg) on the impurity removal of barley grains, especially on the removal of pigment (Fig. 5). Under 5 levels of C18 purification, the average recoveries of cinerin II were 87.93 %∼90.65 %, and the recoveries of cinerin II with increased dosage of C18 had little difference; the average recoveries of pyrethrin II were 87.41 %∼89.82 %, and the recoveries of pyrethrin II with increased dosage of C18 had little difference; the average recoveries of jasmolin II were 86.35 %∼91.06 %, and the recoveries of jasmolin II with increased dosage of C18 had little difference; the average recoveries of cinerin I were 48.39 %∼80.01 %, when the dosage of C18 was increased to 70 mg, the recovery decreased to 48.39 %; the average recoveries of pyrethrin I were 83.28 %∼87.72 %, and the recoveries of pyrethrin I with increased dosage of C18 had little difference; the average recoveries of jasmolin I were 75.32 %∼85.83 %, when the dosage of C18 was increased to 70 mg, the recovery decreased to 75.32 %. Using C18 as purification agent, the pigment removal effect is not good, increasing the dosage still can’t effectively remove pigment. Under 5 levels of GCB purification, they have a good effect on chlorophyll removal, and the best recovery rate is 20 mg GCB, cinerin II was 95.33 %, pyrethrin II was 91.58 %, jasmolin II was 90.67 %, cinerin I was 90.92 %, pyrethrin I was 92.35 %, jasmolin I was 90.46 %, respectively. The recovery rate of pyrethrins components decreased slightly with increasing or decreasing the dosage of GCB. Under 5 levels of PSA purification, the average recoveries of cinerin II were 89.34 %∼90.57 %, and the recoveries of cinerin II with increased dosage of PSA had little difference; the average recoveries of pyrethrin II were 84.17 %∼86.98 %, and the recoveries of pyrethrin II with increased dosage of PSA had little difference; the average recoveries of jasmolin II were 78.63 %∼83.12 %, and the recoveries of jasmolin II with increased dosage of PSA had little difference; the average recoveries of cinerin I were 78.32 %∼84.02 %, and the recoveries of cinerin I with increased dosage of PSA had little difference; the average recoveries of pyrethrin I were 82.12 %∼ 87.31 %, and the recoveries of pyrethrin I with increased dosage of PSA had little difference; the average recoveries of jasmolin I were 80.22 %∼ 87.41 %, and the recoveries of jasmolin I with increased dosage of PSA had little difference. As a purifying agent with PSA, the effect of pigment removal is poor, and the increase in dosage still does not effectively remove the pigment. Under 5 levels of Florisil purification, the average recoveries of cinerin II were 79.09 %∼81.34 %, and the recoveries of cinerin II with increased dosage of Florisil had little difference; the average recoveries of pyrethrin II were 82.12 %∼84.16 %, and the recoveries of pyrethrin II with increased dosage of Florisil had little difference; the average recoveries of jasmolin II were 80.61 %∼84.17 %, and the recoveries of jasmolin II with increased dosage of Florisil had little difference; the average recoveries of cinerin I were 28.76 %∼85.03 %, and when the dosage of Florisil was increased to 70 mg, the recovery decreased to 28.76 %; the average recoveries of pyrethrin I were 84.17 %∼86.87 %, and the recoveries of pyrethrin I with increased dosage of Florisil had little difference; the average recoveries of jasmolin I were 69.42 %∼83.05 %, and when Florisil dosage was increased to 50 mg, the recovery rate increased to 83.05 %. Florisil as a purifier, the pigment removal effect is not good, increasing the dosage still can’t effectively remove pigment. Considering the protection of the instrument, pigment removal effect and recovery rate, GCB can purify the barley grains better.
Stem sample: According to the impurity removal effect and recovery rate of a single purification agent, the combination of PSA (50 mg, 100 mg, 150 mg) and GCB (10 mg, 20 mg, 30 mg) was selected in this part of the experiment to purify highland barley stem samples (Fig. 6). When GCB dosage was 10 mg, PSA dosage was increased from 50 mg to 150 mg, the average recoveries of cinerin II, pyrethrin II and jasmolin II were 76.74 %∼79.28 %, 76.12 %∼78.95 % and 72.12 %∼75.79 % respectively, and the average recoveries of cinerin I, pyrethrin I and jasmolin I were 76.97 %∼80.71 %, 76.94 %∼79.68 % and 78.74 %∼84.41 % respectively. Its recovery rate is lower than that of GCB alone. When GCB dosage was 20 mg, PSA dosage was increased from 50 mg to 150 mg, the average recoveries of cinerin II, pyrethrin II and jasmolin II were 77.32 %∼82.75 %, 72.85 %∼75.60 % and 68.27 %∼72.63 % respectively, and the average recoveries of cinerin I, pyrethrin I and jasmolin I were 65.23 %∼80.45 %, 75.66 %∼77.88 % and 77.88 %∼80.27 % respectively. When GCB dosage was 30 mg, PSA dosage was increased from 50 mg to 150 mg, the average recoveries of cinerin II, pyrethrin II and jasmolin II were 71.19 %∼75.42 %, 70.40 %∼72.92 % and 66.28 %∼73.19 % respectively, and the average recoveries of cinerin I, pyrethrin I and jasmolin I were 73.90 %∼79.03 %, 73.90 %∼76.49 % and 65.52 %∼79.23 % respectively. Its recovery rate is lower than that of 20 mg GCB alone. In conclusion, 20 mg GCB was selected to purify the grains and stems of highland barley in this experiment.
