Synthesis of new 1,2,3-triazole linked benzimidazolidinone: Single crystal X-ray structure, biological activities evaluation and molecular docking studies
⁎Corresponding author. haalghulikah@pnu.edu.sa (Hanan Al-Ghulikah)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
A novel series of 1,2,3-triazole-benzimidazolidinone hybrid derivatives were designed and synthesized via click reaction, between various aryl azide and a terminal alkyne bearing a benzimidazolidinone moiety. All newly synthesized compounds, were efficiently characterized using 1H NMR, 13C NMR and HRMS. Furthermore, the structure of one precursor 5b was supported by single crystal X-ray diffraction. All synthesized derivatives have been evaluated for their antimicrobial and anti-inflammatory activities. Biological activity tests exhibited that the target structures demonstrate that compounds 5a, 5b and 5f have a high antibacterial activity especially derivative 5b. Besides, the in vitro antifungal results revealed that the strongest inhibition recorded to compound 5b in comparison to other products against A. brasiliensis, A. fumigatus and C. albicans. Biological activity evaluation indicated that the synthesized compounds possess moderate anti-inflammatory effects. The most effective compound in terms of efficacy and potency was 5a. Molecular docking simulation was used to investigate the most active compounds' probable binding mechanisms in order to provide a plausible explanation for their biological activity.
Keywords
1,2,3-triazole-benzimidazolidinone
CuAAC
Antimicrobial
Anti-inflammatory
Docking
1 Introduction
Benzimidazolone present a crucial heterocyclic building blocks that attracted special attention for their pronounced therapeutic applications. The majority of the studies show that benzimidazolone compounds exhibit a variety of pharmacological and biological properties including anti-HIV (Pribut et al., 2019), antinociceptive (Intagliata et al., 2020), cytotoxic (Banerji et al., 2015), antitrichinellosis (Mavrova et al., 2005), antiproliferative (El-Naggar et al., 2018), antituberculosis (Jagannath et al., 2021), antioxidant (Ibrahim et al., 2021) and antibacterial (Saber et al., 2021a).
As reported in literature, authors are interested currently in creating bioactive compounds with good pharmacokinetics features (Chamani et al., 2006), various drug administration (Atena et al., 2021), diversity of drug targeting (Nooshin et al., 2016), potent bioavailability (Narges et al., 2022), efficient pharmacological properties (Maryam et al., 2020), broad spectrum (Najmeh et al., 2021, Sattar et al., 2022), low toxicity, better curative impact (Reza et al., 2022), less adverse effects, have been showing more and more range of clinical applications for the treatment of different diseases (Hamid et al., 2022). In particular, many exemples of drug molecules containing triazole ring are discribed including Triadimefon (Paredes-Zúñiga et al., 2019), Etizolam (Nielsen et al., 2020), Hexaconazole (Worthington, 1991),Triazolam (Miyata et al., 2014), Rizatriptan (Mujahid et al., 2021), Ribavirin (Khalili et al., 2020), Propiconazole (Valadas et al., 2019), Myclobutanil (Fonseca et al., 2020) Rufinamide (Panebianco et al., 2020), Fluconazole (Lu et al., 2021), Carboxyamidotriazole (Shi et al., 2022), Tazobactum (Tamma et al., 2021) Fig. 1.
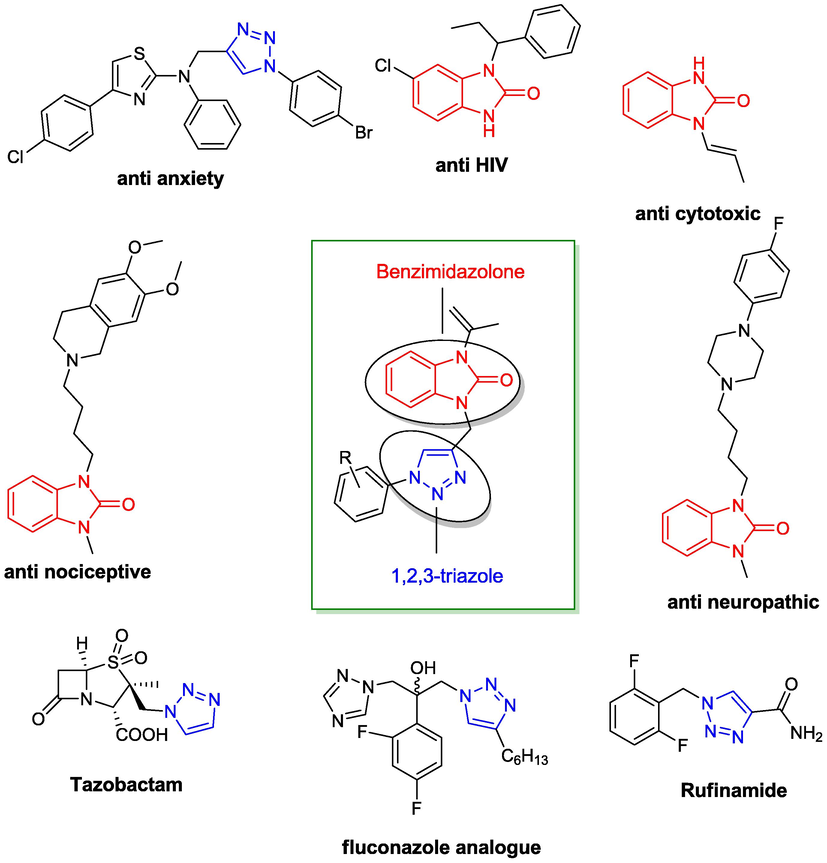
- Exemples of actif drugs containing triazole and benzimidazolidinone scaffold.
Furthermore, the introducing of a triazole ring connected to different pharmacophore fragments can produce new bi/polyfunctional drug molecules. Indeed, 1,2,3-triazole hybrid have attracted the interest of medicinal chemists due to their broad spectrum of applications such as anti-viral (Saroha et al., 2022), antitubercular (Harish, 2021), antimicrobial (Poonia et al., 2022), anti-HIV-1 (Feng et al., 2021), antialzheimer (Zahra et al., 2019), cholinesterase inhibitors (Rania et al., 2021) and antiproliferative agents (Arif et al., 2022). Therefore, the benzimidazolone linked triazole moiety has gained significant attention thanks to their biological properties (Saber et al., 2021b).
Furthermore, the synthesis of 1,2,3-triazole has been extensively researched for its pharmacological effects such as neuroprotective agents (Bozorov et al., 2019, Duarte et al., 2017), anti anxiety-anti inflammatory (Ankali et al., 2021) antibacterial-antileishmanial (Çelik et al., 2021), antichagasic agents (Silva et al., 2021). Huisgen (Rolf et al., 1967) first outlined the reaction between azides and acetylenes as a way of synthesizing these heterocyclic molecules. Due to the high activation energy, these reactions necessitate a high temperature and a long reaction time, in addition to the creation of a mixture of 1,4- and 1,5-regioisomers (Ali et al., 2016). Cu catalyzed azide-alkyne cycloaddition process (CuAAC), also known as the Click reaction, was reported in 2002 to have 100 % regioselectivity (only the 1,4-disubstituted regioisomer is obtained) and excellent yield under mild, safe reaction conditions (Darroudi et al., 2021, Nemallapudi et al., 2021).
Due to the importance of these heterocyclic scaffolds and in connection with these findings, we are interested to prepare a new series of 1,2,3-triazole linked benzimidazolone and evaluating in vitro their biological properties including antimicrobial and anti-inflammatory activities. Significant findings have been made and the structure–activity relationship of these synthesized derivatives was also discussed and further demonstrated by molecular docking simulation.
2 Results and discussion
2.1 Synthesis
The synthesis of hybrid molecules was carried out in four steps. Our synthetic route begins by the preparation of the dipolarophile 3 obtained by propargylation reaction between propargyl bromide and 1-(prop-1-en-2-yl)-1H-benzo[d] imidazol-2(3H)-one 2 which has been prepared from o-phenylenediamine (OPDA) and ethylacetoacetate as starting matérials (Scheme 1). The propargylation of 2 offers the appropriate terminal alkyne 3 in 80 % yield. The 1H NMR spectrum of N-propargylated compound 3, revealed the disappearance of NH signal at δH 10.87 ppm and the appearance of two new characteristic signals at δH 4.69 ppm and δH 2.31 ppm, assigned to the N-CH2 and the alkyne protons of the propargyl unit, respectively.
