Translate this page into:
Study on pharmacodynamic material basis and mechanism of anti-inflammatory effect of Tibetan medicine Gentiana lawrencei var. farreri based on UPLC-Q-TOF/MS combined with network pharmacology and molecular docking
⁎Corresponding authors at: School of Ethnic Medicine, State Key Laboratory of Southwestern Chinese Medicine Resources, Chengdu University of Traditional Chinese Medicine, No. 1166 Liutai Avenue, Chengdu 611137, China (R. Gu). College of Pharmacy, Southwest Minzu University, No. 168 Wenxing Section, Dajian Road, Chengdu611137, China (S.-H. Zhong). 21800010@swun.edu.cn (Shi-Hong Zhong), gurui@cdutcm.edu.cn (Rui Gu)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Gentiana lawrencei var. farreri (GLF) is one of the varieties of the Tibetan medicinal herb “Bang Jian”, known as “Longdanhua” have been employed in Tibetan medicine for the treatment of inflammatory-related diseases. However, the anti-inflammatory effect of its play on the pharmacodynamic material and anti-inflammatory mechanism are still unclear. Therefore, in this study, we proposed UPLC-Q-TOF/MS combined with network pharmacology, molecular docking and in vitro experimental validation to explore the material basis and potential mechanism of the anti-inflammatory effect of GLF. Firstly, 43 compounds of GLF were identified by UPLC-Q-TOF/MS. Next, the potential 6 core active components, 15 core targets and 5 key pathways of GLF for the treatment of inflammation were used to predicted by network pharmacology. Then, molecular docking was used to validate the binding ability of core chemical constituents and core targets of GLF, and the results showed that the 6 core chemical constituents could bind well to 15 core targets, among which isoorientin had the lowest binding energy to AKT1 (−10.5 kcal mol−1). Finally, the promising anti-inflammatory activity of the key component isoorientin was verified by the LPS-induced macrophage inflammation model in RAW264.7 mice. This study revealed the anti-inflammatory pharmacodynamic material basis and mechanism of GLF, which provide a theoretical basis for the development, utilization, and clinical application of GLF.
Keywords
Gentiana lawrencei var. farreri
UPLC-Q-TOF/MS
Pharmacodynamic material basis
Anti-inflammatory
Network pharmacology
Molecular docking
1 Introduction
Inflammation refers to a basic pathological process in which the body is exposed to pathogens, irritants and other harmful stimuli, which is mainly based on defense response (He et al., 2020). Macrophages are one of the immune cells closely associated with the development of inflammation (Gu et al., 2024). Lipopolysaccharide (LPS) is an important component of the cell wall of gram-negative bacteria, and is a powerful activator of macrophages, which can effectively activate macrophages to secrete tumor necrosis factor α (TNF-α), interleukin-6 (IL-6), interleukin-1β (IL-1β), prostaglandin E2 (PGE2), and nitric oxide (NO) and other inflammatory factors (Ren et al., 2017). The massive secretion of these inflammatory factors can cause a systemic inflammatory response in the body. Long term inflammatory reactions can lead to chronic inflammation, which may eventually transform into chronic diseases (Park et al., 2018; Soehnlein and Libby, 2021; Tang et al., 2021). It is well known that most diseases including diabetes (Cheng et al., 2023), cancer (Shadab et al., 2023), neurodegenerative diseases (Anderson et al., 2023), atherosclerosis (Wadley et al., 2013) and inflammatory bowel disease (Rezazadeh et al., 2023) are closely related to the inflammatory response. Severe inflammation may even progress to sepsis, leading to multi-organ dysfunction and ultimately death. Therefore, suppressing inflammation has been recognized as a vital goal in preventing inflammation-mediated human diseases. Currently, the main drugs authorized for clinical use in managing inflammation are non-steroidal anti-inflammatory drugs (NSAIDs) (Patil et al., 2019), as well as glucocorticoid steroidal anti-inflammatory drugs (SAIDS) (Wongrakpanich et al., 2018). However, anti-inflammatory drugs can lead to severe gastrointestinal reactions, liver impairment, and specific damage to the cardiovascular system when taken for a long period of time. In order to improve this situation, more and more researchers are focusing on natural medicines with fewer toxic side effects and have successfully developed a variety of plant-based anti-inflammatory drugs.
Gentiana lawrencei var. farreri (Balf. f.) T. N. Ho (GLF) is a perennial herb of the Gentiana genus in the family Gentianaceae, mainly thrived in Sichuan, Qinghai, Gansu and other high mountain meadows, scrub and beach at 2410–4600 m (Fu et al., 2020). It is a unique species of the Tibet Plateau and a significant component of alpine meadows. GLF is a typical blue-flowered gentian, which is one of the varieties of Tibetan medicine “Bang Jian”, and also the main medicinal material of the classical Tibetan medicines, such as Shiwei Longdanhua Granule (Liu et al., 2023a). And the medicinal part is flower, known as “Longdanhua” have been employed in Tibetan medicine to address pneumonia, hepatitis, acute and chronic tracheobronchitis and other inflammatory diseases (Yang et al., 2012a). At present, research on the chemical constituents of GLF focuses on polysaccharides (Lin et al., 2014), iridoids, flavonoids and triterpenoids (Ma et al., 2023). Pharmacological studies mainly focus on antibacterial and antitumor effects (Sun, 2012), but there is less research on its anti-inflammatory effects, and the pharmacodynamic material and anti-inflammatory mechanism of its anti-inflammatory effects are still unclear.
Phytopharmaceuticals have complex chemical constituents, and traditional methods of isolation and identification are both time-consuming and labor-intensive. UPLC-Q-TOF/MS technology not only has the strong separation ability of liquid chromatography for complex samples, but also has the high sensitivity and selectivity of mass spectrometry, as well as the characteristics of providing relative molecular mass and rich structural information, so that it can rapidly separate and identify complex chemical components in botanicals, and has been widely used in the study of the constituents of medicinal herbs and their qualitative analysis in recent years (Li et al., 2024). Network pharmacology is an emerging discipline that combines fundamental theories from systems biology and network biology. The “chemical constituents-targets-disease” network is constructed to explore the synergistic relationship of drugs, targets, diseases, and the active compounds and molecular mechanisms of traditional Chinese medicine can be effectively predicted (Luo et al., 2023; Zhu et al., 2019). This technique has proven to be powerful in the screening of active compounds, such as Lagotis integra W. W. Smith (Wang et al., 2023), Isodon Japonicus (Xian et al., 2023), and Schisandra chinensis (Lv et al., 2020). Consequently, it is extensively employed in the study of ethnomedicine and traditional Chinese medicine. Molecular docking is a computational software commonly used to explore binding sites and interactions between small and large molecules (Qi et al., 2023). In general, network pharmacology usually requires the collection of target information related to drug chemical constituents in commonly used databases. However, there are currently no databases that include the chemical constituents of GLF. As a result, the UPLC-Q-TOF/MS technique is more comprehensive and reliable.
In this study, we proposed UPLC-Q-TOF/MS combined with network pharmacology, molecular docking technology and in vitro experiments to investigate the material basis and potential mechanism of the anti-inflammatory effects of GLF. Firstly, the UPLC-Q-TOF/MS technique was employed to identify the chemical constituents of GLF. Then, network pharmacology was utilized to analyze and forecast the potential active constituents, targets and action pathways of GLF in the therapy of inflammation. Next, molecular docking technique was utilized to predict the binding capacity of core chemical constituents and core targets of GLF. Lastly, the anti-inflammatory efficacy of its key constituent was verified by LPS-induced RAW264.7 mouse macrophage inflammation model. It is expected to offer a theoretical foundation for the development, utilization and clinical utilization of GLF. The workflow for this study is depicted in Fig. 1.
Overall workflow of the study.
2 Materials and methods
2.1 Materials and reagents
The sample of GLF (flower) was purchased from Tibet Ya Gling Tibetan Medicinal Selling Co., Ltd, and was identified by Professor Rui Gu from Chengdu University of Traditional Chinese Medicine. Before the experiment, the sample materials were dried, pulverized and sieved. Then, stored in a desiccator at room temperature.
Isoorientin was acquired from Chengdu Push Bio-technology Co., Ltd. (Chengdu, China). Dexamethasone was purchased from Shanghai Yien Chemical Technology Co., Ltd. (Shanghai, China). Lipopolysaccharides Purchased from Beijing Solarbio Science & Technology Co., Ltd. (Beijing, China). Acetonitrile, methanol and formic acid were purchased from Fisher Chemicals (Pittsburg, USA). All chemicals and solvents were analytical reagents or chromatographic grade.
