Translate this page into:
Synthesized tin oxide nanoparticles promote apoptosis in human osteosarcoma cells
⁎Corresponding author. 15152353021@163.com (Huaixi Yu)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Complicated protocols have been used to tune the size of tin oxide nanoparticles (SnO2 NPs) in conventional chemical synthesis approaches. In this study, we addressed this issue through the combination of polarity solvent reduction and sonication as a potential strategy for optimizing the size of SnO2 NPs for anticancer applications. Then the anticancer activity and safety of synthesized SnO2 NPs on U-2OS human osteosarcoma cells and normal human osteoblast, NHOst, respectively, were assessed by several assays. SEM and TEM analyses revealed that synthesized SnO2 NPs had a spherical-shaped morphology with an average particle size of about 10 nm. FTIR result showed a broad absorption peak at 649 cm– 1, corresponding to the stretching vibration of the O–Sn–O bond. XRD analysis exhibited eight main peaks, revealing a tetragonal system of rutile-like structure. DLS study showed that synthesized SnO2 NPs had an average hydrodynamic size of 43.93 nm with a zeta potential value of −31.00 mV. Cellular assays displayed that SnO2 NPs mitigate the growth of U-2OS human osteosarcoma cells (IC50: 15.50 µg/mL) with partial cytotoxicity on normal osteoblasts. It was then discovered that SnO2 NPs triggered stress effects by raising the levels of LDH release, ROS, and MDA and lowering those of SOD, CAT activity, and GSH content. Also, it was discovered that SnO2 NPs activated both intrinsic and extrinsic signaling pathways mediated by MMP collapse, elevation of caspase-3, −9, −8 activities and corresponding mRNA, and upregulation of Bax/Bcl-2 mRNA ratio. Overall, this study found that extrinsic and mitochondrial apoptotic pathways can both be activated by synthesized SnO2 NPs to induce apoptosis in human osteosarcoma cells. This finding calls for additional research in the future.
Keywords
Tin oxide
Nanoparticle
Characterization
Apoptosis
Osteosarcoma
1 Introduction
The most prevalent type of primary solid bone tumor, osteosarcoma, is recognized by the existence of malignant mesenchymal cells that generate osteoid as well as immature bone. In adolescence, 15 % of all solid extracranial cancers are osteosarcomas and males typically experience this disorder 1.4 times more often than females. The promising treatment of patients suffering from osteosarcoma requires multidisciplinary supervision (Ritter and Bielack, 2010).
The capability of nanotechnology to improve the early detection, diagnostics, and therapeutic approaches of different cancer cells has recently received overwhelming attention (Ferrari, 2005). Therefore, the application of new strategies that benefit from nanotechnology may open new avenues in the prevention and treatment of osteosarcoma (Alrushaid et al., 2023).
The synthesis of inorganic nanoparticles (NPs) with prominent biomedical characteristics is a crucial challenge and has recently obtained enormous interest (Tuli,Joshi,et al., 2023).
Sonochemistry is known as one of the earliest strategies utilized to prepare nanostructured materials. In this technique, particles go through a chemical reaction following exposure to strong ultrasound radiation (20 kHz–10 MHz) which results in an acoustic cavitation sonochemical process. In comparison with other conventional approaches, the sonochemical method results in the production of NPs with more uniform dispersity, a higher surface area, higher thermal stability, and better phase purity (Low et al., 2022). However, it should be noted that this method needs highly controllable protocols to result in the synthesis of monodisperse NPs with desirable dimensions (Li et al., 2021). Also, surface modification of NPs with different moieties is complicated due to the hydrophobicity of the organic ligands in water. Therefore, much attention has been given to addressing these drawbacks of the sonochemical method by controlling the solvent polarity (Hussain et al., 2020). Therefore, in this paper, we used a combination route for the synthesis of NPs via sonochemical method along with the application of a mixture of water and ethanol for the reduction of the polarity of water as a solvent.
Owing to the elegant biomedical features of metal oxides, they have been using as a future potential stone in cancer therapy through the induction of apoptosis (Shabatina et al., 2023; Saini et al., 2016; Bandyopadhyay et al., 2020). Pure metal oxide of tin oxide (SnO2) has shown promising anticancer activities on a wide range of cancers, including ovarian (Hanna and R. Saad 2021), blood (Ahmadabad et al., 2021), and breast cancer cells (Guo et al., 2021). Hanna et al. showed that SnO2 NPs could induce mitochondria-mediated apoptosis in ovarian cancer cells (Hanna and R. Saad 2021). Also, Ahamed et al. reported that reactive oxygen species (ROS)-mediated apoptosis could be the anticancer mechanism of SnO2 NPs in breast cancer cells (Ahamed et al., 2018).
Though many studies have been published on the anticancer activity of SnO2 NPs, selective apoptosis induction of SnO2 NPs on different types of cancer cells in vitro was not well-explored. As osteosarcoma is one of the most common primary malignant bone tumors and occurs mainly in childhood and young adults, the selective apoptosis induction in these types of tumors could be considered as a promising strategy in modulation of this disorder. Therefore, the main aim of this study was to assess the anticancer effects of SnO2 NPs synthesized through a combination method (sonochemical along with solvent polarity reduction) on U-2OS human osteosarcoma cells.
2 Materials and methods
2.1 Materials
Tin(II) chloride dehydrate (SnCl2·2H2O, ACS reagent, 98 %, product No. 243523), 2, 7-dichlorofluorescein diacetate (DCFDA), and Dulbecco's Modified Eagle's Medium (DMEM) were purchased from Sigma (St. Louis, MO, USA). Fetal bovine serum (FBS) and antibiotics were purchased from Invitrogen (Carlsbad, CA, USA).