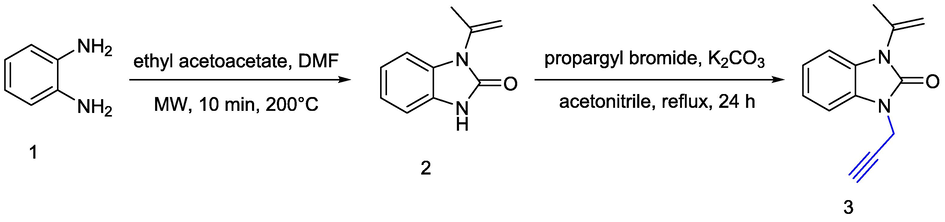
- General method for the synthesis of alkyne 3.
In parallel, aryl azides 4a-f were subjected to our “step-by-step” synthetic procedure, starting from the preparation of diazonium salt by reacting of various aniline with NaNO2 (sodium nitrite) following via nucleophilic aromatic substitution reaction using NaN3 (sodium azide) in situ. We have continued our work to synthesis 1,2,3-triazole by the 1,3-dipolar cycloaddition (CuAAC) starting from 1-(prop-1-en-2-yl)-3-(prop-2-yn-1-yl)-1H-benzo[d]imidazol-2(3H)-one 3 as a dipolarophilic system and aromatic azide 4a-f (Kamalraj et al., 2008) using a catalytic amount of Copper(I) iodideto generate a novel series of 1,2,3-triazole derivatives 5a-f in good yield (Scheme 2).
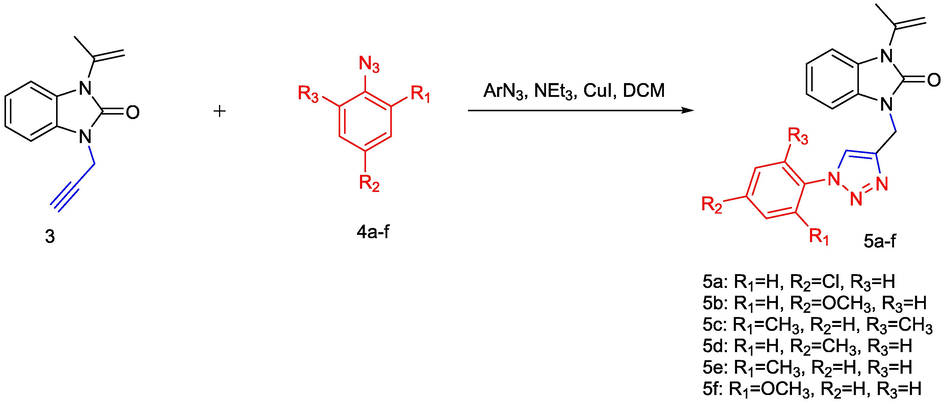
- General method for the synthesis of triazole derivatives 5a-f.
The physical properties of these structures are shown in Table 1.
| Entry | Compd. | Substituent | Yields %a | ||
|---|---|---|---|---|---|
| R1 | R2 | R3 | |||
| 1 | 5a | H | Cl | H | 74 |
| 2 | 5b | H | OCH3 | H | 68 |
| 3 | 5c | CH3 | H | CH3 | 69 |
| 4 | 5d | H | CH3 | H | 70 |
| 5 | 5e | CH3 | H | H | 72 |
| 6 | 5f | OCH3 | H | H | 68 |
A proposed reaction mechanism for the preparation of 1,2,3-triazole ring can be introduced in Scheme 3.
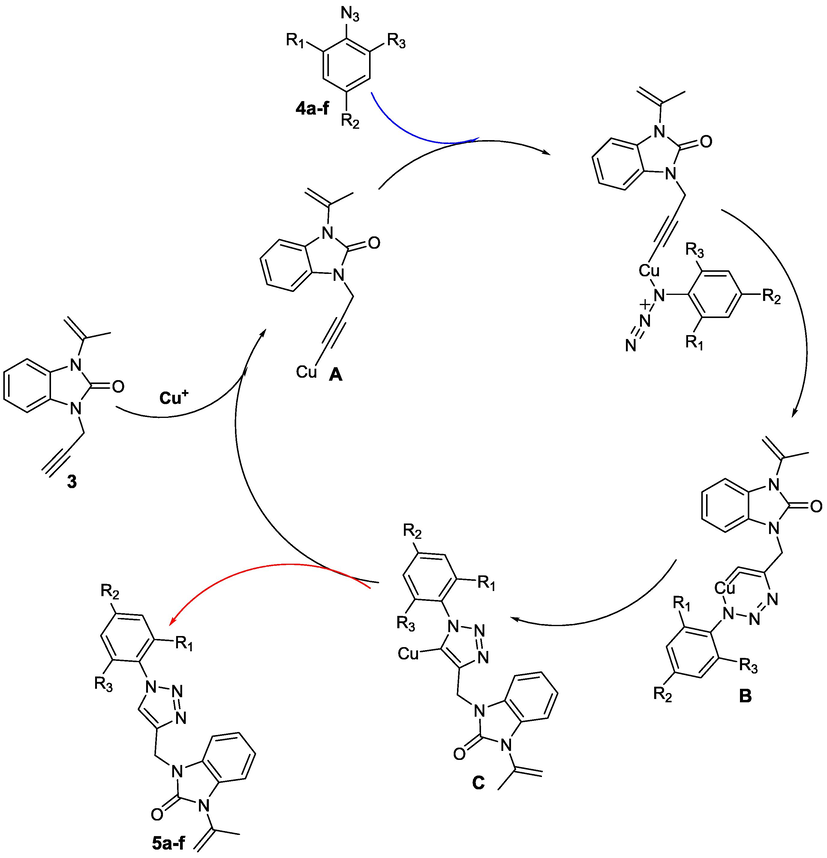
- Proposed mechanism for synthesis of 1,2,3-triazole derivatives 5a-f.
The structures of 1,2,3-triazole prepared 5a-f were fully described by analytical techniques (1H NMR, 13C NMR, RX, HRMS).1H NMR spectrum of compound 5c, showed a characteristic triazolyl proton as a singlet at δH 7.63 ppm due to inductive effect. Furthermore, a sharp singlet assigned to methylene protons was revealed at δH 5.31 ppm and two a doublet of doublet exhibited in the range δH 5.21–5.36 ppm attributed to methylene (CH2 = ) and a triplet at δH 2.23 ppm corresponding to CH3.
The analyse of 13C NMR spectrum of the cycloadducts5a-f displayed two characteristics signals corresponding to C-5 and the quaternary C-4which appear at δC121.8 ppm and δC137.9 ppm respectively. All these observations confirm clearly the regioselectivity of the formation of 1,4-disubstituted 1,2,3-triazole scaffold. Furthermore, the structure and the regioselectivity were performed unambiguously by X-ray diffraction study applied on a single-crystal corresponding to compound 5b as depicted in Fig. 2.
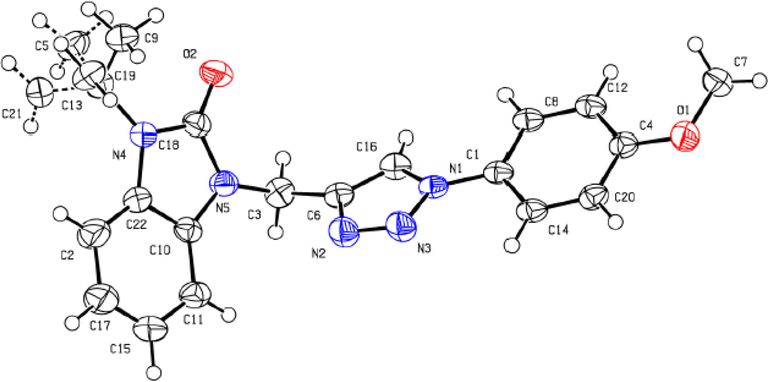
- Ortep diagram of the compound 5b.
2.2 X-ray crystallographic study
Single light yellow plate crystals of 5b were used as supplied. A suitable crystal with dimensions 0.47 × 0.13 × 0.05 mm3 was selected and mounted on a nylon loop in perfluoroether oil on aXcalibur, Atlas, Gemini ultra diffractometer. The crystal was kept at a steady T = 150.00(10) K during data collection. The structure was solved with the ShelXT (Dolomanov et al., 2009) solution program using dual methods and by using Olex2 (Sheldrick et al., 2015a) as the graphical interface. The model was refined withShelXL 2018/3 (Sheldrick et al., 2015b) using full matrix least squares minimization on F2.