2.2 UPLC-Q-TOF/MS analysis of GLF chemical constituents
2.2.1 Laboratory instrument
Instruments for this experiment included: ACQUITY Class 1 plus Ultra High-Performance Liquid chromatograph (Waters, USA), SYNAPT XS High-Resolution Mass Spectrometer (Waters, USA), ACQUITY UPLC CSH C18 (100 mm × 2.1 mm, 1.7 μm, Waters, USA), SB-5200D ultrasonic cleaning machine (Xinyi, China), 1/10000BSA124S Electronic Balance (sartorius, Germany).
2.2.2 Sample preparation
0.5 G of GLF powder was placed in a conical flask, 25 ml of methanol was added, and the extract was sonicated for 30 min. Then, the extract was taken out and cooled, and methanol was added to make up for the weight lost in the extraction process. Samples were filtered with a 0.22 μm microporous filtration membrane for UPLC-Q-TOF/MS analysis (Fan et al., 2023)
2.2.3 UPLC-Q-TOF/MS qualitative analysis
The conditions for chromatographic analysis were as follows: chromatographic separation was performed on an ACQUITY UPLC CSH C18 column (100 mm × 2.1 mm, 1.7 μm), The mobile phase was composed of 0.1 % formic acid solution (solvent A) and acetonitrile solution (solvent B). The gradient elution setup was as follows: 0–7.5 min, 95 %–82 % A; 7.5–10.5 min, 82 %–80 % A, 10.5–15 min, 80 %–75 % A; 15–19.5 min, 75 %–71 % A; 19.5–27 min, 71 %–63 % A; 27–30 min, 63 %–60 % A; 30–34.5 min, 60 %–55 % A; 34.5–36 min, 55 %–52 % A; 36–40.5 min, 52 %–40 % A; 40.5–43.5 min, 40 %–30 % A; 43.5–45.75 min, 30 %–5% A; 45.75–46.5 min, 5 %–95 % A; 46.5–51 min, 95 % A (Li et al., 2024). The flow rate was set at 0.2 ml min−1, the column temperature was 35 ℃, and the injection volume was 2 μL.
The conditions for mass spectrometry analysis were as follows: the capillary voltages were set at 2.5 kV (positive ion mode) and 2.0 kV (negative ion mode). Nitrogen was used as the drying gas, source temperature was set to 150 ℃. The sample cone voltage was 40 V, the collision energy was set to 4 V, and the mass scan range was m/z 50 to 1200 Da.
2.3 Network pharmacology
2.3.1 Collection of active constituents and targets of drugs
The CAS numbers of the GLF chemical components identified by UPLC-Q-TOF/MS were entered into the TCMSP (https://old.tcmsp-e.com/tcmsp.php) platform for searching to screen the chemical constituents with Oral Bioavailability (OB) ≥ 30 % and Drug-Like properties (DL) ≥ 0.18, and the literature was reviewed to complement the chemical ingredients that did not satisfy the OB and DL requirements, but with significant biological activity. Afterward, Swiss Target Prediction (https://www.swisstargetprediction.ch/) and PharmMapper database (https://lilab-ecust.cn/pharmmapper/index.html) were used to import the structures of the chemical ingredients. Then, the targets with probability greater than 0 and Norm Fit greater than 0.9 were selected respectively and imported into the Uniport online website(https://www.uniprot.org/) to be converted to the appropriate gene symbols. Lastly, the targets of the candidate ingredients were determined by combining and deleting duplicates from the obtained targets.
2.3.2 Collection of inflammation-related targets and potential therapeutic targets
GeneCards (https://previous.genecards.org/), and OMIM (https://www.omim.org/) databases were searched for targets related to inflammation with the keyword inflammatory. GeneCards search results were analyzed using the median screening method 4 times to remove a great deal of targets with relevance scores < 3.52. The targets with relevance scores ≥ 3.52 in GeneCards search results were selected and merged with OMIM database search results to obtain inflammation-related targets after removing duplicates. Then, the mutual targets of chemical constituents and diseases were obtained through the Venny (https://bioinfogp.cnb.csic.es/tools/venny/) platform.
2.3.3 Construction of protein–protein interaction networks and screening of core targets
The above obtained “candidate ingredient-disease” common targets were added to the STRING (https://cn.string-db.org/) database to establish the PPI network. The species was limited to “Homo sapiens”, the confidence level was set to 0.4 as a threshold. Subsequently, Cytoscape 3.7.2 software was used to visually analyze the downloaded TSV format result files. And Network Analyzer plug-in was employed to compute the betweenness centrality (BC), closeness centrality (CC) and degree value (Degree). Lastly, common targets above the average of the three were selected as the core targets.
2.3.4 Construction of drug-constituents-targets-disease network
The chemical constituents, common target genes and inflammatory diseases of GLF were imported into Cytoscape 3.7.2, and a “drug-constituents-targets-disease” network diagram of the anti-inflammatory activity of GLF was constructed. The Network Analyzer tool of the software was utilized to calculate network topology parameters. The core anti-inflammatory constituents of GLF were screened according to their degree of size.
2.3.5 GO and KEGG enrichment analysis
The cross-targets were uploaded to the David database (https://david.ncifcrf.gov/) for gene ontology (GO) and Kyoto encyclopedia of genes and genomes (KEGG) pathway enrichment Analysis. The restricted species was Homo sapiens, the screening criterion was P < 0.05. The visualization and mapping were performed using online tools bioinformatics platform (https://www.bioinformatics.com.cn/).
2.4 Molecular docking
The 2D structures of the core ligand compounds were obtained from PubChem database in SDF format and saved in mol2 format after energy minimization using Chem 3D 19.0. The obtained core targets were downloaded from the PDB database (https://www.rcsb.org/) in PDB format files. The PyMoL software was used to remove water molecules and ligands from protein structures. Then, the proteins and components were hydrogenated with AutoDockTools-1.5.7 software and saved as pdbqt format files. Finally, the AutoDock Vina software was utilized for molecular docking. The heat map was drawn by online tools bioinformatics platform (https://www.chiplot.online/) to display the free binding energy and the visual analysis was carried out by PyMoL software.
2.5 Validation of the anti-inflammatory activity of the key constituent of GLF
2.5.1 Cell culture
RAW264.7 cells were purchased from the Type Culture Collection of the Chinese Academy of Sciences (Shanghai, China) and cultured in high-sugar DMEM media supplemented with 10 % fetal bovine serum (FBS) and 1 % penicillin–streptomycin in the fully humidified incubator at 37 ℃ with 5 % CO2.
2.5.2 Cytotoxicity assay
RAW264.7 cells were inoculated in 96-well plates (1 × 104 cells/well) cultured for 24 h. Then, 100 μL of medium containing different concentrations of sample solutions (5 μM-80 μM) was added. After each well’s 24-hour incubation period, the medium was taken out. And after adding 10 % CCK-8 solution, the mixture was incubated at 37 °C for 1 h. At 450 nm, the absorbance was measured.
2.5.3 Determination of nitric oxide and inflammatory cytokines
RAW264.7 cells were inoculated into a 96-well plate (1 × 104 cells/well) and cultured for 24 h. Afterwords, dexamethasone and different concentrations of compounds mixed with LPS (the final concentration of LPS was 1 μg/ml, dexamethasone was 10 μM, and the samples were 10, 20 and 40 μM) were incubated in the complete medium for 24 h. The supernatant was collected to determine the content of NO, TNF-α, IL-6, IL-1β and PGE2. The contents of Nitric oxide (NO) were determined by Griss reagents (Beyotime Chemical Co.), TNF-α, IL-6, IL-1β and PGE2 concentrations were determined by ELISA kit (Multisciences biotech Co.).
2.6 Statistical analysis
All the experiments were performed at least three times, Data were analyzed by GraphPad Prism 8 and showed as mean ± SD. Statistical analysis used one-way ANOVA followed by the Tukey test. All statistical tests with P < 0.05 were taken as statistically significant differences.
