Translate this page into:
Toxic effects of imidacloprid and sulfoxaflor on Rana nigromaculata tadpoles: Growth, antioxidant indices and thyroid hormone-related endocrine system
⁎Corresponding author. plu@gzu.edu.cn (Ping Lu)
-
Received: ,
Accepted: ,
This article was originally published by Elsevier and was migrated to Scientific Scholar after the change of Publisher.
Peer review under responsibility of King Saud University.
Abstract
Imidacloprid and sulfoxaflor have potential damage to nontarget aquatic organisms. However, limited information has been provided on their underlying toxicity effects on Rana nigromaculata tadpoles. Thus, the acute toxicity and chronic effects of imidacloprid and sulfoxaflor on R. nigromaculata tadpoles were studied. Acute toxicity indicted that 96 h for LC50 values of imidacloprid and sulfoxaflor were 173.55 and 427.37 mg/L, respectively. In this research, we explored antioxidant enzymes, some biological indexes, hormone levels and expression of relative tadpole genes involved in thyroid hormone-dependent metaplastic development after exposure for 28 days under 1/10 and 1/100 LC50. Results showed an increase in the activity of antioxidant enzymes such as superoxide dismutase (SOD), catalase (CAT) and glutathione S-transferase (GST) in the tadpoles, and a low bioconcentration level with a bioconcentration factor (BCFs) < 1. The impact on the development of tadpoles was induced by the breakdown of hormonal levels engaged in metamorphosis. According to the real-time PCR results, imidacloprid and sulfoxaflor delayed amphibian metamorphosis by modifying mRNA expression, indicating that imidacloprid and sulfoxaflor may have an endocrine-disrupting effect on R. nigromaculata tadpoles. These findings were indicative of the toxicity of imidacloprid and sulfoxaflor to R. nigromaculata tadpoles.
Keywords
Imidacloprid
Sulfoxaflor
Rana nigromaculata tadpoles
Thyroid disruption
Gene expression
1 Introduction
Rice is one of the major food crops in China and plays an important role in ensuring national food security. Over the past few decades, the output of rice in China has increased rapidly due to improved varieties, advances in farming techniques and the extensive use of chemical fertilizers and pesticides. However, the period when pesticides are often used in agricultural crops corresponds to the period when amphibian larvae breed and develop (Mann et al., 2009). Owing to pesticide runoff and atmospheric drift, amphibian habitats are likely to be influenced (Lehman and Williams, 2010), and amphibian species encounter great risk due to the high penetration into their skin and their susceptibility to environmental chemicals, especially for tadpoles.
Rana nigromaculata tadpoles is a species of amphibians with wide distribution and abundance in China, where they live in various habitats, including agricultural sites. Thus, damage from toxic ingestion starts at birth. Notably, amphibians are known to prey on crop pests, their movement on agricultural land can reduce the occurrence of rice pests (Teng et al., 2016), and tadpoles are the larvae of amphibians that supply the critical link in the transition between lower and higher trophic levels (Junges et al., 2012). R. nigromaculata tadpoles is an excellent model to be used for the assessment of environmental risk due to its naturally low mortality rate with comparatively rapid production times (Zhang et al., 2019). Therefore, it is essential to assess the toxicity of insecticides to R. nigromaculata tadpoles.
Neonicotinoids are extensively employed to protect crops and prevent insect pests due to their promising wide spectrum and highly effective pesticidal activity (Frank and Tooker, 2020; Jacob et al.,2019). Imidacloprid [1-(6-chloro-3-pyridylmethyl)-N-nitroimidazolidin-2-ylideneamine], a neonicotinoid synthetic insecticide, is a stimulator of postsynaptic nicotinic acetylcholine receptors (nAChRs) (Radwan and Mohamed, 2013). It is the most used seed or soil treatment for some agricultural products, such as rice, cereals, maize and potatoes (Badgujar et al., 2013), and has become a widely used and highly effective insecticide (Rios et al., 2017). Sulfoxaflor [[methyl-oxo-[1-[6-(trifluoromethyl) pyridin-3-yl] ethyl]-λ6-sulfanylidene] cyanamide], a fourth-generation neonicotinoid, affects the nAChR of insects in a unique way compared with other neonicotinoids, and it is recognized as a significant new tool in insecticide resistance management programs because of its absence of insecticidal cross resistance (Cutler et al.,2013; Jacob et al., 2019).
Researches has shown that with the application of neonicotinoids around the world, neonicotinoids have been detected worldwide in soil and superficial water on account of their elevated levels of solubility in water and extended period of degradation in soil (Yi et al., 2019). Growing evidence indicated that the widespread use of neonicotinoid insecticides may pose potential risks to amphibians (Saka and Tada, 2021; Hrynyk et al., 2018). Our previous study has reported that when exposed to sublethal concentration of sulfoxaflor, sulfoxaflor induced oxidative stress in zebrafish after different times of exposure (Deng et al., 2022), indicating that sulfoxaflor has potential damage to nontarget organisms. Several studies have suggested that sulfoxaflor presented a risk to honeybees and sub-lethal concentrations of sulfoxaflor may have caused adverse impacts on ants (Wu et al., 2017; Pan et al., 2017), and sulfoxaflor have acute toxicity and sublethal effects to earthworms from oxidative stress and metabolomics (Fang et al., 2018). Meanwhile, researches have reported that imidacloprid exerts sublethal impact on amphibians, for example, imidacloprid inhibited the predator-resistant escape behavior of wood frogs (Lee-Jenkins and Robinson, 2018) and delays the metamorphosis of wood frogs (Robinson et al., 2017). The metamorphosis is regulated by the amphibian thyroid, and the amphibian thyroid also has the capacity to secrete thyroxine (T3) and triiodothyronine (T4) (Carr and Patiño, 2011), having a critical role in the growth, and metabolism of vertebrates (Heijlen et al., 2013). Studies have demonstrated that insecticides not only cause delays in growth and development, but also change the genes related to TH levels in Xenopus laevis tadpoles, leading to the disruption of thyroid endocrine (Li et al., 2016; Liu et al., 2021). However, few studies have been performed to evaluate the impact (especially, sublethal effects) using imidacloprid and sulfoxaflor on R. nigromaculata tadpoles.
Hence, this study aimed to evaluate the risk of imidacloprid and sulfoxaflor on R. nigromaculata tadpoles. In order to assess the impact of imidacloprid and sulfoxaflor, thyroid hormone (TH) levels and genes which are involved in the metabolic pathway, including and deiodinases (dio2 and dio3) and thyroid hormone receptors (TRα and TRβ), and their expression in relation to TH levels were investigated after 28 days exposure. Besides biological indexes including wet weight, snout-to-vent length (SVL), antioxidant enzyme activity and the accumulation of imidacloprid and sulfoxaflor in tadpole was investigated to understand the comprehensive risk to Rana nigromaculata tadpole.