3.4 Evaluation of matrix effects
In the MRM model, the linear relationship is obtained according to the peak area of the target compound (Table 5). The linearity of the method was calculated by preparing the calibration curves of 6 components of pyrethrin in barley plants and soil substrate in the concentration range of 0.01 ∼ 1.0 mg/kg. The linearity of these compounds in the matrix met the requirement of residue analysis (R2 ≥ 0.999). In soil matrix, LOQ of cinerin II, pyrethrin II, jasmolin II, cinerin I, pyrethrin I and jasmolin I was 0.01 mg/kg. The LOQ of cinerin II, pyrethrin II, jasmolin II, cinerin I, pyrethrin I and jasmolin I was 0.02 mg/kg in barley grain and root matter. The LOQ of cinerin II, pyrethrin II, jasmolin II, cinerin I, pyrethrin I and jasmolin I in barley stem matrix was 0.05 mg/kg.
When a compound passes through the mass spectrometer, the matrix components may affect the ionization efficiency. The most likely reason may be due to the energy competition between the analyte and the matrix components. Therefore, the study of matrix effects caused by the matrix is of some value in this study. The ratio of the slope of the matrix standard curve equation to the solvent standard curve equation minus 1 is often used to evaluate matrix effects. Matrix effect is in the range of plus or minus 10 %. Matrix effect can be ignored because its influence on analyte is small. The matrix effect of cinerin II in highland barley root was −12.4 %, which showed stronger signal inhibition compared with soil, highland barley grain and stem. The matrix effect of pyrethrin II in soil and highland barley root was 29.6 % and 21 %, respectively, which showed stronger signal enhancement compared with barley grain and stem. The matrix effect of jasmolin II in soil and barley root was stronger than that in barley grain and stem, which were 28.6 % and 29.8 %, respectively. The matrix effect of cinerin I in soil and highland barley root was 38.1 % and 23.4 %, respectively, which showed stronger signal enhancement compared with barley grain and stem. The matrix effect of pyrethrin I in soil and highland barley root was 36.7 % and 38.3 %, respectively, which showed stronger signal enhancement compared with barley grain and stem. The matrix effect of jasmolin I in grain was −16.3 %, and compared with the other three substrates, the matrix inhibition effect was stronger. 3.5. Validation
This experiment uses the matrix matching standard solution external standard method for quantification, and the accuracy of the method can be expressed by the addition recovery or inter-day precisions. The recovery rate of pyrethrins in blank control area was tested by using soil, barley grain, barley stem and barley root. The soil was recovered at three supplemental levels of 0.01, 0.02 and 0.5 mg/kg, the barley grain and root were recovered at three supplemental levels of 0.02, 0.04 and 1.0 mg/kg, and the barley stem was recovered at three supplemental levels of 0.05, 0.1 and 1.0 mg/kg. The average recovery rate and relative standard deviation (RSD) were calculated for 5 replicates per spiked level. Precision is a prerequisite for determining accuracy, usually expressed by the relative standard deviation, commonly used to express the degree of deviation between the actual measured values obtained after multiple parallel tests on the same group of samples under the same detection method. Evaluate the precision of the method with repeatability and reproducibility (Table 6).
Table 6 shows detailed recovery rates for 3 day spiked samples, which were analyzed to assess inter-day precision. The inter-day recoveries of pyrethrins 6 components in soil were 89.69 %∼95.44 %, RSD ≤ 4.3 %. The inter-day recoveries of pyrethrins 6 components in barley grain were 87.58 %∼94.83 %, RSDS ≤ 3.6 %. The inter-day recovery rates of pyrethrins 6 components in barley stem were 87.72 %∼93.94 %, RSDS ≤ 3.6 %. The inter-day recoveries of pyrethrins 6 components in barley root were 89.71 %∼96.40 %, RSDS ≤ 5.7 %0.3.6. Application to Real Samples.