3 Results
3.1 Identification of the chemical constituents of GLF
UPLC-Q-TOF/MS was employed to perform a fully positive and negative ion scan of the methanol extract of GLF according to the mass spectrometry conditions under 2.2.3, and BPI plots in the positive and negative ion modes were obtained (Fig. 2). Raw data generated by the Waters mass spectrometer were processed using UNIFI software, and the databases (including Waters commercial database and the self-built database) were compared to further analyze and identify the chemical constituents of GLF based on excimer ion peak, retention time and primary and secondary mass spectrometry fragment information. Finally, a total of 43 chemical components were determined (Table 1), including 18 flavonoids, 9 iridoids, 7 triterpenoids, 2 saccharides, 2 organic acids and lignans, 1 steroid, 1 phenolic glycosides and Phenols.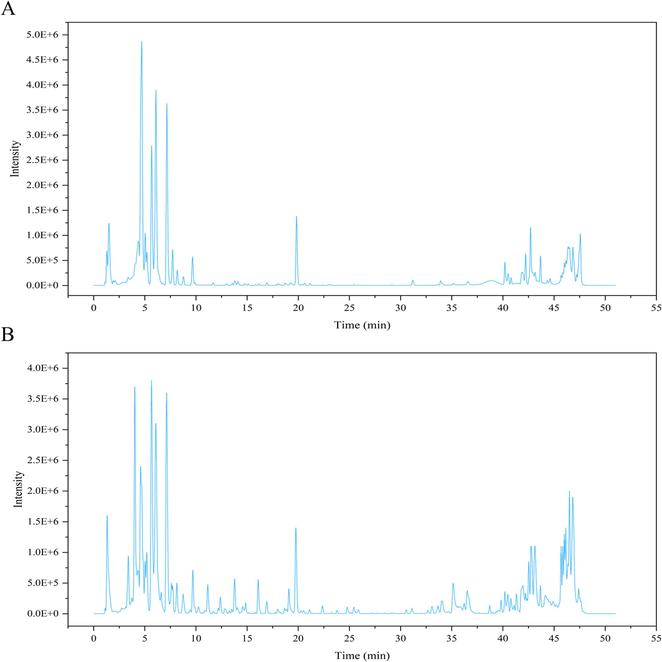
Representative BPI diagram of G. lawrencei var. farreri (GLF) extract in different ionization modes. (A) Positive ion mode. (B) Negative ion mode.
No.
tR/
minIon
speciesformula
Theoretical m/z
Measured
m/z
error/
ppmFragment ions
m/z
Ingredient
nameType
1
1.31
[M−H]−
C18H32O16
504.16903
503.1626
1.8
503.1631, 221.0669, 179.0586, 161.0456, 113.0227, 101.0262, 89.0258
gentianose
saccharides
2
1.32
[M−H]−
C12H22O11
342.11621
341.1098
2.6
179.0586, 101.0262
sucrose
saccharides
3
3.33
[M−H]−
C13H18O8
302.10017
301.0935
2
179.0586, 161.0456, 121.0287
Tachioside
phenolic glycosides
4
3.75
[M−H]−
C27H30O15
594.15847
593.1509
−0.4
503.1158, 375.1258, 213.0763
Isoorientin 2″-O-rhamnoside
Flavonoids
5
4.02
[M−H]−
C16H24O10
376.13695
375.1301
1.2
213.0763, 169.0850, 113.0227
Loganic acid
Iridoids
6
4.04
[M−H]−
C22H34O15
538.18977
537.1834
1.7
358.0641, 151.0748
Gmephiloside
Iridoids
7
4.23
[M−H]−
C33H40O20
756.21129
755.2049
1.1
431.0946, 413.0832
Isovitexin 4′,7-diglucoside
Flavonoids
8
4.58
[M−H]−
C27H30O16
610.15338
609.1465
0.7
489.1070, 429.0800, 298.0502
isoorientin-2″-O-glucopyranoside
Flavonoids
9
4.7
[M−H]−
C27H30O15
594.15847
593.1516
0.6
473.1117, 413.0904, 293.0403
Isovitexin 6″-O-glucoside
Flavonoids
10
4.84
[M + COOH]−
C22H30O14
518.16356
563.1628
1.8
389.1225, 179.0586
6′-O-beta-D-Glucosyl Gentiopicroside
Iridoids
11
5.03
[M + COOH]−
C16H22O10
374.1213
419.1203
2
179.0586, 141.0210
Swertiamarin
Iridoids
12
5.04
[M + Na]+
C23H34O15
550.18977
573.1776
−2.4
149.0623, 121.0661, 85.0315
genipin 1-gentiobioside
Iridoids
13
5.06
[M−H]−
C27H30O16
610.14533
609.1453
2.9
447.0908, 357.0564, 327.0512, 285.0401
Isoorientin 4′ −O-glycoside
Flavonoids
14
5.14
[M−H]−
C19H18O11
422.08491
421.0787
2.6
223.0232, 179.0586, 153.0191
Isomangiferin
Flavonoids
15
5.66
[M−H]−
C22H32O14
520.17921
519.1705
−2.8
455.3521, 341.1068, 179.0586
Swertiapunimarin
Iridoids
16
5.68
[M + COOH]−
C16H20O9
356.11073
401.1095
1.3
327.0512, 179.0539, 149.0612, 113.0227
Gentiopicroside
Iridoids
17
5.79
[M−H]−
C16H22O9
358.12638
357.1194
0.8
357.1323, 195.0657, 125.0261
Sweroside
Iridoids
18
5.93
[M−H]−
C27H30O15
594.15847
593.1509
−0.6
431.0946, 413.0832, 311.0588, 282.0531
Isovitexin 2″-O-glucoside
Flavonoids
19
6.07
[M−H]−
C21H20O11
448.10056
447.0936
0.7
285.0401, 151.0056, 107.0132
Cynaroside
Flavonoids
20
6.6
[M + COOH]−
C28H32O15
608.17412
653.1732
1.3
447.0908, 300.0283, 285.0401
Flavocommelin
Flavonoids
21
6.81
[M−H]−
C28H32O16
624.16903
623.1616
−0.3
443.0959, 323.0598
Isoscoparin-2″-β-D-glucopyranoside
Flavonoids
22
7.13
[M−H]−
C21H20O10
432.10565
431.099
1.5
431.0946, 269.0429, 268.0393
Apigenin 7-glucoside
Flavonoids
23
7.7
[M−H]−
C22H22O11
462.11621
461.1096
1.4
341.0678, 298.0502, 153.0191
Isoscoparin
Flavonoids
24
7.82
[M−H]−
C21H20O11
448.10056
447.0943
2.3
357.0591, 285.0401
Isoorientin
Flavonoids
25
7.89
[M−H]−
C21H20O10
432.10565
431.0994
2.3
341.0678, 311.0588, 283.0648
Isovitexin
Flavonoids
26
9.33
[M + COOH]−
C25H34O14
558.19486
603.1939
1.4
161.0233, 119.0351, 101.0262, 89.0238, 59.0149
Macrophylloside D
Phenols
27
9.44
[M−H]−
C21H20O11
448.10056
447.0941
1.9
429.0800, 357.1323, 327.0512, 285.0401
Kaempferol 3-O-glucoside
Flavonoids
28
9.89
[M−H]−
C28H36O13
580.21559
579.2089
1.1
417.1564, 402.1333, 181.0516, 166.0280
Acanthoside B
Lignans
29
9.89
[M−H]−
C22H26O8
418.16277
417.1561
1.4
402.1333, 181.0516
(+)-syringaresinol
Lignans
30
11.16
[M−H]−
C23H28O13
512.15299
511.1467
1.9
153.0191, 109.0287
Gentiournoside D
Iridoids
31
14.12
[M−H]−
C31H28O14
624.14791
623.1411
0.8
477.0908, 309.0385, 293.0463, 179.0350
7-O-feruloylorientin
Flavonoids
32
15.77
[M−H]−
C30H26O14
610.13226
609.1265
2.4
447.0983, 179.0586, 153.0191
orientin 7-caffeate
Flavonoids
33
31.11
[M−H]−
C16H12O6
300.06339
299.0566
1.7
299.0598, 284.0309
Hispidulin
Flavonoids
34
35.14
[M−H]−
C30H48O5
488.35017
487.3434
1.1
487.3416, 469.3374
Caulophyllogenin
Triterpenoids
35
42.78
[M−H]−
C30H48O4
472.35526
471.3485
1.1
277.2152, 171.1047, 152.9973
Hederagenin
Triterpenoids
36
43.13
[M−H]−
C30H48O4
472.35526
471.3485
1.1
472.3541, 471.3525
Corosolic acid
Triterpenoids
37
44.56
[M + COOH]−
C35H60O6
576.43899
621.4358
−2.3
455.3521
Daucosterol
Steroids
38
44.88
[M + COOH]−
C30H48O2
440.36543
485.3647
2.2
277.2152, 255.2309, 116.9287
Roburic Acid
Triterpenoids
39
46.35
[M−H]−
C16H32O2
256.24023
255.2332
0.9
533.3999, 355.1595, 256.2384, 255.2309
Palmitic acid
Organic acids
40
46.61
[M−H]−
C18H34O2
282.25588
281.2489
0.9
116.9287
Oleic acid
Organic acids
41
46.84
[M−H]−
C30H48O3
456.36035
455.3533
0.6
339.1983, 281.2852, 255.2309
Kouitchenoids A
Triterpenoids
42
47.52
[M−H]−
C30H46O3
454.3447
453.3382
1.7
279.2347, 255.2309,116.9287
Swertialactone-D
Triterpenoids
43
47.81
[M−H]−
C30H48O3
456.36035
455.3537
1.3
456.3591, 455.3521
Ursolic Acid
Triterpenoids
3.2 Network pharmacology
3.2.1 Screening of active constituents and corresponding targets in GLF
According to the results of mass spectrometry analysis, the active constituents of GLF were screened by TCMSP, literature reports. Swertiamarin, genipin 1-gentiobioside, Gentiopicrin, Sweroside, Cynaroside, Flavocommelin, Isoorientin, Isovitexin, Kaempferol 3-O-glucoside, Acanthoside B, (+)-syringaresinol, Hispidulin, Hederagenin, and Corosolic acid, 14 active constituents were selected as candidate constituents. Then, the information of 14 candidate constituents was imported into Swiss TargetPrediction and PharmMapper database for target prediction, and 270 active constituent targets were obtained after weight removal.