2.2.1 Crystal data
C20H19N5O2, Mr = 361.40, monoclinic, I2/a (No. 15), a = 16.4233(16) Å, b = 5.2305(5) Å, c = 40.413(6) Å, b = 93.973(11)°, a = g = 90°, V = 3463.3(7) Å3, T = 150.00(10) K, Z = 8, Z' = 1, m(Cu Ka) = 0.758, 15,059 reflections measured, 3076 unique (Rint = 0.0494) which were used in all calculations. The final wR2 was 0.1231 (all data) and R1 was 0.0451 (I ≥ 2 s(I)).
2.3 Biological activity and molecular modeling
2.3.1 Antibacterial activity
All the newly synthesized 1,2,3-triazole linked benzimidazolidinones derivatives 5a-f were assessed in vitro against a variety of two Gram-positive bacteria (S. aureus and B. subtilis) and two Gram-negative bacteria (S. typhi and E. coli). By measuring the inhibition zone using well agar difusion assay the activity of all the investigated compounds was compared to that of Ampicillin, which was utilized as a standard reference antibiotic (in mm). The inhibiting properties against these four strains are given in Table 2. It was found that the newly generated compounds 5a-f have exerted globally moderate to good inhibitory activity against all of tested bacterial strains (IZ = 8 mm to 23 mm) compared to the reference antibiotic. In detail and starting with the inhibition effect towards gram positive bacteria; against S. aureus, based on zones of inhibition results and a part from the nature of the substituents in the para- and/or ortho-positions of the terminal phenyl, derivative 5f by its ortho-methoxy group showed the highest activity (IZ = 19 mm) compared to the other compounds and Ampicillin (IZ = 17 mm) followed by compound 5b with a methoxy group in the para position which displayed a zone of inhibition value equal to that of Ampicillin while 5c (R = 2,6-diCH3) and 5e (R = 2CH3) showed the lowest antibacterial effects (IZ = 9 mm, IZ = 8 mm respectively). This observation indicates the importance of methoxy groups in addition to triazole and benzimidazolidinone moieties for the S. aureus inhibition and the weak effect of methyl groups. Towards B. subtilis, derivatives 5b (IZ = 19 mm) with its 4-OCH3, 5a (IZ = 18 mm) by its 4-Cl and 5f (IZ = 17 mm) through its 2-OCH3 groups exhibited the highest antibacterial potentials than the reference antibiotic (IZ = 14 mm) and other analogues. Furthermore, with regard to the Gram-negative bacteria: against S. typhi 5b (IZ = 22 mm) was found to be the most active compound compared to its analogs followed by compounds 5a (IZ = 21 mm) and 5f (IZ = 20 mm). So, another time these three compounds showed greater inhibition potential than Ampicillin (IZ = 15 mm). In the same context towards E. coli, derivative 5b besides to 5a showed the highest antibacterial values followed by compound 5f as Table 2 showed. Thus, based on the above findings, we can be inferred that the hybrid molecules combining the two fragments: 1-aryl-1H-1,2,3-triazole and benzimidazolidinone are considered to be a main skeleton for the design of new antibacterial compounds.
| Standard and Test compound |
Zone of inhibition in mm | |||
|---|---|---|---|---|
| S. aureus | B. subtilis | S. typhi | E. coli | |
| 5a:4Cl | 15 | 18 | 21 | 23 |
| 5b: 4OCH3 | 17 | 19 | 22 | 23 |
| 5c: 2,6-diCH3 | 9 | 11 | 14 | 13 |
| 5d:4CH3 | 10 | 10 | 12 | 13 |
| 5e: 2CH3 | 8 | 10 | 12 | 12 |
| 5f:2OCH3 | 19 | 17 | 20 | 22 |
| Ampicillin | 17 | 14 | 15 | 13 |
2.3.2 Molecular docking for antibacterial activity
A Molecular docking test is the finest approach to study the binding affinity of ligands against target protein to more explain the antibacterial activity (Horchani et al, 2020, Horchani et al, 2022a). To estimate the optimal conformational position within the active site of the targeted protein, all of the produced compounds 5a-f were docked against: a crystal structure of a Gram-positive bacteria « Threonine synthase from B. subtilis ATCC 6633 » and a crystal structure of Gram-negative bacteria « MenB-1,4-dihydroxy-2-naphthoyl-CoA synthase from E. coli ATCC 25922 ». All the generated docked complexes were performed and analyzed on the basis of minimum energy values (kcal/mol) and hydrogen/hydrophobic interactions. All the created docked complexes were used to perform and analyze on the basis of minimum energy values (kcal/mol) and hydrogen/hydrophobic interactions.
From the docking results, as depicted in Table 3and Fig. 3, against B. subtilis,compound 5b having an interesting binding energy value is involved in two hydrogen bonds through its benzimidazolidinone and triazole fragment with THR83 and THR86 respectively. Additionally, a Pi-cation interaction is postulated with LYS59 by the para-methoxyphenyl group besides the appearance of some hydrophobic interactions with residues: PHE132 and ILE240. Thus, from the theoretical point of view, based on studies of molecular docking, the complex Threonine synthase-Ligand becomes stable by the formation of interactions between the amino acids forming the active site and the tested molecule, this is the case of our study by noticing the interactions formed and especially the intermolecular hydrogen bonds.
| Compound | Free binding energy of pdb: 6CGQ (kcal mol−1) | Free binding energy of pdb: 3 T88 (kcal mol−1) |
|---|---|---|
| 5a | −7.8 | −7.8 |
| 5b | −7.8 | −7.9 |
| 5c | −7.4 | −7.6 |
| 5d | −7.2 | −7.7 |
| 5e | −7.6 | −7.7 |
| 5f | −7.4 | −7.8 |
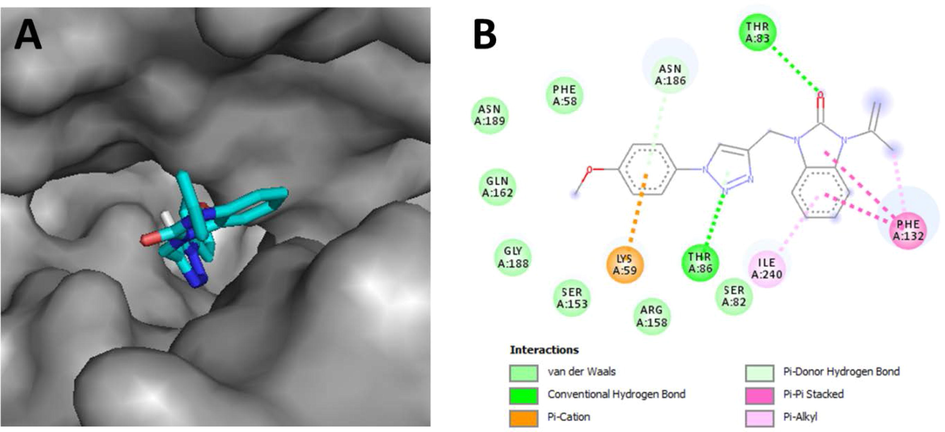
- A) Top view of the accessible surface of Threonine synthase from B. subtilis ATCC 6633 (PDB: 6CGQ) active site. B) Schematic representation of different interactions of 5bwithin the catalytic cavity.
Concerning the complex: E. coli-ligand, docking studies predicted that compound 5b would be the most active, with the lowest energy value (7.80 kcal/mol) when compared to other derivatives. 5b through its triazole pharmacophore showed H-bonding interaction with the side chain of GLN88 and SER161. The 1-isopropenyl-2-benzimidazolidinone fragment in turn displayed a Pi-Sigma interaction withVal108, Alkyl and Pi-Alkyl with LEU106 and Pi-Donor hydrogen bond with TYR97. 5b via its 4-methoxyphenyltriazole moiety exhibited a Pi-Alkyl interaction with VAL159 and Pi-Donor hydrogen bonds with amino acids: THR155 and SER161. Fig. 4. The results of the antibacterial assays are in accordance with the results from the molecular modeling calculations excellently. Thus, the 1,2,3-triazole linked benzimidazolidinone derivative is able to enter the enzyme pocket by showing significant binding interactions.