2 Materials and methods
2.1 Chemical reagent
Analytical imidacloprid (CAS: 138261-41, 99.70%) and sulfoxaflor (CAS: 946578-00-3, 98.0%) standards were obtained from the Ministry of Agriculture, Tianjin Agricultural Environment Station (Tianjin, China). Commercial assay kits (ELISA) for T3 and T4 were obtained from Shanghai Yaji Biological Technology Co., Ltd. (Shanghai, China). Commercial assay kits for superoxide dismutase (SOD), catalase (CAT), and glutathione S-transferase (GST) were obtained from Suzhou Comin Biotech (Suzhou, China). 3-Aminobenzoic acid ethyl ester (MS-222) was sourced from Sigma-Aldrich (USA). Other analytical reagents were acquired from Youpu Reagent Co., Ltd. (Tianjin, China).
2.2 Experimental animals and treatment
R. nigromaculata (Gosner 25–26) tadpoles were purchased from the State Key Laboratory of Environmental Chemistry and Ecotoxicology, Research Center for Eco Environmental Sciences (Beijing, China). The tadpoles were housed in a 20 L aquarium containing charcoal filtered-tap water, adapting to laboratory requirements where the temperature remained 24 ± 1 °C and a cycle of 12 h of light/12 h of darkness, and were fed 2 times by commercial diets every day (Totoro Supplies, Hong Kong, China). The mortality rate was under 5% during the domestication period and refreshed water at 24 h intervals. All experiments were carried out under review and permission of the Guizhou University Laboratory Animal Ethics Committee with the ethics authorization number EAE-GZU-2019-E003.
2.3 Experimental design
2.3.1 Bioconcentration-elimination of imidacloprid and sulfoxaflor
In bioconcentration experiments of imidacloprid and sulfoxaflor, two exposure concentrations were set in accordance with 1/100 and 1/10 of the results of 96 h LC50. One hundred tadpoles were picked at random and moved into glasses filled with 20 L of the corresponding concentration of imidacloprid and sulfoxaflor, separately. The exposed solution was changed every 2 days, each treatment group was set with three repetitions, and other breeding requirements as mentioned above. Tadpoles were randomly removed from each glass after 1, 3, 5, 7, 9, 14, 21, and 28 days of the exposure enrichment stage and 2 h, and 0.5, 1, 2, 3, 4, and 5 days of the water elimination stage. The tadpole and water samples were stored in a refrigerator at –20 °C for liquid chromatography with tandem mass spectrometry (LC-MS/MS) analysis. The bioconcentration and elimination test were carried out according to the guideline of OECD 305 (OECD, 2011).
2.3.2 Acute exposure experiments
Tadpoles were selected and placed randomly into a 5-L flask with 4-L of filtered solution. Imidacloprid and sulfoxaflor solvents were made up with concentration 90–300 mg/L, and 244–645 mg/L, respectively. The concentrations leading to the death of 50% of the experimental groups (LC50) was documented. The whole process was conducted on basis of instructions for testing the environmental security of pesticides in accordance with the OECD guideline 203 (fish, acute toxicity test) (OECD, 2019) and GB/T 31270.18–2014 (amphibian acute toxicity test) (AQSIQ and SAC, 2014).
2.3.3 Toxicity experiment
Sixty tadpoles were picked at random and put in each aquarium with 20 L of the test solution of imidacloprid and sulfoxaflor. The exposure concentration for the toxicity test were 1.74, 17.36 mg/L, and 4.27, 42.73 mg/L (1/100 and 1/10 of the LC50 of tadpole, respectively). Dechlorinated tap water was set as a control, and every treatment had three replicates. The exposures were conducted under the laboratory conditions mentioned above. The exposed solution was to be renewed at 3-day intervals. the tadpoles were fed twice a day. After 1, 7, 14, 21 and 28 days of exposure, the wet weight and SVL of tadpoles were measured, antioxidant enzymes were determined after anesthetization in MS-222 (100 mg/L). After 28 days of exposure, tadpoles were randomly selected and anesthetized using MS-222 (100 mg/L) for TH measurement and gene expression.
2.3.4 Measurement of antioxidant enzyme activities
Ten tadpoles from each aquarium were selected for the analysis (n = 3). After 1, 7, 14, 21 and 28 days of exposure, MS-222 was employed to anesthetize the tadpoles. Tadpoles were homogenized, then centrifuged for 10 min (10,000 g, 4 °C). The supernatant was employed to determine the SOD, CAT and GST activities by using assay kits (Suzhou Comin Biotech).
2.3.5 Levels of TH
After 28 days of exposure, five tadpoles per aquarium were euthanized using MS-222, weighed and placed into a 10 mL centrifuge tube. A proper amount of normal saline was put to the centrifuge tube, homogenized in an ice water bath for 3 min, and then centrifuged for 10 min (4 °C, 3,000 g). Subsequently, T3 and T4 in the supernatant were measured using the ELISA kit.
2.3.6 cDNA synthesis and real-time RT-PCR
All tadpoles were dissected and gathered and TRIzol reagent (Tiangen Biotech, Beijing, China) was employed to extract the total RNA of tadpoles after 28 days of exposure. The concentration of total RNA was determined using a micro spectrophotometer with absorbance at 260 nm (DS-11, 164 DeNovix Inc, USA), and then gel electrophoresis in 1% agarose gel was used to verify the RNA quality. Then RNA was reversely transferred to cDNA in accordance with the manufacturer's instructions with a FastQuant RT kit (Tiangen Biotech, Beijing, China).
A SLightCycler480 II system (Roche, Rotkreuz, Switzerland) was used to confirm and quantify the changes in gene expression. Specific primers are provided in Table 1. Amplification was performed following the protocol of the manufacturer to identify these genes for specific amplification with PCR requirements of 95 °C for 3 min, then 45 cycles of 95 °C for 5 s of annealing at different temperatures (Table 1) and 60 °C for 30 s. The expression of ribosomal protein L8 was normalized using the 2-ΔΔCt method to determine the fold change in gene expression.
Gene
Genbank
accessionPrimer sequences
(5′ to 3′)Size
(bp)Annealing
(°C)
RPL13A
MG844184
Forward: TACCGCAACAAACTCAAATACC
175
60
Revers: CAAAGACCTTCAGTCGCTCC
dio2
MH892455
Forward: TGCCTACAAACAGGTGAAGCT
150
60
Revers: CCAAAGTTGACAACAAGAGGG
dio3
MH892456
Forward: GAGTCCCTGAAGGCGGTCT
127
60
Revers: AGTCCAGGATCCGGCACA
TRα
KC139354.1
Forward: GGGGTGGTCTCAGATGCTATAT
174
60
Revers: TTCAAAGGCGAGGAGGTAAG
TRβ
KC139355.1
Forward: AAGTGAGACCTTAACGCTGAATG
115
60
Revers: TGAAGACAGTGATACGCCCAA
2.4 Extraction and analysis of imidacloprid and sulfoxaflor
2.4.1 Sample pretreatment
The water sample was filtered using a nylon filter with 0.22 µm and moved to a vial for the determination of imidacloprid and sulfoxaflor without pretreatment.