2.2 Synthesis of SnO2 NPs through combination method
SnCl2·2H2O (Merck) (1 M) suspended in 100 mL water–ethanol mixture (10 % ethanol, to reduce the polarity of water solvent) were stirred (500 rpm for 30 min) followed by irradiation by a high intensity ultrasonic processor (750 W; 20 kHz and 6 mm Ti-probe tip) for 1 h. The pH of the medium was regulated by ammonium hydroxide (Sigma Aldrich) and gradually added to reach a range between 9 and 10 to result in the formation of monodispersed NPs. The temperature was also set at 80–90 °C during the sonication system. The samples were then centrifuged (5 min, 3000 rpm/min), washed several times with deionized water, dried at 70 ˚C, and finally calcined at 500 ˚C for 2 h (Rehman et al., 2020).
2.3 Characterization of the synthesized SnO2 NPs
The particle size and the morphological structure were investigated by transmission electron microscope TEM (FEI Tecnai G2 X-TWIN 100 kV) and scanning electron microscope (SEM, MIRA3). Fourier transform infrared (FTIR) was done on a spectrophotometer (FTIR- Tensor 27, Bruker, Germany) in the range of 4000–400 cm−1). Phase identification and crystalline nature of the synthesized SnO2 NPs were measured by X-ray diffraction (XRD) using a diffractometer system (PANalyticalX'pert PRO XRD), Cu as anodic materials [CuKα radiation (λ = 1.54178 Å), 40 kV and 30 mA] with scan step in 2θ range from 20° to 80°. The hydrodynamic radius and zeta potential distribution of SnO2 NPs were measured using dynamic light scattering (DLS, Malvern Instruments, Worcestershire, UK, ZEN 3600 model Nano ZS).
2.4 Cell culture
Human osteosarcoma, U-2OS, and normal human osteoblast, NHOst, cell lines obtained from American Type Culture Collection (ATCC, Manassas, VA, USA), were cultured in DMEM supplemented with 10 % FBS, 1 % antibiotics, 1 mM l-glutamine (Gibco, Grand Island, NY, USA) and 100 µg/mL gentamicin (Gibco, Grand Island, NY, USA) at 37 °C with 5 % CO2.
2.5 Cell viability assay
Cell viability assay was done using a conventional MTT assay. Briefly, cancerous and noncancerous cells seeded into 96-well culture plates with a density of 5 × 103 cells/well were incubated for 24 h. Afterward, SnO2 NPs with different concentrations ranging from 1 to 100 µg/mL were transferred and further incubated for 24 h. Then, 50 μl of MTT (5 mg/mL) was added into each well after removing the culture medium and incubated for 4 h at 37 °C. 100 μl of dimethyl sulfoxide was then added to the wells and the optical density (OD) of the solution was detected at 570 nm on a microplate ELISA reader.
2.6 Lactate dehydrogenase (LDH) release assay
LDH assay was used to evaluate the cytotoxic effects of SnO2 NPs on U-2OS human osteosarcoma cells or NHOst normal cells. Cells seeded in a 96-well plate at a density of 5 × 103 cells/well were exposed to IC50 concentration of SnO2 NPs (15.50 µg/mL) for 24 h. LDH Assay Kit (Shanghai Biyuntian Biotechnology Co., Ltd.) was used to detect the LDH release in cell culture medium according to the manufacturer's protocol and the absorbance of the sample was read at 490 nm using a microplate ELISA reader.
2.7 Reactive oxygen species analysis (ROS)
ROS assay was used to assess the oxidative stress induced by SnO2 NPs in U-2OS human osteosarcoma cells. Cells seeded in a 6-well plate at a density of 3 × 105 cells/well were incubated with IC50 concentration of SnO2 NPs (15.50 µg/mL) for 6 h. Afterward, cells were collected, washed, and suspended in a DCFH-DA solution (20 µM) for 30 min. The cells were then washed and the fluorescence intensity of fluorescent product 2′,7′-dichlorofluorescein (DCF) was read on a SYNERGY-HT multiwell plate reader, Bio-Tek (USA) at excitation and emission wavelengths of 485 and 528 nm, respectively.
2.8 Oxidative stress analysis
Cells with a density of ∼5 × 106 in a 75 cm2 culture flask exposed to IC50 concentration of SnO2 NPs (15.50 µg/mL) were scraped, washed with chilled 1 × PBS, and centrifuged at 500g for 15 min. The pellets were washed and then incubated on ice with 1 × Lysis Buffer (10 mM Tris, pH 7.5, 150 mM NaCl, 0.1 mM EDTA, 0.5 % Triton-100) for 10 min supplemented with appropriate protease inhibitors. Then, the cells were centrifuged at 12 000 g for 10 min and the supernatant was collected. Protein concentration was determined by the Bradford assay (Bonjoch and Tamayo, 2001). Subsequently, superoxide dismutase (SOD, A001-1; Nanjing Jiancheng Bioengineering Institute) and catalase (CAT, A007-2; Nanjing Jiancheng Bioengineering Institute) activities as well as glutathione (GSH, A006-1; Nanjing Jiancheng Bioengineering Institute) and 3,4-Methylenedioxyamphetamine (MDA, A003-1; Nanjing Jiancheng Bioengineering Institute) contents were detected with relevant detection kits according to the protocols recommended by the manufacturer.
2.9 Activity of caspase-3, caspase-9, and caspase-8
The activity levels of caspase-3, caspase-9 and caspase-3 were measured according to the manufacturer's protocol provided with the caspase colorimetric assay kits (Nanjing Jiancheng Bioengineering Institute). Briefly, an aliquot of cell suspension was mixed with the reaction buffers containing the substrates followed by incubating at 37 °C for 2 h and terminating by the addition of a stop buffer. Then, the optical density of samples was read at 405 nm using a microplate ELISA reader.
2.10 Mitochondrial membrane potential (MMP)
Cells were seeded in a 6-well microculture plate at a density of 3 × 105 cells/well and after incubation with IC50 concentration of SnO2 NPs (15.50 µg/mL) for 24 h, then the medium was replaced with reaction buffer provided with MMP kit (Shanghai Yeasen Biotechnology Co., Ltd.). The relative ratio of red to green fluorescence intensity was determined as the level of MMP depolarization.