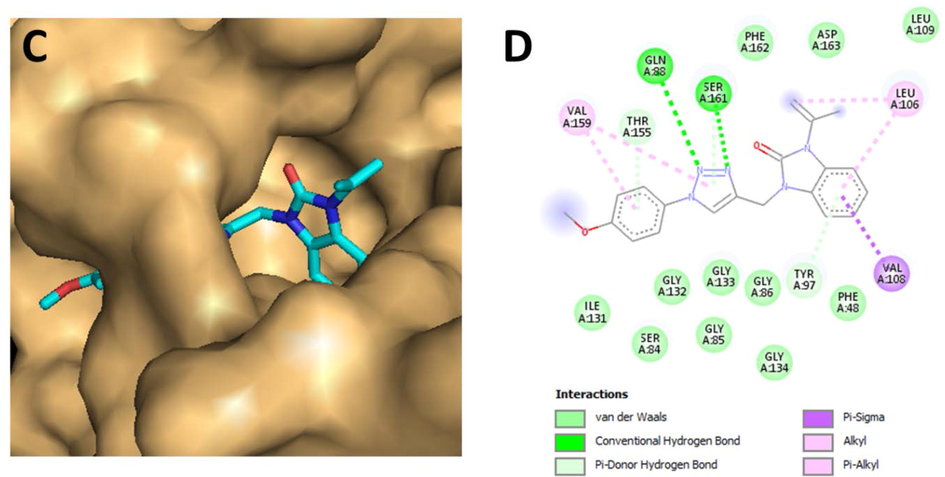
- C) Top view of the accessible surface of Escherichia coli (PDB: 3 T88) active site. D) Schematic representation of different interactions of 5bwithin the catalytic cavity.
2.3.3 Antifungal activity
Using the diffusion method, all newly synthesized compounds were inspected for antifungal activity against two fungi (A. fumigatus and A. brasiliensis) and two yeast strains (C. albicans and C. neoformans) (Murray et al., 1995). Antifungal activity of compounds was compared with the nystatin reference and the results of this study were presented in Table 4. The inhibition zone of strains ranged from 9 mm to 27 mm for fungi and 11 mm to 20 mm for yeast. These results show that more compounds show stronger inhibition against fungal strains. More specifically, the compound 5b with methoxy-groups at the para position of phenyl ring of benzimidazolone exhibit the strongest inhibition compared to other products against A. brasiliensis and A. fumigatus (between 27 mm and 26 mm), which make them even more efficient than the standard nystatin (Table 4). Moreover, the results of the biological activity showed also that compound 5a bearing the p-chloro substituted benzene moiety significantly increase the activity followed by compound 5f having 2-methoxyphenyl group while derivatives 5c, 5d and 5e methyl-substituted did not show good inhibition value against all strains used. In summary, may account for the observed antifungal activity may be due to the chemical structure of the produced compounds having methoxy group or halogen such as chlorine atom in addition to the 1,2,3-triazole linked benzimidazolidinone core.
| Standard and Test compound |
Zone of inhibition in mm | |||
|---|---|---|---|---|
| A. brasiliensis | A. fumigatus | C. albicans | C. neoformans | |
| 5a:4Cl | 26 | 24 | 17 | 19 |
| 5b: 4OCH3 | 27 | 26 | 20 | 17 |
| 5c: 2,6-diCH3 | 11 | 11 | 14 | 13 |
| 5d:4CH3 | 10 | 9 | 12 | 13 |
| 5e: 2CH3 | 10 | 10 | 12 | 11 |
| 5f:2OCH3 | 19 | 20 | 16 | 15 |
| Nystatin | 25 | 24 | 22 | 23 |
2.3.4 In vitro anti-Inflammatory activity
Protein denaturation is a well-known source of inflammation. When a protein is denatured, it loses its secondary and tertiary structures due to external stresses such heat, strong acids or bases, concentrations of inorganic salts, or organic solvents. The majority of biological proteins also lose their biological activity (Mizushima et al., 1968, Grant et al., 1970). In the current study, the inhibition of albumin denaturation method was used to assess the in vitro anti-inflammatory activity of all derivatives 5a-f and the reference drug Aspirin. As shown in Table 5, the results clearly indicate that some derivatives have significantly good inhibition. Besides to the tirazole tethered with benzimidazolidinone, the substituents introduced into the phenyl ring directly related to the triazole heterocycle have an effect on the activity. Indeed, compound 5a with chlorine atom displayed better anti-inflammatory potential at different concentrations compared to its analogues and with values very close to those of Aspirin. This observation demonstrates the importance of halogens in the anti-inflammatory potency in particular the chlorine atom. On the other hand, the 4-methxoy derivative 5b displayed good inhibitory values followed by 2-methxoy derivative 5f while the compounds 5c, 5d and 5e with methyl group showed moderate activity. We can conclude from the above findings that the mesomeric electron donor effect can enhance the anti-inflammation, this is the case of methoxy group and chlorine atom whereas the methyl group exerted just an inductive donor effect that's why did not show significant activity.
| Standard and Test compound |
Concentration µg/mL | ||||
|---|---|---|---|---|---|
| 200 | 400 | 600 | 800 | 1000 | |
| 5a:4-Cl | 57.22 | 64.75 | 65.12 | 67.75 | 73.89 |
| 5b: 4-OCH3 | 52.37 | 61.33 | 62.32 | 65.69 | 70.17 |
| 5c: 2,6-diCH3 | 09.04 | 12.47 | 18.27 | 22.28 | 24.34 |
| 5d:4-CH3 | 10.23 | 12.64 | 15.19 | 18.22 | 20.41 |
| 5e: 2-CH3 | 09.15 | 10.24 | 13.73 | 16.38 | 19.56 |
| 5f:2-OCH3 | 44.12 | 49.24 | 51.85 | 52.14 | 59.69 |
| Aspirin | 59.36 | 66.13 | 67.62 | 69.48 | 75.23 |
2.3.5 Molecular docking for anti-inflammatory activity
Our attention was next directed to assess the selectivity of our developed compounds 5a-f based on molecular docking by comprehending their docking with the inflammatory target receptor (PDB: 3IAZ). Table 6 demonstrates that derivative 5a has the lowest energy value (−7.90 kcal/mol) in comparison with its analogues even compared to the reference ligand Aspirin. According to Fig. 5, it was observed that the most active compound 5a bounded to the active via several interesting interactions. The triazole ring was in interaction with THR430, PRO593 and TYR660 conventional Hydrogen bond, Pi-Donor hydrogen bond, Pi-Alkyl and Pi-Pi T-shaped interactions respectively. Moreover, 5a through its benzimidazolidinone pharmacophore is involved in Alkyl and Pi-Alkyl interactions with VAL591. In addition, the 4-chlorophenyl moiety interacted with PRO655 by an Alkyl interaction. Inspired by the previous observations, molecular modeling agrees well with in vitro results of anti-inflammatory activity.
| Compound | Free binding energy of pdb: 3IAZ (kcal mol−1) |
|---|---|
| 5a | −7.9 |
| 5b | −7.5 |
| 5c | −7.6 |
| 5d | −7.4 |
| 5e | −7.5 |
| 5f | −7.7 |
| Aspirin | −6.3 |
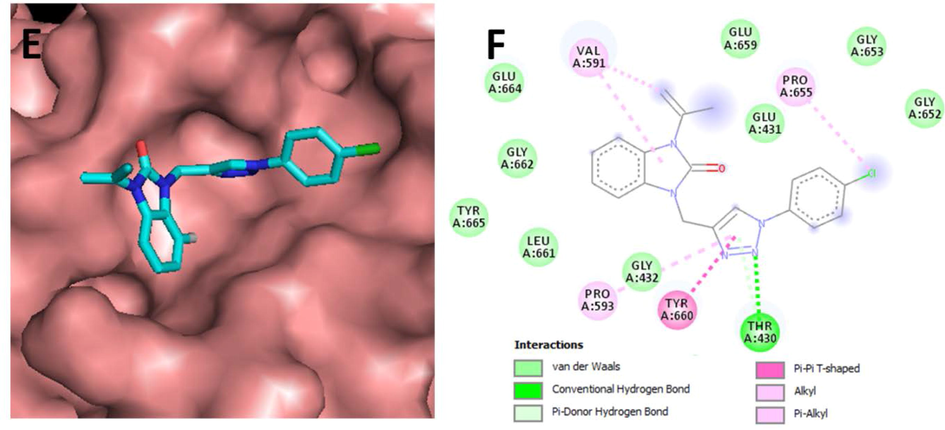
- E) Top view of the accessible surface of inflammatory protein (PDB: 3IAZ) active site. F) Schematic representation of different interactions of 5awithin the catalytic cavity.