For the tadpole samples, 0.20 g of tadpoles were weighed, added to 2 mL of methanol in a plastic centrifuge tube and ground with an electric tissue homogenizer. The tadpole samples were vortexed at 2500 rpm/min for 5 min. Purification agents were added to the samples exposed to imidacloprid (0.2 g MgSO4) and sulfoxaflor (0.2 g MgSO4 and 0.1 g NaCl), then vortexed for 3 min, separately. The samples were collected by centrifugation for 5 min (8000 rpm/min, 4 °C), then 1.5 mL of top-serum was removed and pipetted into round-bottomed flask, and evaporated by rotation (40 °C water bath) until vacuum dried. The evaporated residue was dissolved by the addition of acetonitrile (1.5 mL), supernatant was placed in plastic centrifuge tube, 100 mg of PSA was added, and the sample was swirled for 1 min and the centrifugation was carried out for 3 min. The solution was passed with a 0.22 µm nylon filter and moved to a vial with a lined tube, then analyzed by LC-MS/MS.
2.4.2 Quantification analysis
Imidacloprid and sulfoxaflor in R nigromaculata tadpoles and water were analyzed by liquid chromatography-mass spectrometry LC-MS/MS. The detailed analytical methods for imidacloprid and sulfoxaflor are given in Supporting Information Test S1.
2.5 Statistical analyses
IBM SPSS 26.0 was employed for statistics analysis. For SVL, weight, TH levels, gene expression, and antioxidant enzyme activity, differences in statistics with respect to the exposure and control groups were analyzed by one-way ANOVA, with various letters denoting each statistical difference with respect to the exposure and control groups (p < 0.05). The probit equation was performed to calculate the LC50 and associated 95% confidence intervals. The results presented the mean standard deviation (SD). The bioconcentration factor (BCF) of imidacloprid and sulfoxaflor in tadpoles was calculated using the following equation: where Ct (mg/kg) denoted the concentration of imidacloprid and sulfoxaflor in tadpoles, Cw (mg/L) denoted concentration of imidacloprid and sulfoxaflor in the water.
3 Results and discussion
3.1 Validation of the LC-MS/MS method
Recovery experiments of imidacloprid and sulfoxaflor were carried out. The limit of quantification (LOQ) for every target compound is computed from the signal-to-noise ratio (S/N) around the target peak, defined as 10 times the S/N (Guo et al., 2022). The LOQ of imidacloprid and sulfoxaflor of the method for tadpole was 0.05 mg/kg and 0.3 mg/kg, respectively. The method provided average recoveries of imidacloprid and sulfoxaflor from 76.18% and 96.82%, with relative standard deviations (RSD) from 1.75% to 15.41%, from 82.51% to 112.51%, with RSD from 1.06% to 5.79% in the tadpole, respectively (Table S3 and S5). The calibration curve with R2 > 0.993, was shown in Tables S2 and S4. In conclusion, the approach is well suited for imidacloprid and sulfoxaflor determination.
3.2 Bioconcentration and elimination of imidacloprid and sulfoxaflor
The concentration of imidacloprid and sulfoxaflor in the water was maintained at a constant level as the solution was renewed every two days (Fig. S1). In our study, it was shown that imidacloprid can accumulate rapidly during the biological uptake period and the concentrations of 1.74 and 17.36 mg/L in tadpoles reached the highest level at 7th and 6th days, then kept at a relative steady state until the 28th day (Fig. 1A). The elimination process was not fast, and the concentration of imidacloprid in tadpole at 1.74 and 17.36 mg/L reached the lowest values of 0.015 and 0.08 mg/kg respectively in 5 days after the tadpoles were moved to clean water (Fig. 1A). With the enrichment of sulfoxaflor with the concentration of 4.27 and 42.73 mg/L in tadpoles, the concentrations increased to the level of maximum at 21th and 9th days. The elimination process shown that the concentration of sulfoxaflor in tadpoles at 4.27 and 42.73 mg/L reached to the lowest value of 0.31and 0.61 mg/kg, respectively, in 5 days after the tadpoles were moved to clean water (Fig. 1B). As a result, the research showed that imidacloprid can accumulate rapidly in tadpoles shortly after exposure to sublethal concentrations compared with sulfoxaflor. We also inferred that imidacloprid and sulfoxaflor may continue to decline to undetectable levels in tadpoles over a longer period of time.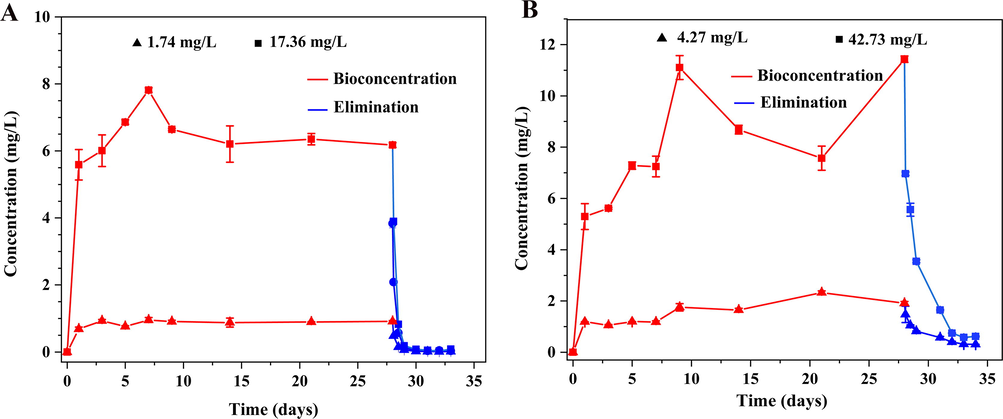
Concentrations of imidacloprid and sulfoxaflor in tadpoles (A) 1.74 and 17.36 mg/L, (B) 4.27 and 42.73 mg/L. (Error bars represent the mean ± SD).
The BCFs of imidacloprid at 1.74 and 17.36 mg/L were 0.559 and 0.39, the BCFs of sulfoxaflor at 4.27 and 42.73 mg/L were 0.359 and 0.191, respectively, these values indicated that imidacloprid and sulfoxaflor have potential accumulation in tadpoles.
3.3 Acute toxicity
For the assessment of the effect of imidacloprid and sulfoxaflor on acute toxicity in R. nigromaculata tadpoles. 96 h-LC50 of imidacloprid and sulfoxaflor was documented. The 96 h-LC50 of imidacloprid and sulfoxaflor was 173.55 and 427.37 mg/L, respectively (Table S1), indicating that imidacloprid have greater toxicity for R. nigromaculata tadpoles than sulfoxaflor. On the basis of their LC50, imidacloprid and sulfoxaflor could be categorized as low-toxicity pesticides to R. nigromaculata tadpoles. Although imidacloprid and sulfoxaflor had low toxicity, our previous study showed that the subacute sulfoxaflor posed impact to organisms (Deng et al., 2022). Therefore, the subacute influence of sulfoxaflor and imidacloprid on R. nigromaculata tadpoles should be assessed.