2.11 Total RNA extraction and quantitative real-time PCR (qRT-PCR)
Cells were seeded in a 6-well microculture plate at a density of 3 × 105 cells/well and after incubation with IC50 concentration of SnO2 NPs (15.50 µg/mL) for 24 h total RNA was extracted by RNA isolation Kit (Huayueyang Biotechnology, China) according to the manufacturer's instructions. After checking the quantity and quality of RNA, the cDNA synthesis kit (Takara, Beijing, China) was used to reverse-transcribe RNA following the manufacturer's instructions. The reactions were done on a Bio-red IQ2 Sequence Detection System and Applied Biosystems software using SYBR Green PCR Master Mix (Applied Biosystems) following the standard protocols. Primer design was based on previous studies (Liang et al., 2012; Punzo et al., 2017). The comparative threshold cycle (Ct) values were calculated and analyzed using the 2−ΔΔCT method, with β-actin as an internal control.
2.12 Statistics analysis
SPSS statistics package was used for statistical analysis between different groups by one-way analysis of variance (*P < 0.05, ** P < 0.01, *** P < 0.001).
3 Result and discussion
3.1 Imaging analysis
SEM and TEM analyses were done to determine the shape and size of the synthesized SnO2 NPs. The morphology of SnO2 NPs was assessed, whereas the size of NPs was determined by the TEM test. SEM images showed that synthesized SnO2 NPs provided monodispersed spherical- or quasi-spherical-shaped morphology (Fig. 1A). TEM analysis revealed that synthesized SnO2 NPs show the average particle size around 10 nm (Fig. 1B). In other words, from TEM analysis, the probable uniform distribution of small sized SnO2 NPs (about 10 nm) is revealed. This can be attributed to the cavitational effects stimulated by ultrasound, which results in the generation of extreme shearing forces, micromixing and turbulence derived from physical influences inserted by the transient deformation of cavities that appeared due to ultrasound (Deosarkar et al., 2013; Choudhari et al., 2020; Karunakaran,Raadha,and Gomathisankar, 2013). Also, solvent polarity reduction not only results in the dispersion of synthesized NPs but also a decrease in the size of NPs (Hussain et al., 2020).
SEM (A) and TEM (B) imaging of synthesized SnO2 NPs through the combination of solvent polarity reduction and sonochemical method.
Sery et al. (Sery et al., 2022) reported that synthesized SnO2 NPs through the sonochemical method had a wide size range of 2 to 30 nm, which makes them complicated for biological applications Karunakaran et al. (Karunakaran,Raadha,and Gomathisankar, 2013) also showed that sonochemically synthesized SnO2 NPs had an average diameter of 10 nm, which is in good agreement with our TEM result. However, they found that the prepared NPs lack finite shape or structure, which might be due to the generation of hotspots through the sonochemical method as the main destructors of the templating effect. Additionally, Zhu et al. reported that sonochemically synthesized SnO2 NPs in an aqueous solution of SnCl4 and azodicarbonamide were ∼3 − 5 nm in size, as detected by TEM analysis (Zhu et al., 2000). However, it should be indicated that ultrasmall NPs (around 3 to 5 nm) are easily filtered by liver and kidney tissues. Therefore, we can conclude that sonochemistry along with solvent polarity reduction could result in the synthesis of SnO2 NPs with monodispersed distribution and a potential size of around 10 nm for biological implementations (Bartucci et al., 2020).
3.2 FTIR measurement
Fig. 2 depicts the FTIR spectrum for synthesized SnO2 NPs in the wavenumber region of 4000–400 cm– 1. The strong absorption peaks at 1635 cm– 1 and 3476 cm−1 attribute to the stretching of H–O–H and O–H bonds, respectively deduced from water molecules (Garg et al., 2020). One broader absorption peak is also visible in the spectrum at 649 cm– 1, corresponding to the stretching vibration of the O–Sn–O bond (Garg et al., 2020; Sery et al., 2022).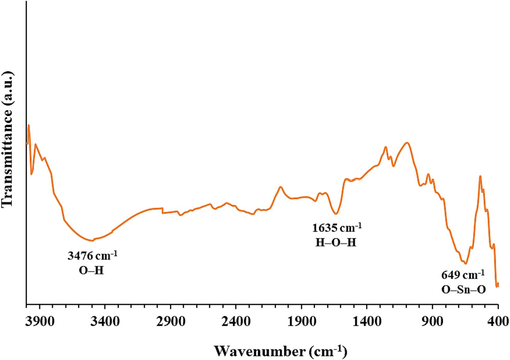
FTIR spectrum of synthesized SnO2 NPs.
This data showed the purity and the lack of crystal lattice defects or disorders in synthesized SnO2 NPs (Garg et al., 2020; Sery et al., 2022).
3.3 XRD analysis
The XRD pattern of the synthesized SnO2 NPs is shown in Fig. 3. The peaks at 2θ values of 27.75°, 33.80°, 37.80°, 52.05°, 54.15°, 62.35°, 66.40°, and 79.01° can be attributed to (1 1 0), (1 0 1), (2 0 0), (2 1 1), (2 2 0), (3 1 0), (3 0 1), and (3 2 1) plans, respectively (Patil et al., 2012; Sery et al., 2022). A matching of the detected and standard (hkl) planes proved that the particle is of SnO2 with a tetragonal system of rutile-like structure, which is in good agreement with the literature values (JCPDS, PDF file No 41–1445, space group P42/MNM, lattice parameters a = 4.7420 Å and c = 3.1900 Å) (Sery et al., 2022).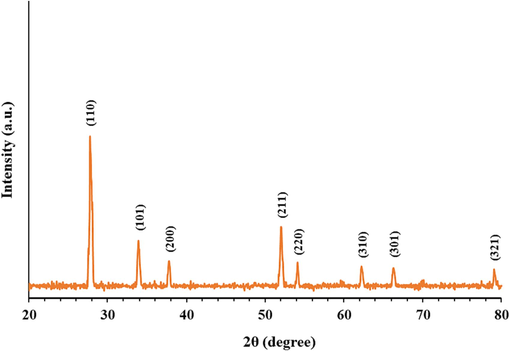
XRD pattern of synthesized SnO2 NPs.