2.3.6 ADMET and predicted physicochemical properties
ADMET properties are known as key steps and appropriate methodologies used in drug development and discovery processes (Horchani et al., 2022b, Ye et al., 2019). Furthermore, the physicochemical properties are important parameters of a compound that influence efficacy, safety, or metabolism and can be predicted using Lipinski’s rule of five (RO5), as summarized in Table 7. The rule of five (RO5) deals with the reliance of active compounds and defines the eight pharmacokinetics parameters named as the molecular weight, H‐bond donors, H‐bond acceptors, log P, number of rotatable bonds, topological polar surface area, molecular volume, percentage of absorption. So, these pharmacokinetic parameters are associated with the acceptable aqueous solubility. For more details, the predicted values of many properties categorized as molecular weight describe the size of the compound. In addition, the value of log P corresponds to the lipophilicity of the molecules which is matched to the solubility of the drug molecule in the aqueous medium. Accordingly, the higher the solubility, the higher the activity of the therapeutic agents. Moreover, the parameters of the H‐bond donor and H‐bond acceptor signalize the quantification of all atoms (N, O) besides to their efficiency in the formation of the hydrogen bond. Thus, the results obtained from the ADMET analysis with ADMETlab 2.0 software unveiled that the predicted compounds 5a-f fully obeyed Lipinski’s rule of five. These findings suggest that these synthesized analogues are feasible in order to serve as a future biological candidates.
| Compd. No. | MWa | nHAb | nHDc | logP(o/w)d | nrotboe | TPSAf | MVg | %Absh |
|---|---|---|---|---|---|---|---|---|
| Acceptable Value | <500 | <10 | <5 | <5 | =<10 | <140 | 500 | 100 % |
| 5a | 365.100 | 6 | 0 | 4.350 | 4 | 57.640 | 355.575 | 89.11 |
| 5b | 361.150 | 7 | 0 | 3.274 | 5 | 66.870 | 366.450 | 85.92 |
| 5c | 358.190 | 6 | 1 | 3.497 | 4 | 64.420 | 377.162 | 86.77 |
| 5d | 345.160 | 6 | 0 | 3.969 | 4 | 57.640 | 357.660 | 89.11 |
| 5e | 345.160 | 6 | 0 | 3.984 | 4 | 57.640 | 357.660 | 89.11 |
| 5f | 361.150 | 7 | 0 | 3.288 | 5 | 66.870 | 366.450 | 85.92 |
3 Conclusion
In summary, we have designed a step-by-step synthetic route to build a novel series of 1,2,3-triazole linked benzimidazolidinone 5a-f which was synthesized in good yields, by click reaction. New hybrid compounds were efficiently characterized by 1H NMR, 13C NMR, HRMS, and the structure of precursor 5b was supported by X-ray crystallographic data. The synthesized compounds 5a-f were evaluated biologically against antimicrobial and anti-inflammatory activities. The biological studies reveal that synthesized compounds may be classified as antimicrobial agents. Biological activity tests showed also that the synthesized compounds have moderate to good activity against anti-inflammatory. In addition, molecular docking simulations confirmed that the most active compounds have been well fitted in the active site of target enzymes via desirable interactions.
4 Experimental section
4.1 Chemistry
4.1.1 General information
The purity of products was confirmed by TLC using aluminium sheets (silica gel 60 F254) purchased from Merck. 1H and 13C NMR spectra were recorded on Bruker AC-400 in deuteriochloroform or DMSO‑d6 solution. Chemical shifts are taken in ppm using TMS as an internal standard. High resolution mass spectra (HRMS) were recorded using Micromass LCT mass spectrometers (ESI technique, positive mode). Melting temperatures were determined in open glass capillaries and are not corrected. All chemical shifts values were reported in ppm and coupling canstants are taken in Hz. Different signals multiplicities are described as following: s (singlet), d (doublet), t (triplet), m (multiplet) etc. All reaction mixture were monitored by TLC and purified by column chromatography using silica gell (40–63 μm).
Reagent and commercial products were purchased from Sigma Aldrich without prior purification. The starting material (2) was formed according to the littérature. (Li et al., 2010).
4.1.2 General procedure a for the synthesis of 1-(prop-1-en-2-yl)-3-(prop-2-yn-1-yl)-1H-benzo[d]imidazol-2(3H)-one 3
To a solution of 1-(prop-1-en-2-yl)-1H-benzo[d]imidazol-2(3H)-one (2) (4 g, 0.023 mol) in acetonitrile (70 mL) was added potassium carbonate (3.87 g, 0.028 mol). The reaction mixture was refluxed at 70 °C for 30 min before addition 1.2 equiv. of propargyl bromide (3.33 g, 0.028 mol). Then, the residue was extracted with ethyl acetate (120 mL × 3). The organic extracts were dried and filtred. The crude was evaporated under vacum and recrystallized from cyclohexane to give 3 (4.75 g, 80 %).
4.1.2.1 1-(prop-1-en-2-yl)-3-(prop-2-yn-1-yl)-1H-benzo[d]imidazol-2(3H)-one (3)
Prepared, according to general procedure A, Yield: 80 %, off-white solid; mp 141–143 °C. Rf = 0.30 [EtOAc/cyclohexane 3:7]. 1H NMR (CDCl3, 400 MHz): δ2.22 (s, 3H, J = 0.8 Hz, CH3), 2.31 (t, 1H, J = 3.2 Hz, CH), 4.69 (d, 2H, J = 3.6 Hz, CH2), 5.20 (d, 1H, J = 0.8 Hz,=CH),5.35 (d, 1H, J = 0.8 Hz,=CH), 7.20–7.11 (m, 4H, Harom). 13C NMR (CDCl3, 75 MHz): δ20.1, 72.7, 30.1,76.6, 108.5, 109.1, 113.3, 121.7, 121,8, 128.6, 128.9, 137.9, 151.9.HRMS, (ESI) calcd C13H12N2NaO[M + Na]+: 235.0802, found 235.0804.
4.1.3 General procedure B for the synthesis of 1,2,3-triazole derivatives (5a-f)
To a mixture of alkyne 3 (400 mg, 1.88 mmol) in DCM (50 mL) and an equimolar amount of aryl azideswas added NEt3 (0.38 mL, 2.82 mmol) and CuI (0.5 mol%). The reaction mixture was stirred at rt for 24 h. The solution was extracted with dichloromethane (60 mL × 3) and dried using Na2SO4. After evaporation of the solvent under vacuum The crude was evaporated under vacuum and recrystallized from cyclohexane to give 5a-f.
4.1.3.1 1-((1-(4-chlorophenyl)-1H-1,2,3-triazol-4-yl)methyl)-3-(prop-1-en-2-yl)-1H-benzo[d]imidazol-2(3H)-one (5a)
Prepared, according to general procedure B, Yield; 74 %, white solid; mp 118–120 °C. Rf = 0.27 [EtOAc/cyclohexane 3:7]. 1H NMR (CDCl3, 400 MHz): δ 2.23 (s, 3H, CH3), 5.20 (d, 1H, =CH), 5.25 (s, 2H, CH2), 5.36 (d, 1H, J = 1,2 Hz, =CH), 7.11 (m, 3H, Harom), 7.33 (m, 1H, Harom), 7.45 (d, 2H, J = 9 Hz, Harom), 7.82 (d, 2H, J = 8.7 Hz, Harom), 8.05 (s, 1H, Htriazole). 13C NMR (CDCl3, 75 MHz): δ 20.2, 36.3,76.58,77.42, 93.7, 108.7, 109.1, 113.2, 120.9, 121.7, 122.0, 128.8, 128.9, 136.5, 137.8, 138.8, 152.5. HRMS, (ESI) calcd C19H16ClN5NaO [M + Na]+: 388.0134, found 388.0128.