3.2.2 Screening of inflammation-related targets and potential therapeutic targets
Inflammatory targets were searched from the GeneCards database and OMIM database, and 1382 inflammatory gene targets were screened according to 2.3.2 conditions. The active constituent targets and disease targets were imported into Venny 2.1 to make Venn diagrams, and 107 intersecting targets were obtained as potential targets for the anti-inflammatory action of GLF for further study (Fig. 3A).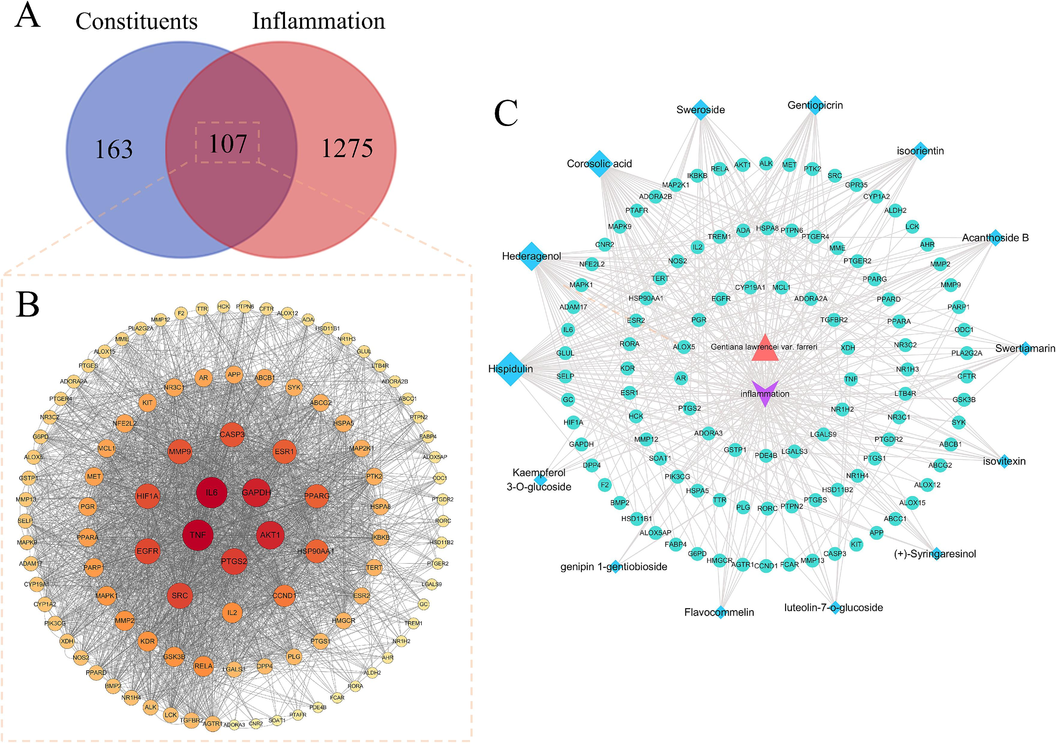
Core constituents and core targets for the treatment of inflammation. (A) Venn diagram of overlapping target genes between GLF constituents and inflammation. (B) Protein–protein interaction (PPI) network analysis of GLF for the treatment of inflammation using STRING database. The size and color of the nodes in the figure is associated with the degree in the network (C) The drug-constituents-targets-disease network of GLF. Blue rhombus nodes indicate bioactive constituents, green circular nodes indicate target proteins, orange triangle nodes indicate GLF drug, purple ellipse nodes represent inflammatory diseases, and the black edge indicated that there was a connection between constituents, targets, drug and disease.
PPI network analysis of anti-inflammatory targets.
To construct a PPI network diagram, 107 potential targets were loaded into the STRING database, and Cytoscape 3.7.2 was used to set the color and size of nodes based on degree size (Fig. 3B). The nodes nearer to the center, larger in size and darker in color in the graph have higher degree values and are more important in the network. According to the network topology model and algorithm, the nodes whose degree, betweenness and closeness values are higher than their average values in the PPI network are regarded as the key nodes, that is the core targets. In our network analysis, there were 15 targets whose degree values, BC values and CC values were greater than 24, 0.0080 and 0.5570, respectively, suggesting that they may be the core targets of anti-inflammatory treatment of GLF. They were TNF, IL6, GAPDH, AKT1, PTGS2, SRC, EGFR, HIF1A, MMP9, CASP3, ESR1, PPARG, HSP90AA1, CCND1 and IL2.
3.2.3 Construction of drug-constituents-targets-disease network
As shown in Fig. 3C, the drug-constituents-targets-disease network of anti-inflammatory-related active constituents and potential therapeutic targets was further constructed using Cytoscape 3.7.2 software. Among them, the top 6 constituents with compound degree values were hispidulin, hederagenin, corosolic acid, sweroside, gentiopicrin and isoorientin, which were hypothesized to be the core constituents of the anti-inflammatory effects of GLF.
3.2.4 GO and KEGG enrichment analysis
David database was applied to obtain the GO and KEGG enrichment of the above intersecting targets. The GO analysis for the anti-inflammatory action of GLF was obtained with screening criteria of P < 0.05. A total of 661 entries were obtained, including 498 cellular Biological Process (BP), 52 Cellular Component (CC) and 111 Molecular Function (MF). The top 10 entries of biological processes, molecular functions and cellular components were visualized in Fig. 4A. GO analysis showed that the BP mainly involved negative regulation of inflammatory response, inflammatory response, response to xenobiotic stimulus, and other processes. The CC mainly included plasma membrane, cytosol, cytoplasm, etc., and the MF mainly involved protein serine/threonine/tyrosine kinase activity, enzyme binding, zinc ion binding protein binding, and so on.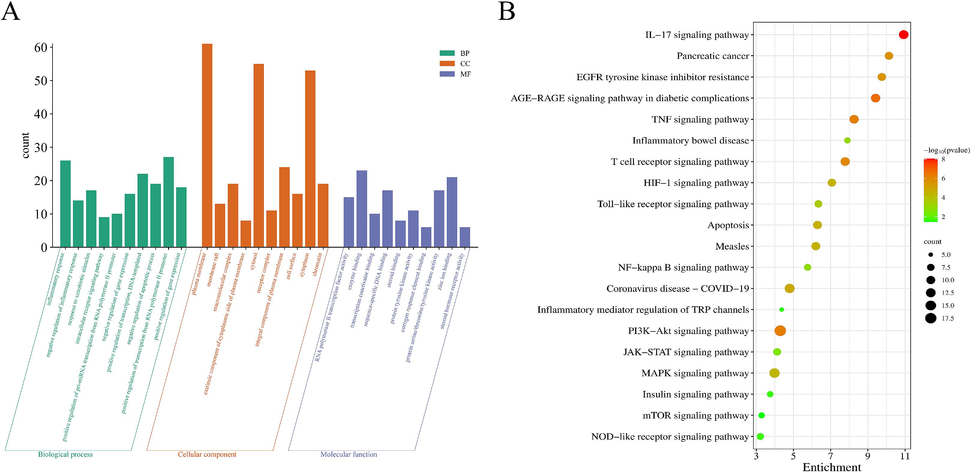
GO and KEGG enrichment analysis. (A) Histograms of GO functional enrichment analysis. (B) Bubble plots of 20 pathways analyzed according to KEGG enrichment.