No other detectable XRD peaks were observed, revealing the high purity of the synthesized SnO2 NPs (Sery et al., 2022). Also, XRD patterns of SnO2 NPs demonstrated high peak intensities, suggesting ideal crystallinity and crystallite-size developments (Sery et al., 2022), which is consistent with the NP size observed by TEM analysis. Also, slight broad peaks were detected for synthesized SnO2 NP samples corroborating the generation of fine nanosized samples (Sery et al., 2022). Therefore, it can be assumed that the sonication of the solution provides us with a high probability of synthesizing particles in the nanometer size.
3.4 Hydrodynamic radius and charge studies
DLS study was also done to measure the hydrodynamic radius and zeta potential values of the synthesized SnO2 NPs. Fig. 4 A shows the hydrodynamic size of the dispersed synthesized SnO2 NPs in solution determined by DLS which was found to be ranged between 34 and 60 nm with an average size of 43.93 nm. The differences in determined size between TEM and DLS techniques could be associated with the slight agglomeration of NPs in the solution. However, a monodisperse distribution of synthesized SnO2 NPs was detected in Fig. 4A, which could be due to the cavitation process induced by the nonchemical approach, which results in large NPs breaking up into homogenous small particles and their slight aggregation (Barbhuiya et al., 2022).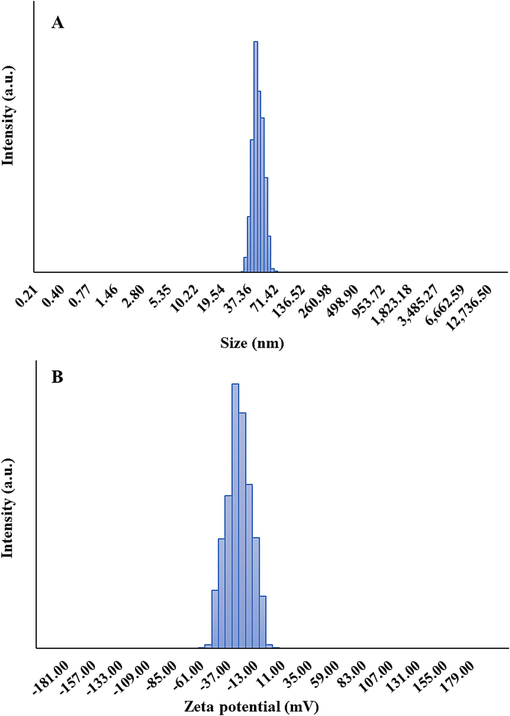
DLS (A) and zeta potential (B) analyses of synthesized SnO2 NPs.
Fig. 4B depicts that the average zeta potential of synthesized SnO2 NPs was −31.00 mV. This strong negative value indicates an apparent particle repulsion, corresponding to the stability of NPs (Manimaran,Muthuvel,and Said, 2023). Generally, zeta potential reveals how electrostatic forces determine the strength of repulsion forces between neighboring NPs. Following the adsorption of OH− ions on the surface of NPs, their zeta potential values become negative, inhibiting the strong NP aggregation and maintaining the colloidal stability (Manimaran,Muthuvel,and Said, 2023; Srivastava and Mukhopadhyay, 2014).
3.5 SnO2 NPs interaction with cells
The anticancer activity of synthesized SnO2 NPs on U-2OS human osteosarcoma cells was evaluated by using the MTT assay. As depicted in Fig. 5A, treatment with synthesized SnO2 NPs for 24 h significantly decreased the proliferation of U-2OS cells in a concentration-dependent fashion in comparison to control cells (*P < 0.05, **P < 0.01, ***P < 0.001), while the growth of human normal osteoblasts NHOst cells (Fig. 5B) was much less influenced by the synthesized SnO2 NPs at the same studied concentrations.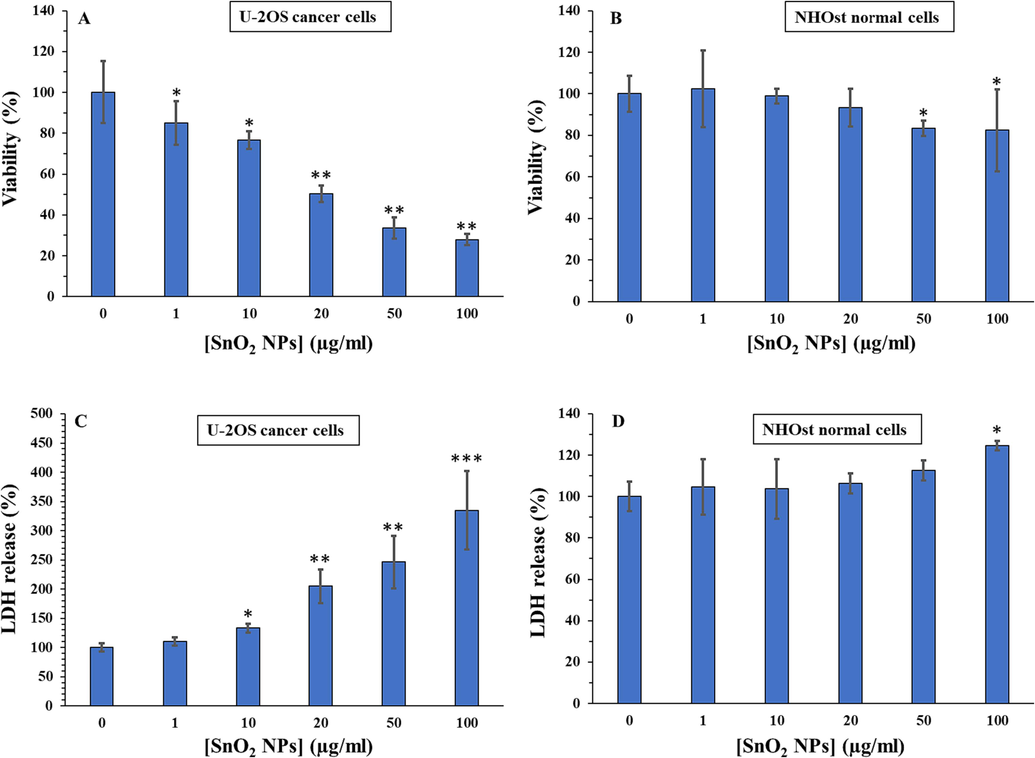
Cytotoxic effects of SnO2 NPs against U-2OS human osteosarcoma cells (A) and human normal osteoblasts NHOst cells (B) determined by MTT assay. Membrane leakage induced by SnO2 NPs in U-2OS human osteosarcoma cells (C) and human normal osteoblasts NHOst cells (D) determined by LDH assay. All experiments were done after 24 h incubation of cells with SnO2 NPs. *P < 0.05, ** P < 0.01, *** P < 0.001 relative to control. Data were presented as mean ± SD of three experiments.