3.4.1 Dissipation dynamics of cinerin II in highland barley plants and soil
The dissipation curve of cinerin II in barley grain, stem and soil was studied (Fig. 8). The regression equation, correlation coefficient and half-life are shown in Table 7. The initial deposition concentrations of cinerin II in soil for first, second and third times were 0.689 mg/kg, 0.703 mg/kg and 0.751 mg/kg, respectively, and the half-lives of cinerin II were 1.45, 1.73 and 1.75 days, respectively. The residue of cinerin II decreased rapidly in soil from 0 to 3 days after administration, and then the decline rate slowed down. The initial deposition concentrations of cinerin II in grain for first, second and third times were 0.264 mg/kg, 0.271 mg/kg and 0.281 mg/kg, respectively, and the half-lives of cinerin II were 0.78, 0.80 and 0.79 days, respectively. The initial deposition concentrations of cinerin II in stem for first, second and third times were 0.180 mg/kg, 0.150 mg/kg and 0.230 mg/kg, respectively, and the half-life of three times of injection was 0.91 days. The stems are sprayed once and twice, no cinerin II was detected after 2 days, which was lower than the minimum detection limit of 0.05 mg/kg cinerin II in the stem.
3.4.2 Dissipation dynamics of pyrethrin II in highland barley plants and soil
The dissipation curve of pyrethrin II in barley grain, stem and soil was studied (Fig. 9). The regression equation, correlation coefficient and half-life are shown in Table 8. The initial deposition concentrations of pyrethrin II in soil for first, second and third times were 0.702 mg/kg, 0.750 mg/kg and 0.900 mg/kg, respectively, and the half-lives of pyrethrin II were 1.63, 2.24 and 2.55 days, respectively. The initial deposition concentrations of pyrethrin II in grain for first, second and third times were 0.282 mg/kg, 0.290 mg/kg and 0.310 mg/kg, respectively, and the half-lives of pyrethrin II were 0.77, 0.79 and 0.83 days, respectively. The initial deposition concentrations of pyrethrin II in stem for first, second and third times were 0.210 mg/kg, 0.230 mg/kg and 0.250 mg/kg, respectively, and the half-life of pyrethrin II were 0.97, 0.91 and 0.87 days, respectively.
3.4.3 Dissipation dynamics of jasmolin II in highland barley plants and soil
The dissipation curve of jasmolin II in barley grain, stem and soil was studied (Fig. 10). The regression equation, correlation coefficient and half-life are shown in Table 9. The initial deposition concentrations of jasmolin II in soil for first, second and third times were 0.498 mg/kg, 0.540 mg/kg and 0.621 mg/kg, respectively, and the half-lives of jasmolin II were 1.86, 2.04 and 2.40 days, respectively. The initial deposition concentrations of jasmolin II in grain for first, second and third times were 0.259 mg/kg, 0.267 mg/kg and 0.265 mg/kg, respectively, and the half-lives of jasmolin II were 0.84, 0.90 and 0.83 days, respectively. The initial deposition concentrations of jasmolin II in stem for first, second and third times were 0.150 mg/kg, 0.160 mg/kg and 0.210 mg/kg, respectively, and the half-life of three times of injection was 0.97 days. The stems are sprayed once and twice, no jasmolin II was detected after 2 days, which was lower than the minimum detection limit of 0.05 mg/kg jasmolin II in the stem.
3.4.4 Dissipation dynamics of cinerin I in highland barley plants and soil
The dissipation curve of cinerin I in barley grain, stem and soil was studied (Fig. 11). The regression equation, correlation coefficient and half-life are shown in Table 10. The initial deposition concentrations of cinerin I in soil for first, second and third times were 0.732 mg/kg, 0.840 mg/kg and 0.900 mg/kg, respectively, and the half-lives of cinerin I were 2.20, 2.22 and 2.57 days, respectively. The initial deposition concentrations of cinerin I in grain for first, second and third times were 0.257 mg/kg, 0.263 mg/kg and 0.266 mg/kg, respectively, and the half-lives of cinerin I were 0.85, 0.83 and 0.85 days, respectively. The initial deposition concentrations of cinerin I in stem for first, second and third times were 0.189 mg/kg, 0.208 mg/kg and 0.250 mg/kg, respectively, and the half-life of three times of injection was 0.86 days. The stems are sprayed once and twice, no cinerin I was detected after 2 days, which was lower than the minimum detection limit of 0.05 mg/kg cinerin I in the stem.