3.4 Oxidative stress effects
The first line of cellular response to antioxidants is catalyzing the disproportionation of O22− to H2O2 and O2, and to prevent the production of superoxide anion radicals by removing reactive oxygen species (ROS) via SOD (Sies, 1997). CAT subsequently degrades H2O2 into oxygen and water. SOD and CAT perform a key role in the antioxidant defense system, with their task being to defend from oxidative stress (Menon and Rozman, 2007). In our research, the SOD activity was significantly increased in exposure of imidacloprid compared with those control group (Fig. 2A, p < 0.05), compared with the SOD activity under the treatment at 1.74 mg/L, the activity was increased in 17.36 mg/L (1, 7, 14, and 21 days), exhibited a time-dependent increase but decreased at 28 days (Fig. 2A, p < 0.05). The CAT activity in 17.36 mg/L was significant increased compared with control (1, 7, 14, 21 and 28 days) (Fig. 2B, p < 0.05), and compared with the CAT activity under the treatment at 1.74 mg/L, the activity was decreased in 17.36 mg/L (14, 21 and 28 days) (Fig. 2B, p < 0.05). The SOD activity was significantly increased compared with the control in the exposure of sulfoxaflor (7, 14, and 21 days, p < 0.05) (Fig. 2C). The CAT activity was all significantly increased in comparison with that under other exposure of sulfoxaflor (Fig. 2D, p < 0.05), and exhibited a time-dependent increase (7, 14, 21, and 28 days).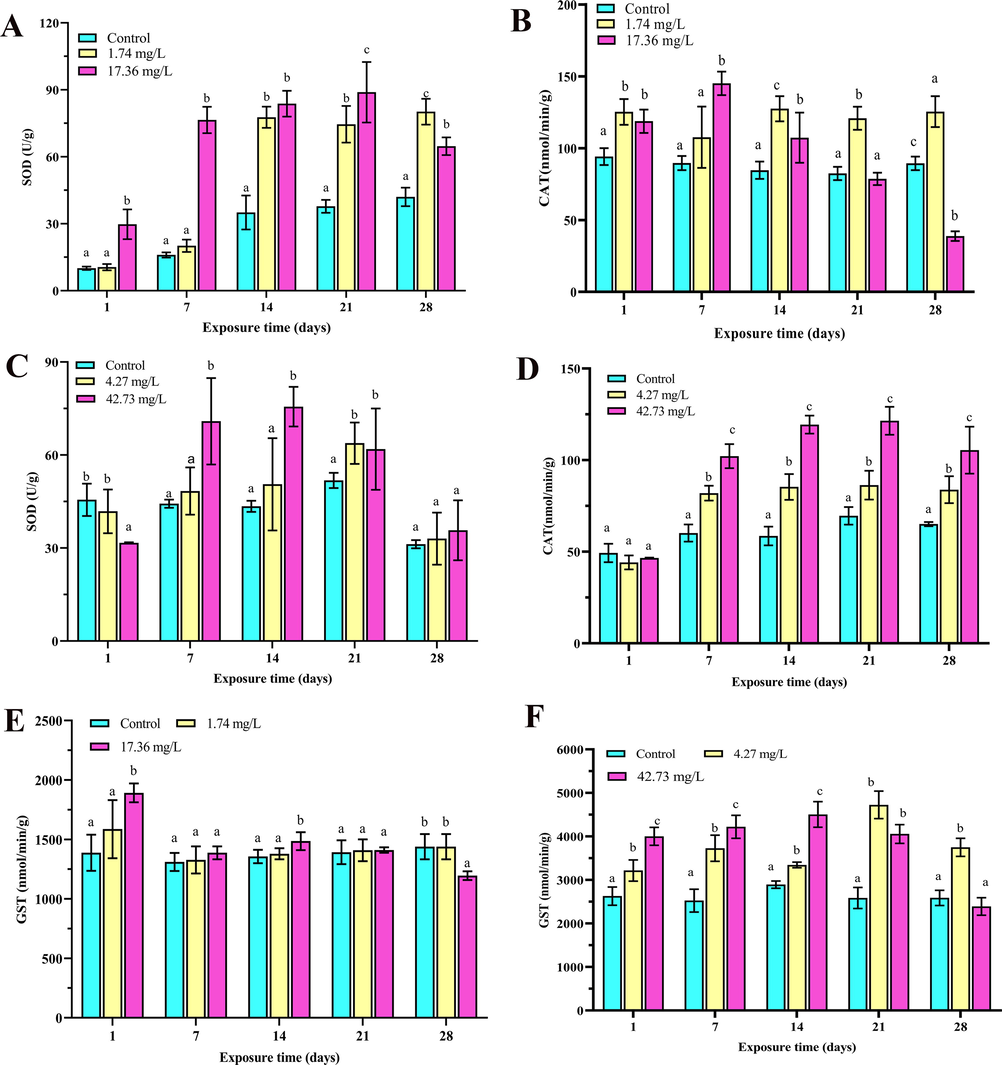
Activity of SOD, CAT and GST in R. nigromaculata tadpoles with exposure to imidacloprid and sulfoxaflor after 1, 7, 14, 21 and 28 days (n = 6). Significant differences between treatments are indicated by different letters, p < 0.05. Error bars represent SDs.
The increase in SOD and CAT activity in the exposure of imidacloprid and sulfoxaflor may be consequence of the increase in ROS, which facilitates the activation of the enzymes (Pinya et al., 2016). Meanwhile, we discovered that higher doses of imidacloprid reduced the SOD activity or CAT activity, which might be attributed to an excessive accumulation of ROS and the suppression of enzymatic activity, resulting in reduced defense response (Rodríguez-Serrano et al., 2006). In return, the enzymes of SOD and CAT were unable to combat the high levels of oxidative stress due to the increased concentrations of imidacloprid, and over oxidative stress might conversely render SOD or CAT inactive activity (Bagnyukova et al., 2006). Researches have reported inhibition of CAT activity in tadpoles with different insecticide exposures (Rutkoski et al., 2021; Sun et al., 2014), which was similar with our results.
GST is a second-stage detoxification enzyme known to catalyze the binding of glutathione (GSH) to exogenous substances and their metabolites (Wu et al., 2007). As for imidacloprid, the concentrations of GST were significantly increased when exposed to 17.36 mg/L (1 and 14 days, p < 0.05) (Fig. 2E). The concentrations of GST were significantly increased in tadpoles exposed to sulfoxaflor in all treatment groups when compared with that in the control (1, 7, 14, 21 and 28 days, p < 0.05) (Fig. 2F). The increase in GST activity may be due to the induction of detoxification by imidacloprid and sulfoxaflor through binding intracellular GSH and harmful metabolites, and imidacloprid and sulfoxaflor induced excessive ROS and themselves may also contribute to the activation of GST. However, when exposed to 28 days, the concentrations of GST exposed to imidacloprid were significantly decreased in 17.36 mg/L (p < 0.05) (Fig. 2E). For sulfoxaflor exposure, the GST levels were significantly decreased to 42.73 mg/L at 21and 28 days when compared with that in 4.27 mg/L (p < 0.05) (Fig. 2F). The study explained that this reduction in activity of the enzyme appears to be associated with an overconsumption of GSH which serves as a precursor and variation in GST formation induced by various intermediary metabolites (Ge et al., 2015). In this research, the GST activity of tadpoles was affected by imidacloprid and sulfoxaflor, indicating that the GST system is probably the pathway of detoxification from imidacloprid and sulfoxaflor in tadpoles.