The enrichment results of KEGG pathway were shown in Fig. 4B, and the number of targets and −lgP values of the enriched pathways were used to measure the degree of KEGG enrichment. It can be seen that a number of pathways were implicated in inflammation occurrence and healing by the screened proteins. The 5 pathways with the lowest p-value were AGE-RAGE signaling pathway in diabetic complications, IL-17 signaling pathway, PI3K-Akt signaling pathway, TNF signaling pathway and T cell receptor signaling pathway. It is speculated that these 5 pathways are the primary mechanisms by which GLF exerts its anti-inflammatory effects.
3.3 Molecular docking validation
The 15 core targets screened in the PPI network were used as receptors, and the top 6 chemical constituents in the “drug-constituents-targets-disease” network were used as ligands. And the PDB ID of the GLF core targets in Table S1. AutoDock Vina was used for molecular docking. A smaller binding energy indicates a stronger binding capacity; a binding energy < -5 kcal·mol−1 indicates a better binding activity, and a binding energy < -7 kcal·mol−1 indicates a strong binding activity(Song et al., 2022). The results of molecular docking were shown in Fig. 5. There were 73 groups with binding energies < -7 kcal·mol−1 and 10 groups with binding energies < -9.5 kcal·mol−1. These results indicated that all constituents could be well bound to the targets. Compared with other target proteins, TNF, AKT1, PTGS2 and EGFR had lower binding energies than other proteins, and isoorientin and AKT1 had the lowest binding energy (−10.5 kcal·mol−1), suggesting that these proteins may be potential anti-inflammatory binding targets of GLF. Isoorientin may be the key anti-inflammatory constituent of GLF. PyMoL was used to visualize the binding sites of constituents and targets with high free binding energy fraction, and it was observed that ligands and receptors could be hydrogen bonded (Fig. 6). In particular, isoorientin formed hydrogen bonds with amino acid residues THR-82, ASN-54, GLU-17 and GLU-85 in AKT1. And it forms hydrogen bonds with the PH-144 and GLN-67 residues of TNF and has a binding affinity.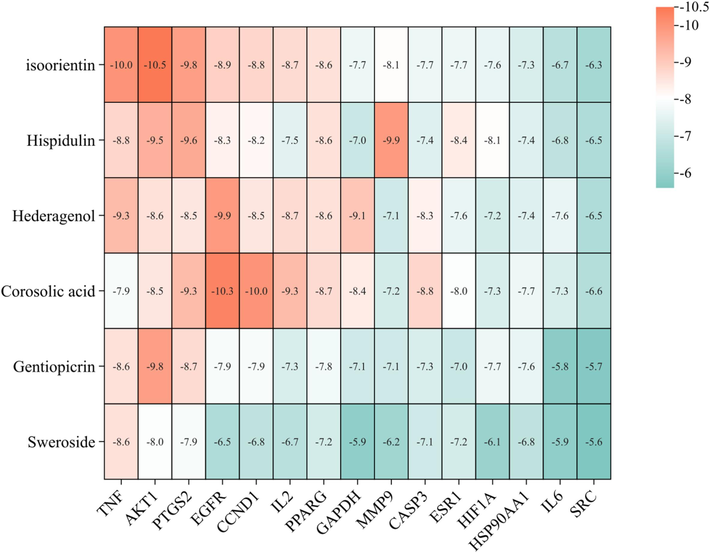
Heatmap of molecular docking scores of core proteins with constituents of GLF.

Docking patterns of some core targets with core constituents.
3.4 Anti-inflammatory activity of isoorientin in LSP-stimulated RAW264.7 cells
Molecular docking results showed that isoorientin was the main active compound in the anti-inflammatory effect of GLF. To further verify the anti-inflammatory action of isoorientin, the effects of different concentrations of isoorientin on cell toxicity of RAW264.7 were evaluated by CCK-8 method. The LPS-induced RAW264.7 cells were treated with different doses of isoorientin, and the expression of NO, TNF-α, IL-6, IL-1β, and PGE2 inflammatory mediators and cytokines in the cell supernatant was also determined to assess the anti-inflammatory effect of isoorientin.
An in vitro cytotoxicity assay was performed using the CCK-8 kit to directly determine the effect of isoorientin on cell viability. Various concentrations of the sample solution (5, 10, 20, 40, 80 μM) were used to treat RAW264.7 cells. Cells in the blank group were not treated with sample solutions. The results showed that there was no cytotoxic effect in the range of 5–80 μM. (Fig. 7A).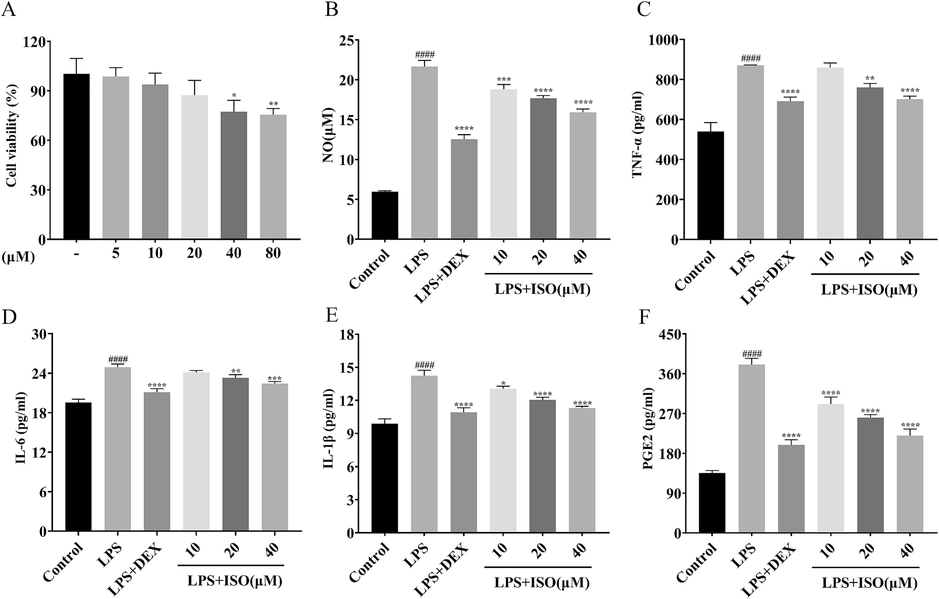
Effect of isoorientin on RAW264.7 cells. (A) Assessment of the cytotoxic effects of different concentrations of isoorientin on RAW264.7 cells using the CCK-8 assay. *p < 0.05, **p < 0.01 vs. control group. (B-F) Effects of isoorientin on NO, TNF-α, IL-6, IL-1β and PGE2 secretion in RAW264.7 cells stimulated by LPS. ####p < 0.0001 vs control group; **p < 0.01, ***p < 0.001, ****p < 0.0001 vs LPS group.
NO is a pro-inflammatory mediator, and LPS stimulation of macrophages to produce NO is a typical marker of a common inflammatory response, so, reducing NO release under inflammatory stimulation is a good strategy to reduce inflammatory response (Tian et al., 2020). Different doses of isoorientin (40, 20, 10 μM) and LPS (1 μg/ml) were co-incubated with RAW264.7 cells. The concentration of NO in the cell supernatant was determined to observe the effect of isoorientin on NO and to evaluate the anti-inflammatory activity of isoorientin. According to Fig. 7B, RAW264.7 cells stimulated with LPS showed significantly higher NO levels in the supernatant than the control group. Compared with the LPS group, NO concentration was significantly lower in the isoorientin group at 40, 20 and 10 μM concentrations in a dose-dependent manner. All concentrations were statistically different from the LPS group.
TNF-α, IL-6, IL-1β, and PGE2 are important pro-inflammatory cytokines in the inflammatory response. Studies have shown that reducing the expression of pro-inflammatory cytokines suppresses the inflammatory response and inhibits oxidative stress (Guo et al., 2021). To further validate the anti-inflammatory activity of isoorientin, we detected the protein expression level of the pro-inflammatory cytokines TNF-α, IL-6, IL-1β and PGE2 by ELISA. According to Fig. 7C–F, compared with the control group, the secretion of the inflammatory factor TNF-α, IL-6, IL-1β, and PGE2 were greatly increased in the LPS group. Compared with the LPS group, the contents of TNF-α, IL-6, IL-1β, and PGE2 in all dose groups of isoorientin showed a tendency to decrease, in which isoorientin with mass concentrations of 40 and 20 μM could significantly inhibit the overproduction of TNF-α, IL-6, IL-1β, and PGE2. These results demonstrated that isoorientin significantly inhibited pro-inflammatory mediator and cytokine release in RAW 264.7 cells induced by LPS.