3.4.5 Dissipation dynamics of pyrethrin I in highland barley plants and soil
The dissipation curve of pyrethrin I in barley grain, stem and soil was studied (Fig. 12). The regression equation, correlation coefficient and half-life are shown in Table 11. The initial deposition concentrations of pyrethrin I in soil for first, second and third times were 0.767 mg/kg, 0.800 mg/kg and 0.871 mg/kg, respectively, and the half-lives of pyrethrin I were 1.83, 2.23 and 2.34 days, respectively. The initial deposition concentrations of pyrethrin I in grain for first, second and third times were 0.293 mg/kg, 0.314 mg/kg and 0.320 mg/kg, respectively, and the half-lives of pyrethrin I were 0.81, 0.79 and 0.81 days, respectively. The initial deposition concentrations of pyrethrin I in stem for first, second and third times were 0.250 mg/kg, 0.259 mg/kg and 0.271 mg/kg, respectively, and the half-life of pyrethrin I were 0.86, 0.85 and 0.87 days, respectively.
3.4.6 Dissipation dynamics of jasmolin I in highland barley plants and soil
The dissipation curve of jasmolin I in barley grain, stem and soil was studied (Fig. 13). The regression equation, correlation coefficient and half-life are shown in Table 12. The initial deposition concentrations of jasmolin I in soil for first, second and third times were 0.518 mg/kg, 0.570 mg/kg and 0.669 mg/kg, respectively, and the half-lives of jasmolin I were 2.18, 2.41 and 2.37 days, respectively. The initial deposition concentrations of jasmolin I in grain for first, second and third times were 0.261 mg/kg, 0.268 mg/kg and 0.270 mg/kg, respectively, and the half-lives of jasmolin I were 0.84, 0.82 and 0.83 days, respectively. The initial deposition concentrations of jasmolin I in stem for first, second and third times were 0.160 mg/kg, 0.179 mg/kg and 0.220 mg/kg, respectively, and the half-life of three times of injection was 0.94 days. The stems are sprayed once and twice, no jasmolin I was detected after 2 days, which was lower than the minimum detection limit of 0.05 mg/kg jasmolin I in the stem.
3.4.7 Terminal residues of pyrethrins in soil
The highest recommended dose of 1.5 % pyrethrins aqueous emulsion in soil (27 g a.i./ hm2) and twice recommended maximum dose (54 g a.i./hm2) were applied for 1 ∼ 3 times. The terminal residue results were shown in Table 13 after 3, 5 and 7 days after the last application.
Low concentration (27 g a.i./ hm2) spray 1 ∼ 3 times, 3 days after the last dose, the terminal residue of cinerin II was 0.093 mg/kg ∼ 0.101 mg/kg; the terminal residue of pyrethrin II was 0.095 mg/kg ∼ 0.110 mg/kg; the terminal residue of jasmolin II was 0.096 mg/kg ∼ 0.099 mg/kg; the terminal residue of cinerin I was 0.084 mg/kg ∼ 0.089 mg/kg; the terminal residue of pyrethrin I was 0.099 mg/kg ∼ 0.124 mg/kg; the terminal residue of jasmolin I was 0.087 mg/kg ∼ 0.094 mg/kg. 7 days after the last application, the terminal residue of 6 components of pyrethrins in soil was less than 0.01 mg/kg.
High concentration (54 g a.i./ hm2) spray 1 ∼ 3 times, 7 days after the last dose, the terminal residue of cinerin II was 0.028 mg/kg ∼ 0.063 mg/kg; the terminal residue of pyrethrin II was 0.045 mg/kg ∼ 0.181 mg/kg; the terminal residue of jasmolin II was 0.043 mg/kg ∼ 0.130 mg/kg; the terminal residue of cinerin I was 0.098 mg/kg ∼ 0.190 mg/kg; the terminal residue of pyrethrin I was 0.057 mg/kg ∼ 0.200 mg/kg; the terminal residue of jasmolin I was 0.065 mg/kg ∼ 0.150 mg/kg.
3.4.8 Terminal residues of pyrethrins in grain
The highest recommended dose of 1.5 % pyrethrins aqueous emulsion in grain (27 g a.i./ hm2) and twice recommended maximum dose (54 g a.i./hm2) were applied for 1 ∼ 3 times. The terminal residue results were shown in Table 14 4 after 1, 3 and 5 days after the last application
Low concentration (27 g a.i./ hm2) spray 1 ∼ 3 times, 1 days after the last dose, the terminal residue of cinerin II was 0.054 mg/kg ∼ 0.058 mg/kg; the terminal residue of pyrethrin II was 0.058 mg/kg ∼ 0.061 mg/kg; the terminal residue of jasmolin II was 0.057 mg/kg ∼ 0.061 mg/kg; the terminal residue of cinerin I was 0.055 mg/kg ∼ 0.062 mg/kg; the terminal residue of pyrethrin I was 0.063 mg/kg ∼ 0.071 mg/kg; the terminal residue of jasmolin I was 0.055 mg/kg ∼ 0.059 mg/kg. 3 days after the last application, the terminal residue of 6 components of pyrethrins in grain was less than 0.02 mg/kg.