Therefore, the IC50 concentration of synthesized SnO2 NPs on U-2OS human osteosarcoma cells was determined to be about 15.50 µg/mL at 24 h.
A previous study has shown that biosynthesized SnO2 NPs with particle size distribution ranging between 5 and 10 nm had significant antiproliferative effects against colorectal and lung cancer cells with IC50 values of 165–208 µg/mL and 138–187 µg/mL, respectively (Tammina et al., 2017). Additionally, Hanna et al. (Hanna and R. Saad 2021) reported that the calculated IC50 value for the sol–gel synthesized SnO2 NPs with a size of around 79 nm against SKOV3 human ovarian cancer cells, was 47.87 µg/mL at 24 h incubation. However, Ahamed et al. (Ahamed et al., 2018) reported that the sol–gel synthesized SnO2 NPs with a size of about 21 nm did not induce cytotoxicity in MCF-7 breast cancer cells up to the concentration 50 µg/mL at 24 and 48 h. Therefore, several parameters may play a key role in the NP-induced cytotoxicity such as the physicochemical properties of NPs, type of cancer or normal cells, and experimental procedures.
LDH enzyme release from cytoplasm into cell culture medium can be appointed as a useful marker of damage to cell membrane integrity (Ahamed et al., 2018). Higher levels of LDH enzyme activity in cell culture media reveal the higher damage of cell membranes induced by NP exposure (Ifijen et al., 2023). We also found that synthesized SnO2 NPs prompted LDH leakage in U-2OS human osteosarcoma cells in a concentration-dependent manner (Fig. 5C), whereas these NPs did not induce a significant LDH release in normal human osteoblast, NHOst, below 100 µg/mL at 24 h (Fig. 5D). Altogether, these data proved that SnO2 NPs show potential to trigger concentration-dependent cytotoxicity in the U-2OS cell line. Our findings are consistent with recent reports demonstrating SnO2 NPs' anticancer effects in several cancer cells (Ahamed et al., 2018; Ahmadabad et al., 2021; Ifijen et al., 2023).
3.6 SnO2 NPs induced oxidative stress in U-2OS human osteosarcoma cells
Induction of oxidative stress through inhibition of antioxidant enzyme activities and depletion of non-enzymatic antioxidants are the most common pathways through which NPs stimulate apoptosis in cancer cells (Akhtar et al., 2012; Rayzah et al., 2023). To maintain the redox balance in cells, antioxidant defense and strict control over the generation of free radicals are essential (Rayzah et al., 2023). The GSH, SOD, and CAT are serving as strong antioxidant molecules in protecting cells from oxidative stress prompted by ROS and other free radicals (Rayzah et al., 2023). Antioxidant enzymes, SOD and CAT, are crucial for shielding cells from the damaging effects of free radicals. GSH plays a crucial role in inhibiting lipid peroxidation (Rayzah et al., 2023). Because cancer cells can express antioxidant molecules, they are able to eliminate the excessive generation of ROS, extending their survival half-life through preventing apoptosis. On the other hand, elevated oxidative stress in cancerous cells may initiate anti-tumorigenic signaling cascades, resulting in tumor cell death (Zeng et al., 2023; Tuli, Kaur, et al. 2023).