3.5 Growth index and TH
3.5.1 Growth indexes
The inhibition of growth is a biomarker of toxicity development (Xu and Huang, 2017). In this research, the effects on biological indexes (weight, SVL) were evaluated when tadpoles were exposed to imidacloprid and sulfoxaflor. For the imidacloprid-treated group, the weight of imidacloprid exposure was significantly decrease (p < 0.05) in comparison to that for control (Fig. 3A) and SVL was significantly decreased (14, 21, and 28 days, p < 0.05) (Fig. 3B). Weight was significantly decrease in all treatments of sulfoxaflor at 21 and 28 days compared with that in the control (Fig. 3C), SVL at 14 days was not significantly difference from that of the control, decreasing at 21 days only by 4.27 mg/L exposure (p < 0.05), and SVL was significantly decreased at 28 days in all treatments of sulfoxaflor (p < 0.05) (Fig. 3D). Some studies have reported that development and metamorphosis in Xenopus laevis were altered by exposing to insecticides (Liu et al., 2021), and the weight of R. nigromaculata tadpoles was significantly decreased after 28 days of exposure to triadimefon (Zhang et al., 2018), which was in accordance with our findings. In our study, reduction in SVL and weight exposed to sulfoxaflor and imidacloprid in all treatments after 28 days of exposure may be ascribed to disturbances in metabolism of the thyroid.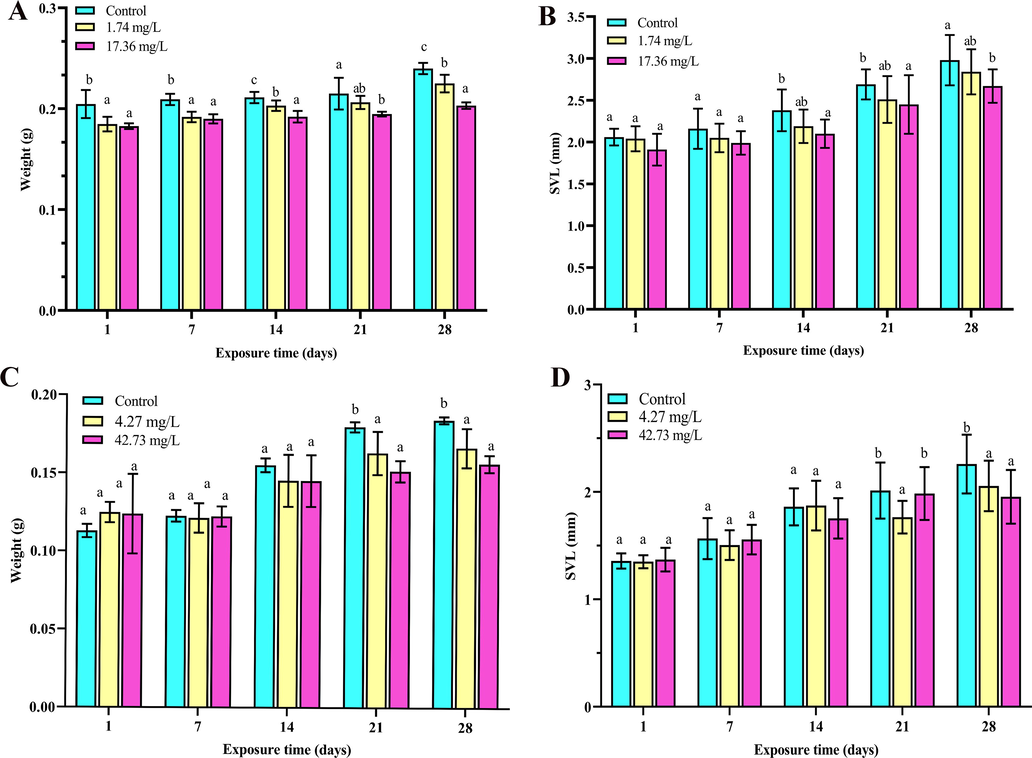
Developmental endpoints of R. nigromaculata tadpoles exposed to different concentrations of imidacloprid and sulfoxaflor for 1, 7, 14, 21 and 28 days (n = 14). Mean weight (A C), SVL (B D). Values represent mean ± SD. Significant differences between treatments are indicated by different letters, p < 0.05. Error bars represent SDs.
3.5.2 Levels of thyroid hormone
The influence of imidacloprid and sulfoxaflor on T3 and T4 concentrations was evaluated. T3 concentrations were observed to decrease significantly during exposure to all treatments of imidacloprid, and T4 concentrations were also significantly decreased in all treatments compared with control (p < 0.05) (Fig. 4A–B). When exposed to 42.73 mg/L of sulfoxaflor, the levels of T3 and T4 were decreased significantly compared with the control (p < 0.05) (Fig. 4C–D). These results clearly showed an endocrine-disrupting effect of imidacloprid and sulfoxaflor on the thyroid system of tadpoles. In our study, significant suppression of metamorphosis development (SVL and weight) in parallel with a decrease in T3 and T4 levels in 28 days was noticed, demonstrating that suppressive effect of imidacloprid and sulfoxaflor on metamorphosis might be attributed to reduction of thyroid hormones levels. A previous study showed that imidacloprid altered TH levels of lizards (Wang et al., 2020). Above all, we concluded that the T3 and T4 synthesis in R. nigromaculata tadpoles was inhibited by imidacloprid and sulfoxaflor.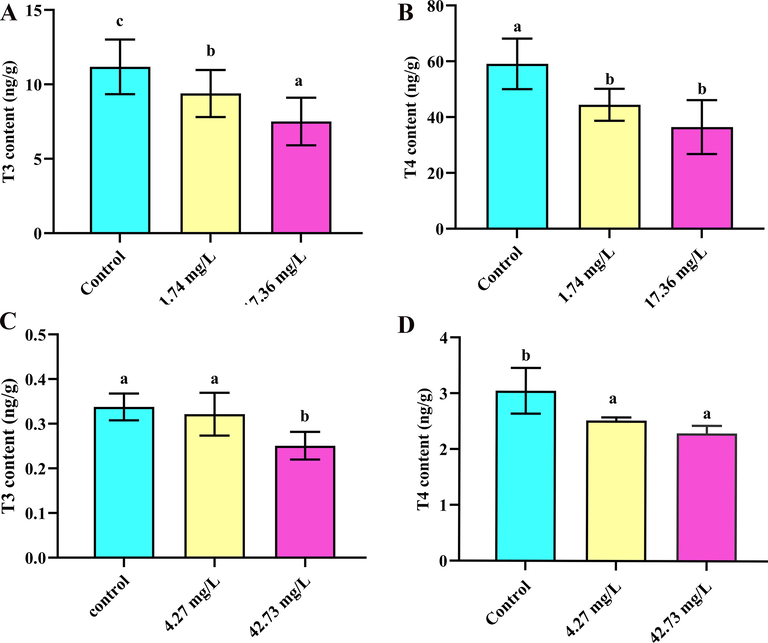
T3 and T4 levels in R. nigromaculata tadpoles exposed to imidacloprid and sulfoxaflor for 28 days (n = 6). Significant differences between treatments are indicated by different letters, p < 0.05. Error bars represent SDs.