High concentration (54 g a.i./ hm2) spray 1 ∼ 3 times, 1 days after the last dose, the terminal residue of cinerin II was 0.157 mg/kg ∼ 0.180 mg/kg; the terminal residue of pyrethrin II was 0.151 mg/kg ∼ 0.180 mg/kg; the terminal residue of jasmolin II was 0.131 mg/kg ∼ 0.151 mg/kg; the terminal residue of cinerin I was 0.150 mg/kg ∼ 0.180 mg/kg; the terminal residue of pyrethrin I was 0.162 mg/kg ∼ 0.170 mg/kg; the terminal residue of jasmolin I was 0.151 mg/kg ∼ 0.162 mg/kg. 5 days after the last application, the terminal residue of 6 components of pyrethrins in grain was less than 0.02 mg/kg.
3.4.9 Terminal residues of pyrethrins in stem
The highest recommended dose of 1.5 % pyrethrins aqueous emulsion in stem (27 g a.i./ hm2) and twice recommended maximum dose (54 g a.i./ hm2) were applied for 1 ∼ 3 times. The terminal residue results were shown in Table 15 after 1, 3 and 5 days after the last application.
Low concentration (27 g a.i./ hm2) spray 1 ∼ 3 times, 3 days after the last application, the terminal residue of 6 components of pyrethrins in stem was less than 0.05 mg/kg.
High concentration (54 g a.i./ hm2) spray 1 ∼ 3 times, 1 days after the last dose, the terminal residue of cinerin II was 0.051 mg/kg ∼ 0.160 mg/kg; the terminal residue of pyrethrin II was 0.110 mg/kg ∼ 0.130 mg/kg; the terminal residue of jasmolin II was 0.051 mg/kg ∼ 0.109 mg/kg; the terminal residue of cinerin I was 0.061 mg/kg ∼ 0.122 mg/kg; the terminal residue of pyrethrin I was 0.130 mg/kg ∼ 0.161 mg/kg; the terminal residue of jasmolin I was 0.059 mg/kg ∼ 0.090 mg/kg. 5 days after the last application, the terminal residue of 6 components of pyrethrins in stem was less than 0.05 mg/kg.
3.5 Degradation in soils at 3 different sites
No residue of pyrethrins was detected in the three soils before the experiment. After detection, the physical and chemical properties of soil are shown in Table 16.
3.5.1 Cinerin II
The half-life of cinerin II in Haibei, Haixi and Haidong soils was 0.95, 0.80 and 1.12 days. The half-life of cinerin II in the three soils changed little, and the degradation of cinerin II in Haixi soil was the fastest. The half-life of sterilized soil increased obviously (Table 17, Fig. 14).
3.5.2 Pyrethrin II
The half-life of pyrethrin II in Haibei, Haixi and Haidong soils was 0.64, 1.14 and 0.88 days. The degradation of pyrethrin II in Haibei soil was the fastest. The half-life of sterilized soil increased obviously (Table 18, Fig. 15).
3.5.3 Jasmolin II
The half-life of jasmolin II in Haibei, Haixi and Haidong soils was 1.22, 1.93 and 1.74 days. The degradation of jasmolin II in Haibei soil was the fastest. The half-life of sterilized soil increased obviously (Table 19, Fig. 16).
3.5.4 Cinerin I
The half-life of cinerin I in Haibei, Haixi and Haidong soils was 2.22, 2.15 and 2.44 days. The half-life of cinerin I in the three soils changed little. The half-life of sterilized soil increased obviously (Table 20,Fig. 17).
3.5.5 Pyrethrin I
The half-life of pyrethrin I in Haibei, Haixi and Haidong soils was 2.61, 2.38 and 3.37 days. The degradation of pyrethrin I in Haibei soil was the fastest. The half-life of sterilized soil increased obviously (Table 21, Fig. 18).
3.5.6 Jasmolin I
The half-life of jasmolin I in Haibei, Haixi and Haidong soils was 4.33, 4.13 and 4.76 days. The half-life of jasmolin I in the three soils changed little. The half-life of sterilized soil increased obviously (Table 22, Fig. 19).
In this study, organic carbon, pH and the half-life of each pyrethrins component at three experimental sites were selected for binary regression analysis (Table 3). Except cinerin II, the other five components were negatively correlated with organic carbon and pH.