4 Discussion
Inflammation is the first line of defense against injury and infection of the body, when an inflammatory response occurs, the body will produce a series of inflammatory cells and inflammatory factors, which may cause body damage or even death when these factors are out of control (Lemmers et al., 2017). GLF is one of the mainstream varieties of Tibetan medicine “Bang Jian”, which is often used to treat various inflammatory diseases. However, the current research on GLF focuses on antibacterial and antitumor aspects, while the bioactive molecules and the underlying mechanisms driving the anti-inflammatory effects are still unclear. Therefore, to explore the potential anti-inflammatory pharmacophoric material of GLF and their mechanism of action, 43 GLF chemical constituents were identified by UPLC-Q-TOF/MS in this study. Next, network pharmacology and molecular docking techniques were utilized to investigate the potential anti-inflammatory effects of GLF. Network pharmacology results indicate that hispidulin, hederagenin, corosolic acid, sweroside, gentiopicrin and isoorientin were the major material basis for the anti-inflammatory effects of GLF. GLF may inhibit TNF, IL6, AKT1, EGFR, PTGS2 and other key targets and regulate the AGE-RAGE signaling pathway in diabetic complications, IL-17 signaling pathway, PI3K-Akt signaling pathway, TNF signaling pathway and T cell receptor signaling pathway to exert anti-inflammatory effects. In addition, the molecular docking results showed that 6 core chemical constituents could be well combined with 15 core targets, among which isoorientin had the lowest binding energy with AKT1 (−10.5 kcal-mol−1). Finally, isoorientin was proved to have good anti-inflammatory activity in vitro, which increased the credibility of our previous research results.
With the high separation performance of liquid chromatography and the powerful identification capability of mass spectrometry, LC-MS technology has been widely used in the qualitative analysis of the chemical constituents of traditional Chinese medicine (Chen et al., 2023a). In a previous study, Yang et al. used column chromatography and nuclear magnetic resonance (NMR), mass spectrometry (MS), and infrared (IR) methods to isolate and identify only 10 components of GLF (Yang et al., 2012b). This traditional method is not only labor-intensive and time-consuming but also identifies fewer components. In this study, we used a more efficient and convenient UPLC-Q-TOF/MS method to identify 43 chemical components from GLF extracts, mainly including flavonoids, iris, triterpenoids, lignans and organic acids. The results revealed the material basis of the medicinal effects of GLF, which is of great significance for the further study of the active constituents.
The “drug-constituents-disease-target” network resulted in the screening of 6 core constituents (hispidulin, hederagenin, corosolic acid, sweroside, gentiopicrin and isoorientin). It is worth noting that these constituents have been shown to have certain anti-inflammatory effects in different inflammatory models. For example, hispidulin has antioxidant and anti-inflammatory properties, and is able to inhibit mast cell-mediated allergic dermatitis by down-regulating histamine release and inflammatory cytokines (Kim et al., 2019; Liu et al., 2020). Hederagenin could reduce inflammatory responses in C28/I2 cells and RAW 264.7 cells by downregulating the expression of PGE2, NO, COX-2, iNOS, TNF-α, IL-1β, and IL-6 (Lee et al., 2015; Shen et al., 2023). Corosolic acid exerts anti-inflammatory effects through inhibition of secretory phospholipase A2IIa activity (Pundalik et al., 2022). Isoorientin reduced inflammation in RAW 264.7 cell line in vitro and carrageenan induced inflammatory animal model systems in vivo (Anilkumar et al., 2017). Sweroside could alleviate inflammation in lipopolysaccharide-induced acute lung injury in mice and gentiopicrin was able to exert a dose-dependent anti-inflammatory effect in LPS-induced acute kidney injury (Shareef and Kathem, 2022; Wang et al., 2021). These studies show that our screening results have high confidence and suggest that these 6 monomeric compounds may be the major constituents of GLF with anti-inflammatory effects.
In addition, to further elucidate the anti-inflammatory mechanism of GLF, we constructed PPI networks. 15 core targets were predicted to interact well with other targets by PPI network analysis. These targets have been shown to be strongly associated with inflammatory diseases in modern studies (Zhu and Hou, 2021). Among these key targets, IL-6 and TNF-α are typical pro-inflammatory cytokines that play a prominent role in the inflammatory response and are often regarded as key anti-inflammatory targets (Majidpoor and Mortezaee, 2022; Muslu et al., 2021; Wang et al., 2022). At present, many drugs have reduced the inflammatory response by inhibiting the expression of IL-6 and TNF-α, such as the commonly used drug aspirin (Shvartsur et al., 2022). Annarita et al. found that AKT1 is associated with cell metabolism, survival migration, and gene expression. In a mouse model of acute inflammation, mice lacking AKT1 inhibited the acute inflammatory response of carrageenan, histamine, and bradykinin (Di Lorenzo et al., 2009). Similar to AKT1, inhibition of EGFR expression attenuated palmitic acid-induced inflammatory response in H9C2 cells (Li et al., 2016). PTGS2, an enzyme induced by pro-inflammatory cytokines, is a key enzyme in the synthesis of PGE2 inflammatory mediators that stimulate cancer cell proliferation, development and metastasis. Studies have shown that inhibition of PTGS2 activity inhibits the expression and activation of the PTGS2/COX2-PGE2 signaling axis during inflammation (Martín-Vázquez et al., 2023). Above all, these studies suggest that all of these key targets we screened play important roles in the inflammatory response and that inhibiting these processes may be an effective therapeutic target for GLF against inflammatory diseases.
KEGG pathway enrichment analysis showed that the anti-inflammatory activity of GLF was mainly involved in AGE-RAGE signaling pathway in diabetic complications, IL-17 signaling pathway, TNF signaling pathway, PI3K-Akt signaling pathway and T cell receptor signaling pathway. And among them, IL-17 signaling pathway, TNF signaling pathway, and PI3K-Akt signaling pathway are common signaling pathways that regulate inflammation. The IL-17 signaling pathway plays an important role in the pathogenesis of inflammatory diseases and many autoimmune. The IL-17 receptor is composed of 5 subunits (IL-17RA-IL17-RE), of which IL-17RA is a common subunit in IL-17R family and widely expressed in a variety of tissues and cells (Mills, 2023; Miossec and Kolls, 2012). Targeting IL-17RA blocks IL-17-mediated proinflammatory pathway (Beringer et al., 2016). TNF signaling pathway is associated with cell survival, death and differentiation, and is involved in the inflammatory response process. For example, Baicalin combined with Echinacoside effectively reversed the mRNA expression of key genes in the TNF signaling pathway, TNF, AKT1, CASP7, CCL5, MAPK14, and MMP3, and thus alleviated the IMQ-induced inflammatory dermatosis (Chen et al., 2023b). The PI3K/Akt pathway is an intracellular signaling pathway that regulates cell growth, death, proliferation, and senescence, and is also a key factor in ulcer healing(Dai et al., 2018). And it has been shown that Isoalantolactone alleviates inflammation by modulating the PI3K-Akt signaling pathway, thereby preventing ethanol-induced gastric ulcers (Zhou et al., 2023). In conclusion, these 5 pathways are tightly linked to the anti-inflammatory effects of GLF, and may be potentially key pathways of its action.
Molecular docking is a computerized target prediction tool which is used for predicting ligand-target interaction, and helps to search for lead compounds with significant activity (Liu et al., 2023b). To further validate the accuracy of our previous web-based pharmacology results, molecular docking of 6 core constituents (hispidulin, hederagenin, corosolic acid, sweroside, gentiopicrin and isoorientin) with 15 core targets (TNF, IL6, GAPDH, AKT1, PTGS2, SRC, EGFR, HIF1A, MMP9, CASP3, ESR1, PPARG, HSP90AA1, CCND1 and IL2) was performed. The results showed that the 6 core constituents were successfully connected to 15 core targets with good binding ability, among which isoorientin had the best binding ability to AKT1 (−10.5 kcal·mol−1), suggesting that isoorientin may be a key constituent in the anti-inflammatory activity exerted by GLF, further validating the accuracy of the above network pharmacological predictions.