3.5.3 Relative expression of TH-dependent genes
The mRNA levels of dio2 and dio3 (encoded as iodothyronine deiodinase (DIO2 and 3)). Type 2 deiodinase plays an essential part in transforming T4 into the more inactive T3, while T4 and T3 are inactivated to the bio-inactive inverted T3 and T2 by type 3 deiodinase, separately (Lorenz et al., 2018). As for imidacloprid, the levels of dio2 gene expression increased at 1.74 mg/L, and significantly downregulated at 17.36 mg/L (p < 0.05) (Fig. 5A). The dio3 gene expression was significantly upregulated at in all treatments (p < 0.05) (Fig. 5B). The results revealed that imidacloprid reduced the dio2 gene expression. which decreased the conversion from T4 to T3 leading to a decrease in T3 levels. In contrast with the results for dio2, the expression of dio3 gene was increased, which inactivated T4 and T3. The downregulation of dio2 expression and the upregulation of dio3 expression by 17.36 mg/L of imidacloprid were in accordance with the inhibition of the T3 and T4 we detected. When exposed to 42.73 mg/L of sulfoxaflor, compared with the control, the dio2 gene expression was not significantly different (p < 0.05, Fig. 5C). Therefore, there was no significant effect of sulfoxaflor on dio2 gene expression at 42.73 mg/L. However, compared with the control, during exposure to 4.27 mg/L, an upregulation was observed (p < 0.05, Fig. 5C). After 28 days exposure, the expression of the dio3 gene was remarkably upregulated in all treatments (p < 0.05) (Fig. 5D). The dio2 expression had no significant changes, and the upregulation of dio3 was in accordance with the levels of T3 and T4 we detected. On the contrary, a previous study observed a downregulation of dio3 in R. nigromaculata tadpoles after 21 days of triazolone exposure, which was likely related to the observed recovery of T3 concentrations (Li et al., 2016).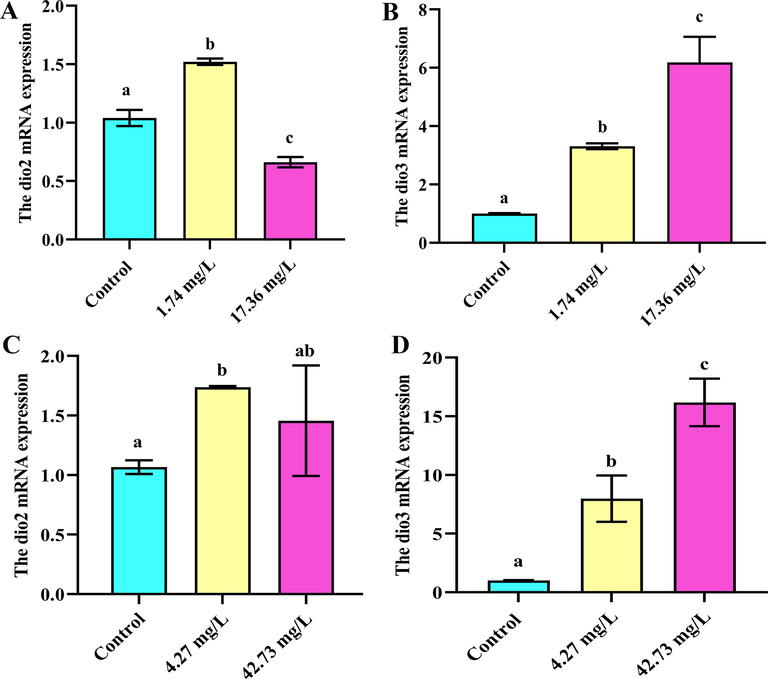
Expression of dio2 and dio3 exposed to imidacloprid and sulfoxaflor after 28 days (n = 3). Significant differences between treatments are indicated by different letters, p < 0.05. Error bars represent SDs.
In our study, the dio3 expression inactivates T4 and T3 and the elevated dio3 expression levels contribute to reduction in TH levels in the target tissue. TH is indispensable to normal growth. For instance, tail muscles die as a result of TH induction; limb muscles grow and differentiate in response to the hormone (Brown and Cai, 2007). As a result, reducing the TH levels in the target tissue might result in suppression of both growth and metamorphosis. On basis of the results of the expression of dio2 and dio3, we inferred that imidacloprid and sulfoxaflor disrupt HPT regulation on the genetic level.