Analyte
OC (a), pH (b)
correlation coefficient (r)
Regression Equation
cinerin II
T1/2 = -12.06 + 0.08a + 1.42b
0.999
pyrethrin II
T1/2 = 306.54–1.41a-34.09b
0.999
jasmolin II
T1/2 = 493.92–2.26a-54.93b
0.999
cinerin I
T1/2 = 41.93–0.16a-4.46b
0.999
pyrethrin I
T1/2 = 145.19–0.58a-16.00b
0.999
jasmolin I
T1/2 = 53.17–0.18a-5.51b
0.999
4 Discussion
Detection of pyrethroids can be used to detect residues in cantaloupe with a multiscale deep spectral network and in seafood based on nanoparticle extraction of pyrethroids (Wongmaneepratip et al., 2022; Yu et al., 2023). However, the detection of pyrethroid residues on barley in Qinghai Plateau has not been reported. Compared with solid phase extraction (SPE), microwave extraction (MAE) and supercritical fluid extraction, QuEChERS method is fast, efficient, time-saving and easy to use (Ya-Hong et al., 2011; Qiu 2014). In addition, the LC-MS/MS method has the characteristics of high sensitivity, high accuracy and low detection limit, while the speed and accuracy of the determination of pyrethrin by thin layer scanning, high performance liquid chromatography and capillary electrophoresis are not good, so this experimental method ensures the rapid and accurate analysis of samples (Yun-Qiao et al., 2016; Li et al., 2020).
The residue of pyrethrins in soil, barley grain and stem had a good relationship with the interval time index after sampling, and the correlation coefficient was 0.9160 ∼ 0.9997. The digestion rate of pyrethroids in soil (T1/2:1.45 ∼ 2.57 days) was slower than that in barley grains (T1/2:0.77 ∼ 0.90 days), and slower than that in barley stem (T1/2:0.85 ∼ 0.97 days). The half-life of pyrethrin applied to two media (tobacco and iron plate) during cigarette production is 1 ∼ 2d and 6 ∼ 10d (Yunchuan et al., 2016), respectively. The half-life of three pyrethrin pesticides was found to be 11 ∼ 13.7d in apple production (Qiang 2013). Pyrethrin levels during postharvest treatment of durum wheat were studied. In the single-dose experiment, pyrethroids I and II had half-lives of 46 and 72 days, respectively, while the double-dose pyrethroids I and II had half-lives of 41 and 53 days, respectively.Pyrethrin levels during postharvest treatment of durum wheat were studied. In the single-dose experiment, pyrethroids I and II had half-lives of 46 and 72 days, respectively, while the double-dose pyrethroids I and II had half-lives of 41 and 53 days, respectively (Caboni et al., 2007). In the process of pyrethrin degradation rate study, Li Haiyun et al. found that the synergistic effect of microbial community is more conducive to the complete degradation of toxic compounds than a single strain. Yaohua Huang et al. found that Y6 strain could effectively remove cypermethrin from soil and mud containing native microorganisms during soil remediation.M. O. Petrova et al. found that high temperature and low humidity increased the degradation rate of pyrethroids by an average of 10 days.This experiment was conducted in the Qinghai Plateau region.Studies have shown that soil physical and chemical properties (organic carbon, pH) can affect the degradation of pesticides (Fengling and Huiqin 2009; Xiaoliang et al., 2009; Yang et al., 2012; Wenjuan and Xiangyang 2019). Organic carbon content and pH were negatively correlated with the degradation of pyrethroid II, jasmin II, octolactone I, pyrethroid I and jasmin I. However, organic carbon content and pH were positively correlated with the degradation of nicotinic acid II in the study of yang et al. (Yang et al., 2012). This may be due to the adsorption of the organic matter. Due to the unique ecology of the region, specific environmental factors such as soil pH, temperature, humidity and microbial communities contribute to differences in degradation efficiency. Therefore, this study evaluated the safety of food and soil environment after pyrethroid spraying for the environmental specificities of this region.
5 Conclusions
An analytical method was developed for the determination of pyrethrins residues in highland barley plants and soil by LC-MS/MS with the QuEChERS technique for the clean-up of barley plants and soil. The method is simple, sensitive, accurate, reliable and widely applicable and can achieve rapid multi-residue determination of pyrethrins to a certain extent;The method was used to determine the actual samples, which indicated that the half-lives of pyrethrins in highland barley grains, stems and soil were 0.77 ∼ 0.90 d, 0.85 ∼ 0.97 d and 1.45 ∼ 2.57 d. At 2 times the recommended high dose, the residue value of each component in the soil was 0.028 ∼ 0.200 mg/kg after 7 days, and the residue value of highland barley grains was less than 0.02 mg/kg after 5 days, and the residue value of highland barley stems was less than 0.05 mg/kg after 3 days. Pyrethrins residue on highland barley is safe;When metal ions are added, the degradation of pyrethrin can be significantly inhibited. Degradation behavior of pyrethrin in soils of three experimental sites showed that organic carbon and pH were important factors affecting the degradation of pyrethrin, and the degradation of the six pyrethroid fractions was more closely related to pH than organic carbon content. This study presents an effective technique for detecting pyrethrin residues in barley and soil in Qinghai Plateau. The findings offer technical assistance for safe pesticide use and residue control while providing a foundation for establishing maximum residue limits of pyrethrins in barley.