Macrophages are one of the immune cells closely associated with the development of inflammation, and NO, TNF-α, IL-6, IL-1β and PGE2 are important pro-inflammatory factors (Gu et al., 2024). The massive secretion of these inflammatory factors can cause systemic inflammatory responses in the body, so the LPS-induced inflammation model of RAW264.7 cells has been widely applied to the evaluation of anti-inflammatory effects of various drugs in modern pharmacology. Based on the results of biological network analysis and molecular docking, we finally chose the isoorientin component with the smallest binding energy to each core target for in vitro anti-inflammatory validation studies. We determined the expression of pro-inflammatory factors (NO, TNF-α, IL-6, IL-1β and PGE2) by LPS-induced macrophage inflammation model in RAW264.7 mice. The results showed a significant decreasing trend of pro-inflammatory mediators and cytokines, suggesting that isoorientin could alleviate LPS-induced inflammation in RAW264.7 cells. Isoorientin is a dietary flavonoid with strong antioxidant, anti-inflammatory and antidiabetic properties is widely distributed in a variety of plants and foods. Pharmacokinetics characterization showed that isoorientin had low bioavailability, but it could be metabolized to monoglucuronide by uridine 5′-diphospho-glucuronosyltransferase and also could be well absorbed through passive diffusion in Caco-2 cell monolayers (Ziqubu et al., 2020). Recently, studies demonstrated that isoorientin is able to suppress the inflammatory response by inhibiting the Galectin-3/NLRP3/IL-1β signaling pathway and the MAPK/NF-κB signaling pathway in mice model of ulcerative colitis (UC) and microglia inflammation model of LPS-induced mouse microglial (BV-2) cells (Chen et al., 2024; Yuan et al., 2014b). Meanwhile, it has also been found that it is able to induce apoptosis and autophagy by inhibiting the PI3K/Akt signaling pathway (Yuan et al., 2014a). In addition, Zhang et al. found that isoorientin reduced the expression of MMP-9 and pro-inflammatory factors TNF-α, IL-6, and IL-1β in the spinal cord of the CCI mice to reduce inflammation by immunofluorescence and western blot analysis (Zhang et al., 2019). These studies again demonstrate the accuracy of the above predictions.
This is the first report about UPLC-Q-TOF/MS combined with network pharmacology, molecular docking and in vitro experiment analysis to interpret the pharmacodynamic material basis and potential mechanisms of the anti-inflammatory effects of GLF. These findings could not only benefit the comprehensive development and utilization of GLF, but also provide a theoretical basis for subsequent clinical applications. But further experimental validation is needed in the future for the subsequent development, utilization and clinical application of GLF.
5 Conclusion
In this study, a total of 43 chemical components were identified using UPLC-Q-TOF/MS technique, which revealed the pharmacodynamic material basis of GLF. Then the anti-inflammatory mechanism of GLF was revealed by network pharmacology and molecular docking analysis, which emphasized that the anti-inflammatory effect of GLF was synergistic through multi-components, multi-targets, and multi-pathways, and finally, the in vitro anti-inflammatory effect of isoorientin was validated based on the molecular docking results selected as a research object. These findings provided a theoretical basis for the future development, utilization, and clinical application of GLF.
CRediT authorship contribution statement
Jie Zhang: Writing – original draft, Validation, Investigation, Formal analysis. Yuan Li: Formal analysis, Conceptualization. Ling Li: Methodology. Jie-Lin Zhang: Validation. Li-Wen Zhong: Investigation. Jia-Yi Zhang: Investigation. Shi-Hong Zhong: Writing – review & editing. Rui Gu: Writing – review & editing, Funding acquisition.
Acknowledgements
We appreciate the help provided by research staff on the field experimental station and laboratory.
Funding
This work was supported by the Ministry of Science and Technology of the People's Republic of China (No. 2019YFC1712305).
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- NLRP3 inflammasome in neurodegenerative disease. Transl. Res.. 2023;252:21-33.
- [CrossRef] [Google Scholar]
- Evaluation of Anti-Inflammatory Properties of Isoorientin Isolated from Tubers of Pueraria tuberosa. Oxid. Med. Cell. Longev.. 2017;2017:1-10.
- [CrossRef] [Google Scholar]
- IL-17 in Chronic Inflammation: From Discovery to Targeting. Trends Mol. Med.. 2016;22:230-241.
- [CrossRef] [Google Scholar]
- UPLC-Q-Exactive-MS Combined with Network Pharmacology to Explore the Antitumor Effect of Polygonatum sibiricum Leaf Tea. J. Food Biochem.. 2023;2023:1-12.
- [CrossRef] [Google Scholar]
- Isoorientin ameliorated ulcerative colitis by inhibiting the Galectin-3/NLRP3/IL-1β signaling pathway. J. Funct. Foods. 2024;121:106394
- [CrossRef] [Google Scholar]
- Topical Application of Baicalin Combined with Echinacoside Ameliorates Psoriatic Skin Lesions by Suppressing the Inflammation-Related TNF Signaling Pathway and the Angiogenesis-Related VEGF Signaling Pathway. ACS Omega. 2023;8:40260-40276.
- [CrossRef] [Google Scholar]
- How inflammation dictates diabetic peripheral neuropathy: An enlightening review. CNS Neurosci. Ther. 2023
- [CrossRef] [Google Scholar]
- Identification of key genes and pathways in meningioma by bioinformatics analysis. Oncol. Lett.. 2018;15:8245-8252.
- [CrossRef] [Google Scholar]
- Akt1 is critical for acute inflammation and histamine-mediated vascular leakage. Proc. Natl. Acad. Sci.. 2009;106:14552-14557.
- [CrossRef] [Google Scholar]
- Fan, J., Nan, C., Deng, W., 2023. Study on chemotype of Tibetan Medicine “Bang Jian” Multi-Origin Species Based on HPLC Fingerprint and UPLC-Q-TOF-MS. Modernization of Traditional Chinese Medicine and Materia Medica-World Science and Technology 25, 3554–3570.
- Analysis of ITS heterozygosity and genetic divergence in Gentiana lawrencei var. farreri. J. Biol.. 2020;37:44-47.
- [Google Scholar]
- Combined targeting of senescent cells and senescent macrophages: A new idea for integrated treatment of lung cancer. Semin. Cancer Biol.. 2024;106–107:43-57.
- [CrossRef] [Google Scholar]
- Network Pharmacology-Based Identification of Potential Targets of Lonicerae japonicae Flos Acting on Anti-Inflammatory Effects. Biomed Res. Int.. 2021;2021:1-18.
- [CrossRef] [Google Scholar]
- NRF2 as a regulator of cell metabolism and inflammation in cancer. Carcinogenesis. 2020;41:405-416.
- [CrossRef] [Google Scholar]
- Hispidulin Inhibits Mast Cell-Mediated Allergic Inflammation through Down-Regulation of Histamine Release and Inflammatory Cytokines. Molecules. 2019;24:2131.
- [CrossRef] [Google Scholar]
- Hederagenin, a major component of Clematis mandshurica Ruprecht root, attenuates inflammatory responses in RAW 264.7 cells and in mice. Int. Immunopharmacol.. 2015;29:528-537.
- [CrossRef] [Google Scholar]
- The anti-inflammatory function of high-density lipoprotein in type II diabetes: A systematic review. J. Clin. Lipidol.. 2017;11:712-724.e5.
- [CrossRef] [Google Scholar]
- EGFR Inhibition Blocks Palmitic Acid-induced inflammation in cardiomyocytes and Prevents Hyperlipidemia-induced Cardiac Injury in Mice. Sci. Rep.. 2016;6:24580.
- [CrossRef] [Google Scholar]
- Comprehensive comparison on antioxidant properties and UPLC-Q-TOF/MS-based metabolomics discrimination between Gentiana veitchiorum and G. szechenyii. Arab. J. Chem.. 2024;17:105695
- [CrossRef] [Google Scholar]
- Determination of polysaccharides content of Gentiana farreri from different producing areas based on anthrone-sulfuric acid method. China Journal of Chinese Materia Medica. 2014;39:2774-2776.
- [Google Scholar]
- Effects of Shiwei Longdanhua formula on LPS induced airway mucus hypersecretion, cough hypersensitivity, oxidative stress and pulmonary inflammation. Biomed. Pharmacother.. 2023;163:114793
- [CrossRef] [Google Scholar]
- Network pharmacology and molecular docking combined with widely targeted metabolomics to elucidate the potential compounds and targets of Euphorbia helioscopia seeds for the treatment of pulmonary fibrosis. Comput. Biol. Med.. 2023;160:107007
- [CrossRef] [Google Scholar]
- Hispidulin: A promising flavonoid with diverse anti-cancer properties. Life Sci.. 2020;259:118395
- [CrossRef] [Google Scholar]
- Mechanism of anti-hyperuricemia of isobavachin based on network pharmacology and molecular docking. Comput. Biol. Med.. 2023;155:106637
- [CrossRef] [Google Scholar]
- Investigation of the active components and mechanisms of Schisandra chinensis in the treatment of asthma based on a network pharmacology approach and experimental validation. Food Funct.. 2020;11:3032-3042.