4.1.3.2 1-((1-(4-methoxyphenyl)-1H-1,2,3-triazol-4-yl)methyl)-3-(prop-1-en-2-yl)-1H-benzo[d]imidazol-2(3H)-one (5b)
Prepared, according to general procedure B, Yield; 68 %, off-white solid;mp 130–132 °C. Rf = 0.27 [EtOAc/cyclohexane 3:7]. 1H NMR (CDCl3, 400 MHz): δ 2,24 (s, 3H, CH3), 3.84 (s, 3H, OCH3), 5.20 (s, 1H, =CH), 5.25 (s, 2H, CH2), 5.35 (s, 1H, =CH), 7.97 (d, 2H, J = 9 Hz, Harom), 7.10 (m, 3H, Harom), 7.35 (m, 1H, Harom), 7.57 (d, 2H, J = 9 Hz, Harom), 7.97 (s, 1H, Htriazole). 13C NMR (CDCl3, 75 MHz): δ 20.2, 36.3, 55.6, 108.7, 109.1, 113.2, 121.7, 122.0, 128.8, 128.9, 129.6, 130.7, 133.4, 137.8, 138.8, 152.5. HRMS, (ESI) calcd C20H19N5NaO2 [M + Na]+: 384.1224, found 384.1220.
4.1.4 1-((1-(2,6-dimethylphenyl)-1H-1,2,3-triazol-4-yl)methyl)-3-(prop-1-en-2-yl)-1H-benzo[d]imidazol-2(3H)-one (5c)
Prepared, according to general procedure B, Yield; 69 %, off-white solid; mp 122–124 °C. Rf = 0.28 [EtOAc/cyclohexane 3:7]. 1H NMR (CDCl3, 400 MHz): δ 1.94 (s, 6H, 2CH3), 2.23 (s, 3H, CH3), 5.21 (s, 1H, =CH), 5.31 (s, 2H, CH2), 5.35 (d, 1H, J = 1,2 Hz, =CH), 7.10 (m, 3H, Harom), 7.13(d,2H, J = 8.4 Hz, Harom),7.24 (m, 2H, Harom), 7.29 (m, 2H, Harom), 7.63 (s, 1H, Htriazole). 13C NMR (CDCl3, 75 MHz): δ 17.4, 20.2, 36.7, 108.7, 109.1, 113.1, 121.6, 121.8, 128.4, 128.9, 129.1, 130.1, 135.3, 135.7, 137.9, 152.6. HRMS, (ESI) calcd C21H21N5NaO [M + Na]+: 382.1637, found 382.1638.
4.1.4.1 1-(prop-1-en-2-yl)-3-((1-(p-tolyl)-1H-1,2,3-triazol-4-yl)methyl)-1H-benzo[d]imidazol-2(3H)-one (5d)
Prepared, according to general procedure B, Yield; 70 %, white solid; mp 137–139 °C. Rf = 0.28 [EtOAc/cyclohexane 3:7]. 1H NMR (CDCl3, 400 MHz): δ 2.24 (s, 3H, CH3), 2.39 (s, 3H, CH3), 5.23 (m, 3H, =CH and CH2), 5.36 (s, 1H, =CH), 7.09 (m, 3H, Harom), 7.27 (d, 2H, J = 8.7 Hz, Harom), 7.35 (d, 1H, J = 5.7 Hz, Harom), 7.56 (d, 2H, J = 8.4 Hz, Harom), 8.14 (s, 1H, Htriazole).13C NMR (CDCl3, 75 MHz): δ 20.2, 21.0, 36.3, 108.8, 109.1, 113.1, 120.4, 121.6, 121.9, 128.8, 129.0, 130.2, 137.9, 138.9, 152.6. HRMS, (ESI) calcd C20H19N5NaO4 [M + Na]+: 368.0482, found 368.0482.
4.1.4.2 1-(prop-1-en-2-yl)-3-((1-(o-tolyl)-1H-1,2,3-triazol-4-yl)methyl)-1H-benzo[d]imidazol-2(3H)-one (5e)
Prepared, according to general procedure B, Yield; 72 %, off-white solid; mp 108–110 °C. Rf = 0.26 [EtOAc/cyclohexane 3:7]. 1H NMR (CDCl3, 400 MHz): δ 2.19 (s, 3H, CH3), 2.23 (s, 3H, CH3), 5.21 (s, 1H, =CH), 5.29 (s, 2H, CH2), 5.35 (d, 1H, J = 1.2 Hz, =CH), 7.12 (m,3H, Harom), 7.28 (d, 1H, J = 1.2 Hz, Harom), 7.29 (m, 1H, Harom), 7.33 (m, 1H, Harom), 7.35 (m, 1H, Harom), 7.39 (m, 1H, Harom), 7.79 (s, 1H, Htriazole). 13C NMR (CDCl3, 75 MHz): δ 17.9; 20.2; 36.4; 108.8; 109.1; 113.2; 121.7; 121.9; 125.9, 126.8, 128.9, 129.1, 129.9, 131.5, 133.7, 137.9, 152.6. HRMS, (ESI) calcd C20H19N5NaO [M + Na]+: 368.1482, found 368.1482.
4.1.4.3 1-((1-(2-methoxyphenyl)-1H-1,2,3-triazol-4-yl)methyl)-3-(prop-1-en-2-yl)-1H-benzo[d]imidazol-2(3H)-one (5f)
Prepared, according to general procedure B, Yield; 68 %, off-white solid; mp 113–115 °C. Rf = 0.24 [EtOAc/cyclohexane 3:7]. 1H NMR (CDCl3, 400 MHz): δ 2.22 (s, 3H, CH3), 3.82 (s, 3H, OCH3), 5.33 (m, 4H, Harom), 7.06 (m, 5H, Harom), 7.36 (m, 2H, Harom), 7.72 (d, 1H, J = 8.0 Hz, Harom), 8.22 (s, 1H, Htriazole). 13C NMR (CDCl3, 75 MHz): δ 20.1, 36.3, 55.9, 109.2, 109.5, 112.2, 121.2, 121.7, 121.9, 122.3, 125.4, 127.6, 129.9, 130.2, 137.7, 151.1. HRMS, (ESI) calcd C20H19N5NaO2[M + Na]+: 384.1428, found 384.1424.
4.2 Biological activity
4.2.1 Antibacterial screening
By using the Agar Well Diffusion Assay technique, the antibacterial properties of the synthesized compounds were assessed against two Gram-positive bacteria, i.e., S. aureus and B. subtilis, and two Gram-negative bacteria, i.e., S. typhi and E. coli. These pathogenic strains were grown for 24 h at 37 °C on nutrient agar. In 10 mL of physiological medium, colonies were suspended, and mixed for 5 min, and the suspensions were calibrated to 0.5 McFarland standard turbidity.Muller Hinton Agar medium plates were covered with one millilitre of bacterial suspension and incubated for 30 min at 37 °C. The seeded agar plates were punched with wells that were approximately 6 mm in diameter, and each test substance was then put to the wells after being reconstituted in DMSO. For all of the testsubstances, DMSO served as the control. The plates were kept at room temperature for two hours to allow the chemicals to diffuse into the agar before the palates were incubated at 37 °C for 24 h. By using a digital calliper, the diameters of the clear zones of inhibition surrounding the sample were measured after 24 h.
4.2.2 Antifungal activity
Yeasts Strains: two yeast strains, C. albicans and C. neoformans, were cultivated on Sabouraud agar for 48 h at 37 °C (the ideal temperature for bacterial growth). The suspensions of pure colonies were then adjusted to 0.5 McFarland standard turbidity after being suspended in 10 mL of physiological medium and well stirred for 5 min. Plates of Sabouraud agar medium received one milliliter of yeast suspension, which was then incubated at 37 °C for 30 min. Then, using a sterile glassy borer, 6 mm diameter wells were drilled into the agar medium.The synthesized compounds were produced in DMSO (1 mg/mL) and added to the appropriate wells; as a control, DMSO was added to one of the wells. For 48 h, these plates were kept in an incubator at 37 °C to promote yeast growth. By using a digital calliper, the diameters of the clear zones of inhibition surrounding the sample were measured after 48 h.
Fungal Strains: two fungi strains, A. brasiliensis and A. fumigatus were cultured on inclined Sabouraud agar in Falcon tube 15 mL at 25 °C for 7 days (optimal temperature for their growing). The fungus strain's spore was measured at 106 spores/mL after being suspended in peptone water. After that, Sabouraud agar medium plates were covered with one millilitre of the fungal suspension and incubated at 25 °C for 30 min. Then, using a sterile glassy borer, 6 mm diameter wells were drilled in agar media. The synthesized compounds were produced in DMSO (1 mg/mL) and added to the appropriate wells; as a control, DMSO was added to one of the wells. For five days, these plates were kept in an incubator at 25 °C to promote fungi development. After five days, a digital caliper was used to measure the diameters of the clear zone of inhibition surrounding the sample.