Therefore, the oxidant and antioxidant levels were examined in the U-2OS cell line treated with SnO2 NPs with a concentration of 15.50 µg/mL at 24 h. ROS (Fig. 6A) and MDA (Fig. 6B) levels were assessed as main biomarkers of oxidative stress, whereas antioxidant status was assessed by analyzing the SOD (Fig. 6C) and CAT (Fig. 6D) activities as well as GSH content (Fig. 6E). The obtained data displayed that oxidant (ROS and MDA) levels were higher in SnO2 NPs-treated cancer cells compared to negative control cells (Fig. 6A and B).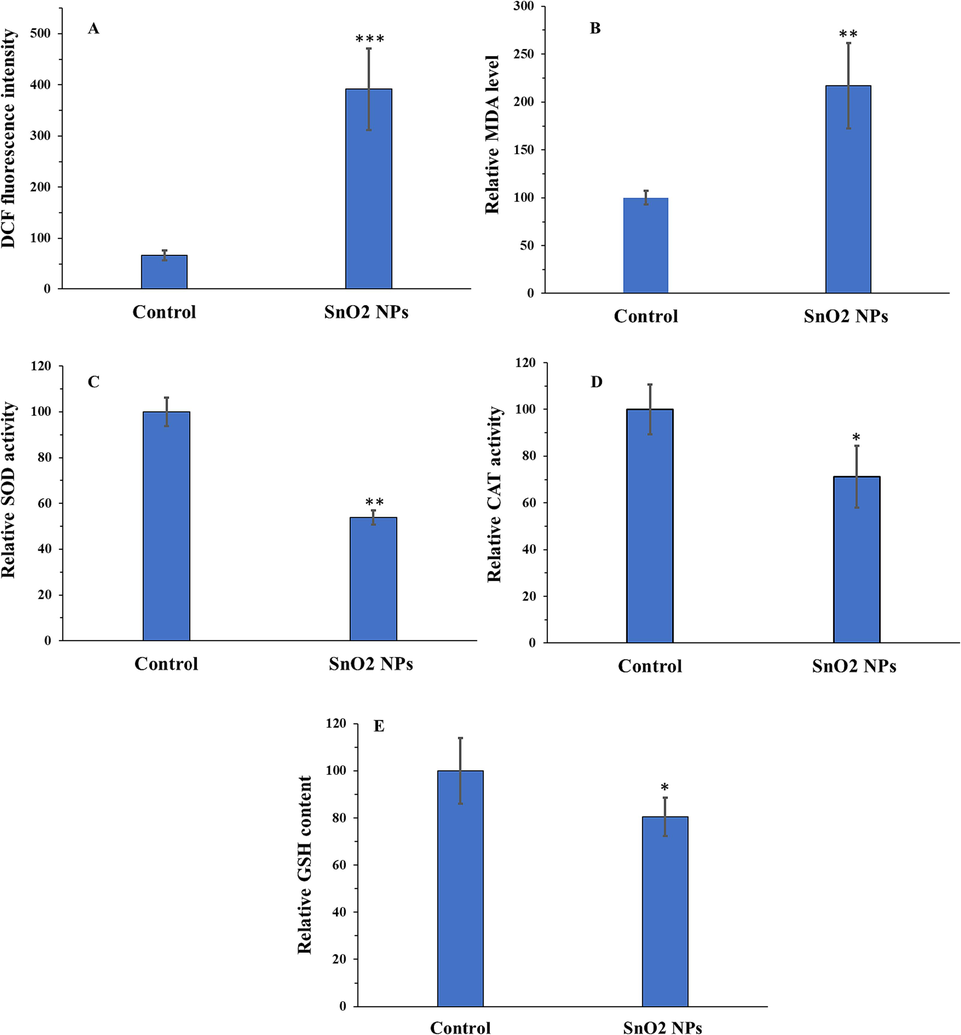
Oxidative stress induction of SnO2 NPs in U-2OS human osteosarcoma cells determined by ROS assay (A), MDA assay (B), SOD assay (C), CAT assay (D), and GSH assay (E). All experiments were done after 24 h incubation of cells with SnO2 NPs. *P < 0.05, ** P < 0.01, *** P < 0.001 relative to control. Data were presented as mean ± SD of three experiments.
Furthermore, CAT and SOD activities as well as the GSH content were remarkably reduced by the IC50 concentration of SnO2 NPs (Fig. 6C-E). These findings indicated that SnO2 NP treatment collapses the antioxidant system of the cells while increasing the ROS and MDA levels, thereby accelerating oxidative stress-manipulated cell death in U-2OS human osteosarcoma cells. These data are in good agreement with a study reported by Ahamed et al. (Ahamed et al., 2018), indicating that SnO2 NPs could induce oxidative stress-mediated anticancer effects through deregulation of enzymatic and non-enzymatic oxidants.
3.7 SnO2 NPs activated both intrinsic and extrinsic signaling pathways
There are two possible ways for NPs to trigger apoptotic cell death: intrinsic or extrinsic pathways (Tuli,Joshi,et al., 2023). Intrinsic mechanisms of apoptosis are activated by non-receptor mediated stimuli that trigger the disruption of several mediators such as MMP, Bax/Bcl-2 ratio, and caspase-9 activity to stimulate apoptosis, whereas extrinsic apoptotic cell death is initiated upon the interaction of anticancer drugs with transmembrane receptors and activation of caspase-8 (Lossi, 2022; Fulda and Debatin, 2006). Metal NPs are potential candidates to induce apoptotic cell death in different types of cancers, including bone, brain, blood, liver, and so on (Tuli,Joshi,et al., 2023). Especially, functionalized SnO2 NPs have been observed to promote anticancer activity in human ovarian cancer cells by overexpression of Bax, and cleaved caspase-3 (Hanna and R. Saad, 2021). In cervical cancer HeLa cells, cell SnO2 NP treatment was shown to deregulate the Bax/Bcl-2 mRNA ratio (Bazsefidpar et al., 2021). Guo et al. explored the downregulation of PI3K/Akt/mTOR/Bax/Bcl-2 in SnO2 NPs- treated breast cancer cells, leading to apoptotic cell death (Guo et al., 2021). Similarly, Jiang et al. (Jiang et al., 2022) observed the apoptotic effect of biosynthesized SnO2-sodium alginate-polyethylene glycol-carvacrol NPs in esophagus squamous cells cancer via upregulation of Bax and caspase 3. Therefore, in this study, for 24 h to assess if synthesized SnO2 NP solution at the IC50 concentration (15.50 µg/mL) could induce apoptotic effects in U-2OS human osteosarcoma cells via intrinsic or extrinsic pathways, MMP, qRT-PCR, and caspase activity assays were performed.