Funding
This research was funded by the Qinghai Province Basic Research Project 2022-ZJ-907 and the National Natural Science Foundation of China (Grant No. 32160659).
Data availability statement
Data is contained within this article
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Dietary intake assessment of pyrethroid residues from okra and eggplant grown in peri-urban areas of Punjab, Pakistan. Environ. Sci. Pollut. Res.. 2020;27:39693-39701.
- [Google Scholar]
- Assessment of genotoxicity of pyrethrin in cultured human lymphocytes. Drug Chem. Toxicol.. 2017;40:251-255.
- [Google Scholar]
- Introduction to pyrethroid insecticides: chemical structures, properties, mode of action and use. Pyrethroid Insecticides 2020
- [CrossRef] [Google Scholar]
- Degradation of pyrethrin residues on stored durum wheat after postharvest treatment. J. Agric. Food Chem.. 2007;55:832-835.
- [CrossRef] [Google Scholar]
- China, M. o. A. a. R. A. o. t. P. s. R. o., 2004. People's Republic of China Agricultural Industry Standard NY/788-2004 Test criteria for pesticide residues. Agricultural quality standard. 5.
- China, M. o. A. a. R. A. o. t. P. s. R. o., 2018. People's Republic of China Agricultural Industry Standard NY/788-2018 Test criteria for pesticide residues. Agricultural quality standard. 5.
- Current research on the safety of pyrethroids used as insecticides. Medicina 2018
- [CrossRef] [Google Scholar]
- Transcriptome profiling reveals mosaic genomic origins of modern cultivated barley. Proc. Natl. Acad. Sci.. 2014;111:13403-13408.
- [CrossRef] [Google Scholar]
- Effect of highland barley β-glucan on starch digestibility in vitro. Shipin Kexue/food Science.. 2018;39:106-111. https://doi.org/kns.cnki.net/kcms/detail/11.2206.TS.20170713.1134.084.html
- [Google Scholar]
- Residue analysis and risk assessment of pyrethrins in open field and greenhouse turnips. Environ. Sci. Pollut. Res.. 2018;25:877-886.
- [CrossRef] [Google Scholar]
- Migration and transformation of pesticides in soil and environmental factors affecting pesticide residues and degradation in soil. Environ. Dev.. 2009;4
- [Google Scholar]
- Understanding the nutrient composition and nutritional functions of highland barley (Qingke): a review. Trends Food Sci. Technol.. 2020;103:109-117.
- [CrossRef] [Google Scholar]
- A modified QuEChERS method for determination of pyrethroid residues in traditional Chinese medicine oral liquids by high-performance liquid chromatography. Molecules. 2019;24:1470.
- [Google Scholar]
- inspection, D. o. A. p., 2016. Standard practice for field tests of pesticide registration residues, Standard practice for field tests of pesticide registration residues.
- Determination of Matrine and oxymatrine in Shiwei KuXiao XunXi powder by HPLC. Strait Pharm. J.. 2020;32:58-61.
- [CrossRef] [Google Scholar]
- Adaptive evolution of the chrysanthemyl diphosphate synthase gene involved in irregular monoterpene metabolism. BMC Evol. Biol.. 2012;12:214.
- [Google Scholar]
- Abuse of chemical substances cause poisoning in dogs and cats: a review. Indian J. Forensic Med. Toxicol.. 2022;16:137-143.
- [Google Scholar]
- Simultaneous determination of pyrethrins residues in teas by ultra-performance liquid chromatography/tandem mass spectrometry. Anal. Chim. Acta. 2010;678:56-62.
- [CrossRef] [Google Scholar]
- A systematic review of highland barley: ingredients, health functions and applications. Grain and Oil Technology.. 2022;005
- [Google Scholar]
- Research status of nutritional value and efficacy of highland barley. Food Res. Dev.. 2020;41:206-211.
- [Google Scholar]
- Sodium channel-directed alkaloids synergize the mosquitocidal and neurophysiological effects of natural pyrethrins. Pestic. Biochem. Physiol.. 2022;186:105171
- [Google Scholar]
- Highland barley starch (Qingke): Structures, properties, modifications, and applications. Int. J. Biol. Macromol.. 2021;185:725-738.