For all vertebrates, it is known that two thyroid hormone receptors (TR), called TRα and TRβ, affect the development of organisms primarily by combining with the TR (Darras et al., 2011). Abnormalities in TRα and TRβ might contribute to a cascade in which THs fail to associate with TRs and activate post-receptor reactions (Yu et al., 2013). In the present study, imidacloprid and sulfoxaflor exposure had a negative effect on TRα expression. When exposed to imidacloprid, gene expression levels of TRα were significantly decreased to 17.36 mg/L compared with the control (p < 0.05) (Fig. 6A), and gene expression levels of TRα showed an increase to 4.27 mg/L and 42.73 mg/L in sulfoxaflor treatments. (p < 0.05) (Fig. 6C). The TRα mRNA levels have a significant change in zebrafish (Wang et al., 2020), which was similar with our results. Imidacloprid and sulfoxaflor exposure also had a negative effect on TRβ expression. In particular, the level of expressions of TRβ was decreased in all treatments of imidacloprid (p < 0.05) (Fig. 6B). Likewise, there was significantly downregulated TRβ expression detected in lizards, which was same as our results (Wang et al., 2020). As for sulfoxaflor exposure, the expression of TRβ was dramatically downregulated in all treatments compared with control group (p < 0.05), and groups have no significant difference from each other (Fig. 6D). The reduced TRβ in tadpoles was associated with the delayed metamorphic response (Navarro-Martín et al., 2014). Accordingly, we concluded that sulfoxaflor and imidacloprid may delay metamorphosis through controlling the expression of TRβ and have an influence on the endocrine system.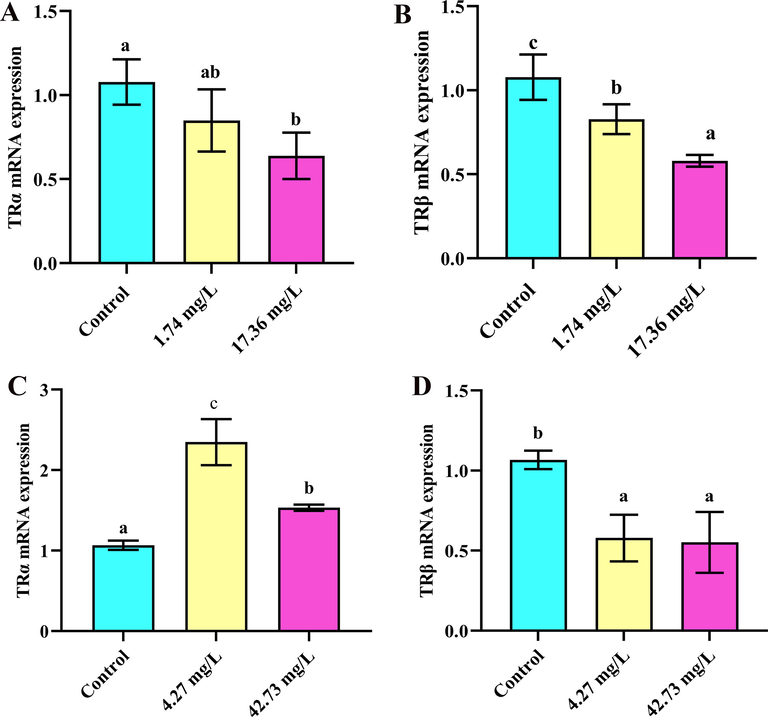
TRα and TRβ expression exposed to imidacloprid and sulfoxaflor after 28 days (n = 3). Significant differences between treatments are indicated by different letters, p < 0.05. Error bars represent SDs.
4 Conclusion
In the present study, the aim of the study was to investigate the impact of exposure to imidacloprid and sulfoxaflor on R. nigromaculata tadpoles. Our results indicated that imidacloprid and sulfoxaflor have shown low-toxicity to R. nigromaculata tadpoles based on the acute toxicity. Oxidative stress was caused when exposed to imidacloprid and sulfoxaflor, respectively, and imidacloprid and sulfoxaflor inhibited the growth of R. nigromaculata tadpoles due to decreased TH levels. Although exposure to imidacloprid and sulfoxaflor showed same trend of the dio3 and TRβ gene expression, there was no significant effect of sulfoxaflor on dio2 gene expression exposed to high level compared with imidacloprid, the gene expression levels of TRα showed an increase in sulfoxaflor but decreased in imidacloprid treatments. On basis of the results of the expression of dio2 and dio3, we inferred that imidacloprid and sulfoxaflor disrupt HPT regulation on the genetic level, and imidacloprid and sulfoxaflor may delay metamorphosis through controlling the expression of TRβ and have an influence on the endocrine system. In sum, imidacloprid and sulfoxaflor probably have negative impacts on the development of R. nigromaculata tadpoles through disruption of the thyroid hormone pathway. This research may contribute to more stringent management of the levels of insecticides in the aquatic environment, providing support for maintaining a healthy ecosystem.
CRediT authorship contribution statement
Xia Zhou: Writing – original draft, Visualization, Data curation. Yao Deng: Formal analysis, Investigation. Ran Wang: Methodology. Fang Wang: Investigation. Honghao Cui: Investigation. Deyu Hu: Funding acquisition, Supervision, Project administration. Ping Lu: Funding acquisition, Supervision, Project administration.
Acknowledgments
This work was supported by Guizhou Provincial Science and Technology Project (ZK [2022] ZD-013 and [2020]4Y100), Postgraduate Education Innovation Program in Guizhou Province (YJSKYJJ [2021]036), and Program of Introducing Talents to Chinese Universities (111 Program, D20023).
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- AQSIQ, 2014. (General Administration of Quality Supervision, Inspection and Quarantine, People’s Republic of China) and SAC (Standardization Administration of China) GB/T 31270.18-2014 (Test Guidelines on Environmental Safety Assessment for Chemical Pesticides (Part 18: Amphibian Acute Toxicity Test)) Standardization Administration of the People’s Republic of China, Beijing, China (2014).
- Immunotoxic effects of imidacloprid following 28 days of oral exposure in BALB/c mice. Environ. Toxicol. Phar.. 2013;35:408e418.
- [Google Scholar]
- Coordinated response of goldfish antioxidant defenses to environmental stress. Aquat. Toxicol.. 2006;78:325-331.
- [CrossRef] [Google Scholar]
- The hypothalamus–pituitary–thyroid axis in teleosts and amphibians: Endocrine disruption and its consequences to natural populations. Gen. Comp. Endocrinol.. 2011;170(2):299-312.
- [CrossRef] [Google Scholar]
- Investigating the mode of action of sulfoxaflor: a fourth-generation neonicotinoid. Pest. Manag. Sci.. 2013;69:607-619.
- [CrossRef] [Google Scholar]
- Thyroid hormone receptors in two model species for vertebrate embryonic development: chicken and Zebrafish. J. Thyroid Res. 2011 402320
- [CrossRef] [Google Scholar]
- Enantioselective bioaccumulation and toxicity of rac-sulfoxaflor in zebrafish (Danio rerio) Sci. Total Environ.. 2022;817
- [CrossRef] [Google Scholar]
- Lethal toxicity and sublethal metabolic interference effects of sulfoxaflor on the earthworm (Eisenia fetida) J. Agric. Food Chem.. 2018;66(45):11902-11908.
- [CrossRef] [Google Scholar]
- Neonicotinoids pose undocumented threats to food webs. Proc. Natl. Acad. Sci. U. S. A.. 2020;117(37):22609-22613.
- [CrossRef] [Google Scholar]
- Oxidative Stress and DNA Damage Induced by Imidacloprid in Zebrafish (Danio rerio) J. Agric. Food Chem.. 2015;63(6):1856-1862.
- [CrossRef] [Google Scholar]
- Insight into the toxic effects, bioconcentration and oxidative stress of acetamiprid on Rana nigromaculata tadpoles. Chemosphere. 2022;305:135380.
- [CrossRef] [Google Scholar]
- Zebrafish as a model to study peripheral thyroid hormone metabolism in vertebrate development. Gen. Comp. Endocrinol.. 2013;188:289-296.
- [CrossRef] [Google Scholar]
- Effect of imidacloprid on the survival of Xenopus tadpoles challenged with wild type frog virus 3. Aquat. Toxicol.. 2018;2018(194):152-158.
- [CrossRef] [Google Scholar]
- The impact of four widely used neonicotinoid insecticides on Tetragonisca angustula (Latreille) (Hymenoptera: Apidae) Chemosphere. 2019;224:65-70.