As determined by the decreased ratio of red to green fluorescence by JC-1 staining, incubation with the SnO2 NP solution at the IC50 concentration (15.50 µg/mL) for 24 h significantly reduced MMP in U-2OS human osteosarcoma cells, compared with negative treated cells (Fig. 7A). Furthermore, All the caspase activities under study found to be increased (Fig. 7B-D) following incubation of U-2OS cells with 15.50 µg/mL SnO2 NPs at 24 h, compared to control cells.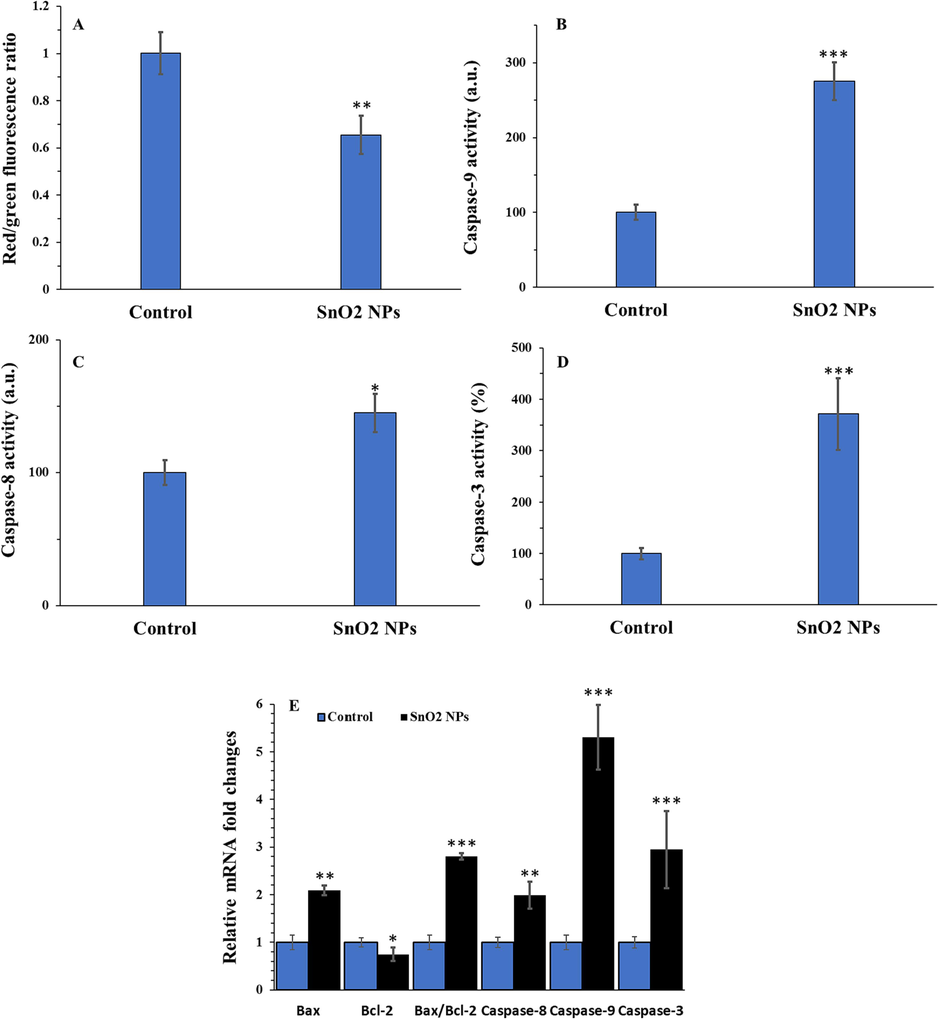
Apoptosis induction of SnO2 NPs in U-2OS human osteosarcoma cells determined by different assays. MMP assay (A), caspase-9 activity assay (B), caspase-8 activity assay (C), caspase-3 activity assay (D), and qRT-PCR assay (E). All experiments were done after 24 h incubation of cells with SnO2 NPs. *P < 0.05, ** P < 0.01, *** P < 0.001 relative to control. Data were presented as mean ± SD of three experiments.
Additionally, the upregulated levels of Bax mRNA, caspase-9 mRNA, caspase-8 mRNA and caspase-3 mRNA, and the reduced level of Bcl-2 mRNA were observed in U-2OS human osteosarcoma cells following incubation with 15.50 µg/mL SnO2 NPs at 24 h (Fig. 7E). These results confirmed the activation of both intrinsic and extrinsic apoptosis signaling pathways by SnO2 NPs in the U-2OS cells. Using leukemia K562 cells, Ahmadabad et al. (Ahmadabad et al., 2021) showed that synthesized SnO2 NPs through hydrothermal method with a size of 60 nm triggered overexpression of caspase-9, −8, −3 and Bax/Bcl2 mRNA ratio. However, the main drawback of this study was the heterogenous distribution of synthesized NPs in solvent as determined by the DLS study.
4 Conclusion
In conclusion, SnO2 NPs with an average size of around 10 nm were produced via a combination of solvent polarity reduction and sonochemical route and well-characterized by different techniques. Then, we demonstrated that synthesized SnO2 NPs, concentration-dependently mitigated the proliferation of U-2OS human osteosarcoma cells through induction of membrane leakage, oxidative stress as well as apoptosis mediated by both intrinsic and extrinsic signaling pathways. These outcomes indicated that synthesized SnO2 NPs could be a promising novel therapeutic nanostructure for the treatment of osteosarcoma. Further investigations are required to discover whether SnO2 NPs can combine with different chemotherapeutics.
CRediT authorship contribution statement
Ruxin Ruan: Methodology, Writing – original draft, Visualization, Resources. Rui Chen: Methodology, Writing – original draft, Visualization, Resources. Huaixi Yu: Conceptualization, Supervision, Writing – review & editing.
Acknowledgments
The authors would like to express their deepest gratitude to the corresponding institutions for the technical support for this study
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Oxidative stress mediated cytotoxicity of tin (IV) oxide (SnO2) nanoparticles in human breast cancer (MCF-7) cells. Colloids Surf. B Biointerfaces. 2018;172:152-160.
- [Google Scholar]
- Hydrothermal method-based synthesized tin oxide nanoparticles: Albumin binding and antiproliferative activity against K562 cells. Mater. Sci. Eng. C. 2021;119:111649
- [Google Scholar]
- Zinc oxide nanoparticles selectively induce apoptosis in human cancer cells through reactive oxygen species. Int. J. Nanomed. 2012:845-857.
- [Google Scholar]
- Cytotoxic effect of green synthesized silver nanoparticles in MCF7 and MDA-MB-231 human breast cancer cells in vitro. Nucleus. 2020;63:191-202.
- [Google Scholar]
- Ultrasound-assisted rapid biological synthesis and characterization of silver nanoparticles using pomelo peel waste. Food Chem.. 2022;385:132602
- [Google Scholar]
- Comparative study of nanoparticle uptake and impact in murine lung, liver and kidney tissue slices. Nanotoxicology. 2020;14:847-865.