- [CrossRef] [Google Scholar]
- Dissipation and residues of pyrethrins in leaf lettuce under greenhouse and open field conditions. Int. J. Environ. Res. Public Health. 2017;14:822.
- [CrossRef] [Google Scholar]
- Comparison of green sample preparation techniques in the analysis of pyrethrins and pyrethroids in baby food by liquid chromatography–tandem mass spectrometry. J. Chromatogr. A. 2017;1497:28-37.
- [Google Scholar]
- Study on the degradation and change regularity of pesticide permethrin in apple. Northwest Agriculture and Forestry University; 2013.
- Qiu, X., 2014. Study on Residue and Dissipation Dynamics of1.3% Matrine AS in Chinese Cabbage and Soil. Northwest A&F University: Xianyang, China.
- Application of the QuEChERS method for the analysis of pyrethrins and pyrethroids in fish tissues. Anal. Bioanal. Chem.. 2010;397:2525-2531.
- [CrossRef] [Google Scholar]
- Detection of 10 pyrethroids residues in tea by multi-wall carbon nanotubes modified QuEChERS method combined with gas chromatography-tandem mass spectrometry. Chromatography. 2022;40:469-476. https://doi.org/kns.cnki.net/kcms/detail/21.1185.O6.20220309.1412.006.html
- [Google Scholar]
- Validation of an analytical method for the quantification of pyrethrins on lemons and apricots using high-performance liquid chromatography/mass spectrometry. J. Environ. Sci. Health B. 2011;46:530-534.
- [CrossRef] [Google Scholar]
- Study on the effect of biochar on the migration of pesticide degradation product trichloropyridinol in soil. Trans. Agric. Eng.. 2019;35:8.
- [Google Scholar]
- Quantification and risk assessment of pyrethroid residues in seafood based on nanoparticle-extraction approach. Food Control. 2022;133:108612
- [Google Scholar]
- A simple solid-phase extraction coupled to high-performance liquid chromatography–UV detection for quantification of pyrethroid residues in fruits and vegetables. Food Anal. Methods. 2012;5:849-855.
- [Google Scholar]
- Effects of soil environmental factors on degradation of residual pesticides. J. Northeast. Agric. Univ.. 2009;40:4.
- [CrossRef] [Google Scholar]
- Modified QuEChERS method based on multi-walled carbon nanotubes coupled with gas chromatography-tandem mass spectrometry for the detection of 10 pyrethroid pesticide residues in tea. Se pu=. Chin. J. Chromatogr.. 2022;40:469-476.
- [Google Scholar]
- Extracting technology of matrine from Vietnamese Sophora root by microwave-assist. Agrochemicals 2011
- [Google Scholar]
- Diversity analysis of the developed qingke (hulless barley) cultivars representing different growing regions of the Qinghai-Tibet Plateau in China using sequencerelated amplified polymorphism (SRAP) markers. Afr. J. Biotechnol.. 2010;9:8530-8538.
- [CrossRef] [Google Scholar]
- Research progress on effects of physicochemical properties on degradation of pesticides. Sci. Technol. Inf.. 2012;000:58-60.
- [Google Scholar]
- Multiscale deepspectra network: detection of pyrethroid pesticide residues on the hami melon. Foods.. 2023;12:1742.
- [Google Scholar]
- Dynamic analysis of pyrethroid insecticide degradation in cigarette production environment. J. Southern Agric.. 2016;47:5.
- [Google Scholar]
- Determination of matrine and oxymatrine in Radix Sophorae Flavescentis by TLC scanning. J. Traditional Chin. Vet. Med. 2016
- [Google Scholar]
- The draft genome of Tibetan hulless barley reveals adaptive patterns to the high stressful Tibetan Plateau. Proc. Natl. Acad. Sci.. 2015;112:1095-1100.
- [CrossRef] [Google Scholar]
- Origin and evolution of qingke barley in Tibet. Nat. Commun.. 2018;9:5433.
- [CrossRef] [Google Scholar]
- Multiresidue determination of pyrethroid pesticide residues in pepper through a modified QuEChERS method and gas chromatography with electron capture detection. Biomed. Chromatogr.. 2016;30:142-148.
- [Google Scholar]
- Pyrethroids toxicity to male reproductive system and offspring as a function of oxidative stress induction: rodent studies. Front. Endocrinol.. 2021;12:656106
- [Google Scholar]
- Physical and viscoelastic properties of different moisture content highland barley kernels. Int. J. Food Eng.. 2017;13:20170186.
- [CrossRef] [Google Scholar]
Appendix A
Supplementary material
Supplementary data to this article can be found online at https://doi.org/10.1016/j.arabjc.2023.105408.
Appendix A
Supplementary material
The following are the Supplementary data to this article:Supplementary data 1
Supplementary data 1