- [CrossRef] [Google Scholar]
- Comparative analysis of eight active components in four Tibetan medicinal materials of Gentianaceae from Qinghai province. Chin. J. Pharm. Anal.. 2023;43:1866-1875.
- [Google Scholar]
- Interleukin-6 in SARS-CoV-2 induced disease: Interactions and therapeutic applications. Biomed. Pharmacother.. 2022;145:112419
- [CrossRef] [Google Scholar]
- The PTGS2/COX2-PGE 2 signaling cascade in inflammation: Pro or anti? A case study with type 1 diabetes mellitus. Int. J. Biol. Sci.. 2023;19:4157-4165.
- [CrossRef] [Google Scholar]
- IL-17 and IL-17-producing cells in protection versus pathology. Nat. Rev. Immunol.. 2023;23:38-54.
- [CrossRef] [Google Scholar]
- Targeting IL-17 and TH17 cells in chronic inflammation. Nat. Rev. Drug Discov.. 2012;11:763-776.
- [CrossRef] [Google Scholar]
- Synthesis, characterization, anti-inflammatory evaluation, molecular docking and density functional theory studies of metal based drug candidate molecules of tenoxicam. Results Chem.. 2021;3:100111
- [CrossRef] [Google Scholar]
- Wood-cultivated ginseng exerts anti-inflammatory effect in LPS-stimulated RAW264.7 cells. Int. J. Biol. Macromol.. 2018;116:327-334.
- [CrossRef] [Google Scholar]
- Animal Models of Inflammation for Screening of Anti-inflammatory Drugs: Implications for the Discovery and Development of Phytopharmaceuticals. Int. J. Mol. Sci.. 2019;20:4367.
- [CrossRef] [Google Scholar]
- Pundalik, S., Hanumappa, K.R., Giresha, A.S., Urs, D., Rajashekarappa, S., Muniyappa, N., Jamballi G, M., Kuaramkote Shivanna, D., S Meti, R., Anekere Dasappa Setty, S., Thippegowda, P.B., Krishnappa, D.K., 2022. Corosolic Acid Inhibits Secretory Phospholipase A2IIa as an Anti-Inflammatory Function and Exhibits Anti-Tumor Activity in Ehrlich Ascites Carcinoma Bearing Mice. J Inflamm Res Volume 15, 6905–6921. https://doi.org/10.2147/JIR.S383441.
- Integrative applications of network pharmacology and molecular docking: An herbal formula ameliorates H9c2 cells injury through pyroptosis. J. Ginseng Res.. 2023;47:228-236.
- [CrossRef] [Google Scholar]
- Autophagy modulates the levels of inflammatory cytokines in macrophages induced by lipopolysaccharide. Chin. J. Cell. Mol. Immunol.. 2017;33:581-585.
- [Google Scholar]
- Imaging Agents for PET of Inflammatory Bowel Disease: A Review. J. Nucl. Med.. 2023;64:1858-1864.
- [CrossRef] [Google Scholar]
- Divergent functions of NLRP3 inflammasomes in cancer: a review. Cell Commun. Signaling. 2023;21:232.
- [CrossRef] [Google Scholar]
- Gentiopicroside ameliorates acute inflammatory kidney injury in a mouse model induced by LPS through hampering NF‐κB, AP‐1 and IRF3 pathways. FASEB J.. 2022;36
- [CrossRef] [Google Scholar]
- Hederagenin Suppresses Inflammation and Cartilage Degradation to Ameliorate the Progression of Osteoarthritis: An In vivo and In vitro Study. Inflammation. 2023;46:655-678.
- [CrossRef] [Google Scholar]
- Low-Dose Aspirin Augments the Anti-Inflammatory Effects of Low-Dose Lithium in Lipopolysaccharide-Treated Rats. Pharmaceutics. 2022;14:901.
- [CrossRef] [Google Scholar]
- Targeting inflammation in atherosclerosis — from experimental insights to the clinic. Nat. Rev. Drug Discov.. 2021;20:589-610.
- [CrossRef] [Google Scholar]
- Network pharmacology and experimental verification based research into the effect and mechanism of Aucklandiae Radix–Amomi Fructus against gastric cancer. Sci. Rep.. 2022;12:9401.
- [CrossRef] [Google Scholar]
- Sun, J., 2012. Studies on the Chemical Constituents and Bioactivity from Gentiana farreri Balf.f.
- Flavonoids with anti-inflammatory activities from Daphne giraldii. Arab. J. Chem.. 2021;14:102962
- [CrossRef] [Google Scholar]
- Anti-inflammatory chemical constituents of Flos Chrysanthemi Indici determined by UPLC-MS/MS integrated with network pharmacology. Food Funct.. 2020;11:6340-6351.
- [CrossRef] [Google Scholar]
- The interactions of oxidative stress and inflammation with vascular dysfunction in ageing: the vascular health triad. Age (Omaha). 2013;35:705-718.
- [CrossRef] [Google Scholar]
- Anti-inflammatory Effects of Sweroside on LPS-Induced ALI in Mice Via Activating SIRT1. Inflammation. 2021;44:1961-1968.
- [CrossRef] [Google Scholar]
- Potential hepatoprotective effects of Cistanche deserticola Y.C. Ma: Integrated phytochemical analysis using UPLC-Q-TOF-MS/MS, target network analysis, and experimental assessment. Front. Pharmacol.. 2022;13:1-21.
- [CrossRef] [Google Scholar]
- Effective materials and mechanisms study of Tibetan herbal medicine Lagotis integra W. W. Smith treating DSS-induced ulcerative colitis based on network pharmacology, molecular docking and experimental validation. J. Ethnopharmacol.. 2023;301:115800
- [CrossRef] [Google Scholar]
- A Comprehensive Review of Non-Steroidal Anti-Inflammatory Drug Use in The Elderly. Aging Dis.. 2018;9:143.
- [CrossRef] [Google Scholar]
- Hypoglycemic effect and active ingredients screening of Isodon Japonicus based on network pharmacology and experimental validation. Arab. J. Chem.. 2023;16:105108
- [CrossRef] [Google Scholar]
- Chemical constituents from Gentiana farreri Balf.f. Chin. Tradit. Patent Med.. 2012;34:506-508.
- [Google Scholar]
- Isoorientin Induces Apoptosis and Autophagy Simultaneously by Reactive Oxygen Species (ROS)-Related p53, PI3K/Akt, JNK, and p38 Signaling Pathways in HepG2 Cancer Cells. J. Agric. Food Chem.. 2014;62:5390-5400.
- [CrossRef] [Google Scholar]
- Isoorientin attenuates lipopolysaccharide-induced pro-inflammatory responses through down-regulation of ROS-related MAPK/NF-κB signaling pathway in BV-2 microglia. Mol. Cell. Biochem.. 2014;386:153-165.
- [CrossRef] [Google Scholar]
- Antinociceptive effect of isoorientin against neuropathic pain induced by the chronic constriction injury of the sciatic nerve in mice. Int. Immunopharmacol.. 2019;75:105753
- [CrossRef] [Google Scholar]
- Isoalantolactone protects against ethanol-induced gastric ulcer via alleviating inflammation through regulation of PI3K-Akt signaling pathway and Th17 cell differentiation. Biomed. Pharmacother.. 2023;160:114315
- [CrossRef] [Google Scholar]
- Molecular mechanism of the anti-inflammatory effects of Sophorae Flavescentis Aiton identified by network pharmacology. Sci. Rep.. 2021;11:1005.
- [CrossRef] [Google Scholar]
- Network Pharmacology Identifies the Mechanisms of Action of Shaoyao Gancao Decoction in the Treatment of Osteoarthritis. Med. Sci. Monit.. 2019;25:6051-6073.
- [CrossRef] [Google Scholar]
- Isoorientin: A dietary flavone with the potential to ameliorate diverse metabolic complications. Pharmacol. Res.. 2020;158:104867
- [CrossRef] [Google Scholar]
Appendix A
Supplementary data
Supplementary data to this article can be found online at https://doi.org/10.1016/j.arabjc.2024.106085.
Appendix A
Supplementary data
The following are the Supplementary data to this article:Supplementary Data 1
Supplementary Data 1