4.2.3 In vitro anti-inflammatory activity
The anti-inflammatory activity of benzimidazolone derivatives 5a–f was determined as below:
Various concentrations of 2 mL of each compound was mixed individually with 3 mL of sodium phosphate buffer saline (pH 6.4) containing 0.2 mL of eggs albumin yielding a final volume of 5 mL. The pure blank does not contain the substrate, but will be added 3 mL of buffer solution. All compounds were re-suspended in the DMSO followed by dilution in the buffer in order that the DMSO does not exceed 1 %. Control was given a comparable volume of double-distilled water. The mixture was then heated at 70 °C for 5 min after being incubated at 37 °C for about 15 min and the absorbance was determined at 660 nm after cooling. To determine absorbance, aspirin (a standard medication) was used as positive control and as a reference medication and handled accordingly. Using the formula listed below, the % inhibition of protein denaturation was determined:
4.3 Molecular docking procedure
Molecular docking simulations were performed by Auto Dock 4.2 program package (Trott et al., 2010). The optimization of all the geometries of compounds was carried out using ACD (3D viewer) software (https://www.filefacts.com/acd3d-viewer-freeware-info). The three dimensional structure of PDB (PDB: 6CGQ) (Petronikolou et al., 2019) and (PDB: 3 T88) (Li et al., 2011) for the antibacterial activityand of PDB (PDB: 3IAZ) (Mir et al., 2009) for the anti-inflammatory potentialwere obtained from the RSCB protein data bank. First, the water molecules were eliminated and the missing hydrogens and Gasteiger charges were then added to the system during the preparation of the receptor input file. Next, AutoDock Tools were used for the preparation of the corresponding ligand and protein files (PDBQT). Then, pre-calculation of the grid maps was performed using Auto Grid for saving a lot of time during docking. The visualization and analysis of interactions were performed using Discovery Studio 2017R2 (https://www.3dsbiovia.com/products/collaborative-science/biovia-discovery-studio/).
4.4 ADMET properties
To predict the pharmacokinetic properties: molecular weight, number of hydrogen-bond acceptors, Number of hydrogen-bond donors, octanol/water partition coefficient, number of rotatable bonds, topological polar surface area, molecular volume and percentage of absorption of all synthesized derivatives, the chemoinformatic tools (database): ADMETlab 2.0 (https://admetmesh.scbdd.com/service/evaluation/cal/) was used.
Acknowledgments
The authors extend their appreciation to Princess Nourah bint Abdulrahman University Researchers Supporting Project number (PNURSP2023R95), Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia.
Fundings
This research was funded by Princess Nourah bint Abdulrahman University Researchers Supporting Project number (PNURSP2023R95), Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- [Bmim]OH mediated Cu-catalyzed azide–alkyne cycloaddition reaction: a potential green route to 1,4-disubstituted 1,2,3-triazoles. Tetrahedron Lett.. 2016;57:5661-5665.
- [CrossRef] [Google Scholar]
- Synthesis and molecular docking of novel 1,3-Thiazole Derived 1,2,3-Triazoles and in vivo biological evaluation for their anti anxiety and anti inflammatory activity. J. Mol. Struct.. 2021;1236:130357
- [CrossRef] [Google Scholar]
- 1,2,3-Triazole Tethered Hybrid Capsaicinoids as Antiproliferative Agents Active against Lung Cancer Cells (A549) ACS Omega. 2022;36:32078-32100.
- [CrossRef] [Google Scholar]
- Oil-in-water nanoemulsions comprising Berberine in olive oil: biological activities, binding mechanisms to human serum albumin or holo-transferrin and QMMD simulations. J. Biomol. Struct. Dyn.. 2021;39:1029-1043.
- [CrossRef] [Google Scholar]
- Synthesis and cytotoxicity studies of 1-propenyl-1,3-dihydro-benzimidazol-2-one. J Chem. Biol.. 2015;8:73-78.
- [CrossRef] [Google Scholar]
- 1,2,3-Triazole-containing hybrids as leads in medicinal chemistry: A recent overview. Bioorg. Med. Chem.. 2019;27:3511-3531.
- [CrossRef] [Google Scholar]
- 1,2,3-triazole derivative: Synthesis, characterization, DFT, molecular docking study and antibacterial-antileishmanial activities. J. Indian Chem. Soc.. 2021;98:100105
- [CrossRef] [Google Scholar]
- Effect of n-alkyl trimethylammonium bromides on folding and stability of alkaline and acid-denatured cytochrome c: a spectroscopic approach. J. Colloid. Interface Sci.. 2006;297:561-569.
- [CrossRef] [Google Scholar]
- An experimental and mechanism study on the regioselective click reaction toward the synthesis of thiazolidinone-triazole. Heliyon.. 2021;7:e06113.
- [Google Scholar]
- Olex2: A complete structure solution, refinement and analysis program. J. Appl. Cryst.. 2009;42:339-341.
- [CrossRef] [Google Scholar]
- Apoptosis induction by 7-chloroquinoline-1,2,3-triazoyl carboxamides in triple negative breast cancer cells. Biomed. Pharmacother.. 2017;91:510-516.
- [CrossRef] [Google Scholar]
- Novel Thiazolidinone/Thiazolo[3,2-a] Benzimidazolone-Isatin conjugates as apoptotic anti-proliferative agents towards breast cancer: one-pot synthesis and in vitro biological evaluation. Molecules.. 2018;23:1420.
- [CrossRef] [Google Scholar]
- 1,2,3-Triazole hybrids with anti-HIV-1 activity. Arch. Pharm.. 2021;354:2000163.
- [CrossRef] [Google Scholar]
- Myclobutanil enantioselective risk assessment in humans through in vitro CYP450 reactions: Metabolism and inhibition studies. Food Chem. Toxicol.. 2020;128:202-211.
- [CrossRef] [Google Scholar]
- Stabilization of serum albumin by anti-inflammatory drugs. Biochem. Pharmacol.. 1970;19:715-722.
- [CrossRef] [Google Scholar]
- Glucokinase activity enhancement by cellulose nanocrystals isolated from jujube seed: A novel perspective for type II diabetes mellitus treatment (In vitro) J. Mol. Struct.. 2022;1269:133803
- [CrossRef] [Google Scholar]
- Coumarin-1,2,3-triazole hybrid molecules: an emerging scaffold for combating drug resistance. Curr. Top Med. Chem.. 2021;21:737-752.
- [CrossRef] [Google Scholar]
- New pyrazolo-triazolo-pyrimidine derivatives as antibacterial agents: design and synthesis, molecular docking and DFT studies. J. Mol. Struct.. 2020;1199:127007
- [CrossRef] [Google Scholar]
- Access to new Schiff bases tethered with pyrazolopyrimidinone as antibacterial agents: design and synthesis, molecular docking and DFT analysis. J. Mol. Struct.. 2022;1248:131523
- [CrossRef] [Google Scholar]
- Synthesis and In Silico Docking Study towards M-Pro of Novel Heterocyclic Compounds Derived from Pyrazolopyrimidinone as Putative SARS-CoV-2 Inhibitors. Molecules. 2022;27:5303.
- [CrossRef] [Google Scholar]
- Microwave-assisted synthesis of (3,5-disubstituted isoxazole)-linked benzimidazolone derivatives: DFT calculations and biological activities. Monatshefte Für Chemie - Chemical Monthly.. 2021;152:523-535.
- [CrossRef] [Google Scholar]
- Discovery of a Highly Selective Sigma-2 Receptor Ligand, 1-(4-(6,7-Dimethoxy-3,4-dihydroisoquinolin-2(1H)-yl)butyl)-3-methyl-1H-benzo[d]imidazol-2(3H)-one (CM398), with Drug-Like Properties and Antinociceptive Effects In Vivo. AAPS J.. 2020;22
- [CrossRef] [Google Scholar]
- Design, Synthesis, and Anti-Tubercular Studies of 10-(phenylsulfonyl)pyrimido[1,2-a]benzimidazol (10H) One Derivatives. New Innovations Chem. Biochem.. 2021;3:66-75.
- [CrossRef] [Google Scholar]
- One-pot synthesis and the fluorescent behavior of 4-acetyl-5-methyl-1,2,3-triazole regioisomers. J. Mol. Struct.. 2008;892:210-215.
- [CrossRef] [Google Scholar]
- Novel coronavirus treatment with ribavirin: Groundwork for an evaluation concerning COVID-19. J. Med. Virol.. 2020;92:740-746.