- [CrossRef] [Google Scholar]
- Toxicity of the fungicide trifloxystrobin on tadpoles and its effect on fish–tadpole interaction. Chemosphere. 2012;87:1348-1354.
- [CrossRef] [Google Scholar]
- Effects of neonicotinoids on putative escape behavior of juvenile wood frogs (Lithobates sylvaticus) chronically exposed as tadpoles. Environ. Toxicol. Chem.. 2018;37 3115e3123
- [CrossRef] [Google Scholar]
- Effects of current-use pesticides on amphibians. In: Sparling D.W., Linder G., Bishop C.A., Krest S., eds. Ecotoxicology of Amphibians and Reptiles (2nd ed.). Pensacola, FL, USA: SETAC Press; 2010. p. :167-202.
- [Google Scholar]
- Waterborne exposure to triadimefon causes thyroid endocrine disruption and developmental delay in Xenopus laevis tadpoles. Aquat. Toxicol.. 2016;177:190-197.
- [CrossRef] [Google Scholar]
- Xenopus laevis tadpoles exposed to metamifop: Changes in growth, behavioral endpoints, neurotransmitters, antioxidant system and thyroid development. Ecotox. Environ. Safe.. 2021;220:12.
- [CrossRef] [Google Scholar]
- The progestin norethisterone affects thyroid hormone-dependent metamorphosis of Xenopus laevis tadpoles at environmentally relevant concentrations. Ecotox. Environ. Safe.. 2018;150:86-95.
- [CrossRef] [Google Scholar]
- Amphibians and agricultural chemicals: review of the risks in a complex environment. Environ. Pollut.. 2009;157:2903-3227.
- [CrossRef] [Google Scholar]
- Oxidative stress, tissue remodeling and regression during amphibian metamorphosis. Comp. Biochem. Physiol. C: Toxicol. Pharmacol.. 2007;145(4):625-631.
- [CrossRef] [Google Scholar]
- Effects of glyphosate-based herbicides on survival, development, growth and sex ratios of wood frogs (Lithobates sylvaticus) tadpoles. I: chronic laboratory exposures to visionmax®. Aquat. Toxicol.. 2014;154:278-290.
- [CrossRef] [Google Scholar]
- OECD, 2011. Guidelines for the Testing of Chemicals, 305: Fish, Bioaccumulation in Fish: Aqueous and Dietary Exposure. OECD (2011).
- OECD, 2019. Guidelines for the Testing of Chemicals, 203: Fish, Acute Toxicity Test. OECD (2019), 10.1787/20745761.
- Toxicity and sublethal effects of sulfoxaflor on the red imported fire ant, Solenopsis invicta. Ecotox. Environ. Safe.. 2017;139:377-383.
- [CrossRef] [Google Scholar]
- Invasive predator snake induces oxidative stress responses in insular amphibian species. Sci. Total Environ.. 2016;566:57.
- [CrossRef] [Google Scholar]
- Imidacloprid induced alterations in enzyme activities and energy reserves of the land snail, Helix aspersa. Ecotox. Environ. Safe.. 2013;95:91-97.
- [CrossRef] [Google Scholar]
- Effects of imidacloprid on Rana catesbeiana immune and nervous system. Chemosphere.. 2017;188:465-469.
- [CrossRef] [Google Scholar]
- Sublethal effects on wood frogs chronically exposed to environmentally relevant concentrations of two neonicotinoid insecticides. Environ Toxicol Chem. 2017;36:1101-1109.
- [CrossRef] [Google Scholar]
- Cadmium effect on oxidative metabolism of pea (Pisum sativum L.) roots. Imaging of reactive oxygen species and nitric oxide accumulation in vivo. Plant Cell Environ.. 2006;29:153.
- [CrossRef] [Google Scholar]
- Cypermethrin- and fipronil-based insecticides cause biochemical changes in Physalaemus gracilis tadpoles. Environ. Sci. Pollut. Res.. 2021;28(4):4377-4387.
- [CrossRef] [Google Scholar]
- Acute and chronic toxicity tests of systemic insecticides, four neonicotinoids and fipronil, using the tadpoles of the western clawed frog Silurana tropicalis. Chemosphere. 2021;270:129418.
- [CrossRef] [Google Scholar]
- Oxidative stress: oxidants and antioxidants. Exp. Physiol.. 1997;82:291-295.
- [CrossRef] [Google Scholar]
- Effects of trilostane and fipronil on the reproductive axis in an early life stage of the Japanese medaka (Oryzias latipes) Ecotoxicology. 2014;23:1044-1054.
- [CrossRef] [Google Scholar]
- Influences of introducing frogs in the paddy fields on soil properties and rice growth. J. Soils Sediments.. 2016;16(1):51-61.
- [CrossRef] [Google Scholar]
- Unraveling the toxic effects of neonicotinoid insecticides on the thyroid endocrine system of lizards. Environ. Pollut.. 2020;258
- [CrossRef] [Google Scholar]
- Combined toxic effects of fludioxonil and triadimefon on embryonic development of zebrafish (Danio rerio) Environ. Pollut.. 2020;260:114105
- [CrossRef] [Google Scholar]
- Antioxidant responses to benzo [a] pyrene, tributyltin and their mixture in the spleen of Sebasticus marmoratu. J. Environ. Sci.. 2007;19:1129-1135.
- [CrossRef] [Google Scholar]
- Acute toxicity and risk assessment of pesticides used in strawberry for controlling aphid to honeybees. Asian. J. Ecotoxicol.. 2017;12:222-227.
- [CrossRef] [Google Scholar]
- Effects of alpha-cypermethrin enantiomers on the growth, biochemical parameters and bioaccumulation in Rana nigromaculata tadpoles of the anuran amphibians. Ecotox. Environ. Safe.. 2017;139:431-438.
- [CrossRef] [Google Scholar]
- Occurrence and distribution of neonicotinoid insecticides in surface water and sediment of the Guangzhou section of the Pearl River, South China. Environ. Pollut.. 2019;251:892-900.
- [CrossRef] [Google Scholar]
- Thyroid endocrine disruption in zebrafish larvae following exposure to hexaconazole and tebuconazole. Aquat. Toxicol.. 2013;138–139:35-42.
- [CrossRef] [Google Scholar]
- Comparison of triadimefon and its metabolite on acute toxicity and chronic effects during the early development of Rana nigromaculata tadpoles. Ecotox. Environ. Safe.. 2018;156:247-254.
- [CrossRef] [Google Scholar]
- Amphibian (Rana nigromaculata) exposed to cyproconazole: Changes in growth index, behavioral endpoints, antioxidant biomarkers, thyroid and gonad development. Aquat. Toxicol.. 2019;208:62-70.
- [CrossRef] [Google Scholar]
Appendix A
Supplementary material
Supplementary data to this article can be found online at https://doi.org/10.1016/j.arabjc.2023.104723.
Appendix A
Supplementary material
The following are the Supplementary data to this article:Supplementary data 1
Supplementary data 1