- [Google Scholar]
- Tin (IV) oxide (SnO2) nanoparticles inhibit the viability of cervical cancer HeLa cells through induction of apoptosis. Journal of Reports in Pharmaceutical Sciences. 2021;10:225.
- [Google Scholar]
- Bonjoch, Nuria Pedrol, and Pilar Ramos Tamayo. 2001. 'Protein content quantification by Bradford method.' in, Handbook of plant ecophysiology techniques (Springer).
- Sonochemical preparation and characterization of rGO/SnO2 nanocomposite: Electrochemical and gas sensing performance. Ceram. Int.. 2020;46:11290-21126.
- [Google Scholar]
- Process intensification of uniform loading of SnO2 nanoparticles on graphene oxide nanosheets using a novel ultrasound assisted in situ chemical precipitation method. Chem. Eng. Process.. 2013;70:48-54.
- [Google Scholar]
- Cancer nanotechnology: opportunities and challenges. Nat. Rev. Cancer. 2005;5:161-171.
- [Google Scholar]
- Extrinsic versus intrinsic apoptosis pathways in anticancer chemotherapy. Oncogene. 2006;25:4798-4811.
- [Google Scholar]
- Garg, Vijay, Harsh Sharma, Divya Rehani, Renu Kumari, Vipin Kumar, Manoj Kumar Tiwari, Shailesh Narain Sharma, and Manish Saxena. 2020. 'Synthesis and Characterization of Temperature Controlled SnO2 Nanoparticles by Solid-state Reaction Method'.
- Exploring the anticancer effects of tin oxide nanoparticles synthesized by pulsed laser ablation technique against breast cancer cell line through downregulation of PI3K/AKT/mTOR signaling pathway. Arab. J. Chem.. 2021;14:103212
- [Google Scholar]
- Induction of mitochondria mediated apoptosis in human ovarian cancer cells by folic acid coated tin oxide nanoparticles. PLoS One. 2021;16:e0258115.
- [Google Scholar]
- Synthesis of various size gold nanoparticles by chemical reduction method with different solvent polarity. Nanoscale Res. Lett.. 2020;15:1-10.
- [Google Scholar]
- Chemical, plant and microbial mediated synthesis of tin oxide nanoparticles: antimicrobial and anticancer potency. Braz. J. Chem. Eng. 2023:1-27.
- [Google Scholar]
- Biosynthesized tin oxide-sodium alginate-polyethylene glycol-carvacrol nanocomposite shows anticancer activity on esophagus squamous carcinoma cells. Process Biochem.. 2022;121:403-412.
- [Google Scholar]
- Microstructures and optical, electrical and photocatalytic properties of sonochemically and hydrothermally synthesized SnO2 nanoparticles. J. Alloy. Compd.. 2013;549:269-275.
- [Google Scholar]
- Sonochemical fabrication of inorganic nanoparticles for applications in catalysis. Ultrason. Sonochem.. 2021;71:105384
- [Google Scholar]
- Matrine induces caspase-dependent apoptosis in human osteosarcoma cells in vitro and in vivo through the upregulation of Bax and Fas/FasL and downregulation of Bcl-2. Cancer Chemother. Pharmacol.. 2012;69:317-331.
- [Google Scholar]
- Sonoproduction of nanobiomaterials–A critical review. Ultrason. Sonochem.. 2022;82:105887
- [Google Scholar]
- Microwave-assisted green synthesis of SnO2 nanoparticles and their photocatalytic degradation and antibacterial activities. Nanotechnology for Environmental Engineering. 2023;8:413-423.
- [Google Scholar]
- Preparation and characterization of SnO2 nanoparticles by hydrothermal route. International Nano Letters. 2012;2:17.
- [Google Scholar]
- Anti-proliferative, pro-apoptotic and anti-invasive effect of EC/EV system in human osteosarcoma. Oncotarget. 2017;8:54459.
- [Google Scholar]
- Syzygium cumini (L.) Extract-Derived Green Titanium Dioxide Nanoparticles Induce Caspase-Dependent Apoptosis in Hepatic Cancer Cells. Plants. 2023;12:3174.
- [Google Scholar]
- Anticandidal and in vitro anti-proliferative activity of sonochemically synthesized indium tin oxide nanoparticles. Sci. Rep.. 2020;10:3228.
- [Google Scholar]
- Evidence of reactive oxygen species (ROS) mediated apoptosis in Setaria cervi induced by green silver nanoparticles from Acacia auriculiformis at a very low dose. Exp. Parasitol.. 2016;160:39-48.
- [Google Scholar]
- Synthesis of pure and doped SnO2 and NiO nanoparticles and evaluation of their photocatalytic activity. Mater. Chem. Phys.. 2022;275:125190
- [Google Scholar]
- Metal and Metal Oxides Nanoparticles and Nanosystems in Anticancer and Antiviral Theragnostic Agents. Pharmaceutics. 2023;15:1181.
- [Google Scholar]
- Biosynthesis of SnO2 nanoparticles using bacterium Erwinia herbicola and their photocatalytic activity for degradation of dyes. Ind. Eng. Chem. Res.. 2014;53:13971-111399.
- [Google Scholar]
- Cytotoxicity study of Piper nigrum seed mediated synthesized SnO2 nanoparticles towards colorectal (HCT116) and lung cancer (A549) cell lines. J. Photochem. Photobiol. B Biol.. 2017;166:158-168.
- [Google Scholar]
- Metal nanoparticles in cancer: from synthesis and metabolism to cellular interactions. Journal of Nanostructure in Chemistry. 2023;13:321-348.
- [Google Scholar]
- Molecular mechanisms behind ROS regulation in cancer: A balancing act between augmented tumorigenesis and cell apoptosis. Arch. Toxicol.. 2023;97:103-120.
- [Google Scholar]
- The interplay of oncogenic signaling, oxidative stress and ferroptosis in cancer. Int. J. Cancer 2023
- [Google Scholar]
- Sonochemical synthesis of SnO2 nanoparticles and their preliminary study as Li insertion electrodes. Chem. Mater.. 2000;12:2557-2566.
- [Google Scholar]