- [CrossRef] [Google Scholar]
- Synthesis of 1-Acyl-3-isopropenylbenzimidazolone Derivatives and Their Activity against Botrytis cinerea. J. Agric. Food. Chem.. 2010;58:2668-2672.
- [CrossRef] [Google Scholar]
- Mechanism of the Intramolecular Claisen Condensation Reaction Catalyzed by MenB, a Crotonase Superfamily Member. Biochemistry.. 2011;50:9532-9544.
- [CrossRef] [Google Scholar]
- Candida albicans targets that potentially synergize with fluconazole. Crit. Rev. Microbiol.. 2021;47:323-337.
- [CrossRef] [Google Scholar]
- A novel view of the separate and simultaneous binding effects of docetaxel and anastrozole with calf thymus DNA: Experimental and in silico approaches. Spectrochim. Acta A Mol. Biomol. Spectrosc.. 2020;228:117528
- [CrossRef] [Google Scholar]
- Synthesis and antitrichinellosis activity of some 2-substituted-[1,3]thiazolo[3,2-a]benzimidazol-3(2H)-ones. Bioorg. Med. Chem.. 2005;13:5550-5559.
- [CrossRef] [Google Scholar]
- The structural basis for the prevention of nonsteroidal antiinflammatory drug-induced gastrointestinal tract damage by the C-lobe of bovine colostrum lactoferrin. Biophys. J.. 2009;97:3178-3186.
- [CrossRef] [Google Scholar]
- The effects of acute treatment with ramelteon, triazolam, and placebo on driving performance, cognitive function, and equilibrium function in healthy volunteers. Psychopharmacology.. 2014;232:2127-2137.
- [CrossRef] [Google Scholar]
- Interaction of anti-inflammatory drugs with serum proteins, especially with some biologically active proteins. J. Pharm. Pharmacol.. 1968;20:169-173.
- [CrossRef] [Google Scholar]
- Fabrication and characterization of rizatriptan loaded pullulan nanofibers as oral fast-dissolving drug system. Mater. Res. Express.. 2021;8:055404
- [CrossRef] [Google Scholar]
- Murray, P. R., Baron, E. J., Pfaller, M. A., Tenover, F. C. and Yolken, R. H., 1995. Manual Clinical Microbiol, sixth ed., ASM Press, Washington DC, pp. 1327-1341.
- Determining the Interaction Behavior of Calf Thymus DNA with Anastrozole in the Presence of Histone H1: spectroscopies and Cell Viability of MCF-7 Cell Line Investigations. DNA Cell Biol.. 2021;40:1039-1051.
- [CrossRef] [Google Scholar]
- Evaluation of the binding effect and cytotoxicity assay of 2-Ethyl-5-(4-methylphenyl) pyramido pyrazole ophthalazine trione on calf thymus DNA: spectroscopic, calorimetric, and molecular dynamics approaches. Luminescence. 2022;37:310-322.
- [CrossRef] [Google Scholar]
- New Methods for Synthesis of 1,2,3-Triazoles: a review. Polycycl. Aromat. Compd.. 2021;1–19
- [CrossRef] [Google Scholar]
- Etizolam: A rapid review on pharmacology, non-medical use and harms. Drug Alcohol Rev.. 2020;39:330-336.
- [CrossRef] [Google Scholar]
- Urea-thiazole/benzothiazole hybrids with a triazole linker: synthesis, antimicrobial potential, pharmacokinetic profile and in silico mechanistic studies. Mol. Divers.. 2022;26:2375-2391.
- [CrossRef] [Google Scholar]
- Investigation of the interaction between human serum albumin and two drugs as binary and ternary systems. Eur. J. Drug Metab. Pharmacokinet.. 2016;41:705-721.
- [CrossRef] [Google Scholar]
- Rufinamide add-on therapy for drug-resistant epilepsy. Cochrane Database Syst. Rev. 2020
- [CrossRef] [Google Scholar]
- Behavioral effects of triadimefon in zebrafish are associated with alterations of the dopaminergic and serotonergic pathways. Prog. Neuro-Psychopharmacol. Biol. Psychiatry.. 2019;92:118-126.
- [CrossRef] [Google Scholar]
- Molecular Basis of Bacillus subtilis ATCC 6633 Self-Resistance to the Phosphono-oligopeptide Antibiotic Rhizocticin. ACS Chem. Biol.. 2019;14:742-750.
- [CrossRef] [Google Scholar]
- Aryl Substituted Benzimidazolones as Potent HIV-1 Non-Nucleoside Reverse Transcriptase Inhibitors. ACS. Med. Chem. Lett.. 2019;10:196-202.
- [CrossRef] [Google Scholar]
- 1H–1,2,3-triazole grafted tacrine-chalcone conjugates as potential cholinesterase inhibitors with the evaluation of their behavioral tests and oxidative stress in mice brain cells. Bioorg. Chem.. 2021;114:105053
- [CrossRef] [Google Scholar]
- Exploring the HSA/DNA/lung cancer cells binding behavior of p-Synephrine, a naturally occurring phenyl ethanol amine with anti-adipogenic activity: multi spectroscopic, molecular dynamic and cellular approaches. J. Mol. Liq.. 2022;368:120826
- [CrossRef] [Google Scholar]
- 1.3-Dipolare Cycloadditionen, XXXII. Kinetik der Additionen organischer Azide an CC-Mehrfachbindungen. Chem. Ber.. 1967;100:2494-2507.
- [CrossRef] [Google Scholar]
- New 1,2,3-triazole containing benzimidazolone derivatives: Syntheses, crystal structures, spectroscopic characterizations, Hirshfeld surface analyses, DFT calculations, anti-corrosion property anticipation, and antibacterial activities. J. Mol. Struct.. 2021;1242:130719
- [CrossRef] [Google Scholar]
- New 1,2,3-triazole containing benzimidazolone derivatives: Syntheses, crystal structures, spectroscopic characterizations, Hirshfeld surface analyses, DFT calculations, anti-corrosion property anticipation, and antibacterial activities. J. Mol. Struct. 2021
- [CrossRef] [Google Scholar]
- a minireview of 1,2,3-triazole hybrids with O-heterocycles as leads in medicinal chemistry. Chem. Biol. Drug Des.. 2022;100:843-869.
- [CrossRef] [Google Scholar]
- Novel perspective into the interaction behavior study of the cyanidin with human serum albumin-holo transferrin complex: Spectroscopic, calorimetric and molecular modeling approaches. J. Mol. Liq.. 2022;356:119042
- [CrossRef] [Google Scholar]
- ShelXT-Integrated space-group and crystal-structure determination. Acta Cryst.. 2015;A71:3-8.
- [CrossRef] [Google Scholar]
- Targeting glutamine utilization to block metabolic adaptation of tumor cells under the stress of carboxyamidotriazole-induced nutrients unavailability. Acta Pharm. Sin. B.. 2022;12:759-773.
- [CrossRef] [Google Scholar]
- Synthesis and in vitro and in silico studies of 1H- and 2H–1,2,3-triazoles as antichagasic agents. Bioorg. Chem.. 2021;116
- [CrossRef] [Google Scholar]
- Modifiable Risk Factors for the Emergence of Ceftolozane-Tazobactam Resistance. Clin. Infect. Dis.. 2021;73:e4599-e4606.
- [CrossRef] [Google Scholar]
- AutoDock Vina: improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J. Comput. Chem.. 2010;31:455-461.
- [CrossRef] [Google Scholar]
- Propiconazole induces abnormal behavior and oxidative stress in zebrafish. Environ. Sci. Pollut. Res. 2019
- [CrossRef] [Google Scholar]
- Synthesis of 1,2,4-triazole compounds related to the fungicides flutriafol and hexaconazole. Pest. Sci.. 1991;31:457-498.
- [CrossRef] [Google Scholar]
- An Integrated Transfer Learning and Multitask Learning Approach for Pharmacokinetic Parameter Prediction. Mol. Pharm.. 2019;16:533-541.
- [CrossRef] [Google Scholar]
- Novel tacrine-coumarin hybrids linked to 1,2,3-triazole as anti-Alzheimer’s compounds: In vitro and in vivo biological evaluation and docking study. Bioorg. Chem.. 2019;83:303-316.
- [CrossRef] [Google Scholar]
Appendix A
Supplementary material
Supplementary data to this article can be found online at https://doi.org/10.1016/j.arabjc.2023.104566.
Appendix A
Supplementary material
The following are the Supplementary data to this article:







